By BRIAN K. HALL 1. From the Department of Biology, Life Sciences Centre, Dalhousie University, Halifax
|
|
|
- Marcia Louisa Austin
- 5 years ago
- Views:
Transcription
1 /. Embryol. exp. Morph. Vol. 58, pp , Printed in Great Britain Company of Biologists Limited 1980 Tissue interactions and the initiation of osteogenesis and chondrogenesis in the neural crest-derived mandibular skeleton of the embryonic mouse as seen in isolated murine tissues and in recombinations of murine and avian tissues By BRIAN K. HALL 1 From the Department of Biology, Life Sciences Centre, Dalhousie University, Halifax SUMMARY Mandibular processes from 9- to 13-day-old embryonic mice formed both bone and cartilage when grafted to the chorioallantoic membranes of host embryonic chicks. Isolated ectomesenchyme, taken from 9-day-old embryos did not form bone or cartilage, while older ectomesenchyme formed both. Recombination of the epithelial and ectomesenchymal components confirmed that the presence of the epithelium was a sufficient stimulus for the initiation of both chondro- and osteogenesis. Recombinations between components of mouse and chick mandibular processes showed that 9-day-old mouse ectomesenchyme could respond to chick epithelium but that, although older murine epithelia could initiate osteogenesis from the avian ectomesenchyme, epithelia from 9-day-old embryos could not. These results indicated that an epithelial-ectomesenchymal interaction was responsible for the initiation of both osteo- and chondrogenesis within the mandibular arch of the mouse; that the interaction began at 10 days of gestation; that the ectomesenchyme was capable of responding at 9 days, but that the epithelium did not acquire its ability to act on the ectomesenchyme until 10 days of gestation. INTRODUCTION Both the cartilaginous and the bony elements in the mandibular skeletons of vertebrate embryos form from ectomesenchymal cells derived from the embryonic neural crest. These cells have their origin within the crests of the neural folds. During neurulation these neural crest cells migrate away from the neural folds. Those cells which originate in the cranial neural crest become preferentially localized within the primordia of the cranio-facial skeleton. The ability of these cells to form the cartilages and bones of the craniofacial skeleton depends upon 1 Author's address; Department of Biology, Life Sciences Centre, Dalhousie University, Halifax, Nova Scotia, Canada, B3H 4J1. 17 EMB 58
2 252 B. K. HALL interactions which they undergo, either during or after, their migration. Thus, in the embryo of the lamprey, Lampetrafluviatilis L., contact with the endoderm is required before chondrogenesis can be initiated (Damas, 1944; Newth, 1951, 1956). While it is known that cranio-facial skeletal elements are of neural crest origin in the fishes (Jollie, 1971; Berkovitz & Moore, 1974; Schaeffer, 1977), the role of tissue interactions in their genesis has not been studied. Interaction of neural crest cells with pharyngeal endoderm is a prerequisite for the initiation of chondrogenesis in the visceral skeleton of the urodele amphibians (Wagner, 1949; Wilde, 1955; Cusimano-Carollo, 1963, 1972; Drews, Kocher-Becker & Drews, 1972; Epperlein, 1974; Corsin, 1975). However, although neural crest cells are known to form bone in the urodeles (de Beer, 1947; Cassin & Capuron, 1977), tissue interactions have not been studied. Nor is there any information for the anuran amphibians (see Horstadius, 1950 and Hall, 1978a for a discussion). The initiation of intramembranous osteogenesis within the scleral, maxillary and mandibular skeletons of the domestic fowl results from inductive interactions between scleral, maxillary, or mandibular epithelia and post-migratory ectomesenchymal cells from the neural crest (Murray, 1943; Tyler & Hall, 1977; Tyler, 1978; Hall, 1978c, b, 1980; Bradamante & Hall, 1980; Tyler & McCobb, 1980). Pre-migratory neural crest cells must interact, (a) with cranial epithelial ectoderm if they are to be able to form Meckel's cartilage later in development (Hall & Tremaine, 1979; Bee & Thorogood, 1980), or (b) with the pigmented retinal epithelium if they are to form scleral cartilage (Reinbold, 1968; Newsome, 1972, 1976; Hall, 1978a). Although the details vary, epithelial-mesenchymal interactions emerge as important prerequisites for the initiation of osteogenesis and chondrogenesis in the sub-mammalian vertebrates. The possible existence of similar interactions in the mammalian embryo has not been explored. Indeed, even the origin of the mammalian cranio-facial skeleton from neural crest cells has not been substantiated experimentally. There is histological evidence for spurs of ectodermal cells leading into the visceral arches (Bartelmez, 1952; Mulnard, 1955; O'Rahilly, 1965; Mayer, 1973); histochemical visualization of presumed neural crest cells (Milaire, 1959; 1974); indications that high doses of vitamin A both arrest migration of cranial neural crest cells (Morriss & Thorogood, 1978) and produce first arch defects such as Treacher Collins syndrome (Poswillo, 1974), and the evidence that ectoderm or teratocarcinomas isolated from rat embryos form both cartilage and bone (Levak-Svajger & Svajger, 1971; Damjanov, Solter & Serman, 1973) - all of which provide circumstantial evidence for contributions to the facial skeleton from the neural crest. The only study known to the author which explores the role of tissue interactions in the development of the mammalian mandibular skeleton is that of Svajger and Levak-Svajger (1976). These workers separated first branchial arch mesenchyme of 13-day rat embryos from their epithelia, organ cultured the mesenchyme, and observed the subsequent development of bone and cartilage.
3 Skeletogenesis in the murine mandible 253 If an epithelial-mesenchymal interaction was involved, it must have taken place before thirteen days of gestation. The aim of the present study was to determine whether an epithelial-mesenchymal (ectomesenchymal) interaction was involved in the initiation ofchondrogenesis and/or osteogenesis within the mandibular skeleton of the embryonic mice. To this end, whole mandibular processes, and mandibular ectomesenchyme, separated from the mandibular epithelium by treatment with proteolytic enzymes, were grafted to the chorioallantoic membranes of host embryonic chicks. (This vascularized site is preferred over organ culture when studying osteogenesis.) The separated components were also either recombined with one another or combined with components isolated from the mandibular processes of embryonic chicks and grafted to chorioallantoic membranes. Histological analysis was used to document the presence or absence of cartilage and bone in the recovered grafts. MATERIALS AND METHODS Animals ICR Swiss albino mice obtained from Bio-breeding Laboratories of Canada, Ottawa, Ontario, were used as the experimental animal. Mice were weaned at 4 weeks of age, housed with three or four of the same sex per cage, and fed Purina lab. chow and water ad Hbidum. Matings were carried out using animals which were at least 8 weeks old. Each male was placed with from one to three females overnight (16.00 to hours), hours was designated as the commencement of day zero of the pregnancy. Pregnancy was determined by presence of a vaginal plug and by weight gain, as measured on alternate days. Embryos Pregnant females were killed by an overdose of ether, the embryos removed by dissection under sterile conditions and aged using the morphological series of stages of Griineberg (1943). Embryos of 9-13 gestational days were used in this study. The 9-day-old embryos had well developed mandibular processes, lacked posterior limb buds, and had unsegmented posterior somitic mesoderm. By 13 days of gestation the embryos were recognizably murine, with five rows of whiskers containing hair follicles, footplates on the limbs and marked segmentation to the distal tips of the tails. Mandibular processes Mandibular processes were dissected from these embryos and either grafted intact, or separated into their epithelial and ectomesenchymal components and then grafted. Separation was achieved by placing the processes into a solution of trypsin and pancreatin in calcium- and magnesium-free Tyrode's solution (257 mg bovine pancreatic trypsin + 43 mg porcine pancreatic pancreatin/
4 254 B. K. HALL ml. both obtained from BDH Chemicals, Toronto, Ontario) for 2 h at 4 C. The mandibular processes were then placed into a solution of the synthetic culture medium BGJ b (GIBCO, Grand Islands, N.Y.) and Horse Serum (1:1) to stop the enzymic digestion and the epithelia separated from the ectomesenchyme by microdissection using sharpened hypodermic needles. A similar separation was performed on mandibular processes obtained from embryonic chicks which had been incubated for 4 days and attained Hamburger & Hamilton (1951) stage 22. Grafting Whole mandibular processes, recombined components from the mouse, or components combined with epithelia or ectomesenchyme from mandibular processes of the chick, were grafted to the chorio-allantoic membranes of 8- or 9-day-old embryonic chicks following the procedure outlined by Hall (1978 c). Briefly, the mandibular process or epithelium was placed onto a square of sterile, black, Millipore filter (0-45/*m porosity, /mi thick, Millipore Filter Corp., Montreal, Quebec), mandibular ectomesenchyme was placed in direct contact with the epithelium and the intact, or recombined processes, grafted for 7 days. Histology Grafts were recovered, dissected away from the chorioallantoic membranes, fixed in neutral buffered formal saline, processed histologically, serially sectioned and stained with haematoxylin, alcian blue and chlorantine fast red (modified from Lison, 1954). RESULTS Intact mandibular processes The chorioallantoic membrane (CAM) has previously been shown to be a graft site which supports the initiation and maintenance of chondrogenesis and osteogenesis from avian mandibular processes (Tyler & Hall, 1977; Hall, 1978, 1980). To determine whether the CAM would support cartilage and bone formation from murine tissue, and to determine whether the initiation of either tissue was dependent upon the age of the donor embryo, whole mandibular processes from mouse embryos of 9-13 gestational days were grafted to the CAMs of host embryonic chicks. Osteogenesis and chondrogenesis were initiated in at least 75 % of the grafts, irrespective of the ages of the mouse embryos (column I, Table 1). Typically, the cartilage consisted of one or two rods of hypertrophic cartilage, with scant amounts of extracellular matrix and an ill-defined perichondrium. The bone typically consisted of one or two ossicles of trabecular, membrane bone, usually separated from the cartilage by intervening connective tissue and often resting on the Millipore Filter substrate.
5 Table 1. Number of grafts forming cartilage and/or membrane bone Intact mandibular processes Age r- embryo Cartilage Bone (days) II Isolated ectomesenchyme Cartilage Bone III Recombined epithelium and ectomesenchyme A Cartilage Bone IVf Mouse ectomesenchyme + chick epithelium A I Cartilage Bone vt Chick ectomesenchyme + mouse epithelium Cartilage Bone * n, number of grafts analysed. t The chick tissues were obtained from embryos of H. H. stage 22 (4 days of incubation). When chick mandibular ectomesenchyme was recombined with chick epithelium and grafted, all grafts formed both cartilage and bone. N
6 256 B. K. HALL Fig. 1. Membrane bone (B) developed when mandibular ectomesenchyme from a 10-day-old embryo was grafted to the chorioallantoic membrane of a host embryonic chick. O, osteocyte; M, Millipore filter substrate. Alcian blue, chlorantine fast red and haematoxylin. Both the large percentage of grafts forming skeleton tissues and the advanced state of differentiation attained by the tissues, indicated that the chorioallantoic membrane was a suitable graft site for these mouse cells. The mandibular epithelium also continued to differentiate and formed a multi-layered squamous epithelia, with numerous buds extending into the underlying ectomesenchyme. Isolated mandibular ectomesenchyme Mandibular ectomesenchyme which had been dissected away from the mandibular epithelium after treatment of the intact mandibular process with trypsin and pancreatin was then grafted. Provided that the embryo was at least 10 days old, cartilage and membrane bone formed in a similar percentage of grafts to that seen when intact mandibular processes were grafted, viz. at least 75 % (see column II, Table 1, Fig. 1). The lack of murine epithelial cells in these grafts attested to the adequacy of the tissue separations. Although the amount of cartilage and bone present was not quantified, it was not noticeably less than that obtained from intact mandibular processes. The ectomesenchyme isolated from the 9-day-old embryos behaved quite differently. Chondrogenesis was not initiated in any graft, and osteogenesis only in one out of the eight grafts. The grafts consisted of a loose meshwork of etomesenchymal cells (Fig. 6). Thus, mandibular ectomesenchyme would only form bone and cartilage if the epithelium was present until 10 days of gestation.
7 Skeletogenesis in the murine mandible 257 Recombined epithelia and ectomesenchyme To confirm that the inability of ectomesenchyme from 9-day-old embryos to form cartilage and bone was a consequence of its isolation from mandibular epithelium, these two components were recombined and grafted to the CAMs of host embryonic chicks. Recombinations of components from 10- to 13-day-old embryos served as a control for the procedure. In these specimens chondrogenesis and osteogenesis were initiated at the same, on a higher rate, than when intact mandibular processes or isolated ectomesenchyme were grafted (column III, Table 1, Fig. 2). Having confirmed that the procedure of recombination did not inhibit skeletogenesis in these older tissues, epithelia and ectomesenchyme from mandibular processes of 9-day-old embryos were recombined and grafted. Both osteogenesis and chondrogenesis were initiated in such grafts (column III, Table 1), indicating that the lack of cartilage and bone in the isolated ectomesenchymes was attributable to the absence of the mandibular epithelia. Heterospecific recombinations Mandibular processes from embryonic mice and from H. H. (Hamburger & Hamilton, 1951) stage-22 embryonic chicks were separated and recombined heterospecifica lly. It is known that mandibular epithelium from the H. H. stage-22 embryonic chick will allow chick mandibular ectomesenchyme to initiate osteogenesis (Tyler & Hall, 1977; Hall, 1978Z?). Would chick mandibular epithelium substitute for murine epithelium in initiating osteogenesis? What of the initiation of chondrogenesis? Chondrogenesis in the chick mandible is not dependent on the presence of the mandibular epithelium. Would the chick epithelium allow the murine ectomesenchyme to form cartilage? In the first recombination, mouse mandibular ectomesenchyme was recombined with chick mandibular epithelium and grafted. Ectomesenchymes from 10- to 13-day-old mice were used as controls. Both bone and cartilage formed in these recombinants (column IV, Table 1, Figs. 3, 4). Cartilage and bone also formed in four out of the nine grafts containing 9-day murine ectomesenchyme (Fig. 7). (In assessing these results all recovered grafts were counted. Given that the amount of epithelial-ectomesenchymal contact varied from graft to graft, the positive results reported represent a minimal estimate.) This recombination indicated that mandibular ectomesenchyme from 9-day-old embryonic mice could respond to chick mandibular epithelium by initiating both chondrogenesis and intramembranous osteogenesis. In the second recombination, H. H. stage-22 chick mandibular ectomesenchyme was recombined with mouse mandibular epithelium of various ages (column V, Table 1). As noted above, the initiation of chondrogenesis in such avian ectomesenchyme is not dependent upon the presence of mandibular
8 258 B. K. HALL
9 Skeletogenesis in the marine mandible 259 Fig. 5. Cartilage (C) and bone (B) both formed when mandibular ectomesenchyme from an H. H. stage-22 chick embryo was combined with mandibular epithelium from a 13-day-old mouse embryo. Contrast the cartilage with that in Figs. 2 and 3. The murine epithelium (e) has also continued to differentiate. M, Millipore filter substrate. Alcian blue, chlorantine fast red, haematoxylin. epithelium. Therefore virtually all of these grafts contained nodules or rods of cartilage (Table 1, Fig. 5). Osteogenesis was also initiated in the ectomesenchyme, provided that the murine epithelium had been obtained from embryos of at least 10 days of gestation (column V, Table 1, Fig. 5). The chick ectomesenchyme also maintained the continued, normal differentiation of the mouse epithelium (Fig. 5). Epithelium from 9-day-old embryos was not able to induce bone formation from the avian ectomesenchyme, but epithelial differentiation was maintained. Therefore (a) the epithelial-ectomesenchymal interaction is neither species nor class-specific; (b) ectomesenchyme from all aged mice is able to respond to chick epithelia by initiating both osteogenesis and chondrogenesis, and (c) the epithelium from the mouse does not acquire the ability to induce osteogenesis FIGURES 2-4 Figs. 2, 3. Cartilage (C) and bone (B) formed when mandibular epithelium and ectomesenchyme from this 12-day-old embryo were recombined (Fig. 2) and when mouse mandibular ectomesenchyme was combined with chick mandibular epithelium (Fig. 3). Note the similarity of the cartilages in the two combinations. Alcian blue, chlorantine fast red and haematoxylin. Fig. 4. A higher magnifications of the bone seen in Fig. 3 to show the trabeculae of bone (B), osteocytes (O), osteoblasts (ob) and flattened ectomesenchymal cells (e) presumed to be osteoprogenitor cells. Alcian blue, chlorantine fast red, haematoxylin.
10 260 B. K. HALL Fig. 6. Ectomessnchyme isolated from 9-day-old embryos and grafted to the chorioallantoic membrane formed a loose meshwork of cells. Neither chondrogenesis nor osteogenesis is seen. Alcian blue, chlorantine fast red and haematoxylin. Fig. 7. Bone formed when mandibular ectomesenchyme from a 9-day-old mouse embryo was combined with mandibular epithelium from an H. H. stage-22 embryonic chick and grafted. Alcian blue, chlorantine fast red and haematoxylin.
11 Skeletogenesis in the marine mandible 261 or chondrogenesis until 10 days of gestation. The lack of bone or cartilage development from ectomesenchyme isolated from 9-day-old embryos reflected the absence of an epithelial signal rather than the inability of the ectomesenchyme to respond to such a signal. DISCUSSION Although a considerable amount is known about chondrogenesis, surprisingly little information is available on the microenvironmental control of the initiation of intramembranous osteogenesis. It is known (a) that calvarial osteogenesis is only initiated after pre-osteogenetic cells have undergone an inductive interaction with the brain or spinal cord (Schowing, 1968); (b) that scleral bones only form in the avian eye after ectomesenchymal cells have interacted with epithelial scleral papillae (Coulombre, Coulombre & Mehta, 1962); (c) that the sub-periosteal bone around the shafts of developing long bones only forms after perichondrial cells have been in contact, either with hypertrophic chondrocytes or with their extracellular products (Shimomura, Yoneda & Suzuki, 1975); and (d) that membrane bones of the avian mandible and maxilla only form after ectomesenchymal cells have interacted with the appropriate epithelia (see Introduction). The dentary of the mouse may now be added to this list. The present study demonstrates that cells of the mandibular process must interact with the mandibular epithelium if they are to initiate the sequence of differentiative events which culminate in the differentiation of osteoblasts and osteocytes, the deposition of osteoid and the mineralization of that osteoid to form bone. That this developmental interaction had not occurred by 9 days of gestation was shown by the fact that ectomesenchyme isolated from such aged embryos failed to form bone (except in one case (column II, Table 1), where epithelial cells were found to have been left on the ectomesenchyme). The reinitiation of osteogenesis in recombinants confirmed that presence of the epithelium was a sufficient osteogenetic stimulus for these ectomesenchymal cells. Therefore, the murine dentary resembles the membrane bones of the avian mandible (Tyler & Hall, 1977) in requiring a mandibular epithelial-ectomesenchymal interaction for its initiation. Unlike Meckel's cartilage in avian embryos, initiation of the murine cartilage was dependent upon the presence of the mandibular epithelium. In the chick the ectomesenchymal cells are already able to form cartilage before they reach the mandibular arch (Tyler & Hall, 1977; Hall & Tremaine, 1979), apparently because of prior interactions with cranial epithelial ectoderm (Bee & Thorogood, 1980). In the urodele amphibians the equivalent interaction is between cells derived from the neural crest and the pharyngeal endoderm (see Introduction). Thus, these three classes of vertebrates (Amphibia, Aves, Mammalia) share epithelial-ectomesenchymal interactions as a common component for the initiation of chondrogenesis in the mandibular skeleton, differing only in the nature of the signalling epithelium. Whether these interactions are mediated via
12 262 B. K. HALL epithelial cell products, as is the initiation of scleral chondrogenesis (Newsome, 1972, 1976), or via direct cell-to-cell contact, remains to be determined. The osteogenetic interaction in the chick requires (a) that the epithelium be vital, and proliferating (Hall, 1980), and (b) the presence of epithelially-derived collagen and glycosaminoglycans (Bradamante & Hall, 1980). It was further shown that these interactions, like those which are responsible for the formation of epidermal structures such as hairs, feathers and teeth (Kollar, 1972; Dhouailly, 1975), were neither species nor class specific. Mouse ectomesenchyme of any age responded to chick epithelium by forming both bone and cartilage. Chick ectomesenchyme responded to mouse epithelium older than 9 days by forming bone and cartilage. From these results I concluded that mouse mandibular ectomesenchyme was capable of responding to epithelial influences at 9 days of gestation but that the murine epithelium was not active until 10 days of gestation. A qualitatively similar temporal pattern has been shown for inductively active epithelia from the chick (Hall, 1978 b). In each combination the bone and cartilage formed were typical of the species providing the ectomesenchyme (cf. Fig. 4 and 5), indicating that, while the epithelium was required to initiate osteo- or chondrogenesis, it played no role in determining the type of cartilage or bone which formed. The identification of these tissue interactions provide a new basis for the study of malformations affecting the mammalian facial skeleton. Failure of these tissues to interact, interaction with fewer than normal ectomesenchymal cells, or delayed interactions, could all alter craniofacial growth and/or morphogenesis. Mutant mice, or embryos from females given teratogens, could be used to provide epithelia or ectomesenchyme for recombination with components from wild-type or untreated embryos to further explore the role of such tissue interactions in development. This research has been supported by the National Sciences and Engineering Research Council of Canada (Grant no. A5O56). The Dalhousie University Research Development Fund in the Sciences provided funds to purchase the CO 2 Incubator. Pilot grafts of whole mandibular process and ectomesenchyme from the older embryos were performed by P. Elliott. Sharon Brunt provided expert technical assistance. REFERENCES BARTELMEZ, G. W. (1952). The proliferation of the neural crest from forebrain levels in the rat. Contrib. Embryol. Carnegie Inst. 37, BEE, J. & THOROGOOD, P. (1980). The role of tissue interactions in the skeletogenic differentiation of avian neural crest cells. Devi Biol. (in the press). BERKOVITZ, B. K. B. & MOORE, M. H. (1974). A longitudinal study of replacement patterns of teeth on the lower jaw and tongue in the rainbow trout, Salmo gairdneri. Archs oral Biol. 19, BRADAMANTE, Z. & HALL, B. K. (1980). The role of epithelial collagen and proteoglycan in the initiation of osteogenesis by avian neural crest cells. Anat. Rec. (in the press). CASSIN, C. & CAPURON, A. (1977). Evolution de la capacite morphogenetique de la region stomodeale chez Pembryon de Pleurodeles waltlii Michah (Amphibien Urodele). Etude par
13 Skeletogenesis in the murine mandible 263 transplantation intrablastocelienne et par culture in vitro. W. Roux's Arch, devl Biol. 181, CORSIN, J. (1975). Differentiation in vitro de cartilage a partir des cretes neurales cephaliques chez Pleurodeles waltlii Michah. J. Embryol. exp. Morph. 33, COULOMBRE, A. J., COULOMBRE, J. L. & MEHTA, H. (1962). The skeleton of the eye. I. Conjunctival papillae and scleral ossicles. Devi Biol. 5, CUSIMANO-CAROLLO, T. (1963). Investigation on the ability of the neural folds to induce a mouth in the Discoglossus pictus embryos. Acta Embryol. Morph. Exp. 6, CUSIMANO-CAROLLO, T. (1972). On the mechanism of the formation of the larval mouth in Discoglossus pictus. Acta Embryol. Morph. exp. 4, DAMAS, H. (1944). Recherches sur le developpement de Lampetrafluviatilis L. Contribution a l'etude de la cephalogenese des vertebres. Archs Biol. 55, DAMJANOV, I., SOLTER, D. & SERMAN, D. (1973). Teratocarcinoma with the capacity for differentiation restricted to neuro-ectodermal tissue. Virchow's Arch. Cell Pathol. 13, DE BEER, G. R. (1947). The differentiation of neural crest cells into visceral cartilages and odontoblasts in Amblystoma, and a re-examination of the germ-layer theory. Proc. R. Soc. (Lond.) B 134, DHOUAILLY, D. (1975). Formation of cutaneous appendages in dermal-epidermal recombinations between reptiles, birds and mammals. W. Roux's Arch, devl Biol. 177, DREWS, U., KOCHER-BECKER, U. & DREWS, U. (1972). The induction of visceral cartilage from cranial neural crest by pharyngeal endoderm in hanging drop cultures and the locomotory behaviour of the neural crest cells during cartilage differentiation. W. Roux Arch. EntwMech. org. 171, EPPERLEIN, H. H. (1974). The ectomesenchymal-endodermal interaction-system (EEIS) of Triturus alpestris in tissue culture. 1. Observations on attachment, migration, and differentiation of neural crest cells. Differentiation 2, GRUNEBERG, H. (1943). The development of some external features in mouse embryos. /. Hered. 34, HALL, B. K. (1978a). Developmental and Cellular Skeletal Biology. New York and London: Academic Press. HALL, B. K. (19786). Initiation of osteogenesis by mandibular mesenchyme of the embryonic chick in response to mandibular and non-mandibular epithelia. Archs oral Biol. 23, HALL, B. K. (1978 C). Grafting organs and tissues to the chorioallantoic membrane of the embryonic chick. Tissue Culture Assoc. Manual 4, HALL, B. K. (1980). Viability and proliferation of epithelia and the initiation of osteogenesis within mandibular ectomesenchyme in the embryonic chick. /. Embryol. exp. Morph. 56, HALL, B. K. & TREMAINE, R. (1979). Ability of neural crest cells from the embryonic chick to differentiate into cartilage before their migration away from the neural tube. Anat. Rec. 194, HAMBURGER, V. & HAMILTON, H. L. (1951). A series of normal stages in development of the chick embryo. /. Morph. 88, HORSTADIUS, S. (1950). The Neural Crest; Its Properties and Derivatives in the Light of Experimental Research. London: Oxford University Press. JOLLIE, M. (1971). Some developmental aspects of the head skeleton of the mm Squalus acanthias foetus. /. Morph. 133, KOLLAR, E. J. (1972). The development of the integument: spatial, temporal and phylogenetic factors. Amer. Zool. 12, LEVAK-SVAJGER, B. & SVAJGER, A. (1971). Differentiation of endodermal tissues in homografts of primitive ectoderm from two-layered rat embryonic shields. Experientia 27, LISON, L. (1954). Alcian blue 8G with chlorantine fast red 5B: a technique for selective staining of mucopolysaccharides. Stain Technol. 29,
14 264 B. K. HALL MAYER, T. C. (1973). The migratory pathway of neural cells into the skin of mouse embryos. Devi Biol. 34, MILAIRE, J. (1959). Predifferenciation cytochemique des divers ebauches cephaliques chez l'embryon des souris. Archs Biol. {Liege) 70, MILAIRE, J. (1974). Histochemical aspects of organogenesis in Vertebrates. In Handbuch d. Histochemie, vol. 8, suppl. 3. pp Stuttgart: Fischer-Verlag. MORRISS, G. A. & THOROGOOD, P. V. (1978). An approach to cranial neural crest cell migration and differentiation in mammalian embryos. In Development of Mammals (ed. M. H. Johnson), vol. 3, pp Amsterdam: North Holland. MULNARD, J. (1955). Contribution a la connaissance des enzymes dans l'ontogenese. Archs Biol. {Liege) 66, MURRAY, P. D. F. (1943). The development of the conjunctival papillae and of the scleral bones in the chick embryo. /. Anat. 11, NEWSOME, D. A. (1972). Cartilage induction by retinal pigmented epithelium of chick embryos. Devi. Biol. 27, NEWSOME, D. A. (1976). In vitro stimulation of cartilage in embryonic chick neural crest cells by products of retinal pigmented epithelium. Devi Biol. 49, NEWTH, D. R. (1951). Experiments on the neural crest of the Lamprey embryo. /. exp. Biol. 28, NEWTH, D. R. (1956). On the neural crest of the Lamprey embryo. /. Embryol. exp. Morph. 4, O'RAHILLY, R. (1965). The optic, vestibulocochlear and terminal vomeronasal neural crest in staged human embryos. In The Structure of the Eye (ed. J. W. Rohen), pp Stuttgart: F. K. Schattauer-Verlag. POSWILLO, D. (1974). Otomandibular deformity: pathogenesis as a guide to reconstruction. J. Maxillofac. Surg. 2, REINBOLD, R. (1968). Role dutapetum dans la differenciation de la sclerotique chez l'embryon de poulet. /. Embryol. exp. Morph. 19, SCHAEFFER, B. (1977). The dermal skeleton in fishes. In Problems in Vertebrate Evolution (ed. S. M. Andrews, R. S. Miles and A. D. Walker), Linn, Soc. Symp. 4, pp SCHOWING, J. (1968). Influence inductrice de l'encephale embryonnaire sur la developpement du crane chez le poulet. III. Mise en evidence du role inducteur de l'encephale dans Posteogenese du crane embryonnaire du poulet. /. Embryol. exp. Morph. 19, SHIMOMURA, Y., YONEDA, T. & SUZUKI, F. (1975). Osteogenesis by chondrocytes from growth cartilage of rat rib. Calc. Tissue Res. 19, SVAJGER, A. & LEVAK-SVAJGER, B. (1976). Differentiation of the first branchial arch mesenchyme of the rat embryo in renal homografts and in organ culture. Bull. Scient. Cons. Acads RSF Yougosl. (A) 21, TYLER, M. S. (1978). Epithelia influences on membrane bone formation in the maxilla of the embryonic chick. Anat. Rec. 192, TYLER, M. S. & HALL, B. K. (1977). Epithelial influences on skeletogenesis in the mandible of the embryonic chick. Anat. Rec. 188, TYLER, M. S. & MCCOBB, D. P. (1980). The genesis of membrane bone in the embryonic chick maxilla: epithelial-mesenchymal tissue recombination studies. J. Embryol. exp. Morph. 56, WAGNER, G. (1949). Die Bedeutung der Neuralleiste fiir die Kopfgestaltung der Amphibienlarven, Untersuchungen an Chimaeren von Triton und Bombinator. Rev. suisse Zool. 56, WILDE, C. E. JR. (1955). The urodele neuroepithelium. The differentiation in vitro of the cranial neural crest. J. exp. Zool. 130, {Received 9 January 1980, revised 14 March 1980)
Proximal distal sequence of development of the skeletal tissues in the penis of rat and the inductive effect of epithelium
 J. Embryol. exp. Morph. 92, 133-143 (1986) 133 Printed in Great Britain The Company of Biologists Limited 1986 Proximal distal sequence of development of the skeletal tissues in the penis of rat and the
J. Embryol. exp. Morph. 92, 133-143 (1986) 133 Printed in Great Britain The Company of Biologists Limited 1986 Proximal distal sequence of development of the skeletal tissues in the penis of rat and the
Epithelial-mesenchymal interactions in the development of chick facial primordia and the target of retinoid action
 Development 99, 341-351 (1987) Printed in Great Britain The Company of Biologists Limited 1987 341 Epithelial-mesenchymal interactions in the development of chick facial primordia and the target of retinoid
Development 99, 341-351 (1987) Printed in Great Britain The Company of Biologists Limited 1987 341 Epithelial-mesenchymal interactions in the development of chick facial primordia and the target of retinoid
Mesenchymal control over elongating and branching morphogenesis in salivary gland development
 J. Embryol. exp. Morph. Vol. 66, pp. 9-, 98 9 Printed in Great Britain Company of Biologists Limited 98 Mesenchymal control over elongating and branching morphogenesis in salivary gland development ByHIROYUKI
J. Embryol. exp. Morph. Vol. 66, pp. 9-, 98 9 Printed in Great Britain Company of Biologists Limited 98 Mesenchymal control over elongating and branching morphogenesis in salivary gland development ByHIROYUKI
The site of action of the ichthyosis locus (ic) in the mouse, as determined by dermal-epidermal recombinations
 /. Embryol. exp. Morph. Vol. 32, 3, pp. 715-721, 1974 715 Printed in Great Britain The site of action of the ichthyosis locus (ic) in the mouse, as determined by dermal-epidermal recombinations BY MARGARET
/. Embryol. exp. Morph. Vol. 32, 3, pp. 715-721, 1974 715 Printed in Great Britain The site of action of the ichthyosis locus (ic) in the mouse, as determined by dermal-epidermal recombinations BY MARGARET
Bone. Development. Tim Arnett. University College London. Department of Anatomy and Developmental Biology
 Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Bone Development Tim Arnett Department of Anatomy and Developmental Biology University College London Bone development Outline Bone composition matrix + mineral Bone formation - intramembranous & endochondral
Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule
 /. Embryol. exp. Morph. Vol. 5, pp. -3, 79 Printed in Great Britain Company of Biologists Limited 79 Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule By H. FUJIMOTO
/. Embryol. exp. Morph. Vol. 5, pp. -3, 79 Printed in Great Britain Company of Biologists Limited 79 Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule By H. FUJIMOTO
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian degeneration
 The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS
 ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM
 LECTURE 4 SKULL NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM CRANIUM NEUROCRANIUM (protective case around brain) VISCEROCRANIUM (skeleton of face) NASOMAXILLARY COMPLEX MANDIBLE (DESMOCRANIUM)
LECTURE 4 SKULL NEUROCRANIUM VISCEROCRANIUM VISCEROCRANIUM VISCEROCRANIUM CRANIUM NEUROCRANIUM (protective case around brain) VISCEROCRANIUM (skeleton of face) NASOMAXILLARY COMPLEX MANDIBLE (DESMOCRANIUM)
Skeletal Development Multiple Cellular Origins. Intramembranous Bone. Endochondrial Bone. Cartilage template of the limb in the Chick wing
 Skeletal Development Multiple Cellular Origins 1 - Paraxial Mesoderm Somite, Sclerotome Axial Skeleton (e.g. vertebra) 2 - Lateral Plate Mesoderm Appendicular Skeleton (e.g. limb) 3 - Neural Crest Head
Skeletal Development Multiple Cellular Origins 1 - Paraxial Mesoderm Somite, Sclerotome Axial Skeleton (e.g. vertebra) 2 - Lateral Plate Mesoderm Appendicular Skeleton (e.g. limb) 3 - Neural Crest Head
Head and Neck Development and Malformations
 Head and Neck Development and Malformations Yang Chai, DDS, PhD Professor George and MaryLou Boone Chair Ostrow School of Dentistry of USC ychai@usc.edu C D E A. B Learning Objectives - Learn cranial neural
Head and Neck Development and Malformations Yang Chai, DDS, PhD Professor George and MaryLou Boone Chair Ostrow School of Dentistry of USC ychai@usc.edu C D E A. B Learning Objectives - Learn cranial neural
An experimental investigation into the early development of the chick elbow joint
 /. Embryol. exp. Morph. Vol. 39,pp. 115-127, 1977 Printed in Great Britain An experimental investigation into the early development of the chick elbow joint By N. HOLDER 1 From the Department of Biology
/. Embryol. exp. Morph. Vol. 39,pp. 115-127, 1977 Printed in Great Britain An experimental investigation into the early development of the chick elbow joint By N. HOLDER 1 From the Department of Biology
A Congenital Defect in the Spinal Cord of the Manx Cat
 Vet, Path. 8: 232-238 (1971) A Congenital Defect in the Spinal Cord of the Manx Cat A. H. MARTIN Department of Anatomy, University of Wisconsin, Madison Wisc. Abstract. The lumbar part of the spinal cords
Vet, Path. 8: 232-238 (1971) A Congenital Defect in the Spinal Cord of the Manx Cat A. H. MARTIN Department of Anatomy, University of Wisconsin, Madison Wisc. Abstract. The lumbar part of the spinal cords
The Occurrence and Morphogenesis of Melanocytes in the Connective Tissues of the PET/MCV Mouse Strain 1
 The Occurrence and Morphogenesis of Melanocytes in the Connective Tissues of the PET/MCV Mouse Strain 1 by STUART E. NICHOLS, JR. 2 and WILLIE M. REAMS, JR. 3 From the Department of Anatomy, Medical College
The Occurrence and Morphogenesis of Melanocytes in the Connective Tissues of the PET/MCV Mouse Strain 1 by STUART E. NICHOLS, JR. 2 and WILLIE M. REAMS, JR. 3 From the Department of Anatomy, Medical College
Tooth development in the 'crooked' mouse
 /. Embryo!, exp. Morph. Vol. 41, pp. 279-287, 1977 279 Printed in Great Britain Company of Biologists Limited 1977 Tooth development in the 'crooked' mouse By J. A. SOFAER 1 From the University of Edinburgh,
/. Embryo!, exp. Morph. Vol. 41, pp. 279-287, 1977 279 Printed in Great Britain Company of Biologists Limited 1977 Tooth development in the 'crooked' mouse By J. A. SOFAER 1 From the University of Edinburgh,
PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis)
 Original Article Buffalo Bulletin (March 2014) Vol.33 No.1 PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis) M. Santhi Lakshmi *, T.S. Chandrasekhara Rao and
Original Article Buffalo Bulletin (March 2014) Vol.33 No.1 PRENATAL DEVELOPMENT OF CARTILAGINOUS VISCEROCRANIUM OF SKULL IN THE BUFFALO (Bubalus bubalis) M. Santhi Lakshmi *, T.S. Chandrasekhara Rao and
Experimental Neoplastic Formation in Embryonic Chick Brains
 Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
Growth and morphogenesis of the fibula in the chick embryo
 /. Embryol. exp. Morph. 75, 101-116 (1983) Printed in Great Britain The Company of Biologists Limited 1983 Growth and morphogenesis of the fibula in the chick embryo By CHARLES W. ARCHER 1, AMATA HORNBRUCH
/. Embryol. exp. Morph. 75, 101-116 (1983) Printed in Great Britain The Company of Biologists Limited 1983 Growth and morphogenesis of the fibula in the chick embryo By CHARLES W. ARCHER 1, AMATA HORNBRUCH
Essentials in Head and Neck Embryology. Part 3 Development of the head, face, and oral cavity
 Essentials in Head and Neck Embryology Part 3 Development of the head, face, and oral cavity Outline General overview of prenatal development Embryonic period phase 1 Formation of bilaminar disk Formation
Essentials in Head and Neck Embryology Part 3 Development of the head, face, and oral cavity Outline General overview of prenatal development Embryonic period phase 1 Formation of bilaminar disk Formation
Interactions of the endocrine system, bone and oral health
 Interactions of the endocrine system, bone and oral health All bones are not equal! Dense high proportion of cortical bone High proportion of trabecular bone Mandible Functions: mastication, respiration,
Interactions of the endocrine system, bone and oral health All bones are not equal! Dense high proportion of cortical bone High proportion of trabecular bone Mandible Functions: mastication, respiration,
The Culture in vitro of Urogenital Organs of Pleurodeles waltlii
 The Culture in vitro of Urogenital Organs of Pleurodeles waltlii by CHARLES L. FOOTE and FLORENCE M. FOOTE 1 From the Laboratoire d'embryologie experimentale du College de France et du C.N.R.S., Paris
The Culture in vitro of Urogenital Organs of Pleurodeles waltlii by CHARLES L. FOOTE and FLORENCE M. FOOTE 1 From the Laboratoire d'embryologie experimentale du College de France et du C.N.R.S., Paris
Laser irradiation of the chick embryo germinal crescent
 /. Embryol exp. Morph. Vol. 26, 1, pp. 31-36, 1971 31 Printed in Great Britain Laser irradiation of the chick embryo germinal crescent By MARTHA FEARON MIMS 1 AND ROBERT GILMORE McKINNELL 2 From the Department
/. Embryol exp. Morph. Vol. 26, 1, pp. 31-36, 1971 31 Printed in Great Britain Laser irradiation of the chick embryo germinal crescent By MARTHA FEARON MIMS 1 AND ROBERT GILMORE McKINNELL 2 From the Department
Positional signalling and the development of the humerus in the chick limb bud
 Development 100, 333-338 (1987) Printed in Great Britain The Company of Biologists Limited 1987 333 Positional signalling and the development of the humerus in the chick limb bud L. WOLPERT and AMATA HORNBRUCH
Development 100, 333-338 (1987) Printed in Great Britain The Company of Biologists Limited 1987 333 Positional signalling and the development of the humerus in the chick limb bud L. WOLPERT and AMATA HORNBRUCH
Osteology. Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College
 Osteology Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College Functions of the Skeletal System: Support Movement Protection Hemopoiesis Electrolyte balance (Ca ++ /PO -3 4 ) Acid-base balance Storage
Osteology Dr. Carmen E. Rexach Anatomy 35 Mt San Antonio College Functions of the Skeletal System: Support Movement Protection Hemopoiesis Electrolyte balance (Ca ++ /PO -3 4 ) Acid-base balance Storage
Further studies on the melanophores of periodic albino mutant of Xenopus laevis
 J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in female chick embryo
 J. Embryol exp. Morph. Vol. 67, pp. 81-87, 1982 g \ Printed in Great Britain Company of Biologists Limited 1982 Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in
J. Embryol exp. Morph. Vol. 67, pp. 81-87, 1982 g \ Printed in Great Britain Company of Biologists Limited 1982 Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in
Skeletal System. Prof. Dr. Malak A. Al-yawer Department of Anatomy/Embryology Section
 Skeletal System Prof. Dr. Malak A. Al-yawer Department of Anatomy/Embryology Section Learning objectives At the end of this lecture, the medical student will be able to: State the embryonic origin of skeletal
Skeletal System Prof. Dr. Malak A. Al-yawer Department of Anatomy/Embryology Section Learning objectives At the end of this lecture, the medical student will be able to: State the embryonic origin of skeletal
FORMATION OF BONE. Intramembranous Ossification. Bone-Lec-10-Prof.Dr.Adnan Albideri
 FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
FORMATION OF BONE All bones are of mesodermal origin. The process of bone formation is called ossification. We have seen that formation of most bones is preceded by the formation of a cartilaginous model,
THE INDUCTION OF HAIR FOLLICLES BY EMBRYONIC DERMAL PAPILLAE*
 THE JouR AL OF INVESTIGATIVE DERMATOLOGY Copyright 1970 by The Williams & Wilkins Co. Vol. 55, No. 6 Printed in U.S.A. THE INDUCTION OF HAIR FOLLICLES BY EMBRYONIC DERMAL PAPILLAE* EDWARD J. KOLLAR, PH.D.
THE JouR AL OF INVESTIGATIVE DERMATOLOGY Copyright 1970 by The Williams & Wilkins Co. Vol. 55, No. 6 Printed in U.S.A. THE INDUCTION OF HAIR FOLLICLES BY EMBRYONIC DERMAL PAPILLAE* EDWARD J. KOLLAR, PH.D.
 Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal
Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal apparatus. Drawings illustrating the human pharyngeal
The embryological basis of craniofacial dysplasias D. E. POSWILLO
 Postgraduate Medical Journal (August 1977) 53, 517-522. The embryological basis of craniofacial dysplasias D. E. POSWILLO D.D.S., D.Sc., F.D.S., F.I.Biol., M.R.C.Path. Department of Teratology, Royal College
Postgraduate Medical Journal (August 1977) 53, 517-522. The embryological basis of craniofacial dysplasias D. E. POSWILLO D.D.S., D.Sc., F.D.S., F.I.Biol., M.R.C.Path. Department of Teratology, Royal College
Pharyngeal Apparatus. Pouches Endoderm Grooves Ectoderm Arch Neural Crest Somitomeres Aortic Arch - Vessel
 Pharyngeal Apparatus Pouches Endoderm Grooves Ectoderm Arch Neural Crest Somitomeres Aortic Arch - Vessel Segmental Organization Humans: Arch 1-4 prominent Arch 5 absent Arch 6 - transient First Arch Face
Pharyngeal Apparatus Pouches Endoderm Grooves Ectoderm Arch Neural Crest Somitomeres Aortic Arch - Vessel Segmental Organization Humans: Arch 1-4 prominent Arch 5 absent Arch 6 - transient First Arch Face
SKELETAL SYSTEM. Jhia Anjela D. Rivera 1,2 1. Department of Biology, College of Science, Polytechnic University of the Philippines 2
 SKELETAL SYSTEM Jhia Anjela D. Rivera 1,2 1 Department of Biology, College of Science, Polytechnic University of the Philippines 2 Department of Biological Sciences, School of Science and Technology, Centro
SKELETAL SYSTEM Jhia Anjela D. Rivera 1,2 1 Department of Biology, College of Science, Polytechnic University of the Philippines 2 Department of Biological Sciences, School of Science and Technology, Centro
Lecture 2: Skeletogenesis
 Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Jilin University School of Stomatology Skeletogenesis Lecture 2: Skeletogenesis Aug. 18, 2015 Yuji Mishina, Ph.D. mishina@umich.edu Student will describe Development of Bone - the general anatomy of bone
Metabolism and Glycogen Formation in the Liver of the Chicken Embryo
 Metabolism and Glycogen Formation in the Liver of the Chicken Embryo by R. j. O'CONNOR 1 From the John Burford Carlill Pathological Laboratories, Westminster School of Medicine INTRODUCTION THE investigations
Metabolism and Glycogen Formation in the Liver of the Chicken Embryo by R. j. O'CONNOR 1 From the John Burford Carlill Pathological Laboratories, Westminster School of Medicine INTRODUCTION THE investigations
Formation of marrow cavity and ossification in mouse limb buds grown in vitro
 /. Embryol. exp. Morph. Vol. 56, pp. 301-307, 1980 301 Printed in Great Britain Company of Biologists Limited 1980 Formation of marrow cavity and ossification in mouse limb buds grown in vitro By D. R.JOHNSON
/. Embryol. exp. Morph. Vol. 56, pp. 301-307, 1980 301 Printed in Great Britain Company of Biologists Limited 1980 Formation of marrow cavity and ossification in mouse limb buds grown in vitro By D. R.JOHNSON
Growth pattern of the sex ducts in foetal mouse hermaphrodites
 /. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
/. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
04 Development of the Face and Neck. Development of the Face Development of the neck
 04 Development of the Face and Neck Development of the Face Development of the neck Development of the face Overview of facial development The fourth week ~ the twelfth week of prenatal development Between
04 Development of the Face and Neck Development of the Face Development of the neck Development of the face Overview of facial development The fourth week ~ the twelfth week of prenatal development Between
The polarity of the dental lamina in the regenerating salamander jaw
 J. Embryol. exp. Morpli. Vol. 30, 3 pp. 635-646, 1973 635 Printed in Great Britain The polarity of the dental lamina in the regenerating salamander jaw ByHEBER T. GRAVER 1 From the Department of Histology
J. Embryol. exp. Morpli. Vol. 30, 3 pp. 635-646, 1973 635 Printed in Great Britain The polarity of the dental lamina in the regenerating salamander jaw ByHEBER T. GRAVER 1 From the Department of Histology
Development of the Pharyngeal Arches
 Development of the Pharyngeal Arches Thomas A. Marino, Ph.D. Temple University School of Medicine Competencies: Upon completion of this section of the course, the student must be able to: 1. Recall the
Development of the Pharyngeal Arches Thomas A. Marino, Ph.D. Temple University School of Medicine Competencies: Upon completion of this section of the course, the student must be able to: 1. Recall the
Department of Anatomy, Glasgow University
 THE UPTAKE OF LABELLED SULPHATE INJECTED INTO THE HOST ANIMAL BY CARTILAGE HOMOGRAFTS By G. M. WYBURN, D.Sc., F.R.F.P.S.G., and P. BACSICH, D.Sc., M.D. Department of Anatomy, Glasgow University INTRODUCTION
THE UPTAKE OF LABELLED SULPHATE INJECTED INTO THE HOST ANIMAL BY CARTILAGE HOMOGRAFTS By G. M. WYBURN, D.Sc., F.R.F.P.S.G., and P. BACSICH, D.Sc., M.D. Department of Anatomy, Glasgow University INTRODUCTION
Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm
 Development of face Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm The ectoderm forms the neural groove, then tube The neural tube lies in the mesoderm
Development of face Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm The ectoderm forms the neural groove, then tube The neural tube lies in the mesoderm
Skeletal Regeneration in Amphibians
 Skeletal Regeneration in Amphibians by R. j. GOSS 1 From the Department of Biology, Brown University, Providence, R.I., U.S.A. WITH ONE PLATE INTRODUCTION THE failure of de novo regeneration of extirpated
Skeletal Regeneration in Amphibians by R. j. GOSS 1 From the Department of Biology, Brown University, Providence, R.I., U.S.A. WITH ONE PLATE INTRODUCTION THE failure of de novo regeneration of extirpated
Gene expression at the pink-eyed dilution (p) locus in the mouse is confirmed to be pigment cell autonomous using recombinant embryonic skin grafts
 /. Embryol. exp. Morph. 87 65-73 (1985) 55 Printed in Great Britain The Company of Biologists Limited 1985 Gene expression at the pink-eyed dilution (p) locus in the mouse is confirmed to be pigment cell
/. Embryol. exp. Morph. 87 65-73 (1985) 55 Printed in Great Britain The Company of Biologists Limited 1985 Gene expression at the pink-eyed dilution (p) locus in the mouse is confirmed to be pigment cell
In vitro development of palatal tissues from embryonic mice
 J. Embryol. exp. Morph. Vol. 38, pp. 19-36, 1977 19 Printed in Great Britain In vitro development of palatal tissues from embryonic mice II. Tissue isolation and recombination studies By MARY S. TYLER
J. Embryol. exp. Morph. Vol. 38, pp. 19-36, 1977 19 Printed in Great Britain In vitro development of palatal tissues from embryonic mice II. Tissue isolation and recombination studies By MARY S. TYLER
CASE OF CONGENITAL CYSTIC EYE AND ACCESSORY LIMB OF THE LOWER EYELID*
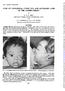 Brit. J. Ophthal. (1966) 50, 409 CASE OF CONGENITAL CYSTIC EYE AND ACCESSORY LIMB OF THE LOWER EYELID* BY N. S. C. RICE Department ofpathology, Institute of Ophthalmology, London, AND S. P. MINWALLA AND
Brit. J. Ophthal. (1966) 50, 409 CASE OF CONGENITAL CYSTIC EYE AND ACCESSORY LIMB OF THE LOWER EYELID* BY N. S. C. RICE Department ofpathology, Institute of Ophthalmology, London, AND S. P. MINWALLA AND
Distinct Roles Of CCN1 And CCN2 In Limb Development
 Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
Anti-mesodermal Activity of a Nitrogen Mustard Derivative
 /. Embryol. exp. Morph., Vol. 11, Part 4, pp. 689-696, December 1963 Printed in Great Britain Anti-mesodermal Activity of a Nitrogen Mustard Derivative by A. JURAND 1 From the Institute of Animal Genetics,
/. Embryol. exp. Morph., Vol. 11, Part 4, pp. 689-696, December 1963 Printed in Great Britain Anti-mesodermal Activity of a Nitrogen Mustard Derivative by A. JURAND 1 From the Institute of Animal Genetics,
Biology 323 Human Anatomy for Biology Majors Lecture 3 Dr. Stuart S. Sumida. Integument Support Structures; Axial Skeleton
 Biology 323 Human Anatomy for Biology Majors Lecture 3 Dr. Stuart S. Sumida Integument Support Structures; Axial Skeleton Integumentary System Functions Protection Sensory Synthesis The Skin Epidermis
Biology 323 Human Anatomy for Biology Majors Lecture 3 Dr. Stuart S. Sumida Integument Support Structures; Axial Skeleton Integumentary System Functions Protection Sensory Synthesis The Skin Epidermis
Evidence for a metameric pattern in the development of the chick humerus
 /. Embryol. exp. Morph. Vol. 61, pp. 19125, 1981 Printed in Great Britain Company of Biologists Limited 1981 Evidence for a metameric pattern in the development of the chick humerus By TRENT D. STEPHENS
/. Embryol. exp. Morph. Vol. 61, pp. 19125, 1981 Printed in Great Britain Company of Biologists Limited 1981 Evidence for a metameric pattern in the development of the chick humerus By TRENT D. STEPHENS
Lecture 1: Embryology of the Limbs (Development of Skeletal and Muscular Systems)
 Lecture 1: Embryology of the Limbs (Development of Skeletal and Muscular Systems) Musculoskeletal Block Editing file Colour Index: Red: Important Gray: Notes Green: Dr. s notes After this lecture you should
Lecture 1: Embryology of the Limbs (Development of Skeletal and Muscular Systems) Musculoskeletal Block Editing file Colour Index: Red: Important Gray: Notes Green: Dr. s notes After this lecture you should
The role of mechanical stimuli induced by prenatal movements in skeletal development
 The role of mechanical stimuli induced by prenatal movements in skeletal development Niamh Nowlan, PhD Department of Bioengineering, Imperial College London Mechanobiology of Bone Mechanoregulation of
The role of mechanical stimuli induced by prenatal movements in skeletal development Niamh Nowlan, PhD Department of Bioengineering, Imperial College London Mechanobiology of Bone Mechanoregulation of
Organogenesis of Heart, Kidney, Nervous System & Sense Organs. [ GLOSSARY ]
![Organogenesis of Heart, Kidney, Nervous System & Sense Organs. [ GLOSSARY ] Organogenesis of Heart, Kidney, Nervous System & Sense Organs. [ GLOSSARY ]](/thumbs/86/94287046.jpg) Organogenesis of Heart, Kidney, Nervous System & Sense Organs. [ GLOSSARY ] Subject : Zoology Course : 3rd Year, B.Sc. Undergraduate UGC Syllabus, Model - 1 Paper No. : Z-305B & Title : Developmental Biology
Organogenesis of Heart, Kidney, Nervous System & Sense Organs. [ GLOSSARY ] Subject : Zoology Course : 3rd Year, B.Sc. Undergraduate UGC Syllabus, Model - 1 Paper No. : Z-305B & Title : Developmental Biology
06 Tooth Development and Eruption
 + 06 Tooth Development and Eruption Tooth development Root development PDL and alveolar bone development Primary tooth eruption and shedding Permanent tooth eruption Q. Where and how tooth starts to form?
+ 06 Tooth Development and Eruption Tooth development Root development PDL and alveolar bone development Primary tooth eruption and shedding Permanent tooth eruption Q. Where and how tooth starts to form?
Development of the skin and its derivatives
 Development of the skin and its derivatives Resources: http://php.med.unsw.edu.au/embryology/ Larsen s Human Embryology The Developing Human: Clinically Oriented Embryology Dr Annemiek Beverdam School
Development of the skin and its derivatives Resources: http://php.med.unsw.edu.au/embryology/ Larsen s Human Embryology The Developing Human: Clinically Oriented Embryology Dr Annemiek Beverdam School
Skeletal Tissues Dr. Ali Ebneshahidi
 Skeletal Tissues Dr. Ali Ebneshahidi Functions of Bones 1. Support and protection: Bones give shape to body structure. Bones provide support to body weight. Certain bones protect vital internal organs
Skeletal Tissues Dr. Ali Ebneshahidi Functions of Bones 1. Support and protection: Bones give shape to body structure. Bones provide support to body weight. Certain bones protect vital internal organs
Chapter 7. Skeletal System
 Chapter 7 Skeletal System 1 Introduction: A. Bones are very active, living tissues B. Each bone is made up of several types of tissues and so is an organ. C. Bone functions include: muscle attachment,
Chapter 7 Skeletal System 1 Introduction: A. Bones are very active, living tissues B. Each bone is made up of several types of tissues and so is an organ. C. Bone functions include: muscle attachment,
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Fig Articular cartilage. Epiphysis. Red bone marrow Epiphyseal line. Marrow cavity. Yellow bone marrow. Periosteum. Nutrient foramen Diaphysis
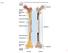 Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
Fig. 7.1 Articular cartilage Epiphysis Red bone marrow Epiphyseal line Marrow cavity Yellow bone marrow Nutrient foramen Diaphysis Site of endosteum Compact bone Spongy bone Epiphyseal line Epiphysis Articular
Skeletal Tissues. Dr. Ali Ebneshahidi
 Skeletal Tissues Dr. Ali Ebneshahidi Functions of Bones 1. Support and protection : Bones give shape to body structure. Bones provide support to body weight. Certain bones protect vital internal organs
Skeletal Tissues Dr. Ali Ebneshahidi Functions of Bones 1. Support and protection : Bones give shape to body structure. Bones provide support to body weight. Certain bones protect vital internal organs
Teeth, orofacial development and
 Teeth, orofacial development and cleft anomalies Miroslav Peterka Variability of jaws in vertebrates. (A) cartilaginous fish shark; (B) an example of a bone fish; (C ) amphibian frog; (D) reptile - turtle;
Teeth, orofacial development and cleft anomalies Miroslav Peterka Variability of jaws in vertebrates. (A) cartilaginous fish shark; (B) an example of a bone fish; (C ) amphibian frog; (D) reptile - turtle;
Functions of the Skeletal System. Chapter 6: Osseous Tissue and Bone Structure. Classification of Bones. Bone Shapes
 Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
Chapter 6: Osseous Tissue and Bone Structure Functions of the Skeletal System 1. Support 2. Storage of minerals (calcium) 3. Storage of lipids (yellow marrow) 4. Blood cell production (red marrow) 5. Protection
Proliferation and migration of primordial germ cells during compensatory growth in mouse embryos
 /. Embryol. exp. Morph. Vol. 64, pp. 133-147, 1981 133 Printed in Great Britain Company of Biologists Limited 1981 Proliferation and migration of primordial germ cells during compensatory growth in mouse
/. Embryol. exp. Morph. Vol. 64, pp. 133-147, 1981 133 Printed in Great Britain Company of Biologists Limited 1981 Proliferation and migration of primordial germ cells during compensatory growth in mouse
The neural cell cycle in the looptail (Lp) mutant mouse
 /. Embryol. exp. Morph. Vol. 32, 3, pp. 697-705, 1974 697 Printed in Great Britain The neural cell cycle in the looptail (Lp) mutant mouse By DORIS B. WILSON 1 AND E. M. CENTER 2 From the Department of
/. Embryol. exp. Morph. Vol. 32, 3, pp. 697-705, 1974 697 Printed in Great Britain The neural cell cycle in the looptail (Lp) mutant mouse By DORIS B. WILSON 1 AND E. M. CENTER 2 From the Department of
Chapter 7 /8 pgs SKELETAL TISSUES AND THE SKELETAL SYSTEM
 Chapter 7 /8 pgs. 189-250 SKELETAL TISSUES AND THE SKELETAL SYSTEM Skeletal Tissue Introduction Bone and cartilage are a specialized types of connective tissue Individual Bones are considered separate
Chapter 7 /8 pgs. 189-250 SKELETAL TISSUES AND THE SKELETAL SYSTEM Skeletal Tissue Introduction Bone and cartilage are a specialized types of connective tissue Individual Bones are considered separate
Embryonic malformations in rats, resulting from maternal diabetes: preliminary observations
 /. Embryol. exp. Morph. Vol. 41, pp. 93-99, 1977 93 Printed in Great Britain Company of Biologists Limited 1977 Embryonic malformations in rats, resulting from maternal diabetes: preliminary observations
/. Embryol. exp. Morph. Vol. 41, pp. 93-99, 1977 93 Printed in Great Britain Company of Biologists Limited 1977 Embryonic malformations in rats, resulting from maternal diabetes: preliminary observations
Due in Lab. Due next week in lab - Scientific America Article Select one article to read and complete article summary
 Due in Lab 1. Skeletal System 33-34 2. Skeletal System 26 3. PreLab 6 Due next week in lab - Scientific America Article Select one article to read and complete article summary Cell Defenses and the Sunshine
Due in Lab 1. Skeletal System 33-34 2. Skeletal System 26 3. PreLab 6 Due next week in lab - Scientific America Article Select one article to read and complete article summary Cell Defenses and the Sunshine
For more information about how to cite these materials visit
 Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
Author(s): University of Michigan Medical School, Department of Cell and Developmental Biology License: Unless otherwise noted, the content of this course material is licensed under a Creative Commons
Development of teeth. 5.DM - Pedo
 Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Skeletal System. Chapter 6.1 Human Anatomy & Physiology
 Skeletal System Chapter 6.1 Human Anatomy & Physiology Overview of Skeletal System Bones Joints Skeletal System Cartilage Tendons (bone to muscle) Ligaments (bone to bone) Function of the Skeletal System
Skeletal System Chapter 6.1 Human Anatomy & Physiology Overview of Skeletal System Bones Joints Skeletal System Cartilage Tendons (bone to muscle) Ligaments (bone to bone) Function of the Skeletal System
The appearance of muscle protein and myofibrils within the embryonic chick limb-bud
 /. Embryol. exp. Morph. Vol. 30, 3, pp. 673-679, 1973 673 Printed in Great Britain The appearance of muscle protein and myofibrils within the embryonic chick limb-bud By P. V. THOROGOOD 1 From the Department
/. Embryol. exp. Morph. Vol. 30, 3, pp. 673-679, 1973 673 Printed in Great Britain The appearance of muscle protein and myofibrils within the embryonic chick limb-bud By P. V. THOROGOOD 1 From the Department
a) They are the most common cause of pediatric kidney failure. b) They are always symptomatic. c) They can be asymmetric.
 Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Pharyngeal apparatus. - At the third week, it is a 3 layered structure: ectoderm, mesoderm and endoderm. This is called trilaminar disc
 Pharyngeal apparatus Remember from the first year embryology - The embryo was disc shaped in the second week of development (this is called embryonic disc) and it is a 2 layered disc (composed of two layers)---bilaminar
Pharyngeal apparatus Remember from the first year embryology - The embryo was disc shaped in the second week of development (this is called embryonic disc) and it is a 2 layered disc (composed of two layers)---bilaminar
evolution and development of primate teeth
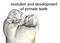 evolution and development of primate teeth diversity of mammalian teeth upper left molars buccal mesial distal lingual Jernvall & Salazar-Ciudad 07 trends in dental evolution many similar single-cusped
evolution and development of primate teeth diversity of mammalian teeth upper left molars buccal mesial distal lingual Jernvall & Salazar-Ciudad 07 trends in dental evolution many similar single-cusped
The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos
 /. Embryol. exp. Morph. Vol. 35, 1, pp. 169-178, 1976 169 Printed in Great Britain The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos By D. R. POLLARD,
/. Embryol. exp. Morph. Vol. 35, 1, pp. 169-178, 1976 169 Printed in Great Britain The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos By D. R. POLLARD,
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue
 The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
The Skeletal System:Bone Tissue Dynamic and ever-changing throughout life Skeleton composed of many different tissues cartilage, bone tissue, epithelium, nerve, blood forming tissue, adipose, and dense
DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiolog
 DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiologist.at an early age- she eloped with a statistician,
DEFINATION Growth was concieved by an anatomist, born to a biologist, delivered by a physician, left on a chemist doorstep, and adopted by a physiologist.at an early age- she eloped with a statistician,
A technique for introducing localized long-lasting implants in the chick embryo
 /. Embryol. exp. Morph. Vol. 39, pp. 261-266, 1977 261 Printed in Great Britain SHORT PAPERS A technique for introducing localized long-lasting implants in the chick embryo By MARIETA B. HEATON 1 From
/. Embryol. exp. Morph. Vol. 39, pp. 261-266, 1977 261 Printed in Great Britain SHORT PAPERS A technique for introducing localized long-lasting implants in the chick embryo By MARIETA B. HEATON 1 From
BONE TISSUE. Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology
 BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
BONE TISSUE Dr. Heba Kalbouneh Associate Professor of Anatomy and Histology BONE FUNCTION Support Protection (protect internal organs) Movement (provide leverage system for skeletal muscles, tendons, ligaments
Anatomy and Physiology Tissue Review
 Anatomy and Physiology Tissue Review OVERVIEW Histology practicals can be rough, especially when access to slides is limited to the lab period. This resource provides an opportunity to learn or review
Anatomy and Physiology Tissue Review OVERVIEW Histology practicals can be rough, especially when access to slides is limited to the lab period. This resource provides an opportunity to learn or review
Elevation of lesioned palatal shelves in vitro
 /. Embryol. exp. Morph. Vol. 54, pp. 229-240, 1979 229 Printed in Great Britain Company of Biologists Limited 1979 Elevation of lesioned palatal shelves in vitro By L. L. BRINKLEY 1 AND M. M. VICKERMAN
/. Embryol. exp. Morph. Vol. 54, pp. 229-240, 1979 229 Printed in Great Britain Company of Biologists Limited 1979 Elevation of lesioned palatal shelves in vitro By L. L. BRINKLEY 1 AND M. M. VICKERMAN
Cartilage Engineering From Autologous Fat For Oro-Facial Deformity Reconstruction - Focus On The Nose
 Cartilage Engineering From Autologous Fat For Oro-Facial Deformity Reconstruction - Focus On The Nose 12-month progress report submitted to AOMS Mr Atheer Ujam PhD student Primary supervisor: Professor
Cartilage Engineering From Autologous Fat For Oro-Facial Deformity Reconstruction - Focus On The Nose 12-month progress report submitted to AOMS Mr Atheer Ujam PhD student Primary supervisor: Professor
Skeleton. Flexibility. Protection of vital organs. Strength
 Skeleton Flexibility Protection of vital organs Strength Skeletal defects Developmental defects Degenerative diseases Fibrous dysplasia (fibrous tissue develops in place of normal bones), Cleidocranial
Skeleton Flexibility Protection of vital organs Strength Skeletal defects Developmental defects Degenerative diseases Fibrous dysplasia (fibrous tissue develops in place of normal bones), Cleidocranial
Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System. Assoc.Prof. E.Elif Güzel, M.D.
 Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System Assoc.Prof. E.Elif Güzel, M.D. Third week of Embryogenesis Primitive streak/pit appears on the epiblast (day
Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System Assoc.Prof. E.Elif Güzel, M.D. Third week of Embryogenesis Primitive streak/pit appears on the epiblast (day
The Skeletal System. Chapter 7a. Skeletal System Introduction Functions of the skeleton Framework of bones The skeleton through life
 The Skeletal System Skeletal System Introduction Functions of the skeleton Framework of bones The skeleton through life Chapter 7a Support Protection Movement Storage areas Minerals Lipids Hemopoiesis
The Skeletal System Skeletal System Introduction Functions of the skeleton Framework of bones The skeleton through life Chapter 7a Support Protection Movement Storage areas Minerals Lipids Hemopoiesis
Fig.9.2. Structure of embryonic brain
 T Chapter 9 Development of Ectodermal Organs he ectoderm gives rise to 3 separate cell populations: neural(plate) ectoderm, neural crest cells, and epiderm (general body ectoderm). A primordium (anlage)
T Chapter 9 Development of Ectodermal Organs he ectoderm gives rise to 3 separate cell populations: neural(plate) ectoderm, neural crest cells, and epiderm (general body ectoderm). A primordium (anlage)
Changes in the distribution of tenascin during tooth development
 Development 101, 289-2% (1987) Printed in Great Britain The Company of Biologists Limited 1987 289 Changes in the distribution of tenascin during tooth development IRMA THESLEFF 1, ELEANOR MACKIE 2, SEPPO
Development 101, 289-2% (1987) Printed in Great Britain The Company of Biologists Limited 1987 289 Changes in the distribution of tenascin during tooth development IRMA THESLEFF 1, ELEANOR MACKIE 2, SEPPO
Biology 340 Comparative Embryology Lecture 11 Dr. Stuart Sumida. Overview of Embryology of the Vertebrate Skull. Emphasis on Amniota
 Biology 340 Comparative Embryology Lecture 11 Dr. Stuart Sumida Overview of Embryology of the Vertebrate Skull Emphasis on Amniota Initial introduction to components parts of a vertebrate head. This lecture
Biology 340 Comparative Embryology Lecture 11 Dr. Stuart Sumida Overview of Embryology of the Vertebrate Skull Emphasis on Amniota Initial introduction to components parts of a vertebrate head. This lecture
Week 14. Development of the Musculoskeletal System
 Week 14 Development of the Musculoskeletal System Skeletal System Derived from: paraxial mesoderm somites and somitomeres sclerotome sclerotome differentiation induced by SHH from notochord and floor plate
Week 14 Development of the Musculoskeletal System Skeletal System Derived from: paraxial mesoderm somites and somitomeres sclerotome sclerotome differentiation induced by SHH from notochord and floor plate
SUPPLEMENTARY FIG. S2. Teratoma. Portion of a teratoma composed of neural tissue. The large cells in the central part correspond to ganglion cells.
 Supplementary Data SUPPLEMENTARY FIG. S1. Teratoma. The tumor is composed predominantly of keratinizing squamous epithelium (Sq), which forms cysts filled with keratin (arrows). The tumor also contains
Supplementary Data SUPPLEMENTARY FIG. S1. Teratoma. The tumor is composed predominantly of keratinizing squamous epithelium (Sq), which forms cysts filled with keratin (arrows). The tumor also contains
strain), were tested by growing explants of neural fold from the posterior Woronzowa,2' 3 implanting pituitaries in postlarval stages, observed
 VOL. 35, 1949 GENETICS: H. C. DALTON 277 DE VELOPMENTAL ANAL YSIS OF GENETIC DIFFERENCES IN PIGMENTATION IN THE AXOLOTL By H. CLARK DALTON DEPARTMENT OF GENETICS, CARNEGIE INSTITUTION OF WASHINGTON, COLD
VOL. 35, 1949 GENETICS: H. C. DALTON 277 DE VELOPMENTAL ANAL YSIS OF GENETIC DIFFERENCES IN PIGMENTATION IN THE AXOLOTL By H. CLARK DALTON DEPARTMENT OF GENETICS, CARNEGIE INSTITUTION OF WASHINGTON, COLD
Maintenance of function in single epithelial cells spatially isolated from similar cells
 J. Embryol. exp. Morph. 90, 409-414 (1985) 409 Printed in Great Britain The Company of Biologists Limited 1985 Maintenance of function in single epithelial cells spatially isolated from similar cells S.
J. Embryol. exp. Morph. 90, 409-414 (1985) 409 Printed in Great Britain The Company of Biologists Limited 1985 Maintenance of function in single epithelial cells spatially isolated from similar cells S.
ANATOMY & PHYSIOLOGY - CLUTCH CH. 8 - BONE AND CARTILAGE.
 !! www.clutchprep.com CONCEPT: BONE CLASSIFICATIONS There are four classifications of bones based on their 1. Long bones are greater in length than in width - Found in the upper and lower limbs (ex: arm,
!! www.clutchprep.com CONCEPT: BONE CLASSIFICATIONS There are four classifications of bones based on their 1. Long bones are greater in length than in width - Found in the upper and lower limbs (ex: arm,
Welcome to ANAT 10A! What is Anatomy? Different levels of Anatomy The Language of Anatomy Pearson Education, Inc.
 Welcome to ANAT 10A! What is Anatomy? Different levels of Anatomy The Language of Anatomy Introduction Anatomy means to dissect: (ANAT 10A) The study of internal & external body structures The study of
Welcome to ANAT 10A! What is Anatomy? Different levels of Anatomy The Language of Anatomy Introduction Anatomy means to dissect: (ANAT 10A) The study of internal & external body structures The study of
Replacement of irradiated epidermis by migration of non-irradiated epidermis in the newt limb: the necessity of healthy epidermis for regeneration
 J. Embryol. exp. Morph. 76, 217-234 (1983) 217 Printed in Great Britain The Company of Biologists Limited 1983 Replacement of irradiated epidermis by migration of non-irradiated epidermis in the newt limb:
J. Embryol. exp. Morph. 76, 217-234 (1983) 217 Printed in Great Britain The Company of Biologists Limited 1983 Replacement of irradiated epidermis by migration of non-irradiated epidermis in the newt limb:
Age factor in the induced metamorphosis of thyroidectomized tadpoles
 /. Embryol exp. Morph. Vol. 25, 3, pp. 331-338, 1971 331 Printed in Great Britain Age factor in the induced metamorphosis of thyroidectomized tadpoles By C. Y. HSU 1, N. W. YU 1 AND H. M. LIANG 1 From
/. Embryol exp. Morph. Vol. 25, 3, pp. 331-338, 1971 331 Printed in Great Britain Age factor in the induced metamorphosis of thyroidectomized tadpoles By C. Y. HSU 1, N. W. YU 1 AND H. M. LIANG 1 From
Organogenesis Part 2. V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination. V. Lateral Plate Mesoderm
 Organogenesis Part 2 V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination V. Lateral Plate Mesoderm chordamesoderm paraxial mesoderm intermediate mesoderm
Organogenesis Part 2 V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination V. Lateral Plate Mesoderm chordamesoderm paraxial mesoderm intermediate mesoderm
