Urea Secretion by the Straight Segment
|
|
|
- Arron Bruce
- 5 years ago
- Views:
Transcription
1 Urea Secretion by the Straight Segment of the Proximal Tubule SATOSHI KAWAMURA and JUHA P. KOKKO From the Department of Internal Medicine, the University of Texas Health Science Center at Dallas, Southwestern Medical School, Dallas, Texas A B S T R A C T Studies utilizing in vitro microperfusion were designed to examine whether urea is actively or passively transported across superficial and juxtamedullary straight segments of rabbit proximal tubules. With perfusate and bath solutions containing 1 nrm urea and electrolytes similar to normal plasma, the efflux (lumento-bath) isotopic permeability (X 10-5 cm s-1) of superficial segments was 1.37±0.16 and of juxtamedullary segments was In the same tubules, the influx (bath-to-lumen) isotopic permeability was 3.70±0.35 in superficial segments and in juxtamedullary segments. Despite net water movement in the opposite direction (0.5 nl mm-' min-1), the influx rate was significantly higher than the efflux rate of urea in both groups. With a low perfusion rate (2 nl/min) and equivalent specific activities of ['4C]urea in bath and perfusate, the collected-to-perfused ratio of ['4C]urea, corrected for volume marker change, was in superficial and in juxtamedullary nephrons, thus indicating net secretion in both segments. In separate studies urea influx was inhibited by hypothermia (decrease from 370 to 280C), by phloretin (0.1 mm in bath), by cyanide (1 nmm), but not by probenecid (0.2 mam). In each case the inhibition was highly significant and reversible. These data suggest that urea is actively secreted by the straight segments of both the superficial and juxtamedullary proximal tubules. These segments may, therefore, contribute significantly to the high urea concentration found at the bend of Henle's loop by micropuncture. This work was submitted in abstract form to the 8th Annual Meeting of the American Society of Nephrology, Washington, D. C., November, Dr. Kawamura's present address is: Second Department of Internal Medicine, Nihon University School of Medicine, Oyaguchi Istabashi, Tokyo, Japan. Receized for puiblication 3 Februiary 1976 and in rcvised formii 1 Juln1e INTRODUCTION The importance of urea in the formiiation of concentrated urine has been enmphasized with the recent description of countercurrent multiplication models in which the inner medullarv structures do not possess active transport nmechanisnms for NaCl (1, 2). These models require that for the kidney to forml maximally concentrated urine a high concentration of urea must be present in the papillary interstitium. To maintain these high concentrations of papillary urea, it has been suggested that urea is "recycled" and thus trapped in the countercurrent flow systems known to exist in the renal medulla. In fact, previous micropuncture studies (3-7) have shown that the concentration of urea is higher in the fluid obtained from the bend of the loop of Henle than can reasonably be expected to exist at the end portions of the p)roximal convoluted tubule. These findings have been generally interpreted to indicate that urea recn cles into the descending limb of Henle. A priori, these findings seem in variance with our demonstration that the descending limb of Henle is relatively impernmeable to influx of urea (8). However, it must be remembered that a relativelx long nephron segment, namely the straight portion of the proximal tubule (pars recta),, is interposed between the proximal convoluted tubule and the descending limb of Henle. No previous studies have examined the mechanism of urea transport across tlis segment. Thus, the purposes of the present studies were to determine the passive permeability characteristics of the pars recta to urea and to examine whether urea is actively secreted or reabsorbed by the pars recta. METHODS All results were obtained by the previously reported technique, in which isolated segments of rabbit nephrons were perfused in vitro (9). In each case tubular segments were obtained from kg female rabbits, fed a standard laboratory diet before guillotine decapitation. Straight seg- The Journal of Clinical Investigation Volume 58 September
2 ments of both superficial (SF) 1 and juxtamedullary (JM) proximal tubules were dissected and perfused by the techniques previously described (10). Similarly, the positive identification of the origin of the straight segment was accomplished by the methods utilized in our previous study (10). Straight segments of SF proximal tubules were obtained from the mid-cortex and juxtamedullary regions while straight segments of JM proximal tubules were obtained exclusively from the outer medulla. Three different types of perfusates were used in these experiments: (a) isosmolal ultrafiltrate of rabbit serum; (b) solution A (Table I), designed to simulate normal ultrafiltrate of rabbit serum; and (c) equilibrium solution (Table I), designed to minimize transtubular fluid movement across SF proximal tubules. The choice of perfusate will be described in the Results section for each experimental protocol. Two different bathing media were used: commercially available rabbit serum or solution A (Table I) to which 5% vol/vol fetal calf serum was added. In addition, varying amounts of urea were added to these solutions as dictated by the specific protocol. Net reabsorption of fluid (C), in nanoliters per millimeter-minute, was calculated by the same equations previously published (11 ): C Vi T0 - (1) L where V. is calculated by dividing the [1"I]iothalamate (Glofil-125, Abbott Laboratories, North Chicago, Ill.) counts per minute of the collected fluid by 'I counts per minute per nanoliter of perfusion fluid and by the time of the collection period; V. is obtained directly by a previously calibrated constant-volume pipette as originally described by Burg (12); L is the length of tubule in millimeters. Pernieability coefficient for urea (Pure,). Permeability coefficients for urea for lumen-to-bath (Plbure.) and for bath-to-lumen (Pblurea) were calculated from the rate of disappearance from or appearance into the luminal fluid of the [14C]urea (10,uCi/ml, ICN Pharmaceuticals, Inc., Irvine, Calif.), added either to the perfusate or to the bath. In this series of experiments the perfusate was solution A while the bath was solution A plus 5% vol/vol fetal calf serum. In one series of studies, bidirectional permeability coefficients were measured on the same tubules by first adding the ["4C]urea to the bath and measuring the influx rate of [14C]urea for three collection periods, followed by three collection periods after change of perfusate (by addition of ["4C]urea), and bath (by deletion of ["4C]urea), and then measuring the efflux rate of ["C] urea. 30 min re-equilibration time was allowed between the changes of the solutions. The order of these studies was randomized to minimize systematic sources of error. Unidirectional plburea was calculated by (13) V1 - V0 lnci*/co* 1 plburea = A L V/V0* + ] (2) where A is the area of the tubule calculated from its length and an assumed inside diameter of 20,um; V, is perfusion rate; 'Abbreziations used in this paper: bl, bath-to-lumen; C, net reabsorption of fluid; Jr, net fluid reabsorption; JM, juxtamedullary; lb, lumen-to-bath; Pure., permeability coefficient for urea; PD, transepithelial potential difference; Qio, temperature coefficient of diffusion; Ourea, reflection coefficient; SF, superficial; V,, perfusion rate. TABLE I Composition of Artificial Solutions Solution A Used* Equilibrium solution m.mi NaCl NaHCO KCI 5 5 Na2HPO4 4 4 Na acetate CaCI MgSO4 1 1 Glucose Alanine 5 5 Urea 1 1 Raffinose 65.1 Osmolality* * Gravimetrically measured. Osmolality measured by standard freezing point depression techniques. V. is collection rate; C+*, counts per minute per nanoliter of isotope in the perfusate; and C.*, counts per minute per nanoliter of isotope in the collected fluid. In those experiments in which V, = V. exactly, the permeability coefficient was calculated according to (13): Plburea = i ln i urea A co* In tubules in which Pblurea was determined together with Plb urea, Pbl urea was calculated by (14), pbl co urea Plb urea 1 Cb* L exp(-aplbure./v)j where C.* is counts per minute per nanoliter of isotope in collected fluid; Cb* is counts per minute per nanoliter of isotope in the bath; A is area of tubule; and V is the mean flow rate, (V, + V.) /2. In experiments in which the permeability coefficient was measured from bath to lumen without the simultaneous determination of lumen-to-bath flux, the following equation was used (14): C pbla Pburea = bn C* (3) (4) A Cb*-ln (5) The radioactivity of '5I was measured by a Packard model 3365 three-channel gamma spectrometer while [I4C] - urea activity was measured with a Packard model 2420 liquid scintillation counter (Packard Instrument Co., Inc., Downers Grove, Ill.). Reflectiont coefficient for lirea. Reflection coefficient for urea, aurea, was measured in these experiments by the "equal concentration" method (15), where urea is equal to the ratio of the increment of net fluid movement induced by an osmotic gradient of urea, A Jzvurea, as compared to net fluid movement induced by an equivalent concentration gradient of nonpermeant compound, raffinose: rjvurea AJvraffinose (6) Proximal Tubule Secretion of Urea 605
3 TABLE II Unidirectional Permeability Coefficients for [14C] Urea, Pur,,, of Proximal Straight Tubules across SF and JM Segments Origin of segment N Length Vi J, Pure& mm nl min-' ml min-' mm-, 10-' cm s- Superficial bl * ±0.07 :1= :10.33 lb : Juxtamedullary bl * 4t lb ±0.08 ± ±0.20 Perfusate was solution A (Table I); bath was solution A plus 5% vol/vol calf serum; n = number of tubules. *P <0.001 (blvs.lb). 606 S. Kawamura and J. P. Kokko In each experiment a total of nine collections were made, three each of: (a) control perfusate; (b) when 50 mosmol/ liter of urea was added to the bath; and (c) when 50 mosmol/liter of raffinose was added to the bath. The order of addition to the bath of urea or raffinose was randomized. Perfusate in each experiment was solution A (Table I). Transtubular potential difference (PD). Transtubular PD was measured by techniques previously reported (16). In summary, Beckman (Beckman Instruments, Inc., Fullerton, Calif.) calomel half-cells were connected to the bath and the perfusion pipette via juxtaposed bridges of 300 mosmol/kg Ringer's solution in 4% agarose. The PD was measured by Keithley model 602 electrometer (Keithley Instruments, Inc., Cleveland, Ohio) and recorded on a Rikadenki model B-261 multipen recorder (Rikadenki Kogyo Co. Ltd., Tokyo, Japan). Transport inhibitors. Various concentrations of three different transport inhibitors were used: probenecid (Merck Sharp & Dohme, Div. of Merck Co., Inc., West Point, Pa.), sodium cyanide (J. T. Baker Chemical Co., Phillipsburg, N. J.) and phloretin (ICN K&K Laboratories Inc., Plainview, N. Y.). The data of each tubule were obtained as the mean of three or four collection periods. The results are expressed as mean+se of number of tubules studied. RESULTS The results of the initial series of studies in which solution A was used as perfusate and in which the unidirectional urea permeabilities were measured are summarized in Table II. The bath-to-lumen urea permeability, 3.70±0.33 x 10- cm/s (n = 18) across the SF segment of the proximal tubules is significantly higher (P <0.001) than the lumen-to-bath permeability, 1.37 ±0.16 X 10' cm/s (n = 13). Similarly, the bath-tolumen permeability of X 10V cm/s (n = 20) (of the JM segments) is higher than the lumen-to-bath permeability, 2.14±0.20 X 10' cm/s (n = 15). The above data, though performed on a large number of tubules, might be criticized for two reasons: the permeability coefficients were measured under conditions in which net fluid movement was not zero; and significant differences exist between permeability coefficients obtained from separate tubules. In view of this, the experiments depicted in Table III were performed. In the top panel both the bath-to-lumen and lumen-tobath permeability coefficients were determined in the same segment sequentially with solution A as the perfusate. In agreement with the previous studies, described in Table II, the results of Table III indicate that influx is greater than efflux of urea. To examine the bidirectional permeabilities in the absence of net water flow, equilibrium solution (Table I) was developed for the SF tubules. Net fluid reabsorption was not statistically different from zero with equilibrium solution as the perfusate. Again, the influx of urea was approximately twice as great as the efflux of urea (Table III). Similarly, the influx of urea in JM tubules was approximately twice as great as the efflux of urea when these two parameters were measured sequentially in the same tubules. The results of these experiments are summarized in Table III when solution A was used as perfusate. For reasons that are not clear, we have been unable to develop an "equilibrium" solution for JM tubules where the net reabsorption of fluid is zero. Consequently, we have not measured the bidirectional urea fluxes in JM tubules when net reabsorption was zero. Net secretion. The possibility of net secretion of urea into the lumen of SF and JM straight segments of proximal tubules was evaluated with solution A as the perfusate and similar solutions with added 5% vol/vol calf serum as the bath. Perfusion rate was slow, varying between 1 and 5 nl/min. Equivalent counts of ["C]urea were added to the perfusion fluid and bath. The concentration of "C was measured in the collected fluid and compared to the counts in the perfusate and the
4 TABLE III Bidirectional Permeability Coefficient for ["4C] Urea in the Two Types of Proximal Straight Tubules Superficial segments; perfusate = solution A Superficial segments; perfusate = equilibrium solution Juxtamedullary segments; perfusate = solution A Exp. Exp. Exp. no. Length Vi J. P,,, no. Length Vi Jr Pure& no. Length Vi J. Pu,e. mm nl min-' nl min-' 10- cm mm ni min-' xi min-' 10- cm mm ni min-' nl min-' 10-5 cm mm-, 5-1 mm-, s-i mm-, 5-I bl lb bl lb bl lb lb bl lb bl lb bl lb lb bl bl bl lb lb bl lb bl lb bl bl lb bl lb bl lb bl bl lb lb lb bl lb bl Mean lb lb lb SE 4±1.65 ± ±0.02 ±0.25 4±1.51 ±0.06 ±0.44 Mean bl bl bl SE ±0.09 ±0.55 ±0.74 ±0.06 ±0.21 ±2.42 ± Bath in each case was solution A plus 5% vol/vol calf serum; bl = bath-to-lumen; lb = lumen-to-bath. Both lb and bl permeabilities were measured in the same tubule. The order of measurement was randomized. bath. At the beginning, bath "C counts were 5% lower than those of the perfusate but on average the two solutions were the same at the time of the collection. A rise in the ratio of ["C]urea counts in collected fluid to perfusate was then compared to the rise in 'I volume marker ratio. The results of these experiments are shown in Fig. 1 and indicate a highly significant (P < 0.001) rise in the "C ratio as compared to the [si]- iothalamate ratio. The mean CF/PF urea "C over CF/ PF iothalamate 'I ratio was 1.07±0.01 in SF-PSF (n = 15, mean length 1.7 mm) and in JM- PSF (n = 12; mean length 1.3 mm). These results indicate net urea secretion in the direction opposite to that of net water movement. Effect of hypothermia. If net urea secretion occurs by energy dependent processes, then it might be predicted that the unidirectional influx would decrease with a decrease in the ambient temperature. To examine this issue, control periods were first obtained at 37 C when the bath-to-lumen ["C]urea flux was measured together with the transtubular potential difference. This was followed by similar measurements at 280C. Bath temperature was then returned to 37 C for a recovery period. The results of these experiments are shown in Fig. 2 and indicate a reversible decrease in both the urea influx and the transtubular PD. The efflux (lumento-bath) urea permeability also decreased significantly (Table IV); however, in absolute terms, the decrease in efflux of urea was not as great as the decrease in influx of urea. Effect of sodium cyanide. The effect of 1 mm NaCN (added to the bath) on the bidirectional urea fluxes and transtubular PD was examined in a series of studies in o o 0 qt * 0% 0 0.X /; 0"0 / *~~~ SF- PST 1.9 /o JM- PST O 0.9 l.o l.l CF/PFV125I] iothalomate FIGURE 1 The correlation between collected fluid (CF) to perfusion fluid (PF) ratios for ['C]urea and ['II]iothalamate in the SF and JM straight segments of proximal tubules (PST). The bath counts of ["C]urea per nanoliter were identical on the average to the counts in the perfusion fluid. Thus, points above the 450 line of identity indicate net secretion of urea. Proximal Tubule Secretion of Urea 607
5 0 5 Q._* 4 z w1 w E 2 3 -ii J 0 F- 2 m -3 >-2 0> ClE --1 I P<O.02 * P< 0.05 SF- PST P<0.05 o P< 0.02 JM-PST (6) (8) T4 T I Pe0O00l Pe0O00l 37 C 280C 37 C BATH TEMPERATURE FIGuRE 2 The effect of hypothermia on the bath-to-lumen and lumen-to-bath permeability for [14C]urea. Closed and open circles refer to SF and JM segments, respectively. P values were calculated by paired t test. which solution A was used as the perfusion fluid while a similar solution plus 5% vol/vol calf serum was used as bathing fluid. Three control periods were obtained first, followed by three experimental periods, started 10 min after addition of NaCN, and finally three recovery periods, obtained after the NaCN had been exchanged out of the bath. The results of these studies indicate a highly significant reversible decrease (P < 0.001, paired t test) in the bath-to-lumen flux in SF proximal straight tubule ( to , and a return to 2.0±0.3 X 10' cm/s, n = 6, (Fig. 3). There was no effect of NaCN on the lumen-to-bath urea fluxes in either of the segments (Table IV). NaCN reversibly decreased the transtubular PD (Fig. 3). The effect of phloretin. The effect of 0.1 mm phloretin was examined by a similar protocol, as was the effect of NaCN. As shown in Fig. 4, phloretin significantly (P < 0.001, paired t) decreased the bath-tolumen influx of urea (from 2.98±0.14 to 2.09±0.14, TABLE IV The Effect of Hypothermia, Sodium Cyanide, and Phloretin on the Lumen-to-Bath Permeation of Urea Control Experimental n Pure. Pure P Hypothermia SF (28 C) JM NaCN, 1 mm SF NS (bath) JM NS Phloretin, 0.1 mm SF NS (bath) JM NS 608 S. Kawamura and J. P. Kokko a zz icn E J u 2 I - 1 m > a--e_1 -T NaCN (Bath) 'O T T T~~~) 0T - I P<0.005 o P<0.001 JM-PST P<0.001 * P<0.05 SF-PST I -A. T I~~~~~ 1 1~~~~ CONTROL EXP RECOVERY FIGuRE 3 The effect of the addition of 10 mm NaCN to the bath on ["C]urea bath-to-lumen influx (top panel) and PD (bottom panel) across the SF and JM segments of proximal straight tubules. and back to x 105), without significant effect on lumen-to-bath fluxes (Table IV). In these studies three SF and three JM straight segments of proximal tubules were used. Simultaneously measured transtubular PD fell minimally but significantly (P < 0.02, paired t), and the net water flow fell from 0.71±0.15 to nl mmn' min-' (P < 0.02, paired t-test). The effect of probenecid. The effect of 0.2 mm probenecid was examined only in the bath-to-lumen direction in four SF proximal straight tubules. The probenecid was added to the bath. In these experiments equilibrium solution was used as the perfusate. There was no effect of probenecid either on the influx of urea 0 a, Q.z 3 Z e W E 2 2 J I O 1 I- m OL I'l 0>aL E 4r r -2f Phloretin (Both) (6) P<0.001 P< 0.02 P<0.02 P< T 1 T CONTROL EXP. RECOVERY FIGURE 4 The effect of the addition of 1 mm phloretin on [14G]urea bath-to-lumen influx (top panel) and PD (bottom panel) across the two types of proximal straight tubules. Results are grouped and similar.
6 TABLE V Reflection Coefficient for Urea across the Two Types of Proximal Straight Tubules Length Bathing medium Vi J.,sr, mm mosmol/liter nl/min ml min-' mm-' SF (n = 5) 0.76 RS ± RS + raffinose ±0.51 ±0.03 RS + urea ± ±0.51 JM (n = 6) 0.60 RS ± ± ±0.02 RS + raffinose ±t ±0.82 ±i0.02 RS + urea ± ±-0.86 ( to 4.95±0.23 X 10' cm/s, P > 0.05, paired t) or on the net absorption of water ( , control to 0.24±0.11 nl/mm min, experimental, P > 0.05, paired t). Reflection coefficient of urea. The reflection coefficient of urea was measured by the equal concentration technique when the net induced water flow was measured with 50 mosmol/liter raffinose added to the bath. Short segments of tubules and rapid perfusion rates were used in these experiments. The calculated reflection coefficient for urea in SF proximal straight tubules was 1.0±0.03 and in JM proximal straight tubules was 0.97±0.02 (Table V). Neither of these numbers is statistically (P > 0.05) different from unity. DISCUSSION It is now generally accepted that transport of urea occurs by passive transport processes both in the proximal convoluted tubule and the descending limb of Henle. However, micropuncture studies have shown that more urea exists in fluid obtained from the bend of the loop of Henle than can be accounted for by filtration (3-7). This suggests that urea enters the nephron somewhere between the end of the proximal convoluted tubule and the end of the descending limb of Henle. Only two general types of epithelia exist between these two points: the pars recta (straight segment of the proximal tubule) and the descending limb of Henle. Direct in vitro studies have suggested that the descending limb of Henle is relatively impermeable to urea (8) ; therefore, it is likely that significant recirculation or re-entry of urea occurs across the pars recta. No previous studies have examined the role of the pars recta with respect to the transport of urea. In view of these evaluations, the present studies were designed to examine the mechanism of urea transport by the pars recta. One way to determine whether active urea secretion exists in the pars recta is to measure simultaneous bidirectional urea fluxes. This approach is not feasible since only one isotope of urea is practical to count by currently available technology, ["C]urea. Another approach is to measure the unidirectional fluxes sequentially in the same tubule; i.e., first calculate the lumento-bath urea flux from the disappearance rate of ["C]- urea added to the perfusate, then change the perfusate, then calculate the bath-to-lumen urea flux from the appearance rate of ["C]urea added to the bath. Initially, a large series of such studies were conducted in which the unidirectional fluxes were measured sequentially, with the same tubule as its own control. The results of these measurements indicate that in both the SF and JM straight segments, the influx of urea is approximately twice as great as the efflux of urea. The inequality of the bath-to-lumen as compared to lumen-tobath flux suggests net secretion of urea. If active secretion of urea exists, then urea concentration of the luminal fluid should rise above the concentration in the bathing and perfusion fluid as the fluid courses through the pars recta. Ideally, these experiments should be designed so that the tubules are perfused slowly, with perfusion fluids and bathing fluids with the same concentration of urea, and then the concentrations of urea in the perfusate, bath, and collected fluid are measured by the same ultra-micro chemical methods. We have been unable to develop a reliable and a reproducible method for the measurement of urea in nanoliter-size samples. Therefore, we used a technique in which equivalent counts of ["C]urea were added to the perfusion fluid and the bath. Since the concentration of urea (1 mm) was the same in the perfusate and the bath, a rise in the counts of "C per nanoliter of collected fluid would represent secretion or concentration of counts due to net fluid reabsorption. Therefore, a disproportionate rise in ["C]urea counts with respect to 'I volume marker counts would represent urea secretion. In fact, this was found, as demonstrated by the results in Fig. 1. The rise in counts is small but statistically significant (P < 0.001). Thus, active secretion of urea is strongly suggested by the inequality of unidirectional urea fluxes and by the net secretion of urea against a concentration gradient. If active secretion of urea exists, then its influx Proximal Tubule Secretion of Urea 609
7 component should be decreased by hypothermia and by some transport inhibitors. The interpretation of temperature effects noted in the present studies is not straightforward. It is known that temperature affects both passive and active transport processes. Temperature can affect transport as a consequence of at least two factors: an effect expressed through free diffusion and an effect expressed through activation energy of membrane permeation. The information concerning temperature dependence of urea permeation across biological membranes is limited. Galey et al. (17) have noted that the temperature coefficient of diffusion, Qio, of urea across the red blood cell membrane is Since urea is passively transported across the red blood cell membrane, a QLo of approximately 1.67 (or a rise in permeability coefficient by a factor of 1.67 when the temperature is elevated by 10 C) establishes precedence for a Qio associated with nonactive transport processes for urea. Wright (personal communication) has determined a Qio of passive urea permeation across the toad bladder of 1.3. Again, the interpretation of these findings is complicated since urea is, in part, transported across the toad bladder by a carrier-mediated process. In our experiment, the temperature was lowered from 37 C to 28 C, close to 10 C. The lumen-to-bath flux decreased by 1.6. A value of 1.6 is consistent with a nonactive process. On the other hand, the active secretory component of urea transport (bath-to-lumen flux minus the lumen-to-bath flux) has a Qio of about 2.0. Thus it is interesting that the effects of temperature on the unidirectional fluxes are asymmetrical. The greater inhibitory effect of temperature lowering on the influx component of urea transport supports the concept that some type of carrier-mediated mechanism is involved in the influx of urea from the bath to the lumen of the pars recta. A purely passive unidirectional flux (a flux describable by Fick equations) should not be inhibited by transport inhibitors, whereas the active component of unidirectional flux may be influenced by an appropriate inhibitor. The effects of three different transport inhibitors were tested. These included cyanide, phloretin, and probenecid. Bidirectional effects on urea transport of cyanide and phloretin were tested, while the effect of probenecid was tested only on the bath-to-lumen flux. Neither cyanide nor phloretin had any effect on the lumen-to-bath flux of urea, whereas both significantly decreased the bath-to-lumen flux of urea. It is of interest that the bath-to-lumen flux was decreased to approximately the same level as the lumen-to-bath flux, suggesting that only the active component was influenced by these two inhibitors. Probenecid had no -effect on the bath-to-lumen flux, suggesting that urea transport does not occur by the same pump responsible for transport of organic acids across the proximal straight segments (18). Though no previous suggestions of urea secretion by the mammalian pars recta have been advanced, the suggestions of active urea transport by the kidney is not without precedent. Clapp (19) and Lassiter et al. (20) have noted that urea reabsorption occurs from the collecting duct against a concentrating gradient in a protein-depleted animal. These findings would suggest that under certain circumstances the collecting duct is capable of active urea reabsorption. No mammalian renal studies have suggested active urea secretion; however, active urea secretion has been demonstrated by frog kidney (21), and carrier-mediated urea transport inhibited by phloretin has been shown in the toad bladder (22, 23). However, phloretin has no effect on transpithelial water permeation across the toad bladder (22). In addition, urea permeability is inhibited by phloretin across the gall bladder (23). The finding of urea secretion by the pars recta does seem to answer an apparent controversy between published in vitro microperfusion and in vivo micropuncture data. We have previously demonstrated by in vitro microperfusion that the descending limb of Henle is relatively impermeable to urea (8). We reasoned that if urea recirculation takes place, the descending limb of Henle does not participate in this process to any major extent. On the other hand, micropuncture studies of antidiuretic rats have revealed urea concentration at the bend of the loop of Henle in the mm range (3-7). We initially felt that these high values of urea are not consistent with a rise in urea concentration purely by water abstraction, but that influx of greater amounts of urea had to be postulated than would be predicted by our previously published urea permeability coefficient of 1.5 X 10' cm/s (8). However, Pennell, et al. (7) used a differential mass balance equation and have shown that it is possible by inward diffusion to raise the urea concentration to higher values than we predicted by our measured permeability coefficients, though the levels that they predicted, 106 mm urea in a 5.0-mm descending limb, fall short of their own experimentally determined concentration of urea. In their calculations, it was assumed that urea entering the descending limb of Henle is 0 mosmol/liter. Our present studies suggest that urea concentration is much higher than zero by two different mechani-sms: (a) passive inward diffusion, and (b) active inward secretion. Since the pars recta courses through the outer strip of the outer medulla, and since the urea concentration in the medullary interstitium is higher than in the cortical interstitium, it then follows tha-t there is a favorable inward concentration gradient for urea diffusion. The second mechanism of urea influx would be by active 610 S. Kawamura and J. P. Kokko
8 urea secretion. It is impossible to make firm quantitative estimations as to how much either of these mechanisms contribute. The reason is that the urea concentration of the outer medullary interstitium is variable and highly dependent on the state of hydration of the animal. Also, since the kidney possesses loops of differing lengths, it is not possible to estimate which fraction of the urea concentration at the bend of the loop is the consequence of urea influx across the pars recta. Nevertheless, these studies do show that one source of the high urea concentration is across the pars recta, and together with the recent mathematical approach of Pennell et al. (7) the present findings provide an explanation for the relatively high concentrations of urea at the bend of the loop. We have previously proposed a model of countercurrent multiplication system, in which high concentrations of papillary urea abstract water out of the thin descending limb of Henle (1). It was postulated that the water abstraction resulted in osmotic equilibration of the thin descending limb fluid and, therefore, generated a NaCl concentration gradient for passive diffusion of NaCl down its concentration gradient. To the extent that urea exists in the descending limb, the magnitude of the NaCl gradient between the loop fluid and adjacent interstitium would be reduced (1). Thus, the maximal amount of NaCl available for passive diffusion out of the thin ascending limb would be reduced (1). However, as discussed in our previous paper (1), the postulated amount of urea entry, consistent with the previous in vivo measurements (3-7), does not negate the operation of the passive equilibration model. Admittedly, the entry of urea into the pars recta would decrease the ability to generate as concentrated a urine as could be generated without the entry of any urea into the descending loop structures. However, the passive model does require that high papillary urea concentrations are maintained to form maximally concentrated urea. Thus, urea must be trapped or recirculated within the medulla. Our previous studies on the thin ascending limb of Henle (14) show that this segment is moderately permeable to urea and therefore would provide a significant pathway for urea recirculation. The present studies would suggest that the pars recta provides another possible mechanism by which urea within the medulla can be conserved without allowing it to "wash out" into the systemic circulation. ACKNOWLEDGMENTS This work was supported in part by U. S. Public Health Service Program Grant P01 HL 11662; National Institute of Arthritis and Metabolic Diseases Research Grant 1 ROI AM 14677; and National Institute of General Medical Sciences Research Fellowship Grant 1 T22 GM REFERENCES 1. Kokko, J. P., and F. C. Rector, Jr Countercurrent multiplication system without active transport in inner medulla. Kidney Int. 2: Stephenson, J. L Concentration of urine in central core model of the renal counterflow system. Kidney Imtt. 2: Lassiter, W. E., C. W. Gottschalk, and M. Mylle Micropuncture study of net transtubular movement of water and urea in nondiuretic mammalian kidney. Am. J. Physiol. 200: Rouffignac, C. de, and F. Morel Micropuncture study of water, electrolytes, and urea movement along the loops of Henle in Psammomys. J. Clin. Invest. 48: Marsh, D. J Solute and water flows in thin limbs of Henle's loop in the hamster kidney. Am. J. Physiol. 218: Jamison, R. L., J. Buerkert, and F. Lacy A micropuncture study of Henle's thin loop in Brattleboro rats. Am. J. Physiol. 224: Pennell, J. P., V. Sanjana, N. R. Frey, and R. L. Jamison The effect of urea infusion on the urinary concentrating mechanism in protein-depleted rats. J. Clin. Invest. 55: Kokko, J. P Urea transport in the proximal tubule and the descending limb of Henle. J. Clin. Invest. 51: Burg, M., J. Grantham, M. Abramow, and J. Orloff Preparation and study of fragments of single rabbit nephrons. Am. J. Physiol. 210: Kawamura, S., M. Imai, D. W. Seldin, and J. P. Kokko Characteristics of salt and water transport in superficial and juxtamedullary straight segments of proximal tubules. J. Clin. Invest. 55: Burg, M. B., and J. Orloff Control of fluid absorption in the renal proximal tubule. J. Clin. Invest. 47: Burg, M. B Perfusion of isolated renal tubules. Yale J. Biol. Med. 45: Grantham, J. J., and M. B. Burg Effect of vasopressin and cyclic AMP on permeability of isolated collected tubules. Am. J. Physio. 211: Imai, M., and J. P. Kokko Sodium chloride, urea and water transport in the thin ascending limb of Henle. Generation of osmotic gradients by passive diffusion of solutes. J. Clin. Invest. 53: Kokko, J. P., M. B. Burg, and J. Orloff Characteristics of NaCl and water transport in the renal proximal tubule. J. Clin. Invest. 50: Kokko, J. P Proximal tubule potential difference. Dependence on glucose, HCO3, and amino acids. J. Clin. Invest. 52: Galey, W. R., J. D. Owen, and A. K. Solomon Temperature dependence of nonelectrolyte permeation across red cell membranes. J. Gen. Physiol. 61: Grantham, J. J., R. L. Irwin, P. B. Qualizza, D. R. Tucker, and F. C. Whittier Fluid secretion in isolated proximal straight renal tubules. Effect of human uremic serum. J. Clin. Invest. 52: Proximal Tubule Secretion of Urea 611
9 19. Clapp, J. R Renal tubular reabsorption of urea in normal and protein-depleted rats. Am. J. Physiol. 210: Lassiter, W. E., M. Mylle, and C. W. Gottschalk Micropuncture study of urea transport in rat renal medulla. Am. J. Physiol. 210: !. Schmidt-Nielsen, B., and Schrauger, C. R Handling of urea and related compounds by the renal tubules of the frog. Am. J. Physiol. 205: Levine, S., N. Franki, and R. M. Hays Effect of phloretin on water and solute movement in the toad bladder. J. Clin. Invest. 52: Wright, E. M., and R. J. Pietras Routes of nonelectrolyte permeation across epithelial membranes. J. Membrane Biol. 17: S. Kawamura and J. P. Kokko
Intrinsic Differences in Various Segments
 Intrinsic Differences in Various Segments of the Proximal Convoluted Tubule HARRY R. JACOBSON and JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas Southwestern Medical School,
Intrinsic Differences in Various Segments of the Proximal Convoluted Tubule HARRY R. JACOBSON and JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas Southwestern Medical School,
Sodium Chloride, Urea, and Water Transport
 Sodium Chloride, Urea, and Water Transport in the Thin Ascending Limb of Henle GENERATION OF OSMOTIC GRADIENTS BY PASSIVE DIFFUSION OF SOLUTES MASASHI IMAI and JUHA P. KOKKO From the Department of Internal
Sodium Chloride, Urea, and Water Transport in the Thin Ascending Limb of Henle GENERATION OF OSMOTIC GRADIENTS BY PASSIVE DIFFUSION OF SOLUTES MASASHI IMAI and JUHA P. KOKKO From the Department of Internal
Calcium and Phosphate Transport in Isolated
 Calcium and Phosphate Transport in Isolated Segments of Rabbit Henle's Loop ANTONINO S. RocHA, JOSE B. MAGALDI, and JUHA P. KOKKO From Unidade de Doencas Renais, Department de Clinica Medica, Hospital
Calcium and Phosphate Transport in Isolated Segments of Rabbit Henle's Loop ANTONINO S. RocHA, JOSE B. MAGALDI, and JUHA P. KOKKO From Unidade de Doencas Renais, Department de Clinica Medica, Hospital
Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion
 Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion OLGA H. BROKL AND WILLIAM H. DANTZLER Department of Physiology, College of Medicine, University of Arizona, Tucson,
Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion OLGA H. BROKL AND WILLIAM H. DANTZLER Department of Physiology, College of Medicine, University of Arizona, Tucson,
Transport characteristics of the thin limbs of Henle
 Kidney International, Vol. 22 (1982), pp. 449 453 Transport characteristics of the thin limbs of Henle JUHA P. KOKKO Department of Internal Medicine, University of Texas Health Science Center, Southwestern
Kidney International, Vol. 22 (1982), pp. 449 453 Transport characteristics of the thin limbs of Henle JUHA P. KOKKO Department of Internal Medicine, University of Texas Health Science Center, Southwestern
Countercurrent system
 Kidney International, Vol. 38 (1990), pp. 695 699 Countercurrent system JEFF M. SANDS and JUHA P. KOKKO Renal Division, Department of Medicine, Emory University School of Medicine, Atlanta, Georgia, USA
Kidney International, Vol. 38 (1990), pp. 695 699 Countercurrent system JEFF M. SANDS and JUHA P. KOKKO Renal Division, Department of Medicine, Emory University School of Medicine, Atlanta, Georgia, USA
Evidence for a Concentration Gradient Favoring Outward
 Evidence for a Concentration Gradient Favoring Outward Movement of Sodium from the Thin Loop of Henle PAUL A. JOHNSTON, CARLOS A. BATTILANA, FRANK B. LACY, and REx L. JAMISON From the Division of Nephrology,
Evidence for a Concentration Gradient Favoring Outward Movement of Sodium from the Thin Loop of Henle PAUL A. JOHNSTON, CARLOS A. BATTILANA, FRANK B. LACY, and REx L. JAMISON From the Division of Nephrology,
Functions of Proximal Convoluted Tubules
 1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
BIOLOGY - CLUTCH CH.44 - OSMOREGULATION AND EXCRETION.
 !! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
!! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
Medullary Thick Ascending Limb of Henle
 Sodium Chloride and Water Transport in the Medullary Thick Ascending Limb of Henle EVIDENCE FOR ACTIVE CHLORIDE TRANSPORT ANTONINO S. RoCHA and JUHA P. KoKKo From the Department of Internal Medicine, The
Sodium Chloride and Water Transport in the Medullary Thick Ascending Limb of Henle EVIDENCE FOR ACTIVE CHLORIDE TRANSPORT ANTONINO S. RoCHA and JUHA P. KoKKo From the Department of Internal Medicine, The
Sodium Chloride and Water Transport
 Sodium Chloride and Water Transport in the Descending Limb of Henle JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas (Southwestern) Medical School, Dallas, Texas 75235 A
Sodium Chloride and Water Transport in the Descending Limb of Henle JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas (Southwestern) Medical School, Dallas, Texas 75235 A
Normal Renal Function
 Normal Renal Function Functions of the Kidney: balances solute and water transport excretes metabolic waste products conserves nutrient regulates acid-base balance secretes hormones that help regulate
Normal Renal Function Functions of the Kidney: balances solute and water transport excretes metabolic waste products conserves nutrient regulates acid-base balance secretes hormones that help regulate
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
Excretion Chapter 29. The Mammalian Excretory System consists of. The Kidney. The Nephron: the basic unit of the kidney.
 Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Calcium Transport in the Thick Ascending Limb of Henle
 Calcium Transport in the Thick Ascending Limb of Henle HETEROGENEITY OF FUNCTION IN THE MEDULLARY AND CORTICAL SEGMENTS WADI N. SUKI, DIANE ROUSE, ROLAND C. K. NG, and JUHA P. KOKKO, Renal Section, Department
Calcium Transport in the Thick Ascending Limb of Henle HETEROGENEITY OF FUNCTION IN THE MEDULLARY AND CORTICAL SEGMENTS WADI N. SUKI, DIANE ROUSE, ROLAND C. K. NG, and JUHA P. KOKKO, Renal Section, Department
Potassium secretion. E k = -61 log ([k] inside / [k] outside).
![Potassium secretion. E k = -61 log ([k] inside / [k] outside). Potassium secretion. E k = -61 log ([k] inside / [k] outside).](/thumbs/80/80478709.jpg) 1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY
 URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY Dilute and concentrated urine 1-Dilute urine : Nephron function continuous reabsorption. Solutes while failing to reabsorbe water in distal tubule and
URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY Dilute and concentrated urine 1-Dilute urine : Nephron function continuous reabsorption. Solutes while failing to reabsorbe water in distal tubule and
Diagram of the inner portions of the kidney
 Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Urinary Physiology. Chapter 17 Outline. Kidney Function. Chapter 17
 Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi
 Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Water, urea, sodium, chloride, and potassium transport in the in vitro isolated perfused papillary collecting duct
 Kidney International, Vol. 22 (1982), pp. 485 491 Water, urea, sodium, chloride, and potassium transport in the in vitro isolated perfused papillary collecting duct ANTONINO S. ROCHA and LUCIA H. KUDO
Kidney International, Vol. 22 (1982), pp. 485 491 Water, urea, sodium, chloride, and potassium transport in the in vitro isolated perfused papillary collecting duct ANTONINO S. ROCHA and LUCIA H. KUDO
Kidney and urine formation
 Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX
 One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
Renal Physiology II Tubular functions
 Renal Physiology II Tubular functions LO. 42, 43 Dr. Kékesi Gabriella Basic points of renal physiology 1. Glomerular filtration (GF) a) Ultrafiltration 2. Tubular functions active and passive a) Reabsorption
Renal Physiology II Tubular functions LO. 42, 43 Dr. Kékesi Gabriella Basic points of renal physiology 1. Glomerular filtration (GF) a) Ultrafiltration 2. Tubular functions active and passive a) Reabsorption
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion.
 The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
Na + Transport 1 and 2 Linda Costanzo, Ph.D.
 Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Salt and Water Balance and Nitrogen Excretion
 Announcements Exam is in class on WEDNESDAY. Bring a #2 pencil and your UFID. You must come to your registered class section (except those with DRC accommodations). Office hours Mon 1-3 pm. Teaching evals:
Announcements Exam is in class on WEDNESDAY. Bring a #2 pencil and your UFID. You must come to your registered class section (except those with DRC accommodations). Office hours Mon 1-3 pm. Teaching evals:
Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction Once the filtrate is formed, the early
Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction Once the filtrate is formed, the early
Osmoregulation and Renal Function
 1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
Sodium and chlorine transport
 Kidney physiology 2 Sodium and chlorine transport The kidneys help to maintain the body's extracellular fluid (ECF) volume by regulating the amount of Na+ in the urine. Sodium salts (predominantly NaCl)
Kidney physiology 2 Sodium and chlorine transport The kidneys help to maintain the body's extracellular fluid (ECF) volume by regulating the amount of Na+ in the urine. Sodium salts (predominantly NaCl)
NORMAL POTASSIUM DISTRIBUTION AND BALANCE
 NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
Nephron Function and Urine Formation. Ms. Kula December 1, 2014 Biology 30S
 Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
11/05/1431. Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate
 Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Potassium recycling EDITORIAL REVIEW
 Kidney International, Vol. 31 (/987), pp. 695 703 EDITORIAL REVIEW Potassium recycling The concept of potassium recycling It has been amply demonstrated by micropuncture in nephrons accessible on the surface
Kidney International, Vol. 31 (/987), pp. 695 703 EDITORIAL REVIEW Potassium recycling The concept of potassium recycling It has been amply demonstrated by micropuncture in nephrons accessible on the surface
Concentrating Mechanism in Protein-Depleted Rats
 The Effect of Urea Infusion on the Urinary Concentrating Mechanism in Protein-Depleted Rats J. PHmuIIP PENNELL, VEERAF SANJANA, NORMAN R. FREY, and REX L. JAMISON From the Division of Nephrology, Department
The Effect of Urea Infusion on the Urinary Concentrating Mechanism in Protein-Depleted Rats J. PHmuIIP PENNELL, VEERAF SANJANA, NORMAN R. FREY, and REX L. JAMISON From the Division of Nephrology, Department
Nephron Anatomy Nephron Anatomy
 Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
41B. Metabolism produces wastes that must be eliminated from the body. This. Renal System Physiology: Computer Simulation
 41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
1. Anatomy / Vascularisation. 2. Urine concentration. 3. Axial heterogeneity of some segments
 Lise BANKIR 1. Anatomy / Vascularisation 2. Urine concentration 3. Axial heterogeneity of some segments Rat kidney. Arterial filling with Microfil silicone rubber Alcian Blue staining Filling of arterial
Lise BANKIR 1. Anatomy / Vascularisation 2. Urine concentration 3. Axial heterogeneity of some segments Rat kidney. Arterial filling with Microfil silicone rubber Alcian Blue staining Filling of arterial
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Physiology (6) 2/4/2018. Rahmeh Alsukkar
 Physiology (6) 2/4/2018 Rahmeh Alsukkar **unfortunately the sheet does not involve the slides. ** the doctor repeat a lot of things from the previous lecture so this sheet will begin from slide 139 to
Physiology (6) 2/4/2018 Rahmeh Alsukkar **unfortunately the sheet does not involve the slides. ** the doctor repeat a lot of things from the previous lecture so this sheet will begin from slide 139 to
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Glomerular Capillary Blood Pressure
 Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
Water movement across nephron segments involved with the countercurrent multiplication system
 Kidnel' International, Vol. 10 (/976). p. 64 81 Water movement across nephron segments involved with the countercurrent multiplication system JUHA P. KOKKO and C. CRAIG TISHER Department of Internal Medicine,
Kidnel' International, Vol. 10 (/976). p. 64 81 Water movement across nephron segments involved with the countercurrent multiplication system JUHA P. KOKKO and C. CRAIG TISHER Department of Internal Medicine,
CONTROLLING THE INTERNAL ENVIRONMENT
 AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
in urinary concentration
 Kidney International, Vol. 2 (1972), p. 297 33 EDITORIAL Physiological role of the ioop of Henle in urinary concentration It is now generally accepted that urinary concentration is achieved via the countercurrent
Kidney International, Vol. 2 (1972), p. 297 33 EDITORIAL Physiological role of the ioop of Henle in urinary concentration It is now generally accepted that urinary concentration is achieved via the countercurrent
Chapter 25 The Urinary System
 Chapter 25 The Urinary System 10/30/2013 MDufilho 1 Kidney Functions Removal of toxins, metabolic wastes, and excess ions from the blood Regulation of blood volume, chemical composition, and ph Gluconeogenesis
Chapter 25 The Urinary System 10/30/2013 MDufilho 1 Kidney Functions Removal of toxins, metabolic wastes, and excess ions from the blood Regulation of blood volume, chemical composition, and ph Gluconeogenesis
Renal Physiology - Lectures
 Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
PARTS OF THE URINARY SYSTEM
 EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
Renal Physiology. April, J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine.
 Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Medullary Thick Ascending Limb of the Rat
 Effects of Potassium on Ammonia Transport by Medullary Thick Ascending Limb of the Rat David W. Good Renal-Electrolyte Physiology Laboratory, Departments ofphysiology & Biophysics and Internal Medicine,
Effects of Potassium on Ammonia Transport by Medullary Thick Ascending Limb of the Rat David W. Good Renal-Electrolyte Physiology Laboratory, Departments ofphysiology & Biophysics and Internal Medicine,
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
November 30, 2016 & URINE FORMATION
 & URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
& URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
A. Incorrect! The urinary system is involved in the regulation of blood ph. B. Correct! The urinary system is involved in the synthesis of vitamin D.
 Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
M.Sc., C.Eng, Ph.D. sodium ions by diffusion. This process is thought to
 Postgraduate Medical Journal (May 1976) 52, 279-284. K. E. BRITTON M.Sc., M.D., M.R.C.P. A 'bootstrap' model of the renal medulla E. R. CARSON M.Sc., C.Eng, Ph.D. P. E. CAGE B.Sc. Department of Nuclear
Postgraduate Medical Journal (May 1976) 52, 279-284. K. E. BRITTON M.Sc., M.D., M.R.C.P. A 'bootstrap' model of the renal medulla E. R. CARSON M.Sc., C.Eng, Ph.D. P. E. CAGE B.Sc. Department of Nuclear
Recent formulations of the urinary concentrating mechanism: A status report
 Kidney International, Vol. 16 (1979), pp. 537 545 EDITORIAL REVIEW Recent formulations of the urinary concentrating mechanism: A status report Since the paper by Wirz, Hargitay, and Kuhn in 1951 [1], the
Kidney International, Vol. 16 (1979), pp. 537 545 EDITORIAL REVIEW Recent formulations of the urinary concentrating mechanism: A status report Since the paper by Wirz, Hargitay, and Kuhn in 1951 [1], the
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Urinary bladder provides a temporary storage reservoir for urine
 Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Urinary System. consists of the kidneys, ureters, urinary bladder and urethra
 Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
Duct Acidification Effects of Potassium, HCO3. Harry R. Jacobson. Internal Medicine, 5323 Harry Hines Blvd., Dallas, Texas 75235
 Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Osmoregulation regulates solute concentrations and balances the gain and loss of water
 Ch 44 Osmoregulation & Excretion Osmoregulation regulates solute concentrations and balances the gain and loss of water Freshwater animals show adaptations that reduce water uptake and conserve solutes
Ch 44 Osmoregulation & Excretion Osmoregulation regulates solute concentrations and balances the gain and loss of water Freshwater animals show adaptations that reduce water uptake and conserve solutes
EXCRETORY SYSTEM E. F. G. H.
 XRTORY SYSTM 1. Label the following parts of the nephron in the diagram below:..... F. G. H. I. J. K. L. 2. Identify the following as either True or False: There is a greater osmotic concentration in the
XRTORY SYSTM 1. Label the following parts of the nephron in the diagram below:..... F. G. H. I. J. K. L. 2. Identify the following as either True or False: There is a greater osmotic concentration in the
Faculty version with model answers
 Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Chapter 23. The Nephron. (functional unit of the kidney
 Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
mid ihsan (Physiology ) GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B **
 (Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
(Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
RENAL PHYSIOLOGY. Physiology Unit 4
 RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
Class XI Chapter 19 Excretory Products and their Elimination Biology
 Class XI Chapter 19 Excretory Products and their Elimination Biology Question 1: Define Glomerular Filtration Rate (GFR) Glomerular filtration rate is the amount of glomerular filtrate formed in all the
Class XI Chapter 19 Excretory Products and their Elimination Biology Question 1: Define Glomerular Filtration Rate (GFR) Glomerular filtration rate is the amount of glomerular filtrate formed in all the
Chapter 44. Osmoregulation and Excretion
 Chapter 44 Osmoregulation and Excretion Overview: A Balancing Act Physiological systems of animals operate in a fluid environment Relative concentrations of water and solutes must be maintained within
Chapter 44 Osmoregulation and Excretion Overview: A Balancing Act Physiological systems of animals operate in a fluid environment Relative concentrations of water and solutes must be maintained within
Superficial Proximal Convoluted Tubule
 Calcium Transport in the Rabbit Superficial Proximal Convoluted Tubule Roland C. K. Ng, Diane Rouse, and Wadi N. Suki Renal Section, Department ofmedicine, The Methodist Hospital and Baylor College ofmedicine,
Calcium Transport in the Rabbit Superficial Proximal Convoluted Tubule Roland C. K. Ng, Diane Rouse, and Wadi N. Suki Renal Section, Department ofmedicine, The Methodist Hospital and Baylor College ofmedicine,
Class XI - Biology Excretory Products and their Elimination
 Class XI - Biology Excretory Products and their Elimination MULTIPLE CHOICE QUESTIONS 1. The following substances are the excretory products in animals. Choose the least toxic form among them? a. Urea
Class XI - Biology Excretory Products and their Elimination MULTIPLE CHOICE QUESTIONS 1. The following substances are the excretory products in animals. Choose the least toxic form among them? a. Urea
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
2) This is a Point and Click question. You must click on the required structure.
 Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Chapter 25: Urinary System
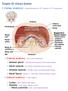 Chapter 25: Urinary System I. Kidney anatomy: retroperitoneal from 12 th thoracic to 3 rd lumbar area A. External anatomy: hilus is the indentation 1. Adrenal gland: in the fat at the superior end of each
Chapter 25: Urinary System I. Kidney anatomy: retroperitoneal from 12 th thoracic to 3 rd lumbar area A. External anatomy: hilus is the indentation 1. Adrenal gland: in the fat at the superior end of each
The kidneys are excretory and regulatory organs. By
 exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
Physiology (5) 2/4/2018. Mohammad jaber
 Physiology (5) 2/4/2018 Mohammad jaber Slides are embedded in the sheet in bold, some figure are not included sorry for any mistake,, lets go.. There are two types of urine : 1-diluted urine ( Excess fluid
Physiology (5) 2/4/2018 Mohammad jaber Slides are embedded in the sheet in bold, some figure are not included sorry for any mistake,, lets go.. There are two types of urine : 1-diluted urine ( Excess fluid
Urinary System. Dr. ZHANG Xiong. Dept. of Physiology. ZJU School of Medicine. QUESTION 6
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
A micropuncture study of Henle s thin loop in Brattleboro
 AMERICAN JOURNAL OF PHYSIOLOGY Vol. 224, No. 1, January 1973. Printed in U.S.A. A micropuncture study of Henle s thin loop in Brattleboro rats REX L. JAMISON, JOHN BUERKERT, AND FRANK LACY Departments
AMERICAN JOURNAL OF PHYSIOLOGY Vol. 224, No. 1, January 1973. Printed in U.S.A. A micropuncture study of Henle s thin loop in Brattleboro rats REX L. JAMISON, JOHN BUERKERT, AND FRANK LACY Departments
After studying this lecture, you should be able to...
 Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
5.Which part of the nephron removes water, ions and nutrients from the blood?
 Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
Uro question 1.While reading a blood test I notice a high level of creatinine, I could assume from this that A) There is a possibility of a UTI B) There is a possibility of diabetes C) There is a possibility
Ch 17 Physiology of the Kidneys
 Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Hill et al. 2004, Fig. 27.6
 Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Long-Term Regulation of Renal Urea Transporters during Antidiuresis
 18 Electrolyte & Blood Pressure 4:18-22, 2006 3) Long-Term Regulation of Renal Transporters during Antidiuresis Dong-Un Kim, M.D. Department of Pediatrics, College of Medicine, The Catholic University
18 Electrolyte & Blood Pressure 4:18-22, 2006 3) Long-Term Regulation of Renal Transporters during Antidiuresis Dong-Un Kim, M.D. Department of Pediatrics, College of Medicine, The Catholic University
Collin College. BIOL Anatomy & Physiology. Urinary System. Summary of Glomerular Filtrate
 Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
Urinary System BIO 250. Waste Products of Metabolism Urea Carbon dioxide Inorganic salts Water Heat. Routes of Waste Elimination
 Urinary System BIO 250 Waste Products of Metabolism Urea Carbon dioxide Inorganic salts Water Heat Routes of Waste Elimination Skin: Variable amounts of heat, salts, and water; small amounts of urea and
Urinary System BIO 250 Waste Products of Metabolism Urea Carbon dioxide Inorganic salts Water Heat Routes of Waste Elimination Skin: Variable amounts of heat, salts, and water; small amounts of urea and
Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron
 Kidney International, Vol. 19 (1981), pp. 797 81 Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron LEON G. FINE and MICHAEL A. KIRSCHENBAUM Division of Nephroiogy,
Kidney International, Vol. 19 (1981), pp. 797 81 Absence of direct effects of prostaglandins on sodium chloride transport in the mammalian nephron LEON G. FINE and MICHAEL A. KIRSCHENBAUM Division of Nephroiogy,
Transport of Ammonia in the Rabbit Cortical Collecting Tubule
 Transport of Ammonia in the Rabbit Cortical Collecting Tubule L. Lee Hamm, D. Trigg, D. Martin, C. Gillespie, and J. Buerkert Renal Division, Department ofmedicine, Washington University School ofmedicine,
Transport of Ammonia in the Rabbit Cortical Collecting Tubule L. Lee Hamm, D. Trigg, D. Martin, C. Gillespie, and J. Buerkert Renal Division, Department ofmedicine, Washington University School ofmedicine,
Diuretics having the quality of exciting excessive excretion of urine. OED. Inhibitors of Sodium Reabsorption Saluretics not Aquaretics
 Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Effect of Prostaglandin E2 on Chloride Transport Across the Rabbit Thick Ascending Limb of Henle
 Effect of Prostaglandin E2 on Chloride Transport Across the Rabbit Thick Ascending Limb of Henle SELECTIVE INHIBITION OF THE MEDULLARY PORTION JOHN B. STOKES, Department ofinternal Medicine, University
Effect of Prostaglandin E2 on Chloride Transport Across the Rabbit Thick Ascending Limb of Henle SELECTIVE INHIBITION OF THE MEDULLARY PORTION JOHN B. STOKES, Department ofinternal Medicine, University
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Homeostatic Regulation
 Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
