UNDERWRITING GUIDE. Secure Advantage HOSPITAL COVERAGE. The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization
|
|
|
- Everett Bennett Clarke
- 6 years ago
- Views:
Transcription
1 UNDERWRITING GUIDE Secure Advantage HOSPITAL COVERAGE The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization
2 TABLE OF CONTENTS Contents Page Secure Advantage Underwriting Guide...2 General Information...2 Tips For Completing An Application...2 Replacement...3 Submitting The Application...3 Reinstatement Procedures...4 Uninsurable s...4 Cancer s...13
3 SECURE ADVANTAGE UNDERWRITING GUIDE We understand the importance agents place on having applications approved and issued as soon as possible! The Underwriting Division is committed to this goal and we will work with you through every step of the Underwriting process to achieve it. Please review this Underwriting Guide carefully. It has been designed to help you to complete an application and help you understand the process of Underwriting and the procedures used to underwrite an Secure Advantage Cash Supplement policy. GENERAL INFORMATION Eligibility: Age 18 through age 85. The age is determined as of the date the application was signed even if there is an age change before a policy is issued. Age of the application: Applications must be received in the Home Office within 14 days of the date signed. Application Date: The application date MUST be the date the application was signed. Backdated applications will NOT be accepted. Effective Dates: Policy effective dates will usually be the application date. You may request effective dates up to 60 days past the application date. No backdating is allowed (i.e., the effective date can never be before the application date.) Signature on the Application: The agent must personally ask and record all answers to the application questions. No other person, including the spouse, may sign on behalf of an applicant. The agent must personally witness the applicant s signature. We do not accept Power of Attorney signatures. C.O.D Business: Equitable does not accept C.O.D business. TIPS FOR COMPLETING AN APPLICATION It is important to complete the application in its entirety because it becomes the basis for the policy (which is a legal contract). If the applicant answers yes to any of the medical questions, the policy will be declined; do not submit the application. Always: Ask each question, exactly as written (don t paraphrase). Record each answer exactly as given. Complete the application legibly and in black ink. Have each applicant initial and date any corrections or mistakes. Use an additional sheet to record any pertinent information you feel would be helpful in evaluating the risk. Have the applicant sign and date any additional sheets. Page 2
4 TIPS FOR COMPLETING AN APPLICATION (CONT.) Never: Use white out or similar substances for corrections or mistakes. Lead" the applicant when they are responding to a question. Ask a general question (i.e. Are you in good health? ) and then answer all of the medical questions on the application as no. Complete an application by telephone or correspondence. The writing agent must be present at the time of application. Allow someone other than the applicant to answer the application questions, unless it is a parent speaking for a child. Answer questions with ditto marks (") or dashes (-). Answer questions with N/A (not applicable). Use abbreviations unless you are sure they are correct. REPLACEMENT If this policy is replacing any other accident and sickness insurance policy, record the policy number and company name on the application, and complete and include a Replacement Notice with the application. SUBMITTING THE APPLICATION The following is required for the Secure Advantage Cash Supplement application: Page 3 Application Premium (including the $25 application fee) Replacement form if replacing any accident and sickness insurance policy (if required in your state) Notice of Our Privacy Policy/Health Information Authorization (HHA & HPN) Medicare Duplication Notice (MDN-10) There may be other forms not listed above that are required in your state. Drafting the Initial Premium Rather than submitting premium with the application, an agent can request to draft the initial monthly premium in the Premium Payment section of the application. When this is selected, unless indicated otherwise, the first premium will be drafted when the policy is issued. Please note: the first premium is drafted when the policy is issued, regardless if the requested effective date is beyond the issue date. Rather than mailing an application, you can use two other methods to reduce time: 1. Fax the application to Use the Upload Applications feature on front page of the website. This is an easy-to-use feature that allows you to upload a copy of a scanned application. Once we receive the faxed or uploaded application, that application will be processed with that day s business received. The original application and premium must be received at the Home Office within 7 days. Upload Applications We will accept your scanned applications in either TIFF images or PDF's of the application. [ click here for multiple files ] Submit
5 REINSTATEMENT PROCEDURES All policies have a standard 31-day grace period followed by a 15-day conservation period. After 31 days, reinstatement applications will be accepted for up to one hundred and fifty (150) days from the date the policy lapsed. After one hundred and fifty (150) days, a new application will have to be completed and approved before a new policy will be issued. For more information, contact the Policyowner Services at If the applicant has taken any of the medications listed below in the past 2 years for the specific condition listed, do not submit the application. Applicants who have taken any of these medications for the related condition in the past 2 years are automatically declined. UNINSURABLE MEDICATIONS Abacavir sulfate hiv/aids Accolate COPD AccuNeb COPD Accupril Congestive heart failure Accuretic Congestive heart failure Aceon Heart disease Acetazolamide Congestive heart failure Actimmune Organ transplant Adalat Heart disease Adrenalin COPD Advair COPD AeroBid COPD Aerolate COPD Aggrenox CVA (stroke) Airet COPD Akineton Parkinson s Albuterol COPD Aldactazide Congestive heart failure Aldactone Congestive heart failure Altace Congestive heart failure/heart disease Alupent COPD Amantadine Parkinson s Amiloride Congestive heart failure Aminophylline COPD Amiodarone Heart disease Amlodipine Heart disease Amyl Nitrite Heart disease Antabuse Alcoholism Apresoline......Congestive heart failure/heart disease Aprida > 50 units per day diabetes Aptivus HIV/AIDS Aricept Alzheimer s, Dementia Artane Parkinson s Atacand Congestive heart failure Atenolol Congestive heart failure Atripla HIV/AIDS Atrovent COPD Avonex Multiple sclerosis Azasan Organ transplant Azathioprine Organ transplant Azmacort COPD Baclofen Multiple sclerosis Becaplermin Skin ulcer Beclomethasone COPD Beclovent COPD Beconase COPD Bendroflumethiazide Congestive heart failure Benztropine Mesylate Parkinson s Bepridil Heart disease Betapace Heart disease Betaseron Organ transplant Biperiden Parkinson s Bisoprolol Congestive heart failure Blocadren Heart disease Brethine COPD Bricanyl COPD Bromocriptine Parkinson s Brondelate COPD Bronkosol COPD Bumetanide Congestive heart failure Bumex Congestive heart failure Campral Alcohol abuse Capoten Congestive heart failure/heart disease Captopril Congestive heart failure/heart disease Carbidopa Parkinson s Cardene Heart disease Carvedilol......Congestive heart failure/heart disease CellCept Organ transplant Chlorothiazide Congestive heart failure Choledyl COPD Chymotrypsin Organ transplant Cilostazol Peripheral Vascular Disease Page 4
6 UNINSURABLE MEDICATIONS (CONT.) Clopidogrel CVA (stroke) Cogentin Parkinson s Cognex Alzheimer s, Dementia Combivent COPD Combivir HIV/AIDS Comtan Parkinson s Copaxone Multiple sclerosis Cordarone Heart disease Coreg Congestive heart failure/heart disease Corgard Heart disease Corzide Heart disease Coumadin....CVA (stroke)/peripheral vascular disease Covera Heart disease Creon Pancreatitis Crixivan HIV/AIDS Cromolyn COPD Cyclosporine Organ transplant Cymbalta Diabetic neuropathy DG COPD Demadex Congestive heart failure Depade Substance abuse Diamox Congestive heart failure Didanosine HIV/AIDS Digitek Congestive heart failure/heart disease Digoxin Congestive heart failure/heart disease Dilatate Heart disease Dilor COPD Diovan Congestive heart failure Dipradam Heart disease Dipyridamole Heart disease Disulfiram Alcoholism Dofetilide Heart disease Donepezil Alzheimer s/dementia Dopar Parkinson s Duloxetine Diabetic neuropathy DuoNeb COPD Dyflex COPD Dylix COPD Dyphylline COPD Dyrenium COPD Ed-Bron COPD Efavirenz HIV/AIDS Eldepryl Parkinson s Elixophyllin COPD Emtriva HIV/AIDS Enalapril Congestive heart failure Enalaprilat Congestive heart failure Enduron Congestive heart failure Entacapone Parkinson s Ephedrine COPD Epinephrine COPD Epivir HIV/AIDS Epogen Kidney failure Epzicom HIV/AIDS Ergoloid Mesylate Alzheimer s/dementia Esidrix Congestive heart failure Ethmozine Heart disease Exelon Alzheimer s/dementia Felodipine...Peripheral Vascular Disease/CVA (stroke) Flecainide Heart disease Flovent COPD Flumazenil Substance abuse Flunisolide COPD Foradil COPD Fortovase HIV/AIDS Foscarnet HIV/AIDS Fosinopril Congestive heart failure Foscavir HIV/AIDS Furosemide Congestive heart failure Galantamine Alzheimer s/dementia Gengraf Organ transplant Hivid HIV/AIDS Humalog > 50 units per day diabetes Humulin > 50 units per day diabetes Hydergine Alzheimer s/dementia Hydralazine.....Congestive heart failure/heart disease Hydrochlorothiazide Congestive heart failure HydroDIURIL Congestive heart failure Hydropres Congestive heart failure Hylenex Organ transplant Iletin > 50 units per day diabetes Imdur Heart disease Imuran Organ transplant Indapamide Congestive heart failure Inderal Heart disease/congestive heart failure Inderide Heart disease Indinavir HIV/AIDS Infergen Organ transplant InnoPran Heart disease Insulin > 50 units per day diabetes Intal COPD Interfero Organ transplant Page 5
7 UNINSURABLE MEDICATIONS (CONT.) Intron Organ transplant Invirase HIV/AIDS Ipratropium COPD Ismo Heart disease Isoetharine COPD Isoproterenol COPD Isoptin Heart disease Isordil Heart disease Isosorbide Heart disease Isoxsuprine Peripheral Vascular Disease Isuprel COPD Jantoven Peripheral vascular disease/ Heart disease/cva (stroke) Jay-Phyl COPD Kaletra HIV/AIDS Kemadrin Parkinson s Ku-Zyme Pancreatitis Lamivudine HIV/AIDS Lanoxicaps Heart disease/ Congestive heart failure Lanoxin Heart disease/ Congestive heart failure Lantus > 50 units per day diabetes Larodopa Parkinson s disease L-Dopa Parkinson s disease Lente > 50 units per day diabetes Levatol Heart disease Levemir > 50 units per day diabetes Levodopa Parkinson s Lexxel Congestive heart failure Lioresal Multiple sclerosis Lipase Pancreatitis Lipram Pancreatitis Lisinopril Heart disease/ Congestive heart failure Lopinavir HIV/AIDS Lopressor Heart disease Lufyllin COPD Lyrica Diabetic neuropathy Maxair COPD Memantine Alzheimer s/dementia Metaproterenol COPD Methyclothiazide Congestive heart failure Metoprolol Heart disease MicroNefrin COPD Microzide Congestive heart failure Midamor Congestive heart failure Minitran Heart disease Mirapex Parkinson s Moduretic Congestive heart failure Monoket Heart disease Moricizine Heart disease Mycophenolate mofetil organ transplant Myfortic Organ transplant Myotrophin ALS/Lou Gehrig s disease Nadolol Heart disease Naloxone Substance abuse Naltrexone Substance abuse Namenda Alzheimer s/dementia Narcan Substance abuse Nelfinavir HIV/AIDS Neoral Organ transplant Neurontin Neuropathy Nevirapine HIV/AIDS Nicardipine Heart disease Nifediac Heart disease Nifedical Heart disease Nifedipine Heart disease Nitrek Heart disease Nitro-Bid Heart disease Nitro-Dur Heart disease Nitrogard Heart disease Nitroglycerin Heart disease Nitrol Heart disease Nitrong Heart disease Nitro paste heart disease Nitro patch heart disease Nitropress Heart disease NitroQuick Heart disease Nitrostat Heart disease NitroTab Heart disease Nitro-Time Heart disease Norpace Heart disease Norvasc Congestive heart failure Norvir HIV/AIDS Novolin > 50 units per day diabetes NovoLog > 50 units per day diabetes Nylidrin Peripheral Vascular Disease Orphenadrine Parkinson s Orthoclone Organ transplant Pacerone Heart disease Palcaps Pancreatitis Page 6
8 UNINSURABLE MEDICATIONS (CONT.) Pancrease Pancreatitis Pancreatin Pancreatitis Pancrecarb Pancreatitis Pancrelipase Pancreatitis Panfil COPD Panocaps Pancreatitis Panokase Pancreatitis Papaverine Peripheral vascular disease Para-Time Peripheral vascular disease Parlodel Parkinson s Paridol Parkinson s Pentoxifylline Peripheral vascular disease Pergolide Parkinson s Permax Parkinson s Persantine Heart disease Plaretase Pancreatitis Plavix Peripheral vascular disease/ CVA (stroke) Plendil Peripheral vascular disease Pletal Peripheral vascular disease Pramipexole Parkinson s Pregabalin Diabetic neuropathy Prinivil Heart disease/ Congestive heart failure ProAir COPD Procainamide Heart disease Procanbid Heart disease Procardia Heart disease Procyclidine Parkinson s Prograf Organ transplant Pronestyl Heart disease Propafenone Heart disease Propranolol Heart disease Proventil COPD Pulmicort COPD Quibron COPD Quinapril Heart disease Quinidex Heart disease Quinidine Heart disease Qvar COPD Ranexa Heart disease Rapamune Organ transplant Razadyne Alzheimer s/dementia Rebif Organ transplant Regranex Skin ulcer Reminyl Alzheimer s/dementia Renese Kidney failure Requip Parkinson s Rescriptor HIV/AIDS Respbid COPD Retrovir HIV/AIDS Revatio Heart disease Revex Substance abuse ReVia Substance abuse Rilutek ALS/Lou Gehrig s disease Riluzole ALS/Lou Gehrig s disease Ritonavir HIV/AIDS Rivastigmine Alzheimer s/dementia Roferon Organ transplant Romazicon Substance abuse Ropinirole Parkinson s Rythmol Heart disease Saluron Congestive heart failure Sandimmune Organ transplant Saquinavir HIV/AIDS Selegiline Parkinson s Serevent COPD Simulect Organ transplant Sinemet Parkinson s Singulair COPD Slo-Bid COPD Slo-Phyllin COPD Sotalol Heart disease Spiriva COPD Spironolactone Congestive heart failure Stavudin HIV/AIDS Sustiva HIV/AIDS Symmetrel Parkinson s Tacrine Alzheimer s/dementia Tambocor Heart disease Tasmar Parkinson s Tenofovir HIV/AIDS Terbutaline COPD Teveten Congestive heart failure Thalitone Cirrhosis Thalomid Organ transplant Theo COPD Theochron COPD Theo-Dur COPD Theolair COPD Theolate COPD Theomax COPD Theophylline COPD Thymoglobulin Organ transplant Page 7
9 UNINSURABLE MEDICATIONS (CONT.) Tiazac Heart disease Ticlid CVA (stroke) Ticlopidine CVA (stroke) Tikosyn Heart disease Tilade COPD Timolide Congestive heart failure/heart disease Tolcapone Parkinson s Toprol Heart disease Tornalate COPD Torsemide Congestive heart failure T-Phyl COPD Tracleer Heart disease Trental Peripheral vascular disease Triamcinolone COPD Trihexyphenidyl Parkinson s Trizivir HIV/AIDS Truvada HIV/AIDS Xolair COPD Xopenex COPD Zalcitabine HIV/AIDS Zaroxolyn Congestive heart failure Zebeta Congestive heart failure Zenapax Organ transplant Zerit HIV/AIDS Ziac Congestive heart failure Ziagen HIV/AIDS Zidovudine HIV/AIDS Zyflo COPD Ultra caps pancreatitis Ultrase Pancreatitis Uniphyl COPD Uniretic Congestive heart failure Valcyte HIV/AIDS Valsartan Congestive heart failure Vanceril COPD Vascor Heart disease Vasodilan Peripheral Vascular Disease Vasotec Congestive heart failure Veldona Organ transplant Velosulin > 50 units per day diabetes Ventavis Heart disease Ventolin COPD Verapamil Heart disease Verelan Heart disease Videx HIV/AIDS Viracept HIV/AIDS Viokase Pancreatitis Viramune HIV/AIDS Viread HIV/AIDS Vitrase Organ transplant Vivitrol Substance abuse Volmax COPD VoSpire COPD Warfarin.....Peripheral vascular disease/cva (stroke) Wellferon HIV/AIDS Wydase Organ transplant Page 8
10 CANCER MEDICATIONS If the applicant has taken any of the medications listed below for the treatment of cancer in the past 2 years, do not submit the application. Applicants who have taken any of these medications in the past 2 years are automatically declined. If the applicant is applying for the First Occurrence Cancer Rider, and has taken any of the medications listed below for the treatment of cancer in the past 10 years, do not apply for the First Occurence Cancer Rider. Applicants who have taken any of these medications in the past 10 years are not eligible for the First Occurrence Cancer Rider. Abarelix Abraxane Adriamycin Adrucil Aldesleukin Alferon Alimta Alkeran Altretamine Anastrozole Aredia Arimidex Aromasin Arranon Arsenic trioxide Asparaginase Avastin BCG Bevacizumab Bexarotene Bexxar Bicalutamide BiCNU Blenoxane Bleomycin Campath Camptosar Capecitabine Capromab pendetide Carac Carboplatin Carmustine Casodex CeeNU Cerubidine Cetuximab Cinacalcet Cisplatin Cladribine Clolar Cosmegen Cytarabine Page 9 Cytomel Cytosar Cytoxan Dacarbazine Dactinomycin Dasatinib DaunoXome Daunorubicin Delatestryl Denileukin Diftitox DepoCyt Docetaxel Doxil Doxorubicin DTIC-dome Efudex Eligard Ellence Eloxatin Elspar Emcyt Epirubicin Erbitux Erlotinib Estramustine Etopophos Etoposide Eulexin Exemestane Fareston Faslodex Femara Floxuridine Fludara Fludarabine phosphate Fluoroplex Fluorouracil Flutamide FUDR Fulvestrant Gefitinib Gemcitabine Gemzar Gleevec Gliadel Goserelin Herceptin Hexalen Histrelin Hycamtin Hydrea Hydroxyurea Idamycin Idarubicin Ifex Ifex/Mesnex Ifosfamide Imatinib Interferon Intron Iressa Irinotecan Ketoconazole Lapatinib Letrozole Leucovorin Leukeran Leuprolide Leustatin Levamisole Liothyronine Liotrix Lomustine Lupron Lysodren Matulane Mechlorethamine Megace Megestrol Melphalan Menest Meprolone Mercaptopurine Methitest Methotrexate Methoxsalen Mithracin Mitomycin Mitoxantrone Mustargen Mutamycin Mylocel Mylotarg Navelbine Neosar Neulasta Nexavar Nilandron Nilutamide Nipent Nizoral Nolvadex Novantrone Octreotide Oncaspar Ontak Onxol Oxaliplatin Paclitaxel Pamidronate Panretin Paraplatin Pegfilgrastim Pemetrexed Photofrin Plenaxis Porfimer
11 Page 10 CANCER MEDICATIONS (CONT.) Prelone Procarbazine Proleukin ProstaScint Purinethol Quadramet Raloxifene Rheumatrex Rituximab Rituxan Roferon Rubex Samarium Sensipar Soltamox Sorafenib Sprycel Stilphostrol Streptozocin Sunitinib Supprelin Sutent Tamoxifen Tarceva Targretin Taxol Taxotere Temodar Temozolomide Temsirolimus Teniposide Teslac Testolactone Testopel TheraCys Thiotepa Thyrogen Thyrolar Thyrotropin Tice BCG Toposar Topotecan Toremifene Trastuzumab Trelstar Tretinoin Trexall Triamcinolone Triptorelin pamoate Trisenox Tykerb Uvadex Vantas Velcade VePesid Vesanoid Viadur Vinblastine Vincasar Vincristine Vinorelbine Vumon Xeloda Zanosar Zevalin Zoladex Zoledronic acid Zometa
12 We re here to serve you! Policyowner Service PO Box 2878, Salt Lake City, Utah
EquiCash UNDERWRITING GUIDE. Equitable &You. The Policy That Pays You Cash In The Event You Have To Be Hospitalized HOSPITAL PLAN
 UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
Acute Lymphocytic Leukemia
 Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Formulary Chemotherapy Agents: (Current as of 6/2018) Therapeutic Class
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
HCPCS Code/ generic (Brand) Name J7506. J8520 capecitabine (Xeloda) 1. J8521 capecitabine. J8530 cyclophosphamide. (Cytoxan) 1
 drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
Chemotherapy 101 for Radiation Oncology Workers
 Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
To help doctors give their patients the best possible care, the American
 Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced
 ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
Form 2023 R2.0: Ovarian Cancer Pre-HSCT Data
 Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Introduction to Antineoplastic Prescribing
 Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites.
 WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
MABCAMPATH mg 3 INJ GENZYME BIOPHARMACEUTICALS. MABCAMPBATH mg 3 INJ GENZYME BIOPHARMACEUTICALS
 ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
Ovarian Cancer. compendia TREATMENT OF
 drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
FIELD GUIDE AND DRUG LIST
 FIELD GUIDE AND DRUG LIST CONTINENTAL CARE INSURANCE Continental Life Insurance Company of Brentwood, Tennessee (CLI) An Aetna Company CLICC01414 090712 FIELD GUIDE Always take enough time with every applicant
FIELD GUIDE AND DRUG LIST CONTINENTAL CARE INSURANCE Continental Life Insurance Company of Brentwood, Tennessee (CLI) An Aetna Company CLICC01414 090712 FIELD GUIDE Always take enough time with every applicant
Guidelines on Chemotherapy-induced Nausea and Vomiting in Pediatric Cancer Patients
 Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
2. Date of most recent breast cancer diagnosis / / 3. Pathlogy: Padget s Phyllodes Tubular Medullary Mucinous
 Breast Cancer Patients: Begin on page 1, then skip to page 3 Lymphoma Patients: Begin on page 2 1. Newly diagnosed with Breast carcinoma Stage: (includes inflammatory breast and newly diagnosed recurrent
Breast Cancer Patients: Begin on page 1, then skip to page 3 Lymphoma Patients: Begin on page 2 1. Newly diagnosed with Breast carcinoma Stage: (includes inflammatory breast and newly diagnosed recurrent
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting
 West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
Chemotherapy and biotherapy: guidelines and recommendations for practice.
 Complete Summary GUIDELINE TITLE Chemotherapy and biotherapy: guidelines and recommendations for practice. BIBLIOGRAPHIC SOURCE(S) Oncology Nursing Society (ONS). Chemotherapy and biotherapy: guidelines
Complete Summary GUIDELINE TITLE Chemotherapy and biotherapy: guidelines and recommendations for practice. BIBLIOGRAPHIC SOURCE(S) Oncology Nursing Society (ONS). Chemotherapy and biotherapy: guidelines
Bladder Cancer Pathways (Urothelial)
 Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer (Urothelial) Pathways
 Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Objectives. Principles of Cancer Treatment. Cancer Treatment Modalities. Cancer Treatment Modalities
 Objectives Principles of Cancer Treatment Juanita Madison, RN, MN, AOCN Oncology Clinical Nurse Specialist Seattle Cancer Care Alliance Describe the principles of cancer treatment Surgery Chemotherapy
Objectives Principles of Cancer Treatment Juanita Madison, RN, MN, AOCN Oncology Clinical Nurse Specialist Seattle Cancer Care Alliance Describe the principles of cancer treatment Surgery Chemotherapy
Leukemia. Treatment of. compendia. Associated ICD-9-CM Codes: Drug & Administration. managedcareoncology.com
 Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
MEDICAL NECESSITY GUIDELINE
 PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data
 Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
UP Health System Marquette Medication Guideline High Alert Drugs
 1 ALTEPLASE SOLR 100 MG ACTIVASE 305010 50242008527 TRUE ALTEPLASE SOLR 50 MG ACTIVASE 305008 50242004413 TRUE AMIODARONE INJ 50 MG/ML CORDARONE IV 305035 63323061609 TRUE AMIODARONE INJ 50 MG/ML CORDARONE
1 ALTEPLASE SOLR 100 MG ACTIVASE 305010 50242008527 TRUE ALTEPLASE SOLR 50 MG ACTIVASE 305008 50242004413 TRUE AMIODARONE INJ 50 MG/ML CORDARONE IV 305035 63323061609 TRUE AMIODARONE INJ 50 MG/ML CORDARONE
REGULATORY INFORMATION
 REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
SCI. SickKids-Caribbean Initiative Enhancing Capacity for Care in Paediatric Cancer and Blood Disorders
 1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A
 Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
Primary malignant neoplasms, not lymphatic or hematopoietic. Secondary malignant neoplasms (i.e.metastatic) Malignant neoplasm, unknown site
 Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Committee Approval Date: December 12, 2014 Next Review Date: July 2015
 Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Who is Bearing the Cost?
 THE COS ST T OF CANCE R DRUGS IN CANADA PA R T 2 Who is Bearing the Cost? KONG KHOO, ROSEMARY COLUCCI, WILLIAM HRYNIUK, ROBERT KAMINO, TANIA REDINA AND COLLEEN SAVAGE Introduction Last year we reported
THE COS ST T OF CANCE R DRUGS IN CANADA PA R T 2 Who is Bearing the Cost? KONG KHOO, ROSEMARY COLUCCI, WILLIAM HRYNIUK, ROBERT KAMINO, TANIA REDINA AND COLLEEN SAVAGE Introduction Last year we reported
Overview of Cancer. Laura Bingell RN Transition Center Nurse for MFP (607)
 Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
COST CONSIDERATIONS Union for International Cancer Control 2014 Review of Cancer Medicines on the WHO List of Essential Medicines!!!!!!!!!
 UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
Cancer Drugs CANCER DRUGS NEW DRUGS AND INDICATIONS OVER THE PAST TEN YEARS
 Cancer Drugs NEW DRUGS AND INDICATIONS OVER THE PAST TEN YEARS by KONG KHOO, ROSEMARY COLUCCI, DANIEL GILLESPIE, JAMES D. GOWING, PIERRE MAJOR, JOSEPH RAGAZ, DAVID SALTMAN, COLLEEN SAVAGE Development of
Cancer Drugs NEW DRUGS AND INDICATIONS OVER THE PAST TEN YEARS by KONG KHOO, ROSEMARY COLUCCI, DANIEL GILLESPIE, JAMES D. GOWING, PIERRE MAJOR, JOSEPH RAGAZ, DAVID SALTMAN, COLLEEN SAVAGE Development of
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Prior Authorization Proposal: Oncology Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Fundamentals of Pharmacology for Veterinary Technicians Chapter 20
 Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Hazard Meds: Navigating the Warning Beacons
 Hazard Meds: Navigating the Warning Beacons Disclosures I have nothing to disclose at this time. Mark Twohey, Pharm.D., BCOP Palmetto Health Richland March 24, 2012 Objectives Understand the reasons for
Hazard Meds: Navigating the Warning Beacons Disclosures I have nothing to disclose at this time. Mark Twohey, Pharm.D., BCOP Palmetto Health Richland March 24, 2012 Objectives Understand the reasons for
Chemotherapy Agents 101
 Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
ALKYLATING AGENTS (22)
 Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Haematology, Oncology and Palliative Care Directorate.
 Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Azacitidine Vidaza Non-transplant myelodysplastic syndrome Funded Funded Funded Funded Funded Funded Not Funded
 Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
1 The Cancer Programs Regulation (AR 242/98) is amended by this Regulation.
 Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
DOXOrubicin, Cyclophosphamide (AC 60/600) 21 day followed by weekly PACLitaxel (80) Therapy (AC-T) 261 CARBOplatin (AUC4-6) Monotherapy-21 days
 Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Protocol Number Tumour Group Protocol Name on NCCP website 22/02/ Lung Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy
 Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Cancer, Heart Attack and Stroke Insurance
 Cancer, Heart Attack and Stroke Insurance AGENT RATE AND UNDERWRITING GUIDE Basic - Monthly AL, DC, ID, IL, IA, KS, KY, LA, ME, MI, MS, NE, NM, NC, ND, OH, OK, PA, SC, TX, WV, WI FOR AGENT USE ONLY UNDERWRITTEN
Cancer, Heart Attack and Stroke Insurance AGENT RATE AND UNDERWRITING GUIDE Basic - Monthly AL, DC, ID, IL, IA, KS, KY, LA, ME, MI, MS, NE, NM, NC, ND, OH, OK, PA, SC, TX, WV, WI FOR AGENT USE ONLY UNDERWRITTEN
Cancer, Heart Attack and Stroke Insurance
 Cancer, Heart Attack and Stroke Insurance AGENT RATE AND UNDERWRITING GUIDE CONNECTICUT - MONTHLY FOR AGENT USE ONLY UNDERWRITTEN BY: Guarantee Trust Life Insurance Company GUARANTEE TRUST LIFE INSURANCE
Cancer, Heart Attack and Stroke Insurance AGENT RATE AND UNDERWRITING GUIDE CONNECTICUT - MONTHLY FOR AGENT USE ONLY UNDERWRITTEN BY: Guarantee Trust Life Insurance Company GUARANTEE TRUST LIFE INSURANCE
DRUG PROPERTIES YOU NEED TO KNOW
 jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
DRUG PROPERTIES YOU NEED TO KNOW
 Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19
 Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249
 Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Anastrozole. Active Ingredient. Brand Name: Arimidex Strength: 1 mg Dosage Form: Tablets Pack Size: 28 Tabs Company: AstraZeneca
 Super Specialty Drugs by Anastrozole Arimidex 1 mg 28 Tabs AstraZeneca Armotraz 1 mg 10 Tabs Cipla Anastrozole Anabrez 1 mg 100 Tabs Sun Decitabine Dacogen 50 mg 1 Vial Eisai Inc. Erlotinib Erlocip-150
Super Specialty Drugs by Anastrozole Arimidex 1 mg 28 Tabs AstraZeneca Armotraz 1 mg 10 Tabs Cipla Anastrozole Anabrez 1 mg 100 Tabs Sun Decitabine Dacogen 50 mg 1 Vial Eisai Inc. Erlotinib Erlocip-150
3/8/2018. Treatment Modalities. Study Resources. Study Resources. New 2018 OCN Test Blueprint
 New 2018 OCN Test Blueprint Treatment Modalities Nancy Thompson, MSN, RN, AOCNS Director of Quality & Clinical Practice Swedish Cancer Institute March 17, 2018 Content Area Sub-Content Areas 2018 Test%
New 2018 OCN Test Blueprint Treatment Modalities Nancy Thompson, MSN, RN, AOCNS Director of Quality & Clinical Practice Swedish Cancer Institute March 17, 2018 Content Area Sub-Content Areas 2018 Test%
Medications currently available to treat Multiple Myeloma include: Current Code Price (AWP) Effective Date. Code Price (AWP)
 drug and administration compendia Compendia information developed and maintained by With each publication Managed Care Oncology s Drug & Compendia will highlight a single medication or a group of medications
drug and administration compendia Compendia information developed and maintained by With each publication Managed Care Oncology s Drug & Compendia will highlight a single medication or a group of medications
Description The following are synthetic cannabinoids requiring prior authorization: dronabinol (Marinol, Syndros ), nabilone (Cesamet )
 Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
A themed review of patient safety incidents involving anti-cancer medicines 1 November June 2008
 A themed review of patient safety incidents involving anti-cancer medicines 1 November 2003 30 June 2008 October 2010 Full Report National Patient Safety Agency 2010. Copyright and other intellectual property
A themed review of patient safety incidents involving anti-cancer medicines 1 November 2003 30 June 2008 October 2010 Full Report National Patient Safety Agency 2010. Copyright and other intellectual property
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care
 Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Disclosure. Objectives. Objectives. Oncologic Emergency. Classification of Emergencies 1 3/2/2015
 Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer
 APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
FREEDOM OF INFORMATION SOUTH EAST SCOTLAND CANCER NETWORK
 Dear Date 06/02/09 Your Ref Our Ref RM/1220 Enquiries to Richard Mutch Extension 89441 Direct Line 0131-536-9441 Direct Fax 0131-536-9009 Email richard.mutch@lhnhslothian.scot.nhs.uk FREEDOM OF INFORMATION
Dear Date 06/02/09 Your Ref Our Ref RM/1220 Enquiries to Richard Mutch Extension 89441 Direct Line 0131-536-9441 Direct Fax 0131-536-9009 Email richard.mutch@lhnhslothian.scot.nhs.uk FREEDOM OF INFORMATION
Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic Therapy
 Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
Clinical Policy: Pemetrexed (Alimta) Reference Number: CP.PHAR.368 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Positively Affecting the Lives of Members Each and Every Day. Volume 14 May Specialty Drug News
 Positively Affecting the Lives of Members Each and Every Day LDI Volume 14 May 2008 Specialty Drug News 2008 Medications to Watch 1. Respiratory syncytial virus (RSV) Numax (motavizumab) Respiratory syncytial
Positively Affecting the Lives of Members Each and Every Day LDI Volume 14 May 2008 Specialty Drug News 2008 Medications to Watch 1. Respiratory syncytial virus (RSV) Numax (motavizumab) Respiratory syncytial
Adjuvant Systemic Therapy in Early Stage Breast Cancer
 Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
AD regimen, 576 adrenal steroid inhibitor. aminoglutethimide, 23 25
 A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
Hazardous Medication List
 Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
InterChina Insight. Partnering up to fight cancer. Strategies for the China oncology market. InterChina Consulting. By Rick Woo April 03, 2013
 Strategies for the China oncology market By Rick Woo InterChina Consulting 英特华投资咨询有限公司 Beijing Shanghai Madrid Washington DC Strategy M&A Advisory IMAP China 2013 InterChina Consulting All Rights Reserved
Strategies for the China oncology market By Rick Woo InterChina Consulting 英特华投资咨询有限公司 Beijing Shanghai Madrid Washington DC Strategy M&A Advisory IMAP China 2013 InterChina Consulting All Rights Reserved
Access To Cancer Drugs In Medicare Part D: Formulary Placement And Beneficiary Cost Sharing In 2006
 Access To Cancer Drugs In Medicare Part D: Formulary Placement And eneficiary Cost Sharing In 2006 Medicare Part D beneficiaries have access to many more cancer drugs than ever before, with low cost sharing.
Access To Cancer Drugs In Medicare Part D: Formulary Placement And eneficiary Cost Sharing In 2006 Medicare Part D beneficiaries have access to many more cancer drugs than ever before, with low cost sharing.
MASCC Guidelines for Antiemetic control: An update
 MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
Subject: Palonosetron Hydrochloride (Aloxi )
 09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
Guidelines for the Use of Anti-Emetics with Chemotherapy
 Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Emetogenicity level 1. Emetogenicity level 2
 Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Genetics in Cancer Therapy. Raju Kucherlapati, Ph.D. Harvard Medical School
 Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY CLASSIFICATION
 GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and
 35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
Topical Immunomodulators
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Topical Immunomodulators Clinical Edit Information Included in this Document Topical Immunomodulators Elidel and Protopic 0.03%
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Topical Immunomodulators Clinical Edit Information Included in this Document Topical Immunomodulators Elidel and Protopic 0.03%
INTRAVENOUS ASCORBATE AND ONCOLOGIC AGENTS
 PAUL S. ANDERSON 2013 INTRAVENOUS ASCORBATE AND ONCOLOGIC AGENTS Updated Data Review and Policies for concurrent use at Anderson Medical Specialty Associates, Southwest College of Naturopathic Medicine
PAUL S. ANDERSON 2013 INTRAVENOUS ASCORBATE AND ONCOLOGIC AGENTS Updated Data Review and Policies for concurrent use at Anderson Medical Specialty Associates, Southwest College of Naturopathic Medicine
Supplementary Figures
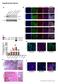 Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY
 ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
DRUGS YOU NEED TO KNOW
 jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
PHARMACY PEARLS CHANIN WRIGHT, PHARMD
 PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
Supplementary Table 1. Summary of FDA approval data for rare cancer indications from December 1987 to May 2011
 Rare Cancer : Lessons from FDA s by Gaddipati et al. Supplementary Table 1 Summary of FDA approval data for rare cancer indications from December 1987 to May 2011 1. mitoxantrone hydrochloride (NOVANTRONE
Rare Cancer : Lessons from FDA s by Gaddipati et al. Supplementary Table 1 Summary of FDA approval data for rare cancer indications from December 1987 to May 2011 1. mitoxantrone hydrochloride (NOVANTRONE
Managements of Chemotherpay Induded Nausea and Vomiting
 REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
Standard Breast Cancer Therapy
 Pt s Who Don t Follow Treatment Standard Breast Cancer Therapy Deborah Thames R.T. (R)(M)(QM) Advancements in Breast Cancer Advancements in Breast Cancer Chemotherapy Full potential was recognized in the
Pt s Who Don t Follow Treatment Standard Breast Cancer Therapy Deborah Thames R.T. (R)(M)(QM) Advancements in Breast Cancer Advancements in Breast Cancer Chemotherapy Full potential was recognized in the
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment
 Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Development trends for new cancer therapeutics and vaccines
 REVIEWS Drug Discovery Today Volume 13, Numbers 1/2 January 2008 Development trends for new cancer therapeutics and vaccines Janice M. Reichert and Julia B. Wenger Tufts Center for the Study of Drug Development,
REVIEWS Drug Discovery Today Volume 13, Numbers 1/2 January 2008 Development trends for new cancer therapeutics and vaccines Janice M. Reichert and Julia B. Wenger Tufts Center for the Study of Drug Development,
1. Purpose Documentation Description.1 4. References 8 5. Cross-References.8 6. Development of the guideline.8
 CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
Appendix 2. Adjuvant Regimens. AC doxorubin 60 mg/m 2 every 3 weeks x 4 cycles Cyclophosphamide 600 mg/m 2
 Appendix 2 Adjuvant Regimens AC doxorubin 60 mg/m 2 every 3 weeks x 4 cycles Cyclophosphamide 600 mg/m 2 CMF IV cyclophosphamide 600 mg/m 2 days 1 & 8 every 4 weeks methotrexate 40 mg/m 2 for 6 cycles
Appendix 2 Adjuvant Regimens AC doxorubin 60 mg/m 2 every 3 weeks x 4 cycles Cyclophosphamide 600 mg/m 2 CMF IV cyclophosphamide 600 mg/m 2 days 1 & 8 every 4 weeks methotrexate 40 mg/m 2 for 6 cycles
OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY)
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
Antineoplastic Agents
 56 Antineoplastic Agents Branimir I. Sikic DRUG LIST GENERIC NAME PAGE GENERIC NAME PAGE Aldesleukin 652 Asparaginase 649 Bleomycin 647 Buserelin 650 Busulfan 642 Carboplatin 652 Carmustine 641 Chlorambucil
56 Antineoplastic Agents Branimir I. Sikic DRUG LIST GENERIC NAME PAGE GENERIC NAME PAGE Aldesleukin 652 Asparaginase 649 Bleomycin 647 Buserelin 650 Busulfan 642 Carboplatin 652 Carmustine 641 Chlorambucil
