Influence of aldosterone on sodium, water and potassium metabolism in chronic renal disease
|
|
|
- Edgar Baldwin
- 5 years ago
- Views:
Transcription
1 Kidney International, Vol. 1 (1972), p Influence of aldosterone on sodium, water and potassium metabolism in chronic renal disease ROBERT W. SCHRIER and EDWARD M. REGAL Department of Medicine and Cardiovascular Research Institute, University of Cal(fornia San Francisco, San Francisco, Caiffornia, and the Department of Metabolism, Walter Reed Army Institute of Research, Washington, D. C. Influence of aldosterone on sodium, water and potassium metabolism in chronic renal disease. To evaluate the physiological importance of aldosterone in chronic renal disease, ii studies were performed in nine patients with renal impairment of varying etiology and severity during periods of normal and low sodium intake. Decreased sodium excretion during sodium restriction was accompanied by increased aldosterone excretion and this anti-natriuretic effect could be reversed by the administration of spironolactone. Constancy of urinary potassium excretion occurred during the period of sodium restriction and may have been related to a combination of diminished distal sodium delivery and increased aldosterone activity. Sodium restriction was also associated with a diminution in renal concentrating capacity and an improvement in renal diluting capacity. Only the improvement in renal diluting capacity, as judged by the minimal urinary osmolality achieved during an acute water load, could be reversed by the administration of spironolactone. These results thus suggest that aldosterone is of physiological importance to the control of urinary sodium, potassium and water excretion in patients with chronic renal disease. Influence de l'aldosterone sur le metabolisme du sodium, de l'eau et du potassium dans l'insuffisance renale chronique. Pour juger de l'importance physiologique de l'aldostérone dans I'msuffisance rénale chronique, onze experiences ont été chez pendant des périodes de régime normal ou pauvre en sodium, sur neuf malades atteints d'insuffisance d'étiologies et de gravité variable. Pendant les périodes de restriction sodée, Ia diminution de l'excrétion du sodium était associée avec une excretion accrue d'aldostérone. Cet effet antinatriuretique pouvait Ctre annulé par l'administration de spironolactone. En periode de restriction sodée, l'excrétion du potassium demeura constante, peut-être sous l'influence combinée d'un apport sodé moindre au niveau du tube distal et d'une activité plus grande de l'aldostérone. La restriction sodée s'accompagnait egalement d'une capacité moindre du rein a concentrer l'urine et d'une meilleure capacité de dilution. Seule l'amélioration de Ia capacité rénale de dilution, jugée par l'osmolalité urinaire minimale atteinte aprés surcharge hydrique, pouvait ëtre annulée par l'administration de spirono- Received for publication September 7, 1971; accepted in revised form November 8, , by the International Society of Nephrology. lactone. Ces résultats suggérent donc que l'aldostérone joue un role physiologique important dans Ic controle de l'élimination urinaire du sodium, du potassium et de l'eau chez les malades atteints d'insuffisance renale chronique. The control of sodium excretion in the presence of normal renal function has been clearly demonstrated to be dependent on factors other than glomerular filtration rate and aldosterone activity [1 4]. Recent studies have demonstrated that a "third factor" may be involved in the regulation of sodium excretion in experimental uremia [51 and in patients with chronic renal failure [6, 7]. Moreover, the physiological role of such a "third factor" may be more important in chronic renal disease since, in contrast to normal subjects, it is possible that aldosterone may not be involved in the adjustments in sodium excretion in patients with chronic renal disease. This possibility is not unlikely since patients with advanced chronic renal disease who are receiving a normal sodium intake have been reported to have increased aldosterone secretory rates [8, 9] and thus may have persistently high endogenous levels of aldosterone. In the presence of already increased aldosterone activity, sodium restriction may not be associated with any significant alternation in either the level of aldosterone activity or the renal response to aldosterone. Some evidence for such a circumstance has been provided by the studies of Slatopoisky et al [6] in which the renal response to alterations in sodium intake in patients with severe chronic renal disease was found to be unaltered by the prior administration of supraphysiological levels of exogenous mineralocorticoid [6]. 156
2 Influence of aldosterone in chronic renal disease 157 Although these results clearly implicate factors other than aldosterone in the regulation of sodium excretion by patients with chronic renal disease [6], they do not exclude an additional contributory role of aldosterone. The present investigation was therefore undertaken to examine whether aldosterone is involved in the renal response to alterations in dietary sodium intake in patients with chronic renal failure. The results suggest that alterations in aldosterone activity do indeed contribute to the renal response to sodium restriction in patients with chronic renal disease, not only with respect to sodium excretion, but also with regards to the renal excretion of potassium and water. Methods Eleven studies were performed in nine patients with chronic renal disease. None of the patients had clinical evidence of uremia, edema, congestive heart failure, urinary tract obstruction or urinary tract infection. Each study consisted of a period on a normal sodium intake (2 meq/kg/day) followed by a period on a low sodium intake (1 to 21 meq/day). Each patient received the same basic diet and fluid intake (3Oml/kg) during periods of normal and low sodium intake. The patients ingested all of the food and fluid offered them throughout the study. A solution of sodium chloride was added to the basic diet during periods of normal sodium intake and was omitted during periods of low sodium intake. The dietary protein intake averaged.75 g/kg/day and the caloric intake ranged from 3 to 45 calories/kg/day, depending on the amount necessary to maintain a constant body weight in each individual patient during the period of normal sodium intake. The periods of normal sodium intake ranged from 7 to 15 days and the periods of low sodium intake ranged from 8 to 25 days. Sodium restriction was instituted when a constant body weight had been noted for at least three consecutive days, and terminated when the urinary sodium concentration had diminished to a low level that was constant for at least three days or when the serum sodium concentration approached 12 meq/liter. Adverse clinical symptomatology was not observed during any of the studies. Aldosterone excretion rates. After at least seven days on a normal sodium intake, three consecutive 24 hour urine collections were obtained for determination of aldosterone excretion rates. Three additional 24 hour urine collections were obtained consecutively at the end of each period of sodium restriction for the same determination. The acid-released conjugate of aldosterone was measured using the double isotope method of Kliman and Petersen [1]. The determinations were done by the Biomedical Assay Laboratory, Worchester, Mass., without knowledge of the sodium intake of the patients. Glomerular filtration rate and volume studies. Endogenous creatinine clearance (Cer) was used as an index of glomerular filtration rate (GFR) in those patients whose Ccr was greater than 1 mi/mm. The mean of the and urea clearance was used as an index of GFR when the Cc, was less than loml/min. Radioactive '311-tagged albumin, 35S-sulfate and tntiated water were used to measure plasma volume, extracellular fluid (ECF) volume and total body water, respectively, during periods of normal and low sodium intake. Sodium and potassium balances. Sodium and potassium balances were undertaken during ten of eleven studies with analyses of both urine and stool. Twelve hour urine volumes were collected from 7:3 AM to 7:3 PM and from 7:3 PM to 7:3 AM. Stool collections were divided into three day periods. A nitricperchloric acid homogenate of stools was analyzed for sodium and potassium content. Blood specimens were obtained every other day. The blood and urine specimens were analyzed for sodium, chloride, potassium, urea, creatinine and osmolality. Sodium, potassium, urea, and creatinine were measured on the Technicon Autoanalyzer, and chloride was measured using an automatic Cotlove titrator. Urine and plasma osmolalities were measured cryoscopically on an Advanced osmometer. Osmolar clearance (Csm) was calculated by the formula: C = urine osmolality (Uosm)/plasma osmolality (Psm) x urine flow (V). Free water clearance (CH2) and free water reabsorption (TcH2O) were calculated respectively as CH2= V C and TCH2O = Cøsm V. Clearances of creatinine and urea were calculated by standard methods. Dilution and concentration tests. Standard dilution and concentration tests were performed at the end of both the normal and low sodium intake periods in the following manner: on the morning of the dilution tests, the patients emptied their bladders and then ingested an oral water load (2 ml/kg) over the next 3 minutes. Four one-hour urine collections were then obtained sequentially and blood specimens were drawn at the midpoint of each urine collection period. Hourly urine volumes were replaced with the oral administration of an equivalent volume of water. Following completion of the dilution test, fluid was restricted for the remainder of the day so that the total
3 158 Schrier/ Regal 24 hour intake did not exceed 3 mi/kg. At 9: PM on the same evening, fluid was completely withdrawn and five units of pitressin tannate in oil, shaken to a fine emulsion, were injected intramuscularly. At 7:3 AM the patients emptied their bladders, three or four one-hour urine collections were obtained consecutively, and blood specimens were drawn at the midpoint of each urine collection period. The patients were then placed on a low sodium intake until the minimal urinary sodium concentration was achieved; the urinary dilution and concentration tests were then repeated at that time. In two studies the period of normal sodium intake was continued after completion of the concentration tests, and the patients were treated with 9-c-fluorohydrocortisone (1.mg) orally every six hours for five days. In a third study desoxycorticosterone acetate (1 mg) was administered intramuscularly for five days. On the fourth and fifth day of mineralocorticoid administration, the dilution and concentration tests were repeated in the same manner as described above. Dietary sodium restriction was then instituted and the concentration and dilution tests were repeated when the minimal urinary sodium concentration was achieved. Spironolactone (75 mg) was then administered orally every six hours for eight days and the dilution and concentration tests were repeated on the seventh and eighth day of its administration. In these same three patients, formation of TCH2O was examined after the performance of the concentration tests on both normal and low sodium intakes. These studies were performed during the infusion of l% mannitol in sodium chloride (45 meq/liter) at 1 mi/mm for 11/2 to 2 hours. Aqueous vasopressin was also administered (5 mu/kg/hr). prolonged sodium deprivation to 28.9 SEM 8.4 tg/ 24 hr. Although some values were within the range of normal after prolonged sodium deprivation, all of these values were increased when compared with the observed excretion rate in the same patient during the period of normal sodium intake. Arterial blood pressure was not significantly different during periods of normal and low sodium intake in any patient. Effect of prolonged sodium restriction on glomerular filtration rate, blood urea nitrogen, body weight, total body water, extracellular fluid and plasma volume (Tables 1 and2). In ten of the eleven studies in nine patients, prolonged dietary sodium restriction was associated with a diminution in GFR. The mean GFR 5 ac 4 U a C 3 Normal Sodium intake Low Results Effect of prolonged sodium restriction on urinary aldosterone excretion in chronic renal disease (Fig. 1). Three consecutive measurements of daily aldosterone excretion were obtained in nine patients after receiving a normal sodium intake for at least one week; similar measurements were then obtained in the same patient on a low sodium intake after a minimal urinary sodium concentration had been achieved (range: 8 to 25 days). The mean values for the three urinary aldosterone excretion rates in each patient on a normal and low sodium diet are shown in Fig. I. The mean value observed on a normal sodium diet was 5.9± SEM I 1g/24 h, and it increased after the period of Fig. 1. Effect of prolonged sodium restriction on a/dos/crone excretion rate. Aldosterone excretion rates were not increased in any of the studies during the normal sodium intake but increased in each patient during sodium restriction.
4 Influence of aldosterone in chronic renal disease 159 Table 1. Summary of patients with chronic renal disease on normal and low sodium intake GFRb mi/mm BUN mg/joo ml Patient Age/Sex Diagnosis a normal low normal low sodium sodium sodium sodium W. M. 37/M Chronic glomerulonephritis J. C. (1) 3/M Polycystic disease J. C. (2) C L. H. 65/M Unilateral kidney N. D. 35/F Chronic pyelonephritis T. M. 2l/M Chronic glomerulonephritis W. B. 31/M Lupus nephritis J. J. 3/M Interstitial nephritis J. P. (1) 24/F Chronic glomerulonephritis J. P. (2) P. N. 2/M Interstitial nephritis Mean P <.5 <.5 Diagnosis confirmed by histological examination of tissue obtained either by renal biopsy or at surgery. h Endogenous creatinine clearance used as an estimate of glomerular filtration rate unless clearance less than 1 mi/mm when the urea clearance -f- (real/rune clearance/2 was used. There was no apparent explanation for the improved renal function during the second study. Table 2. Effect of sodium restriction on body weight, total body water, extracellular fluid volume and plasma volume Weight Total body water ECF volume Plasma volume kg liters liters ml Patient normal low normal low normal low normal low sodium sodium sodium sodium sodium sodium sodium sodium W. M J.C L. H N. D T. M W. B J.J Mean SEM P <.2 <.1 <.1 <.1
5 16 Schrier/Regal in these patients decreased significantly' from 28 to 22 mi/mm. The increase in mean blood urea nitrogen from 48 to 56 mg/loo ml during the period of sodium restriction was not significant. Sodium restriction was associated with a significant reduction in mean body weight from 66.7 to 65.3 kg. Volume measurements in seven of the nine patients revealed a significant, though modest, diminution in total body water in all, and a reduction of extracellular fluid and plasma volume in six of the seven patients. None of the patients developed symptoms of hypovolemia during periods of sodium restriction and a constant fluid intake. Effect of prolonged sodium restriction on sodium and potassium metabolism (Table 3, Fig. 2). The mean daily sodium intake prior to sodium restriction was 138 meq and the mean duration of these control periods was eleven days (range: 7 to 15 days). The mean daily sodium intake during the period of sodium restriction was 11 meq and the mean duration of sodium restriction was 18 days (range: 8 to 25 days). In eight of the nine patients, sodium restriction was discontinued when the minimal urinary sodium concentration was achieved. In these eight patients, Paired student "t" test: a P value <.5 was considered to be statistically significant. although the achievement of a minimal urinary sodium concentration was delayed from 8 to 25 days, sodium balance was eventually achieved in each patient. In the ninth patient (J. P.), the urinary sodium concentration remained high and severe hyponatremia developed rapidly. For this reason, dietary sodium restriction was discontinued after nine days in this patient (Fig. 2). Fecal losses of sodium were generally insignificant during periods of both normal and low sodium intake. The mean daily negative sodium balance for the nine patients was 21 meq, and an average negative cumulative sodium balance of 378 meq was noted during the period of sodium restriction (mean duration: 18 days). Sodium restriction was associated with a slight but significant diminution in mean plasma sodium concentration from 141 to 136 meq/liter, including the marked hyponatremia in J.P. (138 to 123 meq/liter). Diurnal variations in urinary sodium excretion were not observed in any of the nine patients and the 24 hour urine volume was similar during periods of normal and low salt intake. Spironolactone was administered to three patients as the low sodium intake was continued (Table 4). Aldosterone inhibition by spironolactone was associated with an increase in urinary sodium concentration and a decrease in urinary potassium Table 3. Effect of sodium intake Patient Na intake Duration V Minimal UNaVa Fecal Na UNUV losses meq/day Days mi/day on low meq/day meq/day sodium Normal low normal low normal low intake normal low normal low Na Na Na Na Na Na meq/day Na Na Na Na W. M L.H N.D T.M J.P J.C P. N J.J W. B Mean ±sam ±13 ±1 ±1 ±2 ±21 ±245 ±3 ±13 ±4 ±16 ±.3 P <.1 <.1 <.1 a Mean values for entire duration of normal and low sodium periods. b Fecal sodium and potassium losses were not measured and are therefore not accounted for in the balance figures.
6 Influence of aldosterone in chronic renal disease 161 concentration in all three patients. Free-water clearance was decreased in two of the three patients. In the same three patients mineralcorticoid administration (9-c-fluorohydrocortisone in J. C. and P. N.; desoxycorticosterone acetate in J.J.) during a normal sodium intake was associated with a decrease in urinary sodium concentration and an increase in urinary potassium concentration and free-water clearance. During the period of sodium restriction a modest decrease in GFR occurred in all three patients during the administration of spironolactone. Mineralocorticoid administration during a normal sodium intake Fig. 2. Effects of prolonged sodium restriction on plasma sodium concentration, urine flow, Cosm and CH2O. The patient on the left (TM.) demonstrated the ability during sodium restriction to diminish C5m and increase CH2O as V and 'DNa remained unchanged. The patient on the right (J.P.) with "salt-losing nephritis" was unable to diminish Csm and increase CH2O during sodium restriction and thus rapidly developed hyponatremia. 14 Plasma sodium concentration 13 (meq/liter) 12 Urine flow 1 (mi/mm) Csm (mi/mm) Normal salt Low salt intake TM-Chronic glomerulonephritis 14./\ C \ Low salt intake \ J.P-Chronic gloiheonephritis CH2O (mi/mm) U Days -J-l ' 15 2 Days on sodium and potassium balances Mean daily Na K intake UKV Fecal Mean daily K Na balance K losses K balance,neq/day meq/liter meq/day meq/day meq/day meq/day meq/liter normal low normal low normal low normal low normal low normal low Na Na Na Na Na Na Na Na Na Na Na Na Oh b Ob l b <.1 <.5 <.1 <.1 c <.1 <.1 c "s" test performed only on studies with both normal and low sodium values.
7 162 Schrier/Regal Table 4. Effects of spironolactone and mineralocorticoid administration Patient V UNa UK CR2 1Na mi/mm meq/liter meq/liter mi/mm meq/iiter nieq/iiter Conta Spironb Cont Spiron Cont Spiron Cont Spiron Cont Spiron Cont Spiron Spironolactone administration J. C P.N J. J Conte Mineral' Cont Mineral Cont Mineral Cont Mineral Cont Mineral Cont Mineral Mineralocorticoid administration J.C P. N J. J a Cont = Mean values of last 3 days of low sodium intake prior to the diluting and concentrating tests. b Spiron = Mean values after 3 to 5 days of spironolactone treatment and continued low sodium intake. c Cont = Mean value of last 3 days of normal sodium intake prior to the diluting and concentrating tests. ci Mineral = Mean values of first 3 days of mineralocorticoid treatment on normal sodium intake. Table 5. The effect of sodium intake on solute-free water reabsorption (TcH2O) during a mannitol diuresis Patient. Time miii V mi/mm U/Posm Cosm mi/mm TCH2O mi/mm normal low normal low normal low normal low J. C Polycystic disease P. N Interstitial nephritis J. J Interstitial nephritis Mean ± ±.1 SEM ± <.1 <.5 <.1 < , <.1 <.1 <.1 <.1
8 Influence of aldosterone in chronic renal disease 163 was associated with a modest increase in GFR in all three patients. Potassium balance was not altered consistently and the plasma potassium concentration did not differ during the periods of normal and low sodium intake. Fecal potassium loss was not affected by the sodium intake but in some cases rather significant losses were present on both normal and low sodium intakes. In one patient (J. P.) with marked renal impairment (GFR: 3.5 to 5.2ml/min) and severe hyponatremia during sodium restriction, approximately 5 % of the daily potassium intake was excreted in the feces. Effect of prolonged sodium restriction on renal concentrating and diluting capacity (Figs. 3 to 5, Table 5). Impairment of renal concentrating ability was demonstrated in every patient during the period of normal sodium intake (Fig. 3). In five patients (J.P., J.J., N.D., W.M., and P.N.) the maximal urinary osmolality remained below that of plasma Normal Sodium intake after fluid deprivation and vasopressin administration, i.e., so-called vasopressin-resistant hyposthenuria [11]. Prolonged sodium deprivation and decreased solute excretion was not associated with improved concentrating ability in any of the nine patients. In fact, in nine of the eleven studies the maximal urinary osmolality was lower during the period of low sodium intake. The maximal urinary osmolality averaged 35± 39 mosm/kg on a normal sodium diet, and 281 mosm/kg on a low sodium diet (P<.5). The largest decrease in urinary osmolality was observed in the patient with a unilateral kidney and the least impairment of glomerular filtration rate (L. H.). Maximal TH2O formation was examined during mannitol diuresis in three patients on both a normal and low sodium intake (Table 5). On a low sodium intake, hypotonic urine was excreted by all three patients, i.e., TCH2O formation became negative. TCH2O formation became negative in only one patient (J.J.) during mannitol diuresis on a normal sodium intake. The addition of mineralocorticoid to the normal sodium intake did not affect either maximal urinary osmolality or TCH2O formation. The effect of sodium intake on diluting ability is shown in Fig. 4. The minimal urinary osmolality on '.1 6 Normal Sodium intake Low 3 Fig. 3. Effect of prolonged sodium restriction on maximal urinary osmolality. Maximal urinary osmolality diminished in 9 of II patients during sodium restriction. Fig. 4. Effect of prolonged sodium restriction to improve diluting capacity. The minimal urinary osmolality achieved during an acute water load was lower in each study during low sodium as compared to normal sodium intake.
9 164 Schrier/Regal a normal sodium intake was high in all nine patieuts (range: 8 to 23 mosm/kg), but it was significantly lower during sodium restriction. The minimal osmolality averaged 18 9 mosm/kg on the normal sodium intake and 124± 1 mosm/kg on a low sodium intake (P<.l). In three patients the administration of mineralocorticoid during a normal sodium intake was associated with a diminution in minimal urinary osmolality, and the administration of spironolactone during low sodium intake was associated with an increase in minimal urinary osmolality (Fig. 5). r'1 3 Normal sodium + mineralocorticoid I Low sodium Low sodium + spironolactone Ce o 2 I- Ce 1 Ce Jc P. N. J.J o Fig. 5. Role of nzineralocorticoid in i/nprove/nent of diluting capacity during sodium restriction. The administration of 9-a-fluorohydrocortisone or desoxycorticosterone decreased minimal urinary osmolality during a normal sodium intake, while the administration of spironolactone increased minimal urinary osmolality during a low sodium intake. Discussion The present results suggest that alterations in aldosterone activity may be of physiological importance in the renal response to sodium restriction in patients with chronic renal disease. The observed increase in urinary aldosterone excretion during sodium restriction in these patients is compatible with such a thesis. Nevertheless, this observation alone was insufficient to establish a cause and effect relationship between increased aldosterone excretion and decreased urinary sodium excretion. For this reason, studies were undertaken in three patients to assess the effect of a spironolactone-mediated inhibition of the action of aldosterone. In each instance urinary sodium excretion increased substantially during the administration of spironolactone despite continued dietary sodium restriction (Table 4). Since spironolactone is thought to exert its effect on renal tubular sodium reabsorption via direct inhibition of the action of aldosterone [12], the present results would seem to provide strong evidence for an aldosterone-induced enhancement of sodium reabsorption during sodium restriction in chronic renal disease. In addition, the exogenous administration of mineralocorticoids was found to mimic the urinary effects of sodium restriction even in the presence of a normal sodium intake (Table 4). Taken together, therefore, the present results seem to suggest strongly that aldosterone is of physiological importance in the control of sodium metabolism in chronic renal disease. However, one must acknowledge the possibility that alterations in aldosterone activity may be important in the regulation of sodium excretion in patients with moderate renal impairment, but that it exerts very little influence in patients with severe end-stage renal impairment. In this regard, many of the studies of Slatopolsky et al [6] which indicated that a "third factor" might be involved in the regulation of sodium excretion in chronic renal failure were performed in patients with severe renal impairment. In the present study the glomerular filtration rates were 54, 24 and 5 mi/mm in the three patients who received spironolactone during dietary sodium restriction. In all three patients a large increase in urinary sodium excretion was observed during the administration of spironolactone and, in fact, the largest natriuresis was observed in the patient (J.J.) with the lowest filtration rate. These present results are therefore compatible with the thesis that aldosterone is an important regulator of urinary sodium excretion at all levels of glomerular filtration rate in patients with chronic renal disease. However, before this tentative conclusion can be accepted
10 Influence of aldosterone in chronic renal disease 165 finally, additional studies of patients with severe renal impairment will be required. It should also be emphasized that the demonstration of a physiological role of aldosterone in the regulation of sodium excretion in chronic renal disease in no way precludes an additional role of other factors, such as changes in glomerular filtration rate and the activity of "third factor", instead an interaction between all of these factors seems most likely. The possibility that aldosterone might not exert a significant influence on sodium excretion in chronic renal disease was based, at least partially, on the finding that the majority of patients with severe chronic renal disease may exhibit an increased aldosterone secretory rate [8, 9]. These results, [8, 9] however, may be related to methodological problems involved in measuring aldosterone secretory rates in patients with chronic renal disease rather than being indicative of increased aldosterone production. Cope and Pearson [8] have reported that, in contrast to normal subjects, the excretion of the metabolite that is used to measure aldosterone secretion may be delayed considerably longer than 24 hours in patients with chronic renal disease. Since measurements of aldosterone secretion are based on the excretion of the majority of the measured metabolite in 24 hours after the injection of isotope-labeled aldosterone, measurements in chronic renal disease are difficult to interpret even when corrections for the delayed excretion of the metabolite are attempted [8]. Hayslett et al [13] have used an in vitro assay method of adrenal glands in azotemic rats to assess aldosterone production, thereby avoiding any methodological error related to impaired renal excretion. These investigators [13] failed to find any evidence of increased aldosterone production in this experimenai model of renal failure. in the present study repeated measurements of aldosterone excretion in the same patient were used as a qualitative index of adrenal responsiveness to salt restriction in patients with chronic renal disease. Steady-state conditions were insured by maintaining the patients on the same sodium intake for at least one to two weeks prior to measuring the 24 hour excretion rate of aldosterone. During both normal and low dietary sodium intake three consecutive daily measurements were obtained. Under such steadystate conditions and assuming that endogenous aldosterone metabolism remains unaltered, the observed increase in aldosterone excretion should provide a reasonable qualitative index of an increase in aldosterone production. The observed decrease in gbmerular filtration rate during sodium restriction should tend, if anything, to decrease the measured rate of aldosterone excretion; thus changes of filtration rate would not seem to provide an explanation for the observed increase in aldosterone excretion. Although it was not our purpose to examine whether the majority of patients with chronic renal disease are in a state of "hyperaldosteronism", the demonstration of a normal adrenal responsiveness to salt restriction in the present patients is most compatible with the results of Hayslett et al [13] which suggested that aldosterone production was normal in uremia. In the present study, aldosterone excretion was found to be within the range of normal during ingestion of norm& sodium intake. It should be emphasized, however, that a normal rate of aldosterone production could be associated with a normal rate of aldosterone excretion by diseased kidneys in the presence of an elevated plasma concentration of aldosterone. Nevertheless, even if the plasma concentration of aldosterone should be found to be elevated in some patients with chronic renal disease, the present results would seem to somewhat mute the physiological significance of such a finding. In this regard, the most important question concerning the physiological aspects of aldosterone metabolism in chronic renal disease would seem to be whether aldosterone still exerts a significant influence on renal adjustments to alterations in dietary sodium intake. The present finding that the decrease in urinary sodium excretion during sodium restriction may be reversed by spironolactone, and duplicated on a normal sodium intake by exogenous mineralocorticoid administration, supports such a physiological role of aldosterone in chronic renal disease. The concept that chronically diseased kidneys remain responsive to humoral substances which regulate tubular reabsorption is not without precedent. Slatopoisky et al [14] have recently shown that the regulation of tubular phosphate reabsorption in chronic renal disease remains under the control of parathormone activity. Although increased aldosterone activity, enhanced distal sodium reabsorption and increased free-water excretion were consistently observed during sodium restriction in eight patients, in one patient (J.P., Fig. 2) sodium excretion did not diminish significantly and free-water clearance did not increase during sodium restriction. In the presence of a constant fluid intake and urine output this combination of events was associated with the rapid development of hypo-
11 166 Schrier/Regal natremia such as that observed in salt-losing nephritis [ In this same patient sodium restriction on two occasions was associated with a significant increase in aldosterone excretion. Therefore, in contrast to the other eight patients, the presence of a physiological role for aldosterone could be questioned in this patient. The importance of aldosterone in potassium metabolism in chronic renal disease was also examined in the present study. Urinary potassium excretion and the plasma potassium concentration were not significantly different during normal or low sodium intake in the present patients. In view of the findings of increased aldosterone activity and diminished sodium excretion during sodium restriction, a concomitant increase in potassium excretion might have been expected if the rate of sodium delivery to the distal nephron remained unchanged. The constancy of urinary potassium excretion during sodium restriction thus suggested that a decreased rate of sodium delivery to the distal tubule occurred during sodium restriction, and that it obscured the tendency of increased aldosterone activity to increase potassium secretion.2 The decreased rate of glomerular filtration provided a probable mechanism whereby the rate of distal sodium delivery may have been decreased during sodium restriction, but an additional effect of increased proximal sodium reabsorption cannot be excluded [181. The effect of spironolactone to decrease potassium excretion and increase plasma potassium concentration during sodium restriction supported the hypothesis that the constancy of urinary potassium excretion, in spite of decreased distal sodium delivery during sodium restriction, was at least partially related to increased aldosterone activity. In contrast to the renal effect of aldosterone, the excessive fecal losses of potassium in the present patients were not altered during sodium restriction and increased aldosterone activity. This finding confirms the earlier work of Hayes et al [19, 2] who were unable to demonstrate an effect of either sodium restriction or spironolactone on fecal potassium losses in patients with chronic renal disease. The importance of aldosterone in water metabolism in chronic renal disease was also investigated in the 2 Although there is now experimental evidence that the tubular reabsorption of sodium and the tubular secretion of potassium are not necessarily coupled, altered distal sodium delivery in the presence of supraphysiological doses of aldosterone is consistently associated with parallel changes in urinary potassium excretion [4]. present study. Since changes of fluid [21] and protein intake [22] may influence renal concentrating capacity, the present patients were maintained on the same fluid and protein intake throughout the study. Prolonged sodium restriction was found to be associated with a diminution in maximal urinary osmolality and solute-free water reabsorption. Although it has been demonstrated that sodium restriction of three days duration does not affect maximal urinary osmolality in patients with impaired renal function [23], sodium restriction in normal man [24] and dog [25] has been shown to decrease solute-free water reabsorption. The present observations provide the first demonstration of an effect of sodium restriction on the renal concentrating mechanism in chronic renal disease. This defect in renal concentrating capacity during sodium restriction did not seem to be related to increased aldosterone activity since it could not be produced by the administration of exogenous mineralocorticoid in the presence of normal sodium intake. Although Giebisch and Lozano [26] found that the chronic administration of aldosterone was associated with a diminution in solute-free water reabsorption in dogs, the diminished plasma potassium concentration which was observed in their experiments could have been responsible for the renal concentrating defect [27]. In the present patients, prolonged sodium restriction was associated with a diminution in solute-free water reabsorption in the absence of any change in the plasma potassium concentration. A decreased delivery of sodium to the thick ascending limb of the 1oop of Henle, as a result of the diminution in filtration rate, is a possible mechanism for the diminution in renal concentrating capacity during sodium restriction. There is, however, experimental evidence that a decrease in glomerular filtration rate, of a similar degree as that observed in the present study, is associated with an increase, not a decrease, in urinary osmolality in experimental animals undergoing a water diuresis [28, 29]. In hydropenic animals, however, a decrease in sodium delivery to the ascending limb could conceivably decrease rather than increase urine osmolality. In either case, further studies are indicated to define the mechanism of the diminution in renal concentrating capacity that is associated with sodium restriction in chronic renal disease. The present results, however, suggest that this effect is not a result of increased aldosterone activity during sodium restriction. In contrast to the defect in renal concentrating capacity, an improved diluting capacity was observed
12 Influence of aldosterone in chronic renal disease 167 during sodium restriction. The improved diluting capacity could be reversed by the administration of spironolactone despite continued sodium restriction, and it could be duplicated by the administration of exogenous mineralocorticoid and a normal sodium intake. These findings would thus suggest that aldosterone is important in the improved diluting ability during sodium restriction in chronic renal disease. The results however should not be considered to exclude an additional role of diminished solute load per nephron [23]. In summary, the results of the present study suggest that aldosterone is of physiological importance in sodium, potassium and water metabolism in patients with chronic renal disease. The diminution in sodium excretion observed in these patients during sodium restriction was associated with an increase in aldosterone activity, as judged by a qualitively normal increase in aldosterone excretion. This decrease in sodium excretion could be reversed by the administration of spironolactone during continued sodium restriction, and duplicated by the administration of exogenous mineralocorticoid despite a normal sodium intake. Urinary potassium excretion was unaltered during sodium restriction, although the concomitant decrease in glomerular filtration rate suggested a decreased distal delivery of sodium. The kaliuresis and increased plasma potassium concentration during spironolactone administration and sodium restriction suggested that increased aldosterone activity may have allowed a constancy of urinary potassium excretion despite the diminished distal delivery of sodium. Sodium restriction was associated with a diminished maximal urinary osmolality and maximal solute-free water reabsorption which could not be duplicated by exogenous mineralocorticoid administration and a normal sodium intake. However, the improved renal diluting capacity during sodium restriction could be reproduced by the exogenous administration of mineralocorticoid and reversed by spironolactone. Acknowledgements Dr. Schrier is an Established Investigator of the American Heart Association. Reprint requests to Dr. Robert W. Schrier, Department of Medicine, Cardiovascular Research Institute, University of california San Francisco, San Francisco, California 94122, U.S.A. References 1. de Wardener, H. E., Mills, I. H., Clapham, W. F., and Hayter, C. J.: Studies on efferent mechanism of sodium diuresis which follows administration of intravenous saline in dog. Clin. Sci. 21: , Levinsky, N. G., and Lalone, R. C.: Mechanism of sodium diuresis after saline infusion in dog. J. Clin. Invest. 42: , Martino, J. A., and Earley, L. E.: Demonstration of role of physical factors as determinants of natriuretic response to volume expansion. J. Clin. Invest. 46: , Schrier, R. W., and Earley, L. E.: Effects of hematocrit on renal hemodynamics and sodium excretion in hydropenic and volume-expanded dogs. J. Clin. Invest. 49: , Schultze, R. G., Shapiro, H. S., and Bricker, N. S.: Studies on the control of sodium excretion in experimental uremia. J. Clin. Invest. 48: , Slatopolsky, E., Elkan, 1.., Weerts, C., and Bricker, N. 5.: Studies on the characteristics of the control system governing sodium excretion in uremic man. J. Clin. Invest. 47: , Bricker, N. S.: The control of sodium excretion with normal and reduced nephron populations. Am. J. Med. 43: , Cope, C. L., and Pearson, J.: Aldosterone secretion in severe renal failure. Clin. Sd. 25: , Gold, E. M., Kleeman, C. R., Ling, S., Yawata, M., and Maxwell, M.: Sustained aldosterone secretion in chronic renal failure. Clin. Res. 13: 135, Kliman, B., and Petersen, R. E.: Double isotope derivative assay of aldosterone in biological abstracts. J. Biol. Chem. 235: , Tannen, R. L., Regal, E. M., Dunn, M. J., and Schrier, R. W.: Vasopressin-resistant hyposthenuria in advanced chronic renal disease. New Engl. J. Med. 28: , Liddle,. W.: Aldosterone antagonists. Arch. Intern. Med. 12: , Hayslett, J. P., Boyd, J. E., and Epstein, F. H.: Aldosterone production in chronic renal failure. Proc. Soc. Exp. Biol. Med. 13: , Slatopolsky, E., Gradowska, L., Kashemsant, C., Keltncr, R., Manley, C., and Bricker, N. S.: The control of phosphate excretion in uremia. J. Clin. Invest. 45: , Thorn,. W., Koepf, G. F., and Clinton, M.: Renal failure simulating adrenocortical insufficiency. New Engl. J. Med. 231: 76 85, Stanbury, S. W., and Mahler, R. F.: Salt-wasting renal disease. Metabolic observations on a patient with "salt-losing nephritis". Quart. J. Med. 28: , 1959.
13 168 Schrier/ Regal 17. Walker, W. G., Jost, L. J., Johnson, J. R., and Kowarski, A.: Metabolic observations on salt wasting in a patient with renal disease. Am. J. Med. 39: , Weiner, M. W., Weinman, E. J., Kashgarian, M., and Hayslett, J. P.: Accelerated reabsorption in the proximal tubule produced by volume depletion. J. Clin. Invest. 5: , Hayes, C. P., Jr., and Robinson, R. R.: Fecal potassium excretion in patients on chronic intermittent hemodialysis. Trans. Am, Soc. Artif, Intern. Organs 11: , Hayes, C. P., Jr., McLeod, M. E., and Robinson, R. R.: An extrarenal mechanism for the maintenance of potassium balance in severe chronic renal failure. Trans. Assoc. Am. Physicians 8: , Epstein, F. H., Kleeman, C. R., and Hendrikx, A.: The influence of bodily hydration on the renal concentrating process. J. Clin. Invest. 36: , Epstein, F. H., Kleeman, C. R., Pursel, S., and Hendrikx, A.: The effect of feeding protein and urea on the renal concentrating process. J. Clin. Invest. 36: , Adams, D. A., Kleeman, C. R., Bernstein, L. H., and Maxwell, M. H.: An evaluation of maximal water diuresis in chronic renal disease. 11. Effect of variations in sodium intake and excretion. J. Lab. Clin. Med. 58: , Stein, R. M., Levitt, B. H., Goldstein, M. H., Porush, J. G., Eisner, G. M., and Levitt, M. F.: The effects of salt restriction on the renal concentrating operation in normal hydropenic man. J. Clin. invest. 41: , Goldsmith, C., Beasley, H. K., Whalley, P. J., Rector, F. C., Jr., and Seldin, D. W.: The effect of salt deprivation on the urinary concentrating mechanism in the dog. J. Clin. Invest. 4: , Giebisch, G., and Lozano, R.: The effects of adrenal steroids and potassium depletion on the elaboration of an osmotically concentrated urine. J. Clin. Invest. 38: , Hollander, W., Jr., Winters, R. W., Williams, T. F., Bradley, J., Oliver, J., and Welt, L. C.: Defect in the renal tubular reabsorption of water associated with potassium depletion in rats. Am. J. Physiol. 189: , del Greco, and de Wardener, H. E.: The effect on urine osmolarity of a transient reduction in glomerular filtration rate and solute output during a 'water' diuresis. J. Physiol. 131: , Berliner, R. W., and Davidson, D. G.: Production of hypertonic urine in the absence of pituitary antidiuretic hormone. J. Clin. Invest. 36.' , 1957.
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal-Related Questions
 Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
20 Barton et al. Fourteen female mongrel dogs, weighing between 10 to 20 kg, were studied. Light pentobarbital anesthesia
 20 Barton et al. critical determinant of the occurrence and magnitude of natriuresis. It is possible that volume expansion suppresses sodium reabsorption in the loop, and that in the absence of this inhibitory
20 Barton et al. critical determinant of the occurrence and magnitude of natriuresis. It is possible that volume expansion suppresses sodium reabsorption in the loop, and that in the absence of this inhibitory
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
Studies on the Control of Sodium Excretion
 Studies on the Control of Sodium Excretion in Experimental Uremia RAYMOND G. SCHULTZE, HowARD S. SHAPIRo, and NEAL S. BRICKER From the Renal Division, Department of Internal Medicine, Washington University
Studies on the Control of Sodium Excretion in Experimental Uremia RAYMOND G. SCHULTZE, HowARD S. SHAPIRo, and NEAL S. BRICKER From the Renal Division, Department of Internal Medicine, Washington University
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Effect of furosemide on free water excretion in edematous patients with hyponatremia
 Kidney International, VoL 3 (1973), p. 3 34 Effect of furosemide on free water excretion in edematous patients with hyponatremia ROBERT W. SCHRIER, DAVID LEHMAN, BARRY ZACHERLE and LAURENCE E. EARLEY Departments
Kidney International, VoL 3 (1973), p. 3 34 Effect of furosemide on free water excretion in edematous patients with hyponatremia ROBERT W. SCHRIER, DAVID LEHMAN, BARRY ZACHERLE and LAURENCE E. EARLEY Departments
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
nephron alone does not account for the antidiuretic antidiuresis is enhanced by a low sodium intake.
 Journal of Clinical Investigation Vol. 41, No. 11, 1962 THE MECHANISM OF ANTIDIURESIS ASSOCIATED WITH THE ADMINISTRATION OF HYDROCHLOROTHIAZIDE TO PA- TIENTS WITH VASOPRESSIN-RESISTANT DIABETES INSIPIDUS
Journal of Clinical Investigation Vol. 41, No. 11, 1962 THE MECHANISM OF ANTIDIURESIS ASSOCIATED WITH THE ADMINISTRATION OF HYDROCHLOROTHIAZIDE TO PA- TIENTS WITH VASOPRESSIN-RESISTANT DIABETES INSIPIDUS
Faculty version with model answers
 Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
NORMAL POTASSIUM DISTRIBUTION AND BALANCE
 NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
The Control of Phosphate Exeretion in Uremia *
 Journal of Clinical Investigation Vol. 5, No. 5, 19 The Control of Phosphate Exeretion in Uremia * E. SLATOPOLSKY, L. GRADOWSKA, C. KASHEMSANT, R. KELTNER, C. MANLEY, AND N. S. BRICKER t (From the Renal
Journal of Clinical Investigation Vol. 5, No. 5, 19 The Control of Phosphate Exeretion in Uremia * E. SLATOPOLSKY, L. GRADOWSKA, C. KASHEMSANT, R. KELTNER, C. MANLEY, AND N. S. BRICKER t (From the Renal
Fluids and electrolytes
 Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Kidneys in regulation of homeostasis
 Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
** TMP mean page 340 in 12 th edition. Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2:
 QUESTION Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2: Urine flow rate = 1 ml/min Urine inulin concentration = 100 mg/ml Plasma inulin concentration = 2 mg/ml
QUESTION Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2: Urine flow rate = 1 ml/min Urine inulin concentration = 100 mg/ml Plasma inulin concentration = 2 mg/ml
CONCERNING THE EFFECTS OF MAGNESIUM SULFATE ON RENAL FUNCTION, ELECTROLYTE EXCRETION, AND CLEARANCE OF MAGNESIUM
 CONCERNING THE EFFECTS OF MAGNESIUM SULFATE ON RENAL FUNCTION, ELECTROLYTE EXCRETION, AND CLEARANCE OF MAGNESIUM B. I. Heller,, J. F. Hammarsten, F. L. Stutzman J Clin Invest. 1953;32(9):858-861. https://doi.org/10.1172/jci102803.
CONCERNING THE EFFECTS OF MAGNESIUM SULFATE ON RENAL FUNCTION, ELECTROLYTE EXCRETION, AND CLEARANCE OF MAGNESIUM B. I. Heller,, J. F. Hammarsten, F. L. Stutzman J Clin Invest. 1953;32(9):858-861. https://doi.org/10.1172/jci102803.
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
On the Adaptation in Sodium Excretion in Chronic Uremia
 On the Adaptation in Sodium Excretion in Chronic Uremia THE EFFECTS OF "PROPORTIONAL REDUCTION" OF SODIUM INTAKE R. WILLIAM SCHMIDT, JACQUES J. BOURGOIGNIE, and NEAL S. BRICKER From the Division of Nephrology,
On the Adaptation in Sodium Excretion in Chronic Uremia THE EFFECTS OF "PROPORTIONAL REDUCTION" OF SODIUM INTAKE R. WILLIAM SCHMIDT, JACQUES J. BOURGOIGNIE, and NEAL S. BRICKER From the Division of Nephrology,
Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are
 Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D.
 Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Non-protein nitrogenous substances (NPN)
 Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Renal System and Excretion
 Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Counter-Current System Regulation of Renal Functions
 Counter-Current System Regulation of Renal Functions Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most
Counter-Current System Regulation of Renal Functions Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most
1. Urinary System, General
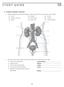 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
CONCENTRATING MECHANISM IN THE DOG*
 THE EFFECT OF SALT DEPRIVATION ON THE URINARY CONCENTRATING MECHANISM IN THE DOG* By CARL GOLDSMITH,t H. KENT BEASLEY, PEGGY J. WHALLEYt FLOYD C. RECTOR, JR. AND DONALD W. SELDIN (From The Department of
THE EFFECT OF SALT DEPRIVATION ON THE URINARY CONCENTRATING MECHANISM IN THE DOG* By CARL GOLDSMITH,t H. KENT BEASLEY, PEGGY J. WHALLEYt FLOYD C. RECTOR, JR. AND DONALD W. SELDIN (From The Department of
Effect of Thoracic Cava Obstruction on Response of Proximal Tubule Sodium Reabsorption to Saline Infusion *
 Journal of Clinical Investigation Vol. 45, No. 2, 1966 Effect of Thoracic Cava Obstruction on Response of Proximal Tubule Sodium Reabsorption to Saline Infusion * WILLIAM J. CIRKSENA,t JOHN H. DIRKS, AND
Journal of Clinical Investigation Vol. 45, No. 2, 1966 Effect of Thoracic Cava Obstruction on Response of Proximal Tubule Sodium Reabsorption to Saline Infusion * WILLIAM J. CIRKSENA,t JOHN H. DIRKS, AND
On the adaptation in potassium excretion associated with nephron reduction in the dog
 On the adaptation in potassium excretion associated with nephron reduction in the dog Raymond G. Schultze,, Sali Caglar, Neal S. Bricker J Clin Invest. 1971;50(5):1061-1068. https://doi.org/10.1172/jci106577.
On the adaptation in potassium excretion associated with nephron reduction in the dog Raymond G. Schultze,, Sali Caglar, Neal S. Bricker J Clin Invest. 1971;50(5):1061-1068. https://doi.org/10.1172/jci106577.
Diuretic Agents Part-2. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
Kidneys and Homeostasis
 16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System)
 NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System) HOMEOSTASIS **Recall HOMEOSTASIS is the steady-state physiological condition of the body. It includes: 1) Thermoregulation:
NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System) HOMEOSTASIS **Recall HOMEOSTASIS is the steady-state physiological condition of the body. It includes: 1) Thermoregulation:
IN THE DOG. by means of a soft indwelling rubber catheter. At the beginning of each study the dogs were given a quantity
 THE EFFECT OF VARIATIONS IN SOLUTE EXCRETION AND VASOPRESSIN DOSAGE ON THE EXCRETION OF WATER IN THE DOG By JACK ORLOFF, HENRY N. WAGNER, JR., AND DOUGLAS G. DAVIDSON (From the Laboratory of Kidney and
THE EFFECT OF VARIATIONS IN SOLUTE EXCRETION AND VASOPRESSIN DOSAGE ON THE EXCRETION OF WATER IN THE DOG By JACK ORLOFF, HENRY N. WAGNER, JR., AND DOUGLAS G. DAVIDSON (From the Laboratory of Kidney and
KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin
![KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin](/thumbs/87/96174332.jpg) Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
New Haven, Conn.) laboratory, having abstained from food and fluids for the preceding 12 hours. In order to assure a constant and
 THE INFLUENCE OF BODILY HYDRATION ON THE RENAL CONCENTRATING PROCESS 1 By FRANKLIN H. EPSTEIN,2 CHARLES R. KLEEMAN, AND ACHIEL HENDRIKX (From the Department of Internal Medicine, Yale University School
THE INFLUENCE OF BODILY HYDRATION ON THE RENAL CONCENTRATING PROCESS 1 By FRANKLIN H. EPSTEIN,2 CHARLES R. KLEEMAN, AND ACHIEL HENDRIKX (From the Department of Internal Medicine, Yale University School
For more information about how to cite these materials visit
 Author(s): Michael Heung, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Michael Heung, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology
 Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper
Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper
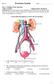
Use the following diagram to answer the next question. 1. In the diagram above, pressure filtration occurs in a. W b. X c. Y d. Z
 Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
The Excretory System. Biology 20
 The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
Urinary Kallikrein Excretion in Hypertensive Man
 Urinary Kallikrein Excretion in Hypertensive Man RELATIONSHIPS TO SODIUM INTAKE AND SODIUM-RETAINING STEROIDS By Harry S. Margolius, David Horwttz, John J. Pisano, and Harry R. Kelser ABSTRACT Urinary
Urinary Kallikrein Excretion in Hypertensive Man RELATIONSHIPS TO SODIUM INTAKE AND SODIUM-RETAINING STEROIDS By Harry S. Margolius, David Horwttz, John J. Pisano, and Harry R. Kelser ABSTRACT Urinary
Renal Regulation of Sodium and Volume. Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM
 Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
Fluid and electrolyte balance, imbalance
 Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
A&P of the Urinary System
 A&P of the Urinary System Week 44 1 Objectives Identify the organs of the urinary system, from a Identify the parts of the nephron (the functional unit List the characteristics of a normal urine specimen.
A&P of the Urinary System Week 44 1 Objectives Identify the organs of the urinary system, from a Identify the parts of the nephron (the functional unit List the characteristics of a normal urine specimen.
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Osmoregulation and Renal Function
 1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
Diagram of the inner portions of the kidney
 Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
The Effect of Saline Infusion on Sodium Reabsorption by the Proximal Tubule of the Dog *
 Journal of Clinical Investigation Vol. 44, No. 7, 1965 The Effect of Saline Infusion on Sodium Reabsorption by the Proximal Tubule of the Dog * JOHN H. DIRKS,t WILLIAM J. CIRKSENA,4 AND ROBERT W. BERLINER
Journal of Clinical Investigation Vol. 44, No. 7, 1965 The Effect of Saline Infusion on Sodium Reabsorption by the Proximal Tubule of the Dog * JOHN H. DIRKS,t WILLIAM J. CIRKSENA,4 AND ROBERT W. BERLINER
Increased sodium reabsorption in the proximal and distal tubule of caval dogs
 Increased sodium reabsorption in the proximal and distal tubule of caval dogs George J. Kaloyanides,, Nancy C. Pablo, Jerome G. Porush J Clin Invest. 1969;48(8):1543-1551. https://doi.org/1.1172/jci1612.
Increased sodium reabsorption in the proximal and distal tubule of caval dogs George J. Kaloyanides,, Nancy C. Pablo, Jerome G. Porush J Clin Invest. 1969;48(8):1543-1551. https://doi.org/1.1172/jci1612.
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
Urinary System and Excretion. Bio105 Lecture 20 Chapter 16
 Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
BIOL 2402 Renal Function
 BIOL 2402 Renal Function Dr. Chris Doumen Collin County Community College 1 Renal Clearance and GFR Refers to the volume of blood plasma from which a component is completely removed in one minute by all
BIOL 2402 Renal Function Dr. Chris Doumen Collin County Community College 1 Renal Clearance and GFR Refers to the volume of blood plasma from which a component is completely removed in one minute by all
Importance of Sodium Intake and Mineralocorticoid Hormone in the Impaired
 Importance of Sodium Intake and Mineralocorticoid Hormone in the Impaired Water Excretion in Adrenal Insufficiency R. C. UFFERMAN and R. W. Sam=IEl From the Department of Medicine and Cardiovascular Research
Importance of Sodium Intake and Mineralocorticoid Hormone in the Impaired Water Excretion in Adrenal Insufficiency R. C. UFFERMAN and R. W. Sam=IEl From the Department of Medicine and Cardiovascular Research
Wales Critical Care & Trauma Network (North) Management of Hyponatraemia in Intensive Care Guidelines
 Wales Critical Care & Trauma Network (North) Management of Hyponatraemia in Intensive Care Guidelines Author: Richard Pugh June 2015 Guideline for management of hyponatraemia in intensive care Background
Wales Critical Care & Trauma Network (North) Management of Hyponatraemia in Intensive Care Guidelines Author: Richard Pugh June 2015 Guideline for management of hyponatraemia in intensive care Background
MIGUEL CHIAPPORI 4. Renal function. Twelve healthy Peruvian males between the ages of 20 and 28 years were studied. None
 ORAL SODIUM LOADING IN NORMAL INDIVIDUALS By KEHL MARKLEY,1 MANUEL BOCANEGRA,2 GUILLERMO MORALES,3 AND MIGUEL CHIAPPORI 4 (From the U. S. Public Health Service, U. S. Department of Health, Education, and
ORAL SODIUM LOADING IN NORMAL INDIVIDUALS By KEHL MARKLEY,1 MANUEL BOCANEGRA,2 GUILLERMO MORALES,3 AND MIGUEL CHIAPPORI 4 (From the U. S. Public Health Service, U. S. Department of Health, Education, and
organs of the urinary system
 organs of the urinary system Kidneys (2) bean-shaped, fist-sized organ where urine is formed. Lie on either sides of the vertebral column, in a depression beneath peritoneum and protected by lower ribs
organs of the urinary system Kidneys (2) bean-shaped, fist-sized organ where urine is formed. Lie on either sides of the vertebral column, in a depression beneath peritoneum and protected by lower ribs
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
SUBJECTS TO THE INGESTION OF ISOTONIC SALINE
 FACTORS INFLUENCING THE DIURETIC RESPONSE OF SEATED SUBJECTS TO THE INGESTION OF ISOTONIC SALINE SOLUTION By WILLIAM H. BIRCHARD AND MAURICE B. STRAUSS (From the Medical Service and Research Laboratory,
FACTORS INFLUENCING THE DIURETIC RESPONSE OF SEATED SUBJECTS TO THE INGESTION OF ISOTONIC SALINE SOLUTION By WILLIAM H. BIRCHARD AND MAURICE B. STRAUSS (From the Medical Service and Research Laboratory,
The Urinary System. BIOLOGY OF HUMANS Concepts, Applications, and Issues. Judith Goodenough Betty McGuire
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
(ethanol) suggests that it is similar to the diuresis following ingestion of water.
 435 J. Physiol. (I946) I04, 435-442 6I2.464.I THE EFFECT OF ETHYL ALCOHOL AND SOME OTHER DIURETICS ON CHLORIDE EXCRETION IN MAN BY M. GRACE EGGLETON AND ISABEL G. SMITH, From the Physiology Department,
435 J. Physiol. (I946) I04, 435-442 6I2.464.I THE EFFECT OF ETHYL ALCOHOL AND SOME OTHER DIURETICS ON CHLORIDE EXCRETION IN MAN BY M. GRACE EGGLETON AND ISABEL G. SMITH, From the Physiology Department,
The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency *
 lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
In nocturnal enuresis
 The role of the kidney In nocturnal enuresis Kostas Kamperis MD PhD Dept of Pediatrics, Section of Nephrology Aarhus University Hospital, Aarhus, Denmark Enuresis prototypes Nocturnal polyuria Bladder
The role of the kidney In nocturnal enuresis Kostas Kamperis MD PhD Dept of Pediatrics, Section of Nephrology Aarhus University Hospital, Aarhus, Denmark Enuresis prototypes Nocturnal polyuria Bladder
On the Mechanism of Polyuria in Potassium Depletion
 On the Mechanism of Polyuria in Potassium Depletion THE ROLE OF POLYDIPSIA TOMAS BERL, STUART L. LINAS, GARY A. AISENBREY, and ROBERT J. ANDERSON From the Division of Renal Diseases, Department of Medicine,
On the Mechanism of Polyuria in Potassium Depletion THE ROLE OF POLYDIPSIA TOMAS BERL, STUART L. LINAS, GARY A. AISENBREY, and ROBERT J. ANDERSON From the Division of Renal Diseases, Department of Medicine,
Functional Renal Physiology and Urine Production
 Functional Renal Physiology and Urine Production Urinalysis can provide insight into hydration status, renal function or dysfunction, systemic disease, and toxic insults. Accurate interpretation of urinalysis
Functional Renal Physiology and Urine Production Urinalysis can provide insight into hydration status, renal function or dysfunction, systemic disease, and toxic insults. Accurate interpretation of urinalysis
Sunday, July 17, 2011 URINARY SYSTEM
 URINARY SYSTEM URINARY SYSTEM Let s take a look at the anatomy first! KIDNEYS: are complex reprocessing centers where blood is filtered through and waste products are removed. Wastes and extra water become
URINARY SYSTEM URINARY SYSTEM Let s take a look at the anatomy first! KIDNEYS: are complex reprocessing centers where blood is filtered through and waste products are removed. Wastes and extra water become
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Renal Pharmacology. Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics BIMM118
 Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
mid ihsan (Physiology ) GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B **
 (Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
(Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
Diuretics having the quality of exciting excessive excretion of urine. OED. Inhibitors of Sodium Reabsorption Saluretics not Aquaretics
 Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
2) This is a Point and Click question. You must click on the required structure.
 Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Urinary system. Lab-7
 Urinary system Lab-7 Excretion: processes that remove wastes and excess materials from the body Urinary system (kidneys): excretes nitrogenous wastes, excess solutes, and water The Kidneys Regulate Water
Urinary system Lab-7 Excretion: processes that remove wastes and excess materials from the body Urinary system (kidneys): excretes nitrogenous wastes, excess solutes, and water The Kidneys Regulate Water
NATURAL HISTORY AND SURVIVAL OF PATIENTS WITH ASCITES. PATIENTS WHO DO NOT DEVELOP COMPLICATIONS HAVE MARKEDLY BETTER SURVIVAL THAN THOSE WHO DEVELOP
 PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
during mannitol diuresis was compared in normal
 Journal of Clinical Investigation Vol. 41, No. 4, 1962 EVIDENCE FOR A DIRECT EFFECT OF SERUM SODIUM CONCENTRATION ON SODIUM REABSORPTION * By CARL GOLDSMITHt FLOYD C. RECTOR, JR. AND DONALD W. SELDIN (From
Journal of Clinical Investigation Vol. 41, No. 4, 1962 EVIDENCE FOR A DIRECT EFFECT OF SERUM SODIUM CONCENTRATION ON SODIUM REABSORPTION * By CARL GOLDSMITHt FLOYD C. RECTOR, JR. AND DONALD W. SELDIN (From
Dr.Nahid Osman Ahmed 1
 1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
DIURETICS-4 Dr. Shariq Syed
 DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
1.&Glomerular/Pressure&Filtration&
 Urine&Formation& Overall&Process&! Urine gets rid of wastes (NH 3, urea, uric acid, creatinine) and other substances (vitamins, penicillin, histamines) found in excess in the blood!! blood is filtered
Urine&Formation& Overall&Process&! Urine gets rid of wastes (NH 3, urea, uric acid, creatinine) and other substances (vitamins, penicillin, histamines) found in excess in the blood!! blood is filtered
Outline Urinary System
 Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
Outline Urinary System. Urinary System and Excretion. Urine. Urinary System. I. Function II. Organs of the urinary system
 Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari
![Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari](/thumbs/90/104323364.jpg) Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Excretion Chapter 29. The Mammalian Excretory System consists of. The Kidney. The Nephron: the basic unit of the kidney.
 Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Diuretic Use in Neonates
 Neonatal Nursing Education Brief: Diuretic Use in the Neonate http://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/ Diuretics
Neonatal Nursing Education Brief: Diuretic Use in the Neonate http://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/ Diuretics
41B. Metabolism produces wastes that must be eliminated from the body. This. Renal System Physiology: Computer Simulation
 41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
Renal physiology D.HAMMOUDI.MD
 Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Renal Functions: Renal Functions: Renal Function: Produce Urine
 Renal Functions: Excrete metabolic waste products Reabsorb vital nutrients Regulate osmolarity: Maintain ion balance Regulate extracellular fluid volume (and thus blood pressure) Renal Functions: Regulate
Renal Functions: Excrete metabolic waste products Reabsorb vital nutrients Regulate osmolarity: Maintain ion balance Regulate extracellular fluid volume (and thus blood pressure) Renal Functions: Regulate
Sodium Reabsorption in the Distal Nephron of Dogs
 Effect of Extracellular Volume Expansion upon Sodium Reabsorption in the Distal Nephron of Dogs CiEAVES M. BiNM= From the Divisions of Nephrology, Departments of Medicine, Duke University Medical Center,
Effect of Extracellular Volume Expansion upon Sodium Reabsorption in the Distal Nephron of Dogs CiEAVES M. BiNM= From the Divisions of Nephrology, Departments of Medicine, Duke University Medical Center,
Human Urogenital System 26-1
 Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
April 08, biology 2201 ch 11.3 excretion.notebook. Biology The Excretory System. Apr 13 9:14 PM EXCRETORY SYSTEM.
 Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Objectives. Objectives
 Objectives Volume regulation entails the physiology of salt content regulation The edematous states reflect the pathophysiology of salt content regulation The mechanisms of normal volume regulation mediate
Objectives Volume regulation entails the physiology of salt content regulation The edematous states reflect the pathophysiology of salt content regulation The mechanisms of normal volume regulation mediate
Structural and Functional Adaptation after Reduction of Nephron Population
 TIlE YALE JOUJRNAL OF BIOLOGY AND MEDICINE 52 (1979), 271-287 Structural and Functional Adaptation after Reduction of Nephron Population FREDRIC 0. FINKELSTEIN AND JOHN P. HAYSLETT Departments of Medicine
TIlE YALE JOUJRNAL OF BIOLOGY AND MEDICINE 52 (1979), 271-287 Structural and Functional Adaptation after Reduction of Nephron Population FREDRIC 0. FINKELSTEIN AND JOHN P. HAYSLETT Departments of Medicine
Pressure Diuresis 9 Sample Student Essays
 Pressure Diuresis 9 Sample Student Essays Below please find assembled consecutively in one document the brief analyses submitted by nine students in Mammalian Physiology 08 to the Teach Yourself Pressure
Pressure Diuresis 9 Sample Student Essays Below please find assembled consecutively in one document the brief analyses submitted by nine students in Mammalian Physiology 08 to the Teach Yourself Pressure
Beneficial Role of Tolvaptan in the Control of Body Fluids Without Reductions in Residual Renal Function in Patients Undergoing Peritoneal Dialysis
 Advances in Peritoneal Dialysis, Vol. 29, 2013 Takefumi Mori, 1,2,3 Ikuko Oba, 1 Kenji Koizumi, 1 Mayumi Kodama, 1 Miwako Shimanuki, 1 Mizuho Tanno, 1 Makiko Chida, 1 Mai Saito, 1 Hideyasu Kiyomoto, 1
Advances in Peritoneal Dialysis, Vol. 29, 2013 Takefumi Mori, 1,2,3 Ikuko Oba, 1 Kenji Koizumi, 1 Mayumi Kodama, 1 Miwako Shimanuki, 1 Mizuho Tanno, 1 Makiko Chida, 1 Mai Saito, 1 Hideyasu Kiyomoto, 1
Depression of Fractional Sodium
 Depression of Fractional Sodium Reabsorption by the Proximal Tubule of the Dog Without Sodium Diuresis STUART S. HOwAmms, BERNAD B. DAVIS, FRANKLYN G. KNOX, FRED S. WRIGHT, and ROBERT W. BERLINER From
Depression of Fractional Sodium Reabsorption by the Proximal Tubule of the Dog Without Sodium Diuresis STUART S. HOwAmms, BERNAD B. DAVIS, FRANKLYN G. KNOX, FRED S. WRIGHT, and ROBERT W. BERLINER From
