Reticular lamina and basilar membrane vibrations in living mouse cochleae
|
|
|
- Dominic Miles
- 5 years ago
- Views:
Transcription
1 Reticular lamina and basilar membrane vibrations in living mouse cochleae Tianying Ren a,1, Wenxuan He a, and David Kemp b a Oregon Hearing Research Center, Department of Otolaryngology, Oregon Health & Science University, Portland, OR 97239; and b University College London Ear Institute, University College London, London WC1E 6BT, United Kingdom Edited by Mario A. Ruggero, Northwestern University, Evanston, IL, and accepted by Editorial Board Member Peter L. Strick July 1, 2016 (received for review May 9, 2016) It is commonly believed that the exceptional sensitivity of mammalian hearing depends on outer hair cells which generate forces for amplifying sound-induced basilar membrane vibrations, yet how cellular forces amplify vibrations is poorly understood. In this study, by measuring subnanometer vibrations directly from the reticular lamina at the apical ends of outer hair cells and from the basilar membrane using a custom-built heterodyne low-coherence interferometer, we demonstrate in living mouse cochleae that the soundinduced reticular lamina vibration is substantially larger than the basilar membrane vibration not only at the best frequency but surprisingly also at low frequencies. The phase relation of reticular lamina to basilar membrane vibration changes with frequency by up to 180 degrees from 135 degrees at low frequencies to -45 degrees at the best frequency. The magnitude and phase differences between reticular lamina and basilar membrane vibrations are absent in postmortem cochleae. These results indicate that outer hair cells do not amplify the basilar membrane vibration directly through a local feedback as commonly expected; instead, they actively vibrate the reticular lamina over a broad frequency range. The outer hair cell-driven reticular lamina vibration collaboratively interacts with the basilar membrane traveling wave primarily through the cochlear fluid, which boosts peak responses at the best-frequency location and consequently enhances hearing sensitivity and frequency selectivity. cochlea outer hair cells hearing cochlear amplifier interferometry As the auditory sensory organ, the mammalian cochlea is able to detect soft sounds at levels as low as 20 μpa, equivalent to eardrum vibrations of <1-pm displacement (1), which is >100-fold smaller than the diameter of a hydrogen atom. The cochlea s remarkable sensitivity is commonly attributed to a micromechanical feedback system, which amplifies soft sounds using forces generated by outer hair cells (2 10). In addition to active bundle movements (2), mammalian outer hair cells are capable of changing their lengths in response to electrical stimulation in vitro (11 14). This electromechanical force production is termed outer hair cell electromotility or somatic motility, which is produced by the membrane protein, prestin (15). Transgenic mice without prestin or with altered prestin suffer from severe hearing loss (4, 16). Targeted inactivation of prestin over well-defined cochlear segments dramatically reduces the traveling wave s peak response (17). Recent micromechanical measurements in sensitive living mouse cochleae showed (18) that electrical stimulation of outer hair cells induces vigorous vibrations of the reticular lamina at the apical surfaces of the outer hair cells. Whereas this experiment demonstrates that outer hair cells are capable of generating substantial forces to vibrate cochlear microstructures in vivo, the lack of frequency selectivity of the electrically induced reticular lamina vibration, however, is inconsistent with the sharp tuning of the basilar membrane vibration. Because electrical stimulation is not a natural stimulus of the auditory system, we here investigate how acoustically induced reticular lamina vibrations interact with basilar membrane vibrations in sensitive living mouse cochleae, using a custom-built scanning heterodyne low-coherence interferometer (Fig. 1 A and B and Materials and Methods). The results from this study do not demonstrate the commonly expected cochlear local feedback but instead indicate a global hydromechanical mechanism for outer hair cells to enhance hearing sensitivity. Results Vibrations of the Reticular Lamina and Basilar Membrane in Sensitive Cochleae. Vibrations of the reticular lamina and basilar membrane measured from a sensitive cochlea are presented in Fig. 1 C J. Below 50 db sound pressure level (SPL) (0 db SPL = 20 μpa), the basilar membrane vibration increased with frequency and reached the maximum at 48 khz (best frequency) and then decreased, forming a sharp peak (Fig. 1C). The peak became broader and shifted toward low frequencies as the sound level increased. The ratios of the basilar membrane displacement to the malleus displacement at low sound levels confirm the best frequency of 48 khz (Fig. 1E). The peak ratio decreased from >100 to 3 as the sound level increased from 20 to 80 db SPL, indicating 30-dB nonlinear compression. The overlapping curves at frequencies below 35 khz (Fig. 1E) indicate the linear growth at low frequencies. The basilar membrane vibration phase (referred to as the malleus phase) decreased with frequency (Fig. 1G). Data in Fig. 1 C and E demonstrate the high sensitivity, sharp tuning, and nonlinearity of basilar membrane responses to ultrasonic sounds in the living mouse cochlea, which are similar to previous measurements in squirrel monkeys (19), gerbils (20 23), chinchillas (17, 24, 25), guinea pigs (26 28), and mice (7, 29, 30). By contrast, the displacement of the reticular lamina vibration (Fig. 1D) was substantially larger than that of the basilar membrane vibration. At 20 db SPL, reticular lamina vibrations were the greatest approximately at the same frequency as the best frequency of the basilar membrane ( 48 khz). Notably, the peak Significance The remarkable sensitivity of mammalian hearing depends on auditory sensory outer hair cells, yet how these cells enhance the hearing sensitivity remains unclear. By measuring subnanometer vibrations directly from motile outer hair cells in living inner ear, we demonstrate that outer hair cells do not directly amplify the basilar membrane vibration by local feedback as commonly expected; instead, they actively vibrate the reticular lamina over a broad frequency range. The outer hair cell-driven reticular lamina vibration interacts with the basilar membrane traveling wave through the cochlear fluid, resulting in maximal vibrations at the best-frequency location, consequently enhancing hearing sensitivity. This finding advances our knowledge of mammalian hearing and may lead to strategies for restoring hearing in patients. Author contributions: T.R. designed research; T.R. and W.H. performed research; T.R., W.H., and D.K. analyzed data; and T.R., W.H., and D.K. wrote the paper. The authors declare no conflict of interest. This article is a PNAS Direct Submission. M.A.R. is a Guest Editor invited by the Editorial Board. 1 To whom correspondence should be addressed. rent@ohsu.edu PNAS August 30, 2016 vol. 113 no. 35
2 Fig. 1. Diagram of the organ of Corti and vibrations of the reticular lamina and basilar membrane in a sensitive mouse cochlea. (A) Intracochlear image through the intact round window membrane in a living mouse. BM, basilar membrane; OSL, osseous spiral lamina. (B) Diagram of the organ of Corti. RL, reticular lamina; IHC, inner hair cell; OHCs, outer hair cells; TM, tectorial membrane; DCs, Deiters cells. (C and D) Displacements of the BM (red) and RL (blue) as a function of frequency. The noise floor (black dotted line in C) is <0.01 nm. BF, best frequency. (E and F) Displacement ratios of the BM and RL to the malleus. (G and H) BM and RL phase referred to the malleus. (I)Theratio of the RL to BM displacement. (J) Phase difference between the RL and BM. displacement of the reticular lamina was 10-fold greater than that of the basilar membrane. At sound levels above 40 db SPL, overlapping curves near the best frequency indicate strong nonlinear compression. Surprisingly, displacements of the reticular lamina at frequencies far below the best frequency were also 10- fold greater than those of the basilar membrane (Fig. 1D). Displacement ratios of the reticular lamina to the malleus in Fig. 1F show a peak magnitude of 1,000 at low sound levels, which is larger than that of the basilar membrane (Fig. 1E). With an increase in sound level from 20 to 80 db SPL, the peak magnitude decreased by 45 db for the reticular lamina vibration (Fig. 1F) and by 30 db for the basilar membrane vibration (Fig. 1E). The phase responses of the reticular lamina (Fig. 1H) were similar to those of the basilar membrane (Fig. 1G). Ratios of the reticular lamina to basilar membrane displacement greater than one (Fig. 1I) confirm that the reticular lamina vibrated more than the basilar membrane at all frequencies and sound levels. Near 48 khz, 30-dB SPL tone-induced reticular lamina vibration was approximately sevenfold greater than that of the basilar membrane. This difference decreased with sound level and approached one at 80 db SPL. At a given sound level, the magnitude ratio decreased with frequency up to 48 khz. The magnitude ratio curves also shifted toward low frequencies with the sound level increase. Surprisingly, the largest-magnitude ratios occurred at frequencies far below the best frequency and at high sound levels, which likely resulted from the saturation near the best frequency. The phase difference of >90 degrees in Fig. 1J indicates that the reticular lamina and basilar membrane vibrated approximately in opposite directions at frequencies below 15 khz. The lack of substantial phase difference near 48 khz, however, demonstrates that the reticular lamina and the basilar membrane moved in the same direction at the best frequency. Vibrations of the Reticular Lamina and Basilar Membrane in Insensitive Cochleae. Under postmortem conditions, the basilar membrane and reticular lamina vibrations (blue lines in Fig. 2 A and B) showed decreased magnitude near the best frequency, peak shift toward low frequencies, and linear growth with the sound level. In contrast with the lack of substantial postmortem change in basilar membrane vibration at frequencies below 30 khz in Fig. 2A, the reticular lamina displacements decreased by 10-fold (or 20 db) over the same frequency range (Fig. 2B). These postmortem magnitude changes were confirmed by the displacement ratios of the basilar membrane and reticular lamina to the malleus in Fig. 2 C and D. In contrast to the basilar membrane phase responses (Fig. 2E), the reticular lamina vibration showed a substantial postmortem phase decrease at low frequencies (Fig. 2F). Compared with sensitive responses, the ratio of the reticular lamina to basilar membrane displacement decreased dramatically and approached one (blue lines in Fig. 2G) in the postmortem cochlea, and the phase difference approached zero (blue lines in Fig. 2H). Thus, under postmortem conditions, the reticular lamina and basilar membrane vibrated synchronously in the same direction and with similar magnitudes at all observed frequencies and sound levels. The Relationship Between the Reticular Lamina and Basilar Membrane Vibrations. Average displacements at the best frequency as a function of the sound level from eight cochleae (Fig. 3A) showed that reticular lamina displacements were significantly greater than those of the basilar membrane at 20, 30, 40, and 50 db SPL (t > 1.89, P < 0.05, n = 8). The displacement difference decreased with the sound level and became insignificant at 60, 70, and 80 db SPL (t < 0.73, P > 0.35, n = 8). The phase of the reticular lamina increased slightly with the sound level up to 70 db SPL (Fig. 3B). In postmortem cochleae, the reticular lamina and basilar membrane vibrations increased proportionally with the sound level (Fig. 3C), whereas the corresponding phase changed insignificantly (Fig. 3D). The average displacement ratio of the reticular lamina to the basilar membrane and the corresponding phase difference from five sensitive and six insensitive cochleae are presented as a NEUROSCIENCE Ren et al. PNAS August 30, 2016 vol. 113 no
3 Discussion In this study, the reticular lamina and basilar membrane vibrations were measured at a longitudinal location 0.85 mm from the cochlear base (red arrows in Fig. 4 A and B) asafunctionof frequency. Because the peak location of the cochlear partition vibration is determined by frequency (31), the change in stimulus frequency from low to high caused the peak of the traveling wave to move from apex to base. Whereas the vibration at 48 khz was Fig. 2. Postmortem changes in reticular lamina and basilar membrane vibrations. Postmortem data were collected immediately after death (blue) and are compared with those measured under sensitive conditions (red) in the same cochlea. (A) Displacements of basilar membrane (BM) as a function of frequency at different sound levels. (B) Reticular lamina (RL) displacements. (C) Displacement ratios of the BM to malleus. (D) Displacement ratios of the RL to malleus. (E and F) BM and RL phase referred to the malleus. (G) Magnitude ratios of the RL to BM. (H) Phase difference between the BM and RL. In addition to poor sensitivity, broad tuning, and linear growth, the magnitude and phase differences between the RL and BM were absent under postmortem conditions. function of frequency in Fig. 3 E H. Because the basilar membrane vibrations at low frequencies can be recorded only at relatively high sound levels due to sharp tuning in sensitive cochleae, the data in Fig. 3 E H were collected at 80 db SPL. Fig. 3 E and F shows that reticular lamina vibration at 10 khz was >fivefold larger than the basilar membrane vibration, with a phase lead of >90 degrees in sensitive cochleae. Near the best frequency, reticular lamina and basilar membrane vibrations had a similar magnitude and phase. The magnitude and phase differences were absent in postmortem cochleae (Fig. 3 G and H). Fig. 3. Differences between the reticular lamina and basilar membrane vibrations. (A) Reticular lamina (RL) and basilar membrane (BM) displacements at different sound levels in sensitive cochleae. (B) RL and BM phase. Phase was normalized to that at 80 db SPL. (C) RL and BM displacements in postmortem cochleae. (D) Insensitive RL and BM phase. (E) Displacement ratios of the RL to BM show that the RL vibrated >fivefold more than the BM at low frequencies. This difference decreased with frequency and approached one near the best frequency. (F) RL phase led the BM phase by 135 degree at low frequencies and lagged the BM phase by -45 degrees at the best frequency (48.08 ± 0.25 khz, n = 5) in sensitive cochleae. (G and H) The magnitude and phase differences were absent in postmortem cochleae Ren et al.
4 Fig. 4. Longitudinal patterns and interaction of the cochlear partition vibrations. The active component of the reticular lamina vibration (green lines in panels C F) was obtained by vector subtraction of the basilar membrane vibration from the measured reticular lamina vibration. (A) Diagram of the conventional traveling wave at 48 khz shows that the vibration was measured at the peak of the traveling wave (red arrow). (B) At a frequency lower than the BF, the vibration was measured at a basal part of the traveling wave. (C) At 40 db SPL, the reticular lamina (RL) and basilar membrane (BM) vibrations were at a small region centered at the BF location. (E) At 70 db SPL, both RL and BM displacements extended from the BF location to the base. (D and F) Near the base, RL phase led BM phase by >120 degrees; they were approximately in-phase at the BF location. (G) Diagram of a single tone-induced cochlear traveling wave. D OHC, the outer hair cell force-induced displacement; D TW, the traveling wave-induced displacement. (H) At the base of the traveling wave, RL and BM move in approximately opposite directions. The active RL movement creates a negative fluid pressure in the scala vestibuli and a positive fluid pressure inside the organ of Corti. (I) At the peak of the measured at the response peak location (Fig. 4A), the vibration at a lower frequency was measured at the basal part of the traveling wave because the response peak moved toward an apical location (Fig. 4B). Therefore, the frequency responses measured in this study can reflect the relationship between the reticular lamina and basilar membrane vibrations at different parts of traveling waves. The longitudinal patterns of the reticular lamina and basilar membrane vibrations at 48 khz were obtained from measured frequency responses (Fig. 1 C, D, G,andH)(Materials and Methods) and are presented by blue and red lines in Fig. 4 C F.Whereas40-dB SPL responses occurred within a 200-μm region(fig. 4C and D), a 70-dB SPL tone-induced vibration extended over the entire region from the response peak to the base (Fig. 4 E and F). The reticular lamina vibration was substantially larger than the basilar membrane vibration not only at the peak (Fig. 4C) but also at the basal part of the traveling wave (Fig. 4E). Phase difference between the reticular lamina and basilar membrane vibrations was >120 degrees at the base (Fig. 4F) but insignificant at the response peak (Fig. 4 D and F). Because outer hair cells sit on the basilar membrane via Deiters cells, they can move passively, following the basilar membrane traveling wave. Outer hair cells can also change their length (11) in response to the membrane potential change caused by sound stimulation (32). Because the reticular lamina vibration is a vector sum of the basilar membrane vibration and the outer hair celldriven movement, the active component of the reticular lamina vibration was obtained by vector subtraction of the basilar membrane vibration from the measured reticular lamina vibration and is shown by green lines in Fig. 4 C F. The overlapping blue and green lines in Fig. 4 C and D indicate that reticular lamina vibration was dominated by the outer hair cell-driven component at 40 db SPL. In Fig. 4E,the green line diverges from the blue line and almost overlaps the red line near the best frequency, reflecting saturation of the outer hair celldriven component at high sound levels near the response peak. These data indicate that acoustically induced reticular lamina vibration is dominated by outer hair cell-driven vibration over the entire traveling wave region. This is confirmed by postmortem changes, which show no substantial difference between the reticular lamina and basilar membrane vibrations (blue lines in Figs. 2 G and H and 3 G and H). The data also show that neither destructive interference between the basilar membrane and reticular lamina vibrations at the base nor constructive interference near the peak is significant (indicated by the negligible gap between blue and green lines in Fig. 4 C F) due to the large-magnitude difference between the reticular lamina and basilar membrane vibrations. Because force production of outer hair cells depends on metabolic energy in vivo and the generated forces act equally on both ends of the cells but in opposite directions, it is expected that the outer hair cell-driven reticular lamina and basilar membrane vibrations are physiologically vulnerable and in opposite directions (13, 33, 34). The lack of postmortem changes in the basilar membrane vibration at low frequencies (Fig. 2 A and C) and approximately in-phase vibrations of the reticular lamina and basilar membrane near the best frequency (Figs. 1J and 3F) indicate that, under physiological conditions, locally generated outer hair cell forces do not drive the basilar membrane effectively, likely because outer hair cell-generated forces could not be coupled back to the basilar membrane (35). When sound causes the stapes to move out toward the middle ear, displacing fluid out of the scala vestibuli, the basilar membrane at the cochlear base reacts by moving up (Fig. 4H). This upward movement of the basilar membrane depolarizes outer hair cells by deflecting hair bundles toward the excitatory direction (35). Due to traveling wave, the RL and BM move in the same direction, which results in the maximal vibration at the apical ends of outer hair cells through constructive interference. Dotted lines show resting positions. NEUROSCIENCE Ren et al. PNAS August 30, 2016 vol. 113 no
5 configuration change in the motor protein, prestin, outer hair cells contract, pulling the reticular lamina toward the basilar membrane (Fig. 4H). Unexpectedly, the present results indicate a 10-fold greater displacement of the reticular lamina than the basilar membrane. Furthermore, the observed phase difference of up to 135 degrees between the two vibrations (Fig. 4F) indicates that they move in approximately opposite directions at the base. The fluid displaced by this opposing reticular lamina motion may have a significant influence on the developing traveling wave. Indeed, electrically evoked reticular lamina vibration has already been shown to initiate a conventional traveling wave (18). The unexpectedly large opposing motion of the reticular lamina must also result in substantial fluid displacements inside the organ of Corti. When the reticular lamina moves toward the basilar membrane (Fig. 4H) at a basal location, the reticular lamina at a more apical location 0.5 wavelength (or 180 degrees) from the basal location moves away from the basilar membrane at the same time. Active differential movements of the reticular lamina could therefore result in longitudinal fluid movements. As the traveling waves propagate forward, these longitudinal fluid movements help transfer energy toward best-frequency locations (6, 36) (Fig. 4G), boosting the peak response. Our interpretation of the present results is also consistent with the following previous studies. Out-of-phase vibrations of the reticular lamina and basilar membrane at low frequencies (Figs. 1J and 3F) are compatible with electrically evoked opposite movements at both ends of outer hair cells (11, 12) and at the reticular lamina (or tectorial membrane) and the basilar membrane in excised cochleae (13, 33, 34). The acoustically induced in-phase vibrations near the best frequency are similar to electrically evoked reticularlaminaandbasilarmembranevibrations(18)andlowfrequency tone-induced vibrations of Hensen s cells and basilar membrane (37). The proposed outer hair cell-driven fluid movement inside the organ of Corti is in agreement with the electrically induced fluid movement inside the tunnel of Corti in vitro (38). Because the outer hair cell-generated pressure inside the organ of Corti acts in all directions, the proposed mechanism can interpret complex vibrations of the organ of Corti (18, 27, 29, 30). Phase inconsistency between our results and others likely resulted from differences in phase detection techniques, animal species, and measurement locations. Whereas the proposed cochlear micromechanical mechanism is well supported by experimental data, it does not exclude other mechanisms (33, 39 49). In summary, heterodyne low-coherence interferometry in living mouse cochleae demonstrates that the sound-induced reticular lamina vibration is substantially larger than basilar membrane vibration and is physiologically vulnerable not only at the best frequency but also at low frequencies and that the reticular lamina and basilar membrane vibrate approximately in the same direction near the best frequency. These results indicate that, contrary to expectations, outer hair cells do not amplify the basilar membrane vibration directly by local feedback. Rather, they actively vibrate the reticular lamina over a broad frequency range. The interaction of the outer hair cell-driven reticular lamina vibration with the basilar membrane traveling wave through the cochlear fluid results in maximum responses at the best-frequency location, which consequently enhances hearing sensitivity and frequency selectivity. Materials and Methods Eighteen male and female CBA/CaJ mice at 3 5 wk of age were used in this study. The animal use protocol was approved by the Oregon Health & Science University Institutional Animal Care and Use Committee. The Scanning Heterodyne Low-Coherence Interferometer. Based on a scanning laser interferometer (50, 51), a scanning heterodyne low-coherence interferometer was built by replacing the light source and related optical and electronic components (18). Taking advantage of the small coherence length, low-coherence interferometers can measure vibration with a high axial resolution (27, 29, 52). Due to the use of the 40-MHz carrier frequency for heterodyne detection, the current low-coherence interferometer has unprecedented sensitivity, wide dynamic range, no 180-degree phase ambiguity, and high temporal resolution. Although the measurement noise floor is below 0.01 nm (black dotted line in Fig. 1C) and heterodyne interferometry does not require a stable optical path length, occasional animal movement disturbed low-level measurements. Thus, data points showing irregular patterns, particularly with <10-dB signal-to-noise ratio, were not presented. The use of low-coherence light and an objective lens with relatively large numerical aperture (NA = 0.42) provides adequate spatial selectivity for measuring the reticular lamina and basilar membrane vibrations through the intact round window membrane in mice. Measurement of the Reticular Lamina and Basilar Membrane Vibrations. Animal anesthesia and surgical procedures were the same as described previously (18). Under direct visualization, low-coherence light from the interferometer was focused on the center of the outer hair cell region of the cochlear partition through the intact round window membrane (Fig. 1A). The distance from the cochlear base to the measurement location was 0.85 mm. The transverse locations of the basilar membrane and reticular lamina were determined by measuring the backscattered light level and the vibration magnitude and phase as a function of the transverse location. When the object beam of the interferometer was focused on the basilar membrane, or reticular lamina, acoustical tones at different frequencies and levels were delivered to the ear canal. The tone frequency was changed from 0.75 to 67.5 khz by <0.75 khz per step. Data Analysis and Statistical Methods. Igor Pro (Version 6.35A5, WaveMetrics) was used for processing and analyzing data. Frequency responses of the reticular lamina and basilar membrane were presented by plotting displacement and phase as a function of frequency. The transfer functions were estimated by the displacement ratio of the reticular lamina or basilar membrane to the malleus at different frequencies. The relationship between the reticular lamina and basilar membrane vibrations was presented by the ratio of the reticular lamina displacement to the basilar membrane displacement and the phase difference obtained by subtracting the basilar membrane phase from the reticular lamina phase. The longitudinal patterns of the reticular lamina and basilar membrane vibrations at the best frequency of the measured location were presented by plotting displacement and phase as a function of the longitudinal location. The longitudinal location was derived from the stimulus frequency according to the frequency location function in mouse cochleae (31) [d = ð logðfþþ= , where d is distance from the base in millimeters and f is frequency in khz]. The grouped results were presented by mean and SE calculated across animals at given frequencies and stimulus levels. Displacement difference between the reticular lamina and basilar membrane vibration at the best frequency was determined using a two-tailed t test, and P value <0.05 was considered statistically significant. ACKNOWLEDGMENTS. We thank Peter Barr-Gillespie and John V. Brigande for valuable comments on the manuscript, Alfred L. Nuttall and other colleagues at Oregon Hearing Research Center for helpful discussion of the data, and Edward Porsov for technical help. This study was funded by NIH Grant R01 DC (to T.R.). 1. Dalhoff E, Turcanu D, Zenner HP, Gummer AW (2007) Distortion product otoacoustic emissions measured as vibration on the eardrum of human subjects. Proc Natl Acad Sci USA 104(5): Hudspeth AJ (2014) Integrating the active process of hair cells with cochlear function. Nat Rev Neurosci 15(9): Kemp DT (1986) Otoacoustic emissions, travelling waves and cochlear mechanisms. Hear Res 22: Dallos P (2008) Cochlear amplification, outer hair cells and prestin. Curr Opin Neurobiol 18(4): Fettiplace R, Hackney CM (2006) The sensory and motor roles of auditory hair cells. Nat Rev Neurosci 7(1): Reichenbach T, Hudspeth AJ (2014) The physics of hearing: Fluid mechanics and the active process of the inner ear. Rep Prog Phys 77(7): Russell IJ, et al. (2007) Sharpened cochlear tuning in a mouse with a genetically modified tectorial membrane. Nat Neurosci 10(2): Peng AW, Ricci AJ (2011) Somatic motility and hair bundle mechanics, are both necessary for cochlear amplification? Hear Res 273(1-2): Dong W, Olson ES (2013) Detection of cochlear amplification and its activation. Biophys J 105(4): Robles L, RuggeroMA (2001) Mechanics ofthe mammalian cochlea. Physiol Rev 81(3): Brownell WE, Bader CR, Bertrand D, de Ribaupierre Y (1985) Evoked mechanical responses of isolated cochlear outer hair cells. Science 227(4683): Ren et al.
6 12. Santos-Sacchi J (1989) Asymmetry in voltage-dependent movements of isolated outer hair cells from the organ of Corti. J Neurosci 9(8): Mammano F, Ashmore JF (1993) Reverse transduction measured in the isolated cochlea by laser Michelson interferometry. Nature 365(6449): Frank G, Hemmert W, Gummer AW (1999) Limiting dynamics of high-frequency electromechanical transduction of outer hair cells. Proc Natl Acad Sci USA 96(8): Zheng J, et al. (2000) Prestin is the motor protein of cochlear outer hair cells. Nature 405(6783): Liberman MC, et al. (2002) Prestin is required for electromotility of the outer hair cell and for the cochlear amplifier. Nature 419(6904): Fisher JA, Nin F, Reichenbach T, Uthaiah RC, Hudspeth AJ (2012) The spatial pattern of cochlear amplification. Neuron 76(5): Ren T, He W, Barr-Gillespie PG (2016) Reverse transduction measured in the living cochlea by low-coherence heterodyne interferometry. Nat Commun 7: Rhode WS (1971) Observations of the vibration of the basilar membrane in squirrel monkeys using the Mössbauer technique. J Acoust Soc Am 49(4):2, Overstreet EH, 3rd, Temchin AN, Ruggero MA (2002) Basilar membrane vibrations near the round window of the gerbil cochlea. J Assoc Res Otolaryngol 3(3): Versteegh CP, van der Heijden M (2012) Basilar membrane responses to tones and tone complexes: Nonlinear effects of stimulus intensity. J Assoc Res Otolaryngol 13(6): van der Heijden M, Versteegh CP (2015) Energy flux in the cochlea: Evidence against power amplification of the traveling wave. J Assoc Res Otolaryngol 16(5): Ren T, Nuttall AL (2001) Basilar membrane vibration in the basal turn of the sensitive gerbil cochlea. Hear Res 151(1-2): Ruggero MA, Narayan SS, Temchin AN, Recio A (2000) Mechanical bases of frequency tuning and neural excitation at the base of the cochlea: Comparison of basilarmembrane vibrations and auditory-nerve-fiber responses in chinchilla. Proc Natl Acad Sci USA 97(22): Recio A, Rich NC, Narayan SS, Ruggero MA (1998) Basilar-membrane responses to clicks at the base of the chinchilla cochlea. J Acoust Soc Am 103(4): Cooper NP, Rhode WS (1992) Basilar membrane mechanics in the hook region of cat and guinea-pig cochleae: Sharp tuning and nonlinearity in the absence of baseline position shifts. Hear Res 63(1-2): Chen F, et al. (2011) A differentially amplified motion in the ear for near-threshold sound detection. Nat Neurosci 14(6): Nilsen KE, Russell IJ (2000) The spatial and temporal representation of a tone on the guinea pig basilar membrane. Proc Natl Acad Sci USA 97(22): Lee HY, et al. (2015) Noninvasive in vivo imaging reveals differences between tectorial membrane and basilar membrane traveling waves in the mouse cochlea. Proc Natl Acad Sci USA 112(10): Gao SS, et al. (2014) Vibration of the organ of Corti within the cochlear apex in mice. J Neurophysiol 112(5): Müller M, von Hünerbein K, Hoidis S, Smolders JW (2005) A physiological placefrequency map of the cochlea in the CBA/J mouse. Hear Res 202(1-2): Corey DP, Hudspeth AJ (1979) Ionic basis of the receptor potential in a vertebrate hair cell. Nature 281(5733): Gummer AW, Hemmert W, Zenner HP (1996) Resonant tectorial membrane motion in the inner ear: Its crucial role in frequency tuning. Proc Natl Acad Sci USA 93(16): Nowotny M, Gummer AW (2011) Vibration responses of the organ of Corti and the tectorial membrane to electrical stimulation. J Acoust Soc Am 130(6): Reichenbach T, Hudspeth AJ (2010) A ratchet mechanism for amplification in lowfrequency mammalian hearing. Proc Natl Acad Sci USA 107(11): Lighthill J (1981) Energy flow in the cochlea. J Fluid Mech 106: Khanna SM, Hao LF (2000) Amplification in the apical turn of the cochlea with negative feedback. Hear Res 149(1-2): Karavitaki KD, Mountain DC (2007) Evidence for outer hair cell driven oscillatory fluid flow in the tunnel of Corti. Biophys J 92(9): Markin VS, Hudspeth AJ (1995) Modeling the active process of the cochlea: Phase relations, amplification, and spontaneous oscillation. Biophys J 69(1): Zwislocki JJ, Kletsky EJ (1979) Tectorial membrane: A possible effect on frequency analysis in the cochlea. Science 204(4393): Neely ST, Kim DO (1983) An active cochlear model showing sharp tuning and high sensitivity. Hear Res 9(2): Allen JB (1980) Cochlear micromechanics a physical model of transduction. J Acoust Soc Am 68(6): Hubbard A (1993) A traveling-wave amplifier model of the cochlea. Science 259(5091): Ghaffari R, Aranyosi AJ, Richardson GP, Freeman DM (2010) Tectorial membrane travelling waves underlie abnormal hearing in Tectb mutant mice. Nat Commun 1: van der Heijden M (2014) Frequency selectivity without resonance in a fluid waveguide. Proc Natl Acad Sci USA 111(40): Wang Y, Steele CR, Puria S (2016) Cochlear outer-hair-cell power generation and viscous fluid loss. Sci Rep 6: Ramamoorthy S, Deo NV, Grosh K (2007) A mechano-electro-acoustical model for the cochlea: Response to acoustic stimuli. J Acoust Soc Am 121(5 Pt1): Liu Y, Gracewski SM, Nam JH (2015) Consequences of location-dependent organ of Corti micro-mechanics. PLoS One 10(8):e Ó Maoiléidigh D, Hudspeth AJ (2013) Effects of cochlear loading on the motility of active outer hair cells. Proc Natl Acad Sci USA 110(14): Ren T (2002) Longitudinal pattern of basilar membrane vibration in the sensitive cochlea. Proc Natl Acad Sci USA 99(26): Ren T (2004) Reverse propagation of sound in the gerbil cochlea. Nat Neurosci 7(4): Hong SS, Freeman DM (2006) Doppler optical coherence microscopy for studies of cochlear mechanics. J Biomed Opt 11(5): NEUROSCIENCE Ren et al. PNAS August 30, 2016 vol. 113 no
Measurement of cochlear power gain in the sensitive gerbil ear
 Received 4 Nov 2 Accepted 3 Feb 2 Published Mar 2 DOI:.38/ncomms226 Measurement of cochlear power gain in the sensitive gerbil ear Tianying Ren,2, Wenxuan He & Peter G. Gillespie,3 The extraordinary sensitivity
Received 4 Nov 2 Accepted 3 Feb 2 Published Mar 2 DOI:.38/ncomms226 Measurement of cochlear power gain in the sensitive gerbil ear Tianying Ren,2, Wenxuan He & Peter G. Gillespie,3 The extraordinary sensitivity
THE INTERPLAY BETWEEN ACTIVE HAIR BUNDLE MECHANICS AND ELECTROMOTILITY IN THE COCHLEA
 451 THE INTERPLAY BETWEEN ACTIVE HAIR BUNDLE MECHANICS AND ELECTROMOTILITY IN THE COCHLEA DÁIBHID Ó MAOILÉIDIGH, FRANK JÜLICHER Max Planck Institute für Physik komplexer Systeme, Nöthnitzerstr. 38, 01187
451 THE INTERPLAY BETWEEN ACTIVE HAIR BUNDLE MECHANICS AND ELECTROMOTILITY IN THE COCHLEA DÁIBHID Ó MAOILÉIDIGH, FRANK JÜLICHER Max Planck Institute für Physik komplexer Systeme, Nöthnitzerstr. 38, 01187
Processing of sounds in the inner ear
 Processing of sounds in the inner ear Sripriya Ramamoorthy Associate Professor, IIT Bombay WiSSAP 2018 Cochlea converts sound into electrical signals [Picture courtesy of Northwestern University] von Bekesy
Processing of sounds in the inner ear Sripriya Ramamoorthy Associate Professor, IIT Bombay WiSSAP 2018 Cochlea converts sound into electrical signals [Picture courtesy of Northwestern University] von Bekesy
Half-Octave Shift in Mammalian Hearing Is an Epiphenomenon of the Cochlear Amplifier
 Half-Octave Shift in Mammalian Hearing Is an Epiphenomenon of the Cochlear Amplifier Sripriya Ramamoorthy 1 *, Alfred L. Nuttall 1,2 1 Oregon Hearing Research Center, Department of Otolaryngology, Oregon
Half-Octave Shift in Mammalian Hearing Is an Epiphenomenon of the Cochlear Amplifier Sripriya Ramamoorthy 1 *, Alfred L. Nuttall 1,2 1 Oregon Hearing Research Center, Department of Otolaryngology, Oregon
Effect of the Attachment of the Tectorial Membrane on Cochlear Micromechanics and Two-Tone Suppression
 1398 Biophysical Journal Volume 106 March 2014 1398 1405 Effect of the Attachment of the Tectorial Membrane on Cochlear Micromechanics and Two-Tone Suppression Julien Meaud * and Karl Grosh Department
1398 Biophysical Journal Volume 106 March 2014 1398 1405 Effect of the Attachment of the Tectorial Membrane on Cochlear Micromechanics and Two-Tone Suppression Julien Meaud * and Karl Grosh Department
HST 721 Lecture 4: Mechanics, electromotility and the cochlear amplifier
 HST 721 Lecture 4: Mechanics, electromotility and the cochlear amplifier 1 Cochlear Mechanics: Measures of Basilar Membrane Motion 2 Cochlear Mechanics: Measures of Basilar Membrane Motion Bekesy s experiments
HST 721 Lecture 4: Mechanics, electromotility and the cochlear amplifier 1 Cochlear Mechanics: Measures of Basilar Membrane Motion 2 Cochlear Mechanics: Measures of Basilar Membrane Motion Bekesy s experiments
Chapter 3: Anatomy and physiology of the sensory auditory mechanism
 Chapter 3: Anatomy and physiology of the sensory auditory mechanism Objectives (1) Anatomy of the inner ear Functions of the cochlear and vestibular systems Three compartments within the cochlea and membranes
Chapter 3: Anatomy and physiology of the sensory auditory mechanism Objectives (1) Anatomy of the inner ear Functions of the cochlear and vestibular systems Three compartments within the cochlea and membranes
Auditory System Feedback
 Feedback Auditory System Feedback Using all or a portion of the information from the output of a system to regulate or control the processes or inputs in order to modify the output. Central control of
Feedback Auditory System Feedback Using all or a portion of the information from the output of a system to regulate or control the processes or inputs in order to modify the output. Central control of
Mechanical Properties of the Cochlea. Reading: Yost Ch. 7
 Mechanical Properties of the Cochlea CF Reading: Yost Ch. 7 The Cochlea Inner ear contains auditory and vestibular sensory organs. Cochlea is a coiled tri-partite tube about 35 mm long. Basilar membrane,
Mechanical Properties of the Cochlea CF Reading: Yost Ch. 7 The Cochlea Inner ear contains auditory and vestibular sensory organs. Cochlea is a coiled tri-partite tube about 35 mm long. Basilar membrane,
Backward Propagation of Otoacoustic Emissions
 40 Review Backward Propagation of Otoacoustic Emissions HE Wenxuan, 1, 2 REN Tianying, 1, 2 1. Oregon Hearing Research Center, Department of Otolaryngology and Head & Neck Surgery, Oregon Health & Science
40 Review Backward Propagation of Otoacoustic Emissions HE Wenxuan, 1, 2 REN Tianying, 1, 2 1. Oregon Hearing Research Center, Department of Otolaryngology and Head & Neck Surgery, Oregon Health & Science
Improving the diagnostic power of otoacoustic emissions. Arturo Moleti Physics Department University of Roma Tor Vergata
 Improving the diagnostic power of otoacoustic emissions Arturo Moleti Physics Department University of Roma Tor Vergata The human ear Ear canal: resonant cavity Middle ear: impedance adapter and pressure
Improving the diagnostic power of otoacoustic emissions Arturo Moleti Physics Department University of Roma Tor Vergata The human ear Ear canal: resonant cavity Middle ear: impedance adapter and pressure
Cochlear anatomy, function and pathology II. Professor Dave Furness Keele University
 Cochlear anatomy, function and pathology II Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of this lecture Focus (2) on the biophysics of the cochlea, the dual roles
Cochlear anatomy, function and pathology II Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of this lecture Focus (2) on the biophysics of the cochlea, the dual roles
An active cochlear model showing sharp tuning and high sensitivity
 Hearing Research, 9 (1983) 123-13 Elsevier Biomedical Press 123 An active cochlear model showing sharp tuning and high sensitivity Stephen T. Neely * and D.O. Kim Box 811, Washington Unitlersit}', St.
Hearing Research, 9 (1983) 123-13 Elsevier Biomedical Press 123 An active cochlear model showing sharp tuning and high sensitivity Stephen T. Neely * and D.O. Kim Box 811, Washington Unitlersit}', St.
Comparing in vitro, in situ, and in vivo experimental data in a three-dimensional model of mammalian cochlear mechanics
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 3676 3681, March 1999 Biophysics Comparing in vitro, in situ, and in vivo experimental data in a three-dimensional model of mammalian cochlear mechanics PAUL J.
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 3676 3681, March 1999 Biophysics Comparing in vitro, in situ, and in vivo experimental data in a three-dimensional model of mammalian cochlear mechanics PAUL J.
In vivo outer hair cell length changes expose the active process in the cochlea
 In vivo outer hair cell length changes expose the active process in the cochlea Dingjun Zha, Fangyi Chen, Sripriya Ramamoorthy, Anders Fridberger, Niloy Choudhury, Steven L Jacques, Ruikang K Wang and
In vivo outer hair cell length changes expose the active process in the cochlea Dingjun Zha, Fangyi Chen, Sripriya Ramamoorthy, Anders Fridberger, Niloy Choudhury, Steven L Jacques, Ruikang K Wang and
Microstructures in the Organ of Corti Help Outer Hair Cells Form Traveling Waves along the Cochlear Coil
 2426 Biophysical Journal Volume 106 June 2014 2426 2433 Microstructures in the Organ of Corti Help Outer Hair Cells Form Traveling Waves along the Cochlear Coil Jong-Hoon Nam* Department of Mechanical
2426 Biophysical Journal Volume 106 June 2014 2426 2433 Microstructures in the Organ of Corti Help Outer Hair Cells Form Traveling Waves along the Cochlear Coil Jong-Hoon Nam* Department of Mechanical
Two-Tone Suppression of Simultaneous Electrical and Mechanical Responses in the Cochlea
 Article Two-Tone Suppression of Simultaneous Electrical and Mechanical Responses in the Cochlea Wei Dong and Elizabeth S. Olson 2, * VA Loma Linda Health Care System and Otolaryngology/Head & Neck Surgery,
Article Two-Tone Suppression of Simultaneous Electrical and Mechanical Responses in the Cochlea Wei Dong and Elizabeth S. Olson 2, * VA Loma Linda Health Care System and Otolaryngology/Head & Neck Surgery,
A truly remarkable aspect of human hearing is the vast
 AUDITORY COMPRESSION AND HEARING LOSS Sid P. Bacon Psychoacoustics Laboratory, Department of Speech and Hearing Science, Arizona State University Tempe, Arizona 85287 A truly remarkable aspect of human
AUDITORY COMPRESSION AND HEARING LOSS Sid P. Bacon Psychoacoustics Laboratory, Department of Speech and Hearing Science, Arizona State University Tempe, Arizona 85287 A truly remarkable aspect of human
FOUR COUNTER-ARGUMENTS FOR SLOW-WAVE OAEs
 FOUR COUNTER-ARGUMENTS FOR SLOW-WAVE OAEs CHRISTOPHER A. SHERA Eaton-Peabody Laboratory, Boston, MA 02114, USA email: shera@epl.meei.harvard.edu ARNOLD TUBIS Institute for Nonlinear Science, La Jolla,
FOUR COUNTER-ARGUMENTS FOR SLOW-WAVE OAEs CHRISTOPHER A. SHERA Eaton-Peabody Laboratory, Boston, MA 02114, USA email: shera@epl.meei.harvard.edu ARNOLD TUBIS Institute for Nonlinear Science, La Jolla,
Structure, Energy Transmission and Function. Gross Anatomy. Structure, Function & Process. External Auditory Meatus or Canal (EAM, EAC) Outer Ear
 Gross Anatomy Structure, Energy Transmission and Function IE N O ME 1 Structure, Function & Process 4 External Auditory Meatus or Canal (EAM, EAC) Outer third is cartilaginous Inner 2/3 is osseous Junction
Gross Anatomy Structure, Energy Transmission and Function IE N O ME 1 Structure, Function & Process 4 External Auditory Meatus or Canal (EAM, EAC) Outer third is cartilaginous Inner 2/3 is osseous Junction
In Vivo Impedance of the Gerbil Cochlear Partition at Auditory Frequencies
 Biophysical Journal Volume 97 September 2009 1233 1243 1233 In Vivo Impedance of the Gerbil Cochlear Partition at Auditory Frequencies Wei Dong * and Elizabeth S. Olson Department of Otolaryngology, Head
Biophysical Journal Volume 97 September 2009 1233 1243 1233 In Vivo Impedance of the Gerbil Cochlear Partition at Auditory Frequencies Wei Dong * and Elizabeth S. Olson Department of Otolaryngology, Head
HST 721 Efferent Control Lecture October 2004
 HST 721 Efferent Control Lecture October 2004 1 Stapedius Muscle Central Circuitry 2 Hypotheses for MEM Function A. Stapedius 1. Extend Dynamic Range - a gain control system 2. Protect the Inner Ear from
HST 721 Efferent Control Lecture October 2004 1 Stapedius Muscle Central Circuitry 2 Hypotheses for MEM Function A. Stapedius 1. Extend Dynamic Range - a gain control system 2. Protect the Inner Ear from
Auditory Physiology PSY 310 Greg Francis. Lecture 30. Organ of Corti
 Auditory Physiology PSY 310 Greg Francis Lecture 30 Waves, waves, waves. Organ of Corti Tectorial membrane Sits on top Inner hair cells Outer hair cells The microphone for the brain 1 Hearing Perceptually,
Auditory Physiology PSY 310 Greg Francis Lecture 30 Waves, waves, waves. Organ of Corti Tectorial membrane Sits on top Inner hair cells Outer hair cells The microphone for the brain 1 Hearing Perceptually,
Vibration of the organ of Corti within the cochlear apex in mice
 J Neurophysiol 2: 92 2, 2. First published June, 2; doi:.2/jn.3.2. Vibration of the organ of Corti within the cochlear apex in mice Simon S. Gao,,2 Rosalie Wang, Patrick D. Raphael, Yalda Moayedi, 3 ndrew
J Neurophysiol 2: 92 2, 2. First published June, 2; doi:.2/jn.3.2. Vibration of the organ of Corti within the cochlear apex in mice Simon S. Gao,,2 Rosalie Wang, Patrick D. Raphael, Yalda Moayedi, 3 ndrew
The Structure and Function of the Auditory Nerve
 The Structure and Function of the Auditory Nerve Brad May Structure and Function of the Auditory and Vestibular Systems (BME 580.626) September 21, 2010 1 Objectives Anatomy Basic response patterns Frequency
The Structure and Function of the Auditory Nerve Brad May Structure and Function of the Auditory and Vestibular Systems (BME 580.626) September 21, 2010 1 Objectives Anatomy Basic response patterns Frequency
Acoustics, signals & systems for audiology. Psychoacoustics of hearing impairment
 Acoustics, signals & systems for audiology Psychoacoustics of hearing impairment Three main types of hearing impairment Conductive Sound is not properly transmitted from the outer to the inner ear Sensorineural
Acoustics, signals & systems for audiology Psychoacoustics of hearing impairment Three main types of hearing impairment Conductive Sound is not properly transmitted from the outer to the inner ear Sensorineural
Systems Neuroscience Oct. 16, Auditory system. http:
 Systems Neuroscience Oct. 16, 2018 Auditory system http: www.ini.unizh.ch/~kiper/system_neurosci.html The physics of sound Measuring sound intensity We are sensitive to an enormous range of intensities,
Systems Neuroscience Oct. 16, 2018 Auditory system http: www.ini.unizh.ch/~kiper/system_neurosci.html The physics of sound Measuring sound intensity We are sensitive to an enormous range of intensities,
Academic and Research Staff Professor Dennis M. Freeman, Dr. Alexander Aranyosi, Dr. Stanley Hong
 Cochlear Mechanics Academic and Research Staff Professor Dennis M. Freeman, Dr. Alexander Aranyosi, Dr. Stanley Hong Graduate Students Chris Bergevin, Roozbeh Ghaffari, Wendy Gu, Kinuko Masaki, Scott Page
Cochlear Mechanics Academic and Research Staff Professor Dennis M. Freeman, Dr. Alexander Aranyosi, Dr. Stanley Hong Graduate Students Chris Bergevin, Roozbeh Ghaffari, Wendy Gu, Kinuko Masaki, Scott Page
sensitivity (db) frequency (khz) I: MONGOLIAN GERBIL FREQUENCY HEARING CHARACTERISTICS
 Supplemental materials to Frequency sensitivity in mammalian hearing from a fundamental nonlinear physics model of the inner ear, by Kanders, K., Lorimer, T., Gomez, F. & Stoop, R. -6-5 sensitivity (db)
Supplemental materials to Frequency sensitivity in mammalian hearing from a fundamental nonlinear physics model of the inner ear, by Kanders, K., Lorimer, T., Gomez, F. & Stoop, R. -6-5 sensitivity (db)
Chapter 11: Sound, The Auditory System, and Pitch Perception
 Chapter 11: Sound, The Auditory System, and Pitch Perception Overview of Questions What is it that makes sounds high pitched or low pitched? How do sound vibrations inside the ear lead to the perception
Chapter 11: Sound, The Auditory System, and Pitch Perception Overview of Questions What is it that makes sounds high pitched or low pitched? How do sound vibrations inside the ear lead to the perception
OtoAcoustic Emissions (OAE s)
 OtoAcoustic Emissions (OAE s) Phenomenon and applications in audiological diagnostics Measurement procedures TEOAE and DPOAE Physiological backgound, functional models Acknowledgment: several illustrations
OtoAcoustic Emissions (OAE s) Phenomenon and applications in audiological diagnostics Measurement procedures TEOAE and DPOAE Physiological backgound, functional models Acknowledgment: several illustrations
Auditory System. Barb Rohrer (SEI )
 Auditory System Barb Rohrer (SEI614 2-5086) Sounds arise from mechanical vibration (creating zones of compression and rarefaction; which ripple outwards) Transmitted through gaseous, aqueous or solid medium
Auditory System Barb Rohrer (SEI614 2-5086) Sounds arise from mechanical vibration (creating zones of compression and rarefaction; which ripple outwards) Transmitted through gaseous, aqueous or solid medium
FIFTH INTERNATIONAL CONGRESS ON SOUND AND VIBRATION DECEMBER 15-18, 1997 ADELAIDE, SOUTH AUSTRALIA. Invited Paper LINEAR RESPONSE OF THE COCHLEA
 FIFTH INTERNATIONAL CONGRESS ON SOUND AND VIBRATION DECEMBER 15-18, 1997 ADELAIDE, SOUTH AUSTRALIA Invited Paper LINEAR RESPONSE OF THE COCHLEA David Alan Bies Visiting Research Fellow Department of Mechanical
FIFTH INTERNATIONAL CONGRESS ON SOUND AND VIBRATION DECEMBER 15-18, 1997 ADELAIDE, SOUTH AUSTRALIA Invited Paper LINEAR RESPONSE OF THE COCHLEA David Alan Bies Visiting Research Fellow Department of Mechanical
Representation of sound in the auditory nerve
 Representation of sound in the auditory nerve Eric D. Young Department of Biomedical Engineering Johns Hopkins University Young, ED. Neural representation of spectral and temporal information in speech.
Representation of sound in the auditory nerve Eric D. Young Department of Biomedical Engineering Johns Hopkins University Young, ED. Neural representation of spectral and temporal information in speech.
Detection of Cochlear Amplification and Its Activation
 Biophysical Journal Volume 5 August 23 67 78 67 Detection of Cochlear Amplification and Its Activation Wei Dong and Elizabeth S. Olson * Otolaryngology, Head and Neck Surgery and Biomedical Engineering,
Biophysical Journal Volume 5 August 23 67 78 67 Detection of Cochlear Amplification and Its Activation Wei Dong and Elizabeth S. Olson * Otolaryngology, Head and Neck Surgery and Biomedical Engineering,
ENT 318 Artificial Organs Physiology of Ear
 ENT 318 Artificial Organs Physiology of Ear Lecturer: Ahmad Nasrul Norali The Ear The Ear Components of hearing mechanism - Outer Ear - Middle Ear - Inner Ear - Central Auditory Nervous System Major Divisions
ENT 318 Artificial Organs Physiology of Ear Lecturer: Ahmad Nasrul Norali The Ear The Ear Components of hearing mechanism - Outer Ear - Middle Ear - Inner Ear - Central Auditory Nervous System Major Divisions
Harmonic distortion on the basilar membrane in the basal turn of the guinea-pig cochlea
 Keywords: Cochlea, Basilar membrane, Hair cell 7006 Journal of Physiology (1998), 509.1, pp. 277 288 277 Harmonic distortion on the basilar membrane in the basal turn of the guinea-pig cochlea Nigel P.
Keywords: Cochlea, Basilar membrane, Hair cell 7006 Journal of Physiology (1998), 509.1, pp. 277 288 277 Harmonic distortion on the basilar membrane in the basal turn of the guinea-pig cochlea Nigel P.
Cochlear anatomy, function and pathology I. Professor Dave Furness Keele University
 Cochlear anatomy, function and pathology I Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of these lectures Introduction to gross anatomy of the cochlea Focus (1) on
Cochlear anatomy, function and pathology I Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of these lectures Introduction to gross anatomy of the cochlea Focus (1) on
Feed-Forward and Feed-Backward Amplification Model from Cochlear Cytoarchitecture: An Interspecies Comparison
 Biophysical Journal Volume 100 January 2011 1 10 1 Feed-Forward and Feed-Backward Amplification Model from Cochlear Cytoarchitecture: An Interspecies Comparison Yong-Jin Yoon, Charles R. Steele, and Sunil
Biophysical Journal Volume 100 January 2011 1 10 1 Feed-Forward and Feed-Backward Amplification Model from Cochlear Cytoarchitecture: An Interspecies Comparison Yong-Jin Yoon, Charles R. Steele, and Sunil
Application of force to the cochlear wall: effect on auditory thresholds, OHC transduction, and DPOAEs
 Application of force to the cochlear wall: effect on auditory thresholds, OHC transduction, and DPOAEs The Auditory Laboratory, Physiology University of Western Australia Greg O Beirne Dept. of Communication
Application of force to the cochlear wall: effect on auditory thresholds, OHC transduction, and DPOAEs The Auditory Laboratory, Physiology University of Western Australia Greg O Beirne Dept. of Communication
Advanced otoacoustic emission detection techniques and clinical diagnostics applications
 Advanced otoacoustic emission detection techniques and clinical diagnostics applications Arturo Moleti Physics Department, University of Roma Tor Vergata, Roma, ITALY Towards objective diagnostics of human
Advanced otoacoustic emission detection techniques and clinical diagnostics applications Arturo Moleti Physics Department, University of Roma Tor Vergata, Roma, ITALY Towards objective diagnostics of human
Auditory Physiology Richard M. Costanzo, Ph.D.
 Auditory Physiology Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture, the student should be able to: 1. Describe the morphology and function of the following structures:
Auditory Physiology Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture, the student should be able to: 1. Describe the morphology and function of the following structures:
SOLUTIONS Homework #3. Introduction to Engineering in Medicine and Biology ECEN 1001 Due Tues. 9/30/03
 SOLUTIONS Homework #3 Introduction to Engineering in Medicine and Biology ECEN 1001 Due Tues. 9/30/03 Problem 1: a) Where in the cochlea would you say the process of "fourier decomposition" of the incoming
SOLUTIONS Homework #3 Introduction to Engineering in Medicine and Biology ECEN 1001 Due Tues. 9/30/03 Problem 1: a) Where in the cochlea would you say the process of "fourier decomposition" of the incoming
Signals, systems, acoustics and the ear. Week 5. The peripheral auditory system: The ear as a signal processor
 Signals, systems, acoustics and the ear Week 5 The peripheral auditory system: The ear as a signal processor Think of this set of organs 2 as a collection of systems, transforming sounds to be sent to
Signals, systems, acoustics and the ear Week 5 The peripheral auditory system: The ear as a signal processor Think of this set of organs 2 as a collection of systems, transforming sounds to be sent to
Force Transmission in the Organ of Corti Micromachine
 Biophysical Journal Volume 98 June 2010 2813 2821 2813 Force Transmission in the Organ of Corti Micromachine Jong-Hoon Nam and Robert Fettiplace* Department of Physiology, University of Wisconsin Medical
Biophysical Journal Volume 98 June 2010 2813 2821 2813 Force Transmission in the Organ of Corti Micromachine Jong-Hoon Nam and Robert Fettiplace* Department of Physiology, University of Wisconsin Medical
Study the Effect of the Quality Factor of the Transient Evoked Oto-acoustic Emissions (TEOAE)
 Research in Otolaryngology 217, 6(4): 47-4 DOI:.923/j.otolaryn.21764.1 Study the Effect of the Quality Factor of the Transient Evoked Oto-acoustic Emissions (TEOAE) Adnan AL-Maamury *, Dhifaf Ahmed Al-Mustansiriyah
Research in Otolaryngology 217, 6(4): 47-4 DOI:.923/j.otolaryn.21764.1 Study the Effect of the Quality Factor of the Transient Evoked Oto-acoustic Emissions (TEOAE) Adnan AL-Maamury *, Dhifaf Ahmed Al-Mustansiriyah
Auditory Periphery! external middle inner. stapes movement initiates a pressure wave in cochlear fluid
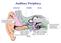 Auditory Periphery! external middle inner sound causes air pressure to increase at eardrum stapes movement initiates a pressure wave in cochlear fluid VIIIth nerve conveys neural signal to cochlear nucleus
Auditory Periphery! external middle inner sound causes air pressure to increase at eardrum stapes movement initiates a pressure wave in cochlear fluid VIIIth nerve conveys neural signal to cochlear nucleus
Acoustics Research Institute
 Austrian Academy of Sciences Acoustics Research Institute Modeling Modelingof ofauditory AuditoryPerception Perception Bernhard BernhardLaback Labackand andpiotr PiotrMajdak Majdak http://www.kfs.oeaw.ac.at
Austrian Academy of Sciences Acoustics Research Institute Modeling Modelingof ofauditory AuditoryPerception Perception Bernhard BernhardLaback Labackand andpiotr PiotrMajdak Majdak http://www.kfs.oeaw.ac.at
Auditory nerve model for predicting performance limits of normal and impaired listeners
 Heinz et al.: Acoustics Research Letters Online [DOI 1.1121/1.1387155] Published Online 12 June 21 Auditory nerve model for predicting performance limits of normal and impaired listeners Michael G. Heinz
Heinz et al.: Acoustics Research Letters Online [DOI 1.1121/1.1387155] Published Online 12 June 21 Auditory nerve model for predicting performance limits of normal and impaired listeners Michael G. Heinz
Deafness and hearing impairment
 Auditory Physiology Deafness and hearing impairment About one in every 10 Americans has some degree of hearing loss. The great majority develop hearing loss as they age. Hearing impairment in very early
Auditory Physiology Deafness and hearing impairment About one in every 10 Americans has some degree of hearing loss. The great majority develop hearing loss as they age. Hearing impairment in very early
external middle inner
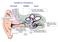 Auditory Periphery external middle inner 3. movement of stapes initiates a pressure wave in cochlear fluid. 1. sound causes air pressure to increase at eardrum 5. auditory nerve conveys neural signal to
Auditory Periphery external middle inner 3. movement of stapes initiates a pressure wave in cochlear fluid. 1. sound causes air pressure to increase at eardrum 5. auditory nerve conveys neural signal to
Effects of Remaining Hair Cells on Cochlear Implant Function
 Effects of Remaining Hair Cells on Cochlear Implant Function N1-DC-2-15QPR1 Neural Prosthesis Program N. Hu, P.J. Abbas, C.A. Miller, B.K. Robinson, K.V. Nourski, F. Jeng, B.A. Abkes, J.M. Nichols Department
Effects of Remaining Hair Cells on Cochlear Implant Function N1-DC-2-15QPR1 Neural Prosthesis Program N. Hu, P.J. Abbas, C.A. Miller, B.K. Robinson, K.V. Nourski, F. Jeng, B.A. Abkes, J.M. Nichols Department
FAST MOTILITY OF ISOLATED MAMMALIAN AUDITORY SENSORY CELLS. Hans P. Zenner, Ulrike Zimmermann and Alfred H. Gitter
 Vol. 149,No. 1, 1987 November 30, 1987 BlOCHEMlCALANDBlOPHYSlCALRESEARCHCOMMUNlCATlONS Pages 304-308 FAST MOTILITY OF ISOLATED MAMMALIAN AUDITORY SENSORY CELLS Hans P. Zenner, Ulrike Zimmermann and Alfred
Vol. 149,No. 1, 1987 November 30, 1987 BlOCHEMlCALANDBlOPHYSlCALRESEARCHCOMMUNlCATlONS Pages 304-308 FAST MOTILITY OF ISOLATED MAMMALIAN AUDITORY SENSORY CELLS Hans P. Zenner, Ulrike Zimmermann and Alfred
THE COCHLEAR AMPLIFIER: IS IT HAIR BUNDLE MOTION OF OUTER HAIR CELLS?
 JOURNAL OF OTOLOGY THE COCHLEAR AMPLIFIER: IS IT HAIR BUNDLE MOTION OF OUTER HAIR CELLS? LI Yi 1, He David Z 2 Abstract Cochlear outer hair cells (OHCs) are involved in a mechanical feedback loop in which
JOURNAL OF OTOLOGY THE COCHLEAR AMPLIFIER: IS IT HAIR BUNDLE MOTION OF OUTER HAIR CELLS? LI Yi 1, He David Z 2 Abstract Cochlear outer hair cells (OHCs) are involved in a mechanical feedback loop in which
Required Slide. Session Objectives
 Auditory Physiology Required Slide Session Objectives Auditory System: At the end of this session, students will be able to: 1. Characterize the range of normal human hearing. 2. Understand the components
Auditory Physiology Required Slide Session Objectives Auditory System: At the end of this session, students will be able to: 1. Characterize the range of normal human hearing. 2. Understand the components
What Drives Mechanical Amplification in the Mammalian Cochlea?
 What Drives Mechanical Amplification in the Mammalian Cochlea? Robert H. Withnell, Lauren A. Shaffer, and David J. Lilly The recent report by Peter Dallos and colleagues of the gene and protein responsible
What Drives Mechanical Amplification in the Mammalian Cochlea? Robert H. Withnell, Lauren A. Shaffer, and David J. Lilly The recent report by Peter Dallos and colleagues of the gene and protein responsible
Educational Module Tympanometry. Germany D Germering
 Educational Module anometry PATH medical Germany D-82110 Germering Our educational modules 1 are made for providing information on how the hearing organ works and which test procedures are used to test
Educational Module anometry PATH medical Germany D-82110 Germering Our educational modules 1 are made for providing information on how the hearing organ works and which test procedures are used to test
I. INTRODUCTION. J. Acoust. Soc. Am. 111 (1), Pt. 1, Jan /2002/111(1)/271/14/$ Acoustical Society of America
 The use of distortion product otoacoustic emission suppression as an estimate of response growth Michael P. Gorga, a) Stephen T. Neely, Patricia A. Dorn, and Dawn Konrad-Martin Boys Town National Research
The use of distortion product otoacoustic emission suppression as an estimate of response growth Michael P. Gorga, a) Stephen T. Neely, Patricia A. Dorn, and Dawn Konrad-Martin Boys Town National Research
Sound and its characteristics. The decibel scale. Structure and function of the ear. Békésy s theory. Molecular basis of hair cell function.
 Hearing Sound and its characteristics. The decibel scale. Structure and function of the ear. Békésy s theory. Molecular basis of hair cell function. 19/11/2014 Sound A type of longitudinal mass wave that
Hearing Sound and its characteristics. The decibel scale. Structure and function of the ear. Békésy s theory. Molecular basis of hair cell function. 19/11/2014 Sound A type of longitudinal mass wave that
The magnificent outer hair cell of Corti's organ
 The magnificent outer hair cell of Corti's organ Yale University J. Santos-Sacchi School of Medicine, New Haver, CT, USA Abstract The outer hair cell is one of two receptor cell types in the organ of Corti.
The magnificent outer hair cell of Corti's organ Yale University J. Santos-Sacchi School of Medicine, New Haver, CT, USA Abstract The outer hair cell is one of two receptor cell types in the organ of Corti.
Hearing Sound. The Human Auditory System. The Outer Ear. Music 170: The Ear
 Hearing Sound Music 170: The Ear Tamara Smyth, trsmyth@ucsd.edu Department of Music, University of California, San Diego (UCSD) November 17, 2016 Sound interpretation in the auditory system is done by
Hearing Sound Music 170: The Ear Tamara Smyth, trsmyth@ucsd.edu Department of Music, University of California, San Diego (UCSD) November 17, 2016 Sound interpretation in the auditory system is done by
Music 170: The Ear. Tamara Smyth, Department of Music, University of California, San Diego (UCSD) November 17, 2016
 Music 170: The Ear Tamara Smyth, trsmyth@ucsd.edu Department of Music, University of California, San Diego (UCSD) November 17, 2016 1 Hearing Sound Sound interpretation in the auditory system is done by
Music 170: The Ear Tamara Smyth, trsmyth@ucsd.edu Department of Music, University of California, San Diego (UCSD) November 17, 2016 1 Hearing Sound Sound interpretation in the auditory system is done by
Physiological basis of sound design. Prof. Dr. med. Eckhard Hoffmann Dipl.-Ing. (FH) Steffen Kreikemeier Aalen University of Applied Sciences
 Physiological basis of sound design Prof. Dr. med. Eckhard Hoffmann Dipl.-Ing. (FH) Steffen Kreikemeier Aalen University of Applied Sciences Index of contents Physiological basis of the inner ear Organ
Physiological basis of sound design Prof. Dr. med. Eckhard Hoffmann Dipl.-Ing. (FH) Steffen Kreikemeier Aalen University of Applied Sciences Index of contents Physiological basis of the inner ear Organ
Efferent-mediated control of basilar membrane motion
 J Physiol 576. (2006) pp 49 54 49 Topical Review Efferent-mediated control of basilar membrane motion N. P. Cooper and J. J. Guinan Jr 2 School of Life Sciences, Keele University, Keele, Staffordshire
J Physiol 576. (2006) pp 49 54 49 Topical Review Efferent-mediated control of basilar membrane motion N. P. Cooper and J. J. Guinan Jr 2 School of Life Sciences, Keele University, Keele, Staffordshire
Trajectory of the Aging Cochlea
 Trajectory of the Aging Cochlea Sumitrajit (Sumit) Dhar Professor & Chair Roxelyn & Richard Pepper Department of Communication Sciences and Disorders Fellow, Hugh Knowles Center for Hearing Science Northwestern
Trajectory of the Aging Cochlea Sumitrajit (Sumit) Dhar Professor & Chair Roxelyn & Richard Pepper Department of Communication Sciences and Disorders Fellow, Hugh Knowles Center for Hearing Science Northwestern
A cochlear AGC model, proposing a new type of cochlear non-linearity
 TEL-AVIV UNIVERSITY The Iby and Aladar Fleischman faculty of engineering The Zandman-Slaner school of Graduate studies A cochlear AGC model, proposing a new type of cochlear non-linearity A thesis submitted
TEL-AVIV UNIVERSITY The Iby and Aladar Fleischman faculty of engineering The Zandman-Slaner school of Graduate studies A cochlear AGC model, proposing a new type of cochlear non-linearity A thesis submitted
The frequency analysis of the cochlea a review of Nobili et al (1998) and Ruggero et al (1992)
 The frequency analysis of the cochlea a review of Nobili et al (1998) and Ruggero et al (1992) by Pedro da Fonseca (pedrofon@mail.telepac.pt) Neuroscience course Presented in 17.12.99 to professor STEPHEN
The frequency analysis of the cochlea a review of Nobili et al (1998) and Ruggero et al (1992) by Pedro da Fonseca (pedrofon@mail.telepac.pt) Neuroscience course Presented in 17.12.99 to professor STEPHEN
Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH).
 Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH). (a), (b), PRPH immunolabelling of cryosections from post-natal day
Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH). (a), (b), PRPH immunolabelling of cryosections from post-natal day
EMANATIONS FROM RESIDUUM OSCILLATIONS IN HUMAN AUDITORY SYSTEM
 EMANATIONS FROM RESIDUUM OSCILLATIONS IN HUMAN AUDITORY SYSTEM V.S. Balaji, N.R.Raajan, S. Rakesh Kumar, Har Narayan Upadhyay School of Electrical & Electronics Engineering, SASTRA University Thanjavur,
EMANATIONS FROM RESIDUUM OSCILLATIONS IN HUMAN AUDITORY SYSTEM V.S. Balaji, N.R.Raajan, S. Rakesh Kumar, Har Narayan Upadhyay School of Electrical & Electronics Engineering, SASTRA University Thanjavur,
Can components in distortion-product otoacoustic emissions be separated?
 Can components in distortion-product otoacoustic emissions be separated? Anders Tornvig Section of Acoustics, Aalborg University, Fredrik Bajers Vej 7 B5, DK-922 Aalborg Ø, Denmark, tornvig@es.aau.dk David
Can components in distortion-product otoacoustic emissions be separated? Anders Tornvig Section of Acoustics, Aalborg University, Fredrik Bajers Vej 7 B5, DK-922 Aalborg Ø, Denmark, tornvig@es.aau.dk David
Otoacoustic Estimates of Cochlear Tuning: Testing Predictions in Macaque
 Otoacoustic Estimates of Cochlear Tuning: Testing Predictions in Macaque Christopher A. Shera, Christopher Bergevin, Radha Kalluri, Myles Mc Laughlin, Pascal Michelet, Marcel van der Heijden, and Philip
Otoacoustic Estimates of Cochlear Tuning: Testing Predictions in Macaque Christopher A. Shera, Christopher Bergevin, Radha Kalluri, Myles Mc Laughlin, Pascal Michelet, Marcel van der Heijden, and Philip
Modelling the micromechanics of the cochlea in Femlab
 Modelling the micromechanics of the cochlea in Femlab R.R.J.J. van Doorn DCT 27.7 Traineeship report Coach(es): Supervisor: Prof. S.J. Elliott Prof. P. Gardonio Prof. H. Nijmeijer Technische Universiteit
Modelling the micromechanics of the cochlea in Femlab R.R.J.J. van Doorn DCT 27.7 Traineeship report Coach(es): Supervisor: Prof. S.J. Elliott Prof. P. Gardonio Prof. H. Nijmeijer Technische Universiteit
MECHANISM OF HEARING
 MECHANISM OF HEARING Sound: Sound is a vibration that propagates as an audible wave of pressure, through a transmission medium such as gas, liquid or solid. Sound is produced from alternate compression
MECHANISM OF HEARING Sound: Sound is a vibration that propagates as an audible wave of pressure, through a transmission medium such as gas, liquid or solid. Sound is produced from alternate compression
What does it mean to analyze the frequency components of a sound? A spectrogram such as that shown here is the usual display of frequency components
 1 2 3 4 What does it mean to analyze the frequency components of a sound? A spectrogram such as that shown here is the usual display of frequency components as a function of time here during the production
1 2 3 4 What does it mean to analyze the frequency components of a sound? A spectrogram such as that shown here is the usual display of frequency components as a function of time here during the production
Effects of Medial Olivocochlear Auditory Reflex Activation on Cochlear Vibration
 University of Iowa Honors Theses University of Iowa Honors Program Spring 2017 Effects of Medial Olivocochlear Auditory Reflex Activation on Cochlear Vibration Alexandra Redfern Shawn Goodman University
University of Iowa Honors Theses University of Iowa Honors Program Spring 2017 Effects of Medial Olivocochlear Auditory Reflex Activation on Cochlear Vibration Alexandra Redfern Shawn Goodman University
Receptors / physiology
 Hearing: physiology Receptors / physiology Energy transduction First goal of a sensory/perceptual system? Transduce environmental energy into neural energy (or energy that can be interpreted by perceptual
Hearing: physiology Receptors / physiology Energy transduction First goal of a sensory/perceptual system? Transduce environmental energy into neural energy (or energy that can be interpreted by perceptual
A Review of the Effectiveness of Otoacoustic Emissions for Evaluating Hearing Status After Newborn Screening
 Otology & Neurotology 34:1058Y1063 Ó 2013, Otology & Neurotology, Inc. A Review of the Effectiveness of Otoacoustic Emissions for Evaluating Hearing Status After Newborn Screening Thomas Janssen ENT-Department,
Otology & Neurotology 34:1058Y1063 Ó 2013, Otology & Neurotology, Inc. A Review of the Effectiveness of Otoacoustic Emissions for Evaluating Hearing Status After Newborn Screening Thomas Janssen ENT-Department,
Evidence and synthesis
 CHAPTER D 9 (Discussion continued) Evidence and synthesis 9.1 Synthesis 9.1/a Outer hair cells as dual detectors Pressure detection and transduction currents The silent current 9.1/b Cancellation effects
CHAPTER D 9 (Discussion continued) Evidence and synthesis 9.1 Synthesis 9.1/a Outer hair cells as dual detectors Pressure detection and transduction currents The silent current 9.1/b Cancellation effects
Synaptopathy Research Uwe Andreas Hermann
 Potential diagnose and basic understanding of hidden hearing loss Synaptopathy Research Uwe Andreas Hermann Motivation Synaptopathy is a current hot topic in the research field because it focuses on a
Potential diagnose and basic understanding of hidden hearing loss Synaptopathy Research Uwe Andreas Hermann Motivation Synaptopathy is a current hot topic in the research field because it focuses on a
Clinical applications of otoacoustic emissions in Industry. Prof. Dr. B. Vinck, MSc, PhD University of Ghent, Belgium
 Clinical applications of otoacoustic emissions in Industry Prof. Dr. B. Vinck, MSc, PhD University of Ghent, Belgium The 25th anniversary of the discovery of otoacoustic emissions (OAEs) [sounds that can
Clinical applications of otoacoustic emissions in Industry Prof. Dr. B. Vinck, MSc, PhD University of Ghent, Belgium The 25th anniversary of the discovery of otoacoustic emissions (OAEs) [sounds that can
Frequency refers to how often something happens. Period refers to the time it takes something to happen.
 Lecture 2 Properties of Waves Frequency and period are distinctly different, yet related, quantities. Frequency refers to how often something happens. Period refers to the time it takes something to happen.
Lecture 2 Properties of Waves Frequency and period are distinctly different, yet related, quantities. Frequency refers to how often something happens. Period refers to the time it takes something to happen.
Chapter 17, Part 2! The Special Senses! Hearing and Equilibrium!
 Chapter 17, Part 2! The Special Senses! Hearing and Equilibrium! SECTION 17-5! Equilibrium sensations originate within the inner ear, while hearing involves the detection and interpretation of sound waves!
Chapter 17, Part 2! The Special Senses! Hearing and Equilibrium! SECTION 17-5! Equilibrium sensations originate within the inner ear, while hearing involves the detection and interpretation of sound waves!
Chapter 17, Part 2! Chapter 17 Part 2 Special Senses! The Special Senses! Hearing and Equilibrium!
 Chapter 17, Part 2! The Special Senses! Hearing and Equilibrium! SECTION 17-5! Equilibrium sensations originate within the inner ear, while hearing involves the detection and interpretation of sound waves!
Chapter 17, Part 2! The Special Senses! Hearing and Equilibrium! SECTION 17-5! Equilibrium sensations originate within the inner ear, while hearing involves the detection and interpretation of sound waves!
I. INTRODUCTION.
 Inner hair cell response patterns: Implications for low-frequency hearing M. A. Cheatham a) and P. Dallos Audiology and Hearing Sciences, Communication Sciences and Disorders, The Hugh Knowles Center,
Inner hair cell response patterns: Implications for low-frequency hearing M. A. Cheatham a) and P. Dallos Audiology and Hearing Sciences, Communication Sciences and Disorders, The Hugh Knowles Center,
Sound Waves. Sensation and Perception. Sound Waves. Sound Waves. Sound Waves
 Sensation and Perception Part 3 - Hearing Sound comes from pressure waves in a medium (e.g., solid, liquid, gas). Although we usually hear sounds in air, as long as the medium is there to transmit the
Sensation and Perception Part 3 - Hearing Sound comes from pressure waves in a medium (e.g., solid, liquid, gas). Although we usually hear sounds in air, as long as the medium is there to transmit the
Unit VIII Problem 9 Physiology: Hearing
 Unit VIII Problem 9 Physiology: Hearing - We can hear a limited range of frequency between 20 Hz 20,000 Hz (human hearing acuity is between 1000 Hz 4000 Hz). - The ear is divided into 3 parts. Those are:
Unit VIII Problem 9 Physiology: Hearing - We can hear a limited range of frequency between 20 Hz 20,000 Hz (human hearing acuity is between 1000 Hz 4000 Hz). - The ear is divided into 3 parts. Those are:
Intro to Audition & Hearing
 Intro to Audition & Hearing Lecture 16 Chapter 9, part II Jonathan Pillow Sensation & Perception (PSY 345 / NEU 325) Fall 2017 1 Sine wave: one of the simplest kinds of sounds: sound for which pressure
Intro to Audition & Hearing Lecture 16 Chapter 9, part II Jonathan Pillow Sensation & Perception (PSY 345 / NEU 325) Fall 2017 1 Sine wave: one of the simplest kinds of sounds: sound for which pressure
Chapter 13 Physics of the Ear and Hearing
 Hearing 100 times greater dynamic range than vision Wide frequency range (20 ~ 20,000 Hz) Sense of hearing Mechanical system that stimulates the hair cells in the cochlea Sensors that produce action potentials
Hearing 100 times greater dynamic range than vision Wide frequency range (20 ~ 20,000 Hz) Sense of hearing Mechanical system that stimulates the hair cells in the cochlea Sensors that produce action potentials
Outer Hair Cells in the Cochlea
 Discrete Dynamics in Nature and Society, Vol. 2, pp. 161-165 Reprints available directly from the publisher Photocopying permitted by license only (C) 1998 OPA (Overseas Publishers Association) N.V. Published
Discrete Dynamics in Nature and Society, Vol. 2, pp. 161-165 Reprints available directly from the publisher Photocopying permitted by license only (C) 1998 OPA (Overseas Publishers Association) N.V. Published
Lecture 3: Perception
 ELEN E4896 MUSIC SIGNAL PROCESSING Lecture 3: Perception 1. Ear Physiology 2. Auditory Psychophysics 3. Pitch Perception 4. Music Perception Dan Ellis Dept. Electrical Engineering, Columbia University
ELEN E4896 MUSIC SIGNAL PROCESSING Lecture 3: Perception 1. Ear Physiology 2. Auditory Psychophysics 3. Pitch Perception 4. Music Perception Dan Ellis Dept. Electrical Engineering, Columbia University
Issues faced by people with a Sensorineural Hearing Loss
 Issues faced by people with a Sensorineural Hearing Loss Issues faced by people with a Sensorineural Hearing Loss 1. Decreased Audibility 2. Decreased Dynamic Range 3. Decreased Frequency Resolution 4.
Issues faced by people with a Sensorineural Hearing Loss Issues faced by people with a Sensorineural Hearing Loss 1. Decreased Audibility 2. Decreased Dynamic Range 3. Decreased Frequency Resolution 4.
Auditory Physiology PSY 310 Greg Francis. Lecture 29. Hearing
 Auditory Physiology PSY 310 Greg Francis Lecture 29 A dangerous device. Hearing The sound stimulus is changes in pressure The simplest sounds vary in: Frequency: Hertz, cycles per second. How fast the
Auditory Physiology PSY 310 Greg Francis Lecture 29 A dangerous device. Hearing The sound stimulus is changes in pressure The simplest sounds vary in: Frequency: Hertz, cycles per second. How fast the
PSY 310: Sensory and Perceptual Processes 1
 Auditory Physiology PSY 310 Greg Francis Lecture 29 A dangerous device. Hearing The sound stimulus is changes in pressure The simplest sounds vary in: Frequency: Hertz, cycles per second. How fast the
Auditory Physiology PSY 310 Greg Francis Lecture 29 A dangerous device. Hearing The sound stimulus is changes in pressure The simplest sounds vary in: Frequency: Hertz, cycles per second. How fast the
9.01 Introduction to Neuroscience Fall 2007
 MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
Distortion product emissions from a cochlear model with nonlinear mechanoelectrical transduction in outer hair cells
 Distortion product emissions from a cochlear model with nonlinear mechanoelectrical transduction in outer hair cells Yi-Wen Liu and Stephen T. Neely Boys Town National Research Hospital, 555 North 3th
Distortion product emissions from a cochlear model with nonlinear mechanoelectrical transduction in outer hair cells Yi-Wen Liu and Stephen T. Neely Boys Town National Research Hospital, 555 North 3th
A computer model of medial efferent suppression in the mammalian auditory system
 A computer model of medial efferent suppression in the mammalian auditory system Robert T. Ferry a and Ray Meddis Department of Psychology, University of Essex, Colchester, CO4 3SQ, United Kingdom Received
A computer model of medial efferent suppression in the mammalian auditory system Robert T. Ferry a and Ray Meddis Department of Psychology, University of Essex, Colchester, CO4 3SQ, United Kingdom Received
HEARING AND PSYCHOACOUSTICS
 CHAPTER 2 HEARING AND PSYCHOACOUSTICS WITH LIDIA LEE I would like to lead off the specific audio discussions with a description of the audio receptor the ear. I believe it is always a good idea to understand
CHAPTER 2 HEARING AND PSYCHOACOUSTICS WITH LIDIA LEE I would like to lead off the specific audio discussions with a description of the audio receptor the ear. I believe it is always a good idea to understand
Sound and Hearing. Decibels. Frequency Coding & Localization 1. Everything is vibration. The universe is made of waves.
 Frequency Coding & Localization 1 Sound and Hearing Everything is vibration The universe is made of waves db = 2log(P1/Po) P1 = amplitude of the sound wave Po = reference pressure =.2 dynes/cm 2 Decibels
Frequency Coding & Localization 1 Sound and Hearing Everything is vibration The universe is made of waves db = 2log(P1/Po) P1 = amplitude of the sound wave Po = reference pressure =.2 dynes/cm 2 Decibels
Can You Hear Me Now?
 An Introduction to the Mathematics of Hearing Department of Applied Mathematics University of Washington April 26, 2007 Some Questions How does hearing work? What are the important structures and mechanisms
An Introduction to the Mathematics of Hearing Department of Applied Mathematics University of Washington April 26, 2007 Some Questions How does hearing work? What are the important structures and mechanisms
SPONTANEOUS OSCILLATIONS IN HAIR CELLS OF THE BULLFROG SACCULUS
 SPONTANEOUS OSCILLATIONS IN HAIR CELLS OF THE BULLFROG SACCULUS Katherine Knisely, D. Bozovic University of California at Los Angeles It has long been observed that there is some form of amplification
SPONTANEOUS OSCILLATIONS IN HAIR CELLS OF THE BULLFROG SACCULUS Katherine Knisely, D. Bozovic University of California at Los Angeles It has long been observed that there is some form of amplification
