TISSUE-SPECIFIC STEM CELLS
|
|
|
- Hilary Terry
- 5 years ago
- Views:
Transcription
1 TISSUE-SPECIFIC STEM CELLS Cell Lineage Mapping of Taste Bud Cells and Keratinocytes in the Mouse Tongue and Soft Palate TADASHI OKUBO, a,b CHERYL CLARK, a BRIGID L.M. HOGAN a a Department of Cell Biology, Duke University Medical Center, Durham, North Carolina, USA; b Center for Integrative Bioscience, National Institutes of Natural Sciences, Okazaki, Japan Key Words. Taste bud Keratinocytes Tongue Progenitor cell Lineage mapping ABSTRACT The epithelium of the mouse tongue and soft palate consists of at least three distinct epithelial cell populations: basal cells, keratinized cells organized into filiform and fungiform papillae, and taste receptor cells present in tight clusters known as taste buds in the fungiform and circumvallate papillae and soft palate. All three cell types develop from the simple epithelium of the embryonic tongue and palate, and are continually replaced in the adult by cell turnover. Previous studies using pulse-chase tritiated thymidine labeling in the adult mouse provided evidence for a high rate of cell turnover in the keratinocytes (5 7 days) and taste buds (10 days). However, little is known about the localization and phenotype of the long-term stem or progenitor cells that give Disclosure of potential conflicts of interest is found at the end of this article. rise to the mature taste bud cells and surrounding keratinocytes in these gustatory tissues. Here, we make use of a tamoxifen-inducible K14-CreER transgene and the ROSA26 LacZ reporter allele to lineage trace the mature keratinocytes and taste bud cells of the early postnatal and adult mouse tongue and soft palate. Our results support the hypothesis that both the pore keratinocytes and receptor cells of the taste bud are derived from a common K14 K5 Trp63 Sox2 population of bipotential progenitor cells located outside the taste bud. The results are also compatible with models in which the keratinocytes of the filiform and fungiform papillae are derived from basal progenitor cells localized at the base of these structures. STEM CELLS 2009;27: INTRODUCTION The tongue and soft palate are gustatory tissues containing numerous taste buds (TBs) to detect taste stimuli. A typical mature TB consists of a tight intragemmal cluster of elongated taste receptor/sensory epithelial cells surrounded by an outer layer of flattened keratinocytes. These form the perigemmal layer and taste pore. TBs are located in several places in the tongue (fungiform, foliate, and circumvallate papillae) and oral cavity (soft palate). Depending on their location, TBs develop either perinatally in the anterior tongue and soft palate or postnatally in the foliate and circumvallate (CV) papillae. In all cases, they arise during development from the local surface epithelium rather than from neuronal or neural crest cells [1, 2]. Indeed, studies in rodents have shown that both the receptor cells of the TBs and the stratified keratinized epithelium of the fungiform papillae in which they reside, as well as the numerous filiform papillae of the tongue, are all derived from the same simple epithelium present in the embryo [3, 4]. Recent studies have begun to identify genetic pathways controlling the differentiation of embryonic tongue epithelium into either TB precursors or stratified, keratinized epithelium. For example, we have found that the Sry-related transcription factor Sox2 is more highly expressed in developing and mature TBs than in the surrounding epithelial cells [5]. Newborn pups hypomorphic for Sox2 lack mature TBs and fungiform papillae. In contrast, when Sox2 is overexpressed in the embryonic tongue epithelium, keratinocyte (filiform) differentiation is completely blocked, with the absence of trichohyalin (AE15 antigen) expression [5]. These findings suggest that the level of Sox2 in the immature tongue epithelial cells is critical for the cell fate decision between TB cells and keratinocytes. Furthermore, Sox2 expression in regenerating CV TBs is dependent on gustatory innervation [6]. Thus, Sox2 also likely plays an important role in the maintenance of adult TB cells. Pax9 is also known to be an important transcription factor for the proper differentiation of filiform papillae [7]. Trp63 (p63), a member of the p53 transcription factor family, is critical for tongue development because Trp63-deficient embryos have only a thin tongue epithelial layer [8]. Thus, gene-targeting studies suggest that complex transcriptional hierarchies and signaling networks regulate the differentiation and maintenance of tongue epithelial cells. In the adult tongue, both filiform papillae and TBs undergo continual turnover. In the case of the keratinocytes of the filiform papillae, pulse-chase tritiated thymidine studies support a model in which long-term, self-renewing stem cells reside in the base of the papillae. The transit amplifying and differentiating descendants move up along the basal lamina and exit into the superficial layers to be shed from the surface over the course of Author contributions: T.O.: conception and design, data collection and analysis, manuscript writing; C.C.: data collection and analysis, B.L.M.H.: conception and design, manuscript writing. Correspondence: Tadashi Okubo, Ph.D., Center for Integrative Bioscience, National Institutes of Natural Sciences, 5 1 Myodaiji-Higashiyama, Okazaki, Aichi Japan. Telephone: ; Fax: ; t.okubo@nibb.ac.jp Received June 28, 2008; accepted for publication November 18, 2008; first published online in STEM CELLS EXPRESS November 26, AlphaMed Press /2009/$30.00/0 doi: /stemcells STEM CELLS 2009;27:
2 Okubo, Clark, Hogan 443 about 5 7 days [9]. In the case of the TBs of the fungiform papillae, previous studies have suggested a turnover time of about 10 days in the adult [10, 11]. However, the localization of the long-term progenitor cells of the TBs and whether they reside exclusively inside or outside the bud are still under debate. For example, studies in the rat suggest that CV TB cells are rapidly renewed from cells outside the bud [12, 13]. In order to learn more about the identify of progenitor cells of the TB and the niche in which they reside, we characterized the expression patterns of the epithelial markers keratin 8 (K8), K14, K5, and Trp63 in mouse gustatory tissues. Based on these observations, we used the tamoxifen-inducible K14-CreER/Rosa26R reporter system [14, 15] to carry out cell lineage mapping studies in the tongue and in the soft palate at early postnatal and adult stages. Our results support a model in which K14 epithelial cells located outside the TBs function as bipotential progenitor cells to continuously generate both mature TB receptor cells and surrounding keratinocytes (perigemmal and taste pore cells). Our findings are also compatible with models in which the keratinocytes of the filiform and fungiform papillae are derived from basal progenitor cells localized to the bottom of these structures. MATERIALS AND METHODS Mice All mice were maintained under a 12-hour light/dark cycle and handled under Institutional Animal Care and Use Committee-approved protocols. Outbred ICR mice were used as controls at the embryonic and postnatal stages. The K14-CreER transgenic mouse line was kindly provided by Barry Stripp [15] and was maintained as hemizygotes. ROSA26 mice (Gt(ROSA)26Sor tm1sor ), in which LacZ is a reporter for Cre recombination, were maintained as homozygous [16]. For genotyping, genomic DNA was extracted from tail tips and assayed using polymerase chain reaction (PCR) primer sets for the Rosa26R allele (5 -AAAGTCGCTCTGAGTT- GTTAT-3, 5 -GCGAAGAGTTTGTCCTCAACC-3, 5 -GGAGC- GGGAGAAATGGATATG-3 ) and for the K14-CreER allele (5 -TCGATGCAACGAGTGATGAG-3, 5 -TTCGGCTATA- CGTAACAGGG-3 ). Tamoxifen Injection K14-CreER males were crossed with Rosa26R females. Tamoxifen (TM) dissolved in corn oil (20 mg/ml) was given by i.p. injection at a dose of 0.25 mg/g body weight (one injection at postnatal day 2 (P2) for the mother, and five injections at 1-day intervals for P30 adult mice). LacZ Staining For each time point, 4 10 independent tongues and soft palates were fixed in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS) for 2 hours after dissection, and then washed in PBS. The tissues were next stained in X-gal solution (5 mm potassium ferrocyanide, 5 mm potassium ferricyanide, 2 mm MgCl, 0.02% NP-40, 1 mg/ml X-gal) at 37 C overnight. Tissues were postfixed in 4% PFA, processed for sectioning at 7 m, and counterstained with eosin. Histology and Immunohistochemistry Tongues and soft palates were fixed in 4% PFA in PBS for 2 hours or overnight at 4 C, processed through the appropriate dehydration steps, and embedded in paraffin. The 7- m thick sections were pretreated by boiling in 10 mm sodium citrate buffer (ph 6.0). For the primary antibody reaction, the sections were incubated with the following antibodies: Trp63 (4A4; Santa Cruz Biotechnology Inc., Santa Cruz, CA, Sox2 (AB5603; Chemicon, Temecula, CA, com), K14 (LL002; Chemicon), K8 (TROMA I; Developmental Studies Hybridoma Bank, Iowa City, IA, dshbwww), K5 (AF138; Covance, Princeton, NJ, covance.com), trichohyalin (AE15, ab58755; Abcam, Cambridge, U.K., and phosphohistone H3 (PHHS) (Ser10) (Upstate, Charlottesville, VA, upstate.com). Signal detection was performed with the following secondary antibodies: AlexaFluor 488, AlexaFluor 546-labeled anti-mouse, anti-rat, and anti-rabbit IgG antibodies (Molecular Probes Inc., Eugene, OR, and the DAKO EnVision system-hrp (DakoCytomation, Glostrup, Denmark, RESULTS Expression of Epithelial Markers in the Development of the Anterior Tongue In the mouse, the TBs and keratinized epithelial structures of the tongue and soft palate develop from the embryonic epithelium with slightly different timing, depending on their location. To follow this development in the anterior tongue we performed immunohistochemistry with several markers at different stages (Fig. 1). K8, a marker of mature intragemmal cells [17, 18], is first expressed in the TBs of the fungiform papillae perinatally (P1), and its level gradually increases (Fig. 1A, 1D, 1G). In contrast, K14 expression is absent from intragemmal cells at P1 but clearly expressed in perigemmal cells and basal keratinocytes (Fig. 1B, 1E, 1H). Likewise, the transcription factor Trp63 is initially expressed throughout the basal layer of the epithelium, including the developing fungiform papillae, but is completely absent from the intragemmal cells at P1 and later (Fig. 1C, 1F, 1I, 1J, 1L). K5, which is coexpressed with K14 and Trp63 in basal cells of other epithelia [19], has a similar expression pattern to Trp63 in the adult tongue (Fig. 1J). The mutually exclusive expression of K8 inside the TB, versus Trp63, K14, and K5 in the perigemmal keratinocytes outside the TB and in basal keratinocytes, is clearly shown by double immunohistochemistry in the adult tongue (Fig. 1G 1J). For example, there is a very sharp boundary between K8 and K14 cells inside and outside the TB (Fig. 1H) as well as between K8 and Trp63 cells (Fig. 1I). The transcription factor Sox2 is clearly expressed at high levels in the mature TB cells, but at lower levels in the basal epithelium of the fungiform and filiform papillae (Fig. 1K). However, unlike K8, it is also present in the nuclei of perigemmal cells, where its expression overlaps with that of Trp63, as shown by double labeling (Fig. 1L). Finally, immunohistochemistry shows that trichohyalin (AE15 antigen), a differentiation marker for keratinocytes of the filiform papillae, is not expressed in fungiform papillae or TB cells (Fig. 1G). To localize mitotic cells in the adult tongue, we used antibodies to PHH3. This shows that, in the anterior tongue, PHH3 cells are present in the perigemmal layer as well as in the Trp63 epithelial basal layer of the filiform papillae (Fig. 1M, 1O). Expression of Epithelial Markers in the Development of the CV Papilla and Soft Palate We next examined the expression of epithelial markers in the CV papilla and soft palate, in which the TBs develop perinatally. At P1, K14 Trp63 CV epithelium is already invaginated into the posterior tongue (Fig. 2A, 2B), but mature TBs have not yet appeared. In contrast, in the perinatal soft palate, distinct K8 TB cells are already differentiated (Fig. 2G, inset) and the expression of K14 and Trp63 is not observed in TBs (Fig. 2G, 2H). At P21, immunohistochemistry shows that many K8 intragemmal cells are present in the TBs at both locations. These cells are completely negative for K14, K5, and Trp63, which are
3 444 Taste Bud Cell and Keratinocyte Cell Lineage Mapping Figure 1. Expression of epithelial markers in the anterior tongue at different stages. Sections of the anterior tongue show expression of keratin 8 (K8) (A, D), K14 (B, E), and Trp63 (C, F) at E16.5 and P1. (G L): Fluorescence immunostaining for epithelial and proliferation markers in fungiform papillae at P21. Shown are expression of K8/AE15 (G), K8/K14 (H), K8/Trp63 (I), Trp63/K5 (J), Sox2 (K), Trp63/Sox2 (L), and phosphohistone H3 (PHH3) (M) merged with differential interference contrast images. Note in particular the sharp boundary between K8 inside and K14 and Trp63 outside the taste bud (TB), and the coexpression of Trp63 and Sox2 in the perigemmal cells around the TB (L). (N, O): Sections of filiform papillae showing expression of Trp63 (N) and PHH3 (O). Arrows indicate TBs. Scale bar, 50 m. Abbreviations: E, embryonic day; P, postnatal day. coexpressed in the epithelium outside the TBs (Fig. 2C, 2D, 2J). As in the tongue, Sox2 is expressed in the TBs of the CV and soft palate in intragemmal cells and in perigemmal cells that are positive for Trp63 (Fig. 2E, 2K). Finally, PHH3 mitotic cells are frequently detected in the basal layer of the CV and soft palate epithelium and in perigemmal cells adjacent to TBs (Fig. 2F, 2L). Taken together, these results in all gustatory tissues are compatible with a model in which TB intragemmal cells are derived from a K14 K5 Trp63 cell population outside the bud. Cell Lineage Mapping in the Postnatal Tongue and Soft Palate To determine the long-term progenitor cells for the maintenance of mature TB cells and keratinocytes in the postnatal mouse, we performed cell lineage mapping experiments to follow the descendants of cells that originally express the K14-CreER transgene. This experiment was carried out at two different times, namely, at P2 and at P30 (Fig. 3A). Since it is difficult to inject TM into pups soon after birth, a single dose was administered to
4 Okubo, Clark, Hogan 445 K14-CreER/Rosa26R compound transgenic pups, but not in littermates lacking the CreER transgene (Fig. 3B 3E). Histological sections of the tongues after 1 week showed that many fungiform TB cells were positive for LacZ activity, even though, as shown in Figure 1, these cells do not themselves express K14 (Fig. 4A, 4B). LacZ staining was also seen in keratinocytes and pore cells immediately surrounding the TBs. The fact that some TBs were completely negative (Fig. 4C) is evidence that the LacZ staining, when present, does not represent endogenous ß-galactosidase activity. Similar patterns of labeling of TB cells and surrounding keratinocytes were seen in the CV and soft palate (Fig. 4D, 4E). In the tongue, LacZ cells were also present in both basal cells and stratified keratinocytes of many filiform papillae (Fig. 4F). When the chase period was extended to 1 month, patches of LacZ cells were still seen in the tongue and soft palate. Significantly, TB cells were still strongly stained, as well as perigemmal cells and adjacent keratinized epithelial cells (pore cells) (Fig. 4G, 4H). Another typical pattern of LacZ staining is illustrated in Figure 4I, which shows labeling of cells of the fungiform papilla and taste pore cells. TBs in the CV (Fig. 4J) and soft palate (Fig. 4K) are also strongly stained. As with the shorter chase period, labeled cells were present in both basal cells and stratified keratinocytes of the fungiform and filiform papillae (Fig. 4I, 4L). Expression of K14 and LacZ overlapped in some cells adjacent to the TB cells and in the basal layer of the filiform papillae (Fig. 4M, 4N). Figure 2. Expression of epithelial markers in the circumvallate papilla and soft palate at different stages. (A F): Sections of circumvallate papilla at P1 (A, B) and P21 (C F). Shown are the expression of keratin 14 (K14) (A) and Trp63 (B) at P1 and the expression of K8/K14 (C), Trp63/K5 (D), Trp63/Sox2 (E), and phosphohistone H3 (PHH3) (F) merged with differential interference contrast (DIC) images. Note the exclusion of K14, Trp63, and K5 from the interior of the taste bud (TB), which contains K8 cells with a sharp boundary in perigemmal cells (C, D). Note also the coexpression of Sox2 and Trp63 in cells around the TBs (E). (G L): Sections of soft palate at P1 (G, H) and P21 (I L). Shown are expression of K14 (G), K8(G, inset), and Trp63 (H) at P1 and expression of K8 (I), K8/K14 (J), Trp63/Sox2 (K), and PHH3 (L) merged with DIC images. Note again the sharp boundary between K8 and K14 cells and the coexpression of Trp63 and Sox2 in perigemmal cells adjacent to the TBs (J, K). Arrows indicate TBs. Scale bar, 50 m. Abbreviation: P, postnatal day. the lactating mother so that the drug would pass to the pups through the milk. By 2 weeks, whole-mount staining revealed patches of LacZ cells throughout the tongue and soft palate of Cell Lineage Mapping in the Adult Tongue and Soft Palate In a second series of experiments, TM was injected five times into individual mice at P30 (Fig. 3A), and the fate of the descendants of K14 cells in which recombination had taken place was followed after 2 weeks and 1 month from the last TM injection (Fig. 3A). This regimen of injection was adopted because control experiments showed that one or two injections of TM did not produce lineage-labeled cells (data not shown), probably due to the inefficiency of the K14-CreER transgene in tongue and soft palate tissue. Other control experiments showed that, 3 days after the last injection, only single cells or small groups of cells were labeled in the basal cells or perigemmal regions of all gustatory tissues such as the fungiform papillae, CV, and soft palate (supporting information Fig. 1A 1D). Significantly, labeling was never seen inside the TBs. After a 2-week and 1-month chase, whole-mount staining revealed patches of LacZ cells in both the tongue and soft palate (Fig. 3C 3E). At both times, no LacZ cells were seen in littermates that did not inherit the K14-CreER transgene (Fig. 3B). Histological analysis was carried out after both chase periods, and serial sections were examined for patterns of LacZ staining. As shown in Figure 5 and summarized in Figure 6, the patterns were very similar to those seen in the first series of experiments carried out in the early postnatal period. Significantly, even after 1 month, labeled receptor cells were found inside the TBs in all locations (fungiform and CV in the tongue and in the soft palate) (Fig. 5). The different staining patterns in the fungiform papillae and TBs were classified and quantified as shown in Figure 6A and Table 1. Typical patterns for the CV are shown in supporting information Figure 2. These results allowed several conclusions to be drawn. First, in all cases where TB cells were labeled, some positive cells were also seen in the surrounding pore cells, as would be expected if the TB cells are continually derived from K14 bipotential cells immediately outside the bud. In no case were only the internal TB receptor cells positive and not the immediate outside (perigemmal) cells. Second, not all the internal cells (intragemmal cells) within
5 446 Taste Bud Cell and Keratinocyte Cell Lineage Mapping Figure 3. Experimental design and whole mount LacZ staining. (A): Time course of cell lineage mapping experiments for postnatal and adult stages. TM, tamoxifen (B, D): Typical examples of whole-mount LacZ staining of tongues in CreER /Rosa26R (B) and CreER /Rosa26R (D) tongues chased for 2 weeks from P30. (E, F): Anterior tongue surface (E) and soft palate (F) of CreER /Rosa26R mice chased for 1 month from P30. Abbreviation: P, postnatal day. positive TBs were labeled (Fig. 5C 5E, 5H inset, 5K), suggesting that several progenitor cells contribute descendants to a single TB [2]. Third, in the longer chases, there were few or no patches (1 of 43 examined after 1 month from P2, and 0 of 24 after 1 month from P30) in which both keratinocytes within the fungiform papillae (i.e., nonperigemmal cells) and the TBs were labeled (Fig. 6A, type 5, Table 1). Cell Lineage Mapping in the Postnatal and Adult Filiform Papillae In the case of the filiform papillae, several different LacZ staining patterns were seen (Figs. 4F, 4L, 5F, 5L, 5M). In most cases, the general pattern of labeling was consistent with the progenitor cells residing in the base of each conical papilla and the transit-amplifying cells moving up along the basal lamina of the walls of the filiform papilla and then exiting into the superficial layer. In some cases (Fig. 6B, type 1), the labeled cells only populated the anterior aspect of the papilla, corresponding approximately to the anterior column of cell proposed by Hume and Potten [9]. In other cases, only the posterior aspect of the papilla was labeled (Fig. 6B, type 2). In some cases (Fig. 6B, type 3, 4) the labeled cells populated both the anterior and posterior aspects of the same or adjacent papillae. Finally, examples were seen (Fig. 6B, type 5) in the which the descendants of labeled progenitor cells populated only the keratinocytes of the interpapillary column [9]. The patterns and their relative frequencies are thus generally consistent with a model in which there is little mixing of epithelial cells between the columns or between one papilla and the next. DISCUSSION Long-Term Progenitor Cells of the Fungiform Papillae and Taste Buds In this study, we used an inducible, Cre recombination-based cell lineage labeling system to trace the origin of TB cells in different gustatory tissues of postnatal and adult mice. Our results support a model in which TB cells in all locations (fungiform papillae, CV, and soft palate) and at both stages are continuously derived from K14 perigemmal cells immediately outside the TB. Moreover, our 1-month chase studies suggest that these K14 cells give rise to mature TB cells over the long term ( 1 month). This is because the TB cells would have turned over three times within the chase period, based on the estimated turnover time of 10 days [10]. Our lineage tracing and PHH3 staining results are consistent with recent findings in the rat suggesting that TBs of the CV papilla are descendants of immediately adjacent K14 cells that can undergo proliferation as judged by 5-bromo-2 -deoxyuridine incorporation [13]. However, these studies did not use lineage labeling and did not address which specific cell types generate TB cells over the long term. The fact that, in our studies, some TBs contain both labeled and unlabeled cells also supports previous conclusions from mosaic analyses in the mouse that more than one progenitor cell contributes to the maintenance of TBs [1, 2]. Thus, our results provide the first clear evidence that the K14 cells that gives rise to mature TB cells are bipotential and give rise to descendants that differentiate into either TB receptor cells or surrounding keratinocytes (perigemmal and pore cells). This conclusion is based on the fact that we never saw labeled TB cells in the absence of staining of keratinocytes (pore cells) around the bud (Fig. 6A, Table 1). It is possible that the staining patterns classified as type 2 and type 4 in Figure 6A are the result of the simultaneous labeling of two adjacent progenitor cells with different potentials one that gives rise exclusively to TB cells and one that generates only local keratinocytes/pore cells. However, two observations argue against the existence of two discrete unipotential populations immediately outside the TB. First, after a short chase period, most of the lineage-labeled cells were single cells, with only a few small clusters (supporting information Fig. 1). Second, according to this model, the type 2 and type 4 lineage-labeling patterns in the fungiform papilla should be relatively rare, while in fact they were seen
6 Okubo, Clark, Hogan 447 Figure 4. Cell lineage analysis after a 1-week chase from P2. (A F): LacZ staining for the tongue and soft palate of K14-CreER /Rosa26R mice dissected at 1 week after tamoxifen administration. (A C): Different examples of fungiform papillae. LacZ staining is seen in taste bud (TB) cells and in the surrounding keratinocyte epithelial cells (pore cells). In the case of circumvallate (CV) (D) and soft palate TBs (E), examples are shown in which adjacent epithelial cells and about half of the taste receptor cells are labeled. (F): Filiform papillae. (G L): Cell lineage analysis after a 1-month chase from P2. Shown are typical staining patterns of fungiform papillae (G I). Note that only a subset of TB cells are stained (G). All TB receptor cells and adjacent epithelial cells are strongly stained (H). The surrounding keratinized epithelial cells are also strongly stained in the absence of TB labeling in (I). (J) TBs in CV; (K) TB in soft palate. (L): Typical labeling of filiform papillae. (M, N): K14/LacZ double staining in fungiform (M) and filiform (N) papillae. Arrows show TBs. Arrowheads show K14/LacZ coexpressing cells. Scale bar, 50 m. Abbreviation: P, postnatal day. more frequently than type 1, in which only keratinocytes around the TBs were labeled. In the case of the keratinocytes making up the bulk of the fungiform papillae, the most frequent lineage-labeling pattern (Fig. 6A, type 3) suggests that these cells are derived from K14 Trp63 cells that reside at the base of the papillae. If this model is correct, then descendants of a single progenitor cell in the base move up along the basal lamina and give rise to differentiating keratinocytes in the superficial layers. It is possible that some of the descendants of the cells in the base of the papilla also give rise to the bipotential K14 progenitor cells around the TBs. Indeed, in short-term chase experiments (2 weeks), there are many cases of the lineage-labeling pattern (Fig. 6A, type 5) that would support this model (Figs. 4B, 5A). However, in a longer chase experiment (1 month), this pattern was only seen once, in a total of 67 fungiform papillae examined
7 448 Taste Bud Cell and Keratinocyte Cell Lineage Mapping Figure 5. Cell lineage analysis after a 2-week chase from P30. (A F): The tongues and soft palate were dissected 2 weeks after tamoxifen injection and stained for LacZ. Shown are typical examples of labeling of taste bud (TB) cells and adjacent keratinized epithelial cells (A), adjacent epithelial cells but not TBs (B), and a subset of TB cells and adjacent perigemmal cells (C). (D): LacZ expression is seen in the TBs, basal cells, and adjacent keratinized cells in the circumvallate (CV) papilla. (E): Some TB cells are stained in the soft palate. (F): Strong LacZ staining in the base of each filiform papillae. (G M): Cell lineage analysis after a 1-month chase from P30. Shown are various staining patterns in the fungiform papillae (G I). LacZ expression is seen in some TB cells and in adjacent epithelial cells (H) (inset shows another example). (J): TBs in the CV papilla; (K): TB in the soft palate. (L, M): Different lineage labeling patterns are seen in the filiform papillae. Arrows show TBs. Scale bar, 50 m. Abbreviation: P, postnatal day. (Table 1). A more parsimonious interpretation is, therefore, that this pattern did result from the chance labeling of two progenitor cells one in the base of the papilla and one local to the TB. To resolve this crucial question, it will be necessary to use a more efficient system than the K14-CreER inducible transgene for lineage labeling of single cells in the tongue. Further experiments will also be needed to characterize the mechanisms determining whether daughter cells give rise to receptor cells or keratinocytes. Relevant to this question, our studies on the dynamics of expression of Trp63 and K14 during development suggest that both of these genes are downregulated in the TB cells but remain on in the progenitor cells. This idea is supported by a recent enhancer analysis indicating that K14 is a direct target of Trp63 ( Np63) [19, 20]. In addition, we found that Sox2 expression is maintained not only in the mature TB, but also in perigemmal cells immediately outside it that also coexpress Trp63 (Figs. 1, 2). Our previous studies have shown that Sox2 plays a critical role in the development of the TB cell lineage and that when Sox2 falls below a threshold, differentiation of the TB lineage is inhibited and fungiform papilla formation is lost [5]. We therefore suggest a model in which the putative bipotential progenitor cells of the TBs are Sox2, Trp63, K14, and K5 (Fig. 6C). Daughters of these cell that move into the TB maintain a high level of Sox2 expression and switch on K8, but lose K14 and Trp63 expression as they become committed to the TB cell lineage. In contrast, other daughters that move into the keratinocyte lineage lose both Sox2 and Trp63 expression and upregulate genes associated with being pore cells. Long-Term Progenitor Cells of the Filiform Papillae Filiform papillae are highly organized, curved, cone-shaped epithelial structures covering the dorsal surface of the tongue. They are tilted so that the anterior face overlaps the posterior. The keratinocytes in the anterior and posterior regions express different soft or hard keratins, such as Krt1 5, Krt2 16, Krt1 17, and keratins recognized by the AE13 antibody [7, 21], possibly because the two faces of the papillae experience different stresses and pressures during the process of mastication.
8 Okubo, Clark, Hogan 449 Figure 6. A possible model based on cell lineage analyses. (A, B): Schematic representation of typical LacZ lineage labeling patterns seen in the fungiform (A) and filiform (B) papillae. (A): Type 1 shows staining only in taste pore cells and adjacent epithelial cells. Type 2 shows staining in a subset of taste bud (TB) cells and adjacent pore and epithelial cells. Type 3 shows staining in both the basal cells of the fungiform papilla and the epithelial cells around the TBs. Type 4 involves labeling of TB cells and surrounding pore cells and adjacent epithelial cells. Type 5 is a combination of Type 3 and 4. (B): Different patterns of labeling seen within the filiform papillae. (C): A possible model based on genetic mapping of keratin 14 (K14) cells in the tongue epithelium. Adjacent to the TBs, K14 Trp63 Sox2 K5 cells (yellow) are bipotential progenitor cells that can generate both TB receptor cells (green) and keratinized pore cells (blue). Daughter cells committed to the TB lineage upregulate Sox2 and K8 expression, and downregulate Trp63 and K14. On the other hand, daughter cells going towards the keratinocyte lineage lose both Sox2 and Trp63 expression. In the filiform and fungiform papillae, longterm progenitor cells (K14 Trp63 K5 Sox2 Low ) are located in the basal layer (orange) and move up and differentiate into K14 Trp63 AE15 filiform keratinocytes (blue). Furthermore, there is some evidence that the Wnt signaling pathway is differentially regulated between the anterior and posterior compartments of the filiform papillae, with higher levels of Lef1 and BAT-gal canonical Wnt reporter expression anteriorly [5, 21]. According to the model previously proposed by others [9], stem cells located in the base of the anterior compartment (position 1) give rise to transit-amplifying daughters that move up along the basal lamina and exit into the superficial layers to differentiate into keratinocytes. According to this model, there is little mixing between the compartments making up the anterior, posterior, buttress and interpapillary columns. Our lineage mapping results generally support this model and the idea that the stem cells reside in niches at the base of the papilla. A Possible Model for the Stem Cell System of Gustatory Tissues In conclusion, we propose a model for the location and niche of long-term stem or progenitor cells with the capacity for selfrenewal and multilineage differentiation in the tongue (Fig. 6C).
9 450 Taste Bud Cell and Keratinocyte Cell Lineage Mapping Table 1. Relative frequency of different LacZ staining patterns in the fungiform and filiform papillae Fungiform papillae P2 to 1 month P30 to 1 month Filiform papillae P2 to 1 month P30 to 1 month Type 1 4/43 3/24 26/111 29/89 Type 2 12/43 3/24 21/111 15/89 Type 3 16/43 12/24 16/111 12/89 Type 4 10/43 6/24 19/111 7/89 Type 5 1/43 0/24 29/111 26/89 The relative frequency is the number of each type/total number of LacZ stained fungiform or filiform papillae. Abbreviation: P, postnatal day. Adjacent to the TBs, K14 Trp63 K5 Sox2 cells are bipotential progenitor cells that can generate both mature TB receptor cells and keratinocytes (pore cells). Daughter cells committed to the taste bud lineage keep high levels of Sox2 expression and switch on K8 expression, and downregulate basal markers such as Trp63, K14, and K5. On the other hand, daughter cells going towards the keratinocyte lineage (pore cells) lose both Sox2 and Trp63 expression. In the filiform and fungiform papillae, longterm progenitor cells located in the basal layer are characterized as Trp63 K14 K5 Sox2 Low, and the daughter cells continuously move up during keratinization (Fig. 6C). We also propose that a similar model is involved in the CV papilla and soft palate. The gustatory tissue of the oral cavity thus provides an accessible experimental system to study in comparison with the stem cells of the hair follicles, epidermis, and intestinal crypt [22, 23]. Further investigations are needed to identify the local cues and transcriptional hierarchy that regulate the proliferation and differentiation capacity of the different cell types in the tongue and oral cavity. ACKNOWLEDGMENTS The antibody to K8 was obtained from the Developmental Studies Hybridoma Bank. This work was supported by NIH grant HL to B.L.M.H. DISCLOSURE OF POTENTIAL CONFLICTS OF INTEREST The authors indicate no potential conflicts of interest. REFERENCES 1 Stone LM, Finger TE, Tam PP et al. Taste receptor cells arise from local epithelium, not neurogenic ectoderm. Proc Natl Acad Sci U S A 1995; 92: Stone LM, Tan SS, Tam PP et al. Analysis of cell lineage relationships in taste buds. J Neurosci 2002;22: Farbman AI. Electron microscope study of the developing taste bud in rat fungiform papilla. Dev Biol 1965;11: Baratz RS, Farbman AI. Morphogenesis of rat lingual filiform papillae. Am J Anat 1975;143: Okubo T, Pevny LH, Hogan BL. Sox2 is required for development of taste bud sensory cells. Genes Dev 2006;20: Suzuki Y. Expression of Sox2 in mouse taste buds and its relation to innervation. Cell Tissue Res 2008;332: Jonker L, Kist R, Aw A et al. Pax9 is required for filiform papilla development and suppresses skin-specific differentiation of the mammalian tongue epithelium. Mech Dev 2004;121: Mills AA, Zheng B, Wang XJ et al. p63 is a p53 homologue required for limb and epidermal morphogenesis. Nature 1999;398: Hume WJ, Potten CS. The ordered columnar structure of mouse filiform papillae. J Cell Sci 1976;22: Beidler LM, Smallman RL. Renewal of cells within taste buds. J Cell Biol 1965;27: Delay RJ, Kinnamon JC, Roper SD. Ultrastructure of mouse vallate taste buds: II. Cell types and cell lineage. J Comp Neurol 1986;253: Hamamichi R, Asano-Miyoshi M, Emori Y. Taste bud contains both short-lived and long-lived cell populations. Neuroscience 2006;141: Asano-Miyoshi M, Hamamichi R, Emori Y. Cytokeratin 14 is expressed in immature cells in rat taste buds. J Mol Histol 2008;39: Vasioukhin V, Degenstein L, Wise B et al. The magical touch: Genome targeting in epidermal stem cells induced by tamoxifen application to mouse skin. Proc Natl Acad Sci U S A 1999;96: Hong KU, Reynolds SD, Watkins S et al. Basal cells are a multipotent progenitor capable of renewing the bronchial epithelium. Am J Pathol 2004;164: Soriano P. Generalized LacZ expression with the ROSA26 Cre reporter strain. Nat Genet 1999;21: Knapp L, Lawton A, Oakley B et al. Keratins as markers of differentiated taste cells of the rat. Differentiation 1995;58: Liebl DJ, Mbiene JP, Parada LF. NT4/5 mutant mice have deficiency in gustatory papillae and taste bud formation. Dev Biol 1999;213: Candi E, Rufini A, Terrinoni A et al. Differential roles of p63 isoforms in epidermal development: Selective genetic complementation in p63 null mice. Cell Death Differ 2006;13: Romano RA, Birkaya B, Sinha S. A functional enhancer of keratin14 is a direct transcriptional target of deltanp63. J Invest Dermatol 2007;127: Rhee H, Polak L, Fuchs E. Lhx2 maintains stem cell character in hair follicles. Science 2006;312: Fuchs E. Skin stem cells: Rising to the surface. J Cell Biol 2008;180: Barker N, van Es JH, Kuipers J et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 2007;449: See for supporting information available online.
The Beauty of the Skin
 The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
Dorsum of the tongue. Oral Part exhibit lingual papillae of the 4 types. Oral Part of Tongue divided into Left & right halves by shallow median groove
 Histology of TONGUE Figure 22.13 Dorsum of the tongue Oral Part of Tongue divided into Left & right halves by shallow median groove Oral Part exhibit lingual papillae of the 4 types a. filiform papillae,
Histology of TONGUE Figure 22.13 Dorsum of the tongue Oral Part of Tongue divided into Left & right halves by shallow median groove Oral Part exhibit lingual papillae of the 4 types a. filiform papillae,
Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a,
 Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a, Transverse sections of E17.5 ovary and mesonephros from Gli1-LacZ reporter embryos (n=3) after LacZ staining (blue). The
Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a, Transverse sections of E17.5 ovary and mesonephros from Gli1-LacZ reporter embryos (n=3) after LacZ staining (blue). The
Supplementary Information
 Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells
 The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells Ralf Kist 1,2, Michelle Watson 1, Moira Crosier 1, Max Robinson 2, Jennifer
The Formation of Endoderm-Derived Taste Sensory Organs Requires a Pax9-Dependent Expansion of Embryonic Taste Bud Progenitor Cells Ralf Kist 1,2, Michelle Watson 1, Moira Crosier 1, Max Robinson 2, Jennifer
SUPPLEMENTARY INFORMATION
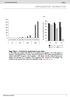 b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
Supplementary Figure 1. Chimeric analysis of inner ears. (A-H) Chimeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears.
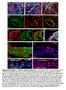 Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
Ahtiainen et al., http :// /cgi /content /full /jcb /DC1
 Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice
 Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
Supporting Information
 Supporting Information Lu et al. 10.1073/pnas.1719109115 SI Materials and Methods Mice. For tamoxifen-induced recombination, mice were given tamoxifenpreparedincornoil(20mg/ml;sigma)at5mgper25gof body
Supporting Information Lu et al. 10.1073/pnas.1719109115 SI Materials and Methods Mice. For tamoxifen-induced recombination, mice were given tamoxifenpreparedincornoil(20mg/ml;sigma)at5mgper25gof body
Tongue In the buccal cavity of the digestive system
 Tongue In the buccal cavity of the digestive system same layers as those of tubular organs Mucosa, submucosa, and muscularis muscularis = the muscularis externa no muscularis mucosa 1 Tongue ling = tongue
Tongue In the buccal cavity of the digestive system same layers as those of tubular organs Mucosa, submucosa, and muscularis muscularis = the muscularis externa no muscularis mucosa 1 Tongue ling = tongue
HUMAN ANATOMY II STUDY NOTES. At the end of this chapter the student should be able to answer the following questions:
 HUMAN ANATOMY II STUDY NOTES CHAPTER ONE The Special Senses Learning objectives At the end of this chapter the student should be able to answer the following questions: 1. What is the gross and histological
HUMAN ANATOMY II STUDY NOTES CHAPTER ONE The Special Senses Learning objectives At the end of this chapter the student should be able to answer the following questions: 1. What is the gross and histological
Supporting Information
 Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines
 Supplemental Data A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines the Initial Trajectory of Mammalian Motor Axons Ivo Lieberam, Dritan Agalliu, Takashi Nagasawa, Johan Ericson, and Thomas M. Jessell
Supplemental Data A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines the Initial Trajectory of Mammalian Motor Axons Ivo Lieberam, Dritan Agalliu, Takashi Nagasawa, Johan Ericson, and Thomas M. Jessell
Taste buds, each of which consists of about 80 specialized
 TISSUE ENGINEERING: Part C Volume 00, Number 00, 2016 ª Mary Ann Liebert, Inc. DOI: 10.1089/ten.tec.2015.0377 METHODS ARTICLE Taste Bud Labeling in Whole Tongue Epithelial Sheet in Adult Mice Nandakumar
TISSUE ENGINEERING: Part C Volume 00, Number 00, 2016 ª Mary Ann Liebert, Inc. DOI: 10.1089/ten.tec.2015.0377 METHODS ARTICLE Taste Bud Labeling in Whole Tongue Epithelial Sheet in Adult Mice Nandakumar
Stem Cells. Induced Stem Cells
 Induced Stem Cells Stem Cells Mouse and human somatic cells can either be reprogrammed to a pluripotent state or converted to another lineage with a combination of transcription factors suggesting that
Induced Stem Cells Stem Cells Mouse and human somatic cells can either be reprogrammed to a pluripotent state or converted to another lineage with a combination of transcription factors suggesting that
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
A classification of epithelial tissues
 A classification of epithelial tissues Ramray Bhat Molecular Reproduction Development and Genetics ramray@iisc.ac.in Textbooks for my portion Molecular Biology of the Cell (Bruce Alberts) 6 th Edition
A classification of epithelial tissues Ramray Bhat Molecular Reproduction Development and Genetics ramray@iisc.ac.in Textbooks for my portion Molecular Biology of the Cell (Bruce Alberts) 6 th Edition
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Recovery of taste organs and sensory function after severe loss from Hedgehog/Smoothened inhibition with cancer drug sonidegib
 Recovery of taste organs and sensory function after severe loss from Hedgehog/Smoothened inhibition with cancer drug sonidegib Archana Kumari a, Alexandre N. Ermilov b, Marina Grachtchouk b, Andrzej A.
Recovery of taste organs and sensory function after severe loss from Hedgehog/Smoothened inhibition with cancer drug sonidegib Archana Kumari a, Alexandre N. Ermilov b, Marina Grachtchouk b, Andrzej A.
Cytological and Histological Study of Adult and Neonate Epidermis in Thick and Thin Skin of Various Anatomical Sites
 Available online on www.ijpqa.com International Journal of Pharmaceutical Quality Assurance 218; 9(2); 174-179 doi: 1.25258/ijpqa.v9i2.13642 ISSN 975 956 Research Article Cytological and Histological Study
Available online on www.ijpqa.com International Journal of Pharmaceutical Quality Assurance 218; 9(2); 174-179 doi: 1.25258/ijpqa.v9i2.13642 ISSN 975 956 Research Article Cytological and Histological Study
Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus
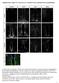 Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus a: Expression of Vimentin, GFAP, Sox2 and Nestin in anterior, central and posterior hypothalamus. In the anterior
Supplementary Figure S1: Tanycytes are restricted to the central/posterior hypothalamus a: Expression of Vimentin, GFAP, Sox2 and Nestin in anterior, central and posterior hypothalamus. In the anterior
glial cells missing and gcm2 Cell-autonomously Regulate Both Glial and Neuronal
 glial cells missing and gcm2 Cell-autonomously Regulate Both Glial and Neuronal Development in the Visual System of Drosophila Carole Chotard, Wendy Leung and Iris Salecker Supplemental Data Supplemental
glial cells missing and gcm2 Cell-autonomously Regulate Both Glial and Neuronal Development in the Visual System of Drosophila Carole Chotard, Wendy Leung and Iris Salecker Supplemental Data Supplemental
Development of teeth. 5.DM - Pedo
 Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Biologic and Materials Sciences, School of Dentistry, University of Michigan, Ann Arbor, Michigan
 THE JOURNAL OF COMPARATIVE NEUROLOGY 409:13 24 (1999) Alterations in Size, Number, and Morphology of Gustatory Papillae and Taste Buds in BDNF Null Mutant Mice Demonstrate Neural Dependence of Developing
THE JOURNAL OF COMPARATIVE NEUROLOGY 409:13 24 (1999) Alterations in Size, Number, and Morphology of Gustatory Papillae and Taste Buds in BDNF Null Mutant Mice Demonstrate Neural Dependence of Developing
Fig. S1. Upregulation of K18 and K14 mrna levels during ectoderm specification of hescs. Quantitative real-time PCR analysis of mrna levels of OCT4
 Fig. S1. Upregulation of K18 and K14 mrna levels during ectoderm specification of hescs. Quantitative real-time PCR analysis of mrna levels of OCT4 (n=3 independent differentiation experiments for each
Fig. S1. Upregulation of K18 and K14 mrna levels during ectoderm specification of hescs. Quantitative real-time PCR analysis of mrna levels of OCT4 (n=3 independent differentiation experiments for each
Skin (Integumentary System) Wheater, Chap. 9
 Skin (Integumentary System) Wheater, Chap. 9 Skin (Integument) Consists of skin and associated derivatives Largest organ of body (21 ft 2 ; 9 lbs.; has 11 miles of blood vessels) Functions: Protection
Skin (Integumentary System) Wheater, Chap. 9 Skin (Integument) Consists of skin and associated derivatives Largest organ of body (21 ft 2 ; 9 lbs.; has 11 miles of blood vessels) Functions: Protection
ANAT3231: lectures overview
 ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
GI Histology Lab 1. Prepared by: Zeina Kalaji
 GI Histology Lab 1 Prepared by: Zeina Kalaji Lip ORAL MUCOSA -Arrow shows labial salivary glands in the submucosa. VERMILLION transitional zone. SKIN Stratified Squamous epithelium, keratinized -Arrow
GI Histology Lab 1 Prepared by: Zeina Kalaji Lip ORAL MUCOSA -Arrow shows labial salivary glands in the submucosa. VERMILLION transitional zone. SKIN Stratified Squamous epithelium, keratinized -Arrow
Cell Birth and Death. Chapter Three
 Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
SUPPLEMENTARY DATA. Supplementary Table 2. Antibodies used for Immunofluoresence. Supplementary Table 3. Real-time PCR primer sequences.
 Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
Journal Club. 03/04/2012 Lama Nazzal
 Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
Journal Club 03/04/2012 Lama Nazzal NOTCH and the kidneys Is an evolutionarily conserved cell cell communication mechanism. Is a regulator of cell specification, differentiation, and tissue patterning.
UNIVERSITY OF NAIROBI
 UNIVERSITY OF NAIROBI UNIVERSITY EXAMINATIONS 2013/2014 LEVEL I MID-SEMESTER II EXAMINATION FOR THE DEGREE OF BACHELOR OF SCIENCE IN NURSING (BScN) AND BACHELOR OF PHARMACY (B.PHARM) MARKING SCHEME HNS101/UPC106:
UNIVERSITY OF NAIROBI UNIVERSITY EXAMINATIONS 2013/2014 LEVEL I MID-SEMESTER II EXAMINATION FOR THE DEGREE OF BACHELOR OF SCIENCE IN NURSING (BScN) AND BACHELOR OF PHARMACY (B.PHARM) MARKING SCHEME HNS101/UPC106:
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
ANAT3231: lectures overview
 ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
Reformation of Taste Buds by Crossed Sensory Nerves in the Rat s Tongue
 Acta physiol. scand. 1970. 79. 88-94 From the Department of Physiology, Veterinarhogskolan, Stockholm, Sweden and the Department of Zoology, University of Michigan, Ann Arbor, Michigan, U.S.A. Reformation
Acta physiol. scand. 1970. 79. 88-94 From the Department of Physiology, Veterinarhogskolan, Stockholm, Sweden and the Department of Zoology, University of Michigan, Ann Arbor, Michigan, U.S.A. Reformation
Retinoid Signaling in Progenitors Controls Specification and Regeneration of the Urothelium
 Article Retinoid Signaling in Progenitors Controls Specification and Regeneration of the Urothelium Devangini Gandhi, 1,8 Andrei Molotkov, 1,8 Ekatherina Batourina, 1,8 Kerry Schneider, 1,8 Hanbin Dan,
Article Retinoid Signaling in Progenitors Controls Specification and Regeneration of the Urothelium Devangini Gandhi, 1,8 Andrei Molotkov, 1,8 Ekatherina Batourina, 1,8 Kerry Schneider, 1,8 Hanbin Dan,
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
THE ORDERED COLUMNAR STRUCTURE OF MOUSE FILIFORM PAPILLAE
 J. Cell Sci. 22, 149-160 (1976) 149 Printed in Great Britain THE ORDERED COLUMNAR STRUCTURE OF MOUSE FILIFORM PAPILLAE WILLIAM J. HUME* AND CHRISTOPHER S. POTTEN Paterson Laboratories, Christie Hospital
J. Cell Sci. 22, 149-160 (1976) 149 Printed in Great Britain THE ORDERED COLUMNAR STRUCTURE OF MOUSE FILIFORM PAPILLAE WILLIAM J. HUME* AND CHRISTOPHER S. POTTEN Paterson Laboratories, Christie Hospital
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling
 The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
Beuling _SupplFig. 1
 euling _SupplFig. 1 Gata6: F: 5 -GGGTGTGG-3 R: 5 -GTGGTTGT-3 hga: F: 5 -GT-3 R: 5 -TTTTTTTT-3 Gcg: F: 5 -TTGGGTGGTTTG-3 R: 5 -TGGGGTGTTGTGGTG-3 Pyy: F: 5 -GGGGGG-3 R: 5 -GGGGGGTG-3 Muc2: F: 5 -TTGTGTGTG-3
euling _SupplFig. 1 Gata6: F: 5 -GGGTGTGG-3 R: 5 -GTGGTTGT-3 hga: F: 5 -GT-3 R: 5 -TTTTTTTT-3 Gcg: F: 5 -TTGGGTGGTTTG-3 R: 5 -TGGGGTGTTGTGGTG-3 Pyy: F: 5 -GGGGGG-3 R: 5 -GGGGGGTG-3 Muc2: F: 5 -TTGTGTGTG-3
Lecture Overview. Chapter 4 Epithelial Tissues Lecture 9. Introduction to Tissues. Epithelial Tissues. Glandular Epithelium
 Visual Anatomy & Physiology First Edition Martini & Ober Chapter 4 Lecture 9 Lecture Overview Introduction to Tissues Location General characteristics Functions Classification Glandular Epithelium 2 Where
Visual Anatomy & Physiology First Edition Martini & Ober Chapter 4 Lecture 9 Lecture Overview Introduction to Tissues Location General characteristics Functions Classification Glandular Epithelium 2 Where
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
Histology Notes -Part 1: Epithelial Tissues
 Introduction Group of cells w/ similar structure & function = TISSUE Four Basic Tissue Types 1. Epithelial-covers 2. Connective-supports 3. Muscular*-produces movement (will discuss in the muscular system
Introduction Group of cells w/ similar structure & function = TISSUE Four Basic Tissue Types 1. Epithelial-covers 2. Connective-supports 3. Muscular*-produces movement (will discuss in the muscular system
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is
 Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Development of Anterior Gustatory Epithelia in the Palate and Tongue Requires Epidermal Growth Factor Receptor
 Developmental Biology 242, 31 43 (2002) doi:10.1006/dbio.2001.0526, available online at http://www.idealibrary.com on Development of Anterior Gustatory Epithelia in the Palate and Tongue Requires Epidermal
Developmental Biology 242, 31 43 (2002) doi:10.1006/dbio.2001.0526, available online at http://www.idealibrary.com on Development of Anterior Gustatory Epithelia in the Palate and Tongue Requires Epidermal
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. The expression of ephrin-b2 H2BGFP persists in the post-hearingonset organ of Corti and is specifically restricted to supporting cells. Sox2 immunolabeling
SUPPLEMENTARY INFORMATION Supplementary Figure 1. The expression of ephrin-b2 H2BGFP persists in the post-hearingonset organ of Corti and is specifically restricted to supporting cells. Sox2 immunolabeling
Epidermis. Integumentary system
 Epidermis the doctor mentioned at the begging of the lecture that the slides is from different sources and has information and details that is enough for us so we don t have to go back and read from the
Epidermis the doctor mentioned at the begging of the lecture that the slides is from different sources and has information and details that is enough for us so we don t have to go back and read from the
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Intravital Microscopic Interrogation of Peripheral Taste Sensation
 Supplementary Information Intravital Microscopic Interrogation of Peripheral Taste Sensation Myunghwan Choi 1, Woei Ming Lee 1,2, and Seok-Hyun Yun 1 * 1 Harvard Medical School and Wellman Center for Photomedicine,
Supplementary Information Intravital Microscopic Interrogation of Peripheral Taste Sensation Myunghwan Choi 1, Woei Ming Lee 1,2, and Seok-Hyun Yun 1 * 1 Harvard Medical School and Wellman Center for Photomedicine,
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Neural stem cells and the neurobiology of ageing. Chen Siyun 1, Dawe G.S. 2
 ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
Lecture Overview. Marieb s Human Anatomy and Physiology. Chapter 4 Tissues: The Living Fabric Epithelial Tissues Lecture 9. Introduction to Tissues
 Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 4 Tissues: The Living Fabric Epithelial Tissues Lecture 9 Lecture Overview Introduction to Tissues Epithelial Tissues Location General characteristics
Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 4 Tissues: The Living Fabric Epithelial Tissues Lecture 9 Lecture Overview Introduction to Tissues Epithelial Tissues Location General characteristics
Epithelial-Derived Brain-Derived Neurotrophic Factor Is Required for Gustatory Neuron Targeting during a Critical Developmental Period
 3354 The Journal of Neuroscience, March 18, 2009 29(11):3354 3364 Development/Plasticity/Repair Epithelial-Derived Brain-Derived Neurotrophic Factor Is Required for Gustatory Neuron Targeting during a
3354 The Journal of Neuroscience, March 18, 2009 29(11):3354 3364 Development/Plasticity/Repair Epithelial-Derived Brain-Derived Neurotrophic Factor Is Required for Gustatory Neuron Targeting during a
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials)
 Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials) Sunny Y. Wong, Allen D. Seol, Po-Lin So, Alexandre N. Ermilov, Christopher K.
Primary Cilia Can Both Mediate and Suppress Hedgehog Pathway- Dependent Tumorigenesis (Supplementary Figures and Materials) Sunny Y. Wong, Allen D. Seol, Po-Lin So, Alexandre N. Ermilov, Christopher K.
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Ophthalmology, Radiation Oncology,
 Supporting Online Material Journal: Nature Neuroscience Article Title: Corresponding Author: All Authors: Affiliations: Tanycytes of the Hypothalamic Median Eminence Form a Diet- Responsive Neurogenic
Supporting Online Material Journal: Nature Neuroscience Article Title: Corresponding Author: All Authors: Affiliations: Tanycytes of the Hypothalamic Median Eminence Form a Diet- Responsive Neurogenic
Nature Medicine doi: /nm.2860
 Supplemental Figure Legends Supplemental Figure 1: Hypomorphic expression of IFT88 results in olfactory signaling proteins no longer localizing to the ciliary layer. (a) ACIII localizes to the cilia and
Supplemental Figure Legends Supplemental Figure 1: Hypomorphic expression of IFT88 results in olfactory signaling proteins no longer localizing to the ciliary layer. (a) ACIII localizes to the cilia and
Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
 Wright et al. BMC Developmental Biology (2015) 15:33 DOI 10.1186/s12861-015-0083-8 RESEARCH ARTICLE Open Access Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
Wright et al. BMC Developmental Biology (2015) 15:33 DOI 10.1186/s12861-015-0083-8 RESEARCH ARTICLE Open Access Cooperative and independent functions of FGF and Wnt signaling during early inner ear development
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/326/5951/443/dc1 Supporting Online Material for The Taste of Carbonation Jayaram Chandrashekar, David Yarmolinsky, Lars von Buchholtz, Yuki Oka, William Sly, Nicholas
www.sciencemag.org/cgi/content/full/326/5951/443/dc1 Supporting Online Material for The Taste of Carbonation Jayaram Chandrashekar, David Yarmolinsky, Lars von Buchholtz, Yuki Oka, William Sly, Nicholas
AP VP DLP H&E. p-akt DLP
 A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Terminology. Terminology. Terminology. Terminology. Terminology. Bromodeoxyuridine
 Kateřina Náměstková, Zuzana Šimonová, Eva Syková Behavioural Brain Research Bromodeoxyuridine : Doublecortin : DCX Glial Fibrillary Acidic Protein : GFAP Trace eye blink conditioning 1 Volume 163 : pp.
Kateřina Náměstková, Zuzana Šimonová, Eva Syková Behavioural Brain Research Bromodeoxyuridine : Doublecortin : DCX Glial Fibrillary Acidic Protein : GFAP Trace eye blink conditioning 1 Volume 163 : pp.
THE sebaceous glands of the rabbit consist of clusters of about ten cells
 79 On the Relationship between Mammary, Sweat, and Sebaceous Glands By D. B. CARLISLE (From the Department of Zoology and Comparative Anatomy, Oxford, and the Plymouth Laboratory of the Marine Biological
79 On the Relationship between Mammary, Sweat, and Sebaceous Glands By D. B. CARLISLE (From the Department of Zoology and Comparative Anatomy, Oxford, and the Plymouth Laboratory of the Marine Biological
SUPPLEMENTAL INFORMATION FOR. PAX7 expression defines germline stem cells in the adult testis
 SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
SUPPLEMENTARY FIG. S2. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of dnscs.
 Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
The Contribution Of Tie2-Lineage Cells To rhbmp-2 Induced Bone Formation
 The Contribution Of Tie2-Lineage Cells To rhbmp-2 Induced Bone Formation Mille P. Kolind, Ph.D 1, Alastair Aiken 1, Kathy Mikulec 1, Lauren Peacock 1, David Little 1,2, Aaron Schindeler, PhD 1,2. 1 Orthopaedic
The Contribution Of Tie2-Lineage Cells To rhbmp-2 Induced Bone Formation Mille P. Kolind, Ph.D 1, Alastair Aiken 1, Kathy Mikulec 1, Lauren Peacock 1, David Little 1,2, Aaron Schindeler, PhD 1,2. 1 Orthopaedic
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and
 Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Generation of a conditional allele of the Kindlin-2 gene. (A) A restriction map of the relevant genomic region of Kindlin-2 (top), the targeting construct
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Generation of a conditional allele of the Kindlin-2 gene. (A) A restriction map of the relevant genomic region of Kindlin-2 (top), the targeting construct
Supplemental Information. Myocardial Polyploidization Creates a Barrier. to Heart Regeneration in Zebrafish
 Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS
 ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
EMBO REPORT SUPPLEMENTARY SECTION. Quantitation of mitotic cells after perturbation of Notch signalling.
 EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature07173 SUPPLEMENTARY INFORMATION Supplementary Figure Legends: Supplementary Figure 1: Model of SSC and CPC divisions a, Somatic stem cells (SSC) reside adjacent to the hub (red), self-renew
doi: 10.1038/nature07173 SUPPLEMENTARY INFORMATION Supplementary Figure Legends: Supplementary Figure 1: Model of SSC and CPC divisions a, Somatic stem cells (SSC) reside adjacent to the hub (red), self-renew
Cell Death & Renewal (part 2)
 17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants
 SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
LESSON ASSIGNMENT. The Human Integumentary and Fascial Systems. After completing this lesson, you should be able to:
 LESSON ASSIGNMENT LESSON 3 The Human Integumentary and Fascial Systems. TEXT ASSIGNMENT Paragraphs 3-1 through 3-14. LESSON OBJECTIVES After completing this lesson, you should be able to: 3-1. Define integumentary
LESSON ASSIGNMENT LESSON 3 The Human Integumentary and Fascial Systems. TEXT ASSIGNMENT Paragraphs 3-1 through 3-14. LESSON OBJECTIVES After completing this lesson, you should be able to: 3-1. Define integumentary
Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System
 Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System Question No. 1 of 10 The lymphatic system is formed early during human development. Which of the following statements
Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System Question No. 1 of 10 The lymphatic system is formed early during human development. Which of the following statements
DEFINING A TISSUE STEM CELL DRIVEN RUNX1/STAT3 SIGNALING AXIS IN EPITHELIAL CANCER
 Manuscript EMBO-2012-81903 DEFINING A TISSUE STEM CELL DRIVEN RUNX1/STAT3 SIGNALING AXIS IN EPITHELIAL CANCER Cornelia Johanna Franziska Scheitz, Tae Seung Lee, David James McDermitt, and Tudorita Tumbar
Manuscript EMBO-2012-81903 DEFINING A TISSUE STEM CELL DRIVEN RUNX1/STAT3 SIGNALING AXIS IN EPITHELIAL CANCER Cornelia Johanna Franziska Scheitz, Tae Seung Lee, David James McDermitt, and Tudorita Tumbar
Skin. Kristine Krafts, M.D.
 Skin Kristine Krafts, M.D. Skin Lecture Objectives Describe the functions of skin. Describe the structure, location and function of the cell types found in epidermis: keratinocytes, melanocytes, Langerhans
Skin Kristine Krafts, M.D. Skin Lecture Objectives Describe the functions of skin. Describe the structure, location and function of the cell types found in epidermis: keratinocytes, melanocytes, Langerhans
Chapter 4 Opener Pearson Education, Inc.
 Chapter 4 Opener Introduction The integumentary system is composed of: Skin Hair Nails Sweat glands Oil glands Mammary glands The skin is the most visible organ of the body Clinicians can tell a lot about
Chapter 4 Opener Introduction The integumentary system is composed of: Skin Hair Nails Sweat glands Oil glands Mammary glands The skin is the most visible organ of the body Clinicians can tell a lot about
Development of retinal synaptic arrays in the inner plexiform layer of dark-reared mice
 /. Embryo/, exp. Morph. Vol. 54, pp. 219-227, 1979 219 Printed in Great Britain Company of Biologists Limited 1977 Development of retinal synaptic arrays in the inner plexiform layer of dark-reared mice
/. Embryo/, exp. Morph. Vol. 54, pp. 219-227, 1979 219 Printed in Great Britain Company of Biologists Limited 1977 Development of retinal synaptic arrays in the inner plexiform layer of dark-reared mice
Histology = the study of tissues. Tissue = a complex of cells that have a common function
 { EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
{ EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2535 Figure S1 SOX10 is expressed in human giant congenital nevi and its expression in human melanoma samples suggests that SOX10 functions in a MITF-independent manner. a, b, Representative
DOI: 10.1038/ncb2535 Figure S1 SOX10 is expressed in human giant congenital nevi and its expression in human melanoma samples suggests that SOX10 functions in a MITF-independent manner. a, b, Representative
Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development
 Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development Jae-Young Kim Department of Medical Science The Graduate School, Yonsei University Pattern Formation and Morphogenesis
Pattern Formation and Morphogenesis of Fungiform Papillae during Mouse Development Jae-Young Kim Department of Medical Science The Graduate School, Yonsei University Pattern Formation and Morphogenesis
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Biology. Dr. Khalida Ibrahim
 Dr. Khalida Ibrahim Biology Histology: Histology: is the study of the tissues of the body. Tissue: group of similar cells combined to perform a common function. The human body is composed of only 4 basic
Dr. Khalida Ibrahim Biology Histology: Histology: is the study of the tissues of the body. Tissue: group of similar cells combined to perform a common function. The human body is composed of only 4 basic
Material and Methods Production and analysis of -gal+ clones Immunostaining Optical projection tomography Statistical analysis
 Material and Methods Production and analysis of β-gal+ clones The α-cardiac actin nlaacz/+ and Cx40 egfp mouse lines used in this study were genotyped as previously reported 1, 2. Double transgenic knock-in
Material and Methods Production and analysis of β-gal+ clones The α-cardiac actin nlaacz/+ and Cx40 egfp mouse lines used in this study were genotyped as previously reported 1, 2. Double transgenic knock-in
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Ras V12 expression in the entire eye-antennal disc does not cause invasive tumours. a, Eye-antennal discs expressing Ras V12 in all cells (marked with GFP, green) overgrow moderately
Supplementary Figure 1. Ras V12 expression in the entire eye-antennal disc does not cause invasive tumours. a, Eye-antennal discs expressing Ras V12 in all cells (marked with GFP, green) overgrow moderately
Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine
 FULL PAPER Anatomy Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine Takashi TAKEUCHI 1) and Tatsuo GONDA 1) 1) Institute of Experimental Animals, Shimane Medical University,
FULL PAPER Anatomy Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine Takashi TAKEUCHI 1) and Tatsuo GONDA 1) 1) Institute of Experimental Animals, Shimane Medical University,
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Table 1. List of primers used in this study
 Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates
 Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates L. Cheng, A. Arata, R. Mizuguchi, Y. Qian, A. Karunaratne, P.A. Gray, S. Arata, S. Shirasawa, M. Bouchard,
Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates L. Cheng, A. Arata, R. Mizuguchi, Y. Qian, A. Karunaratne, P.A. Gray, S. Arata, S. Shirasawa, M. Bouchard,
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Introduction Kit Components Cat. # # of vials Reagent Quantity Storage
 Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
