Fbw7 controls neural stem cell differentiation and progenitor apoptosis via Notch and c-jun
|
|
|
- Shanon McCarthy
- 5 years ago
- Views:
Transcription
1 Fbw7 controls neural stem cell differentiation and progenitor apoptosis via Notch and c-jun Joerg D Hoeck, Anett Jandke, Sophia Maria Blake, Emma Nye, Bradley Spencer-Dene, Sebastian Brandner, Axel Behrens To cite this version: Joerg D Hoeck, Anett Jandke, Sophia Maria Blake, Emma Nye, Bradley Spencer-Dene, et al.. Fbw7 controls neural stem cell differentiation and progenitor apoptosis via Notch and c-jun. Nature Neuroscience, Nature Publishing Group, 2010, < /nn.2644>. <hal > HAL Id: hal Submitted on 10 Apr 2011 HAL is a multi-disciplinary open access archive for the deposit and dissemination of scientific research documents, whether they are published or not. The documents may come from teaching and research institutions in France or abroad, or from public or private research centers. L archive ouverte pluridisciplinaire HAL, est destinée au dépôt et à la diffusion de documents scientifiques de niveau recherche, publiés ou non, émanant des établissements d enseignement et de recherche français ou étrangers, des laboratoires publics ou privés.
2 Fbw7 controls neural stem cell differentiation and progenitor apoptosis via Notch and c- Jun Joerg D Hoeck 1, Anett Jandke 1, Sophia M Blake 1, Emma Nye 2, Bradley Spencer-Dene 2, Sebastian Brandner 3 and Axel Behrens 1, 4 1 Mammalian Genetics Laboratory 2 Experimental Pathology Laboratory Cancer Research UK London Research Institute Lincoln's Inn Fields Laboratories 44 Lincoln's Inn Fields London WC2A 3LY United Kingdom 3 Division of Neuropathology, Department of Neurodegenerative Disease, Institute of Neurology, London, UK 4 Address correspondence to Axel Behrens Tel: Fax: axel.behrens@cancer.org.uk Key words: Fbw7, c-jun, Notch, ubiquitylation, E3 ligase, neural stem cell
3 ABSTRACT Neural stem/progenitor cells (NSCs/NPCs) give rise to neurons, astrocytes, and oligodendrocytes. However, the mechanisms underlying the decision of a stem cell to either self-renew or differentiate are incompletely understood. We demonstrate here that Fbw7 (F-box and WD repeat domain containing 7), the substrate recognition component of an SCF (complex of SKP1, CUL1 and F-box protein)-type E3 ubiquitin ligase, is a key regulator of murine NSC/NPC viability and differentiation. The absence of Fbw7 caused severely impaired stem cell differentiation and increased progenitor cell death. Fbw7 deficiency resulted in accumulation of the SCF(Fbw7) substrates active Notch1 and N-terminally phosphorylated c-jun. Genetic and pharmacological rescue experiments identified c-jun as a key substrate of Fbw7 in controlling progenitor cell viability, whereas inhibition of Notch signalling alleviated the block in stem cell differentiation. Thus Fbw7 controls neurogenesis by antagonising Notch and JNK/c-Jun signalling. 2
4 INTRODUCTION Neural stem cells (NSCs) produce the differentiated cell types of the nervous system, namely neurons and glia. The first NSCs to appear during embryogenesis are neuroepithelial cells. Subsequently, other stem cell populations arise, i.e. radial glia cells (RGCs) at later stages of embryonic development and stem cells with astroglial characteristics in the adult 1,2. NSCs have been suggested as a potential therapeutic tool for neurodegenerative diseases and neural injuries. However, a pre-requisite to manipulate stem cell behaviour is to better understand the mechanisms that govern NSC homeostasis and differentiation. Fbw7 (also known as Fbxw7, hcdc4, hsel-10) is a member of the F-box family of proteins, which function as substrate recognition adaptors for SCF (complex of SKP1, CUL1 and F- box protein)-type ubiquitin ligases 3. SCF(Fbw7) targets several important oncoproteins, including c-myc, c-jun, CyclinE1 and Notch, for ubiquitin-dependent proteolysis and Fbw7 mutations have been detected in a variety of human cancers, indicating that Fbw7 is a tumour suppressor 4 6. Several Fbw7 substrates have been implicated in the control of neural differentiation and viability. The AP-1 transcription factor c-jun is an important regulator of neuronal viability, especially when phosphorylated by the JNK group of MAP kinases 7 9. The Notch signalling pathway controls neural differentiation and is known to maintain NSC character and to inhibit neurogenesis. The Notch-Hes pathway is necessary for self-renewing cell division and thus, maintenance of the neural precursor population However, the significance of the regulation of c-jun, Notch and other substrates by SCF(Fbw7) during neurogenesis has remained unclear to date. In this study we identify the E3 ubiquitin ligase Fbw7 as a key regulator of neural stem cell differentiation and progenitor viability. We demonstrate that Fbw7 acts as a molecular switch that antagonises Notch activity and JNK/c-Jun signalling to enable neural stem cell differentiation and progenitor survival. 3
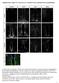
5 RESULTS Conditional inactivation of Fbw7 in the nervous system To investigate the significance of Fbw7 expression and function during neural development, we generated conditional knock-out mice in which exon 5 of fbw7 was flanked by loxp sites (Supplementary Fig. 1a), since mice lacking Fbw7 die during early embryonic development due to placental and vascular defects 14,15. Deletion of exon 5 removes most of the F-box, an essential domain of Fbw7, and in addition disrupts the fbw7 open reading frame preventing production of detectable Fbw7 protein (Jandke et al. in preparation, and below). Floxed fbw7 (fbw7 f/f ) mice were crossed to Nestin-Cre transgenic mice previously shown to provide efficient NSC-specific Cre activity 9, with no recombination detectable in neural crestderived cells 16. In situ hybridisation revealed loss of fbw7 expression in fbw7 f/f ; Nestin-Cre + mutant mice (fbw7 ΔN mice) confirming efficient Cre-mediated deletion (Supplementary Fig. 1b). fbw7 ΔN mice were not obtained at weaning age and died perinatally (data not shown). Fbw7 controls cell number and differentiation in the brain While the overall structure of fbw7 ΔN mutant brains was normal, histological analysis of E18.5 mutant embryos revealed severe defects in neurogenesis. The size and thickness of brain regions such as the cortex was unaffected by the absence of Fbw7 (Supplementary Fig. 2), whereas the cellularity in several regions of the developing fbw7 ΔN brain was significantly reduced. In the E18.5 midbrain tectum and forebrain cortex, cell numbers were decreased by around 30% in areas where committed progenitors and differentiated cells reside, namely the tectal mantle layer (ML) as well as the cortical subventricular zone (SVZ), intermediate zone (IZ) and cortical plate (CP) (Fig. 1a d). Similarly, the cellularity in fbw7 ΔN embryos was decreased in other brain regions such as the cerebellar anlage and the thalamus (Supplementary Fig. 3). In contrast, the cellularity in compartments harbouring immature cells was either unaffected, as in the cortical ventricular zone (VZ), or increased, as seen in 4
6 the tectal VZ (Fig. 1a d). Whilst we could not detect a statistically significant difference in the number of proliferating cells (Fig. 1e,g), the number of apoptotic cells marked by active Caspase 3 was significantly increased in the tectal ML at E16.5 (Fig. 1f,g). To better understand the role of Fbw7 during brain development, we performed in situ hybridisation for fbw7 and immunohistochemistry (IHC) for markers characterising neural stem cells, progenitors and differentiated cells. fbw7 mrna was highly expressed at E14.5 in the cortical VZ and SVZ, indicating a role for Fbw7 in stem cells and progenitors (Fig. 2a). Expression of the neural stem cell and progenitor marker Nestin was already slightly increased in the E14.5 cortex of fbw7 ΔN mice. At E16.5 and E18.5, this difference in Nestin expression was more pronounced (Fig. 2b d). The overall number of proliferating cells in the SVZ was not affected at various stages of development (Supplementary Fig. 4). Immunoreactivity for the radial glia markers glutamate aspartate transporter (GLAST) and Brain lipid binding protein (BLBP) was augmented (Fig. 2c,e,f). Indeed, quantification revealed a significant increase in BLBP-positive cells in the SVZ of the fbw7 ΔN E18.5 cortex (Fig. 2l). In contrast, the abundance of intermediate progenitor cells marked by Tbr2 was diminished in the mutant cortex (Fig. 2g,l). Besides the decrease in Tbr2-positive intermediate progenitors, the proportion of Doublecortin-positive neuronal progenitors was reduced in the mutant SVZ throughout embryonic brain development (Supplementary Fig. 5). Consequently the number of cells expressing markers for neurons, NeuN, Tbr1, Ctip2 and Brn2, was decreased to varying extent (Fig. 2h l). Since there is a substantial reduction in total cell number in areas of differentiated cells (Fig. 1a,c) and a reduced percentage of neurons, the absolute number of neurons is reduced by more than 50% in the fbw7 ΔN cortex. In contrast to neurogenesis, absence of fbw7 did not have a significant effect on gliogenesis. GFAP-positive cells were hardly detectable in the IZ and CP of both the control and the mutant cortex at E18.5 and there was no significant difference in the number of cells expressing the astroglia marker S100 (Supplementary Fig. 6a c). There was also no change 5
7 in the percentage of cells expressing the oligodendroglia marker NG2 (Supplementary Fig. 6d,e). Similar to the cortex, an accumulation of cells expressing markers of immature cells and a reduction in NeuN-positive cells was also seen in the fbw7 ΔN tectum (Supplementary Fig. 7). It is likely that the severe reduction of neurons in fbw7 ΔN brains contributes to the perinatal lethality. Fbw7 controls neurosphere cell survival and differentiation To further investigate the function of Fbw7 in NSCs/NPCs, we prepared neurosphere cultures from E14.5 embryos. Neurospheres consist predominantly of neural progenitors and to a small extent of stem cells and differentiated cells 18. Neurospheres derived from fbw7 ΔN embryos were on average smaller in size and the number of neurospheres was significantly decreased (Fig. 3a c). The loss of Fbw7 function had no significant effect on NSC/NPC proliferation analysed by staining for ph3 and CFSE dilution (Supplementary Fig. 8). Whilst the large majority of cells in both control and fbw7 ΔN neurospheres expressed Nestin, the percentage of cells positive for Musashi-1 (Msi-1), which marks more immature cells, was greatly increased in fbw7 ΔN neurospheres (Fig. 3d,e). Similar to our observations in the cortex, the number of neurosphere cells expressing glial markers was not significantly altered in the absence of Fbw7 (Supplementary Fig. 6f i). Moreover, TUNEL assay revealed substantially increased cell death in fbw7 ΔN neurospheres (Fig. 3f,g). Thus it appears that a decrease in cell differentiation, accompanied by excessive progenitor cell death, reduces the efficiency of fbw7 ΔN neurosphere formation. This predicts that the rescue of cell death would improve neurosphere formation, an experiment we describe below. We next investigated the differentiation potential of Fbw7-deficient NSCs/NPCs. Cultured NSCs/NPCs from control mice readily formed a monolayer of cells after induction of differentiation, but fbw7 ΔN mutant NSCs/NPCs often retained the ball-shaped neurosphere 6
8 morphology (Supplementary Fig. 9), indicative of a problem to undergo the differentiation programme. 5 days after the onset of differentiation, the percentage of Nestin-positive NSCs/NPCs was low in control cultures. In contrast, a higher number of fbw7 ΔN neural cells retained Nestin expression (Fig. 4a,d). To further characterise the Nestin-positive cells that accumulated in fbw7 ΔN differentiation cultures, we investigated the expression of the classical RGC marker RC2. The majority of Nestin-positive cells were also RC2-positive (Fig. 4a,d), and the absence of Fbw7 also led to increased numbers of cells expressing two further RGC markers, BLBP and Vimentin (Supplementary Fig. 10a), indicating that RGCs represent the main immature cell population accumulating in fbw7 ΔN differentiation cultures. Additionally, CD133-positive neuroepithelial stem cells were very rare in control cultures 5 days after the onset of differentiation, whereas in fbw7-deficient cultures CD133-positive cells were readily detectable (Supplementary Fig. 10b). A similar increase in Nestin-positive cells was also observed when adherent NSC cultures were induced to differentiate (Supplementary Fig. 11a d). The accumulation of immature neural cells in fbw7 ΔN neurosphere cultures was also observed after prolonged time (11 days) under differentiation conditions, indicating that Fbw7 inactivation does not only delay, but genuinely blocks efficient neural differentiation (Supplementary Fig. 12). Conversely, the percentage of differentiated cells expressing the neuronal marker Map2 was reduced (Fig. 4b,d) whereas the frequency of astrocytes expressing Connexin43 and oligodendrocytes marked by O4 were not significantly altered in mutant differentiation cultures (Fig. 4b d). Triple IF staining revealed that the vast majority of both control and fbw7 ΔN neurospheres gave rise to differentiated cells of all 3 lineages (Supplementary Fig. 13). Taken together, the absence of Fbw7 results in decreased neurogenesis and an accumulation of cells expressing radial glia markers in cultured neurospheres. Fbw7 antagonises c-jun to regulate neural cell viability 7
9 The fbw7 locus encodes 3 different Fbw7 isoforms which are generated by alternative splicing of the first exon 4. Whereas the α and β fbw7 splice variants were expressed in neurospheres, the γ isoform was undetectable. fbw7 α and β were also present in total adult brain RNA. Notably, other organs showed different fbw7 splicing patterns (Supplementary Fig. 14). Fbw7 protein was detected in control neurosphere extracts but was absent in fbw7 ΔN neurospheres (Fig. 5a). To understand the mechanism of Fbw7 function, we investigated the expression levels of known substrates of SCF(Fbw7). Whilst phosphorylated c-myc and phosphorylated CyclinE protein levels were not substantially altered, there was a significant increase in the levels of active Notch1 and phosphorylated c-jun (Fig. 5a and Supplementary Fig. 11e). Phosphorylated c-jun is an important regulator of neuronal viability 7 9, and we have previously demonstrated that depletion of Fbw7 leads to cell death in PC12 neuronal-like cells due to increased levels of phosphorylated c-jun 19. Thus we investigated whether increased c- Jun function contributed to cell death in the absence of Fbw7 during neurogenesis. As in neurosphere cultures, the expression level of phosphorylated c-jun was also greatly increased in the brains of E18.5 fbw7 ΔN mutant embryos (Fig. 5b). To assess the functional importance of c-jun downstream of Fbw7, one allele of the c-jun gene was inactivated in a fbw7 ΔN -mutant background (fbw7 f/f ; c-jun f/+ ; Nestin-Cre + or fbw7 ΔN ; c-jun ΔN/+ mutant mice). Heterozygous deletion markedly reduced the levels of phosphorylated c-jun in the brain of Fbw7-deficient embryos (Fig. 5b). Genetic reduction of c-jun function significantly rescued the defect in cellularity in the mantle layer of the midbrain tectum, but the number of stem cells in the tectal ventricular zone was still elevated in fbw7 ΔN ; c-jun ΔN/+ embryos (Fig. 5c and Supplementary Fig. 16). Likewise, neurosphere formation in vitro was substantially increased in fbw7 ΔN ; c-jun ΔN/+ cells compared to fbw7 ΔN single mutants, which was accompanied by a considerable reduction in TUNEL-positive apoptotic cells in fbw7 ΔN ; c- 8
10 jun ΔN/+ neurospheres (Fig. 5d,e), indicating that c-jun-mediated cell death is causally involved in defective fbw7 ΔN neurosphere formation. An important mechanism of c-jun-mediated control of neuronal viability is the transcriptional regulation of pro-apoptotic members of the Bcl2 family of proteins mrna levels of c- jun, which is autoregulated by the c-jun protein itself 23, were increased in the absence of Fbw7. Likewise, the expression of the pro-apoptotic genes bad and bim were augmented to varying extents. The expression of the anti-apoptotic bcl2 gene was unaffected (Fig. 5f). As cells with the highest levels of phosphorylated c-jun are expected to undergo apoptosis in neurosphere cultures, it is plausible that this analysis underestimates the effect of Fbw7 inactivation on c-jun-mediated gene activation. Heterozygosity for c-jun reduced the expression of the pro-apoptotic c-jun target genes (Fig. 5f). In summary, negative regulation of c-jun activity by Fbw7 is essential to prevent excessive cell death during neurogenesis. Thus downregulation of c-jun in the fbw7 ΔN background rescued neurosphere formation and the number of apoptotic neurosphere cells in vitro and the reduction in cell number in vivo. Fbw7 antagonises Notch to allow NSC differentiation Heterozygosity for c-jun did not rescue the differentiation defect and the increased number of cells with radial glia identity in differentiating fbw7 ΔN neurosphere cultures, arguing that c-jun protein levels are not critical for Fbw7-regulated neural differentiation (Supplementary Fig. 17). In contrast, deregulated Notch signalling was a possible candidate for the differentiation defects observed in fbw7 ΔN mutants in vivo and in vitro. Overexpression of the activating intracellular fragment of Notch1, the NICD1, has been shown to be sufficient to promote radial glia identity during embryogenesis while abrogation of Notch signalling leads to depletion of RGCs 10,24. However, the molecular mechanism that antagonises Notch activity to allow neural differentiation to occur is not known. The increased protein expression of NICD1 seen by immunoblotting was confirmed by IHC 9
11 (Fig. 6a), indicating that Fbw7 regulates NICD1 levels both in vitro and in vivo. Our observation that BLBP levels were higher in fbw7 ΔN mutant embryos and neurosphere cultures (Fig. 2c,f,l and Supplementary Figs. 7 and 10a) was a first indication that increased Notch immunoreactivity translated into increased Notch function, since BLBP was reported to be a direct target of the Notch signalling pathway 25. To address the functional significance of increased Notch activity in Fbw7-regulated neural differentiation, we performed genetic and pharmacological rescue experiments. One allele of the Notch1 gene 26 was inactivated in a fbw7 ΔN -mutant background (fbw7 f/f ; Notch1 Δ/+ ; Nestin- Cre + or fbw7 ΔN ; Notch1 Δ/+ mutant mice). In addition, we made use of the γ-secretase inhibitor N-[N-(3,5-Difluorophenacetyl-L-Alanyl)]-S-phenylglycine t-butyl ester (DAPT), which prevents the release of the activating intracellular fragment of Notch proteins 27. The deletion of one Notch1 allele in the fbw7 ΔN background reduced the levels of NICD1 (Fig. 6a). Notch1 heterozygosity attenuated the accumulation of BLBP-positive cells and Nestin immunoreactivity and partially rescued the number of NeuN-positive neurons in the fbw7 ΔN - mutant cortex (Fig. 6b d and Supplementary Fig. 18a,b). mrna levels of the classical Notch target genes hes5 and hey1 were augmented in fbw7 ΔN neurosphere cultures, whereas hes1 levels were not significantly altered (Fig. 6e and Supplementary Figs. 11f and 19a). Heterozygosity for Notch1 as well as DAPT treatment resulted in a significant reduction in the expression of hes5 and hey1 (Fig. 6e and Supplementary Fig. 19a). Consequently, we next determined whether aberrant Notch activity contributes to the differentiation defects in fbw7 ΔN neurosphere cultures. Both Notch1 heterozygosity or DAPT treatment resulted in a substantial reduction in the percentage of Nestin-positive cells in fbw7 ΔN mutant differentiation cultures and the number of Map2-positive neurons was significantly increased in fbw7 ΔN ; Notch1 Δ/+ in comparison to fbw7 ΔN cultures (Fig. 6f,g and Supplementary Figs. 18c,d and 19b,c). This suggests that Fbw7 is essential to antagonise 10
12 Notch signalling to allow neural differentiation to occur. DISCUSSION This study identifies Fbw7 as a key regulator of neural progenitor viability and neural stem cell differentiation. We detected increased immunoreactivity for Nestin, GLAST and BLBP in the fbw7-mutant brain, suggesting that Fbw7 deficiency increases the number of immature cells. It is worth noting that GLAST and BLBP are also expressed in some astrocytes postnatally 2,28. Thus it was possible that the supernumerary GLAST-positive and BLBPpositive cells in the fbw7 ΔN cortex were generated due to an increase in astrogliogenesis. However, expression of the astrocyte markers GFAP, S100 and Connexin43 was unaltered in fbw7-mutant cells (Fig. 4b,d and Supplementary Fig. 6a c,f,g). The increased percentage of cells positive for RC2 and other RGC markers in differentiating neurospheres (Fig. 4a,d and Supplementary Fig. 10a) together with the known function of Notch signalling to promote RGC fate 24,29 is in agreement with a role for Fbw7 in RGC development. While we have obtained no evidence for a defect in gliogenesis, Fbw7 deficiency leads to decreased numbers of neurons (Fig. 2h l). The loss of Fbw7 led to a reduction of Tbr2- positive intermediate progenitors (Fig. 2g,l). There was also a significant decrease in Doublecortin-positive neuroblasts in the mutant SVZ throughout embryonic brain development, indicative of a defect in neurogenesis (Supplementary Fig. 5). It is conceivable that Fbw7 deficiency inhibits neural stem cell differentiation at the expense of neuronal progenitors, a phenotype consistent with a gain of Notch function 24. Furthermore, Fbw7 deficiency increases progenitor cell death, which leads to a further reduction in neuronal number. However, a defect in neuronal maturation, and thus marker expression, may also contribute to the fbw7 ΔN phenotype. 11
13 SCF(Fbw7) has several substrates with diverse biological activities. Downregulation of the Fbw7-substrate c-jun in the fbw7 ΔN background rescued the increased number of apoptotic neurosphere cells and the reduced number of cells in areas of differentiated cells in the brain. Regulation of c-jun by Fbw7 is essential to control neural cell viability. Paradoxically, JNK activity is very high in neurons 19,30, and a role for JNK signalling in neuronal migration and maintenance of cytoskeletal integrity of neuronal cells has been described 31,32. Therefore SCF(Fbw7) may function to inactivate apoptotic c-jun-dependent JNK signalling, allowing neural cells to tolerate potentially neurotoxic JNK activity. c-jun is not involved in the regulation of neural differentiation by Fbw7. This process is controlled by Fbw7 through the regulation of Notch activity. Downregulation of the Fbw7- substrate Notch in the fbw7 ΔN background alleviated the block in cell differentiation and rescued neuronal numbers. While it is known that Notch activity needs to be downregulated for neuronal differentiation to occur 12, the mechanism that controls this transition has remained enigmatic. This work shows for the first time that Fbw7 is an important molecular switch that is required for attenuation of Notch activity during neurogenesis. It is noteworthy that SCF(Fbw7) can target various members of the Notch family of proteins 14, making it wellsuited as a general antagonist of Notch function. Decreased Notch pathway activity in mice lacking Notch1, rbp-j (cbf1) or the Notch ligand dll1 as well as in hes1 single and hes1/hes5 double mutants results in precocious neuronal differentiation 12,13. hes5 and hes1 are well known targets of the Notch signalling pathway 33, and are crucial mediators of Notch function in the brain. However, in many instances hes1 and hes5 are differentially regulated. Notch-1 or RBP-J-mutant embryos show reduced expression of hes5, but hes1 expression was normal 13,34. Similarly, γ-secretase inhibition of Notch activity in neural progenitors was reported to have a much stronger effect on hes5 compared to hes1 35. In our mouse model fbw7 deletion induces hes5 mrna increase while having a mild effect on hes1, suggesting that fbw7 affects Notch1 with an overall balance towards hes5. This is in line with previous 12
14 observations in the mouse retina, in which glial cell specification is modulated by hes5 in a hes1-independent manner 36. Fbw7 appears to use distinct substrates to regulate different biological processes, however the molecular mechanism of how this occurs remains unknown. For instance, c-myc protein accumulates in fbw7-deficient hematopoietic stem cells 37. In contrast, Notch1 and c-jun are increased upon fbw7 deletion in mouse embryonic fibroblasts (MEFs), but no effect is found on CyclinE or c-myc in these cells 38. In the developing brain, the main SCF(Fbw7) substrates affected are Notch1 and c-jun, while c-myc and CyclinE protein levels are not greatly altered. One potential explanation could be the relative abundance of Fbw7 isoforms. The Fbw7γ isoform has been shown to mediate degradation of c-myc and CyclinE The absence of Fbw7γ in neural stem cells and progenitors (Supplementary Fig. 14) could therefore impede efficient degradation of CyclinE or c-myc. Thus differential expression of Fbw7 isoforms may contribute to cell-type specific substrate degradation. The function of Fbw7 in stem cells is highly dependent on the organ system. Whereas we demonstrate here that Fbw7 is required for neural differentiation, inactivation of Fbw7 in hematopoietic stem cells (HSCs) causes defective maintenance of quiescence and premature depletion of HSCs 37,42. Thus the lack of Fbw7 leads to opposite functional consequences in HSCs and NSCs. It is plausible that cell-type specific differential regulation of Fbw7 and its substrates is one mechanism that could explain the distinct roles of Fbw7 in different types of stem cells. 13
15 FIGURE LEGENDS Figure 1 Fbw7 controls cell number and survival in the brain. (a) Haematoxylin and eosin (H&E) staining of the E18.5 forebrain from fbw7 f/f and fbw7 ΔN mouse embryos. Rectangles mark comparable regions of the cortex shown below in high magnification. Scale bars: 100 μm. (b) H&E staining of comparable regions of the fbw7 f/f and fbw7 ΔN E18.5 midbrain. Rectangles mark the area of the tectum shown below in high magnification. Scale bars: 200 μm. (c) Histogram showing the relative quantity of cells in the ventricular zone (VZ), subventricular zone (SVZ), intermediate zone (IZ) and the cortical plate (CP) of the fbw7 f/f and fbw7 ΔN E18.5 cortex. Cell number in the fbw7 f/f E18.5 cortex is normalised to 1 (= 100%); n = 3. (d) Histogram showing the relative quantity of cells in the ventricular zone (VZ) and the mantle layer (ML) of the fbw7 f/f and fbw7 ΔN E18.5 tectum. Cell numbers in the fbw7 f/f E18.5 tectum are normalised to 1 (= 100%); n = 5. (e) 3,3'-Diaminobenzidine (DAB) staining for the mitotic marker phosphorylated histone H3 (ph3) on representative sections of the fbw7 f/f and fbw7 ΔN E16.5 tectum. Cells are counterstained with H&E. Scale bars: 50 μm. (f) Immunohistochemistry for active Caspase 3 (Casp3; green) on representative sections of the fbw7 f/f and fbw7 ΔN E16.5 tectum. DNA (blue) is counterstained with DAPI. Scale bars: 50 μm. (g) Quantification of phosphorylated histone H3 (ph3 + ) and active Caspase 3 (Casp3 + ) positive cells in the fbw7 f/f and fbw7 ΔN E16.5 tectum; n = 3. Error bars represent the standard error of the mean (s.e.m.). Statistical significance: n.s.: not significant; *: p 0.05; **: p 0.01; ***: p (unpaired t test). Figure 2 Fbw7 controls stem cell differentiation and neurogenesis in the brain. (a) fbw7 (exon 6 10 specific probe) in situ hybridisation and sense control with H&E counterstain on the fbw7 f/f E14.5 cortex. Scale bars: 50 μm. (b) Immunohistochemistry for Nestin (red) on the 14
16 fbw7 f/f and fbw7 ΔN E14.5 cortex. White rectangles mark areas shown in high magnification in panels at the top. DNA (blue) is counterstained with DAPI. Scale bars: 50 μm. (c) Immunohistochemistry for Nestin (red; left panels) and BLBP (red; right panels) on the fbw7 f/f and fbw7 ΔN E16.5 cortex. White rectangles mark areas shown in high magnification in panels at the top. DNA (blue) is counterstained with DAPI. Scale bars: 50 μm. (d) DAB staining for Nestin on the fbw7 f/f and fbw7 ΔN E18.5 cortex. Black rectangles mark areas shown in high magnification in panels at the top. Cells are counterstained with H&E. Scale bars: 50 μm. (e) Immunohistochemistry for GLAST (green) on the fbw7 f/f and fbw7 ΔN E18.5 cortex. White rectangles mark areas shown in high magnification in panels at the top. Cells are counterstained with DAPI. Scale bars: 50 μm. DAB staining for (f) BLBP, (g) Tbr2, (h) NeuN, (i) Tbr1, (j) Ctip2 and (k) Brn2 on the fbw7 f/f and fbw7 ΔN E18.5 cortex. Black rectangles mark areas shown in high magnification in panels at the top. Cells are counterstained with H&E. Scale bars: 50 μm. (l) Quantification of BLBP-positive cells in the SVZ and Tbr2, NeuN, Tbr1, Ctip2 and Brn2-positive cells in the IZ and the CP of the fbw7 f/f and fbw7 ΔN E18.5 cortex. n = 3. Error bars represent the s.e.m. Statistical significance: *: p 0.05 (unpaired t test); **: p CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. Figure 3 Absence of Fbw7 leads to retention of markers for immature cells and induces apoptosis in vitro. (a) Phase contrast pictures of fbw7 f/f and fbw7 ΔN neurosphere cultures under self-renewal conditions. Arrowheads denote neurospheres. Scale bars: 100 μm. Histograms showing (b) the size (in μm) and (c) the relative quantity of fbw7 f/f and fbw7 ΔN neurospheres. Neurosphere numbers in fbw7 f/f cultures are normalised to 1 (= 100%); n = 4. (d) Immunocytochemistry for Nestin (green)/musashi1 (Msi1; red) on fbw7 f/f and fbw7 ΔN 15
17 neurosphere sections. DNA (blue) was counterstained with DAPI. Scale bars: 100 μm. (e) Quantification of Musashi1-positive cells (Msi1 + ) per neurosphere in fbw7 f/f and fbw7 ΔN neurosphere cultures; n = 3. (f) DAB staining for TUNEL (TdT-mediated dutp-biotin nick end labeling) positive cells on fbw7 f/f and fbw7 ΔN neurosphere sections. Arrowheads denote TUNEL-positive cells. Scale bars: 100 μm. (g) Histogram showing the percentage of TUNEL-positive cells in fbw7 f/f and fbw7 ΔN neurosphere cultures; n = 3. Error bars represent the s.e.m. Statistical significance: **: p 0.01; ***: p (unpaired t test). Figure 4 Absence of Fbw7 blocks differentiation in vitro. Immunocytochemistry for (a) RC2 (green) and Nestin (red), (b) Map2 (green) and Connexin43 (Cx43; red) and (c) O4 on fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days under differentiation conditions. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 50 μm. (d) Quantification of RC2/Nestin, Map2, Cx43 and O4-positive cells in fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days under differentiation conditions. Error bars represent the s.e.m. Statistical significance: n.s.: not significant; *: p 0.05 (unpaired t test). Figure 5 Negative regulation of c-jun by Fbw7 controls neural cell viability. (a) Western blot analysis of Fbw7, c-jun, activated Notch1 (NICD1), threonine 58/serine 62 phosphorylated c- Myc (p-c-myc), threonine 395 phosphorylated cycline and β-actin on protein lysates from fbw7 f/f and fbw7 ΔN neurospheres (cropped images, full-length blots are presented in Supplementary Fig. 15a). (b) Immunohistochemistry for serine 73 phosphorylated c-jun (pc-jun; red) on representative sections of the fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; c-jun ΔN/+ E
18 tectum. DNA (blue) was counterstained with DAPI. Scale bars: 50 μm. (c) Histogram showing the relative quantity of cells in the ventricular zone (VZ) and the mantle layer (ML) of the fbw7 f/f (n = 5), fbw7 ΔN (n = 5) and fbw7 ΔN ; c-jun ΔN/+ (n = 3) E18.5 tectum. Cell numbers in the fbw7 f/f E18.5 tectum are normalised to 1 (= 100%). (d) Histogram showing the relative quantity of neurospheres in fbw7 f/f (n = 4), fbw7 ΔN (n = 4) and fbw7 ΔN ; c-jun ΔN/+ (n = 2) neurosphere cultures after 2 weeks under self-renewal conditions. Neurosphere number in fbw7 f/f neurosphere cultures are normalised to 1 (= 100%). (e) Histogram showing the percentage of TUNEL-positive cells in fbw7 f/f (n = 3), fbw7 ΔN (n = 3) and fbw7 ΔN ; c-jun ΔN/+ (n = 2) neurosphere cultures. (f) qrt-pcr analysis of fbw7, c-jun, bad, bim and bcl2 transcripts in fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; c-jun ΔN/+ neurosphere cells. The data are normalised to gapdh and represented as fold change over RNA levels in fbw7 f/f neurosphere cells, which is set to 1. Error bars represent the s.e.m. Statistical significance: n.s.: not significant; *: p 0.05; **: p 0.01; ***: p (unpaired t test). Figure 6 Fbw7 controls stem cell differentiation by antagonising Notch. (a) Immunohistochemistry for activated Notch1 (NICD1; red) on representative sections of the fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ E18.5 cortex. DNA (blue) was counterstained with DAPI. Scale bars: 50 μm. DAB staining for (b) Nestin and (c) BLBP on the fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ E18.5 cortex. Cells are counterstained with H&E. Scale bars: 50 μm. (d) Quantification of BLBP-positive cells in the IZ and the CP of the fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ E18.5 cortex. n = 3. (e) qrt-pcr analysis of hes5, hey1 and hes1 transcripts in fbw7 f/f, fbw7 f/f ; Notch1 Δ/+, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ neurospheres. The data are normalised to gapdh and represented as fold change over RNA levels in fbw7 f/f neurospheres, which is set to 1. (f) Immunocytochemistry for Nestin (green) on fbw7 f/f, fbw7 f/f ; Notch1 Δ/+, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ neurosphere cultures after 5 days under 17
19 differentiation conditions. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 50 μm. (g) Histogram showing the percentage of Nestin-positive cells in fbw7 f/f, fbw7 f/f ; Notch1 Δ/+, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ neurosphere cultures after 5 days under differentiation conditions. n = 3. Error bars represent the s.e.m. Statistical significance: n.s.: not significant; *: p 0.05; **: p 0.01; ***: p (unpaired t test). CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. SUPPLEMENTARY FIGURE LEGENDS Supplementary Figure 1 Efficient fbw7 deletion in fbw7 ΔN mice. (a) Schematic representation of the targeting construct before and after Cre recombination. Exon 5 of the fbw7 allele is flanked by loxp-sites (fbw7 f ) and excised upon crossing fbw7 f/f mice to transgenic mice expressing Cre recombinase. Conditional knockout of fbw7 in the brain occurs by expressing Cre under the control of a Nestin promoter (fbw7 ΔN ). (b) fbw7 (exon 2 5 specific probe) in situ hybridisation with Giemsa counterstain (blue). Rectangles mark comparable regions of the cortex (1, 1 ) and the eye (2, 2 ) shown below in high magnification for fbw7 f/f and fbw7 ΔN E18.5 heads. Scale bars: 100 μm. Supplementary Figure 2 Cortex size in the fbw7 ΔN brain. (a) Schematic representation of the E18.5 mouse forebrain (fb; green) and midbrain (mb; red) dorsal view. Comparable sagittal sections of fbw7 f/f and fbw7 ΔN E18.5 heads were taken alongside the lateral-medial axis (1, 2, 3) and are shown in (b) stained with H&E. Scale bars: in 1, 580 μm; in 2, 730 μm; in 3,
20 μm. Supplementary Figure 3 Decreased cellularity in the fbw7 ΔN brain. Haematoxylin and eosin (H&E) staining of the E18.5 (a) cerebellum and (b) thalamus from fbw7 f/f and fbw7 ΔN mouse embryos. Rectangles mark comparable regions shown below in high magnification. Scale bars: 50 μm. Supplementary Figure 4 Loss of Fbw7 does not affect proliferation in vivo. DAB staining for the S-phase marker Ki67 on representative sections of the fbw7 f/f and fbw7 ΔN cortex at (a) E10.5, (b) E14.5, (c) E16.5 and (d) E18.5. Cells are counterstained with H&E. Scale bars: 50 μm. (e) Quantification of Ki67-positive cells in the E10.5 fbw7 f/f and fbw7 ΔN cortex and in the SVZ of the E14.5, E16.5 and E18.5 fbw7 f/f and fbw7 ΔN cortex. n = 3. Error bars represent the s.e.m. CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. Supplementary Figure 5 Reduced number of neuroblasts in the fbw7 ΔN cortex. DAB staining for Doublecortin (Dcx) in the (a) E18.5, (b) E16.5 and (c) E14.5 fbw7 f/f and fbw7 ΔN cortex. Black rectangles mark areas shown in high magnification in panels at the top. Cells are counterstained with H&E. Scale bars: 50 μm. (d) Quantification of Dcx-positive cells in the SVZ of the E18.5 (n = 4), E16.5 (n = 3) and E14.5 (n = 3) fbw7 f/f and fbw7 ΔN cortex. Error bars represent the s.e.m. Statistical significance: *: p 0.05; **: p 0.01 (unpaired t test). CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. Supplementary Figure 6 Loss of Fbw7 does not affect gliogenesis. (a) 19
21 Immunohistochemistry for GFAP (red) on representative sections of the fbw7 f/f and fbw7 ΔN E18.5 cortex. DNA (blue) was counterstained with DAPI. Scale bars: 50 μm. DAB staining for (b) S100 and (d) NG2 in the CP of the E18.5 fbw7 f/f and fbw7 ΔN cortex. Red arrowheads denote positive cells. Scale bars: 50 μm. Quantification of (c) S100-positive and (e) NG2- positive cells in the IZ and the CP of the E18.5 fbw7 f/f and fbw7 ΔN cortex. n = 3. DAB staining for (f) S100 and (h) NG2 on fbw7 f/f and fbw7 ΔN neurosphere sections. Red arrowheads denote positive cells. Scale bars: 50 μm. Quantification of (g) S100-positive and (i) NG2-positive cells in fbw7 f/f and fbw7 ΔN neurospheres. n = 3. Error bars represent the s.e.m. Statistical significance: n.s.: not significant (unpaired t test). CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. Supplementary Figure 7 Increased stem cell and decreased neuronal marker expression in the fbw7 ΔN tectum. From left to right: Immunohistochemistry for Musashi1 (Msi1; red), BLBP (red), Nestin (red) and NeuN (green) on representative sections of the fbw7 f/f and fbw7 ΔN E18.5 tectum. DNA (blue) was counterstained with DAPI. ML: mantle layer, VZ: ventricular zone. Scale bars: 50 μm. Supplementary Figure 8 Loss of Fbw7 does not affect proliferation in vitro. (a) Immunocytochemistry for serine 10 phosphorylated histone H3 (ph3; red) on fbw7 f/f and fbw7 ΔN neurosphere sections. DNA (blue) was counterstained with DAPI. Arrowheads denote ph3-positive cells. Scale bars: 50 μm. (b) FACS histograms showing carboxyfluorescein diacetate succinimidyl ester (CFSE) intensity in fbw7 f/f and fbw7 ΔN neurosphere cultures 1 (d1), 3 (d3), 5 (d5) and 7 (d7) days after CFSE staining. u: unstained control. (c) Histogram showing the loss of CFSE intensity (in %) over time (in days). (d) Histogram showing cell division rates of fbw7 f/f and fbw7 ΔN neurosphere cells based on the loss of CFSE intensity. 20
22 Error bars represent the s.e.m. Supplementary Figure 9 fbw7 ΔN neurospheres exhibit differentiation defect. (a) Phase contrast pictures of fbw7 f/f and fbw7 ΔN neurosphere cultures under differentiation conditions. (b) Stainings for DNA (blue) with Hoechst on fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days of differentiation. Scale bars: 50 μm. Supplementary Figure 10 Increased stem cell marker expression in fbw7 ΔN neurosphere differentiation cultures. Immunocytochemistry for (a) Vimentin (green) and BLBP (red) and (b) CD133 (green) on fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days under differentiation conditions. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 100 μm. Supplementary Figure 11 Loss of Fbw7 leads to similar results in adherent NSC cultures in comparison to neurosphere cultures. (a) Phase contrast pictures of fbw7 f/f and fbw7 ΔN adherent NSC cultures under self-renewal conditions. Scale bars: 50 μm. Immunocytochemistry for Nestin (green) on fbw7 f/f and fbw7 ΔN adherent NSC cultures (b) under self-renewal conditions and (c) after 5 days under differentiation conditions. Scale bars: 50 μm. (d) Quantification of Nestin-positive cells in fbw7 f/f and fbw7 ΔN adherent NSC cultures after 5 days under differentiation conditions. (e) Western blot analysis of Fbw7, serine 73 phosphorylated c-jun (p-c-jun), activated Notch1 (NICD1), threonine 58/serine 62 phosphorylated c-myc (p-c- Myc), threonine 395 phosphorylated cycline and β-actin on protein lysates from fbw7 f/f and fbw7 ΔN adherent NSC cultures (cropped images, full-length blots are presented in Supplementary Fig. 15b). (f) qrt-pcr analysis of fbw7, c-jun and hes5 transcripts in cells 21
23 from fbw7 f/f and fbw7 ΔN adherent NSC cultures. The data are normalised to gapdh and represented as fold change over RNA levels in fbw7 f/f adherent NSCs, which is set to 1. Error bars represent the s.e.m. Statistical significance: *: p 0.05 (unpaired t test). Supplementary Figure 12 Increased number of immature cells in fbw7 ΔN neurosphere cultures after prolonged time under differentiation conditions. Immunocytochemistry for Nestin (green) on fbw7 f/f and fbw7 ΔN neurosphere cultures after 11 days of differentiation. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 100 μm. Supplementary Figure 13 Loss of Fbw7 does not affect neurosphere multipotentiality. Immunocytochemistry for Map2 (green), O4 (red) and Connexin43 (Cx43; magenta) on fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days of differentiation. DNA (blue) was counterstained with Hoechst Scale bars: 50 μm. Supplementary Figure 14 fbw7 isoform expression. qrt-pcr analysis showing the relative abundance of fbw7 isoforms α, β and γ in fbw7 f/f neurospheres, adult brain, skeletal muscle and liver normalised to gapdh expression. Expression of fbw α is set to 1. Error bars represent the standard deviation (s.d.). Supplementary Figure 15 Full-length Western Blots. Full-length Western Blots from (a) Fig. 5a and (b) Supplementary Fig. 11e. Molecular weight standards are shown in kda. Supplementary Figure 16 Decreased cellularity in fbw7 ΔN brains is c-jun-dependent. 22
24 Haematoxylin and eosin (H&E) staining of comparable regions of the fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; c-jun ΔN/+ E18.5 midbrain. Rectangles mark the area of the tectum shown below in high magnification. ML: mantle layer, VZ: ventricular zone. Scale bars: 200 μm. Supplementary Figure 17 Differentiation defect in fbw7 ΔN neurosphere cultures is not c-jundependent. Histogram showing the percentage of Nestin-positive cells in fbw7 f/f (n = 8), fbw7 ΔN (n = 11) and fbw7 ΔN c-jun ΔN/+ (n = 2) neurosphere cultures after 5 days of differentiation. Error bars represent the s.e.m. Statistical significance: n.s.: not significant; ***: p (unpaired t test). Supplementary Figure 18 Notch downregulation rescues neuronal numbers. (a) DAB staining for NeuN on the E18.5 fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ cortex. Cells are counterstained with H&E. Scale bars: 50 μm. (b) Quantification of NeuN-positive cells in the IZ and the CP of the E18.5 fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ cortex (n = 3). (c) Immunocytochemistry for Map2 (green) on fbw7 f/f, fbw7 ΔN and fbw7 ΔN ; Notch1 Δ/+ neurosphere cultures after 5 days under differentiation conditions. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 50 μm. (d) Histogram showing the percentage of Map2-positive cells in fbw7 f/f (n = 3), fbw7 ΔN (n = 4) and fbw7 ΔN ; Notch1 Δ/+ (n = 5) neurosphere cultures after 5 days under differentiation conditions. Error bars represent the s.e.m. Statistical significance: *: p 0.05; **: p 0.01 (unpaired t test). CP: cortical plate, IZ: intermediate zone, SVZ: subventricular zone, VZ: ventricular zone. Supplementary Figure 19 Inhibition of Notch signalling alleviates the block in stem cell 23
25 differentiation. (a) qrt-pcr analysis of hes5, hey1 and hes1 transcripts in fbw7 f/f and fbw7 ΔN neurospheres treated with DMSO (control) or 1 μm DAPT. The data are normalised to gapdh and represented as fold change over RNA levels in fbw7 f/f + DMSO neurospheres, which is set to 1. (b) Immunocytochemistry for Nestin (green) on fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days under differentiation conditions treated with DMSO (control) or 1 μm DAPT. White squares mark areas shown in high magnification in panels on the right. DNA (blue) was counterstained with Hoechst Scale bars: 100 μm. (c) Histogram showing the percentage of Nestin-positive cells in fbw7 f/f and fbw7 ΔN neurosphere cultures after 5 days under differentiation conditions treated with DMSO (control) or 1 μm DAPT. n = 5. Error bars represent the s.e.m. Statistical significance: n.s.: not significant; **: p 0.01; ***: p (unpaired t test). ACKNOWLEDGEMENTS We are grateful to the Animal Unit, Equipment Park, FACS Lab and the Experimental Pathology Lab at the London Research Institute for technical help and advice on histology. We are grateful to F. Radke for floxed Notch1 mice. We thank R. Sancho for the data on fbw7 isoform expression in various tissues. We thank C. R. Da Costa, S. Marino and A. Martin- Villalba for critical reading of the manuscript. S.M.B. is supported by a Federation of the Societies of Biochemistry and Molecular Biology (FEBS) Long Term Fellowship. Part of this work is supported by an MRC project grant (85704) to A.B. The London Research Institute is funded by Cancer Research UK. AUTHOR CONTRIBUTIONS 24
26 J.D.H. designed and conducted the majority of the experiments, did the data analyses and cowrote the manuscript. A.J. generated the fbw7 f/f mice and performed the radioactive ISH. S.M.B. performed the experiments on adherent NSC cultures. E.N. did the IHC stainings. B.S.-D. performed the non-radioactive ISH. S.B. gave advice on neurosphere sectioning. A.B. supervised the project, directed the experiments and wrote the manuscript. 25
27 MATERIALS AND METHODS Mouse Lines The fbw7 (floxed exon 5) transgenic construct was electroporated into embryonic stem cells, stable transfectants were selected and mice generated according to standard protocols 7. Details will be described in Jandke et al., in preparation. c-jun f/f, Notch1 f/f and Nestin-Cre mice have been described 9,26. All experiments involving mice were approved by the London Research Institute (LRI) Animal Ethics Committee following Home Office guidelines. Cell culture Neural cells were isolated from E14.5 fore- and midbrains of fbw7 f/f, fbw7 f/f ; Notch1 Δ/+, fbw7 ΔN, fbw7 ΔN ; c-jun ΔN/+ and fbw7 ΔN ; Notch1 Δ/+ mouse embryos. Cells were cultured under self-renewal conditions in Neurobasal Medium (Invitrogen) supplemented with 1% Penicillin/Streptomycin (10000 U/ml; Invitrogen), 1% L-glutamine (200 mm; Invitrogen), 2% B27 supplement (Invitrogen) and human epidermal growth factor (EGF; 20 ng/ml; PeproTech) and fibroblast growth factor (FGF-basic; 20 ng/ml; PeproTech). All experiments were performed using secondary and tertiary neurospheres. Adherent NSC cultures were derived as previously described 43 with minor modifications. Briefly, cells were cultured in Neurobasal Medium (Invitrogen) supplemented with 1% Penicillin/Streptomycin (10000 U/ml; Invitrogen), 1% L-glutamine (200 mm; Invitrogen), 2% B27 supplement (Invitrogen), 1% N-2 supplement (Invitrogen), 20 ng/ml EGF (PeproTech), 20 ng/ml FGF-basic (PeproTech) and 1 μg/ml laminin (Sigma). For differentiation, growth factors were withdrawn from the medium and 10% NeuroCult Differentiation Supplement (StemCell Technologies) was added. Under differentiation conditions, 5 x 10 5 cells were plated on poly-l-ornithine (0.01% solution; Sigma; diluted 1:10 in 150 mm disodium tetraborate; Sigma) coated cover slips. 26
28 Single cell suspensions were made from neurospheres using AccuMax (PAA Laboratories). To assess neurosphere formation, cells were plated in limiting dilutions from 500 to 4 cells in 96-well-plates. Neurospheres were counted after 2 weeks in culture. To examine cell division rates, neurosphere-derived cells were stained with carboxyfluorescein diacetate succinimidyl ester (CFSE) using the CellTrace CFSE Cell Proliferation Kit (Invitrogen) according to the manufacturer s instructions. FACSmeasurements for CFSE intensity were performed every 48 h after CFSE-staining for one week using an LSR II Flow Cytometer (BD). By means of DAPI-staining, viable cells (DAPInegative) were selected. Analysis was carried out using CellQuest Pro (BD) and FlowJo (Tree Star) software. For the inhibition of Notch signalling, neurospheres were treated with 1 μμ N-[N-(3,5- Difluorophenacetyl-L-Alanyl)]-S-phenylglycine t-butyl ester (DAPT; Sigma) for 72 h. Immunohistochemistry and immunocytochemistry Mouse brains were dissected from E10.5, E14.5, E16.5 and E18.5 fbw7 f/f, fbw7 ΔN, fbw7 ΔN ; c- Jun ΔN/+ and fbw7 ΔN ; Notch1 Δ/+ mouse embryos. Brains were fixed in 10% neutral buffered formalin overnight, transferred to 70% ethanol, processed and embedded into paraffin. Neurospheres were fixed in 10% neutral buffered formalin overnight and processed using the Shandon Cytoblock Cell Preparation System (Thermo Fisher Scientific). Sections were cut (for brain sections: sagittally) at 4 μm for Haematoxylin & Eosin (H&E) staining, 3,3'- Diaminobenzidine (DAB) staining, TUNEL (TdT-mediated dutp-biotin nick end labeling) and fluorescent immunohistochemistry. To define comparable sections, brains were cut alongside the medial-lateral axis with every 10 th section stained with H&E. For cell counting, all cells in the tectal ML and VZ as well as in the CP, IZ, SVZ and VZ of the cortex were counted in an area of the same width. Layer boundaries were determined by cell morphology and layer-specific marker expression. 27
29 Cells from neurosphere differentiation cultures were fixed in ice-cold methanol for 3 mins or in 4% paraformaldehyde for 20 mins. For immunohistochemistry and immunocytochemistry, antibodies against activated Notch1 (Abcam), active Caspase3 (R&D), BLBP (Abcam), Brn2 (Santa Cruz Biotechnology), CD133 (Prominin 1; ebioscience), Connexin43 (Zymed), Ctip2 (Abcam), serine 73 phosphorylated c-jun (Cell Signaling), Doublecortin (Santa Cruz Biotechnology), GLAST (LifeSpan Biosciences), serine 10 phosphorylated histone H3 (ph3; Millipore), Ki67 (DAKO), Map2 (Sigma), Musashi1 (Chemicon), Nestin (IHC: Chemicon; ICC: BD (monoclonal); Abcam (polyclonal)), NeuN (Chemicon), NG2 (Chemicon), O4 (Chemicon), RC2 (DSHB), S100 (Abcam), Tbr1 (Abcam), Tbr2 (Abcam) and Vimentin (Abcam) were used. DNA was counterstained with 4'-6-Diamidino-2-phenylindole (DAPI; Sigma) or Hoechst (Sigma). In situ hybridisation Radioactive in situ hybridisation (ISH) was performed by the CRUK in situ hybridisation service at the LRI. The in situ probe corresponded to exon 2 5 of the murine fbw7 cdna. Non-radioactive ISH was performed as previously described 44 with minor modifications. For fbw7 ISH, DIG-labelled 800 bp PCR product (exons 6 to 10) was used. Western Blot Analysis Neurospheres were suspended in cell lysis buffer (Cell Signaling) supplemented with protease inhibitor cocktail (Sigma). Western blot analysis was performed as previously described 45. For immunoblotting, antibodies against activated Notch1 (Abcam), c-jun (Santa Cruz Biotechnology), serine 73 phosphorylated c-jun (Cell Signaling), Fbw7 (LRI/CRUK; Abcam), threonine 58/serine 62 phosphorylated c-myc (Cell Signaling), threonine 395 phosphorylated cycline (Santa Cruz Biotechnology) and β-actin (Sigma) were used. 28
30 qrt-pcr analysis For qrt-pcr analysis, total mrna was isolated from neurospheres using the RNeasy Minikit (QIAGEN) according to the manufacturer s instructions. cdna was synthesized using Invitrogen Superscript III reagents according to the manufacturer s instructions. Quantitative real-time PCR was performed measuring SYBR Green incorporation (Platinum Quantitative PCR SuperMix-UDG w/rox, Invitrogen) on an ABI7900HT (Applied Bioscience). Data were analyzed using the SDS 2.3 software. The following primers were used for murine cell qrt-pcr analysis: F-Bad: 5 -CAGGGAGAAGAGCTGACGTACA-3 R-Bad: 5 -CCACCCCTCCGTGGCTAT-3 F-Bcl2 (beta): 5 -GCTCCCCTGACCTCTCACTCT-3 R-Bcl2 (beta): 5 -CTGGATTCTTGCTCCCTCACA-3 F-Bim: 5 -CCCCTACCTCCCTACAGACAGA-3 R-Bim: 5 -GCGCAGATCTTCAGGTTCCT-3 F-c-Jun: 5 -TGAAAGCTGTGTCCCCTGTC-3 R-c-Jun: 5 -ATCACAGCACATGCCACTTC-3 F-Fbw7α: 5 -CTGACCAGCTCTCCTCTCCATT-3 R-Fbw7α: 5 -GCTGAACATGGTACAAGGCCA-3 F-Fbw7β: 5 -TTGTCAGAGACTGCCAAGCAG-3 R-Fbw7β: 5 -GACTTTGCATGGTTTCTTTCCC-3 F-Fbw7γ: 5 -AACCATGGCTTGGTTCCTGTTG-3 R-Fbw7γ: 5 -CAGAACCATGGTCCAACTTTC-3 F-Fbw7 (exon5): 5 -TTCATTCCTGGAACCCAAAGA-3 R-Fbw7 (exon5): 5 -TCCTCAGCCAAAATTCTCCAGTA-3 29
31 F- Gapdh: 5 - TGAAGCAGGCATCTGAGGG -3 R-Gapdh: 5 -CGAAGGTGGAAGAGTGGGAG -3 F-Hes1: 5 -TCAGCGAGTGCATGAACGA-3 R-Hes1: 5 -TGCGCACCTCGGTGTTAAC-3 F-Hes5: 5 -TGCAGGAGGCGGTACAGTTC-3 R-Hes5: 5 -GCTGGAAGTGGTAAAGCAGCTT-3 F-Hey1: 5 -GGCAGCCCTAAGCACTCTCA-3 R-Hey1: 5 -TTCAGACTCCGATCGCTTACG-3 Statistical Analysis Statistical evaluation was performed by Student s unpaired t test. Data are presented as mean ± standard error of the mean (s.e.m.). *: p 0.05 was considered statistically significant, **: p 0.01 was considered very statistically significant, ***: p was considered extremely statistically significant. 30
32 REFERENCES 1. Merkle, F.T. & Alvarez-Buylla, A. Neural stem cells in mammalian development. Curr Opin Cell Biol 18, (2006). 2. Gotz, M. & Barde, Y.A. Radial glial cells defined and major intermediates between embryonic stem cells and CNS neurons. Neuron 46, (2005). 3. Ho, M.S., Tsai, P.I. & Chien, C.T. F-box proteins: the key to protein degradation. J Biomed Sci 13, (2006). 4. Welcker, M. & Clurman, B.E. FBW7 ubiquitin ligase: a tumour suppressor at the crossroads of cell division, growth and differentiation. Nat Rev Cancer 8, (2008). 5. Tan, Y., Sangfelt, O. & Spruck, C. The Fbxw7/hCdc4 tumor suppressor in human cancer. Cancer Lett 271, 1 12 (2008). 6. Fuchs, S.Y. Tumor suppressor activities of the Fbw7 E3 ubiquitin ligase receptor. Cancer Biol Ther 4, (2005). 7. Behrens, A., Sibilia, M. & Wagner, E.F. Amino-terminal phosphorylation of c-jun regulates stressinduced apoptosis and cellular proliferation. Nat Genet 21, (1999). 8. Besirli, C.G., Wagner, E.F. & Johnson, E.M., Jr. The limited role of NH2-terminal c-jun phosphorylation in neuronal apoptosis: identification of the nuclear pore complex as a potential target of the JNK pathway. J Cell Biol 170, (2005). 9. Raivich, G., et al. The AP-1 transcription factor c-jun is required for efficient axonal regeneration. Neuron 43, (2004). 10. Yoon, K.J., et al. Mind bomb 1-expressing intermediate progenitors generate notch signaling to maintain radial glial cells. Neuron 58, (2008). 11. Mizutani, K., Yoon, K., Dang, L., Tokunaga, A. & Gaiano, N. Differential Notch signalling distinguishes neural stem cells from intermediate progenitors. Nature 449, (2007). 12. Corbin, J.G., et al. Regulation of neural progenitor cell development in the nervous system. J Neurochem 106, (2008). 13. Yoon, K. & Gaiano, N. Notch signaling in the mammalian central nervous system: insights from mouse mutants. Nat Neurosci 8, (2005). 14. Tetzlaff, M.T., et al. Defective cardiovascular development and elevated cyclin E and Notch proteins in mice lacking the Fbw7 F-box protein. Proc Natl Acad Sci U S A 101, (2004). 15. Tsunematsu, R., et al. Mouse Fbw7/Sel-10/Cdc4 is required for notch degradation during vascular development. J Biol Chem 279, (2004). 16. Kramer, E.R., et al. Cooperation between GDNF/Ret and ephrina/epha4 signals for motor-axon pathway selection in the limb. Neuron 50, (2006). 17. Hartfuss, E., Galli, R., Heins, N. & Gotz, M. Characterization of CNS precursor subtypes and radial glia. Dev Biol 229, (2001). 18. Reynolds, B.A. & Rietze, R.L. Neural stem cells and neurospheres--re-evaluating the relationship. Nat Methods 2, (2005). 19. Nateri, A.S., Riera-Sans, L., Da Costa, C. & Behrens, A. The ubiquitin ligase SCFFbw7 antagonizes apoptotic JNK signaling. Science 303, (2004). 20. Bossy-Wetzel, E., Bakiri, L. & Yaniv, M. Induction of apoptosis by the transcription factor c-jun. EMBO J 16, (1997). 21. Whitfield, J., Neame, S.J., Paquet, L., Bernard, O. & Ham, J. Dominant-negative c-jun promotes neuronal survival by reducing BIM expression and inhibiting mitochondrial cytochrome c release. Neuron 29, (2001). 22. Ma, C., et al. dp5/hrk is a c-jun target gene and required for apoptosis induced by potassium deprivation in cerebellar granule neurons. J Biol Chem 282, (2007). 23. Angel, P., Hattori, K., Smeal, T. & Karin, M. The jun proto-oncogene is positively autoregulated by its product, Jun/AP1. Cell 55, (1988). 24. Gaiano, N., Nye, J.S. & Fishell, G. Radial glial identity is promoted by Notch1 signaling in the murine forebrain. Neuron 26, (2000). 25. Anthony, T.E., Mason, H.A., Gridley, T., Fishell, G. & Heintz, N. Brain lipid-binding protein is a direct target of Notch signaling in radial glial cells. Genes Dev 19, (2005). 26. Radtke, F., et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 10, (1999). 27. Selkoe, D. & Kopan, R. Notch and Presenilin: regulated intramembrane proteolysis links development and degeneration. Annu Rev Neurosci 26, (2003). 28. Anthony, T.E., Klein, C., Fishell, G. & Heintz, N. Radial glia serve as neuronal progenitors in all 31
33 regions of the central nervous system. Neuron 41, (2004). 29. Patten, B.A., Peyrin, J.M., Weinmaster, G. & Corfas, G. Sequential signaling through Notch1 and erbb receptors mediates radial glia differentiation. J Neurosci 23, (2003). 30. Coffey, E.T., et al. c-jun N-terminal protein kinase (JNK) 2/3 is specifically activated by stress, mediating c-jun activation, in the presence of constitutive JNK1 activity in cerebellar neurons. J Neurosci 22, (2002). 31. Wang, X., et al. Targeted deletion of the mitogen-activated protein kinase kinase 4 gene in the nervous system causes severe brain developmental defects and premature death. Mol Cell Biol 27, (2007). 32. Chang, L., Jones, Y., Ellisman, M.H., Goldstein, L.S. & Karin, M. JNK1 Is Required for Maintenance of Neuronal Microtubules and Controls Phosphorylation of Microtubule-Associated Proteins. Dev Cell 4, (2003). 33. Ohtsuka, T., et al. Hes1 and Hes5 as notch effectors in mammalian neuronal differentiation. EMBO J 18, (1999). 34. de la Pompa, J.L., et al. Conservation of the Notch signalling pathway in mammalian neurogenesis. Development 124, (1997). 35. Nelson, B.R., Hartman, B.H., Georgi, S.A., Lan, M.S. & Reh, T.A. Transient inactivation of Notch signaling synchronizes differentiation of neural progenitor cells. Dev Biol 304, (2007). 36. Hojo, M., et al. Glial cell fate specification modulated by the bhlh gene Hes5 in mouse retina. Development 127, (2000). 37. Thompson, B.J., et al. Control of hematopoietic stem cell quiescence by the E3 ubiquitin ligase Fbw7. J Exp Med 205, (2008). 38. Mao, J.H., et al. Fbxw7/Cdc4 is a p53-dependent, haploinsufficient tumour suppressor gene. Nature 432, (2004). 39. Welcker, M., Orian, A., Grim, J.E., Eisenman, R.N. & Clurman, B.E. A nucleolar isoform of the Fbw7 ubiquitin ligase regulates c-myc and cell size. Curr Biol 14, (2004). 40. Sangfelt, O., Cepeda, D., Malyukova, A., van Drogen, F. & Reed, S.I. Both SCF(Cdc4alpha) and SCF(Cdc4gamma) are required for cyclin E turnover in cell lines that do not overexpress cyclin E. Cell Cycle 7, (2008). 41. van Drogen, F., et al. Ubiquitylation of cyclin E requires the sequential function of SCF complexes containing distinct hcdc4 isoforms. Mol Cell 23, (2006). 42. Matsuoka, S., et al. Fbxw7 acts as a critical fail-safe against premature loss of hematopoietic stem cells and development of T-ALL. Genes Dev 22, (2008). 43. Pollard, S.M., Conti, L., Sun, Y., Goffredo, D. & Smith, A. Adherent neural stem (NS) cells from fetal and adult forebrain. Cereb Cortex 16 Suppl 1, i (2006). 44. Behrens, A., et al. Impaired intervertebral disc formation in the absence of Jun. Development 130, (2003). 45. Nateri, A.S., Spencer-Dene, B. & Behrens, A. Interaction of phosphorylated c-jun with TCF4 regulates intestinal cancer development. Nature 437, (2005). 32
34
35
36
37
38
39
Supplementary Figure 1
 Supplementary Figure 1 Kif1a RNAi effect on basal progenitor differentiation Related to Figure 2. Representative confocal images of the VZ and SVZ of rat cortices transfected at E16 with scrambled or Kif1a
Supplementary Figure 1 Kif1a RNAi effect on basal progenitor differentiation Related to Figure 2. Representative confocal images of the VZ and SVZ of rat cortices transfected at E16 with scrambled or Kif1a
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus
 Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus a: Expression of Vimentin, GFAP, Sox2 and Nestin in anterior, central and posterior hypothalamus. In the anterior
Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus a: Expression of Vimentin, GFAP, Sox2 and Nestin in anterior, central and posterior hypothalamus. In the anterior
Supplemental Information. Induction of Expansion and Folding. in Human Cerebral Organoids
 Cell Stem Cell, Volume 20 Supplemental Information Induction of Expansion and Folding in Human Cerebral Organoids Yun Li, Julien Muffat, Attya Omer, Irene Bosch, Madeline A. Lancaster, Mriganka Sur, Lee
Cell Stem Cell, Volume 20 Supplemental Information Induction of Expansion and Folding in Human Cerebral Organoids Yun Li, Julien Muffat, Attya Omer, Irene Bosch, Madeline A. Lancaster, Mriganka Sur, Lee
Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse
 Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
Neocortex Zbtb20 / NFIA / Sox9
 Neocortex / NFIA / Sox9 Supplementary Figure 1. Expression of, NFIA, and Sox9 in the mouse neocortex at. The lower panels are higher magnification views of the oxed area. Arrowheads indicate triple-positive
Neocortex / NFIA / Sox9 Supplementary Figure 1. Expression of, NFIA, and Sox9 in the mouse neocortex at. The lower panels are higher magnification views of the oxed area. Arrowheads indicate triple-positive
OSVZ progenitors of human and ferret neocortex are epithelial-like and
 OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling Simone A Fietz, Iva Kelava, Johannes Vogt, Michaela Wilsch-Bräuninger, Denise Stenzel, Jennifer L Fish,
OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling Simone A Fietz, Iva Kelava, Johannes Vogt, Michaela Wilsch-Bräuninger, Denise Stenzel, Jennifer L Fish,
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Cell Birth and Death. Chapter Three
 Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70%
 Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70% Pictures: 9 days after electroporation Red: MAP2 Blue: GFAP Green: GFP The cells were from Embryonic Day 14 Mouse Cerebral Cortex Primary Mouse Hippocampal
Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70% Pictures: 9 days after electroporation Red: MAP2 Blue: GFAP Green: GFP The cells were from Embryonic Day 14 Mouse Cerebral Cortex Primary Mouse Hippocampal
A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers
 A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers Bernard Carré To cite this version: Bernard Carré. A model for calculation
A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers Bernard Carré To cite this version: Bernard Carré. A model for calculation
SUPPLEMENTARY FIG. S2. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of dnscs.
 Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons
 Supplemental Material mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons Antoine de Chevigny, Nathalie Coré, Philipp Follert, Marion Gaudin, Pascal
Supplemental Material mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons Antoine de Chevigny, Nathalie Coré, Philipp Follert, Marion Gaudin, Pascal
TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer
 AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
ErbB4 migrazione I parte. 3- ErbB4- NRG1
 ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
a 0,8 Figure S1 8 h 12 h y = 0,036x + 0,2115 y = 0,0366x + 0,206 Labeling index Labeling index ctrl shrna Time (h) Time (h) ctrl shrna S G2 M G1
 (GFP+ BrdU+)/GFP+ Labeling index Labeling index Figure S a, b, y =,x +, y =,x +,,,,,,,, Time (h) - - Time (h) c d S G M G h M G S G M G S G h Time of BrdU injection after electroporation (h) M G S G M
(GFP+ BrdU+)/GFP+ Labeling index Labeling index Figure S a, b, y =,x +, y =,x +,,,,,,,, Time (h) - - Time (h) c d S G M G h M G S G M G S G h Time of BrdU injection after electroporation (h) M G S G M
Prss56, a novel marker of adult neurogenesis in the mouse brain. - Supplemental Figures 1 to 5- Brain Structure and Function
 Prss56, a novel marker of adult neurogenesis in the mouse brain - Supplemental Figures 1 to 5- Brain Structure and Function Alexandre Jourdon 1,2, Aurélie Gresset 1, Nathalie Spassky 1, Patrick Charnay
Prss56, a novel marker of adult neurogenesis in the mouse brain - Supplemental Figures 1 to 5- Brain Structure and Function Alexandre Jourdon 1,2, Aurélie Gresset 1, Nathalie Spassky 1, Patrick Charnay
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein
 Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Zhu et al, page 1. Supplementary Figures
 Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Journal Club. 03/04/2012 Lama Nazzal
 Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Information
 Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Cell Migration II: CNS Cell Migration. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
In vitro study of the effects of cadmium on the activation of the estrogen response element using the YES screen
 In vitro study of the effects of cadmium on the activation of the estrogen response element using the YES screen Xavier Denier, Jérome Couteau, Magalie Baudrimont, Elisabeth M. Hill, Jeanette Rotchell,
In vitro study of the effects of cadmium on the activation of the estrogen response element using the YES screen Xavier Denier, Jérome Couteau, Magalie Baudrimont, Elisabeth M. Hill, Jeanette Rotchell,
On the empirical status of the matching law : Comment on McDowell (2013)
 On the empirical status of the matching law : Comment on McDowell (2013) Pier-Olivier Caron To cite this version: Pier-Olivier Caron. On the empirical status of the matching law : Comment on McDowell (2013):
On the empirical status of the matching law : Comment on McDowell (2013) Pier-Olivier Caron To cite this version: Pier-Olivier Caron. On the empirical status of the matching law : Comment on McDowell (2013):
GFP/Iba1/GFAP. Brain. Liver. Kidney. Lung. Hoechst/Iba1/TLR9!
 Supplementary information a +KA Relative expression d! Tlr9 5!! 5! NSC Neuron Astrocyte Microglia! 5! Tlr7!!!! NSC Neuron Astrocyte! GFP/Sβ/! Iba/Hoechst Microglia e Hoechst/Iba/TLR9! GFP/Iba/GFAP f Brain
Supplementary information a +KA Relative expression d! Tlr9 5!! 5! NSC Neuron Astrocyte Microglia! 5! Tlr7!!!! NSC Neuron Astrocyte! GFP/Sβ/! Iba/Hoechst Microglia e Hoechst/Iba/TLR9! GFP/Iba/GFAP f Brain
Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were
 Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were electroporated with β- Catenin S33Y in PiggyBac expression
Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were electroporated with β- Catenin S33Y in PiggyBac expression
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
Sox9 is critical for suppression of neurogenesis but not initiation of gliogenesis in the cerebellum. Vong et al.
 Sox9 is critical for suppression of neurogenesis but not initiation of gliogenesis in the cerebellum Vong et al. Vong et al. Molecular Brain (2015) 8:25 DOI 10.1186/s13041-015-0115-0 Vong et al. Molecular
Sox9 is critical for suppression of neurogenesis but not initiation of gliogenesis in the cerebellum Vong et al. Vong et al. Molecular Brain (2015) 8:25 DOI 10.1186/s13041-015-0115-0 Vong et al. Molecular
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. The expression of ephrin-b2 H2BGFP persists in the post-hearingonset organ of Corti and is specifically restricted to supporting cells. Sox2 immunolabeling
SUPPLEMENTARY INFORMATION Supplementary Figure 1. The expression of ephrin-b2 H2BGFP persists in the post-hearingonset organ of Corti and is specifically restricted to supporting cells. Sox2 immunolabeling
s u p p l e m e n ta ry i n f o r m at i o n
 Figure S1 Characterization of tet-off inducible cell lines expressing GFPprogerin and GFP-wt lamin A. a, Western blot analysis of GFP-progerin- or GFP-wt lamin A- expressing cells before induction (0d)
Figure S1 Characterization of tet-off inducible cell lines expressing GFPprogerin and GFP-wt lamin A. a, Western blot analysis of GFP-progerin- or GFP-wt lamin A- expressing cells before induction (0d)
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
Bilateral anterior uveitis secondary to erlotinib
 Bilateral anterior uveitis secondary to erlotinib Lik Thai Lim, Robert Alexander Blum, Chee Peng Cheng, Abdul Hanifudin To cite this version: Lik Thai Lim, Robert Alexander Blum, Chee Peng Cheng, Abdul
Bilateral anterior uveitis secondary to erlotinib Lik Thai Lim, Robert Alexander Blum, Chee Peng Cheng, Abdul Hanifudin To cite this version: Lik Thai Lim, Robert Alexander Blum, Chee Peng Cheng, Abdul
Control of CNS Cell-Fate Decisions by SHP-2 and Its Dysregulation in Noonan Syndrome
 Article Control of CNS Cell-Fate Decisions by SHP-2 and Its Dysregulation in Noonan Syndrome Andrée S. Gauthier, 1,2,3 Olivia Furstoss, 1,2 Toshiyuki Araki, 5 Richard Chan, 5 Benjamin G. Neel, 5 David
Article Control of CNS Cell-Fate Decisions by SHP-2 and Its Dysregulation in Noonan Syndrome Andrée S. Gauthier, 1,2,3 Olivia Furstoss, 1,2 Toshiyuki Araki, 5 Richard Chan, 5 Benjamin G. Neel, 5 David
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary information. Nkx2.1 regulates the generation of telencephalic astrocytes during embryonic
 Supplementary information Nkx2.1 regulates the generation of telencephalic astrocytes during embryonic development Shilpi Minocha 1*, Delphine Valloton 1*, Yvan Arsenijevic 2, Jean-René Cardinaux 3, Raffaella
Supplementary information Nkx2.1 regulates the generation of telencephalic astrocytes during embryonic development Shilpi Minocha 1*, Delphine Valloton 1*, Yvan Arsenijevic 2, Jean-René Cardinaux 3, Raffaella
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
Supplementary Table 1. List of primers used in this study
 Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Information
 Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
(A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939 (5 µm)
 Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses Yichen Shi 1,2, Peter Kirwan 1,2, James Smith 1,2, Hugh P.C. Robinson 3 and Frederick
SUPPLEMENTARY INFORMATION Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses Yichen Shi 1,2, Peter Kirwan 1,2, James Smith 1,2, Hugh P.C. Robinson 3 and Frederick
Supplemental Information. Myocardial Polyploidization Creates a Barrier. to Heart Regeneration in Zebrafish
 Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Supplementary Information
 Supplementary Information Astrocytes regulate adult hippocampal neurogenesis through ephrin-b signaling Randolph S. Ashton, Anthony Conway, Chinmay Pangarkar, Jamie Bergen, Kwang-Il Lim, Priya Shah, Mina
Supplementary Information Astrocytes regulate adult hippocampal neurogenesis through ephrin-b signaling Randolph S. Ashton, Anthony Conway, Chinmay Pangarkar, Jamie Bergen, Kwang-Il Lim, Priya Shah, Mina
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 EGFR inhibition activates signaling pathways (a-b) EGFR inhibition activates signaling pathways (a) U251EGFR cells were treated with erlotinib (1µM) for the indicated times followed
Supplementary Figure 1 EGFR inhibition activates signaling pathways (a-b) EGFR inhibition activates signaling pathways (a) U251EGFR cells were treated with erlotinib (1µM) for the indicated times followed
Supplementary Figure 1. Characterization of ALDH-positive cell population in MCF-7 cells. (a) Expression level of stem cell markers in MCF-7 cells or
 Supplementary Figure 1. Characterization of ALDH-positive cell population in MCF-7 cells. (a) Expression level of stem cell markers in MCF-7 cells or ALDH-positive cell population by qpcr. Data represent
Supplementary Figure 1. Characterization of ALDH-positive cell population in MCF-7 cells. (a) Expression level of stem cell markers in MCF-7 cells or ALDH-positive cell population by qpcr. Data represent
Virtual imaging for teaching cardiac embryology.
 Virtual imaging for teaching cardiac embryology. Jean-Marc Schleich, Jean-Louis Dillenseger To cite this version: Jean-Marc Schleich, Jean-Louis Dillenseger. Virtual imaging for teaching cardiac embryology..
Virtual imaging for teaching cardiac embryology. Jean-Marc Schleich, Jean-Louis Dillenseger To cite this version: Jean-Marc Schleich, Jean-Louis Dillenseger. Virtual imaging for teaching cardiac embryology..
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses
 Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses using an anti-cre antibody; testes at 1 week (left panel),
Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses using an anti-cre antibody; testes at 1 week (left panel),
ErbB4 migrazione II parte
 ErbB4 migrazione II parte Control SVZ cells prefer to migrate on the NRG1 type III substrate the substrate preference of the neuroblasts migrating out of the SVZ explant was evaluated SVZ cells had a strong
ErbB4 migrazione II parte Control SVZ cells prefer to migrate on the NRG1 type III substrate the substrate preference of the neuroblasts migrating out of the SVZ explant was evaluated SVZ cells had a strong
Daily alternating deferasirox and deferiprone therapy for hard-to-chelate β-thalassemia major patients
 Daily alternating deferasirox and deferiprone therapy for hard-to-chelate β-thalassemia major patients Manuela Balocco, Paola Carrara, Valeria Pinto, Gian Luca Forni To cite this version: Manuela Balocco,
Daily alternating deferasirox and deferiprone therapy for hard-to-chelate β-thalassemia major patients Manuela Balocco, Paola Carrara, Valeria Pinto, Gian Luca Forni To cite this version: Manuela Balocco,
From universal postoperative pain recommendations to procedure-specific pain management
 From universal postoperative pain recommendations to procedure-specific pain management Hélène Beloeil, Francis Bonnet To cite this version: Hélène Beloeil, Francis Bonnet. From universal postoperative
From universal postoperative pain recommendations to procedure-specific pain management Hélène Beloeil, Francis Bonnet To cite this version: Hélène Beloeil, Francis Bonnet. From universal postoperative
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events
 Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
ANAT3231: lectures overview
 ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy
 Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Reporting physical parameters in soundscape studies
 Reporting physical parameters in soundscape studies Truls Gjestland To cite this version: Truls Gjestland. Reporting physical parameters in soundscape studies. Société Française d Acoustique. Acoustics
Reporting physical parameters in soundscape studies Truls Gjestland To cite this version: Truls Gjestland. Reporting physical parameters in soundscape studies. Société Française d Acoustique. Acoustics
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software
 Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES 1 2 3 4 SUPPLEMENTARY TABLES Supplementary Table S1. Brain Tumors used in the study Code Tumor Classification Age Gender HuTuP51 Glioblastoma 57 Male HuTuP52 Glioblastoma 53 Male
SUPPLEMENTARY FIGURES 1 2 3 4 SUPPLEMENTARY TABLES Supplementary Table S1. Brain Tumors used in the study Code Tumor Classification Age Gender HuTuP51 Glioblastoma 57 Male HuTuP52 Glioblastoma 53 Male
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH
 EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH Supplementary Figure 1. Supplementary Figure 1. Characterization of KP and KPH2 autochthonous UPS tumors. a) Genotyping of KPH2
EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH Supplementary Figure 1. Supplementary Figure 1. Characterization of KP and KPH2 autochthonous UPS tumors. a) Genotyping of KPH2
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
293T cells were transfected with indicated expression vectors and the whole-cell extracts were subjected
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
SUPPLEMENTARY LEGENDS...
 TABLE OF CONTENTS SUPPLEMENTARY LEGENDS... 2 11 MOVIE S1... 2 FIGURE S1 LEGEND... 3 FIGURE S2 LEGEND... 4 FIGURE S3 LEGEND... 5 FIGURE S4 LEGEND... 6 FIGURE S5 LEGEND... 7 FIGURE S6 LEGEND... 8 FIGURE
TABLE OF CONTENTS SUPPLEMENTARY LEGENDS... 2 11 MOVIE S1... 2 FIGURE S1 LEGEND... 3 FIGURE S2 LEGEND... 4 FIGURE S3 LEGEND... 5 FIGURE S4 LEGEND... 6 FIGURE S5 LEGEND... 7 FIGURE S6 LEGEND... 8 FIGURE
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were
 Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
A new role for meninges as a niche for stem/precursor cells with neural differentiation potential during development up to adulthood
 A new role for meninges as a niche for stem/precursor cells with neural differentiation potential during development up to adulthood Francesco Bifari, MD, PhD Mauro Krampera, MD, PhD Ilaria Decimo, PhD
A new role for meninges as a niche for stem/precursor cells with neural differentiation potential during development up to adulthood Francesco Bifari, MD, PhD Mauro Krampera, MD, PhD Ilaria Decimo, PhD
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Notch pathway molecules are essential for the maintenance, but not the generation, of mammalian neural stem cells
 Notch pathway molecules are essential for the maintenance, but not the generation, of mammalian neural stem cells Seiji Hitoshi, 1,6,9 Tania Alexson, 1 Vincent Tropepe, 1,7 Dorit Donoviel, 2,8 Andrew J.
Notch pathway molecules are essential for the maintenance, but not the generation, of mammalian neural stem cells Seiji Hitoshi, 1,6,9 Tania Alexson, 1 Vincent Tropepe, 1,7 Dorit Donoviel, 2,8 Andrew J.
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
SUPPLEMENTARY INFORMATION
 Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
Automatic spread of attentional response modulation along Gestalt criteria in primary visual cortex
 Automatic spread of attentional response modulation along Gestalt criteria in primary visual cortex Aurel Wannig, Liviu Stanisor, Pieter R. Roelfsema To cite this version: Aurel Wannig, Liviu Stanisor,
Automatic spread of attentional response modulation along Gestalt criteria in primary visual cortex Aurel Wannig, Liviu Stanisor, Pieter R. Roelfsema To cite this version: Aurel Wannig, Liviu Stanisor,
Optimal electrode diameter in relation to volume of the cochlea
 Optimal electrode diameter in relation to volume of the cochlea Dan Gnansia, Thomas Demarcy, Clair Vandersteen, Charles Raffaelli, Nicolas Guevara, Hervé Delingette, Nicholas Ayache To cite this version:
Optimal electrode diameter in relation to volume of the cochlea Dan Gnansia, Thomas Demarcy, Clair Vandersteen, Charles Raffaelli, Nicolas Guevara, Hervé Delingette, Nicholas Ayache To cite this version:
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ).
 Supplementary Figure 1 Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ). (b) Immunoblot analysis of TRIM29 in lung primary
Supplementary Figure 1 Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ). (b) Immunoblot analysis of TRIM29 in lung primary
Nature Neuroscience: doi: /nn.2275
 Supplementary Figure S1. The presence of MeCP2 in enriched primary glial cultures from rat or mouse brains is not neuronal. Western blot analysis of protein extracts from (a) rat glial and neuronal cultures.
Supplementary Figure S1. The presence of MeCP2 in enriched primary glial cultures from rat or mouse brains is not neuronal. Western blot analysis of protein extracts from (a) rat glial and neuronal cultures.
Hematopoiesis. - Process of generation of mature blood cells. - Daily turnover of blood cells (70 kg human)
 Hematopoiesis - Process of generation of mature blood cells - Daily turnover of blood cells (70 kg human) 1,000,000,000,000 total cells 200,000,000,000 red blood cells 70,000,000,000 neutrophils Hematopoiesis
Hematopoiesis - Process of generation of mature blood cells - Daily turnover of blood cells (70 kg human) 1,000,000,000,000 total cells 200,000,000,000 red blood cells 70,000,000,000 neutrophils Hematopoiesis
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
BIO360 Fall 2013 Quiz 1
 BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
Supplementary fig. 1. Crystals induce necroptosis does not involve caspases, TNF receptor or NLRP3. A. Mouse tubular epithelial cells were pretreated
 Supplementary fig. 1. Crystals induce necroptosis does not involve caspases, TNF receptor or NLRP3. A. Mouse tubular epithelial cells were pretreated with zvad-fmk (10µM) and exposed to calcium oxalate
Supplementary fig. 1. Crystals induce necroptosis does not involve caspases, TNF receptor or NLRP3. A. Mouse tubular epithelial cells were pretreated with zvad-fmk (10µM) and exposed to calcium oxalate
