Phoenix Atherectomy System Product Overview /LB
|
|
|
- Octavia Clark
- 5 years ago
- Views:
Transcription
1 Phoenix Atherectomy System Product Overview
2 Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems to tailor the treatment for each patient. Front cutting for direct lesion access Plaque removal Directional cutting ability* Single insertion No need for capital equipment Hybrid Directional Laser Orbital Rotational 16 *Available with Phoenix 2.4 deflecting catheter.
3 The Phoenix Solution Phoenix was created to address physicians clinical concerns and challenges. 1. Endovascular Atherectomy Safety and Effectiveness Study (EASE), ClinicalTrials.gov Identifier NCT (accessed 23Oct2015). Results presented at the Vascular Interventional Advances (VIVA) Conference in October of 2013 (Las Vegas, NV) by Stephen Williams, MD 2. Phoenix Atherectomy device is indicated for vessels 2.5mm in diameter and above 3
4 Phoenix System Components 1. Atherectomy Catheter Distal cutting element rotates at 10,000-12,000 RPM Cutting element made of stainless steel alloy with proprietary carbon coating 2. Battery-Powered Handle Powers rotation of cutting element Small, handheld form factor Compatible with all Phoenix catheter sizes IFU recommended maximum run time is 20 minutes
5 Phoenix System Components 3. Wire Support Clip Serves as system brake Provides for single user operation Integrated torque clip 4. Debris collection bag External bag collects debulked material as it is continuously captured and cleared from body
6 First Hybrid Atherectomy Catheter System Tracking (non-deflecting) Catheters (1.8mm and 2.2mm) OTW system 130cm and 149cm working length Tracks directly over the guidewire- this helps keep catheter centered in vessel lumen (when guidewire is centered) Rotational front cutter restores straight line flow to the foot 1 Deflecting Catheter (2.4mm) OTW system 127cm working length (straight) Combines key features of non-deflecting, tracking catheters with deflection capability Cut initial straight channel, then deflect and rotate catheter tip as needed to debulk larger diameter and/or target eccentric lesions 1. Phoenix Atherectomy Device is indicated for vessels 2.5mm in diameter and above 6
7 Phoenix Indications for Use Indication: The Phoenix Atherectomy System is intended for use in atherectomy of the peripheral vasculature. The system is not intended for use in the coronary, carotid, iliac or renal vasculature. 7
8 Cut, Capture and Clear Mechanism of Action Cut: Front cutter clears tissue in a way that may help reduce potential trauma to the vessel. 1 Capture: Unique cutter head design allows for continuous capture of debulked material. Clear: The internal Archimedes screw allows you to clear plaque without having to remove the catheter and clean out debulked material. 1. Endovascular Atherectomy Safety and Effectiveness Study (EASE), ClinicalTrials.gov Identifier NCT (accessed 23Oct2015). Results presented at the Vascular Interventional Advances (VIVA) Conference in October of 2013 (Las Vegas, NV) by Stephen Williams, MD.
9 Cutting Element Proprietary carbon coating on stainless steel alloy Rotation of blades debulks diseased material Shape of blades serves as impeller to bring material into catheter housing Two flutes on distal cutter 2 nd stage cutter located within housing has 4 flutes Further macerates plaque to facilitate conveyance via Archimedes Screw 9
10 Product Prep
11 Prepping the Phoenix System Simple set-up, prep is similar to other interventional devices Flush wire lumen Attach 10cc syringe filled with heparinized saline to disposal port Flush slowly until saline drips out of proximal guidewire lumen Occlude guidewire port, and flush until saline drips out of distal tip of guidewire lumen Snap catheter into handle Submerge catheter tip into bowl of heparinized saline Turn system ON until saline drips out of disposal port 11 DEVICE PREP IS THE SAME FOR ALL PHOENIX DEVICES
12 Prepping the Phoenix System Simple set-up, prep is similar to other interventional devices Attach disposal bag Attach disposal bag to catheter by pushing tubing into disposal port Assemble catheter Snap integrated wire support clip onto handle Feed wire through the torque device Form a cm support loop that spans from the guidewire exit port of the catheter to the wire support clip 12 Do not use any device found to be broken or non-functional during the preparation steps
13 1.8 and 2.2. Tracking Catheter Product Handling
14 Suggested Tracking Catheter Case Workflow Insertion Insert the appropriately-sized sheath with a cross-cut hemostasis valve using standard techniques Advance a guidewire through the sheath beyond the lesion to be treated, taking care to remain intraluminal Backload the end of the guidewire into the distal tip and out of the proximal end of the Phoenix Atherectomy Catheter guidewire lumen. Insert the distal tip of the Phoenix Atherectomy Catheter into the introducer sheath with the Phoenix System OFF until the tip exits the introducer sheath 14
15 Suggested Tracking Catheter Case Workflow Crossing the lesion and debulking While under fluoroscopy, advance catheter over guidewire until it is positioned proximally to the lesion Confirm that the guidewire is intraluminal, confirm that the distal tip of the guidewire is positioned a minimum of 20 cm from distal tip of Catheter Advancing device Under fluoroscopic guidance, turn system ON using switch on handle Advance catheter at a rate of 1mm/sec carefully through the lesion In highly stenosed lesions or lesions 10cm in length, periodically pause and withdraw the Catheter slightly to allow improved blood flow and plaque removal during cutting. The Phoenix Atherectomy System must remain ON to remove plaque Withdraw the catheter until the distal tip is proximal to the lesion. Image the lumen and repeat cutting through the lesion if desired. 15
16 2.4mm Deflecting Catheter Product Handling
17 Deflecting Mechanism of Action 2.4mm deflecting catheter with nitinol tip outer deflecting sleeve 1. Straight cutting with outer sleeve retracted 2. Deflected cutting with outer sleeve advanced toward tip 17
18 Phoenix 2.4mm Deflecting Mechanism of Action Non-Deflected (Straight) 127 cm working length Straight cutting with slider mechanism in back position Deflected 125 cm working length Rotation knob Deflected cutting with slider mechanism in forward position 18 In deflected position, rotation knob controls tip direction/angle of deflection for directional debulking 360 o of rotation provided through 8 detents or clicks Knob can be rotated in either direction Square shape design makes rotation easier
19 Suggested 2.4 mm Case Workflow Crossing the Lesion and Debulking (Straight Cutting) 1. Verify the catheter is in the Straight Position by moving the slider fully proximal before inserting the catheter into the introducer sheath. 2. While holding the guidewire stationary and using fluoroscopic guidance, advance the Phoenix Atherectomy Catheter distal tip to within a few millimeters proximal to the target lesion. 3. Adjust the guidewire position relative to the lesion as needed. Confirm that the guidewire is intraluminal. 4. Confirm that the distal tip of the wire is positioned a minimum of 20 cm from the distal tip of the catheter. 5. Under fluoroscopic guidance, turn ON the Phoenix Atherectomy System using the switch on the handle. Advance the catheter slowly at a rate of 1 mm/sec and carefully through the lesion. In highly stenosed lesions or lesions > 10 cm in length, periodically pause and withdraw the Catheter slightly to allow improved blood flow and plaque removal during cutting. Continue to advance until the distal tip of catheter has crossed the lesion. 19
20 Suggested 2.4 mm Case Workflow 6. The Phoenix Atherectomy System must remain ON to remove plaque. 7. Withdraw the catheter until the distal tip is proximal to the lesion. Image the lumen and/or repeat straight cutting through the lesion if desired. 8. Turn OFF the Phoenix Atherectomy System with the switch on the handle. 20
21 Suggested 2.4 mm Case Workflow Debulking to Larger Diameter (Deflected Cutting) 1. When moving to deflected cutting, the use of a flexible ( light ) guidewire allows maximum deflection of the Catheter tip. Exchange guidewire if desired. 2. While holding the guidewire stationary and using fluoroscopic guidance, advance the Phoenix Atherectomy Catheter distal tip to within a few millimeters proximal to the target lesion. 3. To Deflect: Using the Slider, slide the outer sheath distal to increase deflection (bend tip) and backward to decrease deflection (straighten tip). The slider features a trigger lock to maintain selected position. 4. To Rotate: Adjust the position of the catheter tip by turning the knob on the outer sheath clockwise or counterclockwise. As the knob is rotated, a tactile click will be felt by the user. A 360 degree rotation of the Catheter tip can be achieved with 8 clicks of the knob. If there is resistance to rotating the tip, decrease deflection (straighten tip) prior to rotating and then re-adjust to desired deflection. 5. Under fluoroscopic guidance, turn ON the Phoenix Atherectomy System. Warning: When the catheter is deflected and the System is ON, do not leave the cutter head stationary or perforation may occur. 21
22 Suggested 2.4 mm Case Workflow Debulking to Larger Diameter (Deflected Cutting) 6. The catheter may be advanced and retracted while at a fixed deflection setting to debulk. Advance the catheter slowly and carefully while debulking. Always monitor the catheter tip deflection position during cutting, in order to ensure the setting does not need to be adjusted as debris is removed and there is less resistance to deflection. Rotate and/or reposition the catheter tip as desired during cutting or between passes. If there is resistance to rotating the tip, decrease deflection (straighten tip). 7. The Phoenix Atherectomy System must remain ON in order to effectively remove plaque. 8. Once the lumen is opened up to the maximum diameter desired, turn OFF the Phoenix Atherectomy System. 9. Retract the Catheter at least 1 cm proximal to the lesion. 10. Perform an angiogram to assess the lumen. 11. Continue debulking if desired and reassess the lumen with an angiogram. 22
23 Points to Consider: Using the Catheter Recognize that cutter head will retract proximally by 2cm when moving from straight to deflected mode (from 127cm working length to 125cm working length) Pay attention to ability of cutter head to rotate In extreme cases of iliac tortuosity or in situations with smaller vessel diameters 1, rotation of cutter head can be impeded when in deflected mode In these situations above, the catheter tip should be placed in straight mode for rotation on distal side of the lesion. Interaction with tortuosity can be overcome if the knob is rotated with cutter head in straight mode mm Phoenix Atherectomy Device is indicated for vessels 3mm in diameter and above 23
24 Suggested Points to Consider: 2.4mm Using Case the Workflow/Cont d Catheter The 2.4mm reduced profile device can cut forward, and, when deflected, backwards through a lesion 2.4mm Deflection Catheter device should be removed only in straight mode through the vessel and should not be run when going through the sheath, particularly through the aortic bifurcation 24
25 Ordering Information
26 Product Offering and Part Numbering Nomenclature P18130K 1.8mm Phoenix Atherectomy System kit, 130cm length P22130K 2.2mm Phoenix Atherectomy System kit, 130cm length P18149K 1.8mm Phoenix Atherectomy System kit, 149cm length P22149K 2.2mm Phoenix Atherectomy System kit, 149cm length PD24127K 2.4mm Phoenix Atherectomy System kit, Deflecting, 127cm length PG14300LF Phoenix Light Support Guidewire, 300cm length P = Phoenix, D = Deflecting 1.8mm, 2.2mm, or 2.4mm = Cutter head diameter 130cm, 149cm, or 127mm = Device working length K = Kit (catheter, handle, clip and bag) 26
27 Guidewires
28 Positioning the Phoenix Guidewire Compatible with the Phoenix Atherectomy System Use with deflecting and tracking catheters Simple to Use Catheter is directed over the guidewire with ease Light support allows the 2.4mm deflecting Phoenix Catheter to deflect over the wire Efficient Vessel Navigation Provides tactile feedback needed to help with vessel navigation Design of wire allows for torque and rotation required to navigate to target lesion
29 Guidewire Selection Physician may select a flexible or light support guidewire when: the 2.4 mm deflecting Phoenix catheter is being used navigating extreme tortuosity Physician may select a supportive or extra supportive guidewire when working distally extra pushability would be helpful for vessel navigation attempting to straighten a tortuous segment and prevent possible vessel interaction the 2.4 mm deflecting catheter is used in non-deflected mode
30 0.014 Guidewires Compatible for Use with the Phoenix Atherectomy System Tracking Catheters 1 Brand Product Length Part Number Wire Support Level Philips Volcano Phoenix Guidewire- Light Support 300 cm PG14300LF Light Support Ev3 Nitrex Guidewire 300 cm N Flexible Asahi Regalia XS1 300 cm PAGP Medium Abbott Vascular Hi-Torque Extra S PORT 300 cm 22235M Supportive Abbott Vascular Hi-Torque Iron Man 300 cm Supportive Abbott Vascular Hi-Torque Standard 300cm 22320H-901 Supportive Asahi Astato XS cm PAGH Supportive CSI VIPER 335cm VPR-GW-14 Supportive Abbott Vascular Hi-Torque Floppy II ES 300cm 22359M Extra Cook Roadrunner 300 cm G08801 RSTF Extra Abbott Vascular Hi-Torque Cross-IT 300cm H CTO (1) Test performed on the bench model.
31 0.014 Guidewires Compatible for Use with the Phoenix Atherectomy System 2.4 mm Deflecting Catheter 1 Brand Product Length Part Number Wire Support Level Philips Volcano Phoenix Guidewire- Light Support 300 cm PG14300LF Light Support Ev3 Nitrex Guidewire 300 cm N Flexible US Endovascular Nitinol Mandrel Wire 300cm USEGS14-300A Flexible Flexible guidewire required to facilitate deflection Using a stiff guidewire will resist deflection and straighten out catheter tip (1) Test performed on the bench model.
32 Phoenix Light Support Guidewire Information Guidewire Data Wire is silicone coated with nitinol core Part number is PG14300LF Diameter:.014 Length: 300cm Tip: 5.5cm Style: Light support Tip Load: 1 to 2 gr For use with the Phoenix tracking and deflecting catheters 32
2.4 mm deflecting 2.4 mm tracking Above the knee Phoenix Below the knee 2.2 mm tracking 1.8 mm tracking
 Deep femoral artery Superficial femoral artery Popliteal artery Phoenix Above the knee Below the knee Atherectomy system Anterior tibial artery Peroneal artery Posterior tibial artery Dorsalis pedis artery
Deep femoral artery Superficial femoral artery Popliteal artery Phoenix Above the knee Below the knee Atherectomy system Anterior tibial artery Peroneal artery Posterior tibial artery Dorsalis pedis artery
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
ROTABLATORTM. Peripheral. Rotational Atherectomy System. Quick Reference Cards
 Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Guide Wires design and selection Abbott Vascular. All rights reserved.
 Guide Wires design and selection the cardiac catheter was...the key in the lock Cournand AF, Nobel lecture December 11, 1956; nowadays, the coronary guidewire is the master key to all locks Colombo et
Guide Wires design and selection the cardiac catheter was...the key in the lock Cournand AF, Nobel lecture December 11, 1956; nowadays, the coronary guidewire is the master key to all locks Colombo et
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Guidewire Selection. Making the Most Out of My Guidewire: LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016
 Making the Most Out of My Guidewire: Guidewire Selection LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016 Brian DeRubertis, MD, FACS Associate Professor of Surgery Division
Making the Most Out of My Guidewire: Guidewire Selection LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016 Brian DeRubertis, MD, FACS Associate Professor of Surgery Division
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
My idea on intimal tracking guide wire selection for Pop-BTK treatment
 LINC 2017 Leipzig January 24-27, 2017 My idea on intimal tracking guide wire selection for Pop-BTK treatment Jos C. van den Berg, MD PhD Ospedale Regionale di Lugano, sede Civico Lugano University of Bern
LINC 2017 Leipzig January 24-27, 2017 My idea on intimal tracking guide wire selection for Pop-BTK treatment Jos C. van den Berg, MD PhD Ospedale Regionale di Lugano, sede Civico Lugano University of Bern
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Galt Medical Corp. A leader in vascular and interventional medical devices. part of the GaltNeedleTech TM Family
 Galt Medical Corp. A leader in vascular and interventional medical devices. part of the GaltNeedleTech TM Family MICRO-INTRODUCERS Micro-Introducer Kits Galt s Micro-Introducer Kits with smooth transition,
Galt Medical Corp. A leader in vascular and interventional medical devices. part of the GaltNeedleTech TM Family MICRO-INTRODUCERS Micro-Introducer Kits Galt s Micro-Introducer Kits with smooth transition,
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
TJF-160F/VF Cleaning and Disinfection Checklist
 TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Appropriate Device Selection for Endovascular Procedures
 Appropriate Device Selection for Endovascular Procedures Thomas M. Shimshak, MD Florida Hospital Heartland Medical Center Sebring, Florida Disclosures Speaker s Bureau: Abbott Vascular Boston Scientific
Appropriate Device Selection for Endovascular Procedures Thomas M. Shimshak, MD Florida Hospital Heartland Medical Center Sebring, Florida Disclosures Speaker s Bureau: Abbott Vascular Boston Scientific
We Catch. What Others Miss
 We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Antegrade techniques for CTO recanalization. Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist
 Antegrade techniques for CTO recanalization Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist can CTOs be reliably opened by PCI? Meta-Analysis of 18,061 Patients Patel V, J Am Coll
Antegrade techniques for CTO recanalization Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist can CTOs be reliably opened by PCI? Meta-Analysis of 18,061 Patients Patel V, J Am Coll
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system.
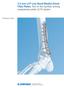 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
Optimal Techniques for Obtaining Large Caliber Arterial Access
 Optimal Techniques for Obtaining Large Caliber Arterial Access Gerald Yong MBBS (Hons) FRACP FSCAI Interventional Cardiologist Royal Perth Hospital Western Australia APCASH 11 October 2014 Disclosure Statement
Optimal Techniques for Obtaining Large Caliber Arterial Access Gerald Yong MBBS (Hons) FRACP FSCAI Interventional Cardiologist Royal Perth Hospital Western Australia APCASH 11 October 2014 Disclosure Statement
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation.
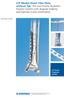 LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Chronic total occlusions (CTOs) are frequently
 Specialty Crossing Devices: Understanding the Learning Curve Technical pearls and new devices for crossing peripheral CTOs. y Usman Javed, MD, and John R. Laird, MD (Courtesy of ard Peripheral Vascular,
Specialty Crossing Devices: Understanding the Learning Curve Technical pearls and new devices for crossing peripheral CTOs. y Usman Javed, MD, and John R. Laird, MD (Courtesy of ard Peripheral Vascular,
Valencia Pedicle Screw Surgical Technique
 Valencia Pedicle Screw Surgical Technique VALENCIA CIRCUIT TABLE OF CONTENTS Design Rationale Indications for Use Surgical Technique 1. Pedicle Preparation 2. Screw Insertion 3. Rod Placement 4. Locking
Valencia Pedicle Screw Surgical Technique VALENCIA CIRCUIT TABLE OF CONTENTS Design Rationale Indications for Use Surgical Technique 1. Pedicle Preparation 2. Screw Insertion 3. Rod Placement 4. Locking
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Home Health Foundation, Inc. To create more permanent IV access for patients undergoing long term IV therapy.
 PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
Conventus CAGE PH Surgical Techniques
 Conventus CAGE PH Surgical Techniques Conventus Orthopaedics The Conventus CAGE PH (PH Cage) is a permanent implant comprised of an expandable scaffold, made from nitinol and titanium, which is deployed
Conventus CAGE PH Surgical Techniques Conventus Orthopaedics The Conventus CAGE PH (PH Cage) is a permanent implant comprised of an expandable scaffold, made from nitinol and titanium, which is deployed
Penumbra INTRACRANIAL ACCESS REDEFINED
 Penumbra INTRACRANIAL ACCESS REDEFINED BENCHMARK Case Examples Stability & Compatibility Atraumatic Navigation Flow Diverter Delivery Pipeline BENCHMARK Stability Without Surpass Full distal shaft radiopacity
Penumbra INTRACRANIAL ACCESS REDEFINED BENCHMARK Case Examples Stability & Compatibility Atraumatic Navigation Flow Diverter Delivery Pipeline BENCHMARK Stability Without Surpass Full distal shaft radiopacity
Catheters and Wires. Dr. Vaibhav Jain MD,DNB,MNAMS Senior Consultant, IR Medanta, the Medicity, Gurgaon
 Catheters and Wires Dr. Vaibhav Jain MD,DNB,MNAMS Senior Consultant, IR Medanta, the Medicity, Gurgaon Guidewires: Guidewires (solid wires navigated within the vascular system / extravascular tract) act
Catheters and Wires Dr. Vaibhav Jain MD,DNB,MNAMS Senior Consultant, IR Medanta, the Medicity, Gurgaon Guidewires: Guidewires (solid wires navigated within the vascular system / extravascular tract) act
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
Chronic Total Occlusion (CTO) Technologies. Re-open vital channels
 Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
Tissue Acquisition. Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract.
 Tissue Acquisition Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract. MAKING A DIFFERENCE TO HEALTH Infinity ERCP Sampling Device STIFFER
Tissue Acquisition Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract. MAKING A DIFFERENCE TO HEALTH Infinity ERCP Sampling Device STIFFER
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
There are multiple endovascular options for treatment
 Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
Indications. The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli
 Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
Ancillary Components with Z-Trak Introduction System
 Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Copyright HMP Communications
 Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Low Bend Distal Tibia Plates
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
PRODUCTS FOR THE DIFFICULT AIRWAY. Courtesy of Cook Critical Care
 PRODUCTS FOR THE DIFFICULT AIRWAY Courtesy of Cook Critical Care EMERGENCY CRICOTHYROTOMY Thyroid Cartilage Access Site Cricoid Cartilage Identify the cricothyroid membrane between the cricoid and thyroid
PRODUCTS FOR THE DIFFICULT AIRWAY Courtesy of Cook Critical Care EMERGENCY CRICOTHYROTOMY Thyroid Cartilage Access Site Cricoid Cartilage Identify the cricothyroid membrane between the cricoid and thyroid
Cook Medical. Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
 Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
New Devices Pedro Pinto Cardoso
 New Devices Pedro Pinto Cardoso HSM, CHLN, CALM for CTOs Guidewires Microcatheters Radiofrequency Pharmacotherapy Pedro Pinto Cardoso, Serviço de Cardiologia HSM CHLN New Guidewires SENTAI family Samurai
New Devices Pedro Pinto Cardoso HSM, CHLN, CALM for CTOs Guidewires Microcatheters Radiofrequency Pharmacotherapy Pedro Pinto Cardoso, Serviço de Cardiologia HSM CHLN New Guidewires SENTAI family Samurai
TRANSRADIAL PERIPHERAL VASCULAR INTERVENTIONS
 TRANSRADIAL PERIPHERAL VASCULAR INTERVENTIONS Cezar Staniloae NYU Heart and Vascular Institute May 27, 2015 Radial Artery is an Ideal Acess Site Easily accessible even in subjects with severe PVD Major
TRANSRADIAL PERIPHERAL VASCULAR INTERVENTIONS Cezar Staniloae NYU Heart and Vascular Institute May 27, 2015 Radial Artery is an Ideal Acess Site Easily accessible even in subjects with severe PVD Major
I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.*
 I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.* Edwards ThruPort Systems The complete system for minimal incision valve surgery (MIVS) Prep Guide MIVS Redefined > THRUPORT SYSTEMS
I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.* Edwards ThruPort Systems The complete system for minimal incision valve surgery (MIVS) Prep Guide MIVS Redefined > THRUPORT SYSTEMS
TABLE OF CONTENTS. 2 (8144 Rev 2)
 1 (8144 Rev 2) TABLE OF CONTENTS Introduction Conventus CAGE TM - Proximal Humerus...3 Indications and Contraindications...4 Surgical Summary...5 Patient Positioning & Approach...6 Surgical Technique Plate
1 (8144 Rev 2) TABLE OF CONTENTS Introduction Conventus CAGE TM - Proximal Humerus...3 Indications and Contraindications...4 Surgical Summary...5 Patient Positioning & Approach...6 Surgical Technique Plate
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
PFNA. With Augmentation Option.
 PFNA. With Augmentation Option. Superior anchor in osteoporotic bone Simple and reproducible procedure through standardized technique Intraoperative decision for augmentation PFNA. Proximal Femoral Nail
PFNA. With Augmentation Option. Superior anchor in osteoporotic bone Simple and reproducible procedure through standardized technique Intraoperative decision for augmentation PFNA. Proximal Femoral Nail
PFNA. With Augmentation Option.
 PFNA. With Augmentation Option. Superior anchor in osteoporotic bone Simple and reproducible procedure through standardized technique Intraoperative decision for augmentation This publication is not intended
PFNA. With Augmentation Option. Superior anchor in osteoporotic bone Simple and reproducible procedure through standardized technique Intraoperative decision for augmentation This publication is not intended
limbsandthings.com Advanced Catheterisation Trainer User Guide For more skills training products visit Limbs & Things Ltd.
 Advanced Catheterisation Trainer Product No: 60150 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
Advanced Catheterisation Trainer Product No: 60150 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
Percutaneous Leads. Directions for Use Content: REV A
 Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
ENG TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS
 TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
PCI for Chronic Total Occlusions
 PCI for Chronic Total Occlusions Chronic Total Occlusions Why not Medical Treatment? Medical Treatment CTO in 891 pts over 24 years High 10% Mortality Low 2 % 1 year 10 years Puma JA, et al. JACC 1994;23:390A
PCI for Chronic Total Occlusions Chronic Total Occlusions Why not Medical Treatment? Medical Treatment CTO in 891 pts over 24 years High 10% Mortality Low 2 % 1 year 10 years Puma JA, et al. JACC 1994;23:390A
CTO: Technique and Tools
 CTO: Technique and Tools S. Hinan Ahmed, MD Associate Professor: Cardiology and Cardiothoracic Surgery Program Director: Interventional Fellowship Program Associate Editor: Cath Cardiov Interventions UT
CTO: Technique and Tools S. Hinan Ahmed, MD Associate Professor: Cardiology and Cardiothoracic Surgery Program Director: Interventional Fellowship Program Associate Editor: Cath Cardiov Interventions UT
3.5 mm Locking Attachment Plate
 For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS
 SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS A NEW STANDARD FOR SMOOTH VASCULAR ACCESS NEEDLE PENETRATION FORCE Needle penetration was measured by the amount of force it took for the needle to
SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS A NEW STANDARD FOR SMOOTH VASCULAR ACCESS NEEDLE PENETRATION FORCE Needle penetration was measured by the amount of force it took for the needle to
Device Evolution. Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance. Where Do We Stand? 4/18/2015
 Disclosure Statement of Financial Interest Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Disclosure Statement of Financial Interest Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Transjugular liver access and biopsy
 Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Distal Radius Surgical Technique Guide
 Distal Radius Surgical Technique Guide About IlluminOss IlluminOss minimally invasive procedure incorporates a PET or Dacron balloon catheter that is delivered through a small pathway into the intramedullary
Distal Radius Surgical Technique Guide About IlluminOss IlluminOss minimally invasive procedure incorporates a PET or Dacron balloon catheter that is delivered through a small pathway into the intramedullary
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures.
 LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
ENDO-EZE TM GENIUS ENDODONTIC SYSTEM WHERE SAFETY MEETS EFFICIENCY
 ENDO-EZE TM GENIUS ENDODONTIC SYSTEM WHERE SAFETY MEETS EFFICIENCY WHERE SAFETY MEETS EFFICIENCY Clinicians have always had to choose between reciprocation and rotary; safety and efficiency. So which do
ENDO-EZE TM GENIUS ENDODONTIC SYSTEM WHERE SAFETY MEETS EFFICIENCY WHERE SAFETY MEETS EFFICIENCY Clinicians have always had to choose between reciprocation and rotary; safety and efficiency. So which do
Versatility and Proven Safety
 Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
WallFlex Biliary Metal Stents
 WallFlex Biliary Metal Stents Please Note: These steps are intended to be for general guidance only. For complete instructions for use, indications, contraindications and warnings, please refer to the
WallFlex Biliary Metal Stents Please Note: These steps are intended to be for general guidance only. For complete instructions for use, indications, contraindications and warnings, please refer to the
TWISTR TM Retrograde Reamer & Cruciate+ Instruments Surgical Technique
 TWISTR TM Retrograde Reamer & Cruciate+ Instruments Surgical Technique Guide Carriage Suture Passing Pin Step 1: Set the TWISTR TM Retrograde Reamer to drill the desired socket / tunnel diameter by twisting
TWISTR TM Retrograde Reamer & Cruciate+ Instruments Surgical Technique Guide Carriage Suture Passing Pin Step 1: Set the TWISTR TM Retrograde Reamer to drill the desired socket / tunnel diameter by twisting
Technique of the VenaSeal System
 Technique of the VenaSeal System Indication & Contraindications VenaSeal Indication: The Sapheon VenaSeal System is intended for the permantent, complete, endovascular adhesive closure of the great saphenous
Technique of the VenaSeal System Indication & Contraindications VenaSeal Indication: The Sapheon VenaSeal System is intended for the permantent, complete, endovascular adhesive closure of the great saphenous
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
OBSOLETED. LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation.
 LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Surgical Technique LCP Small Fragment System This publication
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Surgical Technique LCP Small Fragment System This publication
Effectiveness of IVUS in Complex Cases
 Effectiveness of IVUS in Complex Cases Satoru Sumituji,M.D. Rinku General Medical Center IVUS is can provide images of the vessel wall and the tissue around the vessel which cannot be viewed by angiography.
Effectiveness of IVUS in Complex Cases Satoru Sumituji,M.D. Rinku General Medical Center IVUS is can provide images of the vessel wall and the tissue around the vessel which cannot be viewed by angiography.
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Annals of Vascular Diseases Advance Published Date: June 2, Horie K, et al.
 2016 Annals of Vascular Diseases doi:10.3400/avd.cr.16-00007 Case Report Recanalization of a Heavily Calcified Chronic Total Occlusion in a Femoropopliteal Artery Using a Wingman Crossing Catheter Kazunori
2016 Annals of Vascular Diseases doi:10.3400/avd.cr.16-00007 Case Report Recanalization of a Heavily Calcified Chronic Total Occlusion in a Femoropopliteal Artery Using a Wingman Crossing Catheter Kazunori
limbsandthings.com Advanced Female Catheterisation Trainer User Guide For more skills training products visit Limbs & Things Ltd.
 Advanced Female Catheterisation Trainer Product No: 60155 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
Advanced Female Catheterisation Trainer Product No: 60155 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System.
 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
Endovascular Approach to CTOs: Crossing methods and Devices
 Endovascular Approach to CTOs: Crossing methods and Devices Anish J. Thomas, MD FACC FSCAI Interventional Cardiology Vascular/Endovascular Medicine SSM Heart Institute St. Louis, MO Disclosure Consultant:
Endovascular Approach to CTOs: Crossing methods and Devices Anish J. Thomas, MD FACC FSCAI Interventional Cardiology Vascular/Endovascular Medicine SSM Heart Institute St. Louis, MO Disclosure Consultant:
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
Technique Guide. Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones.
 Technique Guide Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones. Table of Contents Introduction Midface Distractor System 2 Indications
Technique Guide Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones. Table of Contents Introduction Midface Distractor System 2 Indications
Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons
 SCAI Interventional Cardiology Fall Fellows Course 2014 Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons December 7, 2014 8:30 am John S. Douglas Jr, MD Professor of Medicine
SCAI Interventional Cardiology Fall Fellows Course 2014 Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons December 7, 2014 8:30 am John S. Douglas Jr, MD Professor of Medicine
DePuy International Ltd St Anthony s Road Leeds LS11 8DT England Tel: +44 (0) Fax: +44 (0)
 Reference: 1. Data on file at DePuy Spine TM 2008 DePuy Spine International is a joint venture with Biedermann Motech, GmbH. This publication is not intended for distribution in the USA. X-ray on front
Reference: 1. Data on file at DePuy Spine TM 2008 DePuy Spine International is a joint venture with Biedermann Motech, GmbH. This publication is not intended for distribution in the USA. X-ray on front
Cannulated Angled Blade Plate 3.5 and 4.5, 90.
 Cannulated Angled Blade Plate 3.5 and 4.5, 90. Technique Guide This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation. Table of Contents Introduction
Cannulated Angled Blade Plate 3.5 and 4.5, 90. Technique Guide This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation. Table of Contents Introduction
Calcium Removal and Plaque Modification in the Era of DEB and Contemporary Stenting for Femoro- Popliteal Disease
 Calcium Removal and Plaque Modification in the Era of DEB and Contemporary Stenting for Femoro- Popliteal Disease Thomas M. Shimshak, MD Heart and Vascular Center Florida Hospital Heartland Medical Center
Calcium Removal and Plaque Modification in the Era of DEB and Contemporary Stenting for Femoro- Popliteal Disease Thomas M. Shimshak, MD Heart and Vascular Center Florida Hospital Heartland Medical Center
