Enhanced Growth of Small Bowel in Transgenic Mice Expressing Human Insulin-like Growth Factor I
|
|
|
- Melvyn Patterson
- 6 years ago
- Views:
Transcription
1 GASTROENTEROLOGY 1997;112: Enhanced Growth of Small Bowel in Transgenic Mice Expressing Human Insulin-like Growth Factor I KINUKO OHNEDA,* MARTIN H. ULSHEN,, C. RANDALL FULLER,, A. JOSEPH D ERCOLE, and P. KAY LUND*, Departments of *Physiology and Pediatrics and Center for Gastrointestinal Biology and Disease, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina Background & Aims: Growth hormone and insulin-like lates circulating concentrations of IGF-I and local expresgrowth factor I (IGF-I) stimulate small bowel growth. sion of IGF-I in a number of tissues including the The aim of this study was to analyze whether IGF-I bowel. 13 It has long been recognized that GH exerts mediates enterotrophic actions of growth hormone. trophic effects on small bowel. 14,15 However, the extent Methods: IGF-I transgenic mice that overexpress an to which enterotrophic actions of GH are mediated by IGF-I transgene driven by the mouse metallothionein I circulating or locally expressed IGF-I are not well depromoter and are growth hormone deficient were comfined. We reported previously that long-term GH excess pared with wild-type littermates. Growth of small bowel, in transgenic mice overexpressing the bovine GH gene abundance and localization of messenger RNAs for the 16 IGF-I transgene, and insulin-like growth factor binding results in increased bowel length and bowel mass. Deprotein 3 were assayed. Results: Small bowel length spite significant mucosal hyperplasia in GH transgenics, and mass were greater in IGF-I transgenic mice than in crypt cell proliferation did not differ in bowel mucosa wild-type mice. Villus height, crypt depth, and crypt cell of GH transgenics and wild-type (WT) littermates. 16 mitoses were greater in jejunum of transgenics than Therefore, GH may increase the lifespan of mucosal epiwild-type mice, but jejunal disacharidase activities thelial cells. GH transgenics have increased circulating were not increased. The transgene was expressed GH, increased plasma concentrations of IGF-I, and instrongly in villus epithelial cells. Insulin-like growth fac- creased expression of endogenous IGF-I locally within tor binding protein 3 messenger RNA was localized in small bowel. 16 Each may contribute to the enhanced the lamina propria. Regional expression of both corresmall bowel growth. Mathews et al. 17 developed a lated with the increase in mucosal mass. Conclusions: transgenic model of long-term IGF-I excess and GH Effects of IGF-I overexpression on intestinal length and mucosal mass were similar to effects of growth hordeficiency that permits evaluation of the effects of excess mone overexpression observed previously. Excess of IGF-I in a background of GH deficiency. These IGF-I IGF-I increased crypt cell proliferation, whereas excess transgenics have overexpression of a chimeric human of growth hormone did not increase crypt cell proliferation. IGF-I precursor composed of a somatostatin signal pep- IGF-I excess stimulated differentiation of intesti- tide (to facilitate secretion) and coding sequence for the nal epithelial cells less effectively than growth hormone human IGF-I A precursor. 17 Transgene expression is excess. driven by a mouse metallothionein I promoter. The IGF- I transgenics show elevation of circulating IGF-I and nsulin-like growth factor I (IGF-I) plays a central role high-level local expression of transgene-derived IGF-I Iin the regulation of somatic and organ growth during messenger RNA (mrna) in several organs. 17 They also postnatal life. 1,2 Recent evidence indicates that IGF-I can have undetectable levels of plasma GH and a decrease in stimulate the growth of small bowel mucosa because pituitary GH mrna caused by negative feedback effects systemic administration of IGF-I, acting in an endocrine of IGF-I excess. 17 Selective organomegaly of brain, pan- manner, increases small bowel growth and function. 3 6 creas, kidney, spleen, and carcass was observed in these The type 1 IGF-I receptor is expressed throughout small IGF-I transgenics, 17 but effects of excess IGF-I on growth bowel 7 10 and undoubtedly mediates these IGF-I actions. IGF-I also is expressed locally throughout human and Abbreviations used in this paper: GH, growth hormone; IGFBP, rodent gastrointestinal tract 11,12 and, thus, may exert parfactor I; WT, wild type. insulin-like growth factor binding protein; IGF-I, insulin-like growth acrine or autocrine actions on bowel growth and function by the American Gastroenterological Association It is well established that growth hormone (GH) regu /97/$3.00
2 February 1997 SMALL BOWEL GROWTH IN IGF I TRANSGENIC MICE 445 and function of small intestine were not examined in diet. In the other group, the pair-fed group, IGF-I transgenics detail. In the present study, we analyzed mice overex- were pair fed the same daily amount of food as consumed by pressing the same chimeric human IGF-I transgene and WT littermates. This was accomplished by monitoring the ad WT mice for effects on small bowel growth. Our aim libitum daily diet intake of five pairs of WT and IGF-I transgenics for 5 days. IGF-I transgenic mice consumed an was to assess the extent to which long-term excess of average of 5.8 { 0.3 g/day compared with 3.8 { 0.4 g/day IGF-I in a background of GH deficiency could stimulate intake of WT mice. Therefore, in the pair-feeding study, IGFsmall bowel growth. We also studied the abundance and I transgenics and WT mice were provided with 3.8 g/day for localization of IGF-I transgene expression in the small 4 days before the study. bowel to examine whether enterotrophic effects of excess Under anesthesia induced by ketamine hydrochloride (900 IGF-I correlate with local overexpression of IGF-I in mg/g body wt) and xylazine hydrochloride (20 mg/g body wt), bowel. blood was collected by cardiac puncture, and mice were killed It is becoming increasingly clear that insulin-like by exsanguination. The entire small intestine from pylorus to growth factor binding proteins (IGFBPs) have major ileocecal valve was rapidly removed and weighed. The intestine effects on the modulation and/or targeting of IGF-I aclength was subject to uniform tension with a 5-g weight, and its tion. 18 One IGFBP, IGFBP-3, may be particularly relecollected was measured. Segments of small bowel studied were for analyses as follows. For growth analyses, duodevant to the effects of IGF-I on small bowel mucosa. num (from pylorus to the ligament of Treitz), proximal jeju- IGFBP-3 is expressed locally within the small bowel num (one third of small bowel beginning at the ligament of mucosa. 19 Reduced expression of IGFBP-3 in small bowel Treitz), and distal ileum (distal one third of the small bowel has been observed in two situations that induce hyperplaup to the ileocecal valve) were dissected. The length of each sia of small bowel mucosa: adaptive growth in response segment was measured under uniform tension, and then each to proximal small bowel resection 20 and in response to segment was weighed. The remnant mid small bowel segrefeeding after a fast. 19 This has led to the concept that ment was collected for RNA extraction and verification of reduced expression of IGFBP-3 may provide a mecha- transgene expression. In separate groups of ad libitum fed and nism to amplify the enterotrophic actions of IGF-I. 19,20 pair-fed IGF-I transgenics, duodenum, jejunum, and ileum A number of lines of evidence indicate that GH and were collected for extraction of RNA to allow comparisons of IGF-I positively regulate IGFBP-3 expression and that levels of expression of IGF-I and IGFBP-3 in different regions the relative effects of GH and IGF-I on IGFBP-3 may of small bowel. All segments were rapidly frozen in liquid differ in different tissues. 18 Because local expression of nitrogen and stored at 080 C until analysis. Transgenic mice that overexpress the bovine GH gene and IGFBP-3 may influence the enterotrophic actions of extheir WT littermates were evaluated in a pair-fed study as cess IGF-I in the IGF-I transgenics, we compared the described previously. 16 Proximal jejunum was collected for abundance and cellular location of IGFBP-3 mrna in RNA isolation and in situ hybridization histochemistry to small bowel of IGF-I transgenics and WT mice. allow direct comparisons of RNA expression in GH transgenics vs. IGF-I transgenics. Materials and Methods The animal studies were approved by the Institutional Animal Animals and Experimental Design Care and Use Committee of the University of North Caro- Mice overexpressing the human IGF-I transgene and lina at Chapel Hill; study protocols were in compliance with their WT littermates were bred from founder mice generated the Guide for the Care and Use of Laboratory Animals published at the University of North Carolina at Chapel Hill. 21 by the National Institutes of Health. The construct used to establish the IGF-I transgenic lines was iden- Assays of Mucosal Mass and Mucosal tical to that reported by Mathews et al. in Detailed Disaccharidase Activity analyses of small bowel growth described in this study were all performed on IGF-I transgenics of a single line (L52), i.e., Mucosal scrapings from individual segments were prepared mice derived from a single founder, but similar results were and homogenized as described previously. 16 Mucosal obtained in a smaller number of mice from a different line mass was determined by wet weight of the mucosa, weighed (L32). Mice were weaned at 21 days of age and given 25 mmol/ immediately after the scraping, as well as DNA and protein L zinc sulfate in their drinking water until the study at 50 content measured in homogenates. Measurement of DNA content 60 days of age. Transgenic mice were distinguished from WT was performed using a standard fluorescent dye microassay mice before study by their increased body weight. Genotype method. 22 Protein content was determined using the method was confirmed by Northern blot analyses of transgene expression of Lowry et al. 23 Mucosal disaccharidase activity was performed in small bowel after the mice were killed. by modifications of the standard methods. 24,25 A unit of activ- Two separate groups of animals were analyzed. In one, the ity equals one micromole of glucose released per minute. All ad libitum fed group, mice had free access to standard pelleted assays were performed in duplicate.
3 446 OHNEDA ET AL. GASTROENTEROLOGY Vol. 112, No. 2 Histological Analysis of Mucosal Growth Measurement of Crypt Cell Mitoses Crypt cell mitoses were analyzed in jejunum and ileum of pair-fed IGF-I transgenic and WT mice. Bowel segments were collected and fixed in Carnoy s reagent for 3 hours followed by dehydration in 70% ethanol. Tissues were rehydrated and stained with Schiff s reagent (Sigma Chemical Co., St Louis, MO), and crypts were isolated by microdissection. 26 The numbers of mitotic cells per crypt were measured with light microscopy. The measurement was performed for 20 crypts per each specimen by a single investigator who was unaware of the animal groupings. RNA Isolation and Northern Blot Hybridization Total RNA was isolated from different segments of bowel using the guanidine isothiocyanate/cscl method and enriched for poly (A) / RNA by oligo(dt) cellulose chromatography as described previously. 11 Five-microgram aliquots of (poly)a / RNA were denatured with glyoxal and dimethylsulfoxide, size fractionated on 1% agarose gels, and transferred to Gene Screen (New England Nuclear Corp., Boston, MA). Hybridization was performed using antisense RNA probes la- beled with [ 32 P]uridine triphosphate as described previously. 27 The human IGF-I antisense RNA probe was prepared from human IGF-I complementary DNA 28 subcloned into pgem3z. A rat IGFBP-3 probe was prepared from rat IGFBP-3 cdna subcloned into pbst SK /. 19 Each blot was stripped and rehy- bridized with a rat actin complementary RNA probe (Ambion, Austin, TX) to normalize for RNA loading. Abundance of different mrnas was calculated on a scanning densitometer using National Institutes of Health Image software (National Technical Information Service, Springfield, VA). Abundance of IGF-I and IGFBP-3 mrna was normalized for RNA loading by dividing by the abundance of actin mrna. Segments of jejunum (0.5 cm) were opened longitudi- nally and fixed with formalin. Each tissue specimen was em- bedded in paraffin, sectioned, and stained with Mayer s H&E. Villus height and crypt depth were measured on the stained sections using light microscopy with computer-assisted morphometry. Five to ten villi and a similar number of crypts were measured per segment. All measurements were performed by a single investigator who was masked to animal groupings. acetic anhydride (vol/vol, 0.25%), dehydrated through graded alcohols, and air dried. Sense and antisense human IGF-I RNAs and rat IGFBP-3 RNAs labeled with 35 S-uridine triphosphate were prepared as described previously. 27 Probe ( cpm/ slide) was mixed with hybridization buffer containing 75% formamide, and hybridization was performed at 55 C for 18 hours. After hybridization, all slides were treated with ribo- nuclease A (200 mg/ml; Sigma Chemical Co.) at 37 C for 1 hour, followed by washing in 0.51 standard saline citrate (75 mmol/l NaCl and 7.5 mmol/l sodium citrate, ph 7.0) at 55 C. Sections were dehydrated and air dried and then were exposed to Kodak NTB-2 autoradiographic emulsion (Eastman Kodak Co., Rochester, NY) at 4 C for 14 days. Slides were developed, counterstained with Mayer s hematoxylin, and pho- tographed under dark- and bright-field illumination. Specific- ity of hybridization signals observed with antisense probes was verified by two negative controls: absence of hybridization signal with corresponding sense probes and abolition of hybrid- ization signal when slides were pretreated with ribonuclease A (200 mg/ml) before hybridization with antisense probe. Both controls were uniformly negative, and examples of sense controls are shown in Results. IGF-I Radioimmunoassay Serum was extracted with acid-ethanol to remove IGFBPs. IGF-I radioimmunoassay was then performed using IGF-I polyclonal antibody UBK487 as described previously. 29 This antibody recognizes both human and mouse IGF-I. 30 Statistical Analysis Values are expressed as means { SE. To compare values from IGF-I transgenics and WT mice in different bowel seg- ments and in different feeding groups and to allow comparisons of data obtained for IGF-I and GH transgenics, some data are expressed as ratios of the value observed in the transgenic mouse relative to the value in the corresponding age- and sex- matched WT littermate. Statistical comparisons were performed using nonparametric tests: the Mann Whitney U test for independent groups and the Wilcoxon signed rank test for paired values. A P value of õ0.05 was considered statistically significant. Results Body Weight and Daily Diet Intake Body weight of IGF-I transgenics fed ad libitum In Situ Hybridization Histochemistry was 38% greater than that of WT littermates (Table 1). Body weight of pair-fed IGF-I transgenic mice was also Segments of duodenum, jejunum, and ileum (0.5 cm significantly greater than WT mice. Ad libitum diet each) were embedded in OCT compound (Miles, Elkhart, IN) intake was significantly greater in IGF-I transgenics than and frozen in isopentane at 040 C to050 C. Tissues were stored at 080 C before sectioning. Frozen sections (10 mm) WT mice (5.8 { 0.3 vs. 3.8 { 0.4 g/day; P õ 0.01). were prepared using a cryostat. Sections were then fixed with IGF-I transgenics lost 1.1 { 0.2 g body wt during the 4% paraformaldehyde and washed in phosphate-buffered sastriction, whereas WT mice gained 0.6 { 0.2 g body pair-feeding period, which is indicative of nutrient reline. Sections were treated with proteinase K (0.5 mg/ml) for 5 minutes, acetylated with triethylammonium (0.1 mol/l) and wt during the same period.
4 February 1997 SMALL BOWEL GROWTH IN IGF I TRANSGENIC MICE 447 Table 1. Body Weight, Length of Small Intestine, and Wet Weight of Bowel Segments of IGF-I Transgenics and WT Littermates Ad libitum fed Pair fed TG WT TG WT Body weight (g) 27.6 { 1.0 a 20.2 { { 2.0 a 22.4 { 1.8 Small intestine (cm) 50.0 { 1.4 a 37.7 { { 2.3 a 36.5 { 1.1 Duodenum (mg/cm) 44.1 { 2.0 a 34.1 { { 4.6 a 31.9 { 4.7 Jejunum (mg/cm) 54.2 { 2.2 a 35.4 { { 3.8 a 31.6 { 2.8 Ileum (mg/cm) 33.1 { 2.9 a 20.8 { { 1.1 a 20.8 { 1.1 NOTE. Five pairs of mice were studied in each feeding group. Values are expressed as means { SE. TG, IGF-I transgenics. a P õ 0.05 compared with WT. an increase in protein content and not in wet weight or DNA content (data not shown). In both feeding groups, mucosal disacharidase activi- ties did not differ between IGF-I transgenics and WT mice except for total lactase activity, which was significantly greater in WT mice compared with IGF-I transgenics in the pair-fed group (Figure 2). Length and Weight of Bowel The length and wet weight of the entire small intestine and the wet weights of duodenum, jejunum, and ileum were significantly greater in IGF-I transgenics than in WT littermates in both feeding groups (Ta- ble 1). Mucosal Mass and Brush Border Enzyme Activities In the ad libitum feeding group, mucosal wet weight, DNA content, and protein content were signifi- cantly greater in IGF-I transgenics than in WT lit- termates in all bowel segments (Figure 1). The increases in mucosal mass in IGF-I transgenics were more pro- nounced in duodenum and jejunum than ileum. In- creased mass of jejunal mucosa was greater than the increase in body weight. Essentially similar increases in mucosal mass also were observed in IGF-I transgenic mice that were pair fed with WT littermates (data not shown). As in the ad libitum fed animals, the effect was more pronounced in upper small bowel than in ileum. In fact, in pair-fed IGF-I transgenics, ileum showed only Histological Analysis of Mucosal Growth Villus height and crypt depth were measured in jejunum because this segment showed the most dramatic increase in mucosal mass. Both villus height and crypt depth were significantly greater in IGF-I transgenics compared with WT mice from both feeding groups (Figure 3). Crypt depth in the ad libitum fed IGF-I transgenics was significantly greater than in the pair-fed IGF-I transgenics (Figure 3). Epithelial Mitoses in Microdissected Crypts Numbers of crypt cell mitoses were measured in jejunum and ileum of pair-fed mice to assess whether Figure 1. Mass of small bowel mucosa in ad libitum fed IGF-I transgenic mice ( ) and WT littermates ( ), measured as wet weight, DNA content, and protein content per centimeter length, is shown for duodenum (Duod ), jejunum (Jej ), and ileum. Values shown are means { SE (n Å 5). a P õ 0.05 IGF-I transgenics vs. WT mice. b P õ 0.05 for values for ratio of bowel mass IGF-I transgenics/wt mice compared with ratio of body weight for IGF-I transgenics/wt mice.
5 448 OHNEDA ET AL. GASTROENTEROLOGY Vol. 112, No. 2 Figure 2. (A) Sucrase and (B) lactase activities in jejunal mucosa of IGF-I transgenics ( ) and WT littermates ( ). Activity is expressed as milliunits per centimeter of bowel length (total activity ) and as milliunits per gram of protein (specifi activity ). Values shown are means { SE (n Å 10 for sucrase activities of ad libitum [Ad-Lib] fed group; n Å 9 for sucrase activities of pair-fed group; and n Å 5 for lactase activities ). a P õ 0.05 IGF-I transgenics vs. WT mice. changes in mucosal mass were associated with increases in crypt cell proliferation. In jejunum, numbers of mitoses per crypt in IGF-I transgenics (6.2 { 0.9) were significantly greater than values observed in WT mice (4.1 { 0.6; P Å 0.028). However, there was no significant Figure 4. Abundance of human IGF-I transgene derived mrna in small bowel. (A) The top row shows representative autoradiograms of Northern blot hybridization of human IGF-I (higf-i) mrnas in jejunum of IGF-I transgenics (TG ) and WT littermates (WT ). The bottom row shows actin mrna used as a control for RNA loading. (B) The top row shows representative autoradiogram of human IGF-I mrnas in duodenum (Duod ), jejunum (Jej ), and ileum of IGF-I transgenics. The bottom row shows actin mrna used as a control for RNA loading. (C ) Abundance of human IGF-I mrnas in jejunum ( ) and ileum ( ) of IGF-I transgenics expressed as a ratio relative to the abundance in duodenum. Values are expressed as means {SE (n Å 4). a P õ 0.05 vs. values from duodenum. IGF-I mrna abundance in each sample was normalized to actin mrna abundance on the same blots. Ad-Lib, ad libitum. difference in the number of crypt cell mitoses in the ileum of IGF-I transgenic (4.8 { 0.5) and WT mice (4.6 { 0.8; P Å 0.6). Figure 3. Villus height and crypt depth in IGF-I transgenics ( ) and WT littermates ( ). Values shown are means { SE (n Å 4 for ad libitum [Ad Lib] fed group and n Å 5 for pair-fed group). a P õ 0.05 IGF-I transgenics vs. WT mice; b P õ 0.05 ad libitum fed vs. pair-fed IGF-I transgenics. Levels and Localization of Human IGF-I Transgene Expression in Different Bowel Segments As shown in Figure 4A, the human IGF-I transgene mrna was highly expressed in jejunum of IGF-I transgenics, whereas expression was undetectable in WT mice (the human IGF-I complementary RNA probe does not recognize endogenous IGF-I mrna un-
6 February 1997 SMALL BOWEL GROWTH IN IGF I TRANSGENIC MICE 449 Figure 5. In situ hybridization analysis of human IGF-I mrna in jejunum of (A and D) WT littermates and (B, C, E, and F ) IGF-I transgenics fed ad libitum. (A C) Bright-fiel and (D F) dark-fiel photomicrographs of representative autoradiographs of sections of jejunum hybridized with (A, B, D, and E ) an antisense probe complementary to human IGF-I mrna and with (C and F ) a control sense probe are shown (original magnificatio 251). IGF-I mrna was not detectable in bowel of WT mice using the human IGF-I complementary RNA probe (Fig- ure 5). Similar results were observed in sections of duodenum and ileum from ad libitum fed mice and in sections der the hybridization conditions used). Northern hybridization analysis was used to measure the levels of human IGF-I transgene expression in different small bowel segments and, thereby, allows us to assess whether there was a correlation between transgene expression and growth effects. These analyses were performed on pair-fed IGF- I transgenics and WT mice only. The human IGF-I transgene was expressed at high levels in all regions of small bowel of transgenic animals (Figure 4B). Because there was variability in transgene expression in different animals, an abundance of transgene expression in jejunum and ileum of a given transgenic animal was expressed as a ratio of that observed in duodenum of the same animal to facilitate comparisons along the length of small bowel. As shown in Figure 4C, abundance of human IGF-I transgene expression was significantly lower in ileum than in duodenum in both ad libitum and pair-fed transgenics, whereas abundance of transgene expression did not differ between duodenum and jejunum of transgenics. In situ hybridization was performed on sections of jejunum of ad libitum fed IGF-I transgenics and WT mice. Human IGF-I mrna was expressed at very high levels in villus epithelial cells and at much lower, but significant levels, in crypt epithelial cells, lamina propria, and smooth muscle layers (Figures 5 and 6A and C). Figure 6. High-power bright-fiel photographs of (A and C ) human IGF- I and (B and D) IGFBP-3 hybridization signals in (A and B) villi and (C and D) crypt region of jejunum from IGF-I transgenics (original magnifi cation 1501).
7 450 OHNEDA ET AL. GASTROENTEROLOGY Vol. 112, No. 2 Figure 7. Abundance of endogenous IGFBP-3 mrnas in small bowel. (A) The top row shows a representative autoradiogram of IGFBP-3 mrnas in duodenum (Duod ), jejunum (Jej ), and ileum of IGF-I transgenics. The bottom row shows actin mrna used as a control for RNA loading. (B) Abundance of IGFBP-3 mrnas in jejunum ( ) and ileum ( ) of IGF-I transgenics expressed as a ratio relative to the abundance in duodenum. Values are expressed as means { SE (n Å 4). a P õ 0.05 vs. values from duodenum. IGFBP-3 mrna abundance in each sample was normalized to actin mrna abundance on the same blots. Ad-Lib, ad libitum. (C ) The top row shows a representative autoradiogram of IGFBP-3 mrnas in the jejunum of IGF-I transgenic (TG ) and WT littermate (WT ) and bovine GH transgenic and WT littermate. The bottom row shows actin mrna used as a control for RNA loading. (D) Abundance of IGFBP-3 mrnas of human IGF-I transgenics (higf-i) and bovine GH transgenics (bgh ) expressed as a ratio relative to the IGFBP-3 mrna abundance in their corresponding WT littermates. Values are expressed as means { SE (n Å 4). a P õ 0.05 for GH transgenics vs. values from IGF-I transgenics (TG ). All results were normalized by calculation with abundance of actin mrna on the same blot. Note that analyses were performed with GH transgenics and WT mice that were of similar age to IGF-I transgenics and their WT controls and with all samples processed on the same blot. of duodenum, jejunum, and ileum from pair-fed mice (data not shown). Abundance and Localization of IGFBP-3 mrna in Jejunum of IGF-I Transgenic and GH Transgenic Mice Local expression of IGFBP-3 may be an important determinant of IGF-I responsiveness or actions within small bowel. 18,19 Therefore, we assessed the abundance of IGFBP-3 mrna in different small bowel segments of IGF-I transgenic animals. Because local levels or sites of expression of IGFBP-3 in bowel could influence IGF- I action in IGF-I transgenic or in GH transgenic mice, we also compared the abundance and sites of IGFBP-3 mrna expression in jejunum of IGF-I transgenics and WT littermates and GH transgenics and WT littermates. As shown in Figure 7, IGFBP-3 was expressed in all segments of small bowel of IGF-I transgenics. As with the IGF-I transgene, abundance of IGFBP-3 mrna was significantly lower in ileum than in duodenum in both ad libitum fed and pair-fed IGF-I transgenics (Figure 7B). IGFBP-3 mrna abundance in duodenum and jeju- num of IGF-I transgenics did not differ among mice from each feeding group (Figure 7B). In jejunum of both IGF-I and GH transgenics, abundance of IGFBP-3 mrna was greater than in their corresponding age- and sex-matched WT littermates (Figure 7C). However, the ratio of abundance of IGFBP-3 mrna in IGF-I trans- genics vs. WT mice was significantly greater than in GH transgenics vs. WT littermates (Figure 7D). Localization of endogenous IGFBP-3 mrna was evalu- ated on sections of small bowel from IGF-I transgenics and WT mice and from GH transgenics and WT mice. Results from WT littermates of GH transgenics were similar to those from WT littermates of IGF-I transgenics, and thus, data are shown only from a single WT animal. As shown
8 February 1997 SMALL BOWEL GROWTH IN IGF I TRANSGENIC MICE 451 Figure 8. In situ hybridization analysis of endogenous IGFBP-3 mrna in jejunum of (A and D) WT mouse, (B and E ) GH transgenic, and (C and F ) IGF-I transgenic. (A C) Bright-fiel and (D F) dark-fiel photomicrographs of representative autoradiographs of sections of jejunum hybridized with the antisense probe complementary to human IGFBP-3 mrna are shown (original magnificatio 251). was no evidence for expression of IGFBP-3 mrna within mucosal epithelial cells (Figure 6B). High-power photomicrographs of the crypt region show that, al- though the IGF-I transgene is expressed in epithelial cells within the crypt region, it is at lower levels than in villus cells and is not associated with increased IGFBP- 3 expression in adjacent lamina propria (Figure 6C and D). Similar results were observed in sections of duodenum and ileum from ad libitum fed mice and in sections of duodenum, jejunum, and ileum from pair-fed mice (data not shown). Serum IGF-I Levels of IGF-I Transgenics and WT Littermates Serum IGF-I levels were increased in IGF-I transgenics compared with WT littermates in both feeding groups (852.2 { 86.8 vs { 30.1 ng/ml; P Å 0.009; n Å 5 in the ad libitum fed group; and { 72.0 vs { 26.6 ng/ml; P Å 0.009; n Å 5in the pair-fed group). Serum IGF-I concentrations did not differ between ad libitum fed and pair-fed IGF-I transgenics (P Å 0.465). Discussion We have shown that long-term excess of IGF-I caused by expression of a human IGF-I transgene results in sections of jejunum in Figures 6B and 8, endogenous IGFBP-3 mrna was localized only in subepithelial cells within lamina propria. In contrast with localization of transgene-derived human IGF-I mrna, IGFBP-3 mrna was not detected in lamina propria within the crypt region (Figure 6D). The intensity of hybridization signal for IGFBP-3 mrna was markedly stronger in both IGF- I and GH transgenics than WT littermates (Figure 8). Consistent with the results of Northern blot analyses, the intensity of in situ hybridization signal for IGFBP-3 mrna was much stronger in jejunum of IGF-I transgenics than that of GH transgenics (Figure 8). In IGF-I transgenics, the enhanced expression of IGFBP-3 was particularly evident in villus regions adjacent to sites of high-level IGF-I transgene expression (compare Figures 5 and 8). Therefore, we hybridized adjacent sections of jejunum from IGF-I transgenics with either the IGF-I probe or the IGFBP-3 probe to obtain a closer comparison of sites of transgene-derived IGF-I and endogenous IGFBP-3 mrnas. High-power photo- micrographs showed very high-level expression of IGF- I transgene in villus epithelial cells and lower levels of expression in lamina propria cells (Figure 6A). IGFBP-3 mrna was expressed at extremely high levels in lamina propria cells adjacent to villus epithelial cells, and there
9 452 OHNEDA ET AL. GASTROENTEROLOGY Vol. 112, No. 2 in increased length and mass of small bowel in adult somatic growth is more prominent in IGF-I transgenics mice. Excess IGF-I appears to promote an increase in cell than in GH transgenics. number within the small bowel mucosa as shown by an Although the effects on small bowel growth were simiincrease in mucosal DNA and protein content per unit lar in both GH and IGF-I transgenics, the IGF-I length of bowel. Although the brush border sucrase activ- transgenics are GH deficient because of negative feedback ity per unit length of bowel and specific enzyme activity effects of excess IGF-I. 17 This observation suggests that per unit protein were not significantly increased in jeju- IGF-I mediates most of the enterotrophic effects of GH. num of IGF-I transgenics compared with WT lit- One exception appears to be mucosal sucrase activity. In termates, the increased small bowel length in IGF-I GH transgenics, there were significant increases in mucotransgenics resulted in an increase in total sucrase activ- sal sucrase activity expressed as total activity per unit ity, suggesting that excess IGF-I enhances digestive and length of small bowel. 16 In a large group of IGF-I absorptive capabilities of small bowel. The effects of ex- transgenics, sucrase activity did not differ between IGFcess IGF-I remained in transgenics that were nutrient I transgenics and WT mice. In contrast to the IGF-I restricted by pair feeding with WT littermates, indicat- transgenics, GH transgenics did not show any evidence ing that the enterotrophic effects of IGF-I are not depen- of increased crypt cell proliferation. 16 These observations dent on increased nutrient intake. Increased numbers of suggest that GH has greater differentiative actions, crypt cell mitoses in jejunum of IGF-I transgenics sug- whereas IGF-I exerts primarily mitogenic actions. Simigest that excess IGF-I promotes increased crypt cell pro- lar differences in the growth-promoting effects of GH liferation and/or increased size of the proliferative com- and IGF-I have been reported for preadipocytes and partment of the crypts. Consistent with the current growth plate chondrocytes. 32,33 observations, recent reports indicate that systemic IGF- IGF-I transgenics have increased circulating levels of I administered to young adult rats for 14 days results in IGF-I and also local small bowel overexpression of the increased small bowel mass that is associated with in- IGF-I transgene. Therefore, the enterotrophic effects obcreased crypt cell production and increased size of the served in the IGF-I transgenics may be derived from crypt cell proliferative compartment. 31 both endocrine and paracrine/autocrine actions of IGF- The increase in mucosal mass comparing IGF-I I. Our studies do not directly distinguish between the transgenics to WT littermates was greater in proximal endocrine and local effects of IGF-I. However, in IGF-I small bowel than in distal small bowel. This may in part transgenics, enhanced growth of proximal compared with reflect somewhat higher levels of transgene expression in distal small bowel correlates with higher levels of local proximal small bowel. However, systemic IGF-I also had transgene expression. This supports the notion that lo- a more prominent effect in proximal than distal small cally expressed IGF-I is responsible, at least in part, for bowel, 31 indicating that other factors may contribute to observed enterotropic effects. the enhanced effect of IGF-I excess on proximal small It is clear that systemic IGF-I can act in an endocrine bowel. One possibility is higher level expression of type manner to increase small bowel mass of rats after massive 1 IGF-I receptors in proximal small bowel, although this small bowel resection, 3 5 after transplantation of small has not been examined in detail. bowel, 4 and during catabolic weight loss induced by We have reported previously that transgenic mice with dexamethasone treatment, 6 as well as in normal rats. 31 long-term GH excess caused by overexpression of a bobowel In each of these studies, an increased cross-sectional vine GH transgene showed hyperplasia and hypertrophy mass, rather than increased gut length, 6,31 was the of small bowel mucosa. 16 It is possible to compare our dominant effect of IGF-I administration. A major in- previous data in GH transgenics with those reported here crease in small bowel length was observed in IGF-I in the IGF-I transgenics, because both studies used the transgenics. This effect on bowel length may reflect ef- same experimental design and because samples were anaof fects of local IGF-I overexpression in bowel or effects lyzed using the same methods. Body weights of GH early and prolonged IGF-I overexpression throughout transgenics were almost double those of WT littermates, neonatal development. whereas body weights of IGF-I transgenics were only IGFBP-3 has long been established as a GH-dependent 38% greater than WT. However, absolute values of the circulating IGFBP. Studies in GH-deficient trans- length of entire small intestine and mucosal mass of each genic mice and in the original line of IGF-I transgenics small bowel segment were very similar in GH and IGF- provide evidence that IGF-I may be the mediator of GH- I transgenics. These observations suggest that the effect dependent increases in serum IGFBP However, the of transgene on small bowel growth relative to that on regulation of IGFBP-3 expression in IGF-I target organs
10 February 1997 SMALL BOWEL GROWTH IN IGF I TRANSGENIC MICE 453 has not been well studied. Our observation of increased small bowel IGFBP-3 expression in IGF-I transgenics with relative GH deficiency suggests that IGF-I is a primary regulator of IGFBP-3 in small bowel. Further- more, the anatomic proximity of the villus epithelial cells expressing the IGF-I transgene to the villus lamina propria cells that show increased IGFBP-3 mrna sug- gests that IGF-I acts in a paracrine manner to increase IGFBP-3 expression in neighboring lamina propria cells. A number of studies suggest that locally expressed IGFBP-3 in small bowel mucosa may limit enterotrophic effects of IGF-I. 3,5,6,19,20 It is difficult to assess the local rat. Growth 1978; 42: interaction of IGF-I and IGFBP-3 in vivo with certainty, but the expression of IGFBP-3 in the villi of IGF-I transgenics may lead to an inhibition of IGF-I action on mature villus cells and may contribute to the lack of effect of IGF-I excess on sucrase activity. In contrast, our observation that the expression of IGFBP-3 mrna in crypts is undetectable in IGF-I transgenics may explain the prominent effect of IGF-I on crypt cell proliferation, despite the relative low levels of local expression of the 1988; 123: IGF-I transgene in the crypt area. tion of insulin like growth factor 1 receptors in the rat gut. Gut 1993; 34: Lund PK, Moats Staats BM, Hynes MA, Simmons JG, Jansen M, D Ercole AJ, Van Wyk JJ. Somatomedin-C/insulin-like growth factor I and insulin-like growth factor II mrnas in rat fetal and adult tissues. J Biol Chem 1986; 261: Han VK, Lund PK, Lee DC, D Ercole AJ. Expression of soma- tomedin/insulin-like growth factor messenger ribonucleic acids in the human fetus: identification characterization, and tissue distribution. J Clin Endocrinol Metab 1988; 66: Lund PK. Insulin-like growth factors. In: Walsh JH, Dockray GJ, eds. Gut peptides: biochemistry and physiology. In: Martini L, ed. Comprehensive endocrinology. New York: Raven, 1994: Yeh K-Y, Moog F. Hormonal influence on the growth and enzymic differentiation of the small intestine of the hypophysectomized 15. Hart MH, Phares CK, Erdman SH, Grandjean CJ, Park JHY, Vanderhoof JA. Augumentation of postresection mucosal hyperplasia by pleocercoid growth factor (PGF). Analog of human growth hor- mone. Dig Dis Sci 1987; 32: Ulshen MH, Dowling RH, Fuller CR, Zimmermann EM, Lund PK. Enhanced growth of small bowel in transgenic mice overex- pressing bovine growth hormone. Gastroenterology 1993; 104: 17. Mathews LS, Hammer RE, Behringer RR, D Ercole AJ, Bell GI, Brinster RL, Palmiter RD. Growth enhancement of transgenic mice expressing human insulin-like growth factor I. Endocrinology 18. Rechler MM, Brown AL. Insulin-like growth factor binding proteins: gene structure and expression. Growth Regul 1992; 2: References 19. Winesett DE, Ulshen MH, Hoyt EC, Mohapatra NK, Fuller CR, Lund PK. Regulation and localization of the insulin-like growth 1. Baker J, Liu JP, Robertson EJ, Efstratiadis A. Role of insulin-like factor system in small bowel during altered nutrient status. Am growth factors in embryonic and postnatal growth. Cell 1993; J Physiol 1995; 268:G631 G : Albiston AL, Taylor RG, Herington AC, Beveridge DJ, Fuller PJ. 2. Behringer RR, Lewin TM, Quaife CJ, Palmiter RD, Brinster RL, Divergent ileal IGF-I and IGFBP-3 gene expression after small D Ercole AJ. Expression of insulin-like growth factor I stimulates bowel resection: a novel mechanism to amplify IGF action? Mol normal somatic growth in growth hormone-deficien transgenic Cell Endocrinol 1992; 83:R17 R20. mice. Endocrinology 1990; 127: Ye P, Carson J, D Ercole AJ. In vivo actions of insulin-like growth 3. Vanderhoof JA, McCusker RH, Clark R, Mohammadpour H, Blackfactor I (IGF-I) on brain myelination: Studies of IGF-I and IGF wood DJ, Harty RF, Park JH. Truncated and native insulin-like binding protein 1 (IGFBP-1) transgenic mice. J Neurosci 1995; growth factor I enhance mucosal adaptation after jejunoileal re- 15: section. Gastroenterology 1992; 102: CeSarone CF, Bolognesi C, Santi L. Improved microfluorometri 4. Zhang W, Frankel WL, Adamson WT, Roth JA, Mantell MP, Bain DNA determination in biological material using Hoechst. A, Ziegler TR, Smith RJ, Rombeau JL. Insulin-like growth factor Anal Biochem 1979; 100: I improves mucosal structure and function in transplanted rat 23. Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ. Protein measuresmall intestine. Transplantation 1995; 59: ment with the folin phenol reagent. J Biol Chem 1951; 193: Lemmey AB, Martin AA, Read LC, Tomas FM, Owens PC, Ballard 275. FJ. IGF-I and the truncated analogue des-(1-3)igf-i enhance growth 24. Messer M, Dahlqvist A. A one step ultramicromethod for the in rats after gut resection. Am J Physiol 1991; 260:E213 E219. assay of intestinal disaccharidases. Anal Biochem 1966; 14: 6. Read LC, Tomas FM, Howarth GS, Martin AA, Edson KJ, Gillespie CM, Owens PC, Ballard FJ. Insulin-like growth factor I and its N 25. Grand RJ, Chong DA, Isselbacher KJ. Intracellular processing of terminal modifie analogues induce marked gut growth in dexa- disaccharidases: the effect of actinomycin D. Biochim Biophys methasone-treated rats. J Endocrinol 1992; 133: Acta 1972; 261: Young GP, Taranto TM, Jonas HA, Cox AJ, Hogg A, Werther GA. 26. Goodlad RA, Wright NA. Quantitative studies on epithelial replace- Insulin-like growth factors and the developing and mature rat ment in the gut. In: Titchen DA, ed. Techniques in the life scismall intestine: receptors and biological actions. Digestion ences. Digestive physiology. Volume P212. Ireland: Elsevier, 1990; 46(Suppl 2): : Laburthe M, Rouyer Fessard C, Gammeltoft S. Receptors for 27. Zimmermann EM, Sartor RB, McCall RD, Pardo M, Bender D, insulin-like growth factors I and II in rat gastrointestinal epithe- Lund PK. Insulin-like growth factor I and interleukin 1b messenger lium. Am J Physiol 1988; 254:G457 G462. RNA in a rat model of granulomatous enterocolitis and hepatitis. 9. Termanini B, Nardi RV, Finan TM, Parikh I, Korman LY. Insulin- Gastroenterology 1993; 105: like growth factor I receptors in rabbit gastrointestinal tract. Char- 28. Jansen M, van Schaik FMA, Ricker AT, Bullock B, Woods DE, acterization and autoradiographic localization. Gastroenterology Gabbay KH, Nussbaum AL, Sussenbach JS, Van den Brade JL. 1990; 99: Sequence of cdna encoding human insulin-like growth factor I 10. Ryan J, Costigan DC. Determination of the histological distribu- precursor. Nature 1983; 306:
11 454 OHNEDA ET AL. GASTROENTEROLOGY Vol. 112, No Copeland KC, Underwood LE, Van Wyk JJ. Induction of immunore- with altered expression of growth hormone and IGF-I. Endocrinology active somatomedin-c in human serum by growth hormone: dose 1991; 129: response relationships and effect on chromatographic profiles J Clin Endocrinol Metab 1980; 50: Received April 4, Accepted October 8, D Ercole AJ, Applewhite GT, Underwood LE. Evidence that soma- Address requests for reprints to: P. Kay Lund, Ph.D., Department tomedin is synthesized by multiple tissues in fetus. Dev Biol of Physiology, University of North Carolina at Chapel Hill, CB no. 1980; 75: , Chapel Hill, North Carolina Fax: (919) Steeb CB, Trahair JF, Tomas FM, Read LC. Prolonged administra tion of IGF peptides enhances growth of gastrointestinal tissues Supported by National Institutes of Health (NIH) grants DK40247 in normal rats. Am J Physiol 1994; 266:G1090 G1098. (to P.K.L.) and HD08299 (to A.J.D.) and NIH grant DK02131 to the 32. Green H, Morikawa M, Nixon T. A dual effector theory of growth molecular biology and transgenic core facilities of the Center for hormone action. Differentiation 1985; 29: Gastrointestinal Biology and Disease. 33. Isalsson OG, Lindahl A, Nilsson A, Isgaard J. Cellular mecha- The authors thank Eyvonne Bruton and Dr. Louis Underwood for nism(s) for the stimulatory effect of growth hormone on longitudi- assistance with insulin-like growth factor I radioimmunoassays, nal bone growth. In: Isaksson O, Binder C, Hall K, Hokfelt B, Dr. Robert Bagnell for his help with providing photomicrographs, eds. Growth hormone: basic and clinical aspects. Amsterdam: Frank Criscione for technical assistance, Drs. J. Leo Van den Excerpta Medica, 1987: Brande and Marten Jansen for provision of the human insulin-like 34. Camacho Hubner C, Clemmons DR, D Ercole AJ. Regulation of growth factor I complementary DNA, and Drs. Judson J. Van Wyk insulin-like growth factor (IGF) binding proteins in transgenic mice and Ellen Zimmermann for many useful discussions.
Mesenchymal IGF-I overexpression: paracrine effects in the intestine, distinct from endocrine actions
 Am J Physiol Gastrointest Liver Physiol 283: G875 G885, 2002. First published June 5, 2002; 10.1152/ajpgi.00089.2002. Mesenchymal IGF-I overexpression: paracrine effects in the intestine, distinct from
Am J Physiol Gastrointest Liver Physiol 283: G875 G885, 2002. First published June 5, 2002; 10.1152/ajpgi.00089.2002. Mesenchymal IGF-I overexpression: paracrine effects in the intestine, distinct from
Effect of subcutaneous insulin on intestinal adaptation in a rat model of short bowel syndrome
 Pediatr Surg Int (2005) 21: 132 137 DOI 10.1007/s00383-004-1308-2 ORIGINAL ARTICLE Igor Sukhotnik Æ Jorge Mogilner Æ Raanan Shamir Naim Shehadeh Æ Jacob Bejar Æ Mark Hirsh Arnold G. Coran Effect of subcutaneous
Pediatr Surg Int (2005) 21: 132 137 DOI 10.1007/s00383-004-1308-2 ORIGINAL ARTICLE Igor Sukhotnik Æ Jorge Mogilner Æ Raanan Shamir Naim Shehadeh Æ Jacob Bejar Æ Mark Hirsh Arnold G. Coran Effect of subcutaneous
THE SMALL intestinal mucosa exhibits an extremely
 0013-7227/98/$03.00/0 Vol. 139, No. 7 Endocrinology Printed in U.S.A. Copyright 1998 by The Endocrine Society Intestinal Adaptation after Extensive Small Bowel Resection: Differential Changes in Growth
0013-7227/98/$03.00/0 Vol. 139, No. 7 Endocrinology Printed in U.S.A. Copyright 1998 by The Endocrine Society Intestinal Adaptation after Extensive Small Bowel Resection: Differential Changes in Growth
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H.
 UvA-DARE (Digital Academic Repository) Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H. Link to publication Citation for published version (APA):
UvA-DARE (Digital Academic Repository) Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H. Link to publication Citation for published version (APA):
STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT
 GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT ELLIOT WESER, M.D., AND MARY H.
GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT ELLIOT WESER, M.D., AND MARY H.
Parenteral Nutrition. Outline. Potential Biomarkers for Use in Intestinal Adaptation. Jejunum is primary site of digestion and absorption
 Outline Potential Biomarkers for Use in Intestinal Adaptation Kelly A. Tappenden, Ph.D., R.D. Professor of Nutrition and GI Physiology 1. Intestinal Adaptation potential regulators 2. Intestinal mucosal
Outline Potential Biomarkers for Use in Intestinal Adaptation Kelly A. Tappenden, Ph.D., R.D. Professor of Nutrition and GI Physiology 1. Intestinal Adaptation potential regulators 2. Intestinal mucosal
Genotype analysis by Southern blots of nine independent recombinated ES cell clones by
 Supplemental Figure 1 Selected ES cell clones show a correctly recombined conditional Ngn3 allele Genotype analysis by Southern blots of nine independent recombinated ES cell clones by hybridization with
Supplemental Figure 1 Selected ES cell clones show a correctly recombined conditional Ngn3 allele Genotype analysis by Southern blots of nine independent recombinated ES cell clones by hybridization with
IS THERE CIRCADIAN VARIATION IN VILLUS HEIGHT IN RAT SMALL INTESTINE?
 Quarterly Journal of Experimental Physiology (1989) 74, 257-265 Printed in Great Britain IS THERE CIRCADIAN VARIATION IN VILLUS HEIGHT IN RAT SMALL INTESTINE? MICHAEL L. G. GARDNER AND DEBORAH L. STEELE
Quarterly Journal of Experimental Physiology (1989) 74, 257-265 Printed in Great Britain IS THERE CIRCADIAN VARIATION IN VILLUS HEIGHT IN RAT SMALL INTESTINE? MICHAEL L. G. GARDNER AND DEBORAH L. STEELE
Supplemental Data Tamoxifen administration to Vil-Scap- mice.
 Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
Lactase activity is under hormonal control in the intestine of adult rat
 Gut, 1983, 24, 648-652 Lactase activity is under hormonal control in the intestine of adult rat F RAUL, R NORIEGA, E NSI-EMVO, M DOFFOEL, AND J From the Unitd 61 de l'inserm, Strasbourg, France F GRENIER
Gut, 1983, 24, 648-652 Lactase activity is under hormonal control in the intestine of adult rat F RAUL, R NORIEGA, E NSI-EMVO, M DOFFOEL, AND J From the Unitd 61 de l'inserm, Strasbourg, France F GRENIER
General Structure of Digestive Tract
 Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
Ulcerative colitis and Crohn s disease (CD) are
 GASTROENTEROLOGY 2001;120:925 937 Enhanced Survival and Mucosal Repair After Dextran Sodium Sulfate Induced Colitis in Transgenic Mice That Overexpress Growth Hormone KRISTEN L. WILLIAMS,* C. RANDALL FULLER,
GASTROENTEROLOGY 2001;120:925 937 Enhanced Survival and Mucosal Repair After Dextran Sodium Sulfate Induced Colitis in Transgenic Mice That Overexpress Growth Hormone KRISTEN L. WILLIAMS,* C. RANDALL FULLER,
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
LACTOSE TOLERANCE TESTS IN ADULTS WITH NORMAL LACTASE ACTIVITY
 GASTROENTEROLOGY Copyright 1966 by The Williams & Wilkins Co. Vol. 50, No.3 Printed in U.S.A. LACTOSE TOLERANCE TESTS IN ADULTS WITH NORMAL LACTASE ACTIVITY ALBERT D. NEWCOMER, M.D., AND DOUGLAS B. MCGILL,
GASTROENTEROLOGY Copyright 1966 by The Williams & Wilkins Co. Vol. 50, No.3 Printed in U.S.A. LACTOSE TOLERANCE TESTS IN ADULTS WITH NORMAL LACTASE ACTIVITY ALBERT D. NEWCOMER, M.D., AND DOUGLAS B. MCGILL,
EFFECT OF NITROGEN SOURCE ON NUTRITIONAL MANAGEMENT AFTER SMALL BOWEL RESECTION IN RATS
 J. Nutr. Sci. Vitaminol., 24, 167-176, 1978 EFFECT OF NITROGEN SOURCE ON NUTRITIONAL MANAGEMENT AFTER SMALL BOWEL RESECTION IN RATS Tetsuya KISHI, Yasuo IWASAWA, Hiroshi ITOH, and Ichiro CHIBATA1 Department
J. Nutr. Sci. Vitaminol., 24, 167-176, 1978 EFFECT OF NITROGEN SOURCE ON NUTRITIONAL MANAGEMENT AFTER SMALL BOWEL RESECTION IN RATS Tetsuya KISHI, Yasuo IWASAWA, Hiroshi ITOH, and Ichiro CHIBATA1 Department
On the horizon: trophic peptide growth factors as therapy for neonatal short bowel syndrome.
 ISSN: 1472-8222 (Print) 1744-7631 (Online) Journal homepage: http://www.tandfonline.com/loi/iett20 On the horizon: trophic peptide growth factors as therapy for neonatal short bowel syndrome. Patricia
ISSN: 1472-8222 (Print) 1744-7631 (Online) Journal homepage: http://www.tandfonline.com/loi/iett20 On the horizon: trophic peptide growth factors as therapy for neonatal short bowel syndrome. Patricia
Short Bowel Syndrome: Medical management
 Short Bowel Syndrome: Medical management La Sindrome dell'intestino Corto in età pediatrica Brescia 18 marzo 2011 Jon A.Vanderhoof, M.D. Division of Pediatric GI Harvard Medical School Children s Hospital,
Short Bowel Syndrome: Medical management La Sindrome dell'intestino Corto in età pediatrica Brescia 18 marzo 2011 Jon A.Vanderhoof, M.D. Division of Pediatric GI Harvard Medical School Children s Hospital,
Three-dimensional structure of the human small intestinal mucosa in health and disease
 Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
COLLOID DROPLET FORMATION IN DOG THYROID IN VITRO
 COLLOID DROPLET FORMATION IN DOG THYROID IN VITRO Induction by Dibutyryl Cyclic-AMP I. PASTAN and S. HI. WOLLMAN. Froml the National Institute of Arthritis and Metabolic Diseases and the National Cancer
COLLOID DROPLET FORMATION IN DOG THYROID IN VITRO Induction by Dibutyryl Cyclic-AMP I. PASTAN and S. HI. WOLLMAN. Froml the National Institute of Arthritis and Metabolic Diseases and the National Cancer
Non-Invasive Assessment of Intestinal Function
 Overview Non-Invasive Assessment of Intestinal Function Introduction This paper will demonstrate that the 13 C-sucrose breath test ( 13 C-SBT) determines the health and function of the small intestine.
Overview Non-Invasive Assessment of Intestinal Function Introduction This paper will demonstrate that the 13 C-sucrose breath test ( 13 C-SBT) determines the health and function of the small intestine.
What s Golden in Colostrum: Communication from the Dam to the Calf
 What s Golden in : Communication from the Dam to the Calf Mike Van Amburgh Dept. of Animal Science Cornell University Email:mev1@cornell.edu; cell: 607-592-1212 What s Golden in : Communication from the
What s Golden in : Communication from the Dam to the Calf Mike Van Amburgh Dept. of Animal Science Cornell University Email:mev1@cornell.edu; cell: 607-592-1212 What s Golden in : Communication from the
Choosing the right phytase to improve sodium pump function, reduce the catabolism of amino acids and increase protein and glucose uptake
 Choosing the right phytase to improve sodium pump function, reduce the catabolism of amino acids and increase protein and glucose uptake Ha Truong PhD Candidate Poultry Research Foundation Faculty of Veterinary
Choosing the right phytase to improve sodium pump function, reduce the catabolism of amino acids and increase protein and glucose uptake Ha Truong PhD Candidate Poultry Research Foundation Faculty of Veterinary
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Ontogenic Timing Mechanism Initiates the Expression of Rat Intestinal Sucrase Activity
 GASTROENTEROLOGY 1986;9:52-6 Ontogenic Timing Mechanism Initiates the Expression of Rat Intestinal Sucrase Activity KWO-YIH YEH and PETER R. HOLT Division of Gastroenterology, st. Luke's-Roosevelt Hospital
GASTROENTEROLOGY 1986;9:52-6 Ontogenic Timing Mechanism Initiates the Expression of Rat Intestinal Sucrase Activity KWO-YIH YEH and PETER R. HOLT Division of Gastroenterology, st. Luke's-Roosevelt Hospital
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
IMMUNOHISTOCHEMICAL LOCALIZATION OF INSULIN-LIKE GROWTH FACTOR-I RECEPTORS IN THE RAT INTESTINE
 225 Bull Vet Inst Pulawy 50, 225-229, 2006 IMMUNOHISTOCHEMICAL LOCALIZATION OF INSULIN-LIKE GROWTH FACTOR-I RECEPTORS IN THE RAT INTESTINE BERRİN ZIK 1, ARTAY YAGCI 2, KORHAN ALTUNBAS 2 AND MİNE YAKIŞIK
225 Bull Vet Inst Pulawy 50, 225-229, 2006 IMMUNOHISTOCHEMICAL LOCALIZATION OF INSULIN-LIKE GROWTH FACTOR-I RECEPTORS IN THE RAT INTESTINE BERRİN ZIK 1, ARTAY YAGCI 2, KORHAN ALTUNBAS 2 AND MİNE YAKIŞIK
II. The effect of strain and IGF-1 gene transfer using SMGT techniques on some semen characteristics:-
 The experimental work of this study was carried out in poultry farm of the Department of Animal Production, Faculty of Agriculture, Benha University, Egypt with cooperation of Genetic Engineering and Biotechnology
The experimental work of this study was carried out in poultry farm of the Department of Animal Production, Faculty of Agriculture, Benha University, Egypt with cooperation of Genetic Engineering and Biotechnology
EFFECTS OF THE PROSTAGLANDIN ANALOGUE MISOPROSTOL ON CELL PROLIFERATION IN THE CANINE SMALL INTESTINE
 Experimental Physiology (1991), 76, 561-566 Printed in Great Britain EFFECTS OF THE PROSTAGLANDIN ANALOGUE MISOPROSTOL ON CELL PROLIFERATION IN THE CANINE SMALL INTESTINE R. A. GOODLAD, C. Y. LEE, S. LEVIN*
Experimental Physiology (1991), 76, 561-566 Printed in Great Britain EFFECTS OF THE PROSTAGLANDIN ANALOGUE MISOPROSTOL ON CELL PROLIFERATION IN THE CANINE SMALL INTESTINE R. A. GOODLAD, C. Y. LEE, S. LEVIN*
A Scanning Electron Microscope Study of the Effect of an Enterotoxin from Clostridium perfringens 8-6 on Mice of Different Ages
 ~ Journal of General Microbiology (1986), 132, 2893-2898. Printed in Great Britain 2893 A Scanning Electron Microscope Study of the Effect of an Enterotoxin from Clostridium perfringens 8-6 on Mice of
~ Journal of General Microbiology (1986), 132, 2893-2898. Printed in Great Britain 2893 A Scanning Electron Microscope Study of the Effect of an Enterotoxin from Clostridium perfringens 8-6 on Mice of
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
EPITHELIAL CELL KINETICS IN THE SMALL INTESTINE OF THE RAT 60 DAYS AFTER RESECTION OF 70 PER CENT OF THE ILEUM AND JEJUNUM
 GASTROENTEROLOGY Copyright 1971 by The Williams & Wilkins Co. Vol. 60, No.6 Printed in U. S. A. EPITHELIAL CELL KINETICS IN THE SMALL INTESTINE OF THE RAT 60 DAYS AFTER RESECTION OF 70 PER CENT OF THE
GASTROENTEROLOGY Copyright 1971 by The Williams & Wilkins Co. Vol. 60, No.6 Printed in U. S. A. EPITHELIAL CELL KINETICS IN THE SMALL INTESTINE OF THE RAT 60 DAYS AFTER RESECTION OF 70 PER CENT OF THE
Div. of Endocrinology and Metabolism Konyang University, School of Medicine. Byung-Joon Kim M.D., Ph.D.
 Diabetes and Thyroid Center Div. of Endocrinology and Metabolism Konyang University, School of Medicine Byung-Joon Kim M.D., Ph.D. Background Incretin effect and GLP 1 Receptor in L cells TGR5 receptor
Diabetes and Thyroid Center Div. of Endocrinology and Metabolism Konyang University, School of Medicine Byung-Joon Kim M.D., Ph.D. Background Incretin effect and GLP 1 Receptor in L cells TGR5 receptor
Doctor of Philosophy
 Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
What location in the gastrointestinal (GI) tract has tight, or impermeable, junctions between the epithelial cells?
 CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
Effect of Ileo-Jejunal Intestinal Adaptation
 Tohoku J. exp. Med., 1987, 151, 419-428 Effect of Ileo-Jejunal Intestinal Adaptation Colectomy in Dogs Transposition after Total on IWAO SASAKI, TAKASHI TUCHIYA, HIROO NAITO, YUJI FUNAYAMA, MORIHIKO TODA,
Tohoku J. exp. Med., 1987, 151, 419-428 Effect of Ileo-Jejunal Intestinal Adaptation Colectomy in Dogs Transposition after Total on IWAO SASAKI, TAKASHI TUCHIYA, HIROO NAITO, YUJI FUNAYAMA, MORIHIKO TODA,
Small Intestine, Large Intestine and anal cannel
 Small Intestine, Large Intestine and anal cannel 32409 Small intestine Large intestine Small intestine General Structure of the Digestive Tract rat 32409 Epithelium with goblet cells and absorptive cells
Small Intestine, Large Intestine and anal cannel 32409 Small intestine Large intestine Small intestine General Structure of the Digestive Tract rat 32409 Epithelium with goblet cells and absorptive cells
THE EFFECT OF DIET ON LACTASE ACTIVITY IN THE RAT
 GASTROENTEROLOGY Copyright 1971 by The Williams & Wilkins Co. Vol. 60, No.3 Printed in U. S. A. THE EFFECT OF DET ON LACTASE ACTVTY N THE RAT T. D. BOLN, M.B., M.R.C.P., ANNE McKERN, AND A. E. DAVS, M.D.,
GASTROENTEROLOGY Copyright 1971 by The Williams & Wilkins Co. Vol. 60, No.3 Printed in U. S. A. THE EFFECT OF DET ON LACTASE ACTVTY N THE RAT T. D. BOLN, M.B., M.R.C.P., ANNE McKERN, AND A. E. DAVS, M.D.,
Minimal Enteral Nutrition
 Abstract Minimal Enteral Nutrition Although parenteral nutrition has been used widely in the management of sick very low birth weight infants, a smooth transition to the enteral route is most desirable.
Abstract Minimal Enteral Nutrition Although parenteral nutrition has been used widely in the management of sick very low birth weight infants, a smooth transition to the enteral route is most desirable.
Hydrolysis and Absorption in the Small Intestines of Posthatch Chicks
 Hydrolysis and Absorption in the Small Intestines of Posthatch Chicks D. Sklan 1 and Y. Noy Faculty of Agriculture, Hebrew University, Rehovot, Israel 76-100 ABSTRACT In the immediate posthatch period,
Hydrolysis and Absorption in the Small Intestines of Posthatch Chicks D. Sklan 1 and Y. Noy Faculty of Agriculture, Hebrew University, Rehovot, Israel 76-100 ABSTRACT In the immediate posthatch period,
Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine
 FULL PAPER Anatomy Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine Takashi TAKEUCHI 1) and Tatsuo GONDA 1) 1) Institute of Experimental Animals, Shimane Medical University,
FULL PAPER Anatomy Distribution of the Pores of Epithelial Basement Membrane in the Rat Small Intestine Takashi TAKEUCHI 1) and Tatsuo GONDA 1) 1) Institute of Experimental Animals, Shimane Medical University,
Effects of Dietary Lipids on Recovery From Mucosal Injury
 GASTROENTEROLOGY 1990;98:1226-1231 Effects of Dietary Lipids on Recovery From Mucosal Injury JON A. VANDERHOOF, JUNG H. Y. PARK, HAMID MOHAMMADPOUR, and DARCY BLACKWOOD Department of Pediatrics, Creighton
GASTROENTEROLOGY 1990;98:1226-1231 Effects of Dietary Lipids on Recovery From Mucosal Injury JON A. VANDERHOOF, JUNG H. Y. PARK, HAMID MOHAMMADPOUR, and DARCY BLACKWOOD Department of Pediatrics, Creighton
DIRECT AND INDIRECT EFFECTS OF DEXTROSE AND AMINO ACIDS ON GUT MASS
 GASTROENTEROLOGY 72:76-71, 1977 Copyright 1977 by the American Gastroenterological Association Vol. 72, No.4, Part 1 Printed in U.S.A. DRECT AND NDRECT EFFECTS OF DEXTROSE AND AMNO ACDS ON GUT MASS MONROE
GASTROENTEROLOGY 72:76-71, 1977 Copyright 1977 by the American Gastroenterological Association Vol. 72, No.4, Part 1 Printed in U.S.A. DRECT AND NDRECT EFFECTS OF DEXTROSE AND AMNO ACDS ON GUT MASS MONROE
SHORT COMMUNICATION. Human Papillomavirus Type 11 E1 Ú E4 and L1 Proteins Colocalize in the Mouse Xenograft System at Multiple Time Points
 VIROLOGY 214, 259 263 (1995) SHORT COMMUNICATION Human Papillomavirus Type 11 E1 Ú E4 and L1 Proteins Colocalize in the Mouse Xenograft System at Multiple Time Points DARRON R. BROWN,*,,1 JANINE T. BRYAN,
VIROLOGY 214, 259 263 (1995) SHORT COMMUNICATION Human Papillomavirus Type 11 E1 Ú E4 and L1 Proteins Colocalize in the Mouse Xenograft System at Multiple Time Points DARRON R. BROWN,*,,1 JANINE T. BRYAN,
INTESTINAL CALCIUM TRANSPORT: COMPARISON OF DUODENUM AND ILEUM IN VIVO IN THE RAT
 GASTROENTEROLOGY Copyright 1972 by The Williams & Wilkins Co. Vol. 62, No.4 Printed in U.S.A. INTESTINAL CALCIUM TRANSPORT: COMPARISON OF DUODENUM AND ILEUM IN VIVO IN THE RAT M. K. YOUNOSZAI, M.D. AND
GASTROENTEROLOGY Copyright 1972 by The Williams & Wilkins Co. Vol. 62, No.4 Printed in U.S.A. INTESTINAL CALCIUM TRANSPORT: COMPARISON OF DUODENUM AND ILEUM IN VIVO IN THE RAT M. K. YOUNOSZAI, M.D. AND
Nutritional Requirements in Intestinal Failure
 Nutritional Requirements in Intestinal Failure Christopher Duggan, MD, MPH Center for Nutrition Center for Advanced Intestinal Rehabilitation (CAIR) Division of Gastroenterology, Hepatology and Nutrition
Nutritional Requirements in Intestinal Failure Christopher Duggan, MD, MPH Center for Nutrition Center for Advanced Intestinal Rehabilitation (CAIR) Division of Gastroenterology, Hepatology and Nutrition
CONTRACTING ORGANIZATION: University of Southern California Los Angeles, CA 90033
 AD Award Number: W81XWH-07-01-0264 TITLE: Function of Klotho and is MicroRNA in Prostate Cancer and Aging PRINCIPAL INVESTIGATOR: Shao-Yao Ying, Ph.D. CONTRACTING ORGANIZATION: University of Southern California
AD Award Number: W81XWH-07-01-0264 TITLE: Function of Klotho and is MicroRNA in Prostate Cancer and Aging PRINCIPAL INVESTIGATOR: Shao-Yao Ying, Ph.D. CONTRACTING ORGANIZATION: University of Southern California
Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC-
 Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
The Digestive System. Chapter 25
 The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
Dr Ricardo Esquerra Senior R&D Manager, Novus International
 Dr Ricardo Esquerra Senior R&D Manager, Novus International Monogastric nutrition for breeders, broilers and layers Sponsored by: 1 Highlights in Poultry Nutrition Ricardo Esquerra, PhD Senior R&D Manager
Dr Ricardo Esquerra Senior R&D Manager, Novus International Monogastric nutrition for breeders, broilers and layers Sponsored by: 1 Highlights in Poultry Nutrition Ricardo Esquerra, PhD Senior R&D Manager
Nutrition Management in GI Diseases
 Nutrition Management in GI Diseases Aryono Hendarto MD Nutrition & Metabolic Diseases Division Department of Child Health Cipto Mangunkusumo Hospital University of Indonesia 1 Patient s Care 1. Drugs 2.
Nutrition Management in GI Diseases Aryono Hendarto MD Nutrition & Metabolic Diseases Division Department of Child Health Cipto Mangunkusumo Hospital University of Indonesia 1 Patient s Care 1. Drugs 2.
Growth Factors in Inflammatory Bowel Disease
 BASIC SCIENCE REVIEW Growth Factors in Inflammatory Bowel Disease The Actions and Interactions of Growth Hormone and Insulin-Like Growth Factor-I Arianne L. Theiss, BA, Shira Fruchtman, PhD, and P. Kay
BASIC SCIENCE REVIEW Growth Factors in Inflammatory Bowel Disease The Actions and Interactions of Growth Hormone and Insulin-Like Growth Factor-I Arianne L. Theiss, BA, Shira Fruchtman, PhD, and P. Kay
Anatomy & Histology of The Small intestine
 Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Effect of glucose on beta cell proliferation and population size in organ culture of foetal and neonatal rat pancreases
 J. Embryol. exp. Morph. 74, 303-312 (1983) 3Q3 Printed in Great Britain The Company of Biologists Limited 1983 Effect of glucose on beta cell proliferation and population size in organ culture of foetal
J. Embryol. exp. Morph. 74, 303-312 (1983) 3Q3 Printed in Great Britain The Company of Biologists Limited 1983 Effect of glucose on beta cell proliferation and population size in organ culture of foetal
Small intestine. Small intestine
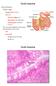 General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
ALTERATION OF LACTASE AND AMINOPEPTIDASE N EXPRESSION IN PORCINE ENTEROCYTES BY ENERGY INTAKE AND ENVIRONMENT
 Quarterly Journal of Experimental Physiology (1986) 71, 443-449 Printed in Great Britain ALTERATION OF LACTASE AND AMINOPEPTIDASE N EXPRESSION IN PORCINE ENTEROCYTES BY ENERGY INTAKE AND ENVIRONMENT M.
Quarterly Journal of Experimental Physiology (1986) 71, 443-449 Printed in Great Britain ALTERATION OF LACTASE AND AMINOPEPTIDASE N EXPRESSION IN PORCINE ENTEROCYTES BY ENERGY INTAKE AND ENVIRONMENT M.
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is
 Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
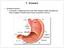 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
The Role of Apoptosis During Intestinal Adaptation After Small Bowel Resection
 The Role of Apoptosis During Intestinal Adaptation After Small Bowel Resection By Carlo F.M. Welters, Femke E. Piersma, David M. Hockenbery, and Erik Heineman Maastricht, The Netherlands; Seattle, Washington;
The Role of Apoptosis During Intestinal Adaptation After Small Bowel Resection By Carlo F.M. Welters, Femke E. Piersma, David M. Hockenbery, and Erik Heineman Maastricht, The Netherlands; Seattle, Washington;
University of Bristol - Explore Bristol Research. Peer reviewed version. Link to published version (if available): /vr.
 Turner, A. E., Rees, G., Barrett, D. C., & Reyher, K. K. (2016). Does inclusion of glutamine in oral rehydration solutions improve recovery from mild to moderate diarrhoea in preweaned calves? Veterinary
Turner, A. E., Rees, G., Barrett, D. C., & Reyher, K. K. (2016). Does inclusion of glutamine in oral rehydration solutions improve recovery from mild to moderate diarrhoea in preweaned calves? Veterinary
Biochemical Studies on the Mineral Components in Sake Yeast. Part V. The Relationship of the Mineral Composition of Yeast to Fermentation
 [Agr, Biol. Chem. Vol. 30, No. 9, p. 925 `930, 1966] Biochemical Studies on the Mineral Components in Sake Yeast Part V. The Relationship of the Mineral Composition of Yeast to Fermentation By Tsuyoshi
[Agr, Biol. Chem. Vol. 30, No. 9, p. 925 `930, 1966] Biochemical Studies on the Mineral Components in Sake Yeast Part V. The Relationship of the Mineral Composition of Yeast to Fermentation By Tsuyoshi
HUMAN NUTRITION: ABSORPTION & ASSIMILATION 14 MAY 2014
 HUMAN NUTRITION: ABSORPTION & ASSIMILATION 14 MAY 2014 In this lesson, we: Absorption Lesson Description Examine and understand absorption Define absorption and describe where it occurs Study the structure
HUMAN NUTRITION: ABSORPTION & ASSIMILATION 14 MAY 2014 In this lesson, we: Absorption Lesson Description Examine and understand absorption Define absorption and describe where it occurs Study the structure
Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis
 Vet. Pathol. 15; 400-406 (1978) Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis G. R. PEARSON. E. F. LOGAN and G. P. BRENNAN Departmcnt
Vet. Pathol. 15; 400-406 (1978) Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis G. R. PEARSON. E. F. LOGAN and G. P. BRENNAN Departmcnt
Digestion and Absorption
 Digestion and Absorption General Considerations - No absorption in esophagus, little in the stomach and vast majority of absorption occurs in small intestine. - The small intestine has specialized structures
Digestion and Absorption General Considerations - No absorption in esophagus, little in the stomach and vast majority of absorption occurs in small intestine. - The small intestine has specialized structures
2. A digestive organ that is not part of the alimentary canal is the: a. stomach b. liver c. small intestine d. large intestine e.
 Chapter 14 The Digestive System and Body Metabolism Review Questions Multiple Choice: 1. Which of the following terms are synonyms? a. Gastrointestinal tract b. Digestive system c. Digestive tract d. Alimentary
Chapter 14 The Digestive System and Body Metabolism Review Questions Multiple Choice: 1. Which of the following terms are synonyms? a. Gastrointestinal tract b. Digestive system c. Digestive tract d. Alimentary
DIGESTIVE TRACT ESOPHAGUS
 DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
Bio& 242 Unit 1 / Lecture 4
 Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
Soluble ADAM33 initiates airway remodeling to promote susceptibility for. Elizabeth R. Davies, Joanne F.C. Kelly, Peter H. Howarth, David I Wilson,
 Revised Suppl. Data: Soluble ADAM33 1 Soluble ADAM33 initiates airway remodeling to promote susceptibility for allergic asthma in early life Elizabeth R. Davies, Joanne F.C. Kelly, Peter H. Howarth, David
Revised Suppl. Data: Soluble ADAM33 1 Soluble ADAM33 initiates airway remodeling to promote susceptibility for allergic asthma in early life Elizabeth R. Davies, Joanne F.C. Kelly, Peter H. Howarth, David
STIMULATION OF INTESTINAL MUCOSAL PROLIFERATION BY VITAMIN D
 GASTROENTEROLOGY 64:977-982, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.5 Printed in U.S.A. STIMULATION OF INTESTINAL MUCOSAL PROLIFERATION BY VITAMIN D STANLEY J. BIRGE, M.D., AND DAVID
GASTROENTEROLOGY 64:977-982, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.5 Printed in U.S.A. STIMULATION OF INTESTINAL MUCOSAL PROLIFERATION BY VITAMIN D STANLEY J. BIRGE, M.D., AND DAVID
SHORT-TERM AND LONG-TERM EFFECT OF OXYTOCIN ON THE MORPHOLOGY AND FUNCTIONAL ACTIVITY OF FEMALE RAT SUBCUTANEOUS ADIPOSE TISSUE. P.
 PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 739 743) SHORT-TERM AND LONG-TERM EFFECT
PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 739 743) SHORT-TERM AND LONG-TERM EFFECT
BIO 139 ANATOMY AND PHYSIOLOGY II
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
R. E. Rompala and T. A. Hoagland. University of Connecticut 3 Storrs ABSTRACT
 EFFECT OF LEVEL OF ALIMENTATION ON VISCERAL ORGAN MASS AND THE MORPHOLOGY AND Na +, K + ADENOSINETRIPHOSPHATASE ACTIVITY OF INTESTINAL MUCOSA IN LAMBS 1,2 R. E. Rompala and T. A. Hoagland University of
EFFECT OF LEVEL OF ALIMENTATION ON VISCERAL ORGAN MASS AND THE MORPHOLOGY AND Na +, K + ADENOSINETRIPHOSPHATASE ACTIVITY OF INTESTINAL MUCOSA IN LAMBS 1,2 R. E. Rompala and T. A. Hoagland University of
BIO 139 ANATOMY AND PHYSIOLOGY II. THE DIGESTIVE SYSTEM LAB ANALOGY PAGES MARY CATHERINE FLATH, Ph.D.
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer.
 Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Prevalence of lactase deficiency in British adults
 Prevalence of lactase deficiency in British adults Gut, 1984, 25, 163-167 ANNE FERGUSON, DOROTHY M MACDONALD, AND W GORDON BRYDON From the Gastro-Intestinal Unit, University of Edinburgh, and Western General
Prevalence of lactase deficiency in British adults Gut, 1984, 25, 163-167 ANNE FERGUSON, DOROTHY M MACDONALD, AND W GORDON BRYDON From the Gastro-Intestinal Unit, University of Edinburgh, and Western General
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Murine Models of. Inflammatory Bowel Disease. Outline. Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center
 Murine Models of Inflammatory Bowel Disease Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center September 19, 2017 Outline Review of IBD in humans Murine models of IBD Tissue
Murine Models of Inflammatory Bowel Disease Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center September 19, 2017 Outline Review of IBD in humans Murine models of IBD Tissue
Small Bowel Obstruction after operation in a severely malnourished man. By: Ms Bounmark Phoumesy
 Small Bowel Obstruction after operation in a severely malnourished man By: Ms Bounmark Phoumesy Normal length of GI tract Normal length(achieved by age 9) Small bowel 600cm (Men: 630 cm; Women: 592 cm)
Small Bowel Obstruction after operation in a severely malnourished man By: Ms Bounmark Phoumesy Normal length of GI tract Normal length(achieved by age 9) Small bowel 600cm (Men: 630 cm; Women: 592 cm)
Enteral and parenteral nutrition in GI failure and short bowel syndrome
 Enteral and parenteral nutrition in GI failure and short bowel syndrome Alastair Forbes University College London Intestinal failure Inadequate functional intestine to allow health to be maintained by
Enteral and parenteral nutrition in GI failure and short bowel syndrome Alastair Forbes University College London Intestinal failure Inadequate functional intestine to allow health to be maintained by
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes
 SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
International Journal of Science, Environment and Technology, Vol. 6, No 3, 2017,
 International Journal of Science, Environment and Technology, Vol. 6, No 3, 2017, 2132 2136 ISSN 2278-3687 (O) 2277-663X (P) HISTOCHEMICAL STUDIES ON INTESTINAL LYMPHOID TISSUES IN KADAKNATH BREED OF POULTRY
International Journal of Science, Environment and Technology, Vol. 6, No 3, 2017, 2132 2136 ISSN 2278-3687 (O) 2277-663X (P) HISTOCHEMICAL STUDIES ON INTESTINAL LYMPHOID TISSUES IN KADAKNATH BREED OF POULTRY
without LOI phenotype by breeding female wild-type C57BL/6J and male H19 +/.
 Sakatani et al. 1 Supporting Online Material Materials and methods Mice and genotyping: H19 mutant mice with C57BL/6J background carrying a deletion in the structural H19 gene (3 kb) and 10 kb of 5 flanking
Sakatani et al. 1 Supporting Online Material Materials and methods Mice and genotyping: H19 mutant mice with C57BL/6J background carrying a deletion in the structural H19 gene (3 kb) and 10 kb of 5 flanking
Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D.
 Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D. I. Growth Hormone (somatotropin): Growth hormone (GH) is a 191 amino acid single chain polypeptide (MW 22,000 daltons). Growth
Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D. I. Growth Hormone (somatotropin): Growth hormone (GH) is a 191 amino acid single chain polypeptide (MW 22,000 daltons). Growth
Chapter 26 The Digestive System
 Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
The Presence of Normal Levels of Serum Immunoreactive Insulin-like Growth Factor 2 (IGF-2) in Patients with Down's Syndrome
 Upsala J Med Sci 89: 274278, 1984 The Presence of Normal Levels of Serum Immunoreactive Insulin-like Growth Factor 2 (IGF-2) in Patients with Down's Syndrome Goran Annerkn,' Gosta Enberg,' and Vicki R.
Upsala J Med Sci 89: 274278, 1984 The Presence of Normal Levels of Serum Immunoreactive Insulin-like Growth Factor 2 (IGF-2) in Patients with Down's Syndrome Goran Annerkn,' Gosta Enberg,' and Vicki R.
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
IN a previous publication (Hewitt, 1954) a description was given of the
 i 9 9 Further Observations on the Histochemistry of Fat Absorption in the Small Intestine of the Rat By W. HEWITT, M.B., B.S. (From the Department of Anatomy, St. Thomas' Hospital Medical School, London,
i 9 9 Further Observations on the Histochemistry of Fat Absorption in the Small Intestine of the Rat By W. HEWITT, M.B., B.S. (From the Department of Anatomy, St. Thomas' Hospital Medical School, London,
Thyroid Gland 甲状腺. Huiping Wang ( 王会平 ), PhD Department of Physiology Rm C541, Block C, Research Building, School of Medicine Tel:
 Thyroid Gland 甲状腺 Huiping Wang ( 王会平 ), PhD Department of Physiology Rm C541, Block C, Research Building, School of Medicine Tel: 88208292 Outline Thyroid Hormones Types Biosynthesis Storage and Release
Thyroid Gland 甲状腺 Huiping Wang ( 王会平 ), PhD Department of Physiology Rm C541, Block C, Research Building, School of Medicine Tel: 88208292 Outline Thyroid Hormones Types Biosynthesis Storage and Release
Effects of methionine-containing dipeptides on α s1 casein expression in bovine mammary epithelial cells *
 Journal of Animal and Feed Sciences, 16, Suppl. 2, 2007, 325 329 Effects of methionine-containing dipeptides on α s1 casein expression in bovine mammary epithelial cells * H.H. Wu 1, J.Y. Yang 1,2, K.
Journal of Animal and Feed Sciences, 16, Suppl. 2, 2007, 325 329 Effects of methionine-containing dipeptides on α s1 casein expression in bovine mammary epithelial cells * H.H. Wu 1, J.Y. Yang 1,2, K.
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Erythropoietin In Vitro
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and
 Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and private study only. The thesis may not be reproduced elsewhere
Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and private study only. The thesis may not be reproduced elsewhere
