A cis Element within Flowering Locus T mrna Determines Its Mobility and Facilitates Trafficking of Heterologous Viral RNA
|
|
|
- Esther King
- 5 years ago
- Views:
Transcription
1 JOURNAL OF VIROLOGY, Apr. 2009, p Vol. 83, No X/09/$ doi: /jvi Copyright 2009, American Society for Microbiology. All Rights Reserved. A cis Element within Flowering Locus T mrna Determines Its Mobility and Facilitates Trafficking of Heterologous Viral RNA Chunyang Li, 1 Ke Zhang, 1,2 Xianwu Zeng, 1,2 Stephen Jackson, 1 Yu Zhou, 1,2 and Yiguo Hong 1 * Warwick HRI, University of Warwick, Warwick CV35 9EF, United Kingdom, 1 and Chengdu Institute of Biological Products, Chengdu, Sichuan , China 2 Received 11 November 2008/Accepted 22 January 2009 The Arabidopsis flowering locus T (FT) gene encodes the mobile florigen essential for floral induction. While movement of the FT protein has been shown to occur within plants, systemic spread of FT mrna remains to be unequivocally demonstrated. Utilizing novel RNA mobility assay vectors based on two distinct movementdefective viruses, Potato virus X and Turnip crinkle virus, and an agroinfiltration assay, we demonstrate that nontranslatable FT mrna, independent of the FT protein, moves throughout Nicotiana benthamiana and mutant Arabidopsis plants and promotes systemic trafficking of viral and green fluorescence protein RNAs. Viral ectopic expression of FT induced flowering in the short-day N. tabacum Maryland Mammoth tobacco under long-day conditions. Recombinant Potato virus X bearing FT RNA spread and established systemic infection more quickly than the parental virus. The cis-acting element essential for RNA movement was mapped to the nucleotides 1 to 102 of the FT mrna coding sequence. These data demonstrate that a plant self-mobile RNA molecule can mediate long-distance trafficking of heterologous RNAs and raise the possibility that FT RNA, along with the FT protein, may be involved in the spread of the floral stimulus throughout the plant. RNA trafficking plays an important role in systemic signaling that controls plant development and defense against pathogen infection (25). Hundreds of RNA transcripts have been recently identified in phloem, suggesting phloem-mobile RNAs may act as long-distance signaling molecules in plants (4, 8). Indeed, systemic movement of a homeobox fusion transcript and gibberellic acid-insensitive RNA regulates leaf architecture (13, 19), a non-cell-autonomous mobile RNA represents a long-distance signal that modulates potato tuber formation (3), and small interfering RNAs are components of intercellular and systemic mobile signals for innate RNA silencing defense (9, 12, 14). RNA trafficking is also critical for plant viruses and viroids to establish systemic infection. It has been demonstrated that an RNA motif directs long-distance trafficking of a small naked RNA viroid (33, 44, 46). Moreover, a short RNA sequence is found to be involved in cell-to-cell movement of a plant viral RNA (24), and replication-independent viral RNA can move over long distances in plants (11). In floral induction, the mobile florigen is encoded by the Arabidopsis flowering locus T (FT) gene. FT transcribes mrna in the leaf, but its encoded FT protein functions in the shoot apices where flowers develop (1, 2, 40). The Arabidopsis FT protein and its orthologues have been shown to be involved in long-distance signaling in floral induction (7, 18, 22, 23, 29, 30, 37). However, whether FT mrna is also capable of systemic spread remains to be demonstrated. We describe novel approaches which show that not only does FT RNA move over * Corresponding author. Mailing address: Warwick HRI, University of Warwick, Wellesbourne, Warwick CV35 9EF, United Kingdom. Phone: Fax: C.L. and K.Z. contributed equally to this study. Published ahead of print on 4 February long distances but, remarkably, also facilitates the systemic spread of heterologous green fluorescent protein (GFP) mrna and different viral RNAs in plants. The FT RNA movement does not rely on the expression of the FT protein. The FT RNA mobility is determined by a cis-acting element localized within nucleotides 1 to 102 of the FT mrna coding sequence. MATERIALS AND METHODS Construction of RMA vectors. The wild-type and mutant Arabidopsis FT genes were reverse transcription-pcr (RT-PCR) amplified using Pfu DNA polymerase and the primers PP354/PP356 or PP356/PP355, digested with BspEI and SalI, and cloned in-frame fused to the GFP coding sequence in the BspEI/SalI sites of PVX and PVX/GFP (38) to produce PVX/FT, PVX/mFT, PVX/GFP-FT, and PVX/GFP-mFT, respectively. Plasmid DNA of PVX/GFP, PVX/GFP-FT, and PVX/GFP-mFT were then digested with SalI and XhoI to remove the coat protein (CP) gene subgenomic RNA promoter and the CP gene and self-ligated to produce PVX/GFP CP, PVX/GFP-FT CP, and PVX/GFP-mFT CP. Expression of the GFP gene from PVX/GFP CP and the wild-type and mutated GFP-FT fusion gene from PVX/GFP-FT CP and PVX/GFP-mFT CP were under the control of an engineered CP subgenomic RNA promoter. For construction of TCV-based RNA mobility assay (RMA) vectors TCV/mFT CP, TCV/GFP-FT CP, and TCV/GFP-mFT CP, the Arabidopsis FT gene was RT- PCR amplified using Pfu DNA polymerase and the primers PP406 or PP407 and PP408, digested with BclI and PmeI, and cloned into the BglII/PmeI sites of TCV/ CP (34) or TCV/GFP CP (47). Using a similar PCR and cloning strategy, a series of TCV/trFT CP-based RMA vectors carrying truncated (tr) FT for mapping the cis-acting element required for FT RNA trafficking were constructed. All RMA constructs were confirmed by nucleotide sequencing. The primers used for the construction of RMA vectors are listed in Table 1. RMAs. RNA transcripts from each movement-deficient recombinant PVX and TCV vector were produced by in vitro transcription as previously described (34), pretreated with RNase-free DNase (Promega), and then mechanically inoculated onto N. benthamiana or A. thaliana ft-10 mutant plants at 5 to 6 or 15 to 16 leaves, respectively, in repeated experiments. N. benthamiana plants aged at only 24 days after sowing seeds and were too young to initiate flowering. For PVXbased RMA, total RNAs (50 ng) extracted from inoculated and newly growing young leaves separately collected at 7 days postinoculation (dpi) were pretreated with RNase-free DNase (Promega) and used for RT-PCR detection (30 cycles) (34) with the primers PP354 and PP356 for FT, PP371 and PP372 for GFP, PP
2 VOL. 83, 2009 cis-acting ELEMENT-MEDIATED FT RNA 3541 TABLE 1. Primers used in this study Primer Sequence (5 3 ) a Origin and modification PP82 CAGTGTTGGCTTGCAAACTAG PVX PP228 GGGTAAGTTTTCCGTATGTTG GFP PP267 AGAAAAGTCATGAAGGTTCTGCTAGCC TCV ACGG PP269 CAAGGTGCGCGAGGTTTACCAATC PVX PP271 CGGCTACCACATCCAAGGAAGG 18S rrna PP272 GAGCTGGAATTACCGCGGCTG 18S rrna PP354 TCAAGATCCGGAATGTCTATAAATATAAG FT, BspEI AGACCC PP355 TCAAGATCCGGATAGTCTATAAATATAAG FT, BspEI AGACCC PP356 AGGAAGAAGTCGACTAAAGTCTTCTTCCTC FT, SalI CGCAG PP371 ATGAGTAAAGGAGAAGAACTTTTCACT GFP GGAG PP372 TTTGTGTCCAAGAATGTTTCCATCTTC GFP PP373 GTATGCTGTTTCCGTTGTGATCTCTGTGAG PVX PP407 TTCAAGTGATCAGTAGTCTATAAATATAAG FT, BclI AGAC PP408 AAGGAAGTTTAAACCTAAAGTCTTCTTCCT FT, PmeI CCGCA PP433 ATGCCTCTCCTACATACACTCAACACAG TCV PP434 GTCTACGACATGTCTCTCTTTGCAG TCV PP473a TTGGCCATAAGTAACCTTTAG FT PP512 TCAAGATGATCATAGTCTATAAATATAAGA FT, BclI GACCC PP513 TTAGTCACGTTTAAACGGCCATAAGTAACC FT, PmeI TTTAG PP514 GTTACTTGATCATAGAGAGAGGTGACTAA FT, BclI TGGCTT PP515 AGGAAGTTTAAACCTAAAGTCTTCTTCCTC FT, PmeI CGCAG PP516 AGATCCGTTTAAACCAAAGTATAGAAGTTC FT, PmeI CTGAG PP517 AACTTCTGATCATGAGTTATGGTGGATCCA FT, BclI GATG PP518 ATCTCAGTTTAAACGGTTGTTCCAGTTGTA FT, PmeI GCAGG PP519 ACAACTTGATCATAGTTTGGCAATGAGATT FT, BclI GTGTG PP520 CACCCTGGTTTAAACACTGTTTGCCTGCCA FT, PmeI AGCTG PP521 GGCAGGTGATCATGATATGCACCAGGGTG GCGCCA FT, BclI a Introduced restriction endonuclease sites are in boldface, and the start and mutated nonsense stop codons are underlined. and PP373 for PVX, and PP271 and PP272 for 18S rrna. Epidermal cells with GFP expression and the movement of GFP-tagged viruses were examined with a Zeiss Axiophot microscope with filters for GFP (excitation, 450 to 490 nm; long-pass emission, 520 nm) or under long-wavelength UV light (Upland UVP model B 100AP) through a yellow filter (Kodak no. 58) and photographed with a Nikon CoolPix 995 digital camera (38). For TCV-based RMAs, inoculated, newly growing young leaves and shoot apices were carefully collected at 7 dpi from mock- and virus-inoculated N. benthamiana and mutant ft-10 A. thaliana plants and used for total RNA extraction. Total RNAs were treated with RNase-free DNase (Promega), and RT-PCR (30 cycles) detection was performed with the primers PP354 and PP356 for FT, PP371 and PP372 for GFP, PP433 and PP434 for TCV RNA, PP267 and PP473a for TCV-mFT RNA, PP267 and PP228 for TCV-GFP RNA, and PP271 and PP272 for 18S rrna. The resultant specific TCV-mFT RT-PCR products were purified and verified by direct sequencing. In two separate mapping experiments, plants were inoculated with recombinant viral RNAs that were produced by in vitro transcription from each TCV/ trft CP-based RMA vectors and pretreated with RNase-free DNase (Promega). Total RNAs were extracted at 7 dpi from inoculated and uninoculated young leaves, pretreated with RNase-free DNase (Promega), and used for RT- PCR (30 cycles) assays with primer sets listed in Table 2. Transient agroinfiltration assay of RNA mobility. The GFP-FT and GFP-mFT fusion genes were isolated from PVX/GFP-FT and PVX/GFP-mFT and inserted into between the Cauliflower mosaic virus 35S promoter and terminator-poly(a) signal sequence of a binary vector in Agrobacterium tumefaciens LBA4404 (34) to produce 35S-GFP-FT-poly(A) and 35S-GFP-mFT-poly(A). In two experiments, TABLE 2. Mapping cis-acting element for FT RNA movement RMA vector a FT RNA 5 3 coordinates Primer pair used for: PCR and cloning leaves of 24-day-old N. benthamiana plants were syringe infiltrated with Agrobacterium cultures carrying 35S-GFP-poly(A) (34), 35S-GFP-FT-poly(A) and 35S- GFP-mFT-poly(A). At 4 days postinfiltration, agroinfiltrated leaf tissues and noninfiltrated newly developed young leaves were collected for protein and RNA analysis. Induction of flowering by viral transient expression of the Arabidopsis FT protein. In three experiments, young short-day (SD) N. tabacum Maryland Mammoth plants were mock inoculated or infected with PVX/FT, PVX/mFT, PVX/ GFP, or PVX/GFP-FT and maintained in an insect-free containment glasshouse at 25 C with a long-day (LD; 16-h) photoperiod. Local and systemic symptoms, including chlorotic lesions on inoculated leaves and mild chlorosis on young leaves, developed after 7 to 14 dpi. All PVX/FT-infected plants started to shoot at 20 dpi and flowered at 35 dpi; this was photographically recorded with a Nikon digital CoolPix 995 camera. Total RNAs (50 ng) extracted from systemically infected young leaves collected at 42 dpi were treated with RNase-free DNase (Promega) and used for RT-PCR detection (30 cycles) (34) with the primers PP82 and PP356 for virus-carried FT RNA. The resultant specific RT- PCR products were purified and verified by direct sequencing. The viral transient FT protein was analyzed by Western blotting detection. Western blotting. To investigate GFP, FT, GFP-FT fusion protein, and PVX CP expression, total proteins were extracted from leaf tissues as described previously (15). Western blot analyses of protein aliquots (10 g) were performed with polyclonal antibodies specifically raised against GFP, the Arabidopsis FT and PVX CP, and detected using a goat anti-rabbit immunoglobulin G conjugated with alkaline phosphatase (Sigma) and BCIP/NBT substrates (Roche) as described previously (39). RESULTS AND DISCUSSION RT-PCR detection Predicted size (bp) of RT-PCR product b TCV/FTn102 CP* PP512/PP513 PP267/PP TCV/FTn201 CP* PP512/PP516 PP267/PP TCV/FTn300 CP* PP512/PP518 PP267/PP TCV/FTn399 CP* PP512/PP520 PP267/PP TCV/FT103c CP PP514/PP515 PP267/PP TCV/FT202c CP PP517/PP515 PP267/PP TCV/FT301c CP PP519/PP515 PP267/PP TCV/FT400c CP PP521/PP515 PP267/PP a *, A stop codon was introduced to replace the native FT start codon;, a stop codon was placed immediately upstream of the truncated FT RNA. Thus, all truncated FT RNAs produced from the eight RMA vectors are nontranslatable. b The predicted size of each RT-PCR product includes TCV and cloning sequences (273 bp) plus the corresponding truncated FT sequence. Arabidopsis FT RNA facilitates the movement of heterologous GFP and viral RNAs in N. benthamiana. To investigate plant RNA trafficking, we developed a GFP-tagged movement-defective Potato virus X (PVX/GFP CP)-based RMA (Fig. 1A). Deletion of viral CP prevented PVX/GFP CP movement and restricted viral RNAs to single N. benthamiana epidermal cells. Thus, unlike the wild-type virus, PVX/GFP CP was incapable of accessing the phloem to move systemically (10). It should be noted that free GFP, once loaded from the mesophyll into the sieve tube, can move over long distances in the phloem (16, 17, 31, 32). PVX/GFP-FT CP and PVX/GFPmFT CP had, respectively, the translatable or nontranslatable Arabidopsis FT that was fused in-frame to the GFP coding sequence (Fig. 1A). Individual epidermal cells expressing transient GFP or GFP-FT fusion protein from PVX/GFP CP, PVX/GFP-mFT CP, or PVX/GFP-FT CP showed green fluorescence (Fig. 1A). However, cell-to-cell movement of PVX/ GFP-FT CP or PVX/GFP-mFT CP or intercellular spread of
3 3542 LI ET AL. J. VIROL. GFP or GFP-FT among epidermal cells was not detected by epifluorescence microscopy (Fig. 1A). We also examined the upper leaves for systemic movement of GFP or GFP-FT protein and these recombinant viruses but failed to observe any GFP in any type of cells. We then used a more sensitive RT-PCR assay to test for RNA movement. Accumulation of recombinant viral RNAs from each vector was readily detected in inoculated leaves by RT-PCR using FT-, GFP-, and PVX-specific primers (Fig. 1A and B). However, no spread of genomic RNA (grna) and subgenomic RNA (sgrna) of PVX/GFP CP to newly formed young leaves occurred (Fig. 1C). In striking contrast, inclusion of FT RNA in PVX/GFP-FT CP enabled GFP-FT sgrna and PVX/GFP-FT CP grna to move systemically from inoculated leaves to young leaves (Fig. 1C). Furthermore, PVX/ GFP-mFT CP grna and sgrnas that contained nonsense mutations precluding FT protein synthesis also spread systemically (Fig. 1C), demonstrating that nontranslatable mft RNA was able to move and promote long-distance trafficking of heterologous GFP and PVX RNAs. FT and mft possessed similar abilities to facilitate the spread of heterologous RNAs, although the levels of mobile RNAs detected in the systemic leaves varied between plants for the FT and mft constructs. The absence of GFP signal in neighboring epidermal cells and upper leaves after FT RNA-mediated movement from lower parts into younger tissues suggests that CP may be required for vascular exit, which could be a novel role for CP in viral movement, although we cannot exclude the possibility that it is due to the limited sensitivity of fluorescence detection of free GFP and GFP-FT fusion proteins. FT and mft RNA movement and FT-mediated heterologous RNA trafficking were also demonstrated in a distinct Turnip crinkle virus (TCV)-based RMA (Fig. 2). In a similar experimental design, we used a GFP-tagged movement-deficient TCV CP-based RMA vector (Fig. 2A) and also showed that the Arabidopsis FT RNA promoted systemic movement of TCV RNA. RT-PCR detection of FT, GFP, and TCV RNA and 18S rrna (18S) in inoculated leaves (Fig. 2B) and particularly in the newly formed young leaves and shoot apices (Fig. 2C) of N. benthamiana plants infected with TCV/ GFP CP, TCV/GFP-FT CP, or TCV/GFP-mFT CP mirrored the results obtained with the PVX-based RMA. Mobility of Arabidopsis FT RNA is independent of viral RNA sequences. The self-mobility of FT RNA and the associated heterologous RNAs was further confirmed by an agroinfil- FIG. 1. (A) PVX-based RMA vectors. Viral 7.0-kb genomic grna, 2.6-kb subgenomic sgrna1, and 1.4-kb GFP-FT sgrna are indicated. The CP gene was deleted. The positions of a stop codon (*) replacing FT start codon in PVX/GFP-mFT CP, and three sets of RT-PCR primers for the detection of FT ( Š [red], PP354/PP356), GFP ( Š [green], PP371/PP372), or PVX ( Š [blue], PP269/PP373) RNAs are indicated. Individual epidermal cells expressing free GFP or GFP-FT fusion protein showed green fluorescence. (B and C) RT-PCR analysis of FT, GFP, and PVX RNA and 18S rrna in inoculated and newly growing young leaves, including shoot apices of N. benthamiana. RNAs were extracted from leaves of plants mock inoculated (mock) or inoculated with PVX/GFP CP (GFP), PVX/GFP-FT CP (GFP-FT), or PVX/GFP-mFT CP (GFP-mFT). The 1-kb DNA ladder and sizes of RT-PCR products are indicated. (D) Construction of 35S promotercontrolled FT gene expression cassettes for a transient agroinfiltration assay of RNA mobility. The position of an introduced stop codon (*) replacing the FT gene start codon in 35S-GFP-mFT-poly(A) is indicated. (E) Expression of free GFP or GFP-FT fusion protein. Total proteins were extracted from N. benthamiana leaves infiltrated with agrobacteria carrying the 35S-controlled gene expression cassette and were analyzed by Western blotting with a GFP-specific antibody. The positions of free GFP and GFP-FT fusion protein are indicated. (F) FT and GFP mrnas in nonagroinfiltrated newly developed systemic young leaves were analyzed by RT-PCR. Samples are from plants with mock infiltration (mock) or infiltrated with agrobacteria carrying 35S-GFPpoly(A) (GFP), 35S-GFP-FT-poly(A) (GFP-FT), or 35S-GFP-mFTpoly(A) (GFP-mFT). The sizes of RT-PCR products, free and GFP-FT fusion proteins, and the prestained protein markers or 1-kb DNA ladder are indicated.
4 VOL. 83, 2009 cis-acting ELEMENT-MEDIATED FT RNA 3543 FIG. 2. The Arabidopsis FT RNA supports TCV RNA movement. (A) GFP-tagged movement-deficient TCV-based RMA vectors. The position of an introduced stop codon (*) replacing the FT gene start codon in TCV/GFP-mFT CP and three sets of RT-PCR primers for detections of FT ( Š [red], PP354/PP356), GFP ( Š [green], PP371/ PP372) or TCV ( Š [blue], PP433/PP434) are indicated. (B and C) RT-PCR analysis of FT, GFP and TCV RNA and 18S rrna (18S) in inoculated (B) and newly formed young leaves and shoot apices (C) of N. benthamiana plants. Total RNAs used as RT-PCR templates were extracted from inoculated or young leaves of plants with mock inoculation (mock) or inoculated with RNA transcripts from TCV/GFP CP (GFP), TCV/GFP-FT CP (GFP-FT), or TCV/GFP-mFT CP (GFPmFT). The 1-kb DNA ladder and the sizes of RT-PCR products are indicated. tration assay. N. benthamiana leaves were infiltrated with Agrobacterium tumefaciens carrying the 35S promoter-controlled GFP or GFP-tagged FT gene expression cassettes, respectively (Fig. 1D). Western blot analysis showed that expression of free GFP occurred in leaves infiltrated with agrobacterium carrying 35S-GFP-poly(A) and 35S-GFP-mFTpoly(A), while the larger GFP-FT fusion protein was expressed from 35S-GFP-FT-poly(A) (Fig. 1E). These data clearly demonstrated that the replacement of the initiation codon with a stop codon in the FT open reading frame blocked the translation of FT protein from the 35S-GFP-mFT construct. We also examined the Arabidopsis FT sequence and found no internal in-frame start codons; thus, it is unlikely that mft RNA would be translated from internal initiation. Consistent with the results obtained from the viral RMAs, following agroinfiltration, both FT (mft) and FT (mft)-gfp RNAs spread systemically to young leaves that had developed on the plant after the agroinfiltration (Fig. 1F). It should be pointed out that systemic movement of FT (mft) and FT (mft)-gfp RNA seems to be stronger in virus-based RMAs than in the agroinflitration assays. This may be due to differences in the site (nucleus versus cytoplasm) and the means of production of the mobile RNA molecules (35S promoter-controlled transcription versus recombinant viral RNA replication and transcription) in agroinfiltrated cells compared to viral inoculated cells. Nevertheless, our data indicate that no viral sequences and proteins are necessary for systemic spread of FT RNA. Viral transient expression of Arabidopsis FT protein promotes floral induction. Arabidopsis FT is required to induce flowering. However, it did not stimulate early flowering in day neutral N. benthamiana expressing GFP-FT, probably due to interference by the GFP fusion. To test this, we infected SD N. tabacum Maryland Mammoth tobacco (21) with PVX/FT (Fig. 3A) and PVX/GFP-FT (see Fig. 6A). Viral ectopic expression of the GFP-FT fusion protein was unable to induce flowering under noninducing LD conditions (Fig. 3B and C); however, all Maryland Mammoth plants infected with PVX/FT that had the capacity to produce free FT protein flowered in LD, while control plants infected with either PVX/mFT carrying a mutated nontranslatable FT mrna or PVX/GFP remained vegetative (Fig. 3B and C). Viral delivery of wild-type and mutated FT RNA was readily detected in systemically infected leaves by RT-PCR and further confirmed by direct sequencing of the specific RT-PCR products (Fig. 3D). Free FT protein expressed from PVX/FT but not from PVX/mFT was also detected by Western blotting with an antiserum specifically raised against FT (Fig. 4D). The fact that PVX/FT could induce flowering but PVX/mFT could not shows that the Arabidopsis FT RNA alone is not sufficient to initiate flowering but that its protein product expressed from PVX/FT is necessary for floral induction. Systemic movement of FT RNA does not require FT protein. The virus-based RMAs, together with the agroinfiltration assay, demonstrate that Arabidopsis FT RNA not only is selfmobile but also can mediate systemic trafficking of heterologous RNAs in N. benthamiana. To eliminate the possibility that endogenous plant-derived FT protein might be involved in facilitation of the FT RNA movement, we used the TCV-based RMA vector TCV/mFT CP containing a nontranslatable FT gene (Fig. 4A). Deletion of the CP gene in TCV CP prevents its spreading from infected cells to distal parts of plants (6). However, in contrast to the PVX-based RMA, TCV is able to infect A. thaliana (36) and TCV CP can establish local infection in this host (unpublished data). We inoculated the Arabidopsis ft-10 T-DNA insertion mutant (43) with RNA transcripts of TCV/mFT CP or TCV/GFP CP. In inoculated leaves, accumulation of recombinant viral RNAs from both RMA vectors was readily detected by RT-PCR, while no specific recombinant viral RNA was detectable in mock-inoculated control plants (Fig. 4B). Not surprisingly, no systemic spread of TCV/GFP CP RNA to noninoculated leaves occurred. However, the nontranslatable mutant mft RNA in TCV/mFT CP moved and facilitated long-distance movement of viral RNAs to young leaves and the shoot apices (Fig. 4B). The nonsense mutation prohibiting FT protein synthesis from
5 3544 LI ET AL. J. VIROL. FIG. 3. Ectopic expression of FT induces flowering. (A) The translatable and mutated (*) nontranslatable Arabidopsis FT coding sequences were cloned into wild-type PVX vector to produce PVX/FT and PVX/mFT, respectively. (B and C) Floral induction caused by viral expression of FT protein. Young SD N. tabacum Maryland Mammoth plants were mock inoculated or infected with PVX/FT or PVX/mFT and grown under a noninducing LD photoperiod. Twelve plants infected by PVX/FT in three separate experiments started bolting at 20 dpi, flowered at 35 dpi, and were photographed at 42 dpi (B and inset image). Tobacco mock inoculated or infected with PVX/mFT, PVX/ GFP, or PVX/GFP-FT did not flower (B and C). (D) Detection of viral transient FT RNA. Viral transient FT RNA was detected by RT-PCR using primers PP82 ( ) and PP356 (Š) in systemic young leaves from two separate plants infected with PVX/mFT (mft) or PVX/FT (FT) but not in a mock-infected plant (mock). The position and the sizes of 1-kb DNA ladder (lane M) are indicated. Direct sequencing of RT- PCR products (648 bp) verified the presence of virally expressed wildtype and mutant FT RNA in flowering and nonflowering plants, respectively. The native FT ATG (underlined) in PVX/FT and its TAG replacement (underlined) together with a nucleotide deletion (doublearrow) in PVX/mFT are indicated. TCV/mFT CP was maintained in the Arabidopsis mutant (Fig. 4C). Moreover, no viral transiently expressed or endogenous FT protein was detected in different leaf tissues of mockinoculated and virus-treated ft-10 mutant plants, a finding consistent with the mutation in ft-10 (Fig. 4D). Long-distance trafficking of virus-derived mft RNA from inoculated leaves to noninoculated leaves and the shoot apices was also detected in ft-1 mutant plants (X. Liu, C. Li, S. Jackson, and Y. Hong, unpublished data). On the other hand, when fusing GFP downstream of FT, the nonsense mutation also eliminates GFP expression (K. Zhang and Y. Hong, unpublished data). Thus, we conclude that the systemic FT RNA trafficking does not require the FT protein. Mapping of the cis-acting element within Arabidopsis FT RNA. To elucidate what controls long-distance spread of FT FIG. 4. (A) Movement-deficient TCV-based RMA vectors. The parental TCV vector and long-distance movement-deficient TCV/ GFP CP were previously constructed (34). The position of the introduced stop codon (*) replacing ATG in the FT gene in TCV/mFT CP and two sets of RT-PCR primers are indicated. (B and C) RT-PCR analysis of FT-TCV and GFP-TCV RNA (top panel) using primers PP267/PP473a ( Š [green]) or PP267/PP228 ( Š [red]) and 18S rrna (bottom panel) in inoculated (In) and newly formed young leaves (M1 and M2) and shoot apices (Ap) of ft-10 A. thaliana mutant plants. RNAs were extracted from tissues of plants mock-inoculated (mock) or inoculated with TCV/GFP CP (GFP) or TCV/mFT CP (mft). The positions and sizes of the 1-kb DNA ladder are indicated. (C) Direct sequencing of mft RT-PCR products (372 bp) generated from total RNA extracted from young leaves of Arabidopsis mutant plants inoculated with TCV/mFT CP. (D) Western blot analysis indicated no endogenous or viral transient FT protein was expressed in the mock-inoculated leaves (mock) or in different leaves (In, M1, and M2) and shoot apices (AP) of ft-10 plants inoculated with TCV/mFT CP. Viral ectopic expression of FT protein (red asterisk) was detected in N. tabacum Maryland Mammoth plants infected with PVX/FT (FT) but not with PVX/mFT (mft), a finding consistent with that the FT protein is required for floral induction (Fig. 3B). Coomassie bluestained gel indicates the equal loading of protein samples. The positions and sizes of the protein markers are indicated. RNA, we constructed a series of TCV/ CP-based RMA vectors carrying truncated nontranslatable FT fragments (Fig. 5A). We found that viral-derived FT RNAs from TCV/ FTn102 CP, TCV/FTn201 CP TCV/FTn300 CP and TCV/ FTn399 CP were capable of movement and facilitating sys-
6 VOL. 83, 2009 cis-acting ELEMENT-MEDIATED FT RNA 3545 FIG. 5. Functional mapping of the cis-acting element that controls the FT RNA movement. (A) TCV/trFT CP-based RMA vectors carrying truncated (tr) nontranslatable Arabidopsis FTs. (B) Detection of virus-derived FT RNA (top panel) by RT-PCR in systemic young leaves (Y) from plants inoculated with TCV/FTn102 CP (n102), TCV/FTn201 CP (n201), TCV/FTn300 CP (n300), or TCV/FTn399 CP (n399); but not from plants inoculated with TCV/FT103c CP (103c), TCV/FT202c CP (202c), TCV/FT301c CP (301c) or TCV/FT400c CP (400c). Recombinant viral RNA of each TCV/trFT CP was readily detectible in inoculated leaves (I). No specific virus-derived FT RNA was detected in mockinoculated plants. RT-PCR analysis of 18S rrna (bottom panel) is included as an RNA control. (C and D) Direct sequencing of RT-PCR products verified the presence of virus-derived truncated FT RNA. Example sequence panels are shown for truncated FT RNA expressed in the young leaves of plants inoculated by TCV/FTn102 CP (C) or in the TCV/FT103c CP-inoculated leaves (D). The TAG stop codon is underlined and TCV and virus-derived FT sequences are indicated. temic movement of TCV RNA. This was evident by positive RT-PCR detections of TCV and virus-derived FT RNAs in the noninoculated, newly developing and growing young leaves (Fig. 5B). All of the truncated FT RNAs possess nucleotides 1 to 102 of FT RNA (Fig. 5A). In striking contrast, FT RNAs generated from TCV/FT103c CP, TCV/FT202c CP, TCV/ FT301c CP, and TCV/FT400c CP that lack the 5 -terminal 102 nucleotides were completely immobile. Consequently, RT- PCRs failed to detect any virus-derived FT-related RNA in the young leaves of virus-treated plants although recombinant viral RNAs accumulated in the inoculated leaves (Fig. 5B). Direct sequencing of RT-PCR products confirmed the identities of the mobile and immobile TCV and virus-derived FT RNA molecules (Fig. 5C and D). Thus, the element required for systemic FT RNA movement was unequivocally mapped to the 102-nucleotide sequence at the 5 terminus of FT mrna. It should be noted that TCV without CP moves readily in dcl2/dcl4 mutated Arabidopsis that is defective in RNA silencing. Thus, the TCV CP is mainly providing a silencing-suppression function which allows virus movement, although it may also be offering some movement support (28). This is consistent with the idea that the CP silencing-suppressor possesses a differential role in viral intercellular spread (35). However, the Arabidopsis FT RNA and indeed FT protein have no effect on RNA silencing (K. Zhang, C. Li, and Y. Hong, unpublished data). Therefore, FT RNA is only providing movement function and the FT RNA-mediated systemic spread of TCV seems to operate with a distinct mechanism. The Arabidopsis FT RNA enhances systemic spread of PVX. The translational and nontranslational Arabidopsis FT coding sequences were fused in-frame with the GFP coding sequence of PVX/GFP to produce PVX/GFP-FT and PVX/GFP-mFT (Fig. 6A), respectively. In repeated experiments, 24-day-old Nicotiana benthamiana plants were mock inoculated (Fig. 6B) or infected with RNA transcripts produced by in vitro transcription from PVX/GFP (Fig. 6C), PVX/GFP-FT (Fig. 6D), or PVX/GFP-mFT (Fig. 6E). Local infection of the inoculated leaves induced GFP-expressing green lesions (ca. 10 lesions per inoculated leaf) 3 to 5 dpi. However, only plants challenged with PVX/GFP-FT or PVX/GFP-mFT quickly established systemic infection at 7 dpi, showing GFP green fluorescence in newly developing young leaves. Development of systemic symptoms in plants infected by PVX/GFP took 2 to 3 extra days. Furthermore, the accumulations of free GFP and GFP-FT fusion proteins (Fig. 6F) and viral CP (Fig. 6G) in the inoculated and systemic young leaves after mock inoculation (mock) or infection of PVX/GFP (GFP), PVX/GFP-FT (GFP- FT), or PVX/GFP-mFT (GFP-mFT) were consistent with the development of viral symptoms. Western blot assays of total proteins extracted from inoculated and systemic young leaves using GFP- and PVX CP-antiserum indicated that only a trace amount of free GFP and PVX CP was detected in the inoculated leaves but not in the systemic young leaves of PVX/GFPinfected plants. However, viral CP as well as the GFP-FT fusion protein or free GFP expressed from PVX/GFP-FT or PVX/GFP-mFT, respectively, could be readily detected in both inoculated and systemic leaves. We provide here compelling evidence that Arabidopsis FT RNA is capable of systemic movement and that FT RNA can also act as a cis transportation carrier for heterologous RNAs. The FT RNA mobile function is independent of the FT protein. Consistent with this idea, an engineered plant RNA virus
7 3546 LI ET AL. J. VIROL. Downloaded from FIG. 6. Arabidopsis FT RNA enhances the systemic spread of PVX. PVX-based FT expression cassettes are schematically represented (A). The translatable and nontranslatable Arabidopsis FT coding sequences were fused in-frame with the GFP coding sequence of PVX/GFP by using the BspEI and SalI sites. The unique XhoI site used to produce CP-defective RMA vectors (Fig. 1A) is indicated. Twenty-four-day-old N. benthamiana plants were mock inoculated (B) or infected with PVX/GFP (C), PVX/GFP-FT (D), or PVX/GFP-mFT (E). (C to E) Local infection of inoculated leaves (IL) induced GFP-expressing lesions. However, only plants challenged with PVX/GFP-FT or PVX/GFP-mFT quickly established systemic infection at 7 dpi, showing GFP green fluorescence in newly developed young leaves (YL) (D and E). Moreover, protein were extracted from inoculated and systemic young leaves of plants with mock (mock) or viral infection, resolved by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, and detected by Western blotting with antiserum raised against GFP (F) and PVX CP (G). The positions of free GFP, GFP-FT fusion protein, and the PVX CP are indicated. The production of viral CP, free GFP, and GFP-FT fusion proteins associated with PVX/GFP (GFP), PVX/GFP-FT (GFP-FT) or PVX/GFP-mFT (GFP-mFT) was consistent with symptom development. These data indicate that the Arabidopsis FT RNA with or without its translatable capacity enhances systemic spread of PVX. on July 19, 2018 by guest carrying Arabidopsis FT RNA spread more quickly than its parental virus to establish infection of young tissues. It should be noted that the CP genes of PVX and TCV participate in virus-plant interactions and contribute to symptom development (5, 10, 20, 45); thus, it was not surprising that the CPdeleted recombinant viruses that were transported through the plant via FT RNA in cis could still not start a proper infection. Moreover, we demonstrate that the systemic mobility of FT RNA is determined by a cis-acting element of 102 nucleotides at the 5 terminus of FT RNA. The positive identification of cis-acting element for FT RNA trafficking provides a unique opportunity to dissect the molecular mechanism that governs cellular RNA signaling in plants. It is possible that the cis-rna sequence may bind host proteins to form an RNA-protein
8 VOL. 83, 2009 cis-acting ELEMENT-MEDIATED FT RNA 3547 complex for FT RNA spread. Indeed, some plant and viral proteins can bind RNAs and facilitate their intercellular and long-distance trafficking (26, 27, 41, 42). The discovery that a host RNA molecule can mediate systemic trafficking of heterologous RNAs is also significant. In particular, facilitation of viral RNA movement by the Arabidopsis FT RNA raises the possibility that viruses and plants might have evolved similar mechanisms for systemic RNA trafficking. There is now collective evidence that the Arabidopsis FT protein and its tomato SFT and rice Hd3a orthologs may act as a non-cell-autonomous flower-inducing signal that can move from the end of the vasculature into the meristematic tissue in the shoot apical meristem (7, 18, 22, 23, 29, 30, 37). Elegant experiments wherein the intercellular movement of FT protein is prevented by either a large C-terminal fusion and/or a nuclear localization signal have demonstrated that FT protein needs to move, and is sufficient on its own, to induce flowering (18, 29). FT- and Hd3a-GFP fusion proteins were found to cross a graft union from a donor scion and reach the apex of the recipient plant (7, 37), and FT-derived peptides have been identified in phloem exudates from Cucurbita (23), suggesting that the protein is transported through the phloem. Once the FT protein enters the apical meristem it interacts with the bzip transcription factor FD to activate floral identity genes such as APETELA 1 (1, 40). However, these studies do not definitely rule out a role for FT mrna as part of the mobile florigenic signal. In this context, it is worthwhile noting that mrna of the rice FT ortholog Hd3a has been detected at extremely low levels in shoot apices of rice even though the Hd3a promoter is not active in the shoot apical meristem (37). The evidence presented here that the Arabidopsis FT RNA is able to move systemically is in contrast to these recent reports where FT RNA movement could not be detected (7, 18, 22, 23, 29, 30), possibly due to the different host plants and experimental systems used. For example, high levels of viral FT expression, even from immobilized viruses in single cells, could increase the levels of systemic FT RNA over the detection limit. Our data raise the possibility that systemic movement of the FT RNA may also contribute to the long-distance florigen signaling, although the FT protein is still required to trigger flowering, as shown by the nonflowering of PVX/mFT-inoculated plants. ACKNOWLEDGMENTS We thank B. Thomas, T. M. A. Wilson, and S. Bright for critical reading of the manuscript and D. Baulcombe for providing the original PVX vector. C.L. is supported by the Warwick University Postgraduate Studentship. This work was supported in part by the Warwick HRI-UK Biotechnology and Biological Sciences Research Council core funding, a Warwick Venture grant, and a Warwick Research Development Fund grant to Y.H. REFERENCES 1. Abe, M., Y. Kobayashi, S. Yamamoto, Y. Daimon, A. Yamaguchi, Y. Ikeda, H. Ichinoki, M. Notaguchi, K. Goto, and T. Araki FD, a bzip protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 309: An, H., C. Roussot, P. Suárez-López, L. Corbesier, C. Vincent, M. Piñeiro, S. Hepworth, A. Mouradov, S. Justin, C. Turnbull, and G. Coupland CONSTANS acts in the phloem to regulate a systemic signal that induces photoperiodic flowering of Arabidopsis. Development 131: Banerjee, A. K., M. Chatterjee, Y. Yu, S.-G. Suh, W. A. Miller, and D. J. Hannapel Dynamics of a mobile RNA of potato involved in a longdistance signaling pathway. Plant Cell 18: Buhtz, A., F. Springer, L. Cappell, D. C. Baulcombe, and J. Kehr Identification and characterization of small RNAs from the phloem of Brassica napus. Plant J. 53: Chapman, S., G. Hills, J. Watts, and D. Baulcombe Mutational analysis of the coat protein gene of potato virus X: effects on virion morphology and viral pathogenicity. Virology 191: Cohen, Y., A. Gisel, and P. C. Zambryski Cell-to-cell and systemic movement of recombinant green fluorescent protein-tagged Turnip crinkle viruses. Virology 273: Corbesier, L., C. Vincent, S. Jang, F. Fornara, Q. Fan, I. Searle, A. Giakountis, S. Farrona, L. Gissot, C. Turnbull, and G. Coupland FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316: Deeken, R., P. Ache, I. Kajahn, J. Klinkenberg, G. Bringmann, and R. Hedrich Identification of Arabidopsis thaliana phloem RNAs provides a search criterion for phloem-based transcripts hidden in complex datasets of microarray experiments. Plant J. 55: Dunoyer, P., C. Himber, and O. Voinnet DICER-LIKE 4 is required for RNA interference and produces the 21-nucleotide small interfering RNA component of the plant cell-to-cell silencing signal. Nature Genetics 37: Fedorkin, O., A. Solovyev, N. Yelina, A. Zamyatnin, Jr., R. Zinovkin, K. Mäkinen, J. Schiemann, and S. Y. Morozov Cell-to-cell movement of potato virus X involves distinct functions of the coat protein. J. Gen. Virol. 82: Gopinath, K., and C. C. Kao Replication-independent long-distance trafficking by viral RNAs in Nicotiana benthamiana. Plant Cell 19: Hamilton, A. J., O. Voinnet, L. Chappell, and D. C. Baulcombe Two classes of short interfering RNA in RNA silencing. EMBO J. 21: Haywood, V., T.-S. Yu, N.-C. Huang, and W. J. Lucas Phloem longdistance trafficking of GIBBERELLIC ACID-INSENSITIVE RNA regulates leaf development. Plant J. 42: Himber, C., P. Dunoyer, G. Moissiard, C. Ritzenthaler, and O. Voinnet Transitivity-dependent and independent cell-to-cell movement of RNA silencing. EMBO J. 22: Hong, Y., K. Saunders, M. R. Hartley, and J. Stanley Resistance to geminivirus infection by virus-induced expression of a plant cytotoxin. Virology 220: Imlau, A., E. Truernit, and N. Sauer Cell-to-cell and long-distance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein into sink tissues. Plant Cell 11: Itaya, A., G. Liang, Y.-M. Woo, R. S. Nelson, and B. Ding Nonspecific intercellular protein trafficking probed by green-fluorescent protein in plants. Protoplasma 213: Jaeger, K. E., and P. A. Wigge FT protein acts as a long-range signal in Arabidopsis. Curr. Biol. 17: Kim, M., W. Canio, S. Kessler, and N. Sinha Developmental changes due to long-distance movement of a homeobox fusion transcript in tomato. Science 293: Kong, Q., J. W. Oh, and A. E. Simon Symptom attenuation by a normally virulent satellite RNA of turnip crinkle virus is associated with the coat protein open reading frame. Plant Cell 7: Lang, A., M. K. Chailakhyan, and I. A. Frolova Promotion and inhibition of flower formation in a day neutral plant in grafts with a short-day plant and a long-day plant. Proc. Natl. Acad. Sci. USA 74: Lifschitz, E., T. Eviatar, A. Rozman, A. Shalit, A. Goldshmidt, Z. Amsellem, J. P. Alvarez, and Y. Eshed The tomato FT ortholog triggers systemic signals that regulate growth and flowering and substitute for diverse environmental stimuli. Proc. Natl. Acad. Sci. USA 103: Lin, M. K., H. Belanger, Y. J. Lee, E. Varkonyi-Gasic, K. Taoka, E. Miura, B. Xoconostle-Cázares, K. Gendler, R. A. Jorgensen, B. Phinney, T. J. Lough, and W. J. Lucas FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 19: Lough, T. J., R. H. Lee, S. J. Emerson, R. L. Forster, and W. J. Lucas Functional analysis of the 5 untranslated region of potexvirus RNA reveals a role in viral replication and cell-to-cell movement. Virology 351: Lough, T. J., and W. J. Lucas Integrative plant biology: role of phloem long-distance macromolecular trafficking. Annu. Rev. Plant Biol. 57: Lucas, W. J Plant viral movement proteins: agents for cell-to-cell trafficking of viral genomes. Virology 344: Lucas, W. J., S. Bouché-Pillon, D. P. Jackson, L. Nguyen, L. Baker, B. Ding, and S. Hake Selective trafficking of KNOTTED1 homeodomain protein and its mrna through plasmodesmata. Science 270: Manfre, A. J., and A. E. Simon Importance of coat protein and RNA silencing in satellite RNA/virus interactions. Virology 379: Mathieu, J., N. Warthmann, F. Küttner, and M. Schmid Export of FT protein from phloem companion cells is sufficient for floral induction in Arabidopsis. Curr. Biol. 17: Notaguchi, M., M. Abe, T. Kimura, Y. Daimon, T. Kobayashi, A. Yamaguchi,
9 3548 LI ET AL. J. VIROL. Y. Tomita, K. Dohi, M. Mori, and T. Araki Long-distance, grafttransmissible action of Arabidopsis FLOWERING LOCUS T protein to promote flowering. Plant Cell Physiol. 49: Oparka, K. J., A. G. Roberts, P. Boevink, S. Santa Cruz, I. Roberts, K. S. Pradel, A. Imlau, G. Kotlizky, N. Sauer, and B. Epel Simple, but not branched, plasmodesmata allow the nonspecific trafficking of proteins in developing tobacco leaves. Cell 97: Peleg, G., D. Malter, and S. Wolf Viral infection enables phloem loading of GFP and long-distance trafficking of the protein. Plant J. 51: Qi, Y., T. Pélissier, A. Itaya, E. Hunt, M. Wassenegger, and B. Ding Direct role of a viroid RNA motif in mediating directional RNA trafficking across a specific cellular boundary. Plant Cell 16: Ryabov, E. V., R. Van Wezel, J. Walsh, and Y. Hong Cell-to-cell, but not long-distance, spread of RNA silencing that is induced in individual epidermal cells. J. Virol. 78: Shi, Y., E. V. Ryabov, R. Van Wezel, C. Li, M. Jin, W. Wang, Z. Fan, and Y. Hong Suppression of local RNA silencing is not sufficient to promote cell-to-cell movement of Turnip crinkle virus in Nicotiana benthamiana. Plant Signal. Behav. 4: Simon, A. E Interaction between Arabidopsis thaliana and virus, p In E. Meyerowitz and C. Sommerville (ed.), Arabidopsis. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY. 37. Tamaki, S., S. Matsuo, H. L. Wong, S. Yokoi, and K. Shimamoto Hd3a protein is a mobile flowering signal in Rice. Science 316: Van Wezel, R., X. Dong, H. Liu, P. Tien, J. Stanley, and Y. Hong Mutations of three cysteine residues in Tomato yellow leaf curl virus-china C2 protein causes dysfunction in pathogenesis and posttranscriptional gene silencing suppression. Mol. Plant-Microbe interact. 15: Van Wezel, R., and Y. Hong Virus survival of RNA silencing without deploying protein-mediated suppression in Nicotiana benthamiana. FEBS Lett. 562: Wigge, P. A., M. C. Kim, K. E. Jaeger, W. Busch, M. Schmid, J. U. Lohmann, and D. Weigel Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309: Xoconostle-Cázares, B., Y. Xiang, R. Ruiz-Medrano, H. L. Wang, J. Monzer, B. C. Yoo, K. C. McFarland, V. R. Franceschi, and W. J. Lucas Plant paralog to viral movement protein that potentiates transport of mrna into the phloem. Science 283: Yoo, B. C., F. Kragler, E. Varkonyi-Gasic, V. Haywood, S. Archer-Evans, Y. M. Lee, T. J. Lough, and W. J. Lucas A systemic small RNA signaling system in plants. Plant Cell 16: Yoo, S. K., K. S. Chung, J. Kim, J. H. Lee, S. M. Hong, S. J. Yoo, S. Y. Yoo, J. S. Lee, and J. H. Ahn CONSTANS activates SUPPRESSOR OF OVEREXPRESSION OF CONSTANS 1 through FLOWERING LOCUS T to promote flowering in Arabidopsis. Plant Physiol. 139: Zhang, X., A. J. Archual, A. A. Amin, and B. Ding A genomic map of viroid RNA motifs critical for replication and systemic trafficking. Plant Cell 20: Zhang, F., and A. E. Simon Enhanced viral pathogenesis associated with a virulent mutant virus or a virulent satellite RNA correlates with reduced virion accumulation and abundance of free coat protein. Virology 312: Zhong, X., X. Tao, J. Stombaugh, N. Leontis, and B. Ding Tertiary structure and function of an RNA motif required for plant vascular entry to initiate systemic trafficking. EMBO J. 26: Zhou, Y., E. Ryabov, X. Zhang, and Y. Hong Influence of viral genes on the cell-to-cell spread of RNA silencing. J. Exp. Bot. 59: Downloaded from on July 19, 2018 by guest
Cell-to-Cell, but Not Long-Distance, Spread of RNA Silencing That Is Induced in Individual Epidermal Cells
 JOURNAL OF VIROLOGY, Mar. 2004, p. 3149 3154 Vol. 78, No. 6 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.6.3149 3154.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Cell-to-Cell,
JOURNAL OF VIROLOGY, Mar. 2004, p. 3149 3154 Vol. 78, No. 6 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.6.3149 3154.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Cell-to-Cell,
Supporting Information
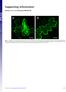 Supporting Information Harries et al. 1.173/pnas.9923916 A Fig. S1. Disruption of microfilaments within epidermal cells after treatment with 5 M Lat. Images of N. benthamiana cells are from plants expressing
Supporting Information Harries et al. 1.173/pnas.9923916 A Fig. S1. Disruption of microfilaments within epidermal cells after treatment with 5 M Lat. Images of N. benthamiana cells are from plants expressing
MINIREVIEW. Activation and Suppression of RNA Silencing by Plant Viruses
 Virology 281, 1 5 (2001) doi:10.1006/viro.2000.0812, available online at http://www.idealibrary.com on MINIREVIEW Activation and Suppression of RNA Silencing by Plant Viruses James C. Carrington, 1 Kristin
Virology 281, 1 5 (2001) doi:10.1006/viro.2000.0812, available online at http://www.idealibrary.com on MINIREVIEW Activation and Suppression of RNA Silencing by Plant Viruses James C. Carrington, 1 Kristin
Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana
 Available online at www.sciencedirect.com R Virology 306 (2003) 33 41 www.elsevier.com/locate/yviro Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana Carole
Available online at www.sciencedirect.com R Virology 306 (2003) 33 41 www.elsevier.com/locate/yviro Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana Carole
Phenomena first observed in petunia
 Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Virus-host interactions
 Virus-host interactions - Strategies viruses use to replicate their genomes in susceptible host cells replication - Strategies viruses use to move their genomes throughout susceptible host plants cell-to-cell
Virus-host interactions - Strategies viruses use to replicate their genomes in susceptible host cells replication - Strategies viruses use to move their genomes throughout susceptible host plants cell-to-cell
Journal of Agricultural Technology 2012 Vol. 8(4): Journal of Agricultural
 Journal of Agricultural Technology 2012 Vol. 8(4): 1389-1395 Journal of Agricultural Available Technology online http://www.ijat-aatsea.com 2012, Vol. 8(4): 1389-1395 ISSN 1686-9141 The effect of the decreased
Journal of Agricultural Technology 2012 Vol. 8(4): 1389-1395 Journal of Agricultural Available Technology online http://www.ijat-aatsea.com 2012, Vol. 8(4): 1389-1395 ISSN 1686-9141 The effect of the decreased
Fine Mapping of a cis-acting Sequence Element in Yellow Fever Virus RNA That Is Required for RNA Replication and Cyclization
 JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow Mosaic Virus Coat Proteins in Nicotiana benthamiana
 Journal of Bacteriology and Virology 2017. Vol. 47, No. 2 p.87 95 http://dx.doi.org/10.4167/jbv.2017.47.2.87 Original Article Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow
Journal of Bacteriology and Virology 2017. Vol. 47, No. 2 p.87 95 http://dx.doi.org/10.4167/jbv.2017.47.2.87 Original Article Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow
Virus survival of RNA silencing without deploying protein-mediated suppression in Nicotiana benthamiana
 FEBS 28167 FEBS Letters 562 (2004) 65^70 Virus survival of RNA silencing without deploying protein-mediated suppression in Nicotiana benthamiana Rene van Wezel, Yiguo Hong Host-Pathogen Interactions, Horticulture
FEBS 28167 FEBS Letters 562 (2004) 65^70 Virus survival of RNA silencing without deploying protein-mediated suppression in Nicotiana benthamiana Rene van Wezel, Yiguo Hong Host-Pathogen Interactions, Horticulture
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
A Viral Movement Protein Prevents Spread of the Gene Silencing Signal in Nicotiana benthamiana
 Cell, Vol. 103, 157 167, September 29, 2000, Copyright 2000 by Cell Press A Viral Movement Protein Prevents Spread of the Gene Silencing Signal in Nicotiana benthamiana Olivier Voinnet, Carsten Lederer,
Cell, Vol. 103, 157 167, September 29, 2000, Copyright 2000 by Cell Press A Viral Movement Protein Prevents Spread of the Gene Silencing Signal in Nicotiana benthamiana Olivier Voinnet, Carsten Lederer,
Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three
 Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three functional domains: a basic domain with a bipartite nuclear localization
Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three functional domains: a basic domain with a bipartite nuclear localization
Anurag Sunpapao, Takashi Nakai, Fang Dong, Tomofumi Mochizuki and Satoshi T. Ohki INTRODUCTION
 Journal of General Virology (2009), 90, 3015 3021 DOI 10.1099/vir.0.013219-0 The 2b protein of cucumber mosaic virus is essential for viral infection of the shoot apical meristem and for efficient invasion
Journal of General Virology (2009), 90, 3015 3021 DOI 10.1099/vir.0.013219-0 The 2b protein of cucumber mosaic virus is essential for viral infection of the shoot apical meristem and for efficient invasion
Lab Tuesday: Virus Diseases
 Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 67-73) and Biocontrol of Crown Gall (p. 113-117), Observation of Viral Movement in Plants (p. 119), and Intro section for Viruses (pp. 75-77).
Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 67-73) and Biocontrol of Crown Gall (p. 113-117), Observation of Viral Movement in Plants (p. 119), and Intro section for Viruses (pp. 75-77).
hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide gel electrophoresis/genetics)
 Proc. Natl. Acad. Sci. USA Vol. 73, No. 6, pp. 242-246, June 976 Microbiology Mapping of the influenza virus genome: Identification of the hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide
Proc. Natl. Acad. Sci. USA Vol. 73, No. 6, pp. 242-246, June 976 Microbiology Mapping of the influenza virus genome: Identification of the hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide
Life Sciences 1A Midterm Exam 2. November 13, 2006
 Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
AP Biology Reading Guide. Concept 19.1 A virus consists of a nucleic acid surrounded by a protein coat
 AP Biology Reading Guide Name Chapter 19: Viruses Overview Experimental work with viruses has provided important evidence that genes are made of nucleic acids. Viruses were also important in working out
AP Biology Reading Guide Name Chapter 19: Viruses Overview Experimental work with viruses has provided important evidence that genes are made of nucleic acids. Viruses were also important in working out
Supplementary Information. Supplementary Figure 1
 Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Lab Tuesday: Virus Diseases
 Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 69-75) and Biocontrol of Crown Gall (p. 115-119), Observation of Viral Movement in Plants (p. 121), and Intro section for Viruses (pp. 77-79).
Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 69-75) and Biocontrol of Crown Gall (p. 115-119), Observation of Viral Movement in Plants (p. 121), and Intro section for Viruses (pp. 77-79).
Supplementary Figure 1 Transcription assay of nine ABA-responsive PP2C. Transcription assay of nine ABA-responsive PP2C genes. Total RNA was isolated
 Supplementary Figure 1 Transcription assay of nine ABA-responsive PP2C genes. Transcription assay of nine ABA-responsive PP2C genes. Total RNA was isolated from 7 day-old seedlings treated with or without
Supplementary Figure 1 Transcription assay of nine ABA-responsive PP2C genes. Transcription assay of nine ABA-responsive PP2C genes. Total RNA was isolated from 7 day-old seedlings treated with or without
Packaging of Brome Mosaic Virus Subgenomic RNA Is Functionally Coupled to Replication-Dependent Transcription and Translation of Coat Protein
 JOURNAL OF VIROLOGY, Oct. 2006, p. 10096 10108 Vol. 80, No. 20 0022-538X/06/$08.00 0 doi:10.1128/jvi.01186-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Packaging of Brome
JOURNAL OF VIROLOGY, Oct. 2006, p. 10096 10108 Vol. 80, No. 20 0022-538X/06/$08.00 0 doi:10.1128/jvi.01186-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Packaging of Brome
Chapter 6- An Introduction to Viruses*
 Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
CHAPTER 4 RESULTS. showed that all three replicates had similar growth trends (Figure 4.1) (p<0.05; p=0.0000)
 CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
Structural vs. nonstructural proteins
 Why would you want to study proteins associated with viruses or virus infection? Receptors Mechanism of uncoating How is gene expression carried out, exclusively by viral enzymes? Gene expression phases?
Why would you want to study proteins associated with viruses or virus infection? Receptors Mechanism of uncoating How is gene expression carried out, exclusively by viral enzymes? Gene expression phases?
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Supplemental Data. Wang et al. (2013). Plant Cell /tpc
 Supplemental Data. Wang et al. (2013). Plant Cell 10.1105/tpc.112.108993 Supplemental Figure 1. 3-MA Treatment Reduces the Growth of Seedlings. Two-week-old Nicotiana benthamiana seedlings germinated on
Supplemental Data. Wang et al. (2013). Plant Cell 10.1105/tpc.112.108993 Supplemental Figure 1. 3-MA Treatment Reduces the Growth of Seedlings. Two-week-old Nicotiana benthamiana seedlings germinated on
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
of Nebraska - Lincoln
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Virology Papers Virology, Nebraska Center for 2002 Efficient Infection of Nicotiana benthamiana by Tomato bushy stunt virus
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Virology Papers Virology, Nebraska Center for 2002 Efficient Infection of Nicotiana benthamiana by Tomato bushy stunt virus
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Leaves turn yellow: small RNAs from viruses silence plant genes such as those involved in photosynthesis. Vitantonio Pantaleo IPSP-CNR
 Consiglio Nazionale delle Ricerche Di.S.Ba Conferenza del Dipartimento di Scienze Bio-agroalimentari del CNR Accademia dei Georgofili, Firenze 24-25 Ottobre 2016 Accademia dei Georgofili Leaves turn yellow:
Consiglio Nazionale delle Ricerche Di.S.Ba Conferenza del Dipartimento di Scienze Bio-agroalimentari del CNR Accademia dei Georgofili, Firenze 24-25 Ottobre 2016 Accademia dei Georgofili Leaves turn yellow:
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Conditional and reversible disruption of essential herpesvirus protein functions
 nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Lecture 2: Virology. I. Background
 Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing
 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing Matthew W. Endres 1, Brian D. Gregory 2,3, Zhihuan Gao 1, Amy Wahba Foreman
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing Matthew W. Endres 1, Brian D. Gregory 2,3, Zhihuan Gao 1, Amy Wahba Foreman
Genetics. Instructor: Dr. Jihad Abdallah Transcription of DNA
 Genetics Instructor: Dr. Jihad Abdallah Transcription of DNA 1 3.4 A 2 Expression of Genetic information DNA Double stranded In the nucleus Transcription mrna Single stranded Translation In the cytoplasm
Genetics Instructor: Dr. Jihad Abdallah Transcription of DNA 1 3.4 A 2 Expression of Genetic information DNA Double stranded In the nucleus Transcription mrna Single stranded Translation In the cytoplasm
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing
 University of Pennsylvania ScholarlyCommons Departmental Papers (Biology) Department of Biology 1-15-2010 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor
University of Pennsylvania ScholarlyCommons Departmental Papers (Biology) Department of Biology 1-15-2010 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor
Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice
 Supplementary Information Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice Gou Takahashi, Channabasavaiah B Gurumurthy,
Supplementary Information Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice Gou Takahashi, Channabasavaiah B Gurumurthy,
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
Transcription and RNA processing
 Transcription and RNA processing Lecture 7 Biology 3310/4310 Virology Spring 2018 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at the
Transcription and RNA processing Lecture 7 Biology 3310/4310 Virology Spring 2018 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at the
PLANT VIRUS SATELLITE AND DEFECTIVE INTERFERING RNAS: New Paradigms for a New Century
 Annu. Rev. Phytopathol. 2004. 42:415 37 doi: 10.1146/annurev.phyto.42.040803.140402 Copyright c 2004 by Annual Reviews. All rights reserved PLANT VIRUS SATELLITE AND DEFECTIVE INTERFERING RNAS: New Paradigms
Annu. Rev. Phytopathol. 2004. 42:415 37 doi: 10.1146/annurev.phyto.42.040803.140402 Copyright c 2004 by Annual Reviews. All rights reserved PLANT VIRUS SATELLITE AND DEFECTIVE INTERFERING RNAS: New Paradigms
Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated by the Movement Protein of Groundnut Rosette Virus
 Virology 260, 98 108 (1999) Article ID viro.1999.9806, available online at http://www.idealibrary.com on Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated
Virology 260, 98 108 (1999) Article ID viro.1999.9806, available online at http://www.idealibrary.com on Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Coronaviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Received 26 January 1996/Returned for modification 28 February 1996/Accepted 15 March 1996
 MOLECULAR AND CELLULAR BIOLOGY, June 1996, p. 3012 3022 Vol. 16, No. 6 0270-7306/96/$04.00 0 Copyright 1996, American Society for Microbiology Base Pairing at the 5 Splice Site with U1 Small Nuclear RNA
MOLECULAR AND CELLULAR BIOLOGY, June 1996, p. 3012 3022 Vol. 16, No. 6 0270-7306/96/$04.00 0 Copyright 1996, American Society for Microbiology Base Pairing at the 5 Splice Site with U1 Small Nuclear RNA
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Journal of Experimental Botany Advance Access published August 24, 2013
 extra Botany Journal of Experimental Botany Advance Access published August 24, 2013 Gated communities: apoplastic and symplastic signals converge at plasmodesmata to control cell fates Yvonne Stahl* and
extra Botany Journal of Experimental Botany Advance Access published August 24, 2013 Gated communities: apoplastic and symplastic signals converge at plasmodesmata to control cell fates Yvonne Stahl* and
Bio 111 Study Guide Chapter 17 From Gene to Protein
 Bio 111 Study Guide Chapter 17 From Gene to Protein BEFORE CLASS: Reading: Read the introduction on p. 333, skip the beginning of Concept 17.1 from p. 334 to the bottom of the first column on p. 336, and
Bio 111 Study Guide Chapter 17 From Gene to Protein BEFORE CLASS: Reading: Read the introduction on p. 333, skip the beginning of Concept 17.1 from p. 334 to the bottom of the first column on p. 336, and
Introduction retroposon
 17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
Virus Transmission Getting Out and In
 Plant Cell Monogr (7) E. Waigmann M. Heinlein: Viral Transport in Plants DOI 10.1007/7089_2006_099/Published online: 17 February 2007 Springer-Verlag Berlin Heidelberg 2007 Virus Transmission Getting Out
Plant Cell Monogr (7) E. Waigmann M. Heinlein: Viral Transport in Plants DOI 10.1007/7089_2006_099/Published online: 17 February 2007 Springer-Verlag Berlin Heidelberg 2007 Virus Transmission Getting Out
Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene
 Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene YUELIN ZHANG, WEIHUA FAN, MARK KINKEMA, XIN LI, AND
Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene YUELIN ZHANG, WEIHUA FAN, MARK KINKEMA, XIN LI, AND
The Role of Lipids in Flowering Development of Arabidopsis Enhanced pah1pah2 Plants. Toshiro Ito 1 & Lee Lishi 2
 The Role of Lipids in Flowering Development of Arabidopsis Enhanced pah1pah2 Plants Toshiro Ito 1 & Lee Lishi 2 Department of Biological Sciences, Faculty of Science, National University of Singapore,
The Role of Lipids in Flowering Development of Arabidopsis Enhanced pah1pah2 Plants Toshiro Ito 1 & Lee Lishi 2 Department of Biological Sciences, Faculty of Science, National University of Singapore,
Supplementary Figures
 Supplementary Figures a miel1-2 (SALK_41369).1kb miel1-1 (SALK_978) b TUB MIEL1 Supplementary Figure 1. MIEL1 expression in miel1 mutant and S:MIEL1-MYC transgenic plants. (a) Mapping of the T-DNA insertion
Supplementary Figures a miel1-2 (SALK_41369).1kb miel1-1 (SALK_978) b TUB MIEL1 Supplementary Figure 1. MIEL1 expression in miel1 mutant and S:MIEL1-MYC transgenic plants. (a) Mapping of the T-DNA insertion
Ali Alabbadi. Bann. Bann. Dr. Belal
 31 Ali Alabbadi Bann Bann Dr. Belal Topics to be discussed in this sheet: Particles-to-PFU Single-step and multi-step growth cycles Multiplicity of infection (MOI) Physical measurements of virus particles
31 Ali Alabbadi Bann Bann Dr. Belal Topics to be discussed in this sheet: Particles-to-PFU Single-step and multi-step growth cycles Multiplicity of infection (MOI) Physical measurements of virus particles
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
VIRUSES. Biology Applications Control. David R. Harper. Garland Science Taylor & Francis Group NEW YORK AND LONDON
 VIRUSES Biology Applications Control David R. Harper GS Garland Science Taylor & Francis Group NEW YORK AND LONDON vii Chapter 1 Virus Structure and 2.2 VIRUS MORPHOLOGY 26 Infection 1 2.3 VIRAL CLASSIFICATION
VIRUSES Biology Applications Control David R. Harper GS Garland Science Taylor & Francis Group NEW YORK AND LONDON vii Chapter 1 Virus Structure and 2.2 VIRUS MORPHOLOGY 26 Infection 1 2.3 VIRAL CLASSIFICATION
RNA-Based Plant Protection
 RNA-Based Plant Protection Host-Induced Gene Silencing to Engineer Resistance to FHB Karl-Heinz Kogel Phytopathologie, Gießen RNA-Based Plant Protection Is there an agronomic potential for application?
RNA-Based Plant Protection Host-Induced Gene Silencing to Engineer Resistance to FHB Karl-Heinz Kogel Phytopathologie, Gießen RNA-Based Plant Protection Is there an agronomic potential for application?
Overview of the Expressway Cell-Free Expression Systems. Expressway Mini Cell-Free Expression System
 Overview of the Expressway Cell-Free Expression Systems The Expressway Cell-Free Expression Systems use an efficient coupled transcription and translation reaction to produce up to milligram quantities
Overview of the Expressway Cell-Free Expression Systems The Expressway Cell-Free Expression Systems use an efficient coupled transcription and translation reaction to produce up to milligram quantities
Assembly and movement of a plant virus carrying a green
 Proc. Natl. Acad. Sci. USA Vol. 93, pp. 6286-6290, June 1996 Plant Biology Assembly and movement of a plant virus carrying a green fluorescent protein overcoat SIMON SANTA CRUZ*, SEAN CHAPMAN, ALISON G.
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 6286-6290, June 1996 Plant Biology Assembly and movement of a plant virus carrying a green fluorescent protein overcoat SIMON SANTA CRUZ*, SEAN CHAPMAN, ALISON G.
Cell-to-Cell and Phloem-Mediated Transport of Potato Virus X: The Role of Virions
 The Plant Cell, Vol. 10, 495 510, April 1998 1998 American Society of Plant Physiologists RESEARCH ARTICLE Cell-to-Cell and Phloem-Mediated Transport of Potato Virus X: The Role of Virions Simon Santa
The Plant Cell, Vol. 10, 495 510, April 1998 1998 American Society of Plant Physiologists RESEARCH ARTICLE Cell-to-Cell and Phloem-Mediated Transport of Potato Virus X: The Role of Virions Simon Santa
Songklanakarin J. Sci. Technol. 36 (1), 21-26, Jan. - Feb Original Article. Anurag Sunpapao*, Tomofumi Mochizuki, and Satoshi T.
 Songklanakarin J. Sci. Technol. 36 (1), 21-26, Jan. - Feb. 2014 http://www.sjst.psu.ac.th Original Article Effect of dicer-like proteins2 and 4 and RNA-dependent RNA polymerase1 as RNA silencing components
Songklanakarin J. Sci. Technol. 36 (1), 21-26, Jan. - Feb. 2014 http://www.sjst.psu.ac.th Original Article Effect of dicer-like proteins2 and 4 and RNA-dependent RNA polymerase1 as RNA silencing components
Transcription and RNA processing
 Transcription and RNA processing Lecture 7 Biology W3310/4310 Virology Spring 2016 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at
Transcription and RNA processing Lecture 7 Biology W3310/4310 Virology Spring 2016 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Innate Immunity & Inflammation
 Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse.
 I. Abstract. Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse. Project Investigators: Drs. William M. Wintermantel and Ivan
I. Abstract. Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse. Project Investigators: Drs. William M. Wintermantel and Ivan
Padmanaban Annamalai, A.L.N. Rao* Department of Plant Pathology, University of California, Riverside, CA , USA
 Virology 338 (2005) 96 111 www.elsevier.com/locate/yviro Replication-independent expression of genome components and capsid protein of brome mosaic virus in planta: A functional role for viral replicase
Virology 338 (2005) 96 111 www.elsevier.com/locate/yviro Replication-independent expression of genome components and capsid protein of brome mosaic virus in planta: A functional role for viral replicase
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Hepadnaviruses: Variations on the Retrovirus Theme
 WBV21 6/27/03 11:34 PM Page 377 Hepadnaviruses: Variations on the Retrovirus Theme 21 CHAPTER The virion and the viral genome The viral replication cycle The pathogenesis of hepatitis B virus A plant hepadnavirus
WBV21 6/27/03 11:34 PM Page 377 Hepadnaviruses: Variations on the Retrovirus Theme 21 CHAPTER The virion and the viral genome The viral replication cycle The pathogenesis of hepatitis B virus A plant hepadnavirus
Sarah A. Kopecky and Douglas S. Lyles*
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4658 4669 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4658 4669.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Contrasting
JOURNAL OF VIROLOGY, Apr. 2003, p. 4658 4669 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4658 4669.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Contrasting
Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains
 Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains Jiro KATAOKA*, Chikara MASUTA* and Yoichi TAKANAMI* Abstract
Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains Jiro KATAOKA*, Chikara MASUTA* and Yoichi TAKANAMI* Abstract
The reovirus genome comprises 10 segments of doublestranded
 Reovirus reverse genetics: Incorporation of the CAT gene into the reovirus genome Michael R. Roner* and Wolfgang K. Joklik *Department of Biological Sciences, Center for Molecular Biology and Biotechnology,
Reovirus reverse genetics: Incorporation of the CAT gene into the reovirus genome Michael R. Roner* and Wolfgang K. Joklik *Department of Biological Sciences, Center for Molecular Biology and Biotechnology,
Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral
 doi:10.1038/nature14171 SUPPLEMENTARY DISCUSSION Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral signaling 1. Stress-induced oligomerization of the extracellular domain of
doi:10.1038/nature14171 SUPPLEMENTARY DISCUSSION Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral signaling 1. Stress-induced oligomerization of the extracellular domain of
Hsp90 Interacts Specifically with Viral RNA and Differentially Regulates Replication Initiation of Bamboo mosaic virus and Associated Satellite RNA
 Hsp90 Interacts Specifically with Viral RNA and Differentially Regulates Replication Initiation of Bamboo mosaic virus and Associated Satellite RNA Ying Wen Huang 1,2, Chung Chi Hu 1, Ming Ru Liou 1, Ban
Hsp90 Interacts Specifically with Viral RNA and Differentially Regulates Replication Initiation of Bamboo mosaic virus and Associated Satellite RNA Ying Wen Huang 1,2, Chung Chi Hu 1, Ming Ru Liou 1, Ban
Problem Set 5 KEY
 2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
Some living things are made of ONE cell, and are called. Other organisms are composed of many cells, and are called. (SEE PAGE 6)
 Section: 1.1 Question of the Day: Name: Review of Old Information: N/A New Information: We tend to only think of animals as living. However, there is a great diversity of organisms that we consider living
Section: 1.1 Question of the Day: Name: Review of Old Information: N/A New Information: We tend to only think of animals as living. However, there is a great diversity of organisms that we consider living
Multi-clonal origin of macrolide-resistant Mycoplasma pneumoniae isolates. determined by multiple-locus variable-number tandem-repeat analysis
 JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
MicroRNA and Male Infertility: A Potential for Diagnosis
 Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection
 DOI: 10.1111/J.1364-3703.2010.00696.X Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection WEI-NA HOU 1,2,, CHENG-GUO
DOI: 10.1111/J.1364-3703.2010.00696.X Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection WEI-NA HOU 1,2,, CHENG-GUO
Viral Genetics. BIT 220 Chapter 16
 Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, MD
 AD Award Number: W81XWH-11-1-0126 TITLE: Chemical strategy to translate genetic/epigenetic mechanisms to breast cancer therapeutics PRINCIPAL INVESTIGATOR: Xiang-Dong Fu, PhD CONTRACTING ORGANIZATION:
AD Award Number: W81XWH-11-1-0126 TITLE: Chemical strategy to translate genetic/epigenetic mechanisms to breast cancer therapeutics PRINCIPAL INVESTIGATOR: Xiang-Dong Fu, PhD CONTRACTING ORGANIZATION:
numbe r Done by Corrected by Doctor
 numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Eukaryotic transcription (III)
 Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Section 6. Junaid Malek, M.D.
 Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Circular RNAs (circrnas) act a stable mirna sponges
 Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004
 Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Arabidopsis: Flower Development and Patterning
 Arabidopsis: Flower Development and Patterning John L Bowman, University of California, Davis, California, USA The development of flowers and floral organs is directed by genetic programmes likely to be
Arabidopsis: Flower Development and Patterning John L Bowman, University of California, Davis, California, USA The development of flowers and floral organs is directed by genetic programmes likely to be
Overview: Chapter 19 Viruses: A Borrowed Life
 Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Certificate of Analysis
 Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Clay nanosheets for topical delivery of RNAi for sustained protection against plant viruses
 In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION VOLUME: 3 ARTICLE NUMBER: 16207 Clay nanosheets for topical delivery of RNAi for sustained protection against plant viruses
In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION VOLUME: 3 ARTICLE NUMBER: 16207 Clay nanosheets for topical delivery of RNAi for sustained protection against plant viruses
Bi 8 Lecture 17. interference. Ellen Rothenberg 1 March 2016
 Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Induction of Particle Polymorphism by Cucumber Necrosis Virus Coat Protein Mutants In Vivo
 JOURNAL OF VIROLOGY, Feb. 2008, p. 1547 1557 Vol. 82, No. 3 0022-538X/08/$08.00 0 doi:10.1128/jvi.01976-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Induction of Particle
JOURNAL OF VIROLOGY, Feb. 2008, p. 1547 1557 Vol. 82, No. 3 0022-538X/08/$08.00 0 doi:10.1128/jvi.01976-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Induction of Particle
