Original Article. Shunichi Shibata 1, Tatsuya Fujimori 2 and Yasuo Yamashita 1
|
|
|
- Alban Whitehead
- 6 years ago
- Views:
Transcription
1 J Med Dent Sci 2006; 53: Original Article An in situ hybridization and histochemical study of development and postnatal changes of mouse mandibular angular cartilage compared with condylar cartilage Shunichi Shibata 1, Tatsuya Fujimori 2 and Yasuo Yamashita 1 1) Maxillofacial Anatomy, Department of Maxillofacial Biology, Graduate School, Tokyo Medical and Dental University 2) Implantology, Department of Masticatory Function Rehabilitation, Graduate School, Tokyo Medical and Dental University To investigate the origin and postnatal changes of mouse mandibular angular cartilage, in situ hybridization for cartilaginous marker proteins, histochemistry for alkaline phosphatase (ALP) and tartrate-resistant acid phosphatase (TRAP), and bromodeoxyuridine (BrDU) analyses were performed. Chondrocytes of the mandibular angular cartilage were derived from ALP-positive progenitor cells and first detected at embryonic day (E) Newly formed chondrocytes rapidly differentiated into hypertrophic chondrocytes and hypertrophic cell zone rapidly extended in subsequent a few days. During this period, bone sialoprotein mrna was more widely expressed than osteopontin mrna in cartilage. Endochondral bone formation started at E 17.5 with the resorption of the bone collar by osteoclasts. These characteristics were consistent with those of the condylar cartilage, although developmental process was day delayed relative to the condylar cartilage. During the postnatal period, contrast to the condylar cartilage, the angular cartilage constantly decreased in volume with advancing age. Reduction of proliferating activity estimated by BrDU incorporation accounts for this phenomenon. Corresponding Author: Shunichi Shibata Maxillofacial Anatomy, Department of Maxillofacial Biology, Graduate School, Tokyo Medical and Dental University , Yushima, Bunkyo-ku, Tokyo , Japan Tel: Fax: S.Shibata.mfa@tmd.ac.jp Received October 12; Accepted December 2, 2005 We demonstrate new structural features of the mandibular angular cartilage that may contribute to a coming research for the secondary cartilage. Key words: secondary cartilage, angular cartilage, in situ hybridization, alkaline phosphatase Introduction During the growing process of mandible, secondary cartilages are formed in condylar, angular and coronoid processes in human, rats and mice The chondrocytes of the mouse mandibular condylar cartilage develop from alkaline phosphatase (ALP)-positive progenitor cells in the periosteum-like tissue after ossifying mandible has formed 6-9. These progenitor cells then rapidly differentiate into hypertrophic chondrocytes which express mrnas for collagen Types I, II and X simultaneously 8,10,11. The hypertrophic cell zone then rapidly extends in subsequent a few days 7,8,10,12,13 and bone sialoprotein (BSP) mrna is more widely expressed than osteopontin (OPN) mrna during this period 13. The endochondral bone formation of this cartilage starts with the resorption of bone collar at E 16 7,14. During postnatal period, the mandibular condylar cartilage continues to grow and function as articular cartilage of the temporo-mandibular joint and some
2 42 S. SHIBATA, T. FUJIMORI and Y. YAMASHITA J Med Dent Sci immunohistochemical studies related to the postnatal changes have been performed 15,16. Meanwhile, in the mandibular angular cartilage, several histological studies in rats 2,3,17-19, in mice 20 and in human 21, and a few histochemical studies for ALP in rats 3 and in mice 9 have been performed. Although this cartilage is also derived from ALP-positive progenitor cells 9, detail developmental process from viewpoints of in situ hybridization has not been described. First, we hypothesized that developmental process of the fetal angular cartilage is similar to that of the condylar cartilage. Furthermore, although rat angular cartilage disappears in postnatal periods because likely of the lack of articular function 2,18, definite reason for disappearance is still unknown. Second, we hypothesized that reduction of proliferation activity and/or acceleration of cartilage resorption may be important for this phenomenon. To confirm these hypotheses, we investigated development and postnatal changes of the mouse mandibular angular cartilage by in situ hybridization of cartilage matrix proteins, incorporation of bromodeoxyuridine (BrDU) and tartrate-resistant acid phosphatase (TRAP) staining, as comparing with the condylar cartilage. Materials and Methods Tissue preparation All animals were maintained in the animal research center in the Tokyo Medical and Dental University and the procedures conformed to the guidelines determined by the University Animal Care Committee. Research protocols conformed to NIH guidelines as stated in the Principles of Laboratory Animal Care (NIH publication No , revised 1985). Ten fetal ICR mice, embryonic day (E) , and twenty postnatal mice, 7-21 day after birth (d 7-21) were used for this study. BrDU was shot (2 ÒM /10g) to each mouse 2 hrs before killing mice. At each time point, the pregnant and postnatal mice were killed by cervical dislocation under ether anesthetization, after which each fetal mouse was killed by cervical dislocation. The heads were then taken and immersed in 4% paraformaldehyde (0.1 M phosphate buffer, ph 7.4) for 1 day at 4 C or 95% ethanol for 3 d at room temperature. Paraformaldehyde-fixed specimens were decalcified with 10% ethylenediamine tetraacetic acid (EDTA) for 7 days at 4 C then routinely embedded in paraffin. Sections of 5 Òm were cut in the coronal plane, perpendicular to the sagittal plane, and parallel to the long axis of the angular or the condylar process of the mandible. Sections were stained with 0.1% toluidine blue (0.1 M phosphate buffer, ph 7.4) for histological observations. Some specimens were embedded in OCT compound (Miles, Elkhart, IN) and frozen with liquid nitrogen in preparation for cryosections. Whole skeletal staining was performed according to the methods of McLeod 22, ethanol-fixed specimens were stained with alcian blue and alizarin red S, then immersed in 1% KOH to dissolve soft tissues. In situ hybridization and immunohistochemistry for BrDU Digoxigenin-labeled crna probes for aggrecan, collagen Types II and X, BSP and OPN were used in previous studies 6,10,13. In situ hybridization using a Nucleic Acid Detection Kit (Roche Diagnostics, Mannheim, Germany) was performed as previously described 13,23. Sections were examined after counterstaining with nuclear fast red. Sense probes were reacted as negative controls. Some in situ hybridization-treated sections were further utilized for immunohistochemistry for BrDU using a HISTOFINE SAB kit (Nichirei, Tokyo, Japan) and a M. O. M kit (Vector Laboratories, Burlingame, CA). After reacting with NBT/BCIP in the Nucleic Acid Detection Kit, sections were then immersed in methanol containing 1% hydrogen peroxide to block endogenous peroxidase activity and further immersed in mouse IgG blocking reagent in the M. O. M kit. Sections were then reacted with anti-brdu mouse monoclonal antibody (Roche Diagnostics, Mannheim, Germany) diluted (1:100) with phosphate buffered saline containing 1% bovine serum albumin The streptavidin-biotin method was then applied to the sections using the HISTOFINE SAB kit, as previously described 8,13. Finally, sections were treated with 3- amino-9-ethylcarbazole (Nichirei, Tokyo, Japan) to reveal any reaction. Normal mouse IgG was reacted instead of the primary antibody as negative controls. Enzyme histochemistry TRAP activity was detected by the hexasotized pararosaniline method described by Lewinson and Silbermann 24. ALP activity was detected by routine azo dye method. Cryosections (8 Òm) made by Cryostat 1720 (Leitz, Wetzlar, Germany) were incubated in a solution with a mixture of Naphtol AS-MX sodium salt (Sigma, St Louis, MO) as a substrate and Fast-blue RR salt (Sigma) diluted in 0.1M Tris-HCl buffer (ph 8.5) at room temperature for 30 min. As negative controls, sec-
3 FETAL MOUSE ANGULAR CARTILAGE 43 tions were incubated in working solution without substrate or containing 25mM levamisole. Histomorphometry Sections cut through the middle plane of the angular or the condylar process were used for histomorphometric analyses and four sets of three serial sections for in situ hybridization were used at each stage. According to Luder et al. 25, the angular and the condylar cartilages were classified as follows: fibrous cell zone (also known as articular zone), polymorphic cell zone, flattened cell zone, upper hypertrophic cell zone, lower hypertrophic cell zone. Total cartilaginous area in this study contained all these zones between lateral and medial bone collars. Especially, the polymorphic cell zone between lateral and medial bone collars was termed as chondroproliferation area. In addition, the zone where chondrocyte lacunae were open was regarded as erosion zone. The length of erosion zone was measured by N.I.H. Image 1.61 in low magnified sections and termed as erosion zone length (Figure 1). The area of the mrna-positive regions for Type II collagen (marker for mature chondrocytes), Type X collagen (marker for hypertrophic chondrocytes) and OPN (marker for deep layer of hypertrophic chondrocytes) were measured using NIH Image The mrna-positive region was easily distinguished from the negative region by color difference. Since there were some overlapping areas, the sum of each area occasionally exceeded the total cartilaginous area. The number of BrDU-positive cells in the chondroproliferation area was counted and normalized number by area was estimated. Only a few chondrocytes in the flattened cell layer were positive for BrDU as previously described 25, but were not counted in this study. The number of TRAP-positive, multinucleated chondrocytes as discriminated from osteoclasts 26 was counted and normalized number by erosion zone length was estimated. Results Whole skeletal staining The mandibular angular cartilage and the condylar cartilage were clearly stained with alcial blue at E 18.5 (Fig. 2). Lines in the figure show the direction of sections for further study. Development of the mandibular angular cartilage At E 14.5, the anlage of the angular cartilage was recognized as mesenchymal cell condensation adjacent to the ossifying mandible (Fig. 3a). This anlage never expressed mrnas for Type II collagen (Fig. 3b), Type X, collagen (Fig.3c), OPN (Fig. 3d) and BSP (Fig. 3e). Cells in the ossifying mandible expressed OPN and BSP mrna (Fig. 3d and e). Cartilage was first recognized at E 15.5 within the anlage (Fig. 3f) and newly formed chondrocytes simultaneously expressed mrnas for these molecules (Figs. 3g-j). At E Fig. 1. Classification of zones in the angular and the condylar cartilages. A: fibrous cell zone, B: polymorphic cell zone, C: flattened cell zone, D: upper hypertrophic cell zone, E: lower hypertrophic cell zone. F: erosion zone. Total cartilaginous area contains all these zones between lateral and medial bone collars. Gray area shows chondroproliferation area. Fig. 2. Whole skeletal staining at E The mandibular angular cartilage (AC) and the condylar cartilage (CC) are clearly recognized. Lines show the direction of sections.
4 44 S. SHIBATA, T. FUJIMORI and Y. YAMASHITA J Med Dent Sci Fig. 3. Development of the angular cartilage at E 14.5 (a-e, p), E 15.5 (f-j, q), E 16.5 (k-o) and E 17.5 (r). (a) The anlage of angular cartilage is recognized as mesenchymal cell condensation (arrow) adjacent to the ossifying mandible (OM). (b-e) The anlage never expresses mrnas for Type II collagen (arrow in b), Type X, collagen (arrow in c), OPN (arrow in d) and BSP (arrow in e). Cells in the ossifying mandible express OPN and BSP mrna (arrowheads in d and e). (f) The angular cartilage is first recognized within the anlage (arrow). Toluidin blue staining. (gj) Newly formed chondrocytes simultaneously express mrnas for Type II collagen (arrow in g), Type X, collagen (arrows in h and inset), OPN (arrow in i) and BSP (arrow in j). (k) The angular cartilage extends in length, especially the hypertrophic cell zone (arrow). Toluidin blue staining. (l-o) Type II collagen mrna is expressed in whole zone (arrow in l). Type X collagen mrna is expressed in the whole hypertrophic cell zone (arrow in m). OPN mrna is restrictedly expressed in the lower hypertrophic cell zone (arrow in n). BSP mrna is expressed in the whole hypertrophic cell zone (arrow in o). (p, q) ALP activity is detected in the mesenchymal condensation (arrow in p) and newly formed cartilage (arrow in q). (r)the endochondral bone formation starts with the resorption of bone collar by TRAP-positive osteoclasts (arrow). MPM: Medial pterygoid nuscle. MM: Masseter muscle. M: Meckel s cartilage. Bar=100 Òm.
5 FETAL MOUSE ANGULAR CARTILAGE , cartilage extended in length, especially the hypertrophic cell zone (Fig. 3k). Type II collagen mrna was expressed in whole zone of this cartilage (Fig. 3l). Type X collagen mrna was expressed in the whole hypertrophic cell zone (Fig. 3m). OPN mrna was restrictedly expressed in the lower hypertrophic cell zone at this stage (Fig. 3n), while BSP mrna was expressed in the whole hypertrophic cell zone (Fig. 3o). Further, ALP activity was detected both in the mesenchymal condensation at E 14.5 (Fig. 3p) and in the newly formed angular cartilage at E 15.5 (Fig. 3q). The endochondral bone formation of this cartilage started with the resorption of bone collar by TRAP-positive osteoclasts at E 17.5 (Fig. 3r). Postnatal changes of the mandibular angular cartilage During postnatal period from E 18.5 to d 21, the angular cartilage decreased in length with advancing age and consequently classification of zones became unclear at d 21. The medio-lateral width of this cartilage hardly increased with advancing age (Fig. 4a, g, m, s). At E 18.5, Type II collagen mrna was expressed from the flattened cell zone to the upper hypertrophic cell zone (Fig. 4b). Type X collagen mrna was mainly expressed in the upper hypertrophic cell zone (Fig. 4c), while OPN mrna was restrictedly expressed in the deep layer of the lower hypertrophic cell zone (Fig. 4d). Although this expression pattern for each mrna was almost similar at d 7 and d 14, each zone expressing mrnas for these molecules reduced in volume (Fig. 4h-j, n-p), and became undistinguishable at d 21 (Fig. 4t-v). Results of histomorphometry for each zone in the angular cartilage were described in Figure 6a. BrDUpositive cells in the chondroproliferation area decreased in number with advancing age (Fig. 4e, k, q, w). TRAP-positive, multinucleated chondroclasts in the erosion zone also decreased in number with advancing age (Fig. 4f, l, r, x). Results of cell counting normalized were described in Figure 6c and d. Postnatal changes of the mandibular condylar cartilage The mandibular condylar cartilage gradually reduced in length but increased in medio-lateral width with advancing age, and consequently maintained its volume at d 21 (Fig. 5a, g, m, s). At E 18.5, Type II collagen mrna was expressed from the flattened cell zone to the upper hypertrophic cell zone (Fig. 5b). Type X collagen mrna was mainly expressed in the upper hypertrophic cell zone (Fig. 5c), while OPN mrna was restrictedly expressed in the deep layer of the lower hypertrophic cell zone (Fig. 5d). This expression pattern for each mrna was almost similar at all stages examined (Figs. 5h-j, n-p, t-v) and each zone expressing these molecules maintained its volume and was clearly distinguishable at d 21. Results of histomorphometry for each zone in the condylar cartilage were described in Figure 6b. BrDU-positive cells in the chondroproliferation area decreased in number with advancing age (Fig. 5e, k, q, w). Considerable amount of BrDU-positive cells remained at d 21 (Fig. 5w), but since the chondroproliferation area also increased in volume, the cell number per this area remarkably reduced. To the contrary TRAP-positive multinucleated chondroclasts were continuously observed in the erosion zone at all stages (Fig. 5f, l, r, x), indicating endochondral bone formation was actively progressing until d 21. Results of cell counting normalized were described in Figure 6c and d. Negative controls for in situ hybridization and histochemistry showed no positive reaction at any time points (data not shown), as previously described 8,13,14. Histomorphometry The total cartilaginous area and each zone constantly decreased in volume in the angular cartilage from E 18.5 to d 21. While the OPN mrna expression area rapidly decreased in volume from E 18.5 to d 14, the Type II collagen mrna expression area tended to decrease slowly in volume as compared to other two areas from d 7 to d 14 (Fig. 6a). Meanwhile, the total cartilaginous area of the mandibular condylar cartilage decreased in volume from E 18.5 to d 14 but recovered from d 14 to d 21, and consequently large amount of volume remained at d 21. The Type II collagen and Type X collagen mrna expressing areas showed similar pattern of changes to the total cartilaginous area, although the OPN expressing area rapidly decreased in volume from E 18.5 to d 14 (Fig. 6b). The number of BrDU-positive cells per chondroproliferation area decreased with advancing age in the angular cartilage but this was similar in the condylar cartilage (Fig. 6c), indicating proliferating activity in the angular cartilage is reduced but still maintains to some extent as the condylar cartilage goes at d 21. The number of TRAP-positive multinucleated chondroclasts per erosion zone length constantly increased in number in the condylar cartilage with advancing age but constantly decreased in the angular cartilage (Fig. 6d).
6 46 S. SHIBATA, T. FUJIMORI and Y. YAMASHITA J Med Dent Sci Fig. 4. Mandibular angular cartilage at E 18.5 (a-f), d 7 (g-l), d14 (m-r), d 21 (s-x). Toluidin blue staining (a, g, m, s) and mrna expression for Type II collagen (b, h, n, t), Type X collagen (c, i, o, u) and OPN (d, j, p, v ). BrDU immunohistochemistry that simultaneously expresses Type II collagen mrna (e, k, q, w). TRAP staining at the erosion zone (f, l, r, x). The angular cartilage decreases in length with age from E 18.5 to d 14 (a, g, m). Type II collagen mrna is expressed from the flattened cell zone to the upper hypertrophic cell zone (arrows in b, h, n). Type X collagen mrna is expressed in the upper hypertrophic cell zone (arrows in c, i, o). OPN mrna is restrictedly expressed in the deep layer of the lower hypertrophic cell zone (arrows in d, j, p). Classification of zones becomes unclear at d 21 (s, t, u, v). BrDU-positive cells in the chondroproliferation area decreased in number with age (e, k, q, w). Note that small anount of BrDU-positive cells similtaneously express Type II collagen mrna (arrows in e). TRAP-positive multinucleated chondroclasts in the erosion zone decreases in number with age (arrows in f, l, r, x). MPM: Medial pterygoid muscle. MM: Masseter muscle. Bar=100 Òm.
7 FETAL MOUSE ANGULAR CARTILAGE 47 Fig. 5. Mandibular condylar cartilage at E 18.5 (a-f), d 7 (g-l), d14 (m-r), d 21 (s-x). Toluidin blue staining (a, g, m, s) and mrna expression for Type II collagen (b, h, n, t), Type X collagen (c, i, o, u) and OPN (d, j, p, v). BrDU immunohistochemistry that simultaneously expresses Type II collagen mrna (e, k, q, w). TRAP staining at the erosion zone (f, l, r, x). The mandibular condylar cartilage reduces in length but increases in medio-lateral width with age (a, g, m, s). Type II collagen mrna is expressed from the flattened cell zone to the upper hypertrophic cell zone (arrows in b, h, n, t). Type X collagen mrna is expressed in the upper hypertrophic cell zone (arrows in c, i, o, u). OPN mrna is restrictedly expressed in the deep layer of the lower hypertrophic cell zone (arrows in d, j, p, v). BrDU-positive cells in the chondroproliferation area decreased in number with age, but considerable amount of these cells remained at d 21 (e, k, q, w). TRAP-positive multinucleated chondroclasts are continuously observed in the erosion zone at all stages (arrows in f, l, r, x). Bar=100 Òm.
8 48 S. SHIBATA, T. FUJIMORI and Y. YAMASHITA J Med Dent Sci Fig. 6. Histomorphometric data during postnatal changes. Data are mean SD (N=4). (a) Changes of areas in the angular cartilage. OPN mrna expression area rapidly decreases in volume from E 18.5 to d 14 (b) Changes of areas in the condylar cartilage. OPN mrna expressing area rapidly decreases in volume from E 18.5 to d 14. Every area shows recovery from d 14 to d 21. (c) The number of BrDUpositive cells per chondroproliferation area. It decreases with age both in the angular cartilage and in the condylar cartilage. (d) The number of chondroclasts per erosion zone length. It constantly increases in the condylar cartilage with age but decreases in the angular cartilage. Discussion In this study, chondrocytes of the mouse mandibular angular cartilage was first detected at E 15.5 and this appearance time was 0.5 to 1 day delayed relative to the condylar cartilage 7,8,10,13. The endochondral bone formation started at E 17.5 in the angular cartilage and this starting time was 1.5 day delayed relative to the condylar cartilage 7,14. However, chondrocytes of the angular cartilage were derived from ALP-positive progenitor cells as previously described 9 and rapidly differentiated into hypertrophic chondrocytes and the hypertrophic cell zone rapidly extended in subsequent a few days. During this period, BSP mrna was more widely expressed than OPN mrna in the newly formed angular cartilage. These characteristics were entirely consistent with those of the condylar cartilage 7-10,13. Rapid differentiation of progenitor cells into hypertrophic chondrocytes is also reported in chick secondary cartilage 27. Therefore, we think that these developing patterns can be regarded as general structural features of secondary cartilage. The principal function of secondary cartilage seems to assist the growth of membrane bone 2,5. Luder et al. 25 indicated that cell enlargement is the most important factor for interstitial growth, and hence rapid expansion of the hypertrophic cell zone in newly formed cartilage is functionally significant for the growth of the angular
9 FETAL MOUSE ANGULAR CARTILAGE 49 process. In both cartilage types, the total cartilaginous area and each zone decreased in volume during E 18.5 to d 14. OPN is expressed in the deep layer of the hypertrophic cell zone (zone of provisional mineralization) at the site of the endochondral bone formation and involved in both provisional mineralization and cartilage resorption by recruiting chondroclasts 13,28,29. Thus rapid reduction of OPN mrma expression area during this period indicates that cartilage resorption rapidly advanced in both cartilage types through the endochondral bone formation. Constant increase of the number of chondroclasts per erosion zone length in the condylar cartilage corresponds with this phenomenon. The condylar cartilage showed a recovery in volume at d 21 despite the constant reduction of proliferating activity in the chondroproliferation area and constant increase of the number of chondroclasts at the erosion zone. Increasing of the medio-lateral width may account for this phenomenon. Meanwhile the angular cartilage constantly decreased in length and did not increase in medio-lateral width, leading to the constant decrease in volume. We have hypothesized two possible reasons for the decrease of angular cartilage in volume; 1) Reduction of proliferating activity. 2) Acceleration of cartilage resorption at the erosion zone. The number of BrDUpositive cells per chondroproliferation area indeed decreased with advancing age, indicating reduced proliferating activity is a reason. Teramoto et al. 30 speculated similar reason for the decrease of condylar cartilage volume under the application of compressive force. However, similar reduced proliferating activity was also recognized in the condylar cartilage that maintained its volume at d 21. The number of chondroclasts per erosion zone length increased in number in the condylar cartilage with advancing age, but rather decreased in the angular cartilage, indicating cartilage resorption was not accelerated in the angular cartilage with advancing age. Therefore, above two possible reasons cannot completely explain the disappearance of the angular cartilage. Silbermann et al. 31 insisted that the condylar cartilage develops from already differentiated progenitor cells called skeletoblasts which are differentiated from embryonic mesenchymal cells, and work as osteochondro progenitor cells that can differentiate both into chondrocytes of secondary cartilage and mature osteoblasts according to circumstances. Several histological/histochemical studies indicated that the condylar cartilage is derived from ALP-positive, Type I collagen mrna expressing progenitor cells continuous to the ossifying mandible 7,8,9,11, and hence we have fundamentally supported Silberman s hypothesis. In vivo, these skeletoblasts seem to differentiate into chondrocytes of the mandibular condyle and osteoblasts of the bone collar 32. Therefore, we hypothesize the possibility that the differentiation of osteochondro progenitor cells (skeletoblasts) into chondrocytes was gradually inhibited in the angular cartilage with advancing age, while the bipotential activity was maintained in the condylar cartilage. Another hypothesis is that the speed of differentiation in every zone is remarkably reduced in the angular cartilage. A pulse-chase study of BrDU incorporation can possibly clarify these hypotheses in the future. Many experimental studies related to the mechanical stress to the condylar cartilage 30,33-35 indicate that cartilage volume tends to decrease by the loss of physiological force, e.g., joint movement, masticatory force and by static compressive force, whereas it tends to increase by adding adequate force such as intermitted force. Therefore, the volume of the condylar cartilage seems to be maintained by adequate mechanical force, since the occlusion of rodents is established around 3 weeks after birth in and the condylar cartilage starts to work as articular cartilage. Meanwhile the angular cartilage does not work as articular cartilage, loss of adequate mechanical force seems to lead the disappearance of this cartilage and similar concepts have been accepted by previous studies 2,18,38. We demonstrate new structural features of the mandibular angular cartilage that may contribute to a coming research for the secondary cartilage. Acknowledgment This work is supported by Grant-in-Aid for Scientific Research (No ) from Ministry of Education, Culture, Sports, Science and Technology of Japan. References 1. Stutzmann J, Petrovic A. Nature et apitudes des cellules du compartiment mitotique des cartilages secondaires de la mandibule et du maxillaire dejeune rat. Expeeiences de culture cytotypique et d7 hormotransolantation. Bull Assoc Anat 1975;59: Vinkka H. Secondary cartilages in the facial skeleton of the rat. Proc Finn Dent Soc [Supple 78] 1982;7: Vilmann H. The mandibular angular cartilage in the rat. Acta Anat 1982;113: Tomo S, Ogita M, Tomo I. Development of mandibular carti-
10 50 S. SHIBATA, T. FUJIMORI and Y. YAMASHITA J Med Dent Sci lages in the rat. Anat Rec 1997; 249: Sperber GH. Craniofacial Development. BC Decker Inc, Hamilton Canada. 2001; Shibata S, Suda N, Fukada K, et al. Mandibular coronoid process in parathyroid hormone-related protein-deficient mice shows ectopic cartilage formation accompanied by abnormal bone modeling. Anat Embryol 2003;207: Shibata S, Suzuki S, Tengan T, et al. A histological study of the developing condylar cartilage of the fetal mouse mandible using coronal sections. Arch Oral Biol 1996;41: Shibata S, Fukada K, Suzuki S, et al. Immunohistochemistry of collagen Types II and X, and enzyme-histochemistry of alkaline phosphatase in the developing condylar cartilage of the fetal mouse mandible. J Anat 1997;191: Miyake T, Cameron AM, Hall BK. Stage-specific expression patterns of alkaline phosphatase during development of the first arch skeleton in inbred C57BL/6 mouse embryos. J Anat 1997;190, Fukada K, Shibata S, Suzuki S, et al. In situ hybridisation study of Type I, II, X collagens and aggrecan mrnas in the developing condylar cartilage of fetal mouse mandible. J Anat 1999;195: Ishii M, Suda N, Tengan T, et al. Immunohistochemical findings type I and Type II collagen in prenatal mouse mandibular condylar anlage compared with the tibial anlage. Arch Oral Biol 1998;43: Ogawa T, Shinokawa H, Fukada K, et al. Localization and inhibitory effect of basic fibroblast growth factor on chondrogenesis in cultured mouse mandibular condyle. J Bone Miner Metab 2003;21: Shibata S, Fukada K, Suzuki S, et al. In situ hybridization and immunohistochemistry of bone sialoprotein and secreted phosphoprotein 1 (osteopontin) in the developing mouse mandibular condylar cartilage compared with limb bud cartilage. J Anat 2002;200: Shibata S, Suzuki S, Yamashita. An ultrastructural study of cartilage resorption at the site of initial endochondral bone formation in the fetal mouse mandibular condyle. J Anat 1997;191: Ashida T. Immunohistochemical studies of types I, II and X collagen in the mandibular cartilage of browing rats. (in Japanese) Jpn J Oral Biol 1996;38: Ohashi N, Ejiri S, Hanada K, et al. Changes in type I, II, X collagen immunoreactivity of the mandibular condylar cartilage in a naturally aging rat madel. J Bone Miner Metab 1997;15: Bhaskar SN. Growth pattern of the rat mandible from 13 days insemination age to 30 days after birth. Am J Anat 1953;92: Moss ML. Functional cranial analysis of the mandibular angular cartilage in the rat. Angle Orthodont 1969;39: Duterloo HS, Jansen HWB. Chondrogenesis and osteogenesis in the mandibular condylar blastema. Trans Eur orthod Soc : Frommer J. Prenatal development of the mandibular joint in mice. Anat Rec 1964;150: Baume LJ. Ontogenesis of the human temporomandibular joint: 1. Development of the condyles. J Dent Res 1962;41: McLeod MJ. Differential staining of cartilage and bone in whole mouse fetuses by alcian blue and alizarin red S. Teratology 1980;22: Shibata S, Fukada K, Imai H, et al. In situ hybridization and immunohistochemistry of versican, aggrecan, and link protein and histochemistry of hyaluronan in the developing mouse limb bud cartilage. J Anat 2003;203: Lewinson D, Silbermann M. Chondroclasts and endothelial cells collaborate in the process of cartilage resorption. Anat Rec 1992;233: Luder HU, Leblond CP, von der Mark K. Cellular stages in cartilage formation as revealed by morphometry, radioautography and Type II collagen immunostaining of the mandibular condyle from weanling rats. Am J Anat 1988;182: Nordahl J, Anderson G, Reinholt FP. Chondroclasts and osteoclasts in bones of young rats: comparison of ultrastructural and functional features. Calcif Tissue Int 1998;63: Buxton PG, Hall B, Archer CW, et al. Secondary chondrocytesderived Ihh stimulates proliferation of periosteal cells during chick development. Development 2003;130: Nomura S, Wills AJ, Edwards DR, et al. Developmental expression of 2ar (osteopontin) and SPARC (osteonectin) RNA as revealed by in situ hybridization. J Cell Biol 1988;106: Chen J, Singh K, Mukherjee BB, et al. Developmental expression of osteopontin (OPN) mrna in rat tissues: evidence for a role for OPN in bone formation and resorption. Matrix 1993;13: Teramoto M, Kaneko S, Shibata S, et al. Effect of compressive forces on extracellular matrix in rat mandibular condylar cartilage. J Bone Miner Metab 2003;21: Silbermsann M, Reddi AH, Hand AR, et al. Further characterization of the extracellular matrix in the mandibular condyle in neonatal mice. J Anat 1987;151: Terashima T. Observations on the structure of the mandibular condyle of the prenatal rabbits (in Japanese). J Stomatol Soc Jpn 1985;52: Copray JCVM, Jansen HWB, Duterloo HS. Effect of compressive force on proliferation and matrix synthesis in mandibular condylar cartilage of the rat in vitro. Arch Oral Biol 1985;30: Axelsson S, Björnsson S, Holmlund A, et al. Metabolic turnover of sulfated glycosaminoglycans and proteoglycans in rabbit temporomandibular joint cartilages with experimentally induced osteoarthrosis. Acta Odont Scand 1994;52: Duterloo HS, Wolters JM. Experiments on the significance of articular function as a stimulating chondrogenic factor for the growth of the secondary cartilages of the rat mandible. Trans Eur Orthod Soc :
An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle
 Okajimas Folia Anat. Jpn., 88(2): 49 55, August, 2011 An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle By Keiichi Kenzaki 1, Kohzo Tsuchikawa
Okajimas Folia Anat. Jpn., 88(2): 49 55, August, 2011 An immunohistochemical study on the localization of type II collagen in the developing mouse mandibular condyle By Keiichi Kenzaki 1, Kohzo Tsuchikawa
FORMATION OF BONE. Intramembranous Ossification. Bone-Lec-10-Prof.Dr.Adnan Albideri
 FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage
 Original Article Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage Tanapan Wattanachai a ; Ikuo Yonemitsu b ; Sawa Kaneko c ; Kunimichi Soma d
Original Article Functional Lateral Shift of the Mandible Effects on the Expression of ECM in Rat Temporomandibular Cartilage Tanapan Wattanachai a ; Ikuo Yonemitsu b ; Sawa Kaneko c ; Kunimichi Soma d
o~ r;'c' - OSTEOARTHRITIS
 Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Bone. Development. Tim Arnett. University College London. Department of Anatomy and Developmental Biology
 Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Original Article. An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle. Atith Manopinivate, Sawa Kaneko and Kunimichi Soma
 J Med Dent Sci 2006; 53: 67 74 Original Article An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle Atith Manopinivate, Sawa Kaneko and Kunimichi Soma Orthodontic Science,
J Med Dent Sci 2006; 53: 67 74 Original Article An Impact of Masticatory Muscle Function on IL-1Ç and SOX9 Expression in Condyle Atith Manopinivate, Sawa Kaneko and Kunimichi Soma Orthodontic Science,
Non commercial use only. An in situ hybridization study of perlecan, DMP1, and MEPE in developing condylar cartilage of the fetal mouse mandible
 European Journal of Histochemistry 2015; volume 59:2553 An in situ hybridization study of perlecan, DMP1, and MEPE in developing condylar cartilage of the fetal mouse mandible and limb bud cartilage K.
European Journal of Histochemistry 2015; volume 59:2553 An in situ hybridization study of perlecan, DMP1, and MEPE in developing condylar cartilage of the fetal mouse mandible and limb bud cartilage K.
Chapter 6: Skeletal System: Bones and Bone Tissue
 Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
Supplemental Tables and Figures. The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate,
 Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
For more information about how to cite these materials visit
 Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
Module 2:! Functional Musculoskeletal Anatomy A! Semester 1! !!! !!!! Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb!
 Functional Musculoskeletal Anatomy A Module 2: Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb Semester 1 1 18. Bone Tissue & Growth of Bones 18.1 Describe the structure of bone tissue
Functional Musculoskeletal Anatomy A Module 2: Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb Semester 1 1 18. Bone Tissue & Growth of Bones 18.1 Describe the structure of bone tissue
ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN
 ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN 1982 Effects of a pulsed electromagnetic field on a mixed chondroplastic
ZUSAMMENSTELLUNG VON WISSENSCHAFTLICHEN PUBLIKATIONEN ZUR WIRKUNG DER PULSIERENDER MAGNETFELDTHERAPIE AUF KNOCHEN- UND KNORPELZELLEN 1982 Effects of a pulsed electromagnetic field on a mixed chondroplastic
Ossification and Bone Remodeling
 Ossification and Bone Remodeling Pre-natal Ossification Embryonic skeleton: fashioned from fibrous membranes or cartilage to accommodate mitosis. 2 types of pre-natal ossification (bone formation) 1.
Ossification and Bone Remodeling Pre-natal Ossification Embryonic skeleton: fashioned from fibrous membranes or cartilage to accommodate mitosis. 2 types of pre-natal ossification (bone formation) 1.
Bone Development. Two Types of OssificaDon 10/18/14. Osteogenesis ( ) bone Dssue formadon Stages. Bones and Skeletal Tissues: Part B
 Bone Development 6 Bones and Skeletal Tissues: Part B Osteogenesis ( ) bone Dssue formadon Stages Bone formadon begins in the 2nd month of development Postnatal bone growth undl early adulthood Bone remodeling
Bone Development 6 Bones and Skeletal Tissues: Part B Osteogenesis ( ) bone Dssue formadon Stages Bone formadon begins in the 2nd month of development Postnatal bone growth undl early adulthood Bone remodeling
BONE TISSUE. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
Bone Formation, Growth, and Remodeling
 Bone Formation, Growth, and Remodeling Pre-natal Ossification Embryonic skeleton: fashioned from fibrous membranes or cartilage to accommodate mitosis. 2 types of pre-natal ossification (bone formation)
Bone Formation, Growth, and Remodeling Pre-natal Ossification Embryonic skeleton: fashioned from fibrous membranes or cartilage to accommodate mitosis. 2 types of pre-natal ossification (bone formation)
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
Lecture 2: Skeletogenesis
 Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Chapter 6 Skeletal System
 Chapter 6 Skeletal System Functions of the skeletal system/bone 1. Support skeletal system is the internal framework of the body 2. Protection protects internal organs 3. Movement muscles & bones work
Chapter 6 Skeletal System Functions of the skeletal system/bone 1. Support skeletal system is the internal framework of the body 2. Protection protects internal organs 3. Movement muscles & bones work
PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis)
 Original Article Buffalo Bulletin (March 2014) Vol.33 No.1 PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis) M. Santhi Lakshmi *, T.S. Chandrasekhara Rao and
Original Article Buffalo Bulletin (March 2014) Vol.33 No.1 PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis) M. Santhi Lakshmi *, T.S. Chandrasekhara Rao and
Non-commercial use only
 European Journal of Histochemistry 2013; volume 57:e39 An immunohistochemical study of matrix proteins in the craniofacial cartilage in midterm human fetuses S. Shibata, 1 Y. Sakamoto, 2 O. Baba, 3 C.
European Journal of Histochemistry 2013; volume 57:e39 An immunohistochemical study of matrix proteins in the craniofacial cartilage in midterm human fetuses S. Shibata, 1 Y. Sakamoto, 2 O. Baba, 3 C.
Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study in the rat
 European Journal of Orthodontics 18 (1996) 3-9 O 1996 European Orthodontic Society Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study
European Journal of Orthodontics 18 (1996) 3-9 O 1996 European Orthodontic Society Growth and type-ll collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: a study
Cartilage. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Growth and repair: Cartilage is a vascular tissues that receives nutrients by diffusion through its matrix, cartilage grow by 2 mechanisms:
 Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Functions of the Skeletal System. Chapter 6: Osseous Tissue and Bone Structure. Classification of Bones. Bone Shapes
 Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
cartilage of the rat
 J. Anat. (1989), 162, pp. 43-51 43 With 4 figures Printed in Great Britain Presence of osteonectin/sparc in mandibular condylar cartilage of the rat J. C. V. M. COPRAY, P. M. JOHNSON*, J. D. DECKER* AND
J. Anat. (1989), 162, pp. 43-51 43 With 4 figures Printed in Great Britain Presence of osteonectin/sparc in mandibular condylar cartilage of the rat J. C. V. M. COPRAY, P. M. JOHNSON*, J. D. DECKER* AND
KEY CONCEPTS Unit 6 THE SKELETAL SYSTEM
 ANATOMY & PHYSIOLOGY 1 (101-805 - AB) PAUL ANDERSON 2011 KEY CONCEPTS Unit 6 THE SKELETAL SYSTEM A Overview of The Skeletal System 1. Definition: Anatomically the SKELETAL SYSTEM consists of bones, cartilages,
ANATOMY & PHYSIOLOGY 1 (101-805 - AB) PAUL ANDERSON 2011 KEY CONCEPTS Unit 6 THE SKELETAL SYSTEM A Overview of The Skeletal System 1. Definition: Anatomically the SKELETAL SYSTEM consists of bones, cartilages,
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Ultrastructure and Immunolocalization of Transforming Growth Factor-Beta in Chondrification of Murine Ligamentous Fibroblasts and Endochondral
 Aeta Histochem, Cytochena. Vol. 30 No, 4 371-379 (1997) Ultrastructure and Immunolocalization of Transforming Growth Factor-Beta in Chondrification of Murine Ligamentous Fibroblasts and Endochondral Calcification
Aeta Histochem, Cytochena. Vol. 30 No, 4 371-379 (1997) Ultrastructure and Immunolocalization of Transforming Growth Factor-Beta in Chondrification of Murine Ligamentous Fibroblasts and Endochondral Calcification
ANATOMY & PHYSIOLOGY - CLUTCH CH. 8 - BONE AND CARTILAGE.
 !! www.clutchprep.com CONCEPT: BONE CLASSIFICATIONS There are four classifications of bones based on their 1. Long bones are greater in length than in width - Found in the upper and lower limbs (ex: arm,
!! www.clutchprep.com CONCEPT: BONE CLASSIFICATIONS There are four classifications of bones based on their 1. Long bones are greater in length than in width - Found in the upper and lower limbs (ex: arm,
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiolog
 DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiologist.at an early age- she eloped with a statistician,
DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiologist.at an early age- she eloped with a statistician,
AAO Foundation Awards Final Report
 401 N. Lindbergh Blvd. St. Louis, MO 63141 Tel.: 314.993.1700, #546 Toll Free: 800.424.2841, #546 Fax: 800.708.1364 Cell: 314.283.1983 E-Mail: rhazel@aaortho.org AAO Foundation Awards Final Report In an
401 N. Lindbergh Blvd. St. Louis, MO 63141 Tel.: 314.993.1700, #546 Toll Free: 800.424.2841, #546 Fax: 800.708.1364 Cell: 314.283.1983 E-Mail: rhazel@aaortho.org AAO Foundation Awards Final Report In an
Osteology. Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College
 Osteology Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College Functions of the Skeletal System: Support Movement Protection Hemopoiesis Electrolyte balance (Ca ++ /PO -3 4 ) Acid-base balance Storage
Osteology Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College Functions of the Skeletal System: Support Movement Protection Hemopoiesis Electrolyte balance (Ca ++ /PO -3 4 ) Acid-base balance Storage
SKELETAL TISSUES CHAPTER 7 INTRODUCTION TO THE SKELETAL SYSTEM TYPES OF BONES
 SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
Distinct Roles Of CCN1 And CCN2 In Limb Development
 Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
Dr. Heba Kalbouneh. Saba Alfayoumi. Heba Kalbouneh
 11 Dr. Heba Kalbouneh Saba Alfayoumi Heba Kalbouneh 2- Bone Bone tissue is also classified into primary bone and secondary bone. In the beginning, the first bone that is deposited by the osteoblasts is
11 Dr. Heba Kalbouneh Saba Alfayoumi Heba Kalbouneh 2- Bone Bone tissue is also classified into primary bone and secondary bone. In the beginning, the first bone that is deposited by the osteoblasts is
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
Regulation of the skeletal mass through the life span
 Regulation of the skeletal mass through the life span Functions of the skeletal system Mechanical protection skull Movement leverage for muscles Mineral metabolism calcium store Erythropoiesis red blood
Regulation of the skeletal mass through the life span Functions of the skeletal system Mechanical protection skull Movement leverage for muscles Mineral metabolism calcium store Erythropoiesis red blood
Matrix Metalloproteinase-9 Regulates Graft Bone Resorption
 Original Article Matrix Metalloproteinase-9 Regulates Graft Bone Resorption Mei Lu a ; A. B. M. Rabie b ABSTRACT Objective: To explore the relationship between bone resorption and matrix metalloproteinase
Original Article Matrix Metalloproteinase-9 Regulates Graft Bone Resorption Mei Lu a ; A. B. M. Rabie b ABSTRACT Objective: To explore the relationship between bone resorption and matrix metalloproteinase
TRACP & ALP double-stain Kit
 Table of Content I. Description... 2 II. Introduction... 2 III. Principles... 2 IV. Kit components... 3 V. Storage... 3 VI. Preparation of reagents... 3 VII. Methods... 4-7 Cell fixation... 4 Activity
Table of Content I. Description... 2 II. Introduction... 2 III. Principles... 2 IV. Kit components... 3 V. Storage... 3 VI. Preparation of reagents... 3 VII. Methods... 4-7 Cell fixation... 4 Activity
Chapter 4. Cartilage and Bone. Li Shu-Lei instructor. Dept. Histology and Embryology, School of Basic Medical Sciences, Jilin University
 Chapter 4 Cartilage and Bone Li Shu-Lei instructor Dept. Histology and Embryology, School of Basic Medical Sciences, Jilin University I Cartilage a specialized connective tissue Characterizers: Cartilage
Chapter 4 Cartilage and Bone Li Shu-Lei instructor Dept. Histology and Embryology, School of Basic Medical Sciences, Jilin University I Cartilage a specialized connective tissue Characterizers: Cartilage
Bone Tissue- Chapter 5 5-1
 Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
Sheets 16&17. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Sheets 16&17 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ossification (formation of bone) - Osteoblasts are responsible for producing the extracellular matrix of the bone and these osteoblasts
Sheets 16&17 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ossification (formation of bone) - Osteoblasts are responsible for producing the extracellular matrix of the bone and these osteoblasts
CHAPTER 6 LECTURE OUTLINE
 CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
Biology. Dr. Khalida Ibrahim
 Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
SKELETAL SYSTEM I NOTE: LAB ASSIGNMENTS for this topic will run over 3 Weeks. A SEPARATE WORKSHEET WILL BE PROVIDED.
 BIO 211; Anatomy and Physiology I REFERENCE: CHAPTER 07 1 Dr. Lawrence Altman Naugatuck Valley Community College LECTURE TOPICS OUTLINE SKELETAL SYSTEM I NOTE: LAB ASSIGNMENTS for this topic will run over
BIO 211; Anatomy and Physiology I REFERENCE: CHAPTER 07 1 Dr. Lawrence Altman Naugatuck Valley Community College LECTURE TOPICS OUTLINE SKELETAL SYSTEM I NOTE: LAB ASSIGNMENTS for this topic will run over
In Vivo Heat-stimulus Triggered Osteogenesis
 In Vivo Heat-stimulus Triggered Osteogenesis Kunihiro Ikuta 1, Hiroshi Urakawa 1, Eiji Kozawa 1, Shunsuke Hamada 1, Naoki Ishiguro 2, Yoshihiro Nishida 1. 1 Nagoya University, Nagoya, Japan, 2 Nagoya Graduated
In Vivo Heat-stimulus Triggered Osteogenesis Kunihiro Ikuta 1, Hiroshi Urakawa 1, Eiji Kozawa 1, Shunsuke Hamada 1, Naoki Ishiguro 2, Yoshihiro Nishida 1. 1 Nagoya University, Nagoya, Japan, 2 Nagoya Graduated
An Introduction to the Skeletal System Skeletal system includes Bones of the skeleton Cartilages, ligaments, and connective tissues
 An Introduction to the Skeletal System Skeletal system includes Bones of the skeleton Cartilages, ligaments, and connective tissues Functions of the Skeletal System Support Storage of minerals (calcium)
An Introduction to the Skeletal System Skeletal system includes Bones of the skeleton Cartilages, ligaments, and connective tissues Functions of the Skeletal System Support Storage of minerals (calcium)
Mandibular condyle: structure properties and growth regulation. Citation Journal of Oral Science and Health, 2014, v. 1 n. 1, p.
 Title Mandibular condyle: structure properties and growth regulation Author(s) Ren, C; Yang, Y Citation, 2014, v. 1 n. 1, p. 1-6 Issued Date 2014 URL http://hdl.handle.net/10722/200447 Rights This work
Title Mandibular condyle: structure properties and growth regulation Author(s) Ren, C; Yang, Y Citation, 2014, v. 1 n. 1, p. 1-6 Issued Date 2014 URL http://hdl.handle.net/10722/200447 Rights This work
Pepsin Solution ready-to-use
 SIE HABEN DIE VISION, WIR HABEN DIE SUBSTANZ. Pepsin Solution Single component Pepsin Solution: only one component refrigerator stable Pepsin is a commonly used digestive enzyme for immunohistochemical
SIE HABEN DIE VISION, WIR HABEN DIE SUBSTANZ. Pepsin Solution Single component Pepsin Solution: only one component refrigerator stable Pepsin is a commonly used digestive enzyme for immunohistochemical
Key words: Collagen synthesis - N-Terminal peptide of type III procollagen - Tumor marker - Liver cancer - Liver cirrhosis
 [Gann, 75, 130-135; February, 1984] HIGH CONCENTRATIONS OF N-TERMINAL PEPTIDE OF TYPE III PROCOLLAGEN IN THE SERA OF PATIENTS WITH VARIOUS CANCERS, WITH SPECIAL REFERENCE TO LIVER CANCER Terumasa HATAHARA,
[Gann, 75, 130-135; February, 1984] HIGH CONCENTRATIONS OF N-TERMINAL PEPTIDE OF TYPE III PROCOLLAGEN IN THE SERA OF PATIENTS WITH VARIOUS CANCERS, WITH SPECIAL REFERENCE TO LIVER CANCER Terumasa HATAHARA,
Two Separated Protocols with the Most Important Comments for Skeletal Staining in Embryonic and Adulthood Period in Laboratory Animals
 May 2014, Volume 11, Number 2 Two Separated Protocols with the Most Important Comments for Skeletal Staining in Embryonic and Adulthood Period in Laboratory Animals Farzane Sadeghi * PhD in Anatomical
May 2014, Volume 11, Number 2 Two Separated Protocols with the Most Important Comments for Skeletal Staining in Embryonic and Adulthood Period in Laboratory Animals Farzane Sadeghi * PhD in Anatomical
NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM
 LECTURE 4 SKULL NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM CRANIUM NEUROCRANIUM (protective case around brain) VISCEROCRANIUM (skeleton of face) NASOMAXILLARY COMPLEX MANDIBLE (DESMOCRANIUM)
LECTURE 4 SKULL NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM CRANIUM NEUROCRANIUM (protective case around brain) VISCEROCRANIUM (skeleton of face) NASOMAXILLARY COMPLEX MANDIBLE (DESMOCRANIUM)
Biology. Dr. Khalida Ibrahim
 Biology Dr. Khalida Ibrahim BONE TISSUE Bone tissue is a specialized form of connective tissue and is the main element of the skeletal tissues. It is composed of cells and an extracellular matrix in which
Biology Dr. Khalida Ibrahim BONE TISSUE Bone tissue is a specialized form of connective tissue and is the main element of the skeletal tissues. It is composed of cells and an extracellular matrix in which
CARTILAGE. Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015
 CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
Peggers Super Summaries Basic Sciences Bone
 Bone Overview & Turnover BONES Function o Support o Protection o Assisting movement o Storage of minerals o Production of red blood cells from marrow Types o Cancellous o Compact with Haversian systems
Bone Overview & Turnover BONES Function o Support o Protection o Assisting movement o Storage of minerals o Production of red blood cells from marrow Types o Cancellous o Compact with Haversian systems
Chapter 6: SKELETAL SYSTEM
 Chapter 6: SKELETAL SYSTEM I. FUNCTIONS A. Support B. Protection C. Movement D. Mineral storage E. Lipid storage (Fig. 6.8b) F. Blood cell production (Fig. 6.4) II. COMPONENTS A. Cartilage 1. Hyaline 2.
Chapter 6: SKELETAL SYSTEM I. FUNCTIONS A. Support B. Protection C. Movement D. Mineral storage E. Lipid storage (Fig. 6.8b) F. Blood cell production (Fig. 6.4) II. COMPONENTS A. Cartilage 1. Hyaline 2.
Versican Expression during Synovial Joint Morphogenesis
 Int. J. Biol. Sci. 2007, 3 380 Short Research Communication Versican Expression during Synovial Joint Morphogenesis International Journal of Biological Sciences ISSN 1449-2288 www.biolsci.org 2007 3(6):380-384
Int. J. Biol. Sci. 2007, 3 380 Short Research Communication Versican Expression during Synovial Joint Morphogenesis International Journal of Biological Sciences ISSN 1449-2288 www.biolsci.org 2007 3(6):380-384
Interactions of the endocrine system, bone and oral health
 Interactions of the endocrine system, bone and oral health All bones are not equal! Dense high proportion of cortical bone High proportion of trabecular bone Mandible Functions: mastication, respiration,
Interactions of the endocrine system, bone and oral health All bones are not equal! Dense high proportion of cortical bone High proportion of trabecular bone Mandible Functions: mastication, respiration,
Development and regeneration of the neonatal digit tip in mice
 Available online at www.sciencedirect.com Developmental Biology 315 (2008) 125 135 www.elsevier.com/developmentalbiology Development and regeneration of the neonatal digit tip in mice Manjong Han a, Xiaodong
Available online at www.sciencedirect.com Developmental Biology 315 (2008) 125 135 www.elsevier.com/developmentalbiology Development and regeneration of the neonatal digit tip in mice Manjong Han a, Xiaodong
A mandibular propulsive appliance modulates collagen-binding integrins distribution in the young rat condylar cartilage
 Biorheology 43 (2006) 293 302 293 IOS Press A mandibular propulsive appliance modulates collagen-binding integrins distribution in the young rat condylar cartilage Mara Rúbia Marques, Denise Hajjar, Virgínia
Biorheology 43 (2006) 293 302 293 IOS Press A mandibular propulsive appliance modulates collagen-binding integrins distribution in the young rat condylar cartilage Mara Rúbia Marques, Denise Hajjar, Virgínia
Skeletal Tissues. Skeletal tissues. Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs.
 Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
What are the parts of the skeletal system? Chapter 6- Part I Bones and Skeletal Tissues. Growth of Cartilage. Bones come in many shapes
 Chapter 6- Part I Bones and Skeletal Tissues Components of the skeletal system Classification of Bone (bone shapes) Functions of bone Bone structure Microscopic structure of bone and bone cells What are
Chapter 6- Part I Bones and Skeletal Tissues Components of the skeletal system Classification of Bone (bone shapes) Functions of bone Bone structure Microscopic structure of bone and bone cells What are
Blood. Hematopoietic Tissue
 Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS
 OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
Chapter 6: Osseous Tissue and Bone Structure
 Chapter 6: Osseous Tissue and Bone Structure I. An Introduction to the Skeletal System, p. 180 Objective: Describe the functions of the skeletal system The skeletal system includes: - bones of the skeleton
Chapter 6: Osseous Tissue and Bone Structure I. An Introduction to the Skeletal System, p. 180 Objective: Describe the functions of the skeletal system The skeletal system includes: - bones of the skeleton
b. Adult bones produce 2.5 million RBCs each second.
 Ch 6 Skeletal System I. Functions of the Skeletal System A. The skeletal system consists of: 1. bones, cartilage, tendons and ligaments B. Living bone is not Gr. dried up 1. It is dynamic and adaptable
Ch 6 Skeletal System I. Functions of the Skeletal System A. The skeletal system consists of: 1. bones, cartilage, tendons and ligaments B. Living bone is not Gr. dried up 1. It is dynamic and adaptable
BONE AND CARTILAGE LIA DAMAYANTI. Department of Histology - FMUI
 1 BONE AND CARTILAGE LIA DAMAYANTI Department of Histology - FMUI Agenda 2 Introduction Cartilage Development of cartilage Growth and repair Components of cartilage Type of Cartilage Agenda 3 Bone Components
1 BONE AND CARTILAGE LIA DAMAYANTI Department of Histology - FMUI Agenda 2 Introduction Cartilage Development of cartilage Growth and repair Components of cartilage Type of Cartilage Agenda 3 Bone Components
BONE LABORATORY DEMONSTRATIONS. These demonstrations are found on the bulletin boards outside the MCO Bookstore.
 BONE LABORATORY DEMONSTRATIONS These demonstrations are found on the bulletin boards outside the MCO Bookstore. COMPACT & TRABECULAR BONE - LM When viewed under the polarizing light microscope, the layering
BONE LABORATORY DEMONSTRATIONS These demonstrations are found on the bulletin boards outside the MCO Bookstore. COMPACT & TRABECULAR BONE - LM When viewed under the polarizing light microscope, the layering
BONE HISTOLOGY SLIDE PRESENTATION
 BONE HISTOLOGY SLIDE PRESENTATION PRESENTED BY: SKELETECH, INC. Clients and Friends: SkeleTech invites you to use these complimentary images for your own presentations or as teaching slides for bone biology.
BONE HISTOLOGY SLIDE PRESENTATION PRESENTED BY: SKELETECH, INC. Clients and Friends: SkeleTech invites you to use these complimentary images for your own presentations or as teaching slides for bone biology.
Human Anatomy & Physiology
 PowerPoint Lecture Slides prepared by Barbara Heard, Atlantic Cape Community College Ninth Edition Human Anatomy & Physiology C H A P T E R 6 Annie Leibovitz/Contact Press Images 2013 Pearson Education,
PowerPoint Lecture Slides prepared by Barbara Heard, Atlantic Cape Community College Ninth Edition Human Anatomy & Physiology C H A P T E R 6 Annie Leibovitz/Contact Press Images 2013 Pearson Education,
Chapter 6 Bones and Bone Tissue Chapter Outline
 Chapter 6 Bones and Bone Tissue Chapter Outline Module 6.1: Introduction to Bones as Organs (Figures 6.1, 6.2, 6.3, 6.4) A. The skeletal system includes the bones, joints, and their associated supporting
Chapter 6 Bones and Bone Tissue Chapter Outline Module 6.1: Introduction to Bones as Organs (Figures 6.1, 6.2, 6.3, 6.4) A. The skeletal system includes the bones, joints, and their associated supporting
Derived copy of Bone *
 OpenStax-CNX module: m57739 1 Derived copy of Bone * Shannon McDermott Based on Bone by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By
OpenStax-CNX module: m57739 1 Derived copy of Bone * Shannon McDermott Based on Bone by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By
Osseous Tissue and Bone Structure
 C h a p t e r 6 Osseous Tissue and Bone Structure PowerPoint Lecture Slides prepared by Jason LaPres Lone Star College - North Harris Copyright 2009 Pearson Education, Inc., publishing as Pearson Benjamin
C h a p t e r 6 Osseous Tissue and Bone Structure PowerPoint Lecture Slides prepared by Jason LaPres Lone Star College - North Harris Copyright 2009 Pearson Education, Inc., publishing as Pearson Benjamin
Oral Histology. Alveolar bone or process: Functions of alveolar bone: Chemical composition: Development of the alveolar process: Dr.
 Oral Histology Lec.12 Alveolar bone or process: Dr. Nada Al-Ghaban Alveolar bone is a specialized part of the mandibular and maxillary bones that forms the primary support structure for teeth. Although
Oral Histology Lec.12 Alveolar bone or process: Dr. Nada Al-Ghaban Alveolar bone is a specialized part of the mandibular and maxillary bones that forms the primary support structure for teeth. Although
Copy and Return to Teacher. The Skeletal System
 Copy and Return to Teacher The Skeletal System The Skeletal System Parts of the skeletal system o Bones (skeleton) o Joints o Cartilages o Ligaments Divided into two divisions o Axial skeleton o Appendicular
Copy and Return to Teacher The Skeletal System The Skeletal System Parts of the skeletal system o Bones (skeleton) o Joints o Cartilages o Ligaments Divided into two divisions o Axial skeleton o Appendicular
Fig Articular cartilage. Epiphysis. Red bone marrow Epiphyseal line. Marrow cavity. Yellow bone marrow. Periosteum. Nutrient foramen Diaphysis
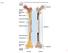 Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:!
 LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student.
 1 CARTILAGE AND BONE The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student. 1. Hyaline cartilage Slide 73 This is a cross section through
1 CARTILAGE AND BONE The purpose of this practical session is to demonstrate cartilage and bone as specialized connective tissues to the student. 1. Hyaline cartilage Slide 73 This is a cross section through
-the emphasis on this section is the structure and function of bone tissue and on the dynamics of its formation and remodeling throughout life.
 Biology 325 Fall 2004 BONES AND SKELETAL TISSUES Introduction -skeleton contains cartilage and bones -the emphasis on this section is the structure and function of bone tissue and on the dynamics of its
Biology 325 Fall 2004 BONES AND SKELETAL TISSUES Introduction -skeleton contains cartilage and bones -the emphasis on this section is the structure and function of bone tissue and on the dynamics of its
The Skeletal System PART A. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Skeletal System 5 PART A The Skeletal System Parts of the skeletal system Bones (skeleton) Joints
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Skeletal System 5 PART A The Skeletal System Parts of the skeletal system Bones (skeleton) Joints
ANATOMY & PHYSIOLOGY 1 ( ) For Intensive Nursing PAUL ANDERSON SAMPLE TEST
 ANATOMY & PHYSIOLOGY 1 (101-805) For Intensive Nursing PAUL ANDERSON SAMPLE TEST 3 2011 1. If calcium levels in the extracellular fluid are too low, parathyroid hormone secretion would and osteoclast activity
ANATOMY & PHYSIOLOGY 1 (101-805) For Intensive Nursing PAUL ANDERSON SAMPLE TEST 3 2011 1. If calcium levels in the extracellular fluid are too low, parathyroid hormone secretion would and osteoclast activity
Development of the mouse mandibles and clavicles in the absence of skeletal myogenesis
 Histol Histopathol (2007) 22: 51-60 http://www.hh.um.es Histology and Histopathology Cellular and Molecular Biology Development of the mouse mandibles and clavicles in the absence of skeletal myogenesis
Histol Histopathol (2007) 22: 51-60 http://www.hh.um.es Histology and Histopathology Cellular and Molecular Biology Development of the mouse mandibles and clavicles in the absence of skeletal myogenesis
The U ltrastructure of Bone
 ANNALS O F CLIN IC A L AND LABORATORY SCIEN CE, Vol. 5, No. 4 Copyright 1975, Institute for Clinical Science The U ltrastructure of Bone MARIJKE E. HOLTROP, M D., Ph D. Department of Orthopedic Surgery,
ANNALS O F CLIN IC A L AND LABORATORY SCIEN CE, Vol. 5, No. 4 Copyright 1975, Institute for Clinical Science The U ltrastructure of Bone MARIJKE E. HOLTROP, M D., Ph D. Department of Orthopedic Surgery,
BIOH111. o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system
 BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
ANATOMY OF THE PERIODONTIUM. Dr. Fatin Awartani
 ANATOMY OF THE PERIODONTIUM Part II Cementum and Alveolar bone Associate Professor Periodontal division King Saud university Cementum Calcified mesenchymal tissue that forms the outer covering of the anatomic
ANATOMY OF THE PERIODONTIUM Part II Cementum and Alveolar bone Associate Professor Periodontal division King Saud university Cementum Calcified mesenchymal tissue that forms the outer covering of the anatomic
Skeletal System worksheet
 Skeletal System worksheet Name Section A: Intro to Skeletal System The skeletal system performs vital functions that enable us to move through our daily lives. Support - The skeleton provides support and
Skeletal System worksheet Name Section A: Intro to Skeletal System The skeletal system performs vital functions that enable us to move through our daily lives. Support - The skeleton provides support and
HRP cytochemistry. Division of Radiooncology, Deutsches Krebsforschungszentrum, Heidelberg, Germany
 HRP cytochemistry WOLF D. KUHLMANN, M.D. Division of Radiooncology, Deutsches Krebsforschungszentrum, 69120 Heidelberg, Germany A range of substrates is available for the cytochemical staining of peroxidase
HRP cytochemistry WOLF D. KUHLMANN, M.D. Division of Radiooncology, Deutsches Krebsforschungszentrum, 69120 Heidelberg, Germany A range of substrates is available for the cytochemical staining of peroxidase
Synovial Chondromatosis of the Temporomandibular Joint: Long-Term Postoperative Follow-Up of the Residual Calcification
 J Med Dent Sci 2003; 50: 133 137 Case Report Synovial Chondromatosis of the Temporomandibular Joint: Long-Term Postoperative Follow-Up of the Residual Calcification Junichi Ishii 1, Koji Kino 2, Junji
J Med Dent Sci 2003; 50: 133 137 Case Report Synovial Chondromatosis of the Temporomandibular Joint: Long-Term Postoperative Follow-Up of the Residual Calcification Junichi Ishii 1, Koji Kino 2, Junji
Influence of orthodontic forces on the distribution of proteoglycans in rat
 J Med Dent Sci 2003; 50: 183 194 Original Article Influence of orthodontic forces on the distribution of proteoglycans in rat hypofunctional periodontal ligament Mayumi Esashika 1, Sawa Kaneko 1, Masaki
J Med Dent Sci 2003; 50: 183 194 Original Article Influence of orthodontic forces on the distribution of proteoglycans in rat hypofunctional periodontal ligament Mayumi Esashika 1, Sawa Kaneko 1, Masaki
Lab Animal Tissue. LEARNING OBJECTIVES: To understand the relationship between the structure and function of different animal tissues
 Name: Bio A.P. PURPOSE: HYPOTHESIS: NONE Lab Animal Tissue BACKGROUND: In animals, groups of closely related cells specialized to perform the same function are called tissues. There are four general classes
Name: Bio A.P. PURPOSE: HYPOTHESIS: NONE Lab Animal Tissue BACKGROUND: In animals, groups of closely related cells specialized to perform the same function are called tissues. There are four general classes
Skeletal System. The skeletal System... Components
 Skeletal System The skeletal System... What are the general components of the skeletal system? What does the skeletal system do for you & how does it achieve these functions? Components The skeletal system
Skeletal System The skeletal System... What are the general components of the skeletal system? What does the skeletal system do for you & how does it achieve these functions? Components The skeletal system
Changes in the temporomandibular joint after mandibular lengthening with different rates of distraction
 Shujuan Zou, DDS, MS Department of Orthodontics Jing Hu, DDS, MS, PhD Dazhang Wang, DDS, FICD Jihua Li, DDS, MS Zhenglong Tang, DDS, MS Department of Oral and Maxillofacial Surgery Huaxi School of Stomatology
Shujuan Zou, DDS, MS Department of Orthodontics Jing Hu, DDS, MS, PhD Dazhang Wang, DDS, FICD Jihua Li, DDS, MS Zhenglong Tang, DDS, MS Department of Oral and Maxillofacial Surgery Huaxi School of Stomatology
Chapter 5 The Skeletal System:Bone Tissue. Functions of Bone. Bones
 Chapter 5 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose,
Chapter 5 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose,
Tissue Reaction to Root Canal Filling Material Embedded Subcutaneously in Rats
 11 Chapter 2 Tissue Reaction to Root Canal Filling Material Embedded Subcutaneously in Rats Takanaga Ochiai Keisuke Nakano Hiromasa Hasegawa Toshiyuki Kawakami Abstract The reaction of subcutaneous connective
11 Chapter 2 Tissue Reaction to Root Canal Filling Material Embedded Subcutaneously in Rats Takanaga Ochiai Keisuke Nakano Hiromasa Hasegawa Toshiyuki Kawakami Abstract The reaction of subcutaneous connective
Original Article. Masako Yanagawa a, Kenji Fueki b and Takashi Ohyama c
 J Med Dent Sci 2004; 51: 115 119 Original Article Influence of length of food platform on masticatory performance in patients missing unilateral mandibular molars with distal extension removable partial
J Med Dent Sci 2004; 51: 115 119 Original Article Influence of length of food platform on masticatory performance in patients missing unilateral mandibular molars with distal extension removable partial
Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis
 Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis Yusuke Kameda, Masahiko Takahata, Tomohiro Shimizu, Hiroki Hamano, Norimasa Iwasaki. Department of Orthopedic Surgery, Hokkaido
Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis Yusuke Kameda, Masahiko Takahata, Tomohiro Shimizu, Hiroki Hamano, Norimasa Iwasaki. Department of Orthopedic Surgery, Hokkaido
