NSF Coming and Going
|
|
|
- Oswald Freeman
- 6 years ago
- Views:
Transcription
1 NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins Papular morphology Dimpling
2 NSF key points To prevent NSF Identify at risk patients: GFR< 30 ml/min, acute For these at risk patients verify Gd is necessary avoid high Gd doses (use 0.1mMol/kg or less) hand inject, experienced technologists For dialysis patients do MRI just before dialysis appt Virtually no new cases in 2008 or 2009! Nonionic linear Gd more reports of NSF Gadobenate Dimeglumine (ionic linear Gd) more reports of death (anaphylaxis)
3 Cases of NSF by Date of Onset* Gadodiamide Gd:DTPA < FDA & EMEA warnings
4
5 NSF: Association With Renal Disease GFR< 30 ml/min Stage I Stage II Stage III Stage IV Stage V GFR Dialysis CKD, chronic kidney disease; egfr, estimated glomerular filtration rate; PD, peritoneal dialysis Issa N et al. Cleve Clin J Med. 2008;75:
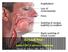
6 NSF: Clinical Images Areas of thick, hardened skin, commonly associated with brawny hyperpigmentation, and preferentially localized to the extremities Extensive thickening of the skin, peau de orange appearance and in some cases, distinct papules and subcutaneous nodules Mendoza FA et al. Sem Arthritis Rheum. 2006;35:
7 some patients with NSF (estimated at 5% or less) have an exceedingly rapid and fulminant disease course. Cowper et al
8 NSF major risk factors GFR < 30ml/min high dose pro-inflammatory thrombosis Biopsy site surgery trauma SLE inpatient >>. outpatients
9 Peau D orange Skin thickening
10 Skin features Papular morphology Dimpling
11 Joint Contractures Jeuvenile NSF Rheumatoid Arthritis
12 Cornell & Columbia All GBCA Total 15/83121 Standard dose 0/74124 High dose 15/ % egfr > 30 ml/min 0/ < egfr < 30 ml/min 2/ % egfr < 15 ml/min (no dialysis) 10/ % C = chronic hemodialysis* 1/ % chronic peritoneal dialysis 0/19 egfr< 30 ml/min w/o ARF 4/ % w/ ARF 11/ % ARF during rising creatinine and no dialysis for > 2 days 11/58 19% Radiology 2008;248
13 Cornell 1 case Exams with high dose Gd & GFR<30 = 284 Hospital MRI = 1 Outpatient MRI = 2 Time to exam = 3d Earlier use of dialysis Columbia 14 cases Exams with high dose Gd & GFR<30 = 507 Hospital MRI = 3 Outpatient MRI = 1 Time to exam = same day Higher threshold to initiate dialysis
14 Cornell & Columbia July 2006 to present July 2006 no Gd for GFR < 15 ml/min Dialysis after Gd in dialysis patients September 2007 careful for GFR < 30 ml/min Gadobenate Dimeglumine instead of Gadodiamide No cases of NSF since May, cardiac arrests and 1 death
15 NSF/NFD papers on Medline 200 n=424 Extrapolated from Jan Aug # of Papers Year
16 80 papers 292 NSF cases all biopsy confirmed 64 papers had data on the relationship of GBCA and NSF involving 243 patients. History of Gd exposure 220 Gd looked for but not found 23 Gd not discussed (16 papers) 49. Total = 292
17 Type of MRI scan (n=112) MRA 57 Brain 7 Pelvis 2 Liver 1 Abdomen 9 Lower extremity 4 MRCP 5 Spine 2 MRV 1 Multiple 23 DSA txp renal art. 1
18 Gadolinium dose Standard Dose (0.1mmol/kg): 23 High Dose (>0.15mmol/kg):164 Dose not specified: 56 Total 243 Multiple doses: 63 with at least 1 high dose: 55 only standard dose noted: 7 no dose noted 1
19 Gd Dose: Case Controlled studies Author # controls # with NSF GBCA dose controls GBCA dose NSF P Kallen ml 80 ml.01 Markmann mmol/kg Coolidge ml 0.23mmol/kg 0.44 mmol/kg 45 ml 0.39mmol/kg
20 Subsystolic Thigh Compression Thigh cuffs with extension tubing Inflation pressure = 60 mmhg Inflate just prior to mask acquisition Venous contamination is delayed calf station to 60 seconds
21 T = 0 Start Injection T = 18 s: 1 st station T = 41 s: 2 nd station 60 mm Hg A Curved Thigh Cuff for Suppressing Venous Enhancement on Peripheral MRA Honglei Zhang et al J. Cardoiovascular MR, 2007 T = 57 s: 3 rd station 57s 1:37min 2:17min 2:57min 3:37min
22 Subclavian Venography Inject both arms simultaneously with 20:1 Gd dilution with normal saline
23 Occluded Right Subclavian Vein
24 Gadolinium Chelate Type Gadodiamide: 163 Gadopentetate Dimeglumine: 10 Gadoversetamide : 5 Multiple: 7 Unknown type: 25 no gadolinium association 23 no mention of gadolinium: 49
25 Risk Factors: Chelate stability Chelates make Gd +++ chemically inert May allow Gd to enter cells (e.g. MultiHanc Allow rapid glomerular filtration half-life for excretion fecal elimination Normal renal function 1.5 hours negligible Moderate egfr hours 6% Severe egfr hours 9% egfr <15 but not on dialysis 34.3 hours Hemodialysis 2.6 hours Peritoneal dialysis 52.7 hours
26 Macrocyclic Complexes - OOC COO - - OOC N N Gd 3+ - OOC N N COO - - OOC N N Dotarem ProHance Gd-DOTA Gd-HP-DO3A COO - - OOC N N N Gd 3+ Gd 3+ N N - OOC OH COO - N Gadovist Gd-DO3A-butrol OH OH OH Linear Complexes COO - COO - - OOC Gd 3+ - OOC N N N COO - COO - CH 3 NHOC Gd 3+ - OOC N N N COO - CONHCH 3 Magnevist Omniscan Gd-DTPA Gd-DTPA-BMA Ionic nonionic
27 Transmetallation (Zn 2+, Cu 2+, Ca 2+, Fe 2+ Gd 2+ Zn 2+ Zn 2+ Gd 2+
28 Stabilty: Macrocyclic vs Linear chelates Kuo JACR 2008; 5:29-35
29 Why not switch to using Macrocyclic GBCA? * Recently cases reported with macrocyclics: ProHance and Gadovist FDA NSF mtg 12/8/09 ** Compounds with protein binding higher relaxivity and biliary excretion, have no cases of NSF reported: FDA NSF mtg 12/8/09 Gado trisodium (Ablavar) Gadoxetate trisodium (Eovist) Gadobenate Dimeglumine (MultiHance)
30 NSF: Change in incidence following a switch in gadolinium agents Altun et al Radiology 2009:253: Screen for at risk patients Gadobenate Dimeglumine maximum dose = 0.05 mmol/kg in at risk patients No cases of NSF in 549 at risk or on dialysis
31 Gadofosveset: 0.05 mmol/kg ~ 10ml 1x1.8x2.5 mm 3 1x1.8x2.5 mm 3 1x1x1 mm 3 Courtesy of Tim Leiner,
32 Risk Factors: Older Age is protective NSF Case Reports NSF Case Reports Columbia Gd MR GFR < < >90 Age (years) Age (years) < >90
33 Typical NSF Presentations (collected from 292 reported cases) Dialysis: 208/292 71% not specified 17 Hemodialysis 150 Peritoneal dialysis 37 CVVH 4 Renal transplant: 57/292 20% failing renal transplant: 34 12%
34 Radiology 2008;248: egfr < 15 ml/min (no dialysis) 10/ % Chronic hemodialysis 1/ %
35 Knopp et al Investigative Radiology 2006;41: Clinical Features of NSF Male: 136 Female: 116 Gender not specified: 30 SUM 45M 53 est 270M 318 Contractions: Limited range of motion: Death reported: 47 Death hastened by NSF: 2
36 US FDA Medwatch Database 2004 to /AdverseDrugEffects/ucm htm ~ 5 million reports outside US AE and NSF events excluded Gadobenate dimeglumine Gadoteridol Gd:DTPA Total AE hospitalized Death Gadodiami Market share Deaths per million doses
37 National Traffic Safety Administration in 1 million chance of death For every 78 miles driven in US
38 Adverse Reactions MRI centers Murphy et al. Academic Radiology 1999;6: Contrast agent: n AE rate Gadoteridol % nonallergi 0.35 total AE (Macrocyclic) 0.14% allergic 0.3 severe AE 0.02% severe Gd:DTPA 0.15 (linear ionic) % total 0.001% severe Gadodiamide % total (linear Gadoteridol nonionic) linear0.00% severe macrocyclic nonionic
39 Adverse Reactions > 1 million patients Murphy et al. Academic Radiology 1999;6: Katayama et al Radiology 1990; 175: total AE severe AE iodine ionic iodine nonionic Macrocyclic Linear ionic linear nonionic
40 Adverse Event Rates in 337,647 patients Ionic contrast agent: 12.66% Nonionic low osmolar agent: 3.13% Katayama et al Radiology 1990;175:616-8
41 Severe adverse events Ionic contrast agent: 0.22% Nonionic low osmolar agent: 0.04% Katayama et al Radiology 1990;175:616-8
42 nonionic contrast media significantly reduce the frequency of severe and potentially lifethreatening ADRs to contrast media at all levels of risk and that use of these media represents the most effective means of increasing the safety of contrast media examinations Katayama et al Radiology
43 Minimize Risk of NSF Ask about renal function Dialysis, Renal transplant If no known renal disease no change If egfr < 30ml/min or acute renal failure Monitor study to ensure Gd is necessary Use single dose Hand injection best technologist +/-Informed consent single dose risk < 1in10,000 If on dialysis schedule schedule MRI just before dialysis If patient has NSF do not give gadolinium
44 Summary NSF is disappearing Avoiding high dose reduces NSF risk Dialysis shortly after MRI reduces NSF risk NSF deaths < allergic reaction deaths Nonionic more reports of NSF but less adverse events & fewer deaths
45 NSF: proposed mechanism
Beyond NSF: Acute GBCA adverse reactions
 Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018)
 Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Administration of Gadolinium Contrast in Adults Procedural Guideline
 Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
A Safety Update on the Gadolinium Chelates
 Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Global reports and findings of NSF and its effects
 Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
MR Contrast Agents. Why Use Contrast Agents in MRI? Why Use Contrast Agents in MRI? US Agents. Understanding and Embracing Change
 Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Seeing is Believing... Look Beneath the Surface
 References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
Gadolinium-Based Contrast Media
 Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
PATIENT CARE AND MRI SAFETY Module 7
 PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
Protocol for iv. iodine and gadolinium contrast studies
 Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research
 University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
Gadolinium-Based Contrast Agents for Magnetic Resonance Imaging (marketed as Magnevist, MultiHance, Omniscan, OptiMARK, ProHance)
 Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
William G. Bradley, Jr, MD, PhD, FACR
 William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
William G. Bradley, Jr, MD, PhD, FACR Professor of Radiology Chair, Dept of Radiology University of California, San Diego Medical Center San Diego, California Learning Objectives Identify emerging safety
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
Incidence of Immediate Gadolinium Contrast Media Reactions
 Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
Health Care Policy and Quality Original Research Prince et al. Immediate Gadolinium Contrast Media Reactions Health Care Policy and Quality Original Research Martin R. Prince 1 Honglei Zhang Zhitong Zou
Contrast-enhanced MRI: how do changing EU regulations impact daily practice?
 Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
Safety considerations in contrast enhanced procedures Carlo Catalano Sapienza University of Rome Contrast-enhanced MRI: how do changing EU regulations impact daily practice? CE MRI: EU regulations - Initial
INTRODUCTION CLINICAL EXPERIENCE CLINICAL EFFICACY PEDIATRIC EXPERIENCE DOSING SUMMARY
 1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
FDA Advisory Committee Briefing Document. Medical Imaging Drugs Advisory Committee. Meeting to be held on February 14, 2013
 FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
Instructions for the safe administration of gadolinium based contrast agents (GBCA). 1. PREADMINISTRATION PRECAUTIONS:
 POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS
 DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, April 2013 Any
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine
 Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
Gadolinium Deposition in the Brain: Summary of Known Science and Recommendations from the International Society for Magnetic Resonance in Medicine Vikas Gulani, MD, PhD 1-4*, Fernando Calamante, PhD 5,6,
Annex III. Amendments to relevant sections of the product information
 Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents
 Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Extracellular gadolinium-based contrast media: An overview
 European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
European Journal of Radiology 66 (2008) 160 167 Extracellular gadolinium-based contrast media: An overview Marie-France Bellin, Aart J. Van Der Molen University Paris-Sud 11, Department of Radiology, University
Special Articles Original Research
 Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
Special Articles Original Research Soulez et al. NSF in Patients With Stage 3 5 CKD Undergoing MRI With GBCA Special Articles Original Research Gilles Soulez 1 Daniel C. Bloomgarden 2,3 Neil M. Rofsky
SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA.
 ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be used
 Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Please see Important Safety Information, including Boxed Warnings for Gadavist, Eovist and Magnevist on the following pages.
 Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance
 Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
Safety and clinical usefulness of gadoteric acid including post-marketing surveillance MRI is an essential diagnostic tool in making definite diagnoses and determining therapeutic strategies. Extracellular
IL PROBLEMA DEL GADOLINIO OGGI
 11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
11-12 Maggio 2018 Rimini IL PROBLEMA DEL GADOLINIO OGGI Il punto di vista del Neuroradiologo pediatrico Dott.ssa Camilla Rossi Espagnet U.O. Neuroradiologia, Dipartimento Immagini, Ospedale Pediatrico
This copy is for personal use only. To order printed copies, contact Purpose: Materials and Methods: Results: Conclusion:
 This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
This copy is for personal use only. To order printed copies, contact reprints@rsna.org No Incidence of Nephrogenic Systemic Fibrosis after Gadobenate Dimeglumine Administration in Patients Undergoing Dialysis
Please see Important Safety Information, including Boxed Warnings for Gadavist and Magnevist below.
 August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
PRODUCT MONOGRAPH DOTAREM. (gadoterate meglumine) Injection. For Intravenous Use
 PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease
 International Nephrology, Article ID 679605, 6 pages http://dx.doi.org/10.1155/2014/679605 Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease Robert F. Hanna, 1 Lee A. Finkelstone, 1 Daniel
International Nephrology, Article ID 679605, 6 pages http://dx.doi.org/10.1155/2014/679605 Clinical Study Nephrogenic Systemic Fibrosis Risk and Liver Disease Robert F. Hanna, 1 Lee A. Finkelstone, 1 Daniel
T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents in Human Whole Blood at 1.5, 3, and 7 T
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2015 T1 Relaxivities of Gadolinium-Based Magnetic Resonance Contrast Agents
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
DOTAREM (gadoterate meglumine) Injection for intravenous use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Hepatobiliary Contrast Agents for Liver MRI
 Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Frequency and Severity of Acute Allergic-Like Reactions to Gadolinium-Containing IV Contrast Media in Children and Adults
 Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Reactions to Gadolinium- Containing Contrast Media MR Imaging Original Research Jonathan R. Dillman 1 James H. Ellis 1 Richard H. Cohan 1 Peter J. Strouse 2 Sophia C. Jan 1 Dillman JR, Ellis JH, Cohan
Annex II. Scientific conclusions
 Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
Annex II Scientific conclusions 31 Scientific conclusions In accordance with Article 107k of Directive 2001/83/EC, the CHMP considered the PRAC recommendation adopted on 6 July 2017. Overall summary of
Figure 1 Figure 1: Energy diagram illustrates formation of a chelate (ML) created by combining two chemicals free Gd 3 (M), which is a metal ion, and
 Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Note: This copy is for your personal, non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, use the Radiology Reprints form at the end of this article.
Side Effects and Complications of Magnetic Resonance Contrast Media
 HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
HOSPITAL CHRONICLES 2012, 7(4): 208 214 Review Side Effects and Complications of Magnetic Resonance Contrast Media Ekaterini Tavernaraki, MD, PhD, Anna Skoula MD, Stylianos Benakis, MD, Dimitrios Exarhos
Public Assessment Report
 Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
Public Assessment Report Increased risk of nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis and gadolinium-containing MRI contrast agents Executive summary 2 Introduction 3 Data assessed
Gadolinium in Humans: A Family of Disorders
 Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
Residents Section Clinical Perspective Semelka et al. Gadolinium in Humans Residents Section Clinical Perspective Residents inradiology Richard C. Semelka 1 Miguel Ramalho 1,2 Mamdoh AlObaidy 1,3 Joana
1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION. 1.1 Central Nervous System. 1.2 Extracranial/Extraspinal Tissues. 1.3 Body
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Gadolinium Deposition:
 Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
Gadolinium Deposition: Imaging Phenomenon or Should we change practice? Gregory Brown FSMRT MR Radiographer, PhD student Centre for Advanced Imaging, University of Queensland, Australia ORCID: 0000-0001-7994-2743
CONTRAINDICATIONS History of severe hypersensitivity reaction to EOVIST (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
Gadolinium and nephrogenic systemic fibrosis
 mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
mini review http://www.kidney-international.org & 2007 International Society of Nephrology Gadolinium and nephrogenic systemic fibrosis T Grobner 1 and FC Prischl 2 1 2nd Department of Medicine/Nephrology,
Policy #: 291 Latest Review Date: February 2013
 Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
Gadolinium Use in Patients with Kidney Disease: A Cause for Concern
 Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
Editorial Gadolinium Use in Patients with Kidney Disease: A Cause for Concern Mark A. Perazella* and Roger A. Rodby *Section of Nephrology, Department of Medicine, Yale University School of Medicine, New
Hepatobiliary Contrast Agents
 Hepatobiliary Contrast Agents SCBT/MR Annual Meeting Salt Lake City September 21, 2016 Scott B. Reeder, MD, PhD Department of Radiology University of Wisconsin Madison, WI Disclosures University of Wisconsin-Madison
Hepatobiliary Contrast Agents SCBT/MR Annual Meeting Salt Lake City September 21, 2016 Scott B. Reeder, MD, PhD Department of Radiology University of Wisconsin Madison, WI Disclosures University of Wisconsin-Madison
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM. Gadoterate meglumine injection. (376.9 mg/ml, equivalent to 0.
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
Imaging in the time of NFD/NSF: do we have to change our routines concerning renal insufficiency?
 DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
DI 10.1007/s10334-007-0071-5 EDITRIAL REVIEW Imaging in the time of FD/SF: do we have to change our routines concerning renal insufficiency? Georg Bongartz Accepted: 7 March 2007 ESMRMB 2007 Abstract To
PRODUCT MONOGRAPH PRIMOVIST. gadoxetate disodium injection mg/ml (0.25 mmol/ml)
 PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
Magnetic resonance imaging (MRI) scans are significantly
 Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Mini-Review Current Status of Gadolinium Toxicity in Patients with Kidney Disease Mark A. Perazella Department of Medicine, Section of Nephrology, Yale University School of Medicine, New Haven, Connecticut
Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance
 Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
Reiter et al. Journal of Cardiovascular Magnetic Resonance 2012, 14:31 REVIEW Open Access Minimizing Risk of Nephrogenic systemic fibrosis in Cardiovascular Magnetic Resonance Theresa Reiter 1*, Oliver
Liver 4 Phase CT Abdomen WO W - NC.A.V.D
 Liver 4 Phase CT Abdomen WO W - NC.A.V.D Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the ALARA
Liver 4 Phase CT Abdomen WO W - NC.A.V.D Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the ALARA
(Non-EKG Gated) CTA Thoracic Aorta = CTA Chest
 (Non-EKG Gated) CTA Thoracic Aorta = CTA Chest Reviewed By: Dan Verdini, MD, Rachael Edwards, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA
(Non-EKG Gated) CTA Thoracic Aorta = CTA Chest Reviewed By: Dan Verdini, MD, Rachael Edwards, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA
Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report
 JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report Arpita Aggarwal
JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Complications of nephrogenic systemic fibrosis following repeated exposure to gadolinium in a man with hypothyroidism: a case report Arpita Aggarwal
Report on the Investigation Results
 This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall Report
During the past decade, nephrogenic systemic fibrosis
 Low Risk for Nephrogenic Systemic Fibrosis in Nondialysis Patients Who Have Chronic Kidney Disease and Are Investigated with Gadolinium-Enhanced Magnetic Resonance Imaging Constantina Chrysochou,* Albert
Low Risk for Nephrogenic Systemic Fibrosis in Nondialysis Patients Who Have Chronic Kidney Disease and Are Investigated with Gadolinium-Enhanced Magnetic Resonance Imaging Constantina Chrysochou,* Albert
PRODUCT INFORMATION NAME OF THE MEDICINE DESCRIPTION. GADOVIST 1.0 (gadobutrol) Physico-chemical properties
 PRODUCT INFORMATION GADOVIST 1.0 (gadobutrol) NAME OF THE MEDICINE Gadovist 1.0 injection is a 1.0 mmol/ml solution of 10-(2,3-Dihydroxy-1- hydroxymethylpropyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic
PRODUCT INFORMATION GADOVIST 1.0 (gadobutrol) NAME OF THE MEDICINE Gadovist 1.0 injection is a 1.0 mmol/ml solution of 10-(2,3-Dihydroxy-1- hydroxymethylpropyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic
CT Pancreas 3 Phase CT Abdomen WO W - NC.A.V
 CT Pancreas 3 Phase CT Abdomen WO W - NC.A.V Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the
CT Pancreas 3 Phase CT Abdomen WO W - NC.A.V Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the
Billing Guide. For Freestanding Imaging Centers Provided by Bayer HealthCare
 Billing Guide For Freestanding Imaging Centers Provided by Bayer HealthCare Introduction Bayer HealthCare Inc. offers this billing guide to freestanding imaging center customers as part of its comprehensive
Billing Guide For Freestanding Imaging Centers Provided by Bayer HealthCare Introduction Bayer HealthCare Inc. offers this billing guide to freestanding imaging center customers as part of its comprehensive
Gadolinium Retention in the Dentate Nucleus and Globus Pallidus Is Dependent on the Class of Contrast Agent 1
 Note: This copy is for your personal non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, contact us at www.rsna.org/rsnarights. Alexander Radbruch,
Note: This copy is for your personal non-commercial use only. To order presentation-ready copies for distribution to your colleagues or clients, contact us at www.rsna.org/rsnarights. Alexander Radbruch,
Review of Hepatobiliary Contrast Agents: Current Applications and Challenges
 REVIEW Review of Hepatobiliary Contrast Agents: Current Applications and Challenges Alex Frydrychowicz, M.D.*, The group of hepatobiliary contrast agents comprises two gadolinium-based contrast agents
REVIEW Review of Hepatobiliary Contrast Agents: Current Applications and Challenges Alex Frydrychowicz, M.D.*, The group of hepatobiliary contrast agents comprises two gadolinium-based contrast agents
PLEASE SCROLL DOWN FOR ARTICLE
 This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
This article was downloaded by:[danish Veterinary and Agricultural Library] [Danish Veterinary and Agricultural Library] On: 24 June 2007 Access Details: [subscription number 773444395] Publisher: Informa
Update. Drug Safety. Welcome to the first issue of the newly launched Drug Safety Update. This
 Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
Latest advice for medicines users The monthly newsletter from the Medicines and Healthcare products Regulatory Agency and its independent advisor the Commission on Human Medicines Volume 1, Issue 1 August
Renal artery disease: MR Techniques and Interpretation
 Renal artery disease: MR Techniques and Interpretation Prof. Dr. Stefan O. Schoenberg Professor and Chairman of Radiology Department of Clinical Radiology and Nuclear Medicine University Medicine Mannheim
Renal artery disease: MR Techniques and Interpretation Prof. Dr. Stefan O. Schoenberg Professor and Chairman of Radiology Department of Clinical Radiology and Nuclear Medicine University Medicine Mannheim
Contrast. 5 Oral Contrast for CT...13
 2 Contrast 1 GI Fluoroscopic Contrast...10 Adult Esophagram/Upper GI... 10 Pediatric Upper GI... 10 Enteroclysis... 10 Double-Contrast Barium Enema... 10 Single-Contrast Barium Enema... 10 Rectum and Pelvic
2 Contrast 1 GI Fluoroscopic Contrast...10 Adult Esophagram/Upper GI... 10 Pediatric Upper GI... 10 Enteroclysis... 10 Double-Contrast Barium Enema... 10 Single-Contrast Barium Enema... 10 Rectum and Pelvic
CT Renal 3 Phase + Pelvis CT Abdomen Pelvis WO W - NC.A.V, Pelvis during V
 CT Renal 3 Phase + Pelvis CT Abdomen Pelvis WO W - NC.A.V, Pelvis during V Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance
CT Renal 3 Phase + Pelvis CT Abdomen Pelvis WO W - NC.A.V, Pelvis during V Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance
Val M. Runge, MD Editor-in-Chief Investigative Radiology
 Val M. Runge, MD Editor-in-Chief Investigative Radiology Patients First RSNA 2012 The classical model of medical care which portrays the authoritative physician evaluating and treating an obedient, non-inquisitive
Val M. Runge, MD Editor-in-Chief Investigative Radiology Patients First RSNA 2012 The classical model of medical care which portrays the authoritative physician evaluating and treating an obedient, non-inquisitive
Contrast Media Guidelines. To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
 APPROVED BY: Director of Radiology Page 1 of 6 Purpose: Contrast Media Guidelines To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
APPROVED BY: Director of Radiology Page 1 of 6 Purpose: Contrast Media Guidelines To define general guidelines for the use of contrast media so that it is used appropriately and safely in imaging studies.
CT Chest HRCT CT Chest WO
 CT Chest HRCT CT Chest WO Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA policies and protocols
CT Chest HRCT CT Chest WO Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA policies and protocols
CTA Pulmonary Embolism CTA Chest W (arterial)
 CTA Pulmonary Embolism CTA Chest W (arterial) Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the
CTA Pulmonary Embolism CTA Chest W (arterial) Reviewed By: Rachael Edwards, MD; Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: January 2019 Contact: (866) 761-4200, Option 1 In accordance with the
Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting
 European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
European Journal of Radiology 66 (2008) 230 234 Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: A summary of the medical literature reporting Dale R. Broome Department of
CTA Chest Pulmonary Embolism & Routine CT Abdomen + Pelvis W CTA Chest W (arterial) & CT Abdomen + Pelvis W (venous)
 CTA Chest Pulmonary Embolism & Routine CT Abdomen + Pelvis W CTA Chest W (arterial) & CT Abdomen + Pelvis W (venous) Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact:
CTA Chest Pulmonary Embolism & Routine CT Abdomen + Pelvis W CTA Chest W (arterial) & CT Abdomen + Pelvis W (venous) Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact:
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, 2 july 2008
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, 2 july 2008
Routine CT Chest + Abdomen WO
 Routine CT Chest + Abdomen WO Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA policies and protocols
Routine CT Chest + Abdomen WO Reviewed By: Anna Ellermeier, MD; Brett Mollard, MD Last Reviewed: August 2018 Contact: (866) 761-4200, Option 1 In accordance with the ALARA principle, TRA policies and protocols
End-Stage Renal Disease. Anna Vinnikova, M.D. Associate Professor of Medicine Division of Nephrology
 End-Stage Renal Disease Anna Vinnikova, M.D. Associate Professor of Medicine Division of Nephrology ESRD : Life with renal replacement therapy CASE: 18 month old male with HUS develops ESRD PD complicated
End-Stage Renal Disease Anna Vinnikova, M.D. Associate Professor of Medicine Division of Nephrology ESRD : Life with renal replacement therapy CASE: 18 month old male with HUS develops ESRD PD complicated
Safety of gadolinium contrast angiography in patients with chronic renal insufficiency
 Safety of gadolinium contrast angiography in patients with chronic renal insufficiency Albert D. Sam II, MD, a Mark D. Morasch, MD, a Jeremy Collins, MD, b Gina Song, MD, b Richard Chen, MD, b and F. Scott
Safety of gadolinium contrast angiography in patients with chronic renal insufficiency Albert D. Sam II, MD, a Mark D. Morasch, MD, a Jeremy Collins, MD, b Gina Song, MD, b Richard Chen, MD, b and F. Scott
DATA SHEET. GADOVIST 1.0 is available as a 1.0 mmol/ml solution and each ml of GADOVIST 1.0 contains mg gadobutrol.
 DATA SHEET 1. PRODUCT NAME (strength pharmaceutical form) GADOVIST 1.0 (1.0 mmol/ml) Solution for Intravenous Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Gadobutrol 604.72 mg/ml (1.0 mmol/ml)
DATA SHEET 1. PRODUCT NAME (strength pharmaceutical form) GADOVIST 1.0 (1.0 mmol/ml) Solution for Intravenous Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Gadobutrol 604.72 mg/ml (1.0 mmol/ml)
Contrast controversies and confusion
 Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
Contrast controversies and confusion Mariya Kobi 1 ; Meir H Scheinfeld 1 ; Seymour Sprayregen 1 ; R Joshua Dym 2 * 1 Department of Radiology, Montefiore Medical Center, USA 2 Department of Radiology, Rutgers
Dialysis Initiation and Optimal Vascular Access: Outcomes and Mortality
 Dialysis Initiation and Optimal Vascular Access: Outcomes and Mortality Shannon H. Norris, BSN, RN June 6, 2018 Dialysis Initiation and Optimal Vascular Access: Outcomes and Mortality DISCUSSION: End Stage
Dialysis Initiation and Optimal Vascular Access: Outcomes and Mortality Shannon H. Norris, BSN, RN June 6, 2018 Dialysis Initiation and Optimal Vascular Access: Outcomes and Mortality DISCUSSION: End Stage
flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric patients. The dose is delivered by manual or power injection.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Update in Nephrology. Case: Question 1. Case presentation. Acute Kidney Injury. For her hypertension management, you decide to:
 Update in Nephrology Chronic Kidney Disease Renoprotection and Proteinuria, ACE and/or ARB Anemia management Update in Nephrology Renal artery stenosis Nephrogenic systemic fibrosis Division of Nephrology
Update in Nephrology Chronic Kidney Disease Renoprotection and Proteinuria, ACE and/or ARB Anemia management Update in Nephrology Renal artery stenosis Nephrogenic systemic fibrosis Division of Nephrology
CONTRAINDICATIONS Clinically important hypersensitivity reactions to DOTAREM. (4)
 M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Chronic Kidney Disease. Dr Mohan B. Biyani A. Professor of Medicine University of Ottawa/Ottawa Hospital
 Chronic Kidney Disease Dr Mohan B. Biyani A. Professor of Medicine University of Ottawa/Ottawa Hospital Health Seminar Series Date 12 May 2013 Objectives Normal functioning of Kidneys. Risk factors to
Chronic Kidney Disease Dr Mohan B. Biyani A. Professor of Medicine University of Ottawa/Ottawa Hospital Health Seminar Series Date 12 May 2013 Objectives Normal functioning of Kidneys. Risk factors to
Innovations in HCC Imaging: MDCT/MRI
 Innovations in HCC Imaging: MDCT/MRI Anthony E. Cheng, M.D. Cardinal MRI Center Cardinal Santos Medical Center, Wilson Street, San Juan Innovations in HCC Imaging: Goals/Objectives MDCT/MRI Learn the diagnostic
Innovations in HCC Imaging: MDCT/MRI Anthony E. Cheng, M.D. Cardinal MRI Center Cardinal Santos Medical Center, Wilson Street, San Juan Innovations in HCC Imaging: Goals/Objectives MDCT/MRI Learn the diagnostic
Reference ID: MultiHance Injection, NDA , SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov _CLEAN.
 MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
Lab Values Explained. working at full strength. Other possible causes of an elevated BUN include dehydration and heart failure.
 Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Special Instructions
 FDA and ACR guidelines are as follows: Special Instructions Safety concerning NSF and gadolinium-based contrast agents (GBCA) Prior to administering MRI contrast (GBCA), any patient who answers yes to
FDA and ACR guidelines are as follows: Special Instructions Safety concerning NSF and gadolinium-based contrast agents (GBCA) Prior to administering MRI contrast (GBCA), any patient who answers yes to
