Metalloproteinase expression is altered in cardiac and skeletal muscle in cancer cachexia
|
|
|
- Ross Reed
- 6 years ago
- Views:
Transcription
1 Am J Physiol Heart Circ Physiol 309: H685 H691, First published June 19, 2015; doi: /ajpheart Metalloproteinase expression is altered in cardiac and skeletal muscle in cancer cachexia Raymond D. Devine, 1,2 Sabahattin Bicer, 3 Peter J. Reiser, 3 Markus Velten, 5 and Loren E. Wold 1,4,6 1 Dorothy M. Davis Heart and Lung Research Institute, The Ohio State University, Columbus, Ohio; 2 Molecular, Cellular and Developmental Biology Graduate Program, The Ohio State University, Columbus, Ohio; 3 Division of Biosciences, College of Dentistry, The Ohio State University, Columbus, Ohio; 4 Department of Physiology and Cell Biology, The Ohio State University, Columbus, Ohio; 5 Department of Anesthesiology and Intensive Care Medicine, Rheinische Friedrich-Wilhelms- University, University Medical Center, Bonn, Germany; and 6 College of Nursing, The Ohio State University, Columbus, Ohio Submitted 11 February 2015; accepted in final form 5 June 2015 Devine RD, Bicer S, Reiser PJ, Velten M, Wold LE. Metalloproteinase expression is altered in cardiac and skeletal muscle in cancer cachexia. Am J Physiol Heart Circ Physiol 309: H685 H691, First published June 19, 2015; doi: /ajpheart Cardiac and skeletal muscle dysfunction is a recognized effect of cancerinduced cachexia, with alterations in heart function leading to heart failure and negatively impacting patient morbidity. Cachexia is a complex and multifaceted disease state with several potential contributors to cardiac and skeletal muscle dysfunction. Matrix metalloproteinases (MMPs) are a family of enzymes capable of degrading components of the extracellular matrix (ECM). Changes to the ECM cause disruption both in the connections between cells at the basement membrane and in cell-to-cell interactions. In the present study, we used a murine model of C26 adenocarcinoma-induced cancer cachexia to determine changes in MMP gene and protein expression in cardiac and skeletal muscle. We analyzed MMP-2, MMP-3, MMP-9, and MMP-14 as they have been shown to contribute to both cardiac and skeletal muscle ECM changes and, thereby, to pathology in models of heart failure and muscular dystrophy. In our model, cardiac and skeletal muscles showed a significant increase in RNA and protein levels of several MMPs and tissue inhibitors of metalloproteinases. Cardiac muscle showed significant protein increases in MMP-2, MMP-3, MMP-9, and MMP-14, whereas skeletal muscles showed increases in MMP-2, MMP-3, and MMP-14. Furthermore, collagen deposition was increased after C26 adenocarcinoma-induced cancer cachexia as indicated by an increased left ventricular picrosirius red-positive-stained area. Increases in serum hydroxyproline suggest increased collagen turnover, implicating skeletal muscle remodeling. Our findings demonstrate that cancer cachexia-associated matrix remodeling results in cardiac fibrosis and possible skeletal muscle remodeling. With these findings, MMPs represent a possible therapeutic target for the treatment of cancer-induced cachexia. matrix metalloproteinase; tissue inhibitors of matrix metalloproteinase; extracellular matrix; cancer cachexia NEW & NOTEWORTHY Our findings represent a significant expansion in the current field of cancer cachexia with an emphasis on targeted therapy. Increased matrix metalloproteinase expression results in cardiac fibrosis and skeletal muscle remodeling in response to C26 adenocarcinoma cell injection. These findings expand the field and introduce potentially new therapeutic strategies. CANCER is a rapidly growing health concern worldwide, with 25% of all deaths attributed to cancer in the United States (31). Address for reprint requests and other correspondence: L. E. Wold, 603 Dorothy M. Davis Heart and Lung Research Institute, The Ohio State Univ., 473 W. 12th Ave., Columbus OH, ( Loren.wold@osumc.edu). During disease progression 30 80% of cancer patients develop cachexia, a syndrome characterized by marked wasting of adipose tissue and skeletal muscle (13). Cachexia not only increases the likelihood of morbidity from cancer but also is directly responsible for 20 40% of deaths in cachectic patients (37). Cachexia is a detrimental syndrome, negatively affecting patient quality of life and the potential for recovery (14, 41). Understanding the molecular mechanisms of cancer cachexia could assist in creating effective therapies to improve survival and quality of life in these patients. Increased serum levels of proinflammatory cytokines, including IL-6 and TNF-, play pivotal roles in tumor-induced muscle wasting (3, 4, 42). These cytokines increase activity of the ubiquitin proteasome and autophagy systems, promoting muscle protein breakdown (10, 19, 24). Ubiquitin activity may play a role in early skeletal muscle wasting; however, the ubiquitin pathway of degradation does not completely explain cachexia-associated pathology. For example, studies in human cancer patients have not correlated increased ubiquitin mrna levels with weight loss (36). Therefore, other degradation systems likely contribute to skeletal muscle wasting in cancer cachexia. In addition to influencing skeletal muscle mass and function, tumor burden also promotes cardiac dysfunction. CD2F1 mice injected with C26 adenocarcinoma cells (5) are a recent established model to study cancer cachexia. In this model, tumor development is directly correlated with reduced cardiac function as measured by ejection fraction and fractional shortening (35). Although in vivo cardiac function was reduced in C26 adenocarcinoma-induced tumor cachexia, in vitro analyses of isolated cardiomyocytes did not indicate cardiac dysfunction at the cellular level (43), suggesting the involvement of the extracellular matrix (ECM) in the development of cardiac dysfunction. Heart samples from human cancer cachexia patients show evidence of myocardial fibrosis (32), potentially associated with matrix metalloproteinase (MMP)-mediated alterations in the ECM. MMPs degrade several components of the ECM, resulting in tissue remodeling processes including angiogenesis (23). In addition, MMPs play active roles in cell proliferation and migration. MMPs are secreted in an inactive form that requires the removal of an inhibitory domain for activation (26). Additionally, endogenous inhibitors of MMPs [tissue inhibitors of metalloproteinases (TIMPs)] inhibit MMP activity by directly binding to the MMP active site. MMPs are secreted by tumor cells, and this facilitates angiogenesis for tumor growth and survival as well as metastasis (11). However, the involvement /15 Copyright 2015 the American Physiological Society H685
2 H686 CANCER CACHEXIA AND MMPs of MMP activity in muscle wasting or dysfunction in cancer cachexia remains unknown. The purpose of the present study was to investigate MMP and TIMP production in skeletal and cardiac muscle in a model of cancer cachexia. We show that tumor-bearing mice have increased MMP levels in both cardiac and skeletal muscle compared with control mice, which may contribute to altered heart and skeletal muscle function associated with cachexia. These findings provide insights into the contribution of MMPs to skeletal muscle wasting and cardiac dysfunction in cancer cachexia. MATERIALS AND METHODS Mice. Adult (10 wk old) female CD2F1 (BALB/c DBA/2 F1) mice weighing g were obtained from Charles River Laboratories. Mice were housed with 1 3 mice/cage and maintained at 25 C under a 12:12-h light-dark cycle with ad libitum access to water and standard rodent chow. All animal care and procedures were approved by the Institutional Animal Care and Use Committee of the Ohio State University. Mouse model of tumor growth. The C26 cell line was maintained in culture and prepared for injection as previously described (34). Mice were randomly selected to receive the cell injection subcutaneously between the scapulae with cells in 0.2 ml of PBS, whereas control mice were injected with PBS alone. Tumor growth was usually palpable by day 12 postinjection, and mice became moribund typically by day 24 postinjection. In this study, mice were euthanized by day 21 postinjection. Mice were euthanized using a ketamine (10 mg/ml)-xylazine (1 mg/ml) cocktail at a volume of 0.01 ml/g body wt ( 0.2 ml), and blood was withdrawn from the brachial artery. The heart, hindlimb muscles, and diaphragm were dissected, snap frozen in liquid nitrogen, and stored at 80 C until biochemical analyses. Samples and preparation. The free wall of the left ventricle (LV), diaphragm, and soleus and extensor digitorum longus (EDL) muscles of control and tumor-bearing mice were used for electrophoretic gel and Western blot analyses. Muscles were dissected and homogenized Fig. 1. RNA expression changes of matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs) in left ventricle (A), diaphragm (B), extensor digitorum longus (EDL; C), and soleus (D) of control and C26 tumor-bearing mice. RNA expression levels were determined using the Livak method with GAPDH serving as the internal control. C t, threshold cycle. *P 0.05 was considered statistically significant. (model PRO200 homogenizer, PRO Scientific, Monroe, CT) in sample buffer (8) (30 l/mg muscle) for 15 s. Homogenates were then centrifuged at 14,000 rpm for 4 min. Supernatants of the samples were collected, heated at 65 C for 2 min, and maintained on ice for 5 min. Samples were maintained in a 40 C freezer until analyzed. Gel electrophoresis. Separating (12% acrylamide with a 200:1 cross-linking ratio) and stacking (4% acrylamide with a 50:1 crosslinking ratio) gels were prepared as previously described (6). Gels were run at constant current (30 ma/gel) for 3 h at 18 C in Hoefer SE600 units (Hoefer Scientific, San Francisco, CA). Gels were stained with Coomassie blue (6). Stained gels were used to determine the loading amount, which was determined by the visualization of actin. Images of the gels were obtained with a digital camera (Spot Insight Firewire, model 18.2, Diagnostic Instruments, Sterling Heights, MI). Gels were analyzed with ImageJ software (National Institutes of Health). Western blot analyses. Gels for immunoblot analysis were run, and proteins were transferred onto nitrocellulose membranes at constant voltage (100 V) for 1 h. Membranes were incubated with 1% BSA in Tris-buffered saline with Tween 20 (TBST) for 1 h at 25 C or overnight at 4 C. Membranes were incubated with primary antibodies for 2 h at 25 C and washed with TBST with gentle shaking three times for 10 min per wash. Membranes were then incubated with secondary antibodies for 1 h at 25 C. Membranes were washed with TBST for 30 min (three 10-min washes with gentle shaking). Color was developed using 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium color development substrate (S3771, Promega, Madison, WI). Finally, membranes were air dried, and digital images were captured as described above for gel imaging. Images were analyzed with ImageJ software. Primary antibodies were anti-mmp-2 clone 2C1 (1:100 diluted), anti-mmp-3 clone F-1 (1:100), anti-mmp9 clone E-11 (1:200), anti-mmp-14 clone C-9 (1:200), anti-timp-1 clone H-150 (1:100), and anti-timp2 clone 3A4 (1:200). All primary antibodies were purchased from Santa Cruz Biotechnology (Dallas, TX). Primary antibodies were diluted with 0.3% BSA in TBST. Secondary antibodies were anti-mouse IgG1 (S3721, Promega, di-
3 CANCER CACHEXIA AND MMPs H687 luted 1:10,000) and anti-rabbit IgG (S3731, Promega, diluted 1:10,000). Secondary antibodies were diluted with 1% BSA in TBST. RNA isolation and real-time quantitative PCR. RNA was isolated from the free wall of the LV, EDL, soleus, and diaphragm using a TissueLyser system in TRIzol reagent (Sigma-Aldrich, St. Louis, MO). RNA was extracted using RNeasy spin column purification (Qiagen, Valencia, CA). RNA was quantitated, and ng of RNA were reverse transcribed using iscript Reverse Transcription Supermix (Bio-Rad, Hercules, CA). Quantitative PCR was performed using a three-step protocol on the CFX96 Real Time System (Bio- Rad), as previously described (43). Briefly, cdna was amplified using real-time quantitative PCR and was normalized based on reference cdna (GAPDH). Data were analyzed using the 2 ( C t) algorithm (where C t is threshold cycle) (18). Picrosirius red quantification. Hearts from control and tumorbearing mice were fixed in paraformaldehyde and then embedded in paraffin. Paraffin blocks were cut in 10- m sections, mounted on glass slides, and rehydrated. After rehydration, slides were placed in 0.1% sirius red and diluted in saturated picric acid for 90 min. After being washed, slides were dehydrated with graded alcohols before being cleared and then mounted in synthetic media (9). For histological analyses, slides from the area below the papillary muscles were identified, and images were taken of the septum and posterior, free, and anterior walls at a magnification of 200 using a BX 41 microscope (Olympus, Hamburg, Germany). Quantification and planimetric analyses were performed using Analysis (Olympus). The picrosirius red positive-stained area was quantified as a percentage of the total myocardial area (12). Hydroxyproline assay. Serum from control and tumor-bearing mice was used to perform a hydroxyproline assay (Sigma-Aldrich). Briefly, samples were hydrolyzed with hydrochloric acid and then spun at 13,000 g. Samples were then treated with chloramine-t to oxidize hydroxyproline, and perchloric acid was then added to remove chloramine-t. Oxidized samples were incubated with 4-(dimethylamino)benzaldehyde (DBMA) to produce a photometric product at 560 nm. Results were acquired using a Bio-Tek Power Wave (Bio-Tek, Winooski, VT) plate reader and calculated using the standard curve of hydroxyproline provided with the assay kit. Statistical analyses. Values from six control mice and six tumorbearing mice are reported as means SE. The two groups were compared using a Student s t-test, where P 0.05 was used to indicate statistical significance. RESULTS Tumor burden increased MMP and TIMP mrna expression in striated muscle. mrna expression of MMPs and TIMPs was determined in skeletal and cardiac muscles of tumorbearing mice to test whether they were elevated in association with cancer cachexia (Fig. 1). In the LV, MMP-2, MMP-3, and MMP-14 were significantly increased in tumor-bearing mice compared with control mice (Fig. 1A). The skeletal muscle response for tumor-bearing mice differed from that of cardiac muscle. In the diaphragm, MMP-2 and MMP-9 were significantly increased (Fig. 1B), whereas both EDL and soleus muscles showed significant increases in MMP-3 (Fig. 1, C and D). Certain striated muscles of tumor-bearing mice had increased levels of TIMP-1 (Fig. 1) compared with those from control mice. The LV had significantly increased TIMP-1 expression, but TIMP-2 was unchanged (Fig. 1A). In skeletal muscle, the diaphragm had significantly increased expression of TIMP-1 but not TIMP-2, whereas the EDL and soleus muscles showed no increases in either TIMP-1 or TIMP-2 (Fig. 1, B D). MMP and TIMP protein levels were increased in tumorbearing mice. In the LV, protein levels of MMP-2, MMP-3, MMP-9, and MMP-14 were significantly increased compared Fig. 2. Quantification of MMP and TIMP protein expression levels in left ventricle (A), diaphragm (B), EDL (C), and soleus (D) of control and C26 tumor-bearing mice. Protein levels are expressed as the ratio between the target protein (MMP or TIMP) and actin, the reference protein. *P 0.05 was considered statistically significant.
4 H688 CANCER CACHEXIA AND MMPs Fig. 3. Representative immunoblots showing changes in MMP-2, MMP-3, MMP-9, and MMP-14 in cardiac and skeletal muscles from control and tumor-bearing mice. with control values (Fig. 2A). The EDL, soleus, and diaphragm demonstrated similar changes in MMP protein levels. The diaphragm, EDL, and soleus had significantly increased expression of MMP-2, MMP-3, and MMP-14 without changes in MMP-9 (Fig. 2, B D). Protein levels of TIMP-2 were increased in tumor-bearing mice. The LV had significantly increased TIMP-2 but not TIMP-1 protein levels (Fig. 2A). The diaphragm, EDL, and soleus showed significant increases in TIMP-2 but not TIMP-1 in a manner similar to cardiac muscle (Fig. 2, B D). Representative immunoblots of MMPs and TIMPs are shown in Figs. 3 and 4. Collagen deposition was increased in the LV of tumorbearing mice. To assess increased collagen deposition in the LV, we analyzed picrosirius red-stained sections. There were Fig. 4. Representative immunoblots showing changes in TIMP-1 and TIMP-2 protein in cardiac and skeletal muscles from control and tumor-bearing mice.
5 CANCER CACHEXIA AND MMPs H689 Fig. 5. Collagen deposition in the left ventricle determined by picrosirius red staining (A) and quantified as a percentage of the total area (B). **P 0.05 was considered statistically significant. significantly increased amounts of collagen deposition in the LV of tumor-bearing mice compared with control mice (Fig. 5). Collagen deposition, as a percentage of the total area, was significantly increased in tumor-bearing mice. Hydroxyproline was increased in the serum of tumor-bearing mice. To assess increased collagen turnover, we analyzed the serum of mice for the collagen byproduct hydroxyproline. There was a significant increase in the amount of hydroxyproline in the serum of tumor-bearing mice (Fig. 6), indicating increased MMP activity and collagen turnover. DISCUSSION In the present study, we found that tumor burden increased the mrna expression and protein amount of MMPs and TIMPs in striated muscle. Upregulation of the ubiquitin proteasome pathway has been established in cancer-induced cachexia (22), but other proteolytic or degradation pathways have not been extensively studied. This study is the first to demonstrate that cancer-induced cachexia in a mouse model is associated with increased expression of MMPs and TIMPs in skeletal and cardiac muscle. This increased expression of MMPs could contribute to the cardiovascular and skeletal muscle pathologies observed in the tumor-burdened mouse. The cardiac phenotype in the cachectic mouse model used in this study exhibited decreased in vivo function but not decreased isolated cardiomyocyte contractility (43). The increased levels of muscle atrophy F-box (MAFbx)/muscle RING finger 1 (MuRF-1) found in our model are expected to negatively affect cardiomyocyte contractility (1, 18). The action of MAFbx/MuRF-1 was demonstrated in a rat model of chronic heart failure, where increases in MAFbx and MuRF-1 resulted in contractile dysfunction that was abrogated by treatment with MAFbx/MuRF-1 small interfering RNA (1). However, we did not observe cardiomyocyte dysfunction 21 days after tumor cell injection in our current model (43). While there are increases in MAFBx/MuRF gene and protein expression in the heart of cachectic mice (34, 43), the lack of changes in cardiomyocyte contractile properties indicates that the functional changes are not entirely due to the ubiquitin system. Increases in MMPs have been implicated in heart failure and morbidity in heart failure patients (15a). The gelatinases MMP-2 and MMP-9 (7, 30) are most commonly associated with alterations of the ECM in heart failure. We observed significant upregulation in both enzymes at the protein level. The collagenase MMP-9 was not increased at the RNA level but was increased at the protein level. MMP-9 protein is often present at a basal level and activated posttranscriptionally during inflammatory states, possibly explaining the discrepancy in RNA levels compared with protein levels. MMP-3 and MMP-14 (muscle type 1-MMP) have not been directly implicated in cardiac remodeling but more often regulate the expression and activity of other MMPs (40). MMP-3 may regulate MMP-2 and MMP-9 in heart failure models, whereas MMP-14 activates MMP-2. TIMP-1 was significantly increased at the RNA level but showed no change in protein expression, whereas TIMP-2 showed no RNA increase but a significant protein increase. TIMP-1 RNA regulation shares some transcription factors with MMP-9 (39), which also showed no RNA increase, possibly indicating certain transcription factors may not be activated in the tumor-burdened mouse. The increases in collagen deposition, determined by both picosirius red staining and hydroxylproline assay, demonstrate that MMPs are actively remodeling the ECM and increasing collagen deposition. This MMP activity and matrix remodeling are most likely responsible for the decreases in wall thickness during contraction, contributing to decreased cardiac function. Skeletal muscle loss has been an intense area of focus within the cachectic field due to the wasting present in both human cancer patients and animal models of cancer. This wasting is due to activation of the ubiquitin proteasome system, primarily from MAFBx and MuRF-1. These two E3 ubiquitin ligases Fig. 6. Quantitation of hydroxyproline in the serum of control and tumorbearing mice. *P 0.05 was considered statistically significant.
6 H690 CANCER CACHEXIA AND MMPs have been heavily studied and regarded as a primary cause of wasting in cachexia. Treatment with the proteasome inhibitor MIG132 has been shown to alleviate cachexia by inhibiting MAFBx and MuRF-1, showing their involvement in wasting (44). The wasting phenotype does not involve a reduction in the total number of muscle fibers or altered force generation per unit of cross-sectional area (25, 29), although others have reported a significant reduction of force/cross-sectional area in the murine C26 model (28) and in human patients (38). Skeletal muscles in some cachectic models are unable to generate new muscle cells due to the inability of satellite cells to mature into myoblasts (16). MMPs can contribute to the differentiation of satellite cells (27). In canine and murine models of muscular dystrophy, MMPs promote the release of satellite cells as well as the release of growth factors that stimulate proliferation (15, 20). There is evidence that MMP-2 and MMP-9 activities contribute to the differentiation of satellite cells in mdx mice. However, MMP-2 and MMP-9 have been shown to degrade the ECM and to prevent muscle regeneration in inflammatory myopathies (20). The extent of MMP upregulation appears to play a role in their specific activity; a small upregulation can release satellite cells and permit differentiation, whereas a more profound increase can disrupt the ECM and prevent differentiation. At the RNA level, only MMP-3 was upregulated in the soleus and EDL muscles, whereas at the protein level, MMP-2, MMP-3, MMP-14, and TIMP-2 were significantly increased. There is posttranslational modification occurring; however, the reason remains unknown. For MMP and TIMP-2, it is possible that skeletal muscle has higher basal amounts of RNA, negating the need for a prolific gene response while maintaining a robust protein response. This aberrant increase in MMPs in the EDL and soleus muscles in the cachectic mouse model may contribute to the inability of satellite cell proliferation by breaking down the surrounding matrix and disrupting normal cell survival signals transmitted through the matrix and between cells. Diaphragm strips from tumor-bearing mice generate decreased peak specific force compared with normal mice (25, 28). Cachectic patients undergo respiratory complications due to failure of the diaphragm (28). The decreased peak specific force of the diaphragm in tumor-bearing mice could be the mechanism by which human patients experience respiratory complications. Increased MMPs in the diaphragm could change the underlying architecture of the ECM, contributing to changes in the physical and mechanical properties of the diaphragm. Additionally, while MMPs perform most of their proteolytic actions extracellularly, they have been shown to localize to sarcomeric proteins in the heart (2), and it is possible that they are able to localize to sarcomeric proteins in other muscle types. In a mouse model of mdx, there was decreased specific force of the diaphragm (17, 33). A separate study (21) showed that decreased force production could be ameliorated by the MMP inhibitor batimastat. However, Kumar et al. (21) investigated several MMPs as well as a disintegrin and metalloproteinases, making the contributions of specific MMPs in respiratory pathology difficult to evaluate. The roles of MMPs in diaphragm and respiratory dysfunction warrant further investigation. In the present study, we demonstrated a significant increase of specific MMPs in cardiac and skeletal muscles at the RNA and protein levels. Currently, the only other studies that have reported changes in MMPs in both cardiac and skeletal muscles were performed in muscular dystrophy models. In the mdx model, increases in MMP-2 and MMP-9 correlated with cardiomyopathy in mice. This cardiac dysfunction is also coupled with skeletal muscle dysfunction. As mentioned above, treatment of mdx mice with batimastat, a broad MMP inhibitor, improved skeletal muscle function and significantly reduced fibrosis (21). MMP-2 and MMP-9 have been most heavily studied in cardiac and skeletal muscles; however, it is known that MMP-3 can regulate the activity of other MMPs, and MMP-14 can activate MMP-2. These MMP-induced MMP expression and activity changes can account for some of the differences in gene expression compared with protein differences. MMPs could cleave other substrates, leading to a protranscription rate while not altering the amount of gene expressed. While we found increased protein expression of TIMP-2, which is capable of inhibiting all MMPs, from our histology and serum assays we can infer that TIMP-2 is no longer providing adequate MMP inhibition. TIMPs, while having a high affinity for MMPs, can bind the active site in a one-to-one ratio, and this can significantly limit their inhibitory activity in cases of marked increases in MMPs. Further research is needed to elucidate the role of MMPs in the cardiac and skeletal muscle dysfunction in cancer cachexia. Determining to what extent MMPs contribute to the pathology will potentially lead to the development of effective treatments for cachectic patients, especially with respect to cardiac complications due to tumor burden. ACKNOWLEDGMENTS The authors thank Diana Norden for assistance with animal husbandry and Cynthia McAllister from the Research Institute at Nationwide Children s Hospital Morphology Core and technicians for help with histology. GRANTS This work was supported National Institutes of Health Grants NR (to L. E. Wold and P. J. Reiser) and ES (to L. E. Wold). DISCLOSURES No conflicts of interest, financial or otherwise, are declared by the author(s). AUTHOR CONTRIBUTIONS Author contributions: R.D.D., S.B., P.J.R., and L.E.W. conception and design of research; R.D.D., S.B., P.J.R., and M.V. performed experiments; R.D.D., S.B., P.J.R., M.V., and L.E.W. analyzed data; R.D.D., S.B., P.J.R., M.V., and L.E.W. interpreted results of experiments; R.D.D., S.B., P.J.R., M.V., and L.E.W. prepared figures; R.D.D., S.B., P.J.R., M.V., and L.E.W. drafted manuscript; R.D.D., S.B., P.J.R., M.V., and L.E.W. edited and revised manuscript; R.D.D., S.B., P.J.R., M.V., and L.E.W. approved final version of manuscript. REFERENCES 1. Adams V, Linke A, Wisloff U, Doring C, Erbs S, Krankel N, Witt CC, Labeit S, Muller-Werdan U, Schuler G, Hambrecht R. Myocardial expression of Murf-1 and MAFbx after induction of chronic heart failure: Effect on myocardial contractility. Cardiovasc Res 73: , Ali MA, Fan X, Schulz R. Cardiac sarcomeric proteins: novel intracellular targets of matrix metalloproteinase-2 in heart disease. Trends Cardiovasc Med 21: , Argiles JM, Busquets S, Lopez-Soriano FJ. Anti-inflammatory therapies in cancer cachexia. Eur J Pharmacol 668, Suppl 1: S81 S86, Argiles JM, Busquets S, Lopez-Soriano FJ. The pivotal role of cytokines in muscle wasting during cancer. Int J Biochem Cell Biol 37: , 2005.
7 CANCER CACHEXIA AND MMPs H Aulino P, Berardi E, Cardillo VM, Rizzuto E, Perniconi B, Ramina C, Padula F, Spugnini EP, Baldi A, Faiola F, Adamo S, Coletti D. Molecular, cellular and physiological characterization of the cancer cachexia-inducing C26 colon carcinoma in mouse. BMC Cancer 10: 363, Bicer S, Reiser PJ. Myosin light chain 1 isoforms in slow fibers from global and orbital layers of canine rectus muscles. Invest Ophthalmol Vis Sci 45: , Blankenberg S, Rupprecht HJ, Poirier O, Bickel C, Smieja M, Hafner G, Meyer J, Cambien F, Tiret L, AtheroGene I. Plasma concentrations and genetic variation of matrix metalloproteinase 9 and prognosis of patients with cardiovascular disease. Circulation 107: , Blough ER, Rennie ER, Zhang F, Reiser PJ. Enhanced electrophoretic separation and resolution of myosin heavy chains in mammalian and avian skeletal muscles. Anal Biochem 233: 31 35, Carrasco FH, Montes GS, Krisztan RM, Shigihara KM, Carneiro J, Junqueira LC. Comparative morphologic and histochemical studies on the collagen of vertebrate arteries. Blood Vessels 18: , Cohen S, Brault JJ, Gygi SP, Glass DJ, Valenzuela DM, Gartner C, Latres E, Goldberg AL. During muscle atrophy, thick, but not thin, filament components are degraded by MuRF1-dependent ubiquitylation. J Cell Biol 185: , Deryugina EI, Quigley JP. Matrix metalloproteinases and tumor metastasis. Cancer Metastasis Rev 25: 9 34, Dewald O, Frangogiannis NG, Zoerlein M, Duerr GD, Klemm C, Knuefermann P, Taffet G, Michael LH, Crapo JD, Welz A, Entman ML. Development of murine ischemic cardiomyopathy is associated with a transient inflammatory reaction and depends on reactive oxygen species. Proc Natl Acad Sci USA 100: , DeWys WD. Nutritional care of the cancer patient. JAMA 244: , Fouladiun M, Korner U, Gunnebo L, Sixt-Ammilon P, Bosaeus I, Lundholm K. Daily physical-rest activities in relation to nutritional state, metabolism, and quality of life in cancer patients with progressive cachexia. Clin Cancer Res 13: , Fukushima K, Nakamura A, Ueda H, Yuasa K, Yoshida K, Takeda S, Ikeda S. Activation and localization of matrix metalloproteinase-2 and -9 in the skeletal muscle of the muscular dystrophy dog (CXMDJ). BMC Musculoskel Disord 8: 54, a.Halade GV, Jin YF, Lindsey ML. Matrix metalloproteinase (MMP)-9: a proximal biomarker for cardiac remodeling and a distal biomarker for inflammation. Pharmacol Ther 139: 32 40, He WA, Berardi E, Cardillo VM, Acharyya S, Aulino P, Thomas- Ahner J, Wang J, Bloomston M, Muscarella P, Nau P, Shah N, Butchbach ME, Ladner K, Adamo S, Rudnicki MA, Keller C, Coletti D, Montanaro F, Guttridge DC. NF- B-mediated Pax7 dysregulation in the muscle microenvironment promotes cancer cachexia. J Clin Invest 123: , Hnia K, Hugon G, Rivier F, Masmoudi A, Mercier J, Mornet D. Modulation of p38 mitogen-activated protein kinase cascade and metalloproteinase activity in diaphragm muscle in response to free radical scavenger administration in dystrophin-deficient Mdx mice. Am J Pathol 170: , Kedar V, McDonough H, Arya R, Li HH, Rockman HA, Patterson C. Muscle-specific RING finger 1 is a bona fide ubiquitin ligase that degrades cardiac troponin I. Proc Natl Acad Sci USA 101: , Khal J, Wyke SM, Russell ST, Hine AV, Tisdale MJ. Expression of the ubiquitin-proteasome pathway and muscle loss in experimental cancer cachexia. Br J Cancer 93: , Kherif S, Lafuma C, Dehaupas M, Lachkar S, Fournier JG, Verdiere- Sahuque M, Fardeau M, Alameddine HS. Expression of matrix metalloproteinases 2 and 9 in regenerating skeletal muscle: a study in experimentally injured and mdx muscles. Dev Biol 205: , Kumar A, Bhatnagar S, Kumar A. Matrix metalloproteinase inhibitor batimastat alleviates pathology and improves skeletal muscle function in dystrophin-deficient mdx mice. Am J Pathol 177: , Lazarus DD, Destree AT, Mazzola LM, McCormack TA, Dick LR, Xu B, Huang JQ, Pierce JW, Read MA, Coggins MB, Solomon V, Goldberg AL, Brand SJ, Elliott PJ. A new model of cancer cachexia: contribution of the ubiquitin-proteasome pathway. Am J Physiol Endocrinol Metab 277: E332 E341, Lemaitre V, D Armiento J. Matrix metalloproteinases in development and disease. Birth Defects Res C Embryo Today 78: 1 10, Mammucari C, Milan G, Romanello V, Masiero E, Rudolf R, Del Piccolo P, Burden SJ, Di Lisi R, Sandri C, Zhao J, Goldberg AL, Schiaffino S, Sandri M. FoxO3 controls autophagy in skeletal muscle in vivo. Cell Metab 6: , Murphy KT, Chee A, Trieu J, Naim T, Lynch GS. Importance of functional and metabolic impairments in the characterization of the C-26 murine model of cancer cachexia. Dis Model Mechan 5: , Nagase H, Visse R, Murphy G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res 69: , Ohtake Y, Tojo H, Seiki M. Multifunctional roles of MT1-MMP in myofiber formation and morphostatic maintenance of skeletal muscle. J Cell Sci 119: , Roberts BM, Ahn B, Smuder AJ, Al-Rajhi M, Gill LC, Beharry AW, Powers SK, Fuller DD, Ferreira LF, Judge AR. Diaphragm and ventilatory dysfunction during cancer cachexia. FASEB J 27: , Sandri M, Sandri C, Gilbert A, Skurk C, Calabria E, Picard A, Walsh K, Schiaffino S, Lecker SH, Goldberg AL. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117: , Shirakabe A, Asai K, Hata N, Yokoyama S, Shinada T, Kobayashi N, Mizuno K. Clinical significance of matrix metalloproteinase (MMP)-2 in patients with acute heart failure. Int Heart J 51: , Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, CA Cancer J Clin 64: 9 29, Springer J, Tschirner A, Haghikia A, von Haehling S, Lal H, Grzesiak A, Kaschina E, Palus S, Potsch M, von Websky K, Hocher B, Latouche C, Jaisser F, Morawietz L, Coats AJ, Beadle J, Argiles JM, Thum T, Foldes G, Doehner W, Hilfiker-Kleiner D, Force T, Anker SD. Prevention of liver cancer cachexia-induced cardiac wasting and heart failure. Eur Heart J 35: , Stedman HH, Sweeney HL, Shrager JB, Maguire HC, Panettieri RA, Petrof B, Narusawa M, Leferovich JM, Sladky JT, Kelly AM. The mdx mouse diaphragm reproduces the degenerative changes of Duchenne muscular dystrophy. Nature 352: , Tian M, Asp ML, Nishijima Y, Belury MA. Evidence for cardiac atrophic remodeling in cancer-induced cachexia in mice. Int J Oncol 39: , Tian M, Nishijima Y, Asp ML, Stout MB, Reiser PJ, Belury MA. Cardiac alterations in cancer-induced cachexia in mice. Int J Oncol 37: , Tisdale MJ. Cachexia in cancer patients. Nat Rev Cancer 2: , Tisdale MJ. Mechanisms of cancer cachexia. Physiol Rev 89: , Toth MJ, Miller MS, Callahan DM, Sweeny AP, Nunez I, Grunberg SM, Der-Torossian H, Couch ME, Dittus K. Molecular mechanisms underlying skeletal muscle weakness in human cancer: reduced myosinactin cross-bridge formation and kinetics. J Appl Physiol 114: , Trim JE, Samra SK, Arthur MJ, Wright MC, McAulay M, Beri R, Mann DA. Upstream tissue inhibitor of metalloproteinases-1 (TIMP-1) element-1, a novel and essential regulatory DNA motif in the human TIMP-1 gene promoter, directly interacts with a 30-kDa nuclear protein. J Biol Chem 275: , Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res 92: , Wang XS. Pathophysiology of cancer-related fatigue. Clin J Oncol Nurs 12: 11 20, White JP, Puppa MJ, Sato S, Gao S, Price RL, Baynes JW, Kostek MC, Matesic LE, Carson JA. IL-6 regulation on skeletal muscle mitochondrial remodeling during cancer cachexia in the ApcMin/ mouse. Skelet Muscle 2: 14, Xu H, Crawford D, Hutchinson KR, Youtz DJ, Lucchesi PA, Velten M, McCarthy DO, Wold LE. Myocardial dysfunction in an animal model of cancer cachexia. Life Sci 88: , Zhang L, Tang H, Kou Y, Li R, Zheng Y, Wang Q, Zhou X, Jin L. MG132-mediated inhibition of the ubiquitin-proteasome pathway ameliorates cancer cachexia. J Cancer Res Clin Oncol 139: , 2013.
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Modeling human cancer cachexia in colon 26 tumor-bearing adult mice
 DOI 10.1007/s13539-014-0141-2 ORIGINAL ARTICLE Modeling human cancer cachexia in colon 26 tumor-bearing adult mice Erin E. Talbert & Gregory A. Metzger & Wei A. He & Denis C. Guttridge Received: 23 December
DOI 10.1007/s13539-014-0141-2 ORIGINAL ARTICLE Modeling human cancer cachexia in colon 26 tumor-bearing adult mice Erin E. Talbert & Gregory A. Metzger & Wei A. He & Denis C. Guttridge Received: 23 December
Running Head: EFFECTS OF RESVERATROL ON CANCER CACHEXIA 1. Effects of resveratrol on cancer cachexia in a mouse model.
 Running Head: EFFECTS OF RESVERATROL ON CANCER CACHEXIA 1 Effects of resveratrol on cancer cachexia in a mouse model Jessica Mansfield The Ohio State University College of Nursing Running Head: EFFECTS
Running Head: EFFECTS OF RESVERATROL ON CANCER CACHEXIA 1 Effects of resveratrol on cancer cachexia in a mouse model Jessica Mansfield The Ohio State University College of Nursing Running Head: EFFECTS
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Cancer Related Fatigue Dan 1. The Effects that Non-steroidal Anti-inflammatory Drugs (NSAID) have on Cancer Related Fatigue in a Mouse Model
 Cancer Related Fatigue Dan 1 The Effects that Non-steroidal Anti-inflammatory Drugs (NSAID) have on Cancer Related Fatigue in a Mouse Model Devon N. Dan The Ohio State University College of Nursing Cancer
Cancer Related Fatigue Dan 1 The Effects that Non-steroidal Anti-inflammatory Drugs (NSAID) have on Cancer Related Fatigue in a Mouse Model Devon N. Dan The Ohio State University College of Nursing Cancer
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Intolerance in Heart Failure
 Novel Targets to Attack Exercise Intolerance in Heart Failure - Skeletal Muscle - Volker Adams, PhD ESC, Paris 3. Aug. 211 UNIVERSITÄT LEIPZIG H E R Z Z E N T R U M Nothing to disclose Myers et al. NEJM
Novel Targets to Attack Exercise Intolerance in Heart Failure - Skeletal Muscle - Volker Adams, PhD ESC, Paris 3. Aug. 211 UNIVERSITÄT LEIPZIG H E R Z Z E N T R U M Nothing to disclose Myers et al. NEJM
Serum cytokine levels in control and tumor-bearing male and female mice at day 15.
 Supplementary Table 1. Serum cytokine levels in control and tumor-bearing male and female mice at day 15. Male Female Cytokine Control C-26 Control C-26 IL-1β 2.0 ± 0.8 9.6 ± 1.5* 1.8 ± 0.2 6.8 ± 1.4*
Supplementary Table 1. Serum cytokine levels in control and tumor-bearing male and female mice at day 15. Male Female Cytokine Control C-26 Control C-26 IL-1β 2.0 ± 0.8 9.6 ± 1.5* 1.8 ± 0.2 6.8 ± 1.4*
OxiSelect HNE-His Adduct ELISA Kit
 Product Manual OxiSelect HNE-His Adduct ELISA Kit Catalog Number STA-334 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined mechanism
Product Manual OxiSelect HNE-His Adduct ELISA Kit Catalog Number STA-334 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined mechanism
1. Cardiomyocytes and nonmyocyte. 2. Extracellular Matrix 3. Vessels שאלה 1. Pathobiology of Heart Failure Molecular and Cellular Mechanism
 Pathobiology of Heart Failure Molecular and Cellular Mechanism Jonathan Leor Neufeld Cardiac Research Institute Tel-Aviv University Sheba Medical Center, Tel-Hashomer שאלה 1 התא הנפוץ ביותר (75%~) בלב
Pathobiology of Heart Failure Molecular and Cellular Mechanism Jonathan Leor Neufeld Cardiac Research Institute Tel-Aviv University Sheba Medical Center, Tel-Hashomer שאלה 1 התא הנפוץ ביותר (75%~) בלב
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
M.1.2_006. TREAT-NMD Activity A07: Accelerate preclinical phase of new therapeutic treatment developement
 TREAT-NMD Activity A07: Accelerate preclinical phase of new therapeutic treatment developement Work package 7.4: Develop standardised protocols and procedures for harmonising and accelerating pre-clinical
TREAT-NMD Activity A07: Accelerate preclinical phase of new therapeutic treatment developement Work package 7.4: Develop standardised protocols and procedures for harmonising and accelerating pre-clinical
Effect of a nutrient mixture on the localization of extracellular matrix proteins in HeLa human cervical cancer xenografts in female nude mice
 Effect of a nutrient mixture on the localization of extracellular matrix proteins in HeLa human cervical cancer xenografts in female nude mice Publication from the Dr. Rath Research Institute Experimental
Effect of a nutrient mixture on the localization of extracellular matrix proteins in HeLa human cervical cancer xenografts in female nude mice Publication from the Dr. Rath Research Institute Experimental
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Muscles, muscle fibres and myofibrils
 Muscles, muscle fibres and myofibrils Properties of Muscle Fiber Types Fast fibers Slow fibers Characteristic IIb IIx IIa Type I V max (speed of shortening) Highest Intermediate Low Resistance to fatigue
Muscles, muscle fibres and myofibrils Properties of Muscle Fiber Types Fast fibers Slow fibers Characteristic IIb IIx IIa Type I V max (speed of shortening) Highest Intermediate Low Resistance to fatigue
Running head: CALCIUM HANDLING PROTEINS IN THE HEART OF TUMOR
 Running head: CALCIUM HANDLING PROTEINS IN THE HEART OF TUMOR BEARING MICE CALCIUM HANDLING PROTEINS IN THE HEART OF TUMOR BEARING MICE Kara C. Brooks The Ohio State University College of Nursing CALCIUM
Running head: CALCIUM HANDLING PROTEINS IN THE HEART OF TUMOR BEARING MICE CALCIUM HANDLING PROTEINS IN THE HEART OF TUMOR BEARING MICE Kara C. Brooks The Ohio State University College of Nursing CALCIUM
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES
 ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES I. Satellite Cells A. Proliferative, myoblastic cells that lie in invaginations in the sarcolemma
ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES I. Satellite Cells A. Proliferative, myoblastic cells that lie in invaginations in the sarcolemma
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Ambient Temperature Stabilization of RNA derived from Jurkat, HeLa and HUVEC Cell Lines for Use in RT-qPCR Assays
 Ambient Temperature Stabilization of RNA derived from Jurkat, HeLa and HUVEC Cell Lines for Use in RT-qPCR Assays C. Litterst 1, H. Martinez, B. Iverson and R. Nuňez 1 Bio-Rad Laboratories, Life Science
Ambient Temperature Stabilization of RNA derived from Jurkat, HeLa and HUVEC Cell Lines for Use in RT-qPCR Assays C. Litterst 1, H. Martinez, B. Iverson and R. Nuňez 1 Bio-Rad Laboratories, Life Science
TITLE: Breast Tumor-Generated Type 1 Collagen Breakdown Fragments Act as Matrikines to Drive Osteolysis
 AD Award Number: W81XWH-08-1-0639 TITLE: Breast Tumor-Generated Type 1 Collagen Breakdown Fragments Act as Matrikines to Drive Osteolysis PRINCIPAL INVESTIGATOR: Ching Hua William Wu PhD. CONTRACTING ORGANIZATION:
AD Award Number: W81XWH-08-1-0639 TITLE: Breast Tumor-Generated Type 1 Collagen Breakdown Fragments Act as Matrikines to Drive Osteolysis PRINCIPAL INVESTIGATOR: Ching Hua William Wu PhD. CONTRACTING ORGANIZATION:
Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value
 Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value Shen Wei 1,a, Chen Juan 2, Li Xiurong 1 and Yin Jie 1 1 Department of Obstetrics and Gynecology,
Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value Shen Wei 1,a, Chen Juan 2, Li Xiurong 1 and Yin Jie 1 1 Department of Obstetrics and Gynecology,
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
SensoLyte Generic MMP Assay Kit *Colorimetric*
 SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury
 J Mol Med 2015 HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury Sara Karsikas; Mikko Myllymäki; Minna Heikkilä; Raija Sormunen; Kari I Kivirikko; Johanna Myllyharju; Raisa
J Mol Med 2015 HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury Sara Karsikas; Mikko Myllymäki; Minna Heikkilä; Raija Sormunen; Kari I Kivirikko; Johanna Myllyharju; Raisa
Chapter Skeletal Muscle Structure and Function
 Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
9/16/2009. Fast and slow twitch fibres. Properties of Muscle Fiber Types Fast fibers Slow fibers
 Muscles, muscle fibres and myofibrils Fast and slow twitch fibres Rat hindlimb muscle ATPase staining at different ph and NADH Muscle fibre shortening velocity lengths/second Properties of Muscle Fiber
Muscles, muscle fibres and myofibrils Fast and slow twitch fibres Rat hindlimb muscle ATPase staining at different ph and NADH Muscle fibre shortening velocity lengths/second Properties of Muscle Fiber
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Tissue repair. (3&4 of 4)
 Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002
![Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002](/thumbs/79/80181612.jpg) Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Supplementary material: Materials and suppliers
 Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Skeletal muscle in the light of its structure
 Mechanism of contraction of Skeletal muscle in the light of its structure By Dr. Mudassar Ali Roomi (MBBS, M. Phil) Muscle Tissue Skeletal Muscle Cardiac Muscle Smooth Muscle Skeletal Muscle Long cylindrical
Mechanism of contraction of Skeletal muscle in the light of its structure By Dr. Mudassar Ali Roomi (MBBS, M. Phil) Muscle Tissue Skeletal Muscle Cardiac Muscle Smooth Muscle Skeletal Muscle Long cylindrical
Warm Up! Test review (already! ;))
 Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Human Leptin ELISA Kit
 Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric*
 SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
Advances in Computer Science Research, volume 59 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016)
 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC-
 Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
The ubiquitin proteasome system in cardiovascular disease Basic mechanisms
 ECS Congress 2010 Stockholm, Sweden 28 Aug 2010 01 Sep 2010 The ubiquitin proteasome system in cardiovascular disease Basic mechanisms Saskia Schlossarek Department of Experimental and Clinical Pharmacology
ECS Congress 2010 Stockholm, Sweden 28 Aug 2010 01 Sep 2010 The ubiquitin proteasome system in cardiovascular disease Basic mechanisms Saskia Schlossarek Department of Experimental and Clinical Pharmacology
International Graduate Research Programme in Cardiovascular Science
 1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
TECHNICAL BULLETIN. Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates
 Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates Catalog Number RAB0011 Storage Temperature 20 C TECHNICAL BULLETIN Product Description
Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates Catalog Number RAB0011 Storage Temperature 20 C TECHNICAL BULLETIN Product Description
Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive
 Online Data Supplement: Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma Dan Cheng, Zheng Xue, Lingling Yi, Huimin Shi, Kan Zhang, Xiaorong Huo, Luke R. Bonser,
Online Data Supplement: Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma Dan Cheng, Zheng Xue, Lingling Yi, Huimin Shi, Kan Zhang, Xiaorong Huo, Luke R. Bonser,
Myocardial lipid accumulation and lipotoxicity in heart failure
 Myocardial lipid accumulation and lipotoxicity in heart failure P. Christian Schulze, MD, PhD New York - Presbyterian Hospital, Columbia University Medical Center, Division of Cardiology, New York, NY,
Myocardial lipid accumulation and lipotoxicity in heart failure P. Christian Schulze, MD, PhD New York - Presbyterian Hospital, Columbia University Medical Center, Division of Cardiology, New York, NY,
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
Expression and clinical significance of ADAM17 protein in esophageal squamous cell carcinoma
 Expression and clinical significance of ADAM17 protein in esophageal squamous cell carcinoma H.B. Liu, Y. Zhu, Q.C. Yang, Y. Shen, X.J. Zhang and H. Chen Department of Pathology First People s Hospital
Expression and clinical significance of ADAM17 protein in esophageal squamous cell carcinoma H.B. Liu, Y. Zhu, Q.C. Yang, Y. Shen, X.J. Zhang and H. Chen Department of Pathology First People s Hospital
Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy
 (Courtesy of Magda Stumpfova. Used with permission.) Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy M.D. Gomes, S.H. Lecker, R.T. Jagoe, A. Navon, and A.L. Goldberg PNAS,
(Courtesy of Magda Stumpfova. Used with permission.) Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy M.D. Gomes, S.H. Lecker, R.T. Jagoe, A. Navon, and A.L. Goldberg PNAS,
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
About This Chapter. Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Pearson Education, Inc.
 About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
removed replaced inflammation scar tissue
 HOMEOSTASIS Normal maintenance and renewal of differentiated cells in many tissues This does NOT involve leukocytes. Leukocytes and inflammation occurs in response to damage NEED FOR REPAIR When tissue
HOMEOSTASIS Normal maintenance and renewal of differentiated cells in many tissues This does NOT involve leukocytes. Leukocytes and inflammation occurs in response to damage NEED FOR REPAIR When tissue
Pathophysiology of heart failure with preserved ejection fraction. Extracellular matrix
 Pathophysiology of heart failure with preserved ejection fraction Extracellular matrix Javier Díez, MD, PhD. Full Professor of Cardiovascular Medicine and Director Division of Cardiovascular Sciences Centre
Pathophysiology of heart failure with preserved ejection fraction Extracellular matrix Javier Díez, MD, PhD. Full Professor of Cardiovascular Medicine and Director Division of Cardiovascular Sciences Centre
The Cardiovascular System and Aging- Is it Built to Fail?
 The Cardiovascular System and Aging- Is it Built to Fail? Francis G. Spinale, MD, PhD Professor of Surgery and Cell Biology and Anatomy University of South Carolina School of Medicine Veterans Affairs
The Cardiovascular System and Aging- Is it Built to Fail? Francis G. Spinale, MD, PhD Professor of Surgery and Cell Biology and Anatomy University of South Carolina School of Medicine Veterans Affairs
Anti-inflammatory properties of SM04690, a small molecule inhibitor of the Wnt pathway as a potential treatment for knee osteoarthritis
 Anti-inflammatory properties of SM04690, a small molecule inhibitor of the Wnt pathway as a potential treatment for knee osteoarthritis V. Deshmukh 1, T. Seo 1, C. Swearingen 1, Y. Yazici 1 1 Samumed,
Anti-inflammatory properties of SM04690, a small molecule inhibitor of the Wnt pathway as a potential treatment for knee osteoarthritis V. Deshmukh 1, T. Seo 1, C. Swearingen 1, Y. Yazici 1 1 Samumed,
Supplementary Figure 1
 VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS. As. MARUSHCHAK M.I.
 BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS As. MARUSHCHAK M.I. Heart attack symptoms Acute MI Measurement of cardiac enzyme levels Measure cardiac enzyme levels at regular
BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS As. MARUSHCHAK M.I. Heart attack symptoms Acute MI Measurement of cardiac enzyme levels Measure cardiac enzyme levels at regular
Skeletal Muscle and the Molecular Basis of Contraction. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
OxiSelect MDA Adduct ELISA Kit
 Revised Protocol Product Manual OxiSelect MDA Adduct ELISA Kit Catalog Number STA-332 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
Revised Protocol Product Manual OxiSelect MDA Adduct ELISA Kit Catalog Number STA-332 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
In Vivo Animal Models of Heart Disease. Why Animal Models of Disease? Timothy A Hacker, PhD Department of Medicine University of Wisconsin-Madison
 In Vivo Animal Models of Heart Disease Timothy A Hacker, PhD Department of Medicine University of Wisconsin-Madison Why Animal Models of Disease? Heart Failure (HF) Leading cause of morbidity and mortality
In Vivo Animal Models of Heart Disease Timothy A Hacker, PhD Department of Medicine University of Wisconsin-Madison Why Animal Models of Disease? Heart Failure (HF) Leading cause of morbidity and mortality
Total Histone H3 Acetylation Detection Fast Kit (Colorimetric)
 Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Catalog Number KA1538 48 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use...
Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Catalog Number KA1538 48 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use...
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS
 STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer
 Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation)
 Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
EpiQuik Total Histone H3 Acetylation Detection Fast Kit (Colorimetric)
 EpiQuik Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Total Histone H3 Acetylation Detection Fast Kit (Colorimetric)
EpiQuik Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Total Histone H3 Acetylation Detection Fast Kit (Colorimetric)
TECHNICAL BULLETIN. Catalog Number RAB0447 Storage Temperature 20 C
 Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
EPIGENTEK. EpiQuik Global Acetyl Histone H3K27 Quantification Kit (Colorimetric) Base Catalog # P-4059 PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE
 EpiQuik Global Acetyl Histone H3K27 Quantification Kit (Colorimetric) Base Catalog # P-4059 PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Acetyl Histone H3K27 Quantification Kit (Colorimetric)
EpiQuik Global Acetyl Histone H3K27 Quantification Kit (Colorimetric) Base Catalog # P-4059 PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Acetyl Histone H3K27 Quantification Kit (Colorimetric)
Collagenase Assay Kit
 Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
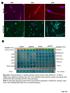 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Differentiation-induced Changes of Mediterranean Fever Gene (MEFV) Expression in HL-60 Cell
 Differentiation-induced Changes of Mediterranean Fever Gene (MEFV) Expression in HL-60 Cell Wenxin Li Department of Biological Sciences Fordham University Abstract MEFV is a human gene that codes for an
Differentiation-induced Changes of Mediterranean Fever Gene (MEFV) Expression in HL-60 Cell Wenxin Li Department of Biological Sciences Fordham University Abstract MEFV is a human gene that codes for an
Matrix metalloproteinases
 Matrix metalloproteinases,, 1. 2. 2-1. Domain 2-2. TIMPs 2-3. MMPs, 3. 4. 1. Collagens, proteoglycans (Extracellular matrix, ECM),,. matrixins matrix metalloproteinases (MMPs) [1, 2]. MMPs,, cytokines
Matrix metalloproteinases,, 1. 2. 2-1. Domain 2-2. TIMPs 2-3. MMPs, 3. 4. 1. Collagens, proteoglycans (Extracellular matrix, ECM),,. matrixins matrix metalloproteinases (MMPs) [1, 2]. MMPs,, cytokines
The Regulation of Skeletal Muscle Protein Turnover During the Progression of Cancer Cachexia in the Apc Min/+ Mouse
 University of South Carolina Scholar Commons Faculty Publications Chemistry and Biochemistry, Department of 9-19-2011 The Regulation of Skeletal Muscle Protein Turnover During the Progression of Cancer
University of South Carolina Scholar Commons Faculty Publications Chemistry and Biochemistry, Department of 9-19-2011 The Regulation of Skeletal Muscle Protein Turnover During the Progression of Cancer
Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary. y Antigens A. antibodies
 Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University
 1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University (Reference NO. 2015-003). 96 Kunming (KM) mice (8 weeks;
1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University (Reference NO. 2015-003). 96 Kunming (KM) mice (8 weeks;
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair
 Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Product Manual. Human LDLR ELISA Kit. Catalog Number. FOR RESEARCH USE ONLY Not for use in diagnostic procedures
 Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle.
 Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
In vitro functional study of Cardiomyocyte
 In vitro functional study of Cardiomyocyte Gwang Hyeon Eom Department of Pharmacology and Medical Research Center for Gene Regulation Chonnam National University Medical School, Gwangju, South Korea Heart
In vitro functional study of Cardiomyocyte Gwang Hyeon Eom Department of Pharmacology and Medical Research Center for Gene Regulation Chonnam National University Medical School, Gwangju, South Korea Heart
Human Troponin I ELISA Kit
 Human Troponin I ELISA Kit Cat. No.:DEIA2358 Pkg.Size:96T Intended use This kit is intended for the quantitative determination of cardiac troponin I in human serum. Measurement of troponin I values are
Human Troponin I ELISA Kit Cat. No.:DEIA2358 Pkg.Size:96T Intended use This kit is intended for the quantitative determination of cardiac troponin I in human serum. Measurement of troponin I values are
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL Mitochondrial Extraction Kit NBP2-29448 Research use only. Not for diagnostic or therapeutic procedures www.novusbio.com P: 303.760.1950 P: 888.506.6887 F: 303.730.1966 technical@novusbio.com
PRODUCT INFORMATION & MANUAL Mitochondrial Extraction Kit NBP2-29448 Research use only. Not for diagnostic or therapeutic procedures www.novusbio.com P: 303.760.1950 P: 888.506.6887 F: 303.730.1966 technical@novusbio.com
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL Nuclear Extraction Kit NBP2-29447 Research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 888.506.6887 - technical@novusbio.com Novus kits are
PRODUCT INFORMATION & MANUAL Nuclear Extraction Kit NBP2-29447 Research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 888.506.6887 - technical@novusbio.com Novus kits are
Ernährung 2006 International Cachexia Workshop Berlin, June 2006
 Activation Peptides ATP E1 ATP Ubiquitin Proteolysis E2 Proteín Proteasome 26S E2 E3 Conjugation Ernährung 2006 International Cachexia Workshop Berlin, June 2006 Antiproteolytic strategies Prof. Dr. Josep
Activation Peptides ATP E1 ATP Ubiquitin Proteolysis E2 Proteín Proteasome 26S E2 E3 Conjugation Ernährung 2006 International Cachexia Workshop Berlin, June 2006 Antiproteolytic strategies Prof. Dr. Josep
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic
 Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall
 Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
Mammalian Tissue Protein Extraction Reagent
 Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
Exercise in Adverse Cardiac Remodeling: of Mice and Men
 Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002
![Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002](/thumbs/79/80181511.jpg) Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Mouse C-peptide EIA. Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY
 Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
c Ischemia (30 min) Reperfusion (8 w) Supplementary Figure bp 300 bp Ischemia (30 min) Reperfusion (4 h) Dox 20 mg/kg i.p.
 a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
