Role of the ubiquitin proteasome system in Bombyx mori nucleopolyhedrovirus infection
|
|
|
- Randolph Tate
- 5 years ago
- Views:
Transcription
1 Journal of General Virology (2011), 92, DOI /vir Role of the ubiquitin proteasome system in Bombyx mori nucleopolyhedrovirus infection Susumu Katsuma, 1 Ayumi Tsuchida, 1 Noriko Matsuda-Imai, 2 WonKyung Kang 2 and Toru Shimada 1 Correspondence Susumu Katsuma katsuma@ss.ab.a.u-tokyo.ac.jp 1 Department of Agricultural and Environmental Biology, Graduate School of Agricultural and Life Sciences, The University of Tokyo, Yayoi 1-1-1, Bunkyo-ku, Tokyo , Japan 2 Molecular Entomology Laboratory, RIKEN ASI, 2-1 Hirosawa, Wako , Japan Received 25 September 2010 Accepted 13 November 2010 The ubiquitin proteasome system plays a central role in the degradation of intracellular proteins and is often required for efficient virus infection. Homologues of ubiquitin are found in all group I nucleopolyhedroviruses (NPVs), but their roles in NPV infection are still unclear. This study found that the specific proteasome inhibitor MG-132 markedly reduced budded virus (BV) production and polyhedrin expression in Bombyx mori NPV (BmNPV)-infected BmN-4 cells. Western blot analysis revealed that treatment of cells with MG-132 resulted in delayed and/or dysregulated viral gene product expression. Application of MG-132 significantly reduced BV production when applied up to 12 h post-infection (p.i.), whereas suppression of polyhedrin expression was almost abolished when applied after 6 h p.i. These results suggested that proteosomal degradation of viral and/or host proteins is required at an early stage of infection for efficient polyhedrin expression. To examine further the possible roles of ubiquitin signalling in BmNPV infection, the baculoviral ubiquitin gene (v-ubi ) was deleted from the BmNPV genome. Deletion of v-ubi affected neither BV production nor polyhedrin expression. Furthermore, Western blots also showed that v-ubi was not required for degradation of IE2, which is known as a target viral protein of the ubiquitin proteasome system. INTRODUCTION Ubiquitin is a small, highly conserved protein that is involved in a number of cellular processes, including the targeting of proteins for degradation (Rechsteiner, 1991). In eukaryotic cells, the ubiquitin-dependent proteolytic system is one of the major routes by which intracellular proteins are selectively destroyed. Misfolded or damaged proteins are specifically marked for destruction by the covalent attachment of ubiquitin. Ubiquitylation of a target protein involves multi-step enzymic reactions catalysed by a cascade of enzymes, including ubiquitin-activating enzyme (E1), ubiquitin-conjugating enzyme (E2) and ubiquitin ligase (E3). After ubiquitin is linked to the protein substrate, a polyubiquitin chain is usually formed in which the C terminus of each ubiquitin unit is linked to a specific lysine residue of the previous ubiquitin. The polyubiquitylated substrates are rapidly recognized and degraded by the 26S proteasome (Ciechanover et al., 2000). The family Baculoviridae is a large family of pathogens that infect arthropods, particularly insects of the order Lepidoptera. Members of the genus Nucleopolyhedrovirus within this family have a large circular, supercoiled, dsdna genome packaged into rod-shaped virions. Nucleopolyhedroviruses Supplementary data are available with the online version of this paper. (NPVs) produce two types of virion, occlusion-derived virions (ODVs) and budded virions (BVs). These two types differ in the origin and composition of their envelopes, as well as their roles in the virus life cycle. ODVs, contained in occlusion bodies (OBs), transmit viruses from insect to insect via oral infection, whereas BVs spread infection to neighbouring cells (Keddie et al., 1989). Homologues of ubiquitin are found in most lepidopteran baculoviruses (Katsuma et al., 2008). Baculoviral ubiquitin protein (v-ubi) is 75 % identical to eukaryotic ubiquitin and appears to be present on the inner surface of viral envelopes (Guarino et al., 1995). v-ubi may be involved in the formation of viral particles, because a v-ubi mutant of Autographa californica multiple NPV (AcMNPV) showed a five- to tenfold reduction in BV production (Reilly & Guarino, 1996). Furthermore, biochemical experiments have suggested that v-ubi may function in baculovirus replication to block the destruction of short-lived proteins by the host degradative pathway (Haas et al., 1996). We have shown previously that three Bombyx mori NPV (BmNPV) proteins, IAP2, PE38 and IE2, have E3 ligase activity using bacterially expressed recombinant proteins (Imai et al., 2003). In addition, transient expression studies have suggested that IE2 is actively degraded by the proteasome, which depends mainly on its own E3 activity G 2011 SGM Printed in Great Britain 699
2 S. Katsuma and others (Imai et al., 2005). To date, however, the roles of v-ubi and viral E3 ligases in BmNPV infection are not well understood. In this study, we examined whether the ubiquitin proteasome system is required for BmNPV infection using a specific proteasome inhibitor and a v-ubi-defective BmNPV. RESULTS Effects of MG-132 on BmNPV infection To examine whether the ubiquitin proteasome system is involved in BmNPV propagation, we examined the effects of the specific proteasome inhibitor MG-132 on BV and OB production. We first assessed the cell viability of MG- 132-treated BmN-4 (BmN) cells to test whether the effects of MG-132 were caused by cytotoxicity. Mock- and BmNPV-infected BmN cells were treated with 5 mm MG- 132, and cellular metabolic activity was measured by cleavage of the tetrazolium salt WST-1. As shown in Supplementary Fig. S1(a) (available in JGV Online), MG- 132 treatment did not exhibit a negative effect on cell survival of BmN cells. Also, no decrease in cell viability compared with DMSO-treated cells was observed during BmNPV infection (Supplementary Fig. S1b). These results indicated that MG-132 treatment at a concentration of 5 mm was not associated with cytotoxicity of BmN cells. Next, we assessed the effects of MG-132 on BV production. As shown in Fig. 1(a), treatment with MG-132 (5 mm) significantly reduced BV production from 24 h postinfection (p.i.), and a tenfold reduction was observed at 72 h p.i. in MG-132-treated cells compared with in DMSOtreated cells. We also observed that treatment with MG-132 (5 mm) completely inhibited OB production at 72 h p.i. in BmNPV-infected BmN cells, whereas a large number of OBs were observed in cells treated with DMSO (control treatment) (Fig. 1b). Western blot analysis indicated that MG-132 markedly inhibited polyhedrin protein expression, suggesting that the reduced OB production by MG-132 was due to the inhibition of polyhedrin synthesis (Fig. 1c). Taken together, these results suggested that the ubiquitin proteasome system is required for efficient production of BVs and OBs during BmNPV infection. Effects of MG-132 on expression of viral gene products and accumulation of ubiquitylated proteins in BmNPV-infected cells We next examined the effects of MG-132 on the expression of BmNPV gene products. Western blot analysis showed that expression of an early gene product, DNA-binding protein (DBP), a late gene product, viral chitinase (v- CHIA), and a very late gene product, polyhedrin, was delayed and/or reduced in MG-132-treated cells compared with in DMSO-treated cells (Fig. 1c). These results indicated that inhibition of the ubiquitin proteasome system results in reduced virus production, coinciding with delayed and/or dysregulated expression of viral gene products. Next, we examined ubiquitylation of proteins expressed in BmNPV-infected cells by Western blotting with antiubiquitin mab. As shown in Fig. 1(c), the ubiquitylated proteins, especially high-molecular-mass proteins, disappeared abruptly from 48 h p.i. in BmNPV-infected cells, whereas treatment with MG-132 resulted in a gradual increase in the accumulation of ubiquitylated proteins throughout the infection. We also observed that expression of the housekeeping protein actin disappeared almost completely from 48 h p.i. in BmNPV-infected DMSOtreated cells, whereas its expression was still observed in MG-132-treated cells, even at 72 h p.i. This suggested that MG-132 treatment delays a global shut-off of host protein synthesis by BmNPV infection. Collectively, these results strongly suggested that the ubiquitin proteasome system is required for efficient BmNPV infection. Effects of v-ubi deletion on virus production in BmN cells To determine further the role of ubiquitin signalling in BmNPV infection, we deleted the v-ubi gene from the BmNPV genome by homologous recombination and successfully isolated a v-ubi-deficient virus, BmUbiD (see Supplementary Fig. S2a, available in JGV Online). Western blot analysis showed that v-ubi deletion did not affect polyhedrin and actin expression (Fig. 2a). Furthermore, we examined the effect of v-ubi deletion on BV production. BmN cells were infected with the wild-type BmNPV T3 isolate or BmUbiD, with or without MG-132 treatment (1 or 5 mm), and yields of BV were determined by plaque assay. The titre of BmUbiD at 72 h p.i. was comparable to that of T3 regardless of treatment or not with MG-132 (Fig. 2b). These results suggested that deletion of v-ubi did not affect BV and OB production in BmNPV-infected BmN cells. To identify more specifically the stage of BmNPV infection targeted by MG-132, we examined BV production or polyhedrin expression after applying the inhibitor at different time points p.i. (0 12 h p.i.). As shown in Fig. 3(a), MG-132 significantly reduced BV production in BmN cells infected with T3 or BmUbiD at all time points tested. In contrast, we observed that suppression of polyhedrin expression by MG-132 was significantly reduced when applied after 6 h p.i. (Fig. 3b). These results suggested that degradation of viral and/or host proteins by the ubiquitin proteasome system at an early stage of BmNPV infection (0 6 h p.i.) is essential for efficient polyhedrin expression. v-ubi is not required for the ubiquitin proteasome-dependent degradation of IE2 In a previous study using confocal microscopy, we observed that MG-132 treatment resulted in IE2 protein accumulation (Imai et al., 2005), suggesting that IE2 is 700 Journal of General Virology 92
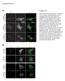
3 Ubiquitin proteasome system in BmNPV-infected cells Fig. 1. Effect of MG-132 on BmNPV infection in BmN cells. (a) BV production. BV titres in BmNPV-infected BmN cells treated with DMSO or MG-132 (5 mm) at 0 h p.i. were measured by plaque assay. (b) Light microscopy observations of mock- or BmNPVinfected BmN cells at 72 h p.i. after treatment at 0 h p.i. with DMSO or MG-132 (5 mm). Bar, 10 mm. (c) Expression of viral gene products and accumulation of ubiquitylated proteins. BmNPV-infected BmN cells were treated with DMSO or MG-132 (5 mm) at 0 h p.i., lysed and immunoblotted with antibodies against ubiquitin (Ub), DNA-binding protein (DBP), viral chitinase (v-chia), polyhedrin (POLH) or actin. actively degraded by the ubiquitin proteasome system. Also, transient expression experiments have shown that the degradation process is seemingly regulated by its own E3 ligase activity (Imai et al., 2005). These results indicate that IE2 is being auto-ubiquitylated and degraded during BmNPV infection. To verify this, we generated a mutant virus, BmIE2CS, expressing an E3 ligase activity-deficient IE2 derivative (IE2C243S; Imai et al., 2003, 2005) (Supplementary Fig. S2b) and examined IE2 expression during BmNPV infection. As reported previously (Imai et al., 2005), we observed that IE2 formed several foci in the nucleus at 2 h p.i. and then gradually diminished after 4 h p.i. in T3-infected BmN cells (see Supplementary Fig. S3, available in JGV Online). As expected, IE2 foci in BmIE2CS-infected cells were brighter compared with those in T3-infected cells. In addition, foci in BmIE2CS-infected cells were clearly detected as late as 12 h p.i., although those in T3-infected cells were rarely detected at this stage (Supplementary Fig. S3). However, it should be noted that the foci in BmIE2CS-infected cells also diminished gradually after 4 h p.i., suggesting that IE2 could be degraded in the absence of its E3 ligase activity. Western blot analysis clearly showed that treatment with MG-132 did not inhibit degradation of IE2CS, indicating that an alternative degradation mechanism is apparently responsible for IE2CS degradation (Fig. 4a). Next, we examined whether v-ubi is involved in IE2 degradation in BmNPV-infected BmN cells. BmN cells were infected with T3, BmUbiD, BmIE2CS or BmIE2D and harvested at 4 or 12 h p.i. As shown in Fig. 4(a, b), we observed that IE2 accumulation was indistinguishable between T3- and BmUbiD-infected cells with or without MG-132 treatment. In BmIE2CS-infected cells, IE2 accumulated strongly at 4 h p.i. and then decreased at 12 h p.i. (Fig. 4b). This was consistent with confocal microscopic 701
4 S. Katsuma and others Fig. 2. Effect of v-ubi deletion on BmNPV infection. (a) Polyhedrin expression. Mock-, T3- or BmUbiD-infected BmN cells were treated with DMSO or MG-132 (5 mm) at 0 h p.i., lysed at 72 h p.i. and immunoblotted with antibodies against polyhedrin (POLH) or actin. (b) BV production. BmN cells infected with T3 or BmUbiD were treated with DMSO or MG-132 (1 or 5 mm) at 0 h p.i. The titres of BV at 72 h p.i. were measured by plaque assay. Fig. 4. IE2 expression in BmN cells infected with wild-type or mutant BmNPV. (a) Effect of MG-132 on IE2 expression. BmN cells infected with T3, BmUbiD (UbiD) or BmIE2CS (IE2CS) were treated with DMSO or MG-132 (MG) at 0 h p.i., lysed at 12 h p.i. and immunoblotted with an anti-ie2 antibody. Western blots of cytoplasmic fractions with the anti-actin antibody are also shown. (b) IE2 expression in T3- or mutant BmNPV-infected cells. Nuclear fractions of BmN cells infected with T3, BmUbiD (UbiD), BmIE2CS (IE2CS) or BmIE2D (IE2D) were harvested at 4 or 12 h p.i., and subjected to Western blotting with an anti-ie2 antibody. Western blots of cytoplasmic fractions with an anti-actin antibody are also shown. observations (Supplementary Fig. S2). On the basis of these results, we concluded that v-ubi is not required for IE2 degradation in BmNPV-infected cells. Fig. 3. BV production and polyhedrin expression after application of MG-132 at different time points p.i. BV production (a) or polyhedrin (POLH) expression (b) was assessed at 72 h p.i. in BmN cells infected with T3 or BmUbiD after application of DMSO (D) or MG-132 (5 mm) at the time points indicated p.i. Polyhedrin expression was visualized by staining with Coomassie Brilliant Blue (CBB). DISCUSSION The ubiquitin proteasome system has a crucial role in the degradation of intracellular proteins and regulates a variety of cellular pathways. Using proteasome inhibitors such as MG-132, viruses belonging to several different families have been shown to utilize or modulate this system to their advantage during their infection cycles (Raaben et al., 2010; Satheshkumar et al., 2009; Teale et al., 2009). In this study, we examined whether the ubiquitin proteasome system is required for efficient BmNPV infection. First, we investigated the effects of MG-132 on OB and BV production and 702 Journal of General Virology 92
5 Ubiquitin proteasome system in BmNPV-infected cells expression of BmNPV gene products. As shown in Fig. 1(a, b), OB and BV production was significantly reduced by MG-132 treatment. It should be noted that the effect of MG-132 on OB production was more prominent than that on BV production. Western blot analysis also showed that treatment of BmNPV-infected cells with MG-132 markedly delayed or reduced expression of early, late and very late gene products of BmNPV (Fig. 1c). These results strongly suggested that the ubiquitin proteasome system plays a role during BmNPV infection. We observed that the suppression of polyhedrin expression was significantly reduced when applied at or after 6 h p.i.; however, decreased BV production was evident even when applied at 12 h p.i. (Fig. 3). This suggested that ubiquitinmediated protein degradation during the early stage of infection has different roles in expression of late and very late gene products. In addition, we observed that ubiquitylated proteins disappeared abruptly from 48 h p.i. in BmNPV-infected cells (Fig. 1c). As these conjugates accumulated in the presence of MG-132 at these time points, this suggested that the ubiquitin proteasome system is responsible for their degradation after 48 h p.i. Surprisingly, we did not observe any phenotypic defects when v-ubi was deleted from the BmNPV genome. This result is inconsistent with that from a study of a closely related AcMNPV (Reilly & Guarino, 1996). The AcMNPV mutant Vubi-FS, with a frameshift mutation in the coding region of v-ubi, produced five- to tenfold fewer BVs than the wild-type, suggesting that v-ubi plays an important role in BV production during AcMNPV infection. This group also showed that AcMNPV v-ubi may function in baculovirus replication to block the destruction of shortlived proteins by the host degradative pathway (Haas et al., 1996). Amino acid differences in some proteins have been shown to result in the phenotypic discrepancies between AcMNPV and BmNPV (Kamita & Maeda, 1997; Katou et al., 2010; Katsuma et al., 2006b; Morishima et al., 1998; Nakanishi et al., 2010). Despite the fact that the amino acid sequences of v-ubi are completely identical between these two viruses, the roles of v-ubi may be different in the infection cycle of each NPV. Host ubiquitin (H-UBI) and v-ubi are known to be structural proteins of BVs and are anchored to the inner face of the BV envelope by covalently linked phospholipids (Guarino et al., 1995). This has led to the hypothesis that BV-associated ubiquitin may be utilized for proteasomedependent degradation of viral and/or host proteins immediately after virus entry. Because IE2 is the only known baculoviral protein degraded via the ubiquitin proteasome system (Imai et al., 2005), we examined IE2 expression in BmUbiD-infected BmN cells. Western blot analysis clearly showed that IE2 accumulation was indistinguishable between T3- and BmUbiD-infected cells, suggesting that H-UBI is utilized for IE2 degradation. v-ubis are more closely related to each other than to animal ubiquitin, and most of the substitutions occur at the same amino acid residues, suggesting that v-ubi has evolved for a specific role in the baculovirus life cycle (Reilly & Guarino, 1996). Although no phenotypic abnormality of BmUbiD was observed in BmN cells, the modern lepidopteran baculovirus has seemingly acquired a v-ubi gene from an ancestral host insect to obtain evolutionary advantages. Further studies using insect larvae instead of cultured cells may help us understand the significance of v-ubi in the BmNPV life cycle. Furthermore, to comprehend the direct contribution of the viral ubiquitin system to BmNPV infection, we are currently generating a mutant BmNPV in which all of the viral ubiquitin system-related genes (e.g. v- ubi, iap2, ie2 and pe38) are inactivated. METHODS Materials. The proteasome inhibitor MG-132 was purchased from Calbiochem and dissolved in DMSO. The final concentration of DMSO in the cell culture medium was 0.1 % (v/v). The ubiquitin mab was obtained from Santa Cruz Biotechnology. Antibodies against BmNPV DBP, polyhedrin, BmChi-h (v-chia) and IE2 were as reported previously (Daimon et al., 2007; Imai et al., 2005; Okano et al., 1999; Shimada et al., 1994). Cell line and virus. The BmN-4 (BmN) cell line was maintained at 26 uc in TC-100 medium with 10 % FBS. The BmNPV T3 isolate (Maeda et al., 1985) was propagated in BmN cells. The ie2 deletion mutant BmIE2D has been reported previously (Gomi et al., 1997). Viruses were propagated in BmN cells, and BV titres were determined by plaque assay. Proteasome inhibition assay. BmN cells were infected with BmNPV at an m.o.i. of 5. After 1 h of incubation, virus-containing culture medium was removed, the cells were washed twice with TC- 100 medium, and fresh medium with MG-132 (5 mm) or DMSO (control) was added (0 h p.i.). A small amount of culture medium was collected at various time points, and BV production was determined by plaque assay. Accumulation of ubiquitylated proteins and expression of viral gene products were examined by SDS-PAGE and Western blotting. Cell viability. BmN cells were infected with BmNPV at an m.o.i. of 5. After 1 h of incubation, virus-containing culture medium was removed and fresh medium with or without MG-132 was added (0 h p.i.). We used the WST-1 assay kit (Roche Applied Science) to assess viable cell numbers as described previously (Katsuma et al., 2005). SDS-PAGE and Western blotting. SDS-PAGE and Western blotting were performed as described previously (Katsuma et al., 2007). Western blot analysis of the ubiquitylated proteins DBP and v- CHIA was carried out using anti-ubiquitin, anti-dbp and anti- BmChi-h antibodies, respectively. Polyhedrin expression was examined by SDS-PAGE as described previously (Katsuma et al., 2007). Construction of the v-ubi deletion mutant by insertion of a b- galactosidase gene cassette. To construct a plasmid for the deletion of v-ubi, a 5.4 kb fragment (nt ; GenBank accession no. L33180) was purified from the BmNPV T3 genomic clone (Maeda & Majima, 1990) and inserted into ptz19r. The plasmid was then digested with BstXI and ligated to a b-galactosidase gene cassette containing the Drosophila melanogaster heat-shock protein promoter (hsp70 lacz cassette). The resultant plasmid was co-transfected with T3 genomic DNA into BmN cells using Cellfectin 703
6 S. Katsuma and others (Invitrogen). The v-ubi-deleted BmNPV (BmUbiD) was isolated by identification of plaques expressing b-galactosidase (Katsuma et al., 2006a). Deletion of v-ubi was confirmed by PCR (see Supplementary Fig. S2a) using the primer set v-ubif (59-AAACGGAACCCGC- AGAGACG-39) and v-ubir (59-CTCCTCGTAATCGTAACACC-39). Construction of BmIE2CS expressing an E3 ligase activitydeficient IE2. A PCR fragment encoding an E3 ligase activitydeficient IE2 (IE2C243S; Imai et al., 2003) was cloned into pbluescript II SK(2) (Stratagene), and designated pbs-ie2cs. The pbs-ie2cs plasmid was co-transfected with the Bsu36I-digested BmIE2D genomic DNA into BmN cells as described previously (Katsuma et al., 2006a). Recombinants identified as white plaques were isolated by plaque assay with agarose overlays containing 400 mg X-Gal per 60 mm dish. To confirm whether the replacement of lacz with the mutated ie2 was performed correctly, the ie2 gene region of isolates with a white plaque phenotype was amplified by PCR (see Supplementary Fig. S2a) using the primer set ie2f (59-ATTCT- TCGTTTGTGACTTCG-39) and ie2r (59-AACTGCAGTTATGGA- TGTACTGCTAACC-39), and DNA sequences were determined using an ABI Prism 3100 DNA sequencer (Applied Biosystems). Immunohistochemistry and confocal microscopy. Infected cells were harvested at designated times and subjected to immunohistochemistry. Immunohistochemistry and confocal microscopy were performed as described by Okano et al. (1999). For immunohistochemistry, anti-ie2 antibody (1 : 200 dilution) and Cy5-conjugated goat anti-rat IgG (1 : 200 dilution; Jackson Immuno Research) or FITCconjugated goat anti-rat IgG (1 : 500 dilution; Cappel) were used. ACKNOWLEDGEMENTS This work was supported by the Professional Program for Agricultural Bioinformatics from MEXT, Japan grant nos and to S. K. and grant no to T. S. REFERENCES Ciechanover, A., Orian, A. & Schwartz, A. L. (2000). Ubiquitinmediated proteolysis: biological regulation via destruction. Bioessays 22, Daimon, T., Katsuma, S. & Shimada, T. (2007). Mutational analysis of active site residues of chitinase from Bombyx mori nucleopolyhedrovirus. Virus Res 124, Gomi, S., Zhou, C. E., Yih, W., Majima, K. & Maeda, S. (1997). Deletion analysis of four of eighteen late gene expression factor gene homologues of the baculovirus, BmNPV. Virology 230, Guarino, L. A., Smith, G. & Dong, W. (1995). Ubiquitin is attached to membranes of baculovirus particles by a novel type of phospholipid anchor. Cell 80, Haas, A. L., Katzung, D. J., Reback, P. M. & Guarino, L. A. (1996). Functional characterization of the ubiquitin variant encoded by the baculovirus Autographa californica. Biochemistry 35, Imai, N., Matsuda, N., Tanaka, K., Nakano, A., Matsumoto, S. & Kang, W. (2003). Ubiquitin ligase activities of Bombyx mori nucleopolyhedrovirus RING finger proteins. JVirol77, Imai, N., Matsumoto, S. & Kang, W. (2005). Formation of Bombyx mori nucleopolyhedrovirus IE2 nuclear foci is regulated by the functional domains for oligomerization and ubiquitin ligase activity. J Gen Virol 86, Kamita, S. G. & Maeda, S. (1997). Sequencing of the putative DNA helicase-encoding gene of the Bombyx mori nuclear polyhedrosis virus and fine-mapping of a region involved in host range expansion. Gene 190, Katou, Y., Yamada, H., Ikeda, M. & Kobayashi, M. (2010). A single amino acid substitution modulates low-ph-triggered membrane fusion of GP64 protein in Autographa californica and Bombyx mori nucleopolyhedroviruses. Virology 404, Katsuma, S., Hatae, N., Yano, T., Ruike, Y., Kimura, M., Hirasawa, A. & Tsujimoto, G. (2005). Free fatty acids inhibit serum deprivationinduced apoptosis through GPR120 in a murine enteroendocrine cell line STC-1. J Biol Chem 280, Katsuma, S., Horie, S., Daimon, T., Iwanaga, M. & Shimada, T. (2006a). In vivo and in vitro analyses of a Bombyx mori nucleopolyhedrovirus mutant lacking functional vfgf. Virology 355, Katsuma, S., Daimon, T., Horie, S., Kobayashi, M. & Shimada, T. (2006b). N-linked glycans of Bombyx mori nucleopolyhedrovirus fibroblast growth factor are crucial for its secretion. Biochem Biophys Res Commun 350, Katsuma, S., Mita, K. & Shimada, T. (2007). ERK- and JNKdependent signaling pathways contribute to Bombyx mori nucleopolyhedrovirus infection. J Virol 81, Katsuma, S., Kawaoka, S., Mita, K. & Shimada, T. (2008). Genomewide survey for baculoviral host homologs using the Bombyx genome sequence. Insect Biochem Mol Biol 38, Keddie, B. A., Aponte, G. W. & Volkman, L. E. (1989). The pathway of infection of Autographa californica nuclear polyhedrosis virus in an insect host. Science 243, Maeda, S. & Majima, K. (1990). Molecular cloning and physical mapping of the genome of Bombyx mori nuclear polyhedrosis virus. J Gen Virol 71, Maeda, S., Kawai, T., Obinata, M., Fujiwara, H., Horiuchi, T., Saeki, Y., Sato, Y. & Furusawa, M. (1985). Production of human a-interferon in silkworm using a baculovirus vector. Nature 315, Morishima, N., Okano, K., Shibata, T. & Maeda, S. (1998). Homologous p35 proteins of baculoviruses show distinctive antiapoptotic activities which correlate with the apoptosis-inducing activity of each virus. FEBS Lett 427, Nakanishi, T., Goto, C., Kobayashi, M., Kang, W., Suzuki, T., Dohmae, N., Matsumoto, S., Shimada, T. & Katsuma, S. (2010). Comparative studies of lepidopteran baculovirus-specific protein FP25K: development of a novel Bombyx mori nucleopolyhedrovirusbased vector with a modified fp25k gene. J Virol 84, Okano, K., Mikhailov, V. S. & Maeda, S. (1999). Colocalization of baculovirus IE-1 and two DNA-binding proteins, DBP and LEF-3, to viral replication factories. J Virol 73, Raaben, M., Posthuma, C. C., Verheije, M. H., te Lintelo, E. H., Kikkert, M., Drijfhout, J. W., Snijder, E. J., Rottier, P. J. M. & de Haan, C. A. M. (2010). The ubiquitin proteasome system plays an important role during various stages in the coronavirus infection cycle. J Virol 84, Rechsteiner, M. (1991). Natural substrates of the ubiquitin proteolytic pathway. Cell 66, Reilly, L. M. & Guarino, L. A. (1996). The viral ubiquitin gene of Autographa californica nuclear polyhedrosis virus is not essential for viral replication. Virology 218, Satheshkumar, P. S., Anton, L. C., Sanz, P. & Moss, B. (2009). Inhibition of the ubiquitin proteasome system prevents vaccinia virus DNA replication and expression of intermediate and late genes. J Virol 83, Journal of General Virology 92
7 Ubiquitin proteasome system in BmNPV-infected cells Shimada, T., Chan, I. C., Noguchi, Y., Nagata, M. & Kobayashi, M. (1994). Structural and functional abnormality of the polyhedrin gene in a polyhedron-deficient mutant of the Bombyx mori nuclear polyhedrosis virus. J Ser Sci Jpn 63, Teale, A., Campbell, S., Buuren, N. V., Magee, W. C., Watmough, K., Couturier, B., Shipclark, R. & Barry, M. (2009). Orthopoxviruses require a functional ubiquitin proteasome system for productive replication. J Virol 83,
Construction and Bioassay of Recombinant AcNPV Containing SpltNPV gp37 Fusion gene
 Construction and Bioassay of Recombinant AcNPV Containing SpltNPV gp37 Fusion gene Chongbi Li 1, *, Zhaofei Li 2, Guanghong Li 2, Yi Pang 2 1 Biopharmaceutical Engineering Center of Zhaoqing University,
Construction and Bioassay of Recombinant AcNPV Containing SpltNPV gp37 Fusion gene Chongbi Li 1, *, Zhaofei Li 2, Guanghong Li 2, Yi Pang 2 1 Biopharmaceutical Engineering Center of Zhaoqing University,
High-Frequency Homologous Recombination between Baculoviruses Involves DNA Replication
 JOURNAL OF VIROLOGY, Dec. 2003, p. 13053 13061 Vol. 77, No. 24 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.24.13053 13061.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. High-Frequency
JOURNAL OF VIROLOGY, Dec. 2003, p. 13053 13061 Vol. 77, No. 24 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.24.13053 13061.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. High-Frequency
Baculovirus Controls Host Catapillars by Manipulating Host Physiology and Behavior
 AGri-Bioscience Monographs, Vol. 5, No. 1, pp. 1 27 (2015) www.terrapub.co.jp/onlinemonographs/agbm/ Baculovirus Controls Host Catapillars by Manipulating Host Physiology and Behavior Susumu Katsuma Department
AGri-Bioscience Monographs, Vol. 5, No. 1, pp. 1 27 (2015) www.terrapub.co.jp/onlinemonographs/agbm/ Baculovirus Controls Host Catapillars by Manipulating Host Physiology and Behavior Susumu Katsuma Department
Bombyx mori Nuclear Polyhedrosis Virus and Autographa californica Nuclear Polyhedrosis Virus
 JOURNAL OF VIROLOGY, JUlY 1991, p. 3625-3632 0022-538X/91/073625-08$02.00/0 Copyright C) 1991, American Society for Microbiology Vol. 65, No. 7 Host Range Expansion by Recombination of the Baculoviruses
JOURNAL OF VIROLOGY, JUlY 1991, p. 3625-3632 0022-538X/91/073625-08$02.00/0 Copyright C) 1991, American Society for Microbiology Vol. 65, No. 7 Host Range Expansion by Recombination of the Baculoviruses
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
Oxford Expression Technologies Ltd
 Oxford Expression Technologies Ltd Founded in 2007 as a spin out from Oxford Brookes University and Natural Environment Research Council Technology based on the insect baculovirus expression vectors (BEVs)
Oxford Expression Technologies Ltd Founded in 2007 as a spin out from Oxford Brookes University and Natural Environment Research Council Technology based on the insect baculovirus expression vectors (BEVs)
NOTES. Received 27 December 2005/Accepted 25 January 2006
 JOURNAL OF VIROLOGY, Apr. 2006, p. 4168 4173 Vol. 80, No. 8 0022-538X/06/$08.00 0 doi:10.1128/jvi.80.8.4168 4173.2006 Copyright 2006, American Society for Microbiology. All Rights Reserved. NOTES Expression
JOURNAL OF VIROLOGY, Apr. 2006, p. 4168 4173 Vol. 80, No. 8 0022-538X/06/$08.00 0 doi:10.1128/jvi.80.8.4168 4173.2006 Copyright 2006, American Society for Microbiology. All Rights Reserved. NOTES Expression
NOTES. In Vivo Induction of Apoptosis Correlating with Reduced Infectivity during Baculovirus Infection. Thomas E. Clarke and Rollie J.
 JOURNAL OF VIROLOGY, Feb. 2003, p. 2227 2232 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2227 2232.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. NOTES In Vivo
JOURNAL OF VIROLOGY, Feb. 2003, p. 2227 2232 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2227 2232.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. NOTES In Vivo
Received 10 May 2004/Accepted 29 June 2004
 JOURNAL OF VIROLOGY, Nov. 2004, p. 12703 12708 Vol. 78, No. 22 0022-538X/04/$08.00 0 DOI: 10.1128/IAI.78.22.12703 12708.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Host
JOURNAL OF VIROLOGY, Nov. 2004, p. 12703 12708 Vol. 78, No. 22 0022-538X/04/$08.00 0 DOI: 10.1128/IAI.78.22.12703 12708.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Host
Nature Medicine: doi: /nm.4322
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
Identification of Mutation(s) in. Associated with Neutralization Resistance. Miah Blomquist
 Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Chapter 5. Viral infections (I)
 Chapter 5. Viral infections (I) 1. Properties of virus - Virus: derived from Latin and means poison or stench (foul odor) - Definition an infectious, potentially pathogenic nucleoprotein entity which reproduces
Chapter 5. Viral infections (I) 1. Properties of virus - Virus: derived from Latin and means poison or stench (foul odor) - Definition an infectious, potentially pathogenic nucleoprotein entity which reproduces
Bombyx mori nucleopolyhedrovirus (BmNPV) Bm64 is required for BV production and per os infection
 Chen et al. Virology Journal (2015) 12:173 DOI 10.1186/s12985-015-0399-9 RESEARCH Bombyx mori nucleopolyhedrovirus (BmNPV) Bm64 is required for BV production and per os infection Lin Chen 1,2, Yunwang
Chen et al. Virology Journal (2015) 12:173 DOI 10.1186/s12985-015-0399-9 RESEARCH Bombyx mori nucleopolyhedrovirus (BmNPV) Bm64 is required for BV production and per os infection Lin Chen 1,2, Yunwang
Large-Scale Production of Porcine Mature Interleukin-18 (IL-18) in Silkworms Using a Hybrid Baculovirus Expression System
 FULL PAPER Immunology Large-Scale Production of Porcine Mature Interleukin-18 (IL-18) in Silkworms Using a Hybrid Baculovirus Expression System Yoshihiro MUNETA 1), Hong Kun ZHAO 2), Shigeki INUMARU 1)
FULL PAPER Immunology Large-Scale Production of Porcine Mature Interleukin-18 (IL-18) in Silkworms Using a Hybrid Baculovirus Expression System Yoshihiro MUNETA 1), Hong Kun ZHAO 2), Shigeki INUMARU 1)
Sciences, China Jiliang University, Hangzhou , PR China. Downloaded from by IP:
 Journal of General Virology (212), 93, 248 2489 DOI 1.199/vir..43836- A baculovirus isolated from wild silkworm encompasses the host ranges of Bombyx mori nucleopolyhedrosis virus and Autographa californica
Journal of General Virology (212), 93, 248 2489 DOI 1.199/vir..43836- A baculovirus isolated from wild silkworm encompasses the host ranges of Bombyx mori nucleopolyhedrosis virus and Autographa californica
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Proteasomes. When Death Comes a Knock n. Warren Gallagher Chem412, Spring 2001
 Proteasomes When Death Comes a Knock n Warren Gallagher Chem412, Spring 2001 I. Introduction Introduction The central dogma Genetic information is used to make proteins. DNA RNA Proteins Proteins are the
Proteasomes When Death Comes a Knock n Warren Gallagher Chem412, Spring 2001 I. Introduction Introduction The central dogma Genetic information is used to make proteins. DNA RNA Proteins Proteins are the
1 Recombinant proteins in insect and mammal cells , F E B R U A R Y 2 3 RD
 1 Recombinant proteins in insect and mammal cells 2 0 1 8, F E B R U A R Y 2 3 RD Bacteria Yeast Insect cells Mammalian Cell growth Medium complexity Rapid (30 min) Rapid (90 min) Slow (18-24h) Slow (24h)
1 Recombinant proteins in insect and mammal cells 2 0 1 8, F E B R U A R Y 2 3 RD Bacteria Yeast Insect cells Mammalian Cell growth Medium complexity Rapid (30 min) Rapid (90 min) Slow (18-24h) Slow (24h)
Replication, Integration, and Packaging of Plasmid DNA following Cotransfection with Baculovirus Viral DNA
 JOURNAL OF VIROLOGY, July 1999, p. 5473 5480 Vol. 73, No. 7 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Replication, Integration, and Packaging of Plasmid
JOURNAL OF VIROLOGY, July 1999, p. 5473 5480 Vol. 73, No. 7 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Replication, Integration, and Packaging of Plasmid
SUPPLEMENTAL FIGURE LEGENDS
 SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
Why chaperone vectors?
 Why chaperone vectors? A protein folding initiative An open discussion with structural biologists Protein Structure Initiative: Pilot Phase Whether the pilot phase achieved its goal depends on how we measure
Why chaperone vectors? A protein folding initiative An open discussion with structural biologists Protein Structure Initiative: Pilot Phase Whether the pilot phase achieved its goal depends on how we measure
Variation in the HindlII Restriction Fragments of DNA from the Chinese Tian Tan Strain of Vaccinia Virus
 J. gen. irol. (1985), 66, 1819-1823. Printed in Great Britain 1819 Key words: vaccinia virus~vaccine~restriction Jragrnent variation ariation in the Hindl Restriction Fragments of DNA from the Chinese
J. gen. irol. (1985), 66, 1819-1823. Printed in Great Britain 1819 Key words: vaccinia virus~vaccine~restriction Jragrnent variation ariation in the Hindl Restriction Fragments of DNA from the Chinese
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Viral Genetics. BIT 220 Chapter 16
 Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer
 AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
33VASTVNGATSANNHGEPPS51PADARPR58
 Pro-rich region Trans-membrane region 214 246 359 381 UL50 1 397 211SSRTAS216PPPPPR222 NLS CR1 CR2 CR3 CR4 UL53 1 376 11RERRS15ALRS19LLRKRRR25 33VASTVNGATSANNHGEPPS51PADARPR58 FIG S1. UL97 phosphorylation
Pro-rich region Trans-membrane region 214 246 359 381 UL50 1 397 211SSRTAS216PPPPPR222 NLS CR1 CR2 CR3 CR4 UL53 1 376 11RERRS15ALRS19LLRKRRR25 33VASTVNGATSANNHGEPPS51PADARPR58 FIG S1. UL97 phosphorylation
VIROLOGY. Engineering Viral Genomes: Retrovirus Vectors
 VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
Supplements. Figure S1. B Phalloidin Alexa488
 Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Supplementary Information
 Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
This training module is required for all personnel listed on an IBC protocol that describes work utilizing viral vectors (both replication competent
 This training module is required for all personnel listed on an IBC protocol that describes work utilizing viral vectors (both replication competent and incompetent) regardless of the biosafety level used
This training module is required for all personnel listed on an IBC protocol that describes work utilizing viral vectors (both replication competent and incompetent) regardless of the biosafety level used
The ubiquitin-proteasome system. in bone biology. University of Nottingham. ECTS PhD Training July 5 th Dr Rob Layfield
 The ubiquitin-proteasome system in bone biology Dr Rob Layfield University of Nottingham ECTS PhD Training July 5 th 2009 Images reproduced from: http://www.bostonbiochem.com/overview.php?prod=ubchains
The ubiquitin-proteasome system in bone biology Dr Rob Layfield University of Nottingham ECTS PhD Training July 5 th 2009 Images reproduced from: http://www.bostonbiochem.com/overview.php?prod=ubchains
Development of a Novel Recombinant Influenza Vaccine in Insect Cells
 Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery. Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito
 Supplementary Information for: Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito Supplementary Methods Supplementary
Supplementary Information for: Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito Supplementary Methods Supplementary
Supplemental information contains 7 movies and 4 supplemental Figures
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
The baculovirus per os infectivity complex
 The baculovirus per os infectivity complex Ke Peng Thesis committee Thesis supervisor Prof. dr. J.M. VLak Personal Chair at the Laboratory of Virology Wageningen University Thesis co-supervisors Dr. M.M.
The baculovirus per os infectivity complex Ke Peng Thesis committee Thesis supervisor Prof. dr. J.M. VLak Personal Chair at the Laboratory of Virology Wageningen University Thesis co-supervisors Dr. M.M.
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Lecture 2: Virology. I. Background
 Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Herpesviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
A new method for the calculation of baculovirus titre using the Agilent 2100 bioanalyzer and the flow cytometry set.
 A new method for the calculation of baculovirus titre using the Agilent 2100 bioanalyzer and the flow cytometry set Application Note Vikash Malde Ian Hunt Ruth Finch Abstract This Application Note provides
A new method for the calculation of baculovirus titre using the Agilent 2100 bioanalyzer and the flow cytometry set Application Note Vikash Malde Ian Hunt Ruth Finch Abstract This Application Note provides
Tyrosine phosphorylation and protein degradation control the transcriptional activity of WRKY involved in benzylisoquinoline alkaloid biosynthesis
 Supplementary information Tyrosine phosphorylation and protein degradation control the transcriptional activity of WRKY involved in benzylisoquinoline alkaloid biosynthesis Yasuyuki Yamada, Fumihiko Sato
Supplementary information Tyrosine phosphorylation and protein degradation control the transcriptional activity of WRKY involved in benzylisoquinoline alkaloid biosynthesis Yasuyuki Yamada, Fumihiko Sato
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Persistence of an Occlusion-Negative Recombinant Nucleopolyhedrovirus in Trichoplusia ni Indicates High Multiplicity of Cellular Infection
 APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Nov. 2001, p. 5204 5209 Vol. 67, No. 11 0099-2240/01/$04.00 0 DOI: 10.1128/AEM.67.11.5204 5209.2001 Copyright 2001, American Society for Microbiology. All Rights
APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Nov. 2001, p. 5204 5209 Vol. 67, No. 11 0099-2240/01/$04.00 0 DOI: 10.1128/AEM.67.11.5204 5209.2001 Copyright 2001, American Society for Microbiology. All Rights
Auxin-Inducible Degron (AID) System Total Set
 Catalog No. BRS-APC011A Auxin-Inducible Degron (AID) System Total Set This AID System Charactoristics Principle of Auxin-Inducible Degron (AID ) System AID plasmid protein 1. Transfection of AID plasmid
Catalog No. BRS-APC011A Auxin-Inducible Degron (AID) System Total Set This AID System Charactoristics Principle of Auxin-Inducible Degron (AID ) System AID plasmid protein 1. Transfection of AID plasmid
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
ab E3 Ligase Auto- Ubiquitilylation Assay Kit
 ab139469 E3 Ligase Auto- Ubiquitilylation Assay Kit Instructions for Use For testing ubiquitin E3 ligase activity through assessment of their ability to undergo auto-ubiquitinylation This product is for
ab139469 E3 Ligase Auto- Ubiquitilylation Assay Kit Instructions for Use For testing ubiquitin E3 ligase activity through assessment of their ability to undergo auto-ubiquitinylation This product is for
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP
 Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
The Zombies of the Scientific Community Viruses and Other Acellular Infectious Agents. Acellular Agents
 viruses protein and nucleic acid viroids RNA virusoids RNA prions proteins The Zombies of the Scientific Community Viruses and Other Acellular Infectious Agents Acellular Agents Viruses major cause of
viruses protein and nucleic acid viroids RNA virusoids RNA prions proteins The Zombies of the Scientific Community Viruses and Other Acellular Infectious Agents Acellular Agents Viruses major cause of
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
CELL BIOLOGY - CLUTCH CH THE IMMUNE SYSTEM.
 !! www.clutchprep.com CONCEPT: OVERVIEW OF HOST DEFENSES The human body contains three lines of against infectious agents (pathogens) 1. Mechanical and chemical boundaries (part of the innate immune system)
!! www.clutchprep.com CONCEPT: OVERVIEW OF HOST DEFENSES The human body contains three lines of against infectious agents (pathogens) 1. Mechanical and chemical boundaries (part of the innate immune system)
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Patricia Fitzgerald-Bocarsly
 FLU Patricia Fitzgerald-Bocarsly October 23, 2008 Orthomyxoviruses Orthomyxo virus (ortho = true or correct ) Negative-sense RNA virus (complementary to mrna) Five different genera Influenza A, B, C Thogotovirus
FLU Patricia Fitzgerald-Bocarsly October 23, 2008 Orthomyxoviruses Orthomyxo virus (ortho = true or correct ) Negative-sense RNA virus (complementary to mrna) Five different genera Influenza A, B, C Thogotovirus
Identification of a hydrophibic domain of HA2 essential to ACCEPTED
 JVI Accepts, published online ahead of print on 30 January 2008 J. Virol. doi:10.1128/jvi.02319-07 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JVI Accepts, published online ahead of print on 30 January 2008 J. Virol. doi:10.1128/jvi.02319-07 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Viral structure م.م رنا مشعل
 Viral structure م.م رنا مشعل Viruses must reproduce (replicate) within cells, because they cannot generate energy or synthesize proteins. Because they can reproduce only within cells, viruses are obligate
Viral structure م.م رنا مشعل Viruses must reproduce (replicate) within cells, because they cannot generate energy or synthesize proteins. Because they can reproduce only within cells, viruses are obligate
Ubiquitin Ligase Activities of Bombyx mori Nucleopolyhedrovirus RING Finger Proteins
 JOURNAL OF VIROLOGY, Jan. 2003, p. 923 930 Vol. 77, No. 2 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.2.923 930.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ubiquitin Ligase
JOURNAL OF VIROLOGY, Jan. 2003, p. 923 930 Vol. 77, No. 2 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.2.923 930.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ubiquitin Ligase
ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2
 ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Conservation of the D domain throughout evolution. Alignment of TRF2 sequences
ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Conservation of the D domain throughout evolution. Alignment of TRF2 sequences
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Conditional and reversible disruption of essential herpesvirus protein functions
 nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
Enhancing the baculovirus expression system with VANKYRIN technology. Kendra Steele, Ph.D.
 Enhancing the baculovirus expression system with VANKYRIN technology Kendra Steele, Ph.D. Insect cells Can express mammalian proteins Similarities with mammalian cells Eukaryotic systems How protein are
Enhancing the baculovirus expression system with VANKYRIN technology Kendra Steele, Ph.D. Insect cells Can express mammalian proteins Similarities with mammalian cells Eukaryotic systems How protein are
Exon 3 of the Human Cytomegalovirus Major Immediate-Early Region Is Required for Efficient Viral Gene Expression and for Cellular Cyclin Modulation
 JOURNAL OF VIROLOGY, June 2005, p. 7438 7452 Vol. 79, No. 12 0022-538X/05/$08.00 0 doi:10.1128/jvi.79.12.7438 7452.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Exon 3 of
JOURNAL OF VIROLOGY, June 2005, p. 7438 7452 Vol. 79, No. 12 0022-538X/05/$08.00 0 doi:10.1128/jvi.79.12.7438 7452.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Exon 3 of
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy
 Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
LESSON 1.4 WORKBOOK. Viral sizes and structures. Workbook Lesson 1.4
 Eukaryotes organisms that contain a membrane bound nucleus and organelles. Prokaryotes organisms that lack a nucleus or other membrane-bound organelles. Viruses small, non-cellular (lacking a cell), infectious
Eukaryotes organisms that contain a membrane bound nucleus and organelles. Prokaryotes organisms that lack a nucleus or other membrane-bound organelles. Viruses small, non-cellular (lacking a cell), infectious
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles
 Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles Amin Feizpour Reinhard Lab Department of Chemistry and the Photonics Center, Boston University, Boston, MA May 2014
Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles Amin Feizpour Reinhard Lab Department of Chemistry and the Photonics Center, Boston University, Boston, MA May 2014
OPEN. Zhong-Jian Guo Xu-Dong Tang 3. , Meng-Han Yu 1, Xian-Yun Dong 1, Wei-Li Wang 2, Ting Tian 1, Xian-Yin Yu 1 &
 www.nature.com/scientificreports Received: 6 March 2017 Accepted: 14 July 2017 Published: xx xx xxxx OPEN Protein composition analysis of polyhedra matrix of Bombyx mori nucleopolyhedrovirus (BmNPV) showed
www.nature.com/scientificreports Received: 6 March 2017 Accepted: 14 July 2017 Published: xx xx xxxx OPEN Protein composition analysis of polyhedra matrix of Bombyx mori nucleopolyhedrovirus (BmNPV) showed
Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts
 Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts (TIG-3 cells) were rendered senescent by either serial passage
Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts (TIG-3 cells) were rendered senescent by either serial passage
number Done by Corrected by Doctor Ashraf
 number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
Supplementary Material
 Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
The Immune System. These are classified as the Innate and Adaptive Immune Responses. Innate Immunity
 The Immune System Biological mechanisms that defend an organism must be 1. triggered by a stimulus upon injury or pathogen attack 2. able to counteract the injury or invasion 3. able to recognise foreign
The Immune System Biological mechanisms that defend an organism must be 1. triggered by a stimulus upon injury or pathogen attack 2. able to counteract the injury or invasion 3. able to recognise foreign
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
293T cells were transfected with indicated expression vectors and the whole-cell extracts were subjected
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
Redacted for Privacy
 AN ABSTRACT OF THE THESIS OF Mary B. Bradford for the degree of Master of Science in Biochemistry/Biophysics presented on June 8. 1990 TITLE: Characterization of the Infection Cycle of the Orgyia pseudotsugata
AN ABSTRACT OF THE THESIS OF Mary B. Bradford for the degree of Master of Science in Biochemistry/Biophysics presented on June 8. 1990 TITLE: Characterization of the Infection Cycle of the Orgyia pseudotsugata
Supplementary Figures
 Supplementary Figures a miel1-2 (SALK_41369).1kb miel1-1 (SALK_978) b TUB MIEL1 Supplementary Figure 1. MIEL1 expression in miel1 mutant and S:MIEL1-MYC transgenic plants. (a) Mapping of the T-DNA insertion
Supplementary Figures a miel1-2 (SALK_41369).1kb miel1-1 (SALK_978) b TUB MIEL1 Supplementary Figure 1. MIEL1 expression in miel1 mutant and S:MIEL1-MYC transgenic plants. (a) Mapping of the T-DNA insertion
Supplementary materials
 Supplementary materials Chemical library from ChemBridge 50,240 structurally diverse small molecule compounds dissolved in DMSO Hits Controls: No virus added μ Primary screening at 20 g/ml of compounds
Supplementary materials Chemical library from ChemBridge 50,240 structurally diverse small molecule compounds dissolved in DMSO Hits Controls: No virus added μ Primary screening at 20 g/ml of compounds
Cbl ubiquitin ligase: Lord of the RINGs
 Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for
 Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
LESSON 4.4 WORKBOOK. How viruses make us sick: Viral Replication
 DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a Case Study
 Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Nature Immunology doi: /ni.3268
 Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Overview of virus life cycle
 Overview of virus life cycle cell recognition and internalization release from cells progeny virus assembly membrane breaching nucleus capsid disassembly and genome release replication and translation
Overview of virus life cycle cell recognition and internalization release from cells progeny virus assembly membrane breaching nucleus capsid disassembly and genome release replication and translation
LESSON 4.6 WORKBOOK. Designing an antiviral drug The challenge of HIV
 LESSON 4.6 WORKBOOK Designing an antiviral drug The challenge of HIV In the last two lessons we discussed the how the viral life cycle causes host cell damage. But is there anything we can do to prevent
LESSON 4.6 WORKBOOK Designing an antiviral drug The challenge of HIV In the last two lessons we discussed the how the viral life cycle causes host cell damage. But is there anything we can do to prevent
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
USING dsrna TO PREVENT IN VITRO AND IN VIVO VIRAL INFECTIONS BY RECOMBINANT BACULOVIRUS
 JBC Papers in Press. Published on March 19, 2003 as Manuscript M212039200 USING dsrna TO PREVENT IN VITRO AND IN VIVO VIRAL INFECTIONS BY RECOMBINANT BACULOVIRUS Victor Julian Valdes +, Alicia Sampieri
JBC Papers in Press. Published on March 19, 2003 as Manuscript M212039200 USING dsrna TO PREVENT IN VITRO AND IN VIVO VIRAL INFECTIONS BY RECOMBINANT BACULOVIRUS Victor Julian Valdes +, Alicia Sampieri
Study of different types of ubiquitination
 Study of different types of ubiquitination Rudi Beyaert (rudi.beyaert@irc.vib-ugent.be) VIB UGent Center for Inflammation Research Ghent, Belgium VIB Training Novel Proteomics Tools: Identifying PTMs October
Study of different types of ubiquitination Rudi Beyaert (rudi.beyaert@irc.vib-ugent.be) VIB UGent Center for Inflammation Research Ghent, Belgium VIB Training Novel Proteomics Tools: Identifying PTMs October
Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication
 Manuscript EMBO-2010-74756 Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication Tsai-Ling Liao, Chung-Yi Wu, Wen-Chi Su, King-Song Jeng and Michael Lai Corresponding
Manuscript EMBO-2010-74756 Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication Tsai-Ling Liao, Chung-Yi Wu, Wen-Chi Su, King-Song Jeng and Michael Lai Corresponding
