Specific Aims Aim 1: Determine whether inhibition of RCE1 will decrease cell growth and viability of PDAC in vivo and in vitro
|
|
|
- Emery Powell
- 5 years ago
- Views:
Transcription
1 Specific Aims Pancreatic ductal adenocarcinoma (PDAC) is a highly aggressive and most common form of pancreatic cancer. Patients with pancreatic cancer have a poor prognosis with a one-year survival of less than 20%. Activating K-Ras mutations occur in over 85% of PDAC cases making it an attractive target for therapy. Inhibitors of the Ras pathway have been FDA approved with the MEK inhibitor, cobimetinib, and PI3K inhibitor, idelalisib, in combination with other therapies being used for the treatment of melanoma and chronic lymphocytic leukemia, respectively. One novel mechanism that may be used to target Ras is the disruption of the plasma membrane localization that is necessary for Ras function. In order for plasma membrane localization to occur, a farnesyl moiety must be added to the cysteine residue of the terminal CAAX motif of Ras, the last three amino acid of the CAAX motif must then be proteolytically cleaved by Ras converting enzyme 1 (RCE1). Finally, isoprenylcysteine carboxyl methyltransferase (ICMT) methylates the C-terminal cysteine to facilitate transport to the plasma membrane. Attempts to target this process with farnesyltransferase inhibitors (FTIs) have failed due to alternate modifications of the CAAX motif in the presence of FTIs and available ICMT inhibitors have limited physical properties for clinical development. However, three RCE1 inhibitors recently derived from previously reported RCE1 inhibitor, NSC1011, have shown promise in disruption of Ras localization in yeast and colon carcinoma cell line, HCT-116. Because of these findings, I hypothesize that RCE1 inhibition will be effective in preventing oncogenic Ras signaling and therefore an effective therapy for pancreatic ductal adenocarcinoma with activating K-Ras mutations. To test this hypothesis I propose the following specific aims: Aim 1: Determine whether inhibition of RCE1 will decrease cell growth and viability of PDAC in vivo and in vitro. I hypothesize that inhibition of RCE1 in pancreatic ductal adenocarcinoma will reduce Ras localization to the plasma membrane and Ras-dependent downstream phosphorylation, which will result in decreased pancreatic adenocarcinoma cell viability, migration and growth. I will treat PDAC cell lines, Panc and Panc 04.03, with the RCE1 inhibitors and determine effects to Ras membrane translocation by immunofluorescent microscopy and cell fractionation. I will also study the efficacy of RCE1 inhibition by observing the phosphorylation of the pathway downstream of Ras such as the phosphorylation of MEK, ERK, PI3K and Akt. I will also measure cell viability and growth by trypan blue assay, migration by scratch test and invasion by matrix invasion assays after RCE1 inhibitor treatment. In addition, as a genetic control I will use RCE1 sirna knockdown to confirm that these findings are specific to RCE1 and not an artifact of the inhibitor. Lastly, I will use these cells in a xenograft mouse model to determine if tumor growth and metastasis is decreased by RCE1 inhibition in vivo. Aim 2: Determine the synergistic effects of targeting RCE1 in combination with inhibitors of downstream effectors in PDAC. Since compensatory mechanisms have been a problem for the efficacy of inhibitors that alter Ras membrane localization, I hypothesize that RCE1 inhibition will have synergistic effects in combination with downstream inhibitors, such as MEK, Akt or PI3K inhibitors, in pancreatic ductal adenocarcinoma. In the PDAC cell lines, I will determine synergistic concentrations of the RCE1 inhibitors in combination with MEK, Akt or PI3K inhibitors as determined by viability and cell growth measured by trypan blue assay. I will then treat PDAC cells with the synergistic concentrations of RCE1 inhibitor and MEK, Akt or PI3K inhibitor and determine the effects on migration and invasion by scratch and matrix invasion assays, respectively. Lastly, I will use xenograft mouse models to determine if the inhibition of RCE1 in combination with an inhibitor of a downstream effector will decrease tumor growth and metastasis in vivo. Since Ras mutations are found in ~25% of human tumors, this mechanism may also be applied to many other cancers types, such as prostate and non-small cell lung cancer. Additionally these studies will provide insight into the role Ras membrane localization plays in malignancies.
2 Significance and Background Pancreatic ductal adenocarcinoma (PDAC) survival statistics illustrate the need for new therapies. With a five year survival of about 8%, pancreatic cancer is the fourth leading cause of cancer-related death in the USA (1). Currently, surgical resection is considered the only treatment to cure pancreatic cancer (2). Unfortunately, since pancreatic cancer can progress with few symptoms, most patients are at an advanced stage at diagnosis and resection is only an option for about 10% of cases (3). Chemotherapy and radiotherapy have only been of marginal benefit and only moderately improved patient survival (4). The standard of care for advanced pancreatic cancer since 1997, gemcitabine, was only able to improve the oneyear survival rate to 18% and extended the median survival duration to 5.65 months (5). Pancreatic ductal adenocarcinoma (PDAC) is the most common type of pancreatic cancer, constituting at least 85% of pancreatic cancers (6). PDAC originates from precursor legions, the most common of which is pancreatic Figure 1: Pancreatic ductal adenocarcinoma (PDAC) arises from pancreatic intraepithelial neoplasia. The pancreatic duct cell, the cell of origin for PDAC, gives rise to pancreatic intraepithelial neoplasia (PanIN) which accumulates genetic mutations as it progresses to invasive carcinoma. Adapted from Hruban RH, Et. Al. (11). intraepithelial neoplasia (PanIN), which arise from ductal epithelial cells (Figure 1) (7). These PanINs are thought to progress from moderate to high-grade dysplasia, and finally to invasive carcinoma (8). Oncogene dependence in PDAC is a rationale for the use of Ras regulation for treatment. Though PDAC is a heterogeneous disease with a mean of somatic mutations in 12 key pathways, K-Ras mutations are found in over 90% of PDAC patients (9, 10). Activating mutations in K-Ras occur in pancreatic duct lesions and the prevalence of these mutations increase with increasing atypia of the duct lesions, it can therefore be inferred that this is an early genetic event that is key for the progression from neoplasia to invasive carcinoma (11). Since there have been many challenges in inhibiting mutated K-Ras, as it has a unique constitutively active conformation, studies have focused on inhibition of the pathway downstream of Ras, such as Akt, mitogen-activated protein kinase (MAPK) and extracellular signal-regulated kinase (ERK) (12). A phase 2 clinical trial of the MAPK/ERK kinase (MEK) inhibitor, selumetinib, in combination with the Food and Drug Administration (FDA) approved epidermal growth factor receptor (EGFR) inhibitor, erlotinib, in previously treated advanced stage PDAC had promising results with a median progression-free survival of 1.9 months and a median overall survival of 7.3 months (13). However, a phase 2 clinical trial to study the effects of selumetinib in combination with the Akt inhibitor, MK-2206, showed no improvement in overall survival (14). Taken together, these clinical studies demonstrated that the inhibition of this pathway is pharmacologically and physiologically possible, however further research is needed to optimize the effective use of these inhibitors in the treatment of pancreatic ductal adenocarcinoma. Inhibition of the transport to the cell membrane impedes Ras activity. In addition to inhibiting downstream targets, the transport and localization of Ras to the membrane as a possible target for inhibition has been investigated (15). Ras must associate with the inner plasma membrane to
3 function, therefore interfering with the transport of Ras to the plasma membrane can suppress Ras activity even if it s constitutively active (16). Ras localization to the cell membrane is a multi-step process (Figure 2). First, Ras undergoes prenylation by farnesyltransferase (FTASE) (Figure 2, A) and subsequently has its terminal AAX motif proteolytically cleaved by Ras converting enzyme 1 (RCE1) (Figure 2, B) before methylation by isoprenylcysteine carboxyl methyltransferase (ICMT) can occur (Figure 2, C) (17). Ras can then be transferred to the recycling endosome or Golgi to be shuttled directly by vesicular transport to the plasma membrane (18). Initial attempt to inhibit this process focused on targeting farnesyltransferase but it was discovered that in the presence of farnesyltransferase inhibitors, Ras is prenylated by geranylgeranyl transferase (GGTASE) (Figure 1, D)(19). RCE1 inhibition can be used to regulate Ras. Blockade of RCE1 can cause mis-localization of Ras and reduce oncogenic transformation of cells (20). A recent study has derived novel RCE1 inhibitors from NSC1011, an 8-Hydroxyquioline-based inhibitor of RCE1 (21). Three of Figure 2: Transport and localization of Ras to the cell membrane requires RCE1, ICMT and farnesylation or geranylgeranylation. Transport to the endoplasmic reticulum (ER) is essential for the removal of the AAX motif, so ICMT can methylate the C-terminal cysteine and facilitate membrane association. Adapted from Papke B and Der CJ (18). which, NSC1011-2, NSC and NSC (Figure 3), show promise in the selective and effective disruption of Ras localization and activity as seen in yeast and colon carcinoma cell line, HCT-116 (21). These compounds were able to inhibit 50% of CAAX cleavage, as measured by a fluorescence-based proteolysis assay in yeast, at concentrations of about 10 μm and did not induce more than a 20% loss in viability of mammalian cells at concentrations of 25 μm after 20 hours, as measured by CellTiter-Blue assay (21). Additionally, these compounds were able to mis-localize Ras as effectively as knockdown of RCE1 as measured by decrease in plasma membrane association to 20-30% as determined by fluorescent microscopy (21). Lastly, these inhibitors showed specificity for RCE1 over functionally similar Ste24 protease, which mediates zinc-dependent CAAX proteolysis in the maturation of lamin A (22), as demonstrated by the decrease in activity of Ste24 by less than 25% and the decrease in activity of RCE1 by about 60% after 10μM treatment with the inhibitors (21). Figure 3: Three novel RCE1 inhibitors recently derived from NSC1011 show promise for the treatment of PDAC. Novel RCE1 inhibitors, NSC1011-2, NSC and NSC , are able to induced 50% inhibition of RCE1 at concentrations less than 10 μm. Adapted from Mohammed I, Et Al. (21) Understanding the effects of RCE1 inhibition may improve health outcomes for PDAC patients and will provide insight on how to better treat PDAC as well as other diseases with K-Ras mutations. Research Plan Aim 1: Determine whether inhibition of RCE1 will decrease cell growth and viability of PDAC in vivo and in vitro. Hypothesis/Rationale: Recently the RCE1 inhibitors derived from the RCE1 inhibitor, NSC1011, have been shown to have specific and effective inhibition of RCE1 in yeast and the HCT-116 colon carcinoma cell line (21). It has been previously published that loss of RCE1 prevents the localization of Ras to the cell membrane and reduces oncogenic transformation of cells (20). I hypothesize the inhibition of RCE1 in pancreatic ductal
4 adenocarcinoma will reduce Ras localization to the plasma membrane and Ras-dependent downstream phosphorylation, which will result in decreased pancreatic adenocarcinoma cell viability, migration, invasion and growth. Approach: Aim 1.1: Determine molecular effects of RCE1 inhibition on PDAC. To determine the molecular and cellular effects of RCE1 inhibition on PDAC cells, the Panc and Panc cell lines will be utilized. Both cell lines are commercially available, have been verified to possess K-Ras oncogenes, form tumors in nude or severe combined immunodeficiency (SCID) mice and were harvested in 1995 from the head-of-the-pancreas of pancreatic adenocarcinoma patients, specifically a 64 year old white female and 70 year old white male, respectively. First, to determine if the RCE1 inhibitors can prevent Ras membrane localization in PDAC cells, I will perform fluorescent microscopy and subcellular fractionation experiments. For the fluorescent microscopy, cells from both cell lines will be seeded at 60% confluency on coverslips, then treated with the RCE1 inhibitors at the concentration determined to be the IC50 for RCE1 activity determined previously (21) or vehicle control for 24 hours. RCE1 and control sirna knockdown will be used a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. The cells on the coverslips will be blocked then probed with K-Ras antibody, DAPI nuclear stain and CellBrite plasma membrane stain. The signal will be visualized using fluorescent microscopy and analyzed for co-localization of the Ras antibody to the membrane using Ziess confocal microscope and ZEN imaging software. For the subcellular fractionation, the ultracentrifugation protocol available through Abcam will be performed on the lysates of 10 million cells from both cell line treated with RCE1 inhibitors at the concentration determined to be the IC50 for RCE1 activity determined previously (21) or vehicle control for 24 hours to separate the nuclear cytoplasmic, mitochondrial and membrane fractions. RCE1 and control sirna knockdown will be used a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. Fractions will be run by SDS-PAGE then transferred to a PVDF membrane. The membrane will be blocked then probed with Ras, GAPDH, cytochrome c, laminin A/C and Na/K ATPase to determine the separation of the cytosolic, mitochondria, nuclear and membrane faction respectively. The signal will be visualized using enhanced chemiluminescence (ECL) reagents then quantified by densitometry using ImageJ software. To determine if the RCE1 inhibitors can prevent Ras-dependent phosphorylation of downstream signaling pathways, I will utilize immunoblotting techniques to analyze phosphorylated phosphatidylinositol-3-kinase (PI3K), Akt, MEK and ERK. I will treat 5 million of each cells treated with RCE1 inhibitors at the IC50 concentration for RCE1 activity determined previously (21) or vehicle control for 24 hours. RCE1 and control sirna knockdown will be used a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. The cells will then be lysed and total protein concentrations will be determined by Bradford assay. Equal total protein amounts will be run by SDS-PAGE and transferred to a PVDF membrane. The membrane will be blocked and probed for phosphorylated and total PI3K, Akt, MEK and ERK, as well as actin to control for loading. The signal will be observed using ECL reagents then quantified by densitometry using ImageJ software. All results will be analyzed for statistical significance using GraphPad Prism software. Aim 1.2: Determine cellular effects of RCE1 inhibition on PDAC. To determine the cellular effects of RCE1 inhibition, I will perform cell viability, cell growth, cell migration and cell invasion assays, as well as in vivo studies in mouse xenograft models to determine the effects on tumor burden, metastasis and survival. First, to determine how RCE1 inhibition affects viability and growth in PDAC cells, I will conduct trypan blue exclusion tests and cell count assays. I will treat 500,000 cells from each cell line with the RCE1 inhibitors at the concentration determined to be the IC50 for RCE1 activity determined previously (21) or vehicle control for 24 hours then viability and cell count will be measured by trypan blue using the Vi-Cell cell counter. The cell growth will be determined from the cell count compared to control. RCE1 and control sirna knockdown will be used as a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. Next, to determine migration and invasion, I will perform scratch tests and matrix invasion assays. Migration will be measured by seeding a confluent monolayer of cell lines treated with the RCE1 inhibitors at the concentration determined to be the IC50 for RCE1 activity determined previously (21) or vehicle control for 24 hours in a tissue culture dish. The surface of the dish will be scratched with a pipette tip and images of the dish will be taken by IncuCyte every 4 hours for 48 hours. RCE1 and control sirna knockdown will be used as a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. Distance between the sides of the scratch will be analyzed by Media Cybernetics Image Pro-Plus software. Invasiveness will be assessed by seeding cells from each cell line treated with the RCE1 inhibitors at the concentration determined to be the IC50 for RCE1 activity determined previously (21) or vehicle control for
5 24 hours at 70% confluency on an upper compartment containing cell permeable membrane coated with basement membrane extract at low, medium and high density available through R&D Systems, cells that degrade the matrix and migrate through the membrane will be stained and counted after 48 hours. RCE1 and control sirna knockdown will be used as a genetic control to ensure results are specific for RCE1 and not an artifact of off-target effects of the inhibitor. Lastly, to determine the effects of RCE1 inhibition on tumor burden, metastasis and survival, I will use a xenograft mouse model. To measure tumor burden and metastasis by luciferin bioluminescence, I will transduce Panc and Panc cells with a luciferase tag. This will allow for non-invasive imaging of the cells using IVIS 100 or IVIS Spectrum in vivo imaging systems provided by the small animal imaging facility at MD Anderson Cancer Center. Dose response assays will be performed on the transduced cells to ensure the vector and luciferase tag have not altered the cells response to the RCE1 inhibitors. For the in vivo experiments, a cell line derived orthotopic xenograft athymic nude mouse model will be utilized as this provides essential stromal support. This also contributes an immune system with intact natural killer cells, B-cells, granulocytes, macrophages and dendritic cells that is only lacking in mature T- cells, while still allowing for engraftment (23). Athymic nude mice grouped into 4 arms containing 5 mice each will be injected orthotopically with 200,000 cells from each cell line tagged with luciferase, once the cells have engrafted as determined by luciferin bioluminescence the mice will be injected intravenously with the RCE1 inhibitor that caused the greatest decrease in viability, cell growth, migration and invasion at the concentration determined to be the IC50 for RCE1 activity determined Figure 4: Schematic for animal studies. Athymic nude mice will be injected orthotopically with luciferase tagged PDAC cells. Once the cells have engrafted, the mouse models will be treated with the RCE1 inhibitor which was most effective in vitro. Tumor burden and metastasis will be tracked by luciferin bioluminescence every 7 days. previously (21) or vehicle control every 24 hours (Figure 4). Survival will be assessed as well as tumor burden and metastasis as measured by luciferin bioluminescence every 7 days for 120 days. Animal studies will be performed in accordance with the Animal Care and Use Committee of the University of Texas M.D. Anderson Cancer Center. Animals will be sacrificed once the tumor has reached a diameter of 1.5cm or at signs of distress, such as inability to access food or water or labored respiration. Expected Outcomes/Alternative Approaches: I expect the use of the novel RCE1 inhibitors and RCE1 knockdown by sirna will significantly decrease Ras phosphorylation of PI3K, Akt, MEK and ERK, as measured by immunoblot, and Ras localization to the cell membrane as measured by fluorescent microscopy and subcellular fractionation in the cell lines. Additionally, I expect the use of the RCE1 inhibitors and RCE1 knockdown by sirna will decrease viability and cell growth as measured by trypan blue assay, migration as measured by the scratch migration assay, and invasion as measured by the matrix invasion assay. Lastly, I expect RCE1 inhibition will decrease tumor burden and metastasis as measured by luciferin bioluminescence and increase survival in the athymic mouse models. As RCE1 inhibition and knockdown have been shown to disrupt Ras localization to the cell membrane (20), it is highly unlikely that there will not be a decrease in co-localization to the membrane in PDAC cells. If this is not seen, the concentrations of inhibitors will be altered as determined by dose response. It is unlikely that a decrease in phosphorylation of PI3K, Akt, MEK and ERK are not seen with RCE1 inhibition or sirna knockdown if there is a decrease in Ras plasma localization, as Ras activity has been shown to be dependent on plasma membrane localization (16). It is additionally unlikely that there will not be a decrease in viability, cell growth, migration or invasiveness of the PDAC cells with the RCE1 inhibitors or sirna knockdown if a decrease in the phosphorylation of PI3K, Akt, MEK and ERK is seen as these are key signaling pathways for survival and proliferation. If either is the case however, I will alter the concentrations and time points at which the assays are measured as determined by dose response and an 8 to 120 hour time course with the RCE1 inhibitors and sirna. If alterations to the concentrations and time point do not produce the expected molecular and cellular effects of RCE1 inhibition, RNASeq and CyTOF will be performed to identify a compensatory mechanism. It is unlikely that I will not see a decrease in tumor burden or metastasis or an increase in survival in athymic nude mice injected orthotopically with PDAC cells then treated with the RCE1 inhibitor that caused the greatest decrease in viability, cell growth, migration and invasion, as these in vitro results should be recapitulated in the in vivo model. If this is the case however, I will alter the concentration and dosing schedule of the inhibitor injections as determined by pharmacokinetic assays. If the cells cannot be tagged with luciferase without altering
6 their response to the RCE1 inhibitors, tumor burden and metastasis will be determined postmortem. Alternatively, cell line derived orthotopic xenograft humanized NOD/SCID mouse models or PDX-1-Cre, LSL- Kras G12D, LSL-Trp53 R172H/- (KPC) genetically engineered mouse models (GEMMs) will be used instead of athymic nude mice, if engraftment of PDAC cells does not occur in athymic nude mice or if cell line derived orthotopic xenograft do not resemble the histopathology of human PDAC (23). In addition, the pancreas and metastatic tumors will be harvested to determine the efficacy of RCE1 inhibition in vivo as measured by the cleavage of terminal AAX motif of Ras and/or Ras membrane localization. These tumors can also be used to identify other proteins that may be acting as compensatory mechanisms in the presence of RCE1 inhibition. Aim 2: Determine the synergistic effects of targeting RCE1 in combination with inhibitors of downstream effectors in PDAC. Hypothesis/Rationale: Previous attempts to target the Ras pathway often lead to compensatory mechanisms that reduce the effectiveness of monotherapies (19). However, combinations of inhibitors targeting multiple nodes within the same signaling pathway, such as the use of an RCE1 inhibitor in combination with a downstream inhibitor, can reduce the likelihood of this occurring. Inhibitors of downstream effectors, such as MEK, Akt and PI3K, are already FDA approved for the treatment of other cancer or are in clinical trials for the treatment of PDAC. Additionally, by using a combination treatment that work synergistically the concentrations of the inhibitors required can be lowered, limiting off target binding and decreasing side effects. I hypothesize that RCE1 inhibition will have synergistic effects in combination with inhibitors of downstream effectors, such as MEK, Akt or PI3K, in pancreatic ductal adenocarcinoma. Approach: Aim 2.1: Determine synergistic effects of RCE1 inhibitors in combination with MEK, Akt or PI3K inhibitors. To determine the concentrations at which RCE1 inhibitors have synergistic effects in combination in MEK, Akt or PI3K inhibitors, I will perform combination dose response assays for viability and cell growth in PDAC cells. I will start the dose response with concentrations that are lower and higher than the reported IC50 of each inhibitor, by half log or log increments. As the RCE1 inhibitors were previously reported to have IC50s of about 10 μm (21), the concentrations of RCE1 inhibitors for the dose response will be 1, 3, 10 and 30 μm. The MEK inhibitor, selumetinib, which has been used in clinical trials for PDAC (13, 14) has an IC50 of 14nM according to the Selleckchem technical datasheet and is able to reduce cell phosphorylation of ERK1/2 in vitro at concentrations of less than 0.5 μm (24) therefore selumetinib at the concentrations of 0.01, 0.03, 0.1, 0.3 and 1 μm will be used in the dose response. Since the Akt inhibitor, MK-2206, has IC50s for Akt 1, 2, and 3 of 8, 12 and 65 nm, respectively, according to the Selleckchem technical datasheet, and all three isoforms have been shown to play a role in chemoresistance (25) MK-2206 at the concentrations of 3, 10, 30, and 100 nm will be used for the dose response. Lastly, the PI3K inhibitor, idelalisib, currently FDA-approved for the treatment of chronic lymphocytic leukemia, has an IC50 of 2.5 nm for the p110δ subunit of PI3K according to the Selleckchem technical datasheet and has been shown to decrease viability of multiple myeloma patient samples by 50% with no effect to healthy cells at concentrations lower than 20μM (26). Therefore, I will use idelalisib at the concentrations of 0.01, 0.1, 1 and 10 μm for the dose response. I will treat 500,000 cells from each cell line with the RCE1 inhibitor and MEK, Akt or PI3K inhibitor, separately and in combination, at the determined concentrations. The viability and cell growth will be measured by trypan blue using the Vi-Cell cell counter. CalcuSyn will be used to analyze the results for synergistic effects. Aim 2.2: Evaluate effects of RCE1 inhibition in combination with MEK, Akt or PI3K inhibition on migration, invasion and in vivo tumor burden, metastasis and survival. I will seed a confluent monolayer of each cell line treated with the RCE1 inhibitor determined to have the most synergistic effects or vehicle control in combination with MEK, Akt, or PI3K inhibitor or vehicle control at the synergistic concentrations determined in a tissue culture dish. The surface of the dish will be scratched and the distance between the sides of the scratch will be assessed as described in Aim 1.2. Next, I will determine the effects of RCE1 inhibition in combination with MEK, Akt or PI3K inhibition on invasiveness by matrix invasion assays. PDAC cells from each cell line will be treated with the most synergistic RCE1 inhibitor or vehicle control in combination with MEK, Akt, or PI3K inhibitor or vehicle control at the determined synergistic concentrations for 24 hours will be seeded at 70% confluency on an upper compartment containing cell permeable membrane coated with basement membrane extract at low, medium and high density available through R&D Systems, cells that degrade the matrix and migrate through the membrane will be stained and counted after 48 hours and invasiveness will be assess as described in Aim 1.2. Lastly, I will use PDAC cells in an orthotopic xenograft mouse model to determine if tumor growth and metastasis
7 are decreased in vivo after treatment with the synergistic RCE1 inhibitor and MEK, Akt, or PI3K inhibitor determined to have the most synergistic effects. Athymic nude mice will be grouped into 8 arms containing 5 mice each which will be injected orthotopically with 200,000 cells from each cell line tagged with luciferase, once the cells have engrafted as determined by luciferin bioluminescence the mice will be injected with RCE1 or the inhibitor of the downstream effector that had resulted in the best effect in vitro at the concentrations determined to have the most synergistic effects, or the combination of both RCE1 inhibitor and downstream effector inhibitor or vehicle control every 24 hours. These concentrations are not to exceed the 120 mg/kg, 100mg/kg and 30mg/kg doses recommended for MK-2206, selumetinib and idelalisib, respectively, according to the Selleckchem technical datasheet. Survival will be assessed as well as tumor burden and metastasis as measured by luciferin bioluminescence, as described in Aim 1.2. Animal studies will be performed in accordance with the Animal Care and Use Committee of the University of Texas M.D. Anderson Cancer Center. Animals will be sacrificed once the tumor has reached a diameter of 1.5cm or at signs of distress, such as inability to access food or water or labored respiration. Expected Outcomes/Alternative Approaches: I expect cell viability, cell growth, migration and invasiveness of PDAC cells to decrease when treated with RCE1 inhibitor in combination with MEK, Akt, or PI3K inhibitors. I expect mice orthotopically injected with PDAC cells then treated with RCE1 inhibitor in combination with MEK, Akt, or PI3K inhibitors to have decreased tumor burden and metastasis and increased survival. It is unlikely that there will not be at least an additive effect of using RCE1 inhibitor in combination with MEK, Akt, or PI3K inhibitors, however there is a possibility that no synergistic effects will be seen, as these inhibitors are targeting the same pathway. In the case that only an additive effect is seen, the inhibitors will be used at their lowest combined concentration that still induces a decrease in viability, cell growth migration and invasion as this should still provide the benefit of reducing the concentration of each inhibitor needed to induce the desired effect. As discussed in the expected outcomes/alternative approaches of Aim 1, it is unlikely that there will not be a decrease in viability, cell growth, migration or invasiveness of the PDAC cells with combination of the RCE1 inhibitor and MEK, Akt, or PI3K inhibitor, even if no decrease in viability, cell growth, migration or invasiveness is seen with RCE1 inhibitor, as MEK, Akt, or PI3K are key signaling pathways for survival and proliferation. If that is the case however, I will alter the time points at which the assays are measured as determined by an 8 to 120 hour time course with the RCE1 inhibitor and MEK, Akt, or PI3K inhibitor. It is unlikely that I will not see a decrease in tumor burden or metastasis or an increase in survival in athymic nude mice injected orthotopically with PDAC cells then treated with the combination of RCE1 inhibitor and ICMT, MEK, PI3K or MAPK, if there is a decrease in viability, cell growth, migration or invasiveness. If this is the case however, I will alter the dosing schedule of the inhibitor injections as determined by pharmacokinetic assays. As mentioned in the alternative approaches in Aim 1, if the cells cannot be tagged with luciferase without altering their response to the RCE1 inhibitors, tumor burden and metastasis will be determined postmortem. Additionally, as mentioned in the alternative approaches in Aim 1, cell line derived orthotopic xenograft humanized NOD/SCID mouse models or PDX-1-Cre, LSL- Kras G12D, LSL-Trp53 R172H/- (KPC) genetically engineered mouse models (GEMMs) will be used instead of athymic nude mice, if engraftment of PDAC cells does not occur in athymic nude mice or if cell line derived orthotopic xenograft do not resemble the histopathology of human PDAC (23,24). Finally, the pancreas and metastatic tumors can be harvested to determine the efficacy of RCE1 and MEK, Akt or PI3K inhibition in vivo and/or to identify compensatory mechanisms, as mentioned in the alternative approaches in Aim 1, if there is no change in survival, tumor burden and/or metastasis. References 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, CA Cancer J Clin. 2016; 66(1): Kamisawa T, Wood LD, Itoi T, Takaori K. Pancreatic Cancer. Lancet. 2016; 388(10039): Hidalgo M, Cascinu S, Kleeff J, et al. Addressing the challenges of pancreatic cancer: future directions for improving outcomes. Pancreatology. 2015;15(1): Adamska A, Domenichini A, Falasca M. Pancreatic Ductal Adenocarcinoma: Current and Evolving Therapies. Int J Mol Sci. 2017; 18(7). 5. Burris HA, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15(6): Conroy T, Bachet JB, Ayav A, et al. Current standards and new innovative approaches for treatment of pancreatic cancer. Eur J Cancer. 2016;57:10-22.
8 7. Fokas E, O'neill E, Gordon-weeks A, Mukherjee S, Mckenna WG, Muschel RJ. Pancreatic ductal adenocarcinoma: From genetics to biology to radiobiology to oncoimmunology and all the way back to the clinic. Biochim Biophys Acta. 2015;1855(1): Muniraj T, Jamidar PA, Aslanian HR. Pancreatic cancer: a comprehensive review and update. Dis Mon. 2013;59(11): Falasca M, Kim M, Casari I. Pancreatic cancer: Current research and future directions. Biochim Biophys Acta. 2016;1865(2): Chiaravalli M, Reni M, O'reilly EM. Pancreatic ductal adenocarcinoma: State-of-the-art 2017 and new therapeutic strategies. Cancer Treat Rev. 2017;60: Hruban RH, Goggins M, Parsons J, Kern SE. Progression model for pancreatic cancer. Clin Cancer Res. 2000;6(8): Dunne RF, Hezel AF. Genetics and Biology of Pancreatic Ductal Adenocarcinoma. Hematol Oncol Clin North Am. 2015;29(4): Mcnally LR, Mezera M, Morgan DE, et al. Current and Emerging Clinical Applications of Multispectral Optoacoustic Tomography (MSOT) in Oncology. Clin Cancer Res. 2016; 14. Chung V, Mcdonough S, Philip PA, et al. Effect of Selumetinib and MK-2206 vs Oxaliplatin and Fluorouracil in Patients With Metastatic Pancreatic Cancer After Prior Therapy: SWOG S1115 Study Randomized Clinical Trial. JAMA Oncol. 2017;3(4): Oettle H. Progress in the knowledge and treatment of advanced pancreatic cancer: from benchside to bedside. Cancer Treat Rev. 2014;40(9): Paulson AS, Tran cao HS, Tempero MA, Lowy AM. Therapeutic advances in pancreatic cancer. Gastroenterology. 2013;144(6): Cox AD, Der CJ, Philips MR. Targeting RAS Membrane Association: Back to the Future for Anti-RAS Drug Discovery?. Clin Cancer Res. 2015;21(8): Simanshu DK, Nissley DV, Mccormick F. RAS Proteins and Their Regulators in Human Disease. Cell. 2017;170(1): Papke B, Der CJ. Drugging RAS: Know the enemy. Science. 2017;355(6330): Vasan N, Boyer JL, Herbst RS. A RAS renaissance: emerging targeted therapies for KRAS-mutated nonsmall cell lung cancer. Clin Cancer Res. 2014;20(15): Wang Y, Kaiser CE, Frett B, Li HY. Targeting mutant KRAS for anticancer therapeutics: a review of novel small molecule modulators. J Med Chem. 2013;56(13): Mohammed I, Hampton SE, Ashall L, et al. 8-Hydroxyquinoline-based inhibitors of the RCE1 protease disrupt Ras membrane localization in human cells. Bioorg Med Chem. 2016;24(2): Tam A, Schmidt WK, Michaelis S. The multispanning membrane protein Ste24p catalyzes CAAX proteolysis and NH2-terminal processing of the yeast a-factor precursor. J Biol Chem. 2001;276(50): Ponz-sarvise M, Tuveson DA, Yu KH. Mouse Models of Pancreatic Ductal Adenocarcinoma. Hematol Oncol Clin North Am. 2015;29(4): Huynh H, Soo KC, Chow PK, Tran E. Targeted inhibition of the extracellular signal-regulated kinase kinase pathway with AZD6244 (ARRY ) in the treatment of hepatocellular carcinoma. Mol Cancer Ther. 2007;6(1): Gdowski A, Panchoo M, Treuren TV, Basu A. Emerging therapeutics for targeting Akt in cancer. Front Biosci (Landmark Ed). 2016;21: Ikeda H, Hideshima T, Fulciniti M, et al. PI3K/p110{delta} is a novel therapeutic target in multiple myeloma. Blood. 2010;116(9):
CLINICAL UPDATE ON K-RAS
 CLINICAL UPDATE ON K-RAS TARGETED THERAPY IN GASTROINTESTINAL CANCERS S. PA N T, 1 J. H U B B A R D, 2 E. M A RT I N E L L I, 3 A N D T. B E K A I I - S A A B 4 SELECTED HIGHLIGHTS 1 Department of Investigational
CLINICAL UPDATE ON K-RAS TARGETED THERAPY IN GASTROINTESTINAL CANCERS S. PA N T, 1 J. H U B B A R D, 2 E. M A RT I N E L L I, 3 A N D T. B E K A I I - S A A B 4 SELECTED HIGHLIGHTS 1 Department of Investigational
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as
 Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
NIH Public Access Author Manuscript Clin Cancer Res. Author manuscript; available in PMC 2015 December 01.
 NIH Public Access Author Manuscript Published in final edited form as: Clin Cancer Res. 2014 December 1; 20(23): 5866 5868. doi:10.1158/1078-0432.ccr-14-1543. p38 MAPK in Pancreatic Cancer: Finding a Protective
NIH Public Access Author Manuscript Published in final edited form as: Clin Cancer Res. 2014 December 1; 20(23): 5866 5868. doi:10.1158/1078-0432.ccr-14-1543. p38 MAPK in Pancreatic Cancer: Finding a Protective
Biol403 MAP kinase signalling
 Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
Robert Newman, Ph.D. and Peiying Yang, Ph.D.
 Robert Newman, Ph.D. and Peiying Yang, Ph.D. Department of Experimental Therapeutics and General Oncology The University of Texas, M.D. Anderson Cancer Center 1 Cardiac glycoside extracts from plants and
Robert Newman, Ph.D. and Peiying Yang, Ph.D. Department of Experimental Therapeutics and General Oncology The University of Texas, M.D. Anderson Cancer Center 1 Cardiac glycoside extracts from plants and
Robert Newman, Ph.D. and Peiying Yang, Ph.D.
 Robert Newman, Ph.D. and Peiying Yang, Ph.D. Department of Experimental Therapeutics and General Oncology The University of Texas, M.D. Anderson Cancer Center Digitalis purpuria: Source of cardiac glycosides
Robert Newman, Ph.D. and Peiying Yang, Ph.D. Department of Experimental Therapeutics and General Oncology The University of Texas, M.D. Anderson Cancer Center Digitalis purpuria: Source of cardiac glycosides
Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC
 Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC Supplementary Table 2. Drug content and loading efficiency estimated with F-NMR and UV- Vis Supplementary Table 3. Complete
Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC Supplementary Table 2. Drug content and loading efficiency estimated with F-NMR and UV- Vis Supplementary Table 3. Complete
Quantification of early stage lesions for loss of p53 should be shown in the main figures.
 Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Cancer Biology Dynamical Cell Systems
 The Institute of Cancer Research PHD STUDENTSHIP PROJECT PROPOSAL PROJECT DETAILS Project Title: SUPERVISORY TEAM Primary Supervisor: The forces behind pancreatic cancer; and changing them as a therapeutic
The Institute of Cancer Research PHD STUDENTSHIP PROJECT PROPOSAL PROJECT DETAILS Project Title: SUPERVISORY TEAM Primary Supervisor: The forces behind pancreatic cancer; and changing them as a therapeutic
SUPPLEMENTARY INFORMATION In format provided by Sebti et al. (NOVEMBER 2011)
 Supplementary Information S4 Clinical trials with farnesyltransferase inhibitors Drug(s) Disease Phase Patients Median Age Tipifarnib CR Clinical response PR HI SD PD MD FT or Prenylation Response rate
Supplementary Information S4 Clinical trials with farnesyltransferase inhibitors Drug(s) Disease Phase Patients Median Age Tipifarnib CR Clinical response PR HI SD PD MD FT or Prenylation Response rate
B Cells Promote Pancreatic Tumorigenesis
 B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
Introduction. Cancer Biology. Tumor-suppressor genes. Proto-oncogenes. DNA stability genes. Mechanisms of carcinogenesis.
 Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
AWARD NUMBER: W81XWH TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers
 AWARD NUMBER: W81XWH-12-1-0420 TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers PRINCIPAL INVESTIGATOR: Ryan B. Corcoran, M.D., Ph.D. CONTRACTING
AWARD NUMBER: W81XWH-12-1-0420 TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers PRINCIPAL INVESTIGATOR: Ryan B. Corcoran, M.D., Ph.D. CONTRACTING
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the
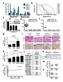 Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
Pancreatic intraepithelial
 Pancreatic intraepithelial neoplasia (PanIN) Markéta Hermanová St. Anne s University Hospital Brno Faculty of Medicine, Masaryk University Precursor lesions of invasive pancreatic cancer Pancreatic intraepithelial
Pancreatic intraepithelial neoplasia (PanIN) Markéta Hermanová St. Anne s University Hospital Brno Faculty of Medicine, Masaryk University Precursor lesions of invasive pancreatic cancer Pancreatic intraepithelial
Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis
 Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis Myron R. Szewczuk Dept. Biomedical and Molecular Sciences, Queen's University, Kingston, K7L 3N6 Ontario, Canada HIGHLIGHTS. An innovative
Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis Myron R. Szewczuk Dept. Biomedical and Molecular Sciences, Queen's University, Kingston, K7L 3N6 Ontario, Canada HIGHLIGHTS. An innovative
Mouse Models of K-RAS- and B-RAFinduced
 Mouse Models of K-RAS- and B-RAFinduced Cancer Martin O. Bergö, Professor Sahlgrenska Cancer Center University of Gothenburg www.gu.se Why models? Why mice? Critical to understanding pathogenesis and identifying
Mouse Models of K-RAS- and B-RAFinduced Cancer Martin O. Bergö, Professor Sahlgrenska Cancer Center University of Gothenburg www.gu.se Why models? Why mice? Critical to understanding pathogenesis and identifying
AACR 101st Annual Meeting 2010, Washington D.C. Experimental and Molecular Therapeutics Section 29; Abstract #3855
 Investigation of the Growth Inhibitory Activity of the MEK Inhibitor ARRY-162 in Combination with Everolimus in a Variety of KRas and PI3K Pathway Mutant Cancers Brian Tunquist, Tyler Risom, Debbie Anderson,
Investigation of the Growth Inhibitory Activity of the MEK Inhibitor ARRY-162 in Combination with Everolimus in a Variety of KRas and PI3K Pathway Mutant Cancers Brian Tunquist, Tyler Risom, Debbie Anderson,
Cancer model Liver metastasis I
 Cancer model Liver metastasis I H. Suemizu, M. Monnai, Y. Ohnishi, M. Ito, N. Tamaoki, and M. Nakamura. 2007. Identification of a key molecular regulator of liver metastasis in human pancreatic carcinoma
Cancer model Liver metastasis I H. Suemizu, M. Monnai, Y. Ohnishi, M. Ito, N. Tamaoki, and M. Nakamura. 2007. Identification of a key molecular regulator of liver metastasis in human pancreatic carcinoma
Cancer therapeutics in the clinical setting. Need for alternate therapeutics. Target multiple cancer pathways. Circumvent the genetic mutations
 Transcriptional factor Snail and MMP-9 signaling axis controls tumor neovascularization, growth and metastasis in mouse model of human ovarian carcinoma Samar Abdulkhalek 1, Olivia Geen 1, Lacey Brodhagen
Transcriptional factor Snail and MMP-9 signaling axis controls tumor neovascularization, growth and metastasis in mouse model of human ovarian carcinoma Samar Abdulkhalek 1, Olivia Geen 1, Lacey Brodhagen
Targeting of the MUC1-C Oncoprotein in Colitis-Associated Colorectal Cancer
 AD Award Number: W81XWH-12-1-0322 TITLE: Targeting of the MUC1-C Oncoprotein in Colitis-Associated Colorectal Cancer PRINCIPAL INVESTIGATOR: Kufe, Donald W., M.D. CONTRACTING ORGANIZATION: Boston, MA 02215-5450
AD Award Number: W81XWH-12-1-0322 TITLE: Targeting of the MUC1-C Oncoprotein in Colitis-Associated Colorectal Cancer PRINCIPAL INVESTIGATOR: Kufe, Donald W., M.D. CONTRACTING ORGANIZATION: Boston, MA 02215-5450
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL. PhD THESIS
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
Supplementary Figure 1. a. b. Relative cell viability. Nature Genetics: doi: /ng SCR shyap1-1 shyap
 Supplementary Figure 1. a. b. p-value for depletion in vehicle (DMSO) 1e-05 1e-03 1e-01 1 0 1000 2000 3000 4000 5000 Genes log2 normalized shrna counts in T0 0 2 4 6 8 sh1 shluc 0 2 4 6 8 log2 normalized
Supplementary Figure 1. a. b. p-value for depletion in vehicle (DMSO) 1e-05 1e-03 1e-01 1 0 1000 2000 3000 4000 5000 Genes log2 normalized shrna counts in T0 0 2 4 6 8 sh1 shluc 0 2 4 6 8 log2 normalized
AP VP DLP H&E. p-akt DLP
 A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
EGFR: fundamenteel en klinisch
 EGFR: fundamenteel en klinisch Guido Lammering MAASTRO Clinic Maastricht, NL What is EGFR?? The EGFR some facts 1186 amino acids 170 kda Expressed by all cells of epithelial origin Increased activation
EGFR: fundamenteel en klinisch Guido Lammering MAASTRO Clinic Maastricht, NL What is EGFR?? The EGFR some facts 1186 amino acids 170 kda Expressed by all cells of epithelial origin Increased activation
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC
 Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplemental information
 Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Development of Rational Drug Combinations for Oncology Indications - An Industry Perspective
 Development of Rational Drug Combinations for Oncology Indications An Industry Perspective Stuart Lutzker, MDPhD Vice President Oncology Early Development Genentech Inc 1 Conventional Oncology Drug Development
Development of Rational Drug Combinations for Oncology Indications An Industry Perspective Stuart Lutzker, MDPhD Vice President Oncology Early Development Genentech Inc 1 Conventional Oncology Drug Development
Molecular targets for altering radiosensitivity: Lessons from Ras as a pre-clinical and clinical model
 Critical Reviews in Oncology/Hematology 55 (2005) 103 116 Molecular targets for altering radiosensitivity: Lessons from Ras as a pre-clinical and clinical model Keith A. Cengel, W. Gillies McKenna Department
Critical Reviews in Oncology/Hematology 55 (2005) 103 116 Molecular targets for altering radiosensitivity: Lessons from Ras as a pre-clinical and clinical model Keith A. Cengel, W. Gillies McKenna Department
CANCER THERAPEUTICS: A NOVEL APPROACH
 CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH-1-1-176 TITLE: Suppression of BRCA2 by Mutant Mitochondrial DNA in Prostate Cancer PRINCIPAL INVESTIGATOR: Hsieh, Jer-Tsong CONTRACTING ORGANIZATION: University of Texas Southwestern
AD Award Number: W81XWH-1-1-176 TITLE: Suppression of BRCA2 by Mutant Mitochondrial DNA in Prostate Cancer PRINCIPAL INVESTIGATOR: Hsieh, Jer-Tsong CONTRACTING ORGANIZATION: University of Texas Southwestern
Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung
 Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung immortalized broncho-epithelial cells (AALE cells) expressing
Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung immortalized broncho-epithelial cells (AALE cells) expressing
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors have presented data demonstrating activation of AKT as a common resistance mechanism in EGFR mutation positive, EGFR TKI resistant
Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors have presented data demonstrating activation of AKT as a common resistance mechanism in EGFR mutation positive, EGFR TKI resistant
Antitumor Activity of CUDC-305, a Novel Oral HSP90 Inhibitor, in Solid and Hematological Tumor Xenograft Models
 Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Cancer Biology Course. Invasion and Metastasis
 Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures
 Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures Supplementary Figure 1 mir-128-3p is highly expressed in chemoresistant, metastatic
Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures Supplementary Figure 1 mir-128-3p is highly expressed in chemoresistant, metastatic
p53 expression in invasive pancreatic adenocarcinoma and precursor lesions
 Malaysian J Pathol 2011; 33(2) : 89 94 ORIGINAL ARTICLE p53 expression in invasive pancreatic adenocarcinoma and precursor lesions NORFADZILAH MY MBBCH,* Jayalakshmi PAILOOR MPath, FRCPath,* RETNESWARI
Malaysian J Pathol 2011; 33(2) : 89 94 ORIGINAL ARTICLE p53 expression in invasive pancreatic adenocarcinoma and precursor lesions NORFADZILAH MY MBBCH,* Jayalakshmi PAILOOR MPath, FRCPath,* RETNESWARI
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Correlation between LKB1 and YAP expression in human lung cancer samples. (a) Representative photos showing LKB1 and YAP immunohistochemical staining in human
Supplementary Figures Supplementary Figure 1 Correlation between LKB1 and YAP expression in human lung cancer samples. (a) Representative photos showing LKB1 and YAP immunohistochemical staining in human
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth.
 Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
(A) Cells grown in monolayer were fixed and stained for surfactant protein-c (SPC,
 Supplemental Figure Legends Figure S1. Cell line characterization (A) Cells grown in monolayer were fixed and stained for surfactant protein-c (SPC, green) and co-stained with DAPI to visualize the nuclei.
Supplemental Figure Legends Figure S1. Cell line characterization (A) Cells grown in monolayer were fixed and stained for surfactant protein-c (SPC, green) and co-stained with DAPI to visualize the nuclei.
RAS Genes. The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes.
 ۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
Isoprenylcysteine carboxylmethyltransferase deficiency exacerbates KRAS-driven pancreatic neoplasia via Notch suppression
 Research article Isoprenylcysteine carboxylmethyltransferase deficiency exacerbates KRAS-driven pancreatic neoplasia via Notch suppression Helen Court, 1,2,3,4 Marc Amoyel, 3 Michael Hackman, 5 Kyoung
Research article Isoprenylcysteine carboxylmethyltransferase deficiency exacerbates KRAS-driven pancreatic neoplasia via Notch suppression Helen Court, 1,2,3,4 Marc Amoyel, 3 Michael Hackman, 5 Kyoung
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
TTI-2341: A Novel Brain-Penetrant, Orally Available, Covalent EGFR Inhibitor for the Treatment of Brain Cancers
 TTI-2341: A Novel Brain-Penetrant, Orally Available, Covalent EGFR Inhibitor for the Treatment of Brain Cancers November 2017 2 EGFR is a Drug Target in Brain Cancer Epidermal growth factor receptor (EGFR)
TTI-2341: A Novel Brain-Penetrant, Orally Available, Covalent EGFR Inhibitor for the Treatment of Brain Cancers November 2017 2 EGFR is a Drug Target in Brain Cancer Epidermal growth factor receptor (EGFR)
Programa Cooperación Farma-Biotech Jornada II: Oncología
 Programa Cooperación Farma-Biotech Jornada II: Oncología Minerval : Treatment of glioma and other types of cancer Vicenç Tur. CEO, Co-founder Barcelona, 13 de abril de 2011 Programa Cooperación Farma-Biotech
Programa Cooperación Farma-Biotech Jornada II: Oncología Minerval : Treatment of glioma and other types of cancer Vicenç Tur. CEO, Co-founder Barcelona, 13 de abril de 2011 Programa Cooperación Farma-Biotech
SUPPLEMENTAL TEXT AND FIGURES
 SUPPLEMENTAL TEXT AND FIGURES Prrx1 isoform switching regulates pancreatic cancer invasion and metastatic colonization Shigetsugu Takano, Maximilian Reichert, Basil Bakir, Koushik K. Das, Takahiro Nishida,
SUPPLEMENTAL TEXT AND FIGURES Prrx1 isoform switching regulates pancreatic cancer invasion and metastatic colonization Shigetsugu Takano, Maximilian Reichert, Basil Bakir, Koushik K. Das, Takahiro Nishida,
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Molecular and genetic analysis of stromal fibroblasts in prostate cancer
 Final report ESMO Translational Research Fellowship 2010-2011 Molecular and genetic analysis of stromal fibroblasts in prostate cancer Michalis Karamouzis Host Institute Department of Biological Chemistry,
Final report ESMO Translational Research Fellowship 2010-2011 Molecular and genetic analysis of stromal fibroblasts in prostate cancer Michalis Karamouzis Host Institute Department of Biological Chemistry,
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Exosomal Del 1 as a potent diagnostic marker for breast cancer : A prospective cohort study
 GBCC 2017: ABS-0017 Exosomal Del 1 as a potent diagnostic marker for breast cancer : A prospective cohort study Soo Jung Lee 1, Jeeyeon Lee 2, Jin Hyang Jung 2, Ho Yong Park 2, Chan Hyeong Lee 3, Pyong
GBCC 2017: ABS-0017 Exosomal Del 1 as a potent diagnostic marker for breast cancer : A prospective cohort study Soo Jung Lee 1, Jeeyeon Lee 2, Jin Hyang Jung 2, Ho Yong Park 2, Chan Hyeong Lee 3, Pyong
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Figure S1, related to Figure 1. Escaper p38a-expressing cancer cells repopulate the tumors (A) Scheme of the mt/mg reporter that expresses a
 Cancer Cell, Volume 33 Supplemental Information Targeting p38a Increases DNA Damage, Chromosome Instability, and the Anti-tumoral Response to Taxanes in Breast Cancer Cells Begoña Cánovas, Ana Igea, Alessandro
Cancer Cell, Volume 33 Supplemental Information Targeting p38a Increases DNA Damage, Chromosome Instability, and the Anti-tumoral Response to Taxanes in Breast Cancer Cells Begoña Cánovas, Ana Igea, Alessandro
Supplementary Figures
 Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
 Rath, N., and Olson, M. (2016) Regulation of pancreatic cancer aggressiveness by stromal stiffening. Nature Medicine, 22(5), pp. 462-463. There may be differences between this version and the published
Rath, N., and Olson, M. (2016) Regulation of pancreatic cancer aggressiveness by stromal stiffening. Nature Medicine, 22(5), pp. 462-463. There may be differences between this version and the published
The 2010 Gastrointestinal Cancers Symposium Oral Abstract Session: Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract
 The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
Plate-Based Assay Methods for the Assessment of Cellular Health
 Plate-Based Assay Methods for the Assessment of Cellular Health Andrew L. Niles, Senior Research Scientist 2012, Promega Corporation. Biological Outcomes in Cell Culture Treatment -Small molecule -Bio-molecule
Plate-Based Assay Methods for the Assessment of Cellular Health Andrew L. Niles, Senior Research Scientist 2012, Promega Corporation. Biological Outcomes in Cell Culture Treatment -Small molecule -Bio-molecule
11/18/2015. Gloria Su, Ph.D. Path G4500. Cancer Statistics 2015 (Siegel et al, CA Cancer J CLIN 2015) The Pancreatic Cancer Burden
 Mouse Modeling for Human Pancreatic Cancer Gloria Su, Ph.D. gs2157@columbia.edu, ICRC 10-04 Associate Professor Departments of Pathology and Otolaryngology/Head and Neck Surgery Columbia University College
Mouse Modeling for Human Pancreatic Cancer Gloria Su, Ph.D. gs2157@columbia.edu, ICRC 10-04 Associate Professor Departments of Pathology and Otolaryngology/Head and Neck Surgery Columbia University College
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
A. Generation and characterization of Ras-expressing autophagycompetent
 Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Research article. Department of Pharmacology and Toxicology, Medical College of Georgia, Georgia Health Sciences University, Augusta, Georgia, USA.
 Research article Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension, and its inhibition attenuates pathologic features of disease Wanli Ma, 1 Weihong Han, 1 Peter
Research article Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension, and its inhibition attenuates pathologic features of disease Wanli Ma, 1 Weihong Han, 1 Peter
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Imaging of glycolytic metabolism in primary glioblastoma cells with
 63 Chapter 5 Imaging of glycolytic metabolism in primary glioblastoma cells with RIMChip 5.1. Introduction Glioblastoma(GBM) is one of the most common brain tumors 1. It is composed of heterogeneous subpopulations
63 Chapter 5 Imaging of glycolytic metabolism in primary glioblastoma cells with RIMChip 5.1. Introduction Glioblastoma(GBM) is one of the most common brain tumors 1. It is composed of heterogeneous subpopulations
FGL2 A new biomarker for cancer in a simple blood test
 FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach
 Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Smoke Like a Man, Survive Like a Woman
 Smoke Like a Man, Survive Like a Woman Sex Specific Differences in Lung Cancer Pulmonary Grand Rounds Philippe R. Montgrain, M.D. May 1, 2008 Objectives 1. Review how lung cancer differs in men and women.
Smoke Like a Man, Survive Like a Woman Sex Specific Differences in Lung Cancer Pulmonary Grand Rounds Philippe R. Montgrain, M.D. May 1, 2008 Objectives 1. Review how lung cancer differs in men and women.
7/6/2015. Cancer Related Deaths: United States. Management of NSCLC TODAY. Emerging mutations as predictive biomarkers in lung cancer: Overview
 Emerging mutations as predictive biomarkers in lung cancer: Overview Kirtee Raparia, MD Assistant Professor of Pathology Cancer Related Deaths: United States Men Lung and bronchus 28% Prostate 10% Colon
Emerging mutations as predictive biomarkers in lung cancer: Overview Kirtee Raparia, MD Assistant Professor of Pathology Cancer Related Deaths: United States Men Lung and bronchus 28% Prostate 10% Colon
Synthesis and Biological Evaluation of Protein Kinase D Inhibitors
 Synthesis and Biological Evaluation of Protein Kinase D Inhibitors Celeste Alverez Topic Seminar October 26, 2013 Celeste Alverez @ Wipf Group 10/26/2013 1 Protein Kinase D (PKD) A novel family of serine/threonine
Synthesis and Biological Evaluation of Protein Kinase D Inhibitors Celeste Alverez Topic Seminar October 26, 2013 Celeste Alverez @ Wipf Group 10/26/2013 1 Protein Kinase D (PKD) A novel family of serine/threonine
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 Award Number: W81XWH-13-1-0198 TITLE: The Role of Akt Isoforms in Colorectal Cancer PRINCIPAL INVESTIGATOR: Jatin Roper CONTRACTING ORGANIZATION: Tufts Medical Center Boston, MA 02111 REPORT DATE: July
Award Number: W81XWH-13-1-0198 TITLE: The Role of Akt Isoforms in Colorectal Cancer PRINCIPAL INVESTIGATOR: Jatin Roper CONTRACTING ORGANIZATION: Tufts Medical Center Boston, MA 02111 REPORT DATE: July
Pancreatic cancer from the past to the future
 Pancreatic cancer from the past to the future Darren Sigal, MD Scripps Clinic Pancreas and Bile Duct Cancer Group Division of Hematology/Oncology Scripps Clinic Pancreas and Bile Duct Cancer Group 1 Objectives
Pancreatic cancer from the past to the future Darren Sigal, MD Scripps Clinic Pancreas and Bile Duct Cancer Group Division of Hematology/Oncology Scripps Clinic Pancreas and Bile Duct Cancer Group 1 Objectives
Discovery and Optimization of Inhibitors of STAT3 Activation for the Treatment of Squamous Cell Carcinoma of the Head and Neck
 Discovery and ptimization of Inhibitors of STAT3 Activation for the Treatment of Squamous Cell Carcinoma of the Head and Neck Feng Zhang Wipf Group Research Topic Seminar 02-09-2013 1 Feng Zhang @ Wipf
Discovery and ptimization of Inhibitors of STAT3 Activation for the Treatment of Squamous Cell Carcinoma of the Head and Neck Feng Zhang Wipf Group Research Topic Seminar 02-09-2013 1 Feng Zhang @ Wipf
GI ONCOLOGY CLINICAL TRIALS AT UCDAVIS
 G ONCOLOGY CLNCAL TRALS AT UCDAVS Version: DECEMBER 2017 Please contact us with any questions or any potential clinical patients Edward Kim, MD/PhD email: jhkim@ucdavis.edu pager: 916-816-4059 May Cho,
G ONCOLOGY CLNCAL TRALS AT UCDAVS Version: DECEMBER 2017 Please contact us with any questions or any potential clinical patients Edward Kim, MD/PhD email: jhkim@ucdavis.edu pager: 916-816-4059 May Cho,
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Information
 Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
The PI3K/AKT axis. Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia. Introduction
 The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
SUPPLEMENTARY FIGURES AND TABLES
 SUPPLEMENTARY FIGURES AND TABLES Supplementary Figure S1: CaSR expression in neuroblastoma models. A. Proteins were isolated from three neuroblastoma cell lines and from the liver metastasis of a MYCN-non
SUPPLEMENTARY FIGURES AND TABLES Supplementary Figure S1: CaSR expression in neuroblastoma models. A. Proteins were isolated from three neuroblastoma cell lines and from the liver metastasis of a MYCN-non
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplemental Figures:
 Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Is it possible to cure patients with liver metastases? Taghizadeh Ali MD Oncologist, MUMS
 Is it possible to cure patients with liver metastases? Taghizadeh Ali MD Oncologist, MUMS Survival Rates of by Stage of Adenocarcinoma of the Colon Liver Resection New Perspective Colorectal cancer liver
Is it possible to cure patients with liver metastases? Taghizadeh Ali MD Oncologist, MUMS Survival Rates of by Stage of Adenocarcinoma of the Colon Liver Resection New Perspective Colorectal cancer liver
Antigen Presentation to T lymphocytes
 Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antibodies and T cell receptors
Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antibodies and T cell receptors
Tissue Factor/PAR2 signaling enhances the malignancy and radiation resistance of lung cancer brain metastases
 Tissue Factor/PAR2 signaling enhances the malignancy and radiation resistance of lung cancer brain metastases Dusten Unruh, PhD 8 th Annual Brain Metastases Research and Emerging Therapy Conference Tissue
Tissue Factor/PAR2 signaling enhances the malignancy and radiation resistance of lung cancer brain metastases Dusten Unruh, PhD 8 th Annual Brain Metastases Research and Emerging Therapy Conference Tissue
Supplemental Table S1
 Supplemental Table S. Tumorigenicity and metastatic potential of 44SQ cell subpopulations a Tumorigenicity b Average tumor volume (mm ) c Lung metastasis d CD high /4 8. 8/ CD low /4 6./ a Mice were injected
Supplemental Table S. Tumorigenicity and metastatic potential of 44SQ cell subpopulations a Tumorigenicity b Average tumor volume (mm ) c Lung metastasis d CD high /4 8. 8/ CD low /4 6./ a Mice were injected
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San
 Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
ab CytoPainter Golgi/ER Staining Kit
 ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
Molecular genetics of PDAC
 Molecular genetics of PDAC Irene Esposito, Melissa Schlitter Pancreatic pathology: Of mice and men Madrid, December 4-6th 2014 Background: Molecular genetic analysis of DNA from paraffin-embedded PDAC
Molecular genetics of PDAC Irene Esposito, Melissa Schlitter Pancreatic pathology: Of mice and men Madrid, December 4-6th 2014 Background: Molecular genetic analysis of DNA from paraffin-embedded PDAC
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast
 Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
OMICS Journals are welcoming Submissions
 OMICS Journals are welcoming Submissions OMICS International welcomes submissions that are original and technically so as to serve both the developing world and developed countries in the best possible
OMICS Journals are welcoming Submissions OMICS International welcomes submissions that are original and technically so as to serve both the developing world and developed countries in the best possible
Annexin V-PE Apoptosis Detection Kit
 Annexin V-PE Apoptosis Detection Kit Catalog Number KA0716 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
Annexin V-PE Apoptosis Detection Kit Catalog Number KA0716 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
CONTRACTING ORGANIZATION: Vanderbilt University Medical Center Nashville, TN
 AD Award Number: W81XWH-04-1-0867 TITLE: A Myc-Driven in Vivo Model of Human Prostate Cancer PRINCIPAL INVESTIGATOR: Simon W. Hayward, Ph.D. CONTRACTING ORGANIZATION: Vanderbilt University Medical Center
AD Award Number: W81XWH-04-1-0867 TITLE: A Myc-Driven in Vivo Model of Human Prostate Cancer PRINCIPAL INVESTIGATOR: Simon W. Hayward, Ph.D. CONTRACTING ORGANIZATION: Vanderbilt University Medical Center
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
