TISSUE-SPECIFIC STEM CELLS
|
|
|
- Barbra Wheeler
- 5 years ago
- Views:
Transcription
1 TISSUE-SPECIFIC STEM CELLS Thrombopoietin Enhances Generation of CD34 Cells from Human Embryonic Stem Cells ANAND S. SRIVASTAVA, a ELENA NEDELCU, b BABAK ESMAELI-AZAD, c RANGNATH MISHRA, d EWA CARRIER a a Moores UCSD Cancer Center, Department of Medicine, University of California San Diego, San Diego, California, USA; b Department of Pathology, University of California Los Angeles, Los Angeles, California, USA; c DNAmicroarray, Inc., San Diego, California, USA; d Division of Nephrology, Department of Medicine, Case Western Reserve University, Cleveland, Ohio, USA Key Words. Human embryonic stem cells CD34 progenitors Hematopoietic progenitors CD31 progenitors Disclosure of potential conflicts of interest is found at the end of this article. ABSTRACT The role of thrombopoietin (TPO) in adult hematopoiesis is well-established. A recent report suggests that TPO and vascular endothelial growth factor (VEGF) play a role in promoting formation of early erythropoietic progenitors in a nonhuman primate embryonic stem cell (ES) model. No such report exists for human ES cells as yet. Because TPO may become an important factor promoting human ES cell-derived hematopoiesis, we sought to investigate whether TPO in combination with VEGF can enhance human ES-derived hematopoiesis in an EB-derived culture system. The emphasis of this work was to demonstrate the molecular mechanisms involved in this process, specifically the role of c-mpl and its ligand TPO. Human ES cells were cultured to the EB state, and EB-derived secondary cultures supporting hematopoietic differentiation were established: condition 1, control (stem cell factor [SCF] and Flt3 ligand [Flt3L]); condition 2, SCF, Flt3L, and TPO; and condition 3, SCF, Flt3L, TPO, and VEGF. Cells were harvested daily, starting at day 2 and continuing until day 8, for reverse transcription-polymerase chain reaction and Western blot. There was no evidence of expression of c-mpl and VEGF receptor on the gene or protein level until day 8, when the formation of well-established hematopoietic colonies began. This correlated with the formation of CD34 /CD31 negative progenitors, mostly found in blast-forming units-erythroid-like colonies. We concluded that TPO and VEGF play an important synergistic role in the formation of early ES-derived hematopoietic progenitors that occurs through the c-mpl and VEGF receptors. STEM CELLS 2007;25: INTRODUCTION Thrombopoietin (TPO), a primary regulator of megakaryocyte and platelet production in vitro and in vivo, acts as a lineagespecific hematopoietic growth factor and exerts effects on early hematopoietic stem cells [1]. There are two lines of evidence supporting TPO in the survival and expansion of hematopoietic stem cells. The first line of evidence comes from in vitro and in vivo experimental studies. In vitro studies demonstrated that TPO supports survival of hematopoietic stem cells (HSCs) and stimulates their proliferation when added in the presence of interleukin 3 or steel factor to the cultures of purified populations of candidate stem cells [2]. TPO stimulates stem cells to generate colony-forming cells of all lineages. TPO not only supports the growth of megakaryocyte progenitors [3] but also enhances the proliferation of erythroid progenitors [4 6]. In vivo studies also point out the crucial role of TPO in the early development of hematopoietic stem cells; knockout mice in which genes coding for TPO or its receptor (c-mpl) were deleted had a profound reduction not only in the number of megakaryocytes and platelets but also in the number of committed myeloid progenitors of all types (primitive progenitors and hematopoietic stem cells). Therefore, it appears that TPO is not only a lineage-specific growth factor but also plays an important role in the proliferation and differentiation of hematopoietic stem cells [7]. Administration of recombinant TPO leads to a significant expansion of colony-forming units of all lineages in both normal and myelosuppressed mice [7, 8]. The second line of evidence comes from clinical studies of patients with amegakaryocytic thrombocytopenia. Recently, it was shown that this disease is caused by the mutations in the c-mpl gene, leading to the unresponsiveness of hematopoietic stem cells to the role of TPO. All these patients developed pancytopenia within the first 3 5 years of life, suggesting that TPO has a role in early hematopoiesis in humans [9]. However, little is known about the role of TPO in the generation of hematopoietic stem cells from human embryonic stem (ES) cells. For the study of recapitulation of early hematopoietic events, the EB differentiation model is frequently used [10]. Previous studies have demonstrated that mouse-derived EB cells generated blast cell colonies and had hematopoietic and endothelial potential when cultured in endothelial cell-conditioned medium in the presence of vascular endothelial growth factor (VEGF) and interleukin 6 (IL-6) [11]. It is thought that the blast colony-forming cells, present in the mouse EBs for approximately 24 hours, represent the in vivo yolk sac progenitor of Correspondence: Ewa Carrier, M.D., Department of Medicine, Pediatrics and UCSD Moores Cancer Center, School of Medicine, University of California San Diego, 9500 Gilman Drive, La Jolla, California 92093, USA. Telephone: ; Fax: ; ecarrier@ucsd.edu Received November 3, 2006; accepted for publication March 8, 2007; first published online in STEM CELLS EXPRESS March 22, AlphaMed Press /2007/$30.00/0 doi: /stemcells STEM CELLS 2007;25:
2 Srivastava, Nedelcu, Esmaeli-Azad et al dual hematopoietic and endothelial potential [12]. In another independent study, these vascular endothelial growth factor receptor 2 (VEGFR2)-positive progenitors generated blood islands composed of round clusters of hematopoietic cells surrounded by both endothelial and hematopoietic cells [13]. Previous studies have shown that hematopoietic differentiation of human ES cells using the EB differentiation model requires bone morphogenic protein (BMP4) in combination with a few other cytokines, such as stem cell factor (SCF), Flt3 ligand (Flt3L), IL-3, IL-6, and granulocyte macrophage-colony stimulating factor [14, 15]. However, a recent report demonstrated that TPO regulates proliferation and differentiation of rhesus EBs to hematopoietic and endothelial progenitors without BMP4 but instead required VEGF, SCF, and Flt3L [16]. TPO alone was not able to stimulate formation of hematopoietic and endothelial progenitors, whereas the addition of VEGF enhanced their generation. In these conditions, the mrna expression of c-mpl was also significantly upregulated, allowing for enhanced responsiveness of early hematopoietic and endothelial progenitors to TPO. It is known that addition of TPO increased formation of primitive hematopoietic progenitors in murine and primate EB differentiation models [16]; however, no such report currently exists for human ES cells. Since EB-derived hematopoiesis represents a good system model with which to study in vitro early hematopoietic events in humans, investigating the role of TPO and VEGF and their respective receptors on early hematopoietic events may enhance future protocols of blood formation from human ES cells. MATERIALS AND METHODS Human ES Cell Culture Conditions Human ES (hes) cell lines (H1 cell line, NIH code WA01) were obtained from WiCell Research Institute (Madison, WI, wicell.org) [10]. hes cells were cultured on mitomycin-treated mouse embryonic fibroblast cells in Dulbecco s modified Eagle s medium (DMEM)/Ham s F-12 medium supplemented with 15% knockout serum replacement (Invitrogen, Carlsbad, CA, ng/ml of human recombinant basic fibroblast growth factor 2 (FGF2) (Invitrogen), 1% nonessential amino acids, and 1% L-glutamine. The cells were fed daily and passaged weekly with 0.05% trypsin, 0.53 mmol/l EDTA (Invitrogen) to maintain undifferentiated growth [17]. EB Formation On the day of passage, the ES cells were treated with 200 U/ml collagenase IV (Invitrogen), counted with a hemocytometer, and transferred at a density of cells per well to a low-attachment plates to allow EB formation by incubation in knockout DMEM supplemented with 20% non-heat-inactivated fetal bovine serum (FBS) (HyClone, Logan, UT, 1% nonessential amino acids, 1 mm L-glutamine, and 0.1 mm -mercaptoethanol. EBs were cultured for 2 days at 37 C and 5% CO 2 in a humidified atmosphere for 48 hours. The number of hebs was counted with a hemocytometer prior to transfer into a secondary medium conducive to hematopoietic differentiation. EB cells mechanically dispersed or obtained by dissociation with collagenase were used for the generation of CD34 cells, colony-forming units (CFU)-C assays, and molecular studies for mrna expression of TPO-R (c-mpl) and VEGFR. Generation of CD34 Cells from EB-Derived Cells After 3 days of culture, DMEM was replaced by Iscove s modified Dulbecco s medium (Gibco-BRL, Gaithersburg, MD, gibcobrl.com) with the same supplements and two additional cytokines (100 ng/ml SCF and 100 ng/ml Flt3L). This culture condition represented the control group. To investigate the role of TPO and VEGF, additional treatment with 100 ng/ml TPO alone (TPOtreated group) or in combination with 100 ng/ml recombinant human VEGF (TPO/VEGF-treated group) was performed. All cytokines were from R&D Systems Inc. (Minneapolis, rndsystems.com). Cytospin preparations were performed after 8 days of culture from these culture conditions, and the CD34 cells were detected by immunohistochemistry as described below. To detect the formation of double endothelio-hematopoietic and endothelial progenitors, CD31 staining was performed on all examined slides, which were read by primary and secondary (blinded) reviewers and compared against positive and negative controls. Histochemical Analysis and Quantification of hes- Derived CD34 Cells At day 8 of secondary differentiation culture, cells were collected, washed, and used for m-rna, CFU-C assay, and histochemical staining. Indirect immunohistochemistry was performed on cytospin preparations of cell cultures with TPO, TPO/VEGF, and control cultures (SCF and Flt3L). The antibodies used were rat anti-human CD34 and CD31 primary antibody and biotin-labeled anti-rat secondary antibody in a streptavidin horseradish peroxidase detection system. All antibodies were purchased from BD Pharmingen (San Diego, The slides were counterstained with hematoxylin and examined with an Olympus BH-2 microscope (Tokyo, photographs were taken with an Olympus DP11 camera, and membrane staining was correlated with cell morphology. Each group of cells was examined for the presence of cells with CD34 and CD31 membranous stain by a trained hematopathologist and a blinded second reader. The intensity of the staining was scored as follows: 1, weak; 2, moderate; 3, strong. The frequency of CD34 and CD31 cells was recorded for all staining intensities. The mean positivity index was calculated as a mean of all products between frequency of the CD34 and CD31 cells and corresponding staining intensity and correlated with morphology. Analysis of CFU-C Obtained from Plating EB Cells Secondary differentiation cultures were set up with cells dispersed from 2-day-old EBs. Cells were washed, and EB-dispersed cells were plated on 35-mm methylcellulose dishes. Human clonogenic progenitor assay was performed by plating cells obtained from hes-derived EBs into methylcellulose as described by Chadwick et al. [17]. Specific culture conditions were as follows: condition 1, SCF and Flt3L (control); condition 2, SCF, Flt3L, and TPO; and condition 3, SCF, Flt3L, TPO, and VEGF. The hematopoietic colonies were scored every second day up to 8 days and were considered positive when they were composed of 50 cells and exhibited typical CFU morphology. Quantitative comparisons were made between the control (e.g., SCF and Flt3L only) and each experimental condition (e.g., TPO with or without VEGF) with regard to the number of CFU derived per 100 cells from dissociated EB-derived cells (Fig. 1). Analysis of mrna Expression of c-mpl and VEGFR by Reverse Transcription-Polymerase Chain Reaction To detect the kinetics of expression of TPO and VEGF receptors (c-mpl and VEGFR, respectively) on cells subjected to three different culture conditions, cells were isolated daily from day 2 to day 8 of secondary differentiation cultures, and total RNA was isolated from these cells et each time point using an RNeasy Mini Kit according to the manufacturer s protocol (Qiagen, Valencia, CA, Five hundred nanograms of total RNA was subjected to study c-mpl and VEGR gene expression using a single-step reverse transcription-polymerase chain reaction (RT- PCR) kit (Invitrogen). The reaction was set for cdna synthesis and predenaturation for one cycle at 55 C for 30 minutes and finally 94 C for 2 minutes, which was followed by a DNA amplification cycle. The reaction was set for denaturation at 94 C for 15 seconds, annealing at 58 C for 30 seconds, and extension at 72 C for 1 minute for 35 cycles followed by a final extension at 72 C for 5
3 1458 TPO Enhances Hematopoietic Differentiation Figure 1. Generation of EB from human embryonic stem (ES) cells. To achieve EB formation, human ES cells were incubated in the Dulbecco s modified Eagle s medium without fibroblast growth factor 2 for 48 hours. Human ES cells formed the typical round EBs shown in photograph (phase contrast; magnification, 20). minutes. The -actin gene was used as an internal control. The following nucleotide sequences were used for primers: VEGFR forward, 5 -AGCCCAGATTCTCCAGCCTGACTCGG-3 ; VEGFR reverse, 5 -TGGGGCCATTGCTTGAAGCTCTTTGTTC-3 ; c-mpl forward, 5 -TGCCCTGCTTCTGCAGAGGCCTCACT-3 ; c-mpl reverse, 5 -CAGCACCGTGCCCTGCTGTGGTA-3 ; -actin forward, 5 -CTGTCTGGCGGCACCACCAT-3 ; -actin reverse, 5 -GCAAC- TAAGTCATAGTCCGC-3. Analysis of Protein Expression of c-mpl and VEGFR by Western Blot Cells were isolated daily from day 2 to day 8 of secondary differentiation cultures for protein analysis by Western blot. Cell homogenates (15% wt/vol) were prepared in extraction buffer composed of 50 mm PIPES (1,4-piperazinediethanesulfonic acid)/hcl, ph 6.5, 2 mm EDTA, 0.1% 3-([3-cholamidopropyl]dimethylammonio)-1- propanesulfonate, 20 g/ml leupeptin, 10 g/ml pepstatin A, 10 g/ml aprotinin, 5 mm dithiothreitol, 2 mm sodium pyrophosphate, 1mMNa 3 VO 4, and 1 mm NaF, and centrifuged at 2,000g for 10 minutes at 4 C. Protein content in the supernatant was assayed with a DC protein assay (Bio-Rad, Hercules, CA, com). An aliquot of the lysate (20 g of protein) was boiled with SDS sample buffer, resolved on a 4% 12% SDS-polyacrylamide gel electrophoresis gradient gel, and transferred to a 0.2- m nitrocellulose membrane. After blocking in 5% nonfat dry milk in Tris-buffered saline/tween 20 (TBS-T) (20 mm Tris-HCl, ph 7.5, 150 mm NaCl, 0.05% Tween 20) for 1 hour, the membrane was washed three times for 5 minutes each time with TBS-T and incubated overnight at 4 C with anti-human VEGF R2 monoclonal antibody (1:1,000; R&D Systems) or anti-human TPO-R antibody (1:1,000; R&D Systems) in 3% bovine serum albumin in TBS-T. After incubation with a suitable HRP-labeled secondary antibody (1:2,000) and extensive washing, the membrane was exposed to film with an average exposure duration ranging from 10 to 30 seconds. Statistical Analysis Statistical analysis was performed using the SPSS statistical package, version 9.0 (SPSS, Inc., Chicago, To analyze differences in mean values between groups, a two-sided unpaired Student s t test was used. RESULTS Human ES Cell Cultures We have established in vitro cultures of hes cells to study molecular mechanisms involved in the formation of early hematopoietic progenitors. These experiments were done using Figure 2. Number of CFU-C colonies in three different culture conditions. Number of CFUs obtained from human embryonic stem (hes) cells cultured in secondary differentiation media: left column, control media (SCF Flt3L); middle column, TPO added; right column, TPO and VEGF added. Results are presented as mean SEM. Significant differences were determined by Student s t test. Data show that TPO with VEGF significantly enhanced differentiation of hes into hematopoietic progenitors (, p 05). Abbreviations: TPO, thrombopoietin; VEGF, vascular endothelial growth factor. NIH-approved hes cell line H1 (code WA01). Human ES cells were maintained in the undifferentiated state by mitomycintreated murine feeder layer and serum-free media (Invitrogen) as described in Materials and Methods. In this culture condition, we have obtained stable proliferating hes cells, which formed typical, well-defined hes cell colonies (data not shown). Embryoid Body Formation To achieve EB formation, hes cells were incubated in DMEM without FGF2 for 48 hours. hes cells formed typical, round EBs (data not shown). The efficiency of plating was very high, and EBs per hes-plated cells were counted. In culture condition 1, which contained SCF and Flt3L only, the average number of CFU-C per 10 5 hes cells plated was 5. In culture condition 2, in which TPO was added, the mean number of CFU-C was 116 (p.001). In culture condition 3, TPO and VEGF were added together with the SCF and Flt3L, and a mean of 153 CFU-C per 10 5 cells plated were obtained (p.001), suggesting that TPO and VEGF exerted a synergistic effect on the formation of hematopoietic progenitors (Fig. 2); differences between cultures 2 and 3 were statistically significant (p.05). The first evidence of well-formed CFU-C was detected at day 8 of differentiation, which correlated with the expression of c-mpl and VEGFR. Formation of Early Hematopoietic Colonies Secondary differentiation cultures were set up with cells dispersed from 2-day-old EBs. EB-derived cells were washed, and 10 5 EB cells were plated on 35-mm methylcellulose-coated dishes. The condition media used in addition to standard media had SCF and Flt3L (control), TPO alone (medium 2), or TPO and VEGF (medium 3). Since we intended to study molecular mechanisms involved in the early hematopoietic events, CFU-C were scored daily up to day 8 using the method described by Chadwick et al. [17]. The first evidence of well-formed CFU-C colonies was detected at day 8 of differentiation, which coincided with the expression of c-mpl (TPO receptor) and VEGFR, indicating their role in the formation of hematopoietic progenitors. The standard blast-forming units-erythroid (BFU-E) morphology (clustered, large colonies with brown pigment), colonyforming units-granulocyte-macrophage (CFU-GM) (disperse
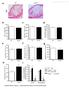
4 Srivastava, Nedelcu, Esmaeli-Azad et al Figure 3. Generation of colony-forming unit (CFU)-C colonies from human embryonic stem (ES) cells. Secondary differentiation cultures were set up with cells dispersed from 2-day-old EBs (48 hours in culture). Cells were washed, and EB-dispersed cells were plated on 35-mm methylcellulose dishes. Human clonogenic progenitor assay was performed by plating cells obtained from human ESderived EBs into methylcellulose as described by Chadwick et al. [17]. The first evidence of well-formed CFU-C colonies was detected at day 8 of differentiation, which coincided with the expression of c-mpl (thrombopoietin receptor) and vascular endothelial growth factor receptor, indicating their role in the formation of hematopoietic progenitors. The standard blast-forming units-erythroid morphology (clustered, large colonies with brown pigment; data not shown), colony-forming unitsgranulocyte-macrophage (disperse colonies with translucent cells), and CFU-E were detected. colonies with translucent cells), and CFU-E were detected (Fig. 1). Eighty percent of the CFU-C colonies corresponded to BFU-E morphology (large colonies composed of blast-like cells with reddish-brown pigment), although CFU-GM and CFU-E colonies were observed as well. No distinct megakaryocyte colonies were noted in these cultures, indicating that TPO alone is not sufficient for their formation. In the recent report by Gaur et al. [18], additional cytokines, such as IL-6 and IL-11, were required in addition to TPO for the formation of definite megakaryocytes, and longer incubation time was required. Quantitation of EB-Derived CD34-Positive Cells by Immunohistochemistry In this study, we wanted to correlate early hematopoietic differentiation with molecular events related to c-mpl and VEGR expression. The expression of the c-mpl receptor and VEGFR was tested daily by RT-PCR from day 2 to day 8 with no evidence of expression of their respective genes and proteins until day 8, suggesting that these receptors are involved in hematopoietic differentiation but not in early differentiation events. The CD31 staining in addition to CD34 staining was performed, with no evidence of CD31 staining, indicating that hematopoietic but not endothelial progenitors were formed in these culture conditions (TPO and VEGF). All slides were reviewed by primary and secondary (blinded) reviewers and discussed with a hematopathologist familiar with this staining. Microscopic examination revealed groups of cells positive for CD34 membranous staining but with different stain intensity. Weak CD34 staining (grade 1) was encountered in larger cells with more abundant cytoplasm and small nuclei, whereas strong staining (grade 3) was observed in smaller cells with a higher nuclear/cytoplasmic ratio (Fig. 4). There was no positive membrane staining for CD31, indicating that in these culture conditions (SCF, IL-3, TPO, and VEGF), endothelial progenitors were not formed. The percentage of cells positively staining for CD34 and the mean positivity index calculated as a mean of all products between frequency of the CD34 cells and corresponding staining intensity were both higher in cytospin preparation obtained from TPO and TPO/VEGF (15% vs. 5% and 10% vs. 3%, respectively), suggesting that TPO/VEGF culture conditions are inductive to the generation of ES-derived CD34 cells (Fig. 5). The two distinct morphologies observed may represent early erythropoietic progenitors (large cells with weak Figure 4. CD34 staining in cells obtained from hematopoietic colonies. CD34 cells derived from human embryonic stem cells showed staining of different intensities. Illustration of the intensity scoring system used for the CD34-positive cells: top left, negative control; top right, weak staining intensity (score 1); bottom left, moderate staining intensity (score 2); bottom right, strong staining intensity (score 3). Weak CD34 staining (score 1) was encountered in larger cells with more abundant cytoplasm and small nuclei, whereas strong staining (score 3) was observed in smaller cells with higher nuclear/cytoplasmic ratios. There was no positive membrane staining for CD31 indicating that in these culture conditions (SCF, interleukin 3, thrombopoietin, and vascular endothelial growth factor), endothelial progenitors were not formed. CD34 staining) and hematopoietic stem cells (small cells with strong CD34 staining), corresponding with the formation of CFU-C colonies. The percentage of staining cells for each culture condition is shown in Figure 5, and the intensity of staining is shown in Figure 6. TPO-Upregulated mrna Expression of VEGFR and c-mpl During Formation of CD34 Cells To investigate the mechanisms through which TPO alone or in combination with VEGF induced differentiation of hes cells to CD34 progenitors, we analyzed mrna expression of c-mpl and VEGFR from RNA obtained on days 2 8 of secondary differentiation cultures. Three culture conditions were established: condition 1, SCF and Flt3L (control); condition 2, SCF, Flt3, and TPO; and condition 3, SCF, Flt3, TPO, and VEGF. RT-PCR analysis showed no expression of c-mpl and VEGFR in progenitors up to seven days in secondary differentiation culture (data not shown). This correlated with the poor formation of CFU-C colonies prior to day 8 and no evidence of megakaryocyte formation. In our culture conditions (TPO and VEGF), the first transition occurred at day 8, when we observed formation of typical CFU-C colonies with predominantly BFU-E morphology, suggesting early erythropoiesis, induced by the combination of TPO and VEGF through their respective receptors. This was correlated with the expression of c-mpl and VEGFR as demonstrated by the RT-PCR (Fig. 7A) and Western blot analysis (Fig. 7). Progenitors obtained from these colonies expressed CD34 but no CD31 staining, indicating that at day 8 of differentiation, only hematopoietic progenitors were formed. Thus, it appears that in human EB-derived cultures, signaling through TPO receptor (c-mpl) activates signaling pathways involved in early hematopoiesis and that VEGF enhances this
5 1460 TPO Enhances Hematopoietic Differentiation Figure 5. Quantification of the human embryonic stem (hes) cellderived CD34 cells by the mean positivity index. The hes-derived CD34 cells were cultured with SCF and Flt3L (column 1, control cultures), thrombopoietin (TPO) alone (column 2), and both vascular endothelial growth factor (VEGF) and TPO (column 3). The highest number of CD34-positive cells was observed in the presence of cultures with TPO and both VEGF and TPO (in addition to SCF and Flt3L). Figure 6. Staining intensity of CD34 cells in three different culture conditions. Percentage of cells displaying strong, moderate, and weak staining was determined. Dotted columns, weak staining; solid columns, moderate staining; striped columns, strong staining. Set 1, thrombopoietin (TPO); set 2, TPO and vascular endothelial growth factor (VEGF); set 3, control (SCF and Flt3L). As shown, culture condition 2 (TPO and VEGF) induced 10% 15% of cells with strong CD34 staining. These cells showed specific morphology, displaying small size and a high nuclear/cytoplasmic ratio, suggesting generation of hematopoietic progenitors. None of these cells displayed CD31 staining (data not shown). mechanism. This process was independent of BMP4, which has been shown in other reports to effectively enhance human ES-derived hematopoiesis [17], but correlated with a recent report on nonhuman primate ES cells in which TPO and VEGF induced early erythroid differentiation [16]. Western Blot Analysis of c-mpl and VEGFR Proteins in Different ES Cell Culture Conditions To study expression of c-mpl and VEGFR on a protein level, Western blot analysis was performed on the progenitors obtained from day 2 to day 8 of secondary differentiation culture in three different conditions as described in Materials and Methods. No expression of c-mpl (Fig. 7B) or VEGFR (Fig. 7C) protein was noted up to day 8, suggesting that signaling through these receptors is initiated at the onset of early hematopoiesis (day 8), whereas signaling through receptors other than c-mpl and VEGF receptors may take place in early differentiation events, such as the hematoendothelial progenitor, sometimes referred to as hemangioblast. Upregulation of these receptors was observed in cultures with TPO, which was further enhanced Figure 7. TPO-R and VEGFR gene and protein expression in EBderived embryonic stem (ES) cell culture. (A): TPO-R and VEGFR gene expression during hematopoietic differentiation of ES cells exposed to different culture conditions by reverse transcription-polymerase chain reaction and exposed to different cytokines. mrna was collected after 8 days of secondary differentiation culture of human embryonic stem (hes) cells. Each sample was subjected to TPO-R and VEGFR gene expression studies. -Actin was used as internal control. Lanes 1 5 show expression of TPO-R; lanes 6 10 show expression of VEGFR. Lane 1, no cytokines; lane 2, Flt3 and SCF; lane 3, thrombopoietin (TPO), SCF and Flt3; lane 4, vascular endothelial growth factor (VEGF); lane 5, VEGF, TPO, SCF, and Flt3; lane 6, no cytokines; lane 7, Flt3 and SCF; lane 8, TPO; lane 9, VEGF, SCF, and Flt3; lane 10, VEGF and TPO. (B): Expression of TPO-R (c-mpl) protein in hes cells at day 8 in different differentiation media by Western blot. Upregulation of these receptors was observed in cultures with TPO, which was further enhanced when VEGF was added. Studies on progenitors prior to 8 days did not show expression of TPO-R and VEGFR (data not shown), which suggests that this signaling pathway operates later in the hematopoietic development. Lane 1, Flt3 and SCF; lane 2, TPO; lane 3, VEGF; lane 4, TPO and VEGF. (C): Expression of the VEGFR protein in hes cells at day 8 in different differentiation media. As in eight a, expression of VEGFR protein was noted on day 8, when the first colony-forming unit-c colonies were formed. No evidence of expression was noted on days 2 7 (data not shown). Lane 1, hes in Flt3 and SCF (control); lane 2, TPO; lane 3, VEGF; lane 4, TPO and VEGF. Abbreviations: TPO-R, thrombopoietin receptor; VEGFR, vascular endothelial growth factor receptor. with VEGF. No c-mpl or VEGFR proteins were detected in culture without these cytokines, indicating that signaling through these receptors in human EB-derived secondary cultures requires both TPO and VEGF. We did not observe megakaryocyte formation in these cultures, which were devoid of IL-6 and IL-11. According to recent report by Gaur et al. [18], formation of megakaryocytes from human ES cells occurs later in secondary differentiation culture. DISCUSSION The role of TPO on hematopoietic differentiation is well-established in the in vitro and in vivo systems, but its role in the human ES-derived hematopoiesis is not known. It was previously shown that TPO promotes hematopoietic differentiation of rhesus monkey ES cells [16]; however, its effect on the generation of CD34 cells from human ES cells has not been established. In this report, we provide experimental evidence supporting hypothesis that TPO has a positive effect on the
6 Srivastava, Nedelcu, Esmaeli-Azad et al survival and expansion of early hematopoietic progenitors derived from human ES cells and that c-mpl receptor plays a role in this process [19, 20]. It is known that patients with congenital amegakaryocytic thrombocytopenia, who have TPO-R mutations, develop not only thrombocytopenia but also pancytopenia [21, 22], suggesting that TPO not only acts as the main regulator of megakaryopoesis but also exerts its effect on early hematopoietic progenitors. In previous studies, the generation of CD34 cells from human ES cells was sixfold enhanced by addition of BMP4 to a mixture of cytokines such as SCF, Flt3L, IL-3, IL-6, and granulocyte-colony-stimulating factor in differentiation media [10]. Similarly, other researchers efficiently induced differentiation of hes cells into CD34 cells by coculturing them with the OP9 mouse bone marrow stromal cell line [23]. It is also well-established that the TPO-R is present on primitive hematopoietic progenitors [4, 24] but not on undifferentiated human ES cells. As the number of CFU-C colonies expanded with TPO and VEGF and most of the colonies found had BFU-E morphology, we hypothesized that the TPO and VEGF combination, in addition to standard hematopoietic cytokines, enhances production of early erythroid progenitors [4]. However, the question remains why megakaryocytes were not formed in those cultures in which TPO was used as a main additive cytokine. In a recent report by Gaur et al., the formation of megakaryocytes from human ES cells was obtained using OP9 stroma-enhanced cultures [18]. TPO alone was not sufficient for the induction of megakaryocytes, and other cytokines, such as IL-6 and IL-11, were required in addition to TPO for megakaryocyte formation. The mrna expression of both c-mpl and VEGFR was significantly upregulated in TPO- and TPO/VEGF-stimulated cell cultures versus cultures containing SCF and Flt3L, suggesting synergy between these cytokines. No expression of c-mpl and VEGF was documented in the control cultures, indicating that TPO and VEGF were essential in promoting expression of their respective receptors, as well as formation of early hematopoietic colonies through these receptors. However, the mechanisms responsible for these effects are not known. TPO-R is one of the important members of the so-called homodimerizing cytokine receptor subfamily that is known to activate many of the same pathways as erythropoietin receptor, JAK2, STAT5, mitogenactivated protein kinase, PI3 kinase [4, 24]. Our findings suggest that TPO may initially act through a different, common receptor on hes cells in preparation for a more specific signaling through its own receptor, when definite hematopoietic progenitors are formed. It is also possible that the hes cells decode the signals transmitted generically through cytokine receptors in similar manner or that TPO is part of the cytokine network regulating the proliferation and differentiation of hes cell-derived uncommitted progenitors [25]. These hypotheses are currently being tested in our laboratory. We demonstrate for the first time that TPO and VEGF induce early hematopoietic differentiation of EB-derived human ES cells and that c-mpl is involved in this process. Understanding of specific culture conditions and signaling pathways will allow optimization of the in vitro differentiation of hes cells toward hematopoietic progenitors and future ES-derived blood products. DISCLOSURE OF POTENTIAL CONFLICTS OF INTEREST The authors indicate no potential conflicts of interest. REFERENCES 1 Sitnicka E, Lin N, Priestley GV et al. The effect of thrombopoietin on the proliferation and differentiation of murine hematopoietic stem cells. Blood 1996;87: Kaushansky K, Lok S, Holly RD et al. Promotion of megakaryocyte progenitor expansion and differentiation by the c-mpl ligand thrombopoietin. Nature 1994;369: Lok S, Kaushansky K, Holly RD et al. Cloning and expression of murine thrombopoietin cdna and stimulation of platelet production in vivo. Nature 1994;369: Kobayashi M, Laver JH, Kato T et al. Recombinant human thrombopoietin (Mpl ligand) enhances proliferation of erythroid progenitors. Blood 1995;86: Kaushansky K, Broudy VC, Grossmann A et al. Thrombopoietin expands erythroid progenitors, increases red cell production, and enhances erythroid recovery after myelosuppressive therapy. J Clin Invest 1995; 96: Kaushansky K, Lin N, Grossmann A et al. Thrombopoietin expands erythroid, granulocyte-macrophage, and megakaryocytic progenitor cells in normal and myelosuppressed mice. Exp Hematol 1996;24: Alexander WS, Roberts AW, Nicola NA et al. Deficiencies in progenitor cells of multiple hematopoietic lineages and defective megakaryocytopoiesis in mice lacking the thrombopoietic receptor c-mpl. Blood 1996; 87: Carver-Moore K, Broxmeyer HE, Luoh SM et al. Low levels of erythroid and myeloid progenitors in thrombopoietin-and c-mpl-deficient mice. Blood 1996;88: Germeshausen M, Schulze H, Gaudig A et al. Congenital amegakaryocytic thrombocytopenia (CAMT): A defect of the thrombopoietin receptor c-mpl]. Klin Padiatr 2001;213: Kaufman DS, Hanson ET, Lewis RL et al. Hematopoietic colonyforming cells derived from human embryonic stem cells. Proc Natl Acad SciUSA2001;98: Jaffredo T, Bollerot K, Sugiyama D et al. Tracing the hemangioblast during embryogenesis: Developmental relationships between endothelial and hematopoietic cells. Int J Dev Biol 2005;49: Yamashita JK. Differentiation and diversification of vascular cells from embryonic stem cells. Int J Hematol 2004;80: Ema M, Faloon P, Zhang WJ et al. Combinatorial effects of Flk1 and Tal1 on vascular and hematopoietic development in the mouse. Genes Dev 2003;17: Cerdan C, Rouleau A, Bhatia M. VEGF-A165 augments erythropoietic development from human embryonic stem cells. Blood 2004;103: Li F, Lu S, Vida L et al. Bone morphogenetic protein 4 induces efficient hematopoietic differentiation of rhesus monkey embryonic stem cells in vitro. Blood 2001;98: Wang Z, Skokowa J, Pramono A et al. Thrombopoietin regulates differentiation of rhesus monkey embryonic stem cells to hematopoietic cells. Ann N Y Acad Sci 2005;1044: Chadwick K, Wang L, Li L et al. Cytokines and BMP-4 promote hematopoietic differentiation of human embryonic stem cells. Blood 2003;102: Gaur M, Kamata T, Wang S et al. Megakaryocytes derived from human embryonic stem cells: A genetically tractable system to study megakaryocytopoiesis and integrin function. J Thromb Haemost 2006;4: Methia N, Louache F, Vainchenker W et al. Oligodeoxynucleotides antisense to the proto-oncogene c-mpl specifically inhibit in vitro megakaryocytopoiesis. Blood 1993;82: Ku H, Yonemura Y, Kaushansky K et al. Thrombopoietin, the ligand for the Mpl receptor, synergizes with steel factor and other early acting cytokines in supporting proliferation of primitive hematopoietic progenitors of mice. Blood 1996;87: Ballmaier M, Germeshausen M, Schulze H et al. c-mpl mutations are the cause of congenital amegakaryocytic thrombocytopenia. Blood 2001;97: Ballmaier M, Germeshausen M, Krukemeier S et al. Thrombopoietin is essential for the maintenance of normal hematopoiesis in humans: Development of aplastic anemia in patients with congenital amegakaryocytic thrombocytopenia. Ann N Y Acad Sci 2003;996: Vodyanik MA, Bork JA, Thomson JA et al. Human embryonic stem cell-derived CD34 cells: Efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood 2005;105: Borge OJ, Ramsfjell V, Cui L et al. Ability of early acting cytokines to directly promote survival and suppress apoptosis of human primitive CD34. Blood 1997;90: Watowich SS, Wu H, Socolovsky M et al. Cytokine receptor signal transduction and the control of hematopoietic cell development. Annu Rev Cell Dev Biol 1996;12:
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT
 168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Physiologic Role of TPO in Thrombopoiesis
 Physiologic Role of TPO in Thrombopoiesis Hiroshi Miyazaki Pharmaceutical Research Laboratory, Kirin Brewery Co., Ltd., Gunma, Japan Key Words. Thrombopoietin Megakaryocyte Megakaryocyte progenitor cell
Physiologic Role of TPO in Thrombopoiesis Hiroshi Miyazaki Pharmaceutical Research Laboratory, Kirin Brewery Co., Ltd., Gunma, Japan Key Words. Thrombopoietin Megakaryocyte Megakaryocyte progenitor cell
Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia
 Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia YOSHINOBU ASANO, SHOICHIRO SHIBATA, SHINJI KOBAYASHI, SEIICHI OKAMURA, YOSHIYUKI NIHO First Department
Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia YOSHINOBU ASANO, SHOICHIRO SHIBATA, SHINJI KOBAYASHI, SEIICHI OKAMURA, YOSHIYUKI NIHO First Department
Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells
 Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells Christopher Sturgeon February 14, 2017 Pluripotent Stem Cells self-renewal hpsc Mesoderm blood cardiovascular muscle Endoderm lung liver
Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells Christopher Sturgeon February 14, 2017 Pluripotent Stem Cells self-renewal hpsc Mesoderm blood cardiovascular muscle Endoderm lung liver
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Haematopoietic stem cells
 Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5
 sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3
 EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
Supplement Material. Spleen weight (mg) LN cells (X106) Acat1-/- Acat1-/- Mouse weight (g)
 Supplement Material A Spleen weight (mg) C Mouse weight (g) 1 5 1 2 9 6 3 2 5 2 1 5 Male LN cells (X16) 4 ** ** Female B 3 2 1 Supplemental Figure I. Spleen weight (A), Inguinal lymph node (LN) cell number
Supplement Material A Spleen weight (mg) C Mouse weight (g) 1 5 1 2 9 6 3 2 5 2 1 5 Male LN cells (X16) 4 ** ** Female B 3 2 1 Supplemental Figure I. Spleen weight (A), Inguinal lymph node (LN) cell number
BMP4/Smad5 dependent stress erythropoiesis is required for the expansion of erythroid progenitors during fetal development
 Available online at www.sciencedirect.com Developmental Biology 317 (2008) 24 35 www.elsevier.com/developmentalbiology BMP4/Smad5 dependent stress erythropoiesis is required for the expansion of erythroid
Available online at www.sciencedirect.com Developmental Biology 317 (2008) 24 35 www.elsevier.com/developmentalbiology BMP4/Smad5 dependent stress erythropoiesis is required for the expansion of erythroid
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Hematopoiesis. BHS Liège 27/1/2012. Dr Sonet Anne UCL Mont-Godinne
 Hematopoiesis BHS Liège 27/1/2012 Dr Sonet Anne UCL Mont-Godinne Hematopoiesis: definition = all the phenomenons to produce blood cells Leukocytes = White Blood Cells Polynuclear = Granulocytes Platelet
Hematopoiesis BHS Liège 27/1/2012 Dr Sonet Anne UCL Mont-Godinne Hematopoiesis: definition = all the phenomenons to produce blood cells Leukocytes = White Blood Cells Polynuclear = Granulocytes Platelet
Chemotherapeutic Susceptibility of Human Bone Marrow Progenitor Cells and Human Myelogenous Leukemia Cells (HMO) in Co-Culture: Preliminary Report
 International Journal of Cell Cloning 2: 254-262 (1984) Chemotherapeutic Susceptibility of Human Bone Marrow Progenitor Cells and Human Myelogenous Leukemia Cells (HMO) in Co-Culture: Preliminary Report
International Journal of Cell Cloning 2: 254-262 (1984) Chemotherapeutic Susceptibility of Human Bone Marrow Progenitor Cells and Human Myelogenous Leukemia Cells (HMO) in Co-Culture: Preliminary Report
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Automated and Standardized Counting of Mouse Bone Marrow CFU Assays
 Automated and Standardized Counting of Mouse Bone Marrow CFU Assays 2 Automated and Standardized Colony Counting Table of Contents 4 Colony-Forming Unit (CFU) Assays for Mouse Bone Marrow 5 Automated Assay
Automated and Standardized Counting of Mouse Bone Marrow CFU Assays 2 Automated and Standardized Colony Counting Table of Contents 4 Colony-Forming Unit (CFU) Assays for Mouse Bone Marrow 5 Automated Assay
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
Nature Immunology: doi: /ni.3412
 Supplementary Figure 1 Gata1 expression in heamatopoietic stem and progenitor populations. (a) Unsupervised clustering according to 100 top variable genes across single pre-gm cells. The two main cell
Supplementary Figure 1 Gata1 expression in heamatopoietic stem and progenitor populations. (a) Unsupervised clustering according to 100 top variable genes across single pre-gm cells. The two main cell
Feasibility of hyperthermia as a purging modality in autologous bone marrow transplantation Wierenga, Pieter Klaas
 University of Groningen Feasibility of hyperthermia as a purging modality in autologous bone marrow transplantation Wierenga, Pieter Klaas IMPORTANT NOTE: You are advised to consult the publisher's version
University of Groningen Feasibility of hyperthermia as a purging modality in autologous bone marrow transplantation Wierenga, Pieter Klaas IMPORTANT NOTE: You are advised to consult the publisher's version
Hematopoietic Growth Factors Colony Stimulating Factors. Erythropoietin (Epoetin alfa). Granulocyte-macrophage colonystimulating factor (G-CSF).
 Hematopoietic Growth Factors Colony Stimulating Factors. Erythropoietin (Epoetin alfa). Granulocyte colony-stimulating factor(g-csf). Granulocyte-macrophage colonystimulating factor (G-CSF). Interleukin-11
Hematopoietic Growth Factors Colony Stimulating Factors. Erythropoietin (Epoetin alfa). Granulocyte colony-stimulating factor(g-csf). Granulocyte-macrophage colonystimulating factor (G-CSF). Interleukin-11
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Introduction Kit Components Cat. # # of vials Reagent Quantity Storage
 Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
The Role of Recombinant Interleukin 11 in Megakaryocytopoiesis
 The Role of Recombinant Interleukin 11 in Megakaryocytopoiesis K.J. Turner, S. Neben, N. Weich, R.G. Schaub, S.J. Goldrnan Departments of Immunology and Hematopoiesis and Preclinical Biology, Genetics
The Role of Recombinant Interleukin 11 in Megakaryocytopoiesis K.J. Turner, S. Neben, N. Weich, R.G. Schaub, S.J. Goldrnan Departments of Immunology and Hematopoiesis and Preclinical Biology, Genetics
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells
 Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Schumacher et al. Journal of Hematology & Oncology (2015) 8:64 DOI /s
 Schumacher et al. Journal of Hematology & Oncology (2015) 8:64 DOI 10.1186/s13045-015-0152-2 JOURNAL OF HEMATOLOGY & ONCOLOGY RESEARCH Angptl4 is upregulated under inflammatory conditions in the bone marrow
Schumacher et al. Journal of Hematology & Oncology (2015) 8:64 DOI 10.1186/s13045-015-0152-2 JOURNAL OF HEMATOLOGY & ONCOLOGY RESEARCH Angptl4 is upregulated under inflammatory conditions in the bone marrow
Molecular Hematopathology Leukemias I. January 14, 2005
 Molecular Hematopathology Leukemias I January 14, 2005 Chronic Myelogenous Leukemia Diagnosis requires presence of Philadelphia chromosome t(9;22)(q34;q11) translocation BCR-ABL is the result BCR on chr
Molecular Hematopathology Leukemias I January 14, 2005 Chronic Myelogenous Leukemia Diagnosis requires presence of Philadelphia chromosome t(9;22)(q34;q11) translocation BCR-ABL is the result BCR on chr
MPL W515L K mutation
 MPL W515L K mutation BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
MPL W515L K mutation BCR-ABL genotyping The exact chromosomal defect in Philadelphia chromosome is a translocation. Parts of two chromosomes, 9 and 22, switch places. The result is a fusion gene, created
MATERIALS AND METHODS. Cells and cell separation INTRODUCTION. Li Lu,* Zhi-Hua Li,* Jie He,* Yue Ge,* Susan Rice, and Hal E.
 Transduction of recombinant human erythropoietin receptor cdna into daughter progenitors derived from single CD34 3 cord blood cells changes the differentiation profile of daughter progenitors Li Lu,*
Transduction of recombinant human erythropoietin receptor cdna into daughter progenitors derived from single CD34 3 cord blood cells changes the differentiation profile of daughter progenitors Li Lu,*
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Stem cells: units of development and regeneration. Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research.
 Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
Erythropoietin In Vitro
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Rapid Decline of Clonogenic Hemopoietic Progenitors in Semisolid Cultures of Bone Marrow Samples Derived from Patients with Chronic Myeloid Leukemia
 Original Manuscript International Journal of Cell Cloning 7314-321 (1989) Rapid Decline of Clonogenic Hemopoietic Progenitors in Semisolid Cultures of Bone Marrow Samples Derived from Patients with Chronic
Original Manuscript International Journal of Cell Cloning 7314-321 (1989) Rapid Decline of Clonogenic Hemopoietic Progenitors in Semisolid Cultures of Bone Marrow Samples Derived from Patients with Chronic
In Vitro Growth of Erythropoietic Progenitor Cells in Long-%rm Remission of Acute Leukemia
 International Journal of Cell Cloning 4: 186-191 (1986) In Vitro Growth of Erythropoietic Progenitor Cells in Long-%rm Remission of Acute Leukemia C. Peschel, G. Konwalinka, D. Geissler, H. Bmunsteiner
International Journal of Cell Cloning 4: 186-191 (1986) In Vitro Growth of Erythropoietic Progenitor Cells in Long-%rm Remission of Acute Leukemia C. Peschel, G. Konwalinka, D. Geissler, H. Bmunsteiner
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
PU.1 regulates both cytokine-dependent proliferation and differentiation of granulocyte/ macrophage progenitors
 The EMBO Journal Vol.17 No.15 pp.4456 4468, 1998 PU.1 regulates both cytokine-dependent proliferation and differentiation of granulocyte/ macrophage progenitors Rodney P.DeKoter 1,2, Jonathan C.Walsh 3
The EMBO Journal Vol.17 No.15 pp.4456 4468, 1998 PU.1 regulates both cytokine-dependent proliferation and differentiation of granulocyte/ macrophage progenitors Rodney P.DeKoter 1,2, Jonathan C.Walsh 3
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11095 Supplementary Table 1. Summary of the binding between Angptls and various Igdomain containing receptors as determined by flow cytometry analysis. The results were summarized from
doi:10.1038/nature11095 Supplementary Table 1. Summary of the binding between Angptls and various Igdomain containing receptors as determined by flow cytometry analysis. The results were summarized from
Accelerate Your Research with Conversant Bio
 Accelerate Your Research with Conversant Bio 400+ Participating MDs 50+ Partner sites for tissue procurement Continuous expansion of sourcing capabilities Closely monitored chain of custody Full regulatory
Accelerate Your Research with Conversant Bio 400+ Participating MDs 50+ Partner sites for tissue procurement Continuous expansion of sourcing capabilities Closely monitored chain of custody Full regulatory
BCR-ABL - LSK BCR-ABL + LKS - (%)
 Marker Clone BCR-ABL + LSK (%) BCR-ABL + LKS - (%) BCR-ABL - LSK (%) P value vs. BCR-ABL + LKS - P value vs. BCR-ABL - LSK CD2 RM2-5 12.9 ± 3.6 36.7 ± 6.5 19.3 ± 2.4 0.01 0.10 CD5 53-7.3 13.9 ± 3.2 20.8
Marker Clone BCR-ABL + LSK (%) BCR-ABL + LKS - (%) BCR-ABL - LSK (%) P value vs. BCR-ABL + LKS - P value vs. BCR-ABL - LSK CD2 RM2-5 12.9 ± 3.6 36.7 ± 6.5 19.3 ± 2.4 0.01 0.10 CD5 53-7.3 13.9 ± 3.2 20.8
Supporting Information
 Supporting Information Identification of Novel ROS Inducers: Quinone Derivatives Tethered to Long Hydrocarbon Chains Yeonsun Hong,, Sandip Sengupta,, Wooyoung Hur, *, Taebo Sim,, * KU-KIST Graduate School
Supporting Information Identification of Novel ROS Inducers: Quinone Derivatives Tethered to Long Hydrocarbon Chains Yeonsun Hong,, Sandip Sengupta,, Wooyoung Hur, *, Taebo Sim,, * KU-KIST Graduate School
Nature Genetics: doi: /ng Supplementary Figure 1. HOX fusions enhance self-renewal capacity.
 Supplementary Figure 1 HOX fusions enhance self-renewal capacity. Mouse bone marrow was transduced with a retrovirus carrying one of three HOX fusion genes or the empty mcherry reporter construct as described
Supplementary Figure 1 HOX fusions enhance self-renewal capacity. Mouse bone marrow was transduced with a retrovirus carrying one of three HOX fusion genes or the empty mcherry reporter construct as described
Nature Genetics: doi: /ng Supplementary Figure 1
 Supplementary Figure 1 MSI2 interactors are associated with the riboproteome and are functionally relevant. (a) Coomassie blue staining of FLAG-MSI2 immunoprecipitated complexes. (b) GO analysis of MSI2-interacting
Supplementary Figure 1 MSI2 interactors are associated with the riboproteome and are functionally relevant. (a) Coomassie blue staining of FLAG-MSI2 immunoprecipitated complexes. (b) GO analysis of MSI2-interacting
Product Manual. Omni-Array Sense Strand mrna Amplification Kit, 2 ng to 100 ng Version Catalog No.: Reactions
 Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Done By : WESSEN ADNAN BUTHAINAH AL-MASAEED
 Done By : WESSEN ADNAN BUTHAINAH AL-MASAEED Acute Myeloid Leukemia Firstly we ll start with this introduction then enter the title of the lecture, so be ready and let s begin by the name of Allah : We
Done By : WESSEN ADNAN BUTHAINAH AL-MASAEED Acute Myeloid Leukemia Firstly we ll start with this introduction then enter the title of the lecture, so be ready and let s begin by the name of Allah : We
Thawing MEFs (Mouse Embryonic Fibroblasts (MEFs)
 1 FEEDER CULTURES The function of feeder cultures is to support the undifferentiated growth of hpsc. Typically primary fibroblasts are used for this purpose. We prepare our mouse feeder cells from ICR
1 FEEDER CULTURES The function of feeder cultures is to support the undifferentiated growth of hpsc. Typically primary fibroblasts are used for this purpose. We prepare our mouse feeder cells from ICR
Hematology 101. Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD
 Hematology 101 Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD Hematocrits Plasma White cells Red cells Normal, Hemorrhage, IDA, Leukemia,
Hematology 101 Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD Hematocrits Plasma White cells Red cells Normal, Hemorrhage, IDA, Leukemia,
Blood & Blood Formation
 Module IB Blood & Blood Formation Histology and Embryology Martin Špaček, MD (m.spacek@centrum.cz) http://www.lf3.cuni.cz/histologie Approximately 7% of a person's weight is blood (about 5 L) Blood consists
Module IB Blood & Blood Formation Histology and Embryology Martin Špaček, MD (m.spacek@centrum.cz) http://www.lf3.cuni.cz/histologie Approximately 7% of a person's weight is blood (about 5 L) Blood consists
Annals of Oncology Advance Access published January 10, 2005
 Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Nature Medicine doi: /nm.3150
 Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Effects of Mast Cell Growth Factor on Ara-C Mediated Acute Myeloid Leukemia Cell Killing
 Effects of Mast Cell Growth Factor on Ara-C Mediated Acute Myeloid Leukemia Cell Killing A. Tafuri, L. De Felice, M. G. Mascolo, T. Valentini, M. T. Petrucci, M. C. Petti University La Sapienza of Rome,
Effects of Mast Cell Growth Factor on Ara-C Mediated Acute Myeloid Leukemia Cell Killing A. Tafuri, L. De Felice, M. G. Mascolo, T. Valentini, M. T. Petrucci, M. C. Petti University La Sapienza of Rome,
International Journal of Cell Cloning (1991)
 Concise Review International Journal of Cell Cloning 9542-547 (1991) Hybrid Cytokines as Hematopoietic Growth Factors Douglas E. Williamsa, Linda S. Park", Hal E. Broxmeyerb, Li Lub 'Immunex Research and
Concise Review International Journal of Cell Cloning 9542-547 (1991) Hybrid Cytokines as Hematopoietic Growth Factors Douglas E. Williamsa, Linda S. Park", Hal E. Broxmeyerb, Li Lub 'Immunex Research and
Lhx2 Expression Promotes Self-Renewal of a Distinct Multipotential Hematopoietic Progenitor Cell in Embryonic Stem Cell-Derived Embryoid Bodies
 Lhx2 Expression Promotes Self-Renewal of a Distinct Multipotential Hematopoietic Progenitor Cell in Embryonic Stem Cell-Derived Embryoid Bodies Lina Dahl, Karin Richter, Anna-Carin Hägglund, Leif Carlsson*
Lhx2 Expression Promotes Self-Renewal of a Distinct Multipotential Hematopoietic Progenitor Cell in Embryonic Stem Cell-Derived Embryoid Bodies Lina Dahl, Karin Richter, Anna-Carin Hägglund, Leif Carlsson*
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Plasmids Western blot analysis and immunostaining Flow Cytometry Cell surface biotinylation RNA isolation and cdna synthesis
 Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis
 212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Generation and validation of mtef4-knockout mice.
 Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Advances in Computer Science Research, volume 59 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016)
 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
The Ufm1-activating enzyme Uba5 is indispensable for erythroid differentiation in mice
 Supplementary information The Ufm1-activating enzyme Uba5 is indispensable for erythroid differentiation in mice Kanako Tatsumi 1, 2, Harumi Yamamoto-Mukai 2, Ritsuko Shimizu 3, Satoshi Waguri 4, Yu-Shin
Supplementary information The Ufm1-activating enzyme Uba5 is indispensable for erythroid differentiation in mice Kanako Tatsumi 1, 2, Harumi Yamamoto-Mukai 2, Ritsuko Shimizu 3, Satoshi Waguri 4, Yu-Shin
Human Cathepsin D ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver
 Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver YUKHI AIBA, MAKIO OGAWA Ralph H. Johnson Department of Veterans Affairs Medical Center, Department of Medicine,
Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver YUKHI AIBA, MAKIO OGAWA Ralph H. Johnson Department of Veterans Affairs Medical Center, Department of Medicine,
Corning BioCoat Matrigel Invasion Chamber
 Corning BioCoat Matrigel Invasion Chamber Catalog No. 354480, 354481 Guidelines for Use Discovery Labware, Inc., Two Oak Park, Bedford, MA 01730, Tel: 1.978.442.2200 (U.S.) CLSTechServ@Corning.com www.corning.com/lifesciences
Corning BioCoat Matrigel Invasion Chamber Catalog No. 354480, 354481 Guidelines for Use Discovery Labware, Inc., Two Oak Park, Bedford, MA 01730, Tel: 1.978.442.2200 (U.S.) CLSTechServ@Corning.com www.corning.com/lifesciences
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features
 Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells
 CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
human Total Cathepsin B Catalog Number: DY2176
 human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
Key Words. AC133 antigen CD34 antigen Cord blood Peripheral blood 2003;21:
 Stem Cells Original Article Differences Between Peripheral Blood and Cord Blood in the Kinetics of Lineage-Restricted Hematopoietic Cells: Implications for Delayed Platelet Recovery Following Cord Blood
Stem Cells Original Article Differences Between Peripheral Blood and Cord Blood in the Kinetics of Lineage-Restricted Hematopoietic Cells: Implications for Delayed Platelet Recovery Following Cord Blood
Differentiation Ability of Peripheral Blood Cells from Patients with Acute Leukemia or Blast Crisis in Chronic Myelocytic Leukemia"
 Differentiation Ability of Peripheral Blood Cells from Patients with Acute Leukemia or Blast Crisis in Chronic Myelocytic Leukemia" Hoelzer, D.,l, Harriss, E. B.l, Kurrle, E.l, Schmücker, H.l, Hellriegel,
Differentiation Ability of Peripheral Blood Cells from Patients with Acute Leukemia or Blast Crisis in Chronic Myelocytic Leukemia" Hoelzer, D.,l, Harriss, E. B.l, Kurrle, E.l, Schmücker, H.l, Hellriegel,
Essential Medium, containing 10% fetal bovine serum, 100 U/ml penicillin and 100 µg/ml streptomycin. Huvec were cultured in
 Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Stem cells and Cancer. John Glod. December 2, 2009
 Stem cells and Cancer John Glod Lehigh University Lehigh University December 2, 2009 The Tumor Microenvironment Littlepage et al Cancer Cell 2005 Cancer Stem Cells A small group of cells within the larger
Stem cells and Cancer John Glod Lehigh University Lehigh University December 2, 2009 The Tumor Microenvironment Littlepage et al Cancer Cell 2005 Cancer Stem Cells A small group of cells within the larger
Department of Pharmaceutical Sciences, School of Pharmacy, Northeastern University, Boston, MA 02115, USA 2
 Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
(PDGF), 9 ( -2 (FGF-2), SMO
 Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
Low Cell Binding Property of LIPIDURE -COAT
 Technical Note_1 ver.1 Low Cell Binding Property of LIPIDURE -COAT 1. LIPIDURE -COAT MULTI DISH A-6MD (Cat. No. 51011617) 2. Cell; NIH 3T3 (Fibroblast, mouse) 1. 10 %CS-DMEM; DMEM (Dulbecco's Modified
Technical Note_1 ver.1 Low Cell Binding Property of LIPIDURE -COAT 1. LIPIDURE -COAT MULTI DISH A-6MD (Cat. No. 51011617) 2. Cell; NIH 3T3 (Fibroblast, mouse) 1. 10 %CS-DMEM; DMEM (Dulbecco's Modified
Genetics and Cancer Ch 20
 Genetics and Cancer Ch 20 Cancer is genetic Hereditary cancers Predisposition genes Ex. some forms of colon cancer Sporadic cancers ~90% of cancers Descendants of cancerous cells all cancerous (clonal)
Genetics and Cancer Ch 20 Cancer is genetic Hereditary cancers Predisposition genes Ex. some forms of colon cancer Sporadic cancers ~90% of cancers Descendants of cancerous cells all cancerous (clonal)
SUPPLEMENTARY INFORMATION GENOTOXICITY. In vitro Genotoxicity Studies
 SUPPLEMENTARY INFORMATION GENOTOXICITY In vitro Genotoxicity Studies The in vitro immortalisation (IVIM) assay relies on the induction of a survival advantage by insertional activation of cellular proto-oncogenes,
SUPPLEMENTARY INFORMATION GENOTOXICITY In vitro Genotoxicity Studies The in vitro immortalisation (IVIM) assay relies on the induction of a survival advantage by insertional activation of cellular proto-oncogenes,
Luminescent platforms for monitoring changes in the solubility of amylin and huntingtin in living cells
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
PNGase F Instruction Manual
 PNGase F Instruction Manual Catalog Number 170-6883 Bio-Rad Laboratories, 2000 Alfred Nobel Dr., Hercules, CA 94547 4006094 Rev A Table of Contents Section 1 Introduction...1 Section 2 Kit Components and
PNGase F Instruction Manual Catalog Number 170-6883 Bio-Rad Laboratories, 2000 Alfred Nobel Dr., Hercules, CA 94547 4006094 Rev A Table of Contents Section 1 Introduction...1 Section 2 Kit Components and
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplemental Information
 Electronic Supplementary Material (ESI) for Food & Function. This journal is The Royal Society of Chemistry 2016 Supplemental Information Supplementary Materials and Methods Materials Assay kits of total
Electronic Supplementary Material (ESI) for Food & Function. This journal is The Royal Society of Chemistry 2016 Supplemental Information Supplementary Materials and Methods Materials Assay kits of total
