Quantified Co-Expression Analysis of Central Amygdala Sub-Populations
|
|
|
- Ambrose McBride
- 5 years ago
- Views:
Transcription
1 This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. Research Article: New Research Cognition and Behavior Quantified Co-Expression Analysis of Central Amygdala Sub-Populations Kenneth M. McCullough 1, Filomene G. Morrison 2,3,4, Jakob Hartmann 1, William A. Carlezon 1 and Kerry J. Ressler 1 1 Division of Depression & Anxiety Disorders, Department of Psychiatry, McLean Hospital, Harvard Medical School, Boston, MA 2 Behavioral Science Division, National Center for PTSD 3 VA Boston Healthcare System, Boston, MA 4 Department of Psychiatry, Boston University School of Medicine, Boston, MA DOI: /ENEURO Received: 5 January 2018 Accepted: 12 January 2018 Published: 24 January 2018 Author contributions: K.M. designed research; K.M. performed research; K.M. contributed unpublished reagents/analytic tools; K.M., F.G.M., J.H., W.A.C.J., and K.J.R. analyzed data; K.M., F.G.M., J.H., W.A.C.J., and K.J.R. wrote the paper. Funding: nih R01 MH MH Funding: nik # F31 MH Conflict of Interest: Authors declare no conflict of interests. Support was provided by NIH (R01 MH to KJR and MH to WAC) and Cohen Veteran Biosciences foundation. FGM s contribution to this work was supported by NIMH award # F31 MH105237, and current support is provided by NIMH award # 5T32MH Correspondence should be addressed to Kerry Ressler, McLean Hospital, Oaks Building 104b, Mailstop 212, 115 Mill Street, Belmont, MA , USA. TEL: , FAX: , kressler@mclean.harvard.edu Cite as: eneuro 2018; /ENEURO Alerts: Sign up at eneuro.org/alerts to receive customized alerts when the fully formatted version of this article is published. Accepted manuscripts are peer-reviewed but have not been through the copyediting, formatting, or proofreading process. Copyright 2018 McCullough et al. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International license, which permits unrestricted use, distribution and reproduction in any medium provided that the original work is properly attributed.
2 Quantified Co-Expression Analysis of Central Amygdala Sub-Populations Quantified Co-Expression Analysis of Central Amygdala McCullough KM 1 ; Morrison FG 2-4 ; Hartmann J 1 ; Carlezon WA Jr 1 ; Ressler KJ 1 1 Division of Depression & Anxiety Disorders, McLean Hospital, Department of Psychiatry, Harvard Medical School, Boston MA 2 National Center for PTSD, Behavioral Science Division 3 VA Boston Healthcare System 4 Boston University School of Medicine, Department of Psychiatry Corresponding Author: Kerry Ressler, MD, PhD McLean Hospital Oaks Building 104b, Mailstop Mill Street Belmont, MA TEL: FAX: kressler@mclean.harvard.edu Pages: 13 Figures: 7 Tables: 2 Supplementary: 0 Words Abstract:159 Words Introduction: 595 Words Discussion: 955 Acknowledgements: Support was provided by NIH (R01 MH to KJR and MH to WAC) and Cohen Veteran Biosciences foundation. FGM s contribution to this work was supported by NIMH award # F31 MH105237, and current support is provided by NIMH award # 5T32MH The authors declare no competing financial interests. 1
3 Abstract Molecular identification and characterization of fear controlling circuitries is a promising path towards developing targeted treatments of fear-related disorders. Three-color in situ hybridization analysis was used to determine whether somatostatin (Sst), neurotensin (Nts), corticotropin releasing factor (Crf), tachykinin 2 (Tac2), protein kinase c delta (Prkcd), and dopamine receptor 2 (Drd2) mrna co-localize in male mouse amygdala neurons. Expression and colocalization was examined across capsular (CeC), lateral (CeL), and medial (CeM) compartments of the central amygdala. The greatest expression of Prkcd and Drd2 were found in CeC and CeL. Crf was expressed primarily in CeL while Sst, Nts, and Tac2 expressing neurons were distributed between CeL and CeM. High levels of co-localization were identified between Sst, Nts, Crf, and Tac2 within the CeL while little co-localization was detected between any mrnas within the CeM. These findings provide a more detailed understanding of the molecular mechanisms that regulate the development and maintenance of fear and anxiety behaviors. Significance Statement Functional and behavioral analysis of central amygdala microcircuits has yielded significant insights into the role of this nucleus in fear and anxiety related behaviors. However, precise molecular and locational description of examined populations is lacking. This publication provides a quantified regionally precise description of the expression and co-expression of six frequently examined central amygdala population markers. Most revealing, within the most commonly examined region, the posterior CeL, four of these markers are extensively coexpressed suggesting the potential for experimental redundancy. This data clarifies circuit interaction and function and will increase relevance and precision of future cell-type specific reports. Introduction The amygdala comprises a wide array of molecularly, electrophysiologically and functionally distinct cell populations(cassell et al., 1999; Ciocchi et al., 2010; Yu et al., 2016). Recent evidence suggests that distinct sub-populations play differential roles in fear and extinction learning(herry et al., 2008; Haubensak et al., 2010; Li et al., 2013a; Kim et al., 2017). The characterization of molecularly identifiable neuronal populations is an important early step in developing improved treatments for fear and anxiety related disorders(mccullough et al., 2016). Previous work has shown that the central lateral amygdala (CeL) contains a mutually inhibitory circuit that gates fear expression via the inhibition of central medial amygdala (CeM) output neurons(herry et al., 2008; Ehrlich et al., 2009; Ciocchi et al., 2010; Letzkus et al., 2015). The CeL is often conflated with the 2
4 central capsular division of the amygdala (CeC) even though these regions have distinct projection patterns and potentially different roles in fear and anxiety; in the present manuscript we discuss these two regions separately(jolkkonen and Pitkanen, 1998; Bourgeais et al., 2001). The protein kinase c delta (PKC-δ, Prkcd) expressing neuron population has previously been shown to directly inhibit CeM output neurons, reducing activity in response to conditioned stimuli (CS) following fear conditioning and thus playing an important role in fear extinction learning(herry et al., 2008; Ciocchi et al., 2010; Haubensak et al., 2010; Cai et al., 2014). The somatostatin (SOM, Sst) expressing population appears to be a counterpart of the PKC-δ population; activity of SOM neurons increases in response to CS following fear conditioning and activity in this population is both necessary and sufficient for the production of fear and defensive behaviors(li et al., 2013b; Penzo et al., 2014; Yu et al., 2016). The tachykinin 2 (TAC2, Tac2) population plays a complementary role to the SOM population; activity of the Tac 2 expressing population is both necessary and sufficient for fear learning(andero et al., 2014; Andero et al., 2016). In addition to SOM and TAC2, other neuropeptides have been implicated as playing critical roles in fear circuitry. In particular corticotropin releasing factor (CRF, Crf) and neurotensin (NTS, Nts) expressing neurons are expressed in populations ideally situated and connected to participate in the central amygdala fear controlling circuit(petrovich and Swanson, 1997). Both NTS and CRF have been shown to play important roles in fear learning and expression(merali et al., 1998; Thompson et al., 2004; Yamauchi et al., 2007; Shilling and Feifel, 2008; Gafford and Ressler, 2015). Dopamine plays a critical role in fear and extinction learning. Specifically, the differential distributions of dopamine receptors may have important implications for mediating fear behaviors(de la Mora et al., 2010; Abraham et al., 2014; Kwon et al., 2015). The dopamine receptor 2 (DRD2, Drd2), has been suggested to label a large population of neurons implicated in the development and maintenance of fear behaviors(perez de la Mora et al., 2012; Kim et al., 2017). Considering the large numbers of CeA cell populations that play parallel or complementary roles in fear behaviors, it is important to determine the extent to which these populations overlap. While much work has been completed identifying markers for behaviorally relevant neuronal populations, less has been done to examine the extent to which each of these populations is distinguishable on the basis of gene expression patterns. In the present investigation three-color in situ hybridization was used to determine the extent of overlap in expression of Prkcd, Sst, Nts, Tac2, Crf, and Drd2. Importantly, significant differences are found in distribution and overlap across the anterior-posterior axis of the CeA, thus results are provided as both compressed across the CeA and split into 3
5 Anterior (A/P ) and Posterior (A/P -1.3 to -1.8) fractions(mcdonald, 2003). Results suggest that within the CeC, Prkcd and Drd2 label large nonoverlapping populations. Within the posterior CeL, Sst, Tac2, Nts and Crf populations largely overlap. Of these Sst labels the largest population that contains the others markers to varying extents. Within the CeL, the Prkcd and Drd2 populations largely do not overlap with each other or the other populations examined. The CeM has moderately sized Sst, Tac2, Nts and Crf populations, but is largely devoid of Prkcd and Drd2 labeled cells. Notably, unlike within the CeL, within the CeM, the Sst, Tac2, Nts, and Crf populations largely do not overlap suggesting important differences in the functional populations labeled by these markers in the CeL and CeM. Materials and Methods Animals C57BL/6J mice were obtained from Jackson Laboratories (Bar Harbor, ME). All ten male mice were adult (10 week) at the time of tissue collection. All mice were group housed and maintained on a 12hr:12hr light:dark cycle. Mice were housed in a temperature-controlled colony and given unrestricted access to food and water. All procedures conformed to National Institutes of Health guidelines and were approved by McLean Hospital Institutional Animal Care and use Committee. All animals were killed and tissue collected together during light cycle at zeitgeber 3:00. RNA Scope Staining Staining for mrna of interest was accomplished using RNA Scope Fluorescent Multiplex 2.5 labeling kit. Probes utilized for staining are: mm-nts-c1, mmnts-c2, mm-tac2-c1, mm-tac2-c2, mm-sst-c1, mm-sst-c2, mm-crh-c1, mm,prkcd- C1, mm-prkcd-c3, mmdrd2-c3. Brains were extracted and snap-frozen in methyl-butane on dry ice. Sections were taken at a width of 16μm. Staining procedure was completed to manufacturers specifications. Image acquisition Images were acquired with experimenter blinded to probes used. Sixteen-bit images of each section were acquired on a Leica SP8 confocal microscope using a 10x objective. Within a sample images were acquired with identical settings for laser power, detector gain, and amplifier offset. Images were acquired as a z- stack of 10 steps of.5 μm each. Max intensity projections were then created and analyzed. Data Analysis The expression and co-expression of mrna of different markers of interest was quantified in three areas central capsular amygdala (CeC), central lateral amygdala (CeL), and central medial amygdala (CeM). Images (approximate 4
6 area) of regions were taken bilaterally from a minimum of one section from each of four animals for each marker pair (n= >8 amygdala/ marker pair). Individual cells were identified based upon DAPI staining of the nucleus. Cells were determined to be expressing marker when more than five fluorescent dots or an area of staining sufficient to contain five dots was clearly associated with a single nucleus. The width of a cell was considered to be twice the diameter of the nucleus. The distribution of cells across CeA nuclei was determined by dividing the number of labeled cells in a nucleus by the total number of labeled cells across all nuclei. The percentage of cells in a nucleus expressing a certain mrna was determined by dividing the number of positive cells in a nucleus by the total number of DAPI labeled nuclei in the nucleus. Experimenters blinded to the identity of probes completed all counts. Statistical Analysis Determination of the percentage of cells within a sub-compartment expressing marker of interest was accomplished by dividing the total number of cells expressing the marker by the total number of DAPI positive nuclei in the area and multiplying by 100. Determination of the percentage of a labeled population found in a certain sub-compartment was accomplished by dividing the number of labeled cells in a compartment by the total number of labeled cells found in all compartments and multiplying by 100. Statistical analysis of the whether a labeled population was significantly different than the co-expressing component of that population was performed using the non-parametric Mann-Whitney test with Graphpad s Prism software package. Results Patterns of mrna expression observed with in situ staining for colocalization of three marker experiments were identical to those observed from single labeling of each marker. Staining patterns were consistent with those observed in the literature and with those produced by the Allen Brain Institute. All six probes produced strong staining in the central CeA. Each marker was examined individually to characterize its distribution across sub-compartments of the CeA. Determination of sub-compartment location was accomplished through examination of DAPI staining patterns. As the most popular available brain atlases (Paxinos and Allen Institute) differ somewhat on the locations of CeA sub-compartments across the anteriorposterior (A-P) axis of the amygdala the reference atlas provided through the Allen Brain Institute was used as a primary guide(lein et al., 2007). Distribution of labeled cells. The distribution of total cells expressing mrnas of interest was examined across CeA sub-compartments (Figure 1A-B) Prkcd staining was almost entirely contained within the CeC (49.8 +/- 3.6% of labeled cells) and CeL (44.4 +/- 3.8%) with only minority populations found within the CeM (5.7 +/- 2.8%). Drd2 was 5
7 strongly expressed within the CeC (43.3 +/- 3.3%) and CeL (32.4 +/- 3.3%) with a smaller population within the CeM (24.3 +/- 2.8%). Crf was primarily expressed within CeL (69.9 +/- 3.8) with smaller populations in CeM (20.7 +/- 5.1%) and CeC (9.4 +/- 4.1%). Sst, Tac2, and Nts populations are primarily found in CeL (51.3 +/- 3.7%, /- 6.0%, and /- 4.6% respectively) and CeM (42.0 +/- 5.9%, /- 5.9%, and /- 4.0%, respectively) with only small numbers of cells labeled in CeC (6.7 +/- 0.8%, 2.3 +/- 1.3% and 7.2 +/- 2.4%, respectively). Prevalence of labeled cells Single labeling by marker mrnas was examined to determine their prevalence within a sub-compartment (Figure 1C). This was completed by determining the proportion of labeled cells to the total number of DAPI positive cells within a compartment. Drd2 and Prkcd label large proportions of cells within the CeC (28.1 +/- 2.0% of total DAPI positive nuclei and /- 2.5%, respectively) while other markers Sst, Tac2, Nts and Crf label minority populations (5.2 +/- 0.6%, 1.0 +/- 0.5%, 2.4 +/- 0.4%, 3.2 +/- 0.3%, respectively). Prkcd, Drd2, Sst, Tac2, Nts and Crf each label significant populations within the CeL (25.1 +/- 2.9%, /- 2.3%, /- 1.8%, /- 1.9%, /- 1.4%, and /- 1.5%, respectively). Sst, Tac2, and Nts label moderate populations within the CeM (11.1 +/- 0.6%, /- 1.0%, and /- 0.9%, respectively) while Prkcd, Drd, and Crf label smaller proportions of cells (1.1 +/- 0.2%, 8.7 +/- 1.3% and 4.1 +/- 0.5%, respectively). Co-localization of CeA markers Co-localization among markers was examined within each CeA subcompartment. Triple-labeled images were analyzed only for co-localization between pairs of markers due to practical limitations on the number of probe combinations. Additionally, although co-localization was examined at a variety of anterior - posterior positions (A/P -.8 to -1.8), co-expression data is presented in Figures 1-7 is collapsed across A-P -0.8 to This may lead to an underestimation of the co-localization of some markers at certain positions (discussed below), but nonetheless provides an indication of overall colocalization between markers in the CeA. Tables 1 & 2 provide quantification of expression and co-localization at both anterior (A/P -0.8 to -1.2) and posterior (A/P -1.3 to -1.8) positions. Sst / Tac 2 / Prkcd Sst appears to mark the largest population of CeL cells (Figure 1C and Figure 2C). This population overlaps to a great extent with Tac2 (Figure 2D) within the CeL, but not the CeC or CeM (Figure 2F). Quantification of coexpression reveals that the total Sst labeled population is significantly larger than the co-expressing Sst/ Tac2 population in all sub-compartments; however, within the CeL the Tac2 population is not significantly different from the co-expressing 6
8 Sst/Tac2 population (Figure 2G). These data suggest that the larger Sst population may entirely contain the Tac2 population at this A-P range. Prkcd exhibits a typical dense CeC and CeL expression (Figure 2E). Within the CeL total Sst, Tac2 and Prkcd populations are larger than co-expressing populations, suggesting these RNAs mark separate populations (Figure 2H and 2I). Within CeC and CeM total populations are significantly larger than co-expressing populations except in cases where total population is very small. Crf / Nts / Prkcd Crf labels a large population of CeL cells with somewhat sparser labeling in the CeM (Figure 3C). Nts marks a large population within the CeL and a moderate population within the CeM (Figure 3D). Quantification of co-expression reveals the total Crf population is significantly larger than the co-expressing Crf/Nts population in the CeL and CeM; however, within the CeL the Nts population is not significantly different from the co-expressing Crf/Nts population (Figure 3G). This suggests that within the CeL the Nts population may be contained within the Crf population while within the CeM these populations are distinct. Prkcd demonstrates a similar expression pattern to that seen in Figure 2 (Figure 3E). The Prkcd population is separately expressed from the Crf and Nts populations in all areas where an appreciable number of marked cells are found (Figure 3H and 3I). Nts / Sst Examination of co-expression of Nts and Sst reveals similar patterns. Within the CeL the Nts population appears to be contained within the Sst population (Figure 4A- 4F). While within the CeM these mrnas mark distinct populations (Figure 4F). Crf / Tac 2 Likewise, when Crf and Tac2 are examined for co-expression, neither of the total labeled populations is significantly larger than the co-labeled Crf/Tac2 populations (Figure 5C-5F). However, within the CeM these mrnas mark separate populations (Figure 5C-5F). These results suggest a hierarchical organization within the CeL wherein Sst > Crf ~Tac2 >Nts. This is in contrast to the CeM where all total labeled populations are found to be significantly different than their co-labeling with any other marker. Examination of co-expression at a variety of A-P positions reveals that the zone of highest co-expression between Sst, Tac2, Nts and Crf is constrained to A-P -1.3 to Examination of more anterior positions (A-P-.8 to -1.2) demonstrates that these populations are co-expressed at lower rates and found in different sub-compartments in the anterior CeA. 7
9 Prkcd / Nts / Drd2 At anterior positions (A-P~-.9) Drd2 labels a large population of CeC cells while Prkcd cells are found in a cluster in the ventral aspect of the CeC (Figure 6C and 6E). At this A-P position Nts is found primarily within the CeM (Figure 6D). These populations largely do not overlap (Figure 6F-I). Crf / Tac 2 / Prkcd / Sst At a similar A-P position (~-.8) Crf densely labels the CeL (Figure 7C). Very little Tac2 staining is found within the CeL; however, labeled Tac2 cells are found in the CeM and the dorsal aspect of the main intercalated mass (Im) located ventrally to the BLA (Figure 7D). Prkcd is found in the ventral CeC (Figure 7E). These populations largely do not overlap (Figure 7F). Slightly more posteriorly (A-P~-1.2), the densely labeled CeL seen in more posterior sections begins to appear (Figure 7G-L). Sst densely labels the CeL and more sparsely the CeM (Figure 7I). Prkcd begins to form the typical CeC and CeL expression pattern (Figure 7K); however, Tac2 does not densely label the CeL at this position and markers continue to be co-expressed at low levels (Figure 7J-L). These results highlight that the zone of dense co-expression in CeL is constrained to more posterior aspects of the CeA. The overall percentages of co-expression for all mrna pairs examined at anterior and posterior positions is presented in Table 1A and 1B. Table 2 contains quantification of total numbers of labeled and co-labeled cells as well as labeled cells as a percent of total DAPI labeled cells. Descriptive summary of localization and co-localization of examined CeA mrna markers is found in Figure 7M. Discussion The central amygdala plays a pivotal role in the control of a wide range of behaviors, including those reflecting fear and anxiety(maren and Fanselow, 1996; Johansen et al., 2011; Pare and Duvarci, 2012; Fadok et al., 2017; Lezak et al., 2017). As such, the connectivity, cytoarchitecture and expression profiles of cells in the various subdivisions of this nucleus have been widely studied, especially in rats(mcdonald, 1982, 1984; Jolkkonen and Pitkanen, 1998; McDonald, 1998; Cassell et al., 1999). To date, a number of molecularly identified populations have been described as playing distinct roles in the control of behavior. Additionally, the distributions of these populations have been mapped using immunohistochemistry or in situ hybridization(cassell et al., 1986; Cassell et al., 1999; Andero et al., 2014; Andero et al., 2016). However, minimal data is available on the extent to which these population markers overlap, especially in mice, leading to ambiguity in the specificity of identified and manipulated populations. The present data provide a novel and important advance by examining the co-expression of Prkcd, Sst, Nts, Tac2, Crf, and Drd2 in the CeA of mice. Additionally, this data begins to address the critical need for 8
10 parsimonious description of both molecular and locational identity of neuron populations examined in the CeA. Our methods for identifying cells expressing an mrna involved a binary system so that all cells reaching minimum cut off (5 fluorescent puncta within twice the nuclear diameter) were considered to be expressing. Thus, the representation of low expressing transcripts may be under sampled in exchange for increased confidence. This is most relevant for Drd2, which has the greatest apparent range in expression strength. All animals were the same age and brains were obtained under the same conditions at the same time. This approach was used to characterize the baseline identity of cells; however, it ignores a wealth of data concerning levels of expression at the time of sacrifice and dynamic (e.g. circadian or following behavior) changes in expression level. One clear example is that both Tac2 and Sst are clearly expressed at different levels in different populations. Cells appear to express Tac2 and Sst at both high (bright) and moderate/low (dimmer) levels within the same sub-nucleus. Future studies examining static differences and dynamic changes in mrna expression level may yield important information regarding the functional roles of these mrnas. Our data confirm in mice previous immunohistochemical analyses conducted in rats by demonstrating that within the CeL there is a high degree of overlap between Sst, Nts, Tac2, and Crf. Remarkably this overlap is observed only within a constrained posterior section of the CeL between A-P -1.4 and Examination of these populations across the A-P axis suggests that Crf most consistently marks a CeL population while Sst, Nts, and Tac2 most consistently label cells in the anterior CeM before densely marking the CeL at posterior positions. Within the posterior CeL, these populations are highly overlapping. Sst expressing cells represent the largest population containing the majority of cells expressing Nts, Tac2, and Crf. This is in contrast to the CeM where these populations are consistently non-overlapping. An important consideration for the examination of these populations across the A-P axis is the inconsistency across currently available mouse brain atlases. For example, at anterior positions the Allen Brain atlas identifies the location of the dense Crf population as the CeL while the Paxinos & Franklin (2001) atlas identifies this region as the interstitial nucleus of the posterior limb of the anterior commissure (IPAC). While these may be semantic differences, the consistency of nucleus identification has important implication for the quantification of co-expression. Our decision to adhere more closely to the Allen Brain Atlas Reference Atlas may have lead to an underestimation of the extent of co-expression of examined markers within the CeL. Drd2 appears to consistently mark a large CeC and CeL population that is contiguous with the Ast. This is in contrast to Prkcd, which at anterior positions marks a very ventral population of CeC cells before moving more dorsally to mark a very constrained population of CeC and CeL cells at posterior positions. 9
11 Consistent with previously published work, neither the Prkcd nor the Drd2 populations is highly overlapping with any others examined. This finding validates the identification of these populations as potentially markers for functionally distinct sub populations(kim et al., 2017). Literature identifying functionally distinct CeA populations has been inconsistent in identifying precisely the CeA sub-compartment where neurons of interest reside. Such a specific delineation is especially critical in the case of Sst, Nts, Tac2, and Crf where the identification of these populations within the CeL may be redundant to previous work. Conversely, lack of co-expression in the CeM highlighted by the present findings may indicate a more specialized role for these cells. Future studies using intersectional approaches may yield clear and parsimonious descriptions of the distinct functional roles of single expressing and co-expressing populations in the CeL and CeM(Dymecki et al., 2010; Hirsch et al., 2013; Jensen and Dymecki, 2014; Okaty et al., 2015). Additionally, unlike in the CeL where examined populations label a majority of total cells, in the CeM, examined populations make up less than half of total cells indicating many additional populations that remain to be described. These results represent a starting point in a more comprehensive characterization of the many possible markers for CeA sub-populations. The receptors of the protein products of several of the mrnas examined may be promising markers for specific sub-populations (Crfr1, Crfr2, Tacr2, Sstr1-5). Additionally, further research into the co-expression of various neuropeptides and other identified markers such as PACAP (pituitary adenylate cyclase-activating polypeptide), vasoactive intestinal peptide, cholecystokinin, neuropeptide Y, dynorphin, enkephalin, and substance P (all of which have also been shown to also play important roles in fear and anxiety behaviors) will, in the future, be necessary to identify the extent to which additional populations co-localize within the CeA. These types of analyses provide a more thorough understanding of the molecular basis of amygdala function and may facilitate the development of innovative approaches, such as those target specific cell types by exploiting their unique patterns of receptor expression, to treat fear and anxiety-related disorders. Figure Legends Figure 1. Distribution of examined mrnas across CeA sub-compartments. A. Graphical representation of labeled cell distribution across CeA subcompartments. B. Tabular results of data represented in A. Each column presents distribution of all counted labeled cells in each CeA sub compartment +/- S.E.M. C. Labeled cells as percentage of total cells in sub-compartment. Labeled cell counts presented as a percentage of total DAPI positive nuclei examined within a nucleus +/- S.E.M. 10
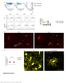
12 Figure 2. Co-expression of Sst, Tac2 and Prkcd (A-P -1.5). A. Map of area examined. B. DAPI stain (grey) of area examined. C. Sst expression (green) is found strongly in the CeL and CeM. D. Tac2 expression (red) is found strongly in the CeL and CeM. E. Prkcd (cyan) expression is found strongly in the CeC and CeL. F. Overlay of B-E reveals strong overlap in expression of Sst and Tac2 in CeL but not CeM. Prkcd does not highly co-express in any area. Scale Bar indicates 50 μm. G. Quantification of single expressing cells and co-expressing Sst and Tac2 cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. H. Quantification of single expressing cells and co-expressing Tac2 and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each subcompartment. I. Quantification of single expressing cells and co-expressing Sst and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. Data presented as mean + / - SEM where * = p<.05 difference between single and double labeled populations (Mann-Whitney U test.). Figure 3. Co-expression of Crf, Nts and Prkcd (A-P -1.5). A. Map of area examined. B. DAPI stain (grey) of area examined. C. Crf expression (green) is found strongly in the CeL and moderately in the CeM. D. Nts expression (red) is found strongly in the CeL and CeM. E. Prkcd expression (cyan) is found strongly in the CeC and CeL. F. Overlay of B-E reveals strong overlap in expression of Crf and Nts in CeL but not CeM. Prkcd does not highly co-express in any area. Scale Bar indicates 50 μm. G. Quantification of single expressing cells and coexpressing Crf and Nts cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. H. Quantification of single expressing cells and co-expressing Crf and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each subcompartment. I. Quantification of single expressing cells and co-expressing Nts and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. Data presented as mean + / - SEM where * = p<.05 difference between single and double labeled populations (Mann-Whitney U test.). Figure 4. Co-expression of Sst and Nts, (A-P -1.5). A. Map of area examined B-E. B. DAPI stain (grey) of area examined. C. Sst expression (green) is found strongly in the CeL and CeM. D. Nts expression (red) is found strongly in the CeL and CeM. E. Overlay of B-E reveals strong overlap in expression of Sst and Nts in CeL but not CeM. Scale Bar indicates 50 μm. F. Quantification of single expressing cells and co-expressing Sst and Nts cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. Data presented as mean + / - SEM where * = p<.05 difference between single and double labeled populations (Mann-Whitney U test.). 11
13 Figure 5. Co-expression of Crf and Tac2. (A-P -1.5) A. Map of area examined B-E. B. DAPI stain (grey) of area examined. C. Crf expression (green) is found strongly in the CeL and CeM. D. Tac2 expression (red) is found strongly in the CeL and CeM. E. Overlay of B-D reveals strong overlap in expression of Crf and Tac2 in CeL but not CeM. Scale Bar indicates 50 μm. F. Quantification of single expressing cells and co-expressing Crf and Tac2 cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. Data presented as mean + / - SEM where * = p<.05 difference between single and double labeled populations (Mann-Whitney U test.). Figure 6. Co-expression of Prkcd, Nts, and Drd2 (A-P -.9) Examination of markers at anterior positions within CeA reveals differential distributions across sub-compartments and reduced co-expression. A. Map of area examined (A-P ~ -.9). B. DAPI stain (grey) of area examined. C. Prkcd expression (green) is found strongly in ventral CeC. D. Nts expression (red) is found strongly in the CeM with limited expression in medial ventral CeC. E. Drd2 expression (cyan) is found strongly in the more dorsal elements of the CeC. F. Overlay of B-E reveals limited overlap in expression of any marker examined. Scale Bar indicates 50 μm. G. Quantification of single expressing cells and coexpressing Nts and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. H. Quantification of single expressing cells and co-expressing Drd2 and Prkcd cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each subcompartment. I. Quantification of single expressing cells and co-expressing Nts and Drd2 cells in CeC, CeL and CeM. Bars represent the mean number of (co)expressing cells in each sub-compartment. Data presented as mean + / - SEM where * = p<.05 difference between single and double labeled populations (Mann-Whitney U test.). Figure 7. Co-expression of Crf, Tac 2, and Prkcd, and Sst, Tac 2, and Prkcd (A-P -.8, and -1.22) A. Map of area examined (A-P ~ -.8). B. DAPI stain (grey) of area examined. C. Crf expression (green) is found strongly in CeL. D. Tac2 expression (red) is found in the CeM. E. Prkcd expression (cyan) is found strongly in ventral CeC. F. Overlay of B-E reveals limited overlap in expression of any marker examined. Scale Bar indicates 200 μm. G. Map of area examined (A-P ~ -1.2). H. DAPI stain (grey) of area examined. I. Sst expression (green) is found strongly in CeL and CeM. J. Tac2 expression (red) is found in the CeL and CeM. K. Prkcd expression (cyan) is found strongly in CeL and CeC with limited expression in the CeM. L. Overlay of H-K reveals limited overlap in expression of any marker examined. Scale Bar indicates 50 μm. M. Summary figure demonstrating localization and co-localization of examined CeA mrna markers. 12
14 Table 1. Co-expression of examined mrna s across CeA sub-compartment in anterior and posterior CeA. A. Co-expression of markers of interest in anterior CeA between A-P -.8 and Parent population labeled on vertical column (total cells labeled). Co-expressed population labeled on horizontal column (total co-labeled cells). B. Co-expression of markers of interest in aposterior CeA between A-P -1.3 and Parent population labeled on vertical column (total cells labeled). Co-expressed population labeled on horizontal column (total co-labeled cells). Table 2. Expression and co-expression of examined mrna s across CeA sub-compartment and anterior and posterior axis. Values presented of average number of positive cells found within a subcompartment in either anterior or posterior region. Additionally, these values are presented as a percentage of total DAPI stained nuclei in each area. Abraham AD, Neve KA, Lattal KM (2014) Dopamine and extinction: a convergence of theory with fear and reward circuitry. Neurobiology of learning and memory 108: Andero R, Dias BG, Ressler KJ (2014) A Role for Tac2, NkB, and Nk3 Receptor in Normal and Dysregulated Fear Memory Consolidation. Neuron. Andero R, Daniel S, Guo JD, Bruner RC, Seth S, Marvar PJ, Rainnie D, Ressler KJ (2016) Amygdala-Dependent Molecular Mechanisms of the Tac2 Pathway in Fear Learning. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 41: Bourgeais L, Gauriau C, Bernard JF (2001) Projections from the nociceptive area of the central nucleus of the amygdala to the forebrain: a PHA-L study in the rat. The European journal of neuroscience 14: Cai H, Haubensak W, Anthony TE, Anderson DJ (2014) Central amygdala PKCdelta(+) neurons mediate the influence of multiple anorexigenic signals. Nature neuroscience 17: Cassell MD, Gray TS, Kiss JZ (1986) Neuronal architecture in the rat central nucleus of the amygdala: a cytological, hodological, and immunocytochemical study. The Journal of comparative neurology 246: Cassell MD, Freedman LJ, Shi C (1999) The intrinsic organization of the central extended amygdala. Annals of the New York Academy of Sciences 877:
15 Ciocchi S, Herry C, Grenier F, Wolff SB, Letzkus JJ, Vlachos I, Ehrlich I, Sprengel R, Deisseroth K, Stadler MB, Muller C, Luthi A (2010) Encoding of conditioned fear in central amygdala inhibitory circuits. Nature 468: de la Mora MP, Gallegos-Cari A, Arizmendi-Garcia Y, Marcellino D, Fuxe K (2010) Role of dopamine receptor mechanisms in the amygdaloid modulation of fear and anxiety: Structural and functional analysis. Progress in neurobiology 90: Dymecki SM, Ray RS, Kim JC (2010) Mapping cell fate and function using recombinase-based intersectional strategies. Methods in enzymology 477: Ehrlich I, Humeau Y, Grenier F, Ciocchi S, Herry C, Luthi A (2009) Amygdala inhibitory circuits and the control of fear memory. Neuron 62: Fadok JP, Krabbe S, Markovic M, Courtin J, Xu C, Massi L, Botta P, Bylund K, Muller C, Kovacevic A, Tovote P, Luthi A (2017) A competitive inhibitory circuit for selection of active and passive fear responses. Nature 542: Gafford GM, Ressler KJ (2015) GABA and NMDA receptors in CRF neurons have opposing effects in fear acquisition and anxiety in central amygdala vs. bed nucleus of the stria terminalis. Hormones and behavior 76: Haubensak W, Kunwar PS, Cai H, Ciocchi S, Wall NR, Ponnusamy R, Biag J, Dong HW, Deisseroth K, Callaway EM, Fanselow MS, Luthi A, Anderson DJ (2010) Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature 468: Herry C, Ciocchi S, Senn V, Demmou L, Muller C, Luthi A (2008) Switching on and off fear by distinct neuronal circuits. Nature 454: Hirsch MR, d'autreaux F, Dymecki SM, Brunet JF, Goridis C (2013) A Phox2b::FLPo transgenic mouse line suitable for intersectional genetics. Genesis (New York, NY : 2000) 51: Jensen P, Dymecki SM (2014) Essentials of recombinase-based genetic fate mapping in mice. Methods in molecular biology (Clifton, NJ) 1092: Johansen JP, Cain CK, Ostroff LE, LeDoux JE (2011) Molecular mechanisms of fear learning and memory. Cell 147: Jolkkonen E, Pitkanen A (1998) Intrinsic connections of the rat amygdaloid complex: projections originating in the central nucleus. The Journal of comparative neurology 395: Kim J, Zhang X, Muralidhar S, LeBlanc SA, Tonegawa S (2017) Basolateral to Central Amygdala Neural Circuits for Appetitive Behaviors. Neuron 93: e1465. Kwon OB, Lee JH, Kim HJ, Lee S, Lee S, Jeong MJ, Kim SJ, Jo HJ, Ko B, Chang S, Park SK, Choi YB, Bailey CH, Kandel ER, Kim JH (2015) Dopamine Regulation of Amygdala Inhibitory Circuits for Expression of Learned Fear. Neuron 88: Lein ES et al. (2007) Genome-wide atlas of gene expression in the adult mouse brain. Nature 445: Letzkus JJ, Wolff SB, Luthi A (2015) Disinhibition, a Circuit Mechanism for Associative Learning and Memory. Neuron 88:
16 Lezak KR, Missig G, Carlezon WA, Jr. (2017) Behavioral methods to study anxiety in rodents. Dialogues in clinical neuroscience 19: Li H, Penzo MA, Taniguchi H, Kopec CD, Huang ZJ, Li B (2013a) Experiencedependent modification of a central amygdala fear circuit. Nat Neurosci 16: Li H, Penzo MA, Taniguchi H, Kopec CD, Huang ZJ, Li B (2013b) Experiencedependent modification of a central amygdala fear circuit. Nature neuroscience 16: Maren S, Fanselow MS (1996) The amygdala and fear conditioning: has the nut been cracked? Neuron 16: McCullough KM, Morrison FG, Ressler KJ (2016) Bridging the Gap: Towards a celltype specific understanding of neural circuits underlying fear behaviors. Neurobiology of learning and memory 135: McDonald AJ (1982) Cytoarchitecture of the central amygdaloid nucleus of the rat. The Journal of comparative neurology 208: McDonald AJ (1984) Neuronal organization of the lateral and basolateral amygdaloid nuclei in the rat. The Journal of comparative neurology 222: McDonald AJ (1998) Cortical pathways to the mammalian amygdala. Progress in neurobiology 55: McDonald AJ (2003) Is there an amygdala and how far does it extend? An anatomical perspective. Annals of the New York Academy of Sciences 985:1-21. Merali Z, McIntosh J, Kent P, Michaud D, Anisman H (1998) Aversive and appetitive events evoke the release of corticotropin-releasing hormone and bombesinlike peptides at the central nucleus of the amygdala. The Journal of neuroscience : the official journal of the Society for Neuroscience 18: Okaty BW, Freret ME, Rood BD, Brust RD, Hennessy ML, debairos D, Kim JC, Cook MN, Dymecki SM (2015) Multi-Scale Molecular Deconstruction of the Serotonin Neuron System. Neuron 88: Pare D, Duvarci S (2012) Amygdala microcircuits mediating fear expression and extinction. Curr Opin Neurobiol 22: Penzo MA, Robert V, Li B (2014) Fear conditioning potentiates synaptic transmission onto long-range projection neurons in the lateral subdivision of central amygdala. The Journal of neuroscience : the official journal of the Society for Neuroscience 34: Perez de la Mora M, Gallegos-Cari A, Crespo-Ramirez M, Marcellino D, Hansson AC, Fuxe K (2012) Distribution of dopamine D(2)-like receptors in the rat amygdala and their role in the modulation of unconditioned fear and anxiety. Neuroscience 201: Petrovich G, Swanson L (1997) Projections from the lateral part of the central amygdalar nucleus to the postulated fear conditioning circuit. Brain research 763:
17 Shilling PD, Feifel D (2008) The neurotensin-1 receptor agonist PD blocks fear-potentiated startle. Pharmacology, biochemistry, and behavior 90: Thompson BL, Erickson K, Schulkin J, Rosen JB (2004) Corticosterone facilitates retention of contextually conditioned fear and increases CRH mrna expression in the amygdala. Behavioural brain research 149: Yamauchi R, Wada E, Kamichi S, Yamada D, Maeno H, Delawary M, Nakazawa T, Yamamoto T, Wada K (2007) Neurotensin type 2 receptor is involved in fear memory in mice. Journal of neurochemistry 102: Yu K, Garcia da Silva P, Albeanu DF, Li B (2016) Central Amygdala Somatostatin Neurons Gate Passive and Active Defensive Behaviors. The Journal of neuroscience : the official journal of the Society for Neuroscience 36:
18
19
20
21
22
23
24
25 Table 1 A Anterior B Posterior Parent Population CeC Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd Parent Population CeC Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd Parent Population CeL Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd Parent Population CeL Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd Parent Population CeM Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd Parent Population CeM Sst (%) Tac2 (%) Crf (%) Nts (%) Prkcd (%) Drd2 (%) Sst Tac Crf Nts Prkcd Drd
26 Table 2 CeC CeL CeM Anterior Posterior Anterior Posterior Anterior Posterior Average # % Total Average # % Total Average # % Total Average # % Total Average # % Total Average # % Total Crf % % % % % % Nts % % % % % % Prkcd % % % % % % Tac % % % % % % Sst % % % % % % Drd % % % % % % Crf / Nts % % % % % % Crf / Prkcd % % % % % % Crf / Tac % % % % % % Crf / Sst % % % % % % Crf / Drd % % % % % % Nts / Prkcd % % % % % % Nts / Tac % % % % % % Nts / Sst % % % % % % Nts / Drd % % % % % % Prkcd / Tac % % % % % % Prkcd / Sst % % % % % % Prkcd / Drd % % % % % % Tac2 / Sst % % % % % % Tac 2 / Drd % % % % % % Sst / Drd % % % % % % 1
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature21682 Supplementary Table 1 Summary of statistical results from analyses. Mean quantity ± s.e.m. Test Days P-value Deg. Error deg. Total deg. χ 2 152 ± 14 active cells per day per mouse
doi:10.1038/nature21682 Supplementary Table 1 Summary of statistical results from analyses. Mean quantity ± s.e.m. Test Days P-value Deg. Error deg. Total deg. χ 2 152 ± 14 active cells per day per mouse
Basolateral to Central Amygdala Neural Circuits for Appetitive Behaviors
 Article Basolateral to Central Amygdala Neural Circuits for Appetitive Behaviors Highlights d Several genetically distinct CeA neurons mediate appetitive behaviors d d d BLA Ppp1r1b + neurons project to
Article Basolateral to Central Amygdala Neural Circuits for Appetitive Behaviors Highlights d Several genetically distinct CeA neurons mediate appetitive behaviors d d d BLA Ppp1r1b + neurons project to
ZNZ Advanced Course in Neuroscience Mon Limbic System II. David P. Wolfer MD
 ZNZ Advanced Course in Neuroscience Mon 05.05.2014 Limbic System II David P. Wolfer MD Institute of Anatomy, University of Zurich Institute for Human Movement Sciences and Sport, ETH Zurich http://www.dpwolfer.ch
ZNZ Advanced Course in Neuroscience Mon 05.05.2014 Limbic System II David P. Wolfer MD Institute of Anatomy, University of Zurich Institute for Human Movement Sciences and Sport, ETH Zurich http://www.dpwolfer.ch
Nature Neuroscience doi: /nn Supplementary Figure 1. Characterization of viral injections.
 Supplementary Figure 1 Characterization of viral injections. (a) Dorsal view of a mouse brain (dashed white outline) after receiving a large, unilateral thalamic injection (~100 nl); demonstrating that
Supplementary Figure 1 Characterization of viral injections. (a) Dorsal view of a mouse brain (dashed white outline) after receiving a large, unilateral thalamic injection (~100 nl); demonstrating that
Nature Neuroscience: doi: /nn Supplementary Figure 1. Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons
 Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Retrieving fear memories, as time goes by
 Molecular Psychiatry (2016) 21, 1027 1036 2016 Macmillan Publishers Limited All rights reserved 1359-4184/16 www.nature.com/mp REVIEW FH Do Monte 1, GJ Quirk 1,BLi 2 and MA Penzo 3 Research in fear conditioning
Molecular Psychiatry (2016) 21, 1027 1036 2016 Macmillan Publishers Limited All rights reserved 1359-4184/16 www.nature.com/mp REVIEW FH Do Monte 1, GJ Quirk 1,BLi 2 and MA Penzo 3 Research in fear conditioning
Hormonal gain control of a medial preoptic area social reward circuit
 CORRECTION NOTICE Nat. Neurosci. 20, 449 458 (2017) Hormonal gain control of a medial preoptic area social reward circuit Jenna A McHenry, James M Otis, Mark A Rossi, J Elliott Robinson, Oksana Kosyk,
CORRECTION NOTICE Nat. Neurosci. 20, 449 458 (2017) Hormonal gain control of a medial preoptic area social reward circuit Jenna A McHenry, James M Otis, Mark A Rossi, J Elliott Robinson, Oksana Kosyk,
Nature Neuroscience: doi: /nn Supplementary Figure 1. Splenic atrophy and leucopenia caused by T3 SCI.
 Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
SUPPLEMENTARY INFORMATION
 doi:1.138/nature9553 Supplementary Table 1. Overlap of neuronal marker and PKC- expression in CEl. Marker/PKC- PKC- Marker Gad65 87.4±4.7 5.3±12.6 CRH 1.2±1. 16.9±15.2 Dyn 1.9±1.2 4.5±2.9 Enk 42.8±7.4
doi:1.138/nature9553 Supplementary Table 1. Overlap of neuronal marker and PKC- expression in CEl. Marker/PKC- PKC- Marker Gad65 87.4±4.7 5.3±12.6 CRH 1.2±1. 16.9±15.2 Dyn 1.9±1.2 4.5±2.9 Enk 42.8±7.4
Research Paper. Christopher W. Butler, 1 Yvette M. Wilson, 1 Jenny M. Gunnersen, and Mark Murphy
 Research Paper Tracking the fear memory engram: discrete populations of neurons within amygdala, hypothalamus, and lateral septum are specifically activated by auditory fear conditioning Christopher W.
Research Paper Tracking the fear memory engram: discrete populations of neurons within amygdala, hypothalamus, and lateral septum are specifically activated by auditory fear conditioning Christopher W.
Prof. Anagnostaras, Lecture 7: Fear
 Historical views that thought and emotion were processed separately in the brain Prof. Anagnostaras, Lecture 7: So far, fear is the best understood What is fear? Dictionary: A feeling of agitation and
Historical views that thought and emotion were processed separately in the brain Prof. Anagnostaras, Lecture 7: So far, fear is the best understood What is fear? Dictionary: A feeling of agitation and
Limbic system outline
 Limbic system outline 1 Introduction 4 The amygdala and emotion -history - theories of emotion - definition - fear and fear conditioning 2 Review of anatomy 5 The hippocampus - amygdaloid complex - septal
Limbic system outline 1 Introduction 4 The amygdala and emotion -history - theories of emotion - definition - fear and fear conditioning 2 Review of anatomy 5 The hippocampus - amygdaloid complex - septal
Emotion I: General concepts, fear and anxiety
 C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
SUPPLEMENTARY INFORMATION
 Encoding of conditioned fear in central amygdala inhibitory circuits Stephane Ciocchi 1,*, Cyril Herry 1,,*, François Grenier 1, Steffen B.E. Wolff 1, Johannes J. Letzkus 1, Ioannis Vlachos 3, Ingrid Ehrlich
Encoding of conditioned fear in central amygdala inhibitory circuits Stephane Ciocchi 1,*, Cyril Herry 1,,*, François Grenier 1, Steffen B.E. Wolff 1, Johannes J. Letzkus 1, Ioannis Vlachos 3, Ingrid Ehrlich
NIH Public Access Author Manuscript Nat Neurosci. Author manuscript; available in PMC 2006 September 5.
 NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
Abstract. R. Roesler 1, J. Quevedo 1, C. Rodrigues 1, M. Madruga 1, M.R.M. Vianna 1 and M.B.C. Ferreira 2
 Brazilian Amygdaloid Journal non-nmda of Medical receptors and Biological and memory Research expression (1999) 32: 349-353 ISSN 0100-879X Short Communication 349 Increased training prevents the impairing
Brazilian Amygdaloid Journal non-nmda of Medical receptors and Biological and memory Research expression (1999) 32: 349-353 ISSN 0100-879X Short Communication 349 Increased training prevents the impairing
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Nature Neuroscience: doi: /nn.4642
 Supplementary Figure 1 Recording sites and example waveform clustering, as well as electrophysiological recordings of auditory CS and shock processing following overtraining. (a) Recording sites in LC
Supplementary Figure 1 Recording sites and example waveform clustering, as well as electrophysiological recordings of auditory CS and shock processing following overtraining. (a) Recording sites in LC
Microarray profiling of gene expression after sleep deprivation and recovery sleep 03/05/2008
 Microarray profiling of gene expression after sleep deprivation and recovery sleep 03/05/2008 1. OVERVIEW...1 2. BEHAVIORAL CONDITIONS...1 3. LASER MICRODISSECTION/MICROARRAY...2 3.1. REGIONS OF INTEREST...
Microarray profiling of gene expression after sleep deprivation and recovery sleep 03/05/2008 1. OVERVIEW...1 2. BEHAVIORAL CONDITIONS...1 3. LASER MICRODISSECTION/MICROARRAY...2 3.1. REGIONS OF INTEREST...
Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex
 Supplementary Information Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex Luc Gentet, Yves Kremer, Hiroki Taniguchi, Josh Huang, Jochen Staiger and Carl
Supplementary Information Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex Luc Gentet, Yves Kremer, Hiroki Taniguchi, Josh Huang, Jochen Staiger and Carl
Stress and Emotion. Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated
 Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
Neurons of the Bed Nucleus of the Stria Terminalis (BNST)
 Neurons of the Bed Nucleus of the Stria Terminalis (BNST) Electrophysiological Properties and Their Response to Serotonin DONALD G. RAINNIE a Harvard Medical School and Department of Psychiatry, Brockton
Neurons of the Bed Nucleus of the Stria Terminalis (BNST) Electrophysiological Properties and Their Response to Serotonin DONALD G. RAINNIE a Harvard Medical School and Department of Psychiatry, Brockton
To: Network of European Neuroscience Schools (NENS) Final report. Daniela Busti
 To: Network of European Neuroscience Schools (NENS) Final report Daniela Busti e-mail address: daniela.busti@i-med.ac.at Contact information of home institution: Prof. Francesco Ferraguti, Innsbruck Medical
To: Network of European Neuroscience Schools (NENS) Final report Daniela Busti e-mail address: daniela.busti@i-med.ac.at Contact information of home institution: Prof. Francesco Ferraguti, Innsbruck Medical
MECHANISMS INVOLVED IN STORAGE OF FEAR IN LATERAL AMYGDALA A COMPUTATIONAL STUDY
 MECHANISMS INVOLVED IN STORAGE OF FEAR IN LATERAL AMYGDALA A COMPUTATIONAL STUDY A Thesis presented to the Faculty of the Graduate School University of Missouri Columbia In Partial Fulfillment of the Requirement
MECHANISMS INVOLVED IN STORAGE OF FEAR IN LATERAL AMYGDALA A COMPUTATIONAL STUDY A Thesis presented to the Faculty of the Graduate School University of Missouri Columbia In Partial Fulfillment of the Requirement
Genetic Analysis of Anxiety Related Behaviors by Gene Chip and In situ Hybridization of the Hippocampus and Amygdala of C57BL/6J and AJ Mice Brains
 Genetic Analysis of Anxiety Related Behaviors by Gene Chip and In situ Hybridization of the Hippocampus and Amygdala of C57BL/6J and AJ Mice Brains INTRODUCTION To study the relationship between an animal's
Genetic Analysis of Anxiety Related Behaviors by Gene Chip and In situ Hybridization of the Hippocampus and Amygdala of C57BL/6J and AJ Mice Brains INTRODUCTION To study the relationship between an animal's
Introduction to Systems Neuroscience. Nov. 28, The limbic system. Daniel C. Kiper
 Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction. Laura Gunter
 Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction Laura Gunter The Brain as the Regulatory Center for Appetite The brain is the integration center
Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction Laura Gunter The Brain as the Regulatory Center for Appetite The brain is the integration center
The Neuroscience of Addiction: A mini-review
 The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
Zhu et al, page 1. Supplementary Figures
 Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
<student name> Undergraduate Research Grant Proposal
 Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1
 Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h)
 Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
Supplementary Information. Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent
 Supplementary Information Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent Authors: Susanne Kossatz a, Christian Brand a, Stanley Gutiontov b, Jonathan T.C. Liu c, Nancy
Supplementary Information Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent Authors: Susanne Kossatz a, Christian Brand a, Stanley Gutiontov b, Jonathan T.C. Liu c, Nancy
Emotional Memory, PTSD, and Clinical Intervention Updates
 Emotional Memory, PTSD, and Clinical Intervention Updates Wen Cai, MD, Ph.D. Chief Medical Officer--La Frontera Arizona Clinical Associate Professor--Psychiatry and Psychology University of Arizona College
Emotional Memory, PTSD, and Clinical Intervention Updates Wen Cai, MD, Ph.D. Chief Medical Officer--La Frontera Arizona Clinical Associate Professor--Psychiatry and Psychology University of Arizona College
Supplementary Figure 1. Nature Neuroscience: doi: /nn.4547
 Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Amygdala Mechanisms of Fear Suppression
 Amygdala Mechanisms of Fear Suppression By Alon Amir A Dissertation submitted to the Graduate School-Newark Rutgers, The State University of New Jersey in partial fulfillment of the requirements for the
Amygdala Mechanisms of Fear Suppression By Alon Amir A Dissertation submitted to the Graduate School-Newark Rutgers, The State University of New Jersey in partial fulfillment of the requirements for the
Nature Neuroscience: doi: /nn Supplementary Figure 1. Trial structure for go/no-go behavior
 Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Neurotransmitter Systems I Identification and Distribution. Reading: BCP Chapter 6
 Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Interactions between BNST PACAP stress system and Estrous Cycling in Non-overiectomized Female Rats
 University of Vermont ScholarWorks @ UVM UVM Honors College Senior Theses Undergraduate Theses 2017 Interactions between BNST PACAP stress system and Estrous Cycling in Non-overiectomized Female Rats Beniah
University of Vermont ScholarWorks @ UVM UVM Honors College Senior Theses Undergraduate Theses 2017 Interactions between BNST PACAP stress system and Estrous Cycling in Non-overiectomized Female Rats Beniah
Investigation of the role of nesfatin-1/nucb2 in the central nervous system. Ph.D. thesis Katalin Könczöl
 Investigation of the role of nesfatin-1/nucb2 in the central nervous system Ph.D. thesis Katalin Könczöl Semmelweis University János Szentágothai Doctoral School of Neurosciences Supervisor: Official reviewers:
Investigation of the role of nesfatin-1/nucb2 in the central nervous system Ph.D. thesis Katalin Könczöl Semmelweis University János Szentágothai Doctoral School of Neurosciences Supervisor: Official reviewers:
Anxiety and Related Disorders: Neurobiology and Treatment
 Anxiety and Related Disorders: Neurobiology and Treatment Kerry Ressler, MD,PhD James and Patricia Poitras Chair in Psychiatry Chief, Division of Depression & Anxiety Disorders McLean Hospital Professor
Anxiety and Related Disorders: Neurobiology and Treatment Kerry Ressler, MD,PhD James and Patricia Poitras Chair in Psychiatry Chief, Division of Depression & Anxiety Disorders McLean Hospital Professor
Brain Mechanisms of Emotion 1 of 6
 Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Inception, Total Recall, & The Brain: An Introduction to Neuroscience Part 2. Neal G. Simon, Ph.D. Professor Dept. of Biological Sciences
 Inception, Total Recall, & The Brain: An Introduction to Neuroscience Part 2 Neal G. Simon, Ph.D. Professor Dept. of Biological Sciences http://www.youtube.com/watch?v=wfmlgeh dije Summary from September
Inception, Total Recall, & The Brain: An Introduction to Neuroscience Part 2 Neal G. Simon, Ph.D. Professor Dept. of Biological Sciences http://www.youtube.com/watch?v=wfmlgeh dije Summary from September
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References
 File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) 108 (b-c) (d) (e) (f) (g)
 Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
Basal Ganglia Anatomy, Physiology, and Function. NS201c
 Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
SOM Husse et al. Supplementary online material. Synaptotagmin10-Cre, a driver to disrupt clock genes in the SCN
 SOM Husse et al. Supplementary online material Synaptotagmin10-Cre, a driver to disrupt clock genes in the SCN Jana Husse, Xunlei Zhou, Anton Shostak, Henrik Oster and Gregor Eichele SOM Husse et al.,
SOM Husse et al. Supplementary online material Synaptotagmin10-Cre, a driver to disrupt clock genes in the SCN Jana Husse, Xunlei Zhou, Anton Shostak, Henrik Oster and Gregor Eichele SOM Husse et al.,
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
CURRCULUM VITAE MAHSA MOADDAB
 CURRCULUM VITAE MAHSA MOADDAB EDUCATION Ph.D. M.Sc. B.Sc. 2010-2014 Department of Physiology, School of Medical Science, University of Otago, Dunedin, New Zealand. 2006-2009 Department of Biology, Faculty
CURRCULUM VITAE MAHSA MOADDAB EDUCATION Ph.D. M.Sc. B.Sc. 2010-2014 Department of Physiology, School of Medical Science, University of Otago, Dunedin, New Zealand. 2006-2009 Department of Biology, Faculty
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Quantification of myelin fragments in the aging brain (a) Electron microscopy on corpus callosum is shown for a 18-month-old wild type mice. Myelin fragments (arrows) were detected
Supplementary Figure 1 Quantification of myelin fragments in the aging brain (a) Electron microscopy on corpus callosum is shown for a 18-month-old wild type mice. Myelin fragments (arrows) were detected
Neurobiology of Addiction
 Neurobiology of Addiction Domenic A. Ciraulo, MD Director of Alcohol Pharmacotherapy Research Center for Addiction Medicine Department of Psychiatry Massachusetts General Hospital Disclosure Neither I
Neurobiology of Addiction Domenic A. Ciraulo, MD Director of Alcohol Pharmacotherapy Research Center for Addiction Medicine Department of Psychiatry Massachusetts General Hospital Disclosure Neither I
ISIS NeuroSTIC. Un modèle computationnel de l amygdale pour l apprentissage pavlovien.
 ISIS NeuroSTIC Un modèle computationnel de l amygdale pour l apprentissage pavlovien Frederic.Alexandre@inria.fr An important (but rarely addressed) question: How can animals and humans adapt (survive)
ISIS NeuroSTIC Un modèle computationnel de l amygdale pour l apprentissage pavlovien Frederic.Alexandre@inria.fr An important (but rarely addressed) question: How can animals and humans adapt (survive)
Neurotransmitters and Neuromodulators
 Oliver von Bohlen und Halbach and Rolf Dermietzel Neurotransmitters and Neuromodulators Handbook of Receptors and Biological Effects 2nd completely revised and enlarged edition WILEY- VCH WILEY-VCH Verlag
Oliver von Bohlen und Halbach and Rolf Dermietzel Neurotransmitters and Neuromodulators Handbook of Receptors and Biological Effects 2nd completely revised and enlarged edition WILEY- VCH WILEY-VCH Verlag
Although humans possess a number of cognitive abilities that
 REVIEW doi:10.1038/nature14188 From circuits to behaviour in the amygdala Patricia H. Janak 1,2 & Kay M. Tye 3 The amygdala has long been associated with emotion and motivation, playing an essential part
REVIEW doi:10.1038/nature14188 From circuits to behaviour in the amygdala Patricia H. Janak 1,2 & Kay M. Tye 3 The amygdala has long been associated with emotion and motivation, playing an essential part
NIH Public Access Author Manuscript Nat Neurosci. Author manuscript; available in PMC 2012 November 25.
 NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. ; 14(10): 1247 1249. doi:10.1038/nn.2899. A category-specific response to animals in the right human amygdala Florian
NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. ; 14(10): 1247 1249. doi:10.1038/nn.2899. A category-specific response to animals in the right human amygdala Florian
Cellular and molecular mechanisms of memory storage Synaptic plasticity and Alzheimer s disease Reward learning and addiction
 Brian Wiltgen, Ph.D. Curriculum Vitae University of Virginia Department of Psychology Charlottesville, VA 22904 Telephone: (434) 924-1458 E-mail: bw4fh@virginia.edu RESEARCH INTERESTS Cellular and molecular
Brian Wiltgen, Ph.D. Curriculum Vitae University of Virginia Department of Psychology Charlottesville, VA 22904 Telephone: (434) 924-1458 E-mail: bw4fh@virginia.edu RESEARCH INTERESTS Cellular and molecular
Morphology, PKC expression, and synaptic responsiveness of different types of rat central lateral amygdala neurons
 J Neurophysiol 18: 3196 325, 212. First published September 12, 212; doi:1.1152/jn.514.212. Morphology, PKC expression, and synaptic responsiveness of different types of rat central lateral amygdala neurons
J Neurophysiol 18: 3196 325, 212. First published September 12, 212; doi:1.1152/jn.514.212. Morphology, PKC expression, and synaptic responsiveness of different types of rat central lateral amygdala neurons
Final published version:
 GABA and NMDA receptors in CRF neurons have opposing effects in fear acquisition and anxiety in central amygdala vs. bed nucleus of the stria terminalis Georgette Gafford, Emory University Kerry Ressler,
GABA and NMDA receptors in CRF neurons have opposing effects in fear acquisition and anxiety in central amygdala vs. bed nucleus of the stria terminalis Georgette Gafford, Emory University Kerry Ressler,
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
What is the Role of the Amygdala in Long Term Memory? Jack Pemment. University of Mississippi
 LT Memory and the Amygdala 1 Running Head: Role of the amygdala in long term memory What is the Role of the Amygdala in Long Term Memory? Jack Pemment University of Mississippi LT Memory and the Amygdala
LT Memory and the Amygdala 1 Running Head: Role of the amygdala in long term memory What is the Role of the Amygdala in Long Term Memory? Jack Pemment University of Mississippi LT Memory and the Amygdala
processes in the central nervous system (CNS), affecting many of the during the course of ethanol treatment. Ethanol stimulates the release of
 INTRODUCTION INTRODUCTION Neuroscience research is essential for understanding the biological basis of ethanol-related brain alterations and for identifying the molecular targets for therapeutic compounds
INTRODUCTION INTRODUCTION Neuroscience research is essential for understanding the biological basis of ethanol-related brain alterations and for identifying the molecular targets for therapeutic compounds
Memory retention the synaptic stability versus plasticity dilemma
 Memory retention the synaptic stability versus plasticity dilemma Paper: Abraham, Wickliffe C., and Anthony Robins. "Memory retention the synaptic stability versus plasticity dilemma." Trends in neurosciences
Memory retention the synaptic stability versus plasticity dilemma Paper: Abraham, Wickliffe C., and Anthony Robins. "Memory retention the synaptic stability versus plasticity dilemma." Trends in neurosciences
Systems Neuroscience November 29, Memory
 Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Supplemental data Supplemental Figure Legends Supplemental Figure 1. Supplemental Figure 2.
 Supplemental data Supplemental Figure Legends Supplemental Figure 1. Analysis of deletion of AMPK!2 in POMC and AgRP neurons in control and POMC!2KO and AgRP!2KO mice. (A) mmunofluorescence analysis for
Supplemental data Supplemental Figure Legends Supplemental Figure 1. Analysis of deletion of AMPK!2 in POMC and AgRP neurons in control and POMC!2KO and AgRP!2KO mice. (A) mmunofluorescence analysis for
F0 F1 F2. Ancestral influences on descendant generations: a case study using the olfactory system in rodents. Brian Dias, PhD.
 Ancestral influences on descendant generations: a case study using the olfactory system in rodents Brian Dias, PhD. F0 F1 F2 Assistant Professor, Emory University School of Medicine Department of Psychiatry
Ancestral influences on descendant generations: a case study using the olfactory system in rodents Brian Dias, PhD. F0 F1 F2 Assistant Professor, Emory University School of Medicine Department of Psychiatry
Nature Neuroscience: doi: /nn Supplementary Figure 1. Visualization of AT1a-positive cells using AT1a lacz/+ mouse.
 Supplementary Figure 1 Visualization of AT1a-positive cells using AT1a lacz/+ mouse. (a f) Immunohistochemical detection of β-gal in the mouse brain. Coronal sections at the respective anteroposterior
Supplementary Figure 1 Visualization of AT1a-positive cells using AT1a lacz/+ mouse. (a f) Immunohistochemical detection of β-gal in the mouse brain. Coronal sections at the respective anteroposterior
Complexity Made Simple
 Complexity Made Simple Bussey-Saksida Touch Screen Chambers for Rats and Mice Tasks translate directly to well established monkey and human touch models e.g. CANTAB High throughput screening of multiple
Complexity Made Simple Bussey-Saksida Touch Screen Chambers for Rats and Mice Tasks translate directly to well established monkey and human touch models e.g. CANTAB High throughput screening of multiple
Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism
 Research article Related Commentary, page 2697 Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism Subhash C. Pandey,
Research article Related Commentary, page 2697 Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism Subhash C. Pandey,
Molecular Biology of Memory Storage: The Persistence of Memory
 Molecular Biology of Memory Storage: The Persistence of Memory The Study of Memory Has Two Parts: (1) The Systems Problem of Memory: Where in the brain is memory stored? (2) The Molecular Problem of Memory:
Molecular Biology of Memory Storage: The Persistence of Memory The Study of Memory Has Two Parts: (1) The Systems Problem of Memory: Where in the brain is memory stored? (2) The Molecular Problem of Memory:
Fig. 4. The activity of Pkc -transduced neurons is required for enhanced learning. After gene transfer, rats were tested on [] vs. +.
![Fig. 4. The activity of Pkc -transduced neurons is required for enhanced learning. After gene transfer, rats were tested on [] vs. +. Fig. 4. The activity of Pkc -transduced neurons is required for enhanced learning. After gene transfer, rats were tested on [] vs. +.](/thumbs/90/102196023.jpg) Research Interests Advanced cognitive learning is encoded in distributed circuits that span multiple forebrain areas. Further, synaptic plasticity and neural network theories hypothesize that essential
Research Interests Advanced cognitive learning is encoded in distributed circuits that span multiple forebrain areas. Further, synaptic plasticity and neural network theories hypothesize that essential
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
A New Era of Migraine Management: The Challenging Landscape in Prevention
 Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Resistance to forgetting associated with hippocampus-mediated. reactivation during new learning
 Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Degree of freedom problem
 KINE 4500 Neural Control of Movement Lecture #1:Introduction to the Neural Control of Movement Neural control of movement Kinesiology: study of movement Here we re looking at the control system, and what
KINE 4500 Neural Control of Movement Lecture #1:Introduction to the Neural Control of Movement Neural control of movement Kinesiology: study of movement Here we re looking at the control system, and what
Michael A. McDannald January 9, 2018
 McDannald 1 Michael A. McDannald January 9, 2018 Department of Psychology Boston College McGuinn Hall 514 Chestnut Hill, MA 02467 michael.mcdannald@bc.edu mcdannaldlab.org Active NIH grants: 2 Publications:
McDannald 1 Michael A. McDannald January 9, 2018 Department of Psychology Boston College McGuinn Hall 514 Chestnut Hill, MA 02467 michael.mcdannald@bc.edu mcdannaldlab.org Active NIH grants: 2 Publications:
Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC
 1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
BIOMED 509. Executive Control UNM SOM. Primate Research Inst. Kyoto,Japan. Cambridge University. JL Brigman
 BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/1/10/e1500775/dc1 Supplementary Materials for Structural-functional connectivity deficits of neocortical circuits in the Fmr1 /y mouse model of autism Matthias
advances.sciencemag.org/cgi/content/full/1/10/e1500775/dc1 Supplementary Materials for Structural-functional connectivity deficits of neocortical circuits in the Fmr1 /y mouse model of autism Matthias
KINE 4500 Neural Control of Movement. Lecture #1:Introduction to the Neural Control of Movement. Neural control of movement
 KINE 4500 Neural Control of Movement Lecture #1:Introduction to the Neural Control of Movement Neural control of movement Kinesiology: study of movement Here we re looking at the control system, and what
KINE 4500 Neural Control of Movement Lecture #1:Introduction to the Neural Control of Movement Neural control of movement Kinesiology: study of movement Here we re looking at the control system, and what
Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral Amygdala Lesions in Rats?
 Neurobiology of Learning and Memory 76, 268 283 (2001) doi:10.1006/nlme.2001.4042, available online at http://www.idealibrary.com on Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral
Neurobiology of Learning and Memory 76, 268 283 (2001) doi:10.1006/nlme.2001.4042, available online at http://www.idealibrary.com on Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral
THE ORGANIZATION OF AMYGDALOPETAL PROJECTIONS FROM THE LATERAL HYPOTHALAMUS AND PREOPTIC AREA IN THE RAT
 ACTA NEUROBIOL. EXP. 1977, 37: 247-252 THE ORGANIZATION OF AMYGDALOPETAL PROJECTIONS FROM THE LATERAL HYPOTHALAMUS AND PREOPTIC AREA IN THE RAT Liliana NITECKA, Olgierd NARKIEWICZ and Czeslaw JAKIEL Department
ACTA NEUROBIOL. EXP. 1977, 37: 247-252 THE ORGANIZATION OF AMYGDALOPETAL PROJECTIONS FROM THE LATERAL HYPOTHALAMUS AND PREOPTIC AREA IN THE RAT Liliana NITECKA, Olgierd NARKIEWICZ and Czeslaw JAKIEL Department
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
The Adolescent Developmental Stage
 The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
Rescue of Impaired Fear Extinction and Normalization of Cortico-Amygdala Circuit Dysfunction in a Genetic Mouse Model by Dietary Zinc Restriction
 13586 The Journal of Neuroscience, October 13, 2010 30(41):13586 13596 Behavioral/Systems/Cognitive Rescue of Impaired Fear Extinction and Normalization of Cortico-Amygdala Circuit Dysfunction in a Genetic
13586 The Journal of Neuroscience, October 13, 2010 30(41):13586 13596 Behavioral/Systems/Cognitive Rescue of Impaired Fear Extinction and Normalization of Cortico-Amygdala Circuit Dysfunction in a Genetic
Food restriction: enhancing effects on drug reward and striatal cell signaling
 Food restriction: enhancing effects on drug reward and striatal cell signaling K.D. Carr Departments of Psychiatry & Pharmacology NYU School of Medicine Common Neural Substrates for Incentive-Motivating
Food restriction: enhancing effects on drug reward and striatal cell signaling K.D. Carr Departments of Psychiatry & Pharmacology NYU School of Medicine Common Neural Substrates for Incentive-Motivating
Insular Cortex-Amygdala Dialogue During Taste Recognition Memory Formation
 The highlight for April is by Dr. Federico Bermudez-Rattoni from the Institute of Cellular Physiology, National University of Mexico in Mexico City, Mexico. Dr. Bermudez-Rattoni s work over the past 25
The highlight for April is by Dr. Federico Bermudez-Rattoni from the Institute of Cellular Physiology, National University of Mexico in Mexico City, Mexico. Dr. Bermudez-Rattoni s work over the past 25
Ph.D. in Neuroscience, Mississippi Medical Center, Jackson, MS USA.
 Loai A. Alzghoul University of Jordan Department of Physiology and Biochemistry P.O.BOX 13408, University of Jordan Amman 11942, Jordan. EMAIL: l.zghoul@ju.edu.jo Education: Dec 2012 Dec 2009 Ph.D. in
Loai A. Alzghoul University of Jordan Department of Physiology and Biochemistry P.O.BOX 13408, University of Jordan Amman 11942, Jordan. EMAIL: l.zghoul@ju.edu.jo Education: Dec 2012 Dec 2009 Ph.D. in
The storage and recall of memories in the hippocampo-cortical system. Supplementary material. Edmund T Rolls
 The storage and recall of memories in the hippocampo-cortical system Supplementary material Edmund T Rolls Oxford Centre for Computational Neuroscience, Oxford, England and University of Warwick, Department
The storage and recall of memories in the hippocampo-cortical system Supplementary material Edmund T Rolls Oxford Centre for Computational Neuroscience, Oxford, England and University of Warwick, Department
Rebecca M Shansky, PhD Northeastern University 360 Huntington Ave 125 Nightingale Hall Boston, MA
 curriculum vitae Rebecca M Shansky, PhD Northeastern University 360 Huntington Ave 125 Nightingale Hall Boston, MA 02215 r.shansky@neu.edu (617) 373 6819 CAREER HISTORY Sept 2011 present Assistant Professor
curriculum vitae Rebecca M Shansky, PhD Northeastern University 360 Huntington Ave 125 Nightingale Hall Boston, MA 02215 r.shansky@neu.edu (617) 373 6819 CAREER HISTORY Sept 2011 present Assistant Professor
Murine GRPR and Stathmin Control in Opposite Directions both Cued Fear Extinction and Neural Activities of the Amygdala and Prefrontal Cortex
 Murine GRPR and Stathmin Control in Opposite Directions both Cued Fear Extinction and Neural Activities of the Amygdala and Prefrontal Cortex Guillaume Martel, Charles Hevi, Alexandra Wong, Ko Zushida,
Murine GRPR and Stathmin Control in Opposite Directions both Cued Fear Extinction and Neural Activities of the Amygdala and Prefrontal Cortex Guillaume Martel, Charles Hevi, Alexandra Wong, Ko Zushida,
Structural basis for the role of inhibition in facilitating adult brain plasticity
 Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
Anatomy of the basal ganglia. Dana Cohen Gonda Brain Research Center, room 410
 Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila
 Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila Angelique Lamaze 1, Arzu Öztürk-Çolak 2, Robin Fischer 3, Nicolai Peschel 3, Kyunghee Koh 2 and James E.C. Jepson 1* 1 UCL Institute
Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila Angelique Lamaze 1, Arzu Öztürk-Çolak 2, Robin Fischer 3, Nicolai Peschel 3, Kyunghee Koh 2 and James E.C. Jepson 1* 1 UCL Institute
