Regulation of Fat Cell Mass by Insulin in Drosophila melanogaster
|
|
|
- Malcolm Randall
- 5 years ago
- Views:
Transcription
1 MOLECULAR AND CELLULAR BIOLOGY, Dec. 2009, p Vol. 29, No /09/$12.00 doi: /mcb Copyright 2009, American Society for Microbiology. All Rights Reserved. Regulation of Fat Cell Mass by Insulin in Drosophila melanogaster Justin R. DiAngelo and Morris J. Birnbaum* The Institute for Diabetes, Obesity and Metabolism, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania Received 26 May 2009/Returned for modification 20 July 2009/Accepted 30 September 2009 A phylogenetically conserved response to nutritional abundance is an increase in insulin signaling, which initiates a set of biological responses dependent on the species. Consequences of augmented insulin signaling include developmental progression, cell and organ growth, and the storage of carbohydrates and lipids. Here, we address the evolutionary origins of insulin s positive effects on anabolic lipid metabolism by selectively modulating insulin signaling in the fat body of the fruit fly, Drosophila melanogaster. Analogous to the actions of insulin in higher vertebrates, those in Drosophila include expansion of the insect fat cell mass both by increasing the adipocyte number and by promoting lipid accumulation. The ability of insulin to accomplish the former depends on its capacity to bring about phosphorylation and inhibition of the transcription factor Drosophila FOXO (dfoxo) and the serine/threonine protein kinase shaggy, the fly ortholog of glycogen synthase kinase 3 (GSK3). Increasing the amount of triglyceride per cell also depends on the phosphorylation of shaggy but is independent of dfoxo. Thus, the findings of this study provide evidence that the control of fat mass by insulin is a conserved process and place dfoxo and shaggy/gsk3 downstream of the insulin receptor in controlling adipocyte cell number and triglyceride storage, respectively. * Corresponding author. Mailing address: 322 Clinical Research Building, 415 Curie Blvd., Philadelphia, PA Phone: (215) Fax: (215) birnbaum@mail.med.upenn.edu. Published ahead of print on 12 October The recent epidemic of obesity, insulin resistance, and type 2 diabetes mellitus (T2DM) has renewed interest in fundamental principles of metabolic homeostasis. Much attention has been directed to the appropriate control of carbohydrate metabolism, as elevated glucose is the cardinal, defining sign of diabetes mellitus. Nonetheless, an integrated and finely tuned balance of lipid storage and breakdown is also essential for metabolic well-being. Defects in lipid metabolism not only contribute directly to a number of diseases, such as the inherited lipodystrophies, the metabolic syndrome, and atherosclerosis, but also are an integral component of T2DM. In fact, it has been argued persuasively that T2DM is regarded as a disorder of glucose metabolism for purely historical reasons and that lipid abnormalities are as or more important to the pathogenesis and consequences of the disease (33). Thus, a refined, mechanistic understanding of the normal control of lipid storage and mobilization is critical. The major regulators of lipid synthesis and breakdown are glucose and the peptide hormone insulin. It is now clear that insulin signals through a canonical pathway that is phylogenetically conserved, controlling growth, development, and ageing in invertebrates (Caenorhabditis elegans and Drosophila melanogaster) and metabolic outputs in vertebrates (rodents and humans) (17, 37). For example, in mammals, insulin regulates hepatic, muscle, and adipocyte glucose metabolism in a manner dependent on signaling through phosphatidylinositol 3-kinase (PI3K) and its downstream intermediary, Akt, also known as protein kinase B (12, 44). This same pathway has also been implicated in insulin s action in the central nervous system to modulate carbohydrate metabolism and energy balance (39). Much less clarity exists concerning the relevant insulin-signaling pathway(s) in the regulation of lipid metabolism, particularly that occurring in liver. Although there is consensus that insulin promotes triglyceride synthesis in liver by augmenting the expression and activity of the lipogenic transcription factor sterol response element binding protein 1c (SREBP1c), data regarding additional relevant signaling pathways have been conflicting (46). For example, there is support for either atypical protein kinase C or Akt as the major transducer of prolipogenic signals, and both FoxO1 and FoxA2 have been suggested as major alternative relevant transcription factors in addition to SREBP1c (31, 32, 50, 57, 59). Even more so than hepatocytes, adipocytes are capable of varying their levels of stored neutral lipid over a wide range by altering synthesis, accumulation, oxidation, and mobilization. The total fat mass contained within adipose tissue is also subject to regulation of the number of fat cells. Whereas there is substantial understanding of the signaling pathways governing differentiation into fat cells, the key factors determining the number of cells representing the precursor stem cell pool are not known. Nonetheless, insulin and/or insulin-like growth factor 1 is an important extracellular agent influencing adipogenesis (41). The utility of D. melanogaster as a relevant model system with which to investigate and understand components of human biology, including the insulin-signaling pathway, is now well established (3, 45). Using the fly, considerable recent effort has been directed toward both defining the physiological role of insulin and delineating its downstream signaling pathways that control cell size and number as well as longevity (17, 51). A model has emerged in which insulin serves as a phylogenetically conserved nutritional sensor, in invertebrates controlling such primitive functions as growth and development but in vertebrates having evolved and broadened to be the prime regulator of anabolic functions, such as the synthesis and redistribution of simple nutrients into long-term energy stores. It is also likely that insulin exerts influence on the control of Drosophila metabolism (3). For example, flies deficient in insulin-like peptides display increases in extracellular reducing sugars that can be abolished by ectopic expression of an insu- 6341
2 6342 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. lin-like peptide gene (43). However, less attention has been given to the roles of these same molecules in lipid metabolism. Flies with a loss of insulin-like peptide-producing cells or mutations in the gene for Drosophila insulin receptor (dinr) or the scaffold protein chico (the Drosophila ortholog of insulin receptor substrate) have a significant increase in triglyceride (4, 8, 52). While reminiscent of the human obesity that predisposes to insulin-resistant states, an increase in total triglyceride is the opposite of the typical consequence of insulin-deficient states in mice and humans (44). This has called into question the relevance of Drosophila as a model for mammalian lipid metabolism. Nonetheless, a number of proteins and pathways integral to the control of lipid metabolism in higher organisms have homologs in flies, in some cases with demonstrably comparable functions. For example, the Drosophila genome encodes both a lipid binding protein and a triglyceride lipase quite similar to proteins expressed in human adipocytes and critical to hormone-regulated lipolysis (20, 21). Moreover, these enzymes appear to participate in the control of lipid storage in flies in a manner analogous to that in mammals (22). Like the human liver, the Drosophila fat body packages lipids into lipoprotein particles for transport to peripheral tissues (10). Thus, lipid metabolism in insects may be more similar to that in mammals than once imagined. It is possible that the accumulation of lipid in flies deficient in insulin signaling may be misleading, possibly as a result of nonautonomous effects on energy metabolism. Consistent with this idea, larval fat body clones overexpressing dinr or the catalytic subunit of PI3K (Dp110) appear to accumulate more fat than controls, although this has been ascertained only indirectly by light or electron microscopy (7). In mice, deletion of the gene encoding insulin receptor substrate 2, which is homologous to chico, results in increased fat mass; this effect is thought to be secondary to a nonautonomous effect of hypothalamic insulin signaling on energy balance (9). In this study, we address the issue of control of lipid metabolism in Drosophila, specifically considering the role of the insulin-signaling pathway in the fly s main nutrient storage organ, the fat body. We find that in flies, as in mammals, insulin is a positive regulator of fat cell mass, acting through changes in both cell number and lipid storage. We further go on to utilize Drosophila genetics to dissect downstream signaling and show distinct pathways by which insulin regulates the number of fat cells versus their triglyceride content. MATERIALS AND METHODS Fly genetics. The following fly strains were used in this study: UASdInR A1325D (Bloomington 8263), UAS-GFP (Bloomington 5194), yolk-gal4 (18), to-gal4 (14), UAS-dTOR TED (24), UAS-dFOXO-TM (27), UAS-sggS9A (5), and UAS-RBF (15). Flies were grown on standard cornmeal-dextrose medium (128.4 g dextrose, 9.3 g agar, 61.2 g cornmeal, and 32.4 g Lake States dried Torula type b yeast per liter) supplemented with dry yeast (Sigma, St. Louis, MO), and crosses were performed at 25 C. To control for effects of the yolk-gal4/takeout (to)-gal4 driver, flies carrying each driver expressing green fluorescent protein (GFP) were compared to those carrying the driver expressing the given experimental transgene. To control for the genetic background and effects of the transgene alone, yolk-gal4/fm7 or to-gal4/cyo virgin females were crossed with males carrying the given experimental transgene and the resulting progeny carrying the driver and transgene were compared to their siblings carrying the transgene alone. To control for the genetic background in experiments including flies with more than one transgene, yolk-gal4/fm7; sternopleural/ virgin females were crossed with UAS-RBF, UAS-dTOR TED, UAS-dFOXO-TM, or UAS-sggS9A males. Yolk- Gal4/y; sternopleural/transgene males were then crossed with UAS-dInR A1325D virgin females, and the resulting siblings were analyzed. Control animals were cultured in the same vials as the experimental animals to normalize for larval crowding. Protein, triglyceride, and DNA assays. Adult virgin female flies aged 4 to 5 days, males aged 7 to 10 days, or fat body-enriched preparations of tissues dissected from these animals, including the fat body cells that remained attached to the cuticles, were homogenized in lysis buffer containing 140 mm NaCl, 50 mm Tris-HCl, ph 7.4, 0.1% Triton X, and 1 protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany). Protein concentrations were measured using the bicinchoninic acid protein assay kit (Pierce, Rockford, IL), triglyceride concentrations were measured using the triglyceride LiquiColor kit (Stanbio Laboratory, Boerne, TX), and the total DNA content was measured using the Quant-iT double-stranded DNA high-sensitivity assay kit (Molecular Probes, Eugene, OR) according to the manufacturers instructions. Each experiment was performed at least three times, and error bars in the figures represent standard errors of the means (SEM). Nile Red staining and cell counting. For Nile Red staining, a 10% stock solution of Nile Red (Sigma-Aldrich, St. Louis, MO) in dimethyl sulfoxide was diluted 1:5,000 in a mixture of 1 phosphate-buffered saline, 30% glycerol, and 2 g/ml DAPI (4,6-diamidino-2-phenylindole; Roche Molecular Biochemicals, Indianapolis, IN). Four to six adult virgin female flies aged 4 to 5 days were dissected into this working solution, and cuticles with fat body cells attached were mounted whole and visualized using a Leica DM IRE2 confocal microscope. For each genotype, three to seven images of multiple fat bodies throughout the slide were taken randomly, and quantitation was performed with each image by using ImageJ 1.38x software (National Institutes of Health). For cell counting experiments, fat body cells attached to the cuticle were labeled with GFP genetically by expressing GFP under the control of the yolk-gal4 driver and the cells were dissociated from one another and the cuticle in 0.5% trypsin-edta for 4 h with rocking. After 4 h, almost all of the dissociated cells were GFP positive. The majority of the GFP signal was found in the dissociated cells, with very little GFP found attached to the cuticle, suggesting that most of the fat body cells had been dissociated from one another and the cuticle at this time point. Cell numbers were determined by counting the total number of dissociated cells with a hemocytometer. Antibodies. The phospho-drosophila Akt (phospho-dakt) and total dakt antibodies were purchased from Cell Signaling Technology, Inc. (Beverly, MA), and used at a 1:1,000 dilution. The E7 beta-tubulin antibody from Michael Klymkowsky was obtained from the Developmental Studies Hybridoma Bank maintained by the Department of Biological Sciences, University of Iowa, Iowa City. Horseradish peroxidase-conjugated secondary antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Immunoblot analysis. Four- to five-day-old virgin female flies were dissected into phosphate-buffered saline, and fat body-enriched preparations of the cuticles with the fat body cells that remained attached were homogenized in lysis buffer containing 2% sodium dodecyl sulfate, 60 mm Tris-HCl, ph 6.8, 1 protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany), and 1 phosphatase inhibitor cocktail 1 (Sigma, St. Louis, MO). The lysates were then centrifuged for 15 min at 13,000 rpm and 4 C. Protein concentrations were determined using the bicinchoninic acid protein assay kit (Pierce, Rockford, IL). Equal amounts of protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto nitrocellulose membranes (Whatman, Florham Park, NJ). Detection was performed using ECL reagents according to the instructions supplied by the manufacturer (Amersham Pharmacia Biotech). Each experiment was performed at least three times, and a representative blot is shown (see Fig. 4). Statistical analysis. Unpaired Student s t tests were used for statistical analysis. RESULTS Insulin signaling promotes triglyceride storage in the Drosophila fat body. In order to understand the role of insulin in regulating triglyceride storage, we took a tissue-specific approach to manipulate insulin signaling in the Drosophila fat body, the major triglyceride storage organ of the fly. The insect fat body is an organ functionally analogous to the mammalian liver and adipose tissue and is present during both the larval and adult stages of development. The larval fat body stores
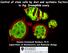
3 VOL. 29, 2009 INSULIN ACTION ON FAT MASS IN DROSOPHILA 6343 Downloaded from FIG. 1. Insulin signaling regulates lipid storage in the adult fat body. (A and B) Triglyceride/protein ratios in yolk-gal4 dinr A1325D 4- to 5-day-old adult virgin females compared to those in yolk-gal4 GFP (A) and UAS-dInR A1325D / (B) controls. (C and D) Triglyceride/protein ratios in to-gal4 dinr A1325D 7- to 10-day-old adult males compared to those in yolk-gal4 GFP (C) and UAS-dInR A1325D/ (D) controls. Each experiment was performed at least three times; values are means SEM. *, P 0.05, and **, P 0.01 by unpaired Student s t test. nutrients as glycogen and triglyceride and, during pupation, breaks down to supply the animal with enough energy to undergo metamorphosis (25, 36). The adult fat body is derived from a separate cell lineage from the larval fat body, and its cells are not easily identified until 3 to 4 days after eclosion (1, 26). To turn on insulin signaling in this tissue, we expressed an activated form of the insulin receptor, dinr A1325D, in the female and male fat bodies by using the Gal4-upstream activation sequence (UAS) system with the yolk-gal4 and to-gal4 drivers, respectively. These drivers are expressed at high levels specifically in both the head and abdominal fat bodies by day 3, with lower-level expression starting at the end of pupal development for yolk-gal4 and starting at eclosion for to-gal4 (14, 18; J. R. DiAngelo and M. J. Birnbaum, unpublished observations). Expression of dinr A1325D in the female fat body increased triglycerides (Fig. 1A and B). A similar phenotype was observed when dinr A1325D was expressed in the male fat body (Fig. 1C and D). Together, these results suggest that activating insulin signaling in the fat body augments triglyceride storage. Insulin increases adult fat body cell number. In increasing total triglyceride, insulin signaling may be affecting the number of fat body cells, the amount of triglyceride in each cell, or both. If the increase in triglycerides from expressing dinr A1325D in the fat body was due to more fat per cell, then this increase in fat may result from larger or more abundant lipid droplets. To test this possibility directly, fat bodies were stained with the neutral lipid stain Nile Red. Activating insulin signaling only slightly increased the cytoplasmic area occupied by lipid droplets (Fig. 2). This finding is consistent with the trend toward an increase in triglyceride/protein and triglyceride/dna ratios in dinr A1325D -expressing fat bodies (Fig. 3C and E) (see below). While these data suggest that insulin does promote lipid accumulation in each cell, the increase in droplet area appears to be insufficient to account for the entire expansion in lipid mass measured in the dinr A1325D -expressing animals. To assess the effect of insulin signaling on fat cell number, we dissociated fat body tissue enzymatically and counted the cells. Activating insulin signaling in the fat body led to an on November 18, 2018 by guest
4 6344 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. FIG. 2. Insulin increases the area covered by lipid droplets. (A to D) DAPI (A and C) and Nile Red (B and D) staining of isolated fat bodies from 4- to 5-day-old yolk-gal4 GFP and yolk-gal4 dinr A1325D adult virgin females, respectively. Scale bars equal 75 m. (E) Quantification of the mean area covered by lipid based on Nile Red staining of yolk-gal4 GFP and yolk-gal4 dinr A1325D fat bodies is expressed in arbitrary relative units. Each experiment was performed at least three times, and values are means SEM. *, P increase in the number of cells compared to tissues from the control animals (Fig. 3A). The total DNA content of dissected fat bodies reflected the number of cells, increasing to the same extent as the cell number in the dinr A1325D -expressing fat body, and was therefore used as a surrogate measure of cell number (Fig. 3B and D). dinr A1325D did not significantly increase the protein/dna ratio in a normally proliferative fat body, consistent with there being no profound effect on fat cell
5 VOL. 29, 2009 INSULIN ACTION ON FAT MASS IN DROSOPHILA 6345 FIG. 3. Insulin signaling regulates fat cell number in the adult fat body. (A) Number of fat body cells dissociated after 4hin0.5% trypsin-edta from fat bodies dissected from 4- to 5-day-old adult yolk-gal4 GFP, dinr A1325D virgin females or yolk-gal4 GFP control animals. (B) Total DNA content of fat bodies dissected from 4- to 5-day-old adult yolk-gal4 dinr A1325D virgin females or yolk-gal4 GFP control animals. (C) Triglyceride/protein, protein/dna, and triglyceride/dna ratios in fat bodies dissected from yolk-gal4 dinr A1325D 4- to 5-day-old adult virgin females or yolk-gal4 GFP control animals. (D) Total DNA content of fat bodies dissected from 4- to 5-day-old adult yolk-gal4 dinr A1325D virgin females or UAS-dInR A1325D/ control animals. (E) Triglyceride/protein, protein/dna, and triglyceride/dna ratios in fat bodies dissected from yolk-gal4 dinr A1325D 4- to 5-day-old adult virgin females or UAS-dInR A1325D / control animals. Each experiment was performed at least three times, and values are means SEM. *, P 0.05; **, P 0.01.
6 6346 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. size independent of triglyceride accumulation (Fig. 3C and E). As mentioned above, triglyceride/protein and triglyceride/ DNA ratios tended toward an increase, again reflecting the modest increase in triglyceride per cell apparent from the light microscopy analysis (Fig. 2 and 3C and E). Overall, these data indicate that insulin positively regulates cell number in the adult fat body and that this regulation contributes significantly to the enhancement in triglyceride storage. Insulin promotes triglyceride storage independently of changes in fat cell number. Since overexpressing dinr A1325D in fat bodies revealed slightly larger lipid droplets than those in controls, we hypothesized that insulin signaling could regulate fat storage independently of changes in cell number. To test this hypothesis directly, we uncoupled proliferation from metabolism by holding the fat body cell number constant through inhibiting the cell cycle via overexpression of the Drosophila E2F corepressor retinoblastoma family homolog (RBF) (15). While RBF expression alone had no effect on the fat body DNA content or the triglyceride/protein, protein/dna, or triglyceride/dna ratio (Fig. 4A and B), RBF expression completely suppressed the increase in the fat body DNA content induced by dinr A1325D (Fig. 3B and D and 4C). These data are most consistent with the idea that insulin signaling enhanced fat cell number by stimulating cell proliferation. Utilization of RBF also provided a strategy to assess the effect of insulin signaling on lipid levels independently of changes in fat cell number. To accomplish this assessment, we measured fat body triglycerides in animals that expressed both dinr A1325D and RBF. These flies displayed increased triglyceride/protein ratios and, as there was no change in the DNA content, enhanced levels of triglyceride storage per cell (Fig. 4D). The ratio of protein to DNA was unchanged (Fig. 4D). These phenotypes were not caused by a reduction in insulin signaling due to the same driver s activating two effector lines at once, as levels of phospho-dakt increased in animals expressing both dinr A1325D and RBF compared to those in animals expressing RBF alone and the signal was comparable to that from driving of dinr A1325D alone (Fig. 4E). These data indicate that insulin signaling exerts effects on lipid storage independently of the fat body cell number. dfoxo and shaggy act downstream of dinr to regulate fat cell storage by different mechanisms. The insulin-signaling pathway regulates metabolism and growth by at least three distinct pathways downstream of dakt: the Drosophila target of rapamycin (dtor), shaggy (sgg)/glycogen synthase kinase 3 (GSK3), and transcription factor Drosophila FOXO (dfoxo) pathways (16, 17, 37, 38). In order to determine which of these are important to triglyceride accumulation, we performed epistasis experiments with the adult fat body. Overexpression of a dominant-inhibitory form of dtor (UAS-dTOR TED ) (24) both alone and in a background of activated insulin signaling did not affect whole-animal triglyceride levels, DNA content, or the triglyceride/protein, protein/dna, or triglyceride/dna ratios (Fig. 5A and B and 6A and B). In contrast, though expressing an active form of dfoxo, triple mutant dfoxo (dfoxo-tm), alone in the adult fat body had no effect on adult triglyceride levels, dfoxo-tm expression in a background of active insulin signaling suppressed the dinr A1325D -induced triglyceride storage (Fig. 5C and D). In these experiments, we utilized a form of dfoxo rendered active by mutation of the three dakt phosphorylation sites to alanine (27, 28, 40). To examine the requirement of sgg inhibition for the effects of insulin signaling on triglyceride storage, we overexpressed a constitutively active form of sgg. sgg is also regulated by wingless, raising the potential problem of distinguishing the effects of this pathway from those of insulin signaling. To avoid this ambiguity, we utilized a transgenic fly line that expresses a form of sgg in which the Akt phosphorylation site serine 9 has been mutated to alanine (sggs9a); this protein is resistant to the inhibitory effects of insulin signaling while normally responsive to wingless (5, 38). While overexpressing sggs9a alone in the adult fat body had no effect on triglyceride, in a background of activated insulin signaling, there was a trend for partial suppression of enhanced triglyceride storage that did not reach statistical significance (Fig. 5E and F). Thus, these data suggest that insulin signals to specific downstream targets to regulate fat storage. As discussed above, the reduction in fat mass resultant from introducing constitutively active dfoxo or sgg could have been due to changes in cell number, triglyceride storage, or both. To resolve this issue, the fat body triglyceride content was normalized with respect to either the protein or the DNA content (Fig. 6). Both dfoxo-tm and sggs9a antagonized the increase in DNA, that is, cell number, generated by overexpression of dinr A1325D in the adult fat body (Fig. 6C and E). However, FOXO-TM had no effect on the ratio of triglyceride to protein, triglyceride to DNA, or protein to DNA, while sggs9a substantially decreased the first two ratios (Fig. 6D and F). These data suggest that suppression of dfoxo acts downstream of dinr to regulate fat cell number exclusively, whereas inhibition of sgg is important for the control of both the cell number and the amount of triglyceride per cell. DISCUSSION In this study, we have shown that activating insulin signaling in the Drosophila fat body promotes triglyceride storage. These data contrast with those obtained when insulin signaling is decreased in the whole animal, during which triglyceride levels are elevated (4, 30, 52). However, the data in this study are consistent with the long-recognized role of insulin in vertebrate anabolic metabolism and the findings of previous studies using clonal overexpression of insulin-signaling pathway members in the Drosophila fat body (7). A number of possibilities may explain these differences. In insulin-deficient flies, extracellular sugar is increased, which may promote lipid storage by an insulin-independent mechanism (8, 43). It is also possible that the increased fat levels in flies in which insulin signaling is diminished may be secondary to the altered reproductive state of the animals. C. elegans nematodes with mutations in the insulin-signaling pathway enter a diapause state called dauer in which the worms decrease their reproductive fitness and increase their fat stores and life span (reviewed in references 6, 48, and 51). In Drosophila, some dinr and chico mutants also display decreased reproductive fitness and increased life span, raising the possibility that the modified fat content in these animals may be secondary to the other diapause-like phenotypes (4, 13, 52). Lastly, it is possible that altered insulin signaling in neurons may lead to secondary effects on nutrient balance by a mechanism analogous to that in rodents (39). In any case, the study results presented above demonstrate clearly
7 VOL. 29, 2009 INSULIN ACTION ON FAT MASS IN DROSOPHILA 6347 FIG. 4. Insulin regulates triglyceride storage independently of cell number. (A) Total DNA content of fat bodies dissected from 4- to 5-day-old adult yolk-gal4 RBF virgin females or yolk-gal4 GFP control animals. (B) Triglyceride/protein, protein/dna, and triglyceride/dna ratios in fat bodies dissected from yolk-gal4 RBF 4- to 5-day-old adult virgin females or yolk-gal4 GFP control animals. (C) Total DNA content of fat bodies dissected from 4- to 5-day-old adult yolk-gal4 (RBF, dinr A1325D ) virgin females or yolk-gal4 RBF control animals. (D) Triglyceride/ protein, protein/dna, and triglyceride/dna ratios in fat bodies dissected from yolk-gal4 (RBF, dinr A1325D ) 4- to 5-day-old adult virgin females or yolk-gal4 RBF control animals. (E) Representative immunoblot of protein extracts made from fat bodies dissected from yolk-gal4 RBF, dinr A1325D 4- to 5-day-old virgin females or yolk-gal4 RBF and yolk-gal4 dinr A1325D control animals. Each experiment was performed at least three times, and values are means SEM. *, P 0.05; P-dAkt, phospho-dakt.
8 6348 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. Downloaded from FIG. 5. Differential signaling downstream of dinr regulates triglyceride levels. (A) Triglyceride/protein ratios in individual yolk- Gal4 dtor TED 4- to 5-day-old virgin females compared to those in yolk-gal4 GFP controls. (B) Triglyceride/protein ratios in individual yolk-gal4 dinr A1325D, dtor TED 4- to 5-day-old virgin females compared to those in yolk-gal4 dinr A1325D controls. (C) Triglyceride/protein ratios in individual yolk-gal4 dfoxo-tm 4- to 5-day-old virgin females or yolk-gal4 GFP controls. (D) Triglyceride/protein ratios in individual yolk-gal4 dinr A1325D, dfoxo-tm 4- to 5-day-old virgin females compared to those in yolk-gal4 dinr A1325D controls. (E) Triglyceride/protein ratios in individual yolk-gal4 sggs9a 4- to 5-day-old virgin females compared to those in yolk-gal4 GFP controls. (F) Triglyceride/protein ratios in individual yolk-gal4 dinr A1325D, sggs9a 4- to 5-day-old virgin females compared to those in yolk-gal4 dinr A1325D controls. Each experiment was performed at least three times, and values are means SEM. **, P on November 18, 2018 by guest that in Drosophila, as in mammals, insulin signaling serves to promote fat storage in a cell-autonomous manner. In the adult fat body, expression of an active insulin receptor promoted lipid storage through changes in both the fat cell number and the amount of lipid per cell. This pattern is reminiscent of the behavior of vertebrate preadipocyte cell lines, in which insulin or insulin-like growth factor 1 is critical for proliferation, differentiation, and storage of triglyceride following adipose conversion. In Drosophila, the adult fat body is distinct from the larval one, being derived from a separate developmental lineage (1, 26). Insulin signaling augments cell size and endoreplication in the larval fat body, but it is uncertain how
9 VOL. 29, 2009 INSULIN ACTION ON FAT MASS IN DROSOPHILA 6349 FIG. 6. dfoxo and sgg act downstream of dinr to regulate fat body cell number, while sgg acts downstream of dinr to regulate the amount of triglyceride per cell. Shown are the total DNA contents (A, C, and E) and triglyceride/protein, protein/dna, and triglyceride/dna ratios (B, D, and F) in fat bodies dissected from yolk-gal4 dinr A1325D, dtor TED, yolk-gal4 dinr A1325D, dfoxo-tm, and yolk-gal4 dinr A1325D, sggs9a 4- to 5-day-old adult virgin females, respectively, or yolk-gal4 dinr A1325D control animals. Each experiment was performed at least three times, and values are means SEM. *, P 0.05; **, P the same stimulus increases cell number in the adult (7). Proliferation of a committed precursor is a likely explanation, but as the nature of that cell in regard to the adult fat body is not known, a role for enhanced differentiation cannot be excluded. Manipulation of insulin signaling in other nonendoreplicating, mitotic cells of the larva leads to changes in cell number (11, 19, 56). In any case, the clearly discernible phenotype of increased fat cell number as well as triglyceride levels in response to expression of an active version of the insulin receptor allowed an assessment of downstream signaling by standard genetic strategies. Not surprisingly, abolishing the ability of insulin to promote dakt-dependent phosphorylation and
10 6350 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. inhibition of dfoxo in the adult fat body also eliminated the increase in cell number. In both mammals and Drosophila, FoxO1 resides downstream of and is inhibited by insulin signaling (2). In response to insulin, Akt phosphorylates FoxO1, causing it to be shuttled out of the nucleus and degraded (55). Among the many biological actions attributed to FoxO, positive regulation of proliferation, life span, and stress resistance takes place in both flies and mammals (2). For example, expression of dfoxo in the Drosophila eye or wing decreases organ size by affecting solely cell number and not cell size (28, 40). The role of suppression of FoxO as a mediator of the metabolic actions of insulin appears less clear. Its importance in the mammalian liver, in which inhibition of FoxO is a major means by which insulin represses gluconeogenic gene expression (23), has been most firmly established. The expression of both glucose 6-phosphatase and phosphoenolpyruvate carboxykinase genes is elevated during fasting, at least in part through the action of FoxO, and this effect is reversed by insulin-dependent phosphorylation catalyzed by Akt. The role of FoxO in hepatic lipid metabolism is more controversial (23, 31, 59). Manipulation of FoxO1 activity in the murine liver results in changes in triglyceride content, but it is unclear whether these changes are a direct effect or secondary to feedback modulation in insulin signaling. In contrast, there is a well-defined role for FoxO in cellular differentiation, including adipogenesis. The results of antagonizing transcriptional activation by FoxO1 explain a significant component of Akt s function in supporting adipocyte differentiation (35). However, even in this situation, the requirement for FoxO may well be in mitotic expansion, i.e., the several rounds of division that preadipocytes undergo prior to differentiation (35, 58). These observations are consistent with the data presented herein that dfoxo-tm antagonizes the insulin-dependent increase in fat body cell number, emphasizing the conservation of this pathway in the regulation of adipose mass. While the results of the experiments described herein indicate a role for dfoxo in regulating cell number and not the amount of fat per cell, a previous study has documented that dfoxo-tm overexpression in the Drosophila head fat body leads to enlarged lipid droplets (27). While the same active dfoxo (dfoxo-tm) transgene was used in both studies, this discrepancy may be explained by the difference in the methodologies employed. Hwangbo et al. utilized histological methods to measure lipid droplet size, whereas the study described herein relied on biochemical measurements of fat body triglycerides (27). It is possible that the larger lipid droplets observed in dfoxo-tm-expressing fat bodies in the previous study (27) may not correlate with a net gain of triglyceride, providing an explanation for the different results observed here by measuring triglycerides directly. Different Gal4 drivers were also used in these two studies, and their different temporal and spatial control of dfoxo-tm expression may be responsible for the differing results. The absence of effect of dominant-inhibitory dtor on lipid accumulation downstream of insulin is somewhat surprising, given the suggestion that the TOR pathway is important to lipid metabolism. In addition to regulating cell and organ growth in Drosophila, a role for the TOR pathway in metabolism has been appreciated recently. A hypomorphic mutation of dtor results in reduced fat body triglyceride levels, with a concomitant decrease in the expression of the lipogenic Drosophila fatty acid synthase gene and an increase in the expression of the brummer lipase gene (30). The mutants also have decreased hemolymph sugar levels, suggesting a glucose-regulatory role for the TOR pathway in addition to the control of lipid storage (30). Evidence implicating the targets of TOR, S6K and 4E-BP, in regulating energy homeostasis also exists. Drosophila 4E-BP (d4e-bp) mutants are starvation sensitive due to decreased lipid levels; conversely, overexpression of an active form of d4e-bp increases whole-animal triglyceride levels (53). This role of d4e-bp in regulating lipid storage is consistent with that observed in mammals (54). Thus, although TOR signaling has been shown to regulate lipid storage, the signal activating TOR in this process is unknown. Given the findings presented above indicating a lack of effect of dominant-inhibitory TOR on insulin-dependent triglyceride accumulation, it is likely that TOR in flies regulates lipids in response to a stimulus such as amino acids rather than insulin. In contrast to the profound effect of active dfoxo on cell number in the fat body, the ability of nonphosphorylatable sgg to suppress the insulin-dependent increase in triglyceride content was completely unanticipated. GSK3 is a signaling intermediate in at least two pathways, the Wnt/wingless and the insulin/pi3k pathways, although phosphorylation by Akt is important only to the latter (34). The role of GSK3 in regulating mammalian adipocyte differentiation is related to Wnt signaling and thus is not relevant to the effects on lipid accumulation found in this study (29, 42). A genetic deficiency in GSK3 partially suppresses diabetes in animal models, but this finding has not been linked to alterations in lipid metabolism (49). A function for GSK3 as a determinant of lipid storage levels in either the mammalian adipocyte or the mammalian hepatocyte has not been ruled out, and the strong conservation of insulinsignaling pathways between Drosophila and higher vertebrates suggests that closer examination of this possibility is warranted. Interestingly, GSK3 phosphorylates SREBPs, causing recruitment of the ubiquitin ligase SCF Fbw7 and targeting for ubiquitination and degradation by the proteosome (47). This phenomenon has been studied in regard to its role in the modulation of lipogenesis occurring with the cell cycle but has not been evaluated for its relevance to metabolic signaling by insulin, nor has it been tested genetically in liver. In summary, this study demonstrates that insulin promotes triglyceride storage in the Drosophila fat body by regulating both cell number and lipid accumulation. Moreover, the complete effect of insulin depends upon the inhibition of signaling through both dfoxo and sgg, raising the novel possibility that the latter is a key signaling intermediate in insulin s critical role in anabolic lipid metabolism. ACKNOWLEDGMENTS We thank Sara Cherry, Marc Tatar, Nick Dyson, and the Bloomington Stock Center for providing fly strains. We also thank Eric Rulifson, Sara Cherry, and members of the Birnbaum lab for helpful discussions, as well as Ed Williamson and the Children s Hospital of Philadelphia Pathology Confocal Core for technical assistance with the Nile Red staining. This work was supported by NIH grant DK56886 to M.J.B. J.R.D. is a recipient of the National Research Service Award for Training in Cell and Molecular Biology (T32-GM07229) and a predoctoral fellowship from the American Heart Association.
11 VOL. 29, 2009 INSULIN ACTION ON FAT MASS IN DROSOPHILA 6351 REFERENCES 1. Aguila, J. R., J. Suszko, A. G. Gibbs, and D. K. Hoshizaki The role of larval fat cells in adult Drosophila melanogaster. J. Exp. Biol. 210: Arden, K. C FOXO animal models reveal a variety of diverse roles for FOXO transcription factors. Oncogene 27: Baker, K. D., and C. S. Thummel Diabetic larvae and obese flies emerging studies of metabolism in Drosophila. Cell Metab. 6: Bohni, R., J. Riesgo-Escovar, S. Oldham, W. Brogiolo, H. Stocker, B. F. Andruss, K. Beckingham, and E. Hafen Autonomous control of cell and organ size by CHICO, a Drosophila homolog of vertebrate IRS1-4. Cell 97: Bourouis, M Targeted increase in shaggy activity levels blocks wingless signaling. Genesis 34: Braeckman, B. P., K. Houthoofd, and J. R. Vanfleteren Insulin-like signaling, metabolism, stress resistance and aging in Caenorhabditis elegans. Mech. Ageing Dev. 122: Britton, J. S., W. K. Lockwood, L. Li, S. M. Cohen, and B. A. Edgar Drosophila s insulin/pi3-kinase pathway coordinates cellular metabolism with nutritional conditions. Dev. Cell 2: Broughton, S. J., M. D. Piper, T. Ikeya, T. M. Bass, J. Jacobson, Y. Driege, P. Martinez, E. Hafen, D. J. Withers, S. J. Leevers, and L. Partridge Longer lifespan, altered metabolism, and stress resistance in Drosophila from ablation of cells making insulin-like ligands. Proc. Natl. Acad. Sci. USA 102: Burks, D. J., J. Font de Mora, M. Schubert, D. J. Withers, M. G. Myers, H. H. Towery, S. L. Altamuro, C. L. Flint, and M. F. White IRS-2 pathways integrate female reproduction and energy homeostasis. Nature 407: Canavoso, L. E., Z. E. Jouni, K. J. Karnas, J. E. Pennington, and M. A. Wells Fat metabolism in insects. Annu. Rev. Nutr. 21: Chen, C., J. Jack, and R. S. Garofalo The Drosophila insulin receptor is required for normal growth. Endocrinology 137: Cho, H., J. Mu, J. K. Kim, J. L. Thorvaldsen, Q. Chu, E. B. Crenshaw, K. H. Kaestner, M. S. Bartolomei, G. I. Shulman, and M. J. Birnbaum Insulin resistance and a diabetes mellitus-like syndrome in mice lacking the protein kinase Akt2 (PKB beta). Science 292: Clancy, D. J., D. Gems, L. G. Harshman, S. Oldham, H. Stocker, E. Hafen, S. J. Leevers, and L. Partridge Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science 292: Dauwalder, B., S. Tsujimoto, J. Moss, and W. Mattox The Drosophila takeout gene is regulated by the somatic sex-determination pathway and affects male courtship behavior. Genes Dev. 16: Du, W., M. Vidal, J. E. Xie, and N. Dyson RBF, a novel RB-related gene that regulates E2F activity and interacts with cyclin E in Drosophila. Genes Dev. 10: Edgar, B. A How flies get their size: genetics meets physiology. Nat. Rev. Genet. 7: Garofalo, R. S Genetic analysis of insulin signaling in Drosophila. Trends Endocrinol. Metab. 13: Georgel, P., S. Naitza, C. Kappler, D. Ferrandon, D. Zachary, C. Swimmer, C. Kopczynski, G. Duyk, J. M. Reichhart, and J. A. Hoffmann Drosophila immune deficiency (IMD) is a death domain protein that activates antibacterial defense and can promote apoptosis. Dev. Cell 1: Goberdhan, D. C., N. Paricio, E. C. Goodman, M. Mlodzik, and C. Wilson Drosophila tumor suppressor PTEN controls cell size and number by antagonizing the Chico/PI3-kinase signaling pathway. Genes Dev. 13: Gronke, S., M. Beller, S. Fellert, H. Ramakrishnan, H. Jackle, and R. P. Kuhnlein Control of fat storage by a Drosophila PAT domain protein. Curr. Biol. 13: Gronke, S., A. Mildner, S. Fellert, N. Tennagels, S. Petry, G. Muller, H. Jackle, and R. P. Kuhnlein Brummer lipase is an evolutionary conserved fat storage regulator in Drosophila. Cell Metab. 1: Gronke, S., G. Muller, J. Hirsch, S. Fellert, A. Andreou, T. Haase, H. Jackle, and R. P. Kuhnlein Dual lipolytic control of body fat storage and mobilization in Drosophila. PLoS Biol. 5:e Gross, D. N., A. P. van den Heuvel, and M. J. Birnbaum The role of FoxO in the regulation of metabolism. Oncogene 27: Hennig, K. M., and T. P. Neufeld Inhibition of cellular growth and proliferation by dtor overexpression in Drosophila. Genesis 34: Hoshizaki, D. K Fat-cell development, p In L. I. Gilbert, K. Iatrou, and S. S. Gill (ed.), Comprehensive molecular insect science. Elsevier, Amsterdam, The Netherlands. 26. Hoshizaki, D. K., R. Lunz, M. Ghosh, and W. Johnson Identification of fat-cell enhancer activity in Drosophila melanogaster using P-element enhancer traps. Genome 38: Hwangbo, D. S., B. Gershman, M. P. Tu, M. Palmer, and M. Tatar Drosophila dfoxo controls lifespan and regulates insulin signalling in brain and fat body. Nature 429: Junger, M. A., F. Rintelen, H. Stocker, J. D. Wasserman, M. Vegh, T. Radimerski, M. E. Greenberg, and E. Hafen The Drosophila forkhead transcription factor FOXO mediates the reduction in cell number associated with reduced insulin signaling. J. Biol. 2: Longo, K. A., W. S. Wright, S. Kang, I. Gerin, S. H. Chiang, P. C. Lucas, M. R. Opp, and O. A. MacDougald Wnt10b inhibits development of white and brown adipose tissues. J. Biol. Chem. 279: Luong, N., C. R. Davies, R. J. Wessells, S. M. Graham, M. T. King, R. Veech, R. Bodmer, and S. M. Oldham Activated FOXO-mediated insulin resistance is blocked by reduction of TOR activity. Cell Metab. 4: Matsumoto, M., S. Han, T. Kitamura, and D. Accili Dual role of transcription factor FoxO1 in controlling hepatic insulin sensitivity and lipid metabolism. J. Clin. Investig. 116: Matsumoto, M., W. Ogawa, K. Akimoto, H. Inoue, K. Miyake, K. Furukawa, Y. Hayashi, H. Iguchi, Y. Matsuki, R. Hiramatsu, H. Shimano, N. Yamada, S. Ohno, M. Kasuga, and T. Noda PKC in liver mediates insulin-induced SREBP-1c expression and determines both hepatic lipid content and overall insulin sensitivity. J. Clin. Investig. 112: McGarry, J. D What if Minkowski had been ageusic? An alternative angle on diabetes. Science 258: McManus, E. J., K. Sakamoto, L. J. Armit, L. Ronaldson, N. Shpiro, R. Marquez, and D. R. Alessi Role that phosphorylation of GSK3 plays in insulin and Wnt signalling defined by knockin analysis. EMBO J. 24: Nakae, J., T. Kitamura, Y. Kitamura, W. H. Biggs III, K. C. Arden, and D. Accili The forkhead transcription factor Foxo1 regulates adipocyte differentiation. Dev. Cell 4: Nelliot, A., N. Bond, and D. K. Hoshizaki Fat-body remodeling in Drosophila melanogaster. Genesis 44: Oldham, S., and E. Hafen Insulin/IGF and target of rapamycin signaling: a TOR de force in growth control. Trends Cell Biol. 13: Papadopoulou, D., M. W. Bianchi, and M. Bourouis Functional studies of shaggy/glycogen synthase kinase 3 phosphorylation sites in Drosophila melanogaster. Mol. Cell. Biol. 24: Plum, L., M. Schubert, and J. C. Bruning The role of insulin receptor signaling in the brain. Trends Endocrinol. Metab. 16: Puig, O., M. T. Marr, M. L. Ruhf, and R. Tjian Control of cell number by Drosophila FOXO: downstream and feedback regulation of the insulin receptor pathway. Genes Dev. 17: Rosen, E. D., and O. A. MacDougald Adipocyte differentiation from the inside out. Nat. Rev. Mol. Cell Biol. 7: Ross, S. E., N. Hemati, K. A. Longo, C. N. Bennett, P. C. Lucas, R. L. Erickson, and O. A. MacDougald Inhibition of adipogenesis by Wnt signaling. Science 289: Rulifson, E. J., S. K. Kim, and R. Nusse Ablation of insulin-producing neurons in flies: growth and diabetic phenotypes. Science 296: Saltiel, A. R., and C. R. Kahn Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414: Schlegel, A., and D. Y. Stainier Lessons from lower organisms: what worms, flies, and zebrafish can teach us about human energy metabolism. PLoS Genet. 3:e Shimomura, I., Y. Bashmakov, S. Ikemoto, J. D. Horton, M. S. Brown, and J. L. Goldstein Insulin selectively increases SREBP-1c mrna in the livers of rats with streptozotocin-induced diabetes. Proc. Natl. Acad. Sci. USA 96: Sundqvist, A., M. T. Bengoechea-Alonso, X. Ye, V. Lukiyanchuk, J. Jin, J. W. Harper, and J. Ericsson Control of lipid metabolism by phosphorylation-dependent degradation of the SREBP family of transcription factors by SCF(Fbw7). Cell Metab. 1: Swanson, M. M., and D. L. Riddle Critical periods in the development of the Caenorhabditis elegans dauer larva. Dev. Biol. 84: Tanabe, K., Z. Liu, S. Patel, B. W. Doble, L. Li, C. Cras-Meneur, S. C. Martinez, C. M. Welling, M. F. White, E. Bernal-Mizrachi, J. R. Woodgett, and M. A. Permutt Genetic deficiency of glycogen synthase kinase-3 corrects diabetes in mouse models of insulin resistance. PLoS Biol. 6:e Taniguchi, C. M., T. Kondo, M. Sajan, J. Luo, R. Bronson, T. Asano, R. Farese, L. C. Cantley, and C. R. Kahn Divergent regulation of hepatic glucose and lipid metabolism by phosphoinositide 3-kinase via Akt and PKC /. Cell Metab. 3: Tatar, M., A. Bartke, and A. Antebi The endocrine regulation of aging by insulin-like signals. Science 299: Tatar, M., A. Kopelman, D. Epstein, M. P. Tu, C. M. Yin, and R. S. Garofalo A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292: Teleman, A. A., Y. W. Chen, and S. M. Cohen E-BP functions as a metabolic brake used under stress conditions but not during normal growth. Genes Dev. 19: Tsukiyama-Kohara, K., F. Poulin, M. Kohara, C. T. DeMaria, A. Cheng, Z. Wu, A. C. Gingras, A. Katsume, M. Elchebly, B. M. Spiegelman, M. E. Harper, M. L. Tremblay, and N. Sonenberg Adipose tissue reduction in mice lacking the translational inhibitor 4E-BP1. Nat. Med. 7:
12 6352 DIANGELO AND BIRNBAUM MOL. CELL. BIOL. 55. Van Der Heide, L. P., M. F. Hoekman, and M. P. Smidt The ins and outs of FoxO shuttling: mechanisms of FoxO translocation and transcriptional regulation. Biochem. J. 380: Weinkove, D., T. P. Neufeld, T. Twardzik, M. D. Waterfield, and S. J. Leevers Regulation of imaginal disc cell size, cell number and organ size by Drosophila class I(A) phosphoinositide 3-kinase and its adaptor. Curr. Biol. 9: Wolfrum, C., E. Asilmaz, E. Luca, J. M. Friedman, and M. Stoffel Foxa2 regulates lipid metabolism and ketogenesis in the liver during fasting and in diabetes. Nature 432: Yun, S. J., E. K. Kim, D. F. Tucker, C. D. Kim, M. J. Birnbaum, and S. S. Bae Isoform-specific regulation of adipocyte differentiation by Akt/ protein kinase B. Biochem. Biophys. Res. Commun. 371: Zhang, W., S. Patil, B. Chauhan, S. Guo, D. R. Powell, J. Le, A. Klotsas, R. Matika, X. Xiao, R. Franks, K. A. Heidenreich, M. P. Sajan, R. V. Farese, D. B. Stolz, P. Tso, S. H. Koo, M. Montminy, and T. G. Unterman FoxO1 regulates multiple metabolic pathways in the liver: effects on gluconeogenic, glycolytic, and lipogenic gene expression. J. Biol. Chem. 281:
Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld
 Journal of Biology BioMed Central Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld Address: Department of Genetics, Cell Biology, and Development,
Journal of Biology BioMed Central Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld Address: Department of Genetics, Cell Biology, and Development,
Requires Signaling though Akt2 Independent of the. Transcription Factors FoxA2, FoxO1, and SREBP1c
 Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Genetic analysis of insulin signaling in Drosophila
 156 Review Genetic analysis of insulin signaling in Drosophila Robert S. Garofalo Studies in the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans have revealed that components
156 Review Genetic analysis of insulin signaling in Drosophila Robert S. Garofalo Studies in the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans have revealed that components
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Supplementary Information
 Supplementary Information Akt regulates hepatic metabolism by suppressing a Foxo1 dependent global inhibition of adaptation to nutrient intake Mingjian Lu 1, Min Wan 1, Karla F. Leavens 1, Qingwei Chu
Supplementary Information Akt regulates hepatic metabolism by suppressing a Foxo1 dependent global inhibition of adaptation to nutrient intake Mingjian Lu 1, Min Wan 1, Karla F. Leavens 1, Qingwei Chu
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
Growth and Differentiation Phosphorylation Sampler Kit
 Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
Phospho-AKT Sampler Kit
 Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Metabolic integration and Regulation
 Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Metabolic Syndrome. DOPE amines COGS 163
 Metabolic Syndrome DOPE amines COGS 163 Overview - M etabolic Syndrome - General definition and criteria - Importance of diagnosis - Glucose Homeostasis - Type 2 Diabetes Mellitus - Insulin Resistance
Metabolic Syndrome DOPE amines COGS 163 Overview - M etabolic Syndrome - General definition and criteria - Importance of diagnosis - Glucose Homeostasis - Type 2 Diabetes Mellitus - Insulin Resistance
UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Regulation of SREBP1c Expression by mtor Signaling in Hepatocytes
 Kobe J. Med. Sci., Vol. 55, No. 2, pp. E45-E52, 29 Regulation of SREBP1c Expression by mtor Signaling in Hepatocytes MOTOTSUGU TAKASHIMA 1, WATARU OGAWA 1,*, AKI EMI 1, and MASATO KASUGA 1,2 1 Department
Kobe J. Med. Sci., Vol. 55, No. 2, pp. E45-E52, 29 Regulation of SREBP1c Expression by mtor Signaling in Hepatocytes MOTOTSUGU TAKASHIMA 1, WATARU OGAWA 1,*, AKI EMI 1, and MASATO KASUGA 1,2 1 Department
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
What systems are involved in homeostatic regulation (give an example)?
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
KEY CONCEPT QUESTIONS IN SIGNAL TRANSDUCTION
 Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DIVISION OF SCIENCE
 THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DIVISION OF SCIENCE THE REGULATION OF LIPID STORAGE BY THE SPLICING FACTOR TRANSFORMER2 (TRA2) IN DROSOPHILA CEZARY MIKOLUK SPRING 2017 A thesis
THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DIVISION OF SCIENCE THE REGULATION OF LIPID STORAGE BY THE SPLICING FACTOR TRANSFORMER2 (TRA2) IN DROSOPHILA CEZARY MIKOLUK SPRING 2017 A thesis
Cytoplasmic activated protein kinase Akt regulates lipiddroplet accumulation in Drosophila nurse cells
 RESEARCH ARTICLE 4731 Development 133, 4731-4735 (2006) doi:10.1242/dev.02659 Cytoplasmic activated protein kinase Akt regulates lipiddroplet accumulation in Drosophila nurse cells Natalia Vereshchagina
RESEARCH ARTICLE 4731 Development 133, 4731-4735 (2006) doi:10.1242/dev.02659 Cytoplasmic activated protein kinase Akt regulates lipiddroplet accumulation in Drosophila nurse cells Natalia Vereshchagina
Lecture 10. G1/S Regulation and Cell Cycle Checkpoints. G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint
 Lecture 10 G1/S Regulation and Cell Cycle Checkpoints Outline: G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint Paper: The roles of Fzy/Cdc20 and Fzr/Cdh1
Lecture 10 G1/S Regulation and Cell Cycle Checkpoints Outline: G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint Paper: The roles of Fzy/Cdc20 and Fzr/Cdh1
Problem Set 5 KEY
 2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Src-INACTIVE / Src-INACTIVE
 Biology 169 -- Exam 1 February 2003 Answer each question, noting carefully the instructions for each. Repeat- Read the instructions for each question before answering!!! Be as specific as possible in each
Biology 169 -- Exam 1 February 2003 Answer each question, noting carefully the instructions for each. Repeat- Read the instructions for each question before answering!!! Be as specific as possible in each
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
NJ To whom correspondence should be addressed: Department of Neuroscience,
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 287, NO. 39, pp. 32406 32414, September 21, 2012 2012 by The American Society for Biochemistry and Molecular Biology, Inc. Published in the U.S.A. Interaction between
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 287, NO. 39, pp. 32406 32414, September 21, 2012 2012 by The American Society for Biochemistry and Molecular Biology, Inc. Published in the U.S.A. Interaction between
Molecular mechanisms of metabolic regulation by insulin in Drosophila
 Biochem. J. (2010) 425, 13 26 (Printed in Great Britain) doi:10.1042/bj20091181 13 REVIEW ARTICLE Molecular mechanisms of metabolic regulation by insulin in Drosophila Aurelio A. TELEMAN 1 German Cancer
Biochem. J. (2010) 425, 13 26 (Printed in Great Britain) doi:10.1042/bj20091181 13 REVIEW ARTICLE Molecular mechanisms of metabolic regulation by insulin in Drosophila Aurelio A. TELEMAN 1 German Cancer
THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals
 Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
ENERGY FROM INGESTED NUTREINTS MAY BE USED IMMEDIATELY OR STORED
 QUIZ/TEST REVIEW NOTES SECTION 1 SHORT TERM METABOLISM [METABOLISM] Learning Objectives: Identify primary energy stores of the body Differentiate the metabolic processes of the fed and fasted states Explain
QUIZ/TEST REVIEW NOTES SECTION 1 SHORT TERM METABOLISM [METABOLISM] Learning Objectives: Identify primary energy stores of the body Differentiate the metabolic processes of the fed and fasted states Explain
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Regulation of Lipid Homeostasis: Lipid Droplets
 Regulation of Lipid Homeostasis: Lipid Droplets Bernd Helms Article Brand Recter Hendrik Mertens 1 The Basics I: FAs The Basics II: FA Activation 2 Basics III: TG-FA Interplay. Why? Adipocytes 3 Foam cells
Regulation of Lipid Homeostasis: Lipid Droplets Bernd Helms Article Brand Recter Hendrik Mertens 1 The Basics I: FAs The Basics II: FA Activation 2 Basics III: TG-FA Interplay. Why? Adipocytes 3 Foam cells
Daniela Drummond-Barbosa, Ph.D. Department of Biochemistry and Molecular Biology
 Control of stem cells by diet and systemic factors in the Drosophila ovary Daniela Drummond-Barbosa, Ph.D. Department of Biochemistry and Molecular Biology Multiple levels of stem cell regulation local
Control of stem cells by diet and systemic factors in the Drosophila ovary Daniela Drummond-Barbosa, Ph.D. Department of Biochemistry and Molecular Biology Multiple levels of stem cell regulation local
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Protein kinases are enzymes that add a phosphate group to proteins according to the. ATP + protein OH > Protein OPO 3 + ADP
 Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Week 3 The Pancreas: Pancreatic ph buffering:
 Week 3 The Pancreas: A gland with both endocrine (secretion of substances into the bloodstream) & exocrine (secretion of substances to the outside of the body or another surface within the body) functions
Week 3 The Pancreas: A gland with both endocrine (secretion of substances into the bloodstream) & exocrine (secretion of substances to the outside of the body or another surface within the body) functions
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic
 Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
18. PANCREATIC FUNCTION AND METABOLISM. Pancreatic secretions ISLETS OF LANGERHANS. Insulin
 18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila
 SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila Ayako Kohyama-Koganeya, 1,2 Takuji Nabetani, 1 Masayuki Miura, 2,3 Yoshio Hirabayashi
SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila Ayako Kohyama-Koganeya, 1,2 Takuji Nabetani, 1 Masayuki Miura, 2,3 Yoshio Hirabayashi
PHY MUSCLE AND EXERCISE. LECTURE 2: Introduction to Exercise Metabolism
 PHY3072 - MUSCLE AND EXERCISE LECTURE 2: Introduction to Exercise Metabolism Learning objectives: - Outline sources of metabolic substrates (fuels), describe when they are used - Relationship between oxidative
PHY3072 - MUSCLE AND EXERCISE LECTURE 2: Introduction to Exercise Metabolism Learning objectives: - Outline sources of metabolic substrates (fuels), describe when they are used - Relationship between oxidative
The Drosophila insulin/igf receptor controls growth and size by modulating PtdInsP 3 levels
 Development 129, 4103-4109 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7960 4103 The Drosophila insulin/igf receptor controls growth and size by modulating PtdInsP 3 levels
Development 129, 4103-4109 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7960 4103 The Drosophila insulin/igf receptor controls growth and size by modulating PtdInsP 3 levels
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
MBB317. Dr D MANGNALL OBESITY. Lecture 2
 MBB317 Dr D MANGNALL OBESITY Lecture 2 When the structure of the insulin receptor was first discovered it was assumed that the active beta subunit tyrosine kinase would phosphorylate some intracellular
MBB317 Dr D MANGNALL OBESITY Lecture 2 When the structure of the insulin receptor was first discovered it was assumed that the active beta subunit tyrosine kinase would phosphorylate some intracellular
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
In The Name Of God. In The Name Of. EMRI Modeling Group
 In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
20S Proteasome Activity Assay Kit
 20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
Obesity frequently leads to insulin-resistance that, in turn, produces
 Bifurcation of insulin signaling pathway in rat liver: mtorc1 required for stimulation of lipogenesis, but not inhibition of gluconeogenesis Shijie Li, Michael S. Brown 1, and Joseph L. Goldstein 1 Department
Bifurcation of insulin signaling pathway in rat liver: mtorc1 required for stimulation of lipogenesis, but not inhibition of gluconeogenesis Shijie Li, Michael S. Brown 1, and Joseph L. Goldstein 1 Department
Master class Biomolecular Sciences Molecular Cell Biology.
 Master class Biomolecular Sciences Molecular Cell Biology. 04-09-08: Paul van Bergen en Henegouwen. Clathrin-indep Endocytosis 11-09-08: Willem Stoorvogel. Endocytosis and MHC classii 18-09-08: X-track
Master class Biomolecular Sciences Molecular Cell Biology. 04-09-08: Paul van Bergen en Henegouwen. Clathrin-indep Endocytosis 11-09-08: Willem Stoorvogel. Endocytosis and MHC classii 18-09-08: X-track
Receptor mediated Signal Transduction
 Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Introduction to Cancer Biology
 Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
Introduction to Cancer Biology Robin Hesketh Multiple choice questions (choose the one correct answer from the five choices) Which ONE of the following is a tumour suppressor? a. AKT b. APC c. BCL2 d.
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
Drosophila PTEN Regulates Cell Growth and Proliferation through PI3K-Dependent and -Independent Pathways
 Developmental Biology 221, 404 418 (2000) doi:10.1006/dbio.2000.9680, available online at http://www.idealibrary.com on Drosophila PTEN Regulates Cell Growth and Proliferation through PI3K-Dependent and
Developmental Biology 221, 404 418 (2000) doi:10.1006/dbio.2000.9680, available online at http://www.idealibrary.com on Drosophila PTEN Regulates Cell Growth and Proliferation through PI3K-Dependent and
Homeostatic Control Systems
 Homeostatic Control Systems In order to maintain homeostasis, control system must be able to Detect deviations from normal in the internal environment that need to be held within narrow limits Integrate
Homeostatic Control Systems In order to maintain homeostasis, control system must be able to Detect deviations from normal in the internal environment that need to be held within narrow limits Integrate
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
Supplementary Figures
 J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
Phenomena first observed in petunia
 Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
LABORATÓRIUMI GYAKORLAT SILLABUSZ SYLLABUS OF A PRACTICAL DEMOSTRATION. financed by the program
 TÁMOP-4.1.1.C-13/1/KONV-2014-0001 projekt Az élettudományi-klinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program
TÁMOP-4.1.1.C-13/1/KONV-2014-0001 projekt Az élettudományi-klinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA. 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT. Acc1 AGCAGATCCGCAGCTTG ACCTCTGCTCGCTGAGTGC
 Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
Lecture: CHAPTER 13 Signal Transduction Pathways
 Lecture: 10 17 2016 CHAPTER 13 Signal Transduction Pathways Chapter 13 Outline Signal transduction cascades have many components in common: 1. Release of a primary message as a response to a physiological
Lecture: 10 17 2016 CHAPTER 13 Signal Transduction Pathways Chapter 13 Outline Signal transduction cascades have many components in common: 1. Release of a primary message as a response to a physiological
effects on organ development. a-f, Eye and wing discs with clones of ε j2b10 show no
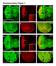 Supplementary Figure 1. Loss of function clones of 14-3-3 or 14-3-3 show no significant effects on organ development. a-f, Eye and wing discs with clones of 14-3-3ε j2b10 show no obvious defects in Elav
Supplementary Figure 1. Loss of function clones of 14-3-3 or 14-3-3 show no significant effects on organ development. a-f, Eye and wing discs with clones of 14-3-3ε j2b10 show no obvious defects in Elav
Insulin worms its way into the spotlight
 PERSPECTIVE Insulin worms its way into the spotlight Donald W. Nelson 2 and Richard W. Padgett 1,3 1 Waksman Institute, Department of Molecular Biology and Biochemistry, Cancer Institute of New Jersey,
PERSPECTIVE Insulin worms its way into the spotlight Donald W. Nelson 2 and Richard W. Padgett 1,3 1 Waksman Institute, Department of Molecular Biology and Biochemistry, Cancer Institute of New Jersey,
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Chapt 15: Molecular Genetics of Cell Cycle and Cancer
 Chapt 15: Molecular Genetics of Cell Cycle and Cancer Student Learning Outcomes: Describe the cell cycle: steps taken by a cell to duplicate itself = cell division; Interphase (G1, S and G2), Mitosis.
Chapt 15: Molecular Genetics of Cell Cycle and Cancer Student Learning Outcomes: Describe the cell cycle: steps taken by a cell to duplicate itself = cell division; Interphase (G1, S and G2), Mitosis.
Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy
 Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003)
 MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
ab Adipogenesis Assay Kit (Cell-Based)
 ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
Lecture 34. Carbohydrate Metabolism 2. Glycogen. Key Concepts. Biochemistry and regulation of glycogen degradation
 Lecture 34 Carbohydrate Metabolism 2 Glycogen Key Concepts Overview of Glycogen Metabolism Biochemistry and regulation of glycogen degradation Biochemistry and regulation of glycogen synthesis What mechanisms
Lecture 34 Carbohydrate Metabolism 2 Glycogen Key Concepts Overview of Glycogen Metabolism Biochemistry and regulation of glycogen degradation Biochemistry and regulation of glycogen synthesis What mechanisms
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy se
 A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis
 Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis MUDr. Jiří Vachtenheim, CSc. CELL CYCLE - SUMMARY Basic terminology: Cyclins conserved proteins with homologous regions; their cellular
Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis MUDr. Jiří Vachtenheim, CSc. CELL CYCLE - SUMMARY Basic terminology: Cyclins conserved proteins with homologous regions; their cellular
BIO360 Fall 2013 Quiz 1
 BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
Rabbit Polyclonal antibody to NFkB p65 (v-rel reticuloendotheliosis viral oncogene homolog A (avian))
 Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Module C CHEMISTRY & CELL BIOLOGY REVIEW
 Module C CHEMISTRY & CELL BIOLOGY REVIEW Note: This module is provided for A&P courses that do not have a prerequisite class which includes chemistry and cell biology. Content covered by required prerequisite
Module C CHEMISTRY & CELL BIOLOGY REVIEW Note: This module is provided for A&P courses that do not have a prerequisite class which includes chemistry and cell biology. Content covered by required prerequisite
A Central Role of MG53 in Metabolic Syndrome. and Type-2 Diabetes
 A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
Supplementary Information
 Supplementary Information Supplementary s Supplementary 1 All three types of foods suppress subsequent feeding in both sexes when the same food is used in the pre-feeding test feeding. (a) Adjusted pre-feeding
Supplementary Information Supplementary s Supplementary 1 All three types of foods suppress subsequent feeding in both sexes when the same food is used in the pre-feeding test feeding. (a) Adjusted pre-feeding
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3
 Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Yiying Zhang, PhD Research Scientist. Research Summary:
 Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 25 Metabolism and Nutrition Metabolic Reactions Metabolism refers to all of the chemical reactions taking place in the body. Reactions that break
Principles of Anatomy and Physiology 14 th Edition CHAPTER 25 Metabolism and Nutrition Metabolic Reactions Metabolism refers to all of the chemical reactions taking place in the body. Reactions that break
Head of College Scholars List Scheme. Summer Studentship Report Form
 Head of College Scholars List Scheme Summer Studentship 2019 Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the
Head of College Scholars List Scheme Summer Studentship 2019 Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
An important function of the central nervous
 Perspectives in Diabetes Insulin Signaling in the Central Nervous System A Critical Role in Metabolic Homeostasis and Disease From C. elegans to Humans Daniel Porte, Jr., 1,2,3 Denis G. Baskin, 3 and Michael
Perspectives in Diabetes Insulin Signaling in the Central Nervous System A Critical Role in Metabolic Homeostasis and Disease From C. elegans to Humans Daniel Porte, Jr., 1,2,3 Denis G. Baskin, 3 and Michael
STRATEGIES FOR TREATMENT OR PREVENTION OF DIABETES USING A CHEMICAL THAT MIMICS EXERCISE
 STRATEGIES FOR TREATMENT OR PREVENTION OF DIABETES USING A CHEMICAL THAT MIMICS EXERCISE DelShawn Fowler Faculty Mentor: Jonathan Fisher, Ph.D. Saint Louis University; Harris-Stowe State University Abstract
STRATEGIES FOR TREATMENT OR PREVENTION OF DIABETES USING A CHEMICAL THAT MIMICS EXERCISE DelShawn Fowler Faculty Mentor: Jonathan Fisher, Ph.D. Saint Louis University; Harris-Stowe State University Abstract
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Zoë Glover. BSc Hons. A thesis submitted to Lancaster University in fulfilment of the requirements for the degree of Doctor of Philosophy.
 Acer, a homologue of the human angiotensin-1 converting enzyme, modulates the response of sleep, glycogen storage, lifespan, fecundity and stress resistance to diet in Drosophila melanogaster By Zoë Glover
Acer, a homologue of the human angiotensin-1 converting enzyme, modulates the response of sleep, glycogen storage, lifespan, fecundity and stress resistance to diet in Drosophila melanogaster By Zoë Glover
Principles of cell signaling Lecture 4
 Principles of cell signaling Lecture 4 Johan Lennartsson Molecular Cell Biology (1BG320), 2014 Johan.Lennartsson@licr.uu.se 1 Receptor tyrosine kinase-induced signal transduction Erk MAP kinase pathway
Principles of cell signaling Lecture 4 Johan Lennartsson Molecular Cell Biology (1BG320), 2014 Johan.Lennartsson@licr.uu.se 1 Receptor tyrosine kinase-induced signal transduction Erk MAP kinase pathway
Structural Variation and Medical Genomics
 Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
100 mm Sucrose. +Berberine +Quinine
 8 mm Sucrose Probability (%) 7 6 5 4 3 Wild-type Gr32a / 2 +Caffeine +Berberine +Quinine +Denatonium Supplementary Figure 1: Detection of sucrose and bitter compounds is not affected in Gr32a / flies.
8 mm Sucrose Probability (%) 7 6 5 4 3 Wild-type Gr32a / 2 +Caffeine +Berberine +Quinine +Denatonium Supplementary Figure 1: Detection of sucrose and bitter compounds is not affected in Gr32a / flies.
Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus
 Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM
 5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM Introduction: Variety of hormones and other molecules regulate the carbohydrates metabolism. Some of these have already been cited in previous sections.
5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM Introduction: Variety of hormones and other molecules regulate the carbohydrates metabolism. Some of these have already been cited in previous sections.
