Prior Authorization Proposal: Oncology Agents
|
|
|
- Elinor Marsh
- 5 years ago
- Views:
Transcription
1 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon Phone Fax Prior Authorization Proposal: Oncology Agents Purpose for the Proposal: The purpose of the prior authorization (PA) proposal is to ensure medically appropriate use of antineoplastic agents, both those recently approved and those approved by the United States Food and Drug Administration (FDA) in the future. Background: Oncology is a rapidly growing area of drug development. In 2017 alone, the FDA approved 12 novel drug therapies as well as 2 gene therapies for oncology. 1-3 Additional indications beyond the first FDA approval are often studied in clinical trials for these agents as well and many obtain subsequent new or expanded indication approvals. Off-label use beyond FDA-approved indications may be supported by Centers for Medicare and Medicaid Services (CMS) compendia. One of the most commonly used guidelines for oncology is the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology (NCCN Guidelines ), which are supported by CMS as a compendia for use in the determination of a medically-accepted indication of drugs and biologicals used offlabel in an anticancer chemotherapeutic regimen. 4,5 Both oral and parenteral oncology drugs are typically studied in multiple indications, may have safety concerns, and are generally of high cost. Therefore, a broad PA policy which requires use to align with FDA-approved or NCCN-supported indications would ensure appropriate use of these agents. Proposal and Methods: A new Antineoplastics preferred drug list (PDL) class was created and all agents which applied (whether previously in a PDL class or not) were added to the class. This proposal to require PA encompasses any new start of an antineoplastic agent approved within the past 10 years. Drugs with an original FDA-approval date prior to January 2008, or subsequently approved new formulations of these older agents, are exempted from this criteria given the increased clinical experience with these agents. The CMS Approval Date and National Drug Data File (NDDF) Add Date were utilized to identify antineoplastic drugs by unique generic name with a date for either field greater than or equal to 1/1/2008 for agents proposed to require PA. Any unique generic antineoplastic agents with any formulation identified with those parameters prior to 1/1/2008 are proposed to not require PA. A list of the medications proposed to require PA (n=85 unique generic name drugs) is provided in the proposed PA (Appendix 1) while the medications proposed to not require PA (n=108 unique generic name drugs) is provided in Appendix 2. Recommendation: Implement PA criteria to ensure medically appropriate use for all drugs in the Antineoplastics PDL class originally approved by the FDA on 1/1/2008 or later (Appendix 1). Author: Julia Page, PharmD January 2019
2 In future, any antineoplastic new molecular entities and new formulations of drugs which already require PA will be added to the Antineoplastics PDL class upon approval by the Food and Drug Administration (FDA), and PA criteria will be updated to incorporate these new agents at subsequent P&T meetings. Appendix 1: Proposed Prior Authorization Criteria Oncology Agents Goal(s): To ensure appropriate use for oncology medications based on FDA-approved and compendia-recommended (i.e., National Comprehensive Cancer Network [NCCN]) indications. Length of Authorization: Up to 1 year Requires PA: Both pharmacy and physician-administered claims for drugs listed in Table 1 Covered Alternatives: Current PMPDP preferred drug list per OAR at Searchable site for Oregon FFS Drug Class listed at Approval Criteria 1. What diagnosis is being treated? Record ICD10 code. 2. Is the request for any continuation of therapy? Yes: Approve for length of therapy or 12 months, whichever is less. No: Go to #3 3. Is the diagnosis funded by OHP? Yes: Go to #4 No: Pass to RPh. Deny; not funded by the OHP.
3 Approval Criteria 4. Is the indication FDA-approved for the requested drug? Note: This includes all information required in the FDAapproved indication, including but not limited to the following as applicable: diagnosis, stage of cancer, biomarkers, place in therapy, and use as monotherapy or combination therapy. 5. Is the indication recommended by National Comprehensive Cancer Network (NCCN) Guidelines for the requested drug? Note: This includes all information required in the NCCN recommendation, including but not limited to the following as applicable: diagnosis, stage of cancer, biomarkers, place in therapy, and use as monotherapy or combination therapy. Yes: Pass to RPh. Approve for length of therapy or 12 months, whichever is less. Yes: Pass to RPh. Approve for length of therapy or 12 months, whichever is less. No: Go to #5 No: Pass to RPh. Deny; medical appropriateness. Table 1. Oncology agents which apply to this policy Generic Name abemaciclib VERZENIO abiraterone acet,submicronized YONSA abiraterone acetate ZYTIGA acalabrutinib CALQUENCE ado-trastuzumab emtansine KADCYLA afatinib dimaleate GILOTRIF alectinib HCl ALECENSA apalutamide ERLEADA asparaginase (Erwinia chrysan) ERWINAZE atezolizumab TECENTRIQ avelumab BAVENCIO axicabtagene ciloleucel YESCARTA axitinib INLYTA belinostat BELEODAQ
4 bendamustine HCl bendamustine HCl bendamustine HCl binimetinib blinatumomab bosutinib brentuximab vedotin brigatinib cabazitaxel cabozantinib s-malate cabozantinib s-malate carfilzomib cemiplimab-rwlc ceritinib cobimetinib fumarate copanlisib di-hcl crizotinib dabrafenib mesylate dacomitinib daratumumab degarelix acetate dinutuximab durvalumab duvelisib elotuzumab enasidenib mesylate encorafenib enzalutamide eribulin mesylate everolimus everolimus gilteritinib glasdegib ibrutinib idelalisib ingenol mebutate inotuzumab ozogamicin ipilimumab BENDAMUSTINE HCL BENDEKA TREANDA MEKTOVI BLINCYTO BOSULIF ADCETRIS ALUNBRIG JEVTANA CABOMETYX COMETRIQ KYPROLIS LIBTAYO ZYKADIA COTELLIC ALIQOPA XALKORI TAFINLAR VIZIMPRO DARZALEX FIRMAGON UNITUXIN IMFINZI COPIKTRA EMPLICITI IDHIFA BRAFTOVI XTANDI HALAVEN AFINITOR AFINITOR DISPERZ XOSPATA DAURISMO IMBRUVICA ZYDELIG PICATO BESPONSA YERVOY
5 ivosidenib ixazomib citrate larotrectinib lenvatinib mesylate lorlatinib midostaurin moxetumomab pasudotox-tdfk necitumumab neratinib maleate niraparib tosylate nivolumab obinutuzumab ofatumumab olaparib olaratumab omacetaxine mepesuccinate osimertinib mesylate palbociclib panobinostat lactate pazopanib HCl pembrolizumab pertuzumab pomalidomide ponatinib HCl pralatrexate ramucirumab regorafenib ribociclib succinate ribociclib succinate/letrozole romidepsin romidepsin rucaparib camsylate ruxolitinib phosphate siltuximab sipuleucel-t/lactated ringers sonidegib phosphate talimogene laherparepvec talazoparib TIBSOVO NINLARO VITRAKVI LENVIMA LORBRENA RYDAPT LUMOXITI PORTRAZZA NERLYNX ZEJULA OPDIVO GAZYVA ARZERRA LYNPARZA LARTRUVO SYNRIBO TAGRISSO IBRANCE FARYDAK VOTRIENT KEYTRUDA PERJETA POMALYST ICLUSIG FOLOTYN CYRAMZA STIVARGA KISQALI KISQALI FEMARA CO-PACK ISTODAX ROMIDEPSIN RUBRACA JAKAFI SYLVANT PROVENGE ODOMZO IMLYGIC TALZENNA
6 tisagenlecleucel trabectedin trametinib dimethyl sulfoxide trifluridine/tipiracil HCl vandetanib vandetanib vemurafenib venetoclax venetoclax vismodegib ziv-aflibercept KYMRIAH YONDELIS MEKINIST LONSURF CAPRELSA VANDETANIB ZELBORAF VENCLEXTA VENCLEXTA STARTING PACK ERIVEDGE ZALTRAP P&T/DUR Review: 1/2019 (JP) Implementation: TBD References: 1. FDA approves CAR-T cell therapy to treat adults with certain types of large B-cell lymphoma. United States Food and Drug Administration Website. Updated March 21, Accessed August 20, FDA approval brings first gene therapy to the United States. United States Food and Drug Administration Website. Updated March 26, Accessed August 20, Novel Drug Approvals for United States Food and Drug Administration Website. Updated February 2, Accessed August 20, Compendia. CMS.gov. Available from: Accessed August 20, NCCN Compendium Revision Request. CMS.gov. Available from: Accessed August 20, Appendix 2. Antineoplastic Agents Proposed to Not Require Prior Authorization Generic Name aldesleukin PROLEUKIN alemtuzumab CAMPATH alitretinoin PANRETIN
7 Generic Name altretamine aminolevulinic acid HCl aminolevulinic acid HCl anastrozole anastrozole arsenic trioxide azacitidine azacitidine BCG live BCG live bexarotene bexarotene bicalutamide bicalutamide bleomycin sulfate bleomycin sulfate bortezomib bortezomib busulfan busulfan busulfan capecitabine capecitabine carboplatin carmustine carmustine in polifeprosan 20 cetuximab chlorambucil cisplatin cladribine clofarabine clofarabine cyclophosphamide cyclophosphamide cytarabine cytarabine/pf dacarbazine HEXALEN AMELUZ LEVULAN ANASTROZOLE ARIMIDEX TRISENOX AZACITIDINE VIDAZA BCG (TICE STRAIN) THERACYS BEXAROTENE TARGRETIN BICALUTAMIDE CASODEX BLEO 15K BLEOMYCIN SULFATE BORTEZOMIB VELCADE BUSULFAN BUSULFEX MYLERAN CAPECITABINE XELODA CARBOPLATIN BICNU GLIADEL ERBITUX LEUKERAN CISPLATIN CLADRIBINE CLOFARABINE CLOLAR CYCLOPHOSPHAMIDE NEOSAR CYTARABINE CYTARABINE DACARBAZINE
8 Generic Name dactinomycin COSMEGEN dactinomycin DACTINOMYCIN dasatinib SPRYCEL daunorubicin HCl DAUNORUBICIN HCL daunorubicin/cytarabine lipos VYXEOS decitabine DACOGEN decitabine DECITABINE docetaxel DOCEFREZ docetaxel DOCETAXEL docetaxel TAXOTERE doxorubicin HCl ADRIAMYCIN doxorubicin HCl ADRIAMYCIN RDF doxorubicin HCl DOXORUBICIN HCL doxorubicin HCl RUBEX doxorubicin HCl peg-liposomal DOXIL doxorubicin HCl peg-liposomal DOXORUBICIN HCL LIPOSOME doxorubicin HCl peg-liposomal LIPODOX doxorubicin HCl peg-liposomal LIPODOX 50 epirubicin HCl ELLENCE epirubicin HCl EPIRUBICIN HCL erlotinib HCl TARCEVA estramustine phosphate sodium EMCYT etoposide ETOPOSIDE etoposide TOPOSAR etoposide phosphate ETOPOPHOS exemestane AROMASIN exemestane EXEMESTANE floxuridine FLOXURIDINE fludarabine phosphate FLUDARABINE PHOSPHATE fluorouracil ADRUCIL fluorouracil CARAC fluorouracil EFUDEX fluorouracil FLUOROURACIL fluorouracil TOLAK flutamide FLUTAMIDE fulvestrant FASLODEX gefitinib IRESSA
9 Generic Name gemcitabine HCl gemcitabine HCl hydroxyurea hydroxyurea idarubicin HCl idarubicin HCl ifosfamide ifosfamide ifosfamide/mesna imatinib mesylate imatinib mesylate interferon alfa-2b,recomb. interferon gamma-1b,recomb. irinotecan HCl irinotecan HCl irinotecan liposomal ixabepilone kit Y-90/ibritumomab/h.albumin lapatinib ditosylate lenalidomide letrozole letrozole lomustine mechlorethamine HCl mechlorethamine HCl megestrol acetate melphalan melphalan melphalan HCl melphalan HCl melphalan HCl/betadex sbes mercaptopurine mercaptopurine methotrexate methotrexate sodium methotrexate sodium methotrexate sodium/pf GEMCITABINE HCL GEMZAR HYDREA HYDROXYUREA IDAMYCIN PFS IDARUBICIN HCL IFEX IFOSFAMIDE IFOSFAMIDE-MESNA GLEEVEC IMATINIB MESYLATE INTRON A ACTIMMUNE CAMPTOSAR IRINOTECAN HCL ONIVYDE IXEMPRA ZEVALIN TYKERB REVLIMID FEMARA LETROZOLE GLEOSTINE MUSTARGEN VALCHLOR MEGESTROL ACETATE ALKERAN MELPHALAN ALKERAN MELPHALAN HCL EVOMELA MERCAPTOPURINE PURIXAN XATMEP METHOTREXATE TREXALL METHOTREXATE
10 Generic Name methotrexate sodium/pf mitomycin mitomycin mitotane mitoxantrone HCl nelarabine nilotinib HCl nilutamide nilutamide oxaliplatin paclitaxel paclitaxel protein-bound panitumumab pegaspargase peginterferon alfa-2b pemetrexed disodium pentostatin plicamycin procarbazine HCl rituximab/hyaluronidase,human sorafenib tosylate streptozocin sunitinib malate tamoxifen citrate tamoxifen citrate temozolomide temozolomide temsirolimus teniposide thioguanine thiotepa thiotepa topotecan HCl topotecan HCl toremifene citrate trastuzumab tretinoin METHOTREXATE SODIUM MITOMYCIN MUTAMYCIN LYSODREN MITOXANTRONE HCL ARRANON TASIGNA NILANDRON NILUTAMIDE OXALIPLATIN PACLITAXEL ABRAXANE VECTIBIX ONCASPAR SYLATRON ALIMTA NIPENT MITHRACIN MATULANE RITUXAN HYCELA NEXAVAR ZANOSAR SUTENT SOLTAMOX TAMOXIFEN CITRATE TEMODAR TEMOZOLOMIDE TORISEL TENIPOSIDE TABLOID TEPADINA THIOTEPA HYCAMTIN TOPOTECAN HCL FARESTON HERCEPTIN TRETINOIN
11 Generic Name valrubicin vinblastine sulfate vincristine sulfate vincristine sulfate vincristine sulfate liposomal vinorelbine tartrate vinorelbine tartrate vorinostat VALSTAR VINBLASTINE SULFATE VINCASAR PFS VINCRISTINE SULFATE MARQIBO NAVELBINE VINORELBINE TARTRATE ZOLINZA
MEDICAL NECESSITY GUIDELINE
 PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Re: Integrated Oncology Management Program with evicore healthcare: Update on codes requiring precertification
 Ajani Nimmagadda, MD Senior Medical Director , , Re: Integrated Oncology Management Program with evicore
Ajani Nimmagadda, MD Senior Medical Director , , Re: Integrated Oncology Management Program with evicore
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Self Administered Oncology Agents Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Self Administered Oncology Agents Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Formulary Chemotherapy Agents: (Current as of 6/2018) Therapeutic Class
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
Chemotherapy 101 for Radiation Oncology Workers
 Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Overview of Cancer. Laura Bingell RN Transition Center Nurse for MFP (607)
 Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
Acute Lymphocytic Leukemia
 Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
OPENING KEYNOTE: PRECISION MEDICINE AT THE INFLECTION POINT. Session PM1, March 5, 2018 Damon Hostin, CEO, Precision Medicine Alliance, LLC
 OPENING KEYNOTE: PRECISION MEDICINE AT THE INFLECTION POINT Session PM1, March 5, 2018 Damon Hostin, CEO, Precision Medicine Alliance, LLC 1 Conflicts of Interest Damon Hostin Has no real or apparent conflicts
OPENING KEYNOTE: PRECISION MEDICINE AT THE INFLECTION POINT Session PM1, March 5, 2018 Damon Hostin, CEO, Precision Medicine Alliance, LLC 1 Conflicts of Interest Damon Hostin Has no real or apparent conflicts
Genetics in Cancer Therapy. Raju Kucherlapati, Ph.D. Harvard Medical School
 Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year
 ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA
 ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA
 ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
Protocol Number Tumour Group Protocol Name on NCCP website 22/02/ Lung Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy
 Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249
 Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Self Administered Oncology Agents Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Self Administered Oncology Agents Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
DOXOrubicin, Cyclophosphamide (AC 60/600) 21 day followed by weekly PACLitaxel (80) Therapy (AC-T) 261 CARBOplatin (AUC4-6) Monotherapy-21 days
 Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Self Administered Oncology Agents Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Self Administered Oncology Agents Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Objectives
 Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Pharmacy Medical Necessity Guidelines: Oral Cancer Medications
 Pharmacy Medical Necessity Guidelines: Effective: February 18, 2019 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: February 18, 2019 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Description The following are synthetic cannabinoids requiring prior authorization: dronabinol (Marinol, Syndros ), nabilone (Cesamet )
 Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data
 Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Primary malignant neoplasms, not lymphatic or hematopoietic. Secondary malignant neoplasms (i.e.metastatic) Malignant neoplasm, unknown site
 Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Committee Approval Date: December 12, 2014 Next Review Date: July 2015
 Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
ANTIEMETICS UTILIZATION MANAGEMENT CRITERIA
 ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced
 ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
Subject: Palonosetron Hydrochloride (Aloxi )
 09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
To help doctors give their patients the best possible care, the American
 Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19
 Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Hazardous Medication List
 Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Guidelines on Chemotherapy-induced Nausea and Vomiting in Pediatric Cancer Patients
 Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites.
 WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting
 Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Subject: Fosnetupitant-Palonosetron (Akynzeo) IV
 09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A
 Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
HCPCS Code/ generic (Brand) Name J7506. J8520 capecitabine (Xeloda) 1. J8521 capecitabine. J8530 cyclophosphamide. (Cytoxan) 1
 drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
AD regimen, 576 adrenal steroid inhibitor. aminoglutethimide, 23 25
 A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
Bladder Cancer (Urothelial) Pathways
 Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
SCI. SickKids-Caribbean Initiative Enhancing Capacity for Care in Paediatric Cancer and Blood Disorders
 1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
Bladder Cancer Pathways (Urothelial)
 Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Pharmacy Policy Bulletin
 Pharmacy Policy Bulletin Title: Policy #: Oncology Agents Rx.01.67 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product line, group, or contract. Some
Pharmacy Policy Bulletin Title: Policy #: Oncology Agents Rx.01.67 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product line, group, or contract. Some
INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY]
![INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY] INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY]](/thumbs/85/92112268.jpg) Commercial Medical Oncology Program Review Code List 3rd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of
Commercial Medical Oncology Program Review Code List 3rd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of
Supplementary Figures
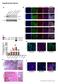 Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy
 Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy Faculty Disclosures Dr. Holle has provided content expertise on patient education materials for Innocrin Pharmaceuticals
Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy Faculty Disclosures Dr. Holle has provided content expertise on patient education materials for Innocrin Pharmaceuticals
Ovarian Cancer. compendia TREATMENT OF
 drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting
 West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care
 Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
CPT Service Description Effective Date
 Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi )
 09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
ALKYLATING AGENTS (22)
 Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Specialty Drug List - Sorted by Therapeutic Class Developed for the Mississippi Division of Medicaid by Mercer
 ANTI-INFECTIVE ABELCET 100 MG/20 ML VIAL 4/1/2017 ANTI-INFECTIVE AMBISOME 50 MG VIAL 4/1/2017 ANTI-INFECTIVE ANCOBON 250 MG CAPSULE 4/1/2017 ANTI-INFECTIVE ANCOBON 500 MG CAPSULE 4/1/2017 ANTI-INFECTIVE
ANTI-INFECTIVE ABELCET 100 MG/20 ML VIAL 4/1/2017 ANTI-INFECTIVE AMBISOME 50 MG VIAL 4/1/2017 ANTI-INFECTIVE ANCOBON 250 MG CAPSULE 4/1/2017 ANTI-INFECTIVE ANCOBON 500 MG CAPSULE 4/1/2017 ANTI-INFECTIVE
Form 2023 R2.0: Ovarian Cancer Pre-HSCT Data
 Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care
 Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care Presented as a Live Webinar Wednesday, April 13, 2016 12:00 p.m. 1:00 p.m. ET Tuesday,
Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care Presented as a Live Webinar Wednesday, April 13, 2016 12:00 p.m. 1:00 p.m. ET Tuesday,
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY
 ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment
 Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Clinical Policy: Aprepitant (Emend) Reference Number: CP.PMN.19 Effective Date: 11/06 Last Review Date: 08/17
 Clinical Policy: (Emend) Reference Number: CP.PMN.19 Effective Date: 11/06 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Emend) Reference Number: CP.PMN.19 Effective Date: 11/06 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
The Royal Marsden NHS Foundation Trust Medicines Formulary. NICE register for medicines initiated at the trust. Type and code. Publication.
 NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
MABCAMPATH mg 3 INJ GENZYME BIOPHARMACEUTICALS. MABCAMPBATH mg 3 INJ GENZYME BIOPHARMACEUTICALS
 ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
Cancer Therapy Update in 2017
 Cancer Therapy Update in 2017 Editor: Le Wang, M.D., Ph.D Medical Oncology and Hematology (Board-Certified) Ibrutinib (Imbruvica) Pharmacyclics 1 /19/2017 Lymphoma Marginal Zone Lymphoma (MZL), Relapsed/Refractory
Cancer Therapy Update in 2017 Editor: Le Wang, M.D., Ph.D Medical Oncology and Hematology (Board-Certified) Ibrutinib (Imbruvica) Pharmacyclics 1 /19/2017 Lymphoma Marginal Zone Lymphoma (MZL), Relapsed/Refractory
COST CONSIDERATIONS Union for International Cancer Control 2014 Review of Cancer Medicines on the WHO List of Essential Medicines!!!!!!!!!
 UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UNDERWRITING GUIDE. Secure Advantage HOSPITAL COVERAGE. The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization
 UNDERWRITING GUIDE Secure Advantage HOSPITAL COVERAGE The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization TABLE OF CONTENTS Contents Page Secure Advantage Underwriting Guide...2
UNDERWRITING GUIDE Secure Advantage HOSPITAL COVERAGE The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization TABLE OF CONTENTS Contents Page Secure Advantage Underwriting Guide...2
NICE-approved drugs for specialities which fall outside of the services currently provided by the Trust
 NICE-approved drugs for specialities which fall outside of the services currently provided by the Trust Drug NICE-approved use NICE TA link Abiraterone Abiraterone Adalimumab Adalimumab Adalimumab Adefovir
NICE-approved drugs for specialities which fall outside of the services currently provided by the Trust Drug NICE-approved use NICE TA link Abiraterone Abiraterone Adalimumab Adalimumab Adalimumab Adefovir
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer
 APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
Introduction to Antineoplastic Prescribing
 Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
The Royal Marsden NHS Foundation Trust Medicines Formulary. NICE register for medicines initiated at the trust. Type and code. Publication.
 NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
Clinical Tools and Resources for Self-Study and Patient Education
 Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
EquiCash UNDERWRITING GUIDE. Equitable &You. The Policy That Pays You Cash In The Event You Have To Be Hospitalized HOSPITAL PLAN
 UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY CLASSIFICATION
 GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
StrandAdvantage Tissue-Specific Cancer Genomic Tests. Empowering Crucial First-Line Therapy Decisions for Your Patient
 StrandAdvantage Tissue-Specific Cancer Genomic Tests Empowering Crucial First-Line Therapy Decisions for Your Patient Harness the power of precision medicine with StrandAdvantage Precision medicine in
StrandAdvantage Tissue-Specific Cancer Genomic Tests Empowering Crucial First-Line Therapy Decisions for Your Patient Harness the power of precision medicine with StrandAdvantage Precision medicine in
Haematology, Oncology and Palliative Care Directorate.
 Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
REGULATORY INFORMATION
 REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
Chemotherapy-Induced Nausea and Vomiting: Strategies for Achieving Optimal Control
 Chemotherapy-Induced Nausea and Vomiting Strategies for Achieving Learning Objectives Describe the challenges of assessing nausea in patients undergoing chemotherapy and the impact of nausea and also vomiting
Chemotherapy-Induced Nausea and Vomiting Strategies for Achieving Learning Objectives Describe the challenges of assessing nausea in patients undergoing chemotherapy and the impact of nausea and also vomiting
intolerance to, contraindication to, or therapeutic failure on a minimum 3 month trial of Inflectra*
 What s New Medical Pharmaceutical Policy March 2018 Updates MBP 5.0 Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)- Updated policy Remicade (infliximab), Inflectra (infliximab-dyyb)
What s New Medical Pharmaceutical Policy March 2018 Updates MBP 5.0 Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)- Updated policy Remicade (infliximab), Inflectra (infliximab-dyyb)
Exposure Control Plan for Cytotoxic Substances
 Exposure Control Plan Department of www.rms.ubc.ca UBC-RMS-OHS-ECP-17-001 Effective date: December 8, 2017 Review date: NA Supersedes: NA 1. PURPOSE Exposure Control Plan for Cytotoxic Substances The University
Exposure Control Plan Department of www.rms.ubc.ca UBC-RMS-OHS-ECP-17-001 Effective date: December 8, 2017 Review date: NA Supersedes: NA 1. PURPOSE Exposure Control Plan for Cytotoxic Substances The University
Adult Intravenous Systemic Anticancer Therapy (SACT) Section A. SUMMARY of SCHEME QIPP Reference
 CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
Azacitidine Vidaza Non-transplant myelodysplastic syndrome Funded Funded Funded Funded Funded Funded Not Funded
 Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
Guideline Update on Antiemetics
 Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Managements of Chemotherpay Induded Nausea and Vomiting
 REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
Corporate Medical Policy
 Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
Clinical Policy: Pemetrexed (Alimta) Reference Number: CP.PHAR.368 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Pharmacy Management Drug Policy
 SUBJECT: Oncology Clinical Review Prior Authorization (Oncology-CRPA) Rx Drugs POLICY NUMBER: Pharmacy-33 EFFECTIVE DATE: 10/13 LAST REVIEW DATE: 11/12/2018 If the member s subscriber contract excludes
SUBJECT: Oncology Clinical Review Prior Authorization (Oncology-CRPA) Rx Drugs POLICY NUMBER: Pharmacy-33 EFFECTIVE DATE: 10/13 LAST REVIEW DATE: 11/12/2018 If the member s subscriber contract excludes
Chemotherapy Agents 101
 Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
Leukemia. Treatment of. compendia. Associated ICD-9-CM Codes: Drug & Administration. managedcareoncology.com
 Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
DRUG PROPERTIES YOU NEED TO KNOW
 Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Clinical Therapeutic Intelligence Report: 2015 Year in Review
 Clinical Therapeutic Intelligence Report: 2015 Year in Review This issue highlights the many ground-breaking therapies approved by the U.S. Food and Drug Administration over the course of 2015. Highlights
Clinical Therapeutic Intelligence Report: 2015 Year in Review This issue highlights the many ground-breaking therapies approved by the U.S. Food and Drug Administration over the course of 2015. Highlights
Pharmacy Management Drug Policy
 SUBJECT: Oncology Clinical Review Prior Authorization (Oncology-CRPA) Rx Drugs POLICY NUMBER: Pharmacy-33 EFFECTIVE DATE: 10/13 LAST REVIEW DATE: 03/22/2018 If the member s subscriber contract excludes
SUBJECT: Oncology Clinical Review Prior Authorization (Oncology-CRPA) Rx Drugs POLICY NUMBER: Pharmacy-33 EFFECTIVE DATE: 10/13 LAST REVIEW DATE: 03/22/2018 If the member s subscriber contract excludes
Welcome. Coding uidelines Coding Guidelines Coding. Coding Guidelines Coding Guidelines. Contact Us. Edition #1 March 2018
 Coding Guidelines Coding Guidelines uidelines Coding Guidelines Coding Coding Coding Guidelines Guidelines Coding uidelines Coding Guidelines Coding Coding Chemotherapy Guidelines and Radiation Coding
Coding Guidelines Coding Guidelines uidelines Coding Guidelines Coding Coding Coding Guidelines Guidelines Coding uidelines Coding Guidelines Coding Coding Chemotherapy Guidelines and Radiation Coding
Guidelines for the Use of Anti-Emetics with Chemotherapy
 Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Opening Address: A Quick Survey Thinking 25 Years Backwards & Forwards
 Opening Address: A Quick Survey Thinking 25 Years Backwards & Forwards Jeffrey M. Bockman, PhD Vice President Defined Health 24 th Annual Cancer Progress Conference March 4 th 5 th, 2014 March 4 th 5 th,
Opening Address: A Quick Survey Thinking 25 Years Backwards & Forwards Jeffrey M. Bockman, PhD Vice President Defined Health 24 th Annual Cancer Progress Conference March 4 th 5 th, 2014 March 4 th 5 th,
Supplementary Table 1. Summary of FDA approval data for rare cancer indications from December 1987 to May 2011
 Rare Cancer : Lessons from FDA s by Gaddipati et al. Supplementary Table 1 Summary of FDA approval data for rare cancer indications from December 1987 to May 2011 1. mitoxantrone hydrochloride (NOVANTRONE
Rare Cancer : Lessons from FDA s by Gaddipati et al. Supplementary Table 1 Summary of FDA approval data for rare cancer indications from December 1987 to May 2011 1. mitoxantrone hydrochloride (NOVANTRONE
