Biology 1. Worksheet I
|
|
|
- Caroline Garrett
- 6 years ago
- Views:
Transcription
1 Biology 1 Worksheet I
2 Question #1 What does the first law of thermodynamics state? Energy cannot be created or destroyed, only transformed No energy transformation is 100% efficient How is this law related to the efficiency of photosynthesis? Photosynthesis is an energy transformation process that converts solar energy into chemical energy (food) Photosynthesis is only 1-2% efficient
3 Question #2 What does the second law of thermodynamics state? Entropy increases Give a biological example of this law in action. An increase in entropy is an increase in randomness or disorder. Decomposition is an example of an increase in entropy.
4 Question #3 Describe the difference between potenial energy and kinetic energy Potential energy is Energy of position Energy that is stored in chemical bonds Kinetic energy is motion Energy of chemical bonds are broken Energy that is released when
5 Question #4 Pyramid of Biomass Trophic Levels 100 Pounds The Sun 1000 Pounds Consumers Heterotrophs 10,000 Pounds Photosynthesis (P/S) Producers Autotrophs
6 Question #5 Autotrophic organisms produce their own food using solar energy. They do not need to feed on other organisms Autotrophs - Producers Diatoms: Marine Phytoplankton Autotrophic organisms are photosynthetic
7 Question #5 Heterotrophs feed on other organisms. They include both herbivores (primary consumers) and carnivores (secondary consumers) Primary Consumers - Heterotrophs
8 Question #5 Secondary Consumers - Heterotrophs The mountain lion is a seconday consumer or carnivore. On a simple three trophic (feeding) level pyramid of biomass, the mountain lion is at the 3 rd trophic level.
9 Question #5 Omnivores are both primary and secondary consumers Omnivores eat both plants and meat Coyotes, bears, bears and raccoons are omnivores
10 Question #6 Based on the information in the pyramid of biomass, would you feed a starving country rice and beans or steak and lamb chops? Why Rice and beans You can feed 10X more people on a vegetarian diet (people feeding as herbivores) 10,000 pounds of rice and beans can support 1,000 pounds of cows and sheep which can support 100 pounds of humans 10,000 pounds of rice an beans can support 1,000 pounds of humans (10X more!)
11 Questions # 20 and #21 Monosaccharides Carbohydrates Blood Sugar = Glucose Fruit Sugar = Fructose Milk Monosaccharide = Galactose Disaccharides Sucrose = Glucose + Fructose Lactose = Glucose + Galactose Maltose = Glucose + Glucose Polysaccharides plants Starch = Storage form of glucose in Glycogen = Storage form of glucose in animals Cellulose = Plant cell walls (the fiber in your diet)
12 Questions #22 and #24 Why is the shape of a protein important? Protein shape determines protein function What does the term denaturization mean? To denature a protein is to alter its shape Proteins can be denatured by altering the changing the ph or the temperature If an enzyme is denatured what would be the consequence? function The enzyme would not longer
13 Question #25 Describe the relationship between enzymes, ph, and temperature. Use the terms protein, denature, shape, and function in your discussion. Enzymes are specialized proteins The shape of a protein determines its function If you alter the ph or temperature of the enzyme you will denature it If you denature the enzyme you will alter its structure This of you denature the enzyme it will not function
14 Question #26 If you were the head of the MSAC cross country team would you feed them a pasta dinner or prime rib the night before the race? I would feed them a pasta dinner. Pasta is a carbohydrate. Carbohydrates are the primary source of energy in the diet. Prime rib is protein (meat). The role of protein it the diet is growth maintenance and repair.
15 Question #30 Amino acids contain an element that is not found in carbohydrates and lipids. What element is this? nitrogen (N) The bond that forms between two amino acids is called a peptide bond A chain of amino acids is called The primary structure of a protein is its specific sequence of amino acids a protein or polypeptide
16 1 Pharynx Common passageway for food and air 2 Esophagus 3 Liver Gall bladder Pancreas Duodenum First (First Section Section) Jejunum (Middle Section) Ileum (Last Section) Small Intestine Appendix 5
Standard B-3: The student will demonstrate an understanding of the flow of energy within and between living systems.
 B-3.1 Summarize the overall process by which photosynthesis converts solar energy into chemical energy and interpret the chemical equation for the process. Taxonomy Level: 2.4-B and 2.1-B Understand Conceptual
B-3.1 Summarize the overall process by which photosynthesis converts solar energy into chemical energy and interpret the chemical equation for the process. Taxonomy Level: 2.4-B and 2.1-B Understand Conceptual
Molecule - two or more atoms held together by covalent bonds. Ex. = water, H O
 ORGANIC CHEMISTRY NOTES Why study carbon? ORGANIC CHEMISTRY NOTES Why study carbon? * All of life is built on carbon * Cells are made up of about 72% water 3% salts (NaCl, and K) 25% carbon compounds which
ORGANIC CHEMISTRY NOTES Why study carbon? ORGANIC CHEMISTRY NOTES Why study carbon? * All of life is built on carbon * Cells are made up of about 72% water 3% salts (NaCl, and K) 25% carbon compounds which
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic
 Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Biochemistry: Macromolecules
 1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
Introduction to Macromolecules. If you were to look at the nutrition label of whole milk, what main items stick out?
 Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins
 I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
Do Now: Sort the following into the order of life from smallest to largest:
 Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Lec 3a- BPK 110 Human Nutr.:Current Iss.
 Lec 3a- BPK 110 Human Nutr.:Current Iss. 1. Overview Carbohydrates (CHO) 2. Types of Carbohydrates 3. Why to Include Carbohydrates in Your Diet? 4. Digestion, Absorption and Transport of Carbohydrates
Lec 3a- BPK 110 Human Nutr.:Current Iss. 1. Overview Carbohydrates (CHO) 2. Types of Carbohydrates 3. Why to Include Carbohydrates in Your Diet? 4. Digestion, Absorption and Transport of Carbohydrates
Nutrients, Enzymes and Digestion Lesson 4: Digestion and Absorption. Digestive Tract and Accessory Organs
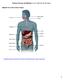 Nutrients, Enzymes and Digestion Lesson 4: Digestion and Absorption Digestive Tract and Accessory Organs http://highered.mheducation.com/sites/0072495855/student_view0/chapter26/animation organs_of_digestion.html
Nutrients, Enzymes and Digestion Lesson 4: Digestion and Absorption Digestive Tract and Accessory Organs http://highered.mheducation.com/sites/0072495855/student_view0/chapter26/animation organs_of_digestion.html
B. Element - each different kind of atom is a different element 1. Examples: C = carbon H = hydrogen
 I. Chemistry study of what substances are made of and how they change and combine Structural Formula A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron
I. Chemistry study of what substances are made of and how they change and combine Structural Formula A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron
2.3 Carbon Compounds 12/19/2011 BIOLOGY MRS. MICHAELSEN. Lesson Overview. Carbon Compounds The Chemistry of Carbon. Lesson Overview.
 2.3 The Chemistry of Carbon A. Carbon atoms have four valence electrons 1. Form strong covalent bonds with many other elements: H, O, P, S, N. 2. Living organisms are made up of carbon and these other
2.3 The Chemistry of Carbon A. Carbon atoms have four valence electrons 1. Form strong covalent bonds with many other elements: H, O, P, S, N. 2. Living organisms are made up of carbon and these other
IB Biology BIOCHEMISTRY. Biological Macromolecules SBI3U7. Topic 3. Thursday, October 4, 2012
 + IB Biology SBI3U7 BIOCHEMISTRY Topic 3 Biological Macromolecules Essential Questions: 1.What are the 4 main types of biological macromolecules and what is their function within cells? 2.How does the
+ IB Biology SBI3U7 BIOCHEMISTRY Topic 3 Biological Macromolecules Essential Questions: 1.What are the 4 main types of biological macromolecules and what is their function within cells? 2.How does the
Carbohydrates. 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds.
 Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
Can you explain that monomers are smaller units from which larger molecules are made?
 Biological molecules Can you explain that all living things have a similar biochemical basis? Can you explain that monomers are smaller units from which larger molecules are made? Can you describe polymers
Biological molecules Can you explain that all living things have a similar biochemical basis? Can you explain that monomers are smaller units from which larger molecules are made? Can you describe polymers
Definition of a Carbohydrate
 * Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
* Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
1.3.1 Function of Food. Why do we need food?
 1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
Carbon Compounds (2.3) (Part 1 - Carbohydrates)
 Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Name: Per. HONORS: Molecules of Life
 Name: Per. HONORS: Molecules of Life Carbohydrates, proteins, and fats are classes of organic molecules that are essential to the life processes of all living things. All three classes of molecules are
Name: Per. HONORS: Molecules of Life Carbohydrates, proteins, and fats are classes of organic molecules that are essential to the life processes of all living things. All three classes of molecules are
Name: Period: Date: Testing for Biological Macromolecules Lab
 Testing for Biological Macromolecules Lab Introduction: All living organisms are composed of various types of organic molecules, such as carbohydrates, starches, proteins, lipids and nucleic acids. These
Testing for Biological Macromolecules Lab Introduction: All living organisms are composed of various types of organic molecules, such as carbohydrates, starches, proteins, lipids and nucleic acids. These
Chapter 1. Chemistry of Life - Advanced TABLE 1.2: title
 Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Dehydration Synthesis and Hydrolysis Reactions. ne_content/animations/reaction_types.ht ml
 Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
Ch 2 Molecules of life
 Ch 2 Molecules of life Think about (Ch 2, p.2) 1. Water is essential to life. If there is water on a planet, it is possible that life may exist on the planet. 2. Water makes up the largest percentage by
Ch 2 Molecules of life Think about (Ch 2, p.2) 1. Water is essential to life. If there is water on a planet, it is possible that life may exist on the planet. 2. Water makes up the largest percentage by
Importance of Nutrition
 The EAT WELL Plate Canada s food guide Food pyramid Importance of Nutrition Energy for body metabolism (nerve impulses, contraction of muscles, repair and replacement of cells Raw materials for building
The EAT WELL Plate Canada s food guide Food pyramid Importance of Nutrition Energy for body metabolism (nerve impulses, contraction of muscles, repair and replacement of cells Raw materials for building
The. Crash Course. Basically, almost all living things are made up of these 4 Elements: - Carbon (C) - Nitrogen (N) - Hydrogen (H) - Oxygen (O)
 The Biochemistry Crash Course Basically, almost all living things are made up of these 4 Elements: - Carbon (C) - Nitrogen (N) - Hydrogen (H) - Oxygen (O) This exercise is designed to familiarize you with
The Biochemistry Crash Course Basically, almost all living things are made up of these 4 Elements: - Carbon (C) - Nitrogen (N) - Hydrogen (H) - Oxygen (O) This exercise is designed to familiarize you with
2.3: Carbon- Based Molecules
 2.3: Carbon- Based Molecules Carbon-based molecules are the foundation of life. Bonding Properties of Carbon Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. 1 3,
2.3: Carbon- Based Molecules Carbon-based molecules are the foundation of life. Bonding Properties of Carbon Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. 1 3,
Neatness 0 1 Accuracy Completeness Lab Class Procedure Total Lab Score
 New Paltz High School Science Department Name:.... Due Date:... Lab Title:.Big Ol Biomolecules..Lab #... Lab Partners: Your Lab Score will be based on the following: Neatness: All labs must be well-written
New Paltz High School Science Department Name:.... Due Date:... Lab Title:.Big Ol Biomolecules..Lab #... Lab Partners: Your Lab Score will be based on the following: Neatness: All labs must be well-written
Molecules of Life. Carbohydrates Lipids Proteins Nucleic Acids
 Molecules of Life Carbohydrates Lipids Proteins Nucleic Acids Molecules of Life All living things are composed of the following basic elements: Carbon Hydrogen Oxygen Nitrogen Phosphorous Sulfur Remember
Molecules of Life Carbohydrates Lipids Proteins Nucleic Acids Molecules of Life All living things are composed of the following basic elements: Carbon Hydrogen Oxygen Nitrogen Phosphorous Sulfur Remember
Metabolism is the sum of body processes inside living cells that sustain life and health.
 Keeping it Simple Nutrition in General: Metabolism is the sum of body processes inside living cells that sustain life and health. The registered dietician role is to carry out major responsibilities in
Keeping it Simple Nutrition in General: Metabolism is the sum of body processes inside living cells that sustain life and health. The registered dietician role is to carry out major responsibilities in
Name a property of. water why is it necessary for life?
 02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds:
 Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic compounds
Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic compounds
Six Nutrients. Nutrients: substances in food that your body needs to stay healthy. Carbohydrates Protein Fat Minerals Vitamins Water
 Nutrients Six Nutrients Nutrients: substances in food that your body needs to stay healthy Carbohydrates Protein Fat Minerals Vitamins Water Water Function: most essential nutrient Helps digest and absorb
Nutrients Six Nutrients Nutrients: substances in food that your body needs to stay healthy Carbohydrates Protein Fat Minerals Vitamins Water Water Function: most essential nutrient Helps digest and absorb
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Carbohydrates: The Energy Nutrient Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license.
 Carbohydrates: The Energy Nutrient 2001 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license. CARBOHYDRATES Functions of Carbohydrates 1. Energy 2.
Carbohydrates: The Energy Nutrient 2001 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license. CARBOHYDRATES Functions of Carbohydrates 1. Energy 2.
Carbohydrates. Organic compounds which comprise of only C, H and O. C x (H 2 O) y
 Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
The Digestive System. 1- Carbohydrates 2- Proteins 3- Lipids 4- Water 5- Vitamins 6- Minerals 7- Fibers
 I. Type of food: The Digestive System 1- Carbohydrates 2- Proteins 3- Lipids 4- Water 5- Vitamins 6- Minerals 7- Fibers 1- Carbohydrates: are energy foods (sugars). They are made of C,H, and O atoms. They
I. Type of food: The Digestive System 1- Carbohydrates 2- Proteins 3- Lipids 4- Water 5- Vitamins 6- Minerals 7- Fibers 1- Carbohydrates: are energy foods (sugars). They are made of C,H, and O atoms. They
Carbohydrates Dr. Ameerah M. Zarzoor
 Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
1) Four main feeding mechanisms of animals a) Suspension feeders i) (1) Humpback whales b) Substrate feeders i)
 1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
UNDERSTANDING THE DIGESTIVE SYSTEM
 UNDERSTANDING THE DIGESTIVE SYSTEM Understanding Digestion The key to solving food and digestive issues starts with an understanding of how the digestive and intestinal system works If you know what is
UNDERSTANDING THE DIGESTIVE SYSTEM Understanding Digestion The key to solving food and digestive issues starts with an understanding of how the digestive and intestinal system works If you know what is
UNDERSTANDING THE DIGESTIVE SYSTEM
 Slide 1 UNDERSTANDING THE DIGESTIVE SYSTEM Slide 2 Understanding Digestion The key to solving food and digestive issues starts with an understanding of how the digestive and intestinal system works If
Slide 1 UNDERSTANDING THE DIGESTIVE SYSTEM Slide 2 Understanding Digestion The key to solving food and digestive issues starts with an understanding of how the digestive and intestinal system works If
Ch18. Metabolism. Chemical processes that maintain life. From the Greek metabole change." version 1.0
 Ch18 Metabolism Chemical processes that maintain life. From the Greek metabole change." version 1.0 Nick DeMello, PhD. 2007-2015 Ch18 Metabolism Metabolism Defined Metabolic Pathways Energy stored as ATP
Ch18 Metabolism Chemical processes that maintain life. From the Greek metabole change." version 1.0 Nick DeMello, PhD. 2007-2015 Ch18 Metabolism Metabolism Defined Metabolic Pathways Energy stored as ATP
Food Chains and Webs --- "What's for dinner?"
 Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
INORGANIC COMPOUNDS. Ex: Water. Compounds that may be essential to life, but are not necessarily found in living things.
 INORGANIC COMPOUNDS Compounds that may be essential to life, but are not necessarily found in living things. Ex: Water Other example: CO2 - ¾ of earth - 90% of living tissue WATER Water is a POLAR compound.
INORGANIC COMPOUNDS Compounds that may be essential to life, but are not necessarily found in living things. Ex: Water Other example: CO2 - ¾ of earth - 90% of living tissue WATER Water is a POLAR compound.
Energy Flow Through an Ecosystem
 Energy Flow Through an Ecosystem Food Chains, Food Webs, Energy Pyramids 1 Ecosystems An ecosystem is all of the organisms living in an area together with their physical environment. Ex. An oak forest,
Energy Flow Through an Ecosystem Food Chains, Food Webs, Energy Pyramids 1 Ecosystems An ecosystem is all of the organisms living in an area together with their physical environment. Ex. An oak forest,
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
Unit 3 Maintaining Dynamic Equilibrium I Topic: Human Digestive System Page 1 of 13. The Chemical Foundation of Digestion
 Page 1 of 13 The Chemical Foundation of Digestion All organisms, regardless of size or complexity, have some method to obtain the essential nutrients they need for survival. Heterotrophs: Organisms that
Page 1 of 13 The Chemical Foundation of Digestion All organisms, regardless of size or complexity, have some method to obtain the essential nutrients they need for survival. Heterotrophs: Organisms that
Chapter 2. What is life? Reproduction. All living things are made of cells
 What is life? Chapter 2 The Nature of Life All living things are made of cells Composed of one or more cells ossess inherited information (DNA) Reproduce Develop respond to the environment Assimilate and
What is life? Chapter 2 The Nature of Life All living things are made of cells Composed of one or more cells ossess inherited information (DNA) Reproduce Develop respond to the environment Assimilate and
1. Which nutrient is so vital to health that you wouldn't live more than a few days without it? A) vitamins B) water C) minerals D) protein
 Nutrition & You, 4e (Blake) Chapter 1 What Is Nutrition? Legend: For sentences / statements that DO NOT HAVE answer choices, Answer A if TRUE Answer B IF False 1. Which nutrient is so vital to health that
Nutrition & You, 4e (Blake) Chapter 1 What Is Nutrition? Legend: For sentences / statements that DO NOT HAVE answer choices, Answer A if TRUE Answer B IF False 1. Which nutrient is so vital to health that
CARBOHYDRATES. By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM
 CARBOHYDRATES By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
CARBOHYDRATES By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
CARBOHYDRATES. Produce energy for living things Atoms? Monomer Examples? Carbon, hydrogen, and oxygen in 1:2:1 ratio.
 CARBOHYDRATES Produce energy for living things Atoms? Carbon, hydrogen, and oxygen in 1:2:1 ratio Monomer Examples? Sugars, starches MONOSACCHARIDES--- main source of energy for cells Glucose Know formula?
CARBOHYDRATES Produce energy for living things Atoms? Carbon, hydrogen, and oxygen in 1:2:1 ratio Monomer Examples? Sugars, starches MONOSACCHARIDES--- main source of energy for cells Glucose Know formula?
2. In terms of appearance, what is the main difference between a monomer, dimer and a polymer?
 Biology Ms. Ye Name Date Block Monomers vs. Polymers 1. The prefix mono- means one. The prefix di- means two. The prefix poly- means many. Based on the given definitions, label the pictures of paperclips
Biology Ms. Ye Name Date Block Monomers vs. Polymers 1. The prefix mono- means one. The prefix di- means two. The prefix poly- means many. Based on the given definitions, label the pictures of paperclips
The Chemical Building Blocks of Life. Chapter 3
 The Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
The Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
Chemistry of Carbon. All living things rely on one particular type of molecule: carbon
 Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Biological Molecules
 The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
Biochemistry. Definition-
 Biochemistry Notes Biochemistry Definition- the scientific study of the chemical composition of living matter AND of the chemical processes that go on in living organisms. Biochemistry Facts 1. The human
Biochemistry Notes Biochemistry Definition- the scientific study of the chemical composition of living matter AND of the chemical processes that go on in living organisms. Biochemistry Facts 1. The human
Chemistry of Carbon. Building Blocks of Life
 Chemistry of Carbon Building Blocks of Life 2007-2008 Why study Carbon? All of life is built on carbon Cells ~72% 2 O ~25% carbon compounds carbohydrates lipids proteins nucleic acids ~3% salts Na, Cl,
Chemistry of Carbon Building Blocks of Life 2007-2008 Why study Carbon? All of life is built on carbon Cells ~72% 2 O ~25% carbon compounds carbohydrates lipids proteins nucleic acids ~3% salts Na, Cl,
The Atoms of Life. What are other elements would you expect to be on this list? Carbon Hydrogen Nitrogen Oxygen Phosphorous Sulfur (sometimes)
 Macromolecules The Atoms of Life The most frequently found atoms in the body are Carbon Hydrogen Nitrogen Oxygen Phosphorous Sulfur (sometimes) What are other elements would you expect to be on this list?
Macromolecules The Atoms of Life The most frequently found atoms in the body are Carbon Hydrogen Nitrogen Oxygen Phosphorous Sulfur (sometimes) What are other elements would you expect to be on this list?
The Carbon Atom (cont.)
 Organic Molecules Organic Chemistry The chemistry of the living world. Organic Molecule a molecule containing carbon and hydrogen Carbon has 4 electrons in its outer shell and can share electrons with
Organic Molecules Organic Chemistry The chemistry of the living world. Organic Molecule a molecule containing carbon and hydrogen Carbon has 4 electrons in its outer shell and can share electrons with
Competitive Inhibitor
 is a substance that reduces the activity of an enzyme by entering the active site in place of the substrate whose structure it mimics. Competitive Inhibitor Identify the following molecule: Polysaccharide
is a substance that reduces the activity of an enzyme by entering the active site in place of the substrate whose structure it mimics. Competitive Inhibitor Identify the following molecule: Polysaccharide
Essential Components of Food
 Essential Components of Food The elements of life living things are mostly (98%) made of 6 elements: C carbon H hydrogen O oxygen P phosphorus N nitrogen S sulphur -each element makes a specific number
Essential Components of Food The elements of life living things are mostly (98%) made of 6 elements: C carbon H hydrogen O oxygen P phosphorus N nitrogen S sulphur -each element makes a specific number
Biological Molecules
 Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
FOOD. Why do we need food? What's in our food? There are 3 trace elements, Iron (Fe), Copper (Cu) and Zinc (Zn).
 Why do we need food? FOOD 1. As a source of energy keeps our cells and us alive. 2. To make chemicals for our metabolic reactions. 3. As raw materials for growth and repair of our cells and body. What's
Why do we need food? FOOD 1. As a source of energy keeps our cells and us alive. 2. To make chemicals for our metabolic reactions. 3. As raw materials for growth and repair of our cells and body. What's
Organic Molecules Worksheet: Read through each section and answer the following questions.
 Name: Date: Period: Organic Molecules Worksheet: Read through each section and answer the following questions. Organic molecules are the molecules that exist in all living things. They are life s building
Name: Date: Period: Organic Molecules Worksheet: Read through each section and answer the following questions. Organic molecules are the molecules that exist in all living things. They are life s building
2-3 Carbon Compounds 10/22/2013. The Chemistry of Carbon. More Carbon. Chemistry (cont) More Macromolecules. Macromolecules
 The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
Macromolecules. Macromolecules. What are the macromolecules? Organic molecules. The human body uses complex organic molecules known as macromolecules.
 Macromolecules Macromolecules Biochemistry The human body uses complex organic molecules known as macromolecules. Macro - long or large It is a large molecule that is made up of smaller units joined together.
Macromolecules Macromolecules Biochemistry The human body uses complex organic molecules known as macromolecules. Macro - long or large It is a large molecule that is made up of smaller units joined together.
Macromolecules Chapter 2.3
 Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
5. Groups A and B in the table below contain molecular formulas of compounds.
 1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
Food Chains and Webs --- "What's for dinner?"
 Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some animals eat other animals.
Digestive System. Part A Multiple Choice. 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin.
 Digestive System Part A Multiple Choice 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin. 2. The presence of large numbers of mitochondria in the cells
Digestive System Part A Multiple Choice 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin. 2. The presence of large numbers of mitochondria in the cells
Not long ago the world was caught up in an anti-carbohydrate craze.
 Carbohydrates Not long ago the world was caught up in an anti-carbohydrate craze. What was or is the problem? Why are carbohydrates so unpopular? Carbohydrates are sugar compounds that plants make when
Carbohydrates Not long ago the world was caught up in an anti-carbohydrate craze. What was or is the problem? Why are carbohydrates so unpopular? Carbohydrates are sugar compounds that plants make when
Macronutrients : Carbohydrates. Structure, sources and function
 Macronutrients : Carbohydrates Structure, sources and function As part of this course, students learn about the structure, sources and function of carbohydrates. They learn to identify carbohydrates in
Macronutrients : Carbohydrates Structure, sources and function As part of this course, students learn about the structure, sources and function of carbohydrates. They learn to identify carbohydrates in
Lecture Outline Chapter 4- Part 2: The Carbohydrates
 Lecture Outline Chapter 4- Part 2: The Carbohydrates I Types of Carbohydrates If someone told you "My carbohydrate intake is too high", what would you assume about what they're eating? A. SIMPLE CARBOHYDRATES:
Lecture Outline Chapter 4- Part 2: The Carbohydrates I Types of Carbohydrates If someone told you "My carbohydrate intake is too high", what would you assume about what they're eating? A. SIMPLE CARBOHYDRATES:
Chapter 15 Food and Digestion
 Chapter 15 Food and Digestion 15.1A Food and Energy Functions of Nutrients 1. 2. 3. 4. Calories = amt. of energy in food RDA depends on age, gender, size and activity level Types of Nutrients (includes
Chapter 15 Food and Digestion 15.1A Food and Energy Functions of Nutrients 1. 2. 3. 4. Calories = amt. of energy in food RDA depends on age, gender, size and activity level Types of Nutrients (includes
Biology: Life on Earth Chapter 3 Molecules of life
 Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
2.3: Carbon-Based Molecules Notes
 2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
Energy and Food Webs
 Energy and Food Webs Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some
Energy and Food Webs Food Chains and Webs --- "What's for dinner?" Every organism needs to obtain energy in order to live. For example, plants get energy from the sun, some animals eat plants, and some
Lesson 2. Biological Molecules. Introduction to Life Processes - SCI 102 1
 Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together.
 Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
Carbohydrates, Lipids, Proteins, and Nucleic Acids
 Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Tiny structures that carry out cellular functions (cell parts) Ex: nucleus, mitochondria, ribosomes
 ALL living things are Building from smallest to LARGEST: Organelles- Cells- Tiny structures that carry out cellular functions (cell parts) Ex: nucleus, mitochondria, ribosomes The basic unit of structure
ALL living things are Building from smallest to LARGEST: Organelles- Cells- Tiny structures that carry out cellular functions (cell parts) Ex: nucleus, mitochondria, ribosomes The basic unit of structure
Biology 12 - Biochemistry Practice Exam
 Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Organic Compounds. (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids
 Organic Compounds (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids Carbon s Bonding Behavior Outer shell of carbon has 4 electrons; can hold 8 Each carbon atom can form covalent bonds with
Organic Compounds (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids Carbon s Bonding Behavior Outer shell of carbon has 4 electrons; can hold 8 Each carbon atom can form covalent bonds with
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS
 BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
BIOCHEMISTRY NOTES Pre AP
 BIOCHEMISTRY NOTES Pre AP I. Chemistry study of what are made of and how they (text pages 35 43) A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron B.
BIOCHEMISTRY NOTES Pre AP I. Chemistry study of what are made of and how they (text pages 35 43) A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron B.
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Biomolecules are organic molecules produced by living organisms which consists mainly of the following elements:
 Biomolecules are organic molecules produced by living organisms which consists mainly of the following elements: These elements are non-metals which combine in various ways to form biomolecules through
Biomolecules are organic molecules produced by living organisms which consists mainly of the following elements: These elements are non-metals which combine in various ways to form biomolecules through
Topic 3: The chemistry of life (15 hours)
 Topic : The chemistry of life (5 hours). Chemical elements and water.. State that the most frequently occurring chemical elements in living things are carbon, hydrogen, oxygen and nitrogen...2 State that
Topic : The chemistry of life (5 hours). Chemical elements and water.. State that the most frequently occurring chemical elements in living things are carbon, hydrogen, oxygen and nitrogen...2 State that
5.2 Lipids 5.21 Triglycerides 5.22 Phospholipids 5.23 Wax 5.24 Steroids. 5.3 Proteins 5.4 Nucleic Acids
 BIOCHEMISTRY Class Notes Summary Table of Contents 1.0 Inorganic and Organic Compounds 2.0 Monomers and Polymers 3.0 Dehydration (Condensation) Synthesis 4.0 Hydrolysis Reaction 5.0 Organic Compounds 5.1
BIOCHEMISTRY Class Notes Summary Table of Contents 1.0 Inorganic and Organic Compounds 2.0 Monomers and Polymers 3.0 Dehydration (Condensation) Synthesis 4.0 Hydrolysis Reaction 5.0 Organic Compounds 5.1
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
Biological Chemistry. Is biochemistry fun? - Find it out!
 Biological Chemistry Is biochemistry fun? - Find it out! 1. Key concepts Outline 2. Condensation and Hydrolysis Reactions 3. Carbohydrates 4. Lipids 5. Proteins 6. Nucleic Acids Key Concepts: 1. Organic
Biological Chemistry Is biochemistry fun? - Find it out! 1. Key concepts Outline 2. Condensation and Hydrolysis Reactions 3. Carbohydrates 4. Lipids 5. Proteins 6. Nucleic Acids Key Concepts: 1. Organic
OBJECTIVE. that carbohydrates, fats, and proteins play in your body.
 OBJECTIVE Describe the roles that carbohydrates, fats, and proteins play in your body. JOurnall How was your weekend? What did you do? Did you practice any of the lessons you have learned thus far? What
OBJECTIVE Describe the roles that carbohydrates, fats, and proteins play in your body. JOurnall How was your weekend? What did you do? Did you practice any of the lessons you have learned thus far? What
Biological Molecules 1
 Biological Molecules 1 Overview Macromolecules Monomers and polymers The four classes of biological molecules Lipids Saturated, unsaturated, trans fats Phospholipids Steroids Carbohydrates Monosaccharides,
Biological Molecules 1 Overview Macromolecules Monomers and polymers The four classes of biological molecules Lipids Saturated, unsaturated, trans fats Phospholipids Steroids Carbohydrates Monosaccharides,
Food Chains and Webs GLOSSARY. autotrophs: organisms, such as plants, that are capable of making their own food.
 Food Chains and Webs GLOSSARY autotrophs: organisms, such as plants, that are capable of making their own food. biological magnification: the process by which small amounts of a poisonous chemical become
Food Chains and Webs GLOSSARY autotrophs: organisms, such as plants, that are capable of making their own food. biological magnification: the process by which small amounts of a poisonous chemical become
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry!
 Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
BIOMOLECULES. Ms. Bosse Fall 2015
 BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
Carbohydrates are a large group of organic compounds occurring in and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1).
 Carbohydrates are a large group of organic compounds occurring in and and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1). Why we study carbohydrates 1) carbohydrates are the
Carbohydrates are a large group of organic compounds occurring in and and including,, and. They contain hydrogen and oxygen in the same ratio as (2:1). Why we study carbohydrates 1) carbohydrates are the
Organic Chemistry. Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl.
 Organic Chemistry Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl. Organic Compounds - have carbon bonded to other atoms and determine structure/function
Organic Chemistry Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl. Organic Compounds - have carbon bonded to other atoms and determine structure/function
Carbon Compounds. Lesson Overview. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview Carbon Compounds Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from
Lesson Overview Carbon Compounds Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from
