Name: Period: Date: Day What happens to the temperature in a compost bin during decomposition? Interpret: Be able to interpret graphs.
|
|
|
- Randall Day
- 5 years ago
- Views:
Transcription
1 Review for 7 th Science 1 st Semester Exam Answer the questions. Day What happens to the temperature in a compost bin during decomposition? Increases Interpret: Be able to interpret graphs. 23. What happens to the organic matter during decomposition? It is broken down into useful compounds and nutrients. 24. What energy transformation occurs as biomass decays in a compost bin? Chemical energy Thermal energy 25. What materials in a compost bin will be recycled into soil-enriching nutrients? Biomass material from living & once living organisms. (Ex: newspapers, fruits, vegetables). 26. During decomposition, what does the bacteria in the soil do? Changes Nitrogen into a useable form. 27. Where does a herbivore get it nutrients from in its habitat? By eating plants that receive its energy from the Sun. 28. In a compost bin, what process leads to the cycling of organic matter? Decomposition 29. What gas do plants release as a result of photosynthesis? O2 - Oxygen 30. What energy transformation occurs during photosynthesis? Radiant energy Chemical energy Page 1 of 7
2 31. Glucose helps maintain life on our planet by storing what type of energy? Chemical energy 32. The diagram provided illustrates the process of photosynthesis, including the substances used by and produced by plants. What is the identity of the chemical represented by the question mark in the diagram? Glucose 33. Show how energy flows through a food chain? Sun Producers Herbivores (Primary Consumers) Carnivores (Secondary Consumers) Top Predators (Tertiary Consumers) Identify: Producers, Consumers, Herbivores, & Carnivores in a Food Chain or Web. 34. Define Producer: Organisms that make complex, energy-containing biomolecules from simple inorganic molecules using energy captured from light. 35. Define Consumer: An organism that must consume other organisms for nutrients. 36. Define Herbivore: Organisms that consume only plants. 37. Define Carnivore: Animals that eat other animals. Page 2 of 7
3 38. What element is found in all organic compounds? Carbon 39. What is Denitrification? Bacteria returns nitrogen gas to the atmosphere. 40. What is the difference between a primary, secondary, and tertiary consumer? Primary: feeds on producers. Secondary: eats other animals that eat plants. Tertiary: eats secondary consumers. Identify: the paths that energy would flow through an ecosystem diagram. An example of an ecosystem diagram is below. Page 3 of 7
4 41. A diagram is provided. Which portion of the diagram represents the level with the most available energy? Producers 42. Why are bacteria so important to the nitrogen cycle? It helps turn nitrogen gas into a useable form for plants and animals. Identify: Using a portion of the Periodic Table or an illustration of chemical compounds be able to identify if a compound is organic or inorganic. 43. What element must a compound contain to be organic? Carbon Page 4 of 7
5 44. What are the elements found in organic compounds? (CHNOPS) Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, Sulfur 45. The table provides information on the function of different digestive structures. Which of the digestive structures in the table can cause a chemical change to occur? Stomach releases acid to break food into a smaller new substance. 46. What are Carbohydrates made of? Simple sugars. List the smaller molecule that each large molecule is broken down into by digestion. 47. Lipid to Fatty acids 48. Carbohydrate to Simple sugars 49. Proteins to Amino acids 50. What is a chemical change? A change that alters the identity of a substance, resulting in a new substance or substance with different properties. 51. List examples of chemical changes during digestion? -Stomach adding acids to breakdown food into a new substance. -Saliva adding enzymes to breakdown starch. 52. What is a physical change? A change to a substance without forming a new substance, such as changing size or state of matter. Page 5 of 7
6 53. List examples of physical changes during digestion? -Crushing/cutting food in your mouth. 54. What are indicators a new substance has been formed? Gas production; color change; precipitate (solid) formed; produce heat/light. 55. What is the formula used to calculate work? W=Force (F) x Distance (D) 56. Label the pyramid for work. ***OMIT*** Three situations in which force is applied to a box are illustrated. Describe what work is being done in each situation and how they relate to each other. 57. Situation 1: Yes work lifted a distance of 1 m. (same as Situation 3) 58. Situation 2: No work no movement. 59. Situation 3: Yes work lifted a distance of 1 m. (same as Situation 1) Page 6 of 7
7 60. When an object is lifted directly from the ground rather than moved with an inclined plane, what will be increased? Force 61. Work is measured in what? Joules (J) 62. What two things have to happen for work to take place? 1. Force applied 2. Shows movement (distance) Page 7 of 7
PHOTOSYNTHESIS (7.5A)
 PHOTOSYNTHESISS (7.5A) 1) What kind of energy is necessary to initiate the process of photosynthesis? A. radiant B. heat C. electrical D. wind 2) What happens to the radiant energy absorbed by plants during
PHOTOSYNTHESISS (7.5A) 1) What kind of energy is necessary to initiate the process of photosynthesis? A. radiant B. heat C. electrical D. wind 2) What happens to the radiant energy absorbed by plants during
Warm-Up. Distinguish between an element and a compound. Element. Compound
 Warm-Up Distinguish between an element and a compound. Element Compound Warm-Up Distinguish between an element and a compound. Element Cannot be broken down into anything smaller Found on the Periodic
Warm-Up Distinguish between an element and a compound. Element Compound Warm-Up Distinguish between an element and a compound. Element Cannot be broken down into anything smaller Found on the Periodic
1st 9 Weeks Test Review Sheet
 1st 9 Weeks Test Review Sheet 1. Which two are reactants of photosynthesis? a. glucose and water b. carbon dioxide and water c. glucose and oxygen d. sunlight and ATP energy 2. Photosynthesis makes which
1st 9 Weeks Test Review Sheet 1. Which two are reactants of photosynthesis? a. glucose and water b. carbon dioxide and water c. glucose and oxygen d. sunlight and ATP energy 2. Photosynthesis makes which
Chemical Compounds in Cells
 Questions: Five study Questions EQ: What are the functions of carbohydrates, lipids, proteins, and nucleic acids? All things are made of tiny particles known as elements and compounds. One reason why cells
Questions: Five study Questions EQ: What are the functions of carbohydrates, lipids, proteins, and nucleic acids? All things are made of tiny particles known as elements and compounds. One reason why cells
Biology Milestone: Unit 2 Topics (Energy Transformations)
 Biology Milestone: Unit 2 Topics (Energy Transformations) Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Population density is found by dividing the number
Biology Milestone: Unit 2 Topics (Energy Transformations) Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Population density is found by dividing the number
C H O N P S. Name : Color the Elements on the Periodic Table as listed below
 Name : Unit 9: and Changes in Digestion (if found, please return to Room 714) 7.6A Organic Compounds Identify that organic compounds contain CHONPS. Organic Compounds contain what? 6 1 8 7 15 16 C H O
Name : Unit 9: and Changes in Digestion (if found, please return to Room 714) 7.6A Organic Compounds Identify that organic compounds contain CHONPS. Organic Compounds contain what? 6 1 8 7 15 16 C H O
Standard B-3: The student will demonstrate an understanding of the flow of energy within and between living systems.
 B-3.1 Summarize the overall process by which photosynthesis converts solar energy into chemical energy and interpret the chemical equation for the process. Taxonomy Level: 2.4-B and 2.1-B Understand Conceptual
B-3.1 Summarize the overall process by which photosynthesis converts solar energy into chemical energy and interpret the chemical equation for the process. Taxonomy Level: 2.4-B and 2.1-B Understand Conceptual
Energy Flow Through an Ecosystem
 Energy Flow Through an Ecosystem Food Chains, Food Webs, Energy Pyramids 1 Ecosystems An ecosystem is all of the organisms living in an area together with their physical environment. Ex. An oak forest,
Energy Flow Through an Ecosystem Food Chains, Food Webs, Energy Pyramids 1 Ecosystems An ecosystem is all of the organisms living in an area together with their physical environment. Ex. An oak forest,
Chapter 3 CELL PROCESSES AND ENERGY
 Chapter 3 CELL PROCESSES AND ENERGY Section 1: Chemical Compounds in Cells Elements= Any substance that cannot be broken down into a simpler form Made up of only one kind of atom Found in the body Carbon
Chapter 3 CELL PROCESSES AND ENERGY Section 1: Chemical Compounds in Cells Elements= Any substance that cannot be broken down into a simpler form Made up of only one kind of atom Found in the body Carbon
Recycle me! 4.7. Decomposers
 .7 Recycle me! Tyrannosaurus rex stalked the Earth over 6 million years ago. We have found the bones, but what happened to the atoms that made up its flesh? What will happen to the atoms in your body when
.7 Recycle me! Tyrannosaurus rex stalked the Earth over 6 million years ago. We have found the bones, but what happened to the atoms that made up its flesh? What will happen to the atoms in your body when
Chemical Formulas. Chemical Formula CH 3 COCHCHOCHClCHNH Lewis Dot Structure
 Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
Organic Compounds. B-3.5 Students will be able to summarize the functions of proteins, carbohydrates, and fats in the human body.
 Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
Unit 4- Energy & Cell Processes
 Unit 4- Energy & Cell Processes Energy in the ecosystem ATP Photosynthesis Cellular Respiration Fermentation By: Mrs. Stahl Biology Background knowledge Unit Layout Energy- where does it all come from?
Unit 4- Energy & Cell Processes Energy in the ecosystem ATP Photosynthesis Cellular Respiration Fermentation By: Mrs. Stahl Biology Background knowledge Unit Layout Energy- where does it all come from?
Chemical Compounds in Cells
 Cell Structure and Function Name Date Class Cell Structure and Function Guided Reading and Study Chemical Compounds in Cells This section identifies the basic building blocks of cells. It also explains
Cell Structure and Function Name Date Class Cell Structure and Function Guided Reading and Study Chemical Compounds in Cells This section identifies the basic building blocks of cells. It also explains
Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together.
 Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
5. Groups A and B in the table below contain molecular formulas of compounds.
 1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
An example of a carbohydrate A) 1 B) 2 C) 3 D) 4
 1. Which chemical formula represents a carbohydrate? A) CH4 B) C3H7O2N C) Cl2H22O11 D) CO2 2. Base your answer to the following question on the diagram below. For each of the following phrases, select
1. Which chemical formula represents a carbohydrate? A) CH4 B) C3H7O2N C) Cl2H22O11 D) CO2 2. Base your answer to the following question on the diagram below. For each of the following phrases, select
Carbon Compounds (2.3) (Part 1 - Carbohydrates)
 Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Unit 2: Cellular Chemistry, Structure, and Physiology Module 2: Cellular Chemistry
 Unit 2: Cellular Chemistry, Structure, and Physiology Module 2: Cellular Chemistry NC Essential Standard: 1.2.1 Explain how cells use buffers to regulate cell ph 4.1.1 Compare the structure and functions
Unit 2: Cellular Chemistry, Structure, and Physiology Module 2: Cellular Chemistry NC Essential Standard: 1.2.1 Explain how cells use buffers to regulate cell ph 4.1.1 Compare the structure and functions
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic
 Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Name Date Class. 2. Is the following sentence true or false? Food is required for the body to. maintain homeostasis, keeping a steady internal state.
 CHAPTER 11 FOOD AND DIGESTION SECTION 11 1 Food and Energy (pages 370-380) This section tells about the six nutrients needed by the body. It also describes the Food Guide Pyramid and how to read labels
CHAPTER 11 FOOD AND DIGESTION SECTION 11 1 Food and Energy (pages 370-380) This section tells about the six nutrients needed by the body. It also describes the Food Guide Pyramid and how to read labels
The building blocks for this molecule are A) amino acids B) simple sugars C) fats D) molecular bases
 1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
Proteins their functions and uses revision 4
 Proteins their functions and uses revision 4 48 minutes 48 marks Page of 4 Q. The diagram shows the digestive system. (a) Complete the following sentences about digestive enzymes. (i) Amylase works in
Proteins their functions and uses revision 4 48 minutes 48 marks Page of 4 Q. The diagram shows the digestive system. (a) Complete the following sentences about digestive enzymes. (i) Amylase works in
Cell Processes Review
 1. Most green algae are able to obtain carbon dioxide from the environment and use it to synthesize organic compounds. This activity is an example of 1) hydrolysis 2) saprophytism 3) cellular respiration
1. Most green algae are able to obtain carbon dioxide from the environment and use it to synthesize organic compounds. This activity is an example of 1) hydrolysis 2) saprophytism 3) cellular respiration
The Structure and Function of Biomolecules
 The Structure and Function of Biomolecules The student is expected to: 9A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic
The Structure and Function of Biomolecules The student is expected to: 9A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic
4.1 Cycling of Matter Date: Cycling of Organic and Inorganic Matter. Build your Own Notes:
 4.1 Cycling of Matter Date: Build your Own Notes: Use these topics as guidelines to create your own notes for 4.1 from pages 83 84 Study Notes/Questions Cycling of Organic and Inorganic Matter Matter is
4.1 Cycling of Matter Date: Build your Own Notes: Use these topics as guidelines to create your own notes for 4.1 from pages 83 84 Study Notes/Questions Cycling of Organic and Inorganic Matter Matter is
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS
 BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
Mid Term Review. 1. step 1, only 3. both step 1 and step 2 2. step 2, only 4. neither step 1 nor step 2
 Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
BIO 2 GO! CARBOHYDRATES. Organic molecules that contain carbon, hydrogen, and oxygen
 BIO 2 GO! 3112 CARBOHYDRATES Organic molecules that contain carbon, hydrogen, and oxygen All living things need carbohydrates to produce energy and to build the organism. Those organisms that cannot make
BIO 2 GO! 3112 CARBOHYDRATES Organic molecules that contain carbon, hydrogen, and oxygen All living things need carbohydrates to produce energy and to build the organism. Those organisms that cannot make
Ms. Golub & Ms. Sahar Date: Unit 2- Test #1
 Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins
 I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
MOLECULES. reflect. You might not want to eat a grasshopper, but you could!
 reflect If you ever travel to another country, you are bound to encounter unfamiliar foods. For example, in Mexico you might be offered a plate of grasshoppers. In Japan, salamanders could be on the menu.
reflect If you ever travel to another country, you are bound to encounter unfamiliar foods. For example, in Mexico you might be offered a plate of grasshoppers. In Japan, salamanders could be on the menu.
2.3: Carbon-Based Molecules Notes
 2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
Cycling of Organic and Inorganic Matter
 Cycling of Matter Build your Own Notes: Use these topics as guidelines to create your own notes from the page given udy Notes/Questions Cycling of Organic and Inorganic Matter Organic matter always contains
Cycling of Matter Build your Own Notes: Use these topics as guidelines to create your own notes from the page given udy Notes/Questions Cycling of Organic and Inorganic Matter Organic matter always contains
Food Chains & Food Webs. **Test on Tuesday, April 29, 2014**
 Name: Mrs. Merenda Date: Reading/Class & Homework Class work: Tuesday, April 22 Food Chains & Food Webs **Test on Tuesday, April 29, 2014** use your glossary to locate definitions #1-3 read booklet pp.
Name: Mrs. Merenda Date: Reading/Class & Homework Class work: Tuesday, April 22 Food Chains & Food Webs **Test on Tuesday, April 29, 2014** use your glossary to locate definitions #1-3 read booklet pp.
Food Chains and Webs
 Food Chains and Webs Let's start with reviewing the parts of a food chain. If you need help, click on the animal. producer consumer decompser If you feel comfortable enough to continue, click here. Plants
Food Chains and Webs Let's start with reviewing the parts of a food chain. If you need help, click on the animal. producer consumer decompser If you feel comfortable enough to continue, click here. Plants
TIPS TO PREPARE FOR THE BIOLOGY 1 st SEMESTER FINAL EXAM:
 TIPS TO PREPARE FOR THE BIOLOGY 1 st SEMESTER FINAL EXAM: FINAL EXAM DETAILS: 80 questions Multiple choice Will assess your mastery of the biological concepts covered in Units 1 and 2 Will assess your
TIPS TO PREPARE FOR THE BIOLOGY 1 st SEMESTER FINAL EXAM: FINAL EXAM DETAILS: 80 questions Multiple choice Will assess your mastery of the biological concepts covered in Units 1 and 2 Will assess your
Guided Inquiry Skills Lab. Additional Lab 1 Making Models of Macromolecules. Problem. Introduction. Skills Focus. Materials.
 Additional Lab 1 Making Models of Macromolecules Guided Inquiry Skills Lab Problem How do monomers join together to form polymers? Introduction A small number of elements make up most of the mass of your
Additional Lab 1 Making Models of Macromolecules Guided Inquiry Skills Lab Problem How do monomers join together to form polymers? Introduction A small number of elements make up most of the mass of your
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry!
 Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
TEST NAME:Cells and Health TEST ID: GRADE:08 - Eighth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY: School Assessment
 TEST NAME:Cells and Health TEST ID:1326431 GRADE:08 - Eighth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY: School Assessment Cells and Health Page 1 of 15 Student: Class: Date: 1. Which best
TEST NAME:Cells and Health TEST ID:1326431 GRADE:08 - Eighth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY: School Assessment Cells and Health Page 1 of 15 Student: Class: Date: 1. Which best
Prerequisite Knowledge: Students should have already been introduced to the inputs and outputs of photosynthesis.
 www.ngsslifescience.com. Topic: Metabolism Chemistry Model Summary: Students will learn act out polymerization by performing dehydration synthesis and hydrolysis using chemistry models. Students will also
www.ngsslifescience.com. Topic: Metabolism Chemistry Model Summary: Students will learn act out polymerization by performing dehydration synthesis and hydrolysis using chemistry models. Students will also
A. There are about 100 elements; 25 of them are necessary for life. B. Carbon atoms can form long chains, leading to a huge number of possible
 Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
Molecules. Background
 Background A molecule is a group of two or more atoms. Compounds are also two or more atoms. Compounds are made from different types of atoms. can be made of different types of atoms or can also be made
Background A molecule is a group of two or more atoms. Compounds are also two or more atoms. Compounds are made from different types of atoms. can be made of different types of atoms or can also be made
Name Date Class. This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels.
 Food and Digestion Name Date Class Food and Energy This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels. Use Target Reading Skills
Food and Digestion Name Date Class Food and Energy This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels. Use Target Reading Skills
Properties of Water. 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin.
 Name: ate: 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin. 1. Which of these enzymes function in the most similar ph range?.
Name: ate: 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin. 1. Which of these enzymes function in the most similar ph range?.
Chapter 2. Living and Non-Living Things Interact in Ecosystems
 Chapter 2 Living and Non-Living Things Interact in Ecosystems 2.1 Types of Interactions in Ecosystems Symbiosis PSymbiosis is the relationship between two different species in an ecosystem. Sometimes this
Chapter 2 Living and Non-Living Things Interact in Ecosystems 2.1 Types of Interactions in Ecosystems Symbiosis PSymbiosis is the relationship between two different species in an ecosystem. Sometimes this
5th Grade Ecosystem Dynamics
 Slide 1 / 89 Slide 2 / 89 5th Grade Ecosystem Dynamics 2015-11-02 www.njctl.org Slide 3 / 89 Table of Contents: Ecosystem Dynamics Click on the topic to go to that section Food Webs Decomposers Cycles
Slide 1 / 89 Slide 2 / 89 5th Grade Ecosystem Dynamics 2015-11-02 www.njctl.org Slide 3 / 89 Table of Contents: Ecosystem Dynamics Click on the topic to go to that section Food Webs Decomposers Cycles
Unit 2 - Characteristics of Living Things
 Living Environment Answer Key to Practice Exam- Parts A and B-1 1. A fully functioning enzyme molecule is arranged in a complex three-dimensional shape. This shape determines the A) specific type of molecule
Living Environment Answer Key to Practice Exam- Parts A and B-1 1. A fully functioning enzyme molecule is arranged in a complex three-dimensional shape. This shape determines the A) specific type of molecule
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Nutrients and Digestion
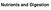 Nutrients and Digestion Nutrition what is needed to be taken in to keep the body healthy Essential Nutrients Carbohydrates Fats Proteins Minerals Vitamins Water Carbohydrates Types of sugars combined in
Nutrients and Digestion Nutrition what is needed to be taken in to keep the body healthy Essential Nutrients Carbohydrates Fats Proteins Minerals Vitamins Water Carbohydrates Types of sugars combined in
Carbon Compounds. Lesson Overview. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview Carbon Compounds Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from
Lesson Overview Carbon Compounds Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from
Bell Work. b. is wrong because combining two glucose molecules requires energy, it does not release energy
 Bell Work How is energy made available to the cell to move large starch molecules across the cell membrane through the process of endocytosis? a. removing a phosphate from ATP b. combining two glucose
Bell Work How is energy made available to the cell to move large starch molecules across the cell membrane through the process of endocytosis? a. removing a phosphate from ATP b. combining two glucose
***Non-living things may show one or more of these Characteristics, but NEVER ALL of them
 -Living things are highly ORGANIZED -Living things are made up of one or more CELLS -Living things use ENERGY -Living things GROW and develop -Living things RESPOND to changes in the environment -Living
-Living things are highly ORGANIZED -Living things are made up of one or more CELLS -Living things use ENERGY -Living things GROW and develop -Living things RESPOND to changes in the environment -Living
Environmental Literacy Project Michigan State University. Lesson 2.3: Materials Plants Are Made Of
 Environmental Literacy Project Michigan State University Lesson 2.3: Materials Plants Are Made Of Benchmark Scale Power of Ten Large Scale: Farm field Decimal Style Large scale Larger 10 5 10 4 10 3 Larger
Environmental Literacy Project Michigan State University Lesson 2.3: Materials Plants Are Made Of Benchmark Scale Power of Ten Large Scale: Farm field Decimal Style Large scale Larger 10 5 10 4 10 3 Larger
Photosynthesis and Respiration. The BIG Idea All cells need energy and materials for life processes.
 Photosynthesis and Respiration Objectives Explain why cells need energy. Summarize how energy is captured and stored. Describe how plants and animals get energy. The BIG Idea All cells need energy and
Photosynthesis and Respiration Objectives Explain why cells need energy. Summarize how energy is captured and stored. Describe how plants and animals get energy. The BIG Idea All cells need energy and
Year 9 AQA GCSE Biology Revision Checklist
 Year 9 AQA GCSE Biology Revision Checklist Use this booklet to help you with your revision in preparation for your year 9 Biology exam and final examinations. This is the work that you will have covered
Year 9 AQA GCSE Biology Revision Checklist Use this booklet to help you with your revision in preparation for your year 9 Biology exam and final examinations. This is the work that you will have covered
Biochemistry Name: Practice Questions
 Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
2 3 Carbon Compounds Slide 1 of 37
 1 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with
1 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with
Name # Class Regents Review: Characteristics of Life and Biochemistry
 Name # Class Regents Review: Characteristics of Life and Biochemistry 6. Some processes that occur in a cell are listed below. A. utilize energy B. detect changes in the environment C. rearrange and synthesize
Name # Class Regents Review: Characteristics of Life and Biochemistry 6. Some processes that occur in a cell are listed below. A. utilize energy B. detect changes in the environment C. rearrange and synthesize
Macromolecules Chapter 2.3
 Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Assembly of ribosomes begins here. Shapes, supports, and protects the cell
 Semester Review Identify the kingdoms that are able to perform cellular respiration. Assembly of ribosomes begins here Shapes, supports, and protects the cell 1 Contrast passive & active transport Describe
Semester Review Identify the kingdoms that are able to perform cellular respiration. Assembly of ribosomes begins here Shapes, supports, and protects the cell 1 Contrast passive & active transport Describe
BIOMOLECULES. Ms. Bosse Fall 2015
 BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells
 Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells Objectives Summarize the characteristics of organic compounds. Compare the structures and function of different types of biomolecules.
Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells Objectives Summarize the characteristics of organic compounds. Compare the structures and function of different types of biomolecules.
PROCTOR VERSION. 2.9 B: Movement of Carbon, Nitrogen, Phosphorus and Water Quiz
 1. A person s blood glucose level is affected by the sugars contained in food. Blood glucose levels are controlled by the hormone insulin via a homeostatic feedback mechanism. A person eats a meal containing
1. A person s blood glucose level is affected by the sugars contained in food. Blood glucose levels are controlled by the hormone insulin via a homeostatic feedback mechanism. A person eats a meal containing
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
Biochemistry Regents Practice
 iochemistry Regents Practice Name: Date: 1. The diagram below represents a sequence of events that occurs in living things. 3. Which statement describes a similarity between all enzymes, antibodies, and
iochemistry Regents Practice Name: Date: 1. The diagram below represents a sequence of events that occurs in living things. 3. Which statement describes a similarity between all enzymes, antibodies, and
Energy and Food Webs
 Energy and Food Webs Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some
Energy and Food Webs Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some
ANIMAL NUTRITION 24 APRIL 2013
 ANIMAL NUTRITION 24 APRIL 2013 Lesson Description In this lesson, we: Look at nutrition in various animals o Herbivores, Carnivores and Omnivores Study the structure of the human digestive system Look
ANIMAL NUTRITION 24 APRIL 2013 Lesson Description In this lesson, we: Look at nutrition in various animals o Herbivores, Carnivores and Omnivores Study the structure of the human digestive system Look
Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients.
 Lesson 1 Nutrition and Your Health What Is Nutrition? Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients. Nutrition Nutrition is the
Lesson 1 Nutrition and Your Health What Is Nutrition? Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients. Nutrition Nutrition is the
Biomolecules. Organic compounds of life
 Biomolecules Organic compounds of life TEKS 9A: Students will Compare the structure and function of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids 9D: Students
Biomolecules Organic compounds of life TEKS 9A: Students will Compare the structure and function of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids 9D: Students
Biology 1. Worksheet I
 Biology 1 Worksheet I Question #1 What does the first law of thermodynamics state? Energy cannot be created or destroyed, only transformed No energy transformation is 100% efficient How is this law related
Biology 1 Worksheet I Question #1 What does the first law of thermodynamics state? Energy cannot be created or destroyed, only transformed No energy transformation is 100% efficient How is this law related
Biology. Slide 1 of 37. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Biomolecules. 1) Carbohydrate Facts. Types of Biomolecules. Carbohydrate Facts. What are Biomolecules? 12/25/13. Two Types of Carbohydrates
 Biomolecules What are Biomolecules? Organic compounds made by living things Also called biochemicals Some are very large There are thousands of different biomolecules, but are separated into 4 categories
Biomolecules What are Biomolecules? Organic compounds made by living things Also called biochemicals Some are very large There are thousands of different biomolecules, but are separated into 4 categories
2 3 Carbon Compounds (Macromolecules)
 2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
Compounds of Life Biological Molecules
 Compounds of Life Biological Molecules By Joseph A. Castellano, Ph.D. RESEED Silicon Valley Reference: Focus on Physical Science, Glencoe/McGraw-Hill, Columbus, Ohio, 2007, Pages 438-442. This textbook
Compounds of Life Biological Molecules By Joseph A. Castellano, Ph.D. RESEED Silicon Valley Reference: Focus on Physical Science, Glencoe/McGraw-Hill, Columbus, Ohio, 2007, Pages 438-442. This textbook
CP Biology Chapter 2: Molecules of Life Name Amatuzzi #1: Carbohydrates pp Period Homework
 Amatuzzi #1: Carbohydrates pp. 46-47 Period 1. Which elements make up carbohydrates? a. In which ratio? 2. How do living things use most of their carbohydrates? 3. How do cells get energy from carbs? a.
Amatuzzi #1: Carbohydrates pp. 46-47 Period 1. Which elements make up carbohydrates? a. In which ratio? 2. How do living things use most of their carbohydrates? 3. How do cells get energy from carbs? a.
Chapter 3- Organic Molecules
 Chapter 3- Organic Molecules CHNOPS Six of the most abundant elements of life (make up 95% of the weight of all living things)! What are they used for? Structures, enzymes, energy, hormones, DNA How do
Chapter 3- Organic Molecules CHNOPS Six of the most abundant elements of life (make up 95% of the weight of all living things)! What are they used for? Structures, enzymes, energy, hormones, DNA How do
The Digestive System
 The Digestive System What is the digestive system? The digestive system is where the digestion of food and liquids occurs. Our bodies are not suited to absorbing the energy contained in food as they are
The Digestive System What is the digestive system? The digestive system is where the digestion of food and liquids occurs. Our bodies are not suited to absorbing the energy contained in food as they are
Macromolecules. The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1.
 Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
B i o c h e m i s t r y N o t e s
 14 P a g e Carbon Hydrogen Nitrogen Oxygen Phosphorus Sulfur ~Major ~Found in all ~Found in most ~Found in all component of all organic organic molecules. molecules. ~Major structural atom in all organic
14 P a g e Carbon Hydrogen Nitrogen Oxygen Phosphorus Sulfur ~Major ~Found in all ~Found in most ~Found in all component of all organic organic molecules. molecules. ~Major structural atom in all organic
HUMAN NUTRITION 08 MAY 2013
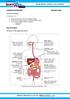 HUMAN NUTRITION 08 MAY 2013 Lesson Description In this lesson, we: Study the structure of the human digestive system Look at the processes used in the human digestive system Look at the two different types
HUMAN NUTRITION 08 MAY 2013 Lesson Description In this lesson, we: Study the structure of the human digestive system Look at the processes used in the human digestive system Look at the two different types
Unit 1: Science of Life 1. Define the following terms: Hypothesis: Testable explanation for a phenomenon
 UCS BIOLOGY STUDY GUIDE FOR 1 ST SEMESTER MIDTERM EXAM 2014-2015 Unit 1: Science of Life 1. Define the following terms: Hypothesis: Testable explanation for a phenomenon Experiment: an orderly procedure
UCS BIOLOGY STUDY GUIDE FOR 1 ST SEMESTER MIDTERM EXAM 2014-2015 Unit 1: Science of Life 1. Define the following terms: Hypothesis: Testable explanation for a phenomenon Experiment: an orderly procedure
GOZO COLLEGE SECONDARY SCHOOL
 Embracing Diversity GOZO COLLEGE SECONDARY SCHOOL Half Yearly Exams 2014-2015 Subject: BIOLOGY TRACK 2 Form: 4 Time: 2 Hours Name: Class: Section A Section B Question No. 1 2 3 4 5 6 1 2 3 4 5 Max mark
Embracing Diversity GOZO COLLEGE SECONDARY SCHOOL Half Yearly Exams 2014-2015 Subject: BIOLOGY TRACK 2 Form: 4 Time: 2 Hours Name: Class: Section A Section B Question No. 1 2 3 4 5 6 1 2 3 4 5 Max mark
Do Now: Sort the following into the order of life from smallest to largest:
 Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Living Environment. Scientific Inquiry Exam
 Name: Class: 1. Which elements are present in all organic compounds? 1) nitrogen and carbon 3) hydrogen and oxygen 2) nitrogen and oxygen 4) hydrogen and carbon 2. Which substances are inorganic compounds?
Name: Class: 1. Which elements are present in all organic compounds? 1) nitrogen and carbon 3) hydrogen and oxygen 2) nitrogen and oxygen 4) hydrogen and carbon 2. Which substances are inorganic compounds?
Reading 3.2 Why do different food molecules provide different amounts of energy?
 Reading 3.2 Why do different food molecules provide different amounts of energy? Getting Started The following four items have carbohydrate, protein, and fat molecules. Honey has simple sugars. Rice has
Reading 3.2 Why do different food molecules provide different amounts of energy? Getting Started The following four items have carbohydrate, protein, and fat molecules. Honey has simple sugars. Rice has
UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg
 UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg. 146-173 Name Date Class Warm-up: List the percentages of each: Total Fats Saturated Fats Carbohydrates Protein What biomolecule would cholesterol be classified
UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg. 146-173 Name Date Class Warm-up: List the percentages of each: Total Fats Saturated Fats Carbohydrates Protein What biomolecule would cholesterol be classified
Digestive System. Science 7 HMS 2016
 Digestive System Science 7 HMS 2016 ??? What is the role of the digestive system? 1. Breaks down food into molecules the body can use 2. Molecules are absorbed into the blood stream to be carried to the
Digestive System Science 7 HMS 2016 ??? What is the role of the digestive system? 1. Breaks down food into molecules the body can use 2. Molecules are absorbed into the blood stream to be carried to the
BIOMOLECULES. (AKA MACROMOLECULES) Name: Block:
 BIOMOLECULES (AKA MACROMOLECULES) Name: Block: BIOMOLECULES POGIL All living things share the same chemical building blocks and depend on chemical processes for survival. Life without carbon (C) would
BIOMOLECULES (AKA MACROMOLECULES) Name: Block: BIOMOLECULES POGIL All living things share the same chemical building blocks and depend on chemical processes for survival. Life without carbon (C) would
Molecules of Life. Carbohydrates Lipids Proteins Nucleic Acids
 Molecules of Life Carbohydrates Lipids Proteins Nucleic Acids Molecules of Life All living things are composed of the following basic elements: Carbon Hydrogen Oxygen Nitrogen Phosphorous Sulfur Remember
Molecules of Life Carbohydrates Lipids Proteins Nucleic Acids Molecules of Life All living things are composed of the following basic elements: Carbon Hydrogen Oxygen Nitrogen Phosphorous Sulfur Remember
C3.1 What is the difference between intensive and organic farming? 1. Recall that many chemicals in living things are natural polymers
 C3 :Food matters C3.1 What is the difference between intensive and organic farming? 1. Recall that many chemicals in living things are natural polymers Potato, Pasta, Bread Carbohydrate Polymer = starch
C3 :Food matters C3.1 What is the difference between intensive and organic farming? 1. Recall that many chemicals in living things are natural polymers Potato, Pasta, Bread Carbohydrate Polymer = starch
Food Chains and Webs --- "What's for dinner?"
 Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
There are four classes of biological macromolecules: Proteins, lipids, carbohydrates and nucleic acids
 There are four classes of biological macromolecules: Proteins, lipids, carbohydrates and nucleic acids Discuss with your shoulder partner: What do these words mean? So What Is A Macromolecule? A very
There are four classes of biological macromolecules: Proteins, lipids, carbohydrates and nucleic acids Discuss with your shoulder partner: What do these words mean? So What Is A Macromolecule? A very
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Science 7 Chapter 2 Section 1
 Science 7 Chapter 2 Section 1 T Y P E S O F I N T E R A C T I O N S Everything is connected One of the key ideas in ecology is that everything is connected to everything else. Each part of the environment
Science 7 Chapter 2 Section 1 T Y P E S O F I N T E R A C T I O N S Everything is connected One of the key ideas in ecology is that everything is connected to everything else. Each part of the environment
Aim 19: Cellular Respiration
 1. During the process of cellular respiration, energy is released from A) carbon dioxide B) oxygen atoms C) water molecules D) chemical bonds 2. The energy used to obtain, transfer, and transport materials
1. During the process of cellular respiration, energy is released from A) carbon dioxide B) oxygen atoms C) water molecules D) chemical bonds 2. The energy used to obtain, transfer, and transport materials
I. ATP: Energy In A Molecule
 I. ATP: Energy In A Molecule All food is broken down by the body into small molecules through digestion By the time food reaches your bloodstream, it has been broken down into nutrient molecules that can
I. ATP: Energy In A Molecule All food is broken down by the body into small molecules through digestion By the time food reaches your bloodstream, it has been broken down into nutrient molecules that can
1) Four main feeding mechanisms of animals a) Suspension feeders i) (1) Humpback whales b) Substrate feeders i)
 1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
