Insulin receptors and renal sodium handling in hypertensive fructose-fed rats
|
|
|
- Barry Melton
- 5 years ago
- Views:
Transcription
1 Kidney International, Vol. (3), pp Insulin receptors and renal sodium handling in hypertensive fructose-fed rats CRISTIANA CATENA,ALESSANDRO CAVARAPE, MARILEDA NOVELLO, GILBERTA GIACCHETTI, and LEONARDO A. SECHI Clinica Medica, Hypertension Unit, Department of Experimental and Clinical Pathology and Medicine, University of Udine, Udine, Italy; and Clinica di Endocrinologia, University of Ancona, Ancona, Italy Insulin receptors and renal sodium handling in hypertensive fructose-fed rats. Background. Insulin resistance and hypertension are present in Sprague-Dawley rats fed a fructose-enriched diet. In these rats, insulin might elevate blood pressure via an antinatriuretic action. Methods. To investigate the sodium-insulin interaction in fructose-fed rats, we compared insulin sensitivity, insulin receptor binding, and insulin receptor mrna levels in the kidney and skeletal muscle of rats that were fed standard rat chow or a fructose-enriched diet (%) with either low (.7%), normal (.3%), or high (7.5%) NaCl concentrations for 3 weeks. Results. Systolic blood pressure increased in the fructose-fed rats receiving the normal and high-salt diet, but not the low-salt diet. When the rats were fed the low-salt diet, the rate of glucose infusion required to maintain euglycemia during a hyperinsulinemic clamp and insulin receptor number and mrna levels in skeletal muscle were lower in fructose-fed than control rats. High-salt diet decreased significantly the rate of glucose disposal during the clamp and muscular insulin receptor number and mrna levels in control, but not fructose-fed rats. During the low-salt diet, renal insulin receptor number and mrna levels were comparable in fructose-fed and control rats and hyperinsulinemia had comparable acute antinatriuretic effects in the two groups; when the rats were maintained on the high-salt diet, the expected decrease in renal insulin receptor number and mrna levels occurred in control but not fructose-fed rats and, consistent with this finding, the antinatriuretic response to hyperinsulinemia was blunted only in controls. An inverse relationship between dietary NaCl content and renal insulin receptor mrna levels was observed in control but not fructose-fed rats. Conclusion. -fed rats appear to have lost the feedback mechanism that limits insulin-induced sodium retention through a down-regulation of the renal insulin receptor when the dietary NaCl content is increased. This abnormality might possibly contribute to the elevation of blood pressure in these rats. Key words: hyperinsulinemic clamp, insulin binding, insulin sensitivity, messenger RNA, salt. Received for publication April 3, 3 and in revised form June, 3 Accepted for publication July 17, 3 C 3 by the International Society of Nephrology The association between hyperinsulinemia, insulin resistance, and essential hypertension [1, ] has led to hypothesize an involvement of insulin in the pathogenesis of this disorder [3]. Abnormal glucose metabolism with decreased insulin sensitivity has been demonstrated also in some hypertensive rodent models such as the Dahl salt-sensitive [], spontaneously hypertensive [5], Milan hypertensive [], and fructose-fed rat [7], suggesting that similar mechanisms might occur in humans and in animal models [3]. Chronic fructose feeding in normal rats causes hypertension in association with insulin resistance and abnormal plasma lipid profile [7]. Recovery of normal insulin sensitivity by physical training [] or administration of drugs that improve sensitivity to insulin such as metformin [9] and troglitazone [1] and reduction of circulating insulin levels by administration of somatostatin [11] normalize blood pressure in fructose-fed rats, supporting the hypothesis of a contribution of insulin to blood pressure increase. Different mechanisms could possibly account for the prohypertensive action of insulin. These mechanisms include activation of the sympathetic nervous system [1], growth-promoting activity on vascular smooth muscle cells [13], increase in intracellular calcium level [1], and increase in renal sodium reabsorption [15]. With regard to the latter mechanism, experimental studies have shown that insulin increases sodium reabsorption by acting on several distinct segments of the renal tubule [1 1]. We have previously demonstrated an inverse relationship between dietary salt intake and renal insulin receptor density and mrna levels, suggesting the existence of a feedback mechanism that could limit insulin-induced sodium retention when extracellular fluid volume is expanded [19] and that this relationship is lost in a strain of rats with hypertension associated with insulin resistance []. The present study was designed to evaluate whether dietary salt intake affects the functional responses to insulin in the kidney and skeletal muscle of rats with fructoseinduced hypertension. We measured the renal and 13
2 1 Catena et al: Insulin and sodium in fructose-hypertension metabolic responses to an euglycemic-hyperinsulinemic clamp, insulin receptor binding parameters, and insulin receptor mrna levels in fructose-fed and control rats maintained on diets with different salt content. METHODS All experiments were performed in male Sprague- Dawley rats (Banting Kingman, Fremont, CA, USA) weighing 15 to 1 g that were housed in climatecontrolled conditions with a 1-hour light/1-hour dark cycle and provided tap water ad libitum. We performed three sets of rat experiments using two different protocols, as performed in previous studies [1]. Protocol A Rats were fed standard rat chow for 3 days and were then divided into four groups: rats fed standard chow containing a low-salt concentration (NaCl.7%) (Harlan Teklad, Madison, WI, USA); rats fed standard chow containing a high-salt concentration (NaCl 7.5%); rats fed a fructose-enriched diet (fructose %, fat 5%, casein %, cellulose 7%, vitamins 1%) (Harlan Teklad) containing a low-salt concentration; and rats fed a fructoseenriched diet containing a high-salt concentration. Body weight and systolic blood pressure were measured every other day []. Systolic blood pressure was measured in conscious, prewarmed (light lamp), restrained rats by the tail-cuff method with plethysmography and a physiograph recorder (Pulse Amplifier) (ITTC Life Sciences, Woodland Hills, CA, USA). For these measurements, rats were trained adequately before the study. Two distinct sets of experiments were conducted in which, after 3 weeks on the experimental diets, rats were prepared for the euglycemic-hyperinsulinemic clamp and rats were killed by decapitation; in the latter group trunk blood was collected in ethylenediaminetetraacetic acid (EDTA) and skeletal muscle (always the same muscle groups from lower limbs) and kidneys were removed and snap-frozen in liquid nitrogen for insulin receptor binding study. Protocol B Rats were fed standard rat chow for 3 days and were then divided into six groups: rats fed standard chow containing low-salt (NaCl.7%), normal-salt (NaCl.3%), or high-salt concentration (NaCl 7.5%); rats fed a fructose-enriched diet (%) containing low-salt, normal-salt, or high-salt concentration. After 3 weeks, rats were decapitated, trunk blood was collected for measurement of plasma glucose, insulin, and triglycerides, and skeletal muscle and kidneys removed and snapfrozen for total RNA isolation. Euglycemic-hyperinsulinemic clamp study The euglycemic-hyperinsulinemic clamp was used to determine insulin sensitivity and to evaluate the renal effects of insulin in rats that were housed and fed as in protocol A, following a procedure that has been described previously []. On the day of the experiment, rats were anesthetized with an intraperitoneal injection of 1 mg/kg of thiobutabarbital (Inactin) (Andrew Lockwood, Sturtevant, WI, USA) and placed on a heated table to maintain rectal temperature at 37 C. Animals underwent tracheostomy and breathed spontaneously. Thereafter, they were prepared for acute experimentation as described previously []. Briefly, catheters were placed in the right jugular vein for insulin infusion, the left jugular vein for the infusion of glucose and saline, and the right femoral artery for blood sampling and blood pressure monitoring via a Gemini 77 pressure transducer (Ugo Basile, Comerio, Italy). A flanged catheter was placed in the bladder through a suprapubic incision for urine collection. To replace fluid losses during the surgical preparation, rats received a constant intravenous infusion of plasma substitute (Hespan, % hetastarch in.9% NaCl) (Du Pont Pharmaceuticals, Wilmington, DE, USA) at a rate of ll/min until a total volume of.5% body weight was administered. The rate of the infusion was subsequently adjusted to match the urinary output and was not significantly different between control and fructose-fed rats fed either low- or high-salt diet. Urine was collected from the bladder in preweighed polypropylene tubes over a 5-minute period for determination of baseline urine flow rate and sodium concentration. Blood was obtained every 15 minutes for glucose measurement (Accu-check II) (Boheringer Mannheim, Indianapolis, IN, USA) and at the beginning and end of this interval for sodium determination. After the 5-minute baseline period, rats received an insulin load of 5 mu/kg followed by a continuous insulin infusion of mu/kg per minute. As expected, this infusion produced comparable plasma insulin levels in fructose-fed and control rats fed with both low-salt and high-salt chow. After injection of the insulin bolus, a 5% glucose solution was infused for a -minute stabilization interval, during which blood glucose was determined at 5-minute intervals and the rate of glucose infusion was adjusted to maintain blood glucose at the average level measured at baseline. Thereafter, a second 5-minute experimental period was begun, during which urine flow rate, urinary and plasma sodium concentration, and blood glucose concentration were determined, and the rate of glucose infusion was adjusted as indicated above. The glucose disposal rate was calculated during the 5 minutes of the experimental period from the amount of glucose required to maintain euglycemia. Insulin receptor binding studies The distribution and binding characteristics of insulin receptors in skeletal muscle and kidney were assessed in rats that were housed and fed as in protocol A, by an situ autoradiographic technique associated with
3 Catena et al: Insulin and sodium in fructose-hypertension 15 computerized microdensitometry, following a procedure that has been described previously and to which readers are referred for further details [19]. Briefly, adjacent tissue sections ( lm thick) were cut on a cryostat, liophylized, preincubated twice for 1 minutes in KCl (3 mmol/l), and incubated for 1 minutes in a buffer containing pmol/l 15 I-Tyr-insulin ( Ci/mmol) (DuPont-NEN, Boston, MA, USA) in the presence of increasing concentrations (from 1 pmol/l to 1 mmol/l) of unlabeled insulin (Humulin R) (Eli-Lilly, Indianapolis, IN, USA), then rinsed in ice-cold buffer, and dried for hours in a stream of cool air. In the kidney, regional analysis of insulin binding was performed in renal cortex, and outer and inner renal medulla by use of film autoradiographs obtained by exposing the tissue sections on LKB-Ultrofilm (Leica, Inc., Deerfield, IL, USA). Optical density in the different regions was measured by computerized microdensitometry. Scatchard analysis of equilibrium binding data was done with the Ligand program of Munson and Rodbard and resulted in curvilinear profiles indicating either two classes of insulin receptors (R1: high-affinity, low-capacity; R: low-affinity, highcapacity) or a negative cooperative hormone-receptor interaction [19]. In all the experiments, data were analyzed for a two-site model. Insulin receptor mrna studies The levels of insulin receptor mrna in skeletal muscle and kidney were assessed in 3 rats that were housed and fed as in protocol B. Total RNA was isolated from frozen tissue, as described previously [19]. The RNA pellet was dissolved in sterile water and quantitated by ultraviolet absorbance at / nm. A.9 kb 3 P-labeled antisense insulin receptor crna probe encoding the 5 end of the rat insulin receptor cdna was synthesized using a PpMuI linearized rat insulin receptor clone, T3 RNA polymerase, and 3 P-CTP ( Ci/mmol). Probe purification was performed by spin column centrifugation using a G-5 Sephadex RNA Purification Quick Spin Column (Boheringer Mannheim). Insulin receptor mrna levels were measured by slotblot hybridization analysis. Nitrocellulose filters were prehybridized in a solution containing 5% formamide, standard sodium citrate (SSC), 1 Denhardt s, 1 mmol/l Tris (ph 7.), lg/ml sheared denatured salmon sperm DNA, and.% sodium dodecyl sulfate (SDS) for to hours at 5 C. Hybridization was performed using fresh prehybridization solution, to which 3 P-labeled rat insulin receptor probe was added at a final concentration of.5 to 3. 1 cpm/ml of hybridization solution, and incubated for 1 to hours at 5 C. Following hybridization, the membranes were washed for 3 minutes at 5 Cin SSC-.1% SDS,.5 SSC-.1% SDS,.1 SSC-.5% SDS, and finally in.1 SSC-.5% SDS. Autoradiographs were obtained by exposing the filters to Cronex x-ray film (Du Pont) with an intensifying screen at C for 3 to 5 days and were analyzed by scanning densitometry (GS7 Imaging Densitometer) (Bio-Rad, Hercules, CA, USA). To ensure equivalent loading conditions, duplicate blots were prepared and hybridized with a 3 P-labeled oligonucleotide probe complementary to bases 11 to 3 of S ribosomal RNA [19]. Probe purification was performed by spin column centrifugation using a G-5 Sephadex DNA Purification Quick Spin Column. After prehybridization, hybridization was performed in a solution containing SSC, 5 Denhardt s, % SDS, and 1% Ppi, to which 3 P-labeled probe was added at a final concentration of 1. 1 cpm/ml of hybridization solution, and incubating for 1 to hours at C. Plasma glucose, insulin, triglycerides, and serum and urinary sodium Sodium concentrations in serum and urine were measured by flame photometry (Klina Flame) (Beckman, Fullerton, CA, USA). Blood glucose concentration in trunk blood was determined by the glucose oxidase method (Beckman Glucose Analyzer) (Beckman). Plasma triglycerides were assayed enzymatically by an automated method. Plasma insulin was measured by radioimmunoassay (Behring, Marburg, Germany) []. Statistical analysis Data are expressed as mean ± SE. Comparisons among groups were done by Student t test for unpaired data or analysis of variance (ANOVA) for multiple comparisons or repeated measures (GB-Stat) (Dynamic Microsystems, Inc., Silver Spring, MD, USA) when appropriate. Correlations are expressed by the correlation coefficient r. Differences were considered as statistically significant when P was less than.5. RESULTS Body weight, systolic blood pressure, and plasma glucose, insulin, and triglycerides As partially reported in a previous study [1], plasma triglyceride levels were significantly greater in the fructose-fed rats independent of dietary salt content, whereas body weight, fasting plasma glucose, and fasting plasma insulin did not differ significantly among groups (Table 1). Systolic blood pressure levels were significantly greater in the fructose-fed rats that received either the normal or high-salt diet but no significant difference was observed in the fructose-fed rats that received the low-salt diet (Table 1).
4 1 Catena et al: Insulin and sodium in fructose-hypertension Table 1. Body weight, systolic blood pressure, plasma triglycerides, and fasting plasma glucose and insulin levels in control and fructose-fed rats maintained on low-salt, normal-salt, and high-salt diet Control rats -fed rats Low-salt Normal-salt High-salt Low-salt Normal-salt High-salt Parameter (N = ) (N = ) (N = ) (N = ) (N = ) (N = ) Body weight g 9 ± 5 ± ± ± 1 ± 5 5 ± 9 Systolic blood pressure mm Hg 173 ± ± 175 ± 1 ± 1 ± a 1 ± a Plasma triglycerides mmol/l 1.1 ± ±.1.93 ±.7 1. ±.3 a 1.9 ±.19 a 1. ±. b Plasma glucose mmol/l. ±.1. ±..7 ±.1.7 ±.. ±.1.7 ±. Plasma insulin mmol/l 7 ± ± 57 ± ± 51 7 ± 91 ± 11 Values are means ± SE. Systolic blood pressure measurements are those obtained on the day of sacrifice. Comparisons were done by two-way analysis of variance (ANOVA). a P <.5; b P <.1 vs. the respective control. Table. Effect of euglycemic-hyperinsulinemic clamp on systolic blood pressure, glucose disposal rate, and urinary sodium excretion in control and fructose-fed rats fed low-salt and high-salt Control rats -fed rats Low-salt High-salt Low-salt High-salt Parameter (N = ) (N = ) (N = ) (N = ) Systolic blood pressure mm Hg Baseline 15 ± 1 ± 3 1 ± 1 ± b,d Experimental 15 ± 3 11 ± 159 ± 3 1 ± 3 b,d Plasma glucose mmol/l Baseline.9 ±.3 7. ±. 7.1 ±. 7. ±. Experimental 7. ±..9 ±.5.9 ±.3.7 ±.5 Plasma insulin pmol/l Baseline 593 ± 5 55 ± 39 ± 9 ± 5 Experimental 579 ± 3 f 3 ± 37 f 91 ± f 77 ± 359 f Glucose utilization rate (mg/kg)/min Experimental 3.7 ± ±.1 c 1. ±. a 11.9 ± 3.5 Serum sodium mmol/l Baseline 15 ± 1 ± 1 1 ± 3 15 ± 1 Experimental 15 ± 1 15 ± 17 ± 1 17 ± Urine flow ll/min Baseline 9.7 ± ±.7 d 1.5 ± 3.5. ±.5 d Experimental.1 ±. e 3.5 ± ± 3.3 e. ±.5 e UNaV lmol/min Baseline.11 ±. 1.9 ± 1.55 d.1 ±. 1.7 ± 1.3 d Experimental. ±. e 15.3 ± ±.1 e 11.9 ± 1.39 e UnaV is urinary sodium excretion. Values are means ± SE. Values of systolic blood pressure, plasma glucose, plasma insulin, and serum sodium are the average of measurements done at the beginning ( minute) and at the end (5 minutes) of the baseline and experimental period. Comparisons were done by two-way analysis of variance (ANOVA). a P <.; b P <.1 vs. the respective control; c P <.5; d P <.1 vs. low salt diet; e P <.5; f P <.1 vs. baseline. Euglycemic-hyperinsulinemic clamp study During the clamp study, plasma insulin levels increased seven- to eightfold, euglycemia was maintained adequately in all groups, and blood pressure did not change significantly in any group. During the low-salt diet, the amount of glucose infused to maintain euglycemia during the hyperinsulinemic clamp was significantly less in fructose-fed rats than controls, indicating reduced sensitivity to insulin; high-salt diet decreased significantly insulin-induced glucose utilization in control but not fructose-fed rats (Table ). Baseline urine flow rate and urinary sodium excretion were comparable in fructosefed and control rats fed both low- and high-salt chow. During the low-salt diet, hyperinsulinemia caused in both fructose-fed and control rats a significant reduction of urine flow rate and urine sodium excretion. When rats were fed the high-salt diet, hyperinsulinemia maintained its antidiuretic and antinatriuretic effect in fructose-fed, but not control rats (Table ). Insulin receptor binding studies Radiolabeled insulin binding was homogeneously distributed in the skeletal muscle of fructose-fed and control rats fed both low- and high-salt diet. Analysis of binding data showed a significantly decreased Bmax of the R1 (high-affinity, low-capacity receptor) in the muscle of fructose-fed rats maintained on a low-salt diet as compared to controls, but no difference in the Kd of R1 and in both the Kd and Bmax of R (low-affinity, high-capacity receptor). When rats were fed high-salt, a significant decrease in the Bmax of R1 was observed in control but not fructose-fed rats,
5 Catena et al: Insulin and sodium in fructose-hypertension 17 Table 3. Insulin binding parameters in skeletal muscle and kidney of control and fructose-fed rats fed low-salt and high-salt diet Control rats -fed rats Low-salt High-salt Low-salt High-salt Parameter (N = ) (N = ) (N = ) (N = ) Skeletal muscle R1 Kd, 1 1 M.3 ±.9. ± 1.1. ± 1.. ± 1.5 Bmax, 1 1 receptors/mm ± ± 1.5 b 9. ± 1.5 a.7 ± 1. R Kd, 1 7 M 3.1 ± ± ± 1.7. ±. Bmax, 1 13 receptors/mm 3. ±..9 ±.1.3 ± ± 1. Kidney R1 Kd, 1 1 M 3. ± ± ± ± 1.3 Bmax, 1 1 receptors/mm 3.5 ± ± 1.7 b. ± ± 1.7 R Kd, 1 7 M 3.1 ±. 3.7 ± 1..9 ± ± 1.1 Bmax, 1 13 receptors/mm 3.9 ± ±.1 5. ±.. ± 1.5 R1 is high-affinity, low-capacity insulin binding site; R is low-affinity, high-capacity insulin binding site. Binding parameters (Kd, dissociation constant; Bmax, maximum binding capacity) were derived from Scatchard analysis of equilibrium binding data from competition experiments performed on adjacent tissue sections that, after incubation with radioligand, were placed in a gamma counter. Values are means ± SE. Comparisons were done by two-way analysis of variance (ANOVA). a P <.5 vs. the respective control; b P <.5 vs. low-salt diet. whereas other binding parameters remained unchanged (Table 3). In the kidney, insulin binding was more abundant in the renal cortex than medulla and the distribution was comparable in all four groups of rats. Analysis of binding data showed no differences in the Kd and Bmax of both insulin receptor sites between fructose-fed and control rats maintained on a low-salt diet. When control rats were fed high-salt, a significant decrease in the Bmax of R1 was observed in comparison to control rats fed lowsalt, whereas other binding parameters did not change. In fructose-fed rats, the high-salt diet did not affect the Bmax and Kd of both insulin receptor sites (Table 3). Analysis of regional binding to renal cortex, outer medulla, and inner medulla showed that insulin receptor density was comparable in all regions of the kidneys obtained from fructose-fed and control rats fed a low-salt diet. High-salt diet decreased insulin receptor density in all regions of the kidney in control but not fructose-fed rats (Fig. 1). Insulin receptor mrna studies Insulin receptor mrna levels in the muscle were significantly lower in fructose-fed than control rats fed low and normal-salt diet. High-salt diet induced a significant decrease in insulin receptor mrna levels in the muscle of control but not fructose-fed rats (Fig. ). In the kidney of fructose-fed and control rats maintained on a low-salt diet, insulin receptor mrna levels were comparable. High-salt diet decreased significantly renal insulin receptor mrna in control but not fructose-fed rats (Fig. ). In both muscle and kidney of control rats, insulin receptor mrna levels were inversely correlated with dietary salt content (r =.59, P <.1, and r =.39, P <.1, respectively) but no relationship was observed in fructose-fed rats. DISCUSSION The findings of this study demonstrate that the density and mrna levels of insulin receptors in the kidney of fructose-fed rats do not differ from controls when rats are fed low and normal-salt diet. High-salt diet decreases the insulin receptor number and mrna levels in the kidney of control rats, as expected from the results of previous studies [19], but this does not occur in rats fed a fructoseenriched diet. Consistent with receptor findings, the acute antinatriuretic effect of insulin is significantly decreased by the high-salt diet in controls but not fructose-fed rats, suggesting that the feedback mechanism that normally limits insulin-induced sodium reabsorption when dietary salt is increased is lacking in this experimental model of hypertension associated with insulin resistance. Different from the kidney, evaluation of the effects of insulin in a tissue, such as the skeletal muscle, that has a primary role in the metabolic actions of the hormone, demonstrates that insulin sensitivity is significantly reduced in fructose-fed rats fed low and normal-salt diet as compared to controls. Decreased insulin-stimulated glucose utilization in fructose-fed rats is associated with decreased insulin receptor number and mrna levels, providing a possible molecular explanation for insulin resistance and indicating tissue-specific regulation of the insulin receptor in this model. Similar to the kidney, the high-salt diet down-regulates muscular insulin receptor number and mrna levels in control but not fructose-fed rats.
6 1 Catena et al: Insulin and sodium in fructose-hypertension A B C 1 Control 1 Control 1 Control P <.1 P <.1 P < Fig. 1. Effect of varying concentrations of unlabeled insulin on binding of 15 I-insulin in cortex (A), outer medulla (B), and inner medulla (C) of kidneys obtained from control and fructose-fed rats fed low-salt ( ) or high-salt ( ) diet. Data were obtained by computerized microdensitometry and are means ± SE of six animals in each group. For each rat, the area below the curve was calculated and the average of the groups compared with Student t test. Insulin receptor density was comparable in fructose-fed and control rats fed a low-salt diet. High-salt diet decreased significantly insulin receptor binding in all renal regions in control but not fructose-fed rats. Possible contribution of insulin to the pathogenesis of arterial hypertension has been suggested following the demonstration of hyperinsulinemia and insulin resistance in essential hypertensive patients [1, ] as well as hypertensive rat models [ ]. Chronic fructose feeding induces hypertension associated with insulin resistance and compensatory hyperinsulinemia in normal rats [7] and this model has been largely employed to investigate the mechanisms of insulin-induced hypertension. Some controversy about fructose-induced hypertension is mainly related to the possibility to induce hyperinsulinemia [1, 3] and hypertension [3] and many potential explanations have been offered, such as differences in the technique used to measure blood pressure [3, ], rat strains [5], and dietary components (e.g., lipids or sodium) [, 7]. More specifically, in our and in other authors [3] hands, fasting hyperinsulinemia cannot be obtained by chronically feeding rats a fructoseenriched diet and, as indicated above, this fact resides among the many disputes regarding this experimental model. In both protocol A and protocol B and independent of salt intake, the fructose-fed rats had greater average fasting insulin levels than controls, although these differences did not reach statistical significance. An explanation might be that the reduced sample size could have limited the power of the study to detect such a difference (b-error). Most important, however, we have previously demonstrated that plasma insulin is significantly increased in fructose-fed rats as compared to controls after a standard oral glucose load [1], suggesting that hyperinsulinemia might be more relevant in the postprandial state. In our opinion, the fructose-model is definitely an hyperinsulinemic rat model of hypertension and, as such, findings on these rats might be extrapolated to human conditions in which hypertension is associated with hyperinsulinemia. In the consideration of possible mechanisms that could mediate the prohypertensive action of insulin, particular attention has been paid to the renal action of the hormone. Insulin, in fact, activates renal sodium reabsorption during acute infusions [15], but the exact site of the tubular action of insulin is unclear and previous reports have variously indicated an effect in the proximal tubule [1], distal tubule [17], and loop of Henle [1]. Indirect
7 Catena et al: Insulin and sodium in fructose-hypertension 19 Scanner units Scanner units A 9 3 B P<.5 P<.5 P<. P<. P<.5 Control Control P<.5 Low salt Normal salt High salt Low salt Normal salt High salt Fig.. Bar graphs of insulin receptor mrna in skeletal muscle (A) and kidney (B) of control and fructose-fed rats maintained on a low-salt, normal-salt, and high-salt diet. Data were obtained from slot-blot analysis performed using a.9 kb 3 P-labeled antisense insulin receptor crna probe. Optical density was determined by computerized densitometry and normalized for S ribosomal RNA. Data are the means ± SE of six animals in each group. In skeletal muscle and kidney of control rats, insulin receptor mrna levels were progressively decreased by an increasing dietary salt content. In fructose-fed rats, no significant changes in insulin receptor mrna levels were observed with different dietary salt content. evidence of the possible relevance of insulin in the regulation of sodium balance was provided by experiments in which an inverse relationship between dietary sodium intake and renal insulin receptor number and mrna levels was demonstrated [19], indicating the existence of a feedback mechanism that limits insulin-induced sodium retention when extracellular fluid volume is expanded. Of particular interest is that, in our present experiments, a significant and comparable insulin-induced antinatriuretic effect occurred in fructose-fed and control rats during a low-salt diet, and that this effect normally disappeared in control but not fructose-fed rats during a highsalt diet. Evaluation of renal insulin receptor binding and mrna data indicate that this difference was mediated at the level of insulin receptor, inasmuch as the high-salt diet down-regulated both receptor number and mrna levels in control but not fructose-fed rats, thereby suggesting a molecular explanation for this observation. It must be acknowledged, however, that other mechanisms beside abnormal regulation of renal insulin receptors by salt might have contributed to impaired natriuresis in fructose-fed rats maintained on a high-salt diet, including changes in the renin-angiotensin system, atrial natriuretic peptides, nitric oxide production, activity of renal nerves, and intrarenal hemodynamics. Particularly relevant in mediating insulin-induced sodium retention might be the role of angiotensin II. In fact, fructose hypertension has been demonstrated to be, at least in part, angiotensin II-dependent [] and inappropriate suppression of angiotensin II levels in fructose-fed rats maintained on a high-salt diet might contribute to persistence of insulin-induced renal sodium reabsorption. However, given our present demonstration of an effect of dietary salt mediated at the level of insulin receptor gene expression, this should not be the case. In fact, we have previously demonstrated that renal insulin receptor binding and mrna levels are relatively insensitive to chronic angiotensin II infusion in both normal and streptozotocin-diabetic rats [9], facts that clearly argue against a role for this peptide in the modulation of renal sensitivity to insulin. It is interesting to notice that the abnormal response of insulin receptor mrna to high-sodium was not uniquely found in the kidney but was present also in the muscle of fructose-fed rats. Although changes in mrna do not necessarily indicate changes in the transcription of the gene, this observation might suggest the presence of a ubiquitous defect in the transcriptional response of the gene to manipulation of dietary salt in these rats. This defect might achieve different pathophysiologic relevance depending on the functional role of the specific tissue and might explain why despite being insulin resistant, the fructose-fed rats fed a low-salt diet do not have hypertension. Although the functional role of the abnormal response of renal insulin receptors to dietary salt in fructose-fed rats remains to be clarified, it may be related to the issue of salt-dependence of fructose-induced hypertension. This issue has been investigated in previous studies and although Donnelly, Ho, and Reaven [3] reported that the blood pressure response to a fructose-enriched diet was not modified by either reducing salt intake or unilateral nephrectomy and Iyer and Katovich [31] showed that hypertension in fructose-fed rats does not occur directly via an increase in fluid volume, Johnson, Zhang, and Kotchen [] and, more recently, Nishimoto et al [7] demonstrated that blood pressure was not affected by high dietary carbohydrate when rats were maintained
8 17 Catena et al: Insulin and sodium in fructose-hypertension on a low-salt diet. The present study argues in favor of salt-dependence of fructose hypertension, inasmuch as hypertension did not occur in fructose-fed rats that were fed a low-salt diet. Broadly defined, insulin resistance refers to a subnormal response to a physiologic concentration of insulin, but this term is commonly used to describe an impairment of insulin-stimulated glucose disposal, as evaluated by the euglycemic-hyperinsulinemic clamp. In our study, insulinstimulated glucose disposal was significantly lower in fructose-fed than control rats, indicating insulin resistance in tissues involved in the metabolic actions of the hormone. Different cellular mechanisms can cause insulin resistance [3], including defects in insulin receptors, signal transduction pathways, and postreceptor defects at the level of substrates of phosphorylation or effector molecules such as glucose transporters and enzymes involved in glucose metabolism. Previous studies suggested that insulin resistance in fructose-fed rats is not due to a decrease in insulin-stimulated glucose uptake by skeletal muscle [33] and no changes in insulin receptor binding and tyrosine kinase activity were found in the liver of these rats [3]. Postreceptor defects, including decreased phosphorylation of insulin receptor substrate (IRS-1) and decreased association of IRS-1 with phosphatidylinositol 3-kinase and phosphotyrosine-phosphatase have been reported in more recent studies [35]. We have found that both insulin receptor number and mrna levels are significantly decreased in the skeletal muscle of fructose-fed rats fed low and normal-salt diet as compared to controls. This finding indicates a possible mechanism for insulin resistance in fructose-fed rats, reasonably explaining why insulin-induced glucose utilization is decreased in these rats when fed low and normal-salt diet. Our observation of salt-dependence of insulin-induced glucose utilization and muscular insulin receptor number and mrna levels in control rats is consistent with the findings of a recent study [3] in which rats fed an % NaCl diet showed decreased sensitivity to insulin in both skeletal muscle and liver. Similar association between salt and sensitivity to insulin has been reported in both normotensive and hypertensive humans independent of obesity [37]. In our study, insulin resistance in fructose-fed rats maintained on a low or normal-salt diet was limited to the skeletal muscle, where receptor number and mrna levels were significantly decreased, whereas in the kidney no difference was found in comparison to controls. Consistent with our data, comparable density of insulin receptors was found in all nephron segments of kidneys obtained from fructose-hypertensive and normotensive rats [3]. Different findings in muscle and kidney should not be surprising because it is known that resistance to the action of insulin may differ among different tissues. For instance, it has been shown that spontaneously hypertensive rats have a decreased sensitivity to insulin while they maintain the same antinatriuretic response to this hormone as their normotensive controls Wistar-Kyoto [, 39]. The present study confirms, in fructose-fed rats, the possibility of tissue-specific regulation of insulin receptors and suggests a possible mechanism to explain the different functional response to insulin in muscle and kidney. CONCLUSION This study identifies a possible mechanism to link insulin resistance to hypertension in rats fed a fructoseenriched diet and demonstrates tissue-specific regulation of insulin receptor in this model. The fructose-fed rats appear to have lost the capability to down-regulate insulin receptors in the kidney when dietary salt intake is chronically increased, leading to the persistence of an antinatriuretic effect of the hormone. This abnormality might possibly contribute to elevation of blood pressure in these rats. ACKNOWLEDGMENTS This research was supported by a Consiglio Nazionale delle Ricerche grant (9.3.CT) and a M.U.R.S.T. (Ministero dell Università e della Ricerca Scientifica) grant. L.A.S. was the recipient of a Ferrero Foundation grant. C.C. was the recipient of a fellowship from the Italian Society of Internal Medicine. Reprint requests to Leonardo A. Sechi, M.D., Clinica Medica, Hypertension Unit, DPMSC, University of Udine, Ospedale Civile, Padiglione Medicine, 331 Udine, Italy. Sechi@uniud.it REFERENCES 1. DENKER PS, POLLOCK VE. Fasting serum insulin levels in essential hypertension: A meta-analysis. Arch Int Med 15:19 151, 199. FERRANNINI E, BUZZIGOLI G, BONADONNA R, et al: Insulin resistance in essential hypertension. N Engl J Med 317:35 35, REAVEN GM: Insulin resistance, hyperinsulinemia, hypertriglyceridemia, and hypertension: Parallels between human disease and rodent models. Diabetes Care 1:195, SECHI LA, GRIFFIN CA, ZINGARO L, et al: Glucose metabolism and insulin receptor binding and mrna levels in tissues of Dahl hypertensive rats. Am J Hypertens 1:13 13, MONDON CE, REAVEN GM: Evidence of abnormalities of insulin metabolism in rats with spontaneous hypertension. Metabolism 37:33 35, 19. DALL AGLIO E, TOSINI P, FERRARI P, et al: Abnormalities of insulin and lipid metabolism in Milan hypertensive rats. Am J Hypertens : , HWANG I-S, HO H, HOFFMAN BB, REAVEN GM: -induced insulin resistance and hypertension in rats. Hypertension 1:51 51, 197. REAVEN GM, HO H, HOFFMAN BB: Attenuation of fructose-induced hypertension in rats by exercise training. Hypertension 1:19 13, VERMA S, BHANOT S, MCNEILL JH: Antihypertensive effects of metformin in fructose-fed hyperinsulinemic, hypertensive rats. J Pharmacol Exp Ther 71: , LEE MK, MILES PDG, KHOURSHEED M, et al: Metabolic effects of troglitazone on fructose-induced insulin resistance in the rat. Diabetes 3: , REAVEN GM, HO H, HOFFMAN BB: Somatostatin inhibition of fructose-induced hypertension. Hypertension 1:117 1, 199
9 Catena et al: Insulin and sodium in fructose-hypertension ROWE JW, YOUNG JB, MINAKER KL, et al: Effects of insulin and glucose infusions on sympathetic nervous activity in normal man. Diabetes 3:19 5, PFEILE B, DISCHANEIT H: Effects of insulin on growth of cultured arterial smooth muscle cells. Diabetologia :155 15, PERSHADSINGH HA, MCDONALD JM: Direct addition of insulin inhibits a high affinity Ca+-ATPase in isolated adipocyte plasma membranes. Nature 1:95 97, DE FRONZO RA : The effect of insulin on renal sodium metabolism. A review with clinical implications. Diabetologia 1:11 171, BAUM M: Insulin stimulates volume absorption in the rabbit proximal convoluted tubule. J Clin Invest 79:11 119, DE FRONZO RA, GOLDBERG M, AGUS Z: The effects of glucose and insulin on renal electrolyte transport. J Clin Invest 5:3 9, KIRCHNER KA: Insulin increases loop segment chloride reabsorption in the euglycemic rat. Am J Physiol 55:F1 F113, SECHI LA, GRIFFIN CA, SCHAMBELAN M: Effect of dietary sodium chloride on insulin receptor number and mrna levels in the kidney of the rat. Am J Physiol :F31 F3, 199. SECHI LA, GRIFFIN CA, GIACCHETTI G, et al: Abnormalities of insulin receptors in spontaneously hypertensive rats. Hypertension 7:955 91, GIACCHETTI G, SECHI LA, GRIFFIN CA, et al: The renin-angiotensin system in rats with fructose-induced hypertension. J Hypertens 1:95 7,. VALENTIN JP, SECHI LA, HUMPHREYS MH: Blunted effect of ANP on hematocrit and plasma volume in streptozotocin-induced diabetes mellitus in rats. Am J Physiol :R5 R591, BRANDS MW, GARRITY CA, HOLMAN MG, et al: High-fructose diet does not raise -hour mean arterial pressure in rats. Am J Hypertens 7:1 19, 199. FERRARI AU, DAFFONCHIO A, ALBERGATI F, et al: Intra-arterial pressure alterations during tail-cuff blood pressure measurements in normotensive and hypertensive rats. J Hypertens :99 911, PREUSS MB, PREUSS HG: The effect of sucrose and sodium on blood pressure in various substrains of Wistar rats. Lab Invest 13:11 17, 19. JOHNSON MD, ZHANG HY, KOTCHEN TA: Sucrose does not raise blood pressure in rats maintained on a low salt intake. Hypertension 1:779 75, NISHIMOTO Y, TOMIDA T, MATSUI H, et al: Decrease in renal medullary endothelial nitric oxide synthase of fructose-fed, saltsensitive hypertensive rats. Hypertension :19 19,. KOBAYASHI R, NAGANO M, NAKAMURA F, et al: Role of angiotensin II in high fructose induced left ventricular hypertrophy in rats. Hypertension 1: , SECHI LA, GRIFFIN CA, ZINGARO L, et al: Effects of angiotensin II on insulin receptor binding and mrna levles in normal and diabetic rats. Diabetologia :77 777, DONNELLY R, HO H, REAVEN GM: Effects of low sodium diet and unilateral nephrectomy on the development of carbohydrateinduced hypertension. Blood pressure :1 19, IYER SN, KATOVICH MJ: feeding in rats is not associated with sodium retention. Am J Hypertens 9:11 13, SECHI LA, BARTOLI E: Mechanisms of insulin resistance leading to hypertension: What we can learn from experimental models. J Invest Med 5:3 51, TOBEY TA, MONDON CE, ZAVARONI I, REAVEN GM: Mechanisms of insulin resistance in fructose-fed rats. Metabolism 31: 1, DEUTSCH DD, JEN KL, GRUNBERGER G: Regulation of hepatic insulin receptor tyrosine kinase in rat models of mild insulin resistance. J Lab Clin Med 1:1 5, BEZERRA RM, UENO M, SILVA MS, et al: A high fructose diet affects the early steps of insulin action in muscle and liver of rats. J Nutr 13: , 3. OGIHARA T, ASANO T, ANDO K, et al: Insulin resistance with enhanced insulin signaling in high-salt diet-fed rats. Diabetes 5:573 53, ROCCHINI AP: The relationship of sodium sensitivity to insulin resistance. Am J Med Sci 37(Suppl 1):S75 S, FERAILLE E, MARSHY S, BARLET-BAS C, et al: Insulin unresponsiveness of tubular monovalent cation transport during fructoseinduced hypertension in rats. Clin Sci :93 99, FINCH D, DAVIS G, BOWER J, KIRCHNER K: Effect of insulin on renal sodium handling in hypertensive rats. Hypertension 15:51 51, 199
Effect of Insulin on Renal Sodium Handling in Hypertensive Rats. David Finch, Gary Davis, John Bower, and Kent Kirchner
 514 Effect of Insulin on Renal Sodium Handling in Hypertensive Rats David Finch, Gary Davis, John Bower, and Kent Kirchner Spontaneously hypertensive rats have reduced peripheral insulin sensitivity. To
514 Effect of Insulin on Renal Sodium Handling in Hypertensive Rats David Finch, Gary Davis, John Bower, and Kent Kirchner Spontaneously hypertensive rats have reduced peripheral insulin sensitivity. To
Salt Sensitivity: Mechanisms, Diagnosis, and Clinical Relevance
 Salt Sensitivity: Mechanisms, Diagnosis, and Clinical Relevance Matthew R. Weir, MD Professor and Director Division of Nephrology University of Maryland School of Medicine Overview Introduction Mechanisms
Salt Sensitivity: Mechanisms, Diagnosis, and Clinical Relevance Matthew R. Weir, MD Professor and Director Division of Nephrology University of Maryland School of Medicine Overview Introduction Mechanisms
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
Outline Urinary System
 Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
Effects of angiotensin II on insulin receptor binding and mrna levels in normal and diabetic rats
 Diabetologia (1997) 4: 77 777 Springer-Verlag 1997 Effects of angiotensin II on insulin receptor binding and mrna levels in normal and diabetic rats L.A. Sechi 1,2, C. A. Griffin 2, L. Zingaro 1, J.-P.Valentin
Diabetologia (1997) 4: 77 777 Springer-Verlag 1997 Effects of angiotensin II on insulin receptor binding and mrna levels in normal and diabetic rats L.A. Sechi 1,2, C. A. Griffin 2, L. Zingaro 1, J.-P.Valentin
Sunday, July 17, 2011 URINARY SYSTEM
 URINARY SYSTEM URINARY SYSTEM Let s take a look at the anatomy first! KIDNEYS: are complex reprocessing centers where blood is filtered through and waste products are removed. Wastes and extra water become
URINARY SYSTEM URINARY SYSTEM Let s take a look at the anatomy first! KIDNEYS: are complex reprocessing centers where blood is filtered through and waste products are removed. Wastes and extra water become
Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate
 Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
A high-fructose diet induces changes in pp185 phosphorylation in muscle and liver of rats
 Fructose Brazilian diet Journal induces of Medical changes and in Biological rat pp185 Research () 33: 1421-1427 ISSN -879X Short Communication 1421 A high-fructose diet induces changes in pp185 phosphorylation
Fructose Brazilian diet Journal induces of Medical changes and in Biological rat pp185 Research () 33: 1421-1427 ISSN -879X Short Communication 1421 A high-fructose diet induces changes in pp185 phosphorylation
WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure
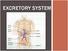 EXCRETORY SYSTEM WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure These wastes include: Carbon dioxide Mostly through breathing
EXCRETORY SYSTEM WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure These wastes include: Carbon dioxide Mostly through breathing
Outline Urinary System. Urinary System and Excretion. Urine. Urinary System. I. Function II. Organs of the urinary system
 Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Urinary System and Excretion. Bio105 Lecture 20 Chapter 16
 Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
2) This is a Point and Click question. You must click on the required structure.
 Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Role of Minerals in Hypertension
 Role of Minerals in Hypertension Lecture objectives By the end of the lecture students will be able to Define primary and secondary hypertention and their risk factors. Relate role of minerals with hypertention.
Role of Minerals in Hypertension Lecture objectives By the end of the lecture students will be able to Define primary and secondary hypertention and their risk factors. Relate role of minerals with hypertention.
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Regulation of Sodium and Volume. Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM
 Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin
![KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin](/thumbs/87/96174332.jpg) Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
The ability of the kidneys to regulate extracellular fluid volume by altering sodium
 REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
Human Urogenital System 26-1
 Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Renal System and Excretion
 Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
Ch17-18 Urinary System
 Ch17-18 Urinary System Main Function: Filter the blood Other Functions: maintain purity and consistency of internal fluids eliminates nitrogenous wastes, toxins, and drugs from the body regulates blood
Ch17-18 Urinary System Main Function: Filter the blood Other Functions: maintain purity and consistency of internal fluids eliminates nitrogenous wastes, toxins, and drugs from the body regulates blood
Functions of the Urinary System
 The Urinary System Functions of the Urinary System Elimination of waste products Nitrogenous wastes Toxins Drugs Regulate aspects of homeostasis Water balance Electrolytes Acid-base balance in the blood
The Urinary System Functions of the Urinary System Elimination of waste products Nitrogenous wastes Toxins Drugs Regulate aspects of homeostasis Water balance Electrolytes Acid-base balance in the blood
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
The Urinary System. Copyright 2003 Pearson Education, Inc. publishing as Benjamin Cummings
 The Urinary System Functions of the Urinary System Elimination of waste products Nitrogenous wastes Toxins Drugs Functions of the Urinary System Regulate aspects of homeostasis Water balance Electrolytes
The Urinary System Functions of the Urinary System Elimination of waste products Nitrogenous wastes Toxins Drugs Functions of the Urinary System Regulate aspects of homeostasis Water balance Electrolytes
April 08, biology 2201 ch 11.3 excretion.notebook. Biology The Excretory System. Apr 13 9:14 PM EXCRETORY SYSTEM.
 Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Kidneys and Homeostasis
 16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
Renal-Related Questions
 Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
PARTS OF THE URINARY SYSTEM
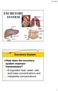 EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
Biology Slide 1 of 36
 Biology 1 of 36 38 3 The Excretory System 2 of 36 Functions of the Excretory System 1.Function: process which eliminates metabolic wastes 3 of 36 Functions of the Excretory System (The skin excretes excess
Biology 1 of 36 38 3 The Excretory System 2 of 36 Functions of the Excretory System 1.Function: process which eliminates metabolic wastes 3 of 36 Functions of the Excretory System (The skin excretes excess
NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System)
 NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System) HOMEOSTASIS **Recall HOMEOSTASIS is the steady-state physiological condition of the body. It includes: 1) Thermoregulation:
NOTES: CH 44 Regulating the Internal Environment (Homeostasis & The Urinary System) HOMEOSTASIS **Recall HOMEOSTASIS is the steady-state physiological condition of the body. It includes: 1) Thermoregulation:
The Urinary System. BIOLOGY OF HUMANS Concepts, Applications, and Issues. Judith Goodenough Betty McGuire
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
Kidney and urine formation
 Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
organs of the urinary system
 organs of the urinary system Kidneys (2) bean-shaped, fist-sized organ where urine is formed. Lie on either sides of the vertebral column, in a depression beneath peritoneum and protected by lower ribs
organs of the urinary system Kidneys (2) bean-shaped, fist-sized organ where urine is formed. Lie on either sides of the vertebral column, in a depression beneath peritoneum and protected by lower ribs
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D.
 Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Chapter 11 Lecture Outline
 Chapter 11 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. Permission required for reproduction
Chapter 11 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. Permission required for reproduction
28/04/2013 LEARNING OUTCOME C13 URINARY SYSTEM STUDENT ACHIEVEMENT INDICATORS STUDENT ACHIEVEMENT INDICATORS URINARY SYSTEM & EXCRETION
 LEARNING OUTCOME C13 Analyse the functional interrelationships of the structures of the urinary system Learning Outcome C13 URINARY SYSTEM STUDENT ACHIEVEMENT INDICATORS Students who have fully met this
LEARNING OUTCOME C13 Analyse the functional interrelationships of the structures of the urinary system Learning Outcome C13 URINARY SYSTEM STUDENT ACHIEVEMENT INDICATORS Students who have fully met this
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
19. RENAL PHYSIOLOGY ROLE OF THE URINARY SYSTEM THE URINARY SYSTEM. Components and function. V BS 122 Physiology II 151 Class of 2011
 19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
The Urinary System 15PART A. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART A Functions of the Urinary System Elimination of waste products Nitrogenous
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART A Functions of the Urinary System Elimination of waste products Nitrogenous
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
I. Metabolic Wastes Metabolic Waste:
 I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
EFFECT OF ACUTE SALINE LOADING ON THE ERYTH ROCYTE SODIUM TRANSPORT IN CHILDREN
 Acta Medica et Biologica Vol. 36, No. 109-113, 1989 EFFECT OF ACUTE SALINE LOADING ON THE ERYTH ROCYTE SODIUM TRANSPORT IN CHILDREN M. UCHIYAMA I. SATOKATA T. AIKAWA K. SAKAI Department 0/ pediatrics,
Acta Medica et Biologica Vol. 36, No. 109-113, 1989 EFFECT OF ACUTE SALINE LOADING ON THE ERYTH ROCYTE SODIUM TRANSPORT IN CHILDREN M. UCHIYAMA I. SATOKATA T. AIKAWA K. SAKAI Department 0/ pediatrics,
Interrelationship between Angiotensin Catecholamines. Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D.
 Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
NIH Public Access Author Manuscript Kidney Int. Author manuscript; available in PMC 2013 November 01.
 NIH Public Access Author Manuscript Published in final edited form as: Kidney Int. 2013 May ; 83(5): 779 782. doi:10.1038/ki.2012.468. Need to quickly excrete K +? Turn off NCC Alicia A. McDonough 1 and
NIH Public Access Author Manuscript Published in final edited form as: Kidney Int. 2013 May ; 83(5): 779 782. doi:10.1038/ki.2012.468. Need to quickly excrete K +? Turn off NCC Alicia A. McDonough 1 and
Urinary system. Lab-7
 Urinary system Lab-7 Excretion: processes that remove wastes and excess materials from the body Urinary system (kidneys): excretes nitrogenous wastes, excess solutes, and water The Kidneys Regulate Water
Urinary system Lab-7 Excretion: processes that remove wastes and excess materials from the body Urinary system (kidneys): excretes nitrogenous wastes, excess solutes, and water The Kidneys Regulate Water
5. Maintaining the internal environment. Homeostasis
 5. Maintaining the internal environment Homeostasis Blood and tissue fluid derived from blood, flow around or close to all cells in the body. Blood and tissue fluid form the internal environment of the
5. Maintaining the internal environment Homeostasis Blood and tissue fluid derived from blood, flow around or close to all cells in the body. Blood and tissue fluid form the internal environment of the
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Renal Functions: Renal Functions: Renal Function: Produce Urine
 Renal Functions: Excrete metabolic waste products Reabsorb vital nutrients Regulate osmolarity: Maintain ion balance Regulate extracellular fluid volume (and thus blood pressure) Renal Functions: Regulate
Renal Functions: Excrete metabolic waste products Reabsorb vital nutrients Regulate osmolarity: Maintain ion balance Regulate extracellular fluid volume (and thus blood pressure) Renal Functions: Regulate
Use the following diagram to answer the next question. 1. In the diagram above, pressure filtration occurs in a. W b. X c. Y d. Z
 Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Regulating the Internal Environment. AP Biology
 Regulating the Internal Environment 2006-2007 Conformers vs. Regulators Two evolutionary paths for organisms regulate internal environment maintain relatively constant internal conditions conform to external
Regulating the Internal Environment 2006-2007 Conformers vs. Regulators Two evolutionary paths for organisms regulate internal environment maintain relatively constant internal conditions conform to external
November 30, 2016 & URINE FORMATION
 & URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
& URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
Moderate (20%) fructose-enriched diet stimulates saltsensitive hypertension with increased salt retention and decreased renal nitric oxide
 ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X Moderate (20%) fructose-enriched diet stimulates saltsensitive hypertension with increased salt retention and decreased renal nitric oxide Kevin L.
ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X Moderate (20%) fructose-enriched diet stimulates saltsensitive hypertension with increased salt retention and decreased renal nitric oxide Kevin L.
Class XI Chapter 19 Excretory Products and their Elimination Biology
 Class XI Chapter 19 Excretory Products and their Elimination Biology Question 1: Define Glomerular Filtration Rate (GFR) Glomerular filtration rate is the amount of glomerular filtrate formed in all the
Class XI Chapter 19 Excretory Products and their Elimination Biology Question 1: Define Glomerular Filtration Rate (GFR) Glomerular filtration rate is the amount of glomerular filtrate formed in all the
5.Which part of the nephron removes water, ions and nutrients from the blood?
 Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
CIE Biology A-level Topic 14: Homeostasis
 CIE Biology A-level Topic 14: Homeostasis Notes Communication is essential for the survival of organism as all living organisms must be able to detect and respond to changes in both their internal and
CIE Biology A-level Topic 14: Homeostasis Notes Communication is essential for the survival of organism as all living organisms must be able to detect and respond to changes in both their internal and
General Anatomy of Urinary System
 General Anatomy of Urinary System URINARY SYSTEM ORGANS Kidneys (2) Ureters (2) Urinary bladder Urethra KIDNEY FUNCTIONS Control blood volume and composition KIDNEY FUNCTIONS Filter blood plasma, eliminate
General Anatomy of Urinary System URINARY SYSTEM ORGANS Kidneys (2) Ureters (2) Urinary bladder Urethra KIDNEY FUNCTIONS Control blood volume and composition KIDNEY FUNCTIONS Filter blood plasma, eliminate
AJH 1998;11: by the American Journal of Hypertension, Ltd /98/$19.00
 AJH 1998;11:8 13 Acute Effects of Intravenous Sodium Chloride Load on Calcium Metabolism and on Parathyroid Function in Patients With Primary Aldosteronism Compared With Subjects With Essential Hypertension
AJH 1998;11:8 13 Acute Effects of Intravenous Sodium Chloride Load on Calcium Metabolism and on Parathyroid Function in Patients With Primary Aldosteronism Compared With Subjects With Essential Hypertension
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Excretion and Waste Management. Biology 30S - Miss Paslawski
 Excretion and Waste Management Biology 30S - Miss Paslawski Lesson 1 Waste Products and Organs 2 3 Excretion Excretion: Process by which dissolved metabolic wastes are separated from body fluids and removed
Excretion and Waste Management Biology 30S - Miss Paslawski Lesson 1 Waste Products and Organs 2 3 Excretion Excretion: Process by which dissolved metabolic wastes are separated from body fluids and removed
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1
 BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
A. Incorrect! The urinary system is involved in the regulation of blood ph. B. Correct! The urinary system is involved in the synthesis of vitamin D.
 Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
1. Urinary System, General
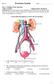
 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
Urinary system. Urinary system
 INTRODUCTION. Several organs system Produce urine and excrete it from the body Maintenance of homeostasis. Components. two kidneys, produce urine; two ureters, carry urine to single urinary bladder for
INTRODUCTION. Several organs system Produce urine and excrete it from the body Maintenance of homeostasis. Components. two kidneys, produce urine; two ureters, carry urine to single urinary bladder for
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Urinary System. Analyze the Anatomy and Physiology of the urinary system
 Urinary System Analyze the Anatomy and Physiology of the urinary system Kidney Bean-shaped Located between peritoneum and the back muscles (retroperitoneal) Renal pelvis funnelshaped structure at the beginning
Urinary System Analyze the Anatomy and Physiology of the urinary system Kidney Bean-shaped Located between peritoneum and the back muscles (retroperitoneal) Renal pelvis funnelshaped structure at the beginning
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
What is excretion? Excretion is the removal of metabolic waste from the body.
 Excretion What is excretion? Excretion is the removal of metabolic waste from the body. Excretion in Plants Plants produce very little waste products. Plants lose oxygen and water vapour through the stomata.
Excretion What is excretion? Excretion is the removal of metabolic waste from the body. Excretion in Plants Plants produce very little waste products. Plants lose oxygen and water vapour through the stomata.
Nephron Anatomy Nephron Anatomy
 Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
BIOLOGY - CLUTCH CH.44 - OSMOREGULATION AND EXCRETION.
 !! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
!! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
Cornerstone: A Collection of Scholarly and Creative Works for Minnesota State University, Mankato. Minnesota State University, Mankato
 Minnesota State University, Mankato Cornerstone: A Collection of Scholarly and Creative Works for Minnesota State University, Mankato Theses, Dissertations, and Other Capstone Projects 2009 The Increase
Minnesota State University, Mankato Cornerstone: A Collection of Scholarly and Creative Works for Minnesota State University, Mankato Theses, Dissertations, and Other Capstone Projects 2009 The Increase
Chapter 23. The Nephron. (functional unit of the kidney
 Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
A&P of the Urinary System
 A&P of the Urinary System Week 44 1 Objectives Identify the organs of the urinary system, from a Identify the parts of the nephron (the functional unit List the characteristics of a normal urine specimen.
A&P of the Urinary System Week 44 1 Objectives Identify the organs of the urinary system, from a Identify the parts of the nephron (the functional unit List the characteristics of a normal urine specimen.
renoprotection therapy goals 208, 209
 Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Toxic Waste?! So now we ve eaten we have to get rid of the waste!
 Toxic Waste?! So now we ve eaten we have to get rid of the waste! Excretory/Urinary System Notes Continuation of digestion food is transferred into nutrients and passes through the body as waste 7.7B transformation
Toxic Waste?! So now we ve eaten we have to get rid of the waste! Excretory/Urinary System Notes Continuation of digestion food is transferred into nutrients and passes through the body as waste 7.7B transformation
Renal Physiology - Lectures
 Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus
 Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Structures of the Excretory System include: ü Skin ü Lung ü Liver ü Kidneys ü Ureter ü Urinary Bladder ü Urethra
 Excretory System Structures of the Excretory System include: ü Skin ü Lung ü Liver ü Kidneys ü Ureter ü Urinary Bladder ü Urethra Function of the Excretory System The function of the excretory system is
Excretory System Structures of the Excretory System include: ü Skin ü Lung ü Liver ü Kidneys ü Ureter ü Urinary Bladder ü Urethra Function of the Excretory System The function of the excretory system is
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate
 Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
(Received 22 July 1957) It is now generally accepted that the unequal distribution of ions between cells
 190 J. Physiol. (I958) I40, I90-200 THE EFFECT OF ALTERATIONS OF PLASMA SODIUM ON THE SODIUM AND POTASSIUM CONTENT OF MUSCLE IN THE RAT By F. 0. DOSEKUN AND D. MENDEL From the Department of Physiology,
190 J. Physiol. (I958) I40, I90-200 THE EFFECT OF ALTERATIONS OF PLASMA SODIUM ON THE SODIUM AND POTASSIUM CONTENT OF MUSCLE IN THE RAT By F. 0. DOSEKUN AND D. MENDEL From the Department of Physiology,
Refer to the figure below, a diagram of a renal tubule, to answer the following questions.
 1. The digestion and utilization of which nutrient creates the greatest need for osmoregulation by the kidneys? a. protein b. starch c. fat d. oil e. cellulose 2. Which of the following is true of urea?
1. The digestion and utilization of which nutrient creates the greatest need for osmoregulation by the kidneys? a. protein b. starch c. fat d. oil e. cellulose 2. Which of the following is true of urea?
The Excretory System. Biology 20
 The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
P215 Spring 2018: Renal Physiology Chapter 18: pp , Chapter 19: pp ,
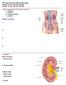 P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion.
 The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
BLOCK REVIEW Renal Physiology. May 9, 2011 Koeppen & Stanton. EXAM May 12, Tubular Epithelium
 BLOCK REVIEW Renal Physiology Lisa M. HarrisonBernard, Ph.D. May 9, 2011 Koeppen & Stanton EXAM May 12, 2011 Tubular Epithelium Reabsorption Secretion 1 1. 20, 40, 60 rule for body fluid volumes 2. ECF
BLOCK REVIEW Renal Physiology Lisa M. HarrisonBernard, Ph.D. May 9, 2011 Koeppen & Stanton EXAM May 12, 2011 Tubular Epithelium Reabsorption Secretion 1 1. 20, 40, 60 rule for body fluid volumes 2. ECF
A. Correct! Flushing acids from the system will assist in re-establishing the acid-base equilibrium in the blood.
 OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
Ch. 44 Regulating the Internal Environment
 Ch. 44 Regulating the Internal Environment 2006-2007 Conformers vs. Regulators Two evolutionary paths for organisms regulate internal environment maintain relatively constant internal conditions conform
Ch. 44 Regulating the Internal Environment 2006-2007 Conformers vs. Regulators Two evolutionary paths for organisms regulate internal environment maintain relatively constant internal conditions conform
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Excretory System. Biology 2201
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System. Excretory System
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
