Epstein-Barr virus and nasopharyngeal carcinoma
|
|
|
- Buddy Mills
- 6 years ago
- Views:
Transcription
1 Chinese Journal of Cancer Review Epstein-Barr virus and nasopharyngeal carcinoma Lawrence S. Young 1 and Christopher W. Dawson 2 Abstract Since its discovery 50 years ago, Epstein-Barr virus (EBV) has been linked to the development of cancers originating from both lymphoid and epithelial cells. Approximately 95% of the world s population sustains an asymptomatic, life-long infection with EBV. The virus persists in the memory B-cell pool of normal healthy individuals, and any disruption of this interaction results in virus-associated B-cell tumors. The association of EBV with epithelial cell tumors, specifically nasopharyngeal carcinoma (NPC) and EBVpositive gastric carcinoma (EBV-GC), is less clear and is currently thought to be caused by the aberrant establishment of virus latency in epithelial cells that display premalignant genetic changes. Although the precise role of EBV in the carcinogenic process is currently poorly understood, the presence of the virus in all tumor cells provides opportunities for developing novel therapeutic and diagnostic approaches. The study of EBV and its role in carcinomas continues to provide insight into the carcinogenic process that is relevant to a broader understanding of tumor pathogenesis and to the development of targeted cancer therapies. Key words Nasopharyngeal carcinoma, EBV, latency, gastric carcinoma Infection is involved in the development of an estimated 20% of human cancers approximately 2 million cases per year [1]. Studies on the role of infection in cancer continue to reveal the underlying mechanisms that drive the oncogenic process and to highlight opportunities for therapeutic and prophylactic intervention. Epstein- Barr virus (EBV) was the first human tumor virus to be discovered and has yielded significant insight into both the pathogenesis of cancer and the natural history of persistent herpesvirus infection. Infection with EBV is very common, affecting all human populations [2,3], and is largely asymptomatic and life-long. Early in the course of primary infection, EBV infects B lymphocytes, though it is unknown where B lymphocytes are infected and whether this infection also involves epithelial cells of the upper respiratory tract. To achieve long-term persistence in vivo, EBV colonizes the memory B-cell pool, where it establishes latent infection characterized by the expression of a limited subset of viral genes known as latent genes [4]. There are several well-described forms of EBV latency, each of which the virus uses at different stages of the viral life cycle and which are also reflected in the patterns of latency observed in Authors Affiliations: 1 Warwick Medical School, The University of Warwick, Coventry, CV4 7AL, UK; 2 School of Cancer Sciences, University of Birmingham, Vincent Drive, Edgbaston, Birmingham, B15 2TT, UK. Corresponding Author: Lawrence S. Young, Warwick Medical School, The University of Warwick, Coventry, CV4 7AL, UK. Tel: ; L.S.Young@warwick.ac.uk. doi: /cjc various EBV-associated malignancies [2,5]. Furthermore, during its life cycle, EBV must periodically replicate to generate infectious virus for transmission to other susceptible hosts, with epithelial sites in the oropharynx and salivary glands appearing to be the major sites for viral replication. Thus, the natural history of persistent EBV infection appears to resemble that of other herpesviruses in requiring distinct cell lineages to manifest the latent and replicative forms of the viral life cycle. The dual tropism of EBV infection is also reflected in the types of tumor associated with the virus. EBV can colonize the memory B-cell pool in vivo and efficiently transform resting B cells into permanent, latently infected lymphoblastoid cell lines (LCLs) in vitro characteristics mirrored in the various malignancies of B-cell origin that are closely associated with EBV infection [2,3,5]. However, it is the undifferentiated form of nasopharyngeal carcinoma (NPC) that shows the most consistent worldwide association with EBV. Furthermore, a subset of gastric adenocarcinomas and certain salivary gland carcinomas are also infected with EBV. Hence, epithelial infection with EBV can result in malignant transformation, suggesting that targeted approaches should be considered for preventive and therapeutic intervention. Historical Background In 1964, Tony Epstein and Yvonne Barr identified herpesvirustype particles by electron microscopy in a subpopulation of Burkitt CACA Chinese Anti-Cancer Association 581
2 lymphoma (BL)-derived tumor cells in vitro. Only two years later, Old et al. [6] described antibodies in the serum of African patients with BL that recognized antigens prepared from BL cell lines. Surprisingly, similar BL antigen-specific antibodies were also present in serum from patients with carcinomas of the post-nasal space, and these antibodies were found in a high proportion of patients from both Africa and the US. This led the authors to suggest, the high frequency of positive sera among patients with carcinoma of the post-nasal space indicates the desirability of searching for similar particles in cultures of this class of tumor also. [6]. A more specific immunofluorescence assay for detecting antibodies against EBVencoded replicative antigens was developed by de Schryver et al. [7], and this assay confirmed the association of elevated antibody titers against EBV-encoded viral capsid antigen (VCA) and membrane antigen (MA) with NPC. In 1970, zur Hausen et al. [8] showed that EBV DNA was present in extracts from NPC tumors using DNA hybridization technology. Further serological analysis showed an association between EBV antibody titers and NPC tumor stage [9] and revealed VCA-specific IgA levels as a potential prognostic indicator [10]. In 1973, EBV DNA was detected by in situ hybridization in NPC tumor cells but not in infiltrating lymphoid cells [11]. Based on the apparent specificity of the association between VCA IgA levels and NPC, Zeng et al. [12] embarked on a mass serological screening program in 1980 in Wuzhou City in China, where these EBV-specific antibodies were found to be useful for early detection of NPC and where subsequent prospective analysis revealed the presence of IgA VCA up to 41 months prior to the clinical diagnosis of NPC. Analysis of terminal repeats (TRs) in the EBV genome in NPCs by Southern blot hybridization demonstrated that resident viral genomes were monoclonal, providing important evidence that EBV infection occurred before the clonal expansion of the malignant cell population [13]. EBV Infection and NPC The World Health Organization (WHO) has classified NPC into two main histological types keratinizing squamous cell carcinoma (type I) and non-keratinizing squamous cell carcinoma (types II and III) based on the tumor cells appearance under the light microscope. The non-keratinizing type is further subdivided into differentiated non-keratinizing carcinoma (type II) and undifferentiated carcinoma (type III), and these type II and type III tumors are predominantly EBV-positive [14,15]. Well-differentiated keratinizing NPC (type I) accounts for less than 20% of all NPC cases worldwide, and this tumor type is relatively rare in southern China [15,16]. However, the association of EBV with the more differentiated, WHO type I form of NPC has been found particularly in the geographical regions with a high incidence of undifferentiated NPC [14]. In NPC, the virus exists in a latent state, exclusively in the tumor cells and absent from the surrounding lymphoid infiltrate. However, the interaction between the prominent lymphoid stroma found in undifferentiated NPC and adjacent carcinoma cells appears to be crucial for the continued growth of malignant NPC cells. Carcinomas with similar features to undifferentiated NPC have been described at other sites including the thymus, tonsils, lungs, stomach, skin, or uterine cervix and are often referred to as undifferentiated carcinomas of nasopharyngeal type (UCNT) or lymphoepitheliomas. The morphological similarities of UCNTs to undifferentiated NPC prompted several groups to examine UCNTs for the presence EBV. UCNTs of the stomach are consistently EBVpositive [17], whereas the association of the other UCNTs with EBV is less strong. EBV was found in thymic epithelial tumors from Chinese but not Western patients [18]. Salivary gland UCNTs are associated with EBV in Greenland Eskimos and Chinese but not in Caucasian patients [19], and several case reports have demonstrated the absence of EBV from UCNTs arising in the uterine cervix and breast [20, 21]. EBV Latent Gene Expression in Virusassociated Tumors Much of our understanding of the biology of EBV relates to its interaction with B lymphocytes. EBV can readily infect and transform normal resting B lymphocytes in vitro, a model system that has provided important insight into the virus biology and behavior. The LCLs generated by in vitro infection of B lymphocytes carry multiple copies of circular, extrachromosomal viral DNA (episomes) in every cell and express a limited set of latent proteins, including 6 nuclear antigens (EBNAs 1, 2, 3A, 3B, 3C, and -LP) and 3 latent membrane proteins (LMPs 1, 2A, and 2B) [2,3,5]. In addition to the latent proteins, LCLs also show abundant expression of the small nonpolyadenylated EBV-encoded small RNAs (EBER) 1 and EBER2; these are expressed in all forms of latent EBV infection and have served as excellent targets to detect EBV in tumors. Transcripts from the BamHI-A region (Bam A) of the viral genome, originally referred to as BamHI-A rightward transcripts (BARTs), are also detected in LCLs, and these, along with transcripts from the BamHI H rightward reading frame 1 (BHRF1) region, encode micrornas (mirnas). This pattern of latent EBV gene expression is referred to as the Latency III form of EBV infection and is characteristic in the majority of EBV-associated lymphomas arising in immunosuppressed patients. Studies examining EBV latent gene expression in virusassociated tumors and cell lines derived from BL biopsies revealed at least 2 additional forms of EBV latency. EBNA1 is the only EBV protein consistently observed in EBV-positive BL tumors, along with the EBER and BamHI-A transcripts; this form of latency is referred to Latency I [2,3,5]. A variant of Latency I, with expression of BHRF1 and EBNAs 3A, 3B, and 3C, is observed in approximately 5% 10% of EBV-positive BL tumors [22]. It appears that in these tumors, the selective pressure to down-regulate EBNA2 expression has occurred via deletion of the EBNA2 gene rather than through the switch in viral promoter usage observed in the conventional BL scenario. Another form of EBV latency, Latency II, was originally identified in biopsies of NPC and subsequently found in cases of EBV-associated Hodgkin lymphoma (HL) (Figure 1) [23-26]. Here, expression of the EBERs, EBNA1, and BamHI-A transcripts is accompanied by expression of LMP1 and LMP2A/B. While this Latency II pattern of EBV latent gene expression is a consistent feature of virus-associated HL, LMP1 expression in NPC is variable, with only approximately 20% of biopsies being unequivocally positive for LMP1 at the protein level [3]. 582 Chin J Cancer; 2014; Vol. 33 Issue 12 Chinese Journal of Cancer
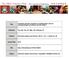
3 EBERs EBNA1 LMP1 LMP2A Figure 1. Epstein-Barr virus (EBV) latent gene expression in nasopharyngeal carcinoma (NPC). In situ hydridization to the abundant EBV-encoded small RNAs (EBER) transcripts (left, upper panel) is the standard approach to detect EBV infection in cells and tissues. Immunohistochemical staining of NPC confirms EBNA1 expression in every tumor cell (right, upper panel). The expression of latent membrane protein 1 (LMP1) and LMP2A in NPC biopsies (lower panels) is more variable. The prominent lymphoid infiltrate in NPC is believed to contribute to the growth and survival of the tumor cells. (magnification, 200) The mechanisms underlying differential LMP1 expression in NPC and the consequent effects on the NPC phenotype remain unknown. These different forms of EBV latent infection are a reflection of the cellular environment and the complex interplay between host regulatory factors and viral promoters that drive EBV latent gene expression. In Latency III, the different EBNAs are encoded by individual mrnas generated by differential splicing of the same long (over 100 kb), rightward primary transcript expressed from one of two promoters (Cp or Wp) located close together in the BamHI-C and BamHI-W regions of the genome [27]. A switch from Wp to Cp occurs early in B-cell infection as a consequence of the transactivating effects of both EBNA1 and EBNA2 on Cp. LMP transcripts are expressed from separate promoters in the BamHI-N region of the EBV genome, with the leftward LMP1 and rightward LMP2B mrnas apparently controlled by the same bidirectional promoter sequence (ED-L1) that also responds to transactivation by EBNA2 [2,27]. The LMP2A promoter is also regulated by EBNA2. Both LMP2A and LMP2B transcripts cross the TRs into the U1 region, thus requiring the circularization of the genome for transcription. Circularization occurs by homologous recombination of the TRs, resulting in fused termini of unique length a feature that has been used as a measure of EBV clonality, on the assumption that fused TRs with an identical number of repeats denote expansions of a single infected progenitor cell [13]. However, EBV clonality postinfection may be a consequence of the selective growth advantage achieved by optimal LMP2A expression over a minimal number of TRs [28]. In the more restricted forms of EBV latency observed in NPC and HL, EBNA1 transcription is driven from the TATA-less Qp promoter [29], and an alternative promoter, L1-TR, located in the terminal repeats is responsible for LMP1 expression [30]. These forms of EBV latency reflect the different Chin J Cancer; 2014; Vol. 33 Issue
4 transcription programs that EBV employs as the virus transits through the germinal center B-cell reaction to finally colonize the resting memory B-cell compartment [4]. The adoption of these forms of latency in EBV-associated tumors is therefore assumed to represent the manifestation of inappropriate viral transcription programs as a consequence of both the host cell environment and the selection pressures associated with oncogenic progression. The Function of EBV Latent Genes Expressed in NPC Understanding the function of the EBV latent genes expressed in NPC is clearly essential in determining the role of viral infection in the carcinogenic process. Furthermore, a detailed appreciation of the function of these EBV latent genes could provide opportunities to develop novel diagnostic and therapeutic approaches. EBNA1 and the LMPs While the EBNA1 protein has an essential role in replication and segregation of the EBV episome, it has recently been found to protect cells from apoptosis, enhance cell survival, and directly contribute to the tumorigenic phenotype [31]. These effects manifest through a variety of mechanisms including destabilization of p53, disruption of promyelocytic leukaemia (PML) nuclear bodies, and modulation of various signaling pathways. LMP1 is the major transforming protein of EBV, behaving as a classical oncogene in rodent fibroblast transformation assays and being essential for EBV-induced B-cell transformation in vitro [32]. LMP1 has pleiotropic effects when expressed in cells, resulting in induction of cell surface adhesion molecules and activation antigens, up-regulation of antiapoptotic proteins (Bcl-2, A20), and stimulation of cytokine production [interleukin (IL)-6, IL-8] [33]. LMP1 functions as a constitutively activated member of the tumor necrosis factor receptor (TNFR) superfamily, activating a number of signaling pathways (NF-κB, ERK-MAPK, JNK/AP1, PI3K) in a ligand-independent manner [34]. The expression of LMP1 in epithelial cells is associated with a range of phenotypic effects including hyperproliferation, induction of proinflammatory cytokines, resistance to apoptosis, and enhanced motility [33]. The role of LMP1 in the pathogenesis of NPC remains speculative. There is considerable variation in the reported expression of LMP1 in NPC biopsies and a general consensus that approximately 20% 40% of tumors express LMP1 at the protein level [3]. In one study, all 6 early, preinvasive NPC (NPC in situ) lesions analyzed expressed the LMP1 protein, arguing for a crticial role of LMP1 in the early pathogenesis of NPC [35]. The possibility of geographical variation in LMP1 expression in NPC has been suggested and is supported by studies in North Africa, where LMP1 is more prevalent in the juvenile form of NPC [36]. The two proteins encoded by the LMP2 gene, LMP2A and LMP2B, share 12 hydrophobic membrane-spanning domains and a short cytoplasmic C-terminus, but it is the unique immunoreceptor tyrosine-based activation motif (ITAM) within the cytoplasmic N-terminal domain of LMP2A that seems responsible for this protein s functional effects [37]. LMP2A appears to drive B-cell proliferation and survival in the absence of signaling through the B-cell receptor (BCR), an effect that may be relevant to the ability of EBV to colonize the memory B-cell pool [38,39]. LMP2A is required for the successful outgrowth of EBV-infected epithelial cells in vitro [28] and can induce anchorage-independent growth, enhance cell adhesion and cell motility, and inhibit epithelial cell differentiation [40-44]. Many of these effects are a consequence of LMP2A s ability to activate the PI3K/ Akt and β-catenin pathways. LMP2A can also induce the epithelialto-mesenchymal transition (EMT), which is associated with the acquisition of stem cell like properties [45]. LMP2A and LMP2B play a role in resistance to the antiviral effects of interferons α and β in epithelial cells [46], and LMP2A modulates NF-κB and STAT signaling in EBV-infected epithelial cells through effects on IL-6 transcription and secretion [47]. Unlike LMP1, LMP2A expression is more consistent in NPC. Studies using reverse transcription-polymerase chain reaction (RT- PCR) confirmed expression of LMP2A mrna in more than 98% of NPC cases, whereas expression of LMP2B was lower and mirrored LMP1 [23,48]. Immunohistochemical staining has confirmed expression of LMP2A protein in more than 50% of NPC cases [45,49]. Interestingly, this consistent expression of LMP2 in NPC is reflected in serological responses such that the majority of NPC patients have detectable IgG responses to LMP2A/2B [50]. EBERs and the BamHI-A region Two small non-polyadenylated (non-coding) RNAs, EBERs 1 and 2, are highly expressed in all forms of EBV latency and serve as sensitive targets for detecting EBV infection in cells and tissues [2,3,5]. The EBERs assemble into stable ribonucleoprotein particles with the auto-antigen La and ribosomal protein L22 and bind the interferoninducible, double-stranded RNA-activated protein kinase protein kinase RNA-activated (PKR) [51]. The interaction of EBERs with retinoic acid inducible gene 1 (RIG-1) induces type I interferon production, an effect that may be counteracted by other viral genes such as LMP1 and LMP2A/B [52]. EBERs are also reported to induce insulin-like growth factor 1 (IGF-1) in NPC cell lines [53]. This effect is associated with enhanced growth of NPC cell lines and its relevance to NPC is supported by the frequent expression of IGF-1 in tumor biopsies. A group of abundantly expressed RNAs encoded by the BamHI-A region of the EBV genome were originally identified in NPC but were subsequently found to be expressed in other EBVassociated malignancies and in the peripheral blood of healthy individuals [2,3,5,54,55]. These highly spliced transcripts, commonly referred to as the BARTs (BamHI-A rightward transcripts), are a cluster of 22 mirnas that generate 44 mature mirnas [56]. These mirnas target and thereby regulate a variety of viral and cellular transcripts [55, 57]. Thus, specific BART mirnas have been found to protect against apoptosis and contribute to immune evasion. The BART mirnas also appear crucial in the regulation of EBV gene expression, targeting both latent (LMP1, LMP2A) and lytic genes (BALF5). Because of their stability in serum, mirnas have been 584 Chin J Cancer; 2014; Vol. 33 Issue 12 Chinese Journal of Cancer
5 recognized as potential cancer biomarkers, and recent studies support the possible use of serum BART mirnas in the detection and prognosis of NPC [58, 59]. It appears that the BART mirnas in serum reside in cell-derived exosomes that are able to deliver mirnas to other non-ebv infected cells [60, 61]. This raises the intriguing possibility that BART mirnas may contribute to the development of NPC by influencing the behavior of stromal and immune cells. Another transcript generated from the BamHI-A region is BARF1, which encodes a 31-kDa protein that was originally identified as an early antigen expressed upon induction of the EBV lytic cycle. However, BARF1 is a secreted protein expressed during latency in EBV-associated NPC and gastric carcinoma (GC) [62,63]. BARF1 shares limited homology with the human colony stimulating factor 1 (CSF-1) receptor (c-fms oncogene) and competes for its natural ligand, macrophage colony-stimulating factor (M-CSF or CSF-1), thereby modulating immune cell growth and function [64]. BARF1 displays oncogenic activity when expressed in rodent fibroblasts and simian primary epithelial cells [65]. Engineered expression of BARF1 in the context of a recombinant EBV enhanced the growth and tumorigenicity of virus-negative NPC cell lines, implicating BARF1 in the pathogenesis of NPC [66]. In addition, serum BARF1 may be a useful diagnostic marker for NPC [67]. EBV Strain Variation EBV isolates from different regions of the world or from patients with different virus-associated diseases are remarkably similar when their genomes are compared with restriction fragment length polymorphism analysis [3,5,68]. However, variations in repeat regions of the EBV genome are observed among different EBV isolates. Analysis of the EBV genome in a number of BL cell lines revealed gross deletions, some of which account for biological differences. More specifically, P3HR-1 virus, which is non-transforming, has a deletion of the EBNA2-encoding gene [3, 5, 68]. Strain variation over the EBNA2-encoding (BamHI-WYH) region of the EBV genome permits all virus isolates to be classified as either type 1 (EBV-1, B95.8- like) or type 2 (EBV-2, Jijoye-like) [68]. This genomic variation results in the production of two antigenically distinct forms of the EBNA2 protein, which share only 50% amino acid homology. Similar allelic polymorphisms (with 50% 80% sequence homology, depending on the locus) related to the EBV type occur in a subset of latent genes, namely those encoding EBNA-LP, EBNA3A, EBNA3B, and EBNA3C [69]. These differences have functional consequences as EBV-2 isolates are less efficient in B-lymphocyte transformation assays in vitro compared with EBV-1 isolates [70]. A combination of virus isolation and sero-epidemiological studies suggests that type 1 virus isolates are predominant (but not exclusively so) in many Western countries, whereas both types are widespread in equatorial Africa, New Guinea, and perhaps certain other regions [68, 71]. In addition to this broad distinction between EBV types 1 and 2, there is also minor heterogeneity within each virus type. Individual strains have been identified on the basis of changes compared with B95.8, ranging from single base mutations to extensive deletions [68]. While infection with multiple strains of EBV was originally thought to be confined to immunologically compromised patients, more recent studies demonstrate that normal, healthy patients who are seropositive can be infected with multiple EBV isolates that vary in abundance and presence over time [72]. Co-infection of the host with multiple virus strains could have evolutionary benefit to EBV, creating diversity through genetic recombination. Such intertypic recombination has been reported in HIV-infected patients and in the Chinese population, and appears to arise via recombination of multiple EBV strains during the intense EBV replication that results from immunosuppression [73]. The possible contribution of EBV strain variation to virusassociated tumors remains contentious. Many studies have failed to establish an epidemiological association between EBV strains and disease and suggest that the specific EBV gene polymorphisms detected in virus-associated tumors occur with similar frequencies in EBV isolates from healthy virus carriers from the same geographic region [3,71]. However, these studies focused on specific regions of the EBV genome rather than on comparing the entire viral DNA sequence. More recent work has used next-generation sequencing (NGS) to analyze EBV isolates from NPC biopsies. These studies have confirmed that, while there is a high level of overall similarity among the NPC-derived virus strains with the prototypical EBV genome, variation exists in viral genes that might result in functional differences [74,75]. In this regard, an LMP1 variant containing a 10 amino acid deletion (residues 343 to 352) was originally identified in Chinese NPC biopsies and has oncogenic and other functional properties distinct from those of the B95.8 LMP1 gene [76-78]. It is therefore likely that variation in LMP1 and other EBV genes can contribute to the risk of developing virus-associated tumors. However, more biological studies using well-defined EBV variants are required, along with more detailed NGS comparisons of tumor-derived EBV strains versus those derived from healthy donors. NPC Pathogenesis The presence of monoclonal EBV episomes in NPC indicates that viral infection precedes the clonal expansion of malignant cells [13]. However, epithelial infection may not be the initiating event in virusassociated carcinogenesis, as tonsils from patients with infectious mononucleosis (IM) and normal nasopharyngeal biopsies from individuals at high risk of developing NPC lack evidence of epithelial EBV infection [2]. EBV infection as detected by in situ hybridization to the abundantly expressed non-polyadenylated EBER RNAs has been demonstrated in high-grade (severe dysplastic and carcinoma in situ), preinvasive lesions in the nasopharynx, but not in low-grade disease or histologically normal nasopharyngeal epithelium [35,79]. Both high-grade and carcinoma in situ lesions carry monoclonal EBV genomes [35]. Multiple genetic changes have been found in NPC, with frequent deletion of regions on chromosomes 3p, 9p, 11q, 13q, and 14q and promoter hypermethylation of specific genes on chromosomes 3p (RASSF1A, RARβ2) and 9p (p16, p15, p14, DAP-kinase) [80,81]. Both 3p and 9p deletions have been identified in the absence of EBV infection in low-grade dysplastic lesions and in normal nasopharyngeal epithelium from individuals at high risk Chin J Cancer; 2014; Vol. 33 Issue
6 of developing NPC, suggesting that these genetic events occur early in NPC pathogenesis and may predispose to subsequent EBV infection [79, 82]. This possibility is supported by in vitro data demonstrating that stable EBV infection of epithelial cells requires an altered, undifferentiated cellular environment [83] and that cyclin D1 overexpression (a consequence of p16 deletion on chromosome 9p and amplification of the cyclin D1 locus on chromosome 11q) facilitates persistent EBV infection of immortalized nasopharyngeal epithelial cells [84]. Thus, a scheme has been proposed whereby loss of heterozygosity occurs early in NPC pathogenesis because of exposure to environmental cofactors such as dietary components (i.e., salted fish), creating low-grade, preinvasive lesions that become susceptible to EBV infection after additional genetic and epigenetic events (Figure 2). Once infected, EBV latent genes provide growth and survival benefits, resulting in the development of NPC. Additional genetic and epigenetic changes occur after EBV infection. EBV and GC GC remains the second leading cause of death from cancer. Although incidence and mortality have been falling for over 50 years, the number of deaths from this tumor will inevitably continue to increase globally because of population growth and aging in highrisk countries. As approximately 10% of GC comprise cells latently infected with EBV, EBV-associated GC (EBV-GC) may represent the most common form of EBV-associated malignancy [85, 86]. These EBV- GC tumors display a restricted pattern of EBV latent gene expression (EBERs, EBNA1, LMP2A, the BART mirnas, BARF1 ), similar to NPC [63,87]. The association of EBV and GC varies significantly with geography, which may be attributed to ethnic and genetic differences [85,86]. EBV-GCs have distinct phenotypic and clinical characteristics compared to EBV-negative GC tumors, including loss of p16 expression, promoter methylation of p73, expression of wildtype p53, distinct patterns of allelic loss, and improved overall survival time [88-91]. Comprehensive genomic and molecular characterization of GC has confirmed that EBV-GC is a distinct entity with a high CpG island methylator (CIMP) phenotype and apparent driver mutations in the PIK3CA and ARID1A genes [92, 93]. As in NPC, the precise role of EBV in the pathogenesis of GC remains to be determined, but the absence of EBV infection Normal epithelium Low-grade preinvasive lesion High-grade preinvasive lesion Nasopharyngeal carcinoma HE EBER Metastasis LOH on chromosome 3p and 9p Inactivation of RASSF1A and CDKN2A EBV latent infection Telomerase dysregulation BCL2 overexpression LOH on 14q, 11q, 13q and 16a Inactivation of EDNRB and TSLC1? Other genetic changes (for example, in TP53 and E-cadherin) Figure 2. Schematic representation of NPC pathogenesis. This model proposes that loss of heterozygosity (LOH) occurs early in NPC pathogenesis, possibly as a result of exposure to environmental cofactors such as dietary components (e.g., salted fish). This results in low-grade, preinvasive lesions that, after additional genetic and epigenetic events, become susceptible to stable EBV infection. Once cells have become infected, EBV latent genes provide growth and survival benefits, resulting in the development of NPC. Additional genetic and epigenetic changes occur after EBV infection. 586 Chin J Cancer; 2014; Vol. 33 Issue 12 Chinese Journal of Cancer
7 in premalignant gastric lesions supports the contention that viral infection is a relatively late event in gastric carcinogenesis [94]. Epidemiological studies suggest that EBV-GC is related to birth order, high salt intake, and exposure to metal dust, but these factors may vary geographically (e.g., between Japan and Colombia), supporting the need for more detailed investigation [85, 86]. Distinct EBV strains are suggested to play a role in EBV-GC development, though this could reflect geographical variation in the prevalence of EBV strains in the population [95]. Conclusions EBV was discovered over 50 years ago, and its DNA was fully sequenced in The widespread nature of EBV infection testifies to the intimate interaction between the virus and the immune system that is characterised by an asymptomatic, life-long infection resident within the memory B-cell compartment. Perturbing this interaction, as occurs in various forms of immunosuppression, results in EBVassociated B-cell tumors. As for the association of EBV with NPC and EBV-GC, the contribution of the virus is less clear but appears to be a consequence of the aberrant establishment of virus latency References [1] de Martel C, Ferlay J, Franceschi S, et al. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis. Lancet Oncol, 2012,13: [2] Young LS, Rickinson AB. Epstein-Barr virus: 40 years on. Nat Rev Cancer, 2004,4: [3] Tao Q, Young LS, Woodman CB, et al. Epstein-Barr virus (EBV) and its associated human cancers genetics, epigenetics, pathobiology and novel therapeutics. Front Biosci, 2006,11: [4] Thorley-Lawson DA. Epstein-Barr virus: exploiting the immune system. Nat Rev Immunol, 2001,1: [5] Rickinson AB, Kieff E. Epstein-Barr Virus. In: Knipe DM, Howley PM, eds. Fields Virology. Philadelphia: Lippincott Williams and Wilkins, 2001: [6] Old LJ, Boyse EA, Oettgen E, et al. Precipitating antibody in human serum to an antigen prersent in cultured Burkitt s lymphoma cells. Proc Natl Acad Sci U S A, 1966,56: [7] de Schryver A, Friberg S, Klein G, et al. Epstein-Barr virusassociated antibody patterns in carcinoma of the post-nasal space. Clin Exp Immunol, 1969,5: [8] zur Hausen H, Schulte-Holthausen H, Klein G, et al. EBV DNA in biopses of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature, 1970,228: [9] Henle W, Henle G, Ho HC, et al. Antibodies to Epstein-Barr virus in nasopharyngeal carcinoma, other head and neck neoplasms, and control groups. J Natl Cancer Inst, 1970,44: [10] Henle W, Ho JH, Henle G, et al. Nasopharyngeal carcinoma: significance of changes in Epstein-Barr virus-related antibody patterns following therapy. Int J Cancer, 1977,20: [11] Wolf H, zur Hausen H, Becker V. EB-viral genomes in epitelial nasopharyngeal carcinoma cells. Nature New Biol, 1973,244:245- in epithelial cells that have already undergone premalignant genetic changes. Whatever the nature of these interactions and the precise role of EBV in the carcinogenic process, there is clearly the opportunity to exploit this association for the clinical benefit of patients. Novel therapeutic approaches using virus reactivation [96], gene therapy [97], or therapeutic vaccination [98] augur well for our ability to effectively target EBV-associated carcinomas, and mass screening programs to identify patients with early-stage NPC are already underway in Hong Kong [99]. These studies are paradigms for the development of targeted cancer therapies and diagnostics and further confirm the far-reaching value of tumor virology to the whole cancer field. Acknowledgments The authors studies are supported by Cancer Research UK and the European Commission s FP6 Life-Sciences-Health Programme (INCA Project: LSHC-CT ). We thank Mu-Sheng Zeng for helpful comments and for providing the EBER in situ hybridisation panel in Figure 1. Received: ; acdepted: [12] Zeng Y, Zhang LG, Wu YC, et al. Prospective studies on nasopharyngeal carcinoma in Epstein-Brr vírus IgA/VCA antibodypositive persons in Wuzhou City, China. Int J Cancer, 1985,36: [13] Raab-Traub N, Flynn K. The structure of the termini of the Epstein- Barr virus as a marker of clonal cellular proliferation. Cell, 1986,47: [14] Pathmanathan R, Prasad U, Chandrika G, et al. Undifferentiated, nonkeratinizing, and squamous cell carcinoma of the nasopharynx. Variants of Epstein-Barr virus neoplasia. Am J Pathol, 1995,146: [15] Wei K, Xu Y, Liu J, et al. Histopathological classification of nasopharyngeal carcinoma. Asian Pacific J Cancer Prev, 2011,12: [16] Ji X, Zhang W, Xie C, et al. Nasopharyngeal carcinoma risk by histologic type in central China: impact of smoking, alcohol and family history. Int J Cancer, 2011,129: [17] Shibata D, Tokunaga M, Uemura Y, et al. Association of Epstein- Barr virus with undifferentiated gastric carcinomas with intense lymphoid infiltration. Lymphoepithelioma-like carcinoma. Am J Pathol, 1991,139: [18] Fujii T, Kawai T, Saito K, et al. EBER-1 expression in thymic carcinoma. Acta Pathol Jpn, 1993,43: [19] Raab-Traub N, Rajadurai P, Flynn K, et al. Epstein-Barr virus infection in carcinoma of the salivary gland. J Virol, 1991,65: [20] Weinberg E, Hoisington S, Eastman AY, et al. Uterine cervical lymphoepithelial-like carcinoma. Absence of Epstein-Barr virus genomes. Am J Clin Pathol, 1993,99: Chin J Cancer; 2014; Vol. 33 Issue
8 [21] Dadmanesh F, Peterse JL, Sapino A, et al. Lymphoepitheliomalike carcinoma of the breast: lack of evidence of Epstein-Barr virus infection. Histopathology, 2001,38: [22] Kelly G, Bell A, Rickinson AB. Epstein-Barr virus-associated Burkitt lymphomagenesis selects for downregulation of the nuclear antigen EBNA1. Nat Med, 2002,8: [23] Brooks L, Yao QY, Rickinson AB, et al. Epstein-Barr virus latent gene transcription in nasopharyngeal carcinoma cells: coexpression of EBNA1, LMP1, and LMP2 transcripts. J Virol, 1992,66: [24] Deacon EM, Pallesen G, Niedobitek G, et al. Epstein-Barr virus and Hodgkin s disease: transcriptional analysis of virus latency in the malignant cells. J Exp Med, 1993,177: [25] Pallesen G, Hamilton-Dutoit SJ, Rowe M, et al. Expression of Epstein-Barr virus latent gene products in tumour cells of Hodgkin s disease. Lancet, 1991,337: [26] Young LS, Dawson CW, Clark D, et al. Epstein-Barr virus gene expression in nasopharyngeal carcinoma. J Gen Virol, 1988,69: [27] Speck SH, Strominger JL. Transcription of Epstein-Barr virus in latently infected, growth-transformed lymphocytes. In: Klein G, ed. Advances in viral oncology. New York: Raven Press, 1989: [28] Moody CA, Scott RS, Su T, et al. Length of Epstein-Barr virus termini as a determinant of epithelial cell clonal emergence. J Virol, 2003,77: [29] Tempera I, Klichinsky M, Lieberman PM. EBV latency types adopt alternative chromatin conformations. PLoS Pathog, 2011,7:e [30] Chen H, Lee JM, Zong Y, et al. Linkage between STAT regulation and Epstein-Barr virus gene expression. J Virol, 2001,75: [31] Frappier L. Role of EBNA1 in NPC tumourigenesis. Sem Cancer Biol, 2012,22: [32] Wang D, Liebowitz D, Kieff E. An EBV membrane protein expressed in immortalised lymphocytes transforms established rodent cells. Cell, 1985,43: [33] Dawson CW, Port RJ, Young LS. The role of EBV-encoded latent membrane proteins LMP1 and LMP2 in the pathogenesis of nasopharyngeal carcinoma. Sem Cancer Biol, 2012,22: [34] Mosialos G, Birkenbach M, Yalamanchili R, et al. The Epstein-Barr virus transforming protein LMP1 engages signaling proteins for the tumor necrosis factor receptor family. Cell, 1995,80: [35] Pathmanathan R, Prasad U, Sadler R, et al. Clonal proliferations of cells infected with Epstein-Barr virus in preinvasive lesions related to nasopharyngeal carcinoma. N Engl J Med, 1995, 333: [36] Khabir A, Karray H, Rodriguez S, et al. EBV latent membrane protein 1 abundance correlates with patient age but not with metastatic behavior in North African nasopharyngeal carcinomas. Virol J, 2005,2:39. [37] Fruehling S, Longnecker R. The immunoreceptor tyrosine-based activation motif of Epstein-Barr virus LMPA2 is essential for blocking BCR-mediated signal transduction. J Virol, 1997,235: [38] Miller CL, Burkhardt AL, Lee JH, et al. Integral membrane protein 2 of Epstein-Barr virus regulates reactivation from latency through dominant negative effects on protein-tyrosine kinases. Immunity, 1995,2: [39] Caldwell RG, Wilson JB, Anderson SJ, et al. Epstein-Barr virus LMP2A drives B-cell development and survival in the absence of normal B cell receptor signals. Immunity, 1998, [40] Scholle F, Bendt KM, Raab-Traub N. Epstein-Barr virus LMP2A transforms epithelial cells, inhibits cell differentiation, and activates Akt. J Virol, 2000,74: [41] Morrison JA, Raab-Traub N. Roles of the ITAM and PY motifs of Epstein-Barr virus latent membrane protein 2A in the inhibition of epithelial cell differentiation and activation of beta-catenin signaling. J Virol, 2005,79: [42] Fukuda M, Longnecker R. Epstein-Barr virus latent membrane protein 2A mediates transformation through constitutive activation of the Ras/PI3-K/Akt pathway. J Virol, 2007,81: [43] Allen MD, Young LS, Dawson CW. The Epstein-Barr virus-encoded LMP2A and LMP2B proteins promote epithelial cell spreading and motility. J Virol, 2005,79: [44] Lu J, Lin WH, Chen SY, et al. Syk tyrosine kinase mediates Epstein- Barr virus latent membrane protein 2A-induced cell migration in epithelial cells. J Biol Chem, 2006,281: [45] Kong QL, Hu LJ, Cao JY, et al. Epstein-Barr virus-encoded LMP2A induces an epithelial-mesenchymal transition and increases the number of side population stem-like cancer cells in nasopharyngeal carcinoma. PLoS Pathog, 2010, 6:e [46] Shah KM, Stewart SE, Wei W, et al. The EBV-encoded latent membrane proteins, LMP2A and LMP2B, limit the actions of interferon by targeting interferon receptors for degradation. Oncogene, 2009,28: [47] Stewart S, Dawson CW, Takada K, et al. Epstein-Barr virus-encoded LMP2A regulates viral and cellular gene expression by modulation of the NF-kappaB transcription factor pathway. Proc Natl Acad Sci U S A, 2004,101: [48] Busson P, McCoy R, Sadler R, et al. Consistent transcription of the Epstein-Barr virus LMP2 gene in nasopharyngeal carcinoma. J Virol, 1992,66: [49] Heussinger N, Büttner M, Ott G, et al. Expression of the Epstein-Barr virus (EBV)-encoded latent membrane protein 2A (LMP2A) in EBVassociated nasopharyngeal carcinoma. J Pathol, 2004,203: [50] Paramita DK, Fatmawati C, Juwana H, et al. Humoral immune responses to Epstein-Barr virus encoded tumor associated proteins and their putative extracellular domains in nasopharyngeal carcinoma patients and regional controls. J Med Virol, 2011,4: [51] Takada K. Role of EBER and BARF1 in nasopharyngeal carcinoma (NPC) tumorigenesis. Sem Cancer Biol, 2012,22: [52] Samanta M, Iwakiri D, Kanda T, et al. EB virus-encoded RNAs are recognised by RIG-1 and activate signalling to induce type I IFN. EMBO J, 2006,25: [53] Iwakiri D, Sheen TS, Chen JY, et al. Epstein-Barr virus-encoded small RNA induces insulin-like growth factor 1 and supports growth of nasopharyngeal carcinoma-derived cell lines. Oncogene, 2005, 24: Chin J Cancer; 2014; Vol. 33 Issue 12 Chinese Journal of Cancer
9 [54] Hitt MM, Allday MJ, Hara T, et al. EBV gene expression in an NPCrelated tumour. EMBO J, 1989,8: [55] Marquitz AR, Raab-Traub N. The role of mirnas and EBV BARTS in NPC. Sem Cancer Biol, 2012,22: [56] Pfeffer S, Zavolan M, Grasser FA, et al. Identification of virusencoded micrornas. Science, 2004,304: [57] Lo AKF, Dawson CW, Jin DY, et al. The pathological roles of BART mirnas in nasopharyngeal carcinoma. J Pathol, 2012,227: [58] Wong AM, Kong KL, Tsang JW, et al. Profiling of Epstein-Barr virus-encoded micrornas in nasopharyngeal carcinoma reveals potential biomarkers and oncomirs. Cancer, 2012,118: [59] Chan JY, Gao W, Ho WK, et al. Overexpression of Epstein- Barr virus-encoded microrna-bart7 in undifferentiated nasopharyngeal carcinoma. Anticancer Res, 2012,32: [60] Pegtel DM, Cosmopoulos K, Thorley-Lawson DA, et al. Functional delivery of viral mirnas via exosomes. Proc Natl Acad Sci U S A, 2010,107: [61] Meckes DG, Shair KHY, Marquitz AR, et al. Human tumor virus utilizes exosomes for intercellular communication. Proc Natl Acad Sci U S A, 2010,107: [62] Decaussin G, Sbih-Lammali F, de Turenne-Tessier M, et al. Expression of BARF1 gene encoded by Epstein-Barr virus in nasopharyngeal carcinoma biopsies. Cancer Res, 2000,60; [63] zur Hausen A, Brink AA, Craanen ME, et al. Unique transcription pattern of Epstein-Barr virus (EBV) in EBV-carrying gastric adenocarcinomas: expression of the transforming BARF1 gene. Cancer Res, 2000,60: [64] Shim AHR, Chang RA, Chen X, et al. Multipronged attenuation of macrophage-colony stimulating factor signaling by Epstein-Barr virus BARF1. Proc Natl Acad Sci U S A, 2012,109: [65] Sheng W, Decaussin G, Sumner S, et al. N-terminal domain of BARF1 gene encoded by Epstein-Barr virus is essential for malignant transformation of rodent fibroblasts and activation of BCL-2. Oncogene, 2001,20: [66] Seto E, Ooka T, Middeldorp J, et al. Reconstruction of nasopharyngeal carcinoma-type infection induced tumorigenicity. Cancer Res, 2008,68: [67] Houali K, Wang X, Shimizu Y, et al. A new diagnostic marker for secreted Epstein-Barr virus-encoded LMP1 and BARF1 oncoproteins in the serum and saliva of patients with nasopharyngeal carcinoma. Clin Cancer Res, 2007,13: [68] Tzellos S, Farrell PJ. Epstein-Barr virus sequence variation biology and disease. Pathogens, 2012,1: [69] Sample J, Young L, Martin B, et al. Epstein-Barr virus types 1 and 2 differ in their EBNA-3A, EBNA-3B, and EBNA-3C genes. J Virol, 1990,64: [70] Rickinson AB, Young LS. Rowe M. Influence of the Epstein-Barr vírus nuclear antigen EBNA2 on the growth phenotype of virustransformed B cells. J Virol, 1987, 61: [71] Khanim F, Yao QY, Niedobitek G, et al. Analysis of Epstein- Barr virus gene polymorphisms in normal donors and in virusasssociated tumors from different geographic locations. Blood, 1996,88: [72] Tierney RJ, Edwards RH, Sitki-Green, D, et al. Multiple Epstein-Barr virus strains in patients with infectious mononucleosis: comparison of ex vivo samples with in vitro isolates by use of heteroduplex tracking assays. J Infect Dis, 2006,193: [73] Midgley RS, Blake NW, Yao QY, et al. Novel intertypic recombinants of Epstein-Barr virus in the Chinese population. J Virol, 2000,74: [74] Liu P, Fang X, Feng Z, et al. Direct sequencing and characterization of a clinical isolate of Epstein-Barr virus from nasopharyngeal carcinoma tissue by using next-generation sequencing technology. J Virol, 2011,85: [75] Kwok H, Wu CW, Palser AL, et al. Genomic diversity of Epstein-Barr virus genomes isolated from primary nasopharyngeal carcinoma biopsy samples. J Virol, 2014,88: [76] Edwards RH, Seillier-Moiseiwitsch F, Raab-Traub N. Signature amino acid changes in latent membrane protein 1 distinguish Epstein-Barr virus strains. Virology, 1999,261: [77] Dawson CW, Eliopoulos AG, Blake SM, et al. Identification of functional differences between prototype Epstein-Barr virusencoded LMP1 and a nasopharyngeal carcinoma-derived LMP1 in human epithelial cells. Virology, 2000,272: [78] Fielding CA, Sandvej K, Mehl AM, et al. Epstein-Barr virus LMP-1 natural sequence variants differ in their potential to activate cellular signaling pathways. J Virol, 2001,75: [79] Chan ASC, To KF, Lo KW, et al. High frequency of chromosome 3p deletion in histologically normal nasopharyngeal epithelia from Southern Chinese. Cancer Res, 2000,60: [80] Lo KW, Chung GTY, To KF. Deciphering the molecular genetic basis of NPC through molecular, cytogenetic, and epigenetic approaches. Sem Cancer Biol, 2012,22: [81] Lung HL, Cheung AKL, Ko JMY, et al. Deciphering the molecular genetic basis of NPC through functional approaches. Sem Cancer Biol, 2012,22: [82] Chan ASC, To KF, Lo KW, et al. Frequent chromosome 9p losses in histologically normal nasopharyngeal epithelia from Southern Chinese. Int J Cancer, 2002,102: [83] Knox PG, Li QX, Rickinson AB, Young LS. In vitro production of stable Epstein-Barr virus-positive epithelial cell clones which resemble the virus: cell interaction observed in nasopharyngeal carcinoma. Virology, 1996,215: [84] Tsang CM, Yip YL, Lo KW, et al. Cyclin D1 overexpression supports stable EBV infection in nasopharyngeal epithelial cells. Proc Natl Acad Sci U S A, 2012,109:E3473-E3482. [85] Lee JH, Kim SH, Han SH, et al. Clinicopathological and molecular characteristics of Epstein-Barr virus-associated gastric carcinoma: a meta-analysis. J Gastroent Hepatol, 2009,24: [86] Murphy G, Pfeiffer R, Camargo MC, et al. Meta-analysis show that prevalence of Epstein-Barr virus-positive gastric cancer differs based on sex and anatomic location. Gastroenterology, 2009,137: [87] Imai S, Koizumi S, Sugiura M, et al. Gastric carcinoma: monoclonal epithelial malignant cells expressing Epstein-Barr virus latent infection protein. Proc Natl Acad Sci U S A, 1994,91: [88] Lee HS, Chang MS, Yang HK, et al. Epstein-Barr virus-positive Chin J Cancer; 2014; Vol. 33 Issue
10 gastric carcinoma has a distinct protein expression profile in comparison with Epstein-Barr virus-negative carcinoma. Clin Cancer Res, 2004, 10: [89] Schneider BG, Gulley ML, Eagan P, et al. Loss of p16/cdkn2a tumour suppressor protein in gastric adenocarcinoma is associated with Epstein-Barr virus and anatomic location in the body of the stomach. Hum Pathol, 2000,31: [90] Ushiku T, Chong JM, Uozaki H, et al. p73 gene promoter methylation in Epstein-Barr virus-associated gastric carcinoma. Int J Cancer, 2006,120: [91] van Rees BP, Caspers E, zur Hausen A, et al. Different pattern of allelic loss in Epstein-Barr virus-positive gastric cancer with emphasis on the p53 tumour suppressor pathway. Am J Pathol, 2002,161: [92] Wang K, Yuen ST, Lee SP, et al. Whole-genome sequencing and comprehensive molecular profiling identify new driver mutations in gastric cancer. Nat Genet, 2014, 46: [93] Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature, 2014, 513: [94] zur Hausen A, van Rees BP, van Beek J, et al. Epstein-Barr virus in gastric carcinomas and gastric stump carcinomas: a late event in gastric carcinogenesis. J Clin Pathol, 2004, 57: [95] Corvalan A, Ding S, Koriyama C, et al. Association of a distinctive strain of Epstein-Barr virus with gastric cancer. Int J Cancer, 2006, 118: [96] Wildeman MA, Novalic Z, Verkuijlen SA, et al. Cytolytic virus activation therapy for Epstein-Barr virus-driven tumors. Clin Cancer Res, 2012,18: [97] Li JH, Shi W, Chia M, et al. Efficacy of targeted FasL in nasopharyngeal carcinoma. Mol Ther, 2003,8: [98] Taylor GS, Jia H, Harrington K, et al. A recombinant modified vaccinia ankara vaccine encoding Epstein-Barr virus (EBV) target antigens: a phase I trial in UK patients with EBV-positive cancer. Clin Cancer Res, 2014,20: [99] Chan KC, Hung EC, Woo JK, et al. Early detection of nasopharyngeal carcinoma by plasma Epstein-Barr virus DNA analysis in a surveillance program. Cancer, 2013, 119: Chin J Cancer; 2014; Vol. 33 Issue 12 Chinese Journal of Cancer
EBV infection B cells and lymphomagenesis. Sridhar Chaganti
 EBV infection B cells and lymphomagenesis Sridhar Chaganti How EBV infects B-cells How viral genes influence the infected B cell Differences and similarities between in vitro and in vivo infection How
EBV infection B cells and lymphomagenesis Sridhar Chaganti How EBV infects B-cells How viral genes influence the infected B cell Differences and similarities between in vitro and in vivo infection How
An Update on Epstein-Barr Virus and Nasopharyngeal Carcinoma.
 -1- An Update on Epstein-Barr Virus and Nasopharyngeal Carcinoma. Epstein-Barr Virus Bayardo Perez-Ordoñez, MD, FRCPC Department of Pathology University Health Network Department of Pathobiology and Laboratory
-1- An Update on Epstein-Barr Virus and Nasopharyngeal Carcinoma. Epstein-Barr Virus Bayardo Perez-Ordoñez, MD, FRCPC Department of Pathology University Health Network Department of Pathobiology and Laboratory
Nasopharynx. 1. Introduction. 1.1 General Information and Aetiology
 Nasopharynx 1. Introduction 1.1 General Information and Aetiology The nasopharynx is the uppermost, nasal part of the pharynx. It extends from the base of the skull to the upper surface of the soft palate.
Nasopharynx 1. Introduction 1.1 General Information and Aetiology The nasopharynx is the uppermost, nasal part of the pharynx. It extends from the base of the skull to the upper surface of the soft palate.
Head and Neck Squamous Subtypes
 1 Head and Neck Squamous Subtypes Adel K. El-Naggar, M.D., Ph.D. The University of Texas MD Anderson Cancer Center, Houston, Texas HNSCC 5 th -6 th most common cancer 400,000/year 50% mortality Considerable
1 Head and Neck Squamous Subtypes Adel K. El-Naggar, M.D., Ph.D. The University of Texas MD Anderson Cancer Center, Houston, Texas HNSCC 5 th -6 th most common cancer 400,000/year 50% mortality Considerable
Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
Herpesviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Gastric Carcinoma with Lymphoid Stroma: Association with Epstein Virus Genome demonstrated by PCR
 Gastric Carcinoma with Lymphoid Stroma: Association with Epstein Virus Genome demonstrated by PCR Pages with reference to book, From 305 To 307 Irshad N. Soomro,Samina Noorali,Syed Abdul Aziz,Suhail Muzaffar,Shahid
Gastric Carcinoma with Lymphoid Stroma: Association with Epstein Virus Genome demonstrated by PCR Pages with reference to book, From 305 To 307 Irshad N. Soomro,Samina Noorali,Syed Abdul Aziz,Suhail Muzaffar,Shahid
Epstein-Barr virus and immunity
 Epstein-Barr virus and immunity Elena Kashuba, PhD Associate Professor Department of Microbiology, Tumor and Cell Biology (MTC) Karolinska Institutet 1 Epstein-Barr virus EBV Everybody s virus 200 nm Enveloped
Epstein-Barr virus and immunity Elena Kashuba, PhD Associate Professor Department of Microbiology, Tumor and Cell Biology (MTC) Karolinska Institutet 1 Epstein-Barr virus EBV Everybody s virus 200 nm Enveloped
W orldwide, gastric carcinoma is one of the leading
 487 ORIGINAL ARTICLE Epstein-Barr virus in gastric carcinomas and gastric stump carcinomas: a late event in gastric carcinogenesis A zur Hausen, B P van Rees, J van Beek, M E Craanen, E Bloemena, G J A
487 ORIGINAL ARTICLE Epstein-Barr virus in gastric carcinomas and gastric stump carcinomas: a late event in gastric carcinogenesis A zur Hausen, B P van Rees, J van Beek, M E Craanen, E Bloemena, G J A
THE Epstein Barr virus (EBV) is an important factor
 Vol. 333 No. 11 EBV IN PREINVASIVE LESIONS RELATED TO NASOPHARYNGEAL CARCINOMA 693 CLONAL PROLIFERATIONS OF CELLS INFECTED WITH EPSTEIN BARR VIRUS IN PREINVASIVE LESIONS RELATED TO NASOPHARYNGEAL CARCINOMA
Vol. 333 No. 11 EBV IN PREINVASIVE LESIONS RELATED TO NASOPHARYNGEAL CARCINOMA 693 CLONAL PROLIFERATIONS OF CELLS INFECTED WITH EPSTEIN BARR VIRUS IN PREINVASIVE LESIONS RELATED TO NASOPHARYNGEAL CARCINOMA
mirna Dr. S Hosseini-Asl
 mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
IJPHCS Open Access: e-journal
 EPSTEIN-BARR VIRUS (EBV) AND ITS ASSOCIATION WITH NASOPHARYNGEAL CARCINOMA (NPC): A SYSTEMATIC REVIEW Sri Ganesh Muthiah 1, Lye MS 1* 1 Department of Community Health, Faculty of Medicine and Health Sciences,
EPSTEIN-BARR VIRUS (EBV) AND ITS ASSOCIATION WITH NASOPHARYNGEAL CARCINOMA (NPC): A SYSTEMATIC REVIEW Sri Ganesh Muthiah 1, Lye MS 1* 1 Department of Community Health, Faculty of Medicine and Health Sciences,
Action and Mechanism of Epstein Barr virus Latent Membrane Protein1. induced Immortalization of Mouse Embryonic Fibroblasts *
 21 1 21 1 16-20 2006 1 VIROLOGICA SINICA January 2006 EB 1 MEF * ** 410078 Action and Mechanism of Epstein Barr virus Latent Membrane Protein1 induced Immortalization of Mouse Embryonic Fibroblasts * HE
21 1 21 1 16-20 2006 1 VIROLOGICA SINICA January 2006 EB 1 MEF * ** 410078 Action and Mechanism of Epstein Barr virus Latent Membrane Protein1 induced Immortalization of Mouse Embryonic Fibroblasts * HE
Seminars in Cancer Biology
 Seminars in Cancer Biology 19 (2009) 377 388 Contents lists available at ScienceDirect Seminars in Cancer Biology journal homepage: www.elsevier.com/locate/semcancer Review Burkitt s lymphoma: The Rosetta
Seminars in Cancer Biology 19 (2009) 377 388 Contents lists available at ScienceDirect Seminars in Cancer Biology journal homepage: www.elsevier.com/locate/semcancer Review Burkitt s lymphoma: The Rosetta
BIT 120. Copy of Cancer/HIV Lecture
 BIT 120 Copy of Cancer/HIV Lecture Cancer DEFINITION Any abnormal growth of cells that has malignant potential i.e.. Leukemia Uncontrolled mitosis in WBC Genetic disease caused by an accumulation of mutations
BIT 120 Copy of Cancer/HIV Lecture Cancer DEFINITION Any abnormal growth of cells that has malignant potential i.e.. Leukemia Uncontrolled mitosis in WBC Genetic disease caused by an accumulation of mutations
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San
 Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
Smoking, human papillomavirus infection, and p53 mutation as risk factors in oropharyngeal cancer: a case-control study
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Smoking, human papillomavirus infection, and p53 as risk factors in oropharyngeal cancer: a case-control study PKS Chan *, JSY Chor, AC Vlantis, TL
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Smoking, human papillomavirus infection, and p53 as risk factors in oropharyngeal cancer: a case-control study PKS Chan *, JSY Chor, AC Vlantis, TL
Complete genomic sequence of Epstein-Barr virus in nasopharyngeal carcinoma cell line C666-1
 Title Complete genomic sequence of Epstein-Barr virus in nasopharyngeal carcinoma cell line C666-1 Author(s) Tso, KK; Yip, KY; Mak, CK; Cheung, ST Citation Infectious Agents and Cancer, 2013, v. 8 n. 1,
Title Complete genomic sequence of Epstein-Barr virus in nasopharyngeal carcinoma cell line C666-1 Author(s) Tso, KK; Yip, KY; Mak, CK; Cheung, ST Citation Infectious Agents and Cancer, 2013, v. 8 n. 1,
VIRUSES AND CANCER Michael Lea
 VIRUSES AND CANCER 2010 Michael Lea VIRAL ONCOLOGY - LECTURE OUTLINE 1. Historical Review 2. Viruses Associated with Cancer 3. RNA Tumor Viruses 4. DNA Tumor Viruses HISTORICAL REVIEW Historical Review
VIRUSES AND CANCER 2010 Michael Lea VIRAL ONCOLOGY - LECTURE OUTLINE 1. Historical Review 2. Viruses Associated with Cancer 3. RNA Tumor Viruses 4. DNA Tumor Viruses HISTORICAL REVIEW Historical Review
Epstein Barr virus and oncogenesis: from latent genes to tumours
 (2003) 22, 5108 5121 & 2003 Nature Publishing Group All rights reserved 0950-9232/03 $25.00 www.nature.com/onc Lawrence S Young*,1 and Paul G Murray 1 1 Cancer Research UK Institute for Cancer Studies,
(2003) 22, 5108 5121 & 2003 Nature Publishing Group All rights reserved 0950-9232/03 $25.00 www.nature.com/onc Lawrence S Young*,1 and Paul G Murray 1 1 Cancer Research UK Institute for Cancer Studies,
The role of Epstein-Barr virus infection in the pathogenesis of nasopharyngeal carcinoma
 VIROLOGICA SINICA 2015, 30 (2): 107-121 DOI 10.1007/s12250-015-3592-5 REVIEW The role of Epstein-Barr virus infection in the pathogenesis of nasopharyngeal carcinoma Chi Man Tsang, Sai Wah Tsao * Department
VIROLOGICA SINICA 2015, 30 (2): 107-121 DOI 10.1007/s12250-015-3592-5 REVIEW The role of Epstein-Barr virus infection in the pathogenesis of nasopharyngeal carcinoma Chi Man Tsang, Sai Wah Tsao * Department
Chapter 9, Part 1: Biology of Cancer and Tumor Spread
 PATHOPHYSIOLOGY Name Chapter 9, Part 1: Biology of Cancer and Tumor Spread I. Cancer Characteristics and Terminology Neoplasm new growth, involves the overgrowth of tissue to form a neoplastic mass (tumor).
PATHOPHYSIOLOGY Name Chapter 9, Part 1: Biology of Cancer and Tumor Spread I. Cancer Characteristics and Terminology Neoplasm new growth, involves the overgrowth of tissue to form a neoplastic mass (tumor).
EBV Infection and Immunity. Andrew Hislop Institute for Cancer Studies University of Birmingham
 EBV Infection and Immunity Andrew Hislop Institute for Cancer Studies University of Birmingham EBV Introduction Large ds DNA virus Spread by saliva contact Lifelong infection Predominantly B-lymphotropic
EBV Infection and Immunity Andrew Hislop Institute for Cancer Studies University of Birmingham EBV Introduction Large ds DNA virus Spread by saliva contact Lifelong infection Predominantly B-lymphotropic
K R N Baumforth, L S Young, K J Flavell, C Constandinou, P G Murray
 J Clin Pathol: Mol Pathol 1999;52:307 322 307 Demystified... Biomedical Research Laboratories, School of Health Sciences, University of Wolverhampton, Wolverhampton WV1 1DJ, UK K R N Baumforth K J Flavell
J Clin Pathol: Mol Pathol 1999;52:307 322 307 Demystified... Biomedical Research Laboratories, School of Health Sciences, University of Wolverhampton, Wolverhampton WV1 1DJ, UK K R N Baumforth K J Flavell
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
number Done by Corrected by Doctor Maha Shomaf
 number 19 Done by Waseem Abo-Obeida Corrected by Abdullah Zreiqat Doctor Maha Shomaf Carcinogenesis: the molecular basis of cancer. Non-lethal genetic damage lies at the heart of carcinogenesis and leads
number 19 Done by Waseem Abo-Obeida Corrected by Abdullah Zreiqat Doctor Maha Shomaf Carcinogenesis: the molecular basis of cancer. Non-lethal genetic damage lies at the heart of carcinogenesis and leads
SEROLOGIC MARKERS OF EPSTEIN BARR VIRUS INFECTION AND NASOPHARYNGEAL CARCINOMA
 SEROLOGIC MARKERS OF EPSTEIN BARR VIRUS INFECTION AND NASOPHARYNGEAL CARCINOMA SEROLOGIC MARKERS OF EPSTEIN BARR VIRUS INFECTION AND NASOPHARYNGEAL CARCINOMA IN TAIWANESE MEN YIN-CHU CHIEN, M.SC., JEN-YANG
SEROLOGIC MARKERS OF EPSTEIN BARR VIRUS INFECTION AND NASOPHARYNGEAL CARCINOMA SEROLOGIC MARKERS OF EPSTEIN BARR VIRUS INFECTION AND NASOPHARYNGEAL CARCINOMA IN TAIWANESE MEN YIN-CHU CHIEN, M.SC., JEN-YANG
Yamaguchi University School of Medicine, Kogushi, Ube, Yamaguchi. growth in low serum, anchorage-independent growth in soft
 JOURNAL OF VIROLOGY, Sept. 1994, p. 6069-6073 Vol. 68, No. 9 0022-538X/94/$04.00+0 Copyright 1994, American Society for Microbiology Isolation of Epstein-Barr Virus (EBV)-Negative Cell Clones from the
JOURNAL OF VIROLOGY, Sept. 1994, p. 6069-6073 Vol. 68, No. 9 0022-538X/94/$04.00+0 Copyright 1994, American Society for Microbiology Isolation of Epstein-Barr Virus (EBV)-Negative Cell Clones from the
RESEARCH ARTICLE. RASSF1A Gene Methylation is Associated with Nasopharyngeal Carcinoma Risk in Chinese
 DOI:http://dx.doi.org/10.7314/APJCP.2015.16.6.2283 RESEARCH ARTICLE RASSF1A Gene Methylation is Associated with Nasopharyngeal Carcinoma Risk in Chinese Kun Wu 1,2&, Xiao-Ning Xu 3&, Yu Chen 1&, Xiao-Lin
DOI:http://dx.doi.org/10.7314/APJCP.2015.16.6.2283 RESEARCH ARTICLE RASSF1A Gene Methylation is Associated with Nasopharyngeal Carcinoma Risk in Chinese Kun Wu 1,2&, Xiao-Ning Xu 3&, Yu Chen 1&, Xiao-Lin
EBV and Infectious Mononucleosis. Infectious Disease Definitions. Infectious Diseases
 Infectious Disease Definitions Infection when a microorganism invades a host and multiplies enough to disrupt normal function by causing signs and symptoms Pathogencity ability of an organism to cause
Infectious Disease Definitions Infection when a microorganism invades a host and multiplies enough to disrupt normal function by causing signs and symptoms Pathogencity ability of an organism to cause
Prevalence of Anti EBV Antibodies in Adult Patients with Nasopharyngeal Carcinoma During In Isfahan, Iran
 Original Article Prevalence of Anti EBV Antibodies in Adult Patients with Nasopharyngeal Carcinoma During 2003 2007 In Isfahan, Iran Mokhtari M 1, Hashemi Jazi M 2, Davarpanah Jazi AH 3 Abstract Background:
Original Article Prevalence of Anti EBV Antibodies in Adult Patients with Nasopharyngeal Carcinoma During 2003 2007 In Isfahan, Iran Mokhtari M 1, Hashemi Jazi M 2, Davarpanah Jazi AH 3 Abstract Background:
An Epstein-Barr virus-encoded microrna targets PUMA to promote host cell survival
 An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
What are the implications of HPV in the biology of Head and Neck Cancer?
 What are the implications of HPV in the biology of Head and Neck Cancer? Raquel Ajub Moyses Friday, August 2nd, 2013 Disclosure Raquel Ajub Moyses has no significant financial relationship with any commercial
What are the implications of HPV in the biology of Head and Neck Cancer? Raquel Ajub Moyses Friday, August 2nd, 2013 Disclosure Raquel Ajub Moyses has no significant financial relationship with any commercial
Epstein-Barr Virus: Cell Trafficking Is Crucial for Persistence
 Epstein-Barr Virus: Cell Trafficking Is Crucial for Persistence This virus moves between host lymphoid and epithelial cells, switching its tropism while enhancing persistence Lindsey Hutt-Fletcher Herpesviruses,
Epstein-Barr Virus: Cell Trafficking Is Crucial for Persistence This virus moves between host lymphoid and epithelial cells, switching its tropism while enhancing persistence Lindsey Hutt-Fletcher Herpesviruses,
Dr Rodney Itaki Lecturer Anatomical Pathology Discipline. University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology
 Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Downregulation of serum mir-17 and mir-106b levels in gastric cancer and benign gastric diseases
 Brief Communication Downregulation of serum mir-17 and mir-106b levels in gastric cancer and benign gastric diseases Qinghai Zeng 1 *, Cuihong Jin 2 *, Wenhang Chen 2, Fang Xia 3, Qi Wang 3, Fan Fan 4,
Brief Communication Downregulation of serum mir-17 and mir-106b levels in gastric cancer and benign gastric diseases Qinghai Zeng 1 *, Cuihong Jin 2 *, Wenhang Chen 2, Fang Xia 3, Qi Wang 3, Fan Fan 4,
Bihong Zhao, M.D, Ph.D Department of Pathology
 Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Case Report A case of EBV positive diffuse large B-cell lymphoma of the adolescent
 Int J Clin Exp Med 2014;7(1):307-311 www.ijcem.com /ISSN:1940-5901/IJCEM1311029 Case Report A case of EBV positive diffuse large B-cell lymphoma of the adolescent Qilin Ao 2, Ying Wang 1, Sanpeng Xu 2,
Int J Clin Exp Med 2014;7(1):307-311 www.ijcem.com /ISSN:1940-5901/IJCEM1311029 Case Report A case of EBV positive diffuse large B-cell lymphoma of the adolescent Qilin Ao 2, Ying Wang 1, Sanpeng Xu 2,
Lawrence S. Young and Alan B. Rickinson
 EPSTEIN BARR VIRUS: 40 YEARS ON Lawrence S. Young and Alan B. Rickinson Abstract Epstein Barr virus (EBV) was discovered 40 years ago from examining electron micrographs of cells cultured from Burkitt
EPSTEIN BARR VIRUS: 40 YEARS ON Lawrence S. Young and Alan B. Rickinson Abstract Epstein Barr virus (EBV) was discovered 40 years ago from examining electron micrographs of cells cultured from Burkitt
Viruses and cancer: Should we be more afraid?
 Viruses and cancer: Should we be more afraid? Viruses and cancer: Should we be more afraid? During the past 30 years it has become exceedingly clear that several viruses play significant roles in the development
Viruses and cancer: Should we be more afraid? Viruses and cancer: Should we be more afraid? During the past 30 years it has become exceedingly clear that several viruses play significant roles in the development
Epstein-Barr Virus 1
 Epstein-Barr Virus 1 Herpesviruses dsdna, linear, enveloped, 180-200 nm Large genome, codes for 75 viral proteins 50-70% similarity Cross reactivity between HSV and VZV HSV-2 virus particle. Note that
Epstein-Barr Virus 1 Herpesviruses dsdna, linear, enveloped, 180-200 nm Large genome, codes for 75 viral proteins 50-70% similarity Cross reactivity between HSV and VZV HSV-2 virus particle. Note that
2017 CST-Astellas Canadian Transplant Fellows Symposium. EBV Post Transplantation Implications and Approach to Management
 2017 CST-Astellas Canadian Transplant Fellows Symposium EBV Post Transplantation Implications and Approach to Management Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University
2017 CST-Astellas Canadian Transplant Fellows Symposium EBV Post Transplantation Implications and Approach to Management Atul Humar, MD Atul Humar is a Professor in the Department of Medicine, University
Focus on nasopharyngeal carcinoma
 Focus on nasopharyngeal carcinoma Kwok Wai Lo, 1 Ka Fai To, 1 and Dolly P. Huang 1, 2, * 1 Department of Anatomical and Cellular Pathology 2 Sir Y.K. Pao Centre for Cancer Prince of Wales Hospital, The
Focus on nasopharyngeal carcinoma Kwok Wai Lo, 1 Ka Fai To, 1 and Dolly P. Huang 1, 2, * 1 Department of Anatomical and Cellular Pathology 2 Sir Y.K. Pao Centre for Cancer Prince of Wales Hospital, The
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Fayth K. Yoshimura, Ph.D. September 7, of 7 HIV - BASIC PROPERTIES
 1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
Epstein-Barr Virus BART MicroRNAs Are Produced from a Large Intron prior to Splicing
 JOURNAL OF VIROLOGY, Sept. 2008, p. 9094 9106 Vol. 82, No. 18 0022-538X/08/$08.00 0 doi:10.1128/jvi.00785-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Epstein-Barr Virus BART
JOURNAL OF VIROLOGY, Sept. 2008, p. 9094 9106 Vol. 82, No. 18 0022-538X/08/$08.00 0 doi:10.1128/jvi.00785-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Epstein-Barr Virus BART
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Chapter 4 Cellular Oncogenes ~ 4.6 -
 Chapter 4 Cellular Oncogenes - 4.2 ~ 4.6 - Many retroviruses carrying oncogenes have been found in chickens and mice However, attempts undertaken during the 1970s to isolate viruses from most types of
Chapter 4 Cellular Oncogenes - 4.2 ~ 4.6 - Many retroviruses carrying oncogenes have been found in chickens and mice However, attempts undertaken during the 1970s to isolate viruses from most types of
Nasopharyngeal Carcinoma. Rusty Stevens, MD Christopher Rassekh, MD
 Nasopharyngeal Carcinoma Rusty Stevens, MD Christopher Rassekh, MD Introduction Rare in the US, more common in Asia High index of suspicion required for early diagnosis Nasopharyngeal malignancies SCCA
Nasopharyngeal Carcinoma Rusty Stevens, MD Christopher Rassekh, MD Introduction Rare in the US, more common in Asia High index of suspicion required for early diagnosis Nasopharyngeal malignancies SCCA
1. Basic principles 2. 6 hallmark features 3. Abnormal cell proliferation: mechanisms 4. Carcinogens: examples. Major Principles:
 Carcinogenesis 1. Basic principles 2. 6 hallmark features 3. Abnormal cell proliferation: mechanisms 4. Carcinogens: examples Carcinogenesis Major Principles: 1. Nonlethal genetic damage is central to
Carcinogenesis 1. Basic principles 2. 6 hallmark features 3. Abnormal cell proliferation: mechanisms 4. Carcinogens: examples Carcinogenesis Major Principles: 1. Nonlethal genetic damage is central to
Nucleic Acid Testing - Oncology. Molecular Diagnosis. Gain/Loss of Nucleic Acid. Objectives. MYCN and Neuroblastoma. Molecular Diagnosis
 Nucleic Acid Testing - Oncology Molecular Diagnosis Nucleic acid based testing in Oncology Gross alterations in DNA content of tumors (ploidy) Gain/Loss of nucleic acids Markers of Clonality Oncogene/Tumor
Nucleic Acid Testing - Oncology Molecular Diagnosis Nucleic acid based testing in Oncology Gross alterations in DNA content of tumors (ploidy) Gain/Loss of nucleic acids Markers of Clonality Oncogene/Tumor
Molecular Diagnosis. Nucleic acid based testing in Oncology
 Molecular Diagnosis Nucleic acid based testing in Oncology Objectives Describe uses of NAT in Oncology Diagnosis, Prediction, monitoring. Genetics Screening, presymptomatic testing, diagnostic testing,
Molecular Diagnosis Nucleic acid based testing in Oncology Objectives Describe uses of NAT in Oncology Diagnosis, Prediction, monitoring. Genetics Screening, presymptomatic testing, diagnostic testing,
Persistent Infections
 Persistent Infections Lecture 17 Biology 3310/4310 Virology Spring 2017 Paralyze resistance with persistence WOODY HAYES Acute vs persistent infections Acute infection - rapid and self-limiting Persistent
Persistent Infections Lecture 17 Biology 3310/4310 Virology Spring 2017 Paralyze resistance with persistence WOODY HAYES Acute vs persistent infections Acute infection - rapid and self-limiting Persistent
Tumor suppressor genes D R. S H O S S E I N I - A S L
 Tumor suppressor genes 1 D R. S H O S S E I N I - A S L What is a Tumor Suppressor Gene? 2 A tumor suppressor gene is a type of cancer gene that is created by loss-of function mutations. In contrast to
Tumor suppressor genes 1 D R. S H O S S E I N I - A S L What is a Tumor Suppressor Gene? 2 A tumor suppressor gene is a type of cancer gene that is created by loss-of function mutations. In contrast to
Activation of cellular proto-oncogenes to oncogenes. How was active Ras identified?
 Dominant Acting Oncogenes Eugene E. Marcantonio, M.D. Ph.D. Oncogenes are altered forms of normal cellular genes called proto-oncogenes that are involved in pathways regulating cell growth, differentiation,
Dominant Acting Oncogenes Eugene E. Marcantonio, M.D. Ph.D. Oncogenes are altered forms of normal cellular genes called proto-oncogenes that are involved in pathways regulating cell growth, differentiation,
Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy uridine in Human Lymphoblastoid Cells F ro m a Rhabdom yosarcom a*
 A n n a ls o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy
A n n a ls o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy
Expression of Epstein-Barr Virus Gene and Clonality of Infiltrated T Lymphocytes in Epstein-Barr Virus-associated Gastric Carcinoma
 DOI 10.4110/in.2011.11.1.50 pissn 1598-2629 eissn 2092-6685 ORIGINAL ARTICLE Expression of Epstein-Barr Virus Gene and Clonality of Infiltrated T Lymphocytes in Epstein-Barr Virus-associated Gastric Carcinoma
DOI 10.4110/in.2011.11.1.50 pissn 1598-2629 eissn 2092-6685 ORIGINAL ARTICLE Expression of Epstein-Barr Virus Gene and Clonality of Infiltrated T Lymphocytes in Epstein-Barr Virus-associated Gastric Carcinoma
on November 3, 2018 by guest
 JVI Accepted Manuscript Posted Online 19 October 2016 J. Virol. doi:10.1128/jvi.01677-16 Copyright 2016, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10 The long and complicated
JVI Accepted Manuscript Posted Online 19 October 2016 J. Virol. doi:10.1128/jvi.01677-16 Copyright 2016, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10 The long and complicated
Circulating Nucleic Acids in Plasma and Serum: An Overview
 Circulating Nucleic Acids in Plasma and Serum: An Overview Y. M. DENNIS LO Department of Chemical Pathology and Institute of Molecular Oncology, The Chinese University of Hong Kong, Prince of Wales Hospital,
Circulating Nucleic Acids in Plasma and Serum: An Overview Y. M. DENNIS LO Department of Chemical Pathology and Institute of Molecular Oncology, The Chinese University of Hong Kong, Prince of Wales Hospital,
5/1/13. The proportion of thymus that produces T cells decreases with age. The cellular organization of the thymus
 T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer
 Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Karyotype analysis reveals transloction of chromosome 22 to 9 in CML chronic myelogenous leukemia has fusion protein Bcr-Abl
 Chapt. 18 Cancer Molecular Biology of Cancer Student Learning Outcomes: Describe cancer diseases in which cells no longer respond Describe how cancers come from genomic mutations (inherited or somatic)
Chapt. 18 Cancer Molecular Biology of Cancer Student Learning Outcomes: Describe cancer diseases in which cells no longer respond Describe how cancers come from genomic mutations (inherited or somatic)
Cancer Genetics. What is Cancer? Cancer Classification. Medical Genetics. Uncontrolled growth of cells. Not all tumors are cancerous
 Session8 Medical Genetics Cancer Genetics J avad Jamshidi F a s a U n i v e r s i t y o f M e d i c a l S c i e n c e s, N o v e m b e r 2 0 1 7 What is Cancer? Uncontrolled growth of cells Not all tumors
Session8 Medical Genetics Cancer Genetics J avad Jamshidi F a s a U n i v e r s i t y o f M e d i c a l S c i e n c e s, N o v e m b e r 2 0 1 7 What is Cancer? Uncontrolled growth of cells Not all tumors
ANTIBODY TO EPSTEIN-BARR VIRUS IN PATIENTS WITH CARCINOMA OF THE NASOPHARYNX
 Keio J. Med. 26: 79-90, 1977 ANTIBODY TO EPSTEIN-BARR VIRUS IN PATIENTS WITH CARCINOMA OF THE NASOPHARYNX YUKIO INUYAMA*, KAZUYUKI ASAOKA*, YASUO NAKAJIMA*, MASATOSHI HORIUCHI*, KEI TAKASAKI** and RAISUKE
Keio J. Med. 26: 79-90, 1977 ANTIBODY TO EPSTEIN-BARR VIRUS IN PATIENTS WITH CARCINOMA OF THE NASOPHARYNX YUKIO INUYAMA*, KAZUYUKI ASAOKA*, YASUO NAKAJIMA*, MASATOSHI HORIUCHI*, KEI TAKASAKI** and RAISUKE
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
Acta Medica Okayama AUGUST A spectrum of clinical manifestations caused by host immune responses against Epstein-Barr virus infections.
 Acta Medica Okayama Volume 58, Issue 4 2004 Article 1 AUGUST 2004 A spectrum of clinical manifestations caused by host immune responses against Epstein-Barr virus infections. Keiji Iwatsuki Takenobu Yamamoto
Acta Medica Okayama Volume 58, Issue 4 2004 Article 1 AUGUST 2004 A spectrum of clinical manifestations caused by host immune responses against Epstein-Barr virus infections. Keiji Iwatsuki Takenobu Yamamoto
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Hepatitis B virus X gene in the development of hepatocellular carcinoma. Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p.
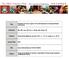 Title Hepatitis B virus X gene in the development of hepatocellular carcinoma Author(s) Ma, NF; Lau, SH; Hu, L; Dong, SS; Guan, XY Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p. 44-47
Title Hepatitis B virus X gene in the development of hepatocellular carcinoma Author(s) Ma, NF; Lau, SH; Hu, L; Dong, SS; Guan, XY Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p. 44-47
oncogenes-and- tumour-suppressor-genes)
 Special topics in tumor biochemistry oncogenes-and- tumour-suppressor-genes) Speaker: Prof. Jiunn-Jye Chuu E-Mail: jjchuu@mail.stust.edu.tw Genetic Basis of Cancer Cancer-causing mutations Disease of aging
Special topics in tumor biochemistry oncogenes-and- tumour-suppressor-genes) Speaker: Prof. Jiunn-Jye Chuu E-Mail: jjchuu@mail.stust.edu.tw Genetic Basis of Cancer Cancer-causing mutations Disease of aging
Section Lectures: Immunology/Virology Time: 9:00 am 10:00 am LRC 105 A & B
 Section Director: Cliff Bellone, Ph.D. Office: Doisy Hall - R 405 Phone: 577-8449 E-Mail: bellonec@slu.edu Lecturers: James Swierkosz, Ph.D. Office: Medical School Rm. 412 Phone: 577-8430 E-Mail: swierkoszje@slu.edu
Section Director: Cliff Bellone, Ph.D. Office: Doisy Hall - R 405 Phone: 577-8449 E-Mail: bellonec@slu.edu Lecturers: James Swierkosz, Ph.D. Office: Medical School Rm. 412 Phone: 577-8430 E-Mail: swierkoszje@slu.edu
What causes cancer? Physical factors (radiation, ionization) Chemical factors (carcinogens) Biological factors (virus, bacteria, parasite)
 Oncogenes What causes cancer? Chemical factors (carcinogens) Physical factors (radiation, ionization) Biological factors (virus, bacteria, parasite) DNA Mutation or damage Oncogenes Tumor suppressor genes
Oncogenes What causes cancer? Chemical factors (carcinogens) Physical factors (radiation, ionization) Biological factors (virus, bacteria, parasite) DNA Mutation or damage Oncogenes Tumor suppressor genes
Methylation status of SOCS1 and SOCS3 in BCR-ABL negative and. JAK2V617F negative chronic myeloproliferative disorders.
 Methylation status of SOCS1 and SOCS3 in BCR-ABL negative and JAK2V617F negative chronic myeloproliferative disorders. To the Editor BCR-ABL negative Chronic Myeloproliferative Disorders (s) are a heterogeneous
Methylation status of SOCS1 and SOCS3 in BCR-ABL negative and JAK2V617F negative chronic myeloproliferative disorders. To the Editor BCR-ABL negative Chronic Myeloproliferative Disorders (s) are a heterogeneous
Review (V1) - The Hallmarks of Cancer
 Review (V1) - The Hallmarks of Cancer Robert A. Weinberg 1 Review (V1) - The Hallmarks of Cancer 2 Review (V1) - Number of somatic mutations in human cancers Top: children vs. adults Numbers in parentheses
Review (V1) - The Hallmarks of Cancer Robert A. Weinberg 1 Review (V1) - The Hallmarks of Cancer 2 Review (V1) - Number of somatic mutations in human cancers Top: children vs. adults Numbers in parentheses
Biologic and Molecular Properties of LMP1: CTARs, Strains, and Beyond. by Bernardo A. Mainou
 Biologic and Molecular Properties of LMP1: CTARs, Strains, and Beyond by Bernardo A. Mainou A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment
Biologic and Molecular Properties of LMP1: CTARs, Strains, and Beyond by Bernardo A. Mainou A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment
The Future of Cancer. Lawrence Tsui Global Risk Products Actuary Swiss Reinsurance Company Hong Kong. Session Number: WBR8
 Lawrence Tsui Global Risk Products Actuary Swiss Reinsurance Company Hong Kong Session Number: WBR8 Agenda Cancer the basics Cancer past and present Cancer the future CANCER THE BASICS Cancer the basics
Lawrence Tsui Global Risk Products Actuary Swiss Reinsurance Company Hong Kong Session Number: WBR8 Agenda Cancer the basics Cancer past and present Cancer the future CANCER THE BASICS Cancer the basics
Viruses. Poxviridae. DNA viruses: 6 families. Herpesviridae Adenoviridae. Hepadnaviridae Papovaviridae Parvoviridae
 Viruses DNA viruses: 6 families Poxviridae Herpesviridae Adenoviridae Hepadnaviridae Papovaviridae Parvoviridae Human herpesviruses Three subfamilies (genome structure, tissue tropism, cytopathologic effect,
Viruses DNA viruses: 6 families Poxviridae Herpesviridae Adenoviridae Hepadnaviridae Papovaviridae Parvoviridae Human herpesviruses Three subfamilies (genome structure, tissue tropism, cytopathologic effect,
Can HPV, cervical neoplasia or. HIV transmission?
 Interactions between HPV and HIV: STIs and HIV shedding, regulation of HPV by HIV, and HPV VLP influence upon HIV Jennifer S. Smith Department of Epidemiology pd University of North Carolina Can HPV, cervical
Interactions between HPV and HIV: STIs and HIV shedding, regulation of HPV by HIV, and HPV VLP influence upon HIV Jennifer S. Smith Department of Epidemiology pd University of North Carolina Can HPV, cervical
by Che-Pei Kung Chapel Hill
 The Epstein-Barr Virus Latent Membrane Protein 1 Regulates Gene Expression by Engaging Multiple Signaling Pathways by Che-Pei Kung A dissertation submitted to the faculty of the University of North Carolina
The Epstein-Barr Virus Latent Membrane Protein 1 Regulates Gene Expression by Engaging Multiple Signaling Pathways by Che-Pei Kung A dissertation submitted to the faculty of the University of North Carolina
Introduction to Cancer Biology
 Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
In Vitro and In Vivo Studies with Epstein-Barr
 A n n a l s o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science In Vitro and In Vivo Studies with Epstein-Barr Virus (EBV)-------A Review
A n n a l s o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science In Vitro and In Vivo Studies with Epstein-Barr Virus (EBV)-------A Review
VIII Curso Internacional del PIRRECV. Some molecular mechanisms of cancer
 VIII Curso Internacional del PIRRECV Some molecular mechanisms of cancer Laboratorio de Comunicaciones Celulares, Centro FONDAP Estudios Moleculares de la Celula (CEMC), ICBM, Facultad de Medicina, Universidad
VIII Curso Internacional del PIRRECV Some molecular mechanisms of cancer Laboratorio de Comunicaciones Celulares, Centro FONDAP Estudios Moleculares de la Celula (CEMC), ICBM, Facultad de Medicina, Universidad
Multistep nature of cancer development. Cancer genes
 Multistep nature of cancer development Phenotypic progression loss of control over cell growth/death (neoplasm) invasiveness (carcinoma) distal spread (metastatic tumor) Genetic progression multiple genetic
Multistep nature of cancer development Phenotypic progression loss of control over cell growth/death (neoplasm) invasiveness (carcinoma) distal spread (metastatic tumor) Genetic progression multiple genetic
Epstein-Barr Virus (EBV) Infection in Epithelial Cells In Vivo: Rare Detection of EBV Replication in Tongue Mucosa but Not in Salivary Glands
 BRIEF REPORT Epstein-Barr Virus (EBV) Infection in Epithelial Cells In Vivo: Rare Detection of EBV Replication in Tongue Mucosa but Not in Salivary Glands Phroso Frangou, Maike Buettner, and Gerald Niedobitek
BRIEF REPORT Epstein-Barr Virus (EBV) Infection in Epithelial Cells In Vivo: Rare Detection of EBV Replication in Tongue Mucosa but Not in Salivary Glands Phroso Frangou, Maike Buettner, and Gerald Niedobitek
Oncogenes and Tumor Suppressors MCB 5068 November 12, 2013 Jason Weber
 Oncogenes and Tumor Suppressors MCB 5068 November 12, 2013 Jason Weber jweber@dom.wustl.edu Oncogenes & Cancer DNA Tumor Viruses Simian Virus 40 p300 prb p53 Large T Antigen Human Adenovirus p300 E1A
Oncogenes and Tumor Suppressors MCB 5068 November 12, 2013 Jason Weber jweber@dom.wustl.edu Oncogenes & Cancer DNA Tumor Viruses Simian Virus 40 p300 prb p53 Large T Antigen Human Adenovirus p300 E1A
Clever tricks EBV employed to modulate innate immunity during latency and lytic infection
 Clever tricks EBV employed to modulate innate immunity during latency and lytic infection Kirsten Kuipers December 22, 2011 About the cover: Upon recognition of PAMPs, activated PRRs initiate innate immune
Clever tricks EBV employed to modulate innate immunity during latency and lytic infection Kirsten Kuipers December 22, 2011 About the cover: Upon recognition of PAMPs, activated PRRs initiate innate immune
Epstein-Barr virus infection in the pathogenesis of nasopharyngeal carcinoma
 248 Institute for Pathology, Friedrich-Alexander- University, Krankenhausstr. 8 10, 91054 Erlangen, Germany G Niedobitek Correspondence to: Dr Niedobitek gerald.niedobitek@ patho.imed.uni-erlangen.de Accepted
248 Institute for Pathology, Friedrich-Alexander- University, Krankenhausstr. 8 10, 91054 Erlangen, Germany G Niedobitek Correspondence to: Dr Niedobitek gerald.niedobitek@ patho.imed.uni-erlangen.de Accepted
Deregulation of signal transduction and cell cycle in Cancer
 Deregulation of signal transduction and cell cycle in Cancer Tuangporn Suthiphongchai, Ph.D. Department of Biochemistry Faculty of Science, Mahidol University Email: tuangporn.sut@mahidol.ac.th Room Pr324
Deregulation of signal transduction and cell cycle in Cancer Tuangporn Suthiphongchai, Ph.D. Department of Biochemistry Faculty of Science, Mahidol University Email: tuangporn.sut@mahidol.ac.th Room Pr324
Epstein-Barr virus (EBV), a linear double-stranded DNA
 Epstein-Barr Virus as a Paradigm in Nasopharyngeal Cancer: From Lab to Clinic Radha Raghupathy, MD, Edwin Pun Hui, MD, and Anthony Tak Cheung Chan, MD, FRCP OVERVIEW Nasopharyngeal carcinoma (NPC) of the
Epstein-Barr Virus as a Paradigm in Nasopharyngeal Cancer: From Lab to Clinic Radha Raghupathy, MD, Edwin Pun Hui, MD, and Anthony Tak Cheung Chan, MD, FRCP OVERVIEW Nasopharyngeal carcinoma (NPC) of the
Human Lung Cancer Pathology and Cellular Biology Mouse Lung Tumor Workshop
 Human Lung Cancer Pathology and Cellular Biology Mouse Lung Tumor Workshop Jan 7 th and 8 th, 2014 Brigitte Gomperts, MD University of California, Los Angeles Lung Structure and Function Airway Epithelial
Human Lung Cancer Pathology and Cellular Biology Mouse Lung Tumor Workshop Jan 7 th and 8 th, 2014 Brigitte Gomperts, MD University of California, Los Angeles Lung Structure and Function Airway Epithelial
7 Omar Abu Reesh. Dr. Ahmad Mansour Dr. Ahmad Mansour
 7 Omar Abu Reesh Dr. Ahmad Mansour Dr. Ahmad Mansour -Leukemia: neoplastic leukocytes circulating in the peripheral bloodstream. -Lymphoma: a neoplastic process in the lymph nodes, spleen or other lymphatic
7 Omar Abu Reesh Dr. Ahmad Mansour Dr. Ahmad Mansour -Leukemia: neoplastic leukocytes circulating in the peripheral bloodstream. -Lymphoma: a neoplastic process in the lymph nodes, spleen or other lymphatic
Introduction. Cancer Biology. Tumor-suppressor genes. Proto-oncogenes. DNA stability genes. Mechanisms of carcinogenesis.
 Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
MicroRNA expression profiling and functional analysis in prostate cancer. Marco Folini s.c. Ricerca Traslazionale DOSL
 MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
The Prototypical Epstein-Barr Virus-Transformed Lymphoblastoid Cell Line IB4 Is an Unusual Variant Containing Integrated but No Episomal Viral DNA
 JOURNAL OF VIROLOGY, JUlY 1991, p. 3958-5963 0022-538X/91/073958-06$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 65, No. 7 The Prototypical Epstein-Barr Virus-Transformed Lymphoblastoid
JOURNAL OF VIROLOGY, JUlY 1991, p. 3958-5963 0022-538X/91/073958-06$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 65, No. 7 The Prototypical Epstein-Barr Virus-Transformed Lymphoblastoid
Cancer genetics
 Cancer genetics General information about tumorogenesis. Cancer induced by viruses. The role of somatic mutations in cancer production. Oncogenes and Tumor Suppressor Genes (TSG). Hereditary cancer. 1
Cancer genetics General information about tumorogenesis. Cancer induced by viruses. The role of somatic mutations in cancer production. Oncogenes and Tumor Suppressor Genes (TSG). Hereditary cancer. 1
Development of Carcinoma Pathways
 The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
MicroRNA dysregulation in cancer. Systems Plant Microbiology Hyun-Hee Lee
 MicroRNA dysregulation in cancer Systems Plant Microbiology Hyun-Hee Lee Contents 1 What is MicroRNA? 2 mirna dysregulation in cancer 3 Summary What is MicroRNA? What is MicroRNA? MicroRNAs (mirnas) -
MicroRNA dysregulation in cancer Systems Plant Microbiology Hyun-Hee Lee Contents 1 What is MicroRNA? 2 mirna dysregulation in cancer 3 Summary What is MicroRNA? What is MicroRNA? MicroRNAs (mirnas) -
