A mandibular propulsive appliance modulates collagen-binding integrins distribution in the young rat condylar cartilage
|
|
|
- Tabitha McBride
- 5 years ago
- Views:
Transcription
1 Biorheology 43 (2006) IOS Press A mandibular propulsive appliance modulates collagen-binding integrins distribution in the young rat condylar cartilage Mara Rúbia Marques, Denise Hajjar, Virgínia Oliveira Crema, Edna Teruko Kimura and Marinilce Fagundes Santos Cell and Developmental Biology Department, Biomedical Sciences Institute, University of São Paulo, São Paulo, Brazil Abstract. We have previously shown that a mandibular propulsive appliance (MPA) stimulates cell proliferation and the synthesis of growth factors in the rat condylar cartilage. The aim of this study was to evaluate the effects of a MPA in the distribution of the integrin subunits α1 andα2 in this cartilage. Twenty eight days-old male Wistar rats were divided into treated (T) and age-matched control groups (C). Treated rats wore the appliance during 3, 5, 7, 9, 11, 15, 20, 30 and 35 days. The condyles were fixed, decalcified and paraffin-embedded. The distribution of α1 andα2 was studied by immunohistochemistry. Alpha1 distribution was uniform along the cartilage, increasing in 48 days-old rats (C20). Treated animals anticipated this increase to the age of 36 days (T9). The number of α2-positive cells was increased in C9 in the anterior condylar region, in C9 and C20 in the middle region and showed no differences in the posterior region. The MPA apparently abolished all variations, leading to a single increase at T30 in all regions. These results suggest that integrins containing the α1 andα2 subunits are modulated by forces promoted by the MPA, participating of the biological response to this therapy. Keywords: Functional orthopedics, condyle, alpha1, alpha2, integrin 1. Introduction The modulation of mandibular growth by the application of mechanical forces has been studied to improve the correction of discrepancies between the maxilla and the mandible, as well as dental malocclusions [19]. It is believed that the use of functional orthopedic appliances by young individuals generate tension in orofacial muscles, which are transmitted to the mandibular condyle, an important growth center of the mandible, generating adaptation. Clinical results obtained with this therapy are well characterized, although the molecular mechanisms underlying this response still remain unclear. The mandibular condyle is part of the temporomandibular joint, a bilateral synovial joint between the mandible and the temporal bones. The young condylar cartilage is a hyaline cartilage covered by a fibrous layer. Proliferative undifferentiated cells, located immediately below this layer, differentiate into chondroblasts, which produce extracellular matrix (ECM), mostly collagen type II and proteoglycans. * Address for correspondence: M.F. Santos, Cell and Developmental Biology Department, Av. Prof. Lineu Prestes 1524 sala 434, São Paulo, SP, Brazil, CEP X/06/$ IOS Press and the authors. All rights reserved
2 294 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage Later on, these cells become entrapped within this matrix, becoming chondrocytes. Hypertrophy of chondrocytes is followed by endochondral ossification. The proliferative compartment in the condylar cartilage is formed by undifferentiated cells and chondroblasts [19,24]. It is possible to modulate growth of the condylar cartilage by the application of forces [6]. Small load alterations such as those promoted by food hardness, for example, affect growth [1]. In cultured rat condylar cartilage hydrostatic compressive forces stimulated glycosaminoglycan (GAG) and DNA synthesis in a magnitude-dependent manner [22], and intermittent compressive loading increased collagen I and fibronectin synthesis [17]. We have previously observed that an intermittent indirect force generated by a mandibular propulsive orthopedic appliance was able to increase the expression of Insulin-like Growth Factors I and II (IGF-I and IGF-II), and cell proliferation in the young rat condylar cartilage [7]. Charlier et al. (1969) also observed an increase in the size of the undifferentiated layer after using this kind of appliance [2]. In our previous study, if this appliance was used for a short period of time (up to 9 days), the effect on mandibular repositioning was reversible. After 11 days of use, the mandibular repositioning was permanent, even after the appliance s withdrawn (unpublished observation). The transmission of forces applied to the condylar chondrocytes is probably mediated by integrins, heterodimeric transmembrane receptors for ECM proteins. Integrins contain two non-covalently associated subunits, α and β, which bind ECM proteins through their extracellular domains [5,9]. Through their cytoplasmic domains integrins usually interact with the cytoskeleton. Although integrins have no intrinsic enzymatic activity, they trigger signaling pathways upon binding to ECM, which might regulate several chondrocyte functions such as differentiation, matrix remodeling, gene expression, responses to mechanical stimulation and cell survival [13]. In normal human articular chondrocytes, integrin activation, consequent to mechanical stimulation in vitro, results in tyrosine phosphorylation of regulatory proteins and subsequent secretion of autocrine and paracrine factors [15]. The expression of integrins in human femoral condyles has been shown to be modulated by health condition and developmental stage [8,15,18,20]. During human growth, α1 and α2 integrin subunits were observed in the proliferative and hypertrophic layers [8], determined mostly by the ECM composition [11]. Condrossarcoma cells, as well as fetal chondrocytes, showed high levels of the collagen-binding α2β1 integrin [12]. We believe that an improved comprehension of how integrins mediate chondrocyte responses to mechanical stimulation, and how cross talk between integrins, ECM, and autocrine/paracrine signaling molecules (such as IGF-I and IGF-II) regulate mechanotransduction are important for further understanding how functional orthopedic appliances promote cartilage remodeling. 2. Materials and methods 2.1. Animals and treatment All experiments were conducted in accordance with the NIH guidelines, and the protocols were approved by the Biomedical Sciences Institute/USP Ethical Committee for Animal Research. Seventy-two male Wistar rats were used, beginning at 28 days of age. They were divided into treated (T) and control age-matched groups (C). Treated animals wore the appliance for 3, 5, 7, 9, 11, 15, 20, 30 and 35 days, from 8:00 a.m. to 6:00 p.m. The appliance was changed when necessary, according to the animal s development. Food and water were available at night, ad libitum. At the time of sacrifice, animals were
3 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage 295 anaesthetized with chloral hydrate (30 mg/100 g of body weight) and perfused with saline and with fixative solution (4% formaldehyde, Sigma, St. Louis, MO, USA). The mandibular condyles were dissected out, post-fixed in 4% formaldehyde at 4 C during 24 h, decalcified with 10% EDTA for 20 days and embedded in Paraplast R. Sagittal 5 µm-thick sections were placed on Poly-L-Lysine-coated slides (Sigma, St. Louis, MO, USA) Orthopedic appliance The appliance was designed as described by Hajjar et al. [7]. Briefly, it consisted of an inclined copper plane that raised the anterior displacement of the mandible every time the animals attempted to close their mouths at the normal position. The appliance was banded 90 at the anterior portion and a rubber tube was glued close, to fit in the upper incisor of the animal. All edges were rounded to avoid mucosal irritation. A soft leather collar was used to prevent the removal of the appliance; control animals wore the collar alone Immunohistochemistry Integrin subunits were investigated by immunohistochemistry, using the avidin biotin peroxidase technique. Rabbit polyclonal anti-α1 (Chemicon, AB1934, Temecula, CA, USA) and anti-α2 (Chemicon, AB1936, Temecula, CA, USA) were used at a 1 : 100 dilution in PBS, overnight at room temperature (RT). After washing in PBS, sections were incubated with the biotin-conjugated goat anti-rabbit secondary antibody (Jackson Immunoresearch Laboratories, West Grove, PA, USA) at a 1 : 250 dilution during 2 hours at RT. The sections were then revealed with 3,3 diamino-benzidine (DAB) in the presence of 0.02% H 2 O 2 for 30 min, mounted with Permount R (Fisher Chemicals) and examined on a Leitz microscope (Aristoplan). As a negative control the omission of the primary antibody was used, and no labeling was observed under these conditions. Pictures were taken using a CCD camera and the Scion Image software (NIH, Bethesda, USA) Counting of labeled cells and statistical analysis The quantification of labeled cells was performed by the evaluation of 900 cells in 3 randomly chosen microscopic fields at the anterior, central and posterior regions of the condylar cartilage per section. The number of labeled cells was expressed as a percentage of the total number of cells observed. Data were submitted to analysis of variance (ANOVA) followed by the Tukey s post-test for multiple comparisons. Main comparisons were made within the control group (between different ages) and between the treated groups and their age-matched controls. A P value of less than 0.05 was considered to be significant. Results were expressed as mean ± standard deviation (SD). 3. Results 3.1. The propulsor appliance modulates α1 integrin subunit distribution During normal development, α1 distribution was almost restricted to the hypertrophic layer in C3 (Fig. 1). From C5 on, α1 staining was distributed throughout the cartilage, including the proliferative
4 296 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage Fig. 1. Distribution of the α1 integrin subunit in the rat condylar cartilage during normal growth (C3 C30) and under therapy with a mandibular propulsive appliance (T3 T30). The α1 subunit was studied by immunohistochemistry using the avidin biotin-peroxidase technique in sagittal sections of the cartilage. T3, T9, T15, T20 and T30 correspond to young animals that wore the appliance for 3, 9, 15, 20 and 30 days, respectively. Each group was sacrificed with their age-matched control groups. In C3, all layers are shown: fibrous (F), undifferentiated (U), chondroblasts (C), mature chondrocytes (MC) and Hypertrophic (H). In detail, the negative control consisting of the omission of the primary antibody (CON). Magnification bar = 100 µm.
5 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage 297 Fig. 2. Quantification of α1-labeled cells in different regions of the rat condylar cartilage: (A) anterior region; (B) central region; and (C) posterior region. The dotted line separates the early and late phases of the therapy. Data are presented as percentage of total cells counted, mean ± SD. # P < when compared to the previous group; P < when compared to the age-matched control group, according to ANOVA and Tukey s post-test. layer. Labeling intensity, however, was variable, mainly in the proliferative compartment and the chondrocyte layer (Fig. 1). The number of α1-positive cells was uniform among the anterior, central and posterior regions of the cartilage, varying from 50 70% (Fig. 2). In the control group, there was an increase in α1-positively labeled cells in the C20 group (100% labeled cells compared to 58.52% in C15). Labeling intensity was increased as well. Both the number of labeled cells and labeling intensity decreased afterwards, returning to initial levels (Figs 1 and 2).
6 298 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage Fig. 3. Distribution of the α2 integrin subunit in the rat condylar cartilage during normal growth (C3 C30) and under therapy with a mandibular propulsive appliance (T3 T30). The α2 subunit was studied by immunohistochemistry using the avidin biotin-peroxidase technique in sagittal sections of the cartilage. T3, T9, T15, T20 and T30 correspond to young animals that wore the appliance for 3, 9, 15, 20 and 30 days, respectively. Each group was sacrificed with their age-matched control groups. In detail, the negative control consisting of the omission of the primary antibody (CON). Magnification bar = 100 µm.
7 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage 299 The appliance promoted a slight increase in labeling intensity at T3, mostly in the hypertrophic layer (Fig. 1). At T9, however, both the number of positive cells and the intensity of the staining were enhanced, comparing to C9 (100% labeled cells vs % in C9) (Figs 1 and 2). From T9 on, the labeling was similar to that observed in the control group, except for T20, which did not show the increase in α1 observed in C The α2 integrin subunit distribution varies according to the cartilage region and is modulated by the propulsor appliance At C3, α2-positive cells were observed mostly in the hypertrophic layer (Fig. 3). At C9 there was a marked increase in the number of positive cells in the anterior and middle regions of the cartilage, but Fig. 4. Quantification of α2-labeled cells in different regions of the rat condylar cartilage: (A) anterior region; (B) central region; and (C) posterior region. The dotted line separates the early and late phases of the therapy. Data are presented as percentage of total cells counted, mean ± SD. # P < when compared to the previous group; P < when compared to the age-matched control group, according to ANOVA and Tukey s post-test.
8 300 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage not in the posterior region (100% labeled cells vs. 67% in C7, Fig. 4). We observed an enhanced labeling intensity as well, and the presence of α2 in all cartilage layers (Fig. 3). This increase in the number of labeled cells was observed at C20 again, but only in the central cartilage region (77% of labeled cells). From C20 on, the number of marked cells gradually returned to the initial levels, being mostly restricted to the hypertrophic layer (Figs 3 and 4). No variations were observed for α2 labeling in the posterior region (Fig. 4). As observed for α1, the staining in T3 was slightly more intense than that observed in the control group (Fig. 3). Curiously, the appliance abolished variations described above in the number of labeled cells, leading to a single increase in number and staining intensity at T30, in the anterior, central and posterior regions (Figs 3 and 4). This difference, observed mainly in the proliferative compartment, was not observed in T Discussion The modulation of the mandibular condylar cartilage growth and adaptation is considered an effective way to correct growth discrepancies between the mandible and maxilla. In ortodonthics, the application of mechanical forces through the use of orthopedic appliances promotes very good clinical results. In this study, we showed that a mandibular propulsor appliance changed the distribution of the integrin subunits α1 and α2, probably corresponding to the collagen-binding integrins α1β1 and α2β1, in the rat condylar cartilage. For both α1 and α2 there was variation in expression and distribution during growth. In control animals, the α1 subunit raised uniformly in all cartilage regions in C20, when the rats were 48 days-old, around puberty. The α2 subunit, on the other hand, was uniformly expressed along the condylar cartilage, but showed some differential distribution with growth. Specifically, there was an increase in C9, but only in the anterior and posterior regions of the cartilage, and another peak in C20, similar to α1, but observed only in the central region. An interesting idea is that while α1 is modulated by hormones and growth factors present in the cartilage during growth, α2 could be related to force, because its expression was higher close to the muscle insertion. However, maybe contrary to this hypothesis, the appliance, which we consider as an intermittent indirect force applied to the cartilage, anticipated the α1 peak (previously in C20) to 9 days of treatment (T9). On the other hand, all α2 variations were abolished by the appliance s use, and a peak of expression appeared at T30, only in the treated group. In general, expression peaks mean a broader distribution, with all layers marked, including the undifferentiated proliferative layer. It is tempting to think that α2 was being replaced by α1 in T9, and that there was a late effect of this therapy after 30 days of treatment, leading to an increase in α2. After 9 days of treatment, it is known that there is a marked increase in cell proliferation, as observed by PCNA (Proliferating Cell Nuclear Antigen) labeling [7]. However, we believe that in order to consolidate this effect on growth, the process of endochondral ossification must be continued, and the use of the appliance ensures that those new cells will differentiate and synthesize ECM proteins, such as collagen II and proteoglycans. If the appliance is removed after 9 days, the mandible does not return to its original position. Several studies showed that the condylar cartilage responds to mechanical stimuli by altering cell proliferation and ECM proteins expression. For example, intermittent compressive forces increased sulfated glycosaminoglycans and collagen synthesis, while continuous compressive forces reduced ECM synthesis [3,4]. Another study showed that compressive forces applied uniformly in vitro to the whole condyle using a pneumatic loading system increased collagen I and fibronectin [17]. One has to consider,
9 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage 301 however, the force direction in these experiments, since it appears to be determinant for the effects on cartilage. Opposite forces such as those exerted by a protrusive appliance compress the anterior portion of the condyle against the temporal articular eminence, decreasing cartilage thickness and aggrecan immunostaining in rats [23]. However, in the same anterior region there are considerable stretching forces compared to the rest of the condyle, due to the muscle intermittent activity. Apparently, ECM composition defines the expression of different integrins in chondrocytes. For example, in vitro, α1 and α2 expression were upregulated by collagens I and II, respectively, in porcine knee articular cartilage [11]. It is possible that, in our study, the increase in α2 after 30 days of treatment was related to a higher amount of collagen type II produced by chondrocytes, after cell proliferation and differentiation. In monkeys and humans, the distribution and expression of collagens I and II vary with age [10,16], suggesting that the normal variation of α1 and α2 in rats could also be associated with ECM composition variations. Corroborating this hypothesis, stretching strain in vitro upregulated cartilaginous collagen II and aggrecan expression, as well as α2 and α5 (fibronectin-binding) integrin subunits [14]. Similar observation was made for the β1 integrin subunit, in vivo [21]. We have shown previously that this mandibular propulsive appliance stimulates the synthesis of IGF-I and IGF-II in the condylar cartilage, closely related to an increase in PCNA labeling [7]. After 9 days of treatment, for example, the expression of IGF-I and IGF-II was increased in the cartilage, which is coincident with the increased expression of α1 observed in the present study. Even in control animals, the expression of IGFs was slightly higher during puberty [7]. It is possible that IGFs affected α1 expression in the condylar cartilage in our study, and this hypothesis deserves further investigation. Alternatively, α1-containing integrins, functioning as mechanoreceptors, might activate signal transduction pathways that regulate gene expression, leading to an increase in IGFs production. In human articular chondrocytes, for example, the fibronectin receptor α5β1 functions as a mechanoreceptor. After mechanical stimulation, there was activation of a signal cascade involving stretch-activated ion channels, the actin cytoskeleton and the tyrosine phosphorylation of several components of focal adhesions. Subsequently, there was secretion of interleukin-4, which acted in an autocrine manner to regulate several cell functions and gene expression [20]. In summary, we have shown that integrin subunits α1 and α2 are expressed in the rat condylar cartilage during normal growth, with varying distribution according to the animal s age. A mandibular propulsive appliance, probably acting as an intermittent force, altered their expression and distribution, suggesting that this modulation may be part of the appliance s effect on a molecular level. The understanding of how forces exerted by orthopedic appliances modulate chondrocyte function is very important to improve our knowledge about this widely employed therapy. Acknowledgements The authors are grateful to Emília Ribeiro for the excellent technical support. This study was supported by the São Paulo State Research Foundation (FAPESP), grants # 97/ and 01/ Mara R. Marques is the recipient of a scholarship from FAPESP (02/ ). References [1] M. Bouvier and M.L. Zimny, Effects of loads on surface morphology of the condylar cartilage of the mandible in rats, Acta Anat. 129 (1997),
10 302 M.R. Marques et al. / Orthopedic therapy modulates integrins in condylar cartilage [2] J.P. Charlier, A. Petrovic and J. Herrmann-Stutzmann, Effects of mandibular hyperpropulsion on the prechondroblastic zone of young rat condyle, Am. J. Orthod. 55 (1969), [3] J.C. Copray and W. Jansen, Cyclic nucleotides and growth regulation of the mandibular condylar cartilage of the rat in vitro, Archs. Oral Biol. 30 (1985), [4] J.C. Copray, H.W. Jansen and H.S. Duterloo, An in-vitro system for studying the effect of variable compressive forces on the mandibular condylar cartilage of the rat, Arch. Oral Biol. 30 (1985), [5] E.H. Danen, Integrins: regulators of tissue function and cancer progression, Curr. Pharm. Des. 11 (2005), [6] A.J. Grodzinsky, M.E. Leventston, J. Moonsoo and E.H. Frank, Cartilage tissue remodeling in response to mechanical forces, Annu. Rev. Biomed. Eng. 02 (2000), [7] D. Hajjar, M.F. Santos and E.T. Kimura, Propulsive appliance stimulates the synthesis of insulin-like growth factors I and II in the mandibular condylar cartilage of young rats, Arch. Oral Biol. 48 (2003), [8] G. Hausler, M. Helmreich, S. Marlovits and M. Egerbacher, Integrins and extracellular matrix proteins in the human childhood and adolescent growth plate, Calcif. Tissue Int. 71 (2002), [9] J. Iqbal and M. Zaidi, Molecular regulation of mechanotransduction, Biochem. Biophys. Res. Commun. 328 (2005), , Review. [10] H. Ishibashi, Y. Takenoshita, K. Ishibashi and M. Oka, Expression of extracellular matrix in human mandibular condyle, Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 81 (1996), [11] S.J. Kim, E.J. Kim, Y.H. Kim, S.B. Hahn and J.W. Lee, The modulation of integrin expression by the extracellular matrix in articular chondrocytes, Yonsei Med. J. 44 (2003), [12] W. Knudson and R.F. Loeser, CD44 and integrin matrix receptors participate in cartilage homeostasis, Cell Mol. Life Sci. 59 (2002), [13] R.F. Loeser, Chondrocyte integrin expression and function, Biorheology 37 (2002), [14] K. Lahiji, A. Polotsky, D.S. Hungerford and C.G. Frondoza, Cyclic strain stimulates proliferative capacity, alpha2 and alpha5 integrin, gene marker expression by human articular chondrocytes propagated on flexible silicone membranes, In Vitro Cell Dev. Biol. Anim. 40 (2004), [15] S.J. Millward-Sadler and D.M. Salter, Integrin-dependent signal cascades in chondrocytes mechanotransduction, Ann. Biom. Eng. 32 (2004), [16] I. Mizoguchi, I. Takahashi, Y. Sasano, M. Kagayama, Y. Kuboki and H. Mitani, Localization of types I, II and X collagen and osteocalcin in intramembranous, endochondral and chondroid bone of rats, Anat. Embryol. 195 (1997), [17] H. Nakai, A. Niimi and M. Ueda, The influence of compressive loading on growth of cartilage of the mandibular condyle in vitro, Arch. Oral Biol. 43 (1998), [18] K. Ostergaard, D.M. Salter, J. Petersen, K. Bendtzen, J. Hvolris and C.B. Andersen, Expression of alpha and beta subunits of the integrin superfamily in articular cartilage from macroscopically normal and osteoarthritic human femoral heads, Ann. Rheum. Dis. 57 (1998), [19] A.G. Petrovic, J. Stutzmann and C. Oudet, Control processes in the postnatal growth of the condylar cartilage of the mandible, in: Determinants of Mandibular form and Growth, J.A. McNamara, Jr., ed., Center for Human Growth and Development, Michigan, 1975, pp [20] D.M. Salter, S.J. Millward-Sadler, G. Nuki and M.O. Wright, Integrin interleukin-4 mechanotransduction pathways in human chondrocytes, Clin. Orthop. Relat. Res. 391(Suppl.) (2001), 49 60, Review. [21] I. Takahashi, K. Onodera, Y. Sasano, I. Mizoguchi, J.W. Bae, H. Mitani, M. Kagayama and H. Mitani, Effect of stretching on gene expression of beta1 integrin and focal adhesion kinase and on chondrogenesis through cell extracellular matrix interactions, Eur. J. Cell Biol. 82 (2003), [22] T. Takano-Yamamoto, S. Soma, K. Nakagawa, Y. Kobayashi, M. Kawakami and M. Sakuda, Comparison of the effects of hydrostatic compressive force on glycosaminoglycan synthesis and proliferation in rabbit chondrocytes from mandibular condylar cartilage, nasal septum, and spheno-occipital synchondrosis in vitro, Am. J. Orthod. Dentofacial Orthop. 99 (1991), [23] M. Teramoto, S. Kaneko, S. Shibata, M. Yanagishita and K. Soma, Effect of compressive forces on extracellular matrix in rat mandibular condylar cartilage, J. Bone Miner. Metab. 21 (2003), [24] V. Visnapuu, T. Peltomaki, K. Isotupa, T. Kantomaa and H. Helenius, Distribution and characterization of proliferative cells in the rat mandibular condyle during growth, Eur. J. Orthod. 22 (2000),
Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage
 Original Article Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage Tanapan Wattanachai a ; Ikuo Yonemitsu b ; Sawa Kaneko c ; Kunimichi Soma d
Original Article Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage Tanapan Wattanachai a ; Ikuo Yonemitsu b ; Sawa Kaneko c ; Kunimichi Soma d
Cartilage. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study in the rat
 European Journal of Orthodontics 18 (1996) 3-9 O 1996 European Orthodontic Society Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study
European Journal of Orthodontics 18 (1996) 3-9 O 1996 European Orthodontic Society Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study
Original Article. An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle. Atith Manopinivate, Sawa Kaneko and Kunimichi Soma
 J Med Dent Sci 2006; 53: 67 74 Original Article An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle Atith Manopinivate, Sawa Kaneko and Kunimichi Soma Orthodontic Science,
J Med Dent Sci 2006; 53: 67 74 Original Article An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle Atith Manopinivate, Sawa Kaneko and Kunimichi Soma Orthodontic Science,
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
o~ r;'c' - OSTEOARTHRITIS
 Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Regulation of the skeletal mass through the life span
 Regulation of the skeletal mass through the life span Functions of the skeletal system Mechanical protection skull Movement leverage for muscles Mineral metabolism calcium store Erythropoiesis red blood
Regulation of the skeletal mass through the life span Functions of the skeletal system Mechanical protection skull Movement leverage for muscles Mineral metabolism calcium store Erythropoiesis red blood
Effects of Occlusal Change on the Histology of the Mandibular Condyle Cartilage of Rats. Xinyun Liu. B.D.S., Sun Yat-sen University, 2016
 Effects of Occlusal Change on the Histology of the Mandibular Condyle Cartilage of Rats by Xinyun Liu B.D.S., Sun Yat-sen University, 2016 Submitted to the Graduate Faculty of the School of Dental Medicine
Effects of Occlusal Change on the Histology of the Mandibular Condyle Cartilage of Rats by Xinyun Liu B.D.S., Sun Yat-sen University, 2016 Submitted to the Graduate Faculty of the School of Dental Medicine
PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION
 A L L O S O U R C E PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION Ryan Delaney MS; Carolyn Barrett BS, MBA; Peter Stevens PhD, MBA AlloSource,
A L L O S O U R C E PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION Ryan Delaney MS; Carolyn Barrett BS, MBA; Peter Stevens PhD, MBA AlloSource,
EXTRACELLULAR MATRIX (pp 9-17)
 EXTRACELLULAR MATRIX (pp 9-17) Extracellular Matrix (ECM) Apart from specific cells, tissues contain matrix of macromolecules in the extracellular space- Extracellular Matrix. ECM is secreted by cells
EXTRACELLULAR MATRIX (pp 9-17) Extracellular Matrix (ECM) Apart from specific cells, tissues contain matrix of macromolecules in the extracellular space- Extracellular Matrix. ECM is secreted by cells
Cell Walls, the Extracellular Matrix, and Cell Interactions (part 1)
 14 Cell Walls, the Extracellular Matrix, and Cell Interactions (part 1) Introduction Many cells are embedded in an extracellular matrix which is consist of insoluble secreted macromolecules. Cells of bacteria,
14 Cell Walls, the Extracellular Matrix, and Cell Interactions (part 1) Introduction Many cells are embedded in an extracellular matrix which is consist of insoluble secreted macromolecules. Cells of bacteria,
Changes in the temporomandibular joint after mandibular lengthening with different rates of distraction
 Shujuan Zou, DDS, MS Department of Orthodontics Jing Hu, DDS, MS, PhD Dazhang Wang, DDS, FICD Jihua Li, DDS, MS Zhenglong Tang, DDS, MS Department of Oral and Maxillofacial Surgery Huaxi School of Stomatology
Shujuan Zou, DDS, MS Department of Orthodontics Jing Hu, DDS, MS, PhD Dazhang Wang, DDS, FICD Jihua Li, DDS, MS Zhenglong Tang, DDS, MS Department of Oral and Maxillofacial Surgery Huaxi School of Stomatology
The Position of Anatomical Porion in Different Skeletal Relationships. Tarek. EL-Bialy* Ali. H. Hassan**
 The Position of Anatomical Porion in Different Skeletal Relationships Tarek. EL-Bialy* Ali. H. Hassan** Abstract Previous research has shown that the position of glenoid fossa differs in different skeletal
The Position of Anatomical Porion in Different Skeletal Relationships Tarek. EL-Bialy* Ali. H. Hassan** Abstract Previous research has shown that the position of glenoid fossa differs in different skeletal
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
Bone. Development. Tim Arnett. University College London. Department of Anatomy and Developmental Biology
 Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
CARTILAGE. Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015
 CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
Nanomechanical Symptoms in Cartilage Precede Histological Osteoarthritis Signs after the Destabilization of Medial Meniscus in Mice
 Nanomechanical Symptoms in Cartilage Precede Histological Osteoarthritis Signs after the Destabilization of Medial Meniscus in Mice Basak Doyran 1, Wei Tong 2, Qing Li 1, Haoruo Jia 2, Xianrong Zhang 3,
Nanomechanical Symptoms in Cartilage Precede Histological Osteoarthritis Signs after the Destabilization of Medial Meniscus in Mice Basak Doyran 1, Wei Tong 2, Qing Li 1, Haoruo Jia 2, Xianrong Zhang 3,
ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN
 ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN 1982 Effects of a pulsed electromagnetic field on a mixed chondroplastic
ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN 1982 Effects of a pulsed electromagnetic field on a mixed chondroplastic
Biology. Dr. Khalida Ibrahim
 Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Immunostaining was performed on tumor biopsy samples arranged in a tissue-microarray format or on
 Supplemental Methods Immunohistochemical Analyses Immunostaining was performed on tumor biopsy samples arranged in a tissue-microarray format or on prostatectomy sections obtained post-study. Briefly,
Supplemental Methods Immunohistochemical Analyses Immunostaining was performed on tumor biopsy samples arranged in a tissue-microarray format or on prostatectomy sections obtained post-study. Briefly,
Mandibular condyle: structure properties and growth regulation. Citation Journal of Oral Science and Health, 2014, v. 1 n. 1, p.
 Title Mandibular condyle: structure properties and growth regulation Author(s) Ren, C; Yang, Y Citation, 2014, v. 1 n. 1, p. 1-6 Issued Date 2014 URL http://hdl.handle.net/10722/200447 Rights This work
Title Mandibular condyle: structure properties and growth regulation Author(s) Ren, C; Yang, Y Citation, 2014, v. 1 n. 1, p. 1-6 Issued Date 2014 URL http://hdl.handle.net/10722/200447 Rights This work
Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit)
 Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit) Cat. No. 280560-N INTRODUCTION Glycosaminoglycans (GAGs) are a major component of the extracellular matrix (ECM)
Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit) Cat. No. 280560-N INTRODUCTION Glycosaminoglycans (GAGs) are a major component of the extracellular matrix (ECM)
Osteochondral regeneration. Getting to the core of the problem.
 Osteochondral regeneration. Getting to the core of the problem. TM TM Bio-mimetic, biointegratable and resorbable Flexible and easy to shape Straightforward one-step procedure Promotes a guided osteo-chondral
Osteochondral regeneration. Getting to the core of the problem. TM TM Bio-mimetic, biointegratable and resorbable Flexible and easy to shape Straightforward one-step procedure Promotes a guided osteo-chondral
Why the dog? Analogy of the anatomy
 Why the dog? Analogy of the anatomy Surgically Induced canine OA models: Anterior (cranial) cruciate ligament transection model Pond MJ, Nuki G. Ann Rheum Dis 1973 (and > 100 others) Meniscal disruption
Why the dog? Analogy of the anatomy Surgically Induced canine OA models: Anterior (cranial) cruciate ligament transection model Pond MJ, Nuki G. Ann Rheum Dis 1973 (and > 100 others) Meniscal disruption
Disclosures: C.B. Raub: None. B.C. Hansen: None. T. Yamaguchi: None. M.M. Temple-Wong: None. K. Masuda: None. R.L. Sah: None.
 En Face Microscopy of Rabbit Knee Articular Cartilage Following Anterior Cruciate Ligament Transection Reveals Early Matrix Damage, Chondrocyte Loss and Cloning Christopher B. Raub, PhD, Bradley C. Hansen,
En Face Microscopy of Rabbit Knee Articular Cartilage Following Anterior Cruciate Ligament Transection Reveals Early Matrix Damage, Chondrocyte Loss and Cloning Christopher B. Raub, PhD, Bradley C. Hansen,
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
contains an antiangiogenesis factor
 CARTILAGE & BONE Cartilage and Bone objectives Student must learn :. What is the meaning of cartilage, and their function, location in human body.. To distinguish the 3 types of cartilage. And their cells,
CARTILAGE & BONE Cartilage and Bone objectives Student must learn :. What is the meaning of cartilage, and their function, location in human body.. To distinguish the 3 types of cartilage. And their cells,
Mesial Step Class I or Class III Dependent upon extent of step seen clinically and patient s growth pattern Refer for early evaluation (by 8 years)
 Orthodontics and Dentofacial Development Overview Development of Dentition Treatment Retention and Relapse Growth of Naso-Maxillary Complex Develops postnatally entirely by intramenbranous ossification
Orthodontics and Dentofacial Development Overview Development of Dentition Treatment Retention and Relapse Growth of Naso-Maxillary Complex Develops postnatally entirely by intramenbranous ossification
Dr. Heba Kalbouneh. Saba Alfayoumi. Heba Kalbouneh
 11 Dr. Heba Kalbouneh Saba Alfayoumi Heba Kalbouneh 2- Bone Bone tissue is also classified into primary bone and secondary bone. In the beginning, the first bone that is deposited by the osteoblasts is
11 Dr. Heba Kalbouneh Saba Alfayoumi Heba Kalbouneh 2- Bone Bone tissue is also classified into primary bone and secondary bone. In the beginning, the first bone that is deposited by the osteoblasts is
2008 JCO, Inc. May not be distributed without permission. Correction of Asymmetry with a Mandibular Propulsion Appliance
 2008 JCO, Inc. May not be distributed without permission. www.jco-online.com CASE REPORT Correction of Asymmetry with a Mandibular Propulsion Appliance JOSÉ AUGUSTO MENDES MIGUEL, DDS, MSC, PHD GUSTAVO
2008 JCO, Inc. May not be distributed without permission. www.jco-online.com CASE REPORT Correction of Asymmetry with a Mandibular Propulsion Appliance JOSÉ AUGUSTO MENDES MIGUEL, DDS, MSC, PHD GUSTAVO
p=0.02 ). Discussion: These results, which expand upon a recently developed chemically defined medium [3], demonstrate that contrary to long-term
![p=0.02 ). Discussion: These results, which expand upon a recently developed chemically defined medium [3], demonstrate that contrary to long-term p=0.02 ). Discussion: These results, which expand upon a recently developed chemically defined medium [3], demonstrate that contrary to long-term](/thumbs/84/90879891.jpg) Synovial Fluid and Physiologic Levels of Cortisol, Insulin, and Glucose in Media Maintain the Homeostasis of Immature Bovine Cartilage Explants over Long Term Culture Michael B. Albro, PhD, Krista M. Durney,
Synovial Fluid and Physiologic Levels of Cortisol, Insulin, and Glucose in Media Maintain the Homeostasis of Immature Bovine Cartilage Explants over Long Term Culture Michael B. Albro, PhD, Krista M. Durney,
Skeletal Tissues. Skeletal tissues. Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs.
 Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets
 Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle
 Okajimas Folia Anat. Jpn., 88(2): 49 55, August, 2011 An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle By Keiichi Kenzaki 1, Kohzo Tsuchikawa
Okajimas Folia Anat. Jpn., 88(2): 49 55, August, 2011 An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle By Keiichi Kenzaki 1, Kohzo Tsuchikawa
Cell Cell Communication
 IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
Which compound is reponsible for the viscous character of the ground substance?
 1 2 Which type of collagen forms the coarse collagen fibres in dense regular and irregular connective tissues? Which compound is reponsible for the viscous character of the ground substance? 3 Which class
1 2 Which type of collagen forms the coarse collagen fibres in dense regular and irregular connective tissues? Which compound is reponsible for the viscous character of the ground substance? 3 Which class
Prof. Steven S. Saliterman. Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ The meniscus is a fibrocartilaginous tissue primarily of type 1 collagen fibers. (In contrast to hyaline cartilage
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ The meniscus is a fibrocartilaginous tissue primarily of type 1 collagen fibers. (In contrast to hyaline cartilage
Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall
 Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
The Correlation Between Neovascularization and Bone Formation in the Condyle During Forward Mandibular Positioning
 Original Article The Correlation Between Neovascularization and Bone Formation in the Condyle During Forward Mandibular Positioning A. B. M. Rabie, MS, Cert Ortho, PhD, FHKAM, FCDSHK a ; F. Y. C. Leung,
Original Article The Correlation Between Neovascularization and Bone Formation in the Condyle During Forward Mandibular Positioning A. B. M. Rabie, MS, Cert Ortho, PhD, FHKAM, FCDSHK a ; F. Y. C. Leung,
The Extracellular Matrices Part II. 2. Elastin fibers. 3. Proteoglycans (PG) and glycosaminoglycans (GAG). 4. Cell-adhesion molecules (CAM).
 The Extracellular Matrices Part II. 2. Elastin fibers. 3. Proteoglycans (PG) and glycosaminoglycans (GAG). 4. Cell-adhesion molecules (CAM). 1 Elastin fibers A network of randomly coiled macromolecules.
The Extracellular Matrices Part II. 2. Elastin fibers. 3. Proteoglycans (PG) and glycosaminoglycans (GAG). 4. Cell-adhesion molecules (CAM). 1 Elastin fibers A network of randomly coiled macromolecules.
Cell Cell Communication
 IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Chapter 15: Signal transduction
 Chapter 15: Signal transduction Know the terminology: Enzyme-linked receptor, G-protein linked receptor, nuclear hormone receptor, G-protein, adaptor protein, scaffolding protein, SH2 domain, MAPK, Ras,
Chapter 15: Signal transduction Know the terminology: Enzyme-linked receptor, G-protein linked receptor, nuclear hormone receptor, G-protein, adaptor protein, scaffolding protein, SH2 domain, MAPK, Ras,
CHONDROTOXICITY OF LOCAL ANESTHETIC
 CHONDROTOXICITY OF LOCAL ANESTHETIC Sport Med 2017 Jas Chahal MD FRCSC MSc MBA University of Toronto NO DISCLOSURES Objectives To understand the clinical presentation and pathogenesis of chondrolysis Differentiate
CHONDROTOXICITY OF LOCAL ANESTHETIC Sport Med 2017 Jas Chahal MD FRCSC MSc MBA University of Toronto NO DISCLOSURES Objectives To understand the clinical presentation and pathogenesis of chondrolysis Differentiate
Disclosures: A.G. Bajpayee: None. A.M. Scheu: None. R.M. Porter: None. A.J. Grodzinsky: None.
 Avidin as a Carrier for Drug Delivery into Cartilage: Electrostatic Interactions Enable Rapid Penetration, Enhanced Uptake, and Retention in Rat Knee Joints Ambika Goel Bajpayee 1, Alfredo M. Scheu 2,
Avidin as a Carrier for Drug Delivery into Cartilage: Electrostatic Interactions Enable Rapid Penetration, Enhanced Uptake, and Retention in Rat Knee Joints Ambika Goel Bajpayee 1, Alfredo M. Scheu 2,
Introduction to Biomedical Engineering
 Introduction to Biomedical Engineering FW 16/17, AUT Biomechanics of tendons and ligaments G. Rouhi Biomechanics of tendons and ligaments Biomechanics of soft tissues The major soft tissues in musculoskeletal
Introduction to Biomedical Engineering FW 16/17, AUT Biomechanics of tendons and ligaments G. Rouhi Biomechanics of tendons and ligaments Biomechanics of soft tissues The major soft tissues in musculoskeletal
Bone Tissue- Chapter 5 5-1
 Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
BIOMATERIALS-TISSUE TISSUE INTERACTIONS: INTRODUCTION
 Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J BIOMATERIALS-TISSUE TISSUE INTERACTIONS: INTRODUCTION M.
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J BIOMATERIALS-TISSUE TISSUE INTERACTIONS: INTRODUCTION M.
Cell Communication. Cell Communication. Communication between cells requires: ligand: the signaling molecule
 Cell Communication Cell Communication Communication between cells requires: ligand: the signaling molecule receptor protein: the molecule to which the ligand binds (may be on the plasma membrane or within
Cell Communication Cell Communication Communication between cells requires: ligand: the signaling molecule receptor protein: the molecule to which the ligand binds (may be on the plasma membrane or within
FORMATION OF BONE. Intramembranous Ossification. Bone-Lec-10-Prof.Dr.Adnan Albideri
 FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
Sheets 16&17. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Sheets 16&17 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ossification (formation of bone) - Osteoblasts are responsible for producing the extracellular matrix of the bone and these osteoblasts
Sheets 16&17 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ossification (formation of bone) - Osteoblasts are responsible for producing the extracellular matrix of the bone and these osteoblasts
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
Bones. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
 Bones Osteocytes : Are responsible for maintenance of bones Present in lacunae, and send processes. Unable to divide. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
Bones Osteocytes : Are responsible for maintenance of bones Present in lacunae, and send processes. Unable to divide. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
1/10/2013. Topic 10: Form and Function (COM) Newton s Laws of Motion. Newton s Laws of Motion. What are? Forces
 /0/203 Topic 0: Form and Function (COM) Forces exerted on vertebrate bodies How do Newton s Laws apply to vertebrate form and function? How do we study forces in functional morphology? What are Ground
/0/203 Topic 0: Form and Function (COM) Forces exerted on vertebrate bodies How do Newton s Laws apply to vertebrate form and function? How do we study forces in functional morphology? What are Ground
Supplementary Figure 1.
 Supplementary Figure 1. Increased expression of cell cycle pathway genes in insulin + Glut2 low cells of STZ-induced diabetic islets. A) random blood glucose measuers of STZ and vehicle treated MIP-GFP
Supplementary Figure 1. Increased expression of cell cycle pathway genes in insulin + Glut2 low cells of STZ-induced diabetic islets. A) random blood glucose measuers of STZ and vehicle treated MIP-GFP
Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location
 Original paper Dental Materials Journal 21 (1): 32-43, 2002 Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location Toshiko INOUE, Hidekazu TAKAHASHI and Fumio
Original paper Dental Materials Journal 21 (1): 32-43, 2002 Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location Toshiko INOUE, Hidekazu TAKAHASHI and Fumio
Spatial and temporal expression of Smad signaling members during the development of mandibular condylar cartilage
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 14: 4967-4971, 2017 Spatial and temporal expression of Smad signaling members during the development of mandibular condylar cartilage DI XIAO 1, RU WANG 1, JING HU
EXPERIMENTAL AND THERAPEUTIC MEDICINE 14: 4967-4971, 2017 Spatial and temporal expression of Smad signaling members during the development of mandibular condylar cartilage DI XIAO 1, RU WANG 1, JING HU
Cephalometric Analysis
 Cephalometric Analysis of Maxillary and Mandibular Growth and Dento-Alveolar Change Part III In two previous articles in the PCSO Bulletin s Faculty Files, we discussed the benefits and limitations of
Cephalometric Analysis of Maxillary and Mandibular Growth and Dento-Alveolar Change Part III In two previous articles in the PCSO Bulletin s Faculty Files, we discussed the benefits and limitations of
Evaluation of directed and random motility in microslides Assessment of leukocyte adhesion in flow chambers
 Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
CHAPTER 6 LECTURE OUTLINE
 CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
2000 European Orthodontic Society. European Journal of Orthodontics 22 (2000)
 European Journal of Orthodontics 22 (2000) 649 656 2000 European Orthodontic Society Computer tomographic and radiographic changes in the temporomandibular joints of two young adults with occlusal asymmetry,
European Journal of Orthodontics 22 (2000) 649 656 2000 European Orthodontic Society Computer tomographic and radiographic changes in the temporomandibular joints of two young adults with occlusal asymmetry,
Thanks to: Signal Transduction. BCB 570 "Signal Transduction" 4/8/08. Drena Dobbs, ISU 1. An Aging Biologist s. One Biologist s Perspective
 BCB 570 "" Thanks to: One Biologist s Perspective Drena Dobbs BCB & GDCB Iowa State University Howard Booth Biology Eastern Michigan University for Slides modified from his lecture Cell-Cell Communication
BCB 570 "" Thanks to: One Biologist s Perspective Drena Dobbs BCB & GDCB Iowa State University Howard Booth Biology Eastern Michigan University for Slides modified from his lecture Cell-Cell Communication
Different Non Surgical Treatment Modalities for Class III Malocclusion
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 9, Issue 6 (Sep.- Oct. 2013), PP 48-52 Different Non Surgical Treatment Modalities for Class III Malocclusion
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 9, Issue 6 (Sep.- Oct. 2013), PP 48-52 Different Non Surgical Treatment Modalities for Class III Malocclusion
Chapter 6: Skeletal System: Bones and Bone Tissue
 Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
Chapter 6 Bones and Bone Tissue Chapter Outline
 Chapter 6 Bones and Bone Tissue Chapter Outline Module 6.1: Introduction to Bones as Organs (Figures 6.1, 6.2, 6.3, 6.4) A. The skeletal system includes the bones, joints, and their associated supporting
Chapter 6 Bones and Bone Tissue Chapter Outline Module 6.1: Introduction to Bones as Organs (Figures 6.1, 6.2, 6.3, 6.4) A. The skeletal system includes the bones, joints, and their associated supporting
DEBRIDEMENT: ANATOMY and PHYSIOLOGY. Professor Donald G. MacLellan Executive Director Health Education & Management Innovations
 DEBRIDEMENT: ANATOMY and PHYSIOLOGY Professor Donald G. MacLellan Executive Director Health Education & Management Innovations ANATOMY and PHYSIOLOGY Epidermal Layers ECM Structure Dermis Structure Skin
DEBRIDEMENT: ANATOMY and PHYSIOLOGY Professor Donald G. MacLellan Executive Director Health Education & Management Innovations ANATOMY and PHYSIOLOGY Epidermal Layers ECM Structure Dermis Structure Skin
Chp. 6: Bones and Skeletal Tissue Student Worksheet. 1. The skeletal system is composed of bones,,, and.
 Chp. 6: Bones and Skeletal Tissue Student Worksheet 1. The skeletal system is composed of bones,,, and. 2. What are 5 functions of the skeletal system? 3. Besides osseous tissue (connective tissue with
Chp. 6: Bones and Skeletal Tissue Student Worksheet 1. The skeletal system is composed of bones,,, and. 2. What are 5 functions of the skeletal system? 3. Besides osseous tissue (connective tissue with
Blood. Hematopoietic Tissue
 Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
Growth and repair: Cartilage is a vascular tissues that receives nutrients by diffusion through its matrix, cartilage grow by 2 mechanisms:
 Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Cartilage Cartilage is a special form of connective tissue; it has the same origin of connective tissue (embryonic mesenchyme). It contains cells
Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Cartilage Cartilage is a special form of connective tissue; it has the same origin of connective tissue (embryonic mesenchyme). It contains cells
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS
 OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
Dr.Heba Kalbouneh. Ragad Alhawi. Dr.Heba Kalbouneh
 9 Dr.Heba Kalbouneh Ragad Alhawi Dr.Heba Kalbouneh "Cartilage" Cartilage is a special form of connective tissue; it has the same origin of connective tissue (embryonic mesenchyme). It contains cells and
9 Dr.Heba Kalbouneh Ragad Alhawi Dr.Heba Kalbouneh "Cartilage" Cartilage is a special form of connective tissue; it has the same origin of connective tissue (embryonic mesenchyme). It contains cells and
Incisor disocclusion in rats affects mandibular condylar cartilage at the cellular level
 Archives of Oral Biology (2004) 49, 393 400 Incisor disocclusion in rats affects mandibular condylar cartilage at the cellular level G.O. Ramirez-Yañez*, T.J. Daley, A.L. Symons, W.G. Young Department
Archives of Oral Biology (2004) 49, 393 400 Incisor disocclusion in rats affects mandibular condylar cartilage at the cellular level G.O. Ramirez-Yañez*, T.J. Daley, A.L. Symons, W.G. Young Department
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
Artigo Original / Original Article
 Artigo Original / Original Article IJD ISSN:1806-146X Mandibular condyle morphology on panoramic radiographs of asymptomatic temporomandibular joints Christiano Oliveira 1 Renata Tarnoschi Bernardo 2 Ana
Artigo Original / Original Article IJD ISSN:1806-146X Mandibular condyle morphology on panoramic radiographs of asymptomatic temporomandibular joints Christiano Oliveira 1 Renata Tarnoschi Bernardo 2 Ana
cartilage of the rat
 J. Anat. (1989), 162, pp. 43-51 43 With 4 figures Printed in Great Britain Presence of osteonectin/sparc in mandibular condylar cartilage of the rat J. C. V. M. COPRAY, P. M. JOHNSON*, J. D. DECKER* AND
J. Anat. (1989), 162, pp. 43-51 43 With 4 figures Printed in Great Britain Presence of osteonectin/sparc in mandibular condylar cartilage of the rat J. C. V. M. COPRAY, P. M. JOHNSON*, J. D. DECKER* AND
Muscles of mastication [part 1]
![Muscles of mastication [part 1] Muscles of mastication [part 1]](/thumbs/76/73586850.jpg) Muscles of mastication [part 1] In this lecture well have the muscles of mastication, neuromuscular function, and its relationship to the occlusion morphology. The fourth determinant of occlusion is the
Muscles of mastication [part 1] In this lecture well have the muscles of mastication, neuromuscular function, and its relationship to the occlusion morphology. The fourth determinant of occlusion is the
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Do Now: 1. Where, specifically, is blood created? Which part of the long bone? 2. Which structures are primarily associated with growth? 3.
 Do Now: 1. Where, specifically, is blood created? Which part of the long bone? 2. Which structures are primarily associated with growth? 3. How could damage to these areas impact bone growth? WRITE AND
Do Now: 1. Where, specifically, is blood created? Which part of the long bone? 2. Which structures are primarily associated with growth? 3. How could damage to these areas impact bone growth? WRITE AND
BONE TISSUE. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student.
 1 CARTILAGE AND BONE The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student. 1. Hyaline cartilage Slide 73 This is a cross section through
1 CARTILAGE AND BONE The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student. 1. Hyaline cartilage Slide 73 This is a cross section through
Essentials in Head and Neck Embryology. Part 3 Development of the head, face, and oral cavity
 Essentials in Head and Neck Embryology Part 3 Development of the head, face, and oral cavity Outline General overview of prenatal development Embryonic period phase 1 Formation of bilaminar disk Formation
Essentials in Head and Neck Embryology Part 3 Development of the head, face, and oral cavity Outline General overview of prenatal development Embryonic period phase 1 Formation of bilaminar disk Formation
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Principles of Genetics and Molecular Biology
 Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
For more information about how to cite these materials visit
 Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
SKELETAL TISSUES CHAPTER 7 INTRODUCTION TO THE SKELETAL SYSTEM TYPES OF BONES
 SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
Temporomandibular Joint Disorders
 Temporomandibular Joint Disorders Introduction Temporomandibular joint disorders, or TMJ disorders, are a group of medical problems related to the jaw joint. TMJ disorders can cause headaches, ear pain,
Temporomandibular Joint Disorders Introduction Temporomandibular joint disorders, or TMJ disorders, are a group of medical problems related to the jaw joint. TMJ disorders can cause headaches, ear pain,
Original Article. Shunichi Shibata 1, Tatsuya Fujimori 2 and Yasuo Yamashita 1
 J Med Dent Sci 2006; 53: 41 50 Original Article An in situ hybridization and histochemical study of development and postnatal changes of mouse mandibular angular cartilage compared with condylar cartilage
J Med Dent Sci 2006; 53: 41 50 Original Article An in situ hybridization and histochemical study of development and postnatal changes of mouse mandibular angular cartilage compared with condylar cartilage
Changes in Pharyngeal Airway Dimensions Due to MARA Treatment in Class II Malocclusions
 University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 5-2013 Changes in Pharyngeal Airway Dimensions Due to MARA Treatment
University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 5-2013 Changes in Pharyngeal Airway Dimensions Due to MARA Treatment
Fig Articular cartilage. Epiphysis. Red bone marrow Epiphyseal line. Marrow cavity. Yellow bone marrow. Periosteum. Nutrient foramen Diaphysis
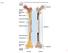 Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
Supplemental Tables and Figures. The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate,
 Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
Temporomandibular Joint Remodeling for the Treatment of Temporomandibular Joint Disorders - A Clinical Case Study
 The Open Rehabilitation Journal, 2009, 2, 43-49 43 Open Access Temporomandibular Joint Remodeling for the Treatment of Temporomandibular Joint Disorders - A Clinical Case Study Rosita Wurgaft 1 and Ricky
The Open Rehabilitation Journal, 2009, 2, 43-49 43 Open Access Temporomandibular Joint Remodeling for the Treatment of Temporomandibular Joint Disorders - A Clinical Case Study Rosita Wurgaft 1 and Ricky
BCM 226 LECTURE SALEMCITY, A.J
 BCM 226 LECTURE SALEMCITY, A.J BIOLOGICAL MEMBRANE Biological membranes are composed of proteins associated with a lipid bilayer matrix. They are the molecular gateway to the cell. Viewed under electron
BCM 226 LECTURE SALEMCITY, A.J BIOLOGICAL MEMBRANE Biological membranes are composed of proteins associated with a lipid bilayer matrix. They are the molecular gateway to the cell. Viewed under electron
Effects of Immobilization on Structure of Cell Layers In Tibial Articular Cartilage
 Effects of Immobilization on Structure of Cell Layers In Tibial Articular Cartilage Effects of Immobilization on Structure of Cell Layers In Tibial Articular Cartilage OGIWARA Yuh* FUJIKAWA Kaoru** OHSAKO
Effects of Immobilization on Structure of Cell Layers In Tibial Articular Cartilage Effects of Immobilization on Structure of Cell Layers In Tibial Articular Cartilage OGIWARA Yuh* FUJIKAWA Kaoru** OHSAKO
Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model
 Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model Averi Leahy, Andrea Foote, Tomoya Uchimura, Li Zeng, PhD. Tufts University, Boston, MA, USA. Disclosures: A. Leahy: None.
Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model Averi Leahy, Andrea Foote, Tomoya Uchimura, Li Zeng, PhD. Tufts University, Boston, MA, USA. Disclosures: A. Leahy: None.
Effects of pulsing electromagnetic fields on cultured cartilage cells.
 Arch Oral Biol. 1993 Jan;38(1):67-74. Autoradiographic study of the effects of pulsed electromagnetic fields on bone and cartilage growth in juvenile rats. Wilmot JJ, Chiego DJ Jr, Carlson DS, Hanks CT,
Arch Oral Biol. 1993 Jan;38(1):67-74. Autoradiographic study of the effects of pulsed electromagnetic fields on bone and cartilage growth in juvenile rats. Wilmot JJ, Chiego DJ Jr, Carlson DS, Hanks CT,
Cartilage. - Cartilage together with long bone form the skeleton and support the body.
 Cartilage - Cartilage is a special type of CT has a firm pliable matrix that can resist mechanical stress, act as a shock absorber. - Cartilage together with long bone form the skeleton and support the
Cartilage - Cartilage is a special type of CT has a firm pliable matrix that can resist mechanical stress, act as a shock absorber. - Cartilage together with long bone form the skeleton and support the
Connective Tissue. Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment.
 Connective Tissue Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment. Connective Tissue Functions Binding and support Protection Insulation Transportation
Connective Tissue Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment. Connective Tissue Functions Binding and support Protection Insulation Transportation
