Intrinsic Differences in Various Segments
|
|
|
- Basil Dorsey
- 5 years ago
- Views:
Transcription
1 Intrinsic Differences in Various Segments of the Proximal Convoluted Tubule HARRY R. JACOBSON and JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas Southwestern Medical School, Dallas, Texas A B S T R A C T Until recently it has not been possible to compare directly the function of superficial and juxtamedullary nephrons. The present studies, using in vitro microperfusion, were designed to examine whether functional differences exist between proximal convoluted tubule segments of superficial and juxtamedullary nephrons. Electrophysiological studies showed that major differences exist between the relative chloride and sodium permeabilities of these segments. In the 1st mm of the superficial proximal convoluted tubule, the permeability to sodium was greater than that to chloride, whereas in the 2nd mm of the superficial proximal convoluted tubule and all later segments, the permeability to chloride was greater than that to sodium. The juxtamedullary proximal convoluted tubule was found to differ from the superficial proximal convoluted tubule in two respects: first, the relative permeabilities to chloride and sodium did not differ in the various segments of the juxtamedullary proximal convoluted tubule; second, the permeability to sodium was greater than to chloride throughout. When perfused with a solution lacking glucose and amino acids, the superficial and juxtamedullary convolutions exhibited the same transepithelial potential change, a reversible decrease to less than - 1 mv. It thus appears that in both convolutions there exists electrogenic sodium transport coupled to the transport of these organic solutes. This differs from pars recta of both of these nephrons, which have been shown to exhibit electrogenic sodium transport independent of organic solutes. However, when perfused with a solution lacking glucose and amino acids but also containing high chloride and low bicarbonate concentrations, the superficial convolution developed a significantly more positive potential than the juxtamedullary. This difference reflects greater relative chloride permeability in the superficial proximal convolution. Received for publication 10 June 1975 and in revised form 5 December These studies show that intrinsic functional differences exist between proximal convoluted tubules obtained from the superficial and juxtamedullary nephron populations. INTRODUCTION It has been recognized for almost 70 years that the mammalian kidney has two major subpopulations of nephrons, based on their location and depth of penetration into the medulla of their loops of Henle (1). Final urine volume and composition depend on the contributions of both these nephron groups, and thus differences in their function may play an important role in what constitutes the final urine. The two nephron populations have been shown to have different filtration rates and different anatomical and functional circulations and differing responses to various changes in physiological parameters such as blood volume (2-6). However, differences between these nephrons can also exist with respect to peritubular environment, intrinsic membrane characteristics, and/or intraluminal fluid constituents. Until recently, experimental observations of renal function have been concerned with the kidney as a whole or with superficial nephrons alone. Only one previous study has directly examined for intrinsic functional differences between nephron segments from superficial and juxtamedullary nephrons. Kawamura et al. (7), using in vitro microperfusion of rabbit nephrons, have recently shown, significant functional differences between straight segments or pars recta obtained from superficial and juxtamedullary nephrons. These investigators have shown differing relative Na and Cl permeabilities, different reflection coefficients for NaCl, and different osmotic water permeabilities. In addition to intrinsic membrane differences, differences in function may exist on the basis of compositional changes of the intraluminal fluid as it courses through the tubule. Kokko has shown (8) that in the isolated perfused superficial proximal convoluted tubule, alteration of the composition of the perfusate with respect The Journal of Clinical Investigation Volume 57 April 1976@
2 to glucose, amino acids, and sodium bicarbonate significantly affects the transepithelial potential difference (PD). It thus was concluded (8) that the latter segment of the proximal tubule differs from the early segment of the proximal tubule in part by virtue of the intraluminal constituents. The fluid in the early segment is similar to ultrafiltrate of plasma, while the fluid in the latter segment has a high concentration of chloride and a low concentration of glucose, amino acids, and HCO3. The purpose of the present studies was twofold: first, to evaluate relative permeabilities to Cl and Na in the superficial and juxtamedullary proximal convolutions; and second, to examine whether all segments of the proximal convoluted tubule have the capacity of electrogenic sodium transport coupled to organic solute transport. METHODS Segments of proximal convoluted tubules of New Zealand white rabbits were isolated and perfused in vitro by techniques described previously (9). Briefly, female New Zealand white rabbits weighing kg were fed a standard laboratory diet and had free access to water before guillotine decapitation. The left kidney was quickly removed and cut into 1-2-mm-thick slices. Tubules were dissected from one of these slices in a dish of chilled rabbit serum kept at ph 7.4 by continuous bubbling with a 95%o Os and 5% C02 gas mixture. Seven different proximal convoluted segments were dissected: (a) early superficial proximal convoluted tubule (SFPCT) 1 less than 1 mm from its glomerulus; (b) the 2nd mm of the SFPCT; (c) mid SFPCT including segments whose glomerular attachment was not identified and were thus probably more than 1 mm from the glomerulus; (b) late SFPCT more than 1 mm from the anatomical pars recta (these were convoluted segments attached to the pars recta but with over 1 mm of clearly convoluted tubule between the perfusion site and the anatomical or relatively straight pars recta) ; (e) the 1st mm of the juxtamedullary proximal convoluted tubule (JMP- CT); (f) the mid JMPCT; and (g) the late JMPCT. All segments from the SFPCT were dissected from the most superficial cortex, and all segments from the JMPCT were dissected from immediately above the cortico-medullary junction. All tubules were perfused in a small Lucite bath containing commercially available rabbit serum kept at 370C and ph 7.4 by continuous bubbling with a 95% 02 and 5% C02 gas mixture. Initial perfusate was either ultrafiltrate of rabbit serum obtained from the same lot as the bathing serum by pressure dialysis with Aminco PM-30 membranes (American Instrument Co., Inc., Silver Spring, Md.) or an artificial solution made to simulate ultrafiltrate. Transepithelial potential differences were measured with the same technique described previously (8), with equivalent bridges of 300 mosm/liter in 4% agarose (PE tubing size 240), connected from the bath and perfusion pipet to saturated KC1 containing Beckman (Beckman Instruments, Inc., 'Abbreziations used in this paper: ET, transtubular potential; F, Faraday's constant; JMPCT, juxtamedullary proximal convoluted tubule; SFPCT, superficial proximal convoluted tubule. Fullerton, Calif.) calomel half-cells. The circuit was completed by placing a voltage reference source and a batteryoperated Keithly model 602 electrometer (Keithly Instruments, Inc., Cleveland, Ohio) in the circuit. This circuit arrangement is completely symmetrical when perfusion and bath solution have the same electrolyte composition. However, the various solutions used in these experiments introduced small liquid junction potentials, which were both calculated and determined experimentally, and then added to or subtracted from the observed PD. When the bath and perfusion solutions differed only in the concentrations of a single salt, the liquid junction potential, in this case a dilution potential, was calculated from the Nernst-Planck equation as modified by Barry and Diamond (10), with concentrations instead of activities: RT(_- A3) a" EL =- lnā F(A1- U3) a' where ju's are mobilities, subscript 1 refers to the cation, subscript 3 refers to the anion, a' and a" are the concentrations of the salt in the two solutions in question, R is the gas constant, T is absolute temperature, and F is Faraday's constant. When the bath and perfusate had differing concentrations of two salts with a common ion, a bi-ionic junction potential developed and was calculated from the Nernst-Planck equation as modified by Barry and Diamond (10): RT~ui1(a'1-a1) +,U2 (a"2-a'2) -,U3(a//3-al3) EL F= - p(a"i-aa') +,U2 (a"2-a 2) -14 (a"3 - a'3) /.Lla-1-/.+s2a a"3 ln a'l+/2a'2+3a' 3 where, refers to the mobility of each ion, subscripts 1, 2, and 3 refer to the ions themselves (in this case Cl, HCOs, and Na), a is the activity coefficient (in this case the concentration) and " and ' refer to the luminal and bath fluids, respectively. Equivalent conductances at 25 C were used instead of mobilities. The limitations and sources of error in these calculations are minimal and have been previously discussed (8). The liquid junction potentials introduced by the various test solutions were also determined experimentally. Fig. 1 shows a schematic representation of the circuit used in measuring the transtubular potential. Following the circuit diagram below the figure reveals that the liquid junctions from E1 through E5 are canceled by those from E. through E1o. Thus, the observed potential is the transtubular potential or ET. The first experimental method used to measure the liquid junction potentials is shown in Fig. 2. This circuit is identical to that used for measuring transtubular potentials with two exceptions: no tubule is in place, and the agarose bridge between the saturated KC1 and the test tube containing bicarbonate in the right-hand side of the figure has been replaced by a saturated KCl agarose bridge. With perfusate and bath containing bicarbonate, the electrometer was centered on zero. Replacing the test tube in the right-hand side of the figure with a test tube containing a solution of different electrolyte composition introduces two liquid junction potentials. The first junction potential between the test solution and the saturated KCl was assumed to be close to zero. The second junction potential between the test solution and the bicarbonate agarose bridge was measured as Functional Heterogeneity of Proximal Convoluted Tubules 819
3 HgCl2-Hg: Sat KCL: 4%Agarose: : 4%Agarose: Bath: Tubule: Perfusion Fluid: E1 E2 E3 E4 E5 ET E6 4% Agarose: : 4% Agarose: Sat KCL : HgC12- Hg Ringer' s E7 E8 E9 E10 FIGURE 1 Schematic representation of the circuit used to measure transepithelial potentials in isolated tubules. a deflection from zero on the electrometer. It is interesting to note that the value of the junction potential with A-50 solution (see Table I for composition) was quite close to the calculated value with the modified Nernst-Planck equation, 3.2 mv versus 3.56 mv, while that of B solution (see Table I) was not, 0.7 mv versus 2.7 mv. The reason for this discrepancy with B solution is unknown. However, to determine if it was related to an effect of agarose on the mobility of HCOM, a second circuit was utilized as shown below. This circuit was also used to test A-50 TABLE I Composition of Artificial Solutions A A-SO B E mm NaCl KCI NaHCO CaCl MgSO Na2HPO Na acetate Glucose Alanine 5 5 Mannitol Sucrose 92 Osmolality of all solutions, H. R. Jacobson and J. P. Kokko solution. HgCl2-Hg: A solution : perfusion fluid: 4% agarose, E1 E2 E3 300 mmol/liter : 300 mmol/liter E4 : 4% agarose 300 mmol/liter E5 : Sat KC1: HgCl2-Hg E6 If the two solutions developing E2 were identical to each other and had the same electrolyte concentrations as the solution, there was no potential in the circuit, and the electrometer was centered at zero. Exchanging the A solution with experimental solutions caused a liquid junction potential to develop at E. With this technique the experimental liquid junction potential for B solution was 1 mv whereas that of A-50 solution was 3.3 mv. Therefore, it appears that there is a major discrepancy between the calculated and experimentally determined liquid junction potential with B solution: 0.7 mv and 1 mv experimentally, and 2.7 mv calculated. The experimental and calculated liquid junction potentials for A-50 solution agree closely: 32 mv and 3.3 mv experimental and 3.56 mv calculated. The differences in the B solution junction potentials cannot be due to artifact introduced by agarose. Because of the discrepancy between the measured and calculated liquid junction potentials with B solution and also because the measurement assumes that a negligible liquid junction potential exists between the saturated KC1 bridge and the test solutions, a further experiment was per-
4 formed. The circuit used in this measurement is shown below: HgC12 : bicarbonate : 4%0 agarose Ei E2 bicarbonate : bicarbonate : Ag/AgCl E3 E4 Although the calomel electrode on the left side of this circuit contains saturated KCl and thus a liquid junction potential may exist at E1, the electrometer was centered on zero to cancel out this possible liquid junction and also the electrode asymetry between the calomel and Ag/AgCl. Changing the solution between E3 and E4 with B solution creates a liquid junction potential between B solution and the bicarbonate bridge at E8. This junction is identical to that occurring during our experiments. The value of this liquid junction potential was measured several times and was found consistently to be between 0.8 and 1.0 mv. It thus appears that the liquid junction potential introduced when our tubules were perfused with B solution was slightly less than 1 mv. In accord with this, the results obtained with B solution as perfusate will be shown corrected for both the calculated and measured liquid junction potentials. At present, the reason for the discrepancy between the calculated and measured liquid junctions with B solution is unknown. Whether the liquid junction potential introduced with B solution is in the range of 1 or 2.7 mv, the differences between the various tubular segments studied are still the same because all observed potentials were corrected in the same way. However, the previously described attempts at defining the true liquid junction potential are important for a different reason. Earlier studies in membrane physiology have either ignored the presence of liquid junction potentials or used calculated values. Insofar as circumstances may exist where liquid junction potentials differ significantly from theoretical values, an attempt must be made to measure these potentials if interpretation of the data requires accurate knowledge of a true transepithelial potential. After initiation of perfusion the tubule was allowed to develop a stable PD before perfusate or bath fluids were changed. The composition of the various solutions used is shown in Table I. Solution A was made to stimulate ultrafiltrate of normal serum with respect to electrolyte and organic solute composition. A-50 solution was identical to A solution except for the removal of 50 mmol/liter NaCl, which was isosmotically replaced with sucrose. Solution E was also identical to A solution except for the removal of glucose and alanine, replaced isosmotically with mannitol. Solution B was identical to solution E except for the removal of about 20 mmol of NaHCO3, replaced with NaCl. B solution was made to simulate in vivo intraluminal fluid in the latter portion of the proximal convolution after the early segment has removed glucose, amino acids, and the majority of the bicarbonate. An isosmotic 50 mmol/liter NaCl gradient from lumen to bath was achieved by exchanging the rabbit serum in the bath with A-50 solution. The PD response in the JMPCT, early SFPCT, and mid SFPCT was also observed when A-50 solution was used as perfusate to examine whether the diffusion potential was symmetrical when equivalent salt gradients were imposed from lumen to bath and bath to lumen. Both early and mid SFPCT and JMPCT were examined with B and E solutions as perfusate to determine specifically the role of glucose, amino acid, and bicarbonate removal on the PD. Whenever a segment was perfused with more than one solution it was reperfused with control A solution until its PD returned to its control value. All tubules that did not return to more than 80% of their control HgC12-Hg : Sat KCL: 4% Agarose: : 4% Agarose: Both: Perfusote: E1 E2 E3 E4 E5 E6 E7 4% Agarose: : 4% Agarose: Sat KCL HgC12 -Hg Sat KCL E8 E9 Elo Ell FIGURE 2 Schematic representation of the circuit used to measure liquid junction potentials. Functional Heterogeneity of Proximal Convoluted Tubules 821
5 TABLE II Transepithelial PD Response to a 50-mM NaCI Gradient from Lumen to Bath Control PD* Experimental PD$ mv mv 1st mm superficial proximal convolution -4.31: : nd mm superficial proximal convolution -4.6: Mid proximal convolution -4.5 ± Late proximal convolution ± st mm juxtamedullary convolution -4.7 ± ± Mid juxtamedullary convolution ± Late juxtamedullary convolution -3.8 ± ±0.4 6 ±SE. Results are * Perfusate, A solution; bath, rabbit serum. Perfusate, A solution; bath, A-50 solution. All experimental PD's corrected for liquid junction by +3.5 mv. PD were discarded (less than 1 in 10). All tubules were perfused at more than 10 nl/min, and never in the range where PD has been described to be flow dependent (11). The calculation of the relative permeability ratios of C1 to Na was done by the Goldman constant field equation: RT PNaENa]l + Pci[CI]b PD- In F PNa[Na]b + Pcl[CI]l where R is the gas constant; T absolute temperature; F the Faraday constant; PN. and Pe, the passive permeability coefficients of Na and Cl; [Na] and [Cl] the concentrations of Na and Cl, and I and b the lumen and bath. The data for each set of tubules is expressed as the mean±sem of the number of tubules studies. With respect to the NaCl gradient experiments, statistics were calculated from absolute potential changes. Data with B and E solution as perfusates were analyzed statistically with resultant PD, not PD changes. Both paired and unpaired t test were utilized when appropriate. RESULTS PD response to removal of NaCI from bath. Table II shows the PD response in the various proximal convoluted segments to an isosomotic 50 mmol/liter NaCl gradient from lumen to bath (A-50 in bath). The early SFPCT (1st mm) and all regions of the JMPCT showed negative PD deflections, indicating greater relative permeability to Na than Cl from lumen to bath. The 2ndmm, mid, and late SFPCT exhibited positive PD deflections, indicating greater relative permeability to Cl than Na from lumen to bath. The early SFPCT and the entire JMPCT were significantly different from the 2nd-mm, mid, and late SFPCT (P < ). The early SFPCT and JMPCT were not statistically different (P > 0.05). The early, mid, and late JMPCT were not different from each other, with all regions exhibiting essentially identical negative PD deflections, indicating greater relative Na than Cl permeability throughout. Symmetry of the PD response to bidirectional salt gradients. As shown in Table III, when the tubules were exposed alternately to a 50 mmol/liter NaCl gradient from lumen to bath and bath to lumen (A-50 as bath and as perfusate), the PD changes were symmetrical. In the early and mid SFPCT there was no statistical difference in the PD changes (P > 0.05). In the JMPCT the PD changes were statistically different (0.5 > P > 0.025), even though the absolute difference in the PD changes was small (3.8 mv PD change with A-50 as bath and 3.3 mv PD change with A-50 as perfusate). PD response to the removal of glucose, alanine, and HCOs from the perfusate. When the early and mid SFPCT and the JMPCT were perfused with solution containing no glucose or alanine (E solution), a significant reversible decrease in the transepithelial PD was observed in all segments (Table IV). All segments exhibited essentially the same response with a reversible decrease in the PD to less than - 1 mv lumen negative. However, with the additional maneuver of NaHCOs removal and its replacement with NaCl (B solution), which creates a Cl concentration gradient from lumen to bath, significant differences were found between the various segments (Table IV). Both the early SFPCT and the JMPCT developed small positive PD's of mv and mv, respectively, lumen positive, with a calculated liquid junction potential correction of mv. These responses were not statistically different from each other (P > 0.05). The mid SFPCT (also with a liquid junction correction of mv), on the other hand, developed a significantly more positive PD of mv when compared to the early SFPCT and JMPCT (P < ). These differences are attributed to the different Cl permeabilities of these segments. The PD's corrected by the experimentally determined liquid junction potential for B solution are also shown in Table IV. Although the differences between the segments are unchanged with a liquid junction correction of mv TABLE I I I Symmetrical PD Response to a 50-mM NaCl Gradient from Lumen to Bath and Bath to Lumen Control APD A-50 APD A-S0 PD 5SE Bath ±SE Perfusate ±SE n mv mv 1st mm superficial convolution -4.4± ± ±i0.41* 5 Mid superficial convolution -4.2 ± ± ± 1.1* 6 Juxtamedullary convolution -5.0± ± t 7 *P > 0.0S. t 0.05 > P > I All PD's corrected for liquid junction potential bath and -3.5 mv with A-50 as perfusate mv with A-So as 822 H. R. Jacobson and J. P. Kokko
6 TABLE IV PD Response to E and B Solutions as Perfusate E solution (s glucose or B solution amino acids) (tcl $HCO) Control (a) Control (n), 1st mm superficial convolution -4.3 ± ±40.2 (6) -4.3± ±0.2 (7)* Mid superficial convolution -4.3i ±0.1 (9) -4.3± ±0.1 (10) Juxtamedullary convolution -5.3±A ±0.2 (12) -5.2± ±0.1 (12) Bath solution was rabbit serum in all experiments. Results are ±SE. * With calculated liquid junction potential of +2.7 mv. t With experimental liquid junction potential of +0.7 mv. With experimental liquid junction potential of +1.0 mv. or + 1 mv instead of mv, the absolute PD values changed importantly to mv to mv for the JMPCT, to mv for the early SFPCT, and to for the mid SFPCT (Table IV). Although not depicted in Table IV, when the early and mid SFPCT and JMPCT were perfused with and bathed in B solution, the PD became zero. DISCUSSION It has been shown previously that the lumen negative transepithelial PD in the superficial proximal convoluted tubule is secondary to active transport processes and not entirely due to diffusion potentials (11). Subsequently Kokko (8) reported that the PD was dependent in a major way on the presence of glucose and amino acids in the luminal fluid. The present studies confirm this concept with respect to the SFPCT and show in addition that the JMPCT also appears to exhibit electrogenic Na transport coupled to organic solute transport. This is based on the observation that removal of glucose and amino acids from the perfusate always caused a less negative transtubular PD in all segments of the proximal convoluted tubule examined (Table IV). It thus appears that all proximal convoluted segments, superficial and juxtamedullary, regardless of distance from the glomerulus, have the capacity to transport sodium electrogenically when glucose and amino acids are present in the luminal fluid. In contrast to both the SFPCT and JMPCT, the pars recta of superficial and juxtamedullary nephrons TABLE V Relative Permeability of Cl to Na 1st mm superficial convolution nd mm superficial convolution 1.7 Mid superficial convolution 2.0 Late superficial convolution 3.2 Juxtamedullary convolution (all segments) 0.49 have been shown to exhibit electrogenic Na transport independent of organic solute transport (8). It is now widely accepted that electrophysiological techniques can be used to measure relative ion permeabilities. This is the first such attempt to measure the relative Cl-to-Na permeabilities along the length of the proximal convoluted tubule, and further to compare the SFPCT to the JMPCT. These studies show first that the SFPCT has different intrinsic functional properties along its length with respect to relative Cl and Na permeabilities. The relative Cl-to-Na permeability increases from glomerulus distally, as shown in Table V. Second, since the JMPCT has never been studied before, these experiments not only describe the relative Cl-to-Na permeabilities in the JMPCT but also show that the JMPCT and SFPCT differ functionally in two major ways: the JMPCT does not appear to have differing Cl-to-Na permeabilities along its length; the JMPCT has a greater relative Na-to-Cl permeability throughout, while the SFPCT has a greater relative Cl-to-Na permeability along the majority of its length. Third, the symmetry of the PD change due to equivalent salt gradients imposed on either side of the tubule suggests that the ions involved in generating the diffusion PD permeate through identical pathways from lumen to bath and bath to lumen, rather than through two membranes of differing permeabilities. The symmetry of the bidirectional diffusion PD's also suggest that the PD changes with imposed salt gradients are not secondary to alteration of the baseline active transport. These studies are therefore in agreement with previous electrophysiological studies of the SFPCT in the dog kidney (12), where it is believed that passive permeabilites determined electrophysiologically with salt gradients reflect the permeability of a low-resistance paracellular pathway. The experiments using a high-cl, low-hcos perfusate (B solution) support the differences in the relative Cl-to- Na permeability of the SFPCT and JMPCT. If the intraluminal fluid in the latter portions of the SFPCT and Functional Heterogeneity of Proximal Convoluted Tubules 823
7 FIGuRE 3 Proposed potential profile of the two populations of proximal tubules of the rabbit: superficial (SF) and juxtamedullary (JM). The pars recta data are from ref 7. JMPCT in vivo are similar, one would expect differences in the in vivo PD's. These PD differences could be significant because of their resultant differing effects on passive transport phenomena. A positive intraluminal PD would act as a driving force for efflux of Na+, while a negative intraluminal PD would retard the efflux of Na'. Direct analysis of this question will require flux measurements in both the SF- and JMPCT with B solution. At present, calculation of the degree of importance of the chloride diffusion potential (or lack of it) in the SFPCT and JMPCT is difficult because the magnitude of the PD is unclear and the chloride permeability coefficients in these segments have not been measured. Barret et al. (14) have shown that in the SFPCT of the rat the first loop accessable to micropuncture has a negative PD, while the segments beyond have a positive PD of approximately + 1 mv. Kokko (8), using the calculated liquid junction potential correction of mv, has previously suggested that the latter portion of the SFPCT has a PD of approximately + 3 mv lumen positive. This previously published + 3 mv PD for the latter portion of the SFPCT is probably too high in view of the discrepancies between the previously calculated and the present directly measured liquid junction potentials (Table IV). With the high-chloride perfusate (B solution) and the directly measured liquid junction PD (in contrast to the calculated value), the PD in the latter segments of the SFPCT is in close agreement with the PD reported in vivo by Barrett et al. (13). In summary, the present studies are the first to establish that the SFPCT and JMPCT have intrinsically different characteristics. In the SFPCT the relative Na-to- Cl permeability varies along the length of the tubule, whereas in the JMPCT the relative Na-to-Cl permeability remains constant along the entire length of the tubule. In the SFPCT, Na is more permeant than Cl in the 1st mm from the glomerulus, whereas in its latter portions Cl is more permeant than Na. On the other hand, Na is more permeant than Cl throughout the JMPCT. These findings suggest that differing diffusion potentials exist along the length of these two populations of nephrons (Fig. 3). We have also shown that the JMPCT is similar to the SFPCT in that it is capable of electrogenic Na transport coupled to organic solute transport, and, therefore, the PD would be negative in early segment of each nephron when glucose and amino acids exist in significant concentrations (Fig. 3). Since the final urine is the composite of all nephron populations and since in various salt-retaining or -excreting models there exists redistribution of filtrate and blood between superficial and juxtamedullary regions of the cortex, it may be argued that the differing characteristics between the superficial and juxtamedullary nephrons contribute differently to final urine composition. ACKNOWLEDGMENTS The authors would like to express their appreciation to Mrs. Virginia Borcoman and Miss Jan Hawkins for their technical assistance. This work was supported in part by U. S. Public Health Service Program Grant P01 HL and National Institute of Arthritis and Metabolic Diseases Research Grant 1 RO1 AM REFERENCES 1. Huber, G. C The morphology and structure of the mammalian renal tubule. Harvey Lect. 5: Bartel, A. G., R. M. Zakheim, L. G. Jones, and A. C. Barger Redistribution of renal blood flow produced by furosemide and ethacrynic acid. Clin. Res. 21: Jamison, R. L Micropuncture study of superficial and juxtamedullary nephrons in the rat. Am. J. Physiol. 218: Horster, M., and K. Thurau Micropuncture studies on the filtration rate of single superficial and juxtamedullary glomeruli in the rat kidney. Pflugers Archiv. Gesamte Physiol. Menschen Tiere. 301: Hollenberg, N. K., M. Epstein, R. D. Guttmann, M. Conroy, R. T. Basch, and J. P. Merrill Effect of Na balance on intrarenal distribution of blood flow in normal man. J. AppI. Physiol. 28: Trueta, J., A. E. Barclay, P. M. Daniel, K. J. Franklin, and M. M. L. Prichard Studies of the Renal Circulation. Blackwell Scientific Publications Ltd., Oxford, England. 187 pp. 7. Kawamura, S., M. Imai, D. W. Seldin, and J. P. Kokko Characteristics of salt and water transport in superficial and juxtamedullary straight segments of proximal tubules. J. Clin. Invest. 55: H. R. Jacobson and J. P. Kokko
8 8. Kokko, J. P Proximal tubule potential difference. Dependence on glucose, HCO., and amino acids. J. Clin. Invest. 52: Burg, M., J. Grantham, M. Abramow, and J. Orloff Preparation and study of fragments of single rabbit nephrons. Am. J. Physiol. 210: Barry, P. H., and J. M. Diamond Junction potentials, electrode standard potentials and other problems in interpreting electrical properties of membranes. J. Membr. Biol. 3: Kokko, J. P., and F. C. Rector Flow dependence of transtubular potential difference in isolated perfused segments of rabbit proximal convoluted tubule. J. Clin. Invest. 50: Boulpaep, E. L., and J. F. Seely Electrophysiology of proximal and distal tubules in the autoperfused dog kidney. Am. J. Physiol. 221: Barratt, L. J., F. C. Rector, J. P. Kokko, and D. W. Seldin Factors governing the transepithelial potential difference across the proximal tubule of the rat kidney. J. Clin. Invest. 53: Functional Heterogeneity of Proximal Convoluted Tubules 825
Duct Acidification Effects of Potassium, HCO3. Harry R. Jacobson. Internal Medicine, 5323 Harry Hines Blvd., Dallas, Texas 75235
 Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Medullary Collecting Duct Acidification Effects of Potassium, HCO3 Concentration, and pco2 Harry R. Jacobson University of Texas Health Science Center, Department of Internal Medicine, 5323 Harry Hines
Diagram of the inner portions of the kidney
 Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion
 Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion OLGA H. BROKL AND WILLIAM H. DANTZLER Department of Physiology, College of Medicine, University of Arizona, Tucson,
Amino acid fluxes in rat thin limb segments of Henle s loop during in vitro microperfusion OLGA H. BROKL AND WILLIAM H. DANTZLER Department of Physiology, College of Medicine, University of Arizona, Tucson,
Volume Reabsorption, Transepithelial Potential Differences, and Ionic Permeability Properties in Mammalian Superficial Proximal Straight Tubules
 Published Online: 1 November, 1974 Supp Info: http://doi.org/10.1085/jgp.64.5.582 Downloaded from jgp.rupress.org on August 26, 2018 Volume Reabsorption, Transepithelial Potential Differences, and Ionic
Published Online: 1 November, 1974 Supp Info: http://doi.org/10.1085/jgp.64.5.582 Downloaded from jgp.rupress.org on August 26, 2018 Volume Reabsorption, Transepithelial Potential Differences, and Ionic
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
Urea Secretion by the Straight Segment
 Urea Secretion by the Straight Segment of the Proximal Tubule SATOSHI KAWAMURA and JUHA P. KOKKO From the Department of Internal Medicine, the University of Texas Health Science Center at Dallas, Southwestern
Urea Secretion by the Straight Segment of the Proximal Tubule SATOSHI KAWAMURA and JUHA P. KOKKO From the Department of Internal Medicine, the University of Texas Health Science Center at Dallas, Southwestern
Calcium and Phosphate Transport in Isolated
 Calcium and Phosphate Transport in Isolated Segments of Rabbit Henle's Loop ANTONINO S. RocHA, JOSE B. MAGALDI, and JUHA P. KOKKO From Unidade de Doencas Renais, Department de Clinica Medica, Hospital
Calcium and Phosphate Transport in Isolated Segments of Rabbit Henle's Loop ANTONINO S. RocHA, JOSE B. MAGALDI, and JUHA P. KOKKO From Unidade de Doencas Renais, Department de Clinica Medica, Hospital
Functions of Proximal Convoluted Tubules
 1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
Na + Transport 1 and 2 Linda Costanzo, Ph.D.
 Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Transport characteristics of in vitro perfused proximal convoluted tubules
 Kidney International, Vol. 22 (/982), pp. 425 433 Transport characteristics of in vitro perfused proximal convoluted tubules HARRY R. JACOBSON The University of Texas Health Science Center, The Southwestern
Kidney International, Vol. 22 (/982), pp. 425 433 Transport characteristics of in vitro perfused proximal convoluted tubules HARRY R. JACOBSON The University of Texas Health Science Center, The Southwestern
Transport characteristics of the thin limbs of Henle
 Kidney International, Vol. 22 (1982), pp. 449 453 Transport characteristics of the thin limbs of Henle JUHA P. KOKKO Department of Internal Medicine, University of Texas Health Science Center, Southwestern
Kidney International, Vol. 22 (1982), pp. 449 453 Transport characteristics of the thin limbs of Henle JUHA P. KOKKO Department of Internal Medicine, University of Texas Health Science Center, Southwestern
Medullary Thick Ascending Limb of Henle
 Sodium Chloride and Water Transport in the Medullary Thick Ascending Limb of Henle EVIDENCE FOR ACTIVE CHLORIDE TRANSPORT ANTONINO S. RoCHA and JUHA P. KoKKo From the Department of Internal Medicine, The
Sodium Chloride and Water Transport in the Medullary Thick Ascending Limb of Henle EVIDENCE FOR ACTIVE CHLORIDE TRANSPORT ANTONINO S. RoCHA and JUHA P. KoKKo From the Department of Internal Medicine, The
Sodium and chlorine transport
 Kidney physiology 2 Sodium and chlorine transport The kidneys help to maintain the body's extracellular fluid (ECF) volume by regulating the amount of Na+ in the urine. Sodium salts (predominantly NaCl)
Kidney physiology 2 Sodium and chlorine transport The kidneys help to maintain the body's extracellular fluid (ECF) volume by regulating the amount of Na+ in the urine. Sodium salts (predominantly NaCl)
MATURATION OF RAT PROXIMAL TUBULE CHLORIDE PERMEABILITY. Michel Baum and Raymond Quigley. Departments of Pediatrics and Internal Medicine
 Articles in PresS. Am J Physiol Regul Integr Comp Physiol (July 28, 2005). doi:10.1152/ajpregu.00257.2005 R-00257-2005.R2 MATURATION OF RAT PROXIMAL TUBULE CHLORIDE PERMEABILITY Michel Baum and Raymond
Articles in PresS. Am J Physiol Regul Integr Comp Physiol (July 28, 2005). doi:10.1152/ajpregu.00257.2005 R-00257-2005.R2 MATURATION OF RAT PROXIMAL TUBULE CHLORIDE PERMEABILITY Michel Baum and Raymond
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
BIOLOGY - CLUTCH CH.44 - OSMOREGULATION AND EXCRETION.
 !! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
!! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
After studying this lecture, you should be able to...
 Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Functional morphology of kidneys Clearance
 Functional morphology of kidneys Clearance Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important
Functional morphology of kidneys Clearance Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important
11/05/1431. Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate
 Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Urinary bladder provides a temporary storage reservoir for urine
 Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX
 One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion.
 The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
Collin College. BIOL Anatomy & Physiology. Urinary System. Summary of Glomerular Filtrate
 Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction Once the filtrate is formed, the early
Early Filtrate Processing Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction Once the filtrate is formed, the early
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Excretion Chapter 29. The Mammalian Excretory System consists of. The Kidney. The Nephron: the basic unit of the kidney.
 Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Homeostatic Regulation
 Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Renal Physiology II Tubular functions
 Renal Physiology II Tubular functions LO. 42, 43 Dr. Kékesi Gabriella Basic points of renal physiology 1. Glomerular filtration (GF) a) Ultrafiltration 2. Tubular functions active and passive a) Reabsorption
Renal Physiology II Tubular functions LO. 42, 43 Dr. Kékesi Gabriella Basic points of renal physiology 1. Glomerular filtration (GF) a) Ultrafiltration 2. Tubular functions active and passive a) Reabsorption
Urinary Physiology. Chapter 17 Outline. Kidney Function. Chapter 17
 Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
The regulation of renal acid secretion: New observations from studies of distal nephron segments
 Kidney International, Vol. 29 (1986), pp. 1099 1109 EDITORIAL REVIEW The regulation of renal acid secretion: New observations from studies of distal nephron segments Forty years ago, in a landmark paper,
Kidney International, Vol. 29 (1986), pp. 1099 1109 EDITORIAL REVIEW The regulation of renal acid secretion: New observations from studies of distal nephron segments Forty years ago, in a landmark paper,
The kidneys are excretory and regulatory organs. By
 exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
A. Correct! Flushing acids from the system will assist in re-establishing the acid-base equilibrium in the blood.
 OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
Kidney and urine formation
 Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Kidney and urine formation Renal structure & function Urine formation Urinary y concentration and dilution Regulation of urine formation 1 Kidney and urine formation 1.Renal structure & function 1)General
Normal Renal Function
 Normal Renal Function Functions of the Kidney: balances solute and water transport excretes metabolic waste products conserves nutrient regulates acid-base balance secretes hormones that help regulate
Normal Renal Function Functions of the Kidney: balances solute and water transport excretes metabolic waste products conserves nutrient regulates acid-base balance secretes hormones that help regulate
Nephron Function and Urine Formation. Ms. Kula December 1, 2014 Biology 30S
 Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate
 Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Renal Physiology - Lectures
 Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
41B. Metabolism produces wastes that must be eliminated from the body. This. Renal System Physiology: Computer Simulation
 41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
41B E X E R C I S E Renal System Physiology: Computer Simulation O B J E C T I V E S 1. To define the following terms: glomerulus, glomerular capsule, renal corpuscle, renal tubule, nephron, proximal convoluted
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Transport across the cell membrane
 Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
1. Anatomy / Vascularisation. 2. Urine concentration. 3. Axial heterogeneity of some segments
 Lise BANKIR 1. Anatomy / Vascularisation 2. Urine concentration 3. Axial heterogeneity of some segments Rat kidney. Arterial filling with Microfil silicone rubber Alcian Blue staining Filling of arterial
Lise BANKIR 1. Anatomy / Vascularisation 2. Urine concentration 3. Axial heterogeneity of some segments Rat kidney. Arterial filling with Microfil silicone rubber Alcian Blue staining Filling of arterial
Potassium secretion. E k = -61 log ([k] inside / [k] outside).
![Potassium secretion. E k = -61 log ([k] inside / [k] outside). Potassium secretion. E k = -61 log ([k] inside / [k] outside).](/thumbs/80/80478709.jpg) 1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
Urinary System. consists of the kidneys, ureters, urinary bladder and urethra
 Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
Urinary System 1 Urinary System consists of the kidneys, ureters, urinary bladder and urethra 2 Location of Kidneys The kidneys which are positioned retroperitoneally lie on either side of the vertebral
fluid, *7 in peritubular capillary plasma and in
 J. Phy8iol. (1977), 273, pp. 765-773 765 With 2 text-ftgurem Printed in Great Britain COMPARISON OF CHLORIDE CONCENTRATION AND OSMOLALITY IN PROXIMAL TUBULAR FLUID, PERITUBULAR CAPILLARY PLASMA AND SYSTEMIC
J. Phy8iol. (1977), 273, pp. 765-773 765 With 2 text-ftgurem Printed in Great Britain COMPARISON OF CHLORIDE CONCENTRATION AND OSMOLALITY IN PROXIMAL TUBULAR FLUID, PERITUBULAR CAPILLARY PLASMA AND SYSTEMIC
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Osmoregulation and Renal Function
 1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
1 Bio 236 Lab: Osmoregulation and Renal Function Fig. 1: Kidney Anatomy Fig. 2: Renal Nephron The kidneys are paired structures that lie within the posterior abdominal cavity close to the spine. Each kidney
Glomerular Capillary Blood Pressure
 Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
MS1 Physiology Review of Na+, K+, H + /HCO 3. /Acid-base, Ca+² and PO 4 physiology
 MS1 Physiology Review of,, / /Acidbase, Ca+² and PO 4 physiology I. David Weiner, M.D. Professor of Medicine and Physiology University of Florida College of Medicine Basic principles Proximal tubule Majority
MS1 Physiology Review of,, / /Acidbase, Ca+² and PO 4 physiology I. David Weiner, M.D. Professor of Medicine and Physiology University of Florida College of Medicine Basic principles Proximal tubule Majority
November 30, 2016 & URINE FORMATION
 & URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
& URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
19. RENAL PHYSIOLOGY ROLE OF THE URINARY SYSTEM THE URINARY SYSTEM. Components and function. V BS 122 Physiology II 151 Class of 2011
 19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
Collin County Community College RENAL PHYSIOLOGY
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
I. Metabolic Wastes Metabolic Waste:
 I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
Question 1: Solution 1: Question 2: Question 3: Question 4: Class X The Excretory System Biology
 A. MULTIPLE CHOICE TYPE: (select the most appropriate option in each case) Book Name: Selina Concise Question 1: Excretion primarily involves (a) removal of all byproducts during catabolism (b) removal
A. MULTIPLE CHOICE TYPE: (select the most appropriate option in each case) Book Name: Selina Concise Question 1: Excretion primarily involves (a) removal of all byproducts during catabolism (b) removal
Transport of Ammonia in the Rabbit Cortical Collecting Tubule
 Transport of Ammonia in the Rabbit Cortical Collecting Tubule L. Lee Hamm, D. Trigg, D. Martin, C. Gillespie, and J. Buerkert Renal Division, Department ofmedicine, Washington University School ofmedicine,
Transport of Ammonia in the Rabbit Cortical Collecting Tubule L. Lee Hamm, D. Trigg, D. Martin, C. Gillespie, and J. Buerkert Renal Division, Department ofmedicine, Washington University School ofmedicine,
Salt and Water Balance and Nitrogen Excretion
 Announcements Exam is in class on WEDNESDAY. Bring a #2 pencil and your UFID. You must come to your registered class section (except those with DRC accommodations). Office hours Mon 1-3 pm. Teaching evals:
Announcements Exam is in class on WEDNESDAY. Bring a #2 pencil and your UFID. You must come to your registered class section (except those with DRC accommodations). Office hours Mon 1-3 pm. Teaching evals:
Nephron Anatomy Nephron Anatomy
 Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Chapter 44. Osmoregulation and Excretion
 Chapter 44 Osmoregulation and Excretion Overview: A Balancing Act Physiological systems of animals operate in a fluid environment Relative concentrations of water and solutes must be maintained within
Chapter 44 Osmoregulation and Excretion Overview: A Balancing Act Physiological systems of animals operate in a fluid environment Relative concentrations of water and solutes must be maintained within
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Renal Pharmacology. Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics BIMM118
 Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
Excretory Lecture Test Questions Set 1
 Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Human Urogenital System 26-1
 Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
CREATININE: is another nitrogenous waste. Creatinine comes from creatinine phosphate in muscle metabolism (a Phosphate-storage molecule)
 BIOLOGY 12 - EXCRETION: CHAPTER NOTES Your cells are constantly carrying out chemical reactions to maintain homeostasis. Many of these chemical reactions produce wastes that must be removed from cells
BIOLOGY 12 - EXCRETION: CHAPTER NOTES Your cells are constantly carrying out chemical reactions to maintain homeostasis. Many of these chemical reactions produce wastes that must be removed from cells
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Functional Profile of the Isolated Uremic Nephron
 Functional Profile of the Isolated Uremic Nephron EVIDENCE OF PROXIMAL TUBULAR "MEMORY" IN EXPERIMENTAL RENAL DISEASE WALTER TRIZNA, NORIMOTO YANAGAWA, YAACOV BAR-KHAYIM, BARBARA HOUSTON, and LEON G. FINE,
Functional Profile of the Isolated Uremic Nephron EVIDENCE OF PROXIMAL TUBULAR "MEMORY" IN EXPERIMENTAL RENAL DISEASE WALTER TRIZNA, NORIMOTO YANAGAWA, YAACOV BAR-KHAYIM, BARBARA HOUSTON, and LEON G. FINE,
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi
 Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
The Urinary System 15PART A. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART A Functions of the Urinary System Elimination of waste products Nitrogenous
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART A Functions of the Urinary System Elimination of waste products Nitrogenous
Use the following diagram to answer the next question. 1. In the diagram above, pressure filtration occurs in a. W b. X c. Y d. Z
 Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Excretory System Workbook
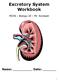 Excretory System Workbook MCHS Biology 20 Mr. Korotash Name: Date: 1 Study the diagram above. Name the structures and indicate their functions by completing the following table: Structure 1. Function 2.
Excretory System Workbook MCHS Biology 20 Mr. Korotash Name: Date: 1 Study the diagram above. Name the structures and indicate their functions by completing the following table: Structure 1. Function 2.
Characterization of Acidification in the Cortical and Medullary Collecting Tubule of the Rabbit
 Characterization of Acidification in the Cortical and Medullary Collecting Tubule of the Rabbit MELVIN E. LASKI and NEIL A. KURTZMAN, with the technical assistance of VICKY MORGAN, Section of Nephrology,
Characterization of Acidification in the Cortical and Medullary Collecting Tubule of the Rabbit MELVIN E. LASKI and NEIL A. KURTZMAN, with the technical assistance of VICKY MORGAN, Section of Nephrology,
Identify and describe. mechanism involved in Glucose reabsorption
 Define tubular reabsorption, tubular secretion, transcellular and paracellular transport. Identify and describe mechanism involved in Glucose reabsorption Describe tubular secretion with PAH transport
Define tubular reabsorption, tubular secretion, transcellular and paracellular transport. Identify and describe mechanism involved in Glucose reabsorption Describe tubular secretion with PAH transport
CONTROLLING THE INTERNAL ENVIRONMENT
 AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
AP BIOLOGY ANIMAL FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROLLING THE INTERNAL ENVIRONMENT KIDNEY AND NEPHRON NEPHRON FUNCTIONS Animal Form & Function Activity #5 page 1 NEPHRON STRUCTURE NEPHRON
Kidney Structure. Renal Lobe = renal pyramid & overlying cortex. Renal Lobule = medullary ray & surrounding cortical labryinth.
 Kidney Structure Capsule Hilum ureter renal pelvis major and minor calyxes renal and vein segmental arteries interlobar arteries arcuate arteries interlobular arteries Medulla renal pyramids cortical/renal
Kidney Structure Capsule Hilum ureter renal pelvis major and minor calyxes renal and vein segmental arteries interlobar arteries arcuate arteries interlobular arteries Medulla renal pyramids cortical/renal
Sodium Chloride and Water Transport
 Sodium Chloride and Water Transport in the Descending Limb of Henle JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas (Southwestern) Medical School, Dallas, Texas 75235 A
Sodium Chloride and Water Transport in the Descending Limb of Henle JuHA P. KoKKo From the Department of Internal Medicine, The University of Texas (Southwestern) Medical School, Dallas, Texas 75235 A
Osmoregulation regulates solute concentrations and balances the gain and loss of water
 Ch 44 Osmoregulation & Excretion Osmoregulation regulates solute concentrations and balances the gain and loss of water Freshwater animals show adaptations that reduce water uptake and conserve solutes
Ch 44 Osmoregulation & Excretion Osmoregulation regulates solute concentrations and balances the gain and loss of water Freshwater animals show adaptations that reduce water uptake and conserve solutes
RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015
 RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015 1. In which abdominal cavity do the kidneys lie? a) Peritoneum. b) Anteperitoneal. c) Retroperitoneal. d) Parietal peritoneal 2. What is the
RNPDC CCNP Anatomy and Physiology: Renal System Pre-Quiz 2015 1. In which abdominal cavity do the kidneys lie? a) Peritoneum. b) Anteperitoneal. c) Retroperitoneal. d) Parietal peritoneal 2. What is the
Calcium Transport in the Thick Ascending Limb of Henle
 Calcium Transport in the Thick Ascending Limb of Henle HETEROGENEITY OF FUNCTION IN THE MEDULLARY AND CORTICAL SEGMENTS WADI N. SUKI, DIANE ROUSE, ROLAND C. K. NG, and JUHA P. KOKKO, Renal Section, Department
Calcium Transport in the Thick Ascending Limb of Henle HETEROGENEITY OF FUNCTION IN THE MEDULLARY AND CORTICAL SEGMENTS WADI N. SUKI, DIANE ROUSE, ROLAND C. K. NG, and JUHA P. KOKKO, Renal Section, Department
Urine Formation. Urinary Physiology Urinary Section pages Urine Formation. Glomerular Filtration 4/24/2016
 Urine Formation Urinary Physiology Urinary Section pages 9-17 Filtrate Blood plasma minus most proteins Urine
Urine Formation Urinary Physiology Urinary Section pages 9-17 Filtrate Blood plasma minus most proteins Urine
Urinary System. Dr. ZHANG Xiong. Dept. of Physiology. ZJU School of Medicine. QUESTION 6
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1)
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1) Dr. Attila Nagy 2017 Functional roles of the kidney 1.Homeostasis of fluid compartments (isosmia, isovolemia, isoionia, isohydria,) 2. Elimination
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (1) Dr. Attila Nagy 2017 Functional roles of the kidney 1.Homeostasis of fluid compartments (isosmia, isovolemia, isoionia, isohydria,) 2. Elimination
Hill et al. 2004, Fig. 27.6
 Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Urinary system. Kidney anatomy Renal cortex Renal. Nephrons
 Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Influence of Peritubular Protein on Solute Absorption
 Influence of Peritubular Protein on Solute Absorption in the Rabbit Proximal Tubule A SPECIFIC EFFECT ON NaCI TRANSPORT CHRISTINE A. BERRY and MARTIN G. COGAN, Cardiovascular Research Institute, and Departments
Influence of Peritubular Protein on Solute Absorption in the Rabbit Proximal Tubule A SPECIFIC EFFECT ON NaCI TRANSPORT CHRISTINE A. BERRY and MARTIN G. COGAN, Cardiovascular Research Institute, and Departments
Chapter 23. The Nephron. (functional unit of the kidney
 Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
Chapter 23 The Nephron (functional unit of the kidney Renal capsule The Nephron Renal cortex Nephron Collecting duct Efferent arteriole Afferent arteriole (a) Renal corpuscle: Glomerular capsule Glomerulus
QUIZ/TEST REVIEW NOTES SECTION 2 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19
![QUIZ/TEST REVIEW NOTES SECTION 2 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 QUIZ/TEST REVIEW NOTES SECTION 2 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19](/thumbs/80/80449526.jpg) 1 QUIZ/TEST REVIEW NOTES SECTION 2 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 Learning Objectives: Differentiate the following processes: filtration, reabsorption, secretion, excretion
1 QUIZ/TEST REVIEW NOTES SECTION 2 RENAL PHYSIOLOGY FILTRATION [THE KIDNEYS/URINARY SYSTEM] CHAPTER 19 Learning Objectives: Differentiate the following processes: filtration, reabsorption, secretion, excretion
BIOL2030 Human A & P II -- Exam 6
 BIOL2030 Human A & P II -- Exam 6 Name: 1. The kidney functions in A. preventing blood loss. C. synthesis of vitamin E. E. making ADH. B. white blood cell production. D. excretion of metabolic wastes.
BIOL2030 Human A & P II -- Exam 6 Name: 1. The kidney functions in A. preventing blood loss. C. synthesis of vitamin E. E. making ADH. B. white blood cell production. D. excretion of metabolic wastes.
Class XI - Biology Excretory Products and their Elimination
 Class XI - Biology Excretory Products and their Elimination MULTIPLE CHOICE QUESTIONS 1. The following substances are the excretory products in animals. Choose the least toxic form among them? a. Urea
Class XI - Biology Excretory Products and their Elimination MULTIPLE CHOICE QUESTIONS 1. The following substances are the excretory products in animals. Choose the least toxic form among them? a. Urea
Membrane Transport. Anatomy 36 Unit 1
 Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
A. Incorrect! The urinary system is involved in the regulation of blood ph. B. Correct! The urinary system is involved in the synthesis of vitamin D.
 Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Human Anatomy - Problem Drill 22: The Urinary System Question No. 1 of 10 1. Which of the following statements about the functions of the urinary system is not correct? Question #01 (A) The urinary system
Sodium Chloride, Urea, and Water Transport
 Sodium Chloride, Urea, and Water Transport in the Thin Ascending Limb of Henle GENERATION OF OSMOTIC GRADIENTS BY PASSIVE DIFFUSION OF SOLUTES MASASHI IMAI and JUHA P. KOKKO From the Department of Internal
Sodium Chloride, Urea, and Water Transport in the Thin Ascending Limb of Henle GENERATION OF OSMOTIC GRADIENTS BY PASSIVE DIFFUSION OF SOLUTES MASASHI IMAI and JUHA P. KOKKO From the Department of Internal
