Information for Healthcare Professionals and other Stakeholders
|
|
|
- Howard Chambers
- 5 years ago
- Views:
Transcription
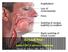
1 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014 Any significant information (eg publication of results of a new study, results of the investigation of a pharmacovigilance case) may cause the issue of a new version of this document. What is Nephrogenic Systemic Fibrosis (NSF)? NSF was first recognized in 1997 in 15 dialyzed patients and described in 2000 [1]. This rare and highly debilitating disorder is characterized by extensive thickening and hardening of the skin associated with skin-colored to erythematous papules that coalesce into erythematous to brawny plaques with a peau d orange appearance. Nodules are sometimes also described. Joint contractures may develop, with patients progressively becoming wheelchair-dependent. Patients often complain of pruritus, causalgia and sharp pains [2]. The distal extremities are the most common area of involvement (with a distribution from ankles to mid-thighs and from wrists to mid-upper arms), followed by the trunk. The lesions are typically symmetrical. It is worth noting that the face and neck are virtually never involved [3]. Markedly reduced renal function (egfr < 30 ml/mn/1.73 m2) is the most important patient-related risk factor for NSF and has been present in almost all reported cases of NSF, and many patients have been on haemodialysis or peritoneal dialysis [4]. NSF can occur in all age-groups and there is no predilection for a geographic region, race or gender. P DOT 1
2 So far, there is no recognized treatment for NSF. It has been suggested that improving renal function may slow down the development of the disease and, in some cases, may reverse its course [2]. The mechanism of this highly debilitating disease remains unknown. Current literature suggests a multifactorial aetiology [5-7]. So far, several factors have been suggested to be frequently associated with the onset of NSF (Table 1): Table 1 Recognized or possibly associated factors for NSF Severe or end-stage renal failure [1-3] Acute kidney insufficiency [4] High cumulative dose of gadolinium chelates [8] High dose of erythropoietin [8, 9] High serum phosphate levels [8, 10, 11] High serum calcium levels [8] Coexisting pro inflammatory event (major surgery, infection, vascular event) [12-14] Iron mobilization [5, 15] Metabolic acidosis [16] (debated: [12, 17]) History of hypothyroidism [18] History of deep venous thrombosis [18-20] Younger age [18] In 2006, two European teams independently suggested a link between the administration of gadolinium (Gd) chelates used as contrast media for magnetic resonance imaging (MRI) P DOT 2
3 and the occurrence of NSF in patients with renal failure [16, 17]. Numerous retrospective analyses rapidly followed and confirmed this temporal link [12, 21, 22]. The time to onset of the symptoms ranges between a few days and a few years following exposure to the gadolinium chelate [4]. The majority of published cases occurred within 2 weeks to 2 months [23]. The link between gadolinium-containing contrast media and NSF is considered probable [16, 17, 24-26]. Awareness of this potential adverse reaction to gadolinium chelates is a major requirement for radiologists and specialists in patients with stage 4 and 5 chronic kidney disease and acute kidney insufficiency who are at higher risk for NSF. In addition, patients with stage 3 chronic kidney disease are at lower risk for NSF [4]. Position of European Health Authorities In February 2007, the European Pharmacovigilance Working Party (PhVWP) of the European Medicines Agency (EMA) initially advised all contrast media marketing authorization holders to add warnings about the possibility that NSF may occur with gadolinium chelates to section 4.4 of the Summary of Product Characteristics (SPC). There are two generally recognized categories of gadolinium chelates: macrocyclic molecules where Gd 3+ is caged in the pre-organised cavity of the ligand (15), and the linear open chain molecules (15). Gadolinium chelates differ in their thermodynamic stability constants and in their kinetic stability. In general, pre-clinical and ex vivo studies have suggested that macrocyclic molecules are more stable than linear molecules (17-21). In December 2007, the Scientific Advisory Group (SAG) for Diagnostics of the CHMP (Committee for Medicinal Products for Human Use) agreed with the PhVWP that the risk of developing NSF depends on the type of gadolinium chelates used, and advised that these agents should be categorized into three groups: High risk: gadoversetamide (OptiMARK ), gadodiamide (Omniscan ) and gadopentetic acid (Magnevist, Magnegita, and Gado-MRT-ratiopharm ); P DOT 3
4 Medium risk: gadofosveset (Vasovist ), gadoxetic acid (Primovist ) and gadobenic acid (MultiHance ); Low risk: gadoteric acid (Dotarem ), gadoteridol (ProHance ) and gadobutrol (Gadovist ). In November 2008 a referral procedure was triggered in order for the CHMP to carry out an assessment of the risk of NSF for the authorized gadolinium chelates, and recommended measures that could be taken to reduce this risk. The final CHMP opinion was issued in November 2009, and was ratified by an European Commission decision in July The conclusions confirmed the classification of the active substances into the three categories of risk, and the CHMP emitted recommendations for the labelling of the different agents to vary according to their risk classification (Table 2). The CHMP recommended also that the prescribing information of all gadolinium chelates should include a statement that the type and dose of contrast agent used should be recorded. In that view, sticky labels removable from the vials and syringes have to be implemented. Finally, the CHMP advised that studies should be performed to evaluate the potential of long-term retention of gadolinium in bone. In addition, MAHs should continue to submit annual cumulative reviews of NSF cases for coordinated assessment until the final study report of the study evaluating gadolinium accumulation in bone and its long-term effect, have been submitted. The evaluation by the CHMP of the 2013 cumulative review of the cases regarding gadoteric acid (Dotarem ) maintained the classification of the product in the low risk class, and endorsed the causality assessment of the reported cases as provided by Guerbet. P DOT 4
5 Table 2 Revised contra-indications and precautions for use of gadolinium-containing contrast agents (CHMP 2010) Risk class LOW RISK: MEDIUM RISK: HIGH RISK: Dotarem, ProHance, Gadovist Primovist, Vasovist, MultiHance Omniscan, Optimark, Magnevist Pregnancy Not recommended, unless benefit risk ratio assessed as favorable Lactation Continuation or suspension 24h according to mother s decision (in consultation with the physician) Discontinuation at least 24h Renal insufficiency (RI), hepatic Precaution in severe Renal insufficiency To be avoided in severe RI and hepatic Contra-indication in severe RI and hepatic transplanted transplantation, dialysis (RI) and hepatic transplanted patients: transplanted patients patients minimum diagnostic dose and minimum 7 If used, minimum diagnostic dose and Precaution in moderate RI patients, according to days between administrations minimum 7 days between benefit risk ratio, minimum diagnostic dose and administrations minimum 7 days between administrations No evidence supporting the use of haemodialysis for preventing or treating NSF in non-dialyzed patients, may be useful in dialyzed patients Paediatric population Precaution in neonate, minimum diagnostic dose and minimum 7 days between administrations* Contra-indication in neonate < 4 weeks Precaution in child < 1 year, minimum diagnostic dose and minimum 7 days between administrations Elderly patient Important to screen patients > 65 years for renal dysfunction Screening of renal function Recommended laboratory test to screen patients for renal dysfunction Mandatory laboratory test to screen all patients for renal dysfunction *In the low and medium risk categories of NSF, DOTAREM provides the largest range of indications for newborns and infants ranging from 0 to 2 years old, for MRI of whole body pathologies as well as for MRI of the brain and spinal diseases and diseases of the vertebral column**.dotarem is not recommended for MR angiography in children under 18 years of age due to insufficient data on the efficacy and safety in this indication. **For more information and because indications may vary from country to country, please refer to your mandatory local SPC. P DOT 5
6 Position of US Health Authorities (FDA) In 2006, the Food and Drug Administration (FDA) alerted the public about cases of NSF reported in patients who received gadolinium chelates. In 2007, the FDA required the addition of a boxed warning about the risk of NSF to the labelling of gadolinium chelates. In December 2009, the safety of gadolinium chelates was reviewed during a Joint Cardiovascular and Renal Drugs and Drug Safety and Risk Management Advisory Committee meeting. Following that, in September 2010, the FDA made a Safety Announcement, requiring new changes in the labelling of gadolinium chelates, falling more closely into line with those requested by European Health Authorities. High-risk gadolinium chelates (Optimark, Omniscan, Magnevist ) are contraindicated in patients with acute kidney injury (AKI) or with chronic severe kidney disease. In addition, the FDA recommended a screening of patients for kidney problems using clinical history and laboratory testing. The use of gadolinium chelates should be avoided in patients having impaired drug elimination, unless this use is necessary; in that case, signs and symptoms of NSF should be monitored after administration. Administration of gadolinium-containing contrast agent should not be repeated in a single imaging session. Dotarem has had a Marketing Authorization in the USA since March 21, Position of the Danish Health and Medicines Authority On 18 September 2009, the Danish Health and Medicines Authority published guidelines for examination by MRI scan of kidney impaired patients using gadolinium-containing P DOT 6
7 contrast agents. On 21 November 2013, a working group established an update of these guidelines aligned with the EMA assessment (July 2010) and even reinforced some particular points listed below: The use of high risk preparations should be avoided in all contexts since usable low risk preparations are available. Medium risk preparations should be used only in case of compelling indications for patients with an egfr below 59 ml/min/1.73 m². Compelling indication means that the diagnostic information is essential and cannot be obtained by non-contrast enhanced scanning or with the use of a low risk preparation. For children 2-18 years, use of MultiHance is not recommended whatever the status of the renal function of the subject is. In contrast, low-risk products can be used without restriction if renal function exceeds 60 ml/min/1.73m². They should only be used for essential diagnostic information in case of egfr< 60 ml/min/1.73m². For children from 0 to 2 years old, the Danish guidelines recommend using Dotarem only. Although Dotarem does not have MR angiography as an approved indication for patients aged less than 18, if an MR angiography must be performed on a compelling indication, the Danish guidelines recommend to use Dotarem in a single dose not exceeding 0.1 mmol/kg. Position of the European Society of Urogenital Radiology (ESUR): Based on a review of the literature, the ESUR s Contrast Media Safety Committee published in 2013 an update of the guidelines on NSF and gadolinium chelates first established in The recommendations are summarized in the table 3: P DOT 7
8 Table 3 Recommendations for use of gadolinium-containing contrast agents (ESUR 2013) Risk class LOW RISK: MEDIUM RISK: HIGH RISK: Dotarem, ProHance, Gadovist Primovist, Vasovist, Ablavar Omniscan, Optimark, Magnevist MultiHance, Eovist Pregnancy Can be used to give essential diagnostic information Contra-indication in pregnant women Lactation Continuation or suspension 24h according to mother s decision (in consultation with the physician) Discontinuation at least 24h Renal insufficiency (RI), hepatic transplantation, dialysis Should be used with caution in patients with Chronic Kidney Disease (CDK) 4 and 5 (GFR<30mL/min), including patients on dialysis, with at least 7 days between 2 administrations. Contra-indication in patients with CDK 4 and 5 (GFR<30mL/min) and with acute renal insufficiency. Should be used with caution in patients with CDK 3 (GFR ml/min, according to benefit risk ratio, minimum diagnostic dose and minimum 7 days between administrations. No evidence supporting the use of haemodialysis for preventing or treating NSF in non-dialyzed patients, But may be useful in dialyzed patients. Paediatric population Precaution in neonate, minimum diagnostic dose and minimum 7 days between administrations Contra-indication in neonate < 4 weeks Precaution in child < 1 year, minimum diagnostic dose and minimum 7 days between administrations Patients with NSF GBCA should only be used if the indication is vital Contra-indication in patients with NSF Screening of renal function Laboratory testing of renal function (egfr) is not mandatory but renal function assessment by questionnaire should be used if serum creatinine is not measured. Mandatory laboratory test to screen all patients for renal dysfunction All patients Use the smallest amount of contrast medium necessary for a diagnostic result. Should never be given in higher doses than 0.1mmol/kg per examination Always record the name and dose of the contrast agent used in the patient records. P DOT 8
9 Guerbet Pharmacovigilance Data Guerbet markets meglumine gadoterate (Dotarem ), a macrocyclic, ionic gadolinium chelate associated with a high thermodynamic and kinetic stabilities [25-29]. The Department of International Pharmacovigilance of Guerbet manages the Adverse Events reported in a context of Dotarem administration in accordance with the regulatory Pharmacovigilance reporting standards; in particular Guideline on Good Pharmacovigilance Practices (GVP) - European Medicines Agency and Heads of Medicines Agencies, For each case reported as NSF and based on the available information, Guerbet has assessed the strength of the diagnosis of NSF and the causality of Dotarem : - the strength of the diagnosis of NSF using the clinicopathological score developed by Yale University (USA) and published by Girardi et al [30] on the basis of their registry of more than 250 cases, and discussing the differential diagnoses of the disease ; - Dotarem causality considering the available and missing information regarding the medical history of the patient, the injection of the sole Dotarem (single-agent case) or other gadolinium chelates (multiple-agent case), the chronology of both the injections and the clinical, biological and imaging events, other possible causative factors such as pro-inflammatory events. With respect to NSF, Guerbet has registered, to date, 19 medically-confirmed cases of patients who developed signs allowing this diagnosis to be considered and who received Dotarem, for more than 43 million of patients who received the product, and more than 850 cases of NSF reported worldwide (source European Medicines Agency, 2010). On the basis of the available information, the diagnosis of NSF is confirmed or consistent according to the Girardi score in only 1/3 of the reported cases, and the causality of Dotarem is unlikely in all cases (Table 4). In all cases the administration of Dotarem preceded the first symptoms, except for two patients who developed NSF after injection of linear gadolinium chelates and prior to Dotarem administration. In both cases, the causality of Dotarem in the disease worsening was assessed as unlikely. P DOT 9
10 Table 4 NSF cases reported for patients having received Dotarem More than 43 million doses sold 19 reported cases of NSF (all sources i.e. health care professionals, authorities, literature) 8 confirmed or very likely cases of NSF Dia Confirmed or consistent diagnosis (Girardi score), information sufficient to rule out the differential diagnoses 11 unconfirmed or doubtful cases of NSF Due to missing information the Girardi score cannot be applied and/or the differential diagnoses cannot be ruled out 0 Single-agent case 8 Multipleagent cases 0 Non qualifiable case* 1 Singleagent case 9 Multipleagent cases 1 Non qualifiable case* *Non qualifiable = unspecified agent received in addition to Dotarem Position Statement At Guerbet, we significantly contribute to improving diagnosis for major disease areas (cardiovascular diseases, cancer, inflammatory and neurodegenerative diseases). We are strongly committed to providing radiologists, cardiologists and healthcare professionals with a comprehensive range of innovative and effective contrast media to achieve their aim to provide optimum diagnosis for their patients. A complete research programme is in progress at Guerbet and in cooperation with recognized academic centres to better understand the mechanism of NSF and thoroughly study the role of physicochemical properties of gadolinium chelates in its pathogenesis. The research programme includes a prospective clinical analysis of the safety of Dotarem. A full collaboration in total transparency with Health Authorities for Pharmacovigilance issues and consistently acting in the best interests of the patients are fundamental principles at Guerbet. This is particularly true in the case of NSF and will remain so. P DOT 10
11 Further information about NSF and gadolinium chelates European Medicines Agency (EMA) US Food and Drug Administration (FDA) European Society of Urogenital Radiology (ESUR) International Center for Nephrogenic Fibrosing Dermopathy Research (ICNFDR) (Yale University) American College of Radiology (ACR) References 1. Cowper, S.E., et al., Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet, (9234): p Cowper, S.E., P.H. Kuo, and R. Bucala, Nephrogenic systemic fibrosis and gadolinium exposure: association and lessons for idiopathic fibrosing disorders. Arthritis Rheum, (10): p Galan, A., S.E. Cowper, and R. Bucala, Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol, (6): p Thomsen, H.S., et al., Nephrogenic systemic fibrosis and gadolinium-based contrast media: updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol. 23(2): p Swaminathan, S. and S.V. Shah, New insights into nephrogenic systemic fibrosis. J Am Soc Nephrol, (10): p Nardone Beatrice, L.A.E., Bohaty Benjamin, West Dennis P., Holzem Kassandra E, Bauer Kimberly Clinical factors associated with nephrogenic systemic fibrosis (NSF): A single site academic center, 2-cohort study. Journal of the American Academy of Dermatology, (4): p. AB Chopra, T., et al., Understanding nephrogenic systemic fibrosis. Int J Nephrol. 2012: p Marckmann, P., et al., Case-control study of gadodiamide-related nephrogenic systemic fibrosis. Nephrol Dial Transplant, (11): p Swaminathan, S., et al., Nephrogenic fibrosing dermopathy and high-dose erythropoietin therapy. Ann Intern Med, (3): p P DOT 11
12 10. Fretellier, N., et al., Hyperphosphataemia sensitizes renally impaired rats to the profibrotic effects of gadodiamide. Br J Pharmacol. 165(4b): p Frenzel, T., et al., Stability of gadolinium-based magnetic resonance imaging contrast agents in human serum at 37 degrees C. Invest Radiol, (12): p Sadowski, E.A., et al., Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology, (1): p Golding, L.P. and J.M. Provenzale, Nephrogenic systemic fibrosis: possible association with a predisposing infection. AJR Am J Roentgenol, (4): p Perazella, M.A., Nephrogenic systemic fibrosis, kidney disease, and gadolinium: is there a link? Clin J Am Soc Nephrol, (2): p Miyamoto, J., et al., Detection of iron deposition in dermal fibrocytes is a useful tool for histologic diagnosis of nephrogenic systemic fibrosis. Am J Dermatopathol. 33(3): p Grobner, T., Gadolinium--a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis? Nephrol Dial Transplant, (4): p Marckmann, P., et al., Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol, (9): p Kallen, A.J., et al., Gadolinium-containing magnetic resonance imaging contrast and nephrogenic systemic fibrosis: a case-control study. Am J Kidney Dis, (6): p Introcaso, C.E., et al., Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis: a case series of nine patients and review of the literature. Int J Dermatol, (5): p Cowper, S.E., Nephrogenic fibrosing dermopathy: the first 6 years. Curr Opin Rheumatol, (6): p Broome, D.R., et al., Gadodiamide-associated nephrogenic systemic fibrosis: why radiologists should be concerned. AJR Am J Roentgenol, (2): p Khurana, A., et al., Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (omniscan). Invest Radiol, (2): p Marckmann, P., An epidemic outbreak of nephrogenic systemic fibrosis in a Danish hospital. Eur J Radiol, (2): p Abraham, J.L. and C. Thakral, Tissue distribution and kinetics of gadolinium and nephrogenic systemic fibrosis. Eur J Radiol, (2): p Morcos, S.K., Extracellular gadolinium contrast agents: differences in stability. Eur J Radiol, (2): p Perazella, M.A. and R.A. Rodby, Gadolinium use in patients with kidney disease: a cause for concern. Semin Dial, (3): p Caravan, P., et al., Gadolinium(III) Chelates as MRI Contrast Agents: Structure, Dynamics, and Applications. Chem Rev, (9): p Laurent, S., et al., Stability of MRI paramagnetic contrast media: a proton relaxometric protocol for transmetallation assessment. Invest Radiol, (2): p Wedeking, P., K. Kumar, and M.F. Tweedle, Dissociation of gadolinium chelates in mice: relationship to chemical characteristics. Magn Reson Imaging, (4): p Girardi, M., et al., Nephrogenic systemic fibrosis: clinicopathological definition and workup recommendations. J Am Acad Dermatol. 65(6): p e7. P DOT 12
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Global reports and findings of NSF and its effects
 Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018)
 Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
A Safety Update on the Gadolinium Chelates
 Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Gadolinium-Based Contrast Media
 Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
Seeing is Believing... Look Beneath the Surface
 References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
Gadolinium-Based Contrast Agents for Magnetic Resonance Imaging (marketed as Magnevist, MultiHance, Omniscan, OptiMARK, ProHance)
 Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research
 University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, 2 july 2008
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, 2 july 2008
Public Assessment Report
 Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
Administration of Gadolinium Contrast in Adults Procedural Guideline
 Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
NSF Coming and Going
 NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
MR Contrast Agents. Why Use Contrast Agents in MRI? Why Use Contrast Agents in MRI? US Agents. Understanding and Embracing Change
 Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
INTRODUCTION CLINICAL EXPERIENCE CLINICAL EFFICACY PEDIATRIC EXPERIENCE DOSING SUMMARY
 1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
Protocol for iv. iodine and gadolinium contrast studies
 Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Please see Important Safety Information, including Boxed Warnings for Gadavist and Magnevist below.
 August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Please see Important Safety Information, including Boxed Warnings for Gadavist, Eovist and Magnevist on the following pages.
 Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents
 Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
FDA Advisory Committee Briefing Document. Medical Imaging Drugs Advisory Committee. Meeting to be held on February 14, 2013
 FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
Beyond NSF: Acute GBCA adverse reactions
 Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
PLEASE SCROLL DOWN FOR ARTICLE
 This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS
 DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
Annex II. Scientific conclusions
 Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
William G. Bradley, Jr, MD, PhD, FACR
 William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
Extracellular gadolinium-based contrast media: An overview
 European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
Annex III. Amendments to relevant sections of the product information
 Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting
 European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
PATIENT CARE AND MRI SAFETY Module 7
 PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
Figure 1 Figure 1: Energy diagram illustrates formation of a chelate (ML) created by combining two chemicals free Gd 3 (M), which is a metal ion, and
 Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance
 Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
Contrast-enhanced MRI: how do changing EU regulations impact daily practice?
 Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance
 Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA.
 ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
Report on the Investigation Results
 This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
PATIENT INFORMATION LEAFLET. DOTAREM mg/ml, solution for injection (Gadoteric acid)
 PATIENT INFORMATION LEAFLET DOTAREM 279.32 mg/ml, solution for injection (Gadoteric acid) Read all this leaflet carefully before you start using this medicine because it contains important information
PATIENT INFORMATION LEAFLET DOTAREM 279.32 mg/ml, solution for injection (Gadoteric acid) Read all this leaflet carefully before you start using this medicine because it contains important information
PRODUCT MONOGRAPH DOTAREM. (gadoterate meglumine) Injection. For Intravenous Use
 PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Side Effects and Complications of Magnetic Resonance Contrast Media
 HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
DOTAREM (gadoterate meglumine) Injection for intravenous use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be used
 Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Gadolinium and nephrogenic systemic fibrosis
 mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
IL PROBLEMA DEL GADOLINIO OGGI
 11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
This copy is for personal use only. To order printed copies, contact Purpose: Materials and Methods: Results: Conclusion:
 This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
PRAC recommendations on signals for update of the product information
 Thursday 22 January 2015 EMA/PRAC/62352/2015 Pharmacovigilance Risk Assessment Committee PRAC recommendations on signals for update of the product information Adopted at the 6-9 January 2015 PRAC 1. Atorvastatin,
Thursday 22 January 2015 EMA/PRAC/62352/2015 Pharmacovigilance Risk Assessment Committee PRAC recommendations on signals for update of the product information Adopted at the 6-9 January 2015 PRAC 1. Atorvastatin,
Gadolinium in Humans: A Family of Disorders
 Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
Poster No.: B-229 Congress: ECR 2011 Scientific Paper. Authors: E. De Kerviler 1, S. Gaillard 2 ; 1 Paris/FR, 2 Villepinte/FR
 Pharmacovigilance based on spontaneous adverse event reporting of meglumine gadoterate (Gd-DOTA) after 15 million administrations and 20 years of clinical use Poster No.: B-229 Congress: ECR 2011 Type:
Pharmacovigilance based on spontaneous adverse event reporting of meglumine gadoterate (Gd-DOTA) after 15 million administrations and 20 years of clinical use Poster No.: B-229 Congress: ECR 2011 Type:
Update. Drug Safety. Welcome to the first issue of the newly launched Drug Safety Update. This
 Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
Special Articles Original Research
 Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
Gadolinium Use in Patients with Kidney Disease: A Cause for Concern
 Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
Nephrogenic Systemic Fibrosis: Possible Association with a Predisposing Infection
 MR Imaging Original Research MR Imaging Original Research Lauren Parks Golding 1,2 James M. Provenzale 3 Golding LP, Provenzale JM Keywords: connective tissue disorders, contrast media, dialysis, infections,
MR Imaging Original Research MR Imaging Original Research Lauren Parks Golding 1,2 James M. Provenzale 3 Golding LP, Provenzale JM Keywords: connective tissue disorders, contrast media, dialysis, infections,
1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION. 1.1 Central Nervous System. 1.2 Extracranial/Extraspinal Tissues. 1.3 Body
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Magnetic resonance imaging (MRI) scans are significantly
 Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Imaging in the time of NFD/NSF: do we have to change our routines concerning renal insufficiency?
 DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
Mechanism of NSF: New evidence challenging the prevailing theory
 Thomas Jefferson University Jefferson Digital Commons Scleroderma Center Faculty Papers Scleroderma Center of Thomas Jefferson University 11-2009 Mechanism of NSF: New evidence challenging the prevailing
Thomas Jefferson University Jefferson Digital Commons Scleroderma Center Faculty Papers Scleroderma Center of Thomas Jefferson University 11-2009 Mechanism of NSF: New evidence challenging the prevailing
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine
 Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
Summary. Correspondence Jerrold L. Abraham. Accepted for publication 17 September 2007
 CLINICAL AND LABORATORY INVESTIGATIONS DOI 10.1111/j.1365-2133.2007.08335.x Dermal inorganic gadolinium concentrations: evidence for in vivo transmetallation and long-term persistence in nephrogenic systemic
CLINICAL AND LABORATORY INVESTIGATIONS DOI 10.1111/j.1365-2133.2007.08335.x Dermal inorganic gadolinium concentrations: evidence for in vivo transmetallation and long-term persistence in nephrogenic systemic
Instructions for the safe administration of gadolinium based contrast agents (GBCA). 1. PREADMINISTRATION PRECAUTIONS:
 POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
Incidence of Immediate Gadolinium Contrast Media Reactions
 Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
During the past decade, nephrogenic systemic fibrosis
 Low Risk for Nephrogenic Systemic Fibrosis in Nondialysis Patients Who Have Chronic Kidney Disease and Are Investigated with Gadolinium-Enhanced Magnetic Resonance Imaging Constantina Chrysochou,* Albert
Low Risk for Nephrogenic Systemic Fibrosis in Nondialysis Patients Who Have Chronic Kidney Disease and Are Investigated with Gadolinium-Enhanced Magnetic Resonance Imaging Constantina Chrysochou,* Albert
CONTRAINDICATIONS History of severe hypersensitivity reaction to EOVIST (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
Gadolinium Deposition:
 Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Safety of Gadoterate Meglumine (Gd-DOTA) as a Contrast Agent for Magnetic Resonance Imaging
 ORIGINAL RESEARCH ARTICLE Drugs R D 2010; 10 (3): 133-145 1179-6901/10/0003-0133 ª 2010 Ishiguchi & Takahashi, publisher and licensee Adis Data Information BV. This is an open access article published
ORIGINAL RESEARCH ARTICLE Drugs R D 2010; 10 (3): 133-145 1179-6901/10/0003-0133 ª 2010 Ishiguchi & Takahashi, publisher and licensee Adis Data Information BV. This is an open access article published
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM. Gadoterate meglumine injection. (376.9 mg/ml, equivalent to 0.
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report
 JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report Arpita Aggarwal
JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report Arpita Aggarwal
Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended.
 Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended Omniscan UK/W/063/pdWS/001 Rapporteur UK Finalisation procedure (day 20)
Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended Omniscan UK/W/063/pdWS/001 Rapporteur UK Finalisation procedure (day 20)
Policy #: 291 Latest Review Date: February 2013
 Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric patients. The dose is delivered by manual or power injection.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
CONTRAINDICATIONS Clinically important hypersensitivity reactions to DOTAREM. (4)
 M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Other EU Activities Contributing to Harmonization of Labeling
 Other EU Activities Contributing to Harmonization of Labeling Dr Laurent Brassart European Medicines Agency Medical Information Sector DIA Labeling Harmonisation 2011 Workshop October 13-14. 2011 Disclaimer
Other EU Activities Contributing to Harmonization of Labeling Dr Laurent Brassart European Medicines Agency Medical Information Sector DIA Labeling Harmonisation 2011 Workshop October 13-14. 2011 Disclaimer
Have you seen a patient like Carol *?
 (linagliptin) 5mg tablets Have you seen a patient like Carol *? *Hypothetical patient profile Carol * : 70 years old Retired schoolteacher *Hypothetical patient profile CAROL*: T2D patient with moderate
(linagliptin) 5mg tablets Have you seen a patient like Carol *? *Hypothetical patient profile Carol * : 70 years old Retired schoolteacher *Hypothetical patient profile CAROL*: T2D patient with moderate
Have you seen a patient like Elaine *?
 (linagliptin) 5mg tablets Have you seen a patient like Elaine *? *Hypothetical patient profile Elaine * : 60 years old Housewife *Hypothetical patient profile ELAINE*: T2D Patient with early signs of kidney
(linagliptin) 5mg tablets Have you seen a patient like Elaine *? *Hypothetical patient profile Elaine * : 60 years old Housewife *Hypothetical patient profile ELAINE*: T2D Patient with early signs of kidney
PRODUCT MONOGRAPH PRIMOVIST. gadoxetate disodium injection mg/ml (0.25 mmol/ml)
 PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
Radiology Reimbursement Information Setting: Hospital Outpatient
 Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
Läkemedelsverkets farmakovigilansdag
 PRAC : Pharmacovigilance Risk Assessment Committee Kommittén för säkerhetsövervakning & riskbedömning av läkemedel Farmakovigilansdagen maj 2017 Ulla Wändel Liminga, PhD Ämnesområdesansvarig farmakologi/toxikologi;
PRAC : Pharmacovigilance Risk Assessment Committee Kommittén för säkerhetsövervakning & riskbedömning av läkemedel Farmakovigilansdagen maj 2017 Ulla Wändel Liminga, PhD Ämnesområdesansvarig farmakologi/toxikologi;
ARTICLE IN PRESS European Journal of Radiology xxx (2011) xxx xxx
 European Journal of Radiology xxx (2011) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Tolerability and diagnostic value
European Journal of Radiology xxx (2011) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Tolerability and diagnostic value
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
Public Assessment Report for paediatric studies submitted in accordance with Article 45 and 46 of Regulation (EC) No1901/2006, as amended
 Public Assessment Report for paediatric studies submitted in accordance with Article 45 and 46 of Regulation (EC) No1901/2006, as amended Gadopentetic acid (Gadopentate) Magnevist UK/W/049/pdWS/001 Rapporteur:
Public Assessment Report for paediatric studies submitted in accordance with Article 45 and 46 of Regulation (EC) No1901/2006, as amended Gadopentetic acid (Gadopentate) Magnevist UK/W/049/pdWS/001 Rapporteur:
Frequency and Severity of Acute Allergic-Like Reactions to Gadolinium-Containing IV Contrast Media in Children and Adults
 Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Hepatobiliary Contrast Agents for Liver MRI
 Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Gadolinium a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?
 Nephrol Dial Transplant (2006) 21: 1104 1108 doi:10.1093/ndt/gfk062 Advance Access publication 23 January 2006 Interesting Case Gadolinium a specific trigger for the development of nephrogenic fibrosing
Nephrol Dial Transplant (2006) 21: 1104 1108 doi:10.1093/ndt/gfk062 Advance Access publication 23 January 2006 Interesting Case Gadolinium a specific trigger for the development of nephrogenic fibrosing
Chapter 9 A 65-Year-Old Woman with Hardened Skin, Joint Contractures, and Yellow Scleral Plaques
 Chapter 9 A 65-Year-Old Woman with Hardened Skin, Joint Contractures, and Yellow Scleral Plaques Jonathan Kay Keywords Nephrogenic systemic fibrosis Chronic kidney disease Contrast media Gadolinium Gadolinium
Chapter 9 A 65-Year-Old Woman with Hardened Skin, Joint Contractures, and Yellow Scleral Plaques Jonathan Kay Keywords Nephrogenic systemic fibrosis Chronic kidney disease Contrast media Gadolinium Gadolinium
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
ASKLEPIOS Course. July 20 21, 2018 Bangkok/Thailand
 This ASKLEPIOS course is implemented with great support and partnership of Guerbet and RCRT Education in partnership Course information This ESOR course aims to discuss the most important topics in neuroradiology.
This ASKLEPIOS course is implemented with great support and partnership of Guerbet and RCRT Education in partnership Course information This ESOR course aims to discuss the most important topics in neuroradiology.
Scientific conclusions
 Annex II Scientific conclusions and grounds for amendment of the summary of product characteristics, labelling and package leaflet presented by the European Medicines Agency 24 Scientific conclusions Overall
Annex II Scientific conclusions and grounds for amendment of the summary of product characteristics, labelling and package leaflet presented by the European Medicines Agency 24 Scientific conclusions Overall
DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC
 DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively.
DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively.
Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease
 International Nephrology, Article ID 679605, 6 pages http://dx.doi.org/10.1155/2014/679605 Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease Robert F. Hanna, 1 Lee A. Finkelstone, 1 Daniel
International Nephrology, Article ID 679605, 6 pages http://dx.doi.org/10.1155/2014/679605 Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease Robert F. Hanna, 1 Lee A. Finkelstone, 1 Daniel
Pro-enkephalin and Renal Disease. Alan H. Wu, Ph.D. University of California, San Francisco 12 th Annual UCSD Biomarkers of Cardiac Disease
 Pro-enkephalin and Renal Disease Alan H. Wu, Ph.D. University of California, San Francisco 12 th Annual UCSD Biomarkers of Cardiac Disease The presenter has received relevant grants and /or consulting
Pro-enkephalin and Renal Disease Alan H. Wu, Ph.D. University of California, San Francisco 12 th Annual UCSD Biomarkers of Cardiac Disease The presenter has received relevant grants and /or consulting
NEWSLETTER October The Pharmacovigilance Risk Assessment Committee July (PRAC) Meeting
 The Pharmacovigilance Risk Assessment Committee (PRAC) Meeting 7-10 July 2014 Finalised safety procedure for methadone products containing povidone Only high molecular weight povidone was associated with
The Pharmacovigilance Risk Assessment Committee (PRAC) Meeting 7-10 July 2014 Finalised safety procedure for methadone products containing povidone Only high molecular weight povidone was associated with
Contrast controversies and confusion
 Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents in Human Whole Blood at 1.5, 3, and 7 T
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Annex IV Scientific conclusions
 Annex IV Scientific conclusions 56 Scientific conclusions On 7 June 2017, the European Commission (EC) was informed of a fatal case of fulminant liver failure in a patient treated with daclizumab in an
Annex IV Scientific conclusions 56 Scientific conclusions On 7 June 2017, the European Commission (EC) was informed of a fatal case of fulminant liver failure in a patient treated with daclizumab in an
PRAC recommendations on signals
 5 February 2015 EMA/PRAC/734433/2014 Corr. 3 Pharmacovigilance Risk Assessment Committee Adopted at the PRAC meeting of 6-9 January 2015 This document provides an overview of the recommendations adopted
5 February 2015 EMA/PRAC/734433/2014 Corr. 3 Pharmacovigilance Risk Assessment Committee Adopted at the PRAC meeting of 6-9 January 2015 This document provides an overview of the recommendations adopted
October 23, Re: Gadolinium Toxicity from GBCAs. Dear Dr. Hamburg:
 October 23, 2012 Commissioner Margaret A. Hamburg, M.D. U.S. Food and Drug Administration 10903 New Hampshire Avenue WO Bldg. 1, Room 2217 Silver Springs, MD 20993 Re: Gadolinium Toxicity from GBCAs Dear
October 23, 2012 Commissioner Margaret A. Hamburg, M.D. U.S. Food and Drug Administration 10903 New Hampshire Avenue WO Bldg. 1, Room 2217 Silver Springs, MD 20993 Re: Gadolinium Toxicity from GBCAs Dear
Nephrogenic Systemic Fibrosis in Denmark A Nationwide Investigation
 A Nationwide Investigation Tina R. Elmholdt 1,2 *, Anne B. B. Olesen 2, Bettina Jørgensen 3, Stinne Kvist 3, Lone Skov 4, Henrik S. Thomsen 5, Peter Marckmann 6,7, Michael Pedersen 1,8 1 Institute of Clinical
A Nationwide Investigation Tina R. Elmholdt 1,2 *, Anne B. B. Olesen 2, Bettina Jørgensen 3, Stinne Kvist 3, Lone Skov 4, Henrik S. Thomsen 5, Peter Marckmann 6,7, Michael Pedersen 1,8 1 Institute of Clinical
Radiology Update 2017
 Radiology Update 2017 John K. Phillips, MD Affiliated Assistant Professor of Radiology University of Tennessee Health Sciences Center Chief, Radiology and Nuclear Medicine VA Memphis Disclosures Financial:
Radiology Update 2017 John K. Phillips, MD Affiliated Assistant Professor of Radiology University of Tennessee Health Sciences Center Chief, Radiology and Nuclear Medicine VA Memphis Disclosures Financial:
