Serotonin Deficiency Exacerbates Acetaminophen-Induced Liver Toxicity In Mice
|
|
|
- Elvin Hensley
- 6 years ago
- Views:
Transcription
1 Serotonin Deficiency Exacerbates Acetaminophen-Induced Liver Toxicity In Mice The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version Accessed Citable Link Terms of Use Zhang, Jingyao, Sidong Song, Qing Pang, Ruiyao Zhang, Lei Zhou, Sushun Liu, Fandi Meng, Qifei Wu, and Chang Liu Serotonin Deficiency Exacerbates Acetaminophen-Induced Liver Toxicity In Mice. Scientific Reports 5 (1): doi: /srep doi: /srep08098 June 7, :29:32 PM EDT This article was downloaded from Harvard University's DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at (Article begins on next page)
2 OPEN SUBJECT AREAS: GASTROINTESTINAL DISEASES TOXICOLOGY Received 4 August 2014 Accepted 6 January 2015 Published 29 January 2015 Correspondence and requests for materials should be addressed to C.L. (liuchangdoctor@ gmail.com) Serotonin Deficiency Exacerbates Acetaminophen-Induced Liver Toxicity In Mice Jingyao Zhang 1, Sidong Song 1, Qing Pang 1, Ruiyao Zhang 1, Lei Zhou 1,2, Sushun Liu 1, Fandi Meng 1, Qifei Wu 3 & Chang Liu 1 1 Department of Hepatobiliary Surgery, The First Affiliated Hospital of Xi an Jiaotong University; NO.277 Yanta West Road, Xi an Shaanxi , People s Republic of China, 2 Departments of Medicine (Division of Molecular and Vascular Biology), Center for Vascular Biology Research, Beth Israel Deaconess Medical Center and Harvard Medical School. Boston. U.S.A, 3 Department of Thoracic Surgery, The First Affiliated Hospital of Xi an Jiaotong University; NO.277 Yanta West Road, Xi an Shaanxi , People s Republic of China. Acetaminophen (APAP) overdose is a major cause of acute liver failure. Peripheral 5-hydroxytryptamine (serotonin, 5-HT) is a cytoprotective neurotransmitter which is also involved in the hepatic physiological and pathological process. This study seeks to investigate the mechanisms involved in APAP-induced hepatotoxicity, as well as the role of 5-HT in the liver s response to APAP toxicity. We induced APAP hepatotoxicity in mice either sufficient of serotonin (wild-type mice and TPH1-/- plus 5- Hydroxytryptophan (5-HTP)) or lacking peripheral serotonin (Tph1-/- and wild-type mice plus p-chlorophenylalanine (PCPA)).Mice with sufficient 5-HT exposed to acetaminophen have a significantly lower mortality rate and a better outcome compared with mice deficient of 5-HT. This difference is at least partially attributable to a decreased level of inflammation, oxidative stress and endoplasmic reticulum (ER) stress, Glutathione (GSH) depletion, peroxynitrite formation, hepatocyte apoptosis, elevated hepatocyte proliferation, activation of 5-HT2B receptor, less activated c-jun NH 2 -terminal kinase (JNK) and hypoxia-inducible factor (HIF)-1a in the mice sufficient of 5-HT versus mice deficient of 5-HT. We thus propose a physiological function of serotonin that serotonin could ameliorate APAP-induced liver injury mainly through inhibiting hepatocyte apoptosis ER stress and promoting liver regeneration. A cetaminophen (N-acetyl-p-aminophenol, APAP), a widely used analgesic and antipyretic drug, is believed to be safe within therapeutic doses. However, an accidental or an intentional APAP overdose often causes acute liver failure with high morbidity and mortality 1,2. The underlying mechanism of APAP hepatotoxicity is thought to be conversion of APAP to N-acetyl-p-benzoquinone imine (NAPQI), which is mediated by members of the cytochrome P450 family, especially CYP2E1. NAPQI can be immediately conjugated with glutathione (GSH) to form the non-toxic metabolites cysteine 3,4. Once the intracellular pool of GSH is exhausted, NAPQI covalently combines with cellular proteins, which results in mitochondrial dysfunction and oxidative stress 5. This type of covalent binding inactivates important functional proteins, leading to hepatic cell death and enzymes like alanine aminotransferase (ALT) release into plasma 6. Both the intracellular (mitochondria) and extracellular (inflammatory cells) reactive oxygen species (ROS) contribute to the liver injury 7,8. Oxidative stress induces c-jun NH 2 -terminal kinase (JNK) activation, which would further promote mitochondrial dysfunction and mitochondrial permeability transition, amplify oxidative stress and lead to sustained JNK activation, and ultimately cause cell death. Previous studies revealed that JNK activation plays a major role in APAP-induced hepatotoxicity which can be inhibited by genetic depletion or pharmaceutic inhibitor Serotonin, as well-known as 5-hydroxytryptamine (5-HT), a small monoamine molecule primarily known as a neurotransmitter, is involved in the regulation of various physiologic procedures including cognition, mood, aggression, mating, feeding, and sleep 12. Peripherally, serotonin mediates vascular contraction and relaxation, cell proliferation, apoptosis, and platelet aggregation. To date, 15 different 5-HT receptor subtypes have been identified 13. Except for its major role in central nervous system, recent studies show that 5-HT plays a crucial role in liver physiology and pathology. To be specific, serotonin is a double-edged sword which is both a friend and a foe to the liver 14. On the one hand, serotonin expedites liver regeneration after partial hepatectomy in various rodent models and promotes tissue repair after ischemia/reperfusion injury 15,16. It is also beneficial to the stabilization of SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
3 hepatic microcirculation and prevents small-for-size liver graft failure 17. Similarly, serotonin receptor agonists improve the sinusoidal perfusion of aged liver and restore the deficient liver regeneration in old mice which shows an effect of age defying 18. Besides, serotonin can also regulate regeneration of the biliary tree by targeting cholangiocytes through an autocrine/paracrine signal way 19. But, on the other hand, studies reveal that serotonin contribute to non-alcoholic fatty liver diseases (NAFLD) in rodent models through its degradation products, the reactive oxygen species 20. Serotonin aggravates viral hepatitis, and plays a crucial role in the progression of hepatic fibrosis 21,22. Serotonin may also facilitate tumor growth of primary liver carcinoma like cholangiocarcinoma and hepatocellular carcinoma 23,24. So far the role of 5-HT in the APAP-induced hepatotoxicity is still unknown. In this study, we focused on the effect of 5-HT on the APAP-induced hepatotoxicity and the relevant mechanisms in mice models. Methods Experimental animals. The study was conducted using male TPH1 knockout mice and wild-type C57BL/6 mice (4 6 weeks, g) (Animal Feeding Center of Xi an Jiaotong University Health Science Center). The TPH12/2 mice were characterized elsewhere 16,17,25. All mice were housed (5 per cage) in clear, pathogen-free polycarbonate cages in the animal care facility, and were fed with a standard animal diet and water ad libitum under controlled temperature conditions with 12 h lightdark cycles. All animal experiments were performed in accordance with the guidelines of China Council on Animal Care and Use. All animal procedures carried out in this study were reviewed, approved, and supervised by the Institutional Animal Care and Use Committee of the Ethics Committee of Xi an Jiaotong University Health Science Center, China. All animals were kept and the experiments were performed in accordance with the European Community guidelines for the use of experimental animals (86/609/EEC). Study design. Mice used in the study were randomly allocated into the following groups: (1) Normal control group: wild type mice and TPH1 knockout mice were treated with 0.9% saline solution through intraperitoneal injection (because of the same results, they served as control group together). (2) WT group: wild type mice were treated with APAP (suspended in saline solution, in a final volume of 0.25 ml) through intraperitoneal injection (i.p). (3) WT1PCPCA group: wild type mice were treated with APAP and p-chlorophenylalanine (PCPA, TPH1-inhibitor, at a dose of 150 mg/kg/day for 3 days by subcutaneous injection before APAP treatment) i.p. as previously reported 25. (4)KO group: TPH1 knockout mice were treated i.p with APAP (TPH1, the rate-limiting enzyme for the synthesis of peripheral serotonin). (5)KO15-HTP group: TPH1knockout mice were treated i.p with APAP and 5- Hydroxytryptophan (5-HTP, precursor of 5-HT which can reload the 5-HT content, at a dose of 75 mg/kg/day for 3 days by subcutaneous injection before APAP administration) as previously reported 19. In another set of experiments, animals (n 5 15 for each group) were randomly divided as described above and monitored for assessing mortality for 5 days after a lethal dose of APAP (600 mg/kg) or saline administration. Measurement of liver function. Serum ALT, AST and alkaline phosphate activities, as well as bilirubin levels were determined by using an Olympus AU5400 Automatic Biochemical Analyzer (Olympus, Tokyo, Japan) in the Department of Inspection, The First Affiliated Hospital of Xi an Jiaotong University 26. Cytokine measurement in murine serum. Levels of serum 5-HT, TNF-a and IL-6 were measured with a commercial ELISA kit following the instructions of the manufacturer (Dakewe, Shenzhen, China). Liver enzymatic activity assay. The liver tissue was homogenized, and tissue myeloperoxidase (MPO), malonaldehyde (MDA), GSH, glutathione peroxidase (GSH-px) activities were measured using the activity assay kits from NanJing JianCheng Bioengineering Institute according to the manufacturer s instructions. Histological study. Liver samples were excised and fixed in 10% formalin solution and embedded in paraffin after completion of the routine follow-up. Serial sections of 5-mm thickness were obtained and stained with Hematoxylin&Eosin (H&E) to evaluate morphology. The results were examined in a blind fashion by two researchers. For electron microscopy examination, liver tissues were prefixed after harvesting immediately with 1.5% glutaraldehyde and 0.8% paraformaldehyde (0.1 mol/l cacodylate buffer) at room temperature and postfixed in an aqueous solution of 1% OsO 4 and 1.5% K 4 (FeCN) 6. Then the specimens were embedded into Epon by routine procedures. Ultrathin sections (50 nm) were contrasted with lead citrate and uranyl acetate and studied with a CM100 Transmission Electron Microscope. Transferase-mediated dutp nick end-labeling (TUNEL) assay. Apoptotic cells were detected by the TUNEL method using an in situ cell detection kit for the detection and quantification of apoptosis at a single-cell level. Staining of tissue sections was performed according to the manufacturer s protocol. Paraffinembedded sections were dewaxed in xylene and rehydrated by passing through a graded series of ethanol solutions, ending with phosphate-buffered saline. Sections were permeabilized with proteinase K (20 mg/ml in 10 mmol/l Tris-HCl, ph ) at 37uC for 15 min. After washing, sections were stained with fluorescent anti- TdT. Sections were viewed and photographed using standard fluorescent microscopic techniques. Immunohistochemistry (IHC). IHC analysis was performed with 4- hydroxynonenal (4-HNE), Nitrotyrosine, TUNEL apoptotic staining, BrdU, Ki-67, PCNA antibodies and HIF-1a, using methods as previously described 27. Mice given BrdUrd (0.5 mg/ml) in drinking water for 4 days were analyzed immunohistochemically for liver nuclear-labeling indices. RNA isolation and quantitative reverse transcription polymerase chain reaction (qrt PCR) analysis. Liver tissue samples from each group were snap-frozen in liquid nitrogen and stored at 270uC until use. Total RNA was isolated from cells using the RNAfast200 Kit (Fastagen Biotech, Shanghai, China). Reverse transcription was performed using the PrimeScript RT reagent Kit (TaKaRa Biotechnology, Dalian, China). The mrna expression was assayed in triplicate and normalized to the b-actin mrna expression. The relative levels were calculated using the Comparative-Ct Method (DDCt method). The primers used in the study were listed in Supplementary Table 1. Western blotting. The monoclonal antibodies used in this research were purchased from Neomarker, Santacruz, Kangcheng for Sigma and Cell signaling. Protein concentration was determined by BCA method. Western blotting was performed as previously described 28. Statistical analysis. The survival and mortality rates are expressed as percentages. The measurement data are expressed as mean 6 standard deviation (SD). Differences between experimental and control groups were assessed by either t test or the analysis of variance (ANOVA), as applicable, using SPSS 18.0 (SPSS, 165 Inc.). A p-value of less than 0.05 was considered statistically significant. Results TPH1-/- mice are more susceptible to acetaminophen-induced hepatotoxicity. Acetaminophen produced a dose-dependent hepatotoxicity. Liver injury was aggravated as the acetaminophen dose increases. TPH1 null mice were more susceptible than wild type mice to acetaminophen-induced hepatotoxicity at the doses of mg/kg, as evidenced by marked increases in serum activities of ALT and AST (Figure 1A). In parallel with serum enzyme activities, more severe necrosis was observed in TPH1 null mice than in wild type mice after acetaminophen administration (Figure 1B). Contemporarily, the depletion of GSH was more rapid in TPH1 knockout mice. (Figure 1C). Then we studied the time course for the effect of APAP on hepatotoxicity in WT and KO mice. Mice were administered 300 mg/kg APAP and sacrificed at 0, 1, 4, 6, 8 and 24 hours after APAP administration. The development of hepatotoxicity was evaluated with the serum levels of ALT and AST and the histopathological evaluation. The time course of the APAP-induced increases in serum ALT and AST is shown in Figure 2. In WT and KO mice, ALT and AST levels were significantly increased by 4 hours. By comparison, the serum ALT levels in the WT mice paralleled those observed in the KO mice but seemed delayed. The serum ALT levels at 6, 8 and 24 hours in the WT mice were significantly lower than those in the KO mice. AST levels in KO mice were significantly higher than those in WT mice at 8 and 24 hours (Figure 2A). In parallel with serum enzyme activities, differences of time-dependent necrosis and hepatic GSH content were also observed in KO and WT mice (Figure 2B and 2C). Serotonin depletion aggravates APAP-induced liver injury in wild type mice and serotonin reloading mitigates hepatotoxicity in TPH1-/- mice. To further validate the role of 5-HT in APAPinduced liver injury, wild type mice were treated with the PCPA before APAP challenge, which has been shown to be a powerful TPH1 enzyme inhibitor 25,29. As demonstrated in Figure 3, PCPA significantly decreased plasma 5-HT levels in wild type mice, which was accompanied by elevations in AST, ALT levels and liver necrosis, and the reduction of 5-day s survival. Together, these SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
4 Figure 1 Dose-response for the effect of APAP on serum ALT and AST, histological examination, liver index and hepatic GSH in Wild-type and TPH1-/- mice. Mice (n 5 6) were given an i.p. injection of acetaminophen ( mg/kg for 24 hours). The liver injury in surviving mice was measured by (A) serum ALT and AST, and Liver tissue were obtained at 24 hours after APAP administration at the dose of 150, 300 and 500 mg/kg. (B) H&E staining, (C) liver index, hepatic necrosis area, and hepatic GSH content were adopted to evaluate the different hepatotoxicity between Wild-type and TPH1-/- mice. The data are presented as mean 6 S.D. *P,.0.05 vs WT group, **P,.0.01 vs WT group. findings show that genetic and pharmacologic inhibition of TPH1 significantly aggravates APAP-induced liver injury. Meanwhile, to substantiate the protective role of serotonin in experimental APAPinduced liver injury, we reloaded Tph1-/- mice with serotonin by injecting its precursor 5-HTP 3 days beforehand. Representative markers of liver injury (AST and ALT) reduced in the serotoninsupplemented group at 24 hours after APAP administration when compared with the KO groups (Figure 3A, 3B, 3C, 3D, 3E and 3F). Taken together, serotonin can also decrease the necrosis region of the liver and improve the 5-day s survival. Figure 2 Time course for the effect of APAP on serum ALT and AST, histological examination, liver index and hepatic GSH in Wild-type and TPH1-/- mice. Mice (n 5 6) were treated with a 300 mg/kg dose of APAP and sacrificed at the indicated times, and serum and liver were collected. The 0 time is the mice in both WT and KO receiving no APAP administration. (A) Serum ALT and AST levels. Liver tissue were obtained at 1, 8, and 24 hours after a dose of 300 mg/kg APAP administration. (B) H&E staining, (C) liver index, hepatic necrosis area and hepatic GSH levels in liver were used to assess the difference between Wild-type and TPH1-/- mice. The data are presented as mean 6 S.D. *P,.0.05 vs WT group, **P,.0.01 vs WT group. SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
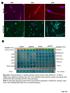
5 Figure 3 Depletion of 5-HT in wild type mice by inhibition of TPH1 aggravating APAP hepatotoxicity while reloading 5-HT in TPH1-/- mice by 5-HTP alleviating liver injury. Four groups including sufficient 5-HT (WT and KO15-HTP groups) and deficient 5-HT (WT1PCPA and KO groups) are recruited to study the role of 5-HT in APAP hepatotoxicity. Mice (n 5 6) were given an i.p. injection of acetaminophen (300 mg/kg for 24 hours), and serum and liver tissues were collected. (A), the concentration of 5-HT in the four groups. (B), Liver index of the mice after APAP administration from the four groups. (C, D), ALT and AST levels in serum. (E), Kaplan-Meier survival curve for mice (n 5 15) in the four groups during 5 days after a single lethal dose of APAP (600 mg/kg). (F), the HE staining of liver sections from four groups and relative percent of necrosis area. (G), the TUNEL staining of hepatocyte apoptosis and corresponding percentage of positive cells. *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. Serotonin decreases cytokines transcription, oxidative stress and APAP-adducts formation in the liver. To assess whether cytokines concentrations induced by APAP in the serum are influenced by 5- HT levels, we measured TNF-a and IL-6 concentrations in serum 24 hours following APAP administration. The result showed that serum levels of TNF-a and IL-6 in the WT and KO15-HTP groups were significantly lower than the mice deficient of 5-HT (Figure 4A). Meanwhile, when detecting the mrna levels of TNF-a and IL-6 in the liver tissues, we found that TNF-a and IL- 6 had significantly higher transcriptional levels in the WT1PCPA and KO groups (Figure 4B). Meanwhile, the present study showed that APAP significantly enhanced oxidative stress (indicated by MPO and MDA levels and 4-HNE expression) in the WT1PCPA and KO groups (Figure 4C and 4E). Simultaneously, glutathione (GSH), not only an indicator of oxidative stress, but also an important role in detoxification of the acetaminophen metabolite NAPQI, was determined to ascertain whether there was a difference in its depletion in the APAP-treated mice. Treatment of wild mice with PCPA or TPH1 knockout produced a significant reduction of GSH and GSH-px levels in liver after APAP administration when compared with the WT or KO15-HTP groups (Figure 4D). It is well established that an APAP overdose induces peroxynitrite formation as indicated by the appearance of NT protein adducts in centrilobular hepatocytes. To investigate whether 5-HT can influence peroxynitrite formation, NT protein adducts were evaluated in the four groups 24 hours after APAP administration. The results showed that a more extensive NT staining of centrilobular hepatocytes was observed in the WT1PCPA and KO groups (Figure 4F). Serotonin inhibits APAP-challenged hepatocyte apoptosis in the liver. Acetaminophen toxicity is often associated with hepatic centrilobular necrosis while hepatocyte apoptosis is a controversial existence. To further analyze whether apoptosis would coexist with necrosis in APAP-induced hepatotoxicity, we detected APAPinduced apoptosis by TUNEL staining and investigated the possible role of Bcl-2 family proteins and caspase-3 under this pathophysiological condition. TUNEL staining showed that liver sections from mice in WT1PCPA and KO groups were more positively stained than mice in the WT and KO15-HTP groups (Figure 3G). Contemporarily, we carried out western blot analysis of phosphorylation Bcl-2 proteins (Bcl-2 and Bcl-xL) and caspase-3 and found an apparent up-regulation in p-bcl-2 and p-bcl-xl expression in APAP-exposed mice. However, mice sufficient with 5-HT effectively show reduced levels of those. On the other hand, APAP-exposure increases neither caspase-3 nor cleaved-caspase-3 expression which was consistent with previous research that caspase- 3 might not play an effect in the APAP-induced liver injury 30 (Figure 5A and 5B). Serotonin promoted endoplasmic reticulum (ER) and inhibited ER stress after APAP challenge. The endoplasmic reticulum (ER) is an important intracellular organelle that carries out multiple physiological functions. Previous studies showed that ER stress was induced during the APAP hepatotoxicity 31. So, we detected the ER stress in the current study to determine whether ER stress can be affected by 5-HT. 24 hours after APAP administration (300 mg/kg), mice were sacrificed and the liver tissue were obtained to assess the ER stress and mitochondria injury. Subsequent analysis by electron SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
6 Figure 4 5-HT mitigates inflammation, oxidative stress and nitrotyrosine formation in the liver after APAP administration. At 24 hours after APAP challenge (300 mg/kg), serum and liver samples were collected from experimental groups to detect the levels of inflammation and oxidative stress. (A). Serum levels of TNF-a and IL-6, (B). The transcriptional levels of TNF-a and IL-6 in the liver tissue, (C). The MDA and MPO levels in the liver, (D). The levels of antioxidants (GSH and GSH-px), (E).The immumohistochemical staining of 4-Hydroxynonenal (4-HNE) and relative staining score, (F). The formation of peroxynitrite detected by immumohistochemical staining of nitrotyrosine protein adducts and relative staining score. n 5 6, *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. microscopy revealed APAP induced ER tumidness, hyperplasia and dissolvent. It could also induce megamitochondria, mitochondria pyknosis, tumidness and flocculent degeneration. These injuries could be alleviated in the wild-type mice when compared with TPH1-/- mice (Figure 6A). Meanwhile, RT-PCR and western bolt analysis revealed that the ER stress related genes such as GRP78, ATF4, CHOP, XBP1 were more highly expressed in the mice e lack of 5-HT (Figure 6B and 6C). Serotonin promoted hepatocyte proliferation in vivo after APAP challenge. As the former study showed that serotonin could promote the liver regeneration and hepatocyte proliferation after partial hepatectomy and hepatic ischemia/reperfusion injury 15,16, we hypothesized that serotonin might also increase hepatocyte proliferation after the APAP overdose. To test this hypothesis, mice were sacrificed at 72 hours after APAP administration (300 mg/kg), and livers were harvested for BrdU, Ki67 and PCNA staining. The tissue from WT1PCPA and KO groups displayed quite a little increase in the number of positive staining hepatocytes, while 5-HT sufficiency can facilitate hepatocyte proliferation (Figure 7A,7B,7C and 7D). Meanwhile, PCNA and Cyclin D1Western blot data indicated that mice lack of 5-HT experienced a lower level of liver regeneration after the APAP overdose and exhibited significantly lower cell proliferation at 72 hours after APAP administration. To investigate the molecular mechanisms underlying the improvement of liver regeneration, we examined whether 5-HT modulated liver proliferation signaling events mainly through the transcription factor STAT3, a key protein in the hepatocyte proliferation by western blot analysis 32,33. Activated STAT3 was higher expressed in the mice with 5-HT when compared with mice lack of 5-HT, which hinted that 5-HT might promote liver regeneration partially by the activation of STAT3. (Figure 7E and 7F) Serotonin inhibited APAP-induced phosphorylation of JNK and activation of CYP2E1. 5-HT receptors are the direct target of 5-HT. So we used RT-PCR to detect the specific 5-HT receptor that was involved in the APAP-induced hepatotoxicity. The result showed that 5-HT2B receptor was more expressed compared with others, which was also confirmed by the Western Blot analysis (Figure 8A). It was also very interesting that previous studies showing activation of 5-HT2B could promote hepatocyte proliferation in mice, which was in accordance with the result in our study 17. It is known that JNK activation is mediated by oxidative stress and plays pathogenic roles in a diverse array of cellular programmes, including cell differentiation, movement, proliferation and death 34. To investigate the mechanisms for APAP-induced necrotic hepatocytes death, we studied the JNK involved in the signal transduction in the hepatic tissues. Western blot analyses showed a significant enhancement in the expression of phospho-jnk after APAP challenge, and sufficient 5-HT could reduce its expression, whereas no significant change was found in the expression of total JNK. Furthermore, we investigated the effect of APAP on CYP2E1 protein level and found that CYP2E1 protein level was increased in the APAP-induced liver tissue while 5- HT could normalize these changes and maintain the normal SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
7 Figure 5 5-HT inhibits the hepatocyte apoptosis mainly by the suppression of BCL-2 proteins phosphorylation. 24 hours after APAP challenge (300 mg/kg), liver samples were collected and proteins were extracted to detect the hepatocyte apoptosis in the APAP hepatotoxicity by western blot analysis. (A, B). Bcl-2, Bcl-XL and caspase-3 protein levels. b-actin was used as an internal control. n 5 6, *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. The gels have been run under the same experimental conditions and the cropped blots images are shown in the figure and the full-length blots are presented in Supplementary Figure S1. physiology of the organs (Figure 8B and 8C). Meanwhile, we detected the HIF-1a, a key protein in the mechanism of APAP-induced liver injury 35 and found that 5-HT could decrease the expression of HIF- 1a by which 5-HT contributed to relieve of APAP-induced hepatotoxicity (Figure 8D and 8E). Discussion Liver is the major site for drug metabolism and elimination, which makes it susceptible to drug toxicity 36. Acetaminophen, the most commonly used analgesic drug, is believed to be safe within therapeutic doses. However, an APAP overdose always produces a centrilobular hepatic necrosis which is the main cause of acute liver failure in the western world 2. Though decades of studies have been launched, the mechanisms of APAP-induced liver injury are not completely known. 5-HT is a small monoamine molecule primarily known for its role as a neurotransmitter. Previous studies showed that peripheral 5-HT could play an important role in the hepatic physiological and pathological process 37,38. This experiment was designed to study the role of 5-HT in acetaminophen-induced hepatotoxicity. We employed mice lacking Tph1, the rate-limiting enzyme for the synthesis of peripheral serotonin, to sort as the 5- HT deficient group. We also used PCPA, an inhibitor of TPH1 as the pharmacological intervention to induce the 5-HT deficiency in the wild type mice. Meanwhile, we used 5-HTP, the precursor of 5-HT, to replenish the 5-HT deficiency in the TPH1-/- mice. Based on both genetic and pharmacological methods, we recruited four major groups, including two rich of 5-HT groups (WT and KO15-HTP groups) and two deficient of 5-HT groups (WT1PCPA and KO Figure 6 5-HT protects the organelles and inhibition of the endoplasmic reticulum (ER) stress after APAP challenge. 24 hours after APAP challenge (300 mg/kg), liver samples were collected to evaluate the ER stress. (A). Representative electron microscopy of ER tumidness, hyperplasia and dissolved, and hepatocyte megamitochondria in the liver tissue after APAP administration. (B). The ER stress associated protein levels showed by western Blot analysis. (C). The transcription and expression levels of ER stress associated gene levels. b-actin was used as an internal control. n 5 6, *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. The gels have been run under the same experimental conditions and the cropped blots images are shown in the figure and the full-length blots are presented in Supplementary Figure S1. SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
8 Figure 7 5-HT facilitates hepatocyte proliferation after APAP challenge. 72 hours after APAP administration (300 mg/kg), liver samples were collected to evaluate the effect of 5-HT on the liver regeneration. (A). Representative immunohistochemistry staining of BrdU, Ki67 and PCNA in the liver. (B D). The quantization of hepatocyte proliferation was launched by calculating the positive cells in a microscopic vision. (E, F) The protein levels of STAT3, Cyclin D1 and PCNA, which were associated with hepatocyte proliferation, were showed by western blot analysis. b-actin was used as an internal control. n 5 6, *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. The gels have been run under the same experimental conditions and the cropped blots images are shown in the figure and the full-length blots are presented in Supplementary Figure S1. groups), to ensure the reliability of the experimental results. Our data showed that depletion of 5-HT using genetic and pharmacologic inhibition of TPH1 resulted in an increased APAP-induced liver injury and a slower recovery. In contrast, supplementation of 5- HT with 5-HTP in TPH1-/- mice resulted in a delayed initiation of liver injury and an accelerated recovery after APAP treatment. Taken together, our data indicate that 5-HT can protect against APAPinduced liver injury. After APAP administration, liver injury was detected in both KO and WT mice. The hepatotoxicity differences between the KO and WT mice were in a time and dose dependent manner. These differences were also observed in the histopathological evaluation of toxicity. Meanwhile, the depletion of glutathione and arylation of proteins by acetaminophen are important events leading to acetaminophen hepatotoxicity. In the present study, TPH1 null mice were more susceptible to acetaminophen-induced glutathione depletion SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
9 Figure 8 5-HT inhibits the activation of JNK to decrease the HIF-1a expression mainly by 5-HT2B receptor. To investigate the potential mechanism involved in the 5-HT protecting APAP hepatotoxicity, liver mrnas and proteins were extracted to detect potential proteins 24 hours after APAP administration (300 mg/kg). (A) RT-PCR was performed to seek the possible 5-HT receptors, and the results showed that 5-HT2B was the target protein which was also confirmed by western blot analysis. (B, C) Western blot analysis of CYP2E1, p-jnk, Total-JNK, 5-HT2B receptor and HIF-1a. b-actin was used as an internal control. (D, E) The immumohistochemical staining of HIF-1a and relative staining score. n 5 6, *P,.0.05 vs WT group, **P,.0.01 vs WT group, { P,.0.05 vs TPH1 knockout group, {{ P,.0.01 vs TPH1 knockout group. The gels have been run under the same experimental conditions and the cropped blots images are shown in the figure and the full-length blots are presented in Supplementary Figure S1. compared with the WT mice. Acetaminophen-induced hepatotoxicity is thought to be mediated by a cytochrome P-450-generated intermediate, NAPQI 30. Therefore, we determined whether TPH1 null mice had an altered metabolism of acetaminophen. Our results on hepatic CYP2E1 analysis using western blot indicate that 5-HT could influence the metabolism of APAP mainly by decreasing the hepatic CYP2E1 expression. Simultaneously, an APAP overdose induces peroxynitrite formation as indicated by the appearance of NT protein adducts in centrilobular hepatocytes 39,40. To investigate whether 5-HT can affect peroxynitrite formation, NT protein adducts were evaluated by immunohistochemistry. Amore extensive NT staining of centrilobular hepatocytes was observed in the WT1PCPA and KO group when compared with the WT and KO15-HTP mice. Oxidative damage has been proposed as a contributing mechanism of acetaminophen hepatotoxicity 41. Oxidative stress occurs in cells when there is a disruption of cellular redox balance 42. Acetaminophen-induced oxidative stress results in lipid peroxidation, oxidation of protein thiols, mitochondrial injury, SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
10 Figure 9 Schematic representation of the proposed role of 5-HT in APAP-induced hepatic injury. After administration into the hepatocytes, APAP is first metabolized by the CYP2E1 and generates the NAPQI, which binds with GSH and also increases ROS and RNS formation to initiate the ER stress which involved in GRP78 and CHOP activation. Increased ROS leads to the JNK phosphorylation to damage the mitochondria, which also promotes the phosphorylation of BCL-2 proteins to induce the apoptosis. Meanwhile, inflammatory factors and HIF-1a can be induced with the oxidative stress. During the hepatic self-repair stage, IL-6/STAT3 pathway can be initiated to promote the cell mitosis and facilitate the liver regeneration. In the present study, we prove that 5-HT can reduce APAP hepatotoxicity by inhibiting APAP metabolism, ROS and RNS formation and promoting the hepatocyte proliferation. altered calcium homeostasis, and DNA damage 43. In the present study, TPH1 null mice were more susceptible than controls to acetaminophen-and NAPQI-induced lipid peroxidation, as evidenced by increased MDA and MPO levels, and reduced GSH and GSH-px in the liver tissues which were also important antioxidant enzymes. The immunohistochemical localization of lipid peroxidation adducts (4- HNE) at early time-points correlated with necrosis of hepatocytes around the central zone. All these data suggested that the greater susceptibility of TPH1 null mice might be partially due to the increased oxidative damage produced by acetaminophen. The role of apoptosis in acetaminophen induced liver injury is controversial 44,45. It is generally accepted that the ultimate form of hepatic damage caused by acetaminophen is necrosis 5. But, according to the latest research, researchers tend to agree with its existence 46,47. During APAP intoxication in the mouse, toxic ROS are generated which can cause apoptotic cell death including JNK activation 10. Both in vivo and in vitro, acetaminophen can induce apoptosis by triggering the release of mitochondrial cytochrome-c and activating the apoptotic pathway 48,49. Several investigators have suggested that the way of cell death induced by acetaminophen may be related to the overall acetaminophen tissue concentrations and intracellular concentrations of ATP 50. So, in this study, we examined whether apoptosis contributes to the induction of acute liver failure caused by APAP and whether 5-HT can affect this pathological process. According to the results of the research, two anti-apoptotic proteins of the Bcl-2 family (Bcl-2 and Bcl-xL) could be phosphorylated by p- JNK. But sufficient 5-HT could decrease its expression along with JNK activation. It has also been reported that caspase inhibitors protect liver from APAP-induced injury, suggesting that apoptosis plays a vital role in initiating the events that lead to hepatic damage, but APAP does not activate execution caspases-3 and -7 51,52.Sowe also investigated the role of caspase-3 after APAP administration and found that caspase-3 was not activated in our experiments. These data suggested that the protection of 5-HT might be partially due to the inhibition of apoptosis of hepatic cells mainly by suppression of Bcl-2 proteins phosphorylation. Endoplasmic reticulum is an important intracellular organelle that carries out multiple physiological functions such as protein folding, posttranslational modifications, biosynthesis of fatty acids and sterols, detoxification of xenobiotics, and the storage of intracellular calcium 53. It is also important in drug metabolism, as most phase I enzymes including cytochrome P450 oxidases and some phase II enzymes localize to the ER 54. Hepatocytes in the liver contain abundant ER, which endows the liver with the capacity to engage in lipid and drug metabolism as well as the production of plasma proteins. ER stress occurs when the amount of protein entering the ER exceeds its folding capacity. Previous studies showed that ER stress in the liver could be induced after APAP administration which contributes to the liver injury. Uzi D et al found that inhibition of CHOP, a key protein of ER stress, could protect liver from APAP hepatotoxicity 31. So,we firstly study the change of hepatocellular subcellular structure by electron microscope. Massive ER swelling, hyperplasia and dilation could be observed in the liver after APAP administration. Meanwhile, real-time PCR and western blot showed that the ER stress could be induced by APAP. And 5-HT could stabilize the structure of organelles and inhibit the ER stress which had the cytoprotection effect as previous studies showed. Liver regeneration is a vital process for survival after a toxic insult. Regeneration ensures the replacement of necrotic cells and the full recovery of organ function 55. Several studies have demonstrated that compensatory liver regeneration after APAP-induced liver injury is a critical determinant of final outcome of APAP overdose The exposure to growth factors such as hepatocyte growth factor results in the expression of cell cycle proteins. The induction of Cyclin D1 is the marker for cell cycle (G1 phase) progression in hepatocytes 33,59. In current investigation, our western blot data showed that a higher level of 5-HT markedly increased the level of Cyclin D1 in the APAP challenged liver tissue. These changes in Cyclin D1 expression were associated with decreased serum ALT/AST and improved liver regeneration in wild type and TPH1-/- plus 5-HTP mice receiving APAP, suggesting that 5-HT likely facilitates activation of Cyclin D1- mediated regeneration pathways. Our data suggest that 5-HT is an SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
11 important factor that promotes hepatocyte regeneration after the APAP overdose. Investigators have shown similar findings in a normothermic ischemia/reperfusion model of injury. Nocito A and others demonstrated that TPH1 knock-out mice had significantly less severe liver injury after ischemia/reperfusion, related to the accelerated hepatocyte proliferation in the knock-out mice 16. Similarly, Lesurtel M and colleagues also demonstrated that serotonin promoted liver regeneration after partial hepatectomy which was mediated by 5-HT2A and 2B subtype serotonin receptors 15. Meanwhile, Tian YH et al found that serotonin could protects small-for-size liver grafts from injury and prevents microcirculation and regeneration through its activation on receptor-2b 17. These findings are similar to ours in that the 5-HT confers protection against a hepatic injury by promoting the hepatocyte proliferation and liver regeneration by the activation of 5-HT 2B receptor. JNK is activated during oxidative stress and plays a central role in APAP-induced hepatotoxicity 60. Hepatic JNK activation is a critical step in the intra-cellular signaling involved in APAP-induced liver injury 10,61. Our data show that 5-HT depletion results in a rapid activation of JNK and that the appropriate supplementation results in a decreased activation of JNK compared with control mice. These data indicate that 5-HT may suppress JNK activation during APAPinduced liver injury. But, whether 5-HT directly affect JNK activation or induce this effect via an upstream regulator is not known. 5-HT is known to signal via multiple 5-HT receptor pathways in the liver. It is possible that 5-HT may use one of these pathways to suppress JNK signaling after APAP-induced liver injury. Meanwhile, HIF-1a was an important protein associated with JNK signaling which was also a key protein in the pathological process of APAP-induced liver injury. HIF-regulated genes include plasminogen activator inhibitor-1 (PAI-1), cell death proteins such as BNIP3 and Nix, which can all contribute to the liver injury 35. According to the study, 5-HT may decrease the expression of HIF-1a to protect APAP hepatotoxicity. In conclusion, we describe a novel physiological role for endogenous serotonin in the protection from APAP-induced liver injury (Figure 9). Considering the pleiotropic effects of serotonin in various physiological processes, we investigated a number of potential mechanisms that might underlie the protection effect of 5-HT. This protect effect of 5-HT on APAP hepatotoxicity was at least partially attributable to a significantly decreased inflammation, oxidative stress, GSH depletion, peroxynitrite formation, hepatocyte apoptosis and ER stress, elevated hepatocyte proliferation, activation of 5-HT2B receptor and less activated JNK and HIF-1a. 1. Reuben, A., Koch, D. G. & Lee, W. M. Drug-induced acute liver failure: Results of a US multicenter, prospective study. Hepatology 52, (2010). 2. Larson, A. M. et al. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology 42, (2005). 3. Jaeschke, H., McGill, M. R. & Ramachandran, A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev 44, (2012). 4. Larson, A. M. Acetaminophen hepatotoxicity. Clin Liver Dis 11, (2007). 5. Hinson, J., Roberts, D. & James, L. Mechanisms of acetaminophen-induced liver necrosis. Handb Exp Pharmacol 196, (2010). 6. Ruepp, S. U., Tonge, R. P., Shaw, J., Wallis, N. & Pognan, F. Genomics and proteomics analysis of acetaminophen toxicity in mouse liver. Toxicol Sci 65, (2002). 7. Agarwal, R. et al. Acetaminophen-induced hepatotoxicity in mice occurs with inhibition of activity and nitration of mitochondrial manganese superoxide dismutase. J Pharmacol Exp Ther 337, (2011). 8. Bajt, M. L. et al. Apoptosis-inducing factor modulates mitochondrial oxidant stress in acetaminophen hepatotoxicity. Toxicol Sci 122, (2011). 9. Bae, M.-A., Pie, J.-E. & Song, B. J. Acetaminophen induces apoptosis of C6 glioma cells by activating the c-jun NH2-terminal protein kinase-related cell death pathway. Mol Pharmacol 60, (2001). 10. Hanawa, N. et al. Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem 283, (2008). 11. Latchoumycandane, C., Goh, C. W., Ong, M. M. & Boelsterli, U. A. Mitochondrial protection by the JNK inhibitor leflunomide rescues mice from acetaminopheninduced liver injury. Hepatology 45, (2007). 12. Mössner, R. & Lesch, K.-P. Role of serotonin in the immune system and in neuroimmune interactions. Brain Behav Immun 12, (1998). 13. Cloëz-Tayarani, I. & Changeux, J.-P. Nicotine and serotonin in immune regulation and inflammatory processes: a perspective. J Leukoc Biol 81, (2007). 14. Lesurtel, M., Soll, C., Humar, B. & Clavien, P.-A. Serotonin: a double-edged sword for the liver? Surgeon 10, (2012). 15. Lesurtel, M. et al. Platelet-derived serotonin mediates liver regeneration. Science 312, (2006). 16. Nocito, A. et al. Platelets and platelet-derived serotonin promote tissue repair after normothermic hepatic ischemia in mice. Hepatology 45, (2007). 17. Tian, Y. et al. Activation of serotonin receptor-2b rescues small-for-size liver graft failure in mice. Hepatology 53, (2011). 18. Furrer, K. et al. Serotonin reverts age-related capillarization and failure of regeneration in the liver through a VEGF-dependent pathway. Proc Natl Acad Sci USA108, (2011). 19. Jang, J. H. et al. Serotonin protects mouse liver from cholestatic injury by decreasing bile salt pool after bile duct ligation. Hepatology 56, (2012). 20. Nocito, A. et al. Serotonin mediates oxidative stress and mitochondrial toxicity in a murine model of nonalcoholic steatohepatitis. Gastroenterology 133, (2007). 21. Lang, P. A. et al. Aggravation of viral hepatitis by platelet-derived serotonin. Nat Med 14, (2008). 22. Ruddell, R. G. et al. A role for serotonin (5-HT) in hepatic stellate cell function and liver fibrosis. Am J Pathol 169, (2006). 23. Alpini, G. et al. Serotonin metabolism is dysregulated in cholangiocarcinoma, which has implications for tumor growth. Cancer Res 68, (2008). 24. Soll, C. et al. Serotonin promotes tumor growth in human hepatocellular cancer. Hepatology 51, (2010). 25. Izikki, M. et al. Tryptophan hydroxylase 1 knockout and tryptophan hydroxylase 2 polymorphism: effects on hypoxic pulmonary hypertension in mice. Am J Physiol Lung Cell Mol Physiol 293, L1045 L1052 (2007). 26. Wang, F., Zhang, P., Jiang, H. & Cheng, S. Gut bacterial translocation contributes to microinflammation in experimental uremia. Dig Dis Sci 57, (2012). 27. Zhang, J.-Y. et al. Protective role of hydrogen-rich water on aspirin-induced gastric mucosal damage in rats. World J Gastroenterol 20, (2014). 28. Qu, K. et al. Negative regulation of transcription factor FoxM1 by p53 enhances oxaliplatin-induced senescence in hepatocellular carcinoma. Cancer Lett 331, (2013). 29. Alexandre, C. et al. Early life blockade of 5-hydroxytryptamine 1A receptors normalizes sleep and depression-like behavior in adult knock-out mice lacking the serotonin transporter. J Neurosci 26, (2006). 30. Das, J., Ghosh, J., Manna, P. & Sil, P. C. Acetaminophen induced acute liver failure via oxidative stress and JNK activation: protective role of taurine by the suppression of cytochrome P450 2E1. Free Radic Res 44, (2010). 31. Uzi, D. et al. CHOP is a critical regulator of acetaminophen-induced hepatotoxicity. J Hepatol 59, (2013). 32. Michalopoulos, G. K. & DeFrances, M. C. Liver regeneration. Science 276, (1997). 33. Fausto, N., Campbell, J. S. & Riehle, K. J. Liver regeneration. Hepatology 43, S45 S53 (2006). 34. Shen, H.-M. & Liu, Z.-G. JNK signaling pathway is a key modulator in cell death mediated by reactive oxygen and nitrogen species. Free Radic Biol Med 40, (2006). 35. Sparkenbaugh, E. M. et al. The role of hypoxia-inducible factor-1a in acetaminophen hepatotoxicity. J Pharmacol Exp Ther 338, (2011). 36. Lammert, C., Bjornsson, E., Niklasson, A. & Chalasani, N. Oral medications with significant hepatic metabolism at higher risk for hepatic adverse events. Hepatology 51, (2010). 37. Clavien, P. Liver regeneration: a spotlight on the novel role of platelets and serotonin. Swiss Med Wkly 138, 361 (2008). 38. Ruddell, R. G., Mann, D. A. & Ramm, G. A. The function of serotonin within the liver. J Hepatol 48, (2008). 39. Knight, T. R., Ho, Y.-S., Farhood, A. & Jaeschke, H. Peroxynitrite is a critical mediator of acetaminophen hepatotoxicity in murine livers: protection by glutathione. J Pharmacol Exp Ther 303, (2002). 40. Knight, T. R., Kurtz, A., Bajt, M. L., Hinson, J. A. & Jaeschke, H. Vascular and hepatocellular peroxynitrite formation during acetaminophen toxicity: role of mitochondrial oxidant stress. Toxicol Sci 62, (2001). 41. Bajt, M. L., Knight, T. R., Lemasters, J. J. & Jaeschke, H. Acetaminophen-induced oxidant stress and cell injury in cultured mouse hepatocytes: protection by N- acetyl cysteine. Toxicol Sci 80, (2004). 42. Murphy, M. How mitochondria produce reactive oxygen species. Biochem J 417, 1 13 (2009). 43. Jaeschke, H. Molecular mechanisms of hepatic ischemia-reperfusion injury and preconditioning. Am J Physiol Gastrointest Liver Physiol 284, G15 G26 (2003). 44. Ray, S. D., Mumaw, V. R., Raje, R. R. & Fariss, M. W. Protection of acetaminophen-induced hepatocellular apoptosis and necrosis by cholesteryl hemisuccinate pretreatment. J Pharmacol Exp Ther 279, (1996). SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
12 45. Gujral, J. S., Knight, T. R., Farhood, A., Bajt, M. L. & Jaeschke, H. Mode of cell death after acetaminophen overdose in mice: apoptosis or oncotic necrosis? Toxicol Sci 67, (2002). 46. Kon, K. et al. Role of apoptosis in acetaminophen hepatotoxicity. J Gastroenterol Hepatol 22, S49 S52 (2007). 47. Malhi, H., Gores, G. J. & Lemasters, J. J. Apoptosis and necrosis in the liver: a tale of two deaths? Hepatology 43, S31 S44 (2006). 48. El-Hassan, H. et al. Involvement of mitochondria in acetaminophen-induced apoptosis and hepatic injury: roles of cytochrome c, Bax, Bid, and caspases. Toxicol Appl Pharmacol 191, (2003). 49. Boulares, A. H., Zoltoski, A. J., Stoica, B. A., Cuvillier, O. & Smulson, M. E. Acetaminophen Induces a Caspase-Dependent and Bcl-xL Sensitive Apoptosis in Human Hepatoma Cells and Lymphocytes. Pharmacol Toxicol 90, (2002). 50. Kon, K., Kim, J. S., Jaeschke, H. & Lemasters, J. J. Mitochondrial permeability transition in acetaminophen-induced necrosis and apoptosis of cultured mouse hepatocytes. Hepatology 40, (2004). 51. Lawson, J. A., Fisher, M. A., Simmons, C. A., Farhood, A. & Jaeschke, H. Inhibition of Fas receptor (CD95)-induced hepatic caspase activation and apoptosis by acetaminophen in mice. Toxicol Appl Pharmacol 156, (1999). 52. Jaeschke, H. & Bajt, M. L. Intracellular signaling mechanisms of acetaminopheninduced liver cell death. Toxicol Sci 89, (2006). 53. Ron, D. & Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8, (2007). 54. Cribb, A. E., Peyrou, M., Muruganandan, S. & Schneider, L. The endoplasmic reticulum in xenobiotic toxicity. Drug Metab Rev 37, (2005). 55. Mehendale, H. M. Tissue repair: an important determinant of final outcome of toxicant-induced injury. Toxicol Pathol 33, (2005). 56. Apte, U. et al. Beta-catenin activation promotes liver regeneration after acetaminophen-induced injury. Am J Pathol 175, (2009). 57. Fontana, R. J. Acute liver failure including acetaminophen overdose. Med Clin North Am 92, (2008). 58. Dalhoff, K. et al. Autoprotection in acetaminophen intoxication in rats: the role of liver regeneration. Pharmacol Toxicol 88, (2001). 59. Albrecht, J. H. & Hansen, L. K. Cyclin D1 promotes mitogen-independent cell cycle progression in hepatocytes. Cell Growth Differ 10, (1999). 60. Henderson, N. C. et al. Critical role of c-jun (NH2) terminal kinase in paracetamol-induced acute liver failure. Gut 56, (2007). 61. Shen, H.-M. & Pervaiz, S. TNF receptor superfamily-induced cell death: redoxdependent execution. FASEB J 20, (2006). Acknowledgments We thank Professor Michelle Bader in Max Planck Institute for Molecular Genetics (Berlin, Germany) for presenting the TPH1 knockout mice. Meanwhile, we thank Doctor Liang Bai in Laboratory of Lipid Metabolism and Atherosclerosis, Xi an Jiaotong University Cardiovascular Research Center for technical assistance and consultant. Author contributions J.Y.Z. designed the experiments, performed most experiments and wrote the paper; S.D.S. and Q.P. discussed the results and generated figures and supported the performance of WB; R.Y.Z. revised the paper and improved the language; L.Z. supported the performance of real time-pcr; S.S.L. contributed the revised manuscript and data analysis. F.D.M. supported the performance of WB and contributed the revised manuscript. Q.F.W. supported the performance of tissue staining; C.L. designed and directed the overall project. All authors reviewed the manuscript. Additional information Supplementary information accompanies this paper at scientificreports Competing financial interests: The authors declare no competing financial interests. How to cite this article: Zhang, J. et al. Serotonin Deficiency Exacerbates Acetaminophen-Induced Liver Toxicity In Mice. Sci. Rep. 5, 8098; DOI: /srep08098 (2015). This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permissionfrom the licenseholder in order toreproduce the material. To view a copy of this license, visit SCIENTIFIC REPORTS 5 : 8098 DOI: /srep
Mechanistic Toxicology
 SECOND EDITION Mechanistic Toxicology The Molecular Basis of How Chemicals Disrupt Biological Targets URS A. BOELSTERLI CRC Press Tavlor & France Croup CRC Press is an imp^t o* :H Taylor H Francn C'r,,jpi
SECOND EDITION Mechanistic Toxicology The Molecular Basis of How Chemicals Disrupt Biological Targets URS A. BOELSTERLI CRC Press Tavlor & France Croup CRC Press is an imp^t o* :H Taylor H Francn C'r,,jpi
The liver in poisoning: what can we learn from animal models?
 The liver in poisoning: what can we learn from animal models? Stephan Krähenbühl Clinical Pharmacology & Toxicology University Hospital 4031 Basel/Switzerland Kraehenbuehl@uhbs.ch Outcome and causes of
The liver in poisoning: what can we learn from animal models? Stephan Krähenbühl Clinical Pharmacology & Toxicology University Hospital 4031 Basel/Switzerland Kraehenbuehl@uhbs.ch Outcome and causes of
Alcoholic hepatitis is a drug-induced disorder
 Alcoholic hepatitis is a drug-induced disorder Gyongyi Szabo, MD, PhD Professor of Medicine University of Massachusetts Medical School Source: 2 Sobernation.com Clinical Progression of ALD Mortality Acute
Alcoholic hepatitis is a drug-induced disorder Gyongyi Szabo, MD, PhD Professor of Medicine University of Massachusetts Medical School Source: 2 Sobernation.com Clinical Progression of ALD Mortality Acute
shehab Moh Tarek ... ManarHajeer
 3 shehab Moh Tarek... ManarHajeer In the previous lecture we discussed the accumulation of oxygen- derived free radicals as a mechanism of cell injury, we covered their production and their pathologic
3 shehab Moh Tarek... ManarHajeer In the previous lecture we discussed the accumulation of oxygen- derived free radicals as a mechanism of cell injury, we covered their production and their pathologic
CHAPTER VII EFFECT OF H. ZEYLANICA, R. NASUTA AND S. CILIATA ON PARACETAMOL - INDUCED HEPATOTOXICITY IN WISTAR RATS
 CHAPTER VII EFFECT OF H. ZEYLANICA, R. NASUTA AND S. CILIATA ON PARACETAMOL - INDUCED HEPATOTOXICITY IN WISTAR RATS 7.1. Introduction Paracetamol is a remarkably safe drug at therapeutic doses, but it
CHAPTER VII EFFECT OF H. ZEYLANICA, R. NASUTA AND S. CILIATA ON PARACETAMOL - INDUCED HEPATOTOXICITY IN WISTAR RATS 7.1. Introduction Paracetamol is a remarkably safe drug at therapeutic doses, but it
Anti-Apoptotic Effects of Cellular Therapy
 Anti-Apoptotic Effects of Cellular Therapy Jason Lapetoda 1, Lee K Landeen, PhD 1, George K Michalopoulos, MD 2, Patricia W Bedard, PhD 1 1 Vital Therapies, Inc., San Diego, CA, USA 2 University of Pittsburgh
Anti-Apoptotic Effects of Cellular Therapy Jason Lapetoda 1, Lee K Landeen, PhD 1, George K Michalopoulos, MD 2, Patricia W Bedard, PhD 1 1 Vital Therapies, Inc., San Diego, CA, USA 2 University of Pittsburgh
Hepatotoxicity and ALT/AST Enzymes Activities Change in Therapeutic and Toxic Doses Consumption of Acetaminophen in Rats
 IBBJ Summer 2017, Vol 3, No 3 Original Article Hepatotoxicity and ALT/AST Enzymes Activities Change in Therapeutic and Toxic Doses Consumption of Acetaminophen in Rats Pouria jarsiah 1, Anahita Nosrati
IBBJ Summer 2017, Vol 3, No 3 Original Article Hepatotoxicity and ALT/AST Enzymes Activities Change in Therapeutic and Toxic Doses Consumption of Acetaminophen in Rats Pouria jarsiah 1, Anahita Nosrati
Supplemental Information
 Electronic Supplementary Material (ESI) for Food & Function. This journal is The Royal Society of Chemistry 2016 Supplemental Information Supplementary Materials and Methods Materials Assay kits of total
Electronic Supplementary Material (ESI) for Food & Function. This journal is The Royal Society of Chemistry 2016 Supplemental Information Supplementary Materials and Methods Materials Assay kits of total
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Acetaminophen (APAP) hepatotoxicity represents a major
 phisms and haplotypes in primary biliary cirrhosis. Clin Gastroenterol Hepatol 2007;5:755 760. 32. Juran BD, Atkinson EJ, Schlicht EM, et al. Interacting alleles of the coinhibitory immunoreceptor genes
phisms and haplotypes in primary biliary cirrhosis. Clin Gastroenterol Hepatol 2007;5:755 760. 32. Juran BD, Atkinson EJ, Schlicht EM, et al. Interacting alleles of the coinhibitory immunoreceptor genes
This student paper was written as an assignment in the graduate course
 77:222 Spring 2005 Free Radicals in Biology and Medicine Page 0 This student paper was written as an assignment in the graduate course Free Radicals in Biology and Medicine (77:222, Spring 2005) offered
77:222 Spring 2005 Free Radicals in Biology and Medicine Page 0 This student paper was written as an assignment in the graduate course Free Radicals in Biology and Medicine (77:222, Spring 2005) offered
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence
 Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Interpretation of the liver hypertrophy in the toxicological evaluation of veterinary medicinal products
 Provisional translation The Food Safety Commission Final decision on September 7, 2017 This English version of the Commission Decision is intended to be reference material to provide convenience for users.
Provisional translation The Food Safety Commission Final decision on September 7, 2017 This English version of the Commission Decision is intended to be reference material to provide convenience for users.
Cysteine Peptide Scientific Review, Dr. S. Dudek, DMV International
 Cysteine Peptide Scientific Review, Dr. S. Dudek, DMV International Ethanol and Glutathione Reduced glutathione plays a critical role in cellular detoxification processes including the metabolism of peroxides,
Cysteine Peptide Scientific Review, Dr. S. Dudek, DMV International Ethanol and Glutathione Reduced glutathione plays a critical role in cellular detoxification processes including the metabolism of peroxides,
Supplementary Figure 1
 Supplementary Figure 1 Expression of apoptosis-related genes in tumor T reg cells. (a) Identification of FOXP3 T reg cells by FACS. CD45 + cells were gated as enriched lymphoid cell populations with low-granularity.
Supplementary Figure 1 Expression of apoptosis-related genes in tumor T reg cells. (a) Identification of FOXP3 T reg cells by FACS. CD45 + cells were gated as enriched lymphoid cell populations with low-granularity.
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):
 Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Summary & conclusion
 Summary & conclusion Cancer is the prime cause of death in developed countries and the second major cause of death in developing world. The early diagnosis is very crucial for the effective treatment of
Summary & conclusion Cancer is the prime cause of death in developed countries and the second major cause of death in developing world. The early diagnosis is very crucial for the effective treatment of
Mechanisms of Cell Injury: Loss of Calcium Homeostasis
 Mechanisms of Cell Injury: Loss of Calcium Homeostasis SCPA610: Cellular Pathology Amornrat N. Jensen, Ph.D. amornrat.nar@mahidol.ac.th Leading questions Why is intracellular calcium important for the
Mechanisms of Cell Injury: Loss of Calcium Homeostasis SCPA610: Cellular Pathology Amornrat N. Jensen, Ph.D. amornrat.nar@mahidol.ac.th Leading questions Why is intracellular calcium important for the
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Cell Injury MECHANISMS OF CELL INJURY
 Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Introduction to pathology lecture 5/ Cell injury apoptosis. Dr H Awad 2017/18
 Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22
 Supplementary Information High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22 Max Gulhane 1, Lydia Murray 1, Rohan Lourie 1, Hui Tong 1, Yong H. Sheng 1, Ran
Supplementary Information High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22 Max Gulhane 1, Lydia Murray 1, Rohan Lourie 1, Hui Tong 1, Yong H. Sheng 1, Ran
Chemical and Biochemical Mechanism Of Cell Injury.
 Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
INTRODUCTION. Liver is the main organ responsible for the major metabolic and secretory functions in the
 INTRODUCTION Liver is the main organ responsible for the major metabolic and secretory functions in the body and hence appears to be a sensitive target site for substances modulating biotransformation
INTRODUCTION Liver is the main organ responsible for the major metabolic and secretory functions in the body and hence appears to be a sensitive target site for substances modulating biotransformation
Pair-fed % inkt cells 0.5. EtOH 0.0
 MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response. Supplementary Results
 Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response Shi-Ying Cai 1, Xinshou Ouyang 1, Yonglin Chen 1, Carol J. Soroka 1, Juxian Wang 2, Albert Mennone 1,
Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response Shi-Ying Cai 1, Xinshou Ouyang 1, Yonglin Chen 1, Carol J. Soroka 1, Juxian Wang 2, Albert Mennone 1,
Do Histological Features Inform DILI mechanisms?
 Do Histological Features Inform DILI mechanisms? Drug-Induced Liver Injury (DILI) Conference XVII June, 2017 David Kleiner, M.D., Ph.D. NCI/Laboratory of Pathology Disclosures No financial or other conflicts
Do Histological Features Inform DILI mechanisms? Drug-Induced Liver Injury (DILI) Conference XVII June, 2017 David Kleiner, M.D., Ph.D. NCI/Laboratory of Pathology Disclosures No financial or other conflicts
A complement-dependent balance between hepatic ischemia/reperfusion injury and liver regeneration in mice
 Research article A complement-dependent balance between hepatic ischemia/reperfusion injury and liver regeneration in mice Songqing He, 1,2 Carl Atkinson, 1 Fei Qiao, 1 Katherine Cianflone, 3 Xiaoping
Research article A complement-dependent balance between hepatic ischemia/reperfusion injury and liver regeneration in mice Songqing He, 1,2 Carl Atkinson, 1 Fei Qiao, 1 Katherine Cianflone, 3 Xiaoping
This student paper was written as an assignment in the graduate course
 77:222 Spring 2005 Free Radicals in Biology and Medicine Page 0 This student paper was written as an assignment in the graduate course Free Radicals in Biology and Medicine (77:222, Spring 2005) offered
77:222 Spring 2005 Free Radicals in Biology and Medicine Page 0 This student paper was written as an assignment in the graduate course Free Radicals in Biology and Medicine (77:222, Spring 2005) offered
Exploring a Link Between Spy1 and Hepatocellular Carcinoma Progression
 University of Windsor Scholarship at UWindsor UWill Discover Undergraduate Conference UWill Discover 2016 Mar 29th, 4:00 PM - 5:00 PM Exploring a Link Between Spy1 and Hepatocellular Carcinoma Progression
University of Windsor Scholarship at UWindsor UWill Discover Undergraduate Conference UWill Discover 2016 Mar 29th, 4:00 PM - 5:00 PM Exploring a Link Between Spy1 and Hepatocellular Carcinoma Progression
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Glutathione Regulation
 The Virtual Free Radical School Glutathione Regulation Dale A. Dickinson 1, Henry Jay Forman 1 and Shelly C. Lu 2 1 University of California, Merced, School of Natural Sciences, P.O. Box 2039, Merced,
The Virtual Free Radical School Glutathione Regulation Dale A. Dickinson 1, Henry Jay Forman 1 and Shelly C. Lu 2 1 University of California, Merced, School of Natural Sciences, P.O. Box 2039, Merced,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
Glutathione / Thioredoxin Nrf2 & Hyperoxia
 Glutathione / Thioredoxin Nrf2 & Hyperoxia Trent E. Tipple, MD Principal Investigator, Center for Perinatal Research The Research Institute at Nationwide Children s Hospital Assistant Professor of Pediatrics,
Glutathione / Thioredoxin Nrf2 & Hyperoxia Trent E. Tipple, MD Principal Investigator, Center for Perinatal Research The Research Institute at Nationwide Children s Hospital Assistant Professor of Pediatrics,
Supplementary Figure 1.
 Supplementary Figure 1. Visualization of endoplasmic reticulum-mitochondria interaction by in situ proximity ligation assay. A) Illustration of targeted proteins in mitochondria (M), endoplasmic reticulum
Supplementary Figure 1. Visualization of endoplasmic reticulum-mitochondria interaction by in situ proximity ligation assay. A) Illustration of targeted proteins in mitochondria (M), endoplasmic reticulum
Assoc. Prof. Dr. Aslı Korkmaz* and Prof. Dr. Dürdane Kolankaya**
 The possible protective effects of some flavonoids that found honey by experimental ischemia/reperfusion (I/R) induced nitrosative damage in kidney of male rats Assoc. Prof. Dr. Aslı Korkmaz* and Prof.
The possible protective effects of some flavonoids that found honey by experimental ischemia/reperfusion (I/R) induced nitrosative damage in kidney of male rats Assoc. Prof. Dr. Aslı Korkmaz* and Prof.
2. List routes of exposure in the order of most rapid response.
 Practice Test questions: 1. What are the two areas of toxicology that a regulatory toxicologist must integrate in order to determine the "safety" of any chemical? 2. List routes of exposure in the order
Practice Test questions: 1. What are the two areas of toxicology that a regulatory toxicologist must integrate in order to determine the "safety" of any chemical? 2. List routes of exposure in the order
Antibodies for Unfolded Protein Response
 Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
Chapter 4. Drug Biotransformation
 Chapter 4 Drug Biotransformation Drug Biotransformation 1 Why is drug biotransformation necessary 2 The role of biotransformation in drug disposition 3 Where do drug biotransformation occur 4 The enzymes
Chapter 4 Drug Biotransformation Drug Biotransformation 1 Why is drug biotransformation necessary 2 The role of biotransformation in drug disposition 3 Where do drug biotransformation occur 4 The enzymes
ulcer healing role 118 Bicarbonate, prostaglandins in duodenal cytoprotection 235, 236
 Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Reperfusion Injury: How Can We Reduce It?
 MI/CAD: Practical Question in Management of AMI Patients Reperfusion Injury: How Can We Reduce It? Hyun-Jai Cho, M.D., Ph.D Cardiovascular Center & Department of Internal Medicine Seoul National University
MI/CAD: Practical Question in Management of AMI Patients Reperfusion Injury: How Can We Reduce It? Hyun-Jai Cho, M.D., Ph.D Cardiovascular Center & Department of Internal Medicine Seoul National University
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Is there a common mechanism of DILI, do we need to know?
 Is there a common mechanism of DILI, do we need to know? Neil Kaplowitz, MD USC Research Center for Liver Disease Los Angeles, California IS THERE A COMMON MECHANISM OF DILI? ANSWER YES & NO Mechanisms
Is there a common mechanism of DILI, do we need to know? Neil Kaplowitz, MD USC Research Center for Liver Disease Los Angeles, California IS THERE A COMMON MECHANISM OF DILI? ANSWER YES & NO Mechanisms
Dr. Hany M. Abo-Haded
 BY Dr. Hany M. Abo-Haded Pediatric Cardiology Unit, Mansoura Universty Children Hospital, Mansoura, Egypt Co-authors: Dina S El-Agamy and Mohamed A Elkablawy Background Doxorubicin (DOX) is an anthracycline
BY Dr. Hany M. Abo-Haded Pediatric Cardiology Unit, Mansoura Universty Children Hospital, Mansoura, Egypt Co-authors: Dina S El-Agamy and Mohamed A Elkablawy Background Doxorubicin (DOX) is an anthracycline
Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury
 Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury Swapan K. Ray, PhD Professor, Department of Pathology, Microbiology, and Immunology USC School of Medicine,
Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury Swapan K. Ray, PhD Professor, Department of Pathology, Microbiology, and Immunology USC School of Medicine,
PUBLICATIONS. cells. J. Physiol. (London) 517P:91P (Manchester, England, UK).
 277 PUBLICATIONS Abstracts Haddad JJ, Land SC (1999). Differential activation of oxygen-responsive transcription factors over fetal-to-neonatal alveolar oxygen tensions in rat fetal distal lung epithelial
277 PUBLICATIONS Abstracts Haddad JJ, Land SC (1999). Differential activation of oxygen-responsive transcription factors over fetal-to-neonatal alveolar oxygen tensions in rat fetal distal lung epithelial
BRIDGE THE GAP A Human Pathways Approach to Disease Research
 BRIDGE THE GAP A Human Pathways Approach to Disease Research BIOMED 21 Brussels December 8 th, 2015 Brigitte Landesmann Systems Toxicology Unit and EURL ECVAM Institute for Health and Consumer Protection
BRIDGE THE GAP A Human Pathways Approach to Disease Research BIOMED 21 Brussels December 8 th, 2015 Brigitte Landesmann Systems Toxicology Unit and EURL ECVAM Institute for Health and Consumer Protection
Supplementary Figure 1
 Supplementary Figure 1 A B mir-141, human cell lines mir-2c, human cell lines mir-141, hepatocytes mir-2c, hepatocytes Relative RNA.1.8.6.4.2 Relative RNA.3.2.1 Relative RNA 1.5 1..5 Relative RNA 2. 1.5
Supplementary Figure 1 A B mir-141, human cell lines mir-2c, human cell lines mir-141, hepatocytes mir-2c, hepatocytes Relative RNA.1.8.6.4.2 Relative RNA.3.2.1 Relative RNA 1.5 1..5 Relative RNA 2. 1.5
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo
 Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo BERNHARD H. LAUTERBURG, GEORGE B. CORCORAN, and JERRY R. MITCHELL, Baylor College of
Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo BERNHARD H. LAUTERBURG, GEORGE B. CORCORAN, and JERRY R. MITCHELL, Baylor College of
The Enhancement of Toxin-Induced Liver Fibrosis in Fatty Liver Disease. Ekihiro Seki, M.D.,Ph.D. University of California San Diego
 The Enhancement of Toxin-Induced Liver Fibrosis in Fatty Liver Disease Ekihiro Seki, M.D.,Ph.D. University of California San Diego - A manufactured chemical. - Does not exist naturally. Carbon tetrachloride
The Enhancement of Toxin-Induced Liver Fibrosis in Fatty Liver Disease Ekihiro Seki, M.D.,Ph.D. University of California San Diego - A manufactured chemical. - Does not exist naturally. Carbon tetrachloride
Screening of novel thalidomide analogues with label-free assays and appling in the in vivo model of hepatocellular carcinoma.
 Screening of novel thalidomide analogues with label-free assays and appling in the in vivo model of hepatocellular carcinoma Lajos István Nagy Ph.D thesis summary Supervisor: László Puskás, PhD, DSc Scientific
Screening of novel thalidomide analogues with label-free assays and appling in the in vivo model of hepatocellular carcinoma Lajos István Nagy Ph.D thesis summary Supervisor: László Puskás, PhD, DSc Scientific
Subject Index. Bcl-2, apoptosis regulation Bone marrow, polymorphonuclear neutrophil release 24, 26
 Subject Index A1, apoptosis regulation 217, 218 Adaptive immunity, polymorphonuclear neutrophil role 31 33 Angiogenesis cancer 178 endometrium remodeling 172 HIV Tat induction mechanism 176 inflammatory
Subject Index A1, apoptosis regulation 217, 218 Adaptive immunity, polymorphonuclear neutrophil role 31 33 Angiogenesis cancer 178 endometrium remodeling 172 HIV Tat induction mechanism 176 inflammatory
Plasma exposure levels from individual mice 4 hours post IP administration at the
 Supplemental Figure Legends Figure S1. Plasma exposure levels of MKC-3946 in mice. Plasma exposure levels from individual mice 4 hours post IP administration at the indicated dose mg/kg. Data represent
Supplemental Figure Legends Figure S1. Plasma exposure levels of MKC-3946 in mice. Plasma exposure levels from individual mice 4 hours post IP administration at the indicated dose mg/kg. Data represent
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
NASH Bench to Bedside
 NASH Bench to Bedside October 2006 Anna Mae Diehl, M.D. Gastroenterology Division Duke University NonAlcoholic Fatty Liver Disease Common ~1/4-1/3 1/3 US adults Outcome highly variable Course indolent
NASH Bench to Bedside October 2006 Anna Mae Diehl, M.D. Gastroenterology Division Duke University NonAlcoholic Fatty Liver Disease Common ~1/4-1/3 1/3 US adults Outcome highly variable Course indolent
Protein isolate from the herb, Phyllanthus niruri, protects liver from acetaminophen induced toxicity
 Protein isolate from the herb, Phyllanthus niruri, protects liver from acetaminophen induced toxicity Author(s): Rajesh Bhattacharjee and Parames C. Sil Vol. 17, No. 1 (2006-01 - 2006-04) Biomedical Research
Protein isolate from the herb, Phyllanthus niruri, protects liver from acetaminophen induced toxicity Author(s): Rajesh Bhattacharjee and Parames C. Sil Vol. 17, No. 1 (2006-01 - 2006-04) Biomedical Research
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Therapies for DILI: NAC, Steroids or NRF-2 activators?
 Therapies for DILI: NAC, Steroids or NRF-2 activators? William M. Lee, MD Professor of Internal Medicine Meredith Mosle Chair in Liver Diseases UT Southwestern Medical Center at Dallas Drug-Induced Liver
Therapies for DILI: NAC, Steroids or NRF-2 activators? William M. Lee, MD Professor of Internal Medicine Meredith Mosle Chair in Liver Diseases UT Southwestern Medical Center at Dallas Drug-Induced Liver
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury
 J Mol Med 2015 HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury Sara Karsikas; Mikko Myllymäki; Minna Heikkilä; Raija Sormunen; Kari I Kivirikko; Johanna Myllyharju; Raisa
J Mol Med 2015 HIF-P4H-2 deficiency protects against skeletal muscle ischemia-reperfusion injury Sara Karsikas; Mikko Myllymäki; Minna Heikkilä; Raija Sormunen; Kari I Kivirikko; Johanna Myllyharju; Raisa
NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update
 NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update 1 Overview The natural growth factor IGF-1 is broken down in the body to IGF-1[1-3] NNZ-2566 is an analogue of IGF-1[1-3] developed
NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update 1 Overview The natural growth factor IGF-1 is broken down in the body to IGF-1[1-3] NNZ-2566 is an analogue of IGF-1[1-3] developed
Acetaminophen (APAP) is a safe analgesic at
 Novel Mechanisms of Protection Against Acetaminophen Hepatotoxicity in Mice by Glutathione and N-Acetylcysteine Chieko Saito, 1 Claudia Zwingmann, 2 and Hartmut Jaeschke 1 Acetaminophen (APAP) overdose
Novel Mechanisms of Protection Against Acetaminophen Hepatotoxicity in Mice by Glutathione and N-Acetylcysteine Chieko Saito, 1 Claudia Zwingmann, 2 and Hartmut Jaeschke 1 Acetaminophen (APAP) overdose
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Biodegradable Zwitterionic Nanogels with Long. Circulation for Antitumor Drug Delivery
 Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment
 Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment Pacific Northwest Bio Meeting Seattle, WA, August 14, 2012 Thomas A. Baillie, PhD, DSc
Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment Pacific Northwest Bio Meeting Seattle, WA, August 14, 2012 Thomas A. Baillie, PhD, DSc
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Acute lung injury in children : from viral infection and mechanical ventilation to inflammation and apoptosis Bern, R.A.
 UvA-DARE (Digital Academic Repository) Acute lung injury in children : from viral infection and mechanical ventilation to inflammation and apoptosis Bern, R.A. Link to publication Citation for published
UvA-DARE (Digital Academic Repository) Acute lung injury in children : from viral infection and mechanical ventilation to inflammation and apoptosis Bern, R.A. Link to publication Citation for published
An Analysis of N-Acetylcysteine Treatment for Acetaminophen Overdose Using A Systems Model of Drug-Induced Liver Injury. Supplementary Materials
 An Analysis of N-Acetylcysteine Treatment for Acetaminophen Overdose Using A Systems Model of Drug-Induced Liver Injury Supplementary Materials Jeffrey L. Woodhead, Brett A. Howell, Yuching Yang, Alison
An Analysis of N-Acetylcysteine Treatment for Acetaminophen Overdose Using A Systems Model of Drug-Induced Liver Injury Supplementary Materials Jeffrey L. Woodhead, Brett A. Howell, Yuching Yang, Alison
NON-ALCOHOLIC STEATOHEPATITIS AND NON-ALCOHOLIC FATTY LIVER DISEASES
 NON-ALCOHOLIC STEATOHEPATITIS AND NON-ALCOHOLIC FATTY LIVER DISEASES Preface Zobair M. Younossi xiii Epidemiology and Natural History of NAFLD and NASH 1 Janus P. Ong and Zobair M. Younossi Understanding
NON-ALCOHOLIC STEATOHEPATITIS AND NON-ALCOHOLIC FATTY LIVER DISEASES Preface Zobair M. Younossi xiii Epidemiology and Natural History of NAFLD and NASH 1 Janus P. Ong and Zobair M. Younossi Understanding
Effect of N-Acetylcysteine on Acetaminophen Toxicity in Mice: Relationship to Reactive Nitrogen and Cytokine Formation
 TOXICOLOGICAL SCIENCES 75, 458 467 (2003) DOI: 10.1093/toxsci/kfg181 Effect of N-Acetylcysteine on Acetaminophen Toxicity in Mice: Relationship to Reactive Nitrogen and Cytokine Formation Laura P. James,*,,1
TOXICOLOGICAL SCIENCES 75, 458 467 (2003) DOI: 10.1093/toxsci/kfg181 Effect of N-Acetylcysteine on Acetaminophen Toxicity in Mice: Relationship to Reactive Nitrogen and Cytokine Formation Laura P. James,*,,1
paracetamol overdose: evidence for early hepatocellular damage
 Gut, 1985, 26, 26-31 Plasma glutathione S-transferase measurements after paracetamol overdose: evidence for early hepatocellular damage G J BECKETT, B J CHAPMAN, E H DYSON, AND J D HAYES From the University
Gut, 1985, 26, 26-31 Plasma glutathione S-transferase measurements after paracetamol overdose: evidence for early hepatocellular damage G J BECKETT, B J CHAPMAN, E H DYSON, AND J D HAYES From the University
Serum microrna biomarkers for human DILI
 CLEARANCE DRUG bioactivation covalent binding Detoxication GLUCURONIDE SULPHATE DRUG + METABOLITE chemical stress mitochondrial dysfunction apoptosis necrosis hepatocyte hypertrophy hepatocyte hyperplasia
CLEARANCE DRUG bioactivation covalent binding Detoxication GLUCURONIDE SULPHATE DRUG + METABOLITE chemical stress mitochondrial dysfunction apoptosis necrosis hepatocyte hypertrophy hepatocyte hyperplasia
OSAS oxidative stress and atherosclerosis Who is the culprit: AHI or ODI
 OSAS oxidative stress and atherosclerosis Who is the culprit: AHI or ODI Arie Wollner MD, FCCP Department of Respiratory Care and Rehabilitation Sheba Medical Center OSA as a major public health problem
OSAS oxidative stress and atherosclerosis Who is the culprit: AHI or ODI Arie Wollner MD, FCCP Department of Respiratory Care and Rehabilitation Sheba Medical Center OSA as a major public health problem
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
DILI: Clinical Pharmacology Considerations for Risk Assessment
 DILI: Clinical Pharmacology Considerations for Risk Assessment Raj Madabushi, PhD Office of Clinical Pharmacology Drug-Induced Liver Injury (DILI) Conference XVII June 06, 2017 Disclaimer: The views expressed
DILI: Clinical Pharmacology Considerations for Risk Assessment Raj Madabushi, PhD Office of Clinical Pharmacology Drug-Induced Liver Injury (DILI) Conference XVII June 06, 2017 Disclaimer: The views expressed
Stimulating healthy tissue regeneration by targeting the 5-HT 2B receptor in chronic liver disease.
 Stimulating healthy tissue regeneration by targeting the 5-HT 2B receptor in chronic liver disease. Mohammad R Ebrahimkhani, Fiona Oakley, Lindsay B Murphy, Jelena Mann, Anna Moles, Maria J Perugorria,
Stimulating healthy tissue regeneration by targeting the 5-HT 2B receptor in chronic liver disease. Mohammad R Ebrahimkhani, Fiona Oakley, Lindsay B Murphy, Jelena Mann, Anna Moles, Maria J Perugorria,
THBA Platform - Bile acid imbalance
 - Bile acid imbalance Bile acids play an important role in maintaining human health by means of signaling molecules in the regulation of bile formation, liver function and metabolism. The detergent effect
- Bile acid imbalance Bile acids play an important role in maintaining human health by means of signaling molecules in the regulation of bile formation, liver function and metabolism. The detergent effect
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Fat, ballooning, plasma cells and a +ANA. Yikes! USCAP 2016 Evening Specialty Conference Cynthia Guy
 Fat, ballooning, plasma cells and a +ANA. Yikes! USCAP 2016 Evening Specialty Conference Cynthia Guy Goals Share an interesting case Important because it highlights a common problem that we re likely to
Fat, ballooning, plasma cells and a +ANA. Yikes! USCAP 2016 Evening Specialty Conference Cynthia Guy Goals Share an interesting case Important because it highlights a common problem that we re likely to
Dietary supplementation in treating non-alcoholic fatty liver disease Dr. Ahmad Saedi Associate Professor School of Nutritional Sciences and
 Dietary supplementation in treating non-alcoholic fatty liver disease Dr. Ahmad Saedi Associate Professor School of Nutritional Sciences and Dietetics Tehran University of Medical Sciences Honorary Academic
Dietary supplementation in treating non-alcoholic fatty liver disease Dr. Ahmad Saedi Associate Professor School of Nutritional Sciences and Dietetics Tehran University of Medical Sciences Honorary Academic
Biology and Medicine
 eissn: 09748369 Anti-oxidative and hepatoprotective effect of beta-carotene on acetaminophen-induced liver damage in rats Biology and Medicine Research Article Volume 4, Issue 3, Pages 134 140, 2012 www.biolmedonline.com
eissn: 09748369 Anti-oxidative and hepatoprotective effect of beta-carotene on acetaminophen-induced liver damage in rats Biology and Medicine Research Article Volume 4, Issue 3, Pages 134 140, 2012 www.biolmedonline.com
Acetaminophen-Induced Hepatotoxicity and Protein Nitration in Neuronal. Nitric Oxide Synthase Knockout Mice
 JPET Fast This article Forward. has not Published been copyedited on and October formatted. 14, The final 2011 version as DOI:10.1124/jpet.111.184192 may differ from this version. Title page Acetaminophen-Induced
JPET Fast This article Forward. has not Published been copyedited on and October formatted. 14, The final 2011 version as DOI:10.1124/jpet.111.184192 may differ from this version. Title page Acetaminophen-Induced
Ginkgo biloba extract postconditioning reduces myocardial ischemia reperfusion injury
 Ginkgo biloba extract postconditioning reduces myocardial ischemia reperfusion injury K. Ran 1, D.-L. Yang 1, Y.-T. Chang 1, K.-M. Duan 2, Y.-W. Ou 2, H.-P. Wang 3 and Z.-J. Li 1 1 Department of Anesthesiology,
Ginkgo biloba extract postconditioning reduces myocardial ischemia reperfusion injury K. Ran 1, D.-L. Yang 1, Y.-T. Chang 1, K.-M. Duan 2, Y.-W. Ou 2, H.-P. Wang 3 and Z.-J. Li 1 1 Department of Anesthesiology,
microrna Presented for: Presented by: Date:
 microrna Presented for: Presented by: Date: 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions
microrna Presented for: Presented by: Date: 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions
Suspected Isoflurane Induced Hepatitis from Cross Sensitivity in a Post Transplant for Fulminant Hepatitis from Halothane.
 ISPUB.COM The Internet Journal of Anesthesiology Volume 25 Number 1 Suspected Isoflurane Induced Hepatitis from Cross Sensitivity in a Post Transplant for Fulminant Hepatitis from Halothane. V Sampathi,
ISPUB.COM The Internet Journal of Anesthesiology Volume 25 Number 1 Suspected Isoflurane Induced Hepatitis from Cross Sensitivity in a Post Transplant for Fulminant Hepatitis from Halothane. V Sampathi,
Role of Yes-Associated Protein (YAP) in Liver Injury and Regeneration following. Acetaminophen Overdose. Samikshya Poudel
 Role of Yes-Associated Protein (YAP) in Liver Injury and Regeneration following Acetaminophen Overdose By Samikshya Poudel Submitted to the graduate degree program in Toxicology and the Graduate Faculty
Role of Yes-Associated Protein (YAP) in Liver Injury and Regeneration following Acetaminophen Overdose By Samikshya Poudel Submitted to the graduate degree program in Toxicology and the Graduate Faculty
Supplementary Figure 1. ETBF activate Stat3 in B6 and Min mice colons
 Supplementary Figure 1 ETBF activate Stat3 in B6 and Min mice colons a pstat3 controls Pos Neg ETBF 1 2 3 4 b pstat1 pstat2 pstat3 pstat4 pstat5 pstat6 Actin Figure Legend: (a) ETBF induce predominantly
Supplementary Figure 1 ETBF activate Stat3 in B6 and Min mice colons a pstat3 controls Pos Neg ETBF 1 2 3 4 b pstat1 pstat2 pstat3 pstat4 pstat5 pstat6 Actin Figure Legend: (a) ETBF induce predominantly
Mechanisms of Cell Injury
 Causes of Cell Injury 1- oxygen deprivation (anoxia) 2- physical agents 3- chemical agents 4- infections agents 5- immunologic reactions 6- genetic defects 7- nutritional imbalances Mechanisms of Cell
Causes of Cell Injury 1- oxygen deprivation (anoxia) 2- physical agents 3- chemical agents 4- infections agents 5- immunologic reactions 6- genetic defects 7- nutritional imbalances Mechanisms of Cell
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Hepato-Protective Effect of Karisalai Karpa Chooranam against Paracetamol Induced Hepato Toxicity in Zebra fish (Daniorerio) Model
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 17, Issue 6 Ver. 12 (June. 2018), PP 51-55 www.iosrjournals.org Hepato-Protective Effect of Karisalai
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 17, Issue 6 Ver. 12 (June. 2018), PP 51-55 www.iosrjournals.org Hepato-Protective Effect of Karisalai
Published on Second Faculty of Medicine, Charles University (http://www.lf2.cuni.cz )
 Published on Second Faculty of Medicine, Charles University (http://www.lf2.cuni.cz ) Biochemistry Submitted by Marie Havlová on 8. February 2012-0:00 Syllabus of Biochemistry Mechanisms of enzyme catalysis.
Published on Second Faculty of Medicine, Charles University (http://www.lf2.cuni.cz ) Biochemistry Submitted by Marie Havlová on 8. February 2012-0:00 Syllabus of Biochemistry Mechanisms of enzyme catalysis.
OxiSelect Hydrogen Peroxide Assay Kit (Colorimetric)
 Product Manual OxiSelect Hydrogen Peroxide Assay Kit (Colorimetric) Catalog Number STA-343 5 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Oxidative stress is a physiological
Product Manual OxiSelect Hydrogen Peroxide Assay Kit (Colorimetric) Catalog Number STA-343 5 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Oxidative stress is a physiological
