Exaggerated prostaglandin biosynthesis and its influence on renal resistance in the isolated hydronephrotic rabbit kidney.
|
|
|
- Hester Burke
- 5 years ago
- Views:
Transcription
1 Exaggerated prostaglandin biosynthesis and its influence on renal resistance in the isolated hydronephrotic rabbit kidney. K Nishikawa,, A Morrison, P Needleman J Clin Invest. 1977;59(6): Research Article Basal and hormone-stimulated prostaglandin biosynthesis was compared in isolated perfused rabbit kidneys with and without ureteral obstruction. At 72 h there was enhanced responsiveness to bradykinin in the ureter-obstructed hydronephrotic kidney. The amount of prostaglandin-like substance released from the perfused kidneys by 25 ng of bradykinin was 533+/-163 ng from the ureter-obstructed, 28+/-4 ng from the contralateral, and 26+/-3 ng from the normal kidney. The enhanced response was also noted with angiotensin II and with norepinephrine. This exaggerated responsiveness by the ureter-obstructed kidney could not be explained by decreased prostaglandin (PG) destruction or by decreased renal peptide inactivation (bradykinin or angiotensin). There was no enhanced PG biosynthesis with exogenous arachidonate, suggesting there was no increase in cyclo-oxygenase activity in the ureter-obstructed kidney. Renal tubular transport of PG from medulla to cortex was apparently not essential for the enhanced PG biosynthesis to hormone stimulation since the same exaggerated responses were noted during perfusion with the ureter ligated. The cyclo-oxygenase inhibitor, indomethacin, increased basal perfusion pressure in the obstructed kidney and enhanced the magnitude and duration of the renal vasoconstriction produced by angiotensin II in the hydronephrotic kidney. These results suggest that the local exaggerated biosynthesis of PG may be occurring in the cortical resistance vessels and may be important to the alteration in blood flow and excretory function that occur in [ ] Find the latest version:
2 Exaggerated Prostaglandin Biosynthesis and Its Influence on Renal Resistance in the Isolated Hydronephrotic Rabbit Kidney KOHEI NISHIKAWA, AUBREY MORRISON, and PHILIP NEEDLEMAN From the Departments of Pharmacology and Internal Medicine, Washington University Medical School, St. Louis, Missouri A B S T R A C T Basal and hormone-stimulated prostaglandin biosynthesis was compared in isolated perfused rabbit kidneys with and without ureteral obstruction. At 72 h there was enhanced responsiveness to bradykinin in the ureter-obstructed hydronephrotic kidney. The amount of prostaglandin-like substance released from the perfused kidneys by 25 ng of bradykinin was 533±+ 163 ng from the ureter-obstructed, 28+4 ng from the contralateral, and 26+3 ng from the normal kidney. The enhanced response was also noted with angiotensin II and with norepinephrine. This exaggerated responsiveness by the ureter-obstructed kidney could not be explained by decreased prostaglandin (PG) destruction or by decreased renal peptide inactivation (bradykinin or angiotensin). There was no enhanced PG biosynthesis with exogenous arachidonate, suggesting there was no increase in cyclo-oxygenase activity in the ureter-obstructed kidney. Renal tubular transport of PG from medulla to cortex was apparently not essential for the enhanced PG biosynthesis to hormone stimulation since the same exaggerated responses were noted during perfusion with the ureter ligated. The cyclo-oxygenase inhibitor, indomethacin, increased basal perfusion pressure in the obstructed kidney and enhanced the magnitude and duration of the renal vasoconstriction produced by angiotensin II in the hydronephrotic kidney. These results suggest that the local exaggerated biosynthesis of PG may be occurring in the cortical resistance vessels and may be important to the alteration in blood flow and excretory function that occur in ureteral obstruction. INTRODUCTION Elevation of ureteral pressure in the cat (1) inhibits vasoconstriction elicited by either neuronal stimulation or exogenous catecholamines. Inhibition of prostaglandin biosynthesis by indomethacin, a known inhibitor of the cyclo-oxygenase (2), diminished the inhibitory effects of increases in ureteral pressure. These observations suggested that the effects of ureteral pressure elevations on renal resistance were in part mediated by de novo prostaglandin biosynthesis. Increasing experimental evidence points to a role for prostaglandins in regulating the renal circulation (3). Knowledge of the endogenous factors which control renal blood flow and glomerular filtration is central to the understanding of the basic excretory function of the kidney. Prostaglandin E2 (PGE2)' exhibits significant vasodepressor function (4), is naturetic (5), and its biosynthesis in ureteral obstruction may contribute to the regulation of regional renal resistance, naturesis, and alterations in excretory function. The current studies were undertaken to examine the changes in renal prostaglandin biosynthesis occurring in a ureteral obstruction model of hydronephrosis in rabbits. METHODS Ureter obstruction. New Zealand male white rabbits weighing kg were anesthetized with pentobarbital sodium (30 mglkg, i.v.). Complete ureter obstruction was 'Abbreviations used in this paper: AII, angiotensin II; BK, bradykinin; CLK, contralateral kidneys; HNK, hydronephrotic kidneys; NE, norepinephrine, PG, prostaglandin(s); PGE2, prostaglandin E2; PLS, prostaglandin-like substance. Received for publication 29 October 1976 and in revised form 11 February The Journal of Clinical Investigation Volume 59 June
3 performed through a small abdominal incision by tying a silk suture around the left ureter near the bladder. The wound was closed, bathed in alcohol, and the rabbits were then treated with penicillin G (20,000 U i.m.). The animals were kept in individual cages and had free access to food and water. Kidney perfusion. 3 days after ligation of the ureter, the animals were anesthetized with pentobarbital sodium (30 mg/kg, i.v.) and treated with heparin (250 U/kg, i.v.). The abdominal cavity was opened, and polyethylene catheters were tied into both renal arteries (PG 160, Clay Adams, Div. of Becton, Dickinson & Co., Parsippany, N. J.), and the kidneys were then removed from the animals. Thus, except where indicated during the perfusion experiments, the ureter of both hydronephrotic (HNK) and contralateral (CLK) kidneys were unobstructed. Both HNK and CLK were separately placed in warming jackets and perfused with oxygenated (95%02-5% CG2) Krebs- Henseleit solution (37 C) and a constant flow rate (12 mlmin); thus, changes in perfusion pressure (measured with a Beckman type pressure transducer on a Beckman R-51 1A recorder [Beckman Instruments, Inc., Fullerton, Calif.]) reflect changes in renal resistance. Renal ischemia was achieved by temporarily diverting (with a three-way stopcock) the flow of perfusion fluid away from the renal artery and directly on to the assay organs. Superfused organ system. Isolated assay tissues (rat stomach strip and chick rectum) were continuously superfused by the renal venous effluent (6). Both tissues were particularly sensitive to prostaglandins, especially of the E type. A mixture of antagonists was added to the superfusion fluid which rendered the assay tissues insensitive to catecholamines, acetylcholine, serotonin, and histamine (7). Indomethacin (10 jag/min) was also perfused directly over the assay tissues to eliminate the possibility that the assay tissues might release prostaglandins (8). The response of the perfused kidney to the drug was determined by injecting the agent as a bolus of ml into the Krebs-Henseleit perfusion media just proximal to the arterial cannula (i.e., through the kidney). The direct effect of the agent on the assay organs was tested (as a bolus injection) by adding the agent to the perfusion fluid after it had passed through the kidney (direct testing). Changes in smooth muscle tension were measured with a Harvard smooth muscle transducer (Harvard Apparatus Co., Inc., Millis, Mass.). Prostaglandin release was estimated by comparing contractions of the rat stomach and chick rectum strips to contractions caused by PGE2 standard on these tissues. The assay organs contracted with a s delay after the bolus injection of agonists and were frequently restandardized to known doses of exogenous E2 directly over the strips to correct for any alterations in sensitivity during experiments. The loss of contractile responses to stimulation with bradykinin (BK), angiotensin II, and norepinephrine after pretreatment of the kidney with indomethacin suggested the substances or substance released into the venous effluent were prostaglandins. Subsequent data obtained after labeling the fatty acid pool with [14C]arachidonic acid indicated that the 14C counts released were essentially all as PGE2 (see below) and that pretreatment of the kidney with indomethacin inhibited release of any 14C counts as PGE2- The agonist hormones were randomly infused with the kidneys with each kidney having a dose-response curve to at least two agonists on any one day. BK and angiotensin II were kept as stock solutions at concentrations of 1 mg/ml in saline and refrigerated at -20'C. The arachidonic acid was kept at -20 C in n K. Nishikawa, A. Morrison, and P. Needleman hexane at a concentration of 5 mg/ml and made up in aqueous solution, ph , each day of use. The agonists (angiotensin II [AII], BK, and norepinephrine [NE]) have direct effects on the tissues. In the case of AII, the results were obtained with AII antagonist [Sarl Ile8]- All infused directly over the strips. This negated the direct effect of AII on the tissues. Since BK, as well as PGE2, directly contracts the rat stomach, it was necessary to abolish with indomethacin the prostaglandin (PG) released into the effluent to visualize the direct effects of any unchanged BK coming through the kidney. The difference between these two values gives the amount of PGE2 released as a consequence of hormone receptor interaction. The correction was only necessary with BK doses greater than 100 mg through the kidney since the kidney appeared to metabolize lesser doses, noted by no contraction of assay organs under the indomethacin-treated kidney. The direct effects of NE at doses used were blocked by phenoxybenzamine. The data obtained by direct bioassay were correlated with acid lipid extractions of the PG from the venous effluent and bioassay of the extract. Radiolabeling techniques. The renal phospholipids were prelabeled by infusion of the kidneys with ['4C]arachidonate (55 mci/mmol, Amersham/Searle Corp., Arlington Heights, Ill.) by the procedure previously described (9). Further elucidation of the nature of the prostaglandin-like materials released from the kidney after hormone stimulation was obtained by extracting the renal venous effluent with ethyl acetate after adjustment to ph 3 with formic acid, evaporating the solvent, then chromatographing (with PG standards) the extract on silica gel-g plates in a chloroform:methanol: acetic acid:water solvent (90:8:1:0.8). Spots corresponding to the locus of a PGE2 standard provided the recovery of a material that contracted the rat stomach and the chick rectum (i.e., the eluted material behaved identically to PGE2 in the bioassay system). Genetic hydronephrotic rats. The rats used were a gift of Dr. Denko Susic, University of Tennessee Memorial Research Centre, Knoxville, Tenn. This strain of Memorial Research Centre/Hydronephrosis rats develop congenital hydronephrosis (10). They were studied by superfusing the isolated rat kidney in a cascade as previously described. The rats studied had unilateral hydronephrosis (by gross morphological examination). This was determined by bivalving the kidneys at the end of the experiments. Grossly nonnal kidneys were compared with grossly abnormal (i.e. HNK) in their response to BK bolus injection through the kidneys. All the results are expressed as mean±se. Statistical significance was determined by Student's t test based on paired data. RESULTS Hormone-stimulated PG release. The isolated perfused kidney removed from a rabbit after 3 days of unilateral ureter obstruction (HNK) exhibited a markedly enhanced response to hormone stimulation when compared to CLK and normal kidneys (i.e., from unoperated animals) (Fig. 1 and Table 1). AII was 10 times more potent in the HNK, inducing renal PG release (Fig. 1A). The amounts of prostaglandin-like substance (PLS) released at the doses of 25 and 100 ng were and ng, respectively, for HNK and 30+6 and 100± 18 ng PLS, respectively, for CLK, P < The response was not
4 FIGURE 1 (A) Comparison of the angiotensin dose-response curve for renal PG release in HNK (i.e. ureter obstructed for 3 days but untied during the perfusion experiment), CLK (i.e. no ureter obstruction), and normal kidney (Norm, i.e., no prior surgery). At 3 days, the obstructed kidney weight was 50% greater than the CLK or normal control (i.e., vs g, vs g [n = 12], respectively). The values shown are the mean ±SE, and n designates the number of kidneys. The PG-like activity in the renal venous effluent was calculated from dose-response curves obtained on the rat stomach strips in assays bracketed with a standard PGE2 dose-response curve. (B) Comparison of renal PG release to BK. (C) Renal conversion of exogenous arachidonate to PGE2. significantly different between CLK and normal kidneys. The perfused HNK was extremely sensitive to the PG-releasing action of BK (Fig. 1B). The response to BK was concentration dependent and the doseresponse curve was shifted sharply to the left and indicated a fold increase in BK potency. The amounts of PLS released by a dose of 25 ng were 533±+163 ng for HNK, 28+4 ng for CLK, and 26+3 ng for normal kidney, P < We found that there was no difference in the BK-stimulated PLS release in ureter-tied vs. ureter-untied (during the perfusion) kidneys. Thus, when comparing the dose of BK needed to release 100 ng of PLS, the ureter-tied HNK required 5+2 ng (n = 4, vs. 6+1 with the ureter-untied [Table I]); whereas the ureter-tied CLK required 150±50 ng (n = 4, vs. 228±27 ng in the ureteruntied CLK [Table II]). Basal release in CLK was 1.7±0.6 ng PGEJ/ml of effluent and in HNK it was 4.3±+1.1 ng PGEJml effluent, P < A comparison of several humoral stimuli including All, BK, and NE (four to six times more potent in the HNK), indicate the existence ofa generalized enhanced renal sensitivity in the HNK (Table I). The PGE2- like substance released by hormone stimulation was abolished by indomethacin (1,ug/ml) pretreatment, as indicated by loss of contractile activity of the assay organs. The experiments presented thus far all relied on TABLE I Comparison of Humoral and Nonhumoral Stimuli for Renal PG Biosynthesis Dose needed to release PLS equivalent to 100 ng PGE2 Treatment Units HNK CLK Normal AII (8) ng 10±2 107± BK (7) ng 6±1 228± NE (6) ng 122± ±100 Arachidonate (5) ng 4,900±1,000 5,900±600 6,800+2,000 ATP (8),umole ± min Ischemia (6), ng PLS released 305± The values are the means+se; the number in parentheses indicates the number of kidneys tested. Prostaglandins in Hydronephrosis 1145
5 TABLE II Effect of Indomethacin on Renal Perfusion Pressure with and without Maintenance of the Ureter Obstruction during the Perfusion Experiment HNK Ureter tuntied + INDO Ureter tied + INDO Ureter untied + INDO Ureter tied + INDO CLK Basal PP, mm Hg ,APP, mm Hg All, ng ±10 39±6 17±4 20±11 27±7 21± ±6 67±5 50±8 78±6 49±7 43±11 56±3 48± ±9 94±9 70±9 91±9 76±16 85±15 75±3 76±7 n Normal kidneys were also tested but were not significantly different from CLK. PP, perfusion pressure; APP, change in perfusion pressure produced by the drug treatment; INDO, 1,ug/ml indomethacin infused continuously through the kidney. bioassay techniques for the identification of PGlike activity in the renal venous effluent after hormone stimulation. Such assay techniques cannot readily distinguish between such arachidonate metabolites as PGE2 or PGF2, or such novel products as 6-keto- PGFiaf (9). We therefore repeated the above experiments in perfused HNK that we pretreated with ["4C]- arachidonic acid to label the renal phospholipid pool (9). Acid-lipid extraction and thin-layer chromatography of the renal venous effluent of such labeled kidneys revealed that [14C]PGE2 was the primary product after BK treatment (Fig. 2). Thus was further documented by uising two solvent systems: (a) benzene:dioxane:acetic acid (60:30:3), and (b) chloroform:methanol:acetic acid:water (90:8: E 0 0 BK T (HNK) AA A2 21 FIGURE 2 Radiochromatogram of the acid-lipid extract of the renal venous effluent after stimulation of a hydronephrotic rabbit kidney with 200 ng of BK. The black spots indicate the migration of known unlabeled standards nin on the same plate K. Nishikawa, A. Morrison, and P. Needleman 1:0.8) for the separation, confirming that PGE2 was the major arachidonate metabolite released. Indomethacin pretreatment completely abolished the PGE2 peak in the radiochromatogram. There was a dosedependent increase of [14C]PGE2 release with increasing concentrations of BK, which was especially apparent in the perfused HNK (Fig. 3). The enhanced sensitivity for PLS release (Fig. 1) to BK in the ureterobstructed compared to the contralateral unobstructed kidney is also strikingly evident when comparing the release of [14C]PGE2 from the prelabeled phospholipid pool (Fig. 3). Nonhormonal renal PG stimulation. Renal PG biosynthesis and release also resulted from ATP administration or a brief ischemic episode. The obstructed kidney was more sensitive to these treatments than the CLK and normal kidneys (Table I). PG release induced by exogenously injected arachidonic acid was not different among HNK, CLK, and normal kidneys (Fig. IC and Table I). This suggests one of two possibilities: (a) that there is no difference in cyclo-oxygenase activity in the HNK as compared with CLK, or normal kidneys, or (b) that there is an intrinsic difference between endogenous and exogenously administered arachidonic acid. Further experiments would be necessary to determine if the latter possibility is correct. Renal perfusion pressure. Angiotensin produced a dose-dependent increase in perfusion pressure in the three types of isolated perfused kidneys (Table II). Indomethacin treatment substantially increased basal perfusion pressure in the obstructed kidney (Table II), and enhanced the magnitude and duration of the renal vasoconstriction produced by All in the HNK (Fig. 4), but the indomethacin was without effect on the CLK (Table IL). Furthermore, there was no significant difference in PLS release or perfusion pressure changes
6 produced by All when the ureter was left obstructed, or if the ureter was untied during the time of perfusion (Table II). This experiment suggests that tubular transport of PG to the cortex is not essential for the supersensitivity since there is no bulk fluid transport along the tubule with complete ligation of the ureter. Congenital hydronephrosis. BK injected as a bolus into the isolated perfused rat kidney with genetic hydronephrosis over a range of ng showed a dose-dependent increase in PLS release (P < 0.02) (Fig. 5). Degradation of All, BK, and PGE2. Some possible explanations for the enhanced PLS release by the HNK would include a decreased rate of PG metabolism, or a decreased rate of peptide destruction. Therefore, the degradation of AII, BK, and PG was determined in the three kidney types by directly testing the contractile response of the agonists on the assay tissues and comparing the contractile response produced by the agonists injected through the indomethacin-treated kidney (Fig. 6). About 50% of the exogenously administered PGE2 and about 80% of the exogenous AII was removed by the perfused kidneys in one transit, but there was no difference between the kidney types (Fig. 6A and B). The HNK exhibited a significant reduction (about two to threefold) in BK metabolism compared to CLK and normal kidney (Fig. 6C), but the shift was not enough to account for the 40-fold I- z LJ Ḏj U- LU 0 C14I LU E 0. 4: _ 1000 _ 100 ng 400ng HNK 40 ng C LK- 2000ng BRADYKININ FIGURE 3 Dose-dependent release of ['4C]PGE2 by BK. The renal venous effluent was collected after BK administration during the time when there was demonstrable bioassayable PLS. The pooled effluent was extracted and chromatographed as described in Methods. The values are the means+se (n = 4). Hg r100hn Jo100 INDDO +\-- (TK)1 HNK (8 CLK (D mmt \ J\ --< Hg.100 INDOT r- -- (TK)J ,000 ANGIOTENSIN ng FIGURE 4 The effect of AII on renal resistance in the presence or absence of indomethacin. (A) HNK; (B) CLK; INDO, l,ug/ml indomethacin; TK, continuously infused through the kidney. The duration of increased resistance of the nonindomethacin-treated CLK with the LO-ng dose ofaii is 5 min. The duration of increased resistance at the other doses can be compared to this time coordinate. increase in PLS release produced by BK in the HNK (Table I, Fig. 1). BK injections less than 100 ng did not appear as intact unchanged peptide in the venous effluent and presumably were completely degraded in the kidney. However, at much lower doses than 100 ng BK, very high concentrations of PLS were released from the HNK. Thus, it is not possible to directly compare the mean effective dose (ED50) for PLS release with the ED50 for destruction. Thus, the dose of BK required for 100 ng PLS release is 6+1 ng BK in HNK and 228±+27 ng BK in CLK (P < 0.01). DISCUSSION These results demonstrate that ureteral obstruction initiates an enhanced basal release of PGE2 which is further exaggerated with peptide hormone stimulation. PG are not stored, but release represents de novo biosynthesis. Therefore, ureteral obstruction in some way increases the capacity of the kidney to synthesize PG. BK and AII markedly shift the dose-response curve to the left for the appearance of PG when determined with bioassay or radiochemical techniques (Figs. 1 and 3). These dose-response curves are not strictly comparable since the [14C]arachidonic acid labels only a small portion of the total phospholipid pool which is then only partially released by hormone stimulation. The data, however, do show that the major PG released is PGE2, and for similar total incorporation of label Prostaglandins in Hydronephrosis 1147
7 c, 200 CLO -J a- bc II BRADYKININ ng FIGuRE 5 BK-induced PG biosynthesis in the congenital hydronephrotic rat. The extent of renal involvement was differentiated after the perfusion experiment by the presence or absence of calyceal dilation and distortion. The values represent the mean±se (n = 6). (i.e. 14C counts in the kidney phospholipids), there is a marked release of 14C counts as PGE2 in the HNK as compared with the CLK. Pretreatment ofthe labeled kidney with indomethacin inhibited release of radioactive or bioassayable PGE2. The observation that exogenous arachidonate produced no enhanced release of PGE2 in HNK as compared with the CLK suggests that there may be no difference in cyclo-oxygenase activity in the two experimental conditions. The sensitivity for PG release in the HNK could also be the result of enhanced phospholipase A2 activity or deficient reacylation which determines the availability of endogenous arachidonic acid for the cyclo-oxygenase. An additional possibility is that exogenous arachidonic acid may not mimic the effects of endogenous arachidonate released by the hormone stimulation. Such a situation could occur because of compartmentation of synthetic enzymes or because of differences in concentration achieved when comparing exogenous and endogenous fatty acid. A marked increase in the number and the triglyceride content of the renal medullary interstitial cell lipid droplets and a decreased rate of PGF, synthesis have been observed with either ureter obstruction or indomethacin treatment in rabbits (11, 12). Contradictory data, however, are available in the rat (13), where a reduction of lipid granularity of interstitial cells was observed with indomethacin. Medullary interstitial cell lipid droplet triglycerides have been advocated as storage pools for PG precursors (14) and the renal vasoconstriction after chronic ureter obstruction was suggested to be due to loss of vasodilator PG (15). Our data show no loss of vasodepressor PG but in fact we observe a marked enhanced PG release (Fig. 1) which can be demonstrated even after 6 wk of ureter obstruction (data not shown). Indeed the primary renal pool of the PG precursor arachidonic acid is in the phospholipids (11) rather than in the triglyceride stored in medullary lipid droplets. Thus, we believe that there is a clear dissociation between renal PG biosynthesis and the triglyceride content and number of interstitial lipid cell droplets. The resistance vessels in the kidney reside mainly in the cortex. However, the major area of PG biosynthesis is in the medulla. This suggests that if medullary PG were influencing cortical resistance, PGEf ng1w _W%# %-ANGIOTENSIN ng BRADYKININ ng PGE2 ng AGOESNn RDKNNn FIGURE 6 Renal clearance of PG, AII, and BK. Standards of PGE2, AII, or BK were injected directly (DIR) across rat fundal stomach strips and the response was compred to that obtained by injecting the standards through the kidney (TK). The rat stomach strip contracts in response to added PGE2, All, or BK. The kidneys were treated with indomethacin, thereby eliminating any renal PLS release and permitting evaluation of the unchanged agonist only. The values represent the mean+se for five experiments with each agonist K. Nishikawa, A. Morrison, and P. Needleman
8 then some pathway of movement from medulla to cortex had to be involved. There was no difference in sensitivity to hormone stimulation whether the ureter of the HNK was ligated or released during the perfusion experiments (Table II). This observation suggests that tubular movement of PG, which has previously been described (16, 17), was not critical for expression of the enhanced sensitivity. Another suggestion is that the PG synthesized in the medulla are transported via vascular channels, e.g., vasa recta (18), into the cortex. The vasa recta which loop downwards towards the papilla are mainly associated with juxtamedullary and medullary nephrons which constitute only about 20% of the total nephron population (19). These ascending vas recta never really become cortical vessels but instead drain into the venous system (20). This means that they are not in close apposition to cortical resistance vessels. Therefore, in the absence of close anatomical interrelationships to afferent and efferent glomerular arterioles, any PG synthesized in medulla and transported to corticomedullary area by vasa recta would have to diffuse into and around the cortical resistance vessels. This region of the kidney is known to have significant PG dehydrogenase activity (21). These two factors, lack of significant cortical flow of vasa recta and high cortical PG dehydrogenase activity, make it very unlikely that the vasa recta play a significant role in medullarycortical transport of PG. Since the major resistance vessels are cortical (22), then any PG-like material which influences cortical resistance must either be synthesized locally in the cortex or be transported to the medulla by some other pathway not yet described. Enhanced basal and hormone-stimulated synthesis in the HNK was also evident by the increase in perfusion pressure produced by indomethacin. Indomethacin also enhanced and prolonged the AII-induced vasoconstriction (Fig. 4 and Table II), suggesting that the drug inhibited the synthesis of a vasodilator PG. Cortical biosynthesis of PG has been demonstrated by Larsson and AnggArd (22) and others (23). This observation, coupled with the previous demonstrations of PG biosynthesis in blood vessel walls (24-28), would suggest that local synthesis in cortical resistance vessels may be occurring. This would fit very nicely with the concepts of local circulatory control involving PG biosynthesis. The isolated Krebs perfused rabbit kidney has markedly compromised excretory function (29, 30). It excretes sodium in the same concentration as that of perfusate and does not concentrate urine. This observation makes it very difficult to interpret the enhanced sensitivity in the context of excretory function. However, the finding of enhanced basal and hormonestimulated release of PGE2, a potent vasodepressor and naturetic substance, may have several potential functions. Firstly, the PGE2 may be a protective mechanism initiated to overcome the severe renal vasoconstriction and decrease in glomerular filtration rate normally associated with chronic hydronephrosis (31-35). Secondly, since PGE2 is known to be naturetic (5), the enhanced PG biosynthesis in hydronephrosis may explain the reduction in sodium reabsorption and naturesis associated with ureter obstruction and could conceivably by involved in the enhanced sodium excretion in postobstructive diuresis. Lastly, since PG have have been shown to inhibit the response to antidiuretic hormone (36); the enhanced release in the HNK may explain in part the reduction in concentrating ability which occurs in obstructive uropathy. ACKNOWLE DGME NTS The authors wish to acknowledge the excellent technical assistance of S. E. Denny and A. Wyche. This work was supported by research grants from the National Institutes of Health, HE-14397, and Postdoctoral Fellowship 1 F32 AM REFERENCES 1. Schramm, L. P. and D. E. Carlson Inhibition of renal vasoconstriction by elevated ureteral pressure. Am. J. Physiol. 228: Vane, J. R Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat. New Biol. 231: McGiff, J. C., K. Crowshaw, and H. D. Itskovitz Prostaglandins and renal function. Fed. Proc. 33: Daniels, E. G., J. W. Hinman, B. E. Leach, and E. E. Muirhead Identification of prostaglandin E2 as the principal vasodepressor lipid of rabbit renal medulla. Nature (Lond.). 215: Johnson, H. H., J. P. Herzog, and D. P. Lauler Effect of prostaglandin E1 on renal hemodynamics, sodium and water excretion. Am. J. Physiol. 213: Herbaczynska-Cedro, K. and J. R. Vane Contribution of intrarenal generation of prostaglandin to autoregulation of renal blood flow in the dog. Circ. Res. 23: Eckenfels, A. and J. R. Vane Prostaglandins, oxygen tension and smooth muscle tone. Br. J. Pharmacol. 45: Ferreira, S. H. and J. R. Vane Prostaglandins: their disappearance from and release into the circulation. Nature (Lond.). 216: Isakson, P. C., A. Raz, and P. Needleman Selective incorporation of 14C arachidonic acid into the phospholipids of intact tissues and subsequent metabolism to 14C-prostaglandins. Prostaglandins. 12: Lozzio, B. B., E. Buonocore, and D. Kentera Radiologic and functional studies in rats with hereditary hydronephrosis. Invest. Urol. 10: Comai, K., S. J. Farber, and J. R. Paulsrud Analyses of renal medullary lipid droplets from normal, hydronephrotic, and indomethacin treated rabbits. Lipids. 10: Comai, K., P. Prose, S. J. Farber, and J. R. Paulsrud Correlation of renal medullary prostaglandin con- Prostaglandins in Hydronephrosis 1149
9 tent and renal interstitial cell lipid droplets. Prostaglandins. 6: Limas, C., C. J. Limas, and M. S. Gesell Effects of indomethacin on renomedullary interstitial cells. Lab. Invest. 34: Danon, A., L. C. T. Chang, B. J. Sweetman, A. S. Nies, and J. A. Oates Synthesis of prostaglandins by the rat renal papilla in vitro. Mechanisms of stimulation by angiotensin II. Biochim. Bipohys. Acta. 388: Vaughan, E. D., Jr., E. J. Sorenson, and J. Y. Gillenwater The renal hemodynamic response of chronic unilateral complete ureteral occlusion. Invest. Urol. 8: Frolich, J. C., T. W. Wilson, B. J. Sweetman, A. S. Nies, K. Carr, J. T. Watson, and J. A. Oates Urinary prostaglandins. Identification and origin.j. Clin. Invest. 55: Frolich, J. C., B. J. Sweetman, K. Carr, and J. A. Oates Prostaglandin synthesis in rabbit renal medulla. Life Sci. 17: Barger, A. C. and J. A. Herd Renal vascular anatomy and distribution of blood flow. Handb. Physiol. (Sect. 8. Renal Physiology) Schmidt-Nielsen, B., and R. O'Dell Structure and concentrating mechanism in the mammalian kidney. Am. J. Physiol. 200: Stein, J The renal circulation. In The Kidney. B. M. Brenner and F. C. Rector, Jr., editors. W. B. Saunders Company, Philadelphia, Pa Larsson, C. and E. Anggard Regional differences in the formation and metabolism of prostaglandin in the rabbit kidney. Eur. J. Pharmacol. 21: Thurau, K Renal hemodynamics. Am. J. Med. 36: Pong, S.-S. and L. Levine Biosynthesis of prostaglandins in rabbit renal cortex. Res. Commun. Chem. Pathol. Pharmacol. 13: Needleman, P., G. R. Marshall, and J. R. Douglas, Jr Prostaglandin release from vasculature by angiotensin II: Dissociation from lipolysis. Eur. J. Pharmacol. 23: Aiken, J. W Effects of prostaglandin synthesis inhibitors on angiotensin tachyphylaxis in the isolated coeliac and mesenteric arteries of the rabbit. Pol. J. Pharmacol. Pharm. 26: Alexander, R. W. and M. A. Gimbrone, Jr Stimulation of prostaglandin E synthesis in cultured human umbilical vein smooth muscle cells. Proc. Natl. Acad. Sci. U. S. A. 73: Terragno, D. A., K. Crowshaw, N. A. Terragno, and J. C. McGiff Prostaglandin synthesis by bovine mesenteric arteries and veins. Circ. Res. 36 (Suppl. I): I-76- I Blumberg, A. L., S. E. Denny, G. R. Marshall, and P. Needleman Blood vessel hormone interactions: angiotensin, bradykinin, and prostaglandins. Am. J. Physiol. Heart Circ. Physiol. 1: H305-H Colina-Chourio, J., J. C. McGiff, M. P. Miller, and A. Nasjletti Possible influence of intrarenal generation of kinins on prostaglandin release from the rabbit perfused kidney. Br. J. Pharmacol. 58: Regoli, D. and R. Gauthier Site of action of angiotensin and other vasoconstrictors on the kidney. Can. J. Physiol. Pharmacol. 49: Idbohm, H. and A. Muren Renal blood flow in experimental hydronephrosis. Acta Physiol. Scand. 38: Kerr, W. S., Jr Effect of complete ureteral obstruction for one week on kidney function. J. Appl. Physiol. 6: Pridgen, W. R., D. M. Woodhead, and R. K. Younger Alterations in renal function produced by ureteral obstruction. Determination of critical obstruction time in relation to renal survival. J. Amer. Med. Assoc. 178: Nagle, R. B., R. E. Bulger, R. E. Cutler, H. R. Jervis, and E. P. Benditt Unilateral obstructive nephropathy in the rabbit. I. Early morphologic, physiologic, and histochemical changes. Lab. Invest. 28: Schubert, G. E., R. Staudhammer, K. Rolle, and U. Kneissler Tubular dimensions and juxtaglomerular granulation index in rat kidneys after unilateral obstruction of the ureter: A study of the morphogenesis of hydronephrosis. Urol. Res. 3: Grantham, J. J. and J. Orloff Effect of prostaglandin E1 on the permeability response of the isolated collecting tubule to vasopressin, adenosine 3',5'- monophosphate, and theophylline. J. Clin. Invest. 47: K. Nishikawa, A. Morrison, and P. Needleman
Contribution of Thromboxane to Renal Resistance Changes in the Isolated Perfused Hydronephrotic Rabbit Kidney
 486 Contribution of Thromboxane to Renal Resistance Changes in the Isolated Perfused Hydronephrotic Rabbit Kidney Akiyoshi Kawasaki and Philip Needleman From the Department of Pharmacology, Washington
486 Contribution of Thromboxane to Renal Resistance Changes in the Isolated Perfused Hydronephrotic Rabbit Kidney Akiyoshi Kawasaki and Philip Needleman From the Department of Pharmacology, Washington
Stimulation of Prostaglandin Biosynthesis by Adenine Nucleotides
 Stimulation of Prostaglandin Biosynthesis by Adenine Nucleotides PROFILE OF PROSTAGLANDIN RELEASE BY PERFUSED ORGANS SyPhilip Needleman, Mark S. Minkes, and James R. Douglas, Jr. ABSTRACT Prostaglandin
Stimulation of Prostaglandin Biosynthesis by Adenine Nucleotides PROFILE OF PROSTAGLANDIN RELEASE BY PERFUSED ORGANS SyPhilip Needleman, Mark S. Minkes, and James R. Douglas, Jr. ABSTRACT Prostaglandin
RENAL PHYSIOLOGY DR.CHARUSHILA RUKADIKAR ASSISTANT PROFESSOR PHYSIOLOGY
 RENAL PHYSIOLOGY DR.CHARUSHILA RUKADIKAR ASSISTANT PROFESSOR PHYSIOLOGY GROSS ANATOMY Location *Bean-shaped *Retroperitoneal *At level of T12 L1 vertebrae. *The right kidney lies slightly inferior to left
RENAL PHYSIOLOGY DR.CHARUSHILA RUKADIKAR ASSISTANT PROFESSOR PHYSIOLOGY GROSS ANATOMY Location *Bean-shaped *Retroperitoneal *At level of T12 L1 vertebrae. *The right kidney lies slightly inferior to left
Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion.
 The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
Copyright 2009 Pearson Education, Inc. Copyright 2009 Pearson Education, Inc. Figure 19-1c. Efferent arteriole. Juxtaglomerular apparatus
 /6/0 About this Chapter Functions of the Kidneys Anatomy of the urinary system Overview of kidney function Secretion Micturition Regulation of extracellular fluid volume and blood pressure Regulation of
/6/0 About this Chapter Functions of the Kidneys Anatomy of the urinary system Overview of kidney function Secretion Micturition Regulation of extracellular fluid volume and blood pressure Regulation of
Prostaglandins and Renal Function: Implications for the Activity of Diuretic Agents
 1 Prostaglandins and Renal Function: Implications for the Activity of Diuretic Agents Downloaded via 148.251.232.83 on October 7, 2018 at 01:21:17 (UTC). See https://pubs.acs.org/sharingguidelines for
1 Prostaglandins and Renal Function: Implications for the Activity of Diuretic Agents Downloaded via 148.251.232.83 on October 7, 2018 at 01:21:17 (UTC). See https://pubs.acs.org/sharingguidelines for
MAJOR FUNCTIONS OF THE KIDNEY
 MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015
 RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015 1. In which abdominal cavity do the kidneys lie? a) Peritoneum. b) Anteperitoneal. c) Retroperitoneal. d) Parietal peritoneal 2. What is the
RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015 1. In which abdominal cavity do the kidneys lie? a) Peritoneum. b) Anteperitoneal. c) Retroperitoneal. d) Parietal peritoneal 2. What is the
Urinary System. consists of the kidneys, ureters, urinary bladder and urethra
 Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
Lab Activity 31. Anatomy of the Urinary System. Portland Community College BI 233
 Lab Activity 31 Anatomy of the Urinary System Portland Community College BI 233 Urinary System Organs Kidneys Urinary bladder: provides a temporary storage reservoir for urine Paired ureters: transport
Lab Activity 31 Anatomy of the Urinary System Portland Community College BI 233 Urinary System Organs Kidneys Urinary bladder: provides a temporary storage reservoir for urine Paired ureters: transport
Kidney Functions Removal of toxins, metabolic wastes, and excess ions from the blood Regulation of blood volume, chemical composition, and ph
 The Urinary System Urinary System Organs Kidneys are major excretory organs Urinary bladder is the temporary storage reservoir for urine Ureters transport urine from the kidneys to the bladder Urethra
The Urinary System Urinary System Organs Kidneys are major excretory organs Urinary bladder is the temporary storage reservoir for urine Ureters transport urine from the kidneys to the bladder Urethra
modulating the tubuloglomerular feed-back mechanism in the canine kidney; Sciences Center, 4200 East Ninth Avenue, Denver, CO 80262, U.S.A.
 J. Physiol. (1986), 380, pp. 35-43 35 With 3 text-figures Printed in Great Britain RENAL VASOCONSTRICTOR RESPONSE TO HYPERTONIC SALINE IN THE DOG: EFFECTS OF PROSTAGLANDINS, INDOMETHACIN AND THEOPHYLLINE
J. Physiol. (1986), 380, pp. 35-43 35 With 3 text-figures Printed in Great Britain RENAL VASOCONSTRICTOR RESPONSE TO HYPERTONIC SALINE IN THE DOG: EFFECTS OF PROSTAGLANDINS, INDOMETHACIN AND THEOPHYLLINE
A. Incorrect! The urinary system is involved in the regulation of blood ph. B. Correct! The urinary system is involved in the synthesis of vitamin D.
 Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Chapter 25 The Urinary System
 Chapter 25 The Urinary System 10/30/2013 MDufilho 1 Kidney Functions Removal of toxins, metabolic wastes, and excess ions from the blood Regulation of blood volume, chemical composition, and ph Gluconeogenesis
Chapter 25 The Urinary System 10/30/2013 MDufilho 1 Kidney Functions Removal of toxins, metabolic wastes, and excess ions from the blood Regulation of blood volume, chemical composition, and ph Gluconeogenesis
Outline the functional anatomy, and the physiological factors, that determine oxygen delivery to the renal medulla.
 2011-2-21 Outline the functional anatomy, and the physiological factors, that determine oxygen delivery to the renal medulla. Oxygen delivery = Blood flow CaO 2 Where Blood flow determined by (arterial
2011-2-21 Outline the functional anatomy, and the physiological factors, that determine oxygen delivery to the renal medulla. Oxygen delivery = Blood flow CaO 2 Where Blood flow determined by (arterial
Urinary System Laboratory
 Urinary System Laboratory 1 Adrenal gland Organs of The Urinary System Renal artery and vein Kidney Ureter Urinary bladder Figure 26.1 2 Urethra Functions of the urinary system organs: Urethra expels urine
Urinary System Laboratory 1 Adrenal gland Organs of The Urinary System Renal artery and vein Kidney Ureter Urinary bladder Figure 26.1 2 Urethra Functions of the urinary system organs: Urethra expels urine
Functions of the kidney
 Physiology of Urinary tract Kidney, Ureter, Urinary bladder Urethra Kidney function Excretion Physiology of volume regulation Functions of the kidney Excretion of dangerous substances endogenous (metabolites):
Physiology of Urinary tract Kidney, Ureter, Urinary bladder Urethra Kidney function Excretion Physiology of volume regulation Functions of the kidney Excretion of dangerous substances endogenous (metabolites):
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Chapter 23. The Nephron. (functional unit of the kidney
 Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
Kidney and urine formation
 Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Human Urogenital System 26-1
 Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Urinary Physiology. Chapter 17 Outline. Kidney Function. Chapter 17
 Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
The Urinary System Pearson Education, Inc.
 26 The Urinary System Introduction The urinary system does more than just get rid of liquid waste. It also: Regulates plasma ion concentrations Regulates blood volume and blood pressure Stabilizes blood
26 The Urinary System Introduction The urinary system does more than just get rid of liquid waste. It also: Regulates plasma ion concentrations Regulates blood volume and blood pressure Stabilizes blood
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
CHAPTER 25 URINARY. Urinary system. Kidneys 2 Ureters 2 Urinary Bladder 1 Urethra 1. functions
 CHAPTER 25 URINARY Kidneys 2 Ureters 2 Urinary Bladder 1 Urethra 1 fluid waste elimination secretion of wastes control blood volume and BP control blood ph electrolyte levels RBC levels hormone production
CHAPTER 25 URINARY Kidneys 2 Ureters 2 Urinary Bladder 1 Urethra 1 fluid waste elimination secretion of wastes control blood volume and BP control blood ph electrolyte levels RBC levels hormone production
5.Which part of the nephron removes water, ions and nutrients from the blood?
 Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
19. RENAL PHYSIOLOGY ROLE OF THE URINARY SYSTEM THE URINARY SYSTEM. Components and function. V BS 122 Physiology II 151 Class of 2011
 19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
1. Urinary System, General
 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
Renal-Related Questions
 Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1
 BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
Excretory Lecture Test Questions Set 1
 Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Basic Functions of the Kidneys
 Dr. Adelina Vlad Basic Functions of the Kidneys Eliminate plasma METABOLIC WASTE PRODUCTS and FOREIGN COMPOUNDS The kidney are the primary means for eliminating metabolic waste products (urea, creatinine,
Dr. Adelina Vlad Basic Functions of the Kidneys Eliminate plasma METABOLIC WASTE PRODUCTS and FOREIGN COMPOUNDS The kidney are the primary means for eliminating metabolic waste products (urea, creatinine,
Urinary bladder provides a temporary storage reservoir for urine
 Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Lab 19 The Urinary System
 Lab 19 The Urinary System Laboratory Objectives Identify and describe the micro- and macroscopic anatomy of the kidney. Track the blood flow in and out of the kidney. Compare blood, glomerular filtrate,
Lab 19 The Urinary System Laboratory Objectives Identify and describe the micro- and macroscopic anatomy of the kidney. Track the blood flow in and out of the kidney. Compare blood, glomerular filtrate,
Figure 26.1 An Introduction to the Urinary System
 Chapter 26 Figure 26.1 An Introduction to the Urinary System Components of the Urinary System Kidney Produces urine Ureter Transports urine toward the urinary bladder Urinary Bladder Temporarily stores
Chapter 26 Figure 26.1 An Introduction to the Urinary System Components of the Urinary System Kidney Produces urine Ureter Transports urine toward the urinary bladder Urinary Bladder Temporarily stores
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Evidence for an In Vivo Antagonism between Vasopressin
 Evidence for an In Vivo Antagonism between Vasopressin and Prostaglandin in the Mammalian Kidney ROBERT J. ANDERSON, TomAs BuRL, Kxrrm M. McDoNALD, and ROBERT W. SCHRIER with the technical assistance of
Evidence for an In Vivo Antagonism between Vasopressin and Prostaglandin in the Mammalian Kidney ROBERT J. ANDERSON, TomAs BuRL, Kxrrm M. McDoNALD, and ROBERT W. SCHRIER with the technical assistance of
KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin
![KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin KD02 [Mar96] [Feb12] Which has the greatest renal clearance? A. PAH B. Glucose C. Urea D. Water E. Inulin](/thumbs/87/96174332.jpg) Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
Renal Physiology MCQ KD01 [Mar96] [Apr01] Renal blood flow is dependent on: A. Juxtaglomerular apparatus B. [Na+] at macula densa C. Afferent vasodilatation D. Arterial pressure (poorly worded/recalled
The Prostaglandin System
 454 The Prostaglandin System A Role in Canine Baroreceptor of Renin Release JOANN L. DATA, JOHN G. GERBER, WILLIAM J. CRUMP, JURGEN C. FROLICH, JOHN W. HOLLIFIELD, AND ALAN S. NIES SUMMARY We used the
454 The Prostaglandin System A Role in Canine Baroreceptor of Renin Release JOANN L. DATA, JOHN G. GERBER, WILLIAM J. CRUMP, JURGEN C. FROLICH, JOHN W. HOLLIFIELD, AND ALAN S. NIES SUMMARY We used the
Human Anatomy Unit 3 URINARY SYSTEM
 Human Anatomy Unit 3 URINARY SYSTEM In Anatomy Today Components Kidneys Ureters Urinary bladder Urethra Functions Storage of urine Bladder stores up to 1 L of urine Excretion of urine Transport of urine
Human Anatomy Unit 3 URINARY SYSTEM In Anatomy Today Components Kidneys Ureters Urinary bladder Urethra Functions Storage of urine Bladder stores up to 1 L of urine Excretion of urine Transport of urine
URINARY SYSTEM. These organs lie posterior or inferior to the. (membrane).
 URINARY SYSTEM I. INTRODUCTION Each kidney is made up of about a million tiny tubules called nephrons. Each nephron individually filters the blood and makes urine and it does the job completely, from start
URINARY SYSTEM I. INTRODUCTION Each kidney is made up of about a million tiny tubules called nephrons. Each nephron individually filters the blood and makes urine and it does the job completely, from start
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
CONTROLLING THE INTERNAL ENVIRONMENT
 AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron
 Kidney International, Vol. 19 (1981), pp. 797 81 Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron LEON G. FINE and MICHAEL A. KIRSCHENBAUM Division of Nephroiogy,
Kidney International, Vol. 19 (1981), pp. 797 81 Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron LEON G. FINE and MICHAEL A. KIRSCHENBAUM Division of Nephroiogy,
2) This is a Point and Click question. You must click on the required structure.
 Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Physiology of Excretory Systems
 Physiology of Excretory Systems Fig 12-2 (a) Urea is formed by the ornithine-urea cycle in most vertebrates. Because ATP is required for the first step, nitrogen excretion in ureotelic animals is more
Physiology of Excretory Systems Fig 12-2 (a) Urea is formed by the ornithine-urea cycle in most vertebrates. Because ATP is required for the first step, nitrogen excretion in ureotelic animals is more
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
describe the location of the kidneys relative to the vertebral column:
 Basic A & P II Dr. L. Bacha Chapter Outline (Martini & Nath 2010) list the three major functions of the urinary system: by examining Fig. 24-1, list the organs of the urinary system: describe the location
Basic A & P II Dr. L. Bacha Chapter Outline (Martini & Nath 2010) list the three major functions of the urinary system: by examining Fig. 24-1, list the organs of the urinary system: describe the location
Histology Urinary system
 Histology Urinary system Urinary system Composed of two kidneys, two ureters, the urinary bladder, and the urethra, the urinary system plays a critical role in: 1- Blood filtration,(filtration of cellular
Histology Urinary system Urinary system Composed of two kidneys, two ureters, the urinary bladder, and the urethra, the urinary system plays a critical role in: 1- Blood filtration,(filtration of cellular
renoprotection therapy goals 208, 209
 Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Chapter 17: Urinary System
 Introduction Chapter 17: Urinary System Organs of the Urinary System REFERENCE FIGURE 17.1 2 kidneys filters the blood 2 ureters transport urine from the kidneys to the urinary bladder Urinary bladder
Introduction Chapter 17: Urinary System Organs of the Urinary System REFERENCE FIGURE 17.1 2 kidneys filters the blood 2 ureters transport urine from the kidneys to the urinary bladder Urinary bladder
P215 Spring 2018: Renal Physiology Chapter 18: pp , Chapter 19: pp ,
 P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
Control of blood tissue blood flow. Faisal I. Mohammed, MD,PhD
 Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog
 Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
Chapter 25: Urinary System
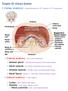 Chapter 25: Urinary System I. Kidney anatomy: retroperitoneal from 12 th thoracic to 3 rd lumbar area A. External anatomy: hilus is the indentation 1. Adrenal gland: in the fat at the superior end of each
Chapter 25: Urinary System I. Kidney anatomy: retroperitoneal from 12 th thoracic to 3 rd lumbar area A. External anatomy: hilus is the indentation 1. Adrenal gland: in the fat at the superior end of each
November 30, 2016 & URINE FORMATION
 & URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
& URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate
 Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
INHIBITION BY CLINICALLY USED DYES OF PROSTAGLANDIN
 Br. J. Pharmac. (1981), 72, 715-721 INHIBITION BY CLINICALLY USED DYES OF PROSTAGLANDIN INACTIVATION IN RAT AND HUMAN LUNG Y.S. BAKHLE Department of Pharmacology, Institute of Basic Medical Sciences, Royal
Br. J. Pharmac. (1981), 72, 715-721 INHIBITION BY CLINICALLY USED DYES OF PROSTAGLANDIN INACTIVATION IN RAT AND HUMAN LUNG Y.S. BAKHLE Department of Pharmacology, Institute of Basic Medical Sciences, Royal
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
AP2, Lab 7 - THE URINARY SYSTEM
 AP2, Lab 7 - THE URINARY SYSTEM I. SYSTEM COMPONENTS (Figs. 25.1 25.4) KIDNEYS Each kidney contains approx. 1,000,000 tubular NEPHRONS which produce FILTRATE from the plasma and then add to or take from
AP2, Lab 7 - THE URINARY SYSTEM I. SYSTEM COMPONENTS (Figs. 25.1 25.4) KIDNEYS Each kidney contains approx. 1,000,000 tubular NEPHRONS which produce FILTRATE from the plasma and then add to or take from
Physiology Lecture 2. What controls GFR?
 Physiology Lecture 2 Too much blood is received by the glomerular capillaries, this blood contains plasma, once this plasma enters the glomerular capillaries it will be filtered to bowman s space. The
Physiology Lecture 2 Too much blood is received by the glomerular capillaries, this blood contains plasma, once this plasma enters the glomerular capillaries it will be filtered to bowman s space. The
Urinary System. BSC 2086 A & P 2 Professor Tcherina Duncombe Palm Beach State College
 Urinary System BSC 2086 A & P 2 Professor Tcherina Duncombe Palm Beach State College Filter plasma, separate and eliminate wastes Functions Regulate blood volume and pressure Regulate osmolarity of body
Urinary System BSC 2086 A & P 2 Professor Tcherina Duncombe Palm Beach State College Filter plasma, separate and eliminate wastes Functions Regulate blood volume and pressure Regulate osmolarity of body
General Anatomy of Urinary System
 General Anatomy of Urinary System URINARY SYSTEM ORGANS Kidneys (2) Ureters (2) Urinary bladder Urethra KIDNEY FUNCTIONS Control blood volume and composition KIDNEY FUNCTIONS Filter blood plasma, eliminate
General Anatomy of Urinary System URINARY SYSTEM ORGANS Kidneys (2) Ureters (2) Urinary bladder Urethra KIDNEY FUNCTIONS Control blood volume and composition KIDNEY FUNCTIONS Filter blood plasma, eliminate
Influence of Prostaglandin E 2, Indomethacin, and Reserpine on Renal Vascular Responses to Nerve Stimulation, Pressor and Depressor Hormones
 72 Influence of Prostaglandin E 2, Indomethacin, and Reserpine on Renal Vascular Responses to Nerve Stimulation, Pressor and Depressor Hormones GREGORY D. FINK, BARRY M. CHAPNICK, MICHAEL R. GOLDBERG,
72 Influence of Prostaglandin E 2, Indomethacin, and Reserpine on Renal Vascular Responses to Nerve Stimulation, Pressor and Depressor Hormones GREGORY D. FINK, BARRY M. CHAPNICK, MICHAEL R. GOLDBERG,
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1)
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1) Dr. Attila Nagy 2017 Functional role of the kidney 1.Homeostasis of fluid compartments (isosmia, isovolemia, isoionia, isohydria,) 2. Elimination
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1) Dr. Attila Nagy 2017 Functional role of the kidney 1.Homeostasis of fluid compartments (isosmia, isovolemia, isoionia, isohydria,) 2. Elimination
QUIZ/TEST REVIEW NOTES SECTION 1 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 CHAPTER 19
![QUIZ/TEST REVIEW NOTES SECTION 1 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 CHAPTER 19 QUIZ/TEST REVIEW NOTES SECTION 1 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 CHAPTER 19](/thumbs/77/74887026.jpg) QUIZ/TEST REVIEW NOTES SECTION 1 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 CHAPTER 19 Learning Objectives: Differentiate the following processes: filtration, reabsorption, secretion,
QUIZ/TEST REVIEW NOTES SECTION 1 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 CHAPTER 19 Learning Objectives: Differentiate the following processes: filtration, reabsorption, secretion,
Osmoregulation and Renal Function
 1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
URINARY SYSTEM ANATOMY
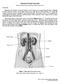 URINARY SYSTEM ANATOMY Adapted from Human Anatomy & Physiology Marieb and Hoehn (9 th ed.) OVERVIEW Metabolism of nutrients by the body produces wastes that must be removed from the body. Although excretory
URINARY SYSTEM ANATOMY Adapted from Human Anatomy & Physiology Marieb and Hoehn (9 th ed.) OVERVIEW Metabolism of nutrients by the body produces wastes that must be removed from the body. Although excretory
Cardiovascular System B L O O D V E S S E L S 2
 Cardiovascular System B L O O D V E S S E L S 2 Blood Pressure Main factors influencing blood pressure: Cardiac output (CO) Peripheral resistance (PR) Blood volume Peripheral resistance is a major factor
Cardiovascular System B L O O D V E S S E L S 2 Blood Pressure Main factors influencing blood pressure: Cardiac output (CO) Peripheral resistance (PR) Blood volume Peripheral resistance is a major factor
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Chapter 32 Excretion
 Chapter 32 Excretion 3.4 Learning Objectives 3.4.6 The Excretory System in Humans 1. Role of the excretory system in homeostasis. 2. Importance of temperature regulation within the body. 3. Outline the
Chapter 32 Excretion 3.4 Learning Objectives 3.4.6 The Excretory System in Humans 1. Role of the excretory system in homeostasis. 2. Importance of temperature regulation within the body. 3. Outline the
BIOH122 Human Biological Science 2
 BIOH122 Human Biological Science 2 Session 16 Urinary System 1 The Kidneys Bioscience Department Endeavour College of Natural Health endeavour.edu.au Session Plan o Functions of Urinary system o The Kidneys:
BIOH122 Human Biological Science 2 Session 16 Urinary System 1 The Kidneys Bioscience Department Endeavour College of Natural Health endeavour.edu.au Session Plan o Functions of Urinary system o The Kidneys:
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Copyright 2010 Pearson Education, Inc. Urinary System
 Urinary System What are the organs that comprise the urinary system? Urinary System Organs Kidneys Urinary bladder Ureters Urethra Hepatic veins (cut) Esophagus (cut) Inferior vena cava Adrenal gland Aorta
Urinary System What are the organs that comprise the urinary system? Urinary System Organs Kidneys Urinary bladder Ureters Urethra Hepatic veins (cut) Esophagus (cut) Inferior vena cava Adrenal gland Aorta
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
A. Correct! Flushing acids from the system will assist in re-establishing the acid-base equilibrium in the blood.
 OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
Urinary System VASTACCESS, INC.
 Urinary System www.vastaccess.com 2 Urinary Tract Kidney Ureter Urinary Bladder Urethra Prostate (male) Membranous (male) Spongy (male) 3 Kidney Relations Suprarenal (Adrenal) Glands Liver Duodenum Transverse
Urinary System www.vastaccess.com 2 Urinary Tract Kidney Ureter Urinary Bladder Urethra Prostate (male) Membranous (male) Spongy (male) 3 Kidney Relations Suprarenal (Adrenal) Glands Liver Duodenum Transverse
Urinary System kidneys, ureters, bladder & urethra
 Urinary System kidneys, ureters, bladder & urethra Kidney Function Filters blood removes waste products conserves salts, glucose, proteins, nutrients and water Produces urine Endocrine functions regulates
Urinary System kidneys, ureters, bladder & urethra Kidney Function Filters blood removes waste products conserves salts, glucose, proteins, nutrients and water Produces urine Endocrine functions regulates
The ability of the kidneys to regulate extracellular fluid volume by altering sodium
 REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D.
 Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM
 RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM RENAL PHYSIOLOGY - ANATOMY Glomerulus + renal tubule Each kidney has 1.3 million nephrons Cortical nephrons (85%) have shorter Loop of Henle than Juxtamedullary nephrons
RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM RENAL PHYSIOLOGY - ANATOMY Glomerulus + renal tubule Each kidney has 1.3 million nephrons Cortical nephrons (85%) have shorter Loop of Henle than Juxtamedullary nephrons
Urinary system. Kidney anatomy Renal cortex Renal. Nephrons
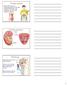 Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Diagram of the inner portions of the kidney
 Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Urinary System. Dr. Ahmed Maher Dr. Ahmed Manhal
 Urinary System Dr. Ahmed Maher Dr. Ahmed Manhal Presentation Map Kidney (cortex & medulla). Nephron. Duct system. Juxtaglomerular apparatus. Ureter, bladder & urethra. Definition & General Structure The
Urinary System Dr. Ahmed Maher Dr. Ahmed Manhal Presentation Map Kidney (cortex & medulla). Nephron. Duct system. Juxtaglomerular apparatus. Ureter, bladder & urethra. Definition & General Structure The
Mechanism: 1- waterretention from the last part of the nephron which increases blood volume, venous return EDV, stroke volume and cardiac output.
 Blood pressure regulators: 1- Short term regulation:nervous system Occurs Within secondsof the change in BP (they are short term because after a while (2-3 days) they adapt/reset the new blood pressure
Blood pressure regulators: 1- Short term regulation:nervous system Occurs Within secondsof the change in BP (they are short term because after a while (2-3 days) they adapt/reset the new blood pressure
Urinary system. Urinary system
 INTRODUCTION. Several organs system Produce urine and excrete it from the body Maintenance of homeostasis. Components. two kidneys, produce urine; two ureters, carry urine to single urinary bladder for
INTRODUCTION. Several organs system Produce urine and excrete it from the body Maintenance of homeostasis. Components. two kidneys, produce urine; two ureters, carry urine to single urinary bladder for
Urinary System and Excretion. Bio105 Lecture 20 Chapter 16
 Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D.
 Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Chapter 24: The Urinary System
 Chapter 24: The Urinary System Overview of kidney functions n Regulation of blood ionic composition n Regulation of blood ph n Regulation of blood volume n Regulation of blood pressure n Maintenance of
Chapter 24: The Urinary System Overview of kidney functions n Regulation of blood ionic composition n Regulation of blood ph n Regulation of blood volume n Regulation of blood pressure n Maintenance of
URINARY SYSTEM CHAPTER 28 I ANATOMY OF THE URINARY SYSTEM. Student Name
 Student Name CHAPTER 28 URINARY SYSTEM L iving produces wastes. Wherever people live or work or play, wastes accumulate. To keep these areas healthy, there must be a method of disposing of these wastes
Student Name CHAPTER 28 URINARY SYSTEM L iving produces wastes. Wherever people live or work or play, wastes accumulate. To keep these areas healthy, there must be a method of disposing of these wastes
BIOL2030 Human A & P II -- Exam 6
 BIOL2030 Human A & P II -- Exam 6 Name: 1. The kidney functions in A. preventing blood loss. C. synthesis of vitamin E. E. making ADH. B. white blood cell production. D. excretion of metabolic wastes.
BIOL2030 Human A & P II -- Exam 6 Name: 1. The kidney functions in A. preventing blood loss. C. synthesis of vitamin E. E. making ADH. B. white blood cell production. D. excretion of metabolic wastes.
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Nephron Anatomy Nephron Anatomy
 Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
BIOLOGY - CLUTCH CH.44 - OSMOREGULATION AND EXCRETION.
 !! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
!! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
CASE 13. What neural and humoral pathways regulate arterial pressure? What are two effects of angiotensin II?
 CASE 13 A 57-year-old man with long-standing diabetes mellitus and newly diagnosed hypertension presents to his primary care physician for follow-up. The patient has been trying to alter his dietary habits
CASE 13 A 57-year-old man with long-standing diabetes mellitus and newly diagnosed hypertension presents to his primary care physician for follow-up. The patient has been trying to alter his dietary habits
