Accessory and Anomalous Atrioventricular Valvar Tissue Causing Outflow Tract Obstruction
|
|
|
- Owen Ball
- 5 years ago
- Views:
Transcription
1 JACC Vol. 32, No. 6 November 15, 1998: PEDIATRIC CARDIOLOGY Accessory and Anomalous Atrioventricular Valvar Tissue Causing Outflow Tract Obstruction Surgical Implications of a Heterogeneous and Complex Problem DOFF B. MCELHINNEY, MD,* V. MOHAN REDDY, MD,* NORMAN H. SILVERMAN, MD, FRANK L. HANLEY, MD* San Francisco, California Objectives. The purpose of this study was to determine the effect of accessory or anomalous atrioventricular valvar apparatus on relief of outflow tract obstruction. Background. Outflow tract obstruction due to accessory tissue or anomalous attachments of the atrioventricular valvar apparatus is an unusual but well-recognized problem. In addition to obstruction, anomalous attachments of the atrioventricular valvar apparatus may interfere with procedures to relieve outflow tract obstruction or perform outflow tract reconstruction. Methods. Since 1992, we have operated on 21 patients (median age 4 years) with systemic (n 13), pulmonary (n 5) or bilateral (n 3) outflow tract obstruction due to accessory atrioventricular valvar tissue and/or anomalous attachments of the subvalvar apparatus. Primary diagnoses were isolated obstruction of the systemic outflow tract or aortic arch (n 7), transposition complexes (n 6), previously repaired atrioventricular septal defect (n 3), functionally single ventricle (n 3) and ventricular septal defect with pulmonary outflow obstruction (n 2). Outflow tract gradients ranged from mm Hg (median 58 mm Hg). Results. Complete relief of obstruction due to atrioventricular valvar anomalies was possible in 14 patients. In six patients, the planned procedure either had to be modified or only partial relief of the obstruction was achieved. In the remaining patient, who had borderline functionally single ventricle heart disease (unbalanced atrioventricular septal defect) and systemic outflow obstruction due to accessory and functional valvar apparatus, support was withdrawn because the parents refused univentricular palliation and the valvar anomalies precluded a Ross-Konno procedure. There were two early deaths. At follow-up ranging from 1 to 66 months (median 27 months), there was one death, and there has been no recurrence of outflow tract obstruction or residual atrioventricular valvar tissue. Conclusions. Outflow tract obstruction caused by accessory or anomalous atrioventricular valvar structures is an uncommon and heterogeneous group of conditions that can have significant surgical implications. In the majority of cases, tailoring of surgical techniques will permit complete relief of obstruction. However, such anomalies may limit standard surgical options and necessitate an innovative approach in some patients. (J Am Coll Cardiol 1998;32:1741 8) 1998 by the American College of Cardiology Outflow tract obstruction due to accessory atrioventricular (AV) valvar tissue or anomalous attachments of the tensor apparatus is an uncommon problem (1 21). In addition to causing obstruction per se, accessory tissue or anomalous attachments of either AV valvar apparatus may interfere with procedures to relieve outflow tract obstruction or perform outflow tract reconstruction in patients with congenital heart disease. Such anomalies may be isolated or found with a wide range of associated lesions, including simple fibromuscular subaortic obstruction, AV septal defects, transposition complexes, and hearts with a functionally single ventricle among others. Although these phenomena have been described in a From the Divisions of *Cardiothoracic Surgery and Pediatric Cardiology, University of California, San Francisco, California. This study was presented at the American College of Cardiology 47th Annual Scientific Session, Atlanta, Georgia, March 29 April 1, Manuscript received March 30, 1998; revised manuscript received July 14, 1998, accepted July 29, Address for correspondence: V. Mohan Reddy, MD, UCSF Medical Center, 505 Parnassus Avenue, M593, San Francisco, California number of autopsy and surgical cases, such reports have generally consisted of only one or two patients (1 21). There have been no consecutive surgical series on the basis of which to evaluate the range of such anomalies and the variability in their surgical management. In our experience, we have encountered a variety of cases in which systemic or pulmonary outflow tract obstruction was caused entirely or in part by accessory AV valvar tissue or anomalous attachments of the tensor apparatus. On several occasions, this problem necessitated modification of the planned surgical procedure, which illustrates the potential significance of such anomalies. To characterize the range of situations in which the AV valvar apparatus contributes to outflow tract obstruction and the surgical implications of such abnormalities, we reviewed our experience with this problem. Methods Between July 1992 and December 1997, a total of 21 patients with outflow tract obstruction due completely or in 1998 by the American College of Cardiology /98/$19.00 Published by Elsevier Science Inc. PII S (98)
2 1742 MCELHINNEY ET AL. JACC Vol. 32, No. 6 ACCESSORY AV VALVAR TISSUE November 15, 1998: Figure 1. Preoperative echocardiogram in patient 11. (Left) Parasternal long axis view shows an abnormal mass of accessory AV valvar tissue (arrow) ballooning into the left ventricular (LV) outflow tract and across the aortic valve during systole. The open leaflets of the aortic valve are indicated by the double arrow. (Middle) Parasternal short axis view shows the abnormal septal commissure (CL) and the accessory tissue above it. (Right) Subcostal sagittal view demonstrates the papillary muscles (PMs) of the mitral valve, the septal commissure and the mass of accessory tissue protruding into the left ventricular outflow tract (arrow). Ao aorta; LA left atrium; PA pulmonary artery; RV right ventricle. part to accessory AV valvar tissue or anomalous attachments of the tensor apparatus were evaluated for surgery at our institution. Patient data are summarized in Table 1. Ages ranged from 3 days to 19 years (median 4 years). Primary diagnoses were complex isolated systemic outflow tract obstruction (n 5), congenitally corrected transposition (n 4), previously repaired AV septal defect (n 3), functionally single ventricle heart disease (n 3; right dominant unbalanced AV septal defect in two cases and tricuspid atresia in one), transposition of the great arteries with a prior Senning procedure (n 2), ventricular septal defect (n 2), Shone s complex (n 1) and interrupted aortic arch (n 1). Ten patients had undergone prior interventions, including AV septal defect repair (n 3), Senning procedure (n 2), palliation or repair of arch obstruction (n 2), univentricular palliation with a systemic-pulmonary arterial shunt and/or bidirectional cavopulmonary anastomosis (n 2) and balloon aortic valvuloplasty (n 1). All patients were evaluated with preoperative echocardiography, and 12 patients underwent preoperative catheterization. Operative techniques were based on operative findings and will be discussed in the Results section. Perioperative data were collected on retrospective review of patient records. Follow-up was obtained by contacting the referring cardiologist. Data are presented throughout as median and range. Results Echocardiographic and surgical findings. All patients were diagnosed by preoperative echocardiography with obstruction to the systemic outflow tract (n 13), the pulmonary outflow tract (n 5) or both (n 3). The median outflow tract gradient was 58 mm Hg (range 20 mm Hg to 110 mm Hg). In 13 cases the left AV valve contributed to outflow tract obstruction (Fig. 1), while the right AV valve contributed in 4 cases, both AV valves contributed in 3 and a common AV valve contributed in 1 patient with a functional single ventricle. One or more additional mechanisms of outflow tract obstruction were present in 18 patients, 17 of whom had at least one additional cause of subvalvar obstruction. There was associated aortic regurgitation in three patients (moderate 2; mild 1) and mitral regurgitation in two (both mild). Diagnostic variables are listed in Table 1. Operative management. Accessory tissue and anomalous septal attachments associated with the systemic AV valve were inspected through the semilunar (usually aortic) valve. In cases of accessory tissue, the valve was often inspected through a left atriotomy as well in order to ensure that resection would not compromise AV valvar function. The tissue was then resected and additional procedures were performed as necessary (Table 2). In the other patients, various procedures and approaches were used. In 13 patients, additional outflow tract myectomy was performed, and in 11 an additional component of membranous obstruction was resected. Patients underwent surgery on moderately hypothermic cardiopulmonary bypass, with aortic and bicaval cannulation. Myocardial protection was performed with cold crystalloid cardioplegia and the aorta was cross-clamped. Circulatory arrest was not employed. Transesophageal echocardiography was used for intraoperative monitoring in all patients. Outcomes. Support was withdrawn in the patient with a severely unbalanced AV septal defect in whom univentricular palliation was refused and a Ross-Konno operation was not possible owing to systemic outflow tract obstruction by functional left AV valvar tissue, and the patient subsequently died. Postmortem examination was not obtained. Of the 20 patients who underwent surgery, there were two early deaths. One of these was a patient with right isomerism, asplenia and subvalvar obstruction of both outflow tracts who underwent a modified Damus procedure and single ventricle palliation, and suffered an unexplained cardiopulmonary arrest 2 days after surgery from which he could not be resuscitated even with a ventricular assist device. The other early death was a patient with corrected transposition and multiple bilateral chordal attachments to the rim of the inlet ventricular septal defect who underwent bidirectional cavopulmonary anastomosis. Four days after surgery he became hypotensive and bradycardic, and the combination of decreased systemic arterial pressure and elevated systemic venous pressure in the superior caval system led to decreased transcapillary pressure in the
3 Table 1. Demographic and Diagnostic Data for 21 Patients With Accessory of Anomalous AV Valvar Apparatus Causing Outflow Tract Obstruction Pt Age (years) Diagnosis Outflow Tract AV Valve Gradient (mm Hg) Mechanism of AV Valvar Apparatus Causing Outflow Tract Obstruction Functional AV Valvar Tissue Other Mechanisms of Obstruction 1 10 TGA, prior Senning, subps P L 100 Multiple adhesions of the anterior mitral leaflet septal and free wall aspects of LVOT No Membranous, dynamic septal bowing into LVOT 2 11 TGA, prior Senning, subps P L 90 Accessory MV tissue No Dynamic septal bowing into LVOT 3 7 Interrupted arch prior palliation, subas S L 55 Tricuspid pouch extruding into LVOT No Membranous 4 10 Corrected TGA, VSD, valvar PS, subps, P L 86 Accessory MV tissue No Membranous, muscular, valvar supravalvar PS 5 15 Congenital AS, subas, aortic regurgitation, S L 20 Accessory MV tissue No Membranous two prior balloon dilatations 6 4 m VSD, double chamber right ventricle, subps P R 60 Large functional papillary muscle supplying entire anterior leaflet of TV anchored at os infundibulum, crossing RVOT Yes Fibrotic muscular os infundibulum, superiorly displaced 7 7 m Right isomerism, right dominant unbalanced AVSD (single ventricle), subas, subps B C 50 subas 36 subps Functional and accessory AV valvar apparatus causing subas and subps 8 3 SubAS, aortic regurgitation, cleft MV S L 110 Abnormal nonfunctional MV chordae running No Membranous, muscular from base of MV to septum 9 9 Complete AVSD, prior repair, subas S L 90 Accessory MV tissue No Membrane, tunnel-type muscular 10 6 Corrected TGA, apicocaval juxtaposition, B B 30 1) Accessory MV tissue & functional chordae Yes Muscular VSD, valvar PS, subps on VSD rim causing subps; 2) MV & TV chordal attachments to rim of VSD prevented Rastelli baffle 11 3 SubAS, aortic regurgitation S L 60 Accessory MV tissue attached to chordal structures of AML, at site of abnormal commissure No Membranous, muscular 12 3 Corrected TGA, inlet VSD, valvar PS, subps S B 100 Multiple functional MV & TV chordae crossing & attaching to rim of inlet VSD prevented Rastelli baffle 13 7 d Coarctation, subas, cleft MV S L 30 Accessory MV tissue No None 14 4 Corrected TGA, VSD, valvar PS, subps B B 45 1) Accessory MV tissue causing subps; 2) TV Both Muscular, valvar chordal attachments to rim of VSD prevented Rastelli baffle 15 3 d Right dominant unbalanced AVSD (borderline single ventricle), valvar AS, subas S L no antegrade flow Accessory & functional MV tissue causing subas (only retrograde flow in ascending aorta), precluded Ross-Konno Both Membranous, hypoplastic LV (muscular), valvar m Complete AVSD (non-downs), azygos continuation of IVC, prior repair, subas 17 6 Complete AVSD, double-orifice left AV valve, prior repair, subas S L 80 Abnormal nonfunctional MV chordal structure & papillary muscle running from base of MV to septum S L 45 Redundant nonfunctional choradeattaching to abnormal papillary muscle and prolapsing into LVOT Both Yes No No None Valvar Membrane, tunnel-type muscular Membrane, tunnel-type muscular 18 3 SubAS, aortic regurgitation S L 55 Abnormal nonfunctional MV chordal structure No Membranous, muscular & papillary muscle running from base of MV to septum 19 6 d Coarctation, subas S R TV chordal attachments to infundibular septum Yes Muscular (interfered with subas repair) 20 4 Tricuspid atresia, d-transposition, subas, S R 80 Residual TV tissue No Restrictive VSD prior single ventricle palliation VSD, subps P R 35 Tricuspid pouch extruding into RVOT during systole No None AS aortic stenosis; AV(SD) atrioventricular (septal defect); C common; L left; LVOT left ventricular outflow tract; MV mitral valve; P pulmonary; PS pulmonary stenosis; R right; RVOT right ventricular outflow tract; S systemic; TGA transposition of the great arteries; TV tricuspid valve; VSD ventricular septal defect. JACC Vol. 32, No. 6 MCELHINNEY ET AL. November 15, 1998: ACCESSORY AV VALVAR TISSUE 1743
4 1744 MCELHINNEY ET AL. JACC Vol. 32, No. 6 ACCESSORY AV VALVAR TISSUE November 15, 1998: Table 2. Operative and Postoperative Data in Patients Undergoing Repair of Outflow Tract Obstruction Due to AV Valvar Anomalies Pt Surgical Management of Accessory/Anomalous AV Valvar Tissue Other Procedures Residual Gradient Follow-up Duration (months) 1 Division of abnormal chordae and reattachment to LVOT myectomy, LVOT membrane resection anterior MV leaflet, release of adhesions between LVOT and anterior MV leaflet 2 Resection of nonfunctional valvar tissue None Resection of nonfunctional valvar tissue LVOT membrane resection Resection of nonfunctional valvar tissue LVOT myectomy (extensive), LVOT membrane resection, PV commissurotomy, supravalvar resection 5 Resection of nonfunctional valvar tissue Ross procedure, LVOT myectomy, LVOT membrane resection 6 Anomalous papillary muscle preserved, muscle resected Circumferential RVOT myectomy, infundibular patch, from around base of papillary muscle VSD closure 7 DKS, resection accessory AV valve tissue causing Subpulmonary myectomy Converted to Early death subpulmonic obstruction Damus 8 Division of nonfunctional chordae LVOT myectomy (extensive), LVOT membrane resection Resection of nonfunctional valvar tissue LVOT myectomy (extensive), LVOT membrane resection, AV plasty (commissurotomy, thinning) 10 Double switch not performed; VSD closed with RV-Ao None & LV-PA continuity 11 Resection of nonfunctional valvar tissue LVOT myectomy (mild), LVOT membrane resection Double switch not performed; BCPA (instead of LV- PA conduit), leaving option of double switch with mechanical valves in future Bidirectional cavopulmonary anastomosis, PA ligated and divided, atrial septectomy PA ligated Early death 13 Resection of nonfunctional valvar tissue Closure of septal MV commissure ( cleft ) 0 Late death (6 months) 14 Double switch not performed; VSD closed with RV-Ao PA ligated and divided, VSD closed, LV-PA conduit PA ligated 20.6 continuity, LV-PA conduit placed to circumvent subps placed 15 Parents refused Norwood operation, support withdrawn n.a. n.a. n.a. 16 Resection of nonfunctional valvar tissue LVOT myectomy (extensive), LVOT membrane resection Resection of nonfunctional chordae and papillary LVOT myectomy (extensive), LVOT membrane resection muscle 18 Resection of nonfunctional valvar tissue LVOT myectomy (extensive), LVOT membrane resection SubAS resection, not completely possible due to TV LVOT myectomy (inadequate), Ross-Konno chordal attachments; Ross-Konno 20 Resection of nonfunctional valvar tissue VSD enlargement Resection of nonfunctional valvar tissue VSD closure Ao aorta; AV aortic valve; BCPA bidirectional cavopulmonary anastomosis; LV left ventricle; PA pulmonary artery; RV right ventricle. Other abbreviations as in Table 1. cerebral circulation and brain damage. He subsequently developed multiorgan failure and support was eventually withdrawn. In 14 patients, the accessory tissue or anomalous attachments of the AV valvar apparatus were resected completely or divided, respectively. In 10 of these patients, the postoperative outflow tract gradient was 10 mm Hg. In two patients, there was residual muscular outflow tract obstruction with gradients of 15 mm Hg and 20 mm Hg. In the two patients with transposition and a prior Senning procedure, there was a residual gradient of 30 mm Hg due to systolic bowing of the interventricular septum into the left ventricular outflow tract. In one patient with right isomerism and a functionally single ventricle heart, only partial relief of obstruction was obtained. There was obstruction of both outflow tracts by functional chordae and accessory tissue, and despite a modified Damus procedure, this patient did not survive. In five patients, the planned surgical procedure could not be performed because of anomalous attachments of functional AV valvar apparatus interfering with outflow tract reconstruction. Three of these patients had corrected transposition of the great arteries, and the plan was to attempt a double-switch procedure if possible, rerouting venous return with a Senning or Mustard baffle and ventricular outflow by means of either an arterial-switch procedure or a Rastelli intraventricular baffle (22). Anomalies of the pulmonary valve (valvar stenosis in two and a bicuspid valve with mild regurgitation in the other) precluded arterial switch, leaving a Rastelli procedure as the only option. However, multiple functional left and right AV valvar chordae were attached to the rim of the ventricular septal defect, effectively precluding a Rastelli procedure without simultaneous replacement of the AV valves. Because the chordal attachments were bilateral, multiple, and diffuse (that is, not into a single papillary muscle), it was not possible to translocate the chordae into the proper ventricle, as we have done with straddling AV valve (23), or to utilize the REV procedure/conal flap method (24,25). The risk of bilateral AV
5 JACC Vol. 32, No. 6 November 15, 1998: MCELHINNEY ET AL. ACCESSORY AV VALVAR TISSUE 1745 valvar dysfunction was unacceptably high. Instead of performing a double switch, all three of these patients underwent the traditional form of repair of corrected transposition, with the right ventricle retained as the systemic ventricle and pulmonary outflow obstruction relieved with a left ventriclepulmonary arterial conduit. There were two other situations in which anomalous attachments of the AV valvar apparatus necessitated a modification of the surgical procedure. In one patient, a Ross-Konno procedure was required because chordal attachments to the outlet septum limited the extent of infundibular septal resection that could be achieved (26). In a patient with doublechambered right ventricle, right ventricular outflow tract obstruction was caused by a large functional papillary muscle that supplied the entire anterior tricuspid valvar leaflet and inserted into the os infundibulum. A significant amount of infundibular muscle was resected, including around the base of the anomalous papillary muscle, but a substantial gradient remained after discontinuation of bypass and evaluation by transesophageal echocardiography. The patient was placed back on bypass, additional muscle was resected and an infundibular patch was placed to augment the outflow tract, resulting in complete relief of obstruction. Follow-up. Patients were followed for 1 to 66 months (median 27 months) postoperatively. There was one late death, 6 months after surgery, in a patient with Shone s complex who required two subsequent operations for supravalvar aortic stenosis (5 months later) and mitral stenosis and regurgitation (6 months later). No other patients underwent reoperation during the follow-up period. On follow-up echocardiographic evaluation, the Doppler-estimated pressure gradient across the affected outflow tract (Table 2) was unchanged in all surviving patients, and no patient had evidence of new or increased AV valvar dysfunction compared with the preoperative period. Discussion We have presented here our experience with 21 patients in whom accessory AV valvar tissue or anomalous attachments of the AV valvar apparatus contributed to outflow tract obstruction or interfered with outflow tract reconstruction. In the majority of cases, the outflow tract of the morphological left ventricle was involved, which reflects the critical role of the left AV valve in the left ventricular outflow tract. Although accessory of anomalous AV valvar tissue was the predominant cause of outflow tract obstruction in most patients, the obstruction was typically complex, with multiple contributing factors, including muscular (n 14), membranous (n 11) and dynamic (n 2) obstruction as well. Consistent with most previously reported cases (1 21), we found that obstruction due to nonfunctional (accessory) tissue can almost always be relieved completely as long as it is identified. In contrast, if functional tissue interferes with outflow tract repair, it has much more significant implications for the approach to repair and possibly for outcome. Accessory/anomalous AV valvar tissue causing outflow tract obstruction. Accessory AV valvar tissue. In the majority of our cases, accessory tissue or anomalous attachments of the AV valves contributed to actual outflow tract obstruction, as opposed to interfering with outflow tract reconstruction. In hearts with normal topology and segmental connections, the most common finding was accessory (nonfunctional) left AV valvar tissue contributing to subaortic obstruction. In two patients, this tissue took the form of a sail-like membrane with chordal attachments to the papillary muscles that would fill with blood during systole and balloon into the left ventricular outflow tract. This type of accessory mitral valve tissue has been described in several reports (1,3,11), and is important because it can cause severe outflow tract obstruction in the dynamic heart, but assumes a less obvious profile in the intraoperative state of chamber relaxation and decompression. In the pre-echocardiography era, there were instances in which such tissue was overlooked on operative inspection, with a fatal consequence (1). With echocardiography, this pattern can be recognized quite readily (Fig. 1). Other than this pattern, accessory tissue in our experience and in the literature seems to constitute a spectrum, which typically has the appearance of mature AV valvar tissue on histopathologic examination (11). In our patients, accessory AV valvar tissue was typically associated with some degree of membranous and/or muscular subaortic obstruction as well. Accessory tissue of the left AV valve appears to be a relatively common cause of left ventricular outflow tract obstruction in both transposition and AV septal defect as well (8,17,18), and should not pose any different management problems than in cases of isolated subaortic obstruction. As in the various case reports describing this phenomenon, resection of the accessory tissue, along with resection of membranous or muscular substrata of obstruction, allowed for essentially complete relief of outflow tract obstruction in all of our cases, with no evidence of resultant AV valvar dysfunction (3,6,7,10,11). We are not aware of any reported complications of resecting accessory AV valvar tissue, although failure to identify such tissue has been known to lead to persistent outflow obstruction and death (1,5 7,19). In two patients with normally related great arteries, systemic or pulmonary outflow tract obstruction was caused by a tricuspid pouch extruding into either the left or right ventricular outflow tract, respectively, during systole. The patient with subaortic obstruction according to this mechanism had elevated right ventricular pressures after prior palliation for interrupted aortic arch at another institution with pulmonary arterial banding and a pulmonary trunk to descending aortic conduit. Outflow tract obstruction due to a tricuspid pouch (sometimes referred to erroneously as a ventricular septal aneurysm) has been described in patients with both normally related and transposed great arteries (8,9,15,16). Rare cases of actual ventricular septal aneurysm causing outflow tract obstruction have also been reported (4,20). Closure of the ventricular septal defect, along with resection of any accessory tissue, sufficiently relieves obstruction due to this mechanism.
6 1746 MCELHINNEY ET AL. JACC Vol. 32, No. 6 ACCESSORY AV VALVAR TISSUE November 15, 1998: Anomalous attachments of the tensor apparatus. Anomalous attachments of the subvalvar AV valvar apparatus causing outflow tract obstruction have been described in a number of reports, both as an isolated anomaly and in association with other types of congenital heart disease (1,2,12). The most common circumstance appears to be AV septal defect or isolated cleft mitral valve, in which chordae run from the edges of the septal commissure ( cleft ) to the septum. These can be divided, as in two patients in our series, and the septal commissure closed without causing AV valvar regurgitation. Attachments of abnormal chordae to the septal wall of the left ventricle have been described in patients without AV septal defect or cleft mitral valve as well, including two in this series, one with transposition and another with isolated left ventricular outflow tract obstruction. As long as the valve is sufficiently supported by normal tensor apparatus, these anomalous attachments can be divided and resected. However, several reports have described an abnormal papillary muscle and chordae inserting into the base of the anterior mitral valvar leaflet with no normal chordae from the free edge of the leaflet (13). In such cases, resection of the abnormal component of the subvalvar apparatus and replacement of the valve or insertion of artificial chordae (expanded polytetrafluoroethylene suture) are essentially the only options. Anomalous attachments of the left AV valve to the septum may be an important cause of subpulmonary outflow tract obstruction in complete transposition of the great arteries as well. Among 166 specimens in the Edwards collection, 21 had subpulmonary (left ventricular) outflow obstruction, 7 of which had anomalous attachments of the left AV valve to the septum (2). In patients with a Senning procedure and leftward septal bowing, the combination of these effects may lead to severe obstruction, as in one of our patients. Anomalous AV valvar tissue interfering with outflow tract reconstruction. Several types of anomalous AV valvar tissue interfered with outflow tract reconstruction. In all but one case, we were forced to abandon our planned outflow tract reconstruction in favor of an approach that would not be compromised by the AV valvar anomalies. In the other case, modification of the planned repair was required. Attachments of functional tensor apparatus to the crest of the septum in transposition complexes. The most common abnormality in this category was the presence of multiple left and right AV valvar chordae diffusely attaching to or crossing the rim of the ventricular septal defect. In three patients with corrected transposition, this precluded a Rastelli intraventricular baffle procedure as part of a double-switch operation (22). These patients all had abnormalities of the pulmonary valve, similarly precluding arterial switch. As a result, they were managed either with standard repair of corrected transposition, leaving the right ventricle as the systemic ventricle, or with bidirectional cavopulmonary anastomosis in order to preserve the option of anatomic repair with a mechanical AV valve at a later date. The fourth patient with corrected transposition in this study had severe valvar pulmonary stenosis and a small ventricular septal defect, so a double-switch procedure was not planned. Chordal attachments to the crest of the ventricular septal defect are not uncommon in patients with transposition complexes such as corrected transposition, complete transposition, and double-outlet right ventricle (24,25). If the pulmonary valve is normal and arterial switch is an option, the anomalous chordal attachments should not have significant implications for outflow tract reconstruction if these attachments are not significantly obstructive. If the valve is not normal, however, such anomalous chordal attachments can be critical. An option that has been employed to baffle the left ventricle to the aorta in patients with transposition or double-outlet right ventricle, pulmonary stenosis and discreet chordal bundles inserting near or on the rim of the ventricular septal defect is the REV procedure, or conal flap method. With this technique, the anomalous chordal insertions are raised from the septum as one or two pedicled flaps of septal wall based away from the defect, and the outlet septum is resected, after which the Rastelli baffle is performed and the flap(s) are secured to the right ventricular aspect of the baffle patch (24,25). In properly selected patients, this method appears to be effective, with a small but important risk of tricuspid regurgitation. However, it is not an option in patients such as ours, in whom the chordal attachments were multiple, diffuse and bilateral. A technique similar to the conal flap method, which we have employed for biventricular repair in patients with AV valvar straddling, is translocation and realignment of anomalously located chordal bundles (23). This approach may also be useful in cases of discrete chordal bundles inserting into the septum at the rim of the defect, but cannot be used safely with the type of complex attachments seen in these cases. Aside from abandoning the Rastelli procedure, the only option for overcoming the anomalous AV valvar insertions in cases with diffuse, multiple and/or bilateral chordae attaching to or crossing the rim of the defect appears to be valve replacement, which is a suboptimal choice in children. Other forms of functional AV valvar apparatus interfering without outflow tract repair. In four other cases, the AV valvar tensor apparatus interfered with potential outflow tract reconstruction. A Ross-Konno procedure was either necessary because chordal attachments to the outlet septum limited the extent of infundibular septal resection that could be achieved (n 1), or was not possible owing to functional AV valvar apparatus obstructing the subaortic region (n 1). The Ross-Konno procedure has proved to be a very important addition to the repertoire of the cardiac surgeon managing systemic outflow tract disease (26). As these two cases illustrate, the Ross or Ross-Konno operations may increase the options for treatment of outflow tract obstruction due to anomalies of the AV valvar apparatus, but such anomalies may also interfere with planned reconstruction using the Ross- Konno procedure. In a patient with right isomerism, multiple chordae from the common AV valve were obstructing both outflow tracts, and the patient died after a modified Damus-Kaye-Stansel proce-
7 JACC Vol. 32, No. 6 November 15, 1998: MCELHINNEY ET AL. ACCESSORY AV VALVAR TISSUE 1747 dure, with residual outflow obstruction. Anomalous AV valvar structures have been shown to contribute to outflow obstruction in a considerable proportion of patients with isomerism (20), although such involvement is more common in left than in right isomerism, and is often due to accessory rather than functional tissue. Unlike anomalies amenable to biventricular repair, there are few good options for managing outflow obstruction attributable to functional AV valvar apparatus in the setting of univentricular heart disease. Residual outflow obstruction and AV valvar regurgitation (a potential consequence of resecting AV valvar tissue) are not well-tolerated and can have significant implications for ventricular function, and relocation of the systemic outflow tract with a conduit (as in the Rastelli procedure) is not feasible in univentricular palliation. Types of AV valvar apparatus causing outflow tract obstruction not included in this study. Two of the most important and most common forms of subaortic obstruction caused in part by the AV valvar apparatus have not been included in this study. Systolic anterior motion of the mitral valve in hypertrophic obstructive cardiomyopathy was not included primarily because this dynamic process has traditionally been considered a functional abnormality, ascribed to the Venturi effect in the setting of severe septal muscular obstruction, and not a structural abnormality of anomalous AV valvar tissue or attachments. It has been proposed, however, that a critical feature of subaortic obstruction due to systolic anterior motion of the mitral valve is anterior displacement of the papillary muscles (27). It is also known that elongation of the mitral valve leaflets are present in patients with this lesion. Thus, this is still considered more a functional than a structural anomaly of the AV valvar apparatus. The other recognized form of AV valve-related outflow tract obstruction that was not included in this report was subaortic obstruction due to the narrowed and elongated left ventricular outflow tract in AV septal defect, which is in part a function of the deficient interventricular septum and consequent displacement of the base of the anterior left AV valvar leaflet (28). Although this can be managed in some cases with reconstruction of the anterior leaflet at its basal attachment (18), this problem is more a function of abnormal development of the AV septum than of the valves or valvar apparatus per se. We did include three patients with previously repaired complete AV septal defect in this series, but all three patients had abnormalities of the subvalvar apparatus that contributed to subaortic obstruction. Although replaced or reconstructed AV valves are not native anomalies, it is worth noting that they can cause left ventricular outflow tract obstruction as well (29,30). Conclusions. Although rare, anomalous functional and accessory AV valvar apparatus are potentially important causes of both pulmonary and systemic outflow tract obstruction. Most often, accessory tissue is involved, and is thus amenable to resection. As others have suggested (11), because accessory AV valvar tissue causing outflow tract obstruction is not a commonly observed condition and may be difficult to identify intraoperatively in the arrested heart, it may be overlooked. Accordingly, its prevalence may be underestimated and it may contribute to residual obstruction and even death (1,5 7,19). This mechanism of obstruction is becoming recognized in an increasing number of cases, in part due to the widespread use and increasing facility with echocardiography, and should be considered regularly as a potential cause of outflow tract obstruction. The relationship between outflow tract obstruction due to AV valvar anomalies and fibrous or muscular causes is not known. It has been proposed that turbulent flow caused by AV valvar tissue in the outflow tract may promote fibrous proliferation and membranous subvalvar obstruction. By the same token, flow disturbances and hemodynamic forces across the AV valvar tissue may stimulate growth of the accessory AV valvar tissue itself. Regardless, there are no reports of which we are aware that accessory tissue recurs after resection. Because accessory tissue can be resected completely and does not appear to redevelop, the outlook for this type of outflow tract obstruction is favorable. However, because accessory tissue is often found with discrete fibromuscular or tunnel type subaortic obstruction, patients may be subject to recurrence of these components. Conversely, patients with outflow tract obstruction due to functional AV valvar tissue may be compromised as a result, either because the obstruction cannot be relieved completely or because the repair of choice cannot be performed. References 1. Sellers RD, Lillehei CW, Edwards JE. Subaortic stenosis caused by anomalies of the atrioventricular valves. J Thorac Cardiovasc Surg 1964;48: Shrivastava S, Tadavarthy SM, Fukuda T, Edwards JE. Anatomic causes of pulmonary stenosis in complete transposition. Circulation 1976;54: Asciutto RJ, Ross-Asciutto NT, Kopf GS, Kleinman CS, Talner NS. Accessory mitral valve tissue causing left ventricular outflow obstruction (two-dimensional echocardiographic diagnosis and surgical approach). Ann Thorac Surg 1986;42: Das SK, Jahnke EJ, Walker WJ. Aneurysm of the membranous septum with interventricular septal defect producing right ventricular outflow obstruction. Circulation 1964;30: Gomes AS, Nath PH, Singhe A, et al. Accessory flaplike tissue causing ventricular outflow obstruction. J Thorac Cardiovasc Surg 1980;80: Hatem J, Sade RM, Taylor A, et al. Supernumerary mitral valve producing subaortic stenosis. Chest 1981;79: Kelly DT, Wulfsberg E, Rowe RD. Discrete subaortic stenosis. Circulation 1972;46: Chin AJ, Yeager SB, Sanders SP, et al. Accuracy of prospective twodimensional echocardiographic evaluation of left ventricular outflow tract in complete transposition of the great arteries. Am J Cardiol 1985;55: Feigl D, Feigl A, Sweetman KM, Lobo FV, Moller JH, Edwards JE. Accessory tissue of the tricuspid valve protruding into the left ventricle through a septal defect. Arch Pathol Lab Med 1986;110: Furuta N, Luhmer I, Hetzer R, Kallfelz HC. Abnormal accessory mitral leaflet simulating left ventricular outflow tract tumor. Thorac Cardiovasc Surg 1983;31: Garrett HE, Spray TL. Accessory mitral valve tissue: an increasingly recognized cause of left ventricular outflow tract obstruction. J Cardiovasc Surg 1990;31: Marino B, Gagliardi MG, Digilio MC, et al. Noonan syndrome: structural abnormalities of the mitral valve causing subaortic obstruction. Eur J Pediatr 1995;154:
8 1748 MCELHINNEY ET AL. JACC Vol. 32, No. 6 ACCESSORY AV VALVAR TISSUE November 15, 1998: Wada Y, Kawai T, Oga K, Oka T. Left ventricular outflow tract obstruction and mitral regurgitation caused by papillary muscle abnormalities: a case report [letter]. J Thorac Cardiovasc Surg 1993;106: Ow EP, DeLeon SY, Freeman JE, et al. Recognition and management of accessory mitral tissue causing severe subaortic stenosis. Ann Thorac Surg 1994;57: Idriss FS, Muster AJ, Paul MH, Backer CL, Mavroudis C. Ventricular septal defect with tricuspid pouch with and without transposition: anatomic and surgical considerations. J Thorac Cardiovasc Surg 1992;103: Kaneko Y, Okabe H, Nagata N, Kanemoto S, Yamada S. Right ventricular obstruction by tricuspid pouch in simple ventricular septal defect. Ann Thorac Surg 1998;65: DeLeon SY, Ilbawi MN, Wilson WR, et al. Surgical options in subaortic stenosis associated with endocardial cushion defects. Ann Thorac Surg 1991;52: Van Arsdell GS, Williams WG, Boutin C, et al. Subaortic stenosis in the spectrum of atrioventricular septal defects: solutions may be complex and palliative. J Thorac Cardiovasc Surg 1995;110: Nanton MA, Belcourt, Gillis DA, Krause VW, Roy DL. Left ventricular outflow tract obstruction owing to accessory endocardial cushion tissue. J Thorac Cardiovasc Surg 1979;78: Van Praagh S, Geva T, Friedberg DZ, et al. Aortic outflow obstruction in visceral heterotaxy: a study based on twenty postmortem cases. Am Heart J 1997;133: Kon MWS, Grech ED, Ho SY, Bennett JG, Collins PD. Anomalous papillary muscle as a cause of left ventricular outflow tract obstruction in an adult. Ann Thorac Surg 1997;63: Reddy VM, McElhinney DB, Silverman NH, Hanley FL. The double switch procedure for anatomical repair of congenitally corrected transposition of the great arteries in infants and children. Eur Heart J 1997;18: Reddy VM, Liddicoat JR, McElhinney DB, Brook MM, van Son JAM, Hanley FL. Biventricular repair of lesions with straddling tricuspid valves using techniques of cordal translocation and realignment. Cardiol Young 1997;7: Lecompte Y, Neveux JY, Leca F, et al. Reconstruction of the pulmonary outflow tract without prosthetic conduit. J Thorac Cardiovasc Surg 1982;84: Niinami H, Imai Y, Sawatari K, Hoshino S, Ishihara K, Aoki M. Surgical management of tricuspid malinsertion in the Rastelli operation: conal flap method. Ann Thorac Surg 1995;59: Reddy VM, Rajasinghe HA, Teitel DF, Haas GS, Hanley FL. Aortoventriculoplasty using the pulmonary autograft: the Ross-Konno procedure. J Thorac Cardiovasc Surg 1996;111: Levine RA, Vlahakes GJ, Lefebvre X, et al. Papillary muscle displacement causes systolic anterior motion of the mitral valve: experimental validation and insights into the mechanism of subaortic obstruction. Circulation 1995;91: Suzuki K, Ho SY, Anderson RH, et al. Morphometric analysis of atrioventricular septal defect with common valve orifice. J Am Coll Cardiol 1998;31: De Canniere D, Jansens JL, Unger P, Le Clerc JL. Left ventricular outflow tract obstruction after mitral valve replacement. Ann Thorac Surg 1997;64: Jebara VA, Mihaileanu S, Acar C, et al. Left ventricular outflow tract obstruction after mitral valve repair: results of the sliding leaflet technique. Circulation 1993;88 Suppl II:II30 4.
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT
 LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
Surgical Treatment for Double Outlet Right Ventricle. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016
 1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
The Rastelli procedure has been traditionally used for repair
 En-bloc Rotation of the Truncus Arteriosus A Technique for Complete Anatomic Repair of Transposition of the Great Arteries/Ventricular Septal Defect/Left Ventricular Outflow Tract Obstruction or Double
En-bloc Rotation of the Truncus Arteriosus A Technique for Complete Anatomic Repair of Transposition of the Great Arteries/Ventricular Septal Defect/Left Ventricular Outflow Tract Obstruction or Double
Double outlet right ventricle: navigation of surgeon to chose best treatment strategy
 Double outlet right ventricle: navigation of surgeon to chose best treatment strategy Jan Marek Great Ormond Street Hospital & Institute of Cardiovascular Sciences, University College London Double outlet
Double outlet right ventricle: navigation of surgeon to chose best treatment strategy Jan Marek Great Ormond Street Hospital & Institute of Cardiovascular Sciences, University College London Double outlet
The modified Konno procedure, or subaortic ventriculoplasty,
 Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
In 1980, Bex and associates 1 first introduced the initial
 Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
The Double Switch Using Bidirectional Glenn and Hemi-Mustard. Frank Hanley
 The Double Switch Using Bidirectional Glenn and Hemi-Mustard Frank Hanley No relationships to disclose CCTGA Interesting Points for Discussion What to do when. associated defects must be addressed surgically:
The Double Switch Using Bidirectional Glenn and Hemi-Mustard Frank Hanley No relationships to disclose CCTGA Interesting Points for Discussion What to do when. associated defects must be addressed surgically:
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions
 Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Anatomy of Atrioventricular Septal Defect (AVSD)
 Surgical challenges in atrio-ventricular septal defect in grown-up congenital heart disease Anatomy of Atrioventricular Septal Defect (AVSD) S. Yen Ho Professor of Cardiac Morphology Royal Brompton and
Surgical challenges in atrio-ventricular septal defect in grown-up congenital heart disease Anatomy of Atrioventricular Septal Defect (AVSD) S. Yen Ho Professor of Cardiac Morphology Royal Brompton and
"Giancarlo Rastelli Lecture"
 "Giancarlo Rastelli Lecture" Surgical treatment of Malpositions of the Great Arteries Pascal Vouhé Giancarlo Rastelli (1933 1970) Cliquez pour modifier les styles du texte du masque Deuxième niveau Troisième
"Giancarlo Rastelli Lecture" Surgical treatment of Malpositions of the Great Arteries Pascal Vouhé Giancarlo Rastelli (1933 1970) Cliquez pour modifier les styles du texte du masque Deuxième niveau Troisième
Tetralogy of Fallot (TOF) with atrioventricular (AV)
 Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
14 Valvular Stenosis
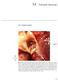 14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
Double Outlet Right Ventricle with Anterior and Left-Sided Aorta and Subpulmonary Ventricular Septal Defect
 Case Report Double Outlet Right Ventricle with Anterior and Left-Sided rta and Subpulmonary Ventricular Septal Defect Luciana Braz Peixoto, Samira Morhy Borges Leal, Carlos Eduardo Suaide Silva, Sandra
Case Report Double Outlet Right Ventricle with Anterior and Left-Sided rta and Subpulmonary Ventricular Septal Defect Luciana Braz Peixoto, Samira Morhy Borges Leal, Carlos Eduardo Suaide Silva, Sandra
Atrioventricular Canal (Septal) Defects. Norman H Silverman MD. D Sc (Med),FACC, FAHA
 Atrioventricular Canal (Septal) Defects Norman H Silverman MD. D Sc (Med),FACC, FAHA Embryology of the A-V Canal Looping NHS. Formation of the Atrial Septum Embryology of the A-V Canal NHS. Development
Atrioventricular Canal (Septal) Defects Norman H Silverman MD. D Sc (Med),FACC, FAHA Embryology of the A-V Canal Looping NHS. Formation of the Atrial Septum Embryology of the A-V Canal NHS. Development
Cardiac Catheterization Cases Primary Cardiac Diagnoses Facility 12 month period from to PRIMARY DIAGNOSES (one per patient)
 PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
Repair of Complete Atrioventricular Septal Defects Single Patch Technique
 Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
An anterior aortoventriculoplasty, known as the Konno-
 The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
TGA Surgical techniques: tips & tricks (Arterial switch operation)
 TGA Surgical techniques: tips & tricks (Arterial switch operation) Seoul National University Children s Hospital Woong-Han Kim Surgical History 1951 Blalock and Hanlon, atrial septectomy 1954 Mustard et
TGA Surgical techniques: tips & tricks (Arterial switch operation) Seoul National University Children s Hospital Woong-Han Kim Surgical History 1951 Blalock and Hanlon, atrial septectomy 1954 Mustard et
Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital
 Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital VSD is one of the most common congenital cardiac abnormalities in the newborn. It can occur as an isolated finding or in combination
Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital VSD is one of the most common congenital cardiac abnormalities in the newborn. It can occur as an isolated finding or in combination
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Congenital Heart Disease An Approach for Simple and Complex Anomalies
 Congenital Heart Disease An Approach for Simple and Complex Anomalies Michael D. Pettersen, MD Director, Echocardiography Rocky Mountain Hospital for Children Denver, CO None Disclosures 1 ASCeXAM Contains
Congenital Heart Disease An Approach for Simple and Complex Anomalies Michael D. Pettersen, MD Director, Echocardiography Rocky Mountain Hospital for Children Denver, CO None Disclosures 1 ASCeXAM Contains
Accessory mitral valve tissue causing left ventricular outflow tract obstruction
 Br Heart 1986; 55: 376-80 Accessory mitral valve tissue causing left ventricular outflow tract obstruction W G MELDRUM-HANNA, T B CARTMILL, R E HAWKER, J M CELERMAJER, C M WRIGHT From the Basser Institute
Br Heart 1986; 55: 376-80 Accessory mitral valve tissue causing left ventricular outflow tract obstruction W G MELDRUM-HANNA, T B CARTMILL, R E HAWKER, J M CELERMAJER, C M WRIGHT From the Basser Institute
Congenital Heart Defects
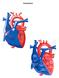 Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
Absent Pulmonary Valve Syndrome
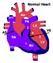 Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Atrioventricular valve repair: The limits of operability
 Atrioventricular valve repair: The limits of operability Francis Fynn-Thompson, MD Co-Director, Center for Airway Disorders Surgical Director, Pediatric Mechanical Support Program Surgical Director, Heart
Atrioventricular valve repair: The limits of operability Francis Fynn-Thompson, MD Co-Director, Center for Airway Disorders Surgical Director, Pediatric Mechanical Support Program Surgical Director, Heart
Common Defects With Expected Adult Survival:
 Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE
 SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
TGA, VSD, and LVOTO. Cheul Lee, MD. Department of Thoracic and Cardiovascular Surgery Sejong General Hospital
 Surgical Management of TGA, VSD, and LVOTO Cheul Lee, MD Department of Thoracic and Cardiovascular Surgery Sejong General Hospital TGA, VSD, and LVOTO Incidence : 0.7% of all CHD 20% of TGA with VSD 4%
Surgical Management of TGA, VSD, and LVOTO Cheul Lee, MD Department of Thoracic and Cardiovascular Surgery Sejong General Hospital TGA, VSD, and LVOTO Incidence : 0.7% of all CHD 20% of TGA with VSD 4%
Anomalous Systemic Venous Connection Systemic venous anomaly
 World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
Recent technical advances and increasing experience
 Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Children with Single Ventricle Physiology: The Possibilities
 Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
Perioperative Management of DORV Case
 Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
IMAGES. in PAEDIATRIC CARDIOLOGY. Abstract. Case
 IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232604 Isolated subpulmonary membrane causing critical neonatal pulmonary stenosis with concordant atrioventricular and ventriculoarterial connections
IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232604 Isolated subpulmonary membrane causing critical neonatal pulmonary stenosis with concordant atrioventricular and ventriculoarterial connections
Heart and Lungs. LUNG Coronal section demonstrates relationship of pulmonary parenchyma to heart and chest wall.
 Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Although most patients with Ebstein s anomaly live
 Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease
 Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Most common fetal cardiac anomalies
 Most common fetal cardiac anomalies Common congenital heart defects CHD % of cardiac defects Chromosomal Infants Fetuses anomaly (%) 22q11 deletion (%) VSD 30 5~10 20~40 10 PS 9 5 (PA w/ VSD) HLHS 7~9
Most common fetal cardiac anomalies Common congenital heart defects CHD % of cardiac defects Chromosomal Infants Fetuses anomaly (%) 22q11 deletion (%) VSD 30 5~10 20~40 10 PS 9 5 (PA w/ VSD) HLHS 7~9
Echocardiographic assessment in Adult Patients with Congenital Heart Diseases
 Echocardiographic assessment in Adult Patients with Congenital Heart Diseases Athanasios Koutsakis Cardiologist, Cl. Research Fellow George Giannakoulas Ass. Professor in Cardiology 1st Cardiology Department,
Echocardiographic assessment in Adult Patients with Congenital Heart Diseases Athanasios Koutsakis Cardiologist, Cl. Research Fellow George Giannakoulas Ass. Professor in Cardiology 1st Cardiology Department,
Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect
 Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect David M. Overman Division of Pediatric Cardiac Surgery The Children s Heart Clinic Chief, Division
Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect David M. Overman Division of Pediatric Cardiac Surgery The Children s Heart Clinic Chief, Division
Avariety of conditions can prevent a successful biventricular
 Which Two Ventricles Cannot Be Used for a Biventricular Repair? Echocardiographic Assessment Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
Which Two Ventricles Cannot Be Used for a Biventricular Repair? Echocardiographic Assessment Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
Echocardiography in Congenital Heart Disease
 Chapter 44 Echocardiography in Congenital Heart Disease John L. Cotton and G. William Henry Multiple-plane cardiac imaging by echocardiography can noninvasively define the anatomy of the heart and the
Chapter 44 Echocardiography in Congenital Heart Disease John L. Cotton and G. William Henry Multiple-plane cardiac imaging by echocardiography can noninvasively define the anatomy of the heart and the
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY
 가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
Surgical Treatment for Atrioventricular Septal Defect. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
5.8 Congenital Heart Disease
 5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
What is the Definition of Small Systemic Ventricle. Hong Ryang Kil, MD Department of Pediatrics, College of Medicine, Chungnam National University
 What is the Definition of Small Systemic Ventricle Hong Ryang Kil, MD Department of Pediatrics, College of Medicine, Chungnam National University Contents Introduction Aortic valve stenosis Aortic coarctation
What is the Definition of Small Systemic Ventricle Hong Ryang Kil, MD Department of Pediatrics, College of Medicine, Chungnam National University Contents Introduction Aortic valve stenosis Aortic coarctation
Surgical Results in Patients With Double Outlet Right Ventricle: A 20-Year Experience
 Surgical Results in Patients With Double Outlet Right Ventricle: A 20-Year Experience John W. Brown, MD, Mark Ruzmetov, MD, Yuji Okada, MD, Palaniswamy Vijay, PhD, MPH, and Mark W. Turrentine, MD Section
Surgical Results in Patients With Double Outlet Right Ventricle: A 20-Year Experience John W. Brown, MD, Mark Ruzmetov, MD, Yuji Okada, MD, Palaniswamy Vijay, PhD, MPH, and Mark W. Turrentine, MD Section
Appendix A.2: Tier 2 Surgical Procedure Terms and Definitions
 Appendix A.2: Tier 2 Surgical Procedure Terms and Definitions Tier 2 surgeries Anomalous Systemic Venous Connection Anomalous Systemic Venous Connection Repair Repair includes a range of surgical approaches,
Appendix A.2: Tier 2 Surgical Procedure Terms and Definitions Tier 2 surgeries Anomalous Systemic Venous Connection Anomalous Systemic Venous Connection Repair Repair includes a range of surgical approaches,
Tricuspid Valve Repair for Ebstein's Anomaly
 Tricuspid Valve Repair for Ebstein's Anomaly Joseph A. Dearani, MD, and Gordon K. Danielson, MD E bstein's anomaly is a malformation of the tricuspid valve and right ventricle that is characterized by
Tricuspid Valve Repair for Ebstein's Anomaly Joseph A. Dearani, MD, and Gordon K. Danielson, MD E bstein's anomaly is a malformation of the tricuspid valve and right ventricle that is characterized by
Transposition of the Great Arteries Preoperative Diagnostic Considerations. John Simpson Evelina Children s Hospital London, UK
 Transposition of the Great Arteries Preoperative Diagnostic Considerations John Simpson Evelina Children s Hospital London, UK Areas to be covered Definitions Scope of occurrence of transposition of the
Transposition of the Great Arteries Preoperative Diagnostic Considerations John Simpson Evelina Children s Hospital London, UK Areas to be covered Definitions Scope of occurrence of transposition of the
Introduction. Study Design. Background. Operative Procedure-I
 Risk Factors for Mortality After the Norwood Procedure Using Right Ventricle to Pulmonary Artery Shunt Ann Thorac Surg 2009;87:178 86 86 Addressor: R1 胡祐寧 2009/3/4 AM7:30 SICU 討論室 Introduction Hypoplastic
Risk Factors for Mortality After the Norwood Procedure Using Right Ventricle to Pulmonary Artery Shunt Ann Thorac Surg 2009;87:178 86 86 Addressor: R1 胡祐寧 2009/3/4 AM7:30 SICU 討論室 Introduction Hypoplastic
Echocardiography in Adult Congenital Heart Disease
 Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
S. Bert Litwin, MD. Preface
 Preface Because of the wide variety of anomalies encountered in congenital heart surgery, a broad understanding of the pathologic anatomy of defects is vitally important to the surgeon. More than in many
Preface Because of the wide variety of anomalies encountered in congenital heart surgery, a broad understanding of the pathologic anatomy of defects is vitally important to the surgeon. More than in many
Techniques for repair of complete atrioventricular septal
 No Ventricular Septal Defect Patch Atrioventricular Septal Defect Repair Carl L. Backer, MD *, Osama Eltayeb, MD *, Michael C. Mongé, MD *, and John M. Costello, MD For the past 10 years, our center has
No Ventricular Septal Defect Patch Atrioventricular Septal Defect Repair Carl L. Backer, MD *, Osama Eltayeb, MD *, Michael C. Mongé, MD *, and John M. Costello, MD For the past 10 years, our center has
CMR for Congenital Heart Disease
 CMR for Congenital Heart Disease * Second-line tool after TTE * Strengths of CMR : tissue characterisation, comprehensive access and coverage, relatively accurate measurements of biventricular function/
CMR for Congenital Heart Disease * Second-line tool after TTE * Strengths of CMR : tissue characterisation, comprehensive access and coverage, relatively accurate measurements of biventricular function/
Surgical Myectomy for HOCM
 Surgical Myectomy for HOCM Volkmar Falk Deutsches Herzzentrum Berlin Different Pathology of HOCM Impact on surgical strategy Said SM Expert Rev Cardiovasc Ther 2013 Different Pathology of HOCM Impact on
Surgical Myectomy for HOCM Volkmar Falk Deutsches Herzzentrum Berlin Different Pathology of HOCM Impact on surgical strategy Said SM Expert Rev Cardiovasc Ther 2013 Different Pathology of HOCM Impact on
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin. Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong
 Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Treatment of congenital aortic valve disease: Neonatal surgical management. Pascal Vouhé - Sick Children Hospital, Paris
 Treatment of congenital aortic valve disease: Neonatal surgical management Pascal Vouhé - Sick Children Hospital, Paris Challenges. valvar lesions. associated lesions. status of left ventricle Valvar lesions.
Treatment of congenital aortic valve disease: Neonatal surgical management Pascal Vouhé - Sick Children Hospital, Paris Challenges. valvar lesions. associated lesions. status of left ventricle Valvar lesions.
Segmental approach to normal and abnormal situs arrangement - Echocardiography -
 Segmental approach to normal and abnormal situs arrangement - Echocardiography - Jan Marek Great Ormond Street Hospital & Institute of Cardiovascular Sciences, University College London No disclosures
Segmental approach to normal and abnormal situs arrangement - Echocardiography - Jan Marek Great Ormond Street Hospital & Institute of Cardiovascular Sciences, University College London No disclosures
3/14/2011 MANAGEMENT OF NEWBORNS CARDIAC INTENSIVE CARE CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 WITH HEART DEFECTS
 CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
S plex of parachute mitral valve, supravalvar ring of the. Shone s Anomaly: Operative Results and Late Outcome
 Shone s Anomaly: Operative Results and Late Outcome Steven F. Bolling, MD, Mark D. Iannettoni, MD, Macdonald Dick 11, MD, Amnon Rosenthal, MD, and Edward L. Bove, MD Sections of Thoracic Surgery and Pediatric
Shone s Anomaly: Operative Results and Late Outcome Steven F. Bolling, MD, Mark D. Iannettoni, MD, Macdonald Dick 11, MD, Amnon Rosenthal, MD, and Edward L. Bove, MD Sections of Thoracic Surgery and Pediatric
Yuichi Shiokawa, MD Anton E. Becker, MD, PhD
 THE LEFT VENTRICULAR OUTFLOW TRACT IN ATRIOVENTRICULAR SEPTAL DEFECT REVISITED: SURGICAL CONSIDERATIONS REGARDING PRESERVATION OF AORTIC VALVE INTEGRITY IN THE PERSPECTIVE OF ANATOMIC OBSERVATIONS Yuichi
THE LEFT VENTRICULAR OUTFLOW TRACT IN ATRIOVENTRICULAR SEPTAL DEFECT REVISITED: SURGICAL CONSIDERATIONS REGARDING PRESERVATION OF AORTIC VALVE INTEGRITY IN THE PERSPECTIVE OF ANATOMIC OBSERVATIONS Yuichi
Since first successfully performed by Jatene et al, the
 Long-Term Predictors of Aortic Root Dilation and Aortic Regurgitation After Arterial Switch Operation Marcy L. Schwartz, MD; Kimberlee Gauvreau, ScD; Pedro del Nido, MD; John E. Mayer, MD; Steven D. Colan,
Long-Term Predictors of Aortic Root Dilation and Aortic Regurgitation After Arterial Switch Operation Marcy L. Schwartz, MD; Kimberlee Gauvreau, ScD; Pedro del Nido, MD; John E. Mayer, MD; Steven D. Colan,
List of Videos. Video 1.1
 Video 1.1 Video 1.2 Video 1.3 Video 1.4 Video 1.5 Video 1.6 Video 1.7 Video 1.8 The parasternal long-axis view of the left ventricle shows the left ventricular inflow and outflow tract. The left atrium
Video 1.1 Video 1.2 Video 1.3 Video 1.4 Video 1.5 Video 1.6 Video 1.7 Video 1.8 The parasternal long-axis view of the left ventricle shows the left ventricular inflow and outflow tract. The left atrium
SURGICAL APPROACHES FOR DOUBLE-OUTLET RIGHT VENTRICLE OR TRANSPOSITION OF THE GREAT ARTERIES ASSOCIATED WITH STRADDLING ATRIOVENTRICULAR VALVES
 SURGICAL APPROACHES FOR DOUBLE-OUTLET RIGHT VENTRICLE OR TRANSPOSITION OF THE GREAT ARTERIES ASSOCIATED WITH STRADDLING ATRIOVENTRICULAR VALVES Alain Serraf, MD ~ Tomohiro Nakamura, MD ~ Fran ois Lacour-Gayet,
SURGICAL APPROACHES FOR DOUBLE-OUTLET RIGHT VENTRICLE OR TRANSPOSITION OF THE GREAT ARTERIES ASSOCIATED WITH STRADDLING ATRIOVENTRICULAR VALVES Alain Serraf, MD ~ Tomohiro Nakamura, MD ~ Fran ois Lacour-Gayet,
Data Collected: June 17, Reported: June 30, Survey Dates 05/24/ /07/2010
 Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Congenital heart disease: When to act and what to do?
 Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
S endocardial cushion defects, with the incidence ranging. Surgical Options in Subaortic Stenosis Associated With Endocardia1 Cushion Defects
 Surgical Options in Subaortic Stenosis Associated With Endocardia1 Cushion Defects Serafin Y. DeLeon, MD, Michel N. Ilbawi, MD, William R. Wilson, Jr, MD, Rene A. Arcilla, MD, Otto G. Thilenius, MD, Saroja
Surgical Options in Subaortic Stenosis Associated With Endocardia1 Cushion Defects Serafin Y. DeLeon, MD, Michel N. Ilbawi, MD, William R. Wilson, Jr, MD, Rene A. Arcilla, MD, Otto G. Thilenius, MD, Saroja
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical)
 September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
Characteristics and Management of Cleft Mitral Valve
 Journal of the American College of Cardiology Vol. 42, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.07.019
Journal of the American College of Cardiology Vol. 42, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.07.019
Cases in Adult Congenital Heart Disease
 Cases in Adult Congenital Heart Disease Sabrina Phillips, MD FACC FASE Associate Professor of Medicine The University of Oklahoma Health Sciences Center No Disclosures I Have Palpitations 18 Year old Man
Cases in Adult Congenital Heart Disease Sabrina Phillips, MD FACC FASE Associate Professor of Medicine The University of Oklahoma Health Sciences Center No Disclosures I Have Palpitations 18 Year old Man
Damus-Kaye-Stansel Procedure: Midterm Follow-up and Technical Considerations
 Damus-Kaye-Stansel Procedure: Midterm Follow-up and Technical Considerations Thomas L. Carter, MD, Richard D. Mainwaring, MD, and John J. Lamberti, MD Division of Cardiac Surgery, Children's Hospital and
Damus-Kaye-Stansel Procedure: Midterm Follow-up and Technical Considerations Thomas L. Carter, MD, Richard D. Mainwaring, MD, and John J. Lamberti, MD Division of Cardiac Surgery, Children's Hospital and
ADULT CONGENITAL HEART DISEASE. Stuart Lilley
 ADULT CONGENITAL HEART DISEASE Stuart Lilley More adults than children have congenital heart disease Huge variety of congenital lesions from minor to major Heart failure, re-operation and arrhythmia are
ADULT CONGENITAL HEART DISEASE Stuart Lilley More adults than children have congenital heart disease Huge variety of congenital lesions from minor to major Heart failure, re-operation and arrhythmia are
The role of intraoperative TOE in congenital cardiac surgery
 The role of intraoperative TOE in congenital cardiac surgery Justiaan Swanevelder Dept of Anaesthesia Groote Schuur and Red Cross War Memorial Children s Hospitals University of Cape Town, South Africa
The role of intraoperative TOE in congenital cardiac surgery Justiaan Swanevelder Dept of Anaesthesia Groote Schuur and Red Cross War Memorial Children s Hospitals University of Cape Town, South Africa
Surgical Treatment of Aortic Arch Hypoplasia
 Surgical Treatment of Aortic Arch Hypoplasia In the early 1990s, 25% of patients could face mortality related to complica-tions of hypertensive disease Early operations and better surgical techniques should
Surgical Treatment of Aortic Arch Hypoplasia In the early 1990s, 25% of patients could face mortality related to complica-tions of hypertensive disease Early operations and better surgical techniques should
An understanding of the many factors involved in the
 Atrioventricular Valve Dysfunction: Evaluation by Doppler and Cross-Sectional Ultrasound Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
Atrioventricular Valve Dysfunction: Evaluation by Doppler and Cross-Sectional Ultrasound Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
HISTORY. Question: What category of heart disease is suggested by this history? CHIEF COMPLAINT: Heart murmur present since early infancy.
 HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
Outline. Congenital Heart Disease. Special Considerations for Special Populations: Congenital Heart Disease
 Special Considerations for Special Populations: Congenital Heart Disease Valerie Bosco, FNP, EdD Alison Knauth Meadows, MD, PhD University of California San Francisco Adult Congenital Heart Program Outline
Special Considerations for Special Populations: Congenital Heart Disease Valerie Bosco, FNP, EdD Alison Knauth Meadows, MD, PhD University of California San Francisco Adult Congenital Heart Program Outline
M/3, cc-tga, PS, BCPC(+) Double Switch Operation
 2005 < Pros & Cons > M/3, cc-tga, PS, BCPC(+) Double Switch Operation Congenitally corrected TGA Atrio-Ventricular & Ventriculo-Arterial discordance Physiologically corrected circulation with the morphologic
2005 < Pros & Cons > M/3, cc-tga, PS, BCPC(+) Double Switch Operation Congenitally corrected TGA Atrio-Ventricular & Ventriculo-Arterial discordance Physiologically corrected circulation with the morphologic
How to Assess and Treat Obstructive Lesions
 How to Assess and Treat Obstructive Lesions Erwin Oechslin, MD, FESC, FRCPC, Director, Congenital Cardiac Centre for Adults Peter Munk Cardiac Centre University Health Network/Toronto General Hospital
How to Assess and Treat Obstructive Lesions Erwin Oechslin, MD, FESC, FRCPC, Director, Congenital Cardiac Centre for Adults Peter Munk Cardiac Centre University Health Network/Toronto General Hospital
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment
 Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Since the Ross procedure was first described in 1967
 Ross-Konno Procedure With Mitral Valve Surgery Norihiko Oka, MD, PhD, Osman Al-Radi, MD, Abdullah A. Alghamdi, MD, Siho Kim, MD, and Christopher A. Caldarone, MD Division of Cardiovascular Surgery, The
Ross-Konno Procedure With Mitral Valve Surgery Norihiko Oka, MD, PhD, Osman Al-Radi, MD, Abdullah A. Alghamdi, MD, Siho Kim, MD, and Christopher A. Caldarone, MD Division of Cardiovascular Surgery, The
Hemodynamic assessment after palliative surgery
 THERAPY AND PREVENTION CONGENITAL HEART DISEASE Hemodynamic assessment after palliative surgery for hypoplastic left heart syndrome PETER LANG, M.D., AND WILLIAM I. NORWOOD, M.D., PH.D. ABSTRACT Ten patients
THERAPY AND PREVENTION CONGENITAL HEART DISEASE Hemodynamic assessment after palliative surgery for hypoplastic left heart syndrome PETER LANG, M.D., AND WILLIAM I. NORWOOD, M.D., PH.D. ABSTRACT Ten patients
Excellence in heart and lung care. Royal Brompton Hospital, Sydney Street, London SW3 6NP
 The Surgical Management of the Bicuspid Aortic Valve in Children Professor D.F Shore Royal Brompton & Harefield NHS Trust Excellence in heart and lung care Royal Brompton Hospital, Sydney Street, London
The Surgical Management of the Bicuspid Aortic Valve in Children Professor D.F Shore Royal Brompton & Harefield NHS Trust Excellence in heart and lung care Royal Brompton Hospital, Sydney Street, London
The Edge-to-Edge Technique f For Barlow's Disease
 The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy
 Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Adel Hasanin Ahmed 1 ASD
 Adel Hasanin Ahmed 1 ASD Atrial septal defect (ASD) is the commonest form of congenital heart disease seen in adults. The commonest form of defect is the secundum ASD, accounting for two thirds of cases,
Adel Hasanin Ahmed 1 ASD Atrial septal defect (ASD) is the commonest form of congenital heart disease seen in adults. The commonest form of defect is the secundum ASD, accounting for two thirds of cases,
ULTRASOUND OF THE FETAL HEART
 ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
Segmental Analysis. Gautam K. Singh, M.D. Washington University School of Medicine St. Louis
 Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve
 OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
Preoperative Echocardiographic Assessment of Uni-ventricular Repair
 Preoperative Echocardiographic Assessment of Uni-ventricular Repair Salem Deraz, MD Pediatric Cardiologist, Aswan Heart Centre Magdi Yacoub Heart Foundation Uni-ventricular repair A single or series of
Preoperative Echocardiographic Assessment of Uni-ventricular Repair Salem Deraz, MD Pediatric Cardiologist, Aswan Heart Centre Magdi Yacoub Heart Foundation Uni-ventricular repair A single or series of
BIVENTRICULAR REPAIR FOR AORTIC ATRESIA OR HYPOPLASIA AND VENTRICULAR SEPTAL DEFECT
 BIVENTRICULAR REPAIR FOR AORTIC ATRESIA OR HYPOPLASIA AND VENTRICULAR SEPTAL DEFECT Richard G. Ohye, MD a Koji Kagisaki, MD a Lisa A. Lee, MD b Ralph S. Mosca, MD a Caren S. Goldberg, MD b Edward L. Bove,
BIVENTRICULAR REPAIR FOR AORTIC ATRESIA OR HYPOPLASIA AND VENTRICULAR SEPTAL DEFECT Richard G. Ohye, MD a Koji Kagisaki, MD a Lisa A. Lee, MD b Ralph S. Mosca, MD a Caren S. Goldberg, MD b Edward L. Bove,
Communication of Mitral Valve with Both Ventricles Associated with Double Outlet Right Ventricle
 Communication of Mitral Valve with Both Ventricles Associated with Double Outlet Right Ventricle By RAJENTDRA TANDON, M.D., JAMES H. MOLLR, MD, AND JESSE E. EDWARDS, M.D. SUMMARY A rare case of an infant
Communication of Mitral Valve with Both Ventricles Associated with Double Outlet Right Ventricle By RAJENTDRA TANDON, M.D., JAMES H. MOLLR, MD, AND JESSE E. EDWARDS, M.D. SUMMARY A rare case of an infant
Breakout Session: Transesophageal Echocardiography
 Breakout Session: Transesophageal Echocardiography Doris Ockert, MD Andrew Schroeder, MD University of Wisconsin School of Medicine and Public Health Jutta Novalija, MD, PhD Medical College of Wisconsin
Breakout Session: Transesophageal Echocardiography Doris Ockert, MD Andrew Schroeder, MD University of Wisconsin School of Medicine and Public Health Jutta Novalija, MD, PhD Medical College of Wisconsin
Cardiac ultrasound protocols
 Cardiac ultrasound protocols IDEXX Telemedicine Consultants Two-dimensional and M-mode imaging planes Right parasternal long axis four chamber Obtained from the right side Displays the relative proportions
Cardiac ultrasound protocols IDEXX Telemedicine Consultants Two-dimensional and M-mode imaging planes Right parasternal long axis four chamber Obtained from the right side Displays the relative proportions
Surgery For Ebstein Anomaly
 Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
The first report of the Society of Thoracic Surgeons
 REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
What is Ebstein Anomaly?
 Echocardiograpnhic Evaluation of : Definition, Detection and Determinants of Outcome P. W. O Leary, M.D. Division of Pediatric Cardiology Mayo Clinic No Conflicts to Disclose What is? Failure of the TV
Echocardiograpnhic Evaluation of : Definition, Detection and Determinants of Outcome P. W. O Leary, M.D. Division of Pediatric Cardiology Mayo Clinic No Conflicts to Disclose What is? Failure of the TV
