Honokiol synergizes chemotherapy drugs in multidrug resistant breast cancer cells via enhanced apoptosis and additional programmed necrotic death
|
|
|
- William Fleming
- 6 years ago
- Views:
Transcription
1 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , 2013 Honokiol synergizes chemotherapy drugs in multidrug resistant breast cancer cells via enhanced apoptosis and additional programmed necrotic death WEI TIAN 1,2*, YONGCHUAN DENG 1,2*, LING LI 3, HAIFEI HE 1,2, JIE SUN 4 and DONG XU 1,2 1 Department of Surgical Oncology, 2 Cancer Institute (The Key Laboratory of Cancer Prevention and Intervention, China National Ministry of Education), The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang , P.R. China; 3 Division of Stem Cell and Leukemia Research, City of Hope National Medical Center, City of Hope, Duarte, CA 91010, USA; 4 Zhejiang Youth College, Hangzhou, Zhejiang , P.R. China Received September 3, 2012; Accepted October 29, 2012 DOI: /ijo Abstract. Multidrug resistance (MDR) is a major challenge in cancer therapy. Apoptosis tolerance is one of the key mechanisms of MDR. Honokiol, a small-molecule pharmacologically active component, exhibits competent cytotoxicity in a variety of human cancer cells through apoptosis and other forms of programmed cell death (such as programmed necrosis). Although much work has been done on its antitumor effects, little attention has been paid on systemic evaluation of efficacy of honokiol combined with other chemotherapeutic agents, especially in drug resistant cell lines. Here, we systematically and quantitatively assess its combinational effect with different chemotherapeutic agents using the combination index (CI) equation. We found that honokiol synergized with chemotherapeutic agents both in sensitive and resistant, solid and non-solid (MCF-7, HL-60, MCF-7/ADR and HL-60/ADR) cell lines. Honokiol (40 µg/ml) induced necrotic cell death in MCF-7/ADR cells with characterized morphological and biochemical features. Co-incubation with honokiol and etoposide (VP-16) activated a complex death modality, which was composed of necrotic cell death and apoptosis. This dual-death pathway was shut down when pretreated with pan-caspase inhibitor (z-vad-fmk) and cyclophilin D inhibitor (cyclosporin A). Western blot analysis results proved that honokiol also enhanced VP-16-induced apoptosis potentially via blocking nuclear factor κb (NF-κB) activation. Our data for the first time quantitatively demonstrate that honokiol synergizes frequently-used chemotherapeutic agents via enhanced apoptosis and additional programmed Correspondence to: Dr Dong Xu, Department of Surgical Oncology, The Second Affiliated Hospital, School of Medicine, Zhejiang University, no. 88 Jiefang Road, Hangzhou, Zhejiang , P.R. China xudongzju@zju.edu.cn * Contributed equally Key words: honokiol, breast cancer, multidrug drug resistance, apoptosis, programmed necrotic death necrotic death. These findings indicate a promising way to circumvent MDR and apoptosis tolerance. Introduction Multidrug drug resistance (MDR) is defined as concomitant existing resistance to several structurally and functionally diverse drugs (1-4). It is a very complicated and dynamical system composed of diverse cellular factors and signal transduction pathways. Major participants of MDR include drug transporter-mediated increased efflux of anticancer agents, evasion of drug-induced apoptosis and others (3,5). Most current chemotherapeutic drugs are substrates or inducers of drug transporters and induce cytotoxicity through apoptosis (1). Basing on this concept, recent research mainly focused on exploring and developing drugs reversing transport-mediated MDR and reactivating apoptosis. But most of these drugs failed to show clinical efficacy (6,7). Since MDR is modulated by a very complicated system, simply inhibiting one or two signal pathways or regulators can not succeed in overcoming it (1). Beside classical type of programmed cell death (apoptosis), other programmed death modalities exist, such as autophagy and programmed necrotic death (necroptosis) (8). Gradually increasing evidence demonstrates cancer cells resistant to current chemotherapeutic drugs are sensitive to drug-induced programmed necrotic death (9-11). Accordingly, contemporaneously activating multiple types of programmed cell death by multi-drug treatment regimen is a promising option to circumvent MDR, especially apoptosis tolerance. Honokiol (HNK) is a small molecule isolated from Chinese traditional medicinal herb Houpu with various pharmacological activities. Among all its medicinal applications, antitumor activity is most extensively studied. Honokiol-induced apoptosis, including caspase-dependent and -independent apoptosis (12), is the most widely investigated and fundamental mechanism of its antitumor activity. Besides, other cell death forms, such as programmed necrotic cell death (9) and paraptosis (13), are also induced by honokiol at certain doses. Our group is devoted to mechanical research on antitumor effects of honokiol. Previous work of our group demonstrated that honokiol induced apop-
2 722 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS tosis in human colorectal carcinoma RKO cells associated with caspase cascade (14,15). Besides, we evaluated honokiol potentiated cytotoxic effect of adriamycin through downregulation of P-gycoprotein (P-gp) in drug resistant breast carcinoma cell line MCF-7/ADR (2). Further, our subsequent study first reported that honokiol induced a distinct programmed cell death modality, programmed necrotic death (9), initiating new insights and greatly expanding the cognition of its antitumor activity. Most of the therapy studies concerning combination effect of two or more anticancer drugs only focus on therapeutic synergy. They lack quantitative data perhaps of composed of pure synergism, additive effect or even moderate antagonism. Although literature is available on antitumor effects of honokiol, little attention has been paid on systemic and quantitative evaluation of efficacy combining honokiol with chemotherapeutic agents, especially in drug resistant cell lines. We assessed the possible combination effects between honokiol and other chemotherapeutic agents and explored optimal synergistic drug combinations through a quantitative analysis method (16). Mechanisms of the synergistic activation are mainly evaluated in the present study. Materials and methods Reagents and chemicals. Honokiol was purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China) with >99% purity. Honokiol power was dissolved in dimethyl sulfoxide (DMSO) at 20 mg/ ml solution and stored at -20 C. RPMI 1640, fetal bovine serum (FBS), and 0.25% trypsin were purchased from Gibco BRL (Invitrogen Co., Grand Island, NY, USA). Cyclosporin A (CsA), etoposide (VP-16) and MTT power were purchased from Sigma Aldrich (St. Louis, MO, USA). Doxorubicin (ADM) and daunorubicin (DNR) were purchased from Pfizer (Actavis Italy S.p.A., Nerviano, MI, Italy). Paclitaxel (taxol) was purchased from Bristol-Myers Squibb (Princeton, NJ, USA). Pan-caspase inhibitor z-vad-fmk was from Beyotime Institute of Biotechnology (Shanghai, China). Annexin V-FITC/PI kit was purchased from Sigma Aldrich. Apotosis DNA Ladder Detection Kit was from KeyGen Biotech (Nanjing, China). Cell culture. Human breast carcinoma cell line MCF-7 and acute promyelocytic leukemia cell line HL-60 were maintained in RPMI 1640 supplemented with 10% FBS. Parallel multidrug resistant cell lines MCF-7/ADR and HL-60/ADR were developed as reported previously (2) and maintained in RPMI 1640 with 1 µg/ml and 100 ng/ml ADM, respectively. Various cells were grown in a humidified CO 2 incubator at 37 C and maintained in exponential growth. Evaluation of drug combination index. Cells were plated in 96-well micro-titer plates of 4,000-8,000 cells/well. After incubation overnight, cells were incubated with drug-free medium, honokiol or chemotherapeutic agent alone or combination of honokiol and chemotherapeutic agent with different rational concentrations. DMSO according to the highest drug concentration served as control. Quadruple wells were prepared for each drug concentration. After 48 h, MTT assays were performed as previously described (2). Viabilities were normalized to untreated samples. Combination index determinations were performed basing on the method of Chou and Talalay (16). Various cytotoxicity levels of the component drugs and combinations were calculated according to equation 1. Combination index values were calculated using equation 2 at each level of cytotoxicity. D 1-FA = D x [FA/(1-FA)] 1/m [1] Combination index (CI) = (D) 1 /(D 1-FA ) 1 + (D) 2 /(D 1-FA ) 2 + α(d) 1 (D) 2 /(D 1-FA ) 1 (D 1-FA ) 2 [2] In the above equations, m (stood for sigmoidity) and D x (is the IC 50 for 50% growth inhibition) were calculated by transforming concentration-effect data to a logarithmic scale. FA stood for the growth-inhibitory effect of each drug. (D) 1 and (D) 2 represented the doses of the combination required to produce survival (1-FA), while (D 1-FA ) 1 and (D 1-FA ) 2 stood for the concentrations of the component drugs required to produce survival (1-FA). α=1 for mutually nonexclusive drugs (17). In analysis, degree of combination effect was determined from the combination index (CI) as follows: CI>1, antagonism; CI=1, additivity; CI<1, synergism. Evaluation of dose reduction index (DRI). Two concentrations of honokiol were chosen for each cell line in the evaluation of DRI, which would produce 90-95% (HNK 1 ) and 70-80% (HNK 2 ) cell survival, respectively. The exact doses of HNK 1 and HNK 2 for each cell line are shown in Table III. After incubation overnight, cells plated in 96-well plates were treated with different chemotherapeutic agents in concentrationgradient combined with either blank control, HNK 1 or HNK 2. After 48 h, MTT assays were performed. IC 50 was calculated by transforming the concentration-effect data to a logarithmic scale. DRI values were determined using equation 3. (IC 50 ) 1 and (IC 50 ) 2 stood for IC 50 of a chemotherapeutic agent without or with honokiol, respectively. Dose Reduction Index (DRI) = (IC 50 ) 1 /(IC 50 ) 2 [3] Cell viability assay. MCF-7/ADR cells were treated with 40 µg/ml honokiol, 100 µg/ml VP-16, or combination, respectively. After 6 h incubation, MTT assay was performed to determine cell viability. Flow cytometric analysis for apoptosis. MCF-7/ADR cells were cultured in 6-well culture plates at 8x10 5 /well in 2 ml media for 24 h and treated with 0.1% DMSO (vehicle control), 40 µg/ml honokiol, 100 µg/ml VP-16, or combination for 6 h. Apoptotic rates were analyzed by flow cytometry using Annexin V-FITC/PI kit according to the manufacturer's instructions. The differentiation among early apoptotic cells, necrotic cells and viable cells were detected as follows: Annexin V positive and PI negative; Annexin V positive and PI positive; and Annexin V negative and PI negative, according to a previous report (9). The experiments were done in triplicate and similar results were obtained in three different experimental setups. Electron microscopy detection of cell death. Cells were seeded and treated as described above for flow cytometric analysis for apoptosis. The exact procedure was processed according to previously reported methods (9).
3 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , Table I. CI values in sensitive and resistant breast cancer cell lines. MCF-7/ADR MCF CI 50 CI 75 CI 85 CI 50 CI 75 CI 85 ADM+HNK VP-16+HNK TAX+HNK CI 50, CI 75 and CI 85 refer to CI values at IC 50, IC 75 and IC 85, respectively. CI>1, antagonism; CI=1, additivity; CI<1, synergism. Table II. CI values in sensitive and resistant leukemia cell lines. HL-60/ADR HL CI 50 CI 75 CI 85 CI 50 CI 75 CI 85 DNR+HNK CI 50, CI 75 and CI 85 refer to CI values at IC 50, IC 75 and IC 85, respectively. CI>1, antagonism; CI=1, additivity; CI<1, synergism. DNA ladder detection. Cells were seeded and treated as indicated above. After treatment, cells were collected and washed twice with chilled PBS. Afterwards, DNA gel electrophoresis was carried out after DNA extraction according to the manufacturer's instructions of DNA Ladder Detection kit. Experimental conditions were tested in triplicate and similar results were obtained at least three times. Western blot analysis for NF-κB translocation detection. For detection of nuclear factor κb (NF-κB) proteins, cells were treated with 0.1% DMSO (vehicle control), 40 µg/ml honokiol, 100 µg/ml VP-16, or combination, respectively, for 2 h. Cells were harvested for isolation of nuclear and cytoplasm extracts using Nuclear Extract Kit (Activ Motif, Carlsbad, CA, USA). The concentration of each protein was then determined by the standard BCA protein assay. About 40 µg of each sample was loaded in gel for immunoblot assay according to standard protocol. After cell lysate preparation, proteins were applied to a 10% SDS-PAGE and electro-transferred to a nitrocellulose membrane. Blots were probed by the proper primary antibodies (mouse monoclonal anti-human NF-κB and rabbit polyclonal anti-human lamin-b and α-tubulin (Cell Signaling Technology Inc., Danvers, MA, USA) overnight at 4 C and then exposed to secondary antibodies (HRP-conjugated anti-rabbit IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 2 h. Visualization was finally detected by chemiluminescence using ECL Plus Western Blotting Substrate (Thermo Fisher Scientific, Rockford, IL, USA). Lamin B and α-tubulin was used to ensure equal loading of protein on the gel. Densitometry was performed to quantify the relative expression levels of NF-κB protein. Statistical analysis. The data represent mean ± SD from three independent experiments. Student's t-test was used for comparison between in vitro responses to different drugs. All analyses were two-tailed. Results Combination effects of honokiol with different chemotherapeutic agents at rational combination doses in different cell lines. We used multiple drug effect analysis method of Chou and Talalay (16) to determine the nature of interaction between honokiol and various chemotherapeutic agents. Four different anticancer drugs (etoposide, doxorubicine, taxol and daunorubicin), commonly used in the treatment of breast cancer and leukemia were selected in the present study. Human breast carcinoma cell line MCF-7, acute promyelocytic leukemia cell line HL-60 and parallel multidrug resistant cell lines MCF-7/ADR, HL-60/ADR were treated with serial dilutions of honokiol and various chemotherapeutic agents at fixed ratios spanning the IC 50 of each drug. Combination index values at different dose-effect levels were calculated on the basis of parameters derived from effect plots of honokiol alone, the chemotherapeutic agent alone and the combination of the two. CI<1, CI=1 and CI>1 indicate synergism, additivity and antagonism effect, respectively. Honokiol synergistically enhanced cytotoxicity of ADM and VP-16 in MCF-7/ADR as the CI values were <1.0 at all dose-effect levels (50, 75 and 85% inhibition rate) (Table I, Fig. 1). CI values for MCF-7/ADR at IC 50 and IC 75 were <1.0 for taxol indicating a synergistic interaction, but CI 85 indicated antagonism (Table I, Fig. 1). The interaction for MCF-7 was synergism between honokiol and ADM at all dose-effect levels, whereas antagonism between honokiol and VP-16. It was synergism between honokiol and taxol for MCF-7 at IC 50 and IC 75 but antagonism at IC 85 (Table I, Fig. 1). Honokiol and daunorubicin (DNR) had synergistic interactions against HL-60 and HL-60/ ADR (Table II, Fig. 1). To compare combination effects among different groups, we calculated the mean CI values [mean CI = (CI 50 + CI 75 + CI 85 )/3] (Fig. 1B). Honokiol synergized most chemotherapeutic agents both in P-gp (MCF-7/ADR) and MRP1 (HL-60/ADR) medi-
4 724 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS Table III. Stated doses of honokiol for evaluation of dose reduction index. HNK 1 (µg/ml) HNK 2 (µg/ml) HL HL-60/ADR 4 5 MCF MCF-7/ADR The two concentrations of honokiol were chosen for each cell line in the evaluation of DRI, which would produce 90-95% (HNK 1 ) and 70-80% (HNK 2 ) cells survival, respectively. Figure 1. Combination effects of honokiol with different chemotherapeutic agents at rational combination doses in different cell lines. After treatment for 48 h, we evaluated (A) the combination index of honokiol with each chemotherapeutic agent at 50, 75 and 85% inhibitory levels, respectively and (B) their average combination index. Degree of combination effect was determined as follows: CI>1, antagonism; CI=1, additivity; CI<1, synergism. ated MDR cancer cells. Surprisingly, the most marked mean CI values existed in honokiol plus VP-16 combination in MCF-7/ ADR and parallel cell line MCF-7, which indicated conflicting effects. Dose reduction of chemotherapeutic drugs after combined treatment with honokiol. DRI parameters represent the order of magnitude of dose reduction which is determined from the relative proportions between the dose of combination and each drug alone at 50% inhibitory effect. Cell viabilities of each cancer cell line after treatments of the two given doses of honokiol (HNK 1 and HNK 2, exact doses are shown in Table III) alone were and 70-80%, respectively. On the other hand, HNK 1 and HNK 2 do no harm to human normal cells, such as normal PBMC (18). Dose-response curves demonstrated dose-dependent growth-inhibition by a single chemotherapeutic agent or combined with a given dose of Honokiol (HNK 1 or HNK 2 ) in all cell lines (Fig. 2). IC 50 was calculated by transforming the concentration-effect data to a logarithmic scale. Based on the IC 50 s, DRI for each chemotherapeutic agent was determined using equation 3. Conspicuous DRI values from 1.85 to were observed in the combination setting compared with chemotherapeutic agents alone except that of MCF-7 treated with VP-16 (Table IV). A dose of 12.5 µg/ml honokiol had no synergistic effect in combination with VP-16 in MCF-7 cell line as the DRI value was (Table IVD). The given doses of honokiol had greater dose-reducing effects in settings of VP-16 treatment in MCF-7/ADR than in MCF-7, and of DNR in HL-60/ADR than in HL-60 (Table IVB and D). Above results indicated that in most combination groups calculated DRI values were >1, whose trendency coincided well with CI values. Conspicuously, the most prominent DRI (17.32) was measured in honokiol plus VP-16 combination in MCF-7/ADR cells, which matched exactly to the lowest CI arisen group (as shown in the section: Combination effects of honokiol with different chemotherapeutic agents at rational combination doses in different cell lines). Accordingly, honokiol plus VP-16 combination treatment in MCF-7/ADR was chosen as representative in assessing synergetic mechanism. Cytotoxicity of honokiol plus VP-16 at highest rational doses towards multidrug resistant MCF-7/ADR cells. Based on the above results, we further tested the potential synergistic effect of honokiol on VP-16 induced MCF-7/ADR cell death at highest rational doses. Concentration of honokiol used in the following experiment was 40 µg/ml, and corresponding rational concentration of VP-16 was 100 µg/ml. After 6 h incubation, cell viabilities after 100 µg/ml VP-16, 40 µg/ ml honokiol, or combination treatment were 82.33±3.73, 56.21±2.97 and 48.18±1.38%, respectively. VP-16 alone only slightly affected cell viability of MCF-7/ADR. Honokiol plus VP-16 had dramatically enhanced cell toxicity compared to that of VP-16 or honokiol alone (P<0.005) (Fig. 3). Honokiol induces programmed necrotic cell death in MCF-7/ADR cells, which is reversed by cyclosporin A. Honokiol (40 µg/ml) induces programmed cell death with necrotic characteristics in MCF-7 cells as we reported previously (9). We hypothesized honokiol at the same concentration could primarily induce programmed necrotic
5 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , Figure 2. Cooperative effects of honokiol at given dose with different chemotherapeutic agents in concentration-gradients. The exact concentrations of honokiol in each cell line are shown in Table I. Cell viabilities were analyzed by MTT assays. These results were representative of three independent experiments. The values are shown as mean ± SD.
6 726 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS Table IV. DRI values for combinations of given dose of honokiol with different chemotherapeutic agents. A, IC 50 (µg/ml) DRI ADM alone Plus HNK 1 Plus HNK 2 Plus HNK 1 Plus HNK 2 MCF MCF-7/ADR B, IC 50 (µg/ml) DRI VP-16 alone Plus HNK 1 Plus HNK 2 Plus HNK 1 Plus HNK 2 MCF MCF-7/ADR C, IC 50 (µg/ml) DRI Taxol alone Plus HNK 1 Plus HNK 2 Plus HNK 1 Plus HNK 2 MCF MCF-7/ADR D, IC 50 (µg/ml) DRI DNR alone Plus HNK 1 Plus HNK 2 Plus HNK 1 Plus HNK 2 HL HL-60/ADR cell death in the parallel drug resistant cell line MCF-7/ADR. This hypothesis was supported by results of DNA ladder detection as only clumping degradation of nuclear DNA was detected after 6 h incubation with 40 µg/ml honokiol (Fig. 4). Compared with normal cells (Fig. 5A) and 100 µg/ml VP-16-treated cells (Fig. 5B), electron microscopy morphological manifestations revealed majority of cells underwent necrotic cell death characterized manifestations of increased cell volume due to swelling of cytoplasmic organelles, dilatation of mitochondria and endoplasmic reticulum, vacuoles and nuclear modifications (Fig. 5C). Not surprisingly, flow cytometry demonstrated that honokiol alone induced a dominant death characterized by loss of plasma membrane Figure 3. Cytotoxicity of honokiol plus VP-16 at highest rational doses towards MCF 7/ADR cells. After treatment with honokiol, VP-16 or combination of drugs for 6 h, cell viabilities were analyzed by MTT assays. Cytotoxicity induced by combination drugs was enhanced compared with each single drug. These results were representative of three independent experiments. The values shown are the mean ± SD. * P<0.001 vs. VP-16- or honokiol-treated cells.
7 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , Figure 4. MCF-7/ADR cells treated with a combination of honokiol and VP-16 demonstrate clumping degradation of nuclear DNA. Internucleosomal DNA fragmentation was identified by DNA gel electrophoresis in MCF-7/ADR cells treated with honokiol 40 µg/ml (C) or combination drugs (D), respectively. No clear DNA ladders were detected in these two groups. Both C and D groups manifested clumping degradation of nuclear DNA. Blank control was used as negative control (B); HL-60 treated with VP µg/ml, as positive control (E). DNA ladder 2000 was used as a marker (A). These results are representative of three independent experiments. integrity (PI-positive), 39.07% of cells were PI-positive (upper quadrants) and 6.16% were Annexin V positive and PI negative (lower right quadrant) (Fig. 6A). We previously demonstrated that honokiol-induced necrotic cell death in MCF-7 cells was modulated by cyclophilin D (a major component of the mitochondrial permeability transition pore, CypD), which can be prevented by cyclosporin A (CsA, inhibitor of CypD) (9). Accordingly, we used Annexin V-FITC/PI assay to determine the effect of CsA on honokiol-induced necrotic cell death in MCF-7/ADR cells. Honokiol-induced necrotic cell death was reversed by 14.97% with pretreatment of 25 µm CsA for 2 h (Fig. 6A). These results indicated honokiol-induced necrotic cell death in MCF-7/ADR was partly associated with CypD. The above results provided substantial evidence for the original assumptions that honokiol at a higher dose (40 µg/ ml) induced dominant programmed necrotic cell death in MCF-7/ADR cells and could be prevented by CsA, which accords with that in the parallel cell line MCF-7 (9). Honokiol and VP-16 combination activates complex cell death pathways. Honokiol (40 µg/ml) induced cell death with necrotic characteristics in MCF-7/ADR cells as demonstrated above. Consequently, we hypothesized the combination of honokiol would enhance the cytotoxic effect of VP-16 through additional necrotic cell death. To authenticate this hypothesis, several markers of cell death modality were analyzed, including DNA ladder, morphological manifestations and Annexin V/ PI staining. Characteristic DNA fragmentation detected by DNA ladder is a direct indication of the existence of apoptosis. We used DNA ladder to determine if apoptosis was the major cell death pathway for MCF-7/ADR cells treated with honokiol plus VP-16. As shown in Fig. 4, no DNA ladder but clumping degradation of nuclear DNA was detected in cells treated with honokiol plus VP-16, which indicated apoptosis was not the major cell death pathway (Fig. 4). Further electron microscopy morphological examinations demonstrated cells treated with VP-16 plus honokiol displayed complex features composed of necrotic cell death (severe vacuolation and dilatation of mitochondria) (Fig. 5D and E) and apoptosis (chromatin condensation and margination) (Fig. 5F). Parts of cells treated with VP-16 exhibited early staged apoptotic manifestations, while most cells had normal morphological manifestations (Fig. 5B). The effects of VP-16 combined with honokiol on induction of cell death were then examined with Annexin V-FITC/PI assay. Inhibition of cell viability was more efficient after incubation with honokiol plus VP-16 (40.96±2.18%) than honokiol (51.90±3.46%) (P < 0.05) (Fig. 6B). Honokiol alone induced MCF-7/ADR cell death with obvious characteristics of necrotic cell death as shown in Honokiol-induced programmed necrotic cell death in MCF 7/ADR cells, which is reversed by cyclosporin A. While treatment with combination of honokiol and VP-16 increased the ratio of cell death, including 37.48% of cells PI positive and 19.97% Annexin V-positive and PI-negative (Fig. 6A), which increased necrosis by 27.60% and apoptosis by 16.80% from basal level. Taken together, these results indicated that the combination treatment of honokiol plus VP-16 induced complex cell death including apoptosis and programmed necrotic cell death in MCF-7/ADR cells. Combination of cyclosporin A and z-vad-fmk shuts down the complex cell death pathways. Above data indicated: i) the combination treatment of VP-16 and honokiol induced programmed cell death containing apoptosis and necrotic cell death in MCF-7/ADR cells; ii) CsA reversed honokiolinduced programmed necrotic death in MCF-7/ADR cells. CsA and z-vad-fmk were used separately or together to confirm whether caspase-dependent apoptosis and necrotic cell death constituted the complex cell death induced by honokiol plus VP-16. After CsA pretreatment, morphological examinations demonstrated that more cells exhibited apoptotic manifestations (Fig. 5G) than that shown in treatment of VP-16 plus honokiol (Fig. 5D-F), whereas still some cells exhibited necrotic manifestations. Moreover, Fig. 6A revealed honokiol plus VP-16-induced necrotic cell death was reversed by 19.97% with CsA, while apoptosis was reversed by z-vad-fmk 13.40%, but when pretreated with both CsA and z-vad-fmk, honokiol plus VP-16-induced necrotic cell death and apoptosis were increasingly reversed by and 15.67%. Viable cells were increased after pretreated with CsA, z-vad-fmk, or both (79.02±8.12, 75.79±6.41 and 84.47±4.31%, respectively) (Fig. 6B). Which indicates that honokiol plus VP-16 induced complex cell death was almost completely shut down by pretreatment with both CsA and z-vad-fmk. These
8 728 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS Figure 5. Combination of honokiol and VP-16 induces a complex death modality with dominant necrotic coupled with apoptotic morphology. (A) Control MCF 7/ADR; (B) 100 µg/ml VP-16-treated cells for 6 h, typical apoptotic appearances such as chromatin condensation and margination compared with control cells; (C) 40 µg/ml honokiol-treated cells with necrotic morphology, irregular clumping of chromatin with appearance of cleared nuclear domains, rounded mitochondria with disrupted internal structures, extensive cytoplasmic vacuolation and autophagic vacuoles containing membranous whorls; (D-F) honokiol plus VP-16 combination treated cells for 6 h; (D) apoptotic appearance resemble that of B; (E) necrotic features similar to that in C; (F) high-power magnifications showing mitochondria with disrupted internal structures, cytoplasmic vacuolation and autophagic vacuoles; (G) cells pretreated with 25 µm CsA for 2 h before combination treatment, more cells exhibited apoptotic manifestations than that in combination treatment of honokiol and VP-16. results indicated combination treatment of VP-16 and honokiol induced programmed cell death contained caspase-dependent apoptosis and programmed necrotic cell death, which were prevented by CsA and z-vad-fmk. Honokiol blocks nuclear translocation of NF-κB induced by VP-16. Activated NF-κB binds to specific DNA sequences in target genes associated with anti-apoptotic effects or expression of multiple drug resistant proteins, such as P-gp, Bcl-2, Bcl-xl and xiap. Constitutively activated NF-κB is associated with cell resistance to various chemotherapeutic agents. Nuclear translocation of NF-κB is momentous in NF-κB signal transduction pathway. Blocking nuclear translocation of NF-κB is a potential target in the reversal of chemoresistance. We used western blot analysis to explore the differences of NF-κB expression in nuclear and cytoplasm after VP-16 incubation with or without honokiol. Although no changes were detected in cytoplasmic NF-κB, expressions of nuclear NF-κB were different between different treatments (Fig. 7A). Expression of nuclear NF-κB was slightly increased after incubated with VP-16 for 2 h (1.16±0.02 fold compared with control), but decreased after incubated with honokiol alone for 2 h (0.55±0.01 fold). And the expression of nuclear NF-κB after co-incubated with VP-16 and honokiol (0.95±0.03 fold) was decreased compared to that of VP-16 treatment alone (P<0.001) (Fig. 7B). These results revealed that honokiol not only decreased the primarily enhanced nuclear expression of NF-κB in multi-drug resistant cell line MCF-7/ADR, but also
9 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , Figure 6. Honokiol plus VP-16 induces complex cell death with dominant necrotic characteristics coupled with apoptotic features, which is reversed by pretreated with CsA, z-vad-fmk or both. (A) Flow cytometric assays for phosphatidyl serine externalization or PI permeability; (B) viable cells (AV negative and PI negative cells) calculated from flow cytometric results; a, 0.1% DMSO (vehicle control); b, cell treated with 40 μg/ml honokiol; c, honokiol (pretreated with CsA); d, VP-16 plus honokiol; e, VP-16 plus honokiol (pretreated with CsA); f, VP-16 plus honokiol (pretreated with z-vad-fmk); g, VP-16 plus honokiol (pretreated with CsA and z-vad-fmk). The drug concentrations and incubation time applied: honokiol 40 µg/ml for 6 h; VP µg/ml for 6 h; CsA 25 µm for 2 h; z-vad-fmk 50 µm for 30 min. These results were representative of three independent experiments. The values are shown as mean ± SD. * P<0.001 vs. control; ** P<0.05 vs. honokiol-treated cells; # P<0.001 vs. VP-16 plus honokiol treated. reversed VP-16-induced nuclear translocation and activation of NF-κB. Discussion Most previous studies concerning combination effect of honokiol with other chemotherapeutic agents only focused on therapeutic synergy or therapeutic enhancement, which manifested as, at a given dose, honokiol strengthened cytotoxicity of other chemotherapeutic agents. But this phenomenon does not exactly signify synergism. It is perhaps composed of pure synergism, additive effect, or even moderate antagonism if the drugs at combination do not produce overlapping toxicity. Consequently, the evaluation of a drug combination as therapeutic synergy seems subjective and lacks quantitative data. Accordingly, we use the combination index equation of Chou and Talalay (16) to quantitatively and
10 730 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS Figure 7. Honokiol attenuates NF-κB activation in VP-16-treated MCF-7/ADR cells. (A) Nuclear and cytoplasmic NF-κB measured by western blot analysis with Lamin B and α-tubulin as internal controls, respectively. (B) Relative folds of nuclear NF-κB compared with negative control. These results are representative of three independent experiments. The values are shown as mean ± SD. * P<0.001 vs. VP-16-treated cells; # P<0.001 vs. honokiol-treated cells. objectively calculate the real combination effects of honokiol with other chemotherapeutic agents in both sensitive and resistant cancer cell lines, solid and none-solid cancer cell lines. Honokiol synergized ADM and taxol in breast carcinoma cell line MCF-7 and with DNR in acute promyelocytic leukemia cell line HL-60. Through evaluation of DRI, we found honokiol at slight toxic concentrations synergized the antitumor effects of ADM and taxol in MCF-7, synergized the antitumor effects of DNR in HL-60. In multidrug resistant cell lines MCF-7/ADR and HL-60/ADR, honokiol also synergized the antitumor effects of ADM, VP-16 or DNR as that shown in the calculation of CI and DRI values. Multiple drug resistance refers to the phenomenon that tumor cells are resistant to various structural and functional different drugs. As we have demonstrated synergistic activity of honokiol with chemotherapeutic agents in MDR cell lines, we aimed to investigate its possible mechanisms, especially associated with overcoming multiple drug resistance. P-gp is the first identified and most well studied ABC transporter, which catalyses the active efflux transport of various structurally unrelated therapeutic agents (19). Our previous report demonstrated at a lower concentration (20 µm, 5 µg/ml), honokiol enhances cytotoxic effect of ADM through downregulation of P-gp (2). These results provide substantial evidence for synergistic mechanism of honokiol at a lower concentration, but possible synergistic mechanism of honokiol at higher concentration is still in need of evaluation. Programmed necrotic cell death (necroptosis) is one of the non-apoptotic mechanisms of cell death, which is also regulated by multiple signal transduction pathways and metabolic processes (8,20-22). Honokiol induces programmed necrotic cell death associated with mitochondrial permeability transition pore in MCF-7 at the concentration 40 µg/ml as we previously reported (9). We presume honokiol also induces necrotic cell death in the parallel drug resistant cell line MCF-7/ADR and the subsequent research data in our present study support the above notion. All evidence including DNA ladder findings, flow cytometric analysis and electron microscopy morphological manifestations, are in conformity with those of MCF-7 (9). In agreement with MCF-7, CsA prevents this necrotic cell death induced by 40 µg/ml honokiol in MCF-7/ADR. These results provide substantial evidence for the original assumptions that honokiol 40 µg/ml induces necrotic cell death which can be prevented by CsA in multi-drug resistant cell line MCF-7/ADR. The substantial pharmacological objective of chemotherapy is inducing tumor cell death. Most chemotherapeutic agents kill tumor cells through apoptosis pathway. Evasion of drug-induced apoptosis is a major barrier for the success of chemotherapy. Most research concentrates on obstructing Bcl-2 family members or associated signal signaling pathways to overcome resistance to apoptosis. Nonetheless, we consider simultaneous activating multiple death pathways may be another choice to overcome apoptosis-escape associated MDR. After demonstrating 40 µg/ml honokiol inducing necrotic cell death in MCF-7/ADR cells, we presume the mechanism of honokiol at a higher concentration synergizes conventional chemotherapy agents and overcomes MDR through additional necrotic cell death. We further assessed the synergistic mechanism of
11 INTERNATIONAL JOURNAL OF ONCOLOGY 42: , Figure 8. Possible mechanisms of honokiol leading to the synergistic effect and overcoming MDR. honokiol (40 µg/ml) with VP-16 (100 µg/ml). Morphological manifestations revealed the complex cell death containing necrotic cell death and apoptosis. Flow cytometry analyzed Annexin V-FITC/PI also demonstrated both cell death pathways were activated after incubation with honokiol and VP-16. While pretreatment with z-vad-fmk or CsA not only partly blocked apoptosis but also reversed necrotic cell death. Supposing that mitochondria permeability transition is a core step in both programmed necrotic cell death and apoptosis (23), it is understandable that CsA, an inhibitor of CypD, has the potency to prevent both programmed necrotic cell death and apoptosis induced by honokiol plus VP-16. z-vad-fmk is a pan-caspase inhibitor, which mainly blocks the cascade of caspase-dependent apoptosis. Consequently, the inhibition of early stage apoptosis by z-vad-fmk can be readily explained. z-vad-fmk can not inhibit honokiol induced necrotic cell death in MCF-7 cells (9), whereas it partly prevents necrotic cell death induced by combination of honokiol and VP-16. A possible explanation is that the combination treatment of honokiol and VP-16 activates some caspase associated pathways, which participate in both cascades of apoptosis and programmed necrotic death. The existence of caspase associated cross-talk between these two cell death modalities enable z-vad-fmk to block both programmed necrotic death and apoptosis. Accordingly, we suppose that these two kinds of cell death constitute a more complex cell death modality and share some corporate signaling pathways. These experimental results support the original hypothesis that honokiol synergized the antitumor effect of VP-16 by inducing enhanced apoptosis and additional necrotic cell death. Transcription factor NF-κB critically regulates a variety of cell physiological and pathological processes (24,25). Aberrant NF-κB activation takes part in development of the hallmarks of cancer (26). One of the most documented functions of NF-κB is its capacity to modulate cancer cell chemoresistance through efflux of drugs and evasion from drug-induced apoptosis by inducing the expression of genes coding anti-apoptotic proteins (such as Bcl-2, Bcl-xl and xiap) (27-29) and ABC families (such as P-gp) (30). Previous reports indicate honokiol suppresses activation of NF-κB and NF-κB-regulated gene expression by inhibition of nuclear translocation and transcriptional activity of NF-κB responsive promoter (31-33). Honokiol downregulates the expression of NF-κB-regulated anti-apoptotic and drug resistance gene products including Bcl-x, Bcl-2, cflip, IAP1, IAP2 (32) and P-gp. Our data indicate VP-16 can induce NF-κB activation in MCF-7/ADR cells, which accord with previous reports (34,35). The data from western blots suggested that honokiol enhanced VP-16-induced apoptosis possibly via block of NF-κB activation, which might lead to subsequent drug resistance and apoptosis tolerance. These results provide evidence for that honokiol by suppressing VP-16-induced NF-κB activation partly contributes to the synergistic effect. The mechanistic findings of this study are restricted to synergistic effect of honokiol with VP-16 in MCF-7/ADR. Combination effects of honokiol and VP-16 are conspicuously
12 732 TIAN et al: HONOKIOL SYNERGIZES CHEMOTHERAPY DRUGS IN MDR BREAST CANCER CELLS different in MCF-7/ADR (synergism) and parental cell line MCF-7 (antagonism). The possible mechanism and associated signal pathways underlying the above phenomenon are still under investigation in our research group. Collectively, according to our previous report (2) and present results, we conclude that the possible mechanisms leading to the synergistic effect and overcoming drug resistance are: i) honokiol downregulates P-gp expression and negatively regulates efflux of chemotherapeutic agents; ii) honokiol is not the substrate of P-gp and keeps sensitivity towards MDR cancer cells; iii) honokiol plus VP-16 induces a very complex death modality composed of programmed necrotic cell death and apoptosis; iv) VP-16 activates NF-κB and subsequent drugresistance or apoptosis tolerance, whereas honokiol block it (Fig. 8). In conclusion, our data demonstrate for the first time that honokiol synergistically enhanced cytotoxicity of frequentlyused chemotherapeutic agents in both sensitive and resistant cancer cell lines. Activating complex cell death pathways through enhanced apoptosis and additional programmed necrotic death may be the principal mechanism underlying the synergistic effect. These findings show a promising way to circumvent multidrug resistance and apoptosis tolerance, which make honokiol a promising novel anticancer drug especially in refractory cancers. Acknowledgements Dong Xu received research grants from National Natural Science Foundation of China (no ) and Zhejiang Provincial Natural Science Foundation of China (no.y ); Jie Sun received research grants from National Natural Science Foundation of China (no ). References 1. Hu X and Xuan Y: Bypassing cancer drug resistance by activating multiple death pathways - a proposal from the study of circumventing cancer drug resistance by induction of necroptosis. Cancer Lett 259: , Xu D, Lu Q and Hu X: Down-regulation of P-glycoprotein expression in MDR breast cancer cell MCF-7/ADR by honokiol. Cancer Lett 243: , Baguley BC: Multiple drug resistance mechanisms in cancer. Mol Biotechnol 46: , Xu D, Tian W and Shen H: Curcumin prevents induced drug resistance: a novel function? Chin J Cancer Res 23: , Gillet JP and Gottesman MM: Mechanisms of multidrug resistance in cancer. Methods Mol Biol 596: 47-76, Gandhi L, Harding MW, Neubauer M, et al: A phase II study of the safety and efficacy of the multidrug resistance inhibitor VX-710 combined with doxorubicin and vincristine in patients with recurrent small cell lung cancer. Cancer 109: , Ruff P, Vorobiof DA, Jordaan JP, et al: A randomized, placebocontrolled, double-blind phase 2 study of docetaxel compared to docetaxel plus zosuquidar (LY335979) in women with metastatic or locally recurrent breast cancer who have received one prior chemotherapy regimen. Cancer Chemother Pharmacol 64: , Kroemer G, Galluzzi L, Vandenabeele P, et al: Classification of cell death: recommendations of the Nomenclature Committee on Cell Death Cell Death Differ 16: 3-11, Li L, Han W, Gu Y, et al: Honokiol induces a necrotic cell death through the mitochondrial permeability transition pore. Cancer Res 67: , Xuan Y and Hu X: Naturally-occurring shikonin analogues - a class of necroptotic inducers that circumvent cancer drug resistance. Cancer Lett 274: , Guidicelli G, Chaigne-Delalande B, Dilhuydy MS, et al: The necrotic signal induced by mycophenolic acid overcomes apoptosis-resistance in tumor cells. PLoS One 4: e5493, Ishitsuka K, Hideshima T, Hamasaki M, et al: Honokiol overcomes conventional drug resistance in human multiple myeloma by induction of caspase-dependent and -independent apoptosis. Blood 106: , Wang Y, Yang Z and Zhao X: Honokiol induces paraptosis and apoptosis and exhibits schedule-dependent synergy in combination with imatinib in human leukemia cells. Toxicol Mech Methods 20: , Wang T, Chen F, Chen Z, et al: Honokiol induces apoptosis through p53-independent pathway in human colorectal cell line RKO. World J Gastroenterol 10: , Chen F, Wang T, Wu YF, et al: Honokiol: a potent chemotherapy candidate for human colorectal carcinoma. World J Gastroenterol 10: , Chou TC and Talalay P: Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 22: 27-55, Bijnsdorp IV, Giovannetti E and Peters GJ: Analysis of drug interactions. Methods Mol Biol 731: , Battle TE, Arbiser J and Frank DA: The natural product honokiol induces caspase-dependent apoptosis in B-cell chronic lymphocytic leukemia (B-CLL) cells. Blood 106: , Steinbach D and Legrand O: ABC transporters and drug resistance in leukemia: was P-gp nothing but the first head of the Hydra? Leukemia 21: , Golstein P and Kroemer G: Cell death by necrosis: towards a molecular definition. Trends Biochem Sci 32: 37-43, Vandenabeele P, Galluzzi L, Vanden Berghe T and Kroemer G: Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 11: , Degterev A and Yuan J: Expansion and evolution of cell death programmes. Nat Rev Mol Cell Biol 9: , Kinnally KW, Peixoto PM, Ryu SY and Dejean LM: Is mptp the gatekeeper for necrosis, apoptosis, or both? Biochim Biophys Acta 1813: , Oeckinghaus A, Hayden MS and Ghosh S: Crosstalk in NF-kappaB signaling pathways. Nat Immunol 12: , Shih VF, Tsui R, Caldwell A and Hoffmann A: A single NFkappaB system for both canonical and non-canonical signaling. Cell Res 21: , Hanahan D and Weinberg RA: Hallmarks of cancer: the next generation. Cell 144: , Catz SD and Johnson JL: Transcriptional regulation of bcl-2 by nuclear factor kappa B and its significance in prostate cancer. Oncogene 20: , Tamatani M, Che YH, Matsuzaki H, et al: Tumor necrosis factor induces Bcl-2 and Bcl-x expression through NFkappaB activation in primary hippocampal neurons. J Biol Chem 274: , Dong QG, Sclabas GM, Fujioka S, et al: The function of multiple IkappaB : NF-kappaB complexes in the resistance of cancer cells to Taxol-induced apoptosis. Oncogene 21: , Bentires-Alj M, Barbu V, Fillet M, et al: NF-kappaB transcription factor induces drug resistance through MDR1 expression in cancer cells. Oncogene 22: 90-97, Arora S, Bhardwaj A, Srivastava SK, et al: Honokiol arrests cell cycle, induces apoptosis, and potentiates the cytotoxic effect of gemcitabine in human pancreatic cancer cells. PLoS One 6: e21573, Ahn KS, Sethi G, Shishodia S, Sung B, Arbiser JL and Aggarwal BB: Honokiol potentiates apoptosis, suppresses osteoclastogenesis, and inhibits invasion through modulation of nuclear factor-kappab activation pathway. Mol Cancer Res 4: , Tse AK, Wan CK, Shen XL, Yang M and Fong WF: Honokiol inhibits TNF-alpha-stimulated NF-kappaB activation and NF-kappaB-regulated gene expression through suppression of IKK activation. Biochem Pharmacol 70: , Arlt A, Vorndamm J, Breitenbroich M, et al: Inhibition of NF-kappaB sensitizes human pancreatic carcinoma cells to apoptosis induced by etoposide (VP16) or doxorubicin. Oncogene 20: , Nakanishi C and Toi M: Nuclear factor-kappab inhibitors as sensitizers to anticancer drugs. Nat Rev Cancer 5: , 2005.
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108
 Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line
 Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line Endah Puji Septisetyani, Muthi Ikawati, Barinta Widaryanti and Edy Meiyanto* ) Cancer Chemoprevention Research Center,
Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line Endah Puji Septisetyani, Muthi Ikawati, Barinta Widaryanti and Edy Meiyanto* ) Cancer Chemoprevention Research Center,
PUMA gene transfection can enhance the sensitivity of epirubicin-induced apoptosis of MCF-7 breast cancer cells
 PUMA gene transfection can enhance the sensitivity of epirubicin-induced apoptosis of MCF-7 breast cancer cells C.-G. Sun 1 *, J. Zhuang 1 *, W.-J. Teng 1, Z. Wang 2 and S.-S. Du 3 1 Department of Oncology,
PUMA gene transfection can enhance the sensitivity of epirubicin-induced apoptosis of MCF-7 breast cancer cells C.-G. Sun 1 *, J. Zhuang 1 *, W.-J. Teng 1, Z. Wang 2 and S.-S. Du 3 1 Department of Oncology,
IMMP8-1. Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells
 IMMP8-1 Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells Assanan Dokmaikaew* Tipaya Ekalaksananan** Dr.Chamsai Pientong** ABSTRACT Androg and IPAD are recently known
IMMP8-1 Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells Assanan Dokmaikaew* Tipaya Ekalaksananan** Dr.Chamsai Pientong** ABSTRACT Androg and IPAD are recently known
Introduction to pathology lecture 5/ Cell injury apoptosis. Dr H Awad 2017/18
 Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Effect of ST2825 on the proliferation and apoptosis of human hepatocellular carcinoma cells
 Effect of ST2825 on the proliferation and apoptosis of human hepatocellular carcinoma cells Y. Deng, J. Sun and L.D. Zhang Department of Hepatobiliary Surgery Institute, Southwest Hospital, Third Military
Effect of ST2825 on the proliferation and apoptosis of human hepatocellular carcinoma cells Y. Deng, J. Sun and L.D. Zhang Department of Hepatobiliary Surgery Institute, Southwest Hospital, Third Military
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Berberine Sensitizes Human Ovarian Cancer Cells to Cisplatin Through mir-93/ PTEN/Akt Signaling Pathway
 Chen Accepted: et al.: February Berberine 24, Sensitizes 2015 Ovarian Cancer Cells to Cisplatin www.karger.com/cpb 956 1421-9778/15/0363-0956$39.50/0 Original Paper This is an Open Access article licensed
Chen Accepted: et al.: February Berberine 24, Sensitizes 2015 Ovarian Cancer Cells to Cisplatin www.karger.com/cpb 956 1421-9778/15/0363-0956$39.50/0 Original Paper This is an Open Access article licensed
[Abstract] Objective: Investigate the effect of KLT on the expression of Fas/Apo-1, FasL and PCNA genes in renal carcinoma cell line (GRC-1).
![[Abstract] Objective: Investigate the effect of KLT on the expression of Fas/Apo-1, FasL and PCNA genes in renal carcinoma cell line (GRC-1). [Abstract] Objective: Investigate the effect of KLT on the expression of Fas/Apo-1, FasL and PCNA genes in renal carcinoma cell line (GRC-1).](/thumbs/90/104150076.jpg) Study on Effect of Kanglaite Injection(KLT) on the Expression of Fas/Apo-1, FasL and PCNA in Renal Carcinoma Cell Lines Wang Junjie, Sun Xinchen, Sheng Wenjiang, Yu Lizhang [Abstract] Objective: Investigate
Study on Effect of Kanglaite Injection(KLT) on the Expression of Fas/Apo-1, FasL and PCNA in Renal Carcinoma Cell Lines Wang Junjie, Sun Xinchen, Sheng Wenjiang, Yu Lizhang [Abstract] Objective: Investigate
Apoptosis Chapter 9. Neelu Yadav PhD
 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Focus Application. Compound-Induced Cytotoxicity
 xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity Featured Study: Using the Time Resolving Function of the xcelligence System to Optimize Endpoint Viability and
xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity Featured Study: Using the Time Resolving Function of the xcelligence System to Optimize Endpoint Viability and
Research Article Ginseng Extract Enhances Anti-cancer Effect of Cytarabine on Human Acute Leukemia Cells
 Advance Journal of Food Science and Technology 7(3): 164-168, 2015 DOI:10.19026/ajfst.7.1286 ISSN: 2042-4868; e-issn: 2042-4876 2015 Maxwell Scientific Publication Corp. Submitted: August 19, 2014 Accepted:
Advance Journal of Food Science and Technology 7(3): 164-168, 2015 DOI:10.19026/ajfst.7.1286 ISSN: 2042-4868; e-issn: 2042-4876 2015 Maxwell Scientific Publication Corp. Submitted: August 19, 2014 Accepted:
Effects of Kanglaite Injedction on Reversing Multiple Drug Resistance (MDR) of Tumor Cells
 Effects of Kanglaite Injedction on Reversing Multiple Drug Resistance (MDR) of Tumor Cells Yang Hua, Wang Xianping, Yu, Linlin, Zheng Shu, Zhejiang Medical University Cancer Institute Abstract Multiple
Effects of Kanglaite Injedction on Reversing Multiple Drug Resistance (MDR) of Tumor Cells Yang Hua, Wang Xianping, Yu, Linlin, Zheng Shu, Zhejiang Medical University Cancer Institute Abstract Multiple
The effect of elemene reversing the multidurg resistance of A549/DDP lung cancer cells
 213 12 33 12 TUMOR Vol. 33, December 213 www.tumorsci.org 161 Basic Research DOI: 1.3781/j.issn.1-7431.213.12. 5 Copyright 213 by TUMOR A549/DDP 1, 2 2 1 3 4 1 1 1. 3619 2. 355 3. 3611 4. 361 elemene cisplatin
213 12 33 12 TUMOR Vol. 33, December 213 www.tumorsci.org 161 Basic Research DOI: 1.3781/j.issn.1-7431.213.12. 5 Copyright 213 by TUMOR A549/DDP 1, 2 2 1 3 4 1 1 1. 3619 2. 355 3. 3611 4. 361 elemene cisplatin
Focus Application. Compound-Induced Cytotoxicity
 xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity For life science research only. Not for use in diagnostic procedures. Featured Study: Using the Time Resolving
xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity For life science research only. Not for use in diagnostic procedures. Featured Study: Using the Time Resolving
Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63
 68 Chin J Cancer Res 22(1):68-72, 2010 www.springerlink.com Original Article Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63 Jing-Wei Wang 1, Yi Liu 2, Hai-mei Tian 2, Wei
68 Chin J Cancer Res 22(1):68-72, 2010 www.springerlink.com Original Article Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63 Jing-Wei Wang 1, Yi Liu 2, Hai-mei Tian 2, Wei
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Multiple mechanisms underlying acquired resistance to taxanes in selected docetaxelresistant MCF-7 breast cancer cells
 Wang et al. BMC Cancer 2014, 14:37 RESEARCH ARTICLE Multiple mechanisms underlying acquired resistance to taxanes in selected docetaxelresistant MCF-7 breast cancer cells Harris Wang 1, The Vo 1, Ali Hajar
Wang et al. BMC Cancer 2014, 14:37 RESEARCH ARTICLE Multiple mechanisms underlying acquired resistance to taxanes in selected docetaxelresistant MCF-7 breast cancer cells Harris Wang 1, The Vo 1, Ali Hajar
Key words: apoptosis, beta-amyrin, cell cycle, liver cancer, tritepenoids
 JBUON 2018; 23(4): 965-970 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Antitumor effects of beta-amyrin in Hep-G2 liver carcinoma cells are
JBUON 2018; 23(4): 965-970 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Antitumor effects of beta-amyrin in Hep-G2 liver carcinoma cells are
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Original Article Bufalin attenuates the proliferation of breast cancer MCF-7 cells in vitro and in vivo by inhibiting the PI3K/Akt pathway
 Int J Clin Exp Med 2016;9(6):10297-10303 www.ijcem.com /ISSN:1940-5901/IJCEM0022878 Original Article Bufalin attenuates the proliferation of breast cancer MCF-7 cells in vitro and in vivo by inhibiting
Int J Clin Exp Med 2016;9(6):10297-10303 www.ijcem.com /ISSN:1940-5901/IJCEM0022878 Original Article Bufalin attenuates the proliferation of breast cancer MCF-7 cells in vitro and in vivo by inhibiting
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
The effect of insulin on chemotherapeutic drug sensitivity in human esophageal and lung cancer cells
 The effect of insulin on chemotherapeutic drug sensitivity in human esophageal and lung cancer cells Published in: Natl Med J China, February 10, 2003; Vol 83, No 3, Page 195-197. Authors: JIAO Shun-Chang,
The effect of insulin on chemotherapeutic drug sensitivity in human esophageal and lung cancer cells Published in: Natl Med J China, February 10, 2003; Vol 83, No 3, Page 195-197. Authors: JIAO Shun-Chang,
RCPA Research Award Final Progress Review
 RCPA Research Award 2010-2011 Final Progress Review Name: Dr Craig Wallington-Beddoe Degree/Institution/Year: PhD, The University of Sydney, Year 2 Research Project Title: New Therapeutic Strategies for
RCPA Research Award 2010-2011 Final Progress Review Name: Dr Craig Wallington-Beddoe Degree/Institution/Year: PhD, The University of Sydney, Year 2 Research Project Title: New Therapeutic Strategies for
ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS
 7 CHAPTER SEVEN ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS 136 7.1 INTRODUCTION Breast cancer is one of the leading causes of death in women (Stephens et al., 2012). Several chemotherapeutic
7 CHAPTER SEVEN ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS 136 7.1 INTRODUCTION Breast cancer is one of the leading causes of death in women (Stephens et al., 2012). Several chemotherapeutic
Development of Drug Resistance and Strategies to Circumvent it: A Brief Look
 Development of Drug Resistance and Strategies to Circumvent it: A Brief Look Jin-Ming Yang, MD, PhD The Cancer Institute of New Jersey Department of Pharmacology UMDNJ-Robert Wood Johnson Medical School
Development of Drug Resistance and Strategies to Circumvent it: A Brief Look Jin-Ming Yang, MD, PhD The Cancer Institute of New Jersey Department of Pharmacology UMDNJ-Robert Wood Johnson Medical School
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Correlation of expression of P-glycoprotein and inhibitor of apoptosis proteins to chemosensitivity in gastrointestinal carcinoma tissues
 [Chinese Journal of Cancer 27:11, Construction 423-427; of a November eukaryotic 2008]; vector 2008 expressing Sun Yat-sen MASPIN University ene and its Cancer effect Center on cell apoptosis in gastric
[Chinese Journal of Cancer 27:11, Construction 423-427; of a November eukaryotic 2008]; vector 2008 expressing Sun Yat-sen MASPIN University ene and its Cancer effect Center on cell apoptosis in gastric
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury
 Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
Cinnamomum Essential Oil Prevents DNA Damage- Induced by Doxorubicin on CHO-K1 Cells
 Indonesian Journal of Cancer Chemoprevention, 2017, 8(1): 27-31 ISSN: 2088 0197 Cinnamomum Essential Oil Prevents DNA Damage- Induced by Doxorubicin on CHO-K1 Cells Layung Sekar Sih Wikanthi, Nindi Wulandari,
Indonesian Journal of Cancer Chemoprevention, 2017, 8(1): 27-31 ISSN: 2088 0197 Cinnamomum Essential Oil Prevents DNA Damage- Induced by Doxorubicin on CHO-K1 Cells Layung Sekar Sih Wikanthi, Nindi Wulandari,
Ethanolic Extract of Moringa oleifera L. Increases Sensitivity of WiDr Colon Cancer Cell Line Towards 5-Fluorouracil
 Indonesian Journal of Cancer Chemoprevention, 2010, 1(2):124-128 Ethanolic Extract of Moringa oleifera L. Increases Sensitivity of WiDr Colon Cancer Cell Line Towards 5-Fluorouracil Kholid Alfan Nur, Herwandhani
Indonesian Journal of Cancer Chemoprevention, 2010, 1(2):124-128 Ethanolic Extract of Moringa oleifera L. Increases Sensitivity of WiDr Colon Cancer Cell Line Towards 5-Fluorouracil Kholid Alfan Nur, Herwandhani
#19 Apoptosis Chapter 9. Neelu Yadav PhD
 #19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
#19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
MOLECULAR MEDICINE REPORTS 13: , 2016
 MOLECULAR MEDICINE REPORTS 13: 3227-3235, 2016 mtor signaling pathway is inhibited downstream of the cyclophilin D-mediated mitochondrial permeability transition in honokiol-triggered regulated necrosis
MOLECULAR MEDICINE REPORTS 13: 3227-3235, 2016 mtor signaling pathway is inhibited downstream of the cyclophilin D-mediated mitochondrial permeability transition in honokiol-triggered regulated necrosis
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 212. Supporting Information for Adv. Funct. Mater., DOI:.2/adfm.2122233 MnO Nanocrystals: A Platform for Integration of MRI and Genuine
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 212. Supporting Information for Adv. Funct. Mater., DOI:.2/adfm.2122233 MnO Nanocrystals: A Platform for Integration of MRI and Genuine
Autophagy induction by low-dose cisplatin: The role of p53 in autophagy
 248 Autophagy induction by low-dose cisplatin: The role of p53 in autophagy KYUNG-HWA CHO 1, JI-HYE PARK 1, KANG-BEOM KWON 2, YOUNG-RAE LEE 2, HONG-SEOB SO 2, KANG-KYOO LEE 3, SAM-YOUN LEE 4, SUN-ROCK
248 Autophagy induction by low-dose cisplatin: The role of p53 in autophagy KYUNG-HWA CHO 1, JI-HYE PARK 1, KANG-BEOM KWON 2, YOUNG-RAE LEE 2, HONG-SEOB SO 2, KANG-KYOO LEE 3, SAM-YOUN LEE 4, SUN-ROCK
McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells
 Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
A novel bfgf antagonist peptide inhibits breast cancer cell growth
 210 A novel bfgf antagonist peptide inhibits breast cancer cell growth QUCHOU LI 1, SUSU GAO 1, YONGLIN YU 1, WENHUI WANG 1, XILEI CHEN 1, RUIXUE WANG 1, TAO LI 1, CONG WANG 1, XIAOKUN LI 1,2 and XIAOPING
210 A novel bfgf antagonist peptide inhibits breast cancer cell growth QUCHOU LI 1, SUSU GAO 1, YONGLIN YU 1, WENHUI WANG 1, XILEI CHEN 1, RUIXUE WANG 1, TAO LI 1, CONG WANG 1, XIAOKUN LI 1,2 and XIAOPING
Annals of Oncology Advance Access published January 10, 2005
 Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Neuronal Death After Hemorrhagic Stroke In Vitro and In Vivo Shares Features of Ferroptosis and Necroptosis
 Neuronal Death After Hemorrhagic Stroke In Vitro and In Vivo Shares Features of Ferroptosis and Necroptosis Marietta Zille, PhD Burke Medical Research Institute Weill Cornell Medicine White Plains, NY
Neuronal Death After Hemorrhagic Stroke In Vitro and In Vivo Shares Features of Ferroptosis and Necroptosis Marietta Zille, PhD Burke Medical Research Institute Weill Cornell Medicine White Plains, NY
Evaluation of the cytotoxicity of the Bithionol - cisplatin combination in a panel of human ovarian cancer cell lines
 Ayyagari et al. BMC Cancer (2017) 17:49 DOI 10.1186/s12885-016-3034-2 RESEARCH ARTICLE Evaluation of the cytotoxicity of the Bithionol - cisplatin combination in a panel of human ovarian cancer cell lines
Ayyagari et al. BMC Cancer (2017) 17:49 DOI 10.1186/s12885-016-3034-2 RESEARCH ARTICLE Evaluation of the cytotoxicity of the Bithionol - cisplatin combination in a panel of human ovarian cancer cell lines
MG132 reverse the malignant characteristics of hypopharyngeal cancer
 MOLECULAR MEDICINE REPORTS 9: 2587-2591, 2014 MG132 reverse the malignant characteristics of hypopharyngeal cancer JUKE MA 1, LIANG YU 1, JIAJUN TIAN 1, YAKUI MU 1, ZHENGHUA LV 1, JIDONG ZOU 1, JIANFENG
MOLECULAR MEDICINE REPORTS 9: 2587-2591, 2014 MG132 reverse the malignant characteristics of hypopharyngeal cancer JUKE MA 1, LIANG YU 1, JIAJUN TIAN 1, YAKUI MU 1, ZHENGHUA LV 1, JIDONG ZOU 1, JIANFENG
Effect of starvation-induced autophagy on cell cycle of tumor cells
 [Chinese Journal of Cancer 27:8, 102-108; August 2008]; 2008 Sun Yat-Sen University Cancer Center Basic Research Paper Effect of starvation-induced autophagy on cell cycle of tumor cells Jun-Na Ge, 1 Dan
[Chinese Journal of Cancer 27:8, 102-108; August 2008]; 2008 Sun Yat-Sen University Cancer Center Basic Research Paper Effect of starvation-induced autophagy on cell cycle of tumor cells Jun-Na Ge, 1 Dan
Effects of COX-2 Inhibitor on the Proliferation of MCF-7 and LTED MCF-7 Cells
 Mahidol University Journal of Pharmaceutical Sciences 2008; 35(1-4): 47-51. Original Article Effects of COX-2 Inhibitor on the Proliferation of MCF-7 and LTED MCF-7 Cells K. Poemsantitham, N. Sookvanichsilp
Mahidol University Journal of Pharmaceutical Sciences 2008; 35(1-4): 47-51. Original Article Effects of COX-2 Inhibitor on the Proliferation of MCF-7 and LTED MCF-7 Cells K. Poemsantitham, N. Sookvanichsilp
ORIGINAL ARTICLE. Hang Huang 1,2, Lin-Jin Li 3, Hai-Bo Zhang 4, An-Yang Wei 4. Summary. Introduction
 JBUON 2017; 22(1): 112-118 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Papaverine selectively inhibits human prostate cancer cell (PC-3) growth
JBUON 2017; 22(1): 112-118 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Papaverine selectively inhibits human prostate cancer cell (PC-3) growth
8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES
 8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES 8.1. Introduction Nanobiotechnology, an emerging field of nanoscience, utilizes nanobased-systems for various biomedical applications.
8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES 8.1. Introduction Nanobiotechnology, an emerging field of nanoscience, utilizes nanobased-systems for various biomedical applications.
ONCOLOGY LETTERS 7: , 2014
 1738 Fermentation supernatants of Lactobacillus delbrueckii inhibit growth of human colon cancer cells and induce apoptosis through a caspase 3 dependent pathway YING WAN 1, YI XIN 1, CUILI ZHANG 1, DACHANG
1738 Fermentation supernatants of Lactobacillus delbrueckii inhibit growth of human colon cancer cells and induce apoptosis through a caspase 3 dependent pathway YING WAN 1, YI XIN 1, CUILI ZHANG 1, DACHANG
Introduction NEOPLASIA. Qun Liu and Yair Gazitt
 NEOPLASIA Potentiation of dexamethasone-, paclitaxel-, and Ad-p53 induced apoptosis by Bcl-2 antisense oligodeoxynucleotides in drug-resistant multiple myeloma cells Qun Liu and Yair Gazitt Overexpression
NEOPLASIA Potentiation of dexamethasone-, paclitaxel-, and Ad-p53 induced apoptosis by Bcl-2 antisense oligodeoxynucleotides in drug-resistant multiple myeloma cells Qun Liu and Yair Gazitt Overexpression
SUPPLEMENTARY INFORMATION
 Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Inhibition of autophagy promotes cell apoptosis induced by the proteasome inhibitor MG-132 in human esophageal squamous cell carcinoma EC9706 cells
 2278 Inhibition of autophagy promotes cell apoptosis induced by the proteasome inhibitor MG-132 in human esophageal squamous cell carcinoma EC9706 cells DONGLEI LIU 1*, MIN GAO 2*, YANG YANG 1, YU QI 1,
2278 Inhibition of autophagy promotes cell apoptosis induced by the proteasome inhibitor MG-132 in human esophageal squamous cell carcinoma EC9706 cells DONGLEI LIU 1*, MIN GAO 2*, YANG YANG 1, YU QI 1,
Caractérisation et méthodes d études de la mort cellulaire par cytométrie en flux
 Caractérisation et méthodes d études de la mort cellulaire par cytométrie en flux Université de Bourgogne Gérard LIZARD - Inserm EA7270 - Equipe Biochimie du Peroxysome, inflammation et Métabolisme Lipidique
Caractérisation et méthodes d études de la mort cellulaire par cytométrie en flux Université de Bourgogne Gérard LIZARD - Inserm EA7270 - Equipe Biochimie du Peroxysome, inflammation et Métabolisme Lipidique
Advances in Computer Science Research, volume 59 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016)
 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
Johannes F Fahrmann and W Elaine Hardman *
 Fahrmann and Hardman Lipids in Health and Disease 2013, 12:36 RESEARCH Open Access Omega 3 fatty acids increase the chemo-sensitivity of B-CLL-derived cell lines EHEB and and of B-PLL-derived cell line
Fahrmann and Hardman Lipids in Health and Disease 2013, 12:36 RESEARCH Open Access Omega 3 fatty acids increase the chemo-sensitivity of B-CLL-derived cell lines EHEB and and of B-PLL-derived cell line
PE Annexin V Apoptosis Detection Kit User Manual KT40001
 PE Annexin V Apoptosis Detection Kit User Manual KT40001 For research use only. Not intended for diagnostic testing. a WuXi AppTec company www.abgent.com.cn PE Annexin-V Apoptosis Detection Kit Product
PE Annexin V Apoptosis Detection Kit User Manual KT40001 For research use only. Not intended for diagnostic testing. a WuXi AppTec company www.abgent.com.cn PE Annexin-V Apoptosis Detection Kit Product
Chloroquine inhibits cell growth and induces cell death in A549 lung cancer cells
 Bioorganic & Medicinal Chemistry 14 (2006) 3218 3222 Chloroquine inhibits cell growth and induces cell death in A549 lung cancer cells Chuandong Fan, a,c Weiwei Wang, a,c Baoxiang Zhao, b, Shangli Zhang
Bioorganic & Medicinal Chemistry 14 (2006) 3218 3222 Chloroquine inhibits cell growth and induces cell death in A549 lung cancer cells Chuandong Fan, a,c Weiwei Wang, a,c Baoxiang Zhao, b, Shangli Zhang
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy
 Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
In Vitro Studies of the Aurora-Kinase Inhibitor MLN8237 in Prostate Cancer Cell Lines
 In Vitro Studies of the Aurora-Kinase Inhibitor MLN837 in Prostate Cancer Cell Lines Nicole V. Julian Mentor: Ana Aparicio, M.D. Special Thanks to Brittany Kleb Department of Genitourinary Medical Oncology
In Vitro Studies of the Aurora-Kinase Inhibitor MLN837 in Prostate Cancer Cell Lines Nicole V. Julian Mentor: Ana Aparicio, M.D. Special Thanks to Brittany Kleb Department of Genitourinary Medical Oncology
Comparison of Cytotoxic Activity of Anticancer Drugs against Various Human Tumor Cell Lines Using In Vitro Cell-Based Approach
 International journal of Biomedical science ORIGINAL ARTICLE Comparison of Cytotoxic Activity of Anticancer Drugs against Various Human Tumor Cell Lines Using In Vitro Cell-Based Approach Leila Florento,
International journal of Biomedical science ORIGINAL ARTICLE Comparison of Cytotoxic Activity of Anticancer Drugs against Various Human Tumor Cell Lines Using In Vitro Cell-Based Approach Leila Florento,
Z.-L. LIU 1, B.-J. JIN 1, C.-G. CHENG 1, F.-X. ZHANG 1, S.-W. WANG 1, Y. WANG 1, B. WU 2. Introduction. Abstract. OBJECTIVE: To observe the reversal
 European Review for Medical and Pharmacological Sciences 2017; 21: 5370-5377 Apatinib resensitizes cisplatin-resistant non-small cell lung carcinoma A549 cell through reversing multidrug resistance and
European Review for Medical and Pharmacological Sciences 2017; 21: 5370-5377 Apatinib resensitizes cisplatin-resistant non-small cell lung carcinoma A549 cell through reversing multidrug resistance and
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Plate-Based Assay Methods for the Assessment of Cellular Health
 Plate-Based Assay Methods for the Assessment of Cellular Health Andrew L. Niles, Senior Research Scientist 2012, Promega Corporation. Biological Outcomes in Cell Culture Treatment -Small molecule -Bio-molecule
Plate-Based Assay Methods for the Assessment of Cellular Health Andrew L. Niles, Senior Research Scientist 2012, Promega Corporation. Biological Outcomes in Cell Culture Treatment -Small molecule -Bio-molecule
Original Article The chemosensitization effect of quercetin on cisplatin induces the apoptosis of human colon cancer HT-29 cell line
 Int J Clin Exp Med 2016;9(2):2285-2292 www.ijcem.com /ISSN:1940-5901/IJCEM0016214 Original Article The chemosensitization effect of quercetin on cisplatin induces the apoptosis of human colon cancer HT-29
Int J Clin Exp Med 2016;9(2):2285-2292 www.ijcem.com /ISSN:1940-5901/IJCEM0016214 Original Article The chemosensitization effect of quercetin on cisplatin induces the apoptosis of human colon cancer HT-29
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Linderalactone inhibits human lung cancer growth b
 JBUON 2019; 24(2): 566-571 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Linderalactone inhibits human lung cancer growth by modulating the expression
JBUON 2019; 24(2): 566-571 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Linderalactone inhibits human lung cancer growth by modulating the expression
Anti-cancer activity of Aya Thambira Chendooram (ATC) in in-vitro cell line against Breast Carcinoma
 International Journal of Advanced Research in Biological Sciences ISSN: 2348-8069 www.ijarbs.com DOI: 10.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 1-2018 Research Article DOI: http://dx.doi.org/10.22192/ijarbs.2018.05.01.010
International Journal of Advanced Research in Biological Sciences ISSN: 2348-8069 www.ijarbs.com DOI: 10.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 1-2018 Research Article DOI: http://dx.doi.org/10.22192/ijarbs.2018.05.01.010
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4):196-203 In Vitro Effect of Thai herbal extracts with
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4):196-203 In Vitro Effect of Thai herbal extracts with
Muse Assays for Cell Analysis
 Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Supporting Information
 Supporting Information Natural small molecule FMHM inhibits lipopolysaccharide-induced inflammatory response by promoting TRAF6 degradation via K48-linked polyubiquitination Ke-Wu Zeng 1, Li-Xi Liao 1,
Supporting Information Natural small molecule FMHM inhibits lipopolysaccharide-induced inflammatory response by promoting TRAF6 degradation via K48-linked polyubiquitination Ke-Wu Zeng 1, Li-Xi Liao 1,
AMPK Phosphorylation Assay Kit
 AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
MECHANISM OF TAXOL-INDUCED APOPTOSIS IN HUMAN BREAST CANCER CELLS
 Chinese Journal of Cancer Research 10(1):32-36, 1998. MECHANISM OF TAXOL-INDUCED APOPTOSIS IN HUMAN BREAST CANCER CELLS Chen Lirong ~ MC Willingham** Zheng Shu ~ll]~j Fan Weimin** ~ Cancer Institute, Zhejiang
Chinese Journal of Cancer Research 10(1):32-36, 1998. MECHANISM OF TAXOL-INDUCED APOPTOSIS IN HUMAN BREAST CANCER CELLS Chen Lirong ~ MC Willingham** Zheng Shu ~ll]~j Fan Weimin** ~ Cancer Institute, Zhejiang
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value
 Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value Shen Wei 1,a, Chen Juan 2, Li Xiurong 1 and Yin Jie 1 1 Department of Obstetrics and Gynecology,
Study on the expression of MMP-9 and NF-κB proteins in epithelial ovarian cancer tissue and their clinical value Shen Wei 1,a, Chen Juan 2, Li Xiurong 1 and Yin Jie 1 1 Department of Obstetrics and Gynecology,
In-vitro assay for Cytotoxicity activity in ethonolic extract of fruit rind of Couropita Guianensis aubl
 ISSN: 2319-7706 Volume 3 Number 10 (2014) pp. 169-176 http://www.ijcmas.com Original Research Article In-vitro assay for Cytotoxicity activity in ethonolic extract of fruit rind of Couropita Guianensis
ISSN: 2319-7706 Volume 3 Number 10 (2014) pp. 169-176 http://www.ijcmas.com Original Research Article In-vitro assay for Cytotoxicity activity in ethonolic extract of fruit rind of Couropita Guianensis
Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells
 Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells Z.Q. Tian 1, Y.Z. Xu 1, Y.F. Zhang 1, G.F. Ma 1, M. He 1 and
Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells Z.Q. Tian 1, Y.Z. Xu 1, Y.F. Zhang 1, G.F. Ma 1, M. He 1 and
Supplementary Figures
 Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Page 39 of 44. 8h LTA & AT h PepG & AT h LTA
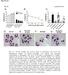 Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Multi-Parameter Apoptosis Assay Kit
 Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Supplementary Data Cyclophilin B Supports Myc and Mutant p53 Dependent Survival of Glioblastoma Multiforme Cells
 Supplementary Data Cyclophilin B Supports Myc and Mutant p53 Dependent Survival of Glioblastoma Multiforme Cells Jae Won Choi, Mark A. Schroeder, Jann N. Sarkaria, and Richard J. Bram 1 Figure S1. Pharmacological
Supplementary Data Cyclophilin B Supports Myc and Mutant p53 Dependent Survival of Glioblastoma Multiforme Cells Jae Won Choi, Mark A. Schroeder, Jann N. Sarkaria, and Richard J. Bram 1 Figure S1. Pharmacological
CHAPTER 4 RESULTS AND DISCUSSION. chemistry, polar substances would dissolve in polar solvents while non-polar substances
 CHAPTER 4 RESULTS AND DISCUSSION 4.1 Extraction yield of Alpinia scabra Samples of A. scabra were dried in an oven as a method of preservation after which the samples were ground to powder. The latter
CHAPTER 4 RESULTS AND DISCUSSION 4.1 Extraction yield of Alpinia scabra Samples of A. scabra were dried in an oven as a method of preservation after which the samples were ground to powder. The latter
HL-60 ATP assay for predicting rat oral toxicity study
 AATEX 14, Special Issue, 699-703 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan HL-60 ATP assay for predicting rat oral toxicity study Yumiko
AATEX 14, Special Issue, 699-703 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan HL-60 ATP assay for predicting rat oral toxicity study Yumiko
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway
 European Review for Medical and Pharmacological Sciences Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway Z. LI 1,2, J. MENG 3, T.-J. XU 4, X.-Y. QIN 5, X.-D.
European Review for Medical and Pharmacological Sciences Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway Z. LI 1,2, J. MENG 3, T.-J. XU 4, X.-Y. QIN 5, X.-D.
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
(14R)-14-hydroxy-4,14-retroretinol (14-HRR)
 Table S7 Atypical retinoids and retinoid-related molecules (RRMs) ame Chemical structure of the ligand(s) Mechanism of action and uses References Anhydroretinol (AR) Modulators of c-raf kinase 1-3 Blocks
Table S7 Atypical retinoids and retinoid-related molecules (RRMs) ame Chemical structure of the ligand(s) Mechanism of action and uses References Anhydroretinol (AR) Modulators of c-raf kinase 1-3 Blocks
Elution of Tumoricidal Doses of Bortezomib from a Resorbable Cement Carrier
 Elution of Tumoricidal Doses of Bortezomib from a Resorbable Cement Carrier Matthew Allen, Vet MB, PhD, Brittani Jones, BS. The Ohio State University, Columbus, OH, USA. Disclosures: M. Allen: 5; Johnson
Elution of Tumoricidal Doses of Bortezomib from a Resorbable Cement Carrier Matthew Allen, Vet MB, PhD, Brittani Jones, BS. The Ohio State University, Columbus, OH, USA. Disclosures: M. Allen: 5; Johnson
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Effect of Organically Grown Curcuma longa (Turmeric) on Leukemic and MCF-7 Cell Lines
 ISSN: 2319-7706 Special Issue-2 (May-2015) pp. 182-186 http://www.ijcmas.com Original Research Article Effect of Organically Grown Curcuma longa (Turmeric) on Leukemic and MCF-7 Cell Lines Priyanka Argade
ISSN: 2319-7706 Special Issue-2 (May-2015) pp. 182-186 http://www.ijcmas.com Original Research Article Effect of Organically Grown Curcuma longa (Turmeric) on Leukemic and MCF-7 Cell Lines Priyanka Argade
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Sample Illustration of the "CompuSyn Report" of Two-Drug Combinations in Vitro
 Sample Illustration of the "CompuSyn Report" of Two-Drug Combinations in Vitro A: Fludelone (FD) in nm (IC 50 was about 2.7 nm), 8 Data Points B: Panaxytriol (PTX) in um (IC 50 was about 3.2 um), 6 Data
Sample Illustration of the "CompuSyn Report" of Two-Drug Combinations in Vitro A: Fludelone (FD) in nm (IC 50 was about 2.7 nm), 8 Data Points B: Panaxytriol (PTX) in um (IC 50 was about 3.2 um), 6 Data
- 1 - Cell types Monocytes THP-1 cells Macrophages. LPS Treatment time (Hour) IL-6 level (pg/ml)
 Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Effects of AFP gene silencing on Survivin mrna expression inhibition in HepG2 cells
 mrna expression inhibition in HepG2 cells Z.L. Fang 1, N. Fang 2, X.N. Han 3, G. Huang 2, X.J. Fu 2, G.S. Xie 2, N.R. Wang 2 and J.P. Xiong 1 1 Department of Medical Oncology, The First Affiliated Hospital
mrna expression inhibition in HepG2 cells Z.L. Fang 1, N. Fang 2, X.N. Han 3, G. Huang 2, X.J. Fu 2, G.S. Xie 2, N.R. Wang 2 and J.P. Xiong 1 1 Department of Medical Oncology, The First Affiliated Hospital
Supporting Information. Non-thermal plasma with 2-deoxy-D-glucose synergistically induces cell death by targeting glycolysis in blood cancer cells
 Supporting Information Non-thermal plasma with 2-deoxy-D-glucose synergistically induces cell death by targeting glycolysis in blood cancer cells Neha Kaushik, 1 Su Jae Lee, 2 Tae Gyu Choi 3, Ku Youn Baik,
Supporting Information Non-thermal plasma with 2-deoxy-D-glucose synergistically induces cell death by targeting glycolysis in blood cancer cells Neha Kaushik, 1 Su Jae Lee, 2 Tae Gyu Choi 3, Ku Youn Baik,
Research on the inhibitory effect of metformin on human oral squamous cell carcinoma SCC-4 and CAL-27 cells and the relevant molecular mechanism.
 Biomedical Research 2017; 28 (14): 6350-6354 ISSN 0970-938X www.biomedres.info Research on the inhibitory effect of metformin on human oral squamous cell carcinoma SCC-4 and CAL-27 cells and the relevant
Biomedical Research 2017; 28 (14): 6350-6354 ISSN 0970-938X www.biomedres.info Research on the inhibitory effect of metformin on human oral squamous cell carcinoma SCC-4 and CAL-27 cells and the relevant
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS
 STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified
 Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Original Article Ginkgo biloba extract induce cell apoptosis and G0/G1 cycle arrest in gastric cancer cells
 Int J Clin Exp Med 2015;8(11):20977-20982 www.ijcem.com /ISSN:1940-5901/IJCEM0015584 Original Article Ginkgo biloba extract induce cell apoptosis and G0/G1 cycle arrest in gastric cancer cells Yu Bai 1,
Int J Clin Exp Med 2015;8(11):20977-20982 www.ijcem.com /ISSN:1940-5901/IJCEM0015584 Original Article Ginkgo biloba extract induce cell apoptosis and G0/G1 cycle arrest in gastric cancer cells Yu Bai 1,
