Gadolinium-Based Contrast Media
|
|
|
- Linda Bishop
- 5 years ago
- Views:
Transcription
1 Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class of coordination or complex compounds consisting of a central metal atom attached to a large molecule, called a ligand Gd +3
2 Purpose Shorten the T1- and T2-relaxation rates of water-based hydrogen protons Water Molecule Gadolinium Agent Molecular tumbling-rate slows T1-relaxation time shortens 1 T1 = r1 [Gd] Relaxivity (r 1) is a measure of the effect on the T1-relaxation rate
3 US Agents Generic Name Gadopentetate dimeglumine Gadoteridol Gadodiamide Gadoversetamide Gadobutrol Gadoterate meglumine Gadobenate dimeglumine Gadoxetate disodium Trade Name Magnevist ProHance Omniscan OptiMARK Gadavist Dotarem MultiHance Eovist Dose Generic Name Trade Name Dose Gadopentetate dimeglumine Gadoteridol Gadodiamide Gadoversetamide Gadobutrol Gadoterate meglumine Magnevist ProHance Omniscan OptiMARK Gadavist Dotarem 0.1 mmol/kg 0.1 mmol/kg 0.1 mmol/kg 0.1 mmol/kg 0.1 mmol/kg 0.1 mmol/kg Gadobenate dimeglumine MultiHance 0.1 mmol/kg Molar Concentration Generic Name Trade Name Concentration Gadopentetate dimeglumine Gadoteridol Gadodiamide Gadoversetamide Gadobutrol Gadoterate meglumine Magnevist ProHance Omniscan OptiMARK Gadavist Dotarem 0.5 molar 0.5 molar 0.5 molar 0.5 molar 1.0 molar 0.5 molar Gadobenate dimeglumine MultiHance 0.5 molar
4 Calculating Volume Delivered Dose is determined by amount of gadolinium not volume of the agent Dose: 0.1 mmol/kg 80 kg patient 0.5 molar agent 1.0 molar agent (80 x 0.1) x 2 = 16 ml (80 x 0.1) = 8 ml Lower volume not lower dose No data to support increased safety due to reduced volume but same dose Increased concentration in the bottle does not translate to increased concentration in tissues (needed for greater effect) Relaxivity 1 T1 = r1 [Gd]
5 Increasing Relaxivity Standard Gd-BOPTA Benzyloxymethyl chain Gd +3 & H20 Weak / Transient Interaction w/proteins Protein 2-fold increase in relaxivity (r1, r2) effectiveness Signal intensity vs TR Higher Signal from the lesion = improved visualization With MultiHance T1=1/(R 10 + r 1C t) Standard Gd Without contrast Courtesy Dr. E. Kanal Relaxivity and Molecular Design MultiHance Eovist (Primovist) Gadavist Ablavar
6 Protein Interaction Relaxivities in Protein-Containing Solutions (1.5 T) Interaction w/protein Relaxation rate (R1) [1/s]* *R1 Relaxation 37 C HSA (g/dl) Various concentration of Proteins Giesel FL, et al. Influence of Human Serum Albumin on Longitudinal and Transverse Relaxation Rates (R1 and R2) of Magnetic Resonance Contrast Agents. Invest Radiol. 2006;41: Comparative Study Gadavist vs. ProHance Phase III data Relaxivity MultiHance (~100% higher ~8) Gadavist (~17% higher ~5) Magnevist, OptiMARK, Omniscan, ProHance (~4) Dotarem (<4) *Relaxivity (r1) in units of L/mmol/sec
7 Colosimo C et al: Detection of intracranial metastases. A multicenter, intrapatient comparison of gadobenate dimeglumine-enhanced MRI with routinely used contrast agents at equal dosage. Invest Radiol 36-2: 72-81, 2001 Dotarem MultiHance 0.1 mmol 0.1 mmol > 2-fold increase in relaxivity Reducing Dose The lowest possible dose of GBCA required to obtain the needed clinical information should be used, and it should generally not exceed the recommended single dose. (Note: the lowest diagnostic dose has not been thoroughly investigated for many indications and caution should be exercised so as not to administer a dose that is too low to provide the diagnostic information sought from the examination). ACR Manual on Contrast Media Version Relaxivity and Dose AJNR Am J Neuroradiol 36: Sep 2015
8 MultiHance 0.1 mmol/kg Dotarem 0.1 mmol/kg MultiHance 0.05 mmol/kg Dotarem 0.1 mmol/kg Relaxivity and Dose AJNR Am J Neuroradiol 36: Sep 2015
9 Clinical Safety Adverse Events Acute: Idiosyncratic / Anaphlactoid Chronic: NSF Gadolinium Retention An anaphylactoid reaction due to Gd-DTPA was observed in a patient who had disposition of asthma bronchiale. Five minutes after injection of Gd-DTPA, the patient developed laryngeal edema and erythema over the whole body. The patient recovered after treatment. It may be advisable to tighten indications for Gd-DTPA study on patients with allergic disposition. Gd-DTPA should be used with the same care against the anaphylactoid reaction as iodinated contrast media. ACR According to the ACR Guidance on MR Safe Practices (pg 15), adverse events after the intravenous injection of gadolinium seem to be more common in patients who had previous reactions to an MR contrast agent. In one study, 16 (21%) of 75 patients who had previous adverse reactions to MR contrast agents reacted to subsequent injections of gadolinium. Patients with asthma seem to be more likely to have an adverse reaction to the administration of a gadolinium-based MR contrast agent Patients with allergies also seemed to be at increased risk (approx times compared with patients without allergies) Patients who have had adverse reactions to iodinated contrast media are more than twice as likely to have an adverse reaction to gadolinium (6.3% of 857 patients)
10 the clinical safety of these agents is to a large extent dependent upon their metabolic stability in vivo. - Val Runge, MD Investigative RadiologyVolume 00, Number 00, Month 2016 Linear Macrocyclic OptiMARK Non-Ionic Magnevist Ionic Omniscan Non-Ionic MultiHance Ionic Gadavist Non-Ionic Dotarem Ionic Eovist (Primovist) Ionic ProHance Non-Ionic Fundam Clin Pharmacol Dec;20(6):563-76
11 Stability Metrics In Vitro Macrocyclic Ionic Linear Non-Ionic Linear Frenzel T, Lengsfeld P, Schirmer H, et al.stability of gadolinium-based magnetic resonance imaging contrast agents in human serum at 37 degrees C.Invest Radiol2008;43: Stability Metrics In Vitro Kinetic Stability: Rate/speed at which the gadolinium dissociates (reported at a ph 1) Thermodynamic Stability: Energy required to release the Gd 3+ ion (reported at a ph 1 or ph 7 as conditional stability") AJNR Am J Neuroradiol 35: Dec 2014
12 Is disassociation and retention of gadolinium a new observation? Increase zinc excretion Compared Omniscan, Magnevist and ProHance Puttagunta, Invest Radiol 1996 Dec;31(12:739-42) Increase zinc excretion Gadolinium-DTPA-BMA caused the highest increase in zinc excretion among the three agents. Gadolinium-HP-DO3A was found to be the most kinetically inert among the three drugs tested. Compared Omniscan, Magnevist and ProHance Puttagunta, Invest Radiol 1996 Dec;31(12:739-42)
13 Radiology Jun;227(3): Gadodiamide administration causes spurious hypocalcemia. CONCLUSION: Gadodiamide administration causes spurious hypocalcemia, particularly at doses of 0.2 mmol/kg or higher and in patients with renal insufficiency. Prince MR, et. al. Retention in bone marrow Omniscan (Gd[DTPA-BMA]) left approximately 4 times (previous study 2.5 times) more Gd behind in bone than did ProHance (Gd[HP-DO3A]).. White, Invest Radiol 2006 Mar;4(3):272-8
14 Tweedle MF. Using Radiotracers to Characterize MRI Contrast Agents. Invest Radiol 2002;37:
15 Radiology Jun 8: Electronically published March year-old female 7 prior administrations of Omniscan (Linear)
16 Electronically published March year-old female 15 prior administrations of ProHance (Macrocyclic) Increased T1-Signal (Brain) Yes Magnevist Omniscan?? MultiHance Gadavist No ProHance Dotarem
17 ACR Statement (March 2017) Intracranial gadolinium retention only recently reported but known for over 10 years some chelates not completely stable Indisputable evidence amount of gadolinium deposited after a single dose is incredibly small Although gadolinium accumulation appears to be dose dependent, no evidence of cellular toxicity nor is there credible evidence of neurologic sequelae after > 300 m human doses worldwide ACR Statement (March 2017) Observing increased signal on T1-weighted images is not a sensitive way to detect retained gadolinium Sensitive quantitative mass spectrometry data from multiple sources show that deposition occurs even with macrocyclic agents (albeit at lower levels) Deposition rates for linear and macrocyclic agents vary with a given class
18 April 2017 Pediatric Radiology April 2017 Pediatric Radiology Retrospective Study 50 patients (mean age 8 yr) - normal renal function - 6 administrations of Dotarem Control: 59 age-matched never having received Gd Measured: - globus pallidus-to-thalamus SI ratio - dentate nucleus-to-pons SI ratio April 2017 Pediatric Radiology
19 April 2017 Pediatric Radiology April 2017 Pediatric Radiology FDA 5/22/17
20 Retained Gadolinium All agents have some level of retention Bone, tissues, brain, etc Stratifies similar to what we see regarding NSF Clinical implications unknown (except for NSF) Radiologists should review orders for gadolinium to ensure they are clinically necessary (FDA statement July 2015) Document: Agent, dose, route and rate (if applicable) for ALL patients (ACR pg 15) NSF Risk increases with decreasing renal function, decreasing agent stability, increasing dose (mmol/kg) and repeat dose Incidence has been greatly reduced by screening patients and selection of agents with higher stability Now generally accepted that GBCA exposure is a necessary factor in the development of NSF (ACR) Sadowski, E. A. et al. Radiology 2007;0: NSF ACR Grouping
21 NSF ACR Grouping Only agents with labeled contraindications Omniscan,OptiMARK, Magnevist NSF Published data: frequency of NSF not equal among all agents (Radiology 2008, /radiol ) Only agents with no unconfounded cases of NSF MultiHance, ProHance, Dotarem Radiology Apr 15: [Epub ahead of print] MultiHance (Linear Ionic) Retrospective Study 401 Patients 303 Dialysis Dependent egfr range: 6-41 Mean Contrast Amount: 24 ml
22 Invest Radiol Nov;51(11): patients with egfr <30 MultiHance No cases of NSF AJR:205, September 2015 AJR:205, September 2015 Conclusion: Study data confirm that both gadoteridol and gadobenate dimeglumine properly belong to the class of GBCAs considered to be associated with the lowest risk of NSF.
23 One More Thing re No NSF MultiHance Eovist Ablavar * * Not commercially available at this time July 2017 July 2017
24 July 2017 July 2017 July 2017
25 July 2017 July 2017 There are things we know that we know. There are known unknowns. That is to say there are things that we now know we don't know. But there are also unknown unknowns. There are things we do not know we don't know. - Donald Rumsfeld
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
MR Contrast Agents. Why Use Contrast Agents in MRI? Why Use Contrast Agents in MRI? US Agents. Understanding and Embracing Change
 Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
A Safety Update on the Gadolinium Chelates
 Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018)
 Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Seeing is Believing... Look Beneath the Surface
 References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
Administration of Gadolinium Contrast in Adults Procedural Guideline
 Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Contrast-enhanced MRI: how do changing EU regulations impact daily practice?
 Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
Beyond NSF: Acute GBCA adverse reactions
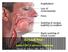 Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
INTRODUCTION CLINICAL EXPERIENCE CLINICAL EFFICACY PEDIATRIC EXPERIENCE DOSING SUMMARY
 1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine
 Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
NSF Coming and Going
 NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
Global reports and findings of NSF and its effects
 Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Gadolinium Deposition:
 Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
Annex II. Scientific conclusions
 Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
Protocol for iv. iodine and gadolinium contrast studies
 Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Report on the Investigation Results
 This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
FDA Advisory Committee Briefing Document. Medical Imaging Drugs Advisory Committee. Meeting to be held on February 14, 2013
 FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
Extracellular gadolinium-based contrast media: An overview
 European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
Gadolinium in Humans: A Family of Disorders
 Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
Gadolinium Retention in the Dentate Nucleus and Globus Pallidus Is Dependent on the Class of Contrast Agent 1
 Note: This copy is for your personal non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, contact us at www.rsna.org/rsnarights. Alexander Radbruch,
Note: This copy is for your personal non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, contact us at www.rsna.org/rsnarights. Alexander Radbruch,
PATIENT CARE AND MRI SAFETY Module 7
 PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
PRODUCT MONOGRAPH DOTAREM. (gadoterate meglumine) Injection. For Intravenous Use
 PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
William G. Bradley, Jr, MD, PhD, FACR
 William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
Hepatobiliary Contrast Agents for Liver MRI
 Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents
 Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Annex III. Amendments to relevant sections of the product information
 Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Please see Important Safety Information, including Boxed Warnings for Gadavist, Eovist and Magnevist on the following pages.
 Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents in Human Whole Blood at 1.5, 3, and 7 T
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research
 University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION. 1.1 Central Nervous System. 1.2 Extracranial/Extraspinal Tissues. 1.3 Body
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Figure 1 Figure 1: Energy diagram illustrates formation of a chelate (ML) created by combining two chemicals free Gd 3 (M), which is a metal ion, and
 Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Please see Important Safety Information, including Boxed Warnings for Gadavist and Magnevist below.
 August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
IL PROBLEMA DEL GADOLINIO OGGI
 11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
DOTAREM (gadoterate meglumine) Injection for intravenous use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
PRODUCT MONOGRAPH PRIMOVIST. gadoxetate disodium injection mg/ml (0.25 mmol/ml)
 PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
Magnetic resonance (MR) imaging
 This copy is for personal use only. To order printed copies, contact reprints@rsna.org Reviews and Commentary n Opinion Carlo Cosimo Quattrocchi, MD, PhD Aart J. van der Molen, MD Published online 10.1148/radiol.2016161626
This copy is for personal use only. To order printed copies, contact reprints@rsna.org Reviews and Commentary n Opinion Carlo Cosimo Quattrocchi, MD, PhD Aart J. van der Molen, MD Published online 10.1148/radiol.2016161626
Incidence of Immediate Gadolinium Contrast Media Reactions
 Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
Gadolinium-Based Contrast Agents for Magnetic Resonance Imaging (marketed as Magnevist, MultiHance, Omniscan, OptiMARK, ProHance)
 Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Frequency and Severity of Acute Allergic-Like Reactions to Gadolinium-Containing IV Contrast Media in Children and Adults
 Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Public Assessment Report
 Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
CONTRAINDICATIONS History of severe hypersensitivity reaction to EOVIST (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM. Gadoterate meglumine injection. (376.9 mg/ml, equivalent to 0.
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
Instructions for the safe administration of gadolinium based contrast agents (GBCA). 1. PREADMINISTRATION PRECAUTIONS:
 POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
Clinical Safety and Diagnostic Value of the Gadolinium Chelate Gadoterate Meglumine (Gd-DOTA)
 ORIGINAL ARTICLE Clinical Safety and Diagnostic Value of the Gadolinium Chelate Gadoterate Meglumine (Gd-DOTA) Christoph U. Herborn, MD,* Elmar Honold, MD, Michael Wolf, Jörn Kemper, MD, Sonja Kinner,
ORIGINAL ARTICLE Clinical Safety and Diagnostic Value of the Gadolinium Chelate Gadoterate Meglumine (Gd-DOTA) Christoph U. Herborn, MD,* Elmar Honold, MD, Michael Wolf, Jörn Kemper, MD, Sonja Kinner,
Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be used
 Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance
 Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
Contrast controversies and confusion
 Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS
 DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
Imaging in the time of NFD/NSF: do we have to change our routines concerning renal insufficiency?
 DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
Central nervous system gadolinium accumulation in patients undergoing periodical contrast MRI screening for hereditary tumor syndromes
 Vergauwen et al. Hereditary Cancer in Clinical Practice (2018) 16:2 DOI 10.1186/s13053-017-0084-7 RESEARCH Central nervous system gadolinium accumulation in patients undergoing periodical contrast MRI
Vergauwen et al. Hereditary Cancer in Clinical Practice (2018) 16:2 DOI 10.1186/s13053-017-0084-7 RESEARCH Central nervous system gadolinium accumulation in patients undergoing periodical contrast MRI
This copy is for personal use only. To order printed copies, contact Purpose: Materials and Methods: Results: Conclusion:
 This copy is for personal use only. To order printed copies, contact reprints@rsna.org Original Research n Pediatric Imaging Pediatric Brain: Repeated Exposure to Linear Gadolinium-based Contrast Material
This copy is for personal use only. To order printed copies, contact reprints@rsna.org Original Research n Pediatric Imaging Pediatric Brain: Repeated Exposure to Linear Gadolinium-based Contrast Material
ASKLEPIOS Course. July 20 21, 2018 Bangkok/Thailand
 This ASKLEPIOS course is implemented with great support and partnership of Guerbet and RCRT Education in partnership Course information This ESOR course aims to discuss the most important topics in neuroradiology.
This ASKLEPIOS course is implemented with great support and partnership of Guerbet and RCRT Education in partnership Course information This ESOR course aims to discuss the most important topics in neuroradiology.
Contrast Media Guidelines. To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
 APPROVED BY: Director of Radiology Page 1 of 6 Purpose: Contrast Media Guidelines To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
APPROVED BY: Director of Radiology Page 1 of 6 Purpose: Contrast Media Guidelines To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA.
 ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting
 European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance
 Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
Summary. Correspondence Jerrold L. Abraham. Accepted for publication 17 September 2007
 CLINICAL AND LABORATORY INVESTIGATIONS DOI 10.1111/j.1365-2133.2007.08335.x Dermal inorganic gadolinium concentrations: evidence for in vivo transmetallation and long-term persistence in nephrogenic systemic
CLINICAL AND LABORATORY INVESTIGATIONS DOI 10.1111/j.1365-2133.2007.08335.x Dermal inorganic gadolinium concentrations: evidence for in vivo transmetallation and long-term persistence in nephrogenic systemic
Review of Hepatobiliary Contrast Agents: Current Applications and Challenges
 REVIEW Review of Hepatobiliary Contrast Agents: Current Applications and Challenges Alex Frydrychowicz, M.D.*, The group of hepatobiliary contrast agents comprises two gadolinium-based contrast agents
REVIEW Review of Hepatobiliary Contrast Agents: Current Applications and Challenges Alex Frydrychowicz, M.D.*, The group of hepatobiliary contrast agents comprises two gadolinium-based contrast agents
ARTICLE IN PRESS European Journal of Radiology xxx (2011) xxx xxx
 European Journal of Radiology xxx (2011) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Tolerability and diagnostic value
European Journal of Radiology xxx (2011) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Tolerability and diagnostic value
SUSPECT ADVERSE REACTION REPORT
 CIOMS FORM SUSPECT ADVERSE REACTION REPORT DE-BFARM-16306260 I. REACTION INFORMATION 1. PATIENT INITIALS 1a. COUNTRY 2. DATE OF BIRTH 2a. AGE 3. SEX 4-6 REACTION ONSET privacy polyneuropathische Schmerzen
CIOMS FORM SUSPECT ADVERSE REACTION REPORT DE-BFARM-16306260 I. REACTION INFORMATION 1. PATIENT INITIALS 1a. COUNTRY 2. DATE OF BIRTH 2a. AGE 3. SEX 4-6 REACTION ONSET privacy polyneuropathische Schmerzen
Contrast. 5 Oral Contrast for CT...13
 2 Contrast 1 GI Fluoroscopic Contrast...10 Adult Esophagram/Upper GI... 10 Pediatric Upper GI... 10 Enteroclysis... 10 Double-Contrast Barium Enema... 10 Single-Contrast Barium Enema... 10 Rectum and Pelvic
2 Contrast 1 GI Fluoroscopic Contrast...10 Adult Esophagram/Upper GI... 10 Pediatric Upper GI... 10 Enteroclysis... 10 Double-Contrast Barium Enema... 10 Single-Contrast Barium Enema... 10 Rectum and Pelvic
Received: 16 October 2015, Accepted: 3 December 2015, Published online in Wiley Online Library: 11 January 2016
 Short communication Received: 16 October 2015, Accepted: 3 December 2015, Published online in Wiley Online Library: 11 January 2016 (wileyonlinelibrary.com) DOI: 10.1002/cmmi.1683 High signal intensity
Short communication Received: 16 October 2015, Accepted: 3 December 2015, Published online in Wiley Online Library: 11 January 2016 (wileyonlinelibrary.com) DOI: 10.1002/cmmi.1683 High signal intensity
Neuroradiology Original Research
 MRI of Multiple Sclerosis Lesions Neuroradiology Original Research Ender Uysal 1 Sukru Mehmet Erturk 1 Hakan Yildirim 1 Feray Seleker 2 Muzaffer Basak 1 Uysal E, Erturk SM, Yildirim H, et al. Keywords:
MRI of Multiple Sclerosis Lesions Neuroradiology Original Research Ender Uysal 1 Sukru Mehmet Erturk 1 Hakan Yildirim 1 Feray Seleker 2 Muzaffer Basak 1 Uysal E, Erturk SM, Yildirim H, et al. Keywords:
Val M. Runge, MD Editor-in-Chief Investigative Radiology
 Val M. Runge, MD Editor-in-Chief Investigative Radiology Patients First RSNA 2012 The classical model of medical care which portrays the authoritative physician evaluating and treating an obedient, non-inquisitive
Val M. Runge, MD Editor-in-Chief Investigative Radiology Patients First RSNA 2012 The classical model of medical care which portrays the authoritative physician evaluating and treating an obedient, non-inquisitive
Special Articles Original Research
 Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
Brain MRI With Single-Dose (0.1 mmol/kg) Gadobutrol at 1.5 T and 3 T: Comparison With 0.15 mmol/kg Gadoterate Meglumine
 euroradiology/head and eck Imaging riginal Research Kramer et al. rain MRI With Gadobutrol Versus Gadoterate Meglumine euroradiology/head and eck Imaging riginal Research Harald Kramer 1 Val M. Runge 2
euroradiology/head and eck Imaging riginal Research Kramer et al. rain MRI With Gadobutrol Versus Gadoterate Meglumine euroradiology/head and eck Imaging riginal Research Harald Kramer 1 Val M. Runge 2
Mechanism of NSF: New evidence challenging the prevailing theory
 Thomas Jefferson University Jefferson Digital Commons Scleroderma Center Faculty Papers Scleroderma Center of Thomas Jefferson University 11-2009 Mechanism of NSF: New evidence challenging the prevailing
Thomas Jefferson University Jefferson Digital Commons Scleroderma Center Faculty Papers Scleroderma Center of Thomas Jefferson University 11-2009 Mechanism of NSF: New evidence challenging the prevailing
PRODUCT INFORMATION DOTAREM
 PRODUCT INFORMATION DOTAREM NAME OF THE MEDICINE DOTAREM (gadoteric acid) paramagnetic contrast medium for Magnetic Resonance Imaging (MRI). The structural formula of gadoteric acid is shown below: The
PRODUCT INFORMATION DOTAREM NAME OF THE MEDICINE DOTAREM (gadoteric acid) paramagnetic contrast medium for Magnetic Resonance Imaging (MRI). The structural formula of gadoteric acid is shown below: The
Reference ID: MultiHance Injection, NDA , SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov _CLEAN.
 MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric patients. The dose is delivered by manual or power injection.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Side Effects and Complications of Magnetic Resonance Contrast Media
 HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
Magnetic Resonance Insights
 Open Access: Full open access to this and thousands of other papers at http://www.la-press.com. Magnetic Resonance Insights Contrast-Enhanced Magnetic Resonance Imaging in Pediatric Patients: Review and
Open Access: Full open access to this and thousands of other papers at http://www.la-press.com. Magnetic Resonance Insights Contrast-Enhanced Magnetic Resonance Imaging in Pediatric Patients: Review and
Renal Tolerance of a Neutral Gadolinium Chelate (gadobutrol) in Patients with Chronic Renal Failure: Results of a Randomized Study 1
 Contrast Media Bernd Tombach, MD Christoph Bremer, MD Peter Reimer, MD Klaus Kisters, MD Roland M. Schaefer, MD Viviane Geens, MD Walter Heindel, MD Index terms: Gadolinium Kidney, failure, 81.699 Kidney,
Contrast Media Bernd Tombach, MD Christoph Bremer, MD Peter Reimer, MD Klaus Kisters, MD Roland M. Schaefer, MD Viviane Geens, MD Walter Heindel, MD Index terms: Gadolinium Kidney, failure, 81.699 Kidney,
Poster No.: B-229 Congress: ECR 2011 Scientific Paper. Authors: E. De Kerviler 1, S. Gaillard 2 ; 1 Paris/FR, 2 Villepinte/FR
 Pharmacovigilance based on spontaneous adverse event reporting of meglumine gadoterate (Gd-DOTA) after 15 million administrations and 20 years of clinical use Poster No.: B-229 Congress: ECR 2011 Type:
Pharmacovigilance based on spontaneous adverse event reporting of meglumine gadoterate (Gd-DOTA) after 15 million administrations and 20 years of clinical use Poster No.: B-229 Congress: ECR 2011 Type:
Gadolinium Magnetic Resonance Contrast Agents Produce Analytic Interference in Multiple Serum Assays
 Clinical Chemistry / GDOLINIUM CONTRST GENT INTERFERENCE Gadolinium Magnetic Resonance Contrast gents Produce nalytic Interference in Multiple Serum ssays Kerry.S. Proctor, MD, 1 Lokinendi V. Rao, PhD,
Clinical Chemistry / GDOLINIUM CONTRST GENT INTERFERENCE Gadolinium Magnetic Resonance Contrast gents Produce nalytic Interference in Multiple Serum ssays Kerry.S. Proctor, MD, 1 Lokinendi V. Rao, PhD,
CONTRAINDICATIONS Clinically important hypersensitivity reactions to DOTAREM. (4)
 M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Safety of Gadoterate Meglumine (Gd-DOTA) as a Contrast Agent for Magnetic Resonance Imaging
 ORIGINAL RESEARCH ARTICLE Drugs R D 2010; 10 (3): 133-145 1179-6901/10/0003-0133 ª 2010 Ishiguchi & Takahashi, publisher and licensee Adis Data Information BV. This is an open access article published
ORIGINAL RESEARCH ARTICLE Drugs R D 2010; 10 (3): 133-145 1179-6901/10/0003-0133 ª 2010 Ishiguchi & Takahashi, publisher and licensee Adis Data Information BV. This is an open access article published
Gadolinium and nephrogenic systemic fibrosis
 mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
October 23, Re: Gadolinium Toxicity from GBCAs. Dear Dr. Hamburg:
 October 23, 2012 Commissioner Margaret A. Hamburg, M.D. U.S. Food and Drug Administration 10903 New Hampshire Avenue WO Bldg. 1, Room 2217 Silver Springs, MD 20993 Re: Gadolinium Toxicity from GBCAs Dear
October 23, 2012 Commissioner Margaret A. Hamburg, M.D. U.S. Food and Drug Administration 10903 New Hampshire Avenue WO Bldg. 1, Room 2217 Silver Springs, MD 20993 Re: Gadolinium Toxicity from GBCAs Dear
This copy is for personal use only. To order printed copies, contact Purpose: Materials and Methods: Results: Conclusion:
 This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
Magnetic resonance imaging (MRI) scans are significantly
 Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Update. Drug Safety. Welcome to the first issue of the newly launched Drug Safety Update. This
 Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml (for multiple use) solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml (for multiple use) solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for
Evangelos Chartampilas Bioclinic Hospital Thessaloniki, Greece
 Evangelos Chartampilas Bioclinic Hospital Thessaloniki, Greece Hepatospecificcontrast agents Gadobenate dimeglumine (Multihance) Gadoxeticacid (Primovist) 3-5% liver uptake 50% liver uptake Hepatobiliary
Evangelos Chartampilas Bioclinic Hospital Thessaloniki, Greece Hepatospecificcontrast agents Gadobenate dimeglumine (Multihance) Gadoxeticacid (Primovist) 3-5% liver uptake 50% liver uptake Hepatobiliary
Improving postoperative MR imaging of pituitary macroadenomas: comparison of full and reduced dose of gadopentetate dimeglumine
 Eur. Radiol. 10, 1068±1072 (2000) Ó Springer-Verlag 2000 European Radiology Original article Improving postoperative MR imaging of pituitary macroadenomas: comparison of full and reduced dose of gadopentetate
Eur. Radiol. 10, 1068±1072 (2000) Ó Springer-Verlag 2000 European Radiology Original article Improving postoperative MR imaging of pituitary macroadenomas: comparison of full and reduced dose of gadopentetate
A WORLD OF KNOWLEDGE FOR MR TECHNOLOGISTS & RADIOGRAPHERS
 SMRT A WORLD OF KNOWLEDGE FOR MR TECHNOLOGISTS & RADIOGRAPHERS SECTION FOR MAGNETIC RESONANCE TECHNOLOGISTS OF THE INTERNATIONAL SOCIETY FOR MAGNETIC RESONANCE IN MEDICINE Home Studies Educational Seminars
SMRT A WORLD OF KNOWLEDGE FOR MR TECHNOLOGISTS & RADIOGRAPHERS SECTION FOR MAGNETIC RESONANCE TECHNOLOGISTS OF THE INTERNATIONAL SOCIETY FOR MAGNETIC RESONANCE IN MEDICINE Home Studies Educational Seminars
PLEASE SCROLL DOWN FOR ARTICLE
 This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
Gadolinium Use in Patients with Kidney Disease: A Cause for Concern
 Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Radiology Reimbursement Information Setting: Hospital Outpatient
 Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
Optimal detection and characterization of brain tumors on
 ORIGINAL RESEARCH Z. Seidl J. Vymazal M. Mechl M. Goyal M. Herman C. Colosimo M. Pasowicz R. Yeung B. Paraniak-Gieszczyk B. Yemen N. Anzalone A. Citterio G. Schneider S. Bastianello J. Ruscalleda Does
ORIGINAL RESEARCH Z. Seidl J. Vymazal M. Mechl M. Goyal M. Herman C. Colosimo M. Pasowicz R. Yeung B. Paraniak-Gieszczyk B. Yemen N. Anzalone A. Citterio G. Schneider S. Bastianello J. Ruscalleda Does
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
