Review. What understanding tendon cell differentiation can teach us about pathological tendon ossification
|
|
|
- Dorthy Shelton
- 6 years ago
- Views:
Transcription
1 Histol Histopathol (2015) 30: Histology and Histopathology Cellular and Molecular Biology Review What understanding tendon cell differentiation can teach us about pathological tendon ossification D. Magne and C. Bougault UMR CNRS 5246, University Claude Bernard-Lyon 1, University of Lyon, Institute for Molecular and Supramolecular Chemistry and Biochemistry, Villeurbanne, France Summary. Tendons are the structures that attach muscles to bones and transmit mechanical forces. Tendon cells are composed of mature tenocytes and a rare population of tendon stem cells. Both cell types ensure homeostasis and repair of tendon extracellular matrix to guarantee its specific mechanical properties. Moreover, tendon cells seem to present a marked potential for trans-differentiation, predominantly into the chondrocyte and osteoblast lineages. In this review article, we first present chronic tendon pathologies associated with abnormal ossification, such as spondyloarthritis and calcifying tendinopathy, and discuss how tendon cell differentiation and transdifferentiation may participate in these diseases. We moreover present the factors known to influence tendon cell differentiation and trans-differentiation, with a particular emphasis on extracellular environment, mechanical stimulation and several soluble factors that can tip the balance toward one or another lineage. A better understanding of the neglected tendon cell biology may be extremely useful to understand the pathological mechanisms of spondyloarthritis and calcifying tendinopathy. Key words: Tenocytes, Tendom stem cells, Transdifferentiation, Pathological ossification Offprint requests to: C. Bougault, UMR CNRS 2546, Villeurbanne France, University Claude Bernard-Lyon 1, University of Lyon, Institute for Molecular and Supramolecular Chemistry and Biochemistry, Villeurbanne, France. carole.bougault@univ-lyon1.fr DOI: /HH Introduction Tendons attach muscles to bones and their essential role is transferring contraction forces. This function explains why tendon physiology and pathology are highly dependent on mechanical stress. As connective tissues, tendons are mainly composed of an organized extracellular matrix (ECM), which confers to the tissue its mechanical properties: high strength and good flexibility and elasticity. In tendons, the ECM represents approximately 95% of the tissue volume, with type I collagen fibers accounting for 95% of all ECM components. Collagen fibers are organized in bundles that are arranged parallel to the forces exerted on the tendon (Fig. 1). This axial alignment is essential for tendon-specific mechanical features [for a review more focused on tendon structure and biomechanics, please read (Wang et al., 2012; Weinreb et al., 2014)]. Overall, the tendon is a poorly cellularized tissue (Tozer and Duprez, 2005). Tendon cells are fibroblastlike cells, which present an elongated morphology. They are stuck between the collagen bundles and align along the long axis of the tendon. The mature differentiated resident cells of tendons are called tenocytes. Tenocytes are the dominant contributors to tendon homeostasis and repair, which is to say the dominant contributors to tendon ECM remodeling. In addition, tendons contain a unique cell population of progenitor cells referred to as tendon stem cells (TSCs). Distinct types of tendon pathologies Tendon injuries can result from an acute trauma or from a chronic degenerative process (Sharma and
2 902 Tendon cells in tendon pathologies Maffulli, 2005). Acute tendon injury causes disruption of tendon ECM continuity and thus dramatic loss of mechanical force transmission between muscle and bone. Chronic tendon diseases are classified in two types: inflammatory enthesitis and tendinopathies (Weinreb et al., 2014). Entheses are the sites of insertion of tendon or ligament to bone through a fibrocartilaginous connection (Fig. 2). Inflammatory enthesitis deals with an inflammation in this area and it is a characteristic of spondyloarthritis. Tendinopathy was traditionally named tendinitis. This term recovers any painful state arising in or around tendons due to mechanical, degenerative or overuse disease. Degeneration and disorganization of the tendon structure may lead to full thickness tear. Tendon pathologies associated with excessive ossification Enthesophytes: spondyloarthritic ossification In spondyloarthritis, inflammation first develops in entheses. This severe inflammation (enthesitis) is followed at late stages by an abnormal ossification spreading from the underlying bone to the tendon or ligament. These excessive ossifications are the so-called enthesophytes. The orthotopic new bone formation at enthesis in the pathological context of spondyloarthritis is relatively similar to the normal age-related process of tendon ossification. Bony spur initiation and development involve both endochondral and intramembranous ossifications. However, enthesophytes look Fig. 1. Achilles tendon of 12-week-old mice (hematoxylin-eosin staining). The double arrow indicates the axis of the main mechanical forces within the tendon (T). Collagen fibers align with this axis and tendon cells are noticeable as dark spots organized in columns between the collagen bundles. Bony (B) and fatty (F) structures can be seen on the top. less well-organized and structures called cartilage metaplasia can appear in tendons and/or bones (Benjamin et al., 2009; Lories and Schett, 2012). Very small spurs are seen in patients with early spondyloarthritis and in normal subjects, whereas large spurs are characteristic of patients with late spondyloarthritis (McGonagle et al., 2008). Calcifying tendinopathy Calcifying tendinopathy (or calcific tendinitis) is a subset of tendinopathy characterized by the presence of calcifications inside tendons. This abnormal ossification deteriorates tendon structure and worsens the clinical symptoms. New bone formation in the pathological context of calcifying tendinopathy is also suggested to resemble endochondral ossification. Fibrocartilage structures emerge in the diseased tendon, and progress toward calcification (Uhthoff et al., 1976; Fenwick et al., 2002). Pathological behavior of tendon cells Increasing evidence indicates that the abnormal ossification of the diseased tendon in calcifying tendinopathy or the diseased enthesis in spondyloarthritis is due to a pathological behavior of tendon cells (Rui et al., 2011b; Lories and Schett, 2012). Both tenocytes and TSCs may be involved in this pathological process. Tenocytes may be responsible for tendon matrix degeneration and degradative events and TSCs may present aberrant functions due to modifications of their microenvironment (Clegg et al., 2007; Lui and Chan, 2011). Several studies reported an altered fate of tendon cells in distinct pathological contexts. For instance, an Fig. 2. Achilles tendon of 12-week-old mice (hematoxylin-eosin staining). The circle indicates the enthesis: site of tendon (T) insertion to bone (B) through a cartilaginous zone (C).
3 Tendon cells in tendon pathologies 903 overuse protocol showed in adult rats that excessive mechanical stimulation increases gene expression of cartilage markers in tendon cells (Archambault et al., 2007). A collagenase-induced tendon injury model was also used to show that TSCs express more chondrocyte and osteoblast markers and less tenocyte markers, compared to healthy TSCs. Moreover, upon stimulation, the trans-differentiation potential of degenerative TSCs toward chondrogenic and osteogenic lineage appears enhanced (Rui et al., 2013a). Analysis of tissue from human Achilles tendon lesions has also featured an upregulation of cartilage matrix and marker genes (de Mos et al., 2009). As discussed below, tendon cells are known to be highly mechanosensitive. Yet, it seems that cyclic strain has no effect on tendon cells isolated from patients with tendinopathy. Thus, degenerative tendon cells present an altered response to mechanical stress (Choi et al., 2014). This aberrant mechanosensitivity may be due to dysregulation in cell-matrix interactions and/or aberrant actin cytoskeleton organization (Kohler et al., 2013). Growth factors likely involved in tendon cell pathological behavior Mediators of pathological behavior of tendon cells have essentially been studied by Lui and colleagues in the model of tendon ossification induced by injection of collagenase into the patellar tendon of rats. Bone morphogenetic proteins (BMPs) have proven to be highly implicated. BMPs are a subfamily belonging to the transforming growth factor (TGF)-β superfamily. The BMP family includes 20 members, which are active as secreted homo- or hetero-dimers. They are particularly well-known for their ability to induce ectopic formation of bone and cartilage. Immunohistochemical staining proved the presence of BMP-2 but also -4 and -7 in ossifying tendons. BMP-2 was especially located around chondrocyte-like cells (Lui et al., 2009; Yee et al., 2011). At the cellular level, higher expression of BMP ligands and specific receptors (BMPRs) has been detected in degenerative TSCs than in healthy ones. Signal transduction through BMPRs results in phosphorylation of downstream Smad pathway targets. Accordingly, upon stimulation, diseased TSC showed enhanced BMP-Smad sensitivity (Lui and Wong, 2013). Moreover, clinical data corroborate the putative role of BMP signaling in tendon pathology associated with abnormal ossification. BMP-2 and -6 precursors have indeed been found in synovial tissue biopsies and activation of the BMP pathway has been detected in enthesis biopsies from spondyloarthritic patients (Lories et al., 2003, 2005). Furthermore, differential expression of BMP-2, -4, -6 and -7 has been shown in patients with degeneration or calcific tendinopathy of the rotator cuff (Neuwirth et al., 2006; Oliva et al., 2011). Alongside the TGF-β superfamily, Wnt proteins are also preponderant regulators of tissue development and maintenance. The Wnt family includes 19 secreted lipidmodified signaling glycoproteins, whose downstream pathways can be canonical (involving the protein β- catenin) or non-canonical. Of interest, activation of the canonical Wnt pathway has also been shown in the rat ossifying tendon model (with an increased expression of Wnt-3a and β-catenin) and in some patients with patellar tendinopathy (Lui et al., 2013). Two cell populations in tendon: tenocytes and tendon stem cells (TSCs) Understanding tendon cell biology and contribution of tenocytes and TSCs to tissue homeostasis and repair should help to understand the development of tendon pathologies. Tendon stem cells TSCs have recently been identified and characterized (Bi et al., 2007). They possess stem cell characteristics of clonogenicity, multipotency and selfrenewal. First identified in human and mouse tendons, TSCs have been isolated from rat and rabbit tendons since then (Rui et al., 2010; Zhang and Wang, 2010b). This unique population may represent 1 to 3% of the tendon cells. Among mouse tendon-derived cells, the percentage of colony-forming cells has been recorded around 3%, and that of slow-cycling cells around 6%. Comparably, the percentage of TSCs within rat tendonderived cells has been estimated at around 1-2%. The multi-differentiation potential of TSCs includes differentiation into tenocytes, adipocytes, chondrocytes and osteoblasts. Their osteogenic, chondrogenic and adipogenic multipotent differentiation capacity appears to be greater than that of bone marrow stromal cells (Bi et al., 2007; Tan et al., 2012). Upon osteogenic stimulation, faster accumulation of calcium and a higher number of nodules have been observed, together with higher expression of osteogenic markers. Upon chondrogenic induction in pellet culture, type II collagen and glycosaminoglycan deposition and chondrogenic marker expression is also increased. Adipogenic conditions induce adipocyte marker expression and more lipid droplets have been observed in TSC than in bone marrow stromal cells. The tenogenic capacity of TSC seems to vary depending on their position within the tissue. TSC are mostly located in the peritendinous zone and the tendon-bone junction, but also in the tendon core (Tan et al., 2013). Tendon core-derived progenitors show a higher tenogenic potential than progenitors from the peritendinous tissue; they express greater levels of tenogenic markers and produce a more tendon-like ECM (Mienaltowski et al., 2014). Tenocytes Tendon-specific mechanical properties rely on an ECM free from defect. The influence of tenocytes on tissue tensile properties is negligible (Hammer et al., 2014) but their role is to maintain ECM integrity.
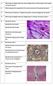
4 904 Tendon cells in tendon pathologies Tenocytes produce large amounts of ECM, in particular type I collagen (Fig. 3), which is the major component of the tendon ECM (about 75% of tendon dry weight). Type I collagen is a heterotrimer helical molecule consisting of two α-1 chains and one α-2 chain. Of note, replacement of the α-2 chain by an α-1 chain, which occurs in the case of osteogenesis imperfecta, results in reduced mechanical strength of the tendons (Chang et al., 2012). Type I collagen fibers are organized with other minor types of collagens, such as the fibrillar type III collagen. Other matrix proteins are also produced by tenocytes, in particular elastin, which forms extensible cross-linked chains that provide elasticity to the ECM, and tenascin C, which is a homohexameric glycoprotein that modulates cell adhesion to the ECM. Tenocytes also produce various proteoglycans, including decorin, which binds to collagen fibers and may affect the collagen fibrillation. In addition, tenocytes are characterized by expression of tenomodulin (Tnmd), also known as tendin. Tenomodulin is a transmembrane glycoprotein belonging to the chondromodulin-1 family that is strongly expressed in tendons and ligaments (Brandau et al., 2001). Tnmd-deficient mice (Docheva et al., 2005) show a decrease in tenocyte proliferation that leads to a reduced tenocyte density. The amount of ECM proteins is not affected, but collagen fibril diameter is abnormal. Therefore, tenomodulin appears crucial for tendon physiology as it seems to regulate tenocyte proliferation and collagen fibrillation (Docheva et al., 2005). Expression of tenocyte specific markers is under the control of one important transcription factor: scleraxis (Scx). This basic helix-loop-helix (bhlh) transcription factor requires dimerization with another bhlh protein for efficient DNA binding to the E-box consensus sequence (Cserjesi et al., 1995). Its expression can be used as a marker of progenitors and mature cells of all tendon tissues (Schweitzer et al., 2001). Scx was shown to regulate in particular the transcription of Col1a1 and Col1a2, coding type I collagen α-1 and α-2 chains (Léjard et al., 2007; Bagchi and Czubryt, 2012). Scleraxis thus appears to be involved in the transcriptional regulation of key ECM components specifically in tendon cells. Scx-deficient mice (Murchison et al., 2007) show severe defects in tendons associated with reduced and disorganized ECM, revealing that scleraxis is crucial for tendon differentiation, especially in force-transmitting tendons, by opposition to muscle-anchoring tendons. Tenogenic differentiation Like other musculoskeletal cells such as myoblasts, chondrocytes and osteoblasts, during embryonic development, tenocytes derive from mesodermal progenitor cells (Tozer and Duprez, 2005). Cell orientation toward the tendon lineage goes along with a persistent expression of the master gene scleraxis and an increasing expression of tenomodulin at the late stages (Schweitzer et al., 2001; Docheva et al., 2005). In addition to scleraxis, another transcription factor called mohawk (Mkx) has recently been shown to be a critical regulator of tenogenic differentiation (Ito et al., 2010). Tendons of Mkx-deficient mice show defects in collagen fibrillation (Ito et al., 2010; Liu et al., 2010), suggesting that mohawk, like scleraxis, is involved in tendon ECM regulation. The more we learn about physiological tenogenic differentiation, the better we will know how to treat tendon pathologies. Any means that could enhance tenogenic differentiation is of particular interest in the context of tendon tissue engineering. Indeed, cell therapy in tendon disease is a current hot spot of the field (Ho et al., 2014). Soluble differentiation factors Fig. 3. Immunofluorescence staining of type I collagen in cultured tendon cells from new- born mouse tail tendons. Nucleus was stained by Hoechst (in blue). All tendon cells are producing type I collagen (in green). Several studies have highlighted the capacity of tendon tissue or tenocytes to orientate stem cell differentiation toward the tenogenic lineage. For instance, indirect co-cultures of bone marrow mesenchymal stem cells (MSCs) with tendon tissue fragments have tenogenic effects (Lovati et al., 2012). Similarly, direct co-culture of MSCs with primary tenocytes upregulates tenocyte markers including type I collagen, tenomodulin and scleraxis (Schneider et al., 2011). Furthermore, numerous intercellular contacts between MSCs and tenocytes have been reported suggesting an extensive exchange of soluble factors. Among soluble tenogenic factors, particular attention was paid to the growth and differentiation factor (GDF) family. GDFs are members of TGF-β superfamily proteins, such as the previously mentioned BMPs. GDF proteins (GDF-1 to GDF-15) are active as secreted homodimers that are particularly involved in development. GDF-5 was in particular shown to promote differentiation of TSCs towards tenocytes, by upregulating ECM proteins such as type I collagen or
5 Tendon cells in tendon pathologies 905 decorin, and tendon specific markers such as scleraxis and tenomodulin, in particular (Holladay et al., 2015; Mienaltowski et al., 2014). These tenogenic effects of GDF-5 are also observed on bone-marrow derived MSCs, adipose tissue-derived stem cells or periodontal ligament cells (Tan et al., 2012; Raabe et al., 2013; Xia et al., 2013). However, GDF-5 is known to affect various other skeletal tissues such as intervertebral disk, cartilage and bone (Mikic, 2004). In some in vitro studies, GDF-5 seemed to reduce adipogenic, chondrogenic and osteogenic pathways along with favoring tenogenic differentiation (Tan et al., 2012a,b; Xia et al., 2013; Holladay et al., 2015). Similarly, GDF-7 (also called BMP-12) was shown to activate tendon transcription factors including scleraxis and mohawk and tendon markers including tenomodulin and decorin in bone marrow MSCs (Violini et al., 2009; Otabe et al., 2015). GDF-7 has tenogenic effects on adipose tissue- derived stem cells comparable to those of GDF-5 (Raabe et al., 2013; Shen et al., 2013). However, GDF-7-induced orientation toward tenocytes lineage seems less frank than that induced by GDF-5 because some chondrogenic and osteogenic markers seem also to be upregulated by GDF-7 (Shen et al., 2013). Extracellular three-dimensional environment Because in situ tenocytes are embedded in a highly specific ECM, the role of tendon matrix components and/or structural organization on tenogenic differentiation is likely crucial. Indeed, tendon-derived decellularized ECM promotes tenogenic differentiation in TSCs while inhibiting osteogenesis (Yin et al., 2013). The tenogenic potential of tendon matrix components on adipose tissue stem cells has been assessed by Yang and colleagues: the addition of urea-extracted fraction of tendon ECM, which contains critical tendon ECM components, to a collagen gel, increases tenogenic markers tenomodulin, scleraxis and tenascin C, while reducing osteogenic markers (Yang et al., 2013). The structural organization of the extracellular environment is also significant for tenogenic differentiation. Tissue-engineered scaffolds should be shaped as dense and aligned fibers to mimic tendon ECM collagen network. Indeed, a synthetic matrix composed of aligned collagen threads has been shown to enhance tenogenic differentiation of MSCs and adipose tissue-derived stem cells, compared to randomly oriented collagen, without osteogenic transdifferentiation (Kishore et al., 2012; Cheng et al., 2014). Mechanical stimulation Tendons are subjected to high mechanical stress and tenocytes are surrounded by a mechanoactive environment. Cyclic stretching has been shown to promote tenogenic differentiation of bone marrow MSCs, with an increase in tenogenic marker expression and tendon ECM deposition (type I collagen and tenascin C, in particular) (Kuo and Tuan, 2008; Morita et al., 2013; Nam et al., 2015). This differentiation was efficiently induced by 8 to 10% uniaxial stretching stimuli and seemed associated with cell orientation angle and cumulative elongation load on the cells (Morita et al., 2013; Nam et al., 2015). RhoA/ROCK, cytoskeletal organization and focal adhesion kinase appears to be part of the signaling pathways that drive mechanical stretchinduced tenogenic differentiation of MSC (Xu et al., 2011, 2012). Furthermore, dynamic stretching seems required to preserve tenocyte phenotype over time. In MSC culture, mechanical stress is necessary to sustain expression of the tenocyte marker scleraxis (Kuo and Tuan, 2008). Similarly, in the absence of mechanical stress, allogeneic tenocytes seeded in tendon scaffolds fail to preserve ECM homeostasis, whereas under cyclic strain, they maintain the construct s tensile properties (Whitlock et al., 2013). Combined strategies for tendon tissue engineering Now that various tenogenic factors are identified, new approaches currently test different combinations of them to further optimize tenogenic differentiation (Fig. 4). The main objective of these studies is to develop functional constructs for tendon tissue engineering. For F i g. 4. F a c t o r s i n f l u e n c i n g t e n o g e n i c differentiation. The transcription factors scleraxis ( scx) and Mohawk ( mkx) are particularly involved in cell orientation toward the tenogenic lineage. Soluble differentiation mediators, extracellular m a t r i x ( E C M ) t h r e e - d i m e n s i o n a l environment and mechanical stimulation can favor tenogenesis in combination. Tendon tissue engineering strategies tend to associate multiple tenogenic elements.
6 906 Tendon cells in tendon pathologies instance, tendon ECM-mimicking scaffolds can be associated with mechanical stimulation to enhance MSC differentiation toward tenocyte lineage (Teh et al., 2013; Yang et al., 2013; Czaplewski et al., 2014). Dynamic culture conditions can favor the intensity and onset of differentiation markers, accelerate tendon matrix deposition and improve tensile properties of the constructs. Mechanical forces and three dimensional environments can be further combined with soluble factors to synergistically enhance stem cell tenogenic differentiation (Raabe et al., 2013; Barsby et al., 2014). Trans-differentiation potential of tendon cells As discussed above, several tendon pathologies are associated with abnormal ossification. Studying chondro- or osteogenic trans-differentiation of tendon cells may provide new tools to address cellular events in pathological context. The multipotency of TSCs has been proven for tenogenic, adipogenic, chondrogenic and osteogenic lineages (Bi et al., 2007; Rui et al., 2010; Zhang and Wang, 2010b; Tan et al., 2012a,b). In addition, mature tenocytes also show an intrinsic transdifferentiation potential and can turn into adipocytes, chondrocytes or osteoblasts (de Mos et al., 2007). Thus, supplemental studies were conducted to isolate the molecular mechanisms that can favor tendon cell transdifferentiation in one or another cell lineage. Proximity of tenogenic and chondrogenic lineages during development During embryonic development, tendon morphogenesis is strongly associated with chondrogenesis. Even if considered as a tendon marker, scleraxis seems to have multiple roles during mouse embryogenesis, and is described as a regulator of gene expression in mesoderm formation and chondrogenesis (Cserjesi et al., 1995; Brown et al., 1999). The balance between sox9 and scleraxis, key transcription factors of chondrocytes and tenocytes respectively, appears to be tightly regulated during the coordination of tendon and skeleton development (Asou et al., 2002). A unique pool of progenitor cells expressing both sox9 and scleraxis has been characterized. These cells can give rise to both chondrocytes and tenocytes and are expected to contribute to the establishment of the junction between cartilage and tendon (Blitz et al., 2013; Sugimoto et al., 2013). Indeed, inactivation of sox9 in those cells causes defects in tendon-bone structure formation. TGF-β F i g. 5. B a l a n c e b e t w e e n t e n o g e n i c differentiation and chondro- or osteogenic t r a n s - d i f f e r e n t i a t i o n. E x t r a c e l l u l a r environment, mechanical stimulation and various soluble factors from GDF, BMP and Wnt families can tip the equilibrium toward one or another lineage. Cellular switch from physiological tenogenesis to pathological chondro- or osteogenesis may lead to abnormal ossification.
7 Tendon cells in tendon pathologies 907 superfamily proteins seem especially involved in regulating the balance between sox9 and scleraxis and therefore in the shift between chondrogenesis and tenogenesis. TGF-β ligands have been shown to regulate the emergence of bone attachment structures from sox9- and scx-positive cells (Blitz et al., 2013). Similarly, TGF-β ligands (including TGF-β1 and 2) down-regulate sox9 and up-regulate scleraxis and tenomodulin in micromass cultures of mesenchymal cells (Lorda-Diez et al., 2009). Chondrogenic trans-differentiation Concerning chondrogenic trans-differentiation of tendon cells, particular attention was paid to the BMP family of growth factors. For instance, injection of BMP-7 into rat Achilles tendon induces transdifferentiation of tendon cells towards chondrocyte-like cells and changes the surrounding ECM composition with an enrichment in cartilage-specific proteins, i.e. glycosaminoglycan and type II collagen (Ozeki et al., 2013). In response to BMP-2, tenogenic marker expression is decreased in TSCs, whereas chondrogenic orientation is enhanced, as shown by an increase in glycosaminoglycan deposition (Rui et al., 2013b). Tendon cells grown in pellet culture in chondrogenic medium have the same differentiation potential as MSCs (Funakoshi and Spector, 2010). Overall, tenocytes proved to have a strong potential for conversion into chondrocytes. Osteogenic trans-differentiation Osteogenic trans-differentiation seems to be greatly influenced by the extracellular three dimensional environment. As tendon decellularized ECM can turn TSC toward tenogenic lineage, bone decellularized ECM can strongly turn them toward osteogenic cells (Yin et al., 2013). Interestingly, alteration of TSC nicheassociated ECM by genetic inactivation of two small proteoglycans led to tendon malformation and abnormal ossification (Bi et al., 2007). In these mice, TSCs switched from tenogenesis to osteogenesis through an ectopic activation of BMP signaling. Lots of cytokines and growth factors are stored within the ECM and the authors suggested that modifications in its composition may possibly perturb the differentiation cocktail delivered onto TSC. Moreover, TSCs can also be induced to transdifferentiate into osteogenic lineage by specific mechanical stretching (Zhang and Wang, 2010a; Shi et al., 2012). Stretch-induced osteogenic response of TSC may be partly mediated by Wnt-5a and by BMP-2. An up-regulation of BMP-2 was observed in stretched TSCs and addition of BMP-2 on cultured TSCs increases osteogenic trans-differentiation, leading to characteristic mineralization (Rui et al., 2011a). The strong osteogenic potential of BMP-2 on tendon cells has been demonstrated several times. For example, overexpression of BMP-2 in tenocytes is sufficient to induce the osteogenic mineralization process (Murray et al., 2010). Similarly, mineralization is induced by addition of BMP-2 in TSC cultures (Rui et al., 2012). BMP-2- mediated osteogenic trans-differentiation of TSCs was also observed in response to prostaglandin E2, suggesting that BMP-Smad pathway may be activated by inflammatory disorders (Zhang and Wang, 2012; Liu et al., 2013). Alternatively, increased mineralization and expression of osteogenic markers have also been observed in TSCs in response to Wnt signaling activation by Wnt-3a (Lui et al., 2013). Finally, tendon cell capacity to trans-differentiate into the osteogenic lineage seems as strong as into the chondrogenic lineage. Therefore, we can conclude a strong chondro- and osteogenic potential, which could be partly explained by a high sensitivity to BMP signals, as a consequence of high expression levels of BMP receptors (Rui et al., 2012). Conclusion The tenocyte phenotype must be tightly regulated to preserve tissue homeostasis and to maintain tendonspecific mechanical properties. Control of tenogenic differentiation of both mature differentiated tenocytes and TSCs appears essential in tendon development and remodeling but also in the repair process. Tendon cells proved to have a strong potential for chondro- or osteogenic trans-differentiation, and the balance between tenogenesis and chondro- or osteogenesis seems tenuous. A set of factors, including the three dimensional environment, mechanical stress, GDF, BMP and Wnt signaling have been shown to participate in this equilibrium (Fig. 5). Different tendon pathologies, including spondyloarthritis and calcifying tendinopathy, cause cartilage metaplasia, which further degenerates into abnormal ossifications. These ectopic calcifications in the tendon core or in enthesis lead to dramatic loss of force transmission between muscle and bone and thus to significant disability for the patients. A better understanding of tendon cell biology therefore deserves investigation. References Archambault J.M., Jelinsky S.A., Lake S.P., Hill A.A., Glaser D.L. and Soslowsky L.J. (2007). Rat supraspinatus tendon expresses cartilage markers with overuse. J. Orthop. Res. 25, Asou Y., Nifuji A., Tsuji K., Shinomiya K., Olson E.N., Koopman P. and Noda M. (2002). Coordinated expression of scleraxis and sox9 genes during embryonic development of tendons and cartilage. J. Orthop. Res. 20, Bagchi R.A. and Czubryt M.P. (2012). Synergistic roles of scleraxis and smads in the regulation of collagen 1α2 gene expression. Biochim. Biophys. Acta 1823, Barsby T., Bavin E.P. and Guest D.J. (2014). Three-dimensional culture and transforming growth factor beta3 synergistically
8 908 Tendon cells in tendon pathologies promote tenogenic differentiation of equine embryo-derived stem cells. Tissue Eng. Part A 20, Benjamin M., Toumi H., Suzuki D., Hayashi K. and McGonagle D. ( ). E v i d e n c e f o r a d i s t i n c t i v e p a t t e r n o f b o n e formation in enthesophytes. Ann. Rheum. Dis. 68, Bi Y., Ehirchiou D., Kilts T.M., Inkson C.A., Embree M.C., Sonoyama W., Li L., Leet A.I., Seo B.M., Zhang L., Shi S. a n d Y o u n g M. F. ( ). I d e n t i f i c a t i o n o f t e n d o n stem/progenitor cells and the role of the extracellular matrix in their niche. Nat. Med. 13, Blitz E., Sharir A., Akiyama H. and Zelzer E. (2013). Tendonbone attachment unit is formed modularly by a distinct pool of scx- and sox9-positive progenitors. Development 140, Brandau O., Meindl A., Fässler R. and Aszódi A. (2001). A novel gene, tendin, is strongly expressed in tendons and ligaments and shows high homology with chondromodulin-i. Dev. Dyn. 221, Brown D., Wagner D., Li X., Richardson J.A. and Olson E.N. (1999). Dual role of the basic helix-loop-helix transcription factor scleraxis in mesoderm formation and chondrogenesis during mouse embryogenesis. Development 126, Chang S.W., Shefelbine S.J. and Buehler M.J. (2012). Structural and mechanical differences between collagen homo- and heterotrimers: Relevance for the molecular origin of brittle bone disease. Biophys. J. 102, Cheng X., Tsao C., Sylvia V.L., Cornet D., Nicolella D.P., Bredbenner T.L. and Christy R.J. (2014). Platelet-derived growth-factor-releasing aligned collagen-nanoparticle fibers promote the proliferation and tenogenic differentiation of adipose-derived stem cells. Acta Biomater. 10, Choi W.J., Park M.S., Park K.H., Courneya J.P., Cho J.S., Schon L.C. and Lee J.W. (2014). Comparative analysis of gene expression in normal and degenerative human tendon cells: Effects of cyclic strain. Foot Ankle Int. 35, C l e g g P. D., S t r a s s b u r g S. a n d S m i t h R. K. ( ). C e l l phenotypic variation in normal and damaged tendons. Int. J. Exp. Pathol. 88, Cserjesi P., Brown D., Ligon K.L., Lyons G.E., Copeland N.G., Gilbert D.J., Jenkins N.A. and Olson E.N. (1995). Scleraxis: A basic helix-loop-helix protein that prefigures skeletal formation during mouse embryogenesis. Development 121, Czaplewski S.K., Tsai T.L., Duenwald-Kuehl S.E., Vanderby R. and Li W.J. (2014). Tenogenic differentiation of human induced pluripotent stem cell-derived mesenchymal stem cells dictated by properties of braided submicron fibrous scaffolds. Biomaterials 35, de Mos M., Koevoet W.J., Jahr H., Verstegen M.M., Heijboer M.P., Kops N., van Leeuwen J.P., Weinans H., Verhaar J.A. and van Osch G.J. (2007). Intrinsic differentiation potential o f a d o l e s c e n t h u m a n t e n d o n t i s s u e : A n i n - v i t r o c e l l differentiation study. BMC Musculoskelet. Disord. 8, 16. de Mos M., Koevoet W., van Schie H.T., Kops N., Jahr H., Verhaar J.A. and van Osch G.J. (2009). In vitro model to study chondrogenic differentiation in tendinopathy. Am. J. Sports Med. 37, Docheva D., Hunziker E.B., Fässler R. and Brandau O. (2005). Tenomodulin is necessary for tenocyte proliferation and tendon maturation. Mol. Cell. Biol. 25, F e n w i c k S., H a r r a l l R., H a c k n e y R., B o r d S., H o r n e r A., Hazleman B. and Riley G. (2002). Endochondral ossification in achilles and patella tendinopathy. Rheumatology (Oxford) 41, F u n a k o s h i T. a n d S p e c t o r M. ( ). C h o n d r o g e n i c d i f f e r e n t i a t i o n a n d l u b r i c i n e x p r e s s i o n o f c a p r i n e infraspinatus tendon cells. J. Orthop. Res. 28, Hammer N., Huster D., Fritsch S., Hädrich C., Koch H., Schmidt P., Sichting F., Wagner M.F. and Boldt A. (2014). Do cells contribute to tendon and ligament biomechanics? PLoS One 9, e Ho J.O., Sawadkar P. and Mudera V. (2014). A review on the use of cell therapy in the treatment of tendon disease and injuries. J. Tissue Eng. 5, Holladay C., Abbah S.A., O'Dowd C., Pandit A. and Zeugolis D.I. (2015). Preferential tendon stem cell response to growth factor supplementation. J. Tissue Eng. Regen. Med. (in press). Ito Y., Toriuchi N., Yoshitaka T., Ueno-Kudoh H., Sato T., Yokoyama S., Nishida K., Akimoto T., Takahashi M., Miyaki S. and Asahara H. (2010). The mohawk homeobox gene is a critical regulator of tendon differentiation. Proc. Natl. Acad. Sci. USA 107, Kishore V., Bullock W., Sun X., Van Dyke W.S. and Akkus O. (2012). Tenogenic differentiation of human mscs induced by t h e t o p o g r a p h y o f e l e c t r o c h e m i c a l l y a l i g n e d c o l l a g e n threads. Biomaterials 33, Kohler J., Popov C., Klotz B., Alberton P., Prall W.C., Haasters F., Müller-Deubert S., Ebert R., Klein-Hitpass L., Jakob F., Schieker M. and Docheva D. (2013). Uncovering the cellular and molecular changes in tendon stem/progenitor cells attributed to tendon aging and degeneration. Aging Cell 12, Kuo C.K. and Tuan R.S. (2008). Mechanoactive tenogenic differentiation of human mesenchymal stem cells. Tissue Eng. Part A 14, Léjard V., Brideau G., Blais F., Salingcarnboriboon R., Wagner G., Roehrl M. H., Noda M., Duprez D., Houillier P. and R o s s e r t J. ( ). S c l e r a x i s a n d n f a t c r e g u l a t e t h e expression of the pro-alpha1(i) collagen gene in tendon fibroblasts. J. Biol. Chem. 282, Liu W., Watson S.S., Lan Y., Keene D.R., Ovitt C.E., Liu H., S c h w e i t z e r R. a n d J i a n g R. ( ). T h e a t y p i c a l homeodomain transcription factor mohawk controls tendon morphogenesis. Mol. Cell. Biol. 30, Liu J., Chen L., Tao X. and Tang K. (2013). Phosphoinositide 3- kinase/akt signaling is essential for prostaglandin e2-induced osteogenic differentiation of rat tendon stem cells. Biochem. Biophys. Res. Commun. 435, Lorda-Diez C.I., Montero J.A., Martinez-Cue C., Garcia-Porrero J.A. and Hurle J.M. (2009). Transforming growth factors beta c o o r d i n a t e c a r t i l a g e a n d t e n d o n d i f f e r e n t i a t i o n i n t h e developing limb mesenchyme. J. Biol. Chem. 284, Lories R.J. and Schett G. (2012). Pathophysiology of new bone formation and ankylosis in spondyloarthritis. Rheum. Dis. Clin. North Am. 38,
9 Tendon cells in tendon pathologies 909 Lories R.J., Derese I., Ceuppens J.L. and Luyten F.P. (2003). Bone morphogenetic proteins 2 and 6, expressed in arthritic synovium, are regulated by proinflammatory cytokines and differentially modulate fibroblast-like synoviocyte apoptosis. Arthritis Rheum. 48, Lories R.J., Derese I. and Luyten F.P. (2005). Modulation of bone morphogenetic protein signaling inhibits the onset and progression of ankylosing enthesitis. J. Clin. Invest. 115, Lovati A. B., Corradetti B., Cremonesi F., Bizzaro D. and Consiglio A.L. (2012). Tenogenic differentiation of equine mesenchymal progenitor cells under indirect co-culture. Int. J. Artif. Organs 35, Lui P.P. and Chan K.M. (2011). Tendon-derived stem cells (tdscs): From basic science to potential roles in tendon pathology and tissue engineering applications. Stem Cell Rev. 7, Lui P.P. and Wong Y. (2013). Higher bmp/smad sensitivity of t e n d o n - d e r i v e d s t e m c e l l s ( t d s c s ) i s o l a t e d f r o m t h e c o l l a g e n a s e - i n d u c e d t e n d o n i n j u r y m o d e l : P o s s i b l e mechanism for their altered fate in vitro. BMC Musculoskelet. Disord. 14, 248. Lui P.P., Chan L.S., Cheuk Y.C., Lee Y.W. and Chan K.M. (2009). Expression of bone morphogenetic protein-2 in the chondrogenic and ossifying sites of calcific tendinopathy and traumatic tendon injury rat models. J. Orthop. Surg. Res. 4, 27. Lui P.P., Lee Y.W., Wong Y.M., Zhang X., Dai K. and Rolf C.G. (2013). Expression of wnt pathway mediators in metaplasic tissue in animal model and clinical samples of tendinopathy. Rheumatology (Oxford) 52, McGonagle D., Wakefield R.J., Tan A.L., D'Agostino M.A., Toumi H., Hayashi K., Emery P. and Benjamin M. (2008). Distinct topography of erosion and new bone formation in achilles tendon enthesitis: Implications for understanding the l i n k b e t w e e n i n f l a m m a t i o n a n d b o n e f o r m a t i o n i n spondylarthritis. Arthritis Rheum. 58, Mienaltowski M.J., Adams S.M. and Birk D.E. (2014). Tendon proper- and peritenon-derived progenitor cells have unique tenogenic properties. Stem Cell Res. Ther. 5, 86. Mikic B. (2004). Multiple effects of gdf-5 deficiency on skeletal tissues: Implications for therapeutic bioengineering. Ann. Biomed. Eng. 32, Morita Y., Watanabe S., Ju Y. and Xu B. (2013). Determination of optimal cyclic uniaxial stretches for stem cell-to-tenocyte differentiation under a wide range of mechanical stretch c o n d i t i o n s b y e v a l u a t i n g g e n e e x p r e s s i o n a n d p r o t e i n synthesis levels. Acta Bioeng. Biomech. 15, Murchison N.D., Price B.A., Conner D.A., Keene D.R., Olson E.N., Tabin C.J. and Schweitzer R. (2007). Regulation of tendon differentiation by scleraxis distinguishes forcet r a n s m i t t i n g t e n d o n s f r o m m u s c l e - a n c h o r i n g t e n d o n s. Development 134, M u r r a y S. J., S a n t a n g e l o K. S. a n d B e r t o n e A. L. ( ). Evaluation of early cellular influences of bone morphogenetic p r o t e i n s 1 2 a n d 2 o n e q u i n e s u p e r f i c i a l d i g i t a l f l e x o r tenocytes and bone marrow-derived mesenchymal stem cells in vitro. Am. J. Vet. Res. 71, Nam H.Y., Pingguan-Murphy B., Amir Abbas A., Mahmood Merican A. and Kamarul T. (2015). The proliferation and tenogenic differentiation potential of bone marrow-derived mesenchymal stromal cell are influenced by specific uniaxial c y c l i c t e n s i l e l o a d i n g c o n d i t i o n s. B i o m e c h. M o d e l. Mechanobiol. (in press). Neuwirth J., Fuhrmann R.A., Veit A., Aurich M., Stonâns I., T r o m m e r T., H o r t s c h a n s k y P., C h u b i n s k a y a S. a n d Mollenhauer J. A. ( 2006). Expression of bioactive bone morphogenetic proteins in the subacromial bursa of patients with chronic degeneration of the rotator cuff. Arthritis Res. Ther. 8, R92. Oliva F., Barisani D., Grasso A. and Maffulli N. (2011). Gene expression analysis in calcific tendinopathy of the rotator cuff. Eur. Cell. Mater. 21, Otabe K., Nakahara H., Hasegawa A., Matsukawa T., Ayabe F., Onizuka N., Inui M., Takada S., Ito Y., Sekiya I., Muneta T., Lotz M. and Asahara H. (2015). Transcription factor mohawk c o n t r o l s t e n o g e n i c d i f f e r e n t i a t i o n o f b o n e m a r r o w mesenchymal stem cells in vitro and in vivo. J. Orthop. Res. 33, 1-8. Ozeki N., Muneta T., Koga H., Katagiri H., Otabe K., Okuno M., Tsuji K., Kobayashi E., Matsumoto K., Saito H., Saito T. and Sekiya I. (2013). Transplantation of achilles tendon treated with bone morphogenetic protein 7 promotes meniscus regeneration in a rat model of massive meniscal defect. Arthritis Rheum. 65, Raabe O., Shell K., Fietz D., Freitag C., Ohrndorf A., Christ H. J., W e n i s c h S. a n d A r n h o l d S. ( ). T e n o g e n i c differentiation of equine adipose-tissue-derived stem cells under the influence of tensile strain, growth differentiation factors and various oxygen tensions. Cell Tissue Res. 352, Rui Y.F., Lui P.P., Li G., Fu S.C., Lee Y.W. and Chan K.M. (2010). Isolation and characterization of multipotent rat tendon-derived stem cells. Tissue Eng. Part A 16, Rui Y.F., Lui P.P., Ni M., Chan L.S., Lee Y.W. and Chan K.M. (2011a). Mechanical loading increased bmp-2 expression which promoted osteogenic differentiation of tendon-derived stem cells. J. Orthop. Res. 29, Rui Y.F., Lui P.P., Chan L.S., Chan K.M., Fu S.C. and Li G. (2011b). Does erroneous differentiation of tendon-derived stem cells contribute to the pathogenesis of calcifying tendinopathy? Chin. Med. J. (Engl) 124, Rui Y.F., Lui P.P., Lee Y.W. and Chan K.M. (2012). Higher bmp r e c e p t o r e x p r e s s i o n a n d b m p i n d u c e d o s t e o g e n i c differentiation in tendon-derived stem cells compared with bone-marrow-derived mesenchymal stem cells. Int. Orthop. 36, Rui Y.F., Lui P.P., Wong Y.M., Tan Q. and Chan K.M. (2013a). Altered fate of tendon-derived stem cells isolated from a failed tendon-healing animal model of tendinopathy. Stem Cells Dev. 22, Rui Y.F., Lui P.P., Wong Y.M., Tan Q. and Chan K.M. (2013b). Bmp-2 stimulated non-tenogenic differentiation and promoted proteoglycan deposition of tendon-derived stem cells (tdscs) in vitro. J. Orthop. Res. 31, Schneider P.R., Buhrmann C., Mobasheri A., Matis U. and Shakibaei M. (2011). Three-dimensional high-density co-
10 910 Tendon cells in tendon pathologies c u l t u r e w i t h p r i m a r y t e n o c y t e s i n d u c e s t e n o g e n i c differentiation in mesenchymal stem cells. J. Orthop. Res. 29, Schweitzer R., Chyung J.H., Murtaugh L.C., Brent A.E., Rosen V., Olson E.N., Lassar A. and Tabin C.J. (2001). Analysis of the tendon cell fate using scleraxis, a specific marker for tendons and ligaments. Development 128, Sharma P. and Maffulli N. (2005). Basic biology of tendon injury and healing. Surgeon 3, Shen H., Gelberman R.H., Silva M.J., Sakiyama-Elbert S.E. and T h o m o p o u l o s S. ( ). B m p 1 2 i n d u c e s t e n o g e n i c differentiation of adipose-derived stromal cells. PLoS One 8, e Shi Y., Fu Y., Tong W., Geng Y., Lui P.P., Tang T., Zhang X. and Dai K. (2012). Uniaxial mechanical tension promoted osteogenic differentiation of rat tendon-derived stem cells (rtdscs) via the wnt5a-rhoa pathway. J. Cell. Biochem. 113, Sugimoto Y., Takimoto A., Akiyama H., Kist R., Scherer G., N a k a m u r a T., H i r a k i Y. a n d S h u k u n a m i C. ( ). Scx+/sox9+ progenitors contribute to the establishment of t h e j u n c t i o n b e t w e e n c a r t i l a g e a n d t e n d o n / l i g a m e n t. Development 140, Tan Q., Lui P.P., Rui Y.F. and Wong Y.M. (2012a). Comparison of potentials of stem cells isolated from tendon and bone marrow for musculoskeletal tissue engineering. Tissue Eng. Part A 18, Tan S.L., Ahmad R.E., Ahmad T.S., Merican A.M., Abbas A.A., N g W. M. a n d K a m a r u l T. ( b ). E f f e c t o f g r o w t h differentiation factor 5 on the proliferation and tenogenic differentiation potential of human mesenchymal stem cells in vitro. Cells Tissues Organs 196, Tan Q., Lui P.P. and Lee Y.W. (2013). In vivo identity of tendon stem cells and the roles of stem cells in tendon healing. Stem Cells Dev. 22, Teh T. K., Toh S. L. and Goh J. C. ( 2013). Aligned fibrous scaffolds for enhanced mechanoresponse and tenogenesis of mesenchymal stem cells. Tissue Eng. Part A 19, T o z e r S. a n d D u p r e z D. ( ). T e n d o n a n d l i g a m e n t : Development, repair and disease. Birth Defects Res. C Embryo Today 75, Uhthoff H.K., Sarkar K. and Maynard J.A. (1976). Calcifying tendinitis: A new concept of its pathogenesis. Clin. Orthop. Relat. Res Violini S., Ramelli P., Pisani L.F., Gorni C. and Mariani P. (2009). Horse bone marrow mesenchymal stem cells express embryo stem cell markers and show the ability for tenogenic differentiation by in vitro exposure to bmp-12. BMC Cell. Biol. 10, 29. Wang J.H., Guo Q. and Li B. (2012). Tendon biomechanics and mechanobiology--a minireview of basic concepts and recent advancements. J. Hand. Ther. 25, ; quiz 141. Weinreb J.H., Sheth C., Apostolakos J., McCarthy M.B., Barden B., Cote M.P. and Mazzocca A.D. (2014). Tendon structure, disease, and imaging. Muscles Ligaments Tendons J. 4, Whitlock P.W., Seyler T.M., Northam C.N., Smith T.L., Poehling G.G., Koman L.A. and Van Dyke M.E. (2013). Effect of cyclic s t r a i n o n t e n s i l e p r o p e r t i e s o f a n a t u r a l l y d e r i v e d, decellularized tendon scaffold seeded with allogeneic tenocytes and associated messenger rna expression. J. Surg. Orthop. Adv. 22, Xia D., Sumita Y., Liu Y., Tai Y., Wang J., Uehara M., Agata H., Kagami H., Fan Z., Asahina I., Wang S. and Tran S.D. (2013). Gdfs promote tenogenic characteristics on human p e r i o d o n t a l l i g a m e n t - d e r i v e d c e l l s i n c u l t u r e a t l a t e passages. Growth Factors 31, Xu B., Song G. and Ju Y. (2011). Effect of focal adhesion kinase on the regulation of realignment and tenogenic differentiation of human mesenchymal stem cells by mechanical stretch. Connect. Tissue Res. 52, Xu B., Song G., Ju Y., Li X., Song Y. and Watanabe S. (2012). Rhoa/rock, cytoskeletal dynamics, and focal adhesion kinase are required for mechanical stretch- induced tenogenic differentiation of human mesenchymal stem cells. J. Cell. Physiol. 227, Yang G., Rothrauff B.B., Lin H., Gottardi R., Alexander P.G. and Tuan R.S. (2013). Enhancement of tenogenic differentiation of human adipose stem cells by tendon-derived extracellular matrix. Biomaterials 34, Yee Lui P.P., Wong Y.M., Rui Y.F., Lee Y.W., Chan L.S. and Chan K.M. (2011). Expression of chondro-osteogenic bmps in ossified failed tendon healing model of tendinopathy. J. Orthop. Res. 29, Yin Z., Chen X., Zhu T., Hu J.J., Song H.X., Shen W.L., Jiang L.Y., Heng B.C., Ji J.F. and Ouyang H.W. (2013). The effect of decellularized matrices on human tendon stem/progenitor cell differentiation and tendon repair. Acta Biomater. 9, Zhang J. and Wang J.H. (2010a). Mechanobiological response of tendon stem cells: Implications of tendon homeostasis and pathogenesis of tendinopathy. J. Orthop. Res. 28, Zhang J. and Wang J.H. (2010b). Characterization of differential properties of rabbit tendon stem cells and tenocytes. BMC Musculoskelet. Disord. 11, 10. Zhang J. and Wang J.H. (2012). Bmp-2 mediates pge(2) - i n d u c e d r e d u c t i o n o f p r o l i f e r a t i o n a n d o s t e o g e n i c differentiation of human tendon stem cells. J. Orthop. Res. 30, Accepted April 8, 2015
Tenogenic Differentiation of Mesenchymal Stem Cells and Their Applications in Tendon Tissue Engineering
 Tenogenic Differentiation of Mesenchymal Stem Cells and Their Applications in Tendon Tissue Engineering Professor Gang Li, MBBS, D Phil (Oxon) Department of Orthopaedics and Traumatology, The Chinese University
Tenogenic Differentiation of Mesenchymal Stem Cells and Their Applications in Tendon Tissue Engineering Professor Gang Li, MBBS, D Phil (Oxon) Department of Orthopaedics and Traumatology, The Chinese University
Introduction to Biomedical Engineering
 Introduction to Biomedical Engineering FW 16/17, AUT Biomechanics of tendons and ligaments G. Rouhi Biomechanics of tendons and ligaments Biomechanics of soft tissues The major soft tissues in musculoskeletal
Introduction to Biomedical Engineering FW 16/17, AUT Biomechanics of tendons and ligaments G. Rouhi Biomechanics of tendons and ligaments Biomechanics of soft tissues The major soft tissues in musculoskeletal
ORTHOPEDICS BONE Recalcitrant nonunions In total hip replacement total knee surgery increased callus volume
 ORTHOPEDICS Orthopedics has to do with a variety of tissue: bone, cartilage, tendon, ligament, muscle. In this regard orthopedic and sports medicine share the same tissue targets. Orthopedics is mostly
ORTHOPEDICS Orthopedics has to do with a variety of tissue: bone, cartilage, tendon, ligament, muscle. In this regard orthopedic and sports medicine share the same tissue targets. Orthopedics is mostly
Tendon stem cells: experimental and clinical perspectives in tendon and tendon-bone junction repair
 Review article Tendon stem cells: experimental and clinical perspectives in tendon and tendon-bone junction repair Pauline Po Yee Lui 1,2,3,, On Tik Wong 1,2 1 Department of Orthopaedics and Traumatology,
Review article Tendon stem cells: experimental and clinical perspectives in tendon and tendon-bone junction repair Pauline Po Yee Lui 1,2,3,, On Tik Wong 1,2 1 Department of Orthopaedics and Traumatology,
Bone. Development. Tim Arnett. University College London. Department of Anatomy and Developmental Biology
 Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION
 A L L O S O U R C E PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION Ryan Delaney MS; Carolyn Barrett BS, MBA; Peter Stevens PhD, MBA AlloSource,
A L L O S O U R C E PROCHONDRIX CARTILAGE RESTORATION MATRIX CONTAINS GROWTH FACTORS NECESSARY FOR HYALINE CARTILAGE REGENERATION Ryan Delaney MS; Carolyn Barrett BS, MBA; Peter Stevens PhD, MBA AlloSource,
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products
 stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
Ming Hao Zheng, PhD, DM, FRC Path Centre For Orthopaedic Research, The University of Western Australia; Paul Anderson MD Orthocell Pty Ltd
 Autologous Tenocyte Implantation for Refractory Tendonopathy Ming Hao Zheng, PhD, DM, FRC Path Centre For Orthopaedic Research, The University of Western Australia; Paul Anderson MD Orthocell Pty Ltd Tendon
Autologous Tenocyte Implantation for Refractory Tendonopathy Ming Hao Zheng, PhD, DM, FRC Path Centre For Orthopaedic Research, The University of Western Australia; Paul Anderson MD Orthocell Pty Ltd Tendon
9/18/18. Welcome- MSK Ultrasound Workshop. Introduction to Musculoskeletal Ultrasound. Acknowledgement of Country. The Workshop.
 Acknowledgement of Country Welcome- MSK Ultrasound Workshop I would like to acknowledge that this meeting is being held on the traditional lands of the Wurundjeri and Boonwurrung people and pay my respect
Acknowledgement of Country Welcome- MSK Ultrasound Workshop I would like to acknowledge that this meeting is being held on the traditional lands of the Wurundjeri and Boonwurrung people and pay my respect
Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells,
 What is a Stem Cell? Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells, and so on. These cells can replicate more of their own kind of cell, but they cannot
What is a Stem Cell? Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells, and so on. These cells can replicate more of their own kind of cell, but they cannot
Supplemental Tables and Figures. The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate,
 Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
Supplemental Tables and Figures The metalloproteinase-proteoglycans ADAMTS7 and ADAMTS12 provide an innate, tendon-specific protective mechanism against heterotopic ossification Timothy Mead et al Supplemental
Diclofenac And Triamcinolone Acetonide Impair Tenocytic Differentiation And Promote Adipocytic Differentiation Of Mesenchymal Stem Cells
 Diclofenac And Triamcinolone Acetonide Impair Tenocytic Differentiation And Promote Adipocytic Differentiation Of Mesenchymal Stem Cells Maritha Fredriksson, Yan Li, Anders Stråman, Lars-Arne Haldosen,
Diclofenac And Triamcinolone Acetonide Impair Tenocytic Differentiation And Promote Adipocytic Differentiation Of Mesenchymal Stem Cells Maritha Fredriksson, Yan Li, Anders Stråman, Lars-Arne Haldosen,
Discovery of a Small Molecule Inhibitor of the Wnt Pathway as a Potential Disease Modifying Treatment for Knee Osteoarthritis
 Discovery of a Small Molecule Inhibitor of the Wnt Pathway as a Potential Disease Modifying Treatment for Knee Osteoarthritis Charlene Barroga, Ph.D., Yong Hu, Ph.D., Vishal Deshmukh, Ph.D., and John Hood,
Discovery of a Small Molecule Inhibitor of the Wnt Pathway as a Potential Disease Modifying Treatment for Knee Osteoarthritis Charlene Barroga, Ph.D., Yong Hu, Ph.D., Vishal Deshmukh, Ph.D., and John Hood,
Most abundant and widely distributed tissues in the body Binds, support, and strengthen body tissues, protect and insulate internal organ, serve as
 Connective tissue Most abundant and widely distributed tissues in the body Binds, support, and strengthen body tissues, protect and insulate internal organ, serve as major transport system, compartmentalizes
Connective tissue Most abundant and widely distributed tissues in the body Binds, support, and strengthen body tissues, protect and insulate internal organ, serve as major transport system, compartmentalizes
Tissue renewal and Repair. Nisamanee Charoenchon, PhD Department of Pathobiology, Faculty of Science
 Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
3rd MuscleTech Network Workshop. Muscle injuries and repair: Current trends in research.
 3rd MuscleTech Network Workshop. Muscle injuries and repair: Current trends in research. CONCLUSIONS AND CLOSING REMARKS Dr. Carles Pedret MUSCLE AND TENDON WORKSHOP HEALTH AND GENERAL POPULATION SPORTS
3rd MuscleTech Network Workshop. Muscle injuries and repair: Current trends in research. CONCLUSIONS AND CLOSING REMARKS Dr. Carles Pedret MUSCLE AND TENDON WORKSHOP HEALTH AND GENERAL POPULATION SPORTS
Despite the unique and important roles of tendons and
 REVIEW JBMR Tendons and Ligaments: Connecting Developmental Biology to Musculoskeletal Disease Pathogenesis Hiroshi Asahara, 1,2 Masafumi Inui, 3 and Martin K Lotz 1 1 Department of Molecular Medicine,
REVIEW JBMR Tendons and Ligaments: Connecting Developmental Biology to Musculoskeletal Disease Pathogenesis Hiroshi Asahara, 1,2 Masafumi Inui, 3 and Martin K Lotz 1 1 Department of Molecular Medicine,
Ricardo E. Colberg, MD, RMSK. PM&R Sports Medicine Physician Andrews Sports Medicine and Orthopedic Center American Sports Medicine Institute
 Ricardo E. Colberg, MD, RMSK PM&R Sports Medicine Physician Andrews Sports Medicine and Orthopedic Center American Sports Medicine Institute Pathophysiology of chronic orthopedic injuries Definition of
Ricardo E. Colberg, MD, RMSK PM&R Sports Medicine Physician Andrews Sports Medicine and Orthopedic Center American Sports Medicine Institute Pathophysiology of chronic orthopedic injuries Definition of
Biology. Dr. Khalida Ibrahim
 Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
Biology Dr. Khalida Ibrahim The cartilage General characteristics: 1. Cartilage is a specialized type of connective tissue (supporting connective tissue). 2. Consists, like other connective tissues, of
Lecture Overview. Connective Tissues. Marieb s Human Anatomy and Physiology. Chapter 4 Tissues: The Living Fabric Connective Tissues Lecture 10
 Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 4 Tissues: The Living Fabric Connective Tissues Lecture 10 Lecture Overview General composition and function of connective tissue Components of
Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 4 Tissues: The Living Fabric Connective Tissues Lecture 10 Lecture Overview General composition and function of connective tissue Components of
BONE TISSUE. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
Connective Tissue. Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment.
 Connective Tissue Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment. Connective Tissue Functions Binding and support Protection Insulation Transportation
Connective Tissue Found everywhere in the body. Most abundant and widely distributed. Never exposed to the outside environment. Connective Tissue Functions Binding and support Protection Insulation Transportation
Cartilage. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Cartilage Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology 1 Cartilage is a specialized type of connective tissue designed to give support, bear weight and withstand tension, torsion and
Re-growing the Skeleton: Approaches in Tissue Engineering and Regenerative Medicine
 Re-growing the Skeleton: Approaches in Tissue Engineering and Regenerative Medicine How we fix things now Total Knee Replacements Fracture Plates Fusing Joints Defining Regenerative Medicine restore form
Re-growing the Skeleton: Approaches in Tissue Engineering and Regenerative Medicine How we fix things now Total Knee Replacements Fracture Plates Fusing Joints Defining Regenerative Medicine restore form
Tissue engineering of cartilage
 Tissue engineering of cartilage Cartilage responds to mechanical forces and is able to remodel in response to the prevailing stress Cartilage, like bone, may respond to mechanical stimulation by increasing
Tissue engineering of cartilage Cartilage responds to mechanical forces and is able to remodel in response to the prevailing stress Cartilage, like bone, may respond to mechanical stimulation by increasing
Chapter 6: Skeletal System: Bones and Bone Tissue
 Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
Chapter 6: Skeletal System: Bones and Bone Tissue I. Functions A. List and describe the five major functions of the skeletal system: 1. 2. 3.. 4. 5.. II. Cartilage A. What do chondroblasts do? B. When
BIOH111. o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system
 BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
An Owner's Guide to Natural Healing. Autologous Conditioned Plasma (ACP)
 An Owner's Guide to Natural Healing Autologous Conditioned Plasma (ACP) Healing after an injury involves a well-orchestrated and complex series of events where proteins in the blood have primary roles,
An Owner's Guide to Natural Healing Autologous Conditioned Plasma (ACP) Healing after an injury involves a well-orchestrated and complex series of events where proteins in the blood have primary roles,
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Prof. Steven S. Saliterman. Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ The meniscus is a fibrocartilaginous tissue primarily of type 1 collagen fibers. (In contrast to hyaline cartilage
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ The meniscus is a fibrocartilaginous tissue primarily of type 1 collagen fibers. (In contrast to hyaline cartilage
EXTRACELLULAR MATRIX (pp 9-17)
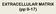 EXTRACELLULAR MATRIX (pp 9-17) Extracellular Matrix (ECM) Apart from specific cells, tissues contain matrix of macromolecules in the extracellular space- Extracellular Matrix. ECM is secreted by cells
EXTRACELLULAR MATRIX (pp 9-17) Extracellular Matrix (ECM) Apart from specific cells, tissues contain matrix of macromolecules in the extracellular space- Extracellular Matrix. ECM is secreted by cells
Soft tissue biomechanics
 Soft tissue biomechanics Caroline Öhman Pula, 22/06-08 TABLE OF CONTENTS Introduction to soft tissues Tendon and ligaments Introduction Composition Function and structure In vitro testing Stress-strain
Soft tissue biomechanics Caroline Öhman Pula, 22/06-08 TABLE OF CONTENTS Introduction to soft tissues Tendon and ligaments Introduction Composition Function and structure In vitro testing Stress-strain
SKELETAL TISSUES CHAPTER 7 INTRODUCTION TO THE SKELETAL SYSTEM TYPES OF BONES
 SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
SKELETAL TISSUES CHAPTER 7 By John McGill Supplement Outlines: Beth Wyatt Original PowerPoint: Jack Bagwell INTRODUCTION TO THE SKELETAL SYSTEM STRUCTURE Organs: Bones Related Tissues: Cartilage and Ligaments
Cellular therapy in bone-tendon interface regeneration
 Organogenesis ISSN: 1547-6278 (Print) 1555-8592 (Online) Journal homepage: http://www.tandfonline.com/loi/kogg20 Cellular therapy in bone-tendon interface regeneration Benjamin B Rothrauff & Rocky S Tuan
Organogenesis ISSN: 1547-6278 (Print) 1555-8592 (Online) Journal homepage: http://www.tandfonline.com/loi/kogg20 Cellular therapy in bone-tendon interface regeneration Benjamin B Rothrauff & Rocky S Tuan
STEM CELLS. Dr Mohammad Ashfaq Konchwalla Consultant Orthopaedic Sports Surgeon
 STEM CELLS Dr Mohammad Ashfaq Konchwalla Consultant Orthopaedic Sports Surgeon www.dubaisportssurgery.com PRACTICE SAUDI GERMAN HOSPITAL, DUBAI MEDCARE HOSPITAL, DUBAI Totipotent cells are cells that can
STEM CELLS Dr Mohammad Ashfaq Konchwalla Consultant Orthopaedic Sports Surgeon www.dubaisportssurgery.com PRACTICE SAUDI GERMAN HOSPITAL, DUBAI MEDCARE HOSPITAL, DUBAI Totipotent cells are cells that can
CARTILAGE. Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015
 CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
CARTILAGE Dr. Emad I Shaqoura M.D, M.Sc. Anatomy Faculty of Medicine, Islamic University-Gaza October, 2015 Introduction Hyaline Cartilage Elastic Cartilage Fibrocartilage Cartilage Formation, Growth,
Spontaneous arthritis and ankylosis in male DBA/ 1 mice: further evidence for a role of behavioral factors in stress-induced arthritis
 Braem et al. Biological Procedures Online 2012, 14:10 Biological Procedures Online SHORT REPORT Open Access Spontaneous arthritis and ankylosis in male DBA/ 1 mice: further evidence for a role of behavioral
Braem et al. Biological Procedures Online 2012, 14:10 Biological Procedures Online SHORT REPORT Open Access Spontaneous arthritis and ankylosis in male DBA/ 1 mice: further evidence for a role of behavioral
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade)
 HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
Which compound is reponsible for the viscous character of the ground substance?
 1 2 Which type of collagen forms the coarse collagen fibres in dense regular and irregular connective tissues? Which compound is reponsible for the viscous character of the ground substance? 3 Which class
1 2 Which type of collagen forms the coarse collagen fibres in dense regular and irregular connective tissues? Which compound is reponsible for the viscous character of the ground substance? 3 Which class
HOLE S ANATOMY CHAPTER 5, PART II Lecture notes
 HOLE S ANATOMY CHAPTER 5, PART II Lecture notes I. Connective Tissue A. Structure 1. have few cells that are spaced apart and can divide; two categories: a. fixed cells cells that are present in tissue
HOLE S ANATOMY CHAPTER 5, PART II Lecture notes I. Connective Tissue A. Structure 1. have few cells that are spaced apart and can divide; two categories: a. fixed cells cells that are present in tissue
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
Sheet #9. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Sheet #9 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Elastic fibers The main function of elastic fibers is to provide elasticity. In other words these fibers are able to restore the original
Sheet #9 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Elastic fibers The main function of elastic fibers is to provide elasticity. In other words these fibers are able to restore the original
AAO Foundation Awards Final Report
 401 N. Lindbergh Blvd. St. Louis, MO 63141 Tel.: 314.993.1700, #546 Toll Free: 800.424.2841, #546 Fax: 800.708.1364 Cell: 314.283.1983 E-Mail: rhazel@aaortho.org AAO Foundation Awards Final Report In an
401 N. Lindbergh Blvd. St. Louis, MO 63141 Tel.: 314.993.1700, #546 Toll Free: 800.424.2841, #546 Fax: 800.708.1364 Cell: 314.283.1983 E-Mail: rhazel@aaortho.org AAO Foundation Awards Final Report In an
ESWT Mechanisms of Action in Veterinary Applications
 ESWT Mechanisms of Action in Veterinary Applications Extracorporeal Shock Wave Technology (ESWT) is a high energy acoustic pressure wave technology that has been demonstrated to treat a variety of musculoskeletal
ESWT Mechanisms of Action in Veterinary Applications Extracorporeal Shock Wave Technology (ESWT) is a high energy acoustic pressure wave technology that has been demonstrated to treat a variety of musculoskeletal
36 1 The Skeletal System Slide 1 of 40
 1 of 40 The Skeleton All organisms need structural support. Unicellular organisms have a cytoskeleton. Multicellular animals have either an exoskeleton (arthropods) or an endoskeleton (vertebrates). 2
1 of 40 The Skeleton All organisms need structural support. Unicellular organisms have a cytoskeleton. Multicellular animals have either an exoskeleton (arthropods) or an endoskeleton (vertebrates). 2
Stem Cells and Sport Medicine
 Stem Cells and Sport Medicine Rehal Abbas Bhojani, MD CAQSM Memorial Hermann Medical Group 2014 Sports Medicine Symposium of the Americas Stem cell biology Overview Potential applications of stem cells
Stem Cells and Sport Medicine Rehal Abbas Bhojani, MD CAQSM Memorial Hermann Medical Group 2014 Sports Medicine Symposium of the Americas Stem cell biology Overview Potential applications of stem cells
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Alginate, tooth-shaped, for constructs, encapsulated pulp cells in, 589 590 Antibiotic paste, triple, change in root length and width
Index Note: Page numbers of article titles are in boldface type. A Alginate, tooth-shaped, for constructs, encapsulated pulp cells in, 589 590 Antibiotic paste, triple, change in root length and width
Connective Tissue Part-2. Dr. Heba Kalbouneh Assistant Professor of Anatomy and Histology
 Connective Tissue Part-2 Dr. Heba Kalbouneh Assistant Professor of Anatomy and Histology 1 Features Composed of cells, fibers and extracellular matrix. Highly vascular Variable regenerative power Originates
Connective Tissue Part-2 Dr. Heba Kalbouneh Assistant Professor of Anatomy and Histology 1 Features Composed of cells, fibers and extracellular matrix. Highly vascular Variable regenerative power Originates
Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model
 Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model Averi Leahy, Andrea Foote, Tomoya Uchimura, Li Zeng, PhD. Tufts University, Boston, MA, USA. Disclosures: A. Leahy: None.
Wnt7a Inhibits Cartilage Matrix Degradation in a Mouse In Vivo Osteoarthritis Model Averi Leahy, Andrea Foote, Tomoya Uchimura, Li Zeng, PhD. Tufts University, Boston, MA, USA. Disclosures: A. Leahy: None.
Bones. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
 Bones Osteocytes : Are responsible for maintenance of bones Present in lacunae, and send processes. Unable to divide. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
Bones Osteocytes : Are responsible for maintenance of bones Present in lacunae, and send processes. Unable to divide. The division of bones anatomically is : long, short, irregular, flat and sesamoid.
General Features. Originates mostly from mesoderm. Composed of cells, fibres and extracellular matrix. Highly vascular. Variable regenerative power.
 Connective Tissue General Features Originates mostly from mesoderm. Composed of cells, fibres and extracellular matrix. Highly vascular. Variable regenerative power. Functions of Connective Tissue Support:
Connective Tissue General Features Originates mostly from mesoderm. Composed of cells, fibres and extracellular matrix. Highly vascular. Variable regenerative power. Functions of Connective Tissue Support:
Functions of the Skeletal System. Chapter 6: Osseous Tissue and Bone Structure. Classification of Bones. Bone Shapes
 Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
Skeletal Tissues. Skeletal tissues. Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs.
 Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
Skeletal Tissues Functions 1) support 2) protection 3) movement Skeletal tissues Frame; muscles, organs and CT attach. Brain, spinal cord, thoracic organs; heart and lungs. Aids muscle contraction; generate
CELL BIOLOGY - CLUTCH CH CELL JUNCTIONS AND TISSUES.
 !! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
!! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
Human Anatomy and Physiology I Laboratory
 Human Anatomy and Physiology I Laboratory Skeletal Tissue: Cartilage and Bone This lab involves study of the laboratory exercise Overview of the Skeleton, Classification and Structure of Bones and Cartilages,
Human Anatomy and Physiology I Laboratory Skeletal Tissue: Cartilage and Bone This lab involves study of the laboratory exercise Overview of the Skeleton, Classification and Structure of Bones and Cartilages,
Osteochondral regeneration. Getting to the core of the problem.
 Osteochondral regeneration. Getting to the core of the problem. TM TM Bio-mimetic, biointegratable and resorbable Flexible and easy to shape Straightforward one-step procedure Promotes a guided osteo-chondral
Osteochondral regeneration. Getting to the core of the problem. TM TM Bio-mimetic, biointegratable and resorbable Flexible and easy to shape Straightforward one-step procedure Promotes a guided osteo-chondral
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
CHAPTER 6 LECTURE OUTLINE
 CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
CHAPTER 6 LECTURE OUTLINE I. INTRODUCTION A. Bone is made up of several different tissues working together: bone, cartilage, dense connective tissue, epithelium, various blood forming tissues, adipose
What is histology? HISTOLOGY
 Introduction to Histology What is histology? HISTOLOGY histo = tissue ogy = study So HISTOLOGY = the study of tissues! What is a TISSUE? Tissues are groups of cells with specialized structural and functional
Introduction to Histology What is histology? HISTOLOGY histo = tissue ogy = study So HISTOLOGY = the study of tissues! What is a TISSUE? Tissues are groups of cells with specialized structural and functional
Disclosures 2/16/2017. Healing of Partial-Thickness Rotator Cuff Lesions. The Problem: Steven P. Arnoczky, DVM. Consultant:
 Healing of Partial-Thickness Rotator Cuff Lesions Balancing Biomechanics and Biology Steven P. Arnoczky, DVM Laboratory for Comparative Orthopaedic Research Michigan State University Disclosures 2 Consultant:
Healing of Partial-Thickness Rotator Cuff Lesions Balancing Biomechanics and Biology Steven P. Arnoczky, DVM Laboratory for Comparative Orthopaedic Research Michigan State University Disclosures 2 Consultant:
PRP Basic Science. Platelets. Definition of PRP 10/4/2011. Questions that this talk aims to answer
 PRP Basic Science Peter J. Moley, MD Hospital for Special Surgery October 5, 2011 Questions that this talk aims to answer 1. What is PRP? 2. What blood components are NOT in PRP? 3. What are the active
PRP Basic Science Peter J. Moley, MD Hospital for Special Surgery October 5, 2011 Questions that this talk aims to answer 1. What is PRP? 2. What blood components are NOT in PRP? 3. What are the active
Pattern of bone resorption after extraction
 Teeth loss Pattern of bone resorption after extraction 50% in 1st year 2/ 3 in first 3 months Reich KM, Huber CD, Lippnig WR, Um C, Watzek G, Tangl S. (2011, 17). Atrophy of Residual Alveolar Ridge following
Teeth loss Pattern of bone resorption after extraction 50% in 1st year 2/ 3 in first 3 months Reich KM, Huber CD, Lippnig WR, Um C, Watzek G, Tangl S. (2011, 17). Atrophy of Residual Alveolar Ridge following
Acute. Chronic. Tendinopathy: Acute vs. Chronic Differences in Anatomy, Symptoms & Treatment
 Tendinopathy: Acute vs. Chronic Differences in Anatomy, Symptoms & Treatment Science and Management Symposium SAM 2018 Marlene Reid, DPM FACFAS, FACFAO, FASPS Tendinopathy Tendinopathy is a failed healing
Tendinopathy: Acute vs. Chronic Differences in Anatomy, Symptoms & Treatment Science and Management Symposium SAM 2018 Marlene Reid, DPM FACFAS, FACFAO, FASPS Tendinopathy Tendinopathy is a failed healing
HISTOLOGY Lecture TWO DR. ASHRAF SAID
 HISTOLOGY Lecture TWO DR. ASHRAF SAID Start Of this lecture TISSUES TISSUE: A DEFINITION A group of connected and interdependent cells that cooperate to perform a specific function CONNECTIVE TISSUE The
HISTOLOGY Lecture TWO DR. ASHRAF SAID Start Of this lecture TISSUES TISSUE: A DEFINITION A group of connected and interdependent cells that cooperate to perform a specific function CONNECTIVE TISSUE The
Cartilage. - Cartilage together with long bone form the skeleton and support the body.
 Cartilage - Cartilage is a special type of CT has a firm pliable matrix that can resist mechanical stress, act as a shock absorber. - Cartilage together with long bone form the skeleton and support the
Cartilage - Cartilage is a special type of CT has a firm pliable matrix that can resist mechanical stress, act as a shock absorber. - Cartilage together with long bone form the skeleton and support the
Growth and repair: Cartilage is a vascular tissues that receives nutrients by diffusion through its matrix, cartilage grow by 2 mechanisms:
 Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Skeletal connective tissues: (cartilage and bone): Cartilage and bone are specialized connective tissues both adapted to serve as skeletal framework in most vertebrates the presence of solid inter cellular
Unit 5: SKELETAL SYSTEM
 Unit 5: SKELETAL SYSTEM (a) NRSG231 Dr. Moattar Raza Rizvi Skeletal System: Contents Functions of the Skeletal System Fracture Structure of Bone Tissue Articulations & Joint Bone Development and Growth
Unit 5: SKELETAL SYSTEM (a) NRSG231 Dr. Moattar Raza Rizvi Skeletal System: Contents Functions of the Skeletal System Fracture Structure of Bone Tissue Articulations & Joint Bone Development and Growth
Tissues organs system organism. pg151
 Histology is the study of tissues A TISSUE is a group of cells, usually of one kind, & their intercellular substance (e.g. intercellular matrix in animal) which are linked together & perform a particular
Histology is the study of tissues A TISSUE is a group of cells, usually of one kind, & their intercellular substance (e.g. intercellular matrix in animal) which are linked together & perform a particular
Equine Regenerative Medicine. Regenerative Medicine IRAP and PRP in the Equine Athlete. Stem Cells. Stem Cells. Veterinary Medical Devices
 Equine Regenerative Medicine Regenerative Medicine IRAP and PRP in the Equine Athlete Victoria Maxwell, DVM, MBA 2018 Potomac Regional Veterinary Conference Hyatt Regency Inner Harbor Baltimore, Maryland
Equine Regenerative Medicine Regenerative Medicine IRAP and PRP in the Equine Athlete Victoria Maxwell, DVM, MBA 2018 Potomac Regional Veterinary Conference Hyatt Regency Inner Harbor Baltimore, Maryland
SCIENCE BASED PRODUCT
 SCIENCE BASED PRODUCT Oct 2006 **RPP has been marketing and selling this type of regenerative medicine since 2014 Evolution of Orthobiologics Single human recombinant growth factors BMP-7 BMP-2 Sterile
SCIENCE BASED PRODUCT Oct 2006 **RPP has been marketing and selling this type of regenerative medicine since 2014 Evolution of Orthobiologics Single human recombinant growth factors BMP-7 BMP-2 Sterile
Calcium alginate/bone marrow mesenchymal stem cells combined with degradation membrane for repair of skin defects
 19 32 2015 08 06 Chinese Journal of Tissue Engineering Research August 6, 2015 Vol.19, No.32 1 2 2 3 4 2 5 ( 1 116044 2 5 110034 3 110024 4 110000) 1 2 3 C 2 15 3 ( ) ( 1 ) ( 2 ) 7 14 21 d 14 21 d 21 d
19 32 2015 08 06 Chinese Journal of Tissue Engineering Research August 6, 2015 Vol.19, No.32 1 2 2 3 4 2 5 ( 1 116044 2 5 110034 3 110024 4 110000) 1 2 3 C 2 15 3 ( ) ( 1 ) ( 2 ) 7 14 21 d 14 21 d 21 d
TECHNICAL RESEARCH DOCUMENTS AND MEDICAL PUBLICATIONS FOR: DAMAGED BONE AND CARTILAGE
 TECHNICAL RESEARCH DOCUMENTS AND MEDICAL PUBLICATIONS FOR: DAMAGED BONE AND CARTILAGE January 2018 TECHNICAL RESEARCH DOCUMENTS AND MEDICAL PUBLICATIONS FOR: DAMAGED BONE AND CARTILAGE Stem cells from
TECHNICAL RESEARCH DOCUMENTS AND MEDICAL PUBLICATIONS FOR: DAMAGED BONE AND CARTILAGE January 2018 TECHNICAL RESEARCH DOCUMENTS AND MEDICAL PUBLICATIONS FOR: DAMAGED BONE AND CARTILAGE Stem cells from
The Tissue Level of Organization
 The Tissue Level of Organization 4.5-4.11 August 31, 2012 4.5 Connective Tissues Describe the general features of connective Describe the structure, location, and function of the various types of connective
The Tissue Level of Organization 4.5-4.11 August 31, 2012 4.5 Connective Tissues Describe the general features of connective Describe the structure, location, and function of the various types of connective
Blood. Hematopoietic Tissue
 Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
Blood Hematopoietic Tissue Is a type of connective tissue in which its cells are suspended in a circulating fluid. Erythrocytes+ leukocytes + platelets (thrombocytes) =formed elements of blood. These formed
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS
 OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
OSSEOUS TISSUE & BONE STRUCTURE PART I: OVERVIEW & COMPONENTS The Skeletal System Skeletal system includes: bones of the skeleton, cartilages, ligaments, and connective tissues What are the functions of
Središnja medicinska knjižnica.
 Središnja medicinska knjižnica Pećina, M., Vukičević, S. (2007) Biological aspects of bone, cartilage and tendon regeneration. International Orthopaedics, 31 (6). pp. 719-720. The original publication
Središnja medicinska knjižnica Pećina, M., Vukičević, S. (2007) Biological aspects of bone, cartilage and tendon regeneration. International Orthopaedics, 31 (6). pp. 719-720. The original publication
LIGAMENTS AND TENDONS
 Harvard-MIT Division of Health Sciences and Technology HST.523J: Cell-Matrix Mechanics Prof. Myron Spector Massachusetts Institute of Technology Harvard Medical School Brigham and Women s s Hospital VA
Harvard-MIT Division of Health Sciences and Technology HST.523J: Cell-Matrix Mechanics Prof. Myron Spector Massachusetts Institute of Technology Harvard Medical School Brigham and Women s s Hospital VA
ORTHOBIOLOGIC TREATMENTS IN BASEBALL. Casey G. Batten MD PBATS - January 19th, 2018
 ORTHOBIOLOGIC TREATMENTS IN BASEBALL Casey G. Batten MD PBATS - January 19th, 2018 The Problem Musculoskeletal injuries are common in sport Pressure to minimize down time, swift return Many injuries involve
ORTHOBIOLOGIC TREATMENTS IN BASEBALL Casey G. Batten MD PBATS - January 19th, 2018 The Problem Musculoskeletal injuries are common in sport Pressure to minimize down time, swift return Many injuries involve
Practical Histology. Lab 3: Connective tissue
 Practical Histology Lab 3: Connective tissue Connective tissues Connective tissue provides structural support for the body by binding cells and tissues together to form organs. It also provides metabolic
Practical Histology Lab 3: Connective tissue Connective tissues Connective tissue provides structural support for the body by binding cells and tissues together to form organs. It also provides metabolic
Rama Nada. - Mousa Al-Abbadi. 1 P a g e
 - 1 - Rama Nada - - Mousa Al-Abbadi 1 P a g e Bones, Joints and Soft tissue tumors Before we start: the first 8 minutes was recalling to Dr.Mousa s duties, go over them in the slides. Wherever you see
- 1 - Rama Nada - - Mousa Al-Abbadi 1 P a g e Bones, Joints and Soft tissue tumors Before we start: the first 8 minutes was recalling to Dr.Mousa s duties, go over them in the slides. Wherever you see
3:30 PM 4:00 PM Hazel Screen, PhD, Queen Mary, University of London The Tendon Interfascicular Matrix Niche
 Discovery to Delivery in Tendon Research: Team Approaches ORS Tendon Section 2018 Conference November 15-17, 2018 Oregon Health and Science University, Portland, Oregon Conference Chairs: Ronen Schweitzer,
Discovery to Delivery in Tendon Research: Team Approaches ORS Tendon Section 2018 Conference November 15-17, 2018 Oregon Health and Science University, Portland, Oregon Conference Chairs: Ronen Schweitzer,
Bone Tissue- Chapter 5 5-1
 Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
Bone Tissue- Chapter 5 5-1 Bone Functions Support Protection Assistance in movement Mineral storage and release Blood cell production Triglyceride storage 5-2 Bone Chemistry Water (25%) Organic Constituent
Diseases of the skeleton
 Diseases of the skeleton Flexibility Protection of vital organs Strength Skeletal defects impact human health Developmental diseases Degenerative diseases Fins regenerate and grow rapidly following amputation
Diseases of the skeleton Flexibility Protection of vital organs Strength Skeletal defects impact human health Developmental diseases Degenerative diseases Fins regenerate and grow rapidly following amputation
FORMATION OF BONE. Intramembranous Ossification. Bone-Lec-10-Prof.Dr.Adnan Albideri
 FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
What are the parts of the skeletal system? Chapter 6- Part I Bones and Skeletal Tissues. Growth of Cartilage. Bones come in many shapes
 Chapter 6- Part I Bones and Skeletal Tissues Components of the skeletal system Classification of Bone (bone shapes) Functions of bone Bone structure Microscopic structure of bone and bone cells What are
Chapter 6- Part I Bones and Skeletal Tissues Components of the skeletal system Classification of Bone (bone shapes) Functions of bone Bone structure Microscopic structure of bone and bone cells What are
Bone (2) Chapter 8. The bone is surrounded by the periosteum, the periosteum consists of two layers: a fibrous outer layer and an innercellular layer.
 Bone (2) Chapter 8 The bone is surrounded by the periosteum, the periosteum consists of two layers: a fibrous outer layer and an innercellular layer. The innercellular layer contains osteoprogenitor cells,
Bone (2) Chapter 8 The bone is surrounded by the periosteum, the periosteum consists of two layers: a fibrous outer layer and an innercellular layer. The innercellular layer contains osteoprogenitor cells,
Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for
 Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for LacZ activity, which reflects Egr1 expression. (A)
Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for LacZ activity, which reflects Egr1 expression. (A)
Connective Tissue (CT)
 Connective Tissue (CT) YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Introduction of CT 1. Origin 2. Compositions
Connective Tissue (CT) YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Introduction of CT 1. Origin 2. Compositions
Lecture 2: Skeletogenesis
 Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Evidence Based Approach to the Treatment of Lower Extremity Tendinopathy. Cage SA, Gallegos DM, Eilers MA, Warner BJ
 Evidence Based Approach to the Treatment of Lower Extremity Tendinopathy Cage SA, Gallegos DM, Eilers MA, Warner BJ Thank you PICO Question Patients Subjects suffering from lower extremity tendinopathy
Evidence Based Approach to the Treatment of Lower Extremity Tendinopathy Cage SA, Gallegos DM, Eilers MA, Warner BJ Thank you PICO Question Patients Subjects suffering from lower extremity tendinopathy
1/10/2013. Topic 10: Form and Function (COM) Newton s Laws of Motion. Newton s Laws of Motion. What are? Forces
 /0/203 Topic 0: Form and Function (COM) Forces exerted on vertebrate bodies How do Newton s Laws apply to vertebrate form and function? How do we study forces in functional morphology? What are Ground
/0/203 Topic 0: Form and Function (COM) Forces exerted on vertebrate bodies How do Newton s Laws apply to vertebrate form and function? How do we study forces in functional morphology? What are Ground
Restore Tendon Function (UK ) L
 Restore Tendon Function (UK ) L OSTENIL TENDON helps to maintain tendon biomechanical properties OSTENIL TENDON contains a solution of hyaluronic acid which has unique properties: Acts as a lubricant
Restore Tendon Function (UK ) L OSTENIL TENDON helps to maintain tendon biomechanical properties OSTENIL TENDON contains a solution of hyaluronic acid which has unique properties: Acts as a lubricant
The effects of high glucose on tendon-derived stem cells: implications of the pathogenesis of diabetic tendon disorders
 /, 2017, Vol. 8, (No. 11), pp: 17518-17528 The effects of high glucose on tendon-derived stem cells: implications of the pathogenesis of diabetic tendon disorders Yu-Cheng Lin 1,*, Ying-Juan Li 2,*, Yun-Feng
/, 2017, Vol. 8, (No. 11), pp: 17518-17528 The effects of high glucose on tendon-derived stem cells: implications of the pathogenesis of diabetic tendon disorders Yu-Cheng Lin 1,*, Ying-Juan Li 2,*, Yun-Feng
Module 2:! Functional Musculoskeletal Anatomy A! Semester 1! !!! !!!! Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb!
 Functional Musculoskeletal Anatomy A Module 2: Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb Semester 1 1 18. Bone Tissue & Growth of Bones 18.1 Describe the structure of bone tissue
Functional Musculoskeletal Anatomy A Module 2: Hard Tissues, Distal Upper Limb & Neurovascular Supply of Upper Limb Semester 1 1 18. Bone Tissue & Growth of Bones 18.1 Describe the structure of bone tissue
Skeletal System. The skeletal System... Components
 Skeletal System The skeletal System... What are the general components of the skeletal system? What does the skeletal system do for you & how does it achieve these functions? Components The skeletal system
Skeletal System The skeletal System... What are the general components of the skeletal system? What does the skeletal system do for you & how does it achieve these functions? Components The skeletal system
Structural Support and Movement. Chapter 36
 Structural Support and Movement Chapter 36 Impacts, Issues Pumping Up Muscles Increasing muscle size and strength with drugs such as andro has unwanted side effects and can damage other organ systems 36.1
Structural Support and Movement Chapter 36 Impacts, Issues Pumping Up Muscles Increasing muscle size and strength with drugs such as andro has unwanted side effects and can damage other organ systems 36.1
