DEVELOPMENT AND DISEASE FGF10 maintains stem cell compartment in developing mouse incisors
|
|
|
- Lindsey Bryant
- 5 years ago
- Views:
Transcription
1 Development 129, (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV DEVELOPMENT AND DISEASE FGF10 maintains stem cell compartment in developing mouse incisors Hidemitsu Harada 1, *, Takashi Toyono 1, Kuniaki Toyoshima 1, Masahiro Yamasaki 2, Nobuyuki Itoh 2, Shigeaki Kato 3, Keisuke Sekine 3 and Hideyo Ohuchi 4 1 Second Department of Oral Anatomy and Cell Biology, Kyushu Dental College, 2-6-1, Manazuru, Kokurakita-ku, Kitakyushu, , Japan 2 Department of Genetic Biochemistry, Graduate School of Pharmaceutical Sciences, Kyoto University, Yoshida-shimo- Adachi-cho, Sakyo-ku, Kyoto City, Kyoto, , Japan 3 Institute of Molecular and Cellular Biosciences, The University of Tokyo, Tokyo , Japan 4 Department of Biological Science and Technology, Faculty of Engineering, University of Tokushima, 2-1 Minami-Jyosanjima-cho, Tokushima City, Tokushima , Japan *Author for correspondence ( hide-h@mail.kyu-dent.ac.jp) Accepted 14 December 2001 SUMMARY Mouse incisors are regenerative tissues that grow continuously throughout life. The renewal of dental epithelium-producing enamel matrix and/or induction of dentin formation by mesenchymal cells is performed by stem cells that reside in cervical loop of the incisor apex. However, little is known about the mechanisms of stem cell compartment formation. Recently, a mouse incisor was used as a model to show that fibroblast growth factor (FGF) 10 regulates mitogenesis and fate decision of adult stem cells. To further illustrate the role of FGF10 in the formation of the stem cell compartment during tooth organogenesis, we have analyzed incisor development in Fgf10-deficient mice and have examined the effects of neutralizing anti-fgf10 antibody on the developing incisors in organ cultures. The incisor germs of FGF10-null mice proceeded to cap stage normally. However, at a later stage, the cervical loop was not formed. We found that the absence of the cervical loop was due to a divergence in Fgf10 and Fgf3 expression patterns at E16. Furthermore, we estimated the growth of dental epithelium from incisor explants of FGF10-null mice by organ culture. The dental epithelium of FGF10-null mice showed limited growth, although the epithelium of wild-type mice appeared to grow normally. In other experiments, a functional disorder of FGF10, caused by a neutralizing anti-fgf10 antibody, induced apoptosis in the cervical loop of developing mouse incisor cultures. However, recombinant human FGF10 protein rescued the cervical loop from apoptosis. Taken together, these results suggest that FGF10 is a survival factor that maintains the stem cell population in developing incisor germs. Key words: Stem cells, FGF10, Incisor development, Epithelialmesenchymal interactions, Mouse INTRODUCTION During embryogenesis, a single fertilized oocyte gives rise to a multicellular organism whose cells and tissues adopt differentiated characteristics or fates to perform the specified functions of each organ of the body. Many tissues and organs maintain a process known as homeostasis: as cells die, either by apoptosis or by injury, they are replenished. Additionally, the epidermis, hair, small intestine and hematopoietic system are all examples of adult tissues that exist naturally in a state of dynamic flux. Even in the absence of injury, these structures continually give rise to new cells, and are able to divide transiently, terminally differentiate and die. These regenerative tissues are maintained by adult stem cells, which have both the capacity to self-renew, to divide and create additional stem cells, and also to differentiate along a specified molecular pathway. In recent years tremendous advances have been made in our understanding of identification of adult stem cells (reviewed by Fuchs and Segre, 2000). However, molecular mechanisms for establishing adult stem cell compartments during embryogenesis have not been examined in detail. The mouse incisor tooth is an excellent model for analyzing certain aspects of stem cell regulation and function (Harada et al., 1999). The continuous eruption of mouse incisors throughout the lifetime of an animal is maintained by the division of germs localized in the cervical loop of the apical region. We have previously shown that stem cells divide rarely and asymmetrically, and one daughter cell remains as an undifferentiated stem cell in the cervical loop, whereas the other cell moves to the incisal end, giving rise to transitamplifying cells. Dental epithelial stem cells in the cervical loop give rise to four cell-lineages inner enamel epithelium (ameloblast cell-lineage), stratum intermedium, stellate reticulum and outer enamel epithelium.
2 1534 H. Harada and others The ability to select among multiple terminal differentiation pathways and to generate cells that can migrate in the selected direction are characteristics that parallel those of the hair follicle bulge or of crypt cells of the small intestine (Rochat et al., 1994; Fuchs and Segre, 2000). These stem cells seem to rely upon mesenchymal cues for their survival and differentiation (Korinek et al., 1998; DasGupta and Fuchs, 1999; Millar et al., 1999). Epithelial-mesenchymal interactions play an essential role in regulating a wide variety of developmental processes, including those of the teeth (Thesleff and Sharpe, 1997; Peters and Balling, 1999; Hogan, 1999). Tissue recombination experiments have shown that the mesenchymal tissue controls advancing morphogenesis after initiation of tooth development and regulates the continuous growth of epithelium in the mouse incisor (Koller and Baird, 1969). Fibroblast growth factors (FGFs) play a crucial role in the developing tooth (Kettunen and Thesleff, 1999). Our recent studies have shown that mouse incisor mesenchymal cells express Fgf3 and Fgf10, and their receptors, Fgfr1b and Fgfr2b, are expressed throughout the dental epithelium (Kettunen et al., 1998; Harada et al., 1999). Furthermore, beadimplantation assays have shown that FGF10 signaling from the mesenchyme indirectly regulated cell division and fate decision of epithelial stem cells by modulating the Notch pathway in the cervical loop via stimulation of lunatic fringe expression. The involvement of FGF10 in tooth morphogenesis was further indicated by the hypoplastic tooth organ in FGF10 null mice (Ohuchi et al., 2000). In the present study, which focuses on mesenchymal molecular cues for forming the stem cell compartment during organogenesis, we used the developing mouse incisor germ cell as a model of regenerative tissues. To further examine the role of FGF10 during incisor development, we have designed two loss-of-function experimental lines. Analysis of developing incisor germ of Fgf10-deficient mice showed that FGF10 is not involved in the early signaling networks that regulate tooth initiation at early morphogenesis, but is involved in the establishment of adults stem cells. Other experiments using neutralizing antibody provided direct evidence that FGF10 prevents apoptosis in the stem cell compartment. We propose a model that demonstrates the formation of adult stem cells during organogenesis and shows that stem cells are maintained as undifferentiated cells by mesenchymal signals. MATERIALS AND METHODS Fgf10-deficient mice Heads from Fgf10 / mice were obtained at desired embryonic stages from intercrosses of heterozygous breeding pairs. Genotyping was performed as described previously (Sekine et al., 1999). Tissues were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS), dehydrated in graded ethanol, embedded in paraffin and sectioned. The sections were stained with Hematoxylin and Eosin. Immunohistochemistry The sections were incubated with rabbit anti-keratin (L-1824, DAKO, Carpinteria, CA) as a primary antibody for 20 minutes at room temperature. The anti-rabbit Vectastain ABC-kit (PK-6101, Vector Laboratories) and the chromogen DAB (Vector Laboratories, Burlingame, CA) were used for antibody detection. The sections were then counterstained with Hematoxylin. Fig. 1. The developing mouse incisor during the initiation (E11), bud (E13), cap (E14), early bell (E16) and late bell (E19.5) stages. The initial stages of morphogenesis are very similar in all teeth. The first morphological sign of incisor development is a thickening of the oral epithelium (E11), which subsequently buds into the underlying mesenchyme (E13, bud stage). At later stages, the developing incisor rotates anteroposteriorly (E14, cap stage) and becomes parallel to the long axis of the incisors (E16, early bell stage). At early bell stage (E16), the cervical loop is seen at the apical end of the labial epithelium. Only the labial epithelium gives rise to the enamelforming ameloblasts (E19.5, late bell stage). Epithelium in dark blue, dental mesenchyme in light blue dots, enamel in red and dentin in green. cl, cervical loop; cm, condensed mesenchyme; d, dentine; dp, dental papilla; e, enamel; eo, enamel organ; m, molar; oe, oral epithelium; p, pulp; vl, vestibular lamina. Organ culture experiments Culture methods were as described previously (Harada et al., 1999). Dissected incisors from the mandible of Fgf10 +/+ or Fgf10 +/ (n=8) and Fgf10 / mice (n=7) at E19 were cultured for 20 days. All pictures were digitized and the growth length of dental epithelium was estimated at intervals of about 5 days by using NIH image 1.62 (public domain program, US National Institute of Health). Averaged growth rate was plotted using Microsoft Excel 98 (Microsoft Corporation, Redmond, WA). For culture of the cervical loop, the mesenchyme or tissue recombinants the incisors were incubated for 4 hours in 2% collagenase (GibcoBRL) in DMEM at 4 C, and the epithelia were mechanically separated from mesenchyme in PBS. The cervical loops were cut from the epithelial sheets with 26G needle. On tissue recombination experiments the cervical loop was placed in contact with mesenchyme. These explants were cultured for 48 hours on the filters coated matrigel (BD Biosciences, MA). The cervical loops were cultured in the medium supplemented with human recombinant FGF10 (R&D systems Inc, Minneapolis, MN) varying in concentration (0-10 ng/ml). Heparin acrylic beads (Sigma) that released FGF10 (25 ng/µl) were placed in cultures of the cervical loop or the apical end of the mesenchyme. Control beads were incubated in bovine serum albumin (BSA, Sigma). The preparation of the beads has been described in detail (Kettunen et al., 1998). For antibody inoculation, anti-fgf3 antibody (SC-135, Santa Cruz Biotechnology, Santa Cruz, CA), anti-fgf10 antibody (SC- 7375, Santa Cruz Biotechnology, Inc) and control goat IgG (SC-2028, Santa Cruz Biotechnology) were added to the culture medium at a concentration of 50 µg/ml. Cell proliferation assays Cell proliferation was localized by mapping the distribution of S-
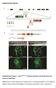
3 FGF10 for stem cells 1535 Fig. 2. Absence of cervical loop in the developing incisor germ of FGF null mice. These pictures show Hematoxylin and Eosin stained sections of lower incisor germs at cap (A,C), early bell (B,D) and late bell (E,G) stages in Fgf10 +/+ embryos (A,B,E,F) and Fgf10 / embryos (B,D,G,H). At cap stage (E14), FGF10 null mice did not show clear morphological abnormalities in incisor development (C). Arrows show the end of labial epithelium. At early bell stage (E16), the epithelial bulge of FGF null mice was smaller than that of wild type (B,D, arrows). At late bell stage (E19), the cervical loop was missing in FGF10 null mice (G,H). F and H show higher magnification of E and G at the region of the apical end. (I-L) Immunostaining of cytokeratin (CK) at late bell stage (E19). At the apical end of wild type, the center region of the stellate reticulum in the cervical loop was strongly CK positive (J). The peripheral region of the stellate reticulum, found in close proximity to basal epithelium, was CK negative (J, smaller asterisk). In mutant incisor, CK-negative stellate reticulum cells were missing around strongly CK-positive cells shown by arrowheads (L). Arrows show the apical end of labial epithelium. The staining of erythrocytes was due to nonspecific binding of antibody (L, larger asterisk). Hematoxylin and Eosin staining of the lower first molar germ in Fgf10 +/+ (M) and Fgf10 / (N) embryos. be, basal epithelium; cl, cervical loop; ie, inner enamel epithelium; oe, outer enamel epithelium; sr, stellate reticulum. Scale bars: 200 µm in A- D,E,G,I,K,M,N; 100 µm in F,H,J,L. phase cells. BrdU (1.5 ml/100g, 5-bromo-2 -deoxyuridine, Roche Diagnostics, Mannheim, Germany) was administered to ether-treated mice, which were sacrificed 2 hours later, and incorporated BrdU was detected immunohistochemically by the BrdU Staining Kit (Oncogene Research Products, Darmstadt, Germany). In situ hybridization For in situ hybridization analysis, digoxigenin-labeled antisense riboprobes for Fgf3 and Fgf10 mrna were synthesized according to the manufacturer s instructions (Roche Diagnostics). Paraffin sections were made as described previously (Kettunen and Thesleff, 1998; Kettunen et al., 1998). The chromogen 4-nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl-phosphate (NBT/BCIP, Roche Diagnostics) was used for color detection. Annexin V staining We used Alexa 488-conjugated Annexin V (Molecular Probes, Eugene, OR), which binds to phosphatidylserine moieties that become exposed on the outer surface of the cell membrane at the onset of apoptosis, to detect apoptotic cells in the explants. The explants were incubated with Hepes buffer (10 mm Hepes, 150 mm NaCl, 5 mm KCl, 1 mm MgCl, 1.8 mm CaCl 2) containing 1 µg/ml Annexin V-Alexa 488 for 20 minutes. After rinsing with Hepes buffer, fluorescence was detected by fluorescent microscopy. RESULTS Absence of Fgf10 causes defect in stem cell compartment in developing mouse incisor A mouse incisor exhibits special developmental systems that allow for regeneration, a capacity not seen in molars. The first morphological sign of incisor development is a thickening of the oral epithelium (Fig. 1, E11), which subsequently buds into the underlying mesenchyme (E13, bud stage). The mesenchymal cells condense around the bud, and during the following cap stage the tooth bud is lain down longitudinally in the mandible (E14, cap stage). The labial epithelium, which produces differentiated ameloblasts, grows longer in length than the lingual epithelium and the cervical loop, which is a region thought to contain a population of self-renewing epithelial stem cells (Harada et al., 1999) formed at the end of the epithelium (E16, early bell stage). Subsequently, unlike in the molar, the incisor germs continue to grow permanently without forming a root (E19.5, late bell stage). Elucidating the molecules that orchestrate specific developmental programs for stem cells is important for determining the key components that regulate stem cells.
4 1536 H. Harada and others Fig. 3. Gene expression patterns of Fgf3 and Fgf10 in the developing lower incisor by in situ hybridization. Both Fgf3 and Fgf10 mrna was co-expressed in the dental papilla at the cap stage (E14) (A,B). At early bell stage (E16), Fgf3 mrna was restricted to the mesenchyme underlying the inner enamel epithelium (D). Fgf3 mrna was not seen around the cervical loop (D, arrowheads) and lingual epithelium (arrow). Cells expressing Fgf10 mrna extended to the mesenchyme neighboring the inner enamel epithelium and covered the cervical loop (C, arrowheads). Scale bar: 200 µm. cl, cervical loop. We studied the role of FGF10 in mesenchymal signaling by analyzing the development of mouse incisors in Fgf10-deficient mice. Morphological defects were not found before E14 (Fig. 2A,C). Tooth germs in Fgf10 +/+, Fgf10 +/ and Fgf10 / mice developed normally until bud, cap and bell stage. However, at E16, the cervical loop of FGF10 null mice was smaller than that of wild type (Fig. 2B,D). At later stages, the cervical loop was missing (Fig. 2E-H). We examined the cervical loop defect in detail by immunostaining with anti-cytokeratin (CK) antibody. The cervical loop consists of a core of stellate reticulum cells surrounded by basal epithelial cells contacting the dental mesenchyme. Within the cervical loop of Fgf10 +/+ or Fgf10 +/ mice, the center of the stellate reticulum was CK positive, whereas the remaining cells were CK negative. Notably, the peripheral stellate reticulum cells (putative stem cells), which are located in close proximity to basal epithelial cells, were CK negative (Fig. 2I,J, a smaller asterisk). However, in FGF10 null mice, although a CK-positive stellate reticulum remained (Fig. 2L, arrowheads), the basal epithelium and CK-negative stellate reticulum cells were missing, verifying the absence of the incisor stem cell population. However, a normal cell-differentiation gradient of enamel-forming ameloblasts and dentin-forming odontoblasts was observed in both incisors and molars of FGF10 null mice (Fig. 2E,G,M,N). To characterize the timing of the morphological inconsistency between the mutant and wild type, we examined the gene expression pattern of Fgf3 and Fgf10 in the developing incisors by in situ hybridization. At E14, Fgf3 and Fig. 4. Development of the apical end of incisors in Fgf10 +/+ and Fgf10 / mice by organ culture and estimation of growing dental epithelium. (A) The dissected incisors from the mandible of Fgf10 +/+ or Fgf10 +/ (n=8) and Fgf10 / mice (n=7) at E19 were cultured for 20 days as described. (B) All pictures were digitized, and the growth length of dental epithelium was estimated using NIH image The graph of average growth rate was created using Microsoft Excel 98. red line, Fgf10 +/+ or Fgf10 +/ ; blue line, Fgf10 /. (C) These explants were cultured for 30 days. Arrows show disruption of the dental epithelium in the mutant. Arrowheads indicate apical end of the dental epithelium in the explants. Scale bars: 200 µm. *P<0.05. Fgf10 mrna were co-expressed in the dental papilla (Fig. 3A,B) suggesting a functional redundancy between the two FGFs in early incisor morphogenesis. After E16, however, cells expressing Fgf10 mrna spread from the zone neighboring the inner enamel epithelium to the apical end and surrounded the cervical loop, and this expression pattern appeared to correlate with the growth of labial epithelium (Fig. 3C). By contrast, cells expressing Fgf3 mrna remained only in dental papilla neighboring the inner enamel epithelium (Fig. 3D). Fgf7 mrna was weakly expressed in the dental follicle at the apical end and not seen in dental papilla (Kettunen et al., 2000). Fgfr1b and Fgfr2b are expressed throughout the dental epithelium (Kettunen et al., 1998; Harada et al., 1999). These characteristic patterns do not change after birth (Harada et al., 1999). Thus, the timing of cervical loop formation correlates well with the divergence of Fgf10 and Fgf3 gene expression patterns.
5 FGF10 for stem cells 1537 Dental epithelium lacking a cervical loop exhibits decreased growth rate To determine the role of existing stem cells, we examined the growth of epithelium that lacked a stem cell compartment. As Fgf10-deficient mice die soon after birth, owing to an absence of lung development (Sekine et al., 1999), we cultured incisor germs dissected from the mandible of E19 mice. The dental epithelium of both mutant and wild-type mice seemed to grow steadily for 20 days (Fig. 4A). However, as shown in Fig. 4B, the epithelial growth rate in mutant mice began to decrease and reached a maximum of approximately 400 µm (Fig. 4B). By contrast, the epithelium of wild-type mice continued to grow. When these explants were cultured beyond 20 days, the explants of mutant type showed disruption of the dental epithelium. Consequently, the length could not be estimated precisely. To investigate difference of epithelial growth pattern between wild type and mutant or between incisor and molar, BrdU labeling analysis was used to detect the dividing cells of dental epithelium in E18 mice (Fig. 5A) and the percentage of BrdU-positive cells was quantified (Fig. 5B). Labeled cells were detected in the dental epithelium of wild type mice in the cervical loop and extended into the inner enamel epithelial zone (Fig. 5A, parts a and b). The percentage of BrdU-positive cells was 24% on average in the cervical loop and 68% in the inner enamel epithelium (Fig. 5B). In the cervical loop the positive cells were located in peripheral stellate reticulum showing putative stem cell population (Harada et al., 1999). The characteristic scattering pattern of positive cells observed in the cervical loop was clearly distinct from the pattern of dense labeled cells in the inner enamel epithelium. In the mutant incisor, the percentage was high (63%) in the apical end of dental epithelium and was nearly identical (68%) to that in inner enamel epithelium of wild type. But, the labeling pattern seen in the cervical loop of the wild type was missing in the epithelium of mutant type (Fig. 5A, parts c and d). The inner enamel epithelium of molar germs showed high percentages (88%, 67.5%) in both wild type and mutant (Fig. 5B), and their patterns of labeled cells were dense (Fig. 5A, parts e and f). These data suggest that a stem cell population does not exist in the dental epithelium of FGF10 null mice and molar germs. As the growth of epithelium that lacks stem cells depends only on proliferation of transit-amplifying cells, which undergo terminal differentiation after several rounds of cell division, the growth of mutant incisors was expected to be limited (Fig. 4B). Furthermore, cell death in the dental epithelium of the mutant involved limit of cell division of transit-amplifying cells (Fig. 4C). Thus, continuous growth would depend on stem cell division induced by mesenchymal FGF10. Fig. 5. Distribution of dividing cells in apical end of dental epithelium. (A) Representative longitudinal section of lower incisors and first molars in Fgf10 +/+ and Fgf10 / embryos at E18 as shown by BrdU labeling. In lower incisor of Fgf10 +/+ embryos (a,b), BrdU-incorporating cells are sparsely distributed in the cervical loop (b) and densely distributed in inner enamel epithelium. In lower incisor of Fgf10 / embryos (c,d), the labeling cells were seen densely at the edge of dental epithelium. The sparse pattern seen in the cervical loop of the Fgf10 +/+ embryo was not observed (arrows in c,d). b and d show higher magnification of a and c at the apical end of the dental epithelium. Black dots outline the dental epithelium. In molar germs of Fgf10 +/+ and Fgf10 / embryos (e,f), BrdU is densely incorporated into the inner dental epithelium. Arrows show the apical end of dental epithelium. (B) Quantification of percentage of BrdU-positive cells at the apical region of dental epithelium in incisors and molars of wild type and mutant. The percentage of BrdU-posisitve cells in the cervical loop in wild type is significantly lower than the percentage in the inner enamel epithelium. Each bar presents data from three samples. Error bars represent standard deviation. ie, inner enamel epithelium; dp, dental papilla; I, incisor; M, molar. Scale bars: 200 µm in a,c,e,f; 100 µm in b,d.
6 1538 H. Harada and others Fig. 6. Effect of anti-fgf10 neutralizing antibody on cultured incisor explants. The explants were cultured for 24 hours in the presence of anti-fgf10 antibody (A-C), anti-fgf3 antibody (D-F), anti-fgf10 antibody and human recombinant FGF10 (G- I), or normal goat IgG (J-L). Apoptotic cells were detected by Annexin V staining. Anti-FGF10 antibody disrupted the cervical loop epithelium (A,B) and caused apoptosis in the cervical loop cells (C). In the presence of anti-fgf3 antibody (D,E) or anti-fgf10 antibody and human recombinant FGF10 (G,H), the cervical loop was recognized. Anti-FGF3 antibody caused partly apoptosis in the inner enamel epithelium and the neighboring mesenchyme, but apoptotic cells were rarely seen in the cervical loop and the surrounding mesenchyme. In the presence of control IgG, few apoptotic cells were detected (K,L). Asterisks show the cervical loop. Scale bar: 200 µm. FGF10 functional disorder causes apoptosis of cervical loop cells To clarify the role of FGF10 in the dental epithelium of incisors, we designed an in vitro loss-of-function experiment using anti-fgf10 neutralizing antibody. When the incisor explants were cultured for 48 hours in the presence of anti- FGF10 antibody, destruction of the cervical loop was observed (Fig. 6A,B). By contrast, in the culture with anti-fgf3 antibody, anti-fgf10 antibody and human recombinant FGF10, control goat IgG at an equal concentration to anti- FGF10 antibody, the cervical loop epithelium maintained a morphological feature (Fig. 6E,H,K). To identify the extent of cell death in the explants, we visualized apoptotic cells using whole-mount Annexin V staining. In the culture with anti-fgf10 antibody, most epithelial cells in the cervical loop reacted with Annexin V (Fig. 6C). Interestingly, apoptotic cells were observed in the neighboring mesenchyme, suggesting that the survival of mesenchymal cells was associated with that of epithelial cells. In the presence of anti-fgf3 antibody, apoptosis occurred in the inner enamel epithelial and the neighboring mesenchymal cells (Fig. 6F). Notably, few apoptotic cells were seen in the cervical loop or the surrounding mesenchyme. Furthermore, human recombinant FGF10 rescued the explants from the destruction of the cervical loop, which was caused by the FGF10 neutralizing antibody (Fig. 6G-I). In the presence of control goat IgG, apoptotic cells were rarely seen (Fig. 6L). We also determined whether FGF10 protein could prevent cell death in the cervical loop epithelium. The isolated cervical loops were cultured in the medium supplemented with recombinant human FGF protein varying concentration (0-10 ng/ml). The cervical loops were disrupted within 48 hours (Fig. 7B,F) in a concentration of 0.1 ng/ml or less and Annexin V bound to many of the cells (Fig. 7B,F ). By contrast, in the culture supplemented with FGF10 (1 or 10 ng/ml), the epithelium maintained its tight appearance (Fig. 7D,G) and few cells bound Annexin V (Fig. 7D,G ). FGF10-releasing beads also rescued the destruction of the cervical loop epithelium (Fig. 7H ). Control beads had no effect on maintaining the morphology or escaping apoptosis (Fig. 7I ). The neutralizing anti-fgf10 antibody (50 ng/ml) disturbed the effects of FGF10 protein (1 ng/ml) as a survival factor. These results indicated that FGF10 plays a role in the maintenance of the stem cell compartment in the developing incisors. The apoptosis of some mesenchymal cells in the presence of anti-fgf10 antibody suggested that it might be due to absence of epithelial signaling (Fig. 6C). To examine whether absence of the cervical loop cause apoptosis of the neighboring mesenchyme, we had the tissue recombination experiments and the culture of mesenchyme in absence of the dental epithelium. When the isolated mesenchyme and cervical loop were placed in contact on the filter and cultured for 2 days, apoptotic cells were rarely seen in the mesenchyme around the cervical loop (Fig. 7L, asterisk). However, the mesenchymal cells distant from the cervical loop underwent apoptosis. A few apoptotic cells were also seen in the cervical loop. When the isolated mesenchyme was cultured, a large number of apoptotic cells were seen (Fig. 7N,N ). FGF10-releasing beads were not effective in preventing the isolated mesenchyme from undergoing apoptosis (Fig. 7O,O ).
7 FGF10 for stem cells 1539 DISCUSSION Mouse incisors erupt throughout the animal s lifetime through the renewal of dental epithelium produced from the cervical loop located in the tooth apex. Slowly and asymmetrically dividing Notch1-expressing stellate reticulum cells, located near the lunatic fringe-expressing basal epithelial cells, have previously been identified as putative stem cells. As molecular signaling during development has been well studied ( the incisor presents an excellent Fig. 7. FGF10 rescued the isolated cervical loop epithelium from apoptosis. The cervical loop epithelium was separated from the mesenchyme (A) and cultured for 48 hours (B,D,F-J) in the medium supplemented with recombinant human FGF10 at a concentration of 0.1 ng/ml (F,F ), 1.0 ng/ml (D,D,E,E ), 10 ng/ml (G,G ), with bovine serum albumin (B,B,C,C ), with FGF10-releasing beads (H,H ), with bovine serum albumin (BSA) -soaked beads (I,I ), or with FGF10 (1 ng/ml) and FGF10 neutralizing antibody (50 µg/ml) (J,J ). Apoptotic cells were detected using Annexin V staining (B -J,L,N,O ). In the presence of FGF10 (1 ng/ml and 10 ng/ml), the outline of the isolated epithelium was clearly maintained (D,G) and apoptotic cells bound to Annexin V were sparse (D,E,G ). Higher magnification (E) shows that the cell morphologies of the basal epithelium and the peripheral stellate reticulum (putative stem cells) in the explant were clearly maintained. The explants cultured with BSA, BSA beads or FGF10 (1 ng/ml) and FGF10 neutralizing antibody (50 µg/ml) showed disrupted epithelium (B,C,I,J) and many apoptotic cells (B,C,I,J ). The shape of cells in the disrupted cervical loop epithelium was not clearly recognized (C) and a large number of cells were bound to Annexin V (C ). The explant in 0.1 ng/ml FGF10 showed the outline clearly (F), but many of these cells underwent apoptosis (F ). (K,P) The isolated mesenchyme (M) was placed in contact with the isolated cervical loop epithelium (A) and cultured for 2 days. In the mesenchyme adherent to the epithelium, apoptosis was rarely seen (L,L ). Dots outline the border between the epithelium and the mesenchyme (L,L ). In the isolated mesenchyme, several apoptotic cells were seen in the presence (O,O ) and absence (N,N ) of FGF10 beads. Scale bars: 200 µm in A,B,D,F-O; 40 µm in C,E.
8 1540 H. Harada and others model for the study of stem cell niche formation, stem cell division and fate decision mechanisms. The cervical loop, including putative stem cells, is not formed in developing incisors of FGF10-null mice. The present study indicates that mutant incisors lacking the cervical loop lose the ability to grow continuously. In situ hybridization of developing incisor germs has revealed differential expression of Fgf10 and Fgf3. After E16 and in conjunction with the growth of dental epithelium, cells expressing Fgf10 mrna spread out from a region underlying the inner enamel epithelium to the apical end of the tooth germ and overlaid the cervical loop. By contrast, cells expressing Fgf3 remained restricted to the regions adjacent to the inner enamel epithelium. Thus, FGF10 rather than FGF3 is responsible for the formation of the cervical loop. Furthermore, our loss-offunction experiments using neutralizing antibodies verify that FGF10 plays a critical role in formation and maintenance of the stem cell population during organogenesis of mouse incisors. Formation of cervical loop is crucial for regeneration of incisors In contrast to molars, mouse incisors exhibit specific developmental systems that allow for regeneration. The putative stem cell compartment of incisors, the cervical loop, is not found in molar germs. In molars, the dental epithelium (Hertwig s epithelial root sheath) migrates downwards to the root base. At this point, the dental epithelium loses the ability to grow (Kaneko et al., 1999). By contrast, the continuously growing molars of rabbits show small epithelial bulges containing cells that behave like stem cells at the apical end of the germs (Starkey, 1963). Our experiments on incisor explants showed that epithelium-lacking stem cells exhibited limited growth. Taken together, these results suggest that formation of the cervical loop during incisor morphogenesis is crucial for developing the capacity to regenerate. FGF10 is involved in the formation of incisor cervical loop in a non-redundant manner with FGF3 Because the expression pattern of Fgf10 mrna is identical to that of Fgf3 during molar development (Kettunen et al., 2000), FGF10 null mice do not exhibit striking molar abnormalities. Likewise, Fgf3-deficient mice do not exhibit gross morphological changes in their teeth (Mansour et al., 1993). These data support the results from our loss-of-function experiments in which antibodies that neutralize FGF3 did not affect incisor explants perniciously. Considering that Fgfr1b and Fgfr2b are expressed throughout dental epithelium (Kettunen et al., 1998; Harada et al., 1999) ( these results suggest that the FGFs function somewhat redundantly. As expected from the phenotype of Fgfr2(IIIb)-deficient mice (Moerlooze et al., 2000), in Fgf3 and Fgf10-double knockout mice, tooth germs would fail to progress beyond the bud stage. In the present study, in situ hybridization in the incisor germs revealed that after E16, the Fgf10-expression zone differed from that of Fgf3. Expression of Fgf3 mrna was restricted to the mesenchyme underlying the inner enamel epithelium, but cells expressing FGF10 extended to the zone covering the cervical loop. Fgf7 mrna was weakly expressed in the dental follicle at outside of the apical end, but not in the dental papilla (Kettunen et al., 2000), suggesting that FGF7 is associated with growth of outer enamel epithelium. Consequently, only FGF10 appears to function in maintaining cervical loop. Kettunen et al. (Kettunen et al., 2000) have found that Fgf10, Fgf3 and Fgf7 mrna are not expressed in the mesenchyme after the later bell stage of molar development. These results suggest that continuous expression of Fgf10 in the mesenchyme of the apical end is crucial for the formation of stem cells during incisor development. Fgf10 may be an essential mesenchymal factor for reciprocal interaction between epithelial and mesenchymal stem cells in developing incisor germs FGF signaling plays an important role in the mediation of epithelial-mesenchymal interactions in a variety of organs. Our previous studies of Fgf10 expression patterns and in vitro application of FGF10-soaked beads suggested that FGF10 acts as a regulatory signal from the mesenchyme to the epithelium during tooth development (Harada et al., 1999; Kettunen et al., 2000). In the present study, when FGF10 neutralizing antibodies caused apoptosis in the cervical loop, some neighboring mesenchymal cells were also apoptotic (Fig. 6C). Furthermore, many of cells in the isolated mesenchyme from the epithelium were apoptotic regardless of the presence of FGF10 protein (Fig. 7N,N,O,O ). Thus, the mesenchymal cell death appears to be attributable to the absence of signaling from the cervical loop. The development of tooth germs is governed by reciprocal epithelial-mesenchymal interaction and the molecular mechanism is very similar to that seen in limbs and hair (Tickle, 1995). The growth of limbs and genital tubercles is governed by the reciprocal interaction between FGF8 and FGF10 (Ohuchi et al., 1997; Xu et al., 1998; Haraguchi et al., 2000). Although Fgf8 mrna was observed in the dental epithelium from its thickening to early bud stage (E12), it was not seen in the developing incisor at a later stage (Kettunen and Thesleff, 1998). Kettunen et al. (Kettunen et al., 2000) have shown that the expression of both Fgf3 and Fgf10 in the mesenchyme depends on dental epithelium, and that FGF4 and FGF8 induce Fgf3 expression in the mesenchyme. In addition, Bei and Maas (Bei and Maas, 1998) have also shown that FGFs (FGF1, FGF2 and FGF8), but not bone morphogenetic protein 4 (BMP4), regulate Fgf3 expression in the dental mesenchyme. The supernatant from an odontogenic epithelial stem cell line originating from the incisor cervical loop stimulates cell proliferation and maintains expression of FGF10 in primary culture of odontogenic mesenchyme (S. Kawano, personal communication). However, FGFs, sonic hedgehog, BMP2, transforming growth factor β1 and Wnt6 expressed in the early dental epithelium did not stimulate Fgf10 expression in the dental mesenchyme. Although the epithelial signals regulating Fgf10 expression in teeth are still unknown, we speculate that a regulatory cascade exists between putative cervical loop molecules and FGF10 expressed in the mesenchyme. Adult stem cells in the mouse incisor seem to maintain an undifferentiated early embryonic stage by epithelial-mesenchymal interaction. Does FGF10 control stem cell survival? The persistence of stem cell populations through adulthood probably depends on the survival of quiescent cells, as well as
9 FGF10 for stem cells 1541 on the ability of cycling cells to self-renew. Quiescent stem cells have been identified in the liver, brain, bone marrow (reviewed by Morrison et al., 1997) and hair bulge (Taylor et al., 2000). However, it is not clear whether these apparently quiescent cells are in G 0 or whether they are moving very slowly through G 1. We have previously identified slow-cycling cells of the incisor cervical loop as putative stem cells in the dental epithelium (Harada et al., 1999). In the present study, BrdU-labeled cells in the cervical loop showed a sparse distribution pattern. Cells escaping from the cell cycle appear to behave like quiescent cells. Although previously we have shown that FGF10 stimulates division of stem cells, we do not yet know whether FGF10 plays a role in the survival of quiescent cells. Increased apoptotic cells are found in hair follicles of FGF10 null mice (Ohuchi et al., 2000). These data are consistent with the present study, which demonstrates that the FGF10- neutralizing antibodies cause apoptosis in the incisor cervical loop epithelium. Furthermore, recombinant human FGF10 could rescue the isolated cervical loop epithelium from apoptosis. The evidence indicates that FGF10 plays a role in stem cell survival and stimulates cell-proliferation; however, other, more complex regulatory mechanisms in the stem cell compartment remain to be studied. We thank Prof. Irma Thesleff (University of Helsinki, Finland) for suggestions and critical reading of the manuscripts. This work was partly funded by grants from the Ministry of Education, Science, Sports and Culture (No , No , No ), the Kato Memorial Bioscience Foundation in Japan and Scandinavia Japan Sasakawa Foundation (No.01-18). REFERENCES Bei, M and Maas, R. (1998). FGFs and BMP4 induce both Msx-1 independent and Msx1-dependent signaling pathways in early tooth development. Development 125, DasGupta, R. and Fuchs, E. (1999). Multiple roles for activated LEF/TCF transcription complexes during hair follicle development and differentiation. Development 126, Fuchs, E. and Segre, J. A. (2000). Stem cells: a new lease on life. Cell 100, Harada, H., Kettunen, P., Jung, H. S., Mustonen, T., Wang, Y. A. and Thesleff, I. (1999). Localization of putative stem cells in dental epithelium and their association with Notch and FGF signaling. J. Cell Biol. 147, Haraguchi, R., Suzuki, K., Murakami, R., Sakai, M., Kamikawa, M., Kengaku, M., Sekine, K., Kawano, H., Kato, S., Ueno, N. and Yamada, G. (2000). Molecular analysis of external genitalia formation: the role of fibroblast growth factor (Fgf) genes during genital tubercle formation. Development 127, Hogan, B. L. M. (1999). Morphogenesis. Cell 96, Kaneko, H., Hashimoto, S., Enokiya, Y., Ogiuchi, H. and Shimono, M. (1999). Cell proliferation and death of Hertwig s epithelial root sheath in the rat. Cell Tissue Res. 298, Kettunen, P., Karavanova, I. and Thesleff, I. (1998). Responsiveness of developing dental tissues to fibroblast growth factors: expression of splicing alternatives of FGFR1, -2, -3, and of FGFR4; and stimulation of cell proliferation by FGF-2, -4, -8, and -9. Dev. Genet. 22, Kettunen, P., Laurikkala, J., Itaranta, P., Vainio, S., Itoh, N. and Thesleff, I. (2000). Associations of FGF-3 and FGF-10 with signaling networks regulating tooth morphogenesis. Dev. Dyn. 219, Kettunen, P. and Thesleff, I. (1998). Expression and function of FGFs-4, -8, and -9 suggest functional redundancy and repetitive use as epithelial signals during tooth morphogenesis. Dev. Dyn. 211, Koller, E. L. and Baird, G. R. (1969). The influence of the dental papilla on the development of tooth shape in embryonic mouse tooth germs. J. Embryol. Exp. Morphol. 21, Korinek, V., Barker, N., Moerer, P., van Donselaar, E., Huls, G., Peters, P. and Clevers, H. (1998). Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat. Genet. 19, Mansour, S. L., Goddard, J. M. and Capecchi, M. R. (1993). Mice homozygous for a targeted disruption of the proto-oncogene int-2 have development defects in the tail and inner ear. Development 117, Millar, S. E., Willert, K., Salinas, P. C., Roelink, H., Nusse, R., Sussman, D. J. and Barsh, G. S. (1999). Wnt signaling in the control of hair growth and structure. Dev. Biol. 207, Morrison, S. J., Shah, N. M. and Anderson, D. A. (1997). Regulatory mechanisms in stem cell biology. Cell 88, Moerlooze, L. D., Spencer-Dene, B., Revest, J.-M., Hajihosseini, M., Rosewell, I. and Dickson, C. (2000). An important role for the lllb isoform of fibroblast growth factor receptor 2 (FGFR2) in mesenchymal-epithelial signaling during mouse organogenesis. Development 127, Ohuchi, H., Hori, Y., Yamasaki, M., Harada, H., Sekine, K., Kato, S. and Itoh, N. (2000). FGF10 acts as a major ligand for FGF receptor 2IIIb in mouse multi-organ development. Biochem. Biophys. Res. Commun. 277, Ohuchi, H., Nakagawa, T., Yamamoto, A., Araga, A., Ohata, T., Ishimaru, Y., Yoshioka, H., Kuwana, T., Nohno, T., Yamasaki, M., Itoh, N. and Noji, S. (1997). The mesenchymal factor, FGF10, initiates and maintains the outgrowth of the chick limb bud through interaction with FGF8, an apical ectodermal factor. Development 124, Peters, H. and Balling, R. (1999). Teeth. Where and how to make them. Trends Genet. 15, Rochat, A., Kobayashi, K. and Barrandon, Y. (1994). Location of stem cells of human hair follicles by clonal analysis. Cell 76, Sekine, K., Ohuchi, H., Fujiwara, M., Yamasaki, M., Yoshizawa, T., Sato, T., Yagishita, N., Matsui, D., Koga, Y., Itoh, N. and Kato, S. (1999). Fgf10 is essential for limb and lung formation. Nat. Genet. 21, Starkey, W. E. (1963). The migration and renewal of tritium labeled cells in the developing enamel organ of rabbits. Br. Dent. J. 115, Taylor, G., Lehrer, M. S., Jensen, P. J., Sun, T. T. and Lavker, R. M. (2000). Involvement of follicular stem cells in forming not only the follicle but also the epidermis. Cell 102, Thesleff, I. and Sharpe, P. (1997). Signaling networks regulating dental development. Mech. Dev. 67, Tickle, C. (1995) Vertebrate limb development. Curr. Opin. Genet. Dev. 5, Xu, X., Weinstein, M., Li, C., Naski, M., Cohen, R. I., Ornitz, D. M., Leder, P. and Deng, C. (1998). Fibroblast-growth-factor-receptor-2 (FGFR2)- mediated reciprocal regulation loop between FGF8 and FGF10 is essential for limb induction. Development 125,
The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling
 The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
The Open Biotechnology Journal, 2008, 2, 111-115 111 The Epithelial-Mesenchymal Interaction Plays a Role in the Maintenance of the Stem Cell Niche of Mouse Incisors via Fgf10 and Fgf9 Signaling Tamaki
The eternal tooth germ is formed at the apical end of continuously growing teeth*
 The eternal tooth germ is formed at the apical end of continuously growing teeth* Hayato Ohshima 1, Naohiro Nakasone 1, 2, Emi Hashimoto 1, Hideo Sakai 1, Kuniko Nakakura-Ohshima 3 and Hidemitsu Harada
The eternal tooth germ is formed at the apical end of continuously growing teeth* Hayato Ohshima 1, Naohiro Nakasone 1, 2, Emi Hashimoto 1, Hideo Sakai 1, Kuniko Nakakura-Ohshima 3 and Hidemitsu Harada
BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor
 BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor Kateryna Kyrylkova 1, Sergiy Kyryachenko 1, Brian Biehs 2 *, Ophir Klein 2, Chrissa Kioussi 1 *, Mark
BCL11B Regulates Epithelial Proliferation and Asymmetric Development of the Mouse Mandibular Incisor Kateryna Kyrylkova 1, Sergiy Kyryachenko 1, Brian Biehs 2 *, Ophir Klein 2, Chrissa Kioussi 1 *, Mark
Development of teeth. 5.DM - Pedo
 Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS
 ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and
 Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
06 Tooth Development and Eruption
 + 06 Tooth Development and Eruption Tooth development Root development PDL and alveolar bone development Primary tooth eruption and shedding Permanent tooth eruption Q. Where and how tooth starts to form?
+ 06 Tooth Development and Eruption Tooth development Root development PDL and alveolar bone development Primary tooth eruption and shedding Permanent tooth eruption Q. Where and how tooth starts to form?
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Ahtiainen et al., http :// /cgi /content /full /jcb /DC1
 Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
Supplemental material JCB Ahtiainen et al., http ://www.jcb.org /cgi /content /full /jcb.201512074 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Distinct distribution of different cell cycle phases in the
CAP STAGE. Ans 1 The following are the stages of tooth development :
 Ans 1 The following are the stages of tooth development : 1. Bud stage 2. Cap stage 3. Bell stage 4. Advanced bell stage 5. Formation of Hertwig s epithelial root sheath BUD STAGE 1. Around the eighth
Ans 1 The following are the stages of tooth development : 1. Bud stage 2. Cap stage 3. Bell stage 4. Advanced bell stage 5. Formation of Hertwig s epithelial root sheath BUD STAGE 1. Around the eighth
Supporting Information
 Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
Supporting Information Plikus et al. 10.1073/pnas.1215935110 SI Text Movies S1, S2, S3, and S4 are time-lapse recordings from individually cultured Period2 Luc vibrissa follicles show that circadian cycles
The Beauty of the Skin
 The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
Supplementary Information
 Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
Supplementary Information Figure S1: Follicular melanocytes in the wound peripheral area migrate to the epidermis in response to wounding stimuli. Dorsal skin of Trp2-LacZ mice stained with X-gal and analyzed
An Integrated Gene Regulatory Network Controls Stem Cell Proliferation in Teeth
 An Integrated Gene Regulatory Network Controls Stem Cell Proliferation in Teeth The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
An Integrated Gene Regulatory Network Controls Stem Cell Proliferation in Teeth The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
Tooth eruption and movement
 Tooth eruption and movement Dr. Krisztián Nagy Diphydont dentition Deciduous dentition primary dentition Diphydont dentition Permanent dentition secondary dentition Mixed Dentition: Presence of both dentitions
Tooth eruption and movement Dr. Krisztián Nagy Diphydont dentition Deciduous dentition primary dentition Diphydont dentition Permanent dentition secondary dentition Mixed Dentition: Presence of both dentitions
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is
 Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Vertebrate Limb Patterning
 Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Tooth development in the 'crooked' mouse
 /. Embryo!, exp. Morph. Vol. 41, pp. 279-287, 1977 279 Printed in Great Britain Company of Biologists Limited 1977 Tooth development in the 'crooked' mouse By J. A. SOFAER 1 From the University of Edinburgh,
/. Embryo!, exp. Morph. Vol. 41, pp. 279-287, 1977 279 Printed in Great Britain Company of Biologists Limited 1977 Tooth development in the 'crooked' mouse By J. A. SOFAER 1 From the University of Edinburgh,
INTRODUCTION. Developmental Biology 229, (2001) doi: /dbio , available online at
 Developmental Biology 229, 443 455 (2001) doi:10.1006/dbio.2000.9955, available online at http://www.idealibrary.com on TNF Signaling via the Ligand Receptor Pair Ectodysplasin and Edar Controls the Function
Developmental Biology 229, 443 455 (2001) doi:10.1006/dbio.2000.9955, available online at http://www.idealibrary.com on TNF Signaling via the Ligand Receptor Pair Ectodysplasin and Edar Controls the Function
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate
 Research article Related Commentary, page 1676 Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate Ritva Rice, 1,2 Bradley Spencer-Dene, 3 Elaine C. Connor, 1
Research article Related Commentary, page 1676 Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate Ritva Rice, 1,2 Bradley Spencer-Dene, 3 Elaine C. Connor, 1
To the root of the stem cell problem. The evolutionary importance of the epithelial stem cell niche during tooth development
 To the root of the stem cell problem The evolutionary importance of the epithelial stem cell niche during tooth development Mark Tummers Developmental Biology Programme Institute of Biotechnology University
To the root of the stem cell problem The evolutionary importance of the epithelial stem cell niche during tooth development Mark Tummers Developmental Biology Programme Institute of Biotechnology University
AMELOGENESIS. Prof. Shaleen Chandra
 AMELOGENESIS Epithelial Enamel Organ Outer Enamel Epithelium Stellate Reticulum Stratum Intermedium Inner Enamel Epithelium Cervical Loop Life Cycle of Ameloblasts Morphogenic stage Organizing Stage Formative
AMELOGENESIS Epithelial Enamel Organ Outer Enamel Epithelium Stellate Reticulum Stratum Intermedium Inner Enamel Epithelium Cervical Loop Life Cycle of Ameloblasts Morphogenic stage Organizing Stage Formative
Changes in the distribution of tenascin during tooth development
 Development 101, 289-2% (1987) Printed in Great Britain The Company of Biologists Limited 1987 289 Changes in the distribution of tenascin during tooth development IRMA THESLEFF 1, ELEANOR MACKIE 2, SEPPO
Development 101, 289-2% (1987) Printed in Great Britain The Company of Biologists Limited 1987 289 Changes in the distribution of tenascin during tooth development IRMA THESLEFF 1, ELEANOR MACKIE 2, SEPPO
Chapter 2 Tooth Development
 Chapter 2 Tooth Development Experimental research on tooth development or odontogenesis is based very largely on the teeth of murine rodents (Butler 1967 ). Pioneering work by Shirley Glasstone on rat
Chapter 2 Tooth Development Experimental research on tooth development or odontogenesis is based very largely on the teeth of murine rodents (Butler 1967 ). Pioneering work by Shirley Glasstone on rat
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
An FGF signaling loop sustains the generation of differentiated progeny from stem cells in mouse incisors
 377 Development 135, 377-385 (2008) doi:10.1242/dev.015081 An FGF signaling loop sustains the generation of differentiated progeny from stem cells in mouse incisors Ophir D. Klein 1,2,3, David B. Lyons
377 Development 135, 377-385 (2008) doi:10.1242/dev.015081 An FGF signaling loop sustains the generation of differentiated progeny from stem cells in mouse incisors Ophir D. Klein 1,2,3, David B. Lyons
Using Dental Stem Cells to Regenerate Tooth Tissue and Whole Tooth Replacement Peretz Rapoport
 and Whole Tooth Replacement Peretz Rapoport Peretz Rapoport graduated January 2018 with a BS in Biology and is starting Touro School of Dental Medicine in fall 2018 Abstract Irreversible dental problems
and Whole Tooth Replacement Peretz Rapoport Peretz Rapoport graduated January 2018 with a BS in Biology and is starting Touro School of Dental Medicine in fall 2018 Abstract Irreversible dental problems
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
2.79J/3.96J/BE.441/HST522J DENTAL TISSUE REPLACEMENT AND REGENERATION
 Massachusetts Institute of Technology Harvard Medical School Brigham and Women s/massachusetts General Hosp. VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J DENTAL TISSUE REPLACEMENT AND REGENERATION
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s/massachusetts General Hosp. VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J DENTAL TISSUE REPLACEMENT AND REGENERATION
ANAT3231: lectures overview
 ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Alginate, tooth-shaped, for constructs, encapsulated pulp cells in, 589 590 Antibiotic paste, triple, change in root length and width
Index Note: Page numbers of article titles are in boldface type. A Alginate, tooth-shaped, for constructs, encapsulated pulp cells in, 589 590 Antibiotic paste, triple, change in root length and width
Msx1 controls inductive signaling in mammalian tooth morphogenesis
 Development 122, 3035-3044 (1996) Printed in Great Britain The Company of Biologists Limited 1996 DEV9494 3035 Msx1 controls inductive signaling in mammalian tooth morphogenesis YiPing Chen, Marianna Bei,
Development 122, 3035-3044 (1996) Printed in Great Britain The Company of Biologists Limited 1996 DEV9494 3035 Msx1 controls inductive signaling in mammalian tooth morphogenesis YiPing Chen, Marianna Bei,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2697 Figure S1 Cytokeratin 5 is a specific marker for basal and intermediate cells in all mouse prostate lobes. (a) Immunofluorescence staining showing co-localization of YFP with p63 in
DOI: 10.1038/ncb2697 Figure S1 Cytokeratin 5 is a specific marker for basal and intermediate cells in all mouse prostate lobes. (a) Immunofluorescence staining showing co-localization of YFP with p63 in
Dentin Formation(Dentinogenesis)
 Lecture four Dr. Wajnaa Oral Histology Dentin Formation(Dentinogenesis) Dentinogenesis begins at the cusp tips after the odontoblasts have differentiated and begin collagen production. Dentinogenesis growth
Lecture four Dr. Wajnaa Oral Histology Dentin Formation(Dentinogenesis) Dentinogenesis begins at the cusp tips after the odontoblasts have differentiated and begin collagen production. Dentinogenesis growth
Lung hypoplasia and neonatal death in Fgf9-null mice identify this gene as an essential regulator of lung mesenchyme
 Development 128, 2095-2106 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV4537 2095 Lung hypoplasia and neonatal death in Fgf9-null mice identify this gene as an essential regulator
Development 128, 2095-2106 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV4537 2095 Lung hypoplasia and neonatal death in Fgf9-null mice identify this gene as an essential regulator
Differential expression patterns of the tight junction-associated proteins occludin and claudins in secretory and mature ameloblasts in mouse incisor
 Med Mol Morphol (2010) 43:102 106 The Japanese Society for Clinical Molecular Morphology 2010 DOI 10.1007/s00795-009-0482-7 ORIGINAL PAPER Masaki Hata Tadafumi Kawamoto Mariko Kawai Toshio Yamamoto Differential
Med Mol Morphol (2010) 43:102 106 The Japanese Society for Clinical Molecular Morphology 2010 DOI 10.1007/s00795-009-0482-7 ORIGINAL PAPER Masaki Hata Tadafumi Kawamoto Mariko Kawai Toshio Yamamoto Differential
evolution and development of primate teeth
 evolution and development of primate teeth diversity of mammalian teeth upper left molars buccal mesial distal lingual Jernvall & Salazar-Ciudad 07 trends in dental evolution many similar single-cusped
evolution and development of primate teeth diversity of mammalian teeth upper left molars buccal mesial distal lingual Jernvall & Salazar-Ciudad 07 trends in dental evolution many similar single-cusped
ANAT3231: lectures overview
 ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
ANAT3231: lectures overview Stem Cell Biology Stem Cell Technology Resources: http://php.med.unsw.edu.au/cell biology/ Essential Cell Biology 3 rd edition Alberts Dr Annemiek Beverdam School of Medical
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Branching morphogenesis of the lung: new molecular insights into an old problem
 86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location
 Original paper Dental Materials Journal 21 (1): 32-43, 2002 Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location Toshiko INOUE, Hidekazu TAKAHASHI and Fumio
Original paper Dental Materials Journal 21 (1): 32-43, 2002 Anisotropy of Tensile Strengths of Bovine Dentin Regarding Dentinal Tubule Orientation and Location Toshiko INOUE, Hidekazu TAKAHASHI and Fumio
Lec. 11 & 12 Dr. Ali H. Murad Dental pulp 1- Coronal pulp
 Lec. 11 & 12 Dr. Ali H. Murad Dental pulp Is the soft connective tissue located in the central portion of each tooth. All pulps have similar morphologic characteristic, such as a soft, gelatinous consistency
Lec. 11 & 12 Dr. Ali H. Murad Dental pulp Is the soft connective tissue located in the central portion of each tooth. All pulps have similar morphologic characteristic, such as a soft, gelatinous consistency
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse
 Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
Temporal Analysis of Ectopic Enamel Production in Incisors From Sprouty Mutant Mice
 JOURNAL OF EXPERIMENTAL ZOOLOGY (MOL DEV EVOL) 312B (2009) Temporal Analysis of Ectopic Enamel Production in Incisors From Sprouty Mutant Mice TOMAS BORAN 1,2, RENATA PETERKOVA 1, HERVE LESOT 3,4,5, DAVID
JOURNAL OF EXPERIMENTAL ZOOLOGY (MOL DEV EVOL) 312B (2009) Temporal Analysis of Ectopic Enamel Production in Incisors From Sprouty Mutant Mice TOMAS BORAN 1,2, RENATA PETERKOVA 1, HERVE LESOT 3,4,5, DAVID
Sonic hedgehog regulates growth and morphogenesis of the tooth
 Development 127, 4775-4785 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3251 4775 Sonic hedgehog regulates growth and morphogenesis of the tooth Hélène R. Dassule 1, Paula
Development 127, 4775-4785 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3251 4775 Sonic hedgehog regulates growth and morphogenesis of the tooth Hélène R. Dassule 1, Paula
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Distinct Role of Transforming Growth Factor-Beta 1 and Fibroblast Growth Factors in Human Amelobastoma Epithelial Cell Proliferation
 Biochemistry and Molecular Biology 2017; 2(1): 1-5 http://www.sciencepublishinggroup.com/j/bmb doi: 10.11648/j.bmb.20170201.11 Distinct Role of Transforming Growth Factor-Beta 1 and Fibroblast Growth Factors
Biochemistry and Molecular Biology 2017; 2(1): 1-5 http://www.sciencepublishinggroup.com/j/bmb doi: 10.11648/j.bmb.20170201.11 Distinct Role of Transforming Growth Factor-Beta 1 and Fibroblast Growth Factors
Supplementary Figure 1. Chimeric analysis of inner ears. (A-H) Chimeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears.
 Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice
 Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
Impact of Sox9 Dosage and Hes1-mediated Notch Signaling in Controlling the Plasticity of Adult Pancreatic Duct Cells in Mice Shinichi Hosokawa 1,3,Kenichiro Furuyama 1,3, Masashi Horiguchi 1,3,Yoshiki
Independent regulation of Dlx2 expression in the epithelium and mesenchyme of the first branchial arch
 Development 127, 217-224 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2450 217 Independent regulation of Dlx2 expression in the epithelium and mesenchyme of the first branchial
Development 127, 217-224 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2450 217 Independent regulation of Dlx2 expression in the epithelium and mesenchyme of the first branchial
Tetrapod Limb Development
 IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
Supporting Information
 Supporting Information Voutilainen et al. 0.07/pnas.06709 SI Materials and Methods Animals. The generation and genotyping of K-da-A (K- da) () and N-κ reporter mice () has been described earlier. oth strains
Supporting Information Voutilainen et al. 0.07/pnas.06709 SI Materials and Methods Animals. The generation and genotyping of K-da-A (K- da) () and N-κ reporter mice () has been described earlier. oth strains
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Introduction Kit Components Cat. # # of vials Reagent Quantity Storage
 Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
Human Pluripotent Stem Cell Cardiomyocyte Differentiation Kit (PSCCDK) Catalog #5901 Introduction Human pluripotent stem cells (hpsc), including embryonic stem cells (ESC) and induced pluripotent stem
Teeth, orofacial development and
 Teeth, orofacial development and cleft anomalies Miroslav Peterka Variability of jaws in vertebrates. (A) cartilaginous fish shark; (B) an example of a bone fish; (C ) amphibian frog; (D) reptile - turtle;
Teeth, orofacial development and cleft anomalies Miroslav Peterka Variability of jaws in vertebrates. (A) cartilaginous fish shark; (B) an example of a bone fish; (C ) amphibian frog; (D) reptile - turtle;
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
o~ r;'c' - OSTEOARTHRITIS
 Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Osteoarthritis and Cartilage (2001) 9, Supplement A, S102-S108 2001 OsteoArthritis Research Society International doi:10.1053/joca.2001.0451, available online at http://www.idealibrary.com on IDE~l Osteoarthritis
Lecture 2 Maxillary central incisor
 Lecture 2 Maxillary central incisor Generally The deciduous tooth appears in the mouth at 3 18 months of age, with 6 months being the average and is replaced by the permanent tooth around 7 8 years of
Lecture 2 Maxillary central incisor Generally The deciduous tooth appears in the mouth at 3 18 months of age, with 6 months being the average and is replaced by the permanent tooth around 7 8 years of
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
SUPPLEMENTARY INFORMATION
 b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats
 Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats Summary of Doctoral Thesis Hidenori Yasuda Graduate School of Veterinary
Analysis on the mechanism of reduced nephron number and the pathological progression of chronic renal failure in Astrin deficient rats Summary of Doctoral Thesis Hidenori Yasuda Graduate School of Veterinary
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Figure (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were
 Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Summary and Concluding Remarks
 Summary and Concluding Remarks Chapter 6 The intestinal epithelium provides an excellent model system for investigating molecular mechanisms regulating cell lineage establishment, stem cell proliferation,
Summary and Concluding Remarks Chapter 6 The intestinal epithelium provides an excellent model system for investigating molecular mechanisms regulating cell lineage establishment, stem cell proliferation,
Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in
 Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in nulliparous (left panel) and InvD6 mouse mammary glands (right
Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in nulliparous (left panel) and InvD6 mouse mammary glands (right
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Interactions between cancer stem cells and their niche govern metastatic colonization
 Correction Interactions between cancer stem cells and their niche govern metastatic colonization Ilaria Malanchi, Albert Santamaria-Martínez, Evelyn Susanto, Hong Peng, Hans-Anton Lehr, Jean-Francois Delaloye
Correction Interactions between cancer stem cells and their niche govern metastatic colonization Ilaria Malanchi, Albert Santamaria-Martínez, Evelyn Susanto, Hong Peng, Hans-Anton Lehr, Jean-Francois Delaloye
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM.
 Supplementary Figure 1 Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM. (a-c) Heterozygous c.216ga mice displayed normal hair bundle morphology at P18. (d-i) Disorganized hair bundles
Supplementary Figure 1 Analysis of hair bundle morphology in Ush1c c.216g>a mice at P18 by SEM. (a-c) Heterozygous c.216ga mice displayed normal hair bundle morphology at P18. (d-i) Disorganized hair bundles
Supplementary Figure 1. Nature Neuroscience: doi: /nn.4547
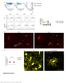 Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Evaluation of directed and random motility in microslides Assessment of leukocyte adhesion in flow chambers
 Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for
 Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for LacZ activity, which reflects Egr1 expression. (A)
Supplemental Figure 1. Egr1 expression in adult Achilles tendons. (A,B) Achilles tendons were isolated from 2 month-old Egr1 +/- mice and stained for LacZ activity, which reflects Egr1 expression. (A)
B. Incorrect! The ectoderm does not produce the dermis. C. Incorrect! The dermis is derived from the mesoderm.
 Human Anatomy - Problem Drill 04: The Integumentary System Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as 1. From the inner cell
Human Anatomy - Problem Drill 04: The Integumentary System Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as 1. From the inner cell
Original Histological and Immunohistochemical Observation of the Furcation Area Formation with the Subpulpal Lobus of Rat Molar
 Eri Osawa et al,: The Furcation Area Formation with the Subpulpal Lobus Journal of Hard Tissue Biology 26[2] (2017) 149-156 2017 The Hard Tissue Biology Network Association Printed in Japan, All rights
Eri Osawa et al,: The Furcation Area Formation with the Subpulpal Lobus Journal of Hard Tissue Biology 26[2] (2017) 149-156 2017 The Hard Tissue Biology Network Association Printed in Japan, All rights
Further studies on the melanophores of periodic albino mutant of Xenopus laevis
 J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
J. Embryol. exp. Morph. 91, 65-78 (1986) 65 Printed in Great Britain The Company of Biologists Limited 1986 Further studies on the melanophores of periodic albino mutant of Xenopus laevis T. FUKUZAWA AND
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions
 CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
The developing murine molar tooth germ provides a powerful
 Conservation of early odontogenic signaling pathways in Aves YiPing Chen*, Yanding Zhang*, Ting-Xing Jiang, Amanda J. Barlow, Tara R. St. Amand, Yueping Hu, Shaun Heaney*, Philippa Francis-West, Cheng-Ming
Conservation of early odontogenic signaling pathways in Aves YiPing Chen*, Yanding Zhang*, Ting-Xing Jiang, Amanda J. Barlow, Tara R. St. Amand, Yueping Hu, Shaun Heaney*, Philippa Francis-West, Cheng-Ming
Figure 1. Morphology of a normal furcation area (hematoxylin and eosin [H&E] staining), a Low magnification view of a furcation area.
![Figure 1. Morphology of a normal furcation area (hematoxylin and eosin [H&E] staining), a Low magnification view of a furcation area. Figure 1. Morphology of a normal furcation area (hematoxylin and eosin [H&E] staining), a Low magnification view of a furcation area.](/thumbs/94/120315208.jpg) 905 Errata that were The following figures are four-color representations of figures incorrectly published in black and white in the July and August issues. Immunohistochemical Expression of Extracellular
905 Errata that were The following figures are four-color representations of figures incorrectly published in black and white in the July and August issues. Immunohistochemical Expression of Extracellular
Transgenic Expression of FGF8 and FGF10 Induces Transdifferentiation of Pancreatic Islet Cells into Hepatocytes and Exocrine Cells
 Biochemical and Biophysical Research Communications 292, 138 143 (2002) doi:10.1006/bbrc.2002.6601, available online at http://www.idealibrary.com on Transgenic Expression of FGF8 and FGF10 Induces Transdifferentiation
Biochemical and Biophysical Research Communications 292, 138 143 (2002) doi:10.1006/bbrc.2002.6601, available online at http://www.idealibrary.com on Transgenic Expression of FGF8 and FGF10 Induces Transdifferentiation
Urogenital Development
 2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
CHAPTER 6 SUMMARIZING DISCUSSION
 CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:!
 LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Expression of Axin2 Indicates a Role for Canonical Wnt Signaling in Development of the Crown and Root During Pre- and Postnatal Tooth Development
 DEVELOPMENTAL DYNAMICS 239:160 167, 2010 SPECIAL ISSUE RESEARCH ARTICLE Expression of Axin2 Indicates a Role for Canonical Wnt Signaling in Development of the Crown and Root During Pre- and Postnatal Tooth
DEVELOPMENTAL DYNAMICS 239:160 167, 2010 SPECIAL ISSUE RESEARCH ARTICLE Expression of Axin2 Indicates a Role for Canonical Wnt Signaling in Development of the Crown and Root During Pre- and Postnatal Tooth
Immature organoids appear after hours.
 THE ESSENTIALS OF LIFE SCIENCE RESEARCH GLOBALLY DELIVERED Allison Ruchinskas, B.S., and James Clinton, Ph.D. ATCC Cell Systems, Gaithersburg, MD INTRODUCTION Figure 1. Mouse small intestinal organoid
THE ESSENTIALS OF LIFE SCIENCE RESEARCH GLOBALLY DELIVERED Allison Ruchinskas, B.S., and James Clinton, Ph.D. ATCC Cell Systems, Gaithersburg, MD INTRODUCTION Figure 1. Mouse small intestinal organoid
Early cell death (FGF) B No RunX transcription factor produced Yes No differentiation
 Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Construction of Nephron by Fusion of Adult Glomeruli to Ureteric Buds with Type V Collagen. Yusuke Murasawa, Pi-chao Wang
 Construction of Nephron by Fusion of Adult Glomeruli to Ureteric Buds with Type V Collagen Yusuke Murasawa, Pi-chao Wang Abstract Although tissue engineering of artificial organs such as skin or cartilage
Construction of Nephron by Fusion of Adult Glomeruli to Ureteric Buds with Type V Collagen Yusuke Murasawa, Pi-chao Wang Abstract Although tissue engineering of artificial organs such as skin or cartilage
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
IMMUNOHISTOCHEMICAL PROFILE OF ODONTOGENIC EPITHELIUM OF DEVELOPING DOG TEETH (CANIS FAMILIARIS)
 IMMUNOHISTOCHEMICAL PROFILE OF ODONTOGENIC EPITHELIUM OF DEVELOPING DOG TEETH (CANIS FAMILIARIS) By SULETTE NEL Submitted in partial fulfilment of the requirements for the degree of Master of Science (Odontology)
IMMUNOHISTOCHEMICAL PROFILE OF ODONTOGENIC EPITHELIUM OF DEVELOPING DOG TEETH (CANIS FAMILIARIS) By SULETTE NEL Submitted in partial fulfilment of the requirements for the degree of Master of Science (Odontology)
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
