Neurochemical mechanism of the gastrointestinal interdigestive migrating motor complex in rats with acute inflammatory stomach ache
|
|
|
- Reynold Campbell
- 5 years ago
- Views:
Transcription
1 NEURL REGENERTION RESERCH Volume 7, Issue 27, September doi: /j.issn [ Xu XL, Li Q, Zhou L, Ru LQ. Neurochemical mechanism of the gastrointestinal interdigestive migrating motor complex in rats with acute inflammatory stomach ache. Neural Regen Res. 2012;7(27): Neurochemical mechanism of the gastrointestinal interdigestive migrating motor complex in rats with acute inflammatory stomach ache Xiaoli Xu 1, Qin Li 2, Lv Zhou 3, Liqiang Ru 2 1 Department of natomy, School of Medicine, Jianghan University, Wuhan , Hubei Province, China 2 Department of Neurobiology, Tongji Medical College, Huazhong University of Science and Technology, Wuhan , Hubei Province, China 3 Department of Physiology, Institute of asic Medical Science, Chinese cademy of Medical Science, eijing , China bstract The normal gastrointestinal interdigestive migrating motor complex cycle was interrupted, and paroxysmal contraction appeared after formaldehyde-induced stomach ache. ctivities of nitric oxide synthase, acetylcholinesterase and vasoactive intestinal peptide neurons were significantly reduced, whereas activities of calcitonin gene-related peptide neurons were significantly increased in the pyloric sphincter muscular layer, myenteric nerve plexus and submucous nerve plexus. Electroacupuncture at Zusanli (ST36) suppressed paroxysmal contraction in rats with formaldehyde-induced stomach ache, and neurons in the enteric nervous system were normal. These results indicated that nitrergic neurons, cholinergic neurons, vasoactive intestinal peptide neurons and calcitonin gene-related peptide neurons in the enteric nervous system may be involved in changes to the gastrointestinal interdigestive migrating motor complex following stomach ache, and that electroacupuncture can regulate this process. Key Words pyloric sphincter; inflammatory pain; interdigestive migrating motor complex; enteric nervous system; electroacupuncture; neural regeneration Xiaoli Xu, ssociate professor, Department of natomy, School of Medicine, Jianghan University, Wuhan , Hubei Province, China Corresponding author: Lv Zhou, Doctoral supervisor, Professor, Department of Physiology, Institute of asic Medical Science, Chinese cademy of Medical Science, eijing , China Luzhou_88@yahoo.com.cn Received: ccepted: (N /WLM) Research Highlights Nitrergic, cholinergic, vasoactive intestinal peptidergic and calcitonin gene-related peptide neurons participate in changes to the gastrointestinal interdigestive migrating motor complex following chemical inflammatory pain. Electroacupuncture has a regulatory effect on these changes. bbreviations NOS, nitric oxide synthase; ChE, acetylcholinesterase; VIP, vasoactive intestinal peptide; CGRP, calcitonin gene-related peptide; MMC, migrating motor complex INTRODUCTION The migrating motor complex (MMC) in the interdigestive phase is a main form of fasting gastrointestinal motility. MMC can be promoted by gastrointestinal myoelectric activity that is initiated by interstitial cells of Cajal between the gastrointestinal longitudinal muscle and circular smooth muscle [1]. MMC is regulated by interneurons of the myenteric plexus in the enteric nervous system [2-5]. previous study has confirmed that a small amount of 2136
2 formaldehyde-induced gastric mucosal lesion can induce c-fos expression in the nociceptive information conduction pathway [6]. When the digestive tract was affected by a nociceptive stimulus, pain was induced following activation of the enteric nervous system, peripheral sympathetic nerve, parasympathetic nerve and non-adrenergic non-cholinergic nerve, resulting in changes in intestinal motility and enteric nervous system function [7]. However, there is a lack of evidence linking function to cell morphology. The pyloric sphincter can regulate and control gastrointestinal motility. Electroacupuncture can regulate gastrointestinal mobility disorder [8]. This study sought to observe gastrointestinal MMC changes, as well as changes in nitrergic, cholinergic, vasoactive intestinal peptide, and calcitonin gene-related peptide neurons in the pyloric sphincter following exposure to acute gastric mucosal lesions, particularly focusing on changes to function and morphology. In addition, we aimed to investigate the regulatory effects of electroacupuncture. RESULTS Quantitative analysis of experimental animals total of 72 rats were assigned to a normal control group (saline by intragastric administration), model group (formaldehyde by intragastric administration), electroacupuncture control group (electroacupuncture stimulus), and electroacupuncture treatment group (formaldehyde + electroacupuncture). total of 72 rats were included in the final analysis. MMC cycle in the rat gastric antrum was ± 5.82 minutes, with significant time phase characteristics. In phase I, there was no contraction wave, and time duration was 5.41 ± 1.07 minutes. In phase II, there was an intermittent contraction wave, and the time duration was 7.13 ± 2.81 minutes, with a contraction frequency of 1.64 ± 0.41 times/minute and a contraction amplitude of ± g. In phase III, there were intense successive contractions, and the time duration was 3.18 ± 0.75 minutes, with a contraction frequency of 3.80 ± 0.40 times/minute and a contraction amplitude of ± g. Phase IV was the transition period of phase III and phase I, and the characteristics were identical to that in phase II; however the time duration was 1.80 ± 0.40 minutes, with a contraction frequency of 1.36 ± 0.33 times/minute and a contraction amplitude of ± g. The MMC cycle in the rat gastric antrum could migrate to duodenum. The MMC cycle in the small intestine was ± 5.21 minutes. In phase I, there was no contraction wave, and time duration was 6.20 ± 0.89 minutes. In phase II, there was an intermittent contraction wave, and the time duration was 7.88 ± 1.93 minutes, with a contraction frequency of 1.83 ± 0.42 times/minute and a contraction amplitude of ± 5.63 g. In phase III, there were intense successive contractions, and the time duration was 3.76 ± 0.75 minutes, with a contraction frequency of ± 1.40 times/minute and a contraction amplitude of ± 6.14 g. Phase IV was the transition period of phase III and phase I, and the characteristics were identical to that in phase II; however, the time duration was 1.52 ± 0.40 minutes (Figure 1). Effects of formaldehyde-induced stomach ache on pain threshold in rats fter intragastric administration of formaldehyde, rats experienced restlessness, rapid breathing, embracing of the abdomen, back arching, and less activity. Electrical measurement methods displayed that the pain threshold of normal rats (7.63 ± 1.85 m) was diminished at 30 minutes following formaldehyde administration, reached a minimum (3.86 ± 0.8 m) at 1 hour, which lasted for 2 hours. From then on, the pain threshold gradually recovered. Thermal measurement methods obtained similar results as electrical measurement methods (Table 1). Pain was relieved at 1-2 hours following formaldehyde administration. MMC characteristics in the rat gastric antrum and duodenum Gastrointestinal motility detection displayed that the Table 1 Changes in pain thresholds in rats with formaldehyde-induced acute inflammatory pain Time of intragastric administration (minute) Pain threshold by electrical measurement method (m) Pain threshold by thermal measurement method (second) ± ± ± ± ±1.70 a 0.53± ±1.50 a 0.43±0.01 a ±0.89 a 0.33±0.05 a ±0.86 a 0.34±0.06 a ±1.04 a 0.33±0.04 a ±1.50 a 0.38±0.06 a ±1.90 a 0.41±0.08 a ± ± ± ±0.01 Results are expressed as mean ± SEM. Twenty-four rats for each method; measured three times at each time point. a P < 0.05, vs. 0 minute intragastric administration group (normal control) (one-way analysis of variance). 2137
3 MMC phase I II III IV Gastric antrum Duodenum Figure 1 Interdigestive migrating motor complex (MMC) from normal conscious control rats. The rat gastric MMC cycle shows significant phase characteristics; phase I with no contraction wave; phase II with intermittent contraction wave; phase III appears to have a strong continuous contraction; phase IV is a transition period. The intestinal MMC cycle amplitude is relatively small. Effects of formaldehyde on the gastric antrum and duodenum MMC, and the regulatory effects of electroacupuncture Formaldehyde was given after three normal MMCs were recorded. We recorded an interrupted gastric MMC cycle, paroxysmal contraction, and increased wave amplitude, at a contraction frequency of 4.64 ± 0.72 times/minute and a contraction amplitude of ± g. The duodenum MMC disappeared, and an incomplete tetanus wave appeared. Electroacupuncture at Zusanli (ST36) suppressed the paroxysmal contraction wave induced by formaldehyde, showing sustained low-amplitude activity. The paroxysmal contraction wave appeared at 1 hour following needle withdrawal (Figure 2). 5 g 1 minute Distribution of nitric oxide synthase (NOS)-, acetylcholinesterase (ChE)-, vasoactive intestinal peptide (VIP)- and calcitonin gene-related peptide (CGRP)-positive neurons in the rat pyloric sphincter, and electroacupuncture intervention Histological detection revealed that the sphincter was surrounded by circular muscle bundles where there were four to seven NOS-positive nerve plexuses and one to five NOS-positive perikarya in each plexus. In thickened circular muscle bundles, there were abundant NOS-positive perikarya, 5 12 perikarya in each plexus, and strong enzymatic activity (+++). bundant NOS-positive fibers and moderate enzymatic activity (++) was visible in the muscular layer, but only a few NOS-positive fibers were found in the lamina propria and mucosa. ChE-positive ganglia (or nerve plexuses) of unequal size and number were observed in the muscularis propria. ChE-positive perikarya were observed, and enzymatic activity was uneven and mainly moderate (++). In the thickened muscular layer, we found five to six plexuses and each plexus had at least 10 cells with a high enzymatic activity (++ +++). The distribution of ChE-positive fibers was similar to that of NOS. In the sphincter, the number of VIP-immunoreactive nerve fibers was more than CGRP-immunoreactive fibers. In the thickened site of the sphincter, VIP-immunoreactive nerve fibers formed a dense fasciculate structure, whereas CGRP-immunoreactive fibers were scattered (Figures 3 6). 1% formaldehyde 2 ml Gastric antrum Duodenum 5 g 1 minute C D Gastric antrum Duodenum 1 minute Figure 2 Effect of formaldehyde and treatment with electroacupuncture on the interdigestive migrating motor complex (MMC) in conscious rats. () Effect of formaldehyde on the gastric antrum and duodenum MMC; the MMC cycles were interrupted, loss of phase nature. () Treatment of electroacupuncture on the MMC. Electroacupuncture suppressed the paroxysmal contraction wave, showing persistence of low-amplitude activity. 5 g Figure 3 Effect of electroacupuncture on nitric oxide synthase (NOS)-positive neurons in rats with gastric inflammatory pain (NDPH staining, light microscope; scale bar: 40 μm). rrows exhibit positive neurons. Compared with the normal control group () and electroacupuncture control group (C), the number of NOS-positive perikarya and enzymatic activity were lower in the submucosal plexus and myenteric nerve plexus of the sphincter in the model group (). bove-mentioned data in the electroacupuncture treatment group (D) were near to the normal control group. 2138
4 following formaldehyde-induced inflammatory pain. fter electroacupuncture at Zusanli, the viabilities of NOS-positive, ChE-positive, VIP-immunoreactive and CGRP-immunoreactive neurons were normal in the rat pyloric sphincter (Table 2). C D Figure 4 Effect of electroacupuncture on acetylcholinesterase (ChE)-positive neurons in rats with gastric inflammatory pain (NDPH staining, light microscope; scale bar: 40 μm). rrows exhibit positive neurons. Compared with the normal control group () and electroacupuncture control group (C), the number of ChE-positive perikarya and enzymatic activity were lower in the submucosal plexus and myenteric nerve plexus of the sphincter in the model group (). bove-mentioned data in the electroacupuncture treatment group (D) were near to the normal control group. C Figure 6 Effect of electroacupuncture on calcitonin gene-related peptide (CGRP)-positive neurons in rats with gastric inflammatory pain (NDPH staining, light microscope; scale bar: 40 μm). rrows exhibit positive neurons. CGRP-positive crude fibers were visible in the sphincter of the normal control group () and electroacupuncture control group (C). The number of CGRP-positive nerve fibers was obviously increased in the model group (), showing strong immunoreactivity. The number of positive nerve fibers was decreased in the electroacupuncture treatment group (D). D C D Table 2 Changes in nitric oxide synthase (NOS) (absorbance), acetylcholinesterase (ChE) (absorbance), vasoactive intestinal peptide (VIP) (absorbance), calcitonin gene-related peptide (CGRP) activities (absorbance) in the pyloric sphincter of formaldehyde-treated rats Group NOS ChE Figure 5 Effect of electroacupuncture on vasoactive intestinal peptide (VIP)-positive neurons in rats with gastric inflammatory pain (NDPH staining, light microscope; scale bar: 40 μm). rrows exhibit positive neurons. The number of VIP-positive nerve fibers was significantly reduced in the model group (), with the presence of low distribution density, weak immunoreactivity, lack of fine reticular fiber structure, and no positive fibers in the lamina propria. The number of positive nerve fibers was increased in the electroacupuncture treatment group (D), and close to the normal control group () and electroacupuncture control group (C). Normal control 0.094± ±0.011 Model 0.045±0.011 b 0.042±0.010 b Electroacupuncture control 0.091± ±0.022 Electroacupuncture treatment 0.102±0.021 b 0.089±0.024 b Results are expressed as mean ± SEM. Ten rats in each group. a P < 0.05, b P < 0.01, vs. normal control group (one-way analysis of variance). Group VIP CGRP Normal control 0.163± ±0.014 Model 0.104±0.014 b 0.156±0.013 a Electroacupuncture control 0.141± ±0.015 Electroacupuncture treatment 0.142±0.010 b 0.108±0.013 a Statistical results displayed that viabilities of NOS-, ChE-positive and VIP-immunoreactive neurons were decreased (P < 0.01), but the number of CGRPimmunoreactive nerve fibers was increased (P < 0.05) DISCUSSION This study established a model of visceral nociception 2139
5 using direct intragastric administration of formaldehyde. In order to avoid the effects of anesthetics on pain threshold and neurotransmitter release, we did not use anesthetics. To identify the effective time course of the animal model, we measured the change in visceral pain threshold using electric/thermal pain measurement methods. In accordance with the theory of somatic-visceral pain afference gathering in the spinal dorsal horn [9], we observed the effects of stomach ache using the measurement method of somatic pain, which can reflect the change in pain threshold. Results showed that the pain threshold decreased at 1 hour following formaldehyde-induced acute injury to the gastric mucosa, and the level of pain changed greatly at 1-2 hours. Therefore, this study observed the correlation of gastrointestinal motility changes and neurochemical changes at 1-2 hours after gastric mucosal injury. The gastrointestinal MMC removes food debris, increases motor coordination, stimulates pancreatic juices and bile secretion and clears the intestines. The MMC is strongly associated with gastrointestinal diseases. Gastroparesis and functional dyspepsia can prolong the MMC cycle and induce contraction deletion of phase III of the MMC cycle. This study recorded the typical MMC activity of the rat stomach and intestine when fasting, and showed stable results and characteristic phases. fter formaldehyde administration, a diffuse inflammatory reaction (bleeding point) appeared in the gastric mucosa, and pain reaction (pain threshold) was decreased. The above-mentioned changes peaked at 1-2 hours. Simultaneously, the normal MMC cycle was interrupted and paroxysmal contraction appeared after formaldehyde-induced stomachache. Formaldehyde-induced gastritis and pain are correlated with gastrointestinal MMC disorder. However, the mechanism of MMC dysfunction remains unclear. The enteric nervous system is embedded in all layers of the pyloric sphincter and adjacent gastrointestinal wall. Thus, we hypothesized that the MMC is regulated by the nerves and its transmitters or gut hormones. In particular, the enteric nervous system most likely mediates sphincter activity and adjusts the MMC. Results from this study verified that the gastrointestinal MMC was disordered after formaldehyde-induced stomach ache, and simultaneously, innervations were also disordered, showing that NOS, ChE and VIP activities were significantly reduced in the sphincter and gastrointestinal wall, whereas CGRP activity was increased. These results indicate that the changes in neurotransmitters in the enteric nervous system may be due to a neurochemical-mediated MMC disorder. Cholinergic nerve release of Ch stimulates contraction of the gastrointestinal muscle (including the pyloric sphincter) and promotes gastrointestinal motility. The decrease in ChE activity most likely represents an increase in Ch activity. Thus, it is inferred that sphincter contraction and regulation of the MMC is Ch-mediated. Nitric oxide, an inhibitory transmitter, is mainly produced or released by myenteric nerve plexus neurons and results in the regulation of gastrointestinal MMC motility. Nitric oxide can induce dilatation of the pyloric sphincter and affect gastric emptying. The decrease in NOS activity observed was most likely due to the increased synthesis or release of nitric oxide by nitroxidergic neurons. The distribution of NOS-positive nerve fibers in the stomach and intestine was obviously different. Distribution of NOS-positive neurons in the sphincter was altered when inflammatory pain appeared. This result suggests that inherent nitroxidergic neuron disorders directly affect the closing and opening function of the sphincter and pathophysiological functions of gastrointestinal motility disorders [8]. VIP acts as a peptidergic neurotransmitter capable of directly inducing gastrointestinal smooth-muscle relaxation, and mediating relaxation of the lower esophageal and internal anal sphincter. VIP-immunoreactive neurons in the enteric nervous system most likely mediated the closing and opening function of the pyloric sphincter, affected gastric emptying, and the regulated MMC [10]. VIP and NOS expression was significantly diminished in the gastric mucosa after ethanol-induced gastric mucosal injury. VIP can promote nitric oxide release. Therefore, the decrease of VIP damages the gastric mucosa by decreasing nitric oxide release and increasing endothelin-1 levels following ethanol-induced gastric mucosal injury or formaldehyde-induced stomach ache [10]. CGRP-regulated gastrointestinal tract function may be associated with gastrointestinal motion and sensory function. CGRP has complicated effects on gastrointestinal smooth muscle function. Results from the present study demonstrated thick CGRP fibers in the sphincter and abundant positive fiber networks in the thickened sphincter. These results indicate that the relaxation and contraction of the sphincter may be associated with CGRP in the enteric nervous system. In summary, some neurotransmitters in the enteric nervous system affected the closing and opening function of the sphincter and gastrointestinal motility, directly regulated the MMC, and participated in the neurological pathophysiology of gastrointestinal motility disorders following acute chemical inflammatory pain. 2140
6 MTERILS ND METHODS Design randomized, controlled animal experiment. Time and setting Experiments were performed at the School of Medicine, Jianghan University, Laboratory of Neurobiology, Tongji Medical College, Huazhong University of Science and Technology, Laboratory of Physiology, Institute of asic Medical Science, and the Chinese cademy of Medical Science, China in October Materials total of 64 healthy, clean, Sprague-Dawley rats aged 3-6 months and weighing about 200 g, of both genders, were supplied by the Department of Experimental Zoology, Tongji Medical College, Huazhong University of Science and Technology, China (license No. SYXK (E) ). ll rats were housed at 20 ± 2 C under subdued light. n additional eight healthy, clean, Sprague-Dawley rats aged 3 6 months and weighing g, of both genders were supplied by the Experimental nimal Center, Chinese cademy of Medical Sciences (license No. SCXK (Jing) ). ll protocols were performed in accordance with the Guidance Suggestions for the Care and Use of Laboratory nimals, formulated by the Ministry of Science and Technology of China [11]. Methods Measurement of pain threshold in rats with formaldehyde-induced visceral pain total of 24 conscious rats were intragastrically administered 2 ml of 1% (v/v) formaldehyde after 12-hour fasting. Fifteen minutes later, pain threshold (the electric current when the animal responded to the stimulus) was measured using the WQ-9E pain threshold detector (eijing Haidian Electronic Medical Instruments, eijing, China). Pain threshold (duration of leg-raising) was measured using a thermal pain threshold detector (Fourth Military Medical University, China). measurement was recorded every 15 minutes for 3 hours. The pain threshold measured before formaldehyde administration was considered as a baseline. Establishment of animal models of formaldehyde-induced visceral pain and electroacupuncture intervention total of 40 rats were equally and randomly assigned to four groups. fter fasting for hours, the conscious rats were intragastrically administered 2 ml saline in the normal control group. The rats in the model group were intragastrically administered 2 ml of 1% (v/v) formaldehyde [6]. The rats in the electroacupuncture control group were intragastrically administered saline. Electroacupuncture at Zusanli was conducted for 50 minutes, with a frequency of 4 16 Hz, using an electroacupuncture apparatus (Shanghai Medical Electronic pparatus, Shanghai, China). The pulsed voltage ranged from 1 V to 5 V, and was increased by 1 V progressively every 10 minutes [8]. In the electroacupuncture treatment group, electroacupuncture at Zusanli was conducted as that in the electroacupuncture control group after formaldehyde treatment. Perfusion and sample collection were performed after experimental procedures in each rat were completed. Gastrointestinal motility recording Eight rats provided by the Experimental nimal Center, Chinese cademy of Medical Sciences, were allowed free access to water after hours of fasting. The rats were intraperitoneally anesthetized with chloral hydrate (10% (v/v), 300 mg/kg), and a strain gauge was used [12]. The manufacture of the strain gauge (Department of Physiology, Institute of asic Medical Science, Chinese cademy of Medical Science) is as follows: a 5 mm 4 mm gauge [12] was consolidated by a copper sheet. guideline was welded at the output site. The specimen was embedded using silica gel. The strain gauge was separately embedded into the gastric antrum (5 mm from the pylorus) and duodenum (2 cm from the pylorus) subserosa. The guideline was subcutaneously fixed on the neck and back. t 5 7 days following surgery, the strain gauge was connected to the X-Y balance recorder (Sichuan Instrument Factory, Sichuan, China) through an electric bridge [12]. Gastrointestinal motility was recorded when the rats were conscious. Three consecutive normal MMCs were recorded first. 2 ml of 1% (v/v) formaldehyde was given intragastrically, and then the gastrointestinal MMC was recorded dynamically for 1 hour. fter perfusion, the materials were collected. In the electroacupuncture treatment group, at least three consecutive normal MMCs were recorded first. 2 ml of 1% (v/v) formaldehyde was given intragastrically, and simultaneously electroacupuncture at Zusanli was conducted. Gastrointestinal MMC was recorded dynamically for 1 hour. fter perfusion, the materials were collected. Stimulus intensity and duration were the same as above. Preparation of animal specimen Rats provided by the Experimental nimal Center, 2141
7 Chinese cademy of Medical Sciences, from the experimental and control groups were intraperitoneally anesthetized with 10% (v/v) chloral hydrate (300 mg/kg). fter the chest was opened, the heart was exposed and perfused with about 100 ml of saline (37 C ) through the ascending aorta. The blood was rapidly removed by washing. The heart was fixed with 400 ml of 4% (w/v) Millioning s paraformaldehyde (4 C ) buffer (ph 7.2). Stomach-pylorus-duodenum specimens were fixed in the same fixative (enzyme stained preparation for 6 hours; peptide stained preparation for 48 hours), and then sliced into full-thickness sections or cryostat sections after the specimens were immersed in 20% (w/v) sucrose buffer (ph 7.2) at 4 C overnight. Horizontal or longitudinal sections (25 μm-thick) were made for enzyme histochemical staining or immunohistochemical staining. Enzyme histochemical staining In accordance with a previous method for histochemical staining of NOS [13], sections were incubated in a mixture of 0.25 mm nitroblue tetrazolium (Sigma, St. Louis, MO, US), 1 mm nicotinamide-adenine dinucleotide phosphate (reduced form; Sigma), 0.25% (v/v) Triton X-100, and 0.1 M Tris-HCl buffer (ph 7.6) at 37 C for 30 minutes. fter washing in PS (0.01 M, ph 7.3), the sections were dehydrated, permeabilized and mounted. In accordance with a previous method for histochemical staining of ChE [14], sections were incubated in a mixture of 12.5 mg acetylthiocholine iodide (Shanghai Dongfeng Reagent Factory, Shanghai, China), 0.06 M sodium acetate, 0.1 M acetic acid, 0.1 M sodium citrate, 30 mm copper sulphate, 5 mm potassium ferricyanide, and 4 mm tetraisopropyl pyrophosphoramide at 37 C for 1.5 hours. fter washing in PS, the sections were dehydrated, permeabilized and mounted. Immunohistochemical staining Following a PS wash, sections were incubated in methanol and H 2 O 2 at 37 C for 1 hour, in 0.25% (v/v) Triton X-100 at 37 C for 1 hour, and 10% (v/v) calf serum at 37 C for 1 hour. Sections were then incubated with rabbit anti-rat VIP monoclonal antibody (1:4 000; Peninsula Lab, Torrance, C, US), rabbit anti-rat CGRP monoclonal antibody (1:6 000; Paesel+Lorei GmbH & Co., Hanau, Germany) at 37 C for 2 hours and 4 C for 48 hours, respectively, and incubated with goat anti-rabbit IgG (1:200; Peninsula Lab) at 37 C for 1 hour, and non-labeled antibody peroxidase-antioxidant enzyme complex (1:400; Peninsula Lab) at 37 C for 1 hour. Sections were developed with 0.05% (w/v) diaminobenzidine-0.01% (v/v) H 2 O 2 -Tris-HCl buffer for 3 4 minutes. Three washes in PS were performed between each step, each for 5 minutes. The sections were placed on a slide, dehydrated, permeabilized and mounted. Imaging analysis Five sections were randomly selected from each rat and observed under a light microscope (10 20 magnification; 3-5 visual fields; Olympus, Tokyo, Japan). Positive results were expressed as +, with four levels of immunoreactivity. The HPIS-1000 high definition color pathology image analysis system (Qianping Yingxiang, Tongji Medical College, Huazhong University of Science and Technology, China) was used to measure the viability of positive neurons in the stomach-pyloric sphincter-intestine specimens. The average value was obtained and expressed as an absorbance. Statistical analysis ll data were expressed as mean ± SEM, and analyzed with SPSS 12.0 software (SPSS, Chicago, IL, US). One-way analysis of variance was used. value of P < 0.05 was considered statistically significant. uthor contributions: Xiaoli Xu and Lv Zhou participated in the study design and concept. ll authors performed experiments. Xiaoli Xu was in charge of data analysis and manuscript writing. Lv Zhou was responsible for manuscript authorization. Conflicts of interest: None declared. Ethical approval: This study was approved by the nimal Ethics Committee, Tongji Medical College, Huazhong University of Science and Technology, China. REFERENCES [1] Holzer P, Schicho R, Holzer-Petsche U, et al. The gut as a neurological organ. Wien Klin Wochenschr. 2001; 113(17-18): [2] Huizinga JD, Robinson TL, Thomsen L. The search for the origin of rhythmicity in intestinal contraction; from tissue to single cells. Neurogastroenterol Motil. 2000;12(1):3-9. [3] Huizinga JD. Neural injury, repair, and adaptation in the GI tract. IV. Pathophysiology of GI motility related to interstitial cells of Cajal. m J Physiol. 1998;275(3 Pt 1): G [4] Dong K, Yu XJ, Li, et al. dvances in mechanisms of postsurgical gastroparesis syndrome and its diagnosis and treatment. Chin J Dig Dis. 2006;7(2): [5] Camilleri M. Diagnosis and treatment of enteric neuromuscular diseases. Clin uton Res. 2003;13(1):
8 [6] Chen LW, Jin GR, Rao ZR, et al. c-fos expression within central nervous system of the rat following gastrointestinal (viceral) noxious stimulation. Shenjing Jiepou Xue Zazhi. 1993;9(2): [7] Lazovic J, Wrzos HF, Yang QX, et al. Regional activation in the rat brain during visceral stimulation detected by c-fos expression and fmri. Neurogastroenterol Motil. 2005;17(4): [8] Lei J, Li Q, Li GC, et al. Electric rat gastrointestinal motility disorder for the adjustment and its neurochemical mechanism. Zhenci Yanjiu. 2005;30(3): [9] Han JS. Essentials of Neuroscience. eijing: Joint Press of eijing Medical University and Peking Union Medical College [10] Chen ZY, Xiang YM, Xiang K, et al. Chronic stress in rat blood and gastrointestinal hormone changes in colonic mucosa. World J Gastroenterol. 2001;9(1): [11] The Ministry of Science and Technology of the People s Republic of China. Guidance Suggestions for the Care and Use of Laboratory nimals [12] Zhou L, Xie YK. strain gauge for measuring gastrointestinal motility in animals. Zhongguo Yingyong Shenglixue Zazhi. 1988;4(4): [13] Dawson TW, redt DS, Fotuhi M, et al. Nitric oxide synthase and neuronal NDPH-disphorase are identical in brain and peripheral tissues. Proc Natl cad Sci U S. 1991;88(17): [14] Li Q, Hu DS, Ru LQ, et al. Chemical and other stomach when the pyloric sphincter inflammatory pain within the enteric nervous system, some of the changes in neural activity in the material. Shenjing Kexue. 2002;18(4): (Edited by Wang HL, Qi JP/Qiu Y/Wang L) 2143
Physiological processes in the GI tract:
 Gastrointestinal physiology for medical students General principal of gastrointestinal function Motility, nervous control and blood circulation Physiological processes in the GI tract: Motility Secretion
Gastrointestinal physiology for medical students General principal of gastrointestinal function Motility, nervous control and blood circulation Physiological processes in the GI tract: Motility Secretion
Gastrointestinal Motility 2: Intestinal and Colonic Motility Jack Grider, Ph.D.
 Gastrointestinal Motility 2: Intestinal and Colonic Motility Jack Grider, Ph.D. OBJECTIVES: 1. Contrast the types of motility in the small intestine. 2. Describe the neural circuits that mediate peristalsis.
Gastrointestinal Motility 2: Intestinal and Colonic Motility Jack Grider, Ph.D. OBJECTIVES: 1. Contrast the types of motility in the small intestine. 2. Describe the neural circuits that mediate peristalsis.
2.4 Autonomic Nervous System
 2.4 Autonomic Nervous System The ANS regulates visceral activities normally outside the realm of consciousness and voluntary control: Circulation. Digestion. Sweating. Pupillary size. The ANS consists
2.4 Autonomic Nervous System The ANS regulates visceral activities normally outside the realm of consciousness and voluntary control: Circulation. Digestion. Sweating. Pupillary size. The ANS consists
Overview of digestion
 Key concepts in Digestion. Overview of digestion introduction to the GI system Prof. Barry Campbell Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl DIGESTION
Key concepts in Digestion. Overview of digestion introduction to the GI system Prof. Barry Campbell Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl DIGESTION
INTRODUCTION TO GASTROINTESTINAL FUNCTIONS
 1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
What part of the gastrointestinal (GI) tract is composed of striated muscle and smooth muscle?
 CASE 29 A 34-year-old man presents to his primary care physician with the complaint of increased difficulty swallowing both solid and liquid foods. He notices that he sometimes has more difficulty when
CASE 29 A 34-year-old man presents to his primary care physician with the complaint of increased difficulty swallowing both solid and liquid foods. He notices that he sometimes has more difficulty when
University of Buea. Faculty of Health Sciences. Programme in Medicine
 Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Eliades, Erlandson, Ruiz UW-L Journal of Undergraduate Research XVII (2014)
 Effects of Hibernation on the Enteric Nervous System of the Thirteenlined Ground Squirrels. Lauren Eliades, Martin Erlandson, Amelia Ruiz Faculty Sponsors: Dr. Sumei Liu and Dr. Scott Cooper, Department
Effects of Hibernation on the Enteric Nervous System of the Thirteenlined Ground Squirrels. Lauren Eliades, Martin Erlandson, Amelia Ruiz Faculty Sponsors: Dr. Sumei Liu and Dr. Scott Cooper, Department
General principles of gastrointestinal motility
 General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
Gastrointestinal Anatomy and Physiology. Bio 219 Napa Valley College Dr. Adam Ross
 Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
Overview of digestion or, gut reactions - to food
 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
20 2 Stomach Fig. 2.1 An illustration showing different patterns of the myenteric plexus peculiar to the regions in the guinea-pig stomach stained wit
 Stomach 2 The stomach is unique in that ICC have a different distribution in proximal and distal regions of the same organ. ICC-CM and ICC-LM are densely distributed throughout the thick circular and longitudinal
Stomach 2 The stomach is unique in that ICC have a different distribution in proximal and distal regions of the same organ. ICC-CM and ICC-LM are densely distributed throughout the thick circular and longitudinal
Overview of digestion or, gut reactions - to food
 1 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
1 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
Gastrointestinal Physiology
 Gastrointestinal Physiology Digestion and Absorption Definition Digestion: i Food dis dissolved d and broken down. Physical digestion: Propulsion and mixing of food by muscle in the alimentary tract. Chemical
Gastrointestinal Physiology Digestion and Absorption Definition Digestion: i Food dis dissolved d and broken down. Physical digestion: Propulsion and mixing of food by muscle in the alimentary tract. Chemical
Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, ,
 IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
,. 1\)... ubj«pcl: Doclo1: M. Khatatbeh Handout # 1 8/4/ Gastreintestinal System. Lecture#: 1. Gastrointestinal Physiology
 Lecture#: 1 Mc:di
Lecture#: 1 Mc:di
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
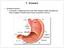 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER
 ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
DIGESTIVE PHYSIOLOGY
 DIGESTIVE PHYSIOLOGY GASTROINTESTINAL PHYSIOLOGY Ingested foodstuff contributes to an increase in the overall weight of the animal, but does not become an integral part of the structure or metabolic activities
DIGESTIVE PHYSIOLOGY GASTROINTESTINAL PHYSIOLOGY Ingested foodstuff contributes to an increase in the overall weight of the animal, but does not become an integral part of the structure or metabolic activities
Introduction to Gastrointestinal Physiology and GI Hormones Jack Grider, Ph.D
 Introduction to Gastrointestinal Physiology and GI Hormones Jack Grider, Ph.D OBJECTIVES: 1. Describe the general organization of the gut. 2. Identify the levels of neural control and describe the interaction
Introduction to Gastrointestinal Physiology and GI Hormones Jack Grider, Ph.D OBJECTIVES: 1. Describe the general organization of the gut. 2. Identify the levels of neural control and describe the interaction
1. GASTROINTESTINAL PHYSIOLOGY
 1. GASTROINTESTINAL PHYSIOLOGY FOOD INGESTION Ingested foodstuff contributes to an increase in the overall weight of the animal, but does not become an integral part of the structure or metabolic activities
1. GASTROINTESTINAL PHYSIOLOGY FOOD INGESTION Ingested foodstuff contributes to an increase in the overall weight of the animal, but does not become an integral part of the structure or metabolic activities
The Autonomic Nervous System
 The Autonomic Nervous System Responsible for control of visceral effectors and visceral reflexes: smooth muscle, glands, the heart. e.g. blood pressure, cardiac output, plasma glucose The autonomic system
The Autonomic Nervous System Responsible for control of visceral effectors and visceral reflexes: smooth muscle, glands, the heart. e.g. blood pressure, cardiac output, plasma glucose The autonomic system
Digestive System Module 4: The Stomach *
 OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
Invited Revie W. Hirschsprung's disease - immunohistochemical findings. Histology and H istopathology
 Histol Histopath (1 994) 9: 615-629 Histology and H istopathology Invited Revie W Hirschsprung's disease - immunohistochemical findings L.T. Larsson Department of Pediatric Surgery, University of Lund,
Histol Histopath (1 994) 9: 615-629 Histology and H istopathology Invited Revie W Hirschsprung's disease - immunohistochemical findings L.T. Larsson Department of Pediatric Surgery, University of Lund,
Asma Karameh. -Shatha Al-Jaberi محمد خطاطبة -
 -2 Asma Karameh -Shatha Al-Jaberi محمد خطاطبة - 1 P a g e Gastrointestinal motilities Chewing: once you introduce the first bolus to the mouth you started what we call chewing reflex appears by muscle
-2 Asma Karameh -Shatha Al-Jaberi محمد خطاطبة - 1 P a g e Gastrointestinal motilities Chewing: once you introduce the first bolus to the mouth you started what we call chewing reflex appears by muscle
Digestive system L 2. Lecturer Dr. Firdous M. Jaafar Department of Anatomy/Histology section
 Digestive system L 2 Lecturer Dr. Firdous M. Jaafar Department of Anatomy/Histology section objectives 1-Describe the general structure of digestive tract: a-mucosa. b-submucosa. c-muscularis externa d-adventitia
Digestive system L 2 Lecturer Dr. Firdous M. Jaafar Department of Anatomy/Histology section objectives 1-Describe the general structure of digestive tract: a-mucosa. b-submucosa. c-muscularis externa d-adventitia
Propulsion and mixing of food in the alimentary tract Chapter 63
 Propulsion and mixing of food in the alimentary tract Chapter 63 Types of GI movements: Propulsive movement-peristalsis Propulsion: controlled movement of ingested foods, liquids, GI secretions, and sloughed
Propulsion and mixing of food in the alimentary tract Chapter 63 Types of GI movements: Propulsive movement-peristalsis Propulsion: controlled movement of ingested foods, liquids, GI secretions, and sloughed
The Autonomic Nervous
 Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 24 The Digestive System Introduction The purpose of this chapter is to Identify the anatomical components of the digestive system as well as their
Principles of Anatomy and Physiology 14 th Edition CHAPTER 24 The Digestive System Introduction The purpose of this chapter is to Identify the anatomical components of the digestive system as well as their
Autonomic Nervous System
 Autonomic Nervous System Autonomic nervous system organization Sympathetic Nervous System division of the autonomic nervous system that arouses the body, mobilizing its energy in stressful situations
Autonomic Nervous System Autonomic nervous system organization Sympathetic Nervous System division of the autonomic nervous system that arouses the body, mobilizing its energy in stressful situations
Alimentary Canal (I)
 Alimentary Canal (I) Esophagus and Stomach (Objectives) By the end of this lecture, the student should be able to discuss the microscopic structure in correlation with the function of the following organs:
Alimentary Canal (I) Esophagus and Stomach (Objectives) By the end of this lecture, the student should be able to discuss the microscopic structure in correlation with the function of the following organs:
Immunohistochemical localization of calcium binding proteins and some neurotransmitters in myenteric plexus of goat stomach
 J. Vet. Sci. (2006),G7(4), 315 319 J O U R N A L O F Veterinar y Science Immunohistochemical localization of calcium binding proteins and some neurotransmitters in myenteric plexus of goat stomach Heungshik
J. Vet. Sci. (2006),G7(4), 315 319 J O U R N A L O F Veterinar y Science Immunohistochemical localization of calcium binding proteins and some neurotransmitters in myenteric plexus of goat stomach Heungshik
Soft palate elevates, closing off the nasopharynx. Hard palate Tongue Bolus Epiglottis. Glottis Larynx moves up and forward.
 The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
Chapter 16. APR Enhanced Lecture Slides
 Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
CHAPTER 15 LECTURE OUTLINE
 CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
For more information about how to cite these materials visit
 Author: John Williams, M.D., Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author: John Williams, M.D., Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Dana Alrafaiah. Dareen Abu Shalbak. Mohammad Almuhtaseb. 1 P a g e
 2 Dana Alrafaiah Dareen Abu Shalbak Mohammad Almuhtaseb 1 P a g e Esophagus: A muscular tube that is 25 cm long, but if measured from the incisors it would be 45cm long. Extends from C6 of cervical vertebra,
2 Dana Alrafaiah Dareen Abu Shalbak Mohammad Almuhtaseb 1 P a g e Esophagus: A muscular tube that is 25 cm long, but if measured from the incisors it would be 45cm long. Extends from C6 of cervical vertebra,
consists of: Muscular, hollow tube (= digestive tract ) + Various accessory organs
 DIGESTIVE SYSTEM consists of: Muscular, hollow tube (= digestive tract ) + Various accessory organs FUNCTION Individual parts function in: ingestion mechanical digestion chemical and enzymatic digestion
DIGESTIVE SYSTEM consists of: Muscular, hollow tube (= digestive tract ) + Various accessory organs FUNCTION Individual parts function in: ingestion mechanical digestion chemical and enzymatic digestion
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Adrenergic fibres in the human intestine
 Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
HISTOLOGY. GIT Block 432 Histology Team. Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih
 HISTOLOGY Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih Color Guide: Black: Slides. Red: Important. Green: Doctor s notes. Blue: Explanation.
HISTOLOGY Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih Color Guide: Black: Slides. Red: Important. Green: Doctor s notes. Blue: Explanation.
Gastrointestinal Physiology Structure and Innervation of the Gastrointestinal Tract Gastrointestinal Smooth Muscle Functions as a Syncytium.
 Gastrointestinal Physiology Structure and Innervation of the Gastrointestinal Tract Structure of the gastrointestinal (GI) tract, Layers of the Gastrointestinal Tract 1. Mucosa: including A lining epithelium,
Gastrointestinal Physiology Structure and Innervation of the Gastrointestinal Tract Structure of the gastrointestinal (GI) tract, Layers of the Gastrointestinal Tract 1. Mucosa: including A lining epithelium,
Chapter 14 The Autonomic Nervous System Chapter Outline
 Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
Chapter 14 The Autonomic Nervous System Chapter Outline Module 14.1 Overview of the Autonomic Nervous System (Figures 14.1 14.3) A. The autonomic nervous system (ANS) is the involuntary arm of the peripheral
Lab activity manual - Histology of the digestive system. Lab activity 1: esophagus stomach - small intestines
 Lab activity manual - Histology of the digestive system Jeanne Adiwinata Pawitan Prerequisite: Histology of the 4 basic tissues In this module we learn about the histology of the digestive system, from
Lab activity manual - Histology of the digestive system Jeanne Adiwinata Pawitan Prerequisite: Histology of the 4 basic tissues In this module we learn about the histology of the digestive system, from
Overview of Gastroenterology
 Harvard-MIT Division of Health Sciences and Technology HST.121: Gastroenterology, Fall 2005 Instructors: Dr. Jonathan Glickman Overview of Gastroenterology Gastroenterology Made Really Simple! Food Waste
Harvard-MIT Division of Health Sciences and Technology HST.121: Gastroenterology, Fall 2005 Instructors: Dr. Jonathan Glickman Overview of Gastroenterology Gastroenterology Made Really Simple! Food Waste
Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.)
 Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.) Peripheral nerves contain both motor and sensory neurons Among the motor neurons, some of these are somatic and innervate skeletal muscles while some
Chp. 16: AUTONOMIC N.S. (In Review: Peripheral N. S.) Peripheral nerves contain both motor and sensory neurons Among the motor neurons, some of these are somatic and innervate skeletal muscles while some
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Functions of the Nervous System
 The Nervous System Functions of the Nervous System 1. Control center for all body activities 2. Responds and adapts to changes that occur both inside and outside the body (Ex: pain, temperature, pregnancy)
The Nervous System Functions of the Nervous System 1. Control center for all body activities 2. Responds and adapts to changes that occur both inside and outside the body (Ex: pain, temperature, pregnancy)
Major Structures of the Nervous System. Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors
 Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
! BIOL 2401! Week 5. Nervous System. Nervous System
 Collin County Community College! BIOL 2401! Week 5 Nervous System 1 Nervous System The process of homeostasis makes sure that the activities that occur in the body are maintained within normal physiological
Collin County Community College! BIOL 2401! Week 5 Nervous System 1 Nervous System The process of homeostasis makes sure that the activities that occur in the body are maintained within normal physiological
(A) Diarrhea. (B) Stomach cramps. (C) Dehydration due to excess fluid loss. (D) A, B, and C are correct. (E) Only answer B is correct.
 Human Anatomy - Problem Drill 21: The Digestive System Question No. 1 of 10 1. A 26-year-old male is treated in the emergency department for severe gastrointestinal disturbance. Which of the following
Human Anatomy - Problem Drill 21: The Digestive System Question No. 1 of 10 1. A 26-year-old male is treated in the emergency department for severe gastrointestinal disturbance. Which of the following
Autonomic Nervous System Dr. Ali Ebneshahidi
 Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
Lab 5 Digestion and Hormones of Digestion. 7/16/2015 MDufilho 1
 Lab 5 Digestion and Hormones of Digestion 1 Figure 23.1 Alimentary canal and related accessory digestive organs. Mouth (oral cavity) Tongue* Parotid gland Sublingual gland Submandibular gland Salivary
Lab 5 Digestion and Hormones of Digestion 1 Figure 23.1 Alimentary canal and related accessory digestive organs. Mouth (oral cavity) Tongue* Parotid gland Sublingual gland Submandibular gland Salivary
number Done by Corrected by Doctor
 number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Stomach. Stomach. Nerve supply. Blood supply. Sympathe0c and parasympathe0c fibers of the autonomic nervous system
 Stomach Nerve supply Sympathe0c and parasympathe0c fibers of the autonomic nervous system Blood supply Celiac trunk, and corresponding veins (part of the hepa0c portal system) Stomach Figure 23.14a Chapter
Stomach Nerve supply Sympathe0c and parasympathe0c fibers of the autonomic nervous system Blood supply Celiac trunk, and corresponding veins (part of the hepa0c portal system) Stomach Figure 23.14a Chapter
Skeletal Muscle. Connective tissue: Binding, support and insulation. Blood vessels
 Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Autonomic Nervous System
 Autonomic Nervous System Touqeer Ahmed PhD 3 rd March, 2017 Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Nervous System Divisions The peripheral nervous system
Autonomic Nervous System Touqeer Ahmed PhD 3 rd March, 2017 Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Nervous System Divisions The peripheral nervous system
Yara Saddam. Amr Alkhatib. Ihsan
 1 Yara Saddam Amr Alkhatib Ihsan NOTE: Yellow highlighting=correction/addition to the previous version of the sheet. Histology (micro anatomy) :- the study of tissues and how they are arranged into organs.
1 Yara Saddam Amr Alkhatib Ihsan NOTE: Yellow highlighting=correction/addition to the previous version of the sheet. Histology (micro anatomy) :- the study of tissues and how they are arranged into organs.
Functions of the Nervous System
 The Nervous System Functions of the Nervous System 1. Control center for all body activities 2. Responds and adapts to changes that occur both inside and outside the body (Ex: pain, temperature, pregnancy)
The Nervous System Functions of the Nervous System 1. Control center for all body activities 2. Responds and adapts to changes that occur both inside and outside the body (Ex: pain, temperature, pregnancy)
Purinergic Neuron-to-Glia Signaling in the Enteric Nervous System Dr. Brian D. Gulbransen
 1 Assistant Professor Neuroscience Program and Department of Physiology Michigan State University East Lansing, Michigan USA The requirement to nourish and sustain an organism, as well as to defend it
1 Assistant Professor Neuroscience Program and Department of Physiology Michigan State University East Lansing, Michigan USA The requirement to nourish and sustain an organism, as well as to defend it
The Nervous System: Autonomic Nervous System Pearson Education, Inc.
 17 The Nervous System: Autonomic Nervous System Introduction The autonomic nervous system: Functions outside of our conscious awareness Makes routine adjustments in our body s systems The autonomic nervous
17 The Nervous System: Autonomic Nervous System Introduction The autonomic nervous system: Functions outside of our conscious awareness Makes routine adjustments in our body s systems The autonomic nervous
Nervous System. Master controlling and communicating system of the body. Secrete chemicals called neurotransmitters
 Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Physiologic Anatomy and Nervous Connections of the Bladder
 Micturition Objectives: 1. Review the anatomical organization of the urinary system from a physiological point of view. 2. Describe the micturition reflex. 3. Predict the lines of treatment of renal failure.
Micturition Objectives: 1. Review the anatomical organization of the urinary system from a physiological point of view. 2. Describe the micturition reflex. 3. Predict the lines of treatment of renal failure.
DIGESTIVE TRACT ESOPHAGUS
 DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
Systems Neuroscience November 21, 2017 The autonomic nervous system
 Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Section: Chapter 5: Multiple Choice. 1. The structure of synapses is best viewed with a(n):
 Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Small intestine. Small intestine
 General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
Composed by Natalia Leonidovna Svintsitskaya, Associate professor of the Chair of Human Anatomy, Candidate of Medicine
 Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
The Nervous System: Neural Tissue Pearson Education, Inc.
 13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
Chapter 17 Nervous System
 Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Morphometry of Myenteric Neurons in Stomach
 Morphometry of Myenteric Neurons in Stomach Narbada Saini and Madhur Gupta Department of Anatomy, Postgraduate Institute of Medical Education and Research, Chandigarh 160012 India Corresponding author:
Morphometry of Myenteric Neurons in Stomach Narbada Saini and Madhur Gupta Department of Anatomy, Postgraduate Institute of Medical Education and Research, Chandigarh 160012 India Corresponding author:
Autonomic Nervous System. Part of the nervous system that controls most of the visceral functions of the body ( Automatically?
 Autonomic Response? Autonomic Nervous System Part of the nervous system that controls most of the visceral functions of the body ------ ( Automatically?) Classification Of CNS Autonomic Nervous System
Autonomic Response? Autonomic Nervous System Part of the nervous system that controls most of the visceral functions of the body ------ ( Automatically?) Classification Of CNS Autonomic Nervous System
THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS
 Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
I. Neural Control of Involuntary Effectors. Chapter 9. Autonomic Motor Nerves. Autonomic Neurons. Autonomic Ganglia. Autonomic Neurons 9/19/11
 Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
What you really need to know about Gastroparesis?
 What you really need to know about Gastroparesis? John M. Wo, MD Division of Gastroenterology/Hepatology Director of GI Motility and Neurogastroenterology 8/3/2016 1/4/2017 1 What you really need to know
What you really need to know about Gastroparesis? John M. Wo, MD Division of Gastroenterology/Hepatology Director of GI Motility and Neurogastroenterology 8/3/2016 1/4/2017 1 What you really need to know
Screening and bioassay of Sympatholytics. Dr. Magdy M. Awny Lecture 4
 Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
Digestive System. - Food is ingested
 11 V. Digestive Processes in the Mouth - Food is ingested - Mechanical digestion begins (chewing) - Salivary amylase begins chemical breakdown of starch - Propulsion is initiated by Deglutition (Swallowing)
11 V. Digestive Processes in the Mouth - Food is ingested - Mechanical digestion begins (chewing) - Salivary amylase begins chemical breakdown of starch - Propulsion is initiated by Deglutition (Swallowing)
Warm Up! Test review (already! ;))
 Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
Name Class Date. KEY CONCEPT The nervous system and the endocrine system provide the means by which organ systems communicate.
 Section 1: How Organ Systems Communicate KEY CONCEPT The nervous system and the endocrine system provide the means by which organ systems communicate. VOCABULARY nervous system central nervous system (CNS)
Section 1: How Organ Systems Communicate KEY CONCEPT The nervous system and the endocrine system provide the means by which organ systems communicate. VOCABULARY nervous system central nervous system (CNS)
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION
 ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
The Nervous System. Autonomic Division. C h a p t e r. PowerPoint Lecture Slides prepared by Jason LaPres North Harris College Houston, Texas
 C h a p t e r 17 The Nervous System Autonomic Division PowerPoint Lecture Slides prepared by Jason LaPres North Harris College Houston, Texas Copyright 2009 Pearson Education, Inc., publishing as Pearson
C h a p t e r 17 The Nervous System Autonomic Division PowerPoint Lecture Slides prepared by Jason LaPres North Harris College Houston, Texas Copyright 2009 Pearson Education, Inc., publishing as Pearson
D) around, bypassing B) toward
 Nervous System Practice Questions 1. Which of the following are the parts of neurons? A) brain, spinal cord, and vertebral column B) dendrite, axon, and cell body C) sensory and motor D) cortex, medulla
Nervous System Practice Questions 1. Which of the following are the parts of neurons? A) brain, spinal cord, and vertebral column B) dendrite, axon, and cell body C) sensory and motor D) cortex, medulla
Our gut reactions to food or, gut reactions - to food
 Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
BI 232: Human Anatomy & Physiology
 BI 232: Human Anatomy & Physiology Roster Business Course Introduction and Syllabus Notecard Name E-mail Why you are taking the course Something interesting you did over break Lecture Tips Use the Study
BI 232: Human Anatomy & Physiology Roster Business Course Introduction and Syllabus Notecard Name E-mail Why you are taking the course Something interesting you did over break Lecture Tips Use the Study
Why would fatty foods aggravate the patient s RUQ pain? What effect does cholecystokinin (CCK) have on gastric emptying?
 CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
Autonomic Nervous System
 Autonomic Nervous System 6 th March, 2015 & 19 th March, 2015 Touqeer Ahmed Ph.D. Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Autonomic Nervous System Role
Autonomic Nervous System 6 th March, 2015 & 19 th March, 2015 Touqeer Ahmed Ph.D. Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Autonomic Nervous System Role
The Autonomic Nervous System Outline of class lecture for Physiology
 The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
Biology 218 Human Anatomy
 Chapter 17 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Overview of the Nervous System (p. 537) 1. The nervous system and the endocrine system are the body s major control and integrating centers.
Chapter 17 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Overview of the Nervous System (p. 537) 1. The nervous system and the endocrine system are the body s major control and integrating centers.
Fig Copyright McGraw-Hill Education. Permission required for reproduction or display. Nucleus. Muscle fiber. Endomysium. Striations.
 Fig. 11.1 Nucleus Muscle fiber Endomysium Striations Ed Reschke 1 Fig. 11.2 Muscle fiber Nucleus I band A band Z disc Mitochondria Openings into transverse tubules Sarcoplasmic reticulum Triad: Terminal
Fig. 11.1 Nucleus Muscle fiber Endomysium Striations Ed Reschke 1 Fig. 11.2 Muscle fiber Nucleus I band A band Z disc Mitochondria Openings into transverse tubules Sarcoplasmic reticulum Triad: Terminal
Anatomy & Histology of The Small intestine
 Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
Nervous Systems: Diversity & Functional Organization
 Nervous Systems: Diversity & Functional Organization Diversity of Neural Signaling The diversity of neuron structure and function allows neurons to play many roles. 3 basic function of all neurons: Receive
Nervous Systems: Diversity & Functional Organization Diversity of Neural Signaling The diversity of neuron structure and function allows neurons to play many roles. 3 basic function of all neurons: Receive
Includes mouth, pharynx, esophagus, stomach, small intestine, large intestine, rectum, anus. Salivary glands, liver, gallbladder, pancreas
 Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Digestive System 7/15/2015. Outline Digestive System. Digestive System
 Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Neural Basis of Motor Control
 Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Drugs Affecting The Autonomic Nervous System(ANS)
 Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
Human Anatomy and Physiology - Problem Drill 15: The Autonomic Nervous System
 Human Anatomy and Physiology - Problem Drill 15: The Autonomic Nervous System Question No. 1 of 10 Which of the following statements is correct about the component of the autonomic nervous system identified
Human Anatomy and Physiology - Problem Drill 15: The Autonomic Nervous System Question No. 1 of 10 Which of the following statements is correct about the component of the autonomic nervous system identified
The Digestive System. Chapter 25
 The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
