Mutagenesis of Asp-569 of Glucosyltransferase I Glucansucrase Modulates Glucan and Oligosaccharide Synthesis
|
|
|
- Collin Thornton
- 5 years ago
- Views:
Transcription
1 APPLIED AND ENVIRONMENTAL MICROBIOLOGY, May 2000, p Vol. 66, No /00/$ Copyright 2000, American Society for Microbiology. All Rights Reserved. Mutagenesis of Asp-569 of Glucosyltransferase I Glucansucrase Modulates Glucan and Oligosaccharide Synthesis VINCENT MONCHOIS, 1 MICHEL VIGNON, 2 AND ROY R. B. RUSSELL 1 * Department of Oral Biology, The Dental School, University of Newcastle upon Tyne, Newcastle upon Tyne, United Kingdom, 1 and Centre de Recherche sur les Macromolécules Végétales, CERMAV-CNRS, affiliated with the Joseph Fourier University of Grenoble, Grenoble Cedex 9, France 2 Received 19 November 1999/Accepted 2 February 2000 Glucansucrases of oral streptococci and Leuconostoc mesenteroides are enzymes of medical and biotechnological interest that synthesize -glucans. They can also synthesize oligosaccharides in the presence of a sugar acceptor. Previous reports have identified an amino acid residue that may affect the structure of the glucan product; therefore, random mutagenesis of the corresponding Asp-569 of Streptococcus downei glucosyltransferase I (GTF-I) was used to further understanding of its involvement in the catalytic mechanism and to evaluate how different amino acids can modulate glucan and oligosaccharide synthesis. GTF-I variants were obtained where Asp-569 was replaced by each of the different possible classes of amino acids. These were expressed in Escherichia coli and purified by means of a His 6 tag. The results showed that the amino acid in position 569 influences the structure of the glucan and the size of the oligosaccharides produced by GTF-I. The results suggest that the amino acid occupying this position is more likely to interact with the acceptor molecules (oligosaccharides or elongating glucan chain) than to be directly involved in glucosyl transfer from sucrose. Engineering of the equivalent position in glucansucrases thus appears to be a good target to expand the range of oligosaccharides synthesized. * Corresponding author. Mailing address: Department of Oral Biology, Dental School, University of Newcastle upon Tyne, Newcastle upon Tyne NE2 4BW, United Kingdom. Phone: Fax: r.r.russell@newcastle.ac.uk. The glucansucrases (EC ; commonly called glucosyltransferases [GTFs]) from oral streptococci are enzymes belonging to glycosidase family 70 that catalyze the transfer of glucosyl units from the cleavage of sucrose to a growing -glucan chain (6). Depending on the enzyme, different sizes and structures of glucan can be produced, and the nature of the linkages between glucosyl units determines the water solubility and properties of the glucan. A higher content of (1-3) linkages is associated with greater insolubility (13, 20). Glucans are of central importance in adhesive interactions in plaque, where they mediate attachment of bacteria to the tooth surface and to other bacteria, thus stabilizing the plaque biofilm, serve as energy stores aiding the survival of plaque bacteria, and modulate the permeability of plaque and hence the level of acid at the enamel surface (1). In addition to synthesis of glucan (with the release of fructose), GTF can hydrolyze sucrose to glucose and fructose and also transfer glucose to fructose, in which case leucrose [5-O-( -D-glucopyranosyl)-D-fructopyranose] is formed. If an acceptor molecule such as maltose is present, it is extended to form a series of glucooligosaccharides (GOS). Oligosaccharides are of potential industrial interest for incorporation in foodstuffs or as prebiotics (14), and we have previously reported that site-directed mutagenesis of GTF can alter the relative balance of the three reaction pathways of synthesis of glucan, hydrolysis, or synthesis of oligosaccharides (12). Further information on the influence on reaction specificity of various amino acid residues in GTF is needed for protein engineering to achieve desired properties as well as providing insights into enzymatic mechanisms and aiding rational design of inhibitors that could have application in dental care products (5, 16). All glucansucrases possess a common pattern of organization (1, 13). They are of high molecular mass (around 160 kda) and have a signal sequence followed by a variable stretch of about 200 amino acids, a highly conserved core region of about 900 amino acids including the catalytic domain, and a C-terminal glucan-binding domain covering about 400 amino acids. Sequence alignment and secondary structure prediction showed that the GTF catalytic domain can be related to the -amylase superfamily (glycosidase family 13), members of which contain a catalytic ( / ) 8 barrel domain (9). GTFs are predicted to contain alternating sheets and helices, though the homologous elements appear to be circularly permuted with respect to those in amylases (9) and amino acids important in catalysis lie outside the main barrel region (12). Amino acids contributing to the active site have been recognized, and invariant Asp and Glu residues, homologous at those present at the C termini of the 4, 5, and 7 strands of -amylases and involved in the catalytic triad, have been found to be essential for GTFs (3, 8, 19). In -amylases, some - loops may play an important role in substrate specificity (10), and a critical Asp residue in a region corresponding to the 7 strand of -amylase has been shown to clearly influence the structure of the glucan produced by Streptococcus mutans GTF-B (18). Shimamura et al. compared sequences of GTF-I, GTF-S, GTF-B, GTF-C, and GTF-D and identified positions where amino acid residues are conserved for the GTFs producing water-insoluble (1-3)-linked glucan but differ from the residues present in GTFs producing a soluble (1-6)-linked glucan (18). In addition, site-directed mutagenesis experiments confirmed that in GTF-B and GTF-D, conversion of an Asp to a Thr residue influenced the structure of the glucan produced (18). In view of the evidence for the importance of an Asp or Thr residue in this position, further analysis of this site should throw light on this phenomenon; we describe mutagenesis of a GTF from Streptococcus downei, one of the mutans group of oral streptococci (4, 17, 21) that produces a water-insoluble glucan containing (1-3) glucosyl linkages. We have reported 1923
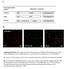
2 1924 MONCHOIS ET AL. APPL. ENVIRON. MICROBIOL. the genetic manipulation of the gtfi gene to facilitate purification of the catalytic core (GTF-Ic) and shown that it retains the properties of the intact enzyme (11). Random mutagenesis of Asp-569 was used to better understand the involvement of this position in the catalytic mechanism and to evaluate how different amino acids can modulate glucan and oligosaccharide synthesis. MATERIALS AND METHODS Mutations at position 569 of GTF-I. Plasmid pgtfic carrying the gene coding for the 905-amino-acid conserved core region of GTF-I (GTF-Ic) fused with a stretch of six histidine residues (11) was used as the template for mutagenesis. This corresponds to amino acids 148 to 1053 of GTF-I from S. downei MFe28 (4, 21). Random and biased random mutagenesis were performed using two sets of primers (set 1, 5 -GATAGCGAAGTACAANNNCTGATTCGTGACATC and 5 -GATGTCACGAATCAGNNNTTGTACTTCGCTATC; set 2, 5 -GATAGC GAAGTACAAWNSCTGATTCGTGACATC and 5 -GATGTCACGAATCA GSNWTTGTACTTCGCTATC) with a Quick-Change site-directed mutagenesis kit (Stratagene). After transformation into Escherichia coli XL1-Blue and selection on Luria-Bertani medium plates containing ampicillin, a two-step screening was undertaken. Recombinant colonies were first patched on 2xYT (16 g of tryptone, 10 g of yeast extract, and 5gofNaCl liter 1 ) containing ampicillin (2xYT Amp) supplemented with 2% sucrose (wt/vol) and 5 mm isopropyl- - D-thiogalactopyranoside (IPTG) at 37 C. Under these conditions, appearance of a white halo around colonies could be observed, the size of which corresponded with GTF activity. For the second screening step, mutants selected on the basis of halo size were propagated overnight at 37 C in 10 ml of 2xYT Amp with 2% glucose (wt/vol) and 5 mm IPTG. After protein extraction by sonication, activity was checked by the dinitrosalicylic acid method (11), and the level of expression was monitored by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and staining with Coomassie blue. The selected mutants were then sequenced with an Applied Biosystems 377 DNA sequencer (Molecular Biology Facility, University of Newcastle). Expression and purification of GTF-Ic mutants. Selected mutant or wild-type GTF-Ic enzymes were expressed by culturing E. coli XL1-Blue carrying the relevant plasmids overnight at 37 C in 15 ml of 2xYT Amp supplemented with 2% (wt/vol) glucose and 5 mm IPTG. Cells were harvested and resuspended in 0.5 ml of 50 mm sodium phosphate buffer (ph 8.0) 300 mm NaCl and treated with lysozyme (0.1 mg ml 1 ) before sonication. Purification of GTF-Ic variants was achieved with an Ni-nitrilotriacetic acid spin column kit (Qiagen) as recommended by the manufacturer. Proteins were eluted in 50 mm sodium phosphate buffer (ph 8.0) containing 300 mm NaCl and 250 mm imidazole and dialyzed overnight at 4 C against 50 mm Tris-HCl buffer (ph 7.0). Protein purity was assayed by SDS-PAGE, and protein concentration was determined by UV absorption at 280 nm using calculated extinction coefficients. Glucan synthesis activity. Specific activity at 37 C was assayed by measuring fructose release over a 30-min period in the presence of 50 g of sucrose liter 1 in 50 mm Tris-HCl buffer (ph 7.0) by the dinitrosalicylic acid method (11). One unit is defined as the amount of enzyme that catalyzed the formation of 1 mol of fructose min 1. After complete depletion of sucrose, concentrations of fructose, glucose, and leucrose released in the reaction medium were assayed by enzymatic methods and high-performance liquid chromatography (HPLC) as previously described (11). Glucan synthesis yield was calculated by subtracting the free glucose concentration from the fructose concentration, since this corresponds to the glucose residues coming from sucrose cleavage and incorporated into glucan. The structure of glucans produced by mutant T569D as well as GTF-Ic were analyzed by 13 C nuclear magnetic resonance (NMR) spectroscopy as previously described (12). The peak assignments were made according to Colson et al. (2). Oligosaccharide synthesis reaction. Oligosaccharide synthesis was undertaken in the presence of sucrose and maltose as acceptors, using a sucrose/maltose molar ratio of 1:5. The sucrose concentration was 50 g liter 1, and the reaction was allowed to continue until all of the sucrose was depleted. Synthesized oligosaccharides were analyzed by HPLC using a C 18 column and water as eluant at a flow rate of 0.7 ml min 1. They were detected using a differential refractometer. FIG. 1. Aligned amino acid sequences of 7 regions of GTFs and related L. mesenteroides dextransucrases. GTF-B, -C, and -D are from S. mutans; GTF-G is from S. gordonii; GTF-S and -I are from S. downei; GTF-J through -M are from S. salivarius. DSR-A and -S are produced by L. mesenteroides (12). Amino acids aligned with Asp-569 from GTF-I are boxed. *, identical residues; :, conserved substitution;, semiconserved substitution; s, invariant catalytic residue;, essential His stabilizing transition state in -amylases (3, 8, 17). structures of the glucans made by all GTFs are not yet known. To investigate how the nature of the amino acid at this position modulates both glucan and oligosaccharide synthesis, replacement of Asp-569 from GTF-Ic with different residues was performed by random and biased random mutagenesis. A double-screening procedure after random mutagenesis allowed isolation, from about 100 colonies, of six different GTF-Ic variants where Asp-569 was replaced by Arg, Val, Leu, His, Glu, or Ala. To cover all different possible classes of amino acids, a similar double-screening procedure following biased random mutagenesis of Asp-569 allowed isolation of further GTF-Ic variants where Asp-569 was replaced by Tyr, Ile, Thr, Ser, or Gln. Wild-type GTF-Ic and the 11 variants were expressed in E. coli XL1-Blue and purified by means of the His 6 tag at their N-terminal end, using an Ni-nitrilotriacetic acid spin column kit as shown in Fig. 2. No major influence on the level of enzyme production in E. coli by the mutations could be detected. Effect of mutations on activity. Activity assays carried out with purified GTF-Ic and variants showed that all mutations affected the specific activity (Table 1). In the case of D569E and D569T, there was only slight loss of activity, but changing Asp to residues such as Asn, His, Val, Ser, Ile, and Ala resulted in a specific activity less than half that of the wild-type enzyme RESULTS Mutagenesis of GTF-I Asp-569. Among streptococcal GTFs and related dextransucrases from Leuconostoc mesenteroides, an Asp residue is always present in sequences of enzymes synthesizing a water-insoluble glucan (Fig. 1). Except for GTF-L and GTF-M, where Ser and Ala residues, respectively, are present, a Thr residue is associated with enzymes synthesizing water-soluble glucan (Fig. 1). Insolubility is thought to be associated with a high content of (1-3) linkages, though the FIG. 2. SDS-PAGE analysis followed by Coomassie blue staining of wildtype GTF-Ic and mutant enzymes purified by affinity chromatography. Single letters indicate amino acids substituted for D in GTF-Ic.
3 VOL. 66, 2000 MUTAGENESIS OF GLUCOSYLTRANSFERASE 1925 TABLE 1. Effects of mutation of Asp-569 on glucan and oligosaccharide synthesis Amino acid Sp act (U mg 1 ) a Distribution of products from sucrose (%) Glucan synthesis b Leucrose synthesis b Hydrolysis b Yield of oligosaccharides c D (wild type) E T N H V S I A R Y L Avg d a Initial velocity determined in the presence of 50 g of sucrose liter 1. One unit is defined as the amount of enzyme that catalyzed the formation of 1 mol of fructose min 1. b One liter of the glucan synthesis reaction mixture contained 50 g of sucrose. c One liter of the oligosaccharide synthesis reaction mixture contained 50 g of sucrose and 10 g of maltose (molecular ratio of 5:1). d All experiments were performed in duplicate. Duplicate results never differed by more than 5%. (mean standard deviation, Umg 1 [Table 1]). Change to Tyr, Arg, or Leu had the greatest effect on enzyme activity. Effects of mutations on glucan synthesis. To investigate the influence of the mutations on the distribution of glucosyl residues deriving from sucrose cleavage during the glucan synthesis reaction, we analyzed the reaction products (Table 1). The rate of sucrose hydrolysis was not significantly modified by any of the mutations, hydrolysis accounting for 16.7% of the sucrose utilization by wild-type GTF-Ic and an average value of 15.7% for the different variants. However, the ratio between the transfer of glucosyl residues to growing glucan chain and to free fructose residues (resulting in leucrose synthesis) was modified by the mutations (Table 1). With GTF-Ic, yields of glucan and leucrose synthesis were 60.8 and 23.9%, respectively. The change of Asp-569 to any other amino acid diverted the transfer of glucosyl residues from fructose toward glucan synthesis. The yield of leucrose synthesis thus decreased to an average value of 11.6% and the yield of glucan synthesis increased to an average value of 73.5%. Effect of mutation on glucan structure. To explore the influence of position 569 on the structure of the polymer produced by GTF-Ic, glucan synthesized by mutant T569D was analyzed. Contrary to GTF-Ic, which produces an insoluble (1-3)-linked glucan (11, 17), mutant T569D synthesized both insoluble and soluble polymer. The 13 C NMR spectrum of the insoluble glucan presented only signals characteristic of glucosyl residues linked through (1-3) linkages. In addition to signals corresponding to carbon 5 (C-5), C-4, and C-2 in the 68- to 76-ppm area, signals arising at 100.5, 84, and 61 ppm corresponded to C-1 involved in (1-3) glucosyl linkage, C-3 involved in (1-3) glucosyl linkage, and nonlinked C-6, respectively (Fig. 3A). The 13 C NMR spectrum of the soluble glucan presented only signals characteristic of glucosyl residues linked through (1-6) linkages (Fig. 3B). In addition to signals corresponding to C-5, C-4, C-3, and C-2 in the 68- to 76-ppm area, signals arising at 98.5, 67, and 61 ppm corresponded to C-1 involved in (1-6) linkage, C-6 involved in (1-6) linkage, and nonlinked C-6, respectively. Effects of mutations on oligosaccharide synthesis in the presence of maltose. In the presence of maltose as an acceptor, oligosaccharides produced by GTF-I were mainly linear GOS of various degrees of polymerization (DP), according to their retention time during HPLC separation on C 18 in comparison with known standard oligosaccharides produced by L. mesenteroides NRRL B-512F dextransucrase (13, 15). GOS of DP n were oligodextrans composed of (n 2) glucosyl residues linked through (1-6) bonds and a maltose residue at the reducing end. With a sucrose/maltose molecular ratio of 5:1, GOS ranging from DP 3 (panose) to DP 7 were produced (Fig. 4). When GOS synthesis was performed with the different GTF-Ic variants, no additional peaks corresponding to the synthesis of new oligosaccharides were observed during HPLC analysis. The total yield of GOS produced by the variants was very close to that obtained with wild-type enzyme (Table 1). Most of the mutations resulted in a change of less than 5% FIG C NMR analyses of the insoluble (A) and the soluble (B) glucan produced by mutant D569T. Peaks were assigned according to the method of Colson et al. (2).
4 1926 MONCHOIS ET AL. APPL. ENVIRON. MICROBIOL. FIG. 4. Effects of mutations on the sizes of oligosaccharides produced in the presence of sucrose and maltose as acceptors. One liter of the oligosaccharide synthesis reaction mixture contained 50 g of sucrose and 10 g of maltose. For each reaction, the total amount of linear GOS was defined as 100%. from the yield obtained with GTF-Ic, and only the D569I variant decreased this yield by 10%. On the contrary, the distribution between the different lengths of GOS and especially the yield of oligodextran of DP 7 (OD 7 ) was strongly influenced by mutations (Fig. 4), these differences being dependent on the nature of the amino acid change. The replacement of Asp-569 by hydroxyl amino acids (Thr and Ser) or by an aliphatic amino acid with a short lateral chain (Ala) increased the yield of OD 7 synthesis 3.5- to 5-fold (Fig. 4). However, an increase in the size of the lateral chain of aliphatic amino acids resulted in a reduction of the OD 7 synthesis yield, from 26% of overall GOS with D569A, 18% with D569V, 4% with D569L, and only 1.5% with D569I. The D569Y change virtually abolished the synthesis of OD 7. Changes to basic residues such as His and Arg had the same small effect as the change to Asn (Fig. 4). DISCUSSION Site-directed mutagenesis of the position corresponding to that studied here in GTF-B and GTF-D of S. mutans has previously been shown to influence the nature of the glucan produced (18). Sequence alignment of GTFs and related L. mesenteroides dextransucrases seemed to indicate that the presence of an Asp residue resulted in the synthesis of insoluble glucan whereas the presence of a Thr residue led to the synthesis of soluble glucan. This suggested that other changes to different amino acids might enlarge GTF specificity. The aim of this study was therefore to find out how this position modulates the activities of glucan and oligosaccharide synthesis by replacing the equivalent Asp-569 of the core region of S. downei GTF-I with different amino acids by random and biased random mutagenesis. As for GTF-B or GTF-D, this position influences the structure of the glucan produced by GTF-I. In addition to the insoluble (1-3)-linked glucan produced by GTF-Ic (11), the variant carrying the D569T mutation also produces a soluble (1-6)-linked glucan, showing that the mutation has affected the orientation of the growing glucan chain mechanism or the transfer of glucosyl residues coming from sucrose cleavage. It was therefore of interest to investigate whether mutation also affected transfer to other acceptors. However, in the presence of maltose as an acceptor, no GOS additional to those produced by GTF-Ic were synthesized by the different variants. The various replacements of Asp-569 of GTF-Ic resulted in a decrease in activity, the magnitude depending on the nature of the substituent residue. Because Shimamura et al. (18) showed that mutating this position did not significantly modify the sucrose binding of GTF, substitutions of Asp-569 are likely to affect the turnover of the reaction. Local structural disturbances that mutations might cause may explain some small variations, but general features can also be noticed. The change of Asp to Thr does not result in a significant loss of activity, 72% of the initial activity being retained. However, the location of the hydroxyl group itself appears to be essential, as the change to Ser resulted in a twofold decrease of activity. The lateral location of the methyl group in Leu is also critical for activity, as D569L reduced the activity 2.6-fold in comparison to D569I, D569V, and D569A variants, which were all similar. In the presence of sucrose, the different mutations had no significant effect on the rate of sucrose hydrolysis. In the presence of sucrose and maltose, the total yield of GOS is also largely unchanged. Position 569 is therefore unlikely to be involved directly in glucosyl transfer from sucrose to acceptors. However, the destination of the glucosyl was affected by the
5 VOL. 66, 2000 MUTAGENESIS OF GLUCOSYLTRANSFERASE 1927 mutations. All mutations of Asp-569 had the same effect in increasing glucan yield at the expense of the leucrose yield. As all mutations of Asp-569 have the same effect, this suggests that only interactions promoted by Asp-569 allow fructose molecules to be bound in a way that results in a high yield of leucrose. Distribution between the different lengths of GOS was also clearly affected by the mutations, indicating the influence of Asp-569 on binding of growing oligosaccharides or glucan. In addition, distribution between GOS was dependent on the nature of the residue replacing Asp-569, suggesting that certain amino acids are able to promote specific interactions with the products. The change to a hydroxyl amino acid increased the yield of longer GOS; the difference between the D569T and D569S mutations may illustrate the importance of its orientation. The size of the aliphatic chain seems also to be important, as D569A and D569V mutations increased the yield of longer GOS while D569L and D569I mutations increased the yield of shorter GOS. Random mutagenesis of Asp-569 from GTF-I has thus shown that the amino acid in this position is more likely to interact with the acceptor molecules (oligosaccharides or elongating glucan) than to be directly involved into the glucosyl transfer from sucrose. Secondary structure predictions locate Asp-569 in a region equivalent to the seventh - loop in -amylase (9), just after the conserved catalytic Asp-567 essential for GTF activity (3). In -amylases, besides loop 7, residues in the fourth and fifth loops are involved in enzyme specificity (7, 10). We have previously shown by mutation of His-355 of GTF-I that it may play a role in a subsite involved in product binding (12). Other residues may also be directly involved in determining the structure of products (18), and this may explain why a single change at Asp-569 is insufficient to drastically modify enzyme specificity. Nevertheless, this study shows that while engineering of this position does not influence the structure of oligosaccharides, it may be an important target in tailoring the size of the oligosaccharides produced. Investigation of a wider range of reaction conditions may make it possible to enhance this effect. Furthermore, modification of the corresponding residue in GTFs that make predominantly other types of linkage [ (1-6) and (1-2)] has the potential to expand the range of GOS that can conveniently be synthesized. ACKNOWLEDGMENTS This work was supported by Wellcome Trust grant and the European project BIOTECH CT REFERENCES 1. Colby, S. M., and R. R. B. Russell Sugar metabolism by mutans streptococci. Soc. J. Appl. Microbiol. Symp. Suppl. 83:80S 88S. 2. Colson, P., H. J. Jennings, and I. C. Smith Composition, sequence, and conformation of polymers and oligomers of glucose as revealed by carbon-13 nuclear magnetic resonance. J. Am. Chem. Soc. 96: Devulapalle, K. S., S. D. Goodman, Q. Gao, A. Hemsley, and G. Mooser Knowledge-based model of a glucosyltransferase from the oral bacterial group of mutans streptococci. Protein Sci. 6: Ferretti, J. J., M. L. Gilpin, and R. R. B. Russell Nucleotide sequence of a glucosyltransferase gene from Streptococcus sobrinus MFe28. J. Bacteriol. 169: Hajishengallis, G., and S. M. Michalek Current status of a mucosal vaccine against dental caries. Oral Microbiol. Immunol. 14: Henrissat, B Glycosidase families. Biochem. Soc. Trans. 26: Jespersen, H. M., E. A. MacGregor, B. Henrissat, M. R. Sierks, and B. Svensson Starch- and glycogen-debranching and branching enzymes: prediction of structural features of the catalytic ( / ) 8 -barrel domain and evolutionary relationship to other amylolytic enzymes. J. Protein Chem. 12: Kato, C., Y. Nakano, M. Lis, and H. K. Kuramitsu Molecular genetic analysis of the catalytic site of Streptococcus mutans glucosyltransferases. Biochem. Biophys. Res. Commun. 189: MacGregor, E. A., H. M. Jespersen, and B. Svensson A circularly permuted -amylase-type ( / ) 8 -barrel structure in glucan-synthesizing glucosyltransferases. FEBS Lett. 378: Matsui, I., and B. Svensson Improved activity and modulated action pattern obtained by random mutagenesis at the fourth beta-alpha loop involved in substrate binding to the catalytic ( / ) 8 -barrel domain of barley -amylase 1. J. Biol. Chem. 272: Monchois, V., M. Arguello-Morales, and R. R. B. Russell Isolation of an active catalytic core of Streptococcus downei MFe28 GTF-I glucosyltransferase. J. Bacteriol. 181: Monchois, V., M. Vignon, and R. R. B. Russell Isolation of key amino acid residues at the N-terminal end of the core region Streptococcus downei glucansucrase, GTF-I. Appl. Microbiol. Biotechnol. 52: Monchois, V., R. M. Willemot, and P. Monsan Glucansucrases: mechanism of action and structure-function relationships. FEMS Microbiol. Rev. 23: Monsan, P., and F. Paul Enzymatic synthesis of oligosaccharides. FEMS Microbiol. Rev. 16: Robyt, J. F., and S. H. Eklund Relative, quantitative effects of acceptors in the reaction of Leuconostoc mesenteroides B-512F dextransucrase. Carbohydr. Res. 121: Russell, R. R. B Control of specific plaque bacteria. Adv. Dent. Res. 8: Russell, R. R. B., M. L. Gilpin, H. Mukasa, and G. Dougan Characterization of glucosyltransferase expressed from a Streptococcus sobrinus gene cloned in Escherichia coli. J. Gen. Microbiol. 133: Shimamura, A., Y. J. Nakano, H. Mukasa, and H. K. Kuramitsu Identification of amino acid residues in Streptococcus mutans glucosyltransferases influencing the structure of the glucan product. J. Bacteriol. 176: Tsumori, H., T. Minami, and H. K. Kuramitsu Identification of essential amino acids in the Streptococcus mutans glucosyltransferases. J. Bacteriol. 179: Walker, G. J Dextrans. Int. Rev. Biochem. 16: Whiley, R. A., R. R. B. Russell, J. M. Hardie, and D. Beighton Streptococcus downei sp. nov. of strains previously described as Streptococcus mutans serotype h. Int. J. Syst. Bacteriol. 38:25 29.
Physiological studies of Leuconostoc mesenteroides strain NRRL B-1149 during cultivation on glucose and fructose media
 Veselin Bivolarski 1 Tonka Vasileva 1 Rishikesh Shukla 2 Arun Goyal 2 Ilia Iliev 1 Authors addresses: 1 Department of Biochemistry and Microbiology, Faculty of Biology, Plovdiv University, Plovdiv, Bulgaria.
Veselin Bivolarski 1 Tonka Vasileva 1 Rishikesh Shukla 2 Arun Goyal 2 Ilia Iliev 1 Authors addresses: 1 Department of Biochemistry and Microbiology, Faculty of Biology, Plovdiv University, Plovdiv, Bulgaria.
VIRULENCE SCHEME OVERVIEW
 VIRULENCE SCHEME OVERVIEW Sucrose Stronger ADHESION; Greater Accumulation Bacterial factors that promote biofilm development ADHESION ACIDOGENICITY Fermentable Carbohydrates Selection for S. mutans and
VIRULENCE SCHEME OVERVIEW Sucrose Stronger ADHESION; Greater Accumulation Bacterial factors that promote biofilm development ADHESION ACIDOGENICITY Fermentable Carbohydrates Selection for S. mutans and
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL
 Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Mutanase of Paenibacillus humicus from Fermented Food Has a Potential for Hydrolysis of Biofilms Synthesized by Streptococcus mutans
 420 Journal of Health Science, 57(5) 420 424 (2011) Research Letter Mutanase of Paenibacillus humicus from Fermented Food Has a Potential for Hydrolysis of Biofilms Synthesized by Streptococcus mutans
420 Journal of Health Science, 57(5) 420 424 (2011) Research Letter Mutanase of Paenibacillus humicus from Fermented Food Has a Potential for Hydrolysis of Biofilms Synthesized by Streptococcus mutans
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Manja Henze, Dorothee Merker and Lothar Elling. 1. Characteristics of the Recombinant β-glycosidase from Pyrococcus
 S1 of S17 Supplementary Materials: Microwave-Assisted Synthesis of Glycoconjugates by Transgalactosylation with Recombinant Thermostable β-glycosidase from Pyrococcus Manja Henze, Dorothee Merker and Lothar
S1 of S17 Supplementary Materials: Microwave-Assisted Synthesis of Glycoconjugates by Transgalactosylation with Recombinant Thermostable β-glycosidase from Pyrococcus Manja Henze, Dorothee Merker and Lothar
ON THE DIFFERENCE IN ADSORPTION ON SEPHADEX GEL OF THE DEXTRANSUCRASE OF STREPTOCOCCUS BOVIS GROWN ON SUCROSE AND GLUCOSE MEDIA
 J. Gen. App!. Microbiol., 34, 213-219 (1988) ON THE DIFFERENCE IN ADSORPTION ON SEPHADEX GEL OF THE DEXTRANSUCRASE OF STREPTOCOCCUS BOVIS GROWN ON SUCROSE AND GLUCOSE MEDIA TOSHIRO HAYASHI, RYO IOROI,*
J. Gen. App!. Microbiol., 34, 213-219 (1988) ON THE DIFFERENCE IN ADSORPTION ON SEPHADEX GEL OF THE DEXTRANSUCRASE OF STREPTOCOCCUS BOVIS GROWN ON SUCROSE AND GLUCOSE MEDIA TOSHIRO HAYASHI, RYO IOROI,*
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular
 Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
Macromolecules Structure and Function
 Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
PAPER No. : 16, Bioorganic and biophysical chemistry MODULE No. : 22, Mechanism of enzyme catalyst reaction (I) Chymotrypsin
 Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Applied Microbiology and Biotechnology. Supplementary Material
 Applied Microbiology and Biotechnology Supplementary Material Biochemical characterization of a GH70 protein from Lactobacillus kunkeei DSM 12361 with two catalytic domains involving branching sucrase
Applied Microbiology and Biotechnology Supplementary Material Biochemical characterization of a GH70 protein from Lactobacillus kunkeei DSM 12361 with two catalytic domains involving branching sucrase
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Previous Class. Today. Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism. Protein Kinase Catalytic Properties
 Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Topic 4 - #2 Carbohydrates Topic 2
 Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
A BEGINNER S GUIDE TO BIOCHEMISTRY
 A BEGINNER S GUIDE TO BIOCHEMISTRY Life is basically a chemical process Organic substances: contain carbon atoms bonded to other carbon atom 4 classes: carbohydrates, lipids, proteins, nucleic acids Chemical
A BEGINNER S GUIDE TO BIOCHEMISTRY Life is basically a chemical process Organic substances: contain carbon atoms bonded to other carbon atom 4 classes: carbohydrates, lipids, proteins, nucleic acids Chemical
Supporting Information
 Supporting Information Dauvillée et al. 10.1073/pnas.0907424106 Fig. S1. Iodine screening of the C. cohnii mutant bank. Each single colony was grown on rich-medium agar plates then vaporized with iodine.
Supporting Information Dauvillée et al. 10.1073/pnas.0907424106 Fig. S1. Iodine screening of the C. cohnii mutant bank. Each single colony was grown on rich-medium agar plates then vaporized with iodine.
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB
 Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Ch. 5 The S & F of Macromolecules. They may be extremely small but they are still macro.
 Ch. 5 The S & F of Macromolecules They may be extremely small but they are still macro. Background Information Cells join small molecules together to form larger molecules. Macromolecules may be composed
Ch. 5 The S & F of Macromolecules They may be extremely small but they are still macro. Background Information Cells join small molecules together to form larger molecules. Macromolecules may be composed
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Short polymer. Dehydration removes a water molecule, forming a new bond. Longer polymer (a) Dehydration reaction in the synthesis of a polymer
 HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
University of Groningen. Glucansucrases of lactobacilli Kralj, Slavko
 University of Groningen Glucansucrases of lactobacilli Kralj, Slavko IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document
University of Groningen Glucansucrases of lactobacilli Kralj, Slavko IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document
Scholars Research Library. Purification and characterization of neutral protease enzyme from Bacillus Subtilis
 Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2012, 2 (4):612-618 (http://scholarsresearchlibrary.com/archive.html) Purification and characterization
Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2012, 2 (4):612-618 (http://scholarsresearchlibrary.com/archive.html) Purification and characterization
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
A. Lipids: Water-Insoluble Molecules
 Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Mutational and biochemical analysis of Lactobacillus reuteri glucansucrase enzymes Meng, Xiangfeng
 University of Groningen Mutational and biochemical analysis of Lactobacillus reuteri glucansucrase enzymes Meng, Xiangfeng IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Mutational and biochemical analysis of Lactobacillus reuteri glucansucrase enzymes Meng, Xiangfeng IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
NOTE: For studying for the final, you only have to worry about those with an asterix (*)
 NOTE: For studying for the final, you only have to worry about those with an asterix (*) (*)1. An organic compound is one that: a. contains carbon b. is slightly acidic c. forms long chains d. is soluble
NOTE: For studying for the final, you only have to worry about those with an asterix (*) (*)1. An organic compound is one that: a. contains carbon b. is slightly acidic c. forms long chains d. is soluble
Macromolecules. The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1.
 Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
Proteins. Amino acids, structure and function. The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka
 Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Supplementary Information
 Supplementary Information Structural basis of improved second generation 3-nitro-tyrosine trna synthetases Richard B. Cooley, Jessica L. Feldman, Camden M. Driggers, Taylor Bundy, Audrey L. Stokes, P.
Supplementary Information Structural basis of improved second generation 3-nitro-tyrosine trna synthetases Richard B. Cooley, Jessica L. Feldman, Camden M. Driggers, Taylor Bundy, Audrey L. Stokes, P.
Abdullah zurayqat. Bahaa Najjar. Mamoun Ahram
 9 Abdullah zurayqat Bahaa Najjar Mamoun Ahram Polysaccharides Polysaccharides Definition and Structure [Greek poly = many; sacchar = sugar] are complex carbohydrates, composed of 10 to up to several thousand
9 Abdullah zurayqat Bahaa Najjar Mamoun Ahram Polysaccharides Polysaccharides Definition and Structure [Greek poly = many; sacchar = sugar] are complex carbohydrates, composed of 10 to up to several thousand
189,311, , ,561, ,639, ,679, Ch13; , Carbohydrates
 Lecture 31 (12/8/17) Reading: Ch7; 258-267 Ch10; 371-373 Problems: Ch7 (text); 26,27,28 Ch7 (study-guide: applying); 2,5 Ch7 (study-guide: facts); 6 NEXT (LAST!) Reading: Chs4,6,8,10,14,16,17,18; 128-129,
Lecture 31 (12/8/17) Reading: Ch7; 258-267 Ch10; 371-373 Problems: Ch7 (text); 26,27,28 Ch7 (study-guide: applying); 2,5 Ch7 (study-guide: facts); 6 NEXT (LAST!) Reading: Chs4,6,8,10,14,16,17,18; 128-129,
What are the molecules of life?
 Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
University of Groningen. Exopolysaccharide synthesis by Lactobacilus reuteri van Geel-Schutten, Gerritdina Hendrika
 University of Groningen Exopolysaccharide synthesis by Lactobacilus reuteri van Geel-Schutten, Gerritdina Hendrika IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if
University of Groningen Exopolysaccharide synthesis by Lactobacilus reuteri van Geel-Schutten, Gerritdina Hendrika IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if
Supplementary Material
 Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
Kinetics analysis of β-fructofuranosidase enzyme. 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction
 Kinetics analysis of β-fructofuranosidase enzyme 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction Enzyme kinetics It is the study of the chemical reactions that are catalyzed by enzymes.
Kinetics analysis of β-fructofuranosidase enzyme 1-Effect of Time Incubation On The Rate Of An Enzymatic Reaction Enzyme kinetics It is the study of the chemical reactions that are catalyzed by enzymes.
Organic molecules are molecules that contain carbon and hydrogen.
 Organic Chemistry, Biochemistry Introduction Organic molecules are molecules that contain carbon and hydrogen. All living things contain these organic molecules: carbohydrates, lipids, proteins, and nucleic
Organic Chemistry, Biochemistry Introduction Organic molecules are molecules that contain carbon and hydrogen. All living things contain these organic molecules: carbohydrates, lipids, proteins, and nucleic
Carbohydrates. Organic compounds which comprise of only C, H and O. C x (H 2 O) y
 Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
BabyBio IMAC columns DATA SHEET DS
 BabyBio IMAC columns DATA SHEET DS 45 655 010 BabyBio columns for Immobilized Metal Ion Affinity Chromatography (IMAC) are ready-to-use for quick and easy purification of polyhistidine-tagged (His-tagged)
BabyBio IMAC columns DATA SHEET DS 45 655 010 BabyBio columns for Immobilized Metal Ion Affinity Chromatography (IMAC) are ready-to-use for quick and easy purification of polyhistidine-tagged (His-tagged)
Why Carbon? What does a carbon atom look like?
 Biomolecules Organic Chemistry In the 1800 s it was believed to be impossible to recreate molecules in a lab Thus, the study of organic chemistry was originally the study of molecules in living organisms
Biomolecules Organic Chemistry In the 1800 s it was believed to be impossible to recreate molecules in a lab Thus, the study of organic chemistry was originally the study of molecules in living organisms
Expression constructs
 Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Chemical Mechanism of Enzymes
 Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Enzyme Catalysis-Serine Proteases
 Enzyme Catalysis-Serine Proteases Concepts to be learned Activation Energy Transition State Example: Proteases Requirements for proteolysis Families of proteases Protein Folds used by proteases for catalysis
Enzyme Catalysis-Serine Proteases Concepts to be learned Activation Energy Transition State Example: Proteases Requirements for proteolysis Families of proteases Protein Folds used by proteases for catalysis
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Bio 12 Chapter 2 Test Review
 Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
I. Polymers & Macromolecules Figure 1: Polymers. Polymer: Macromolecule: Figure 2: Polymerization via Dehydration Synthesis
 I. Polymers & Macromolecules Figure 1: Polymers Polymer: Macromolecule: Figure 2: Polymerization via Dehydration Synthesis 1 Dehydration Synthesis: Figure 3: Depolymerization via Hydrolysis Hydrolysis:
I. Polymers & Macromolecules Figure 1: Polymers Polymer: Macromolecule: Figure 2: Polymerization via Dehydration Synthesis 1 Dehydration Synthesis: Figure 3: Depolymerization via Hydrolysis Hydrolysis:
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B
 3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
9/16/15. Properties of Water. Benefits of Water. More properties of water
 Properties of Water Solid/Liquid Density Water is densest at 4⁰C Ice floats Allows life under the ice Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form Benefits
Properties of Water Solid/Liquid Density Water is densest at 4⁰C Ice floats Allows life under the ice Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form Benefits
Biology. Lectures winter term st year of Pharmacy study
 Biology Lectures winter term 2008 1 st year of Pharmacy study 3 rd Lecture Chemical composition of living matter chemical basis of life. Atoms, molecules, organic compounds carbohydrates, lipids, proteins,
Biology Lectures winter term 2008 1 st year of Pharmacy study 3 rd Lecture Chemical composition of living matter chemical basis of life. Atoms, molecules, organic compounds carbohydrates, lipids, proteins,
Structural analysis of fungus-derived FAD glucose dehydrogenase
 Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
2-2 Properties of Water
 2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN LAJIDE LETURE NTENT INTRDUTIN GLYGEN BREAKDWN (Glycogenolysis) GLYGEN SYNTESIS (Glycogenesis) REGULATIN F GLYGEN METABLISM MAINTENANE F BLD GLUSE DIABETES MELLITUS AND INSULIN INTRDUTIN
BY: RASAQ NURUDEEN LAJIDE LETURE NTENT INTRDUTIN GLYGEN BREAKDWN (Glycogenolysis) GLYGEN SYNTESIS (Glycogenesis) REGULATIN F GLYGEN METABLISM MAINTENANE F BLD GLUSE DIABETES MELLITUS AND INSULIN INTRDUTIN
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
An aldose contains an aldehyde functionality A ketose contains a ketone functionality
 RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
A. Structure and Function 1. Carbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b.
 Biochemistry 2 A. Structure and Function 1. arbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b. in three dimensions (3D) Diagrams in 2D may
Biochemistry 2 A. Structure and Function 1. arbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b. in three dimensions (3D) Diagrams in 2D may
Exams written in pencil or erasable ink will not be re-graded under any circumstances.
 Biochemistry 461, Section I May 21, 1998 Final Exam Prof. Jason D. Kahn Your Printed ame: Your SS#: Your Signature: You have 120 minutes for this exam. The exam has 7 questions, worth 200 points. Do all
Biochemistry 461, Section I May 21, 1998 Final Exam Prof. Jason D. Kahn Your Printed ame: Your SS#: Your Signature: You have 120 minutes for this exam. The exam has 7 questions, worth 200 points. Do all
Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life
 AP Biology Name: Block Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life Most of this chapter is new material. We will discuss it all in detail. Section 1 1. Make an electron distribution
AP Biology Name: Block Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life Most of this chapter is new material. We will discuss it all in detail. Section 1 1. Make an electron distribution
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chapter 1. Chemistry of Life - Advanced TABLE 1.2: title
 Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Honors Biology Chapter 3: Macromolecules PPT Notes
 Honors Biology Chapter 3: Macromolecules PPT Notes 3.1 I can explain why carbon is unparalleled in its ability to form large, diverse molecules. Diverse molecules found in cells are composed of carbon
Honors Biology Chapter 3: Macromolecules PPT Notes 3.1 I can explain why carbon is unparalleled in its ability to form large, diverse molecules. Diverse molecules found in cells are composed of carbon
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase. Biol 405 Molecular Medicine
 Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
SUPPLEMENTAL INFORMATION
 SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
Four melanocyte-stimulating hormones have the following amino acid sequences:
 Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
Chapter 5: The Structure and Function of Large Biological Molecules
 Chapter 5: The Structure and Function of Large Biological Molecules 1. Name the four main classes of organic molecules found in all living things. Which of the four are classified as macromolecules. Define
Chapter 5: The Structure and Function of Large Biological Molecules 1. Name the four main classes of organic molecules found in all living things. Which of the four are classified as macromolecules. Define
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced to express
 Supplementary Figure 1. VapC-mt4 cleaves trna Ala2 in E. coli. Histograms representing the fold change in reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced
Supplementary Figure 1. VapC-mt4 cleaves trna Ala2 in E. coli. Histograms representing the fold change in reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced
Wheat Amino acids & Peptides for Hair Care. INCI Name EU/USA CAS # EINECS # Hydrolyzed wheat protein
 KELYAMIN Wheat Amino acids & Peptides for Hair Care Identification INCI Name EU/USA CAS # EINECS # Hydrolyzed wheat protein 70084-87-6 305-225-0 Composition % Liquid Powder Aqua Hydrolyzed wheat protein
KELYAMIN Wheat Amino acids & Peptides for Hair Care Identification INCI Name EU/USA CAS # EINECS # Hydrolyzed wheat protein 70084-87-6 305-225-0 Composition % Liquid Powder Aqua Hydrolyzed wheat protein
Carbon. p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms
 Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Chapter Three (Biochemistry)
 Chapter Three (Biochemistry) 1 SECTION ONE: CARBON COMPOUNDS CARBON BONDING All compounds can be classified in two broad categories: organic compounds and inorganic compounds. Organic compounds are made
Chapter Three (Biochemistry) 1 SECTION ONE: CARBON COMPOUNDS CARBON BONDING All compounds can be classified in two broad categories: organic compounds and inorganic compounds. Organic compounds are made
Catalysis & specificity: Proteins at work
 Catalysis & specificity: Proteins at work Introduction Having spent some time looking at the elements of structure of proteins and DNA, as well as their ability to form intermolecular interactions, it
Catalysis & specificity: Proteins at work Introduction Having spent some time looking at the elements of structure of proteins and DNA, as well as their ability to form intermolecular interactions, it
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Introduction to Biochemistry Midterm exam )ومن أحياها(
 Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
Chapter 3. Protein Structure and Function
 Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
INTRODUCTION TO ORGANIC COMPOUNDS. Copyright 2009 Pearson Education, Inc.
 INTRODUCTION TO ORGANIC COMPOUNDS 3.1 I can explain why carbon is unparalleled in its ability to form large, diverse molecules. Diverse molecules found in cells are composed of carbon bonded to other elements
INTRODUCTION TO ORGANIC COMPOUNDS 3.1 I can explain why carbon is unparalleled in its ability to form large, diverse molecules. Diverse molecules found in cells are composed of carbon bonded to other elements
Streptococcus mutans
 INFECTION AND IMMUNITY, Aug. 1983, p. 722-727 19-9567/83/8722-6$2./ Copyright C 1983, American Society for Microbiology Vol. 41, No. 2 Genetic Transformation of Putative Cariogenic Properties in Streptococcus
INFECTION AND IMMUNITY, Aug. 1983, p. 722-727 19-9567/83/8722-6$2./ Copyright C 1983, American Society for Microbiology Vol. 41, No. 2 Genetic Transformation of Putative Cariogenic Properties in Streptococcus
Four Classes of Biological Macromolecules. Biological Macromolecules. Lipids
 Biological Macromolecules Much larger than other par4cles found in cells Made up of smaller subunits Found in all cells Great diversity of func4ons Four Classes of Biological Macromolecules Lipids Polysaccharides
Biological Macromolecules Much larger than other par4cles found in cells Made up of smaller subunits Found in all cells Great diversity of func4ons Four Classes of Biological Macromolecules Lipids Polysaccharides
GLYCATION OF PROTEINS IN ESCHERICHIA COLI: EFFECT OF NUTRIENT BROTH INGREDIENTS ON GLYCATION
 Industry GLYCATION OF PROTEINS IN ESCHERICHIA COLI: EFFECT OF NUTRIENT BROTH INGREDIENTS ON GLYCATION R. Dimitrova, R. Mironova, I. Ivanov Institute of Molecular biology, Bulgarian Academy of Sciences
Industry GLYCATION OF PROTEINS IN ESCHERICHIA COLI: EFFECT OF NUTRIENT BROTH INGREDIENTS ON GLYCATION R. Dimitrova, R. Mironova, I. Ivanov Institute of Molecular biology, Bulgarian Academy of Sciences
Point total. Page # Exam Total (out of 90) The number next to each intermediate represents the total # of C-C and C-H bonds in that molecule.
 This exam is worth 90 points. Pages 2- have questions. Page 1 is for your reference only. Honor Code Agreement - Signature: Date: (You agree to not accept or provide assistance to anyone else during this
This exam is worth 90 points. Pages 2- have questions. Page 1 is for your reference only. Honor Code Agreement - Signature: Date: (You agree to not accept or provide assistance to anyone else during this
Chapter 8. Interaction between the phosphatidylinositol 3- kinase SH3 domain and a photocleavable cyclic peptide
 Interaction between the phosphatidylinositol 3- kinase SH3 domain and a photocleavable cyclic peptide 129 Abstract The interaction of the PI3K SH3 domain with a cyclic photocleavable peptide and the linear
Interaction between the phosphatidylinositol 3- kinase SH3 domain and a photocleavable cyclic peptide 129 Abstract The interaction of the PI3K SH3 domain with a cyclic photocleavable peptide and the linear
Molecular characterization and expression analysis of the glucansucrase DSRWC from Weissella cibaria synthesizing a a(1! 6) glucan
 RESEARCH LETTER Molecular characterization and expression analysis of the glucansucrase DSRWC from Weissella cibaria synthesizing a a(1! 6) glucan Hee-Kyoung Kang 1,2, Jong-Suk Oh 3 & Doman Kim 1,2 1 The
RESEARCH LETTER Molecular characterization and expression analysis of the glucansucrase DSRWC from Weissella cibaria synthesizing a a(1! 6) glucan Hee-Kyoung Kang 1,2, Jong-Suk Oh 3 & Doman Kim 1,2 1 The
Lecture 2: Glycogen metabolism (Chapter 15)
 Lecture 2: Glycogen metabolism (Chapter 15) First. Fig. 15.1 Review: Animals use glycogen for ENERGY STORAGE. Glycogen is a highly-branched polymer of glucose units: Basic structure is similar to that
Lecture 2: Glycogen metabolism (Chapter 15) First. Fig. 15.1 Review: Animals use glycogen for ENERGY STORAGE. Glycogen is a highly-branched polymer of glucose units: Basic structure is similar to that
2.1.1 Biological Molecules
 2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
Organic Compounds. Biology-CP Mrs. Bradbury
 Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
Carbon. Isomers. The Chemical Building Blocks of Life
 The Chemical Building Blocks of Life Carbon Chapter 3 Framework of biological molecules consists primarily of carbon bonded to Carbon O, N, S, P or H Can form up to 4 covalent bonds Hydrocarbons molecule
The Chemical Building Blocks of Life Carbon Chapter 3 Framework of biological molecules consists primarily of carbon bonded to Carbon O, N, S, P or H Can form up to 4 covalent bonds Hydrocarbons molecule
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.
 Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Lesson 2. Biological Molecules. Introduction to Life Processes - SCI 102 1
 Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Glycogen Metabolism. BCH 340 lecture 9
 Glycogen Metabolism BC 340 lecture 9 Structure of glycogen Glycogen is homopolysaccharide formed of branched D-glucose units The primary glycosidic bond is 1-4-linkage Each branch is made of 6-12 glucose
Glycogen Metabolism BC 340 lecture 9 Structure of glycogen Glycogen is homopolysaccharide formed of branched D-glucose units The primary glycosidic bond is 1-4-linkage Each branch is made of 6-12 glucose
BIOCHEMISTRY & MEDICINE:
 BIOCHEMISTRY & MEDICINE: INTRODUCTION Biochemistry can be defined as the science of the chemical basis of life (Gk bios "life"). The cell is the structural unit of living systems. Thus, biochemistry can
BIOCHEMISTRY & MEDICINE: INTRODUCTION Biochemistry can be defined as the science of the chemical basis of life (Gk bios "life"). The cell is the structural unit of living systems. Thus, biochemistry can
