BMPR1A signaling is necessary for hair follicle cycling and hair shaft differentiation in mice
|
|
|
- Cuthbert Stewart Boone
- 5 years ago
- Views:
Transcription
1 Development Development Advance Online epress Articles. online First posted publication online on date 17 March 17 March 2004 as /dev Access the most recent version at BMPR1A signaling is necessary for hair follicle cycling and hair shaft differentiation in mice Munehiro Yuhki 1,4, Masahisa Yamada 1,2, *, Masako Kawano 1, Takuji Iwasato 3, Shigeyoshi Itohara 3, Hisahiro Yoshida 5, Masaharu Ogawa 1 and Yuji Mishina 6 1 Laboratory for Cell Culture Development, RIKEN Brain Science Institute, Saitama , Japan 2 Molecular Neuropathology Group, RIKEN Brain Science Institute, Saitama , Japan 3 Laboratory for Behavioral Genetics, RIKEN Brain Science Institute, Saitama , Japan 4 Division of Gene Function in Animals, Nara Institute of Science and Technology, Nara , Japan 5 Immune System Development Group, RIKEN Research Center for Allergy and Immunology, Kanagawa , Japan 6 Laboratory of Reproductive and Developmental Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, NC 27709, USA *Author for correspondence ( masahisa@brain.riken.go.jp) Accepted 7 January 2004 Development 131, Published by The Company of Biologists 2004 doi: /dev Summary Interactions between ectodermal and mesenchymal extracellular signaling pathways regulate hair follicle (HF) morphogenesis and hair cycling. Bone morphogenetic proteins (BMPs) are known to be important in hair follicle development by affecting the local cell fate modulation. To study the role of BMP signaling in the HF, we disrupted Bmpr1a, which encodes the BMP receptor type IA (BMPR1A) in an HF cell-specific manner, using the Cre/loxP system. We found that the differentiation of inner root sheath, but not outer root sheath, was severely impaired in mutant mice. The number of HFs was reduced in the dermis and subcutaneous tissue, and cycling epithelial cells were reduced in mutant mice HFs. Our results strongly suggest that BMPR1A signaling is essential for inner root sheath differentiation and is indispensable for HF renewal in adult skin. Key words: Bone morphogenetic protein, Type I receptor, Hair follicle cycling, Mouse Introduction During skin development, epidermal and dermal cells continuously receive various environmental cues for differentiation and patterning, and finally form tightly interconnected structures. Communication between cells happens through cell-surface receptors, cell-adhesion molecules, extracellular matrix proteins and chemical messaging via secreted molecules (McElwee and Hoffman, 2000). Hair follicles (HFs) are tiny epidermal appendage structures that develop through a series of epitheliummesenchyme interactions. Recent findings obtained from analyzing mutant mice revealed various signaling molecules that are involved in HF development (Huelsken et al., 2001; Karlsson et al., 1999); however, the precise function of each of these molecules in the process of HF development is only partially understood. Tumor necrosis factor receptor; super-family molecules, such as ectodysplasin receptor (EDAR), X-linked ectodysplasin-a2 receptor (XEDAR) and TROY, and their ligand, ectodysplasin (EDA); and other related signal transduction molecules reportedly initiate guard hair follicle and some skin appendage development (Laurikkala et al., 2002; Srivastava et al., 2001; Kojima e al., 2000). Wnt signals and β-catenin were suggested to play important signaling roles in later stages of HF development (Gat et al., 1998; Huelsken et al., 2001; Kishimoto et al., 2000). Bone morphogenetic protein (BMP) signaling has been found to contribute to various developmental processes, including rapid proliferation and morphogenetic movements during the gastrulation period (reviewed by Mishina, 2003; Furuta et al., 1997). During HF development, BMPs are expressed in the hair placode and surrounding mesenchyme, and may participate in stimulating HF induction. For example, BMP family members such as BMP2, BMP3, BMP4 and BMP7 are expressed in the skin and HFs from the embryonic stage to adult (Lyons et al., 1989; Takahashi and Ikeda, 1996; Stelnicki et al., 1998; Wilson et. al., 1999; Botchkarev et al., 2001). The BMP-neutralizing protein Noggin, which interacts with BMP4, triggers HF induction in the hair placode, suggesting that BMP downregulation may be a cue to start HF development (Botchkarev et al., 1999; Botchkarev et al., 2002) (reviewed by Botchkarev, 2003). In hair placode development, BMPs and sonic hedgehog (SHH) expressions are genetically located downstream of β-catenin signaling (Noramly et al., 1999; Huelsken et al., 2001). SHH also regulates HF growth and morphogenesis (Oro and Higgins, 2003; Callahan and Oro, 2001; Chuong et al., 2000; Dlugosz, 1999). β-catenin is necessary in determining the fate of stem cells that form follicular keratinocytes, whereas this is not the case for an epidermal fate decision (Jamora et al., 2003). At the initiation of the anagen phase, follicle progenitor cells of the epidermis induce mesenchymal condensation to form dermal papilla, and then generate proliferating matrix cells.
2 1826 Development 131 (8) Those epidermal cells further differentiate into hair shaft cells. Additionally, some of these stem cells also form the basal layer of the epidermis. Recent evidence suggests that the initiation of feather placodes in chickens is controlled by positive and negative signals mediated by FGFs and BMPs, respectively (Song et al., 1996; Jung et al., 1998). BMPs signaling is exerted through a complex of type I (BMPRI) and type II receptors (BMPRII). BMPR1A (alternatively known as ALK3) is one of three type I receptors for BMPs. Unfortunately, disrupting Bmpr1a in mice blocked mesoderm formation and resulted in intrauterine death before embryonic day 7.5 (E7.5) (Mishina et al., 1995), preventing further investigation into the function of BMP signaling in HF development through BMPR1A, the receptor with the highest affinity for BMP4. In this study, we examined the role of BMP signaling through BMPR1A during generation and maturation of HFs using mutant mice with Bmpr1a deleted in HFs in skin. We found abnormal HF differentiation and reduced cell growth of interfollicular epidermal cells in the fetal skin of these hairspecific Bmpr1a knockout mice (Hair-Bmpr1a KO mice). In postnatal Hair-Bmpr1a KO mice, a reduction in the number of HFs was apparent in 2-week-old and 10-month-old mutant mice which were hairless in affected regions. Epithelial cells in the hair matrix were located separately covering the luminal surface of open hair canals; and, in the hair shaft, inner root sheath (IRS) development was severely impaired. Furthermore, HF epithelial cells of the older mutant mice also had hardly incorporated BrdU. Taken together, our results strongly suggest that BMPR1A signaling is not only essential for the differentiation of the HF in the developmental stage, but also important for epidermal cell proliferation or differentiation in hair cycle renewal during adult life. Materials and methods Generation of mouse lines Emx1-Cre mice and floxed-bmpr1a mice had a mixed 129SvJ:C57BL/6 (3:1) background. Mice were genotyped as previously described (Mishina et al., 2002; Iwasato et al., 2000). For evaluation of the Cre activities, Emx1 Cre/+ mice were bred with CAG- CAT-Z reporter mice (Sakai and Miyazaki, 1997), and double heterozygous (Emx1-Cre/LacZ) mice were obtained. We crossed the female Emx1 Cre/Cre mice with male heterozygous Bmpr1a-null mutant (Bmpr1a +/ ) mice (Mishina et al., 1995) to obtain double heterozygous (Emx1 Cre/+ Bmpr1a +/ ) mice, and further crossed male Emx1 Cre/+ Bmpr1a +/ mice with female homozygous floxed-bmpr1a (Bmpr1a flox/flox ) mice (Mishina et al., 2002) to obtain four types of mice: Emx1 Cre/+ Bmpr1a flox/ ; Emx1 +/+ Bmpr1a flox/ ; Emx1 Cre/+ Bmpr1a flox/+ ; and Emx1 +/+ Bmpr1a flox/+. Emx1 Cre/+ Bmpr1a flox/ mice are Hair-specific Bmpr1a knockout mice, hereafter referred to as Hair- Bmpr1a KO mice. The Hair-Bmpr1a KO mice were viable for 1 year. This study was carried in accordance with the Guide for the Care and Use of Laboratory Animals in the Guidelines of the Society for Neuroscience and authorized by the Animal Care and Use Committee of RIKEN. Immunohistochemistry Frozen cryosections (15 µm) were treated with 3% H 2O 2, incubated with blocking buffer (PBS containing 0.01% Triton X and 1.5% normal goat serum), and incubated overnight at 4 C with monoclonal mouse anti-bromodeoxyuridine (1:100, DAKO), antiphospho-histone H3 (rabbit polyclonal, 1:300, Upstate), anti Met (goat polyclonal antibody, SANTA CRUZ) and PDGFR-α (rat monoclonal antibody, Pharmingen). Sections were incubated in biotinylated antimouse IgG (1:300, Vector Laboratories), horseradish peroxidase conjugated anti-rabbit IgG (MBL) and horseradish peroxidase conjugated anti-rat IgG (Jackson Immunoresearch) antibody diluted in blocking buffer, and DAB reaction. Primary antibodies used were monoclonal anti β-catenin (1:500, Sigma, 15B8), polyclonal antikeratin 5 (1:1000, Covance), polyclonal anti-keratin 6 (K6; kindly provided by Dr Shimomura) (Aoki et al., 2001), anti-traf6 (goat polyclonal antibody, Santa Cruz), anti-notch (goat polyclonal antibody, Santa Cruz) and anti-egfr (goat polyclonal antibody, Santa Cruz). FITC-conjugated goat anti-mouse IgG (1:200, Jackson Immunoresearch), Cy3-conjugated goat anti-rabbit IgG (1:800, Jackson Immunoresearch) and Cy3-conjugated anti-goat IgG (Chemicon) second antibodies were used. Sections were counterstained with Hoechst (Calbiochem). Analysis of apoptosis Apoptotic cells were detected by TUNEL assay on 4% paraformaldehyde fixed cryosections of E15-16 limbs by using the ApopTag Peroxidase kit (Intergen). In situ hybridization In situ hybridization using digoxigenin-labeled (Roche) crna probes was performed on cryosections. Riboprobes for Bmpr1a and Shh were generated as described previously (Mishina et al., 1995; Kato et al., 2001). Skin sections were fixed in 4% formaldehyde, acetylated with 0.5% acetic anhydride in 0.1 M triethanolamine (ph 8.0) for 10 minutes, and rinsed in PBS. The slides were prehybridized in hybridization buffer without crna probe at room temperature for 2 hours and hybridized using a hybridization buffer (50% formamide, 5 SSPE, 1 mg/ml yeast trna, 0.2% SDS) containing 1 µg/ml crna probe at 60 C overnight. Slides were washed in 2 SSC containing 50% formamide at 60 C for 1 hour. Hybridization was detected using an anti-dig Fab (Roche) coupled to alkaline phosphatase using NBT/BCIP. X-gal histochemistry staining Frozen sections (15 µm) were fixed in 4% paraformaldehyde (ph 7.5) for 20 minutes, then washed and incubated with PBS containing 0.01% sodium deoxycholate, 0.02% NP40 and 1 mm MgCl 2 (lacz wash buffer) for 3 hours. For combined X-gal staining with Hematoxylin-Eosin, overnight X-gal staining was processed with PBS containing 1 mg/ml X-gal (5-bromo-4-chloro-3-indoyl-βDgalactopyranoside), 5 mm K-ferrocyanide, 5 mm K-ferricyanide, 0.01% sodium deoxycholate, 0.02% NP40, 1 mm MgCl 2 at 37 C protected from light, then Hematoxylin-Eosin staining was processed. BrdU-labeling experiments BrdU (3 mg/100 g body weight, cell proliferation KIT, Amersham Bioscience) was injected to mice subcutaneously, and skin samples were taken 3 hours after injection, fixed in 4% paraformaldehyde and processed for immunohistochemistry. Electron-micrograph For electronmicroscopic observation of the skin, a postnatal day 30 Hair-Bmpr1a KO mouse and a control mouse (Emx1 Cre/+ Bmpr1a flox/ ) were fixed 2.5% glutaraldehyde and 4% paraformaldehyde. After fixation, the tissue samples were postfixed with 1% (w/v) osmium tetroxide in 0.1 M PB, dehydrated in a graded ethanol series and embedded in epoxy resin (EPON812 and Araldyte CY212, TAAB, Aldermaston, UK). For electron microscopic observation, the skin was sectioned into 80 nm slices with an ultramicrotome (Ultracut-R, Leica, Heidelberg, Germany). Ultrathin sections were stained with uranyl acetate and lead citrate. Electron micrographs recorded on imaging plates through an LEO 912 electron microscope (LEO electron microscopy, Oberkochen, Germany) were scanned and digitized by an FDL 5000 imaging system (Fuji Photofilm, Tokyo, Japan).
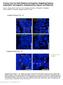
3 BMPR1A signaling and hair follicle cycling 1827 Fig. 1. Analysis of mice expressing Crerecombinase under control of the Emx1 locus. Whole-mount tissue from E13.5 Emx1-Cre/LacZ mice was stained with X-gal (A). Whole-mount tissue from P0 Emx1-Cre/lacZ mice was stained with X-gal (B) and a frozen section from P0 Emx1-Cre/lacZ mice was stained with X-gal, and counterstained with Hematoxylin-Eosin (C,D). Section from P14 Emx1-Cre/lacZ mice was stained with X-gal (E). c, cortex; dp, dermal papilla; ds, dermal sheath; irs, inner root sheath; m, matrix; ors, outer root sheath. Scale bars: 500 µm in A-C; 50 µm in D; 20 µm in E. Results Cre-mediated recombination in skin and Hair- Bmpr1a KO mice To study the function of BMPR1A signaling in the skin, we used a mouse line with a conditional allele of Bmpr1a, in which exon 2 was floxed (Mishina et al., 2002). The Emx1-Cre mouse strain was previously generated by knocking-in Cre recombinase at the Emx1 locus (Iwasato et al., 2000), and this mouse strain expresses Cre in the skin, but not in surrounding tissues, including bone (see below). We bred Emx1-Cre mice with CAG-CAT-Z reporter mice to obtain double heterozygous mice (Emx1-Cre/LacZ mice), and stained histological sections with X-gal. Cre-mediated recombination was expressed over the entire epidermal layer from E13.5 and, 1 day later, guard hair skin follicles around forelimbs and hindlimbs started to express β-galactosidase activity (Fig. 1A, data not shown). In P0 of Emx1-Cre/LacZ mice, Cre-mediated recombination occurred in skin and hair follicles that were derived from the same stem cells in the skin of the limbs (Fig. 1B-D). In HFs, Cre-mediated recombination occurred in a group of postmitotic cells forming the inner root sheath (IRS), outer root sheath (ORS), hair cortex and hair matrix (Fig. 1E). By contrast, Cremediated recombination did not occur in HF mesenchymal components such as the dermal papilla (Fig. 1D) and the dermal sheath (Fig. 1E). Older mice (10 months old) showed the same staining pattern of X-gal (data not shown). Interestingly, Cre activity in these mice was found only in parts of the skin, namely the skin of the limbs, and both the dorsal and ventral side of the basal region of the limbs (Fig. 1A,B, and data not shown). We then crossed the Emx1-Cre mice with floxed-bmpr1a mice to delete exon 2 of Bmpr1a to generate a functional null allele in a hair-specific manner (see Materials and methods). Progeny that were trans-heterozygous for floxed-bmpr1a and Bmpr1a null, and also carried Emx1-Cre (Emx1 Cre/+ Bmpr1a flox/ ; Hair-Bmpr1a KO hereafter) were viable for more than 1 year, but were 10-20% smaller than control mice. Hair loss was observed at Cre-mediated recombination sites of the skin of the limbs (Fig. 2A, arrowheads). The mice showed Cre activity in the same manner as described previously (Fig. 1). Interestingly, these mice also had malformed digits and claws (Fig. 2F,G). Apoptotic cell death was reduced in interdigit areas of E15-16 mutant embryos Fig. 2. Hair-specific ablation of the Bmpr1a gene in mice. Hair- Bmpr1a KO mice (right) lack external hairs at the limbs (arrowheads) at P30 (A). (B-D) The right hindlimbs of control mice at P30: (B) whole limb; (C) instep; (D) outstep. (E-G) Hindlimbs of P30 Hair-Bmpr1a KO mice: (E) whole limb shows an abnormal spreading of first digit and malformed claws in hindlimbs; (F) instep; (G) outstep. (H,I) HE stained section of forelimb from control mice (H) and Hair-Bmpr1a KO mice (I) at E TUNEL analysis in adjacent section of control mice (J) and Hair-Bmpr1a KO mice (K) (see boxed areas in H and I). The number of TUNEL-labeled nuclei in first digit of Hair-Bmpr1a KO mice limb at E15-16 was reduced (K) compared with control mice (J). In situ hybridization analysis of Bmpr1a using an exon 2- specific probe in a section from P30 control (L,M). The expression of Bmpr1a is efficiently abolished in the P30 Hair-Bmpr1a KO mice (N). c, cortex; irs, inner root sheath; m, matrix; me, medulla; ors, outer root sheath. Scale bars: 20 mm in A; 2 mm in B-G; 100 µm in H,I; 50 µm in J-N.
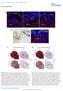
4 1828 Development 131 (8) Fig. 3. Abnormal hair follicle formation in Hair-Bmpr1a KO mice. Hematoxylin-Eosin (HE)-stained sections of hindlimbs from Hair-Bmpr1a KO mice are shown in the developmental stage of HF. First anagen phase of P14 control mice (A). Hair follicles in the control skin retract into dermis, maintaining close contact between the dermal papilla and the epithelial hair shaft in P14 control mice (arrow). Abnormally differentiated hair shafts were observed in P14 Hair-Bmpr1a KO mice (B-D). Retracting epithelial hair shafts that separate from the dermal papilla and form cysts in hair canals were observed in P14 Hair- Bmpr1a KO mice (arrow, B). Section from P30 control mice shows second anagen phase of HF (E). In P30 Hair-Bmpr1a KO mice (F-H), nearly all of the canals have small cysts (arrowhead, H) in the HF. Section from older (10-month-old) control mice was processed with HE staining (I). The hair canals were wider and large cysts (arrowhead) were observed in hair canals in older Hair-Bmpr1a KO mice (J-L). The number of HFs per mm 2 was counted in P14, P30 and mature mice (n=7 per group) (M). Data are given as mean±s.e.m.; *P<0.05; **P<0.01; Student s t-test. d, dermis; s, subcutis; pc, panniculus carnosus. Scale bars: 100 µm in A,B,E,F,I,J; 50 µm in C,D,G,H,K,L. (Fig. 2J,K) that may explain these abnormalities. In situ hybridization data showed that Bmpr1a was expressed throughout the epidermal layer of HF, ORS, IRS and hair matrix regions in control mice (Fig. 2L,M). Yet, the exon 2- specific probe failed to detect expression of Bmpr1a in the mutant, suggesting that Cre-mediated recombination was completed by P14 (Fig. 2N). Hair defects in Hair-Bmpr1a KO mice BMPs are known to be environmental factors for the development of HFs. However, the specific role of BMP signaling through individual receptors in the diversification of epithelial cells and differentiation of HFs function is poorly understood. To explore BMPR1A signaling involvement in the development of HF epithelial cells, we performed morphological analyses during skin development in mutant mice. In mice, initial development of HFs starts from E13 to E18, and extends to postnatal day 14 (P14). In secondary HF development, the postnatal first anagen phase begins around P25 and lasts until P40. The patterning of skin was abnormal in Hair-Bmpr1a KO mice at the end of the initial phase at P14 (Fig. 3A-D). At the end of this first growth phase, around P14, new hair bulbs start to appear in the subcutaneous space of control mice. The morphology of HFs in the skin of Hair- Bmpr1a KO mice at P14 showed impaired development in hair formation during this same period. Most of the hair failed to pierce the epidermal surface, remaining stacked and distorted in the subcutaneous region (Fig. 3B). Only 10% of HFs had hair shafts and these were abnormally shaped (Fig. 3C). The distorted hair canals of most of the hair follicles had ectopic keratinized centers (Fig. 3D). In the second anagen phase at P30, newly generated HFs were observed in the deep layer of the subcutaneous region of control mice (Fig. 3E). Although developing HFs were observed in the subcutaneous region of Hair-Bmpr1a KO mice, the amount was significantly less, and retracting epithelial hair shafts had separated from the dermal papilla (Fig. 3E-H,M). Moreover, the hair shaft of KO mice did not extend to the epidermal ceiling. Instead, small cysts formed in the hair canals (Fig. 3H). Any hair that had developed disappeared during this postnatal hair cycle. In addition, unlike control mice, in the skin of Hair-Bmpr1a KO mice, an insignificant number of newly generated HFs could be found in deeper regions of the subcutaneous skin. (Fig. 3I,J,M). Most of the hair canals had cysts including keratinized debris, which
5 BMPR1A signaling and hair follicle cycling 1829 Fig. 4. Altered proliferation of hair follicle cells in Hair-Bmpr1a KO mice. Phosphorylated histone (PH) immunostaining signals exhibit dividing epithelial cells of HFs from P30 control mice (A). P30 Hair-Bmpr1a KO mice showed that PH-positive cells were located abnormally in hair canals of HF (B). Older (10-month-old) Hair-Bmpr1a KO mice showed a significantly reduced index of PH immunosignals (D) than did control mice (C). BrdU incorporation in Hair-Bmpr1a KO mice (F) was severely reduced compared with control mice (E). The number of PH-positive cells per HF was counted in P14, P30 and older mice (n=24 per group) (G). Data are given as mean±s.e.m.; *P<0.05; Student s t-test. Scale bars: 100 µm in A (left panel), B (left panel) and C-F; 50 µm in A (right panel) and B (right panel). was enlarged and surrounded by the multilayered epithelium (Fig. 3J-L). Hair follicle epidermal cell cycling in Hair-Bmpr1a KO mice To determine whether HF developmental defects in Hair- Bmpr1a KO mice resulted from an impairment of the cell proliferating ability of HF epidermal cells, we further examined HF development during the anagen phase in KO and control mice. Phosphorylated histone (PH) immunostaining signals showed cells to be in the M phase. Control mice had dividing epithelial cells in HFs; however, in Hair-Bmpr1a KO mice, PH-positive cells were abnormally located in HF hair canals at P30 (Fig. 4A,B). The number of PH-positive cells per HF was significantly reduced in Hair-Bmpr1a KO mice at P30 and at P14 (Fig. 4G). In 10-month-old (older) Hair-Bmpr1a KO mice, both PH immunosignals (Fig. 4C,D,G) and BrdU labeling indices (Fig. 4E,F) were significantly reduced. The number of HFs was also significantly reduced in Hair-Bmpr1a KO mice (Fig. 3M). Taken together, our data suggests that the proliferating ability of hair follicle epidermal cells was severely impaired in Hair-Bmpr1a KO mice. Although the loss of the cell proliferating ability may explain the absence of new HF development in Hair-Bmpr1a KO mice, the distorted shapes and appearance of abnormal cysts prompted further analysis of HF epidermal cell differentiation. In fact, the IRS structure, which was clearly detected by anti IRS-keratin 6 antibody in control mice, was severely impaired with observable hair loss in Hair-Bmpr1a KO mice at P14 (compare Fig. 5A,B with E,F). By contrast, the expression of keratin 5, which marks the ORS, appeared normal in KO mice hair follicles at P14 (compare Fig. 5C,D with G,H). The electron density indicated that multiple layers of ORS with trichohyalin granules (th) were located beside a medulla-(me) like structure in Hair-Bmpr1a KO mice at P30 (compare Fig. 5I with J). However, the structure of the IRS, including Henle s layer (he), Huxley s layer (hu) and the cuticle of IRS (ci) disappeared, as did the cortex of the hair shaft (c) in Hair-Bmpr1a KO mice (Fig. 5I,J). These results indicated that the hair of Hair- Bmpr1a KO mice failed to differentiate into the distinct structures of the IRS. This finding suggests that BMPR1A signaling may be the crucial factor in controlling the cell fate of epithelial cells in the matrix. We also carried out additional immunohistochemical experiments to identify differentiation marker(s) of IRS. We found that the expression of notch, an early differentiation marker of IRS (Kopan and Weintraub, 1993), was relatively normal in the epidermal cells in the Hair-Bmpr1a KO mice (Fig. 6). This implies that BMPR1A signaling is necessary for the differentiation of IRS to form follicular keratinocytes, and that stem cells of the hair matrix only differentiate into a single lineage (outer root sheath) in the absence of BMPR1A. Bmpr1a deletion affects the expression of β-catenin and Shh To further explore the mechanism by which BMPR1A signaling acts as a regulator in HF induction, we studied the expression profiles of several factors essential for HF development such as Noggin, Shh and β-catenin (Botchkarev et al., 1999; Kulessa et al., 2000; Callahan and Oro, 2001; Huelsken et al., 2001; Jamora et al., 2003). Shh, the regulator of proliferation in HFs, is also know to be a downstream factor of β-catenin signaling (Chiang et al., 1999; Huelsken et al., 2001). The expression of Shh is normally found at the tip of the placode and is restricted to the anterior or posterior hair matrix in mature anagen HF in the control mice (Fig. 7A,C,E) (Gat et al., 1998). Shh was expressed in the distal hair bulb even in 10-month old Hair-Bmpr1a KO mice (Fig. 7B,D,F). Furthermore, the increased expression area of β-catenin in the KO mice was detected by immunohistological analysis (Fig. 7I,J). Interestingly, the expression of β-catenin in the epidermis of Hair-Bmpr1a KO mice was wider than in control mice (Fig. 7I,J). Translocation of β-catenin into the nucleus associates with β-catenin signaling activity. In Hair-Bmpr1a KO mice, the translocation of β-catenin into the nucleus was significantly (P<0.05) reduced (control mice 3.5±0.5%; Hair-Bmpr1a KO mice, 1.9±0.5%; mean±s.e.m.% of translocated β-catenin per
6 1830 Development 131 (8) Fig. 5. Formation of the IRS in Hair-Bmpr1a KO mice and control mice. Frozen sections of hindlimb skin were prepared from control mice (A-D) and Hair-Bmpr1a KO mice (E-H) at P14. Expression of keratin 6 (A,B,E,F) in the IRS, and expression of keratin 5 (C,D,G,H) in the ORS were detected by immunofluorescence staining. B,D,F and H are merged with Hoechst (blue) for nuclear staining. Formation of IRS characterized by the expression of IRS-keratin 6 was severely reduced in Hair-Bmpr1a KO mice (E,F). (I,J) Ultrastructure of hair follicle in control mice (I) and Hair-Bmpr1a KO mice (J) at P30. Insets show keratin 5 (red) and Hoechst (blue) signals at P30. Absence of IRS cells resulted in proliferation of keratin-5-positive ORS cells. c, cortex; ci, cuticle of IRS; he, Henle s layer; hu, Huxley s layer; irs, inner root sheath; me, medulla; ors, outer root sheath; th, trichohyalin granule. Scale bars: 100 µm in A-H and insets in I,J; 5 µm in I,J. total β-catenin positive cells; n=15 each group). The skin of Hair-Bmpr1a KO mice appears to have normal skin cycling with no abnormalities observed in the basal layer of skin (Fig. 7H,J). These results suggest that the expression of β-catenin is correlated with wider ORS formation in Hair-Bmpr1a KO mice. Reduced nuclear β-catenin localization may be associated with impaired keratin genesis. The expression of several other growth regulators (PDGFRα, TRAF6, EGFR, notch and Met), which are important in the morphogenesis of HF development, was also examined. The expression levels of all these genes in HF epidermal cells in Hair-Bmpr1a KO mice seemed to be comparable with control mice HF (Fig. 6). These results are intriguing because both PH-positive cells and BrdU incorporation indices were severely decreased in the hair bulb in 10-month-old KO mice (Fig. 4D,F,G). These data suggested that expression of Shh or other growth regulators are not associated with the cell proliferating ability of Hair-Bmpr1a KO mice. Discussion A number of studies indicate that BMPs control hair maturation (reviewed by Botchkarev, 2003; Furuta et al., 1997; Jung et al., 1998; Kulessa et al., 2000), however, the nature of BMP signaling contribution through individual receptors remained unknown. In this paper, we demonstrate that epithelial cells forming the hair matrix are individually located and cover the luminal surface of opened hair canals, and that, in the hair shaft, inner root sheath (IRS) development was severely impaired in Hair-Bmpr1a KO mice. Furthermore, epithelial cells that remained HFs in older mutant mice did not significantly incorporate BrdU. Taken together, our results strongly suggest that BMPR1A signaling is not only essential for HF differentiation during developmental, but also important for epidermal cell proliferation and, later, the differentiation in hair cycle renewal in adults. An abnormal pattern of HF structure was observed in Hair- Bmpr1a KO mice in both the first and second anagen phases of hair cycle development. Still, part of the HF induction normally occurred in Hair-Bmpr1a KO mice during the first anagen. Because, the induction of HF starts at about E14 in the limbs, where Emx1-Cre is expressed as early as E13.5, it is likely that BMPR1A signaling is not required to initiate hair follicle genesis. EDA, EDAR and TRAF6 could be the important triggers for this induction (Laurikkala et al., 2002; Srivastava et al., 2001; Kojima et al., 2000). Relatively normal expression levels of these genes found in Hair-Bmpr1a KO mice support this idea. However, we cannot completely exclude the possibility that some of the cells in HFs may retain enough BMPR1A to induce normal HF development. How does BMPR1A signaling contribute to HF development? Kulessa et al. have previously reported that disrupting the BMP signal in HF-specific mice overexpressing Noggin selectively inhibited the formation of the medulla and the hair shaft. However, Hair-Bmpr1a KO mice showed impaired IRS formation, as indicated by a lack of keratin6 expression, and despite the fact that notch expression, an early marker of IRS (Kopan and Weintraub, 1993), was relatively normal. Thus, although IRS progenitor cells in Hair-Bmpr1a
7 BMPR1A signaling and hair follicle cycling 1831 Fig. 6. The expression of growth regulators in Hair-Bmpr1a KO mice and control mice. Expression of EGFR, notch, TRAF6, PDGFRα and Met in HFs from P30 Hair-Bmpr1a KO mice and P30 control mice (as indicated) was analyzed by immunohistochemistry (shown in green for antibodies, Hoechst blue for EGFR, notch and TRAF6; brown for antibodies for PDGFRα and Met). The expression levels of all of these regulators in Hair-Bmpr1a KO mice were comparable with those in control mice. No signal was detected in negative controls (normal goat serum was the control for anti-egfr, notch, TRAF6, and Met goat polyclonal antibodies; no primary antibody was used as a control for anti-pdgfrα monoclonal antibody). Scale bars: 100 µm. KO mice are likely to be generated, further differentiation involving IRS-keratin 6 expression in the absence of BMPR1A signaling was impaired. These results suggest that BMP signaling, which is essential for early differentiation of IRS, may not be transduced by BMPR1A, and that overexpressed Noggin interferes with the other receptors mediating signaling in the early stage of IRS differentiation. We found malformation of HFs, impaired cell cycling of matrix cells, and complete hair loss in the second anagen. Impaired HF development can be seen in P14 Hair-Bmpr1a KO mice because HF formation in these mice was already impaired in the first anagen. We found notch expression in abnormal HF in Hair-Bmpr1a KO mice, which suggests that the commitment to the IRS cell lineage occurs normally. Although, IRS-keratin 6, a late differentiation marker for both Huxley s and Henle s layers, was present in the hair follicle bulb of Hair-Bmpr1a KO mice (Fig. 5E), the structures were missing (Fig. 5J). These differentiation impairments strongly suggest that BMPR1A signaling plays an essential role in the differentiation of IRS. Abnormal HFs in our mice have only multiple layers of ORS and an immature hair shaft that produce cysts composed of keratinized debris. Therefore, our results suggest that the abnormal differentiation of IRS leads to abnormal HF formation that may eventually impair the cell cycling of epithelial cells. From these finding, we suspect that bulge formation, originating from HF stem cells, is also affected in the absence of BMPR1A signaling. But, how does the absence of Bmpr1a contribute to the formation of the bulge structure? The skin of Hair- Bmpr1a KO mice appears to have normal skin cycling with no abnormalities observed in the basal layer of skin except hair loss (Fig. 7H,J). However, bulge structure is therefore likely to unformed in adequate region, which interfere with receiving the key regulator regeneration signals. Interestingly, the expression area of TRAF6, which is genetically down stream of XEDAR or TROY (Laurikkala et al., 2002; Srivastava et al., 2001; Kojima et al., 2000), was increased in the HFs of the Hair-Bmpr1a KO mice compared with control (Fig. 6). The constitutive expression of TRAF6 may represent the inhibition of TRAF6 signaling in the Hair-Bmpr1a KO mice, if feedback regulation is the case. TRAF6 mutant mice are hypohidrotic (anhidrotic), with ectodermal dysplasia (HED) (Naito et al., 2002). Moreover, impaired TRAF6 signaling may contribute to the pathogenesis of a certain type of abnormality in the formation of HF and sebaceous gland that has impaired formation of bulge architecture. Further investigations of Hair-Bmpr1a KO mice and TRAF6 mutant mice might reveal a connection between these two signaling pathways. The development of a perinatal stage-specific disruption system for Bmpr1a in a HF-specific manner would facilitate this comparison. A similar phenotype, in which the inner root sheath is impaired in a similar fashion as, in Hair-Bmpr1a KO mice, was recently observed in mice harboring a mutation of the β- catenin gene (Huelsken et al., 2001), suggesting that β-catenin is necessary for the determination of the cell fate of stem cells to form follicular keratinocytes. Notably, abnormal hair shaft
8 1832 Development 131 (8) Bmpr1a in HFs. The reduction of IRS was obvious in Hair- Bmpr1a KO mice; yet, the ORS formed multiple layers in the mice. In human studies, both BMPR1A and BMPR1B were detected in the HFs in adult and fetal skin (Hwang et al., 2001). Perhaps BMP signaling, through BMPR1B, compensates for the loss of BMPR1A signaling during the generation of the ORS. An unsolved problem of interest is how to define the unique function of BMP signaling through each type I receptor in the HF. Analyzing BMPR1B KO mice may address this question. Taken together, this suggests that BMPR1A signaling has a role in differentiation during maturation of the IRS from hair matrix epithelial cells. Abnormal hair cycling occurs in the absence of BMPR1A signaling, regardless of the relatively normal expression levels of other growth regulation genes for HFs, suggesting that BMPR1A signaling is one of the key components for hair cycling. Note added in proof Since the submission of this paper, one paper has appeared demonstrating that importance of BMP signaling for development of IRS in the mouse (Kobielak et al., 2003). We thank Drs C. Itakura and M. Ray for helpful discussions and careful reading of our manuscript before submission. Thanks to Ms Y. Okumura, S. Yamamoto and B. L. La Madeleine for their expert technical assistance, and to Ms T. Yoshida (Brain Science and Life Technology Research Foundation) for Ultrastructure analysis. The anti-k6 antibody was a very generous gift from Dr Y. Shimomura, and CAG-CAT-lacZ reporter mice were provided by Dr Miyazaki. This work was supported by The Ministry of Education, Culture, Sports and Technology Grant-in-Aid for Young Scientists (B) to M.Y. Fig. 7. Expression of Shh and β-catenin in Hair-Bmpr1a KO mice and control mice. (A-F) A digoxigenin-labeled Shh riboprobe was used on sections of skin from hindlimb of P14 (A,B), P30 (C,D) and older (10-month-old; E,F) mice. (G-J) The expression area of β- catenin was wider in P30 Hair-Bmpr1a KO mice (I,J) than the P30 control mice (G,H). Scale bars: 100 µm in A-G,I; 50 µm in H,J. differentiations, subsequent loss of hair, and cyst formation in widened canals in the β-catenin mutant mice are the same phenotypes as seen in our mice. Interestingly, the expression of cytekeratin 17, as a marker of the ORS, is upregulated in mutant mice. Based upon the results of β-catenin KO mice, which show the loss of BMP and Shh expression in the mice, the expression of BMP and Shh is controlled by β-catenin in a complex process determining the fate of skin stem cells (reviewed by Oro and Scott, 1998; Barsh, 1999; Huelsken et al., 2001). In Hair-Bmpr1a KO mice, the expression areas of β-catenin and Shh were increased. These results suggest that BMPR1A signaling and β-catenin signaling may be responsible for different functions in HF development and presumably interact with each other. These signals may be using a common downstream pathway; however, it is also possible that they act in parallel to regulate HF differentiation. In situ hybridization demonstrated broad expression of References Aoki, N., Sawada, S., Rogers, M. A., Schweizer, J., Shimomura, Y., Tsujimoto, T., Ito, K. and Ito, M. (2001). A novel type II cytokeratin, mk6irs, is expressed in the Huxley and Henle layers of the mouse inner root sheath. J. Invest. Dermatol. 116, Barsh, G. (1999). Of ancient tales and hair less tails. Nat. Genet. 22, Botchkarev, V. A., Botchkareva, N. V., Roth, W., Nakamura, M., Chen, L. H., Herzog, W., Lindner, G., McMahon, J. A., Peters, C., Lauster, R., McMahon, A. P. and Paus, R. (1999). Noggin is a mesenchymally derived stimulator of hair-follicle induction. Nat. Cell. Biol. 1, Botchkarev, V. A., Botchkareva, N. V., Nakamura, M., Huber, O., Funa, K., Lauster, R., Paus, R. and Gilchrest, B. A. (2001). Noggin is required for induction of the hair follicle growth phase in postnatal skin. FASEB J. 15, Botchkarev, V. A., Botchkareva, N. V., Sharov, A. A., Funa, K., Huber, O. and Gilchrest, B. A. (2002). Modulation of BMP signaling by noggin is required for induction of the secondary (nontylotrich) hair follicles. J. Invest. Dermatol. 118, Botchkarev, V. A. (2003). Bone morphogenetic proteins and their antagonists in skin and hair follicle biology. J. Invest. Dermatol. 120, Callahan, C. A. and Oro, A. E. (2001). Monstrous attempts at adnexogenesis: regulating hair follicle progenitors through Sonic hedgehog signaling. Curr. Opin. Genet. Dev. 11, Chiang, C., Swan, R. Z., Grachtchouk, M., Bolinger, M., Litingtung, Y., Robertson, E. K., Cooper, M. K., Gaffield,W., Westphal, H., Beachy, P. A. and Dlugosz, A. A. (1999). Essential role for Sonic hedgehog during hair follicle morphogenesis. Dev. Biol. 205, 1-9. Chuong, C. M., Patel, N., Lin, J., Jung, H. S. and Widelitz, R. B. (2000). Sonic hedgehog signaling pathway in vertebrate epithelial appendage morphogenesis: perspectives in development an evolution. Cell Mol. Life Sci. 57, Dlugosz, A. (1999). The Hedgehog and the hair follicle: growing relationship. J. Clin. Invest. 104, Furuta, Y., Piston, D. W. and Hogan, B. L. M. (1997). Bone morphogenetic
9 BMPR1A signaling and hair follicle cycling 1833 proteins (BMPs) as regulators of dorsal forebrain development. Development 124, Gat, U., DasGupta, R., Degenstein, L. and Fuchs, E. (1998). De novo hair follicle morphogenesis and hair tumors in mice expressing a truncated betacatenin in skin. Cell. 95, Huelsken, J., Vogel, R., Erdmann, B., Cotsarelis, G. and Birchmeier, W. (2001). beta-catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell 105, Hwang, E. A., Lee, H. B. and Tark, K. C. (2001). Comparison of Bone Morphogenetic Protein receptors expression in the fetal and adult skin. Yonsei Med. J. 42, Iwasato, T., Datwani, A., Wolf, A. M., Nishiyama, H., Taguchi, Y., Tonegawa, S., Knopfel, T., Erzurumlu, R. S. and Itohara, S. (2000). Cortex-restricted disruption of NMDAR1 impairs neuronal patterns in the barrel cortex. Nature 406, Jamora, C., DasGupta, R., Kocieniewski, P. and Fuchs, E. (2003). Links between signal transduction, transcription and adhesion in epithelial bud development. Nature 422, Jung, H. S., Francis-West, P. H., Widelitz, R. B., Jiang, T. X., Ting, B. S., Tickle, C., Wolpert, L. and Chuong, C. M. (1998). Local inhibitory action of BMPs and their relationships with activations in feather formation: implication for periodic patterning. Dev. Biol. 196, Karlsson, L., Bondjers, C. and Betsholtz, C. (1999). Roles for PDGF-A and sonic hedgehog in development of mesenchymal components of the hair follicle. Development 126, Kato, M., Seki, N., Sugano, S., Hashimoto, K., Masuho, Y., Muramatsu, M., Kaibuchi, K. and Nakafuku, M. (2001). Identification of sonic hedgehog-responsive genes using cdna microarray. Biochem. Biophys. Res. Commun. 289, Kishimoto, J., Burgeson, R. E. and Morgan, B. A. (2000). Wnt signaling maintains the hair-inducing activity of the dermal papilla. Genes Dev. 14, Kobielak, K., Pasolli, H. A., Alonso, L., Polak, L. and Fuchs, E. (2003). Defining BMP functions in the hair follicle by conditional ablation of BMP receptor IA. J. Cell Biol. 163, Kojima, T., Morikawa, Y., Copeland, N. G., Gilbert, D. J., Jenkins, N. A., Senba, E. and Kitamura, T. (2000). TROY, a newly identified member of the tumor necrosis factor receptor superfamily, exhibits a homology with Edar and is expressed in embryonic skin and hair follicles. J. Biol. Chem. 275, Kopan, R. and Weintraub, H. (1993). Mouse notch: expression in hair follicles correlates with cell fate determination. J. Cell Biol. 121, Kulessa, H., Turk, G. and Hogan, B. L. (2000). Inhibition of Bmp signaling affects growth and differentiation in the anagen hair follicle. EMBO J. 19, Laurikkala, J., Pispa, J., Jung, H. S., Nieminen, P., Mikkola, M., Wang, X., Saarialho-Kere, U., Galceran, J., Grosschedl, R. and Thesleff, I. (2002). Regulation of hair follicle development by the TNF signal ectodysplasin and its receptor Edar. Development. 129, Lyons, K. M., Pelton, R. W. and Hogan, B. L. (1989). Patterns of expression of murine Vgr-1 and BMP-2a RNA suggest that transforming growth factorbeta-like genes coordinately regulate aspects of embryonic development. Genes Dev. 3, McElwee, K. and Hoffmann, R. (2000). Growth factors in early hair follicle morphogenesis. Eur. J. Dermatol. 10, Mishina, Y. (2003). Function of bone morphogrnrtic protein signaling during mouse development, Front. Biosci. 8, d855-d869. Mishina, Y., Suzuki, A., Ueno, N. and Behringer, R. R. (1995). Bmpr encodes a type I bone morphogenetic protein receptor that I essential for gastrulation during mouse embryogenesis. Genes Dev. 9, Mishina,Y., Hanks, M. C., Miura, S., Tallquist, M. D. and Behringer, R. R. (2002). Generation of Bmpr/Alk3 conditional knockout mice. Genesis 32, Naito, A., Yoshida, H., Nishioka, E., Satoh, M., Azuma, S., Yamamoto, T., Nishikawa, S. and Inoue, J. (2002). TRAF6-deficient mice display hypohidrotic ectodermal dysplasia. Proc. Natl. Acad. Sci. USA 99, Noramly, S., Freeman, A. and Morgan, B. A. (1999). beta-catenin signaling can initiate feather bud development. Development 126, Oro, A. E. and Scott, M. P. (1998). Splitting hairs: dissecting roles of signaling systems in epidermal development. Cell 95, Oro, A. E. and Higgins, K. (2003). Hair cycle regulation of Hedgehog signal reception. Dev. Biol. 255, Sakai, K. and Miyazaki, J. (1997). A transgenic mouse line that retains Cre recombinase activity in mature oocytes irrespective of the cre transgene transmission. Biochem. Biophys. Res. Commun. 237, Song, H., Wang, Y. and Goetinck, P. F. (1996). Fibroblast growth factor can replace ectodermal signaling for feather development. Proc. Natl. Acad. Sci. USA 93, Srivastava, A. K., Durmowiczm, M. C., Hartungm, A. J., Hudsonm, J., Ouzts, L. V., Donovan, D. M., Cui, C. Y. and Schlessinger, D. (2001). Ectodysplasin-A1 is sufficient to rescue both hair growth and sweat glands in Tabby mice. Hum. Mol. Genet. 10, Stelnicki, E. J., Longaker, M. T., Holmes, D., Vanderwall, K., Harrison, M. R., Largman, C. and Hoffman, W. Y. (1998). Bone morphogenetic protein-2 induces scar formation and skin maturation in the second trimester fetus. Plast. Reconstr. Surg. 101, Takahashi, H. and Ikeda, T. (1996). Transcripts for two members of the transforming growth factor-beta superfamily BMP-3 and BMP-7 are expressed in developing rat embryos. Dev. Dyn. 207, Wilson, N., Hynd, P. I. and Powell, B. C. (1999). The role of BMP-2 and BMP-4 in follicle initiation and the murine hair cycle. Exp. Dermatol. 8,
The Beauty of the Skin
 The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
Epithelial Bmpr1a regulates differentiation and proliferation in postnatal hair follicles and is essential for tooth development
 Research article 2257 Epithelial Bmpr1a regulates differentiation and proliferation in postnatal hair follicles and is essential for tooth development Thomas Andl 1, Kyung Ahn 3, Alladin Kairo 1, Emily
Research article 2257 Epithelial Bmpr1a regulates differentiation and proliferation in postnatal hair follicles and is essential for tooth development Thomas Andl 1, Kyung Ahn 3, Alladin Kairo 1, Emily
Vertebrate Limb Patterning
 Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Embryonic hair follicle fate change by augmented β-catenin through Shh and Bmp signaling
 RESEARCH REPORT 367 Development 136, 367-372 (2009) doi:10.1242/dev.021295 Embryonic hair follicle fate change by augmented β-catenin through Shh and Bmp signaling Kentaro Suzuki 1,2, Yuji Yamaguchi 3,
RESEARCH REPORT 367 Development 136, 367-372 (2009) doi:10.1242/dev.021295 Embryonic hair follicle fate change by augmented β-catenin through Shh and Bmp signaling Kentaro Suzuki 1,2, Yuji Yamaguchi 3,
Supplementary Information
 Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
Supporting Information
 Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
TISSUE-SPECIFIC STEM CELLS
 TISSUE-SPECIFIC STEM CELLS Bone Morphogenetic Protein Signaling Inhibits Hair Follicle Anagen Induction by Restricting Epithelial Stem/Progenitor Cell Activation and Expansion JIWANG ZHANG, a XI C. HE,
TISSUE-SPECIFIC STEM CELLS Bone Morphogenetic Protein Signaling Inhibits Hair Follicle Anagen Induction by Restricting Epithelial Stem/Progenitor Cell Activation and Expansion JIWANG ZHANG, a XI C. HE,
Localization of Shh expression by Wnt and Eda affects axial polarity and shape of hairs
 Developmental Biology 305 (2007) 246 261 www.elsevier.com/locate/ydbio Localization of Shh expression by Wnt and Eda affects axial polarity and shape of hairs Brigitte Hammerschmidt, Thomas Schlake Max-Planck
Developmental Biology 305 (2007) 246 261 www.elsevier.com/locate/ydbio Localization of Shh expression by Wnt and Eda affects axial polarity and shape of hairs Brigitte Hammerschmidt, Thomas Schlake Max-Planck
Supporting Information
 Supporting Information Voutilainen et al. 0.07/pnas.06709 SI Materials and Methods Animals. The generation and genotyping of K-da-A (K- da) () and N-κ reporter mice () has been described earlier. oth strains
Supporting Information Voutilainen et al. 0.07/pnas.06709 SI Materials and Methods Animals. The generation and genotyping of K-da-A (K- da) () and N-κ reporter mice () has been described earlier. oth strains
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and
 Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Bone Morphogenetic Protein Signaling Regulates Postnatal Hair Follicle Differentiation and Cycling
 American Journal of Pathology, Vol. 165, No. 3, September 2004 Copyright American Society for Investigative Pathology Bone Morphogenetic Protein Signaling Regulates Postnatal Hair Follicle Differentiation
American Journal of Pathology, Vol. 165, No. 3, September 2004 Copyright American Society for Investigative Pathology Bone Morphogenetic Protein Signaling Regulates Postnatal Hair Follicle Differentiation
Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse
 Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Notch signaling in bulge stem cells is not required for selection of hair follicle fate
 RESEARCH REPORT 891 Development 136, 891-896 (2009) doi:10.1242/dev.030700 Notch signaling in bulge stem cells is not required for selection of hair follicle fate Shadmehr Demehri and Raphael Kopan* Notch
RESEARCH REPORT 891 Development 136, 891-896 (2009) doi:10.1242/dev.030700 Notch signaling in bulge stem cells is not required for selection of hair follicle fate Shadmehr Demehri and Raphael Kopan* Notch
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
ErbB4 migrazione I parte. 3- ErbB4- NRG1
 ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling
 The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
Tetrapod Limb Development
 IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
INTRODUCTION. Developmental Biology 229, (2001) doi: /dbio , available online at
 Developmental Biology 229, 443 455 (2001) doi:10.1006/dbio.2000.9955, available online at http://www.idealibrary.com on TNF Signaling via the Ligand Receptor Pair Ectodysplasin and Edar Controls the Function
Developmental Biology 229, 443 455 (2001) doi:10.1006/dbio.2000.9955, available online at http://www.idealibrary.com on TNF Signaling via the Ligand Receptor Pair Ectodysplasin and Edar Controls the Function
Emx2 patterns the neocortex by regulating FGF positional signaling
 Emx2 patterns the neocortex by regulating FGF positional signaling Tomomi Fukuchi-Shimogori and Elizabeth A Grove Presented by Sally Kwok Background Cerebral cortex has anatomically and functionally distinct
Emx2 patterns the neocortex by regulating FGF positional signaling Tomomi Fukuchi-Shimogori and Elizabeth A Grove Presented by Sally Kwok Background Cerebral cortex has anatomically and functionally distinct
BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor
 BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor Kateryna Kyrylkova 1, Sergiy Kyryachenko 1, Brian Biehs 2 *, Ophir Klein 2, Chrissa Kioussi 1 *, Mark
BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor Kateryna Kyrylkova 1, Sergiy Kyryachenko 1, Brian Biehs 2 *, Ophir Klein 2, Chrissa Kioussi 1 *, Mark
Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials)
 Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials) Sunny Y. Wong, Allen D. Seol, Po-Lin So, Alexandre N. Ermilov, Christopher K.
Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials) Sunny Y. Wong, Allen D. Seol, Po-Lin So, Alexandre N. Ermilov, Christopher K.
Tetrapod Limb Development
 Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Cytological and Histological Study of Adult and Neonate Epidermis in Thick and Thin Skin of Various Anatomical Sites
 Available online on www.ijpqa.com International Journal of Pharmaceutical Quality Assurance 218; 9(2); 174-179 doi: 1.25258/ijpqa.v9i2.13642 ISSN 975 956 Research Article Cytological and Histological Study
Available online on www.ijpqa.com International Journal of Pharmaceutical Quality Assurance 218; 9(2); 174-179 doi: 1.25258/ijpqa.v9i2.13642 ISSN 975 956 Research Article Cytological and Histological Study
Abnormal Hair Development and Apparent Follicular Transformation to Mammary Gland in the Absence of Hedgehog Signaling
 Article Abnormal Hair Development and Apparent Follicular Transformation to Mammary Gland in the Absence of Hedgehog Signaling Amel Gritli-Linde, 1, * Kristina Hallberg, 1 Brian D. Harfe, 2 Azadeh Reyahi,
Article Abnormal Hair Development and Apparent Follicular Transformation to Mammary Gland in the Absence of Hedgehog Signaling Amel Gritli-Linde, 1, * Kristina Hallberg, 1 Brian D. Harfe, 2 Azadeh Reyahi,
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2697 Figure S1 Cytokeratin 5 is a specific marker for basal and intermediate cells in all mouse prostate lobes. (a) Immunofluorescence staining showing co-localization of YFP with p63 in
DOI: 10.1038/ncb2697 Figure S1 Cytokeratin 5 is a specific marker for basal and intermediate cells in all mouse prostate lobes. (a) Immunofluorescence staining showing co-localization of YFP with p63 in
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Molecular Mechanisms Regulating Hair Follicle Development
 Molecular Mechanisms Regulating Hair Follicle Development Sarah E. Millar Departments of Dermatology and Cell and Developmental Biology, University of Pennsylvania, Philadelphia, Pennsylvania, U.S.A. Clinical
Molecular Mechanisms Regulating Hair Follicle Development Sarah E. Millar Departments of Dermatology and Cell and Developmental Biology, University of Pennsylvania, Philadelphia, Pennsylvania, U.S.A. Clinical
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Skin stem cells: rising to the surface
 JCB: MINI-REVIEW Skin stem cells: rising to the surface Elaine Fuchs Howard Hughes Medical Institute, Laboratory of Mammalian Cell Biology and Development, The Rockefeller University, New York, NY 10065
JCB: MINI-REVIEW Skin stem cells: rising to the surface Elaine Fuchs Howard Hughes Medical Institute, Laboratory of Mammalian Cell Biology and Development, The Rockefeller University, New York, NY 10065
Tetrapod Limb Development
 Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:!
 LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
Role of Sonic hedgehog signaling in epithelial and mesenchymal development of hair follicles in an organ culture of embryonic mouse skin
 Develop. Growth Differ. (2003) 45, 231 239 Role of Sonic hedgehog signaling in epithelial and mesenchymal development of hair follicles in an organ culture of embryonic mouse skin Daisuke Nanba,* Yasuo
Develop. Growth Differ. (2003) 45, 231 239 Role of Sonic hedgehog signaling in epithelial and mesenchymal development of hair follicles in an organ culture of embryonic mouse skin Daisuke Nanba,* Yasuo
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
INTEGUMENTARY 1-Epidermis, 2-Dermis, Structure of thick and thin skin I- Epidermis . Stratum basale
 INTEGUMENTARY The skin (integument, cutis ) and its derivatives constitute the integumentary system. It form the external covering of the body and is the largest organ of the body. The skin consists of
INTEGUMENTARY The skin (integument, cutis ) and its derivatives constitute the integumentary system. It form the external covering of the body and is the largest organ of the body. The skin consists of
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Cell Birth and Death. Chapter Three
 Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Disruption of Smad4 in Mouse Epidermis Leads to. Depletion of Follicle Stem Cells
 Disruption of Smad4 in Mouse Epidermis Leads to Depletion of Follicle Stem Cells Leilei Yang, Lijuan Wang and Xiao Yang State Key Laboratory of Proteomics, Genetic Laboratory of Development and Diseases,
Disruption of Smad4 in Mouse Epidermis Leads to Depletion of Follicle Stem Cells Leilei Yang, Lijuan Wang and Xiao Yang State Key Laboratory of Proteomics, Genetic Laboratory of Development and Diseases,
Neural stem cells and the neurobiology of ageing. Chen Siyun 1, Dawe G.S. 2
 ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
Developmental Biology
 Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
 Wright et al. BMC Developmental Biology (2015) 15:33 DOI 10.1186/s12861-015-0083-8 RESEARCH ARTICLE Open Access Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
Wright et al. BMC Developmental Biology (2015) 15:33 DOI 10.1186/s12861-015-0083-8 RESEARCH ARTICLE Open Access Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Development Supplementary information
 Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
FGF-regulated BMP signaling is required for eyelid closure and to specify conjunctival epithelial cell fate
 RESEARCH ARTICLE 1741 Development 136, 1741-1750 (2009) doi:10.1242/dev.034082 FGF-regulated BMP signaling is required for eyelid closure and to specify conjunctival epithelial cell fate Jie Huang 1, Lisa
RESEARCH ARTICLE 1741 Development 136, 1741-1750 (2009) doi:10.1242/dev.034082 FGF-regulated BMP signaling is required for eyelid closure and to specify conjunctival epithelial cell fate Jie Huang 1, Lisa
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS
 J. Cell Sci. II, 205-219 (1972) 205 Printed in Great Britain AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS D.F.G.ORWIN AND R.W.THOMSON Wool Research Organisation of N.Z.
J. Cell Sci. II, 205-219 (1972) 205 Printed in Great Britain AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS D.F.G.ORWIN AND R.W.THOMSON Wool Research Organisation of N.Z.
The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells
 The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells Ralf Kist 1,2, Michelle Watson 1, Moira Crosier 1, Max Robinson 2, Jennifer
The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells Ralf Kist 1,2, Michelle Watson 1, Moira Crosier 1, Max Robinson 2, Jennifer
The Role of E2Fs in Mouse Mammary Gland Development. A Senior Honors Thesis. April Sandy. The Ohio State University June 2006
 The Role of E2Fs in Mouse Mammary Gland Development A Senior Honors Thesis Presented in Partial Fulfillment of the Requirements for graduation with distinction in Microbiology in the College of Biological
The Role of E2Fs in Mouse Mammary Gland Development A Senior Honors Thesis Presented in Partial Fulfillment of the Requirements for graduation with distinction in Microbiology in the College of Biological
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Integumentary System. Integumentary System
 1. General aspects a. The integumentary system consists of several organs major organ of the system is the skin other organs are relatively small and they can be considered as specialized structures of
1. General aspects a. The integumentary system consists of several organs major organ of the system is the skin other organs are relatively small and they can be considered as specialized structures of
TISSUE-SPECIFIC STEM CELLS
 TISSUE-SPECIFIC STEM CELLS Cell Lineage Mapping of Taste Bud Cells and Keratinocytes in the Mouse Tongue and Soft Palate TADASHI OKUBO, a,b CHERYL CLARK, a BRIGID L.M. HOGAN a a Department of Cell Biology,
TISSUE-SPECIFIC STEM CELLS Cell Lineage Mapping of Taste Bud Cells and Keratinocytes in the Mouse Tongue and Soft Palate TADASHI OKUBO, a,b CHERYL CLARK, a BRIGID L.M. HOGAN a a Department of Cell Biology,
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice
 Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
BMP signaling in the development of the mouse esophagus and forestomach
 RESEARCH REPORT 4171 Development 137, 4171-4176 (2010) doi:10.1242/dev.056077 2010. Published by The Company of Biologists Ltd BMP signaling in the development of the mouse esophagus and forestomach Pavel
RESEARCH REPORT 4171 Development 137, 4171-4176 (2010) doi:10.1242/dev.056077 2010. Published by The Company of Biologists Ltd BMP signaling in the development of the mouse esophagus and forestomach Pavel
Multiple Roles of Notch Signaling in the Regulation of Epidermal Development
 Article Multiple Roles of Notch Signaling in the Regulation of Epidermal Development Mariko Moriyama, 1 André-Dante Durham, 2 Hiroyuki Moriyama, 1 Kiyotaka Hasegawa, 1 Shin-Ichi Nishikawa, 3 Freddy Radtke,
Article Multiple Roles of Notch Signaling in the Regulation of Epidermal Development Mariko Moriyama, 1 André-Dante Durham, 2 Hiroyuki Moriyama, 1 Kiyotaka Hasegawa, 1 Shin-Ichi Nishikawa, 3 Freddy Radtke,
Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes
 SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
(From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) Materials and Methods
 HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN XI. THF. DISTRIBUTION OF /~-GLucURONIDASE* BY WILLIAM MONTAGNA, PH.D. (From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) PLATES
HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN XI. THF. DISTRIBUTION OF /~-GLucURONIDASE* BY WILLIAM MONTAGNA, PH.D. (From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) PLATES
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
Interactions between cancer stem cells and their niche govern metastatic colonization
 Correction Interactions between cancer stem cells and their niche govern metastatic colonization Ilaria Malanchi, Albert Santamaria-Martínez, Evelyn Susanto, Hong Peng, Hans-Anton Lehr, Jean-Francois Delaloye
Correction Interactions between cancer stem cells and their niche govern metastatic colonization Ilaria Malanchi, Albert Santamaria-Martínez, Evelyn Susanto, Hong Peng, Hans-Anton Lehr, Jean-Francois Delaloye
Effect of Propecia on the Hair Follicle in Male Androgenetic Alopecia: A Confocal Laser Scanning Microscopy and Video Imaging Study
 Effect of Propecia on the Hair Follicle in Male Androgenetic Alopecia: A Confocal Laser Scanning Microscopy and Video Imaging Study Investigators: Department of Dermatology, University of Minnesota, Minneapolis,
Effect of Propecia on the Hair Follicle in Male Androgenetic Alopecia: A Confocal Laser Scanning Microscopy and Video Imaging Study Investigators: Department of Dermatology, University of Minnesota, Minneapolis,
Figure S1. (A) Schematic diagram of dnrar transgene allele. (B) X-Gal staining of testis from
 Figure S1. (A) Schematic diagram of dnrar transgene allele. (B) X-Gal staining of testis from germ cell mutants (dnrar flox/flox, Stra8-Cre +, RARElacZ) (A ), controls (dnrar flox/flox, RARElacZ) (B ),
Figure S1. (A) Schematic diagram of dnrar transgene allele. (B) X-Gal staining of testis from germ cell mutants (dnrar flox/flox, Stra8-Cre +, RARElacZ) (A ), controls (dnrar flox/flox, RARElacZ) (B ),
Supplemental Information. Myocardial Polyploidization Creates a Barrier. to Heart Regeneration in Zebrafish
 Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Polarity and Segmentation. Chapter Two
 Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Ahtiainen et al., http :// /cgi /content /full /jcb /DC1
 Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2535 Figure S1 SOX10 is expressed in human giant congenital nevi and its expression in human melanoma samples suggests that SOX10 functions in a MITF-independent manner. a, b, Representative
DOI: 10.1038/ncb2535 Figure S1 SOX10 is expressed in human giant congenital nevi and its expression in human melanoma samples suggests that SOX10 functions in a MITF-independent manner. a, b, Representative
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Fgf20 governs formation of primary and secondary dermal condensations in developing hair follicles and
 Fgf20 governs formation of primary and secondary dermal condensations in developing hair follicles Sung-Ho Huh, 1,3 Katja Närhi, 2,3 Päivi H. Lindfors, 2 Otso Häärä, 2 Lu Yang, 1 David M. Ornitz, 1,4 and
Fgf20 governs formation of primary and secondary dermal condensations in developing hair follicles Sung-Ho Huh, 1,3 Katja Närhi, 2,3 Päivi H. Lindfors, 2 Otso Häärä, 2 Lu Yang, 1 David M. Ornitz, 1,4 and
CNS Developmental. Anke van Eekelen, PhD. Telethon Institute for Child Health Research
 CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION. Androgen deprivation therapy is the most used treatment of de novo or recurrent
 CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION Stathmin in Prostate Cancer Development and Progression Androgen deprivation therapy is the most used treatment of de novo or recurrent metastatic PCa.
CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION Stathmin in Prostate Cancer Development and Progression Androgen deprivation therapy is the most used treatment of de novo or recurrent metastatic PCa.
Hair Follicle Stem Cells: Molecular Basis of Development and Regeneration
 Farivar and RojhanNejad, 2015. Journal of Genes and Cells, 1(3): p, 57-66 www.imaqpress.com Hair Follicle Stem Cells: Molecular Basis of Development and Regeneration Shirin Farivar 1, 2*, Mahboobeh RojhanNejad
Farivar and RojhanNejad, 2015. Journal of Genes and Cells, 1(3): p, 57-66 www.imaqpress.com Hair Follicle Stem Cells: Molecular Basis of Development and Regeneration Shirin Farivar 1, 2*, Mahboobeh RojhanNejad
Lecture IV. Mechanisms of Neural. Neural Development
 Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
The Wnt/βcatenin signaling pathway
 Wnt signaling is crucial for functioning of the endometrium The Role of Wnt Signaling in Uterus Development (I) and in Homeostasis (II) and Malignancy (III) of the Uterine Endometrium Leen J Blok Erasmus
Wnt signaling is crucial for functioning of the endometrium The Role of Wnt Signaling in Uterus Development (I) and in Homeostasis (II) and Malignancy (III) of the Uterine Endometrium Leen J Blok Erasmus
Epidermal Stem Cells of the Skin
 I ANRV288-CB22-14 ARI 22 June 2006 19:51 R E V I E W S First published online as a Review in Advance on July 11, 2006 E C N A D V A N Annu. Rev. Cell Dev. Biol. 2006. 22:339 73 The Annual Review of Cell
I ANRV288-CB22-14 ARI 22 June 2006 19:51 R E V I E W S First published online as a Review in Advance on July 11, 2006 E C N A D V A N Annu. Rev. Cell Dev. Biol. 2006. 22:339 73 The Annual Review of Cell
Modeling lymphangiogenesis in a three-dimensional culture system
 Modeling lymphangiogenesis in a three-dimensional culture system Françoise Bruyère, Laurence Melen-Lamalle, Silvia Blacher, Guy Roland, Marc Thiry, Lieve Moons, Francis Frankenne, Peter Carmeliet, Kari
Modeling lymphangiogenesis in a three-dimensional culture system Françoise Bruyère, Laurence Melen-Lamalle, Silvia Blacher, Guy Roland, Marc Thiry, Lieve Moons, Francis Frankenne, Peter Carmeliet, Kari
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development
 Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development Jae-Young Kim Department of Medical Science The Graduate School, Yonsei University Pattern Formation and Morphogenesis
Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development Jae-Young Kim Department of Medical Science The Graduate School, Yonsei University Pattern Formation and Morphogenesis
Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats
 Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats Summary of Doctoral Thesis Hidenori Yasuda Graduate School of Veterinary
Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats Summary of Doctoral Thesis Hidenori Yasuda Graduate School of Veterinary
Chapter 4 Opener Pearson Education, Inc.
 Chapter 4 Opener Introduction The integumentary system is composed of: Skin Hair Nails Sweat glands Oil glands Mammary glands The skin is the most visible organ of the body Clinicians can tell a lot about
Chapter 4 Opener Introduction The integumentary system is composed of: Skin Hair Nails Sweat glands Oil glands Mammary glands The skin is the most visible organ of the body Clinicians can tell a lot about
Journal Club. 03/04/2012 Lama Nazzal
 Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION 60611, USA
 Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) (b) (c) (d) (e) (f) (g) .
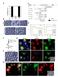 Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) The inhibitory effects of new antibodies (Mab17 and Mab18). They were investigated in in vitro fertilization
Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) The inhibitory effects of new antibodies (Mab17 and Mab18). They were investigated in in vitro fertilization
Skin (Integumentary System) Wheater, Chap. 9
 Skin (Integumentary System) Wheater, Chap. 9 Skin (Integument) Consists of skin and associated derivatives Largest organ of body (21 ft 2 ; 9 lbs.; has 11 miles of blood vessels) Functions: Protection
Skin (Integumentary System) Wheater, Chap. 9 Skin (Integument) Consists of skin and associated derivatives Largest organ of body (21 ft 2 ; 9 lbs.; has 11 miles of blood vessels) Functions: Protection
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
CHAPTER 6 SUMMARIZING DISCUSSION
 CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
Hepatogenesis I Liver development
 Hepatogenesis I Liver development HB 308 George Yeoh Room 2.59 MCS Building yeoh@cyllene.uwa.edu.au Topics Early liver development Tissue interaction - role of morphogens and cytokines Liver enriched transcription
Hepatogenesis I Liver development HB 308 George Yeoh Room 2.59 MCS Building yeoh@cyllene.uwa.edu.au Topics Early liver development Tissue interaction - role of morphogens and cytokines Liver enriched transcription
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
C. elegans Embryonic Development
 Autonomous Specification in Tunicate development Autonomous & Conditional Specification in C. elegans Embryonic Development Figure 8.36 Bilateral Symmetry in the Egg of the Tunicate Styela partita Fig.
Autonomous Specification in Tunicate development Autonomous & Conditional Specification in C. elegans Embryonic Development Figure 8.36 Bilateral Symmetry in the Egg of the Tunicate Styela partita Fig.
Further studies on the melanophores of periodic albino mutant of Xenopus laevis
 J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions
 CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
Journal of Biomedical Science 2011, 18:92
 Journal of Biomedical Science This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Inhibition of
Journal of Biomedical Science This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Inhibition of
Branching morphogenesis of the lung: new molecular insights into an old problem
 86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
Material and Methods Production and analysis of -gal+ clones Immunostaining Optical projection tomography Statistical analysis
 Material and Methods Production and analysis of β-gal+ clones The α-cardiac actin nlaacz/+ and Cx40 egfp mouse lines used in this study were genotyped as previously reported 1, 2. Double transgenic knock-in
Material and Methods Production and analysis of β-gal+ clones The α-cardiac actin nlaacz/+ and Cx40 egfp mouse lines used in this study were genotyped as previously reported 1, 2. Double transgenic knock-in
Distinct Roles Of CCN1 And CCN2 In Limb Development
 Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
Distinct Roles Of CCN1 And CCN2 In Limb Development Jie Jiang, PhD 1, Jessica Ong, BS 1, Faith Hall-Glenn, PhD 2, Teni Anbarchian, BS 1, Karen M. Lyons, PhD 1. 1 University of California, Los Angeles,
