The sarcoendoplasmic reticulum Ca
|
|
|
- Barry Evans
- 5 years ago
- Views:
Transcription
1 SERCA3 Ablation Does Not Impair Insulin Secretion but Suggests Distinct Roles of Different Sarcoendoplasmic Reticulum Ca 2 Pumps for Ca 2 Homeostasis in Pancreatic -cells Abdelilah Arredouani, 1 Yves Guiot, 2 Jean-Christophe Jonas, 1 Lynne H. Liu, 3 Myriam Nenquin, 1 José A. Pertusa, 1 Jacques Rahier, 2 Jean-François Rolland, 1 Gary E. Shull, 3 Martine Stevens, 2 Frank Wuytack, 4 Jean-Claude Henquin, 1 and Patrick Gilon 1 Two sarcoendoplasmic reticulum Ca 2 -ATPases, SERCA3 and SERCA2b, are expressed in pancreatic islets. Immunocytochemistry showed that SERCA3 is restricted to -cells in the mouse pancreas. Control and SERCA3-deficient mice were used to evaluate the role of SERCA3 in -cell cytosolic-free Ca 2 concentration ([Ca 2 ] c ) regulation, insulin secretion, and glucose homeostasis. Basal [Ca 2 ] c was not increased by SERCA3 ablation. Stimulation with glucose induced a transient drop in basal [Ca 2 ] c that was suppressed by inhibition of all SERCAs with thapsigargin (TG) but unaffected by selective SERCA3 ablation. Ca 2 mobilization by acetylcholine was normal in SERCA3-deficient -cells. In contrast, [Ca 2 ] c oscillations resulting from intermittent glucose-stimulated Ca 2 influx and [Ca 2 ] c transients induced by pulses of high K were similarly affected by SERCA3 ablation or TG pretreatment of control islets; their amplitude was increased and their slow descending phase suppressed. This suggests that, during the decay of each oscillation, the endoplasmic reticulum releases Ca 2 that was pumped by SERCA3 during the upstroke phase. SERCA3 ablation increased the insulin response of islets to 15 mmol/l glucose. However, basal and postprandial plasma glucose and insulin concentrations in SERCA3-deficient mice were normal. In conclusion, SERCA2b, but not SERCA3, is involved in basal [Ca 2 ] c regulation in -cells. SERCA3 becomes operative when [Ca 2 ] c rises and is required for normal [Ca 2 ] c oscillations in response to glucose. However, a lack of SERCA3 is insufficient in itself to alter glucose homeostasis or impair insulin secretion in mice. Diabetes 51: , 2002 From the 1 Unité d Endocrinologie et Métabolisme, University of Louvain Faculty of Medicine, Brussels, Belgium; the 2 Unité d Anatomie Pathologique Spéciale, University of Louvain Faculty of Medicine, Brussels, Belgium; the 3 Department of Molecular Genetics, University of Cincinnati College of Medicine, Cincinnati, Ohio; and the 4 Laboratorium voor Fysiologie, Katholieke Universiteit Leuven, Leuven, Belgium. Address correspondence and reprint requests to Dr. P. Gilon, Unité d Endocrinologie et Métabolisme, University of Louvain Faculty of Medicine, UCL 55.30, Av. Hippocrate 55, B-1200 Brussels, Belgium. gilon@endo. ucl.ac.be. Received for publication 8 February 2002 and accepted in revised form 26 July ACh, acetylcholine; [Ca 2 ] c, cytosolic-free Ca 2 concentration; ER, endoplasmic reticulum; IP 3, Ins(1,4,5)P 3 ; SERCA, sarcoendoplasmic reticulum Ca 2 -ATPase; TG, thapsigargin. The sarcoendoplasmic reticulum Ca 2 -ATPase (SERCA) transfers Ca 2 from the cytosol to the lumen of the reticulum. Three SERCA genes (SERCA1 3) have been identified, all of which can generate several alternatively spliced isoforms (1 4). Pancreatic islets express SERCA2b and the three isoforms of SERCA3 (a c) (1,5). SERCA2b is nearly ubiquitous and considered essential for viability (6 8). SERCA3 has a restricted tissue distribution (4,9,10) and shows lower Ca 2 affinity, higher sensitivity to vanadate, and higher ph optimum than SERCA1 or SERCA2 (11). The evidence for distinct physiological roles of the various SERCAs in cells is scant (3,12,13) and difficult to obtain because it requires experiments with genetically modified mouse or cell models or analysis of spontaneously occurring genetic diseases (8). Glucose-stimulated -cells display oscillations of the membrane potential that cause intermittent openings of voltage-dependent Ca 2 channels (14) and induce synchronous cytosolic-free Ca 2 concentration ([Ca 2 ] c ) oscillations (15,16). The endoplasmic reticulum (ER) plays complex roles in -cell Ca 2 homeostasis. Upon stimulation by glucose, it sequesters Ca 2 (17 19), leading to a lowering of [Ca 2 ] c if Ca 2 influx is prevented. Upon stimulation by phospholipase C activating agonists, such as acetylcholine (ACh) (17,20), it massively releases Ca 2. During [Ca 2 ] c oscillations induced by periodic influx of Ca 2, such as those triggered by glucose, the ER takes up Ca 2 during the upstroke of the [Ca 2 ] c rise and releases it slowly when Ca 2 influx stops (21). An impaired [Ca 2 ] c response to glucose is observed in -cells of animal models of type 2 diabetes (22,23). Several studies have suggested that this dysregulation results from an impaired control of [Ca 2 ] c by the SERCAs, in particular SERCA3. Thus, a decreased Ca 2 -ATPase activity has been reported in islets of rats made diabetic by neonatal injection of streptozotocin (24). The activity of all SERCAs was also found to be reduced in spontaneously diabetic db/db mice (25). Other studies have suggested that a deficiency of SERCA3 specifically might be involved. Indeed, a major decrease of the expression of SERCA3 but not of SERCA2b was observed in Goto-Kakizaki rats (5) DIABETES, VOL. 51, NOVEMBER
2 SERCA3 AND [Ca 2 ] c IN PANCREATIC -CELLS and in 90% pancreatectomized rats (26). Several missense mutations of SERCA3 have been detected in type 2 diabetic patients (27). Overexpression of insulin receptor substrate-1, which occurs after prolonged exposure of TC6 cells to insulin (a phenomenon that might occur in hyperinsulinemic patients), inhibited SERCA3 gene expression without affecting that of SERCA2b (28). All of these data therefore suggest that a decreased expression or activity of SERCA3 could contribute to the impaired -cell [Ca 2 ] c and insulin responses observed in type 2 diabetes. In the present study, we studied by immunocytochemistry the specific cell localization of SERCA3 in islets. Then, we addressed the role of SERCA3 in -cell Ca 2 signaling by using the recently generated SERCA3 knockout mice (SERCA3 / ) (29) and their wild-type littermates (SERCA3 / ). The [Ca 2 ] c response to various agents was compared in -cells expressing SERCA2b and SERCA3 (SERCA3 / mice), in cells expressing only SERCA2b (SERCA3 / mice), and in cells treated by thapsigargin (TG) to block all SERCA activity (30). RESEARCH DESIGN AND METHODS Immunodetection of SERCA3 and islet hormones. The pancreas of adult NMRI mice was fixed with 4% paraformaldehyde and embedded in paraffin. Immunodetection (peroxidase-diaminobenzidine technique) of insulin or SERCA3 was performed on serial sections, pretreated (SERCA3) or not (insulin) with microwaves, using a monoclonal anti-insulin antiserum (Novo Biolabs, Temecula, Denmark) or the rabbit anti-serca3 N89 antibody directed against a sequence corresponding to amino acid of the human SERCA3 (31). For double-fluorescent immunodetections of non -cells and SERCA3, the sections were incubated with a cocktail of monoclonal antiglucagon (Novo Biolabs), monoclonal anti-somatostatin (Novo Biolabs), and rabbit anti-serca3 antisera. Detection of SERCA3 by a red immunofluorescent color (Texas Red) required a tyramine amplification step, as described elsewhere (32). Non -cells were immunolabeled in green fluorescent color (fluorescein isothiocyanate). Control experiments without primary antibody never showed any labeling. Animals, solutions, and preparations. The SERCA3 gene contains 22 exons. SERCA3 knockout mice (SERCA3 / ) were generated previously by disrupting exon 5 (29). Age- (4 8 months) and sex-matched homozygous wild-type (SERCA3 / ) and SERCA3 knockout mice (SERCA3 / ) were used. The mutant mice (25 1 g) were indistinguishable from their wild-type littermates (26 1 g) in their gross phenotype and behavior. To evaluate glucose homeostasis of the mice, blood glucose was measured (glucometer; Bayer, Zurich) after 24 h of food deprivation and again 1 and 3 h after ad libitum refeeding. One week later, the same animals were fasted again, and blood glucose was measured after 24, 25, and 27 h of fasting. Plasma insulin was measured as described (33). The medium used for the isolation of pancreatic islets and for [Ca 2 ] c and insulin secretion experiments was a bicarbonate-buffered solution (21). When the concentration of KCl was increased, that of NaCl was decreased accordingly. Ca 2 -free solutions were prepared by substituting MgCl 2 for CaCl 2 and were supplemented with 2 mmol/l EGTA. Islets were obtained by collagenase digestion of the pancreas, and clusters were prepared by dispersion of islet cells in a Ca 2 -free medium (34). Islets were cultered for 1 or 2 days, and cell clusters for 2 days in RPMI 1640 medium containing 10 mmol/l glucose. Semiquantitative radioactive RT-PCR analysis of SERCA2 and SERCA3 mrna. Islet total RNA was extracted, quantified, and reverse transcribed in cdna as described (26). The sense (5 -GTC ATC ATG CTA ATC CTT GTG GC-3 ) and antisense (5 -GGG GGT CTG GAA TGG CA-3 ) primers for amplification of mouse SERCA3 mrna (nucleotides of the cdna, Genbank accession nos. U49393 and U49394) were chosen on opposite sides of the 84-bp region deleted in SERCA3 / mice (29), giving products of 316 and 232 bp in wild-type and knockout mice, respectively. The sense (5 -GGC TTT TAC AGG GCG AGA GT-3 ) and antisense (5 -ACC AGA TTG ACC CAG AGT AAC TG-3 ) primers for mouse SERCA2 were chosen in a region common to both a and b isoforms (from nucleotides 531 in exon 2 to nucleotide 949 in exon 4 of SERCA2 mrna, Genbank accession no. AJ131870), giving rise to a product of 441 bp. SERCA3 cdna was amplified in duplex with cyclophilin as internal control gene (for primers, see ref. 26). Semiquantitative radioactive PCRs were performed as previously described (26), except that the thermal cycle profile was a 10-min denaturing step at 94 C followed by 22 cycles (1-min steps at 94 C, 60 C, and 72 C each) and a final extension step of 10 min at 72 C. Measurements of [Ca 2 ] c and insulin secretion. [Ca 2 ] c was measured at 37 C in cultured islets or clusters of cells loaded with fura-pe3/am (Teflabs, Austin, TX) as previously described (21). When needed, 1 mol/l TG was added to the loading solution. Insulin secretion was measured in a perifusion system using batches of 15 islets (35). Presentation of the results. The experiments are illustrated by traces that are means SE or by representative traces of results obtained with the indicated number of islets or clusters of islet cells from at least three different cultures, or with the indicated number of mice. The statistical significance of differences between means was assessed by unpaired or paired Student s t test as appropriate. Differences between means were considered significant at P RESULTS Immunolocalization of SERCA3. Immunodetection of SERCA3 revealed a strong labeling in islets, but no staining in the exocrine pancreas (Fig. 1A). The close similarity of the immunostaining pattern of serial sections for SERCA3 (Fig. 1A) and insulin (Fig. 1B) indicated that SERCA3 is present in -cells. A minority of islet cells, often localized at the periphery and hence thought to be non -cells, did not display SERCA3 immunoreactivity (arrows in Fig. 1A and B). This was confirmed by double immunodetection of non -cells and SERCA3, which unequivocally revealed that glucagon- or somatostatinsecreting cells were not immunoreactive for SERCA3 (Fig. 1C H). SERCA3 expression is thus limited to -cells. RT-PCR experiments. The expression of SERCA3 and SERCA2 in islet cells was assessed by RT-PCR on total RNA extracted from SERCA3 / and SERCA3 / islets (Fig. 2). As expected, the size of SERCA3 amplicon from SERCA3 / mice was smaller than that of SERCA3 / mice (Fig. 2B). This difference in size results from the excision of the last 84 nucleotides in exon 5, which impairs the pump activity of SERCA3 (29). Under semiquantitative PCR conditions, the levels of the mutated SERCA3 mrna of SERCA3 / islets were much lower than those of the wild-type SERCA3 mrna of SERCA3 / islets (Fig. 2A). However, the expression level of SERCA2 mrna (presumably SERCA2b) relative to the control mrna cyclophilin in SERCA3 / islets was 98.7% of that observed in SERCA3 / islets (n 2). This suggests that the absence of SERCA3 is not compensated for by an overexpression of SERCA2 in islets. Evaluation of the role of SERCA3 at basal [Ca 2 ] c. During perifusion of the islets with a glucose-free medium or a medium containing glucose and the ATP-sensitive K channel opener diazoxide, [Ca 2 ] c was low and stable. There was no significant difference between basal [Ca 2 ] c in SERCA3 / and SERCA3 / islets in all experimental series (pooled data of G0, G3, G0 plus diazoxide, G10 plus diazoxide, and G20 plus diazoxide: 99 3 nmol/l, n 38, in SERCA3 / islets vs nmol/l, n 38, in SERCA3 / islets). Pretreatment of the islets with TG did not increase basal [Ca 2 ] c in SERCA3 / (106 7 nmol/l, n 8) and SERCA3 / islets (78 6 nmol/l, n 7). This suggests that basal [Ca 2 ] c is mainly set by mechanisms other than the SERCAs. We next evaluated the role of SERCA3 in glucosestimulated Ca 2 uptake by the ER. Switching from a 3246 DIABETES, VOL. 51, NOVEMBER 2002
3 A. ARREDOUANI AND ASSOCIATES FIG. 1. Immunolocalization of SERCA3 in the mouse pancreas. A and B: Immunodetection of SERCA3 (A) and insulin (B) on serial sections suggested that SERCA3 and insulin immunoreactivity have a similar localization within the islet. Some peripheral islet cells (arrows) and the exocrine tissue were not stained. (C H) Double immunodetections of SERCA3 in red and glucagon plus somatostatin in green (non -cells) on the same section. Red and green colors were digitally separated in C, D, F, and G, whereas they were digitally combined in E and H. InE and H, a yellow color was seen in restricted areas, where the cytoplasms of red- and green-labeled cells slightly overlapped due to the thickness of the section. However, no overlapping of the two colors was ever observed in whole cells, indicating that non -cells of the islet do not express SERCA3. Original magnifications: A and B: 150; C E: 500; F H: 1,000. glucose-free medium to a medium containing 15 mmol/l glucose induced an initial decrease in [Ca 2 ] c that preceded a [Ca 2 ] c rise in SERCA3 / islets (Fig. 3A). This drop is attributed to Ca 2 uptake by the ER because it was completely prevented by TG. Glucose also transiently lowered [Ca 2 ] c in SERCA3 / islets (Fig. 3B). The amplitude of this initial fall was similar in SERCA3 / (19 2 nmol/l, n 9) and SERCA3 / islets (15 1 nmol/l, n 9). Similar observations were made when the experiments were performed in the presence of diazoxide (not shown). The ER comprises Ins(1,4,5)P 3 (IP 3 )-sensitive and -insensitive Ca 2 stores that are both refilled by SERCAs during exposure to glucose (19,36,37). ACh mobilizes Ca 2 from IP 3 -sensitive stores, whereas TG mobilizes Ca 2 from both stores (19,36). To evaluate the role of SERCA3 in the replenishment of these Ca 2 stores and the maintenance of their Ca 2 content at basal [Ca 2 ] c, islets and clusters of cells were initially perifused with a medium containing Ca 2, diazoxide, and 10 or 20 mmol/l glucose, which maximally stimulates Ca 2 uptake by the ER (19,38). They were then perifused for 5 (islets) or 8 min (clusters of cells) with a Ca 2 -free medium supplemented with 2 mmol/l EGTA to prevent Ca 2 influx. Subsequent addition of ACh triggered a large transient rise in [Ca 2 ] c in islets (Fig. 4A) and clusters (Fig. 4B). In clusters, addition of TG 8 min after removal of ACh induced a small [Ca 2 ] c elevation that reflects release of residual Ca 2, not mobi- DIABETES, VOL. 51, NOVEMBER
4 SERCA3 AND [Ca 2 ] c IN PANCREATIC -CELLS FIG. 2. Comparison of SERCA2 and SERCA3 mrna levels by RT-PCR in isolated islets from SERCA3 / (WT) and SERCA3 / (KO) mice. A: After reverse transcription (RT ) of islet total RNA, SERCA3 and SERCA2 cdnas were amplified for 22 cycles under semiquantitative conditions. Cyclophilin was used as an internal control gene and amplified in duplex with SERCA3. SERCA2 mrna expression was not affected by SERCA3 ablation. Compared with the 316-bp product in islets from wild-type mice, the shorter SERCA3 amplicon in SERCA3 / islets only gave a faint band under these conditions. B: SERCA3 cdna was amplified for 40 cycles to confirm the presence and size of the shorter SERCA3 amplicon in islets from knockout mice. No PCR products were amplified when reverse transcriptase was omitted from the reverse transcription reaction (RT ). The results of both panels are representative of experiments performed on islets from two mice. lized by ACh (right side of Fig. 4B). The magnitude of the [Ca 2 ] c rises induced by ACh and TG was similar in SERCA3 / and SERCA3 / islet cells. The effect of 100 mol/l ACh on [Ca 2 ] c was also tested in islets perifused with a medium containing Ca 2, 20 mmol/l glucose, and 250 mol/l diazoxide. The amplitude of the mobilization of Ca 2 was again similar in SERCA3 / ( nmol/l, n 3) and SERCA3 / islets ( nmol/l, n 3). These results suggest that the IP 3 -sensitive and -insensitive Ca 2 pools of the ER do not require FIG. 3. SERCA3 does not mediate the initial [Ca 2 ] c decrease induced by glucose. Isolated islets from SERCA3 / (A) or SERCA3 / (B) mice were pretreated (dotted line) or not (solid line) with 1 mol/l TG during loading with fura-pe3. After an initial perifusion without glucose (G0), they were perifused with a medium containing 15 mmol/l glucose (G15), as indicated on the top of the panels. The traces show mean changes ( ) around basal [Ca 2 ] c in the absence of glucose. Basal levels were nmol/l, n 9(A, solid line); nmol/l, n 3(A, dotted line); 80 7 nmol/l, n 9(B, solid line); and nmol/l, n 3(B, dotted line). FIG. 4. SERCA3 is not required for refilling IP 3 -sensitive and -insensitive Ca 2 pools in mouse islets. Before the recording, islets (A) or clusters of islet cells (B) were bathed in a medium containing 2.5 mmol/l Ca 2, 100 mol/l diazoxide (Dz), and 10 (G10) or 20 mmol/l glucose (G20), respectively. They were then perifused with a Ca 2 -free medium supplemented with 2 mmol/l EGTA for 5 (islets) or 8 min (clusters of cells) before addition of 100 mol/l ACh. In clusters only, 1 mol/l TG was added as indicated. The traces show mean changes ( ) around basal [Ca 2 ] c levels at the beginning of the recording. Basal levels were nmol/l, n 5(A, solid line); 98 7 nmol/l, n 6(A, dotted line); 41 6 nmol/l, n 4(B, solid line); and 36 9 nmol/l, n 4(B, dotted line). SERCA3 activity to be filled with Ca 2 during glucose stimulation and to retain Ca 2 when [Ca 2 ] c is low. Role of SERCA3 at elevated [Ca 2 ] c. The role of SERCA3 was next evaluated under conditions where [Ca 2 ] c was transiently elevated by depolarizing the plasma membrane with 45 mmol/l K to stimulate Ca 2 influx through voltage-dependent Ca 2 channels. Diazoxide was present throughout to prevent glucose-induced [Ca 2 ] c oscillations (15) and to impose similar membrane potentials in the presence and absence of TG (21). Depolarizing SERCA3 / islets for 30 s induced a large and rapid [Ca 2 ] c rise followed by a slightly ascending plateau and a descending phase characterized by an initial fast component and a secondary slow decline (Fig. 5A, solid line). As previously reported (21), TG pretreatment increased the rising phase to the peak (P 0.05), gave the plateau a slowly descending shape, and suppressed the secondary slow component of the recovery phase (Fig. 5A, dotted line). These changes have been interpreted as evidence that the ER takes up Ca 2 during the upstroke of the [Ca 2 ] c rise and releases it slowly when Ca 2 influx stops (21). Strikingly, the [Ca 2 ] c changes induced by the high K pulse in SERCA3 / islets had a similar shape to that observed in SERCA3 / islets after TG pretreatment (compare Fig. 5A dotted line with Fig. 5B dotted line). In contrast, TG pretreatment of SERCA3 / islets did not 3248 DIABETES, VOL. 51, NOVEMBER 2002
5 A. ARREDOUANI AND ASSOCIATES FIG. 5. SERCA3 influences [Ca 2 ] c changes induced by a 30-s pulse of 45 mmol/l K in mouse islets. Isolated islets from SERCA3 / or SERCA3 / mice were pretreated or not with 1 mol/l TG during loading with fura-pe3. They were then perifused throughout with a medium containing 10 mmol/l glucose (G10) and 100 mol/l diazoxide (Dz). The K concentration was increased from 4.8 to 45 mmol/l when indicated by the horizontal bar. The traces show mean changes ( ) around basal [Ca 2 ] c levels before the pulse of high K. Basal levels were nmol/l, n 7(A and B, solid line); nmol/l, n 6 (B, dotted line and C, solid line); nmol/l, n 5(A, dotted line); and 84 7 nmol/l, n 4(C, dotted line). *P < 0.05 for comparisons of the mean amplitude of the high K -induced elevation in [Ca 2 ] c between groups shown by dotted and solid lines. significantly change the characteristics of the [Ca 2 ] c rise and decrease induced by the K pulse (Fig. 5C). These observations indicate that the difference (P 0.05) in [Ca 2 ] c peak amplitude between SERCA3 / and SERCA3 / islets, illustrated in Fig. 5B, is due to a buffering action of the ER that is mainly mediated by SERCA3 activity. They also show that a major component of the slow recovery phase observed in SERCA3 / islets corresponds to the release of Ca 2 from ER Ca 2 pools that were replenished by SERCA3. A small contribution of SERCA2b is suggested by the minor residual slow recovery [Ca 2 ] c phase in SERCA3 / islets (Fig. 5C). Influence of SERCA3 on glucose-induced [Ca 2 ] c oscillations. Increasing the glucose concentration from 3 to 7 mmol/l triggered a transient rise in [Ca 2 ] c in all islets. Thereafter, [Ca 2 ] c started to oscillate in 12/14 SERCA3 / and 7/13 SERCA3 / islets and returned to basal levels in the others (Fig. 6A and B). In 10 mmol/l glucose, [Ca 2 ] c oscillations occurred in all islets. In 20 mmol/l glucose, [Ca 2 ] c was steadily elevated in 12/14 SERCA3 / and 9/13 SERCA3 / islets, whereas it continued to oscillate in the others. Average [Ca 2 ] c, integrated over the last 5 min at each glucose concentration, was similar in both types of islets (Fig. 6C). However, several features of [Ca 2 ] c oscillations were different. The nadir between oscillations was more elevated in SERCA3 / than SERCA3 / islets, in both 7 mmol/l glucose (120 4 nmol/l, n 12 vs nmol/l, n 7) and 10 mmol/l glucose (156 5 nmol/l, n 14 vs nmol/l, n 13). Therefore, the difference between nadir and basal [Ca 2 ] c at 3 mmol/l glucose was significantly larger in SERCA3 / than SERCA3 / islets ( 1 and 2 in Fig. 6A, B, and D). In contrast, when 20 mmol/l glucose induced a sustained elevation of [Ca 2 ] c, there was no difference between SERCA3 / (233 8 nmol/l, n 12) and SERCA3 / islets ( nmol/l, n 9); therefore, 3 with basal [Ca 2 ] c was similar (141 7 vs nmol/l). The differences between nadirs reflect differences in the amplitude of [Ca 2 ] c oscillations, which was smaller in SERCA3 / than SERCA3 / islets (Fig. 6E). The frequency of [Ca 2 ] c oscillations was similar in the two types of islets during the last 5 min of perifusion with 7 FIG. 6. SERCA3 influences glucose-induced [Ca 2 ] c oscillations in mouse islets. A and B: Isolated islets from SERCA3 / or SERCA3 / mice were perifused with a medium containing 3, 7, 10, and 20 mmol/l glucose (G) as indicated. shows the difference between basal [Ca 2 ] c at 3 mmol/l glucose and the nadir of [Ca 2 ] c oscillations at 7 ( 1) or 10 mmol/l glucose ( 2) or the sustained [Ca 2 ] c elevation at 20 mmol/l glucose ( 3). The traces are representative of results obtained in six (A) and five (B) islets. C: Average [Ca 2 ] c calculated by integrating [Ca 2 ] c over the last 5 min of stimulation with each glucose concentration in all experiments similar to those illustrated in A and B. D: Difference between basal [Ca 2 ] c at 3 mmol/l glucose and the nadir of [Ca 2 ] c oscillations at 7 ( 1) or 10 mmol/l glucose ( 2). Amplitude (E) and frequency (F) of[ca 2 ] c oscillations calculated during the last 5 min of perifusion with 7 or 10 mmol/l glucose. C F: Data are means SE. *P < 0.05, ** P < 0.01 for differences between SERCA3 / and SERCA3 / islets. mmol/l glucose but was much higher in SERCA3 / than SERCA3 / islets in 10 mmol/l glucose (Fig. 6F). The rate of the [Ca 2 ] c changes during the oscillations was also different. The increase and decrease in [Ca 2 ] c during the oscillations were clearly slower in SERCA3 / than SERCA3 / islets (Fig. 7A and B). Similar features were observed in clusters of islet cells in which glucose-induced [Ca 2 ] c oscillations are slower and often more regular than in islets (Fig. 7C and D). Average [Ca 2 ] c integrated over 15 min of perifusion with 20 mmol/l glucose was similar in SERCA3 / ( nmol/l, n 8) and in SERCA3 / cells ( nmol/l, n 10). The nadir between [Ca 2 ] c oscillations was higher (P 0.01) in SERCA3 / than SERCA3 / cells (60 8 vs nmol/l above basal [Ca 2 ] c inaca 2 -free medium, respectively, not shown in Fig. 7C and D). The amplitude of [Ca 2 ] c oscillations was smaller in SERCA3 / than SERCA3 / clusters of cells ( vs. DIABETES, VOL. 51, NOVEMBER
6 SERCA3 AND [Ca 2 ] c IN PANCREATIC -CELLS FIG. 7. SERCA3 influences glucose-induced [Ca 2 ] c oscillations in mouse islets and clusters of islet cells. Isolated islets (A and B) or clusters of islet cells (C E) from SERCA3 / or SERCA3 / mice were perifused with a medium containing 10 (G10) or 20 mmol/l glucose (G20) as indicated. Traces A D are representative of results obtained in six (A) and five (B) islets and in 8 (C) and 10 (D) clusters. E: Average of 8 or 10 [Ca 2 ] c oscillations (1 oscillation taken per each experiment) induced by 20 mmol/l glucose in clusters of islets cells from SERCA3 / and SERCA3 / mice, respectively. Simple averaging could be misleading because the oscillations have different durations. Averaging was thus performed at the onset and end of all oscillations to show the differences in mean rates of [Ca 2 ] c rise and decline in the two groups nmol/l, P 0.05). In addition, differences in the rate of [Ca 2 ] c changes during the oscillations can be seen in the representative experiments shown in Fig. 7C and D and become even more evident when single oscillations from all experiments are pooled (Fig. 7E). At the onset of the oscillation, the [Ca 2 ] c rise is faster and larger in SERCA3 / than SERCA3 / clusters. At the end of the oscillation, the return to basal [Ca 2 ] c is also faster with no slow component. These characteristics of the glucoseinduced [Ca 2 ] c oscillations are similar to those of the [Ca 2 ] c changes induced by pulses of high K (Fig. 5), which highlights the important contribution of SERCA3 in [Ca 2 ] c oscillations resulting from periodic Ca 2 influx. SERCA3 ablation does not impair glucose tolerance and glucose-induced insulin secretion. Blood glucose concentrations were similar in fed SERCA3 / (120 3 mg/dl, n 17) and SERCA3 / mice (111 4 mg/dl, n FIG. 8. SERCA3 ablation does not impair glucose tolerance and glucose-induced insulin secretion. A: SERCA3 / (F and f) and SERCA3 / mice (E and ) were fasted for 24 h. After a first measurement of blood glucose (time zero), the animals were refed ad libitum (F and E) and blood glucose was again measured 1 and 3 h later. One week later, the same animals (f and ) were fasted again, and blood glucose was measured after 24 (time zero), 25, and 27 h of fasting. Results are expressed as differences ( ) from blood glucose at time zero (arrow) for each animal (90 10 mg/dl in SERCA3 / and 89 9 mg/dl in SERCA3 / mice). Values are means SE of results obtained with seven mice for each group. B: Batches of 15 islets from SERCA3 / (F) and SERCA3 / mice (E) were perifused with a medium containing 3, 8, 12, or 15 mmol/l glucose (G) or 100 mol/l tolbutamide (Tolb), as indicated. Insulin secretion is expressed as a percentage of islet insulin content. Values are means SE of seven experiments. 17). There was also no difference after 24 h fasting (90 10 mg/dl, n 14 and 89 9 mg/dl, n 14, two samplings in each mouse). The increase in blood glucose occurring after refeeding was not different between the two groups (Fig. 8A), suggesting that SERCA3 ablation does not impair glucose homeostasis. Plasma insulin was also similar in SERCA3 / ( ng/ml, n 11) and SERCA3 / fed mice ( ng/ml, n 11). Glucose-induced insulin secretion was evaluated in vitro with perifused islets. Stimulation with 15 mmol/l glucose, after 40 min of perifusion with 3 mmol/l glucose, induced biphasic insulin release that was larger in SERCA3 / than SERCA3 / islets (Fig. 8B). Thus, the percentage of insulin secreted over the 30 min of stimulation relative to the islet insulin content was vs %, P After a 20-min period rest at 3 mmol/l glucose, the islets were stimulated by stepwise increases of the glucose concentration to 8 and 12 mmol/l and, eventually, by 100 mol/l tolbutamide (Fig. 8B). During these three periods of stimulation, fractional insulin secretion was not different between SERCA3 / and SERCA3 / islets DIABETES, VOL. 51, NOVEMBER 2002
7 A. ARREDOUANI AND ASSOCIATES DISCUSSION The present study shows that in the mouse pancreas, SERCA3 is present only in insulin-secreting -cells. By using the recently generated SERCA3 knockout mice (29), we also demonstrate that SERCA3 does not contribute to glucose-induced Ca 2 uptake by the ER at basal [Ca 2 ] c but has a specific role in the modulation of [Ca 2 ] c oscillations induced by periodic Ca 2 influx in -cells. Finally, SERCA3 expression is dispensable for normal insulin secretion and glucose homeostasis in mice maintained under normal conditions. The model. Pancreatic islets express SERCA2b and the three SERCA3 isoforms (SERCA3a c) (1,39). The SERCA3 / mouse model permits investigation of the role of all SERCA3 isoforms together, but not that of individual isoforms, because excision of part of exon 5 during processing of the primary transcript blocks the pump activity of all three alternatively spliced isoforms, which are the product of the same gene but differ by no, partial, or total retention of exon 21 (1). RT-PCR did no reveal any obvious difference in the expression level of SERCA2 mrna (presumably SERCA2b) between SERCA3 / and SERCA3 / islets, suggesting that the loss of SERCA3 activity is not compensated for by an overexpression of SERCA2 in islets of SERCA3-deficient mice. Immunolocalization of SERCA3 and islet hormones showed that within the islet, SERCA3 expression is restricted to -cells. This makes the interpretation of [Ca 2 ] c measurements easier because any difference in [Ca 2 ] c changes between SERCA3 / and SERCA3 / islets can be attributed specifically to -cells. The lack of SERCA3 immunoreactivity in pancreatic acini has been documented previously (12). Role of SERCA3 when [Ca 2 ] c is low in -cells. Voltage-dependent Ca 2 channels are the main Ca 2 entry pathway in the electrically excitable -cell (40). A small capacitative Ca 2 entry (through voltage-independent Ca 2 channels) can also be evoked when TG, a SERCA inhibitor, or IP 3 -producing agents deplete the Ca 2 content of the ER (41,42). However, because this entry mechanism inactivates rapidly in a Ca 2 -dependent manner (43), it is best detected upon readdition of Ca 2 to a Ca 2 -free medium. Moreover, Ca 2 influx through this pathway is small and barely increases [Ca 2 ] c in the steady state (42). This probably explains why no significant differences in basal [Ca 2 ] c levels were observed here between islets that were or were not pretreated with TG. This lack of difference also indicates that basal [Ca 2 ] c in -cells is mainly set by Ca 2 extrusion systems other than the SERCAs. Stimulation of -cells with glucose induces an initial TG-inhibitable drop in [Ca 2 ] c that precedes the [Ca 2 ] c rise and is explained by Ca 2 being pumped into the ER (18,42). As this drop was of similar amplitude in SERCA3 / and SERCA3 / mice, we can rule out the intervention of SERCA3 in this phenomenon. Likewise, SERCA3 does not seem to be involved in the glucose-induced replenishment of the IP 3 -sensitive and -insensitive Ca 2 pools of the ER, nor in the maintenance of their Ca 2 content at basal [Ca 2 ] c over time. Indeed, ACh and TG elicited similar Ca 2 mobilizations in SERCA3 / and SERCA3 / -cells preexposed to glucose. Glucoseinduced Ca 2 uptake by the ER at basal [Ca 2 ] c is thus probably mediated by SERCA2b, the other SERCA isoform expressed in -cells, which is compatible with its much higher affinity for Ca 2 (K 1/2 of 0.2 mol/l) relative to that of SERCA3 (K 1/2 of 2 mol/l) (1,10,11,44). A recent study suggested that decreased expression of SERCA2, but not SERCA3, by injection of specific antisense oligonucleotides reduced the Ca 2 content of the ER at basal [Ca 2 ] c (13). Role of SERCA3 in [Ca 2 ] c oscillations induced by periodic Ca 2 influx in -cells. We previously demonstrated that the ER shapes [Ca 2 ] c oscillations induced by periodic Ca 2 influx in -cells (21). It limits the amplitude of the oscillation by rapidly taking up Ca 2 during the upstroke and prolongs the duration of the oscillation by slowly releasing Ca 2 into the cytosol after Ca 2 influx has stopped. These findings are confirmed here by the observation that high K -induced transient [Ca 2 ] c elevations were larger and devoid of a slow recovery phase in islets whose SERCAs were blocked by TG. Importantly, our present results also demonstrate that this effect of the ER can largely be attributed to the activity of SERCA3. Thus, high K -induced transient [Ca 2 ] c elevations in SERCA3 / islets were similar to those in SERCA3 / islets pretreated with TG and were virtually unaffected by TG pretreatment. The present results further show that SERCA3 also plays a role during [Ca 2 ] c oscillations induced by glucose. The amplitude of these oscillations was indeed larger and their nadir lower in SERCA3 / than SERCA3 / islets. As for high K -induced transient [Ca 2 ] c elevations, the larger amplitude of glucose-induced [Ca 2 ] c oscillations likely results from reduced Ca 2 uptake by the ER in SERCA3 / -cells. As a consequence, less or no Ca 2 is slowly released by the ER during the intervals, and the return of [Ca 2 ] c to basal levels is not impeded (the nadir is lower) before the next [Ca 2 ] c oscillation starts. Blockade of all SERCAs by TG is known to augment glucoseinduced [Ca 2 ] c increase by activating a depolarizing store-operated current, which then facilitates the opening of voltage-dependent Ca 2 channels (18,42,45,46). However, selective loss of SERCA3 did not increase average [Ca 2 ] c compared with control islets, probably because the activity of SERCA2 prevented activation of a storeoperated current with a similar amplitude to that induced by TG. A distinct subcellular distribution of various SERCA isoforms has been reported in acinar cells (12) but not in Jurkat T or HEK-293 cells expressing SERCA2b and SERCA3 (44). Spatial separation of SERCAs with distinct Ca 2 affinities might explain the existence of subcompartments of the ER with high and low Ca 2 concentrations (47). Whether the high Ca 2 -affinity SERCA2b, operating mainly at low [Ca 2 ] c, and the low Ca 2 -affinity SERCA3, operating when [Ca 2 ] c increases are localized at distinct sites of the ER best fitting their specific roles, remains to be investigated in -cells. SERCA3 ablation does not impair glucose tolerance and insulin secretion in mice. Glucose-induced [Ca 2 ] c responses are altered in islets from animal models of type 2 diabetes (22,23), and several studies have incriminated a reduced activity of Ca 2 -ATPase (24), all SERCAs (25), or DIABETES, VOL. 51, NOVEMBER
8 SERCA3 AND [Ca 2 ] c IN PANCREATIC -CELLS more specifically SERCA3 (5,26,27). The present experiments with SERCA3 / mice do not support this suggestion. Indeed, SERCA3 / mice were normoglycemic, had a normal insulinemia and body weight, and showed essentially the same response to a refeeding test as SERCA3 / mice. Moreover, perifused islets from SERCA3 / mice displayed a similar or better insulin secretory response to glucose than did islets from their wild-type littermates. The reasons for this are unclear because average [Ca 2 ] c was similar between the two types of islets. Differences in the kinetics of [Ca 2 ] c changes during the oscillations might allow [Ca 2 ] c to stay above the threshold for exocytosis for longer periods of time in SERCA3 / than SERCA3 / -cells. Localized [Ca 2 ] c in zones of exocytosis might also be higher after ablation of the buffering effect of SERCA3. The essentially normal glucose tolerance and insulin secretion of SERCA3 / mice suggest that the mutations or polymorphisms of the SERCA3 gene reported in type 2 diabetic patients (27) are not causally involved in the development of diabetes. The reduced expression or activity of SERCA3 observed in type 2 diabetes could result partly from the associated hyperglycemia. Indeed, culturing rat islets in high glucose concentrations decreased their Ca 2 -ATPase activity (24). Moreover, the large reduction of SERCA3 expression that was observed in rats rendered hyperglycemic by 90% pancreatectomy was completely reversed after normalization of the glycemia by phlorizin treatment (26). Another possibility is that SERCA3 knockout mice have developed compensatory mechanisms that type 2 diabetic patients have not. Indeed, a recent study using mice lacking one copy of the SERCA2 gene indicated that Ca 2 signaling mechanisms exhibit a high degree of plasticity (48). Further studies will be needed to determine whether alteration in SERCA3 activity could increase susceptibility to diabetes development in a predisposing environment. Our data demonstrate that within the pancreas, SERCA3 is expressed in -cells only. They also suggest that Ca 2 uptake by the ER at basal [Ca 2 ] c is attributable to SERCA2b rather than SERCA3. In contrast, the contribution of SERCA3 becomes important when [Ca 2 ] c increases. The distinct roles of the two SERCA isoforms are compatible with their respective affinities for Ca 2 (1,10,11,44). Activation of SERCA3 by a rise in [Ca 2 ] c has an impact on [Ca 2 ] c oscillations induced by periodic Ca 2 influx. Indeed, SERCA3 allows the ER to rapidly buffer Ca 2 during the upstroke of each oscillation, thereby limiting the amplitude of [Ca 2 ] c oscillations. Once Ca 2 influx stops, [Ca 2 ] c decreases, but Ca 2 accumulated in the ER is slowly released into the cytosol, thereby prolonging the [Ca 2 ] c oscillation. These effects of SERCA3 do not seem to have a major impact on insulin secretion. Although differences of the pulsatility of secretion may occur in the absence of SERCA3, no diabetes develops in these mice. Even if SERCA3 seems dispensable for normal insulin response, it might play other roles in -cells. The observation that [Ca 2 ] c oscillations were different in SERCA3 / and SERCA3 / islets corroborates the previous proposal that the membrane potential of -cells is modulated by the ER (21) and suggests that this control involves SERCA3. Several studies reported that the Ca 2 content of the ER is important for synthesis, modifications, and folding of proteins (49,50). It is possible that cyclic elevations of the Ca 2 concentration within the ER during [Ca 2 ] c oscillations play a role in this process (49). ACKNOWLEDGMENTS This work was supported by grant from the Fonds de la Recherche Scientifique Médicale (Brussels), grants and from the Fonds National de la Recherche Scientifique (Brussels), grant ARC 00/ from the General Direction of Scientific Research of the French Community of Belgium, National Institutes of Health grant HL61974, and by the Interuniversity Poles of Attraction Program P5/3/20, Belgian State, Prime Minister s Office, Federal Office for Scientific, Technical and Cultural Affairs. The authors thank Alexandra Fiévet for help with RT- PCR and Fabien Knockaert for insulin assays. P.G. is Senior Research Associate and J.-C.J. Research Associate of the Fonds National de la Recherche Scientifique, Brussels. REFERENCES 1. Dode L, De Greef C, Mountian I, Attard M, Town MM, Casteels R, Wuytack F: Structure of the human sarco/endoplasmic reticulum Ca 2 -ATPase 3 gene: promoter analysis and alternative splicing of the SERCA3 pre-mrna. J Biol Chem 273: , East JM: Sarco(endo)plasmic reticulum calcium pumps: recent advances in our understanding of structure/function and biology. Mol Membr Biol 17: , Misquitta CM, Mack DP, Grover AK: Sarco/endoplasmic reticulum Ca 2 (SERCA)-pumps: link to heart beats and calcium waves. Cell Calcium 25: , Wu KD, Lee WS, Wey J, Bungard D, Lytton J: Localization and quantification of endoplasmic reticulum Ca 2 -ATPase isoform transcripts. Am J Physiol 269:C775 C784, Váradi A, Molnár E, Östenson CG, Ashcroft SJH: Isoforms of endoplasmic reticulum Ca 2 -ATPase are differentially expressed in normal and diabetic islets of Langerhans. Biochem J 319: , Lytton J, Zarain-Herzberg A, Periasamy M, MacLennan DH: Molecular cloning of the mammalian smooth muscle sarco(endo)plasmic reticulum Ca 2 -ATPase. J Biol Chem 264: , Periasamy M, Reed TD, Liu LH, Ji Y, Loukianov E, Paul RJ, Nieman ML, Riddle T, Duffy JJ, Doetschman T, Lorenz JN, Shull GE: Impaired cardiac performance in heterozygous mice with a null mutation in the sarco(endo)plasmic reticulum Ca 2 -ATPase isoform 2 (SERCA2) gene. J Biol Chem 274: , Shull GE: Gene knockout studies of Ca 2 -transporting ATPases. Eur J Biochem 267: , Burk SE, Lytton J, MacLennan DH, Shull GE: cdna cloning, functional expression, and mrna tissue distribution of a third organellar Ca 2 pump. J Biol Chem 264: , Wuytack F, Dode L, Baba-Aissa F, Raeymaekers L: The SERCA3-type of organellar Ca 2 pumps. Biosci Rep 15: , Lytton J, Westlin M, Burk SE, Shull GE, MacLennan DH: Functional comparisons between isoforms of the sarcoplasmic or endoplasmic reticulum family of calcium pumps. J Biol Chem 267: , Lee MG, Xu X, Zeng WZ, Diaz J, Kuo TH, Wuytack F, Racymaekers L, Muallem S: Polarized expression of Ca 2 pumps in pancreatic and salivary gland cells: role in initiation and propagation of [Ca 2 ] i waves. J Biol Chem 272: , Varadi A, Rutter GA: Dynamic imaging of endoplasmic reticulum Ca 2 concentration in insulin-secreting MIN6 cells using recombinant targeted cameleons: roles of sarco(endo)plasmic reticulum Ca 2 -ATPase (SERCA)-2 and ryanodine receptors. Diabetes 51 (Suppl. 1):S190 S201, Henquin JC, Meissner HP: Significance of ionic fluxes and changes in membrane potential for stimulus-secretion coupling in pancreatic -cells. Experientia 40: , Gilon P, Henquin JC: Influence of membrane potential changes on cyto DIABETES, VOL. 51, NOVEMBER 2002
9 A. ARREDOUANI AND ASSOCIATES plasmic Ca 2 concentration in an electrically excitable cell, the insulinsecreting pancreatic -cell. J Biol Chem 267: , Santos RM, Rosario LM, Nadal A, Garcia-Sancho J, Soria B, Valdeolmillos M: Widespread synchronous [Ca 2 ] i oscillations due to bursting electrical activity in single pancreatic islets. Pflügers Archiv 418: , Maechler P, Kennedy ED, Sebo E, Valeva A, Pozzan T, Wollheim CB: Secretagogues modulate the calcium concentration in the endoplasmic reticulum of insulin-secreting cells: studies in aequorin-expressing intact and permeabilized Ins-1 cells. J Biol Chem 274: , Roe MW, Mertz RJ, Lancaster ME, Worley JF, III, Dukes ID: Thapsigargin inhibits the glucose-induced decrease of intracellular Ca 2 in mouse islets of Langerhans. Am J Physiol 266:E852 E862, Tengholm A, Hellman B, Gylfe E: Glucose regulation of free Ca 2 in the endoplasmic reticulum of mouse pancreatic -cells. J Biol Chem 274: , Gilon P, Henquin JC: Mechanisms and physiological significance of the cholinergic control of pancreatic -cell function. Endocr Rev 22: , Gilon P, Arredouani A, Gailly P, Gromada J, Henquin JC: Uptake and release of Ca 2 by the endoplasmic reticulum contribute to the oscillations of the cytosolic Ca 2 concentration triggered by Ca 2 influx in the electrically excitable pancreatic -cell. J Biol Chem 274: , Marie JC, Bailbe D, Gylfe E, Portha B: Defective glucose-dependent cytosolic Ca 2 handling in islets of GK and nstz rat models of type 2 diabetes. J Endocrinol 169: , Henquin JC, Jonas JC, Sato Y, Detimary P, Gilon P: Signal transduction: regulation of insulin secretion by changes in Ca 2 concentration and action in pancreatic -cells. In Advances in Molecular and Cell Biology. E. Edward Bittar, Ed. Stamford, CT, JAI Press, p , Levy J, Zhu ZX, Dunbar JC: The effect of glucose and calcium on Ca 2 -adenosine triphosphatase in pancreatic islets isolated from a normal and a non-insulin-dependent diabetes mellitus rat model. Metabolism 47: , Roe MW, Philipson LH, Frangakis CJ, Kuznetsov A, Mertz RJ, Lancaster ME, Spencer B, Worley JF, Dukes ID: Defective glucose-dependent endoplasmic reticulum Ca 2 sequestration in diabetic mouse islets of Langerhans. J Biol Chem 269: , Jonas JC, Sharma A, Hasenkamp W, Ilkova H, Patane G, Laybutt R, Bonner WS, Weir GC: Chronic hyperglycemia triggers loss of pancreatic beta cell differentiation in an animal model of diabetes. J Biol Chem 274: , Varadi A, Lebel L, Hashim Y, Mehta Z, Ashcroft SJH, Turner R: Sequence variants of the sarco(endo)plasmic reticulum Ca 2 -transport ATPase 3 gene (SERCA3) in caucasian type II diabetic patients (UH Prospective Diabetes Study 48). Diabetologia 42: , Xu GG, Gao Z, Borge PD, Jegier PA, Young RA, Wolf BA: Insulin regulation of -cell function involves a feedback loop on SERCA gene expression, Ca 2 homeostasis, and insulin expression and secretion. Biochemistry 39: , Liu LH, Paul RJ, Sutliff RL, Miller ML, Lorenz JN, Pun RY, Duffy JJ, Doetschman T, Kimura Y, MacLennan DH, Hoying JB, Shull GE: Defective endothelium-dependent relaxation of vascular smooth muscle and endothelial cell Ca 2 signaling in mice lacking sarco(endo)plasmic reticulum Ca 2 -ATPase isoform 3. J Biol Chem 272: , Lytton J, Westlin M, Hanley MR: Thapsigargin inhibits the sarcoplasmic or endoplasmic reticulum Ca 2 -ATPase family of calcium pumps. J Biol Chem 266: , Wuytack F, Papp B, Verboomen H, Raeymaekers L, Dode L, Bobe R, Enouf J, Bokkala S, Authi KS, Casteels R: A sarco/endoplasmic reticulum Ca 2 -ATPase 3-type Ca 2 pump is expressed in platelets, in lymphoid cells, and in mast cells. J Biol Chem 269: , vonwasielewski R, Mengel M, Gignac S, Wilkens L, Werner M, Georgii A: Tyramine amplification technique in routine immunohistochemistry. J Histochem Cytochem 45: , Ravier MA, Eto K, Jonkers FC, Nenquin M, Kadowaki T, Henquin JC: The oscillatory behavior of pancreatic islets from mice with mitochondrial glycerol-3-phosphate dehydrogenase knockout. J Biol Chem 275: , Jonkers FC, Jonas JC, Gilon P, Henquin JC: Influence of cell number on the characteristics and synchrony of Ca 2 oscillations in clusters of mouse pancreatic islet cells. J Physiol 520: , Ravier MA, Gilon P, Henquin JC: Oscillations of insulin secretion can be triggered by imposed oscillations of cytoplasmic Ca 2 or metabolism in normal mouse islets. Diabetes 48: , Miura Y, Gilon P, Henquin JC: Muscarinic stimulation increases Na entry in pancreatic -cells by a mechanism other than the emptying of intracellular Ca 2 pools. Biochem Biophys Res Commun 224:67 73, Rutter GA, Theler JM, Li G, Wollheim CB: Ca 2 stores in insulin-secreting cells: lack of effect of cadp ribose. Cell Calcium 16:71 80, Tengholm A, Hellman B, Gylfe E: The endoplasmic reticulum is a glucosemodulated high-affinity sink for Ca 2 in mouse pancreatic -cells. J Physiol 530: , Varadi A, Molnar E, Ashcroft SJ: A unique combination of plasma membrane Ca 2 -ATPase isoforms is expressed in islets of Langerhans and pancreatic -cell lines. Biochem J 314: , Ashcroft FM, Rorsman P: Electrophysiology of the pancreatic -cell. Prog Biophys Mol Biol 54:87 143, Liu YJ, Gylfe E: Store-operated Ca 2 entry in insulin-releasing pancreatic -cells. Cell Calcium 22: , Miura Y, Henquin JC, Gilon P: Emptying of intracellular Ca 2 stores stimulates Ca 2 entry in mouse pancreatic -cells by both direct and indirect mechanisms. J Physiol 503: , Louzao MC, Ribeiro CMP, Bird GStJ, Putney JW: Cell type-specific modes of feedback regulation of capacitative calcium entry. J Biol Chem 271: , Poch E, Leach S, Snape S, Cacic T, MacLennan DH, Lytton J: Functional characterization of alternatively spliced human SERCA3 transcripts. Am J Physiol 275:C1449 C1458, Worley JF, III, McIntyre MS, Spencer B, Dukes ID: Depletion of intracellular Ca 2 stores activates a maitotoxin-sensitive nonselective cationic current in -cells. J Biol Chem 269: , Worley JF, III, McIntyre MS, Spencer B, Mertz RJ, Roe MW, Dukes ID: Endoplasmic reticulum calcium store regulates membrane potential in mouse islet -cells. J Biol Chem 269: , Montero M, Alvarez J, Scheenen WJ, Rizzuto R, Meldolesi J, Pozzan T: Ca 2 homeostasis in the endoplasmic reticulum: coexistence of high and low [Ca 2 ] subcompartments in intact HeLa cells. J Cell Biol 139: , Zhao XS, Shin DM, Liu LH, Shull GE, Muallem S: Plasticity and adaptation of Ca 2 signaling and Ca 2 -dependent exocytosis in SERCA2( / ) mice. EMBO J 20: , Corbett EF, Michalak M: Calcium, a signaling molecule in the endoplasmic reticulum? Trends Biochem Sci 25: , Guest PC, Bailyes EM, Hutton JC: Endoplasmic reticulum Ca 2 is important for the proteolytic processing and intracellular transport of proinsulin in the pancreatic -cell. Biochem J 323: , 1997 DIABETES, VOL. 51, NOVEMBER
Results and discussion
 Supplementary Figure 1. Assessment of the cross-talk between FuraPE3 and D4ER fluorescent spectra. Cells were perifused with 15 mmol/l glucose (G15) in the presence of 250 µmol/l of the KATP channel opener
Supplementary Figure 1. Assessment of the cross-talk between FuraPE3 and D4ER fluorescent spectra. Cells were perifused with 15 mmol/l glucose (G15) in the presence of 250 µmol/l of the KATP channel opener
Abbreviations: P- paraffin-embedded section; C, cryosection; Bio-SA, biotin-streptavidin-conjugated fluorescein amplification.
 Supplementary Table 1. Sequence of primers for real time PCR. Gene Forward primer Reverse primer S25 5 -GTG GTC CAC ACT ACT CTC TGA GTT TC-3 5 - GAC TTT CCG GCA TCC TTC TTC-3 Mafa cds 5 -CTT CAG CAA GGA
Supplementary Table 1. Sequence of primers for real time PCR. Gene Forward primer Reverse primer S25 5 -GTG GTC CAC ACT ACT CTC TGA GTT TC-3 5 - GAC TTT CCG GCA TCC TTC TTC-3 Mafa cds 5 -CTT CAG CAA GGA
Disorganization of cytoplasmic Ca 2+ oscillations and pulsatile insulin secretion in islets from ob/ob mice
 Diabetologia (2002) 45:1154 1163 DOI 10.1007/s00125-002-0883-9 Disorganization of cytoplasmic Ca 2+ oscillations and pulsatile insulin secretion in islets from ob/ob mice M. A. Ravier 1, J. Sehlin 2, J.
Diabetologia (2002) 45:1154 1163 DOI 10.1007/s00125-002-0883-9 Disorganization of cytoplasmic Ca 2+ oscillations and pulsatile insulin secretion in islets from ob/ob mice M. A. Ravier 1, J. Sehlin 2, J.
Alterations of the Ca 2+ signaling pathway in pancreatic beta-cells isolated from db/db mice
 Protein Cell 214, 5(1):783 794 DOI 1.17/s13238-14-75-7 Alterations of the Ca 2+ signaling pathway in pancreatic beta-cells isolated from db/db mice Kuo Liang 1, Wen Du 2, Jingze Lu 2, Fei Li 1&, Lu Yang
Protein Cell 214, 5(1):783 794 DOI 1.17/s13238-14-75-7 Alterations of the Ca 2+ signaling pathway in pancreatic beta-cells isolated from db/db mice Kuo Liang 1, Wen Du 2, Jingze Lu 2, Fei Li 1&, Lu Yang
a) Primary cultures derived from the pancreas of an 11-week-old Pdx1-Cre; K-MADM-p53
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1. Induction of p53 LOH by MADM. a) Primary cultures derived from the pancreas of an 11-week-old Pdx1-Cre; K-MADM-p53 mouse revealed increased p53 KO/KO (green,
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1. Induction of p53 LOH by MADM. a) Primary cultures derived from the pancreas of an 11-week-old Pdx1-Cre; K-MADM-p53 mouse revealed increased p53 KO/KO (green,
Store-operated influx of Ca 2+ in pancreatic β-cells exhibits graded dependence on the filling of the endoplasmic reticulum
 RESEARCH ARTICLE 2179 Store-operated influx of Ca 2+ in pancreatic β-cells exhibits graded dependence on the filling of the endoplasmic reticulum Oleg Dyachok 1,2 and Erik Gylfe 1, * 1 Department of Medical
RESEARCH ARTICLE 2179 Store-operated influx of Ca 2+ in pancreatic β-cells exhibits graded dependence on the filling of the endoplasmic reticulum Oleg Dyachok 1,2 and Erik Gylfe 1, * 1 Department of Medical
SUR1, sulfonylurea receptor 1; [Ca 2 ] c, cytosolic Ca 2 concentration; KO, knock-out.
![SUR1, sulfonylurea receptor 1; [Ca 2 ] c, cytosolic Ca 2 concentration; KO, knock-out. SUR1, sulfonylurea receptor 1; [Ca 2 ] c, cytosolic Ca 2 concentration; KO, knock-out.](/thumbs/76/73249559.jpg) THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 20, pp. 14768 14776, May 18, 2007 2007 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Overnight Culture Unmasks
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 20, pp. 14768 14776, May 18, 2007 2007 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Overnight Culture Unmasks
A Mathematical Study of the Differential Effects of Two SERCA Isoforms on Ca 2+ Oscillations in Pancreatic Islets
 Bulletin of Mathematical Biology (2008) 70: 1251 1271 DOI 10.1007/s11538-008-9298-1 ORIGINAL ARTICLE A Mathematical Study of the Differential Effects of Two SERCA Isoforms on Ca 2+ Oscillations in Pancreatic
Bulletin of Mathematical Biology (2008) 70: 1251 1271 DOI 10.1007/s11538-008-9298-1 ORIGINAL ARTICLE A Mathematical Study of the Differential Effects of Two SERCA Isoforms on Ca 2+ Oscillations in Pancreatic
Supplementary Materials
 Supplementary Materials 1 Supplementary Table 1. List of primers used for quantitative PCR analysis. Gene name Gene symbol Accession IDs Sequence range Product Primer sequences size (bp) β-actin Actb gi
Supplementary Materials 1 Supplementary Table 1. List of primers used for quantitative PCR analysis. Gene name Gene symbol Accession IDs Sequence range Product Primer sequences size (bp) β-actin Actb gi
SUPPLEMENTAL FIGURE 1
 SUPPLEMENTL FIGURE 1 C Supplemental Figure 1. pproach for removal of snorns from Rpl13a gene. () Wild type Rpl13a exonintron structure is shown, with exo in black and intronic snorns in red rectangles.
SUPPLEMENTL FIGURE 1 C Supplemental Figure 1. pproach for removal of snorns from Rpl13a gene. () Wild type Rpl13a exonintron structure is shown, with exo in black and intronic snorns in red rectangles.
CD31 5'-AGA GAC GGT CTT GTC GCA GT-3' 5 ' -TAC TGG GCT TCG AGA GCA GT-3'
 Table S1. The primer sets used for real-time RT-PCR analysis. Gene Forward Reverse VEGF PDGFB TGF-β MCP-1 5'-GTT GCA GCA TGA ATC TGA GG-3' 5'-GGA GAC TCT TCG AGG AGC ACT T-3' 5'-GAA TCA GGC ATC GAG AGA
Table S1. The primer sets used for real-time RT-PCR analysis. Gene Forward Reverse VEGF PDGFB TGF-β MCP-1 5'-GTT GCA GCA TGA ATC TGA GG-3' 5'-GGA GAC TCT TCG AGG AGC ACT T-3' 5'-GAA TCA GGC ATC GAG AGA
Influence of cell number on the characteristics and synchrony of Ca oscillations in clusters of mouse pancreatic islet cells
 9636 Journal of Physiology (1999), 520.3, pp. 839 849 839 Influence of cell number on the characteristics and synchrony of Ca oscillations in clusters of mouse pancreatic islet cells Fran coise C. Jonkers,
9636 Journal of Physiology (1999), 520.3, pp. 839 849 839 Influence of cell number on the characteristics and synchrony of Ca oscillations in clusters of mouse pancreatic islet cells Fran coise C. Jonkers,
Pancreatic -cells synthesize insulin and secrete it at
 Perspectives in Diabetes Triggering and Amplifying Pathways of Regulation of Insulin Secretion by Glucose Jean-Claude Henquin Glucose stimulates insulin secretion by generating triggering and amplifying
Perspectives in Diabetes Triggering and Amplifying Pathways of Regulation of Insulin Secretion by Glucose Jean-Claude Henquin Glucose stimulates insulin secretion by generating triggering and amplifying
The central importance of glucose homeostasis. Dynamics of Calcium Clearance in Mouse Pancreatic -Cells
 Dynamics of Calcium Clearance in Mouse Pancreatic -Cells Liangyi Chen, 1 Duk-Su Koh, 1,2 and Bertil Hille 1 Pancreatic -cells maintain glucose homeostasis by their regulated Ca 2 -dependent secretion of
Dynamics of Calcium Clearance in Mouse Pancreatic -Cells Liangyi Chen, 1 Duk-Su Koh, 1,2 and Bertil Hille 1 Pancreatic -cells maintain glucose homeostasis by their regulated Ca 2 -dependent secretion of
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Sherman SI, Wirth LJ, Droz J-P, et al. Motesanib diphosphate
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Sherman SI, Wirth LJ, Droz J-P, et al. Motesanib diphosphate
Supplementary Document
 Supplementary Document 1. Supplementary Table legends 2. Supplementary Figure legends 3. Supplementary Tables 4. Supplementary Figures 5. Supplementary References 1. Supplementary Table legends Suppl.
Supplementary Document 1. Supplementary Table legends 2. Supplementary Figure legends 3. Supplementary Tables 4. Supplementary Figures 5. Supplementary References 1. Supplementary Table legends Suppl.
Laboratory of Experimental Medicine, Brussels Free University, B-1070 Brussels', Belgium and Novo Nordisk A/S, DK-2880 Bagsvaerd, Denmark
 Vol. 43, No. 2, October 1997 Pages 233-240 CATONC AND SECRETORY EFFECTS OF 6-O-ACETYL-D-GLUCOSE N RAT PANCREATC SLETS Hassan JJAKL, Ole KRK and Willy J. MALASSE Laboratory of Experimental Medicine, Brussels
Vol. 43, No. 2, October 1997 Pages 233-240 CATONC AND SECRETORY EFFECTS OF 6-O-ACETYL-D-GLUCOSE N RAT PANCREATC SLETS Hassan JJAKL, Ole KRK and Willy J. MALASSE Laboratory of Experimental Medicine, Brussels
Supplemental Data. Shin et al. Plant Cell. (2012) /tpc YFP N
 MYC YFP N PIF5 YFP C N-TIC TIC Supplemental Data. Shin et al. Plant Cell. ()..5/tpc..95 Supplemental Figure. TIC interacts with MYC in the nucleus. Bimolecular fluorescence complementation assay using
MYC YFP N PIF5 YFP C N-TIC TIC Supplemental Data. Shin et al. Plant Cell. ()..5/tpc..95 Supplemental Figure. TIC interacts with MYC in the nucleus. Bimolecular fluorescence complementation assay using
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 U1 inhibition causes a shift of RNA-seq reads from exons to introns. (a) Evidence for the high purity of 4-shU-labeled RNAs used for RNA-seq. HeLa cells transfected with control
Supplementary Figure 1 U1 inhibition causes a shift of RNA-seq reads from exons to introns. (a) Evidence for the high purity of 4-shU-labeled RNAs used for RNA-seq. HeLa cells transfected with control
Phys 173 / BGGN 266. LPA Induced Cl - Oscillations in Xenopus Oocytes. Nini Huynh David Marciano Chisa Suzuki
 Phys 173 / BGGN 266 LPA Induced Cl - Oscillations in Xenopus Oocytes Nini Huynh David Marciano Chisa Suzuki If only we hadn t poked these oocytes, how cute would it be! INTRODUCTION Electrophysiology in
Phys 173 / BGGN 266 LPA Induced Cl - Oscillations in Xenopus Oocytes Nini Huynh David Marciano Chisa Suzuki If only we hadn t poked these oocytes, how cute would it be! INTRODUCTION Electrophysiology in
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used for PCR and qpcr Primer Name
 Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
ISSN: [Bindulekha * et al., 7(1): January, 2018] Impact Factor: 5.164
![ISSN: [Bindulekha * et al., 7(1): January, 2018] Impact Factor: 5.164 ISSN: [Bindulekha * et al., 7(1): January, 2018] Impact Factor: 5.164](/thumbs/96/128285435.jpg) IJESRT INTERNATIONAL JOURNAL OF ENGINEERING SCIENCES & RESEARCH TECHNOLOGY THE INTERRELATIONSHIP OF INSULIN AND Ca 2+ - ATPase MEDIATED INTRACELLULAR CALCIUM HOMEOSTASIS Bindulekha D.S* *Department of
IJESRT INTERNATIONAL JOURNAL OF ENGINEERING SCIENCES & RESEARCH TECHNOLOGY THE INTERRELATIONSHIP OF INSULIN AND Ca 2+ - ATPase MEDIATED INTRACELLULAR CALCIUM HOMEOSTASIS Bindulekha D.S* *Department of
Beta Thalassemia Case Study Introduction to Bioinformatics
 Beta Thalassemia Case Study Sami Khuri Department of Computer Science San José State University San José, California, USA sami.khuri@sjsu.edu www.cs.sjsu.edu/faculty/khuri Outline v Hemoglobin v Alpha
Beta Thalassemia Case Study Sami Khuri Department of Computer Science San José State University San José, California, USA sami.khuri@sjsu.edu www.cs.sjsu.edu/faculty/khuri Outline v Hemoglobin v Alpha
Autonomic regulation of islet hormone secretion
 Autonomic regulation of islet hormone secretion Implications for health and disease Billy & Bree Paper 1: Autonomic regulation of islet hormone secretion : Implications for health and disease By Team BBB
Autonomic regulation of islet hormone secretion Implications for health and disease Billy & Bree Paper 1: Autonomic regulation of islet hormone secretion : Implications for health and disease By Team BBB
Type 2 diabetes develops as a consequence of
 -Cell Function and Viability in the Spontaneously Diabetic GK Rat Information From the GK/Par Colony B. Portha, M.-H. Giroix, P. Serradas, M.-N. Gangnerau, J. Movassat, F. Rajas, D. Bailbe, C. Plachot,
-Cell Function and Viability in the Spontaneously Diabetic GK Rat Information From the GK/Par Colony B. Portha, M.-H. Giroix, P. Serradas, M.-N. Gangnerau, J. Movassat, F. Rajas, D. Bailbe, C. Plachot,
G-PROTEIN-INDEPENDENT ACTIVATION OF AN INWARD Na + CURRENT
 JBC Papers in Press. Published on August 2, 2002 as Manuscript M203888200 1 G-PROTEIN-INDEPENDENT ACTIVATION OF AN INWARD Na + CURRENT BY MUSCARINIC RECEPTORS IN MOUSE PANCREATIC β-cells Jean-François
JBC Papers in Press. Published on August 2, 2002 as Manuscript M203888200 1 G-PROTEIN-INDEPENDENT ACTIVATION OF AN INWARD Na + CURRENT BY MUSCARINIC RECEPTORS IN MOUSE PANCREATIC β-cells Jean-François
Original Article Inositol (1,4,5)-Trisphosphate Dynamics and Intracellular Calcium Oscillations in Pancreatic -Cells
 Original Article Inositol (1,4,5)-Trisphosphate Dynamics and Intracellular Calcium Oscillations in Pancreatic -Cells Natalia A. Tamarina, 1 Andrey Kuznetsov, 1 Christopher J. Rhodes, 2 Vytautas P. Bindokas,
Original Article Inositol (1,4,5)-Trisphosphate Dynamics and Intracellular Calcium Oscillations in Pancreatic -Cells Natalia A. Tamarina, 1 Andrey Kuznetsov, 1 Christopher J. Rhodes, 2 Vytautas P. Bindokas,
Supplementary Table 3. 3 UTR primer sequences. Primer sequences used to amplify and clone the 3 UTR of each indicated gene are listed.
 Supplemental Figure 1. DLKI-DIO3 mirna/mrna complementarity. Complementarity between the indicated DLK1-DIO3 cluster mirnas and the UTR of SOX2, SOX9, HIF1A, ZEB1, ZEB2, STAT3 and CDH1with mirsvr and PhastCons
Supplemental Figure 1. DLKI-DIO3 mirna/mrna complementarity. Complementarity between the indicated DLK1-DIO3 cluster mirnas and the UTR of SOX2, SOX9, HIF1A, ZEB1, ZEB2, STAT3 and CDH1with mirsvr and PhastCons
Objectives. Functions of smooth muscle. Smooth muscle. Smooth Muscle Contraction: Mechanism. Latch state. Smooth muscle contraction
 Objectives Functions of smooth muscle Sompol Tapechum,, M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj hospital อธ บายล กษณะการหดต วของกล ามเน อเร ยบได อธ บายกลไกและป จจ ยท ม ผลต อการหดต
Objectives Functions of smooth muscle Sompol Tapechum,, M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj hospital อธ บายล กษณะการหดต วของกล ามเน อเร ยบได อธ บายกลไกและป จจ ยท ม ผลต อการหดต
Universiteit Leuven, B-3000 Leuven, Belgium
 J. Physiol. (1977), 271, pp. 63-79 63 With 11 text-f guree Printed in Great Britain EXCITATION-CONTRACTION COUPLING IN THE SMOOTH MUSCLE CELLS OF THE RABBIT MAIN PULMONARY ARTERY BY R. CASTEELS, K. KITAMURA,*
J. Physiol. (1977), 271, pp. 63-79 63 With 11 text-f guree Printed in Great Britain EXCITATION-CONTRACTION COUPLING IN THE SMOOTH MUSCLE CELLS OF THE RABBIT MAIN PULMONARY ARTERY BY R. CASTEELS, K. KITAMURA,*
Chapter 4. Estrogen receptor expression in human macrophages
 Chapter 4 Estrogen receptor expression in human macrophages 4.1. Introduction Macrophages respond to estrogen present in their microenvironment and hence should express functional estrogen receptors unless
Chapter 4 Estrogen receptor expression in human macrophages 4.1. Introduction Macrophages respond to estrogen present in their microenvironment and hence should express functional estrogen receptors unless
Peter Walter, UCSF IRE1 Signaling Affects Cell Fate during the Unfolded Protein Response
 Peter Walter, UCSF IRE1 Signaling Affects Cell Fate during the Unfolded Protein Response Jenn Hou Burke Group Literature Seminar November 19 th 2016 Protein Synthesis Pathway 4. Final Destination: proteins
Peter Walter, UCSF IRE1 Signaling Affects Cell Fate during the Unfolded Protein Response Jenn Hou Burke Group Literature Seminar November 19 th 2016 Protein Synthesis Pathway 4. Final Destination: proteins
Homeostasis. Endocrine System Nervous System
 Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells.
 SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
Supporting Information
 Supporting Information Gerasimenko et al..73/pnas.39 SI Materials and Methods Reagents used in this study include Fluo-4/Fura- (Invitrogen), thapsigargin (albiochem), collagenase (Worthington), palmitoleic
Supporting Information Gerasimenko et al..73/pnas.39 SI Materials and Methods Reagents used in this study include Fluo-4/Fura- (Invitrogen), thapsigargin (albiochem), collagenase (Worthington), palmitoleic
Diabetologia 9 Springer-Vcrlag 1993
 Diabetologia (1993) 36:305-309 Diabetologia 9 Springer-Vcrlag 1993 Preferential alteration of oxidative relative to total glycolysis in pancreatic islets of two rat models of inherited or acquired Type
Diabetologia (1993) 36:305-309 Diabetologia 9 Springer-Vcrlag 1993 Preferential alteration of oxidative relative to total glycolysis in pancreatic islets of two rat models of inherited or acquired Type
Nature Biotechnology: doi: /nbt.3828
 Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 18, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 18, 2016 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 18, 2016 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
Alternative insulin delivery systems: how demanding should the patient be?
 Diabetologia (1997) 4: S97 S11 Springer-Verlag 1997 Alternative insulin delivery systems: how demanding should the patient be? K.S. Polonsky, M. M. Byrne, J. Sturis Department of Medicine, The University
Diabetologia (1997) 4: S97 S11 Springer-Verlag 1997 Alternative insulin delivery systems: how demanding should the patient be? K.S. Polonsky, M. M. Byrne, J. Sturis Department of Medicine, The University
Supplementary Table 2. Conserved regulatory elements in the promoters of CD36.
 Supplementary Table 1. RT-qPCR primers for CD3, PPARg and CEBP. Assay Forward Primer Reverse Primer 1A CAT TTG TGG CCT TGT GCT CTT TGA TGA GTC ACA GAA AGA ATC AAT TC 1B AGG AAA TGA ACT GAT GAG TCA CAG
Supplementary Table 1. RT-qPCR primers for CD3, PPARg and CEBP. Assay Forward Primer Reverse Primer 1A CAT TTG TGG CCT TGT GCT CTT TGA TGA GTC ACA GAA AGA ATC AAT TC 1B AGG AAA TGA ACT GAT GAG TCA CAG
Physiology and pharmacology of smooth muscle contractions a brief introduction
 SimVessel: Physiology and pharmacology of smooth muscle contractions a brief introduction The virtual SimVessel Laboratory is for recordings of smooth muscle contractions of small muscle stripes of different
SimVessel: Physiology and pharmacology of smooth muscle contractions a brief introduction The virtual SimVessel Laboratory is for recordings of smooth muscle contractions of small muscle stripes of different
Glucose Homeostasis. Liver. Glucose. Muscle, Fat. Pancreatic Islet. Glucose utilization. Glucose production, storage Insulin Glucagon
 Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
STEIN IN-TERM EXAM -- BIOLOGY APRIL 18, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- APRIL 18, 2019 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There is
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- APRIL 18, 2019 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There is
NIH Public Access Author Manuscript Diabetologia. Author manuscript; available in PMC 2014 February 01.
 NIH Public Access Author Manuscript Published in final edited form as: Diabetologia. 2013 February ; 56(2): 231 233. doi:10.1007/s00125-012-2788-6. Lipotoxicity impairs incretin signalling V. Poitout 1,2
NIH Public Access Author Manuscript Published in final edited form as: Diabetologia. 2013 February ; 56(2): 231 233. doi:10.1007/s00125-012-2788-6. Lipotoxicity impairs incretin signalling V. Poitout 1,2
General Principles of Endocrine Physiology
 General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
Plasma membrane depolarization as a determinant of the first phase of insulin secretion
 Am J Physiol Endocrinol Metab 297: E315 E322, 2009. First published May 26, 2009; doi:10.1152/ajpendo.90981.2008. Plasma membrane depolarization as a determinant of the first phase of insulin secretion
Am J Physiol Endocrinol Metab 297: E315 E322, 2009. First published May 26, 2009; doi:10.1152/ajpendo.90981.2008. Plasma membrane depolarization as a determinant of the first phase of insulin secretion
McAlpine PERK-GSK3 regulates foam cell formation. Supplemental Material. Supplementary Table I. Sequences of real time PCR primers.
 Mclpine PERK-GSK3 regulates foam cell formation Supplemental Material Supplementary Table I. Sequences of real time PCR primers. Primer Name Primer Sequences (5-3 ) Product Size (bp) GRP78 (human) Fwd:
Mclpine PERK-GSK3 regulates foam cell formation Supplemental Material Supplementary Table I. Sequences of real time PCR primers. Primer Name Primer Sequences (5-3 ) Product Size (bp) GRP78 (human) Fwd:
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
Kidney. Heart. Lung. Sirt1. Gapdh. Mouse IgG DAPI. Rabbit IgG DAPI
 a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
Dynamical complexity and temporal plasticity in pancreatic β-cells
 Review Dynamical complexity and temporal plasticity in pancreatic β-cells RICHARD BERTRAM* and ARTHUR SHERMAN Institute of Molecular Biophysics, Florida State University, Tallahassee, FL 32306, USA Mathatical
Review Dynamical complexity and temporal plasticity in pancreatic β-cells RICHARD BERTRAM* and ARTHUR SHERMAN Institute of Molecular Biophysics, Florida State University, Tallahassee, FL 32306, USA Mathatical
Skeletal Muscle. Connective tissue: Binding, support and insulation. Blood vessels
 Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Fig. S4. Current-voltage relations of iglurs. A-C: time courses of currents evoked by 100 ms pulses
 Fig. S1. Immunohistochemical detection of iglur2 protein in single islet cells. A: α cells identified using glucagon-specific antibody express the iglur2 subtype of AMPA receptor. 24 out of 26 identified
Fig. S1. Immunohistochemical detection of iglur2 protein in single islet cells. A: α cells identified using glucagon-specific antibody express the iglur2 subtype of AMPA receptor. 24 out of 26 identified
Ca 2+ controls slow NAD(P)H oscillations in glucose-stimulated mouse pancreatic islets
 J Physiol 572.2 (2006) pp 379 392 379 Ca 2+ controls slow NAD(P)H oscillations in glucose-stimulated mouse pancreatic islets Dan S. Luciani 1,2, Stanley Misler 3 and Kenneth S. Polonsky 3 1 Department
J Physiol 572.2 (2006) pp 379 392 379 Ca 2+ controls slow NAD(P)H oscillations in glucose-stimulated mouse pancreatic islets Dan S. Luciani 1,2, Stanley Misler 3 and Kenneth S. Polonsky 3 1 Department
Pancreatic islets and -cells display oscillations in
 Glucose-Stimulated Oscillations in Free Cytosolic ATP Concentration Imaged in Single Islet -Cells Evidence for a Ca 2 -Dependent Mechanism Edward K. Ainscow and Guy A. Rutter Normal glucose-stimulated
Glucose-Stimulated Oscillations in Free Cytosolic ATP Concentration Imaged in Single Islet -Cells Evidence for a Ca 2 -Dependent Mechanism Edward K. Ainscow and Guy A. Rutter Normal glucose-stimulated
Supporting Information
 ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
Plasmids Western blot analysis and immunostaining Flow Cytometry Cell surface biotinylation RNA isolation and cdna synthesis
 Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 15, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 15, 2018 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 15, 2018 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
c Tuj1(-) apoptotic live 1 DIV 2 DIV 1 DIV 2 DIV Tuj1(+) Tuj1/GFP/DAPI Tuj1 DAPI GFP
 Supplementary Figure 1 Establishment of the gain- and loss-of-function experiments and cell survival assays. a Relative expression of mature mir-484 30 20 10 0 **** **** NCP mir- 484P NCP mir- 484P b Relative
Supplementary Figure 1 Establishment of the gain- and loss-of-function experiments and cell survival assays. a Relative expression of mature mir-484 30 20 10 0 **** **** NCP mir- 484P NCP mir- 484P b Relative
Numerical investigation of phase transition in a cellular network and disease onset
 Numerical investigation of phase transition in a cellular network and disease onset Xujing Wang, Associate Professor Dept of Physics xujingw@uab.edu 934-8186 The question: Is (chronic) disease onset a
Numerical investigation of phase transition in a cellular network and disease onset Xujing Wang, Associate Professor Dept of Physics xujingw@uab.edu 934-8186 The question: Is (chronic) disease onset a
Alteration of Expression of Ca 2+ Signaling Proteins and Adaptation of Ca 2+ Signaling in SERCA2 +/- Mouse Parotid Acini
 Yonsei Med J 49(2):311-321, 2008 DOI 10.3349/ymj.2008.49.2.311 Alteration of Expression of Ca 2+ Signaling Proteins and Adaptation of Ca 2+ Signaling in SERCA2 +/- Mouse Parotid Acini Jong-Hoon Choi, 1
Yonsei Med J 49(2):311-321, 2008 DOI 10.3349/ymj.2008.49.2.311 Alteration of Expression of Ca 2+ Signaling Proteins and Adaptation of Ca 2+ Signaling in SERCA2 +/- Mouse Parotid Acini Jong-Hoon Choi, 1
STUDIES IN ISLETS LACKING THE NA
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 34, pp. 24538 24546, August 24, 2007 2007 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Glucose-induced Cytosolic
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 34, pp. 24538 24546, August 24, 2007 2007 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Glucose-induced Cytosolic
GENE Forward primer Reverse primer FABP4 CCTTTGTGGGGACCTGGAAA TGACCGGATGACGACCAAGT CD68 AATGTGTCCTTCCCACAAGC GGCAGCAAGAGAGATTGGTC
 Published in "" which should be cited to refer to this work. mrna extraction and RT-PCR Total RNA from 5 15 mg of crushed white adipose tissue was isolated using the technique described by Chomczynski
Published in "" which should be cited to refer to this work. mrna extraction and RT-PCR Total RNA from 5 15 mg of crushed white adipose tissue was isolated using the technique described by Chomczynski
Supplemental Data. Short Article. ATF4-Mediated Induction of 4E-BP1. Contributes to Pancreatic β Cell Survival. under Endoplasmic Reticulum Stress
 Cell Metabolism, Volume 7 Supplemental Data Short Article ATF4-Mediated Induction of 4E-BP1 Contributes to Pancreatic β Cell Survival under Endoplasmic Reticulum Stress Suguru Yamaguchi, Hisamitsu Ishihara,
Cell Metabolism, Volume 7 Supplemental Data Short Article ATF4-Mediated Induction of 4E-BP1 Contributes to Pancreatic β Cell Survival under Endoplasmic Reticulum Stress Suguru Yamaguchi, Hisamitsu Ishihara,
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 16, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
YEAR(s) FIELD OF STUDY (if applicable) Faculty de Sciences Univ. of Montpellier, Montpellier, France
 Principal Investigator/Program Director (Last, First, Middle): BIOGRAPHICAL SKETCH Provide the following information for the key personnel in the order listed on Form Page 2. Follow this format for each
Principal Investigator/Program Director (Last, First, Middle): BIOGRAPHICAL SKETCH Provide the following information for the key personnel in the order listed on Form Page 2. Follow this format for each
SUPPLEMENTARY RESULTS
 SUPPLEMENTARY RESULTS Supplementary Table 1. hfpr1- Flpln-CHO hfpr2-flpln-cho pec 50 E max (%) Log( /K A) Log( /K A) N pec 50 E max (%) Log( /K A) Log( /K A) n ERK1/2 phosphorylation fmlp 9.0±0.6 80±7
SUPPLEMENTARY RESULTS Supplementary Table 1. hfpr1- Flpln-CHO hfpr2-flpln-cho pec 50 E max (%) Log( /K A) Log( /K A) N pec 50 E max (%) Log( /K A) Log( /K A) n ERK1/2 phosphorylation fmlp 9.0±0.6 80±7
Where are the normal pacemaker and the backup pacemakers of the heart located?
 CASE 9 A 68-year-old woman presents to the emergency center with shortness of breath, light-headedness, and chest pain described as being like an elephant sitting on her chest. She is diagnosed with a
CASE 9 A 68-year-old woman presents to the emergency center with shortness of breath, light-headedness, and chest pain described as being like an elephant sitting on her chest. She is diagnosed with a
Cell Signaling: Proteins, Pathways and Mechanisms CALL FOR PAPERS. Yukari Takeda, 1 Takao Shimayoshi, 2 George G. Holz, 3 and Akinori Noma 1 1
 Am J Physiol Cell Physiol 30: C337 C347, 206. First published November 25, 205; doi:0.52/ajpcell.00234.205. CALL FOR PAPERS Cell Signaling: Proteins, Pathways and Mechanisms Modeling analysis of inositol,4,5-trisphosphate
Am J Physiol Cell Physiol 30: C337 C347, 206. First published November 25, 205; doi:0.52/ajpcell.00234.205. CALL FOR PAPERS Cell Signaling: Proteins, Pathways and Mechanisms Modeling analysis of inositol,4,5-trisphosphate
The Ca 21 Dynamics of Isolated Mouse b-cells and Islets: Implications for Mathematical Models
 2852 Biophysical Journal Volume 84 May 2003 2852 2870 The Ca 21 Dynamics of Isolated Mouse b-cells and Islets: Implications for Mathematical Models Min Zhang,* Paula Goforth,* Richard Bertram, y Arthur
2852 Biophysical Journal Volume 84 May 2003 2852 2870 The Ca 21 Dynamics of Isolated Mouse b-cells and Islets: Implications for Mathematical Models Min Zhang,* Paula Goforth,* Richard Bertram, y Arthur
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Qualifying Examination (Part I)
 Department of Pharmacology Qualifying Examination (Part I) December 11 & 12, 2003 Please remember that this is a closed-book examination. You must be prepared to answer 4 of the 7 questions. Although not
Department of Pharmacology Qualifying Examination (Part I) December 11 & 12, 2003 Please remember that this is a closed-book examination. You must be prepared to answer 4 of the 7 questions. Although not
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Mechanisms of Cell Injury: Loss of Calcium Homeostasis
 Mechanisms of Cell Injury: Loss of Calcium Homeostasis SCPA610: Cellular Pathology Amornrat N. Jensen, Ph.D. amornrat.nar@mahidol.ac.th Leading questions Why is intracellular calcium important for the
Mechanisms of Cell Injury: Loss of Calcium Homeostasis SCPA610: Cellular Pathology Amornrat N. Jensen, Ph.D. amornrat.nar@mahidol.ac.th Leading questions Why is intracellular calcium important for the
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
BCOR 011 Lecture 19 Oct 12, 2005 I. Cell Communication Signal Transduction Chapter 11
 BCOR 011 Lecture 19 Oct 12, 2005 I. Cell Communication Signal Transduction Chapter 11 External signal is received and converted to another form to elicit a response 1 Lecture Outline 1. Types of intercellular
BCOR 011 Lecture 19 Oct 12, 2005 I. Cell Communication Signal Transduction Chapter 11 External signal is received and converted to another form to elicit a response 1 Lecture Outline 1. Types of intercellular
Basics of skeletal muscle electrophysiology. Tóth András, PhD
 Basics of skeletal muscle electrophysiology Tóth András, PhD Topics Structure Contraction and relaxation Activation Excitation-contraction coupling Action potential Ion channels* Calcium homeostasis Structure
Basics of skeletal muscle electrophysiology Tóth András, PhD Topics Structure Contraction and relaxation Activation Excitation-contraction coupling Action potential Ion channels* Calcium homeostasis Structure
Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches
 Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches during application of 500 µm Ca 2+ at the intracellular
Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches during application of 500 µm Ca 2+ at the intracellular
Supplementary Figure 1 MicroRNA expression in human synovial fibroblasts from different locations. MicroRNA, which were identified by RNAseq as most
 Supplementary Figure 1 MicroRNA expression in human synovial fibroblasts from different locations. MicroRNA, which were identified by RNAseq as most differentially expressed between human synovial fibroblasts
Supplementary Figure 1 MicroRNA expression in human synovial fibroblasts from different locations. MicroRNA, which were identified by RNAseq as most differentially expressed between human synovial fibroblasts
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/106/ra7/dc1 Supplementary Materials for A Crucial Role for RACK1 in the Regulation of Glucose-Stimulated IRE1α Activation in Pancreatic β Cells Yifu Qiu, Ting
www.sciencesignaling.org/cgi/content/full/3/106/ra7/dc1 Supplementary Materials for A Crucial Role for RACK1 in the Regulation of Glucose-Stimulated IRE1α Activation in Pancreatic β Cells Yifu Qiu, Ting
Formylpeptide receptor2 contributes to colon epithelial homeostasis, inflammation, and tumorigenesis
 Supplementary Data Formylpeptide receptor2 contributes to colon epithelial homeostasis, inflammation, and tumorigenesis Keqiang Chen, Mingyong Liu, Ying Liu, Teizo Yoshimura, Wei Shen, Yingying Le, Scott
Supplementary Data Formylpeptide receptor2 contributes to colon epithelial homeostasis, inflammation, and tumorigenesis Keqiang Chen, Mingyong Liu, Ying Liu, Teizo Yoshimura, Wei Shen, Yingying Le, Scott
Silencing neurotransmission with membrane-tethered toxins
 nature methods Silencing neurotransmission with membrane-tethered toxins Sebastian Auer, Annika S Stürzebecher, René Jüttner, Julio Santos-Torres, Christina Hanack, Silke Frahm, Beate Liehl & Inés Ibañez-Tallon
nature methods Silencing neurotransmission with membrane-tethered toxins Sebastian Auer, Annika S Stürzebecher, René Jüttner, Julio Santos-Torres, Christina Hanack, Silke Frahm, Beate Liehl & Inés Ibañez-Tallon
Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells.
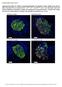 Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells. Pancreata from 16-weeks-old 6J +/+, 6J db/db, KS +/+ and KS db/db mice were harvested, fixed with
Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells. Pancreata from 16-weeks-old 6J +/+, 6J db/db, KS +/+ and KS db/db mice were harvested, fixed with
Cell Injury MECHANISMS OF CELL INJURY
 Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Table S1. Oligonucleotides used for the in-house RT-PCR assays targeting the M, H7 or N9. Assay (s) Target Name Sequence (5 3 ) Comments
 SUPPLEMENTAL INFORMATION 2 3 Table S. Oligonucleotides used for the in-house RT-PCR assays targeting the M, H7 or N9 genes. Assay (s) Target Name Sequence (5 3 ) Comments CDC M InfA Forward (NS), CDC M
SUPPLEMENTAL INFORMATION 2 3 Table S. Oligonucleotides used for the in-house RT-PCR assays targeting the M, H7 or N9 genes. Assay (s) Target Name Sequence (5 3 ) Comments CDC M InfA Forward (NS), CDC M
Closing in on the Mechanisms of Pulsatile Insulin Secretion
 Diabetes Volume 67, March 2018 351 Closing in on the Mechanisms of Pulsatile Insulin Secretion Richard Bertram, 1 Leslie S. Satin, 2 and Arthur S. Sherman 3 Diabetes 2018;67:351 359 https://doi.org/10.2337/dbi17-0004
Diabetes Volume 67, March 2018 351 Closing in on the Mechanisms of Pulsatile Insulin Secretion Richard Bertram, 1 Leslie S. Satin, 2 and Arthur S. Sherman 3 Diabetes 2018;67:351 359 https://doi.org/10.2337/dbi17-0004
Toluidin-Staining of mast cells Ear tissue was fixed with Carnoy (60% ethanol, 30% chloroform, 10% acetic acid) overnight at 4 C, afterwards
 Toluidin-Staining of mast cells Ear tissue was fixed with Carnoy (60% ethanol, 30% chloroform, 10% acetic acid) overnight at 4 C, afterwards incubated in 100 % ethanol overnight at 4 C and embedded in
Toluidin-Staining of mast cells Ear tissue was fixed with Carnoy (60% ethanol, 30% chloroform, 10% acetic acid) overnight at 4 C, afterwards incubated in 100 % ethanol overnight at 4 C and embedded in
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Electrical activity and exocytotic correlates of biphasic insulin secretion from β-cells of canine islets of Langerhans
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 Electrical activity and exocytotic correlates of biphasic insulin secretion from β-cells of canine islets of
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 Electrical activity and exocytotic correlates of biphasic insulin secretion from β-cells of canine islets of
About This Chapter. Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Pearson Education, Inc.
 About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
Chapter 3 subtitles Action potentials
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
Potentiation of Glucose-stimulated Insulin Secretion by the GPR40 PLC TRPC
 Supplementary information Potentiation of Glucose-stimulated Insulin Secretion by the GPR40 PLC TRPC Pathway in Pancreatic -Cells Authors: Hodaka Yamada 1,*, Masashi Yoshida 1,*, Kiyonori Ito 1, Katsuya
Supplementary information Potentiation of Glucose-stimulated Insulin Secretion by the GPR40 PLC TRPC Pathway in Pancreatic -Cells Authors: Hodaka Yamada 1,*, Masashi Yoshida 1,*, Kiyonori Ito 1, Katsuya
Animal Physiology Study Guide
 Animal Physiology Study Guide 1. Which of the following are an example of passive transport? 2. Which active transport? 3. How can you tell? 1. Which of the following are an example of passive transport?
Animal Physiology Study Guide 1. Which of the following are an example of passive transport? 2. Which active transport? 3. How can you tell? 1. Which of the following are an example of passive transport?
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM.
 Supplementary Figure 1 Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM. (a-c) Heterozygous c.216ga mice displayed normal hair bundle morphology at P18. (d-i) Disorganized hair bundles
Supplementary Figure 1 Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM. (a-c) Heterozygous c.216ga mice displayed normal hair bundle morphology at P18. (d-i) Disorganized hair bundles
FIBER TYPES - oxidative metabolism is the main form here - ATPase activity is relatively low
 Cardiac Muscle Physiology Special characteristics of cardiac muscle - Branching and interdigitating cells - At their ends, they are connected by INTERCALATED DISCS - The discs are always at the Z-lines
Cardiac Muscle Physiology Special characteristics of cardiac muscle - Branching and interdigitating cells - At their ends, they are connected by INTERCALATED DISCS - The discs are always at the Z-lines
Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris
 ZOOLOGICAL SCIENCE 21: 131 138 (2004) 2004 Zoological Society of Japan Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris Kazunori Oami* Institute
ZOOLOGICAL SCIENCE 21: 131 138 (2004) 2004 Zoological Society of Japan Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris Kazunori Oami* Institute
Figure S1. Analysis of genomic and cdna sequences of the targeted regions in WT-KI and
 Figure S1. Analysis of genomic and sequences of the targeted regions in and indicated mutant KI cells, with WT and corresponding mutant sequences underlined. (A) cells; (B) K21E-KI cells; (C) D33A-KI cells;
Figure S1. Analysis of genomic and sequences of the targeted regions in and indicated mutant KI cells, with WT and corresponding mutant sequences underlined. (A) cells; (B) K21E-KI cells; (C) D33A-KI cells;
Glucose homeostasis is supported in a complex. Involvement of K ATP Channels From Both a-cells and
 ORIGINAL ARTICLE Tolbutamide Controls Glucagon Release From Mouse Islets Differently Than Glucose Involvement of K ATP Channels From Both a-cells and d-cells Rui Cheng-Xue, 1 Ana Gómez-Ruiz, 1 Nancy Antoine,
ORIGINAL ARTICLE Tolbutamide Controls Glucagon Release From Mouse Islets Differently Than Glucose Involvement of K ATP Channels From Both a-cells and d-cells Rui Cheng-Xue, 1 Ana Gómez-Ruiz, 1 Nancy Antoine,
STEIN IN-TERM EXAM -- BIOLOGY APRIL 19, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- APRIL 19, 2018 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There is
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- APRIL 19, 2018 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There is
Psych 181: Dr. Anagnostaras
 Psych 181: Dr. Anagnostaras Lecture 5 Synaptic Transmission Introduction to synaptic transmission Synapses (Gk., to clasp or join) Site of action of most psychoactive drugs 6.5 1 Synapses Know basic terminology:
Psych 181: Dr. Anagnostaras Lecture 5 Synaptic Transmission Introduction to synaptic transmission Synapses (Gk., to clasp or join) Site of action of most psychoactive drugs 6.5 1 Synapses Know basic terminology:
