Getting to the Root of mirna-mediated Gene Silencing
|
|
|
- Leslie Gilmore
- 5 years ago
- Views:
Transcription
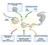
1 Leading Edge Essay Getting to the Root of mirna-mediated Gene Silencing Ana Eulalio, 1 Eric Huntzinger, 1 and Elisa Izaurralde 1, * 1 Max-Planck-Institute for Developmental Biology, Spemannstrasse 35, D Tübingen, Germany *Correspondence: elisa.izaurralde@tuebingen.mpg.de DOI /j.cell MicroRNAs are ~22 nucleotide-long RNAs that silence gene expression posttranscriptionally by binding to the 3 untranslated regions of target mrnas. Although much is known about their biogenesis and biological functions, the mechanisms allowing mirnas to silence gene expression in animal cells are still under debate. Here, we discuss current models for mirna-mediated gene silencing and formulate a hypothesis to reconcile differences. MicroRNAs (mirnas) play important roles in a broad range of biological processes including development, cellular differentiation, proliferation, and apoptosis. Emerging evidence also implicates mirnas in the pathogenesis of human diseases such as cancer and metabolic disorders. At least 100 mirna genes have been identified in invertebrates, and in vertebrates and plants. Computational predictions of mirna targets estimate that each mirna regulates hundreds of different mrnas, suggesting that a large proportion of the transcriptome is subject to mirna regulation (reviewed by Bushati and Cohen, 2007). To perform their regulatory functions mirnas assemble together with Argonaute family proteins into mirnainduced silencing complexes (miriscs). Within these complexes, mirnas guide Argonaute proteins to fully or partially complementary mrna targets, which are then silenced posttranscriptionally (reviewed by Bushati and Cohen, 2007). Despite remarkable progress in our understanding of mirna biogenesis and function, the mechanisms used by mirnas to regulate gene expression remain under debate. Indeed, published studies indicate that mirnas repress protein expression in four distinct ways: (1) cotranslational protein degradation; (2) inhibition of translation elongation; (3) premature termination of translation (ribosome drop-off); and (4) inhibition of translation initiation (Figure 1). In addition, animal mirnas can induce significant degradation of mrna targets despite imperfect mrna-mirna base-pairing (Figure 1). MicroRNAs might also silence their targets by sequestering mrnas in discrete cytoplasmic foci known as mrna processing bodies or P bodies, which exclude the translation machinery. Here, we discuss evidence supporting these different mechanisms of repression by mirnas and the discrepancies between them. Post-initiation Mechanisms Early studies in the worm Caenorhabditis elegans and recent studies in mammalian cell cultures present persuasive evidence that mirnas repress protein synthesis after translation is initiated (Seggerson et al., 2002; Maroney et al., 2006; Nottrott et al., 2006; Petersen et al., 2006). Although these studies differ in the details, their conclusions stem from a common observation: in sucrose sedimentation gradients, mirnas and their targets are associated with polysomes. These polysomes were shown to be actively translating mrna targets because they were sensitive to a variety of conditions that inhibit translation. For example, they dissociate into monosomes or ribosomal subunits following brief incubation with translation inhibitors, such as hippuristanol, puromycin, or pactamycin (Maroney et al., 2006; Nottrott et al., 2006; Petersen et al., 2006). The paradoxical observation that the targets of mirnas appear to be actively translated while the corresponding protein product remains undetectable prompted the proposal that the nascent polypeptide chain might be degraded cotranslationally (Figure 1B; Nottrott et al., 2006). This proposal is, however, based on negative rather than direct positive evidence. For example, the identity of the putative protease remains unknown; and the proteasome was excluded as a possibility because proteasome inhibitors do not restore protein expression from silenced reporters (Nottrott et al., 2006). To investigate how mirnas silence their targets, Petersen et al. (2006) designed a synthetic mirna reporter containing a 3 untranslated region (UTR) with six identical sites partially complementary to a transfected sirna (that is, an mirna mimic). When this reporter was transiently expressed it associated with polysomes, although its expression was repressed by the sirna. But if translation initiation was inhibited, then these ribosomes dissociated more rapidly than those associated with a control (unrepressed) mrna. This led to the suggestion that mirnas cause premature ribosome dissociation or ribosome drop-off (Figure 1A). There is additional evidence that mirnas mediate repression after translation initiation: silencing occurs even when reporter mrna translation is initiated by a 5 UTR containing an internal ribosome entry site (IRES). Because IRESs initiate translation of mrnas independently of the mrna cap structure, these results indicate that mirnas repress translation at a step downstream of cap recognition (Petersen et al., 2006). Inhibition of Translation Initiation In contrast to these studies, Pillai et al. (2005) have shown that mirnas and their targets are not associated with the Cell 132, January 11, Elsevier Inc. 9
2 Figure 1. Mechanisms of mirna-mediated Gene Silencing (A) Postinitiation mechanisms. MicroRNAs (mirnas; red) repress translation of target mrnas by blocking translation elongation or by promoting premature dissociation of ribosomes (ribosome drop-off). (B) Cotranslational protein degradation. This model proposes that translation is not inhibited, but rather the nascent polypeptide chain is degraded cotranslationally. The putative protease is unknown. (C E) Initiation mechanisms. MicroRNAs interfere with a very early step of translation, prior to elongation. (C) Argonaute proteins compete with eif4e for binding to the cap structure (cyan dot). (D) Argonaute proteins recruit eif6, which prevents the large ribosomal subunit from joining the small subunit. (E) Argonaute proteins prevent the formation of the closed loop mrna configuration by an ill-defined mechanism that includes deadenylation. (F) MicroRNA-mediated mrna decay. MicroRNAs trigger deadenylation and subsequent decapping of the mrna target. Proteins required for this process are shown including components of the major deadenylase complex (CAF1, CCR4, and the NOT complex), the decapping enzyme DCP2, and several decapping activators (dark blue circles). (Note that mrna decay could be an independent mechanism of silencing, or a consequence of translational repression, irrespective of whether repression occurs at the initiation or postinitiation levels of translation.) RISC is shown as a minimal complex including an Argonaute protein (yellow) and GW182 (green). The mrna is represented in a closed loop configuration achieved through interactions between the cytoplasmic poly(a) binding protein (PABPC1; bound to the 3 poly(a) tail) and eif4g (bound to the cytoplasmic cap-binding protein eif4e). polysomal fraction in sucrose gradients but rather with the free mrnp pool in mammalian cells, indicating that translation inhibition occurs at the initiation step. Furthermore, in this and other studies, mrnas translated through cap-independent mechanisms (that is, through an IRES) were refractory to repression by mirnas, further supporting the notion that mirnas inhibit capdependent translation initiation (Humphreys et al., 2005; Pillai et al., 2005; Mathonnet et al., 2007; Wakiyama et al., 2007). In agreement with a role for mirnas in blocking translation initiation, Kiriakidou et al. (2007) reported an unexpected observation: the central domain of Argonaute proteins exhibits sequence similarities to the cytoplasmic cap-binding protein eif4e (eukaryotic translation initiation factor 4E), which is essential for cap-dependent translation initiation. eif4e binds to the m 7 Gppp-cap structure of mrnas by stacking the methylated base of the cap between two tryptophans. At the equivalent position of the tryptophans in eif4e, Argonaute proteins have phenylalanines that could mediate a similar interaction. Consistently, Kiriakidou et al. (2007) showed that human Argonaute 2 (AGO2) binds to m 7 GTP on Sepharose beads, and that there is competition for this binding by a methylated cap analog (e.g., m 7 GpppG) but not by unmethylated GpppG. The authors then showed that substituting one or both AGO2 phenylalanines with valine residues abrogated the silencing activity. These results support the idea that mirnas inhibit translation at the cap-recognition step by displacing eif4e from the cap structure (Figure 1C). Evidence suggesting that mirnas inhibit an early translation step (before elongation) was also reported by Chendrimada et al. (2007). Using human cells they showed that AGO2 associates with both eif6 and large ribosomal subunits. By binding to the large ribosomal subunit, eif6 prevents this subunit from prematurely joining with the small ribosomal subunit. Thus, if AGO2 recruits eif6, then the large and small ribosomal subunits might not be able to associate, causing translation to be repressed (Figure 1D; Chendrimada et al., 2007). Although both Kiriakidou et al. (2007) and Chendrimada et al. (2007) present results indicating that mirnas repress translation prior to the elongation step, they propose mutually exclusive underlying mechanisms. Clearly, we need to investigate more fully how eif6 and Argonaute s putative cap-binding domain contribute to mrna silencing. In particular, without further structural information on eukaryotic Argonaute 10 Cell 132, January 11, Elsevier Inc.
3 proteins, another viable interpretation of Kiriakidou s results is that mutating the phenylalanine residues affects the protein activity through an unrelated mechanism. Similarly, eif6 is required for 60S ribosomal subunit biogenesis and its depletion may have secondary effects that we do not yet fully appreciate. mirna-mediated mrna Decay Initial studies reported that animal mir- NAs repress translation without significantly affecting the abundance of target mrnas. More recently, however, several reports showed that animal mir- NAs do induce significant degradation of target mrnas (Bagga et al., 2005; Wu and Belasco, 2005; Behm-Ansmant et al., 2006; Giraldez et al., 2006; Wu et al., 2006; Chendrimada et al., 2007; Eulalio et al., 2007a). In agreement with this, the levels of predicted and validated mirna targets increase in cells in which the mirna pathway is inhibited, for example, by depletion of Dicer or Argonaute proteins (Giraldez et al., 2006; Rehwinkel et al., 2006; Schmitter et al., 2006). Conversely, if specific mirnas are ectopically expressed, then transcripts containing binding sites for those mirnas become less abundant (Lim et al., 2005). Likewise, several examples show that expressing a given mirna correlates with downregulation of transcripts containing complementary binding sites. For example, in zebrafish embryos at the onset of zygotic transcription, the dramatic increase of mir-430 expression correlates with the degradation of a large number of maternal mrnas containing mir-430 binding sites in their 3 UTRs (Giraldez et al., 2006). In animal cells, mirnas cause decay of mrnas not through endonucleolytic cleavage by Argonaute proteins but rather by directing mrnas to the general mrna degradation machinery (except when the mirna is fully complementary to the target). This observation is supported by studies in zebrafish embryos, C. elegans, D. melanogaster, and human cells showing that mirnas accelerate deadenylation and decapping of their targets (Behm-Ansmant et al., 2006; Giraldez et al., 2006; Wu et al., 2006; Eulalio et al., 2007a). Messenger RNA decay mediated by micrornas requires Argonaute proteins, the P body component GW182, the CAF1-CCR4-NOT deadenylase complex, the decapping enzyme DCP2, and several decapping activators including DCP1, Ge-1, EDC3, and RCK/p54 (Figure 1F; Behm-Ansmant et al., 2006; Eulalio et al., 2007a). Current evidence indicates that GW182 is recruited to mirna targets through direct interactions with the Argonaute proteins, contributing to translational repression (reviewed by Ding and Han, 2007; Eulalio et al., 2007b). GW182 also marks the transcript as a target for decay via deadenylation and decapping (reviewed by Ding and Han, 2007; Eulalio et al., 2007b). Although compelling evidence shows that mirnas trigger degradation of their targets, a critical question remains open: Is degradation an independent mechanism by which silencing is accomplished, or is it a consequence of a primary effect on translation? Some evidence suggests that mirna-mediated mrna decay can be uncoupled from translation. In human cells, an mirna target whose translation was inhibited by inserting a strong stem loop structure in its 5 UTR was nevertheless deadenylated in an mirna-dependent manner (Wu et al., 2006). Likewise, in zebrafish embryos and human cell extracts, mirna targets were deadenylated despite having a defective cap structure (Appp-cap) that impairs translation (Mishima et al., 2006; Wakiyama et al., 2007). Furthermore, in D. melanogaster cells and human cell extracts, mirna-mediated mrna decay could occur in the absence of active translation (Eulalio et al., 2007a; Wakiyama et al., 2007). The extent of degradation is clearly specified by the mrna target and not by the mirna itself because the same mirna can either repress translation or induce mrna decay in a target-specific manner (Eulalio et al., 2007a). Alemán et al. (2007) found that whether mirna mimics elicit decay or translational repression depends on the structure of the mirna-mrna duplexes (that is, the number, type, and position of mismatches). Grimson et al. (2007) found that the number of mirna binding sites, the distance separating these sites, their position within the 3 UTR, and the RNA context strongly influence the magnitude of the regulation, although the relative contributions of translational repression and decay in each case were not established. Thus, whether or not mirnas elicit mrna degradation strongly depends on specific features of the mirna-binding site and its RNA context and so, most likely, on the specific complement of proteins associated with a given target. Sequestration in P Bodies Argonaute proteins, mirnas, and mirna targets colocalize to cytoplasmic foci known as P bodies. Additional components of P bodies include GW182, the CAF1-CCR4-NOT deadenylase complex, the decapping enzyme DCP2, decapping activators (e.g., DCP1, EDC3, Ge-1), and the RNA helicase RCK/p54, all of which have been implicated in mirna function (reviewed by Eulalio et al., 2007b; Parker and Sheth, 2007). The detection of Argonaute proteins, mirnas, and mirna targets in P bodies led to a model in which mirna targets get sequestered in P bodies, where they are shielded from the translation machinery and may undergo decay (Eulalio et al., 2007b; Parker and Sheth, 2007). It has been debated whether the localization to P bodies is a cause or a consequence of silencing. However, recent studies demonstrate that the mirna pathway remains unaffected in cells lacking detectable microscopic P bodies (Chu and Rana, 2006; Eulalio et al., 2007c). Thus, although P body components play crucial roles in mirna-mediated silencing, aggregation into microscopic P bodies is not required for mirna function. These results imply that silencing is initiated in the soluble cytoplasmic fraction, and that the localization of the silencing machinery in P bodies is a consequence rather than a cause of silencing (Humphreys et al., 2005; Pillai et al., 2005; Chu and Rana, 2006; Eulalio et al., 2007c). Insights from In Vitro Studies The inhibition of translation initiation by mirnas has been recapitulated in vitro, in cell-free extracts of diverse origin. These include rabbit reticulocyte lysates (Wang et al., 2006), D. melanogaster embryo extracts (Thermann and Hentze, 2007), and extracts from two Cell 132, January 11, Elsevier Inc. 11
4 different mammalian cell lines, mouse Krebs-2 ascites, and human HEK293F cells (Mathonnet et al., 2007; Wakiyama et al., 2007). In these extracts, mirnas silenced translation of m 7 Gppp-capped mrnas but not of transcripts carrying an artificial Appp-cap structure (irrespective of the poly(a) tail). In mouse and human cell extracts, transcripts initiating translation in an IRES-dependent manner (thus cap-independent) were refractory to mirna regulation (Mathonnet et al., 2007; Wakiyama et al., 2007). Few studies have investigated the poly(a) tail s role in silencing; but when this issue was addressed explicitly, nonpolyadenylated mrnas were either silenced in vivo (Pillai et al., 2005; Wu et al., 2006) or were partially or fully refractory to silencing in vivo and in vitro, respectively, regardless of the cap structure (Humphreys et al., 2005; Wang et al., 2006; Wakiyama et al., 2007). Wakiyama et al. (2007) observed a strict requirement for both the cap structure and the poly(a) tail for silencing. Moreover, their extracts recapitulated mirna-mediated deadenylation as previously observed in zebrafish embryos and in human and D. melanogaster cells (Behm-Ansmant et al., 2006; Giraldez et al., 2006; Wu et al., 2006). Based on these observations, Wakiyama et al. (2007) proposed that mirnas trigger deadenylation; consequently in this model, translation is repressed because the cap structure and the poly(a) tail cannot synergize (Figure 1E). It is well established that if the cytoplasmic poly(a)-binding protein (PABPC1) is bound to an mrna poly(a) tail, it can interact with translation initiation factor 4G (eif4g); eif4g is bound to the cap structure through interactions with eif4e. This interaction induces mrna to form a closed loop (Figure 1B), which greatly enhances translation (see Wakiyama et al., 2007 and references therein). Silenced mrnas will not form a closed loop because they are deadenylated. Notably, there is also debate about whether deadenylation is the cause or the consequence of silencing. Wakiyama et al. (2007) found that all mrnas containing mirna-binding sites were deadenylated in an mirna-dependent manner, including those refractory to silencing because of the presence of an Appp-cap or an IRES in their 5 UTR; this finding is in agreement with results reported in zebrafish embryos (Mishima et al., 2006). Deadenylation also occurred in the presence of cycloheximide, suggesting that it does not require ongoing translation. These results were interpreted as evidence that deadenylation causes the translational repression (Wakiyama et al., 2007). Other studies suggest that beyond deadenylation, additional mechanisms cause translational repression. For example, depleting an essential component of the deadenylase complex (that is, NOT1) in D. melanogaster cells prevented mirnamediated mrna decay; however, protein expression was not fully restored indicating that the reporters remained silenced at the translational level (Behm-Ansmant et al., 2006). Similarly, Wu et al. (2006) showed that, in human cells, a reporter mrna in which the poly(a) tail was replaced by a histone-stem loop structure was nevertheless repressed by an mirna, demonstrating that translational repression is not caused by deadenylation. A striking finding has been reported by Mathonnet et al. (2007): adding purified initiation complex eif4f (which includes the cytoplasmic cap-binding protein eif4e, the scaffolding protein eif4g, and the RNA helicase eif4a) counteracted silencing. This fits nicely with the idea that the Argonaute proteins compete with eif4e for binding to the cap structure as proposed by Kiriakidou et al. (2007). The displacement of eif4e would prevent the circularization of the mrna and consequently enhance deadenylation. In summary, although the in vitro studies differ in the details, they all point to a role for mirnas in interfering with early steps of translation initiation. Why So Many Mechanisms? It is hard to reconcile the different reported modes of mirna regulation of gene expression. Perhaps these different modes reflect different interpretations and experimental approaches. Another possibility is that mirnas do indeed silence gene expression via multiple mechanisms. Finally, mirnas might silence gene expression by a common and unique mechanism; and the multiple modes of action represent secondary effects of this primary event. Are We Measuring What Matters in Silencing? In principle, it should be easy to discriminate the post-initiation mode from the initiation mode of mirna-mediated repression by analyzing the position of the silenced mrna reporter in sucrose sedimentation gradients. In the first case, the reporter is expected to cofractionate with polysomes, whereas in the second case it should be present in the free RNP fraction (that is, at the top of the gradient). Unfortunately, these experiments do not give clear-cut results and their interpretation has proven problematic. First, except for EDTA treatments, most conditions that disrupt polysomes (e.g., puromycin, hippuristanol, or pactamycin treatments) do not quantitatively shift mrnas from the polysome fraction to the free mrnp fraction but rather from heavy to lighter fractions of the gradient. This shift could be, in some cases, within experimental variations of the measurements. Second, inhibitory effects of mirnas are in the 2- to 5-fold range. Hence, 50% 20% of the reporter mrna is not silenced. This heterogeneity of the reporter mrna population likely obscures the results. Third, a fraction of repressed mrna may be partially degraded (most studies cannot rule out 10% 15% degradation of the reporter); depending on the degree of regulation, this may represent a significant fraction of the silenced mrna. Given the difficulties mentioned above, it is not surprising that sedimentation profiles do not provide incontrovertible evidence for or against a specific mechanism. Aware of the limitations of this approach, many studies have validated their conclusions using an independent strategy. The use of IRES represents such an independent approach. The results obtained with IRES-containing reporters have been consistent with the analysis of polysome profiles within a given study but have been surprisingly inconsistent between different studies, suggesting an experimental bias. An Experimental Approach Bias? Most evidence that translation initiation is inhibited comes from studies of mrnas synthesized in vitro and incubated in cell-free extracts or transfected into cultured cells. In vivo, 12 Cell 132, January 11, Elsevier Inc.
5 mirna targets are not naked mrnas but exist as ribonucleoprotein particles or mrnps. It is generally accepted that RNA-binding proteins are deposited on mrnas cotranscriptionally or during processing. Therefore, the full complement of proteins associating with mrnas transcribed in vivo is likely to be different from that bound to the same mrna in an in vitro system or following mrna transfection. This may explain some discrepancies observed, for example, between studies by Humphreys et al. (2005) on the one hand and Petersen et al. (2006) on the other. The implication is that RNAbinding proteins strongly influence the final outcome of mirna regulation. Another potential source of discrepancy is the nature of the reporter. Some studies used artificial reporters containing multiple identical mirna-binding sites inserted in a heterologous 3 UTR. These artificial reporters require up to six binding sites for efficient silencing, whereas natural 3 UTRs seldom have six identical, regularly spaced binding sites for the same mirna. Thus, it is possible that the artificial reporters do not fully recapitulate mirna regulation. Yet, when such reporters were used, different mechanisms could be observed; so these reporters alone do not explain all of the discrepancies (Humphreys et al., 2005; Petersen et al., 2006; Wakiyama et al., 2007). Additional experimental differences reside in the use of in vivo transcribed and processed mirnas versus transfected sirnas. Transfected sirnas are believed to act as mirnas, provided that the reporter contains partially complementary binding sites. In human cells, however, it is not clear how the biogenesis pathway or the structure of the double-stranded mirna or sirna intermediate influences which Argonaute protein they associate with. However, studies in D. melanogaster demonstrated that it is the structure of the small RNA duplex rather than the biogenesis pathway that specifies which Argonaute-containing complex is loaded with the mirna or sirna (Tomari et al., 2007). If this were the case also in human cells, endogenous mirnas and transfected sirnas could end up in different Argonaute complexes. There are four human Argonaute paralogs; all are presumed to repress translation through a similar mechanism (although this was never investigated in great detail). Moreover, most studies did not establish which human Argonaute paralog was responsible for the silencing activity. This raises the following question: Do different Argonaute proteins silence partially complementary targets through similar or different molecular mechanisms? The corollary is whether discrepancies between different studies reflect the action of different Argonaute proteins or Argonaute complexes with distinct protein compositions. Indeed, biochemical purification of Argonautecontaining mrna-protein complexes reveals partially nonoverlapping sets of mrnas in association with individual Argonaute proteins, suggesting some degree of specificity in target selection (Beitzinger et al., 2007). Multiple Mechanisms or Multiple Outputs? Studies comparing natural full-length 3 UTRs suggest that the final outcome of mirna regulation depends on the features and sequence of the target s 3 UTR (Behm-Ansmant et al., 2006; Eulalio et al., 2007a; Grimson et al., 2007). In these studies, the contribution of translational repression or mrna degradation to gene silencing differed for each mirna target pair. Stress conditions provide additional evidence that the context of the 3 UTR is important, as binding of a protein known as HuR to the 3 UTR of cationic amino acid transporter 1 (CAT-1) mrna relieves mir-122-mediated silencing. Although it remains to be determined whether HuR directly displaces mir-122 or interferes indirectly with its function in cis, it seems likely that other RNA-binding proteins might also counteract, modulate, or influence the extent and mode of mirna regulation in a target-specific manner. This is supported by the increasing number of examples in which RNA-binding proteins modulate mirna function (reviewed by Leung and Sharp, 2007). Nevertheless, it is still possible that mirnas silence gene expression through a common and unique mechanism, and that the multiple modes of action reflect secondary consequences of this primary event rather than independent mechanisms. These secondary effects will vary in a cell- or target-dependent manner. For instance, translational repression may represent a primary event that in a target- or cell-specific manner may or may not lead to mrna decay. Indeed, proteins bound to the target mrna may influence degradation. Cell-specific effects are also possible: for example, zebrafish nanos1 mrna is deadenylated and degraded by mir-430 in somatic cells yet is refractory to mir-430 regulation in the germline (Mishima et al., 2006). Likewise, one could envisage cotranslational protein degradation as a consequence of a primary event that occurred during translation elongation; the silenced ribosome signals aberrant translation such that the nascent polypeptide chain is degraded. Examples showing nascent polypeptides being cotranslationally degraded have been described (Nottrott et al., 2006 and references therein). Conclusion A growing body of evidence indicates that mirnas play important roles in the pathogenesis of human diseases such as cancer, neurodegenerative and metabolic disorders, and viral infection. A detailed mechanistic understanding of mirna-mediated gene regulation is therefore of critical importance to evaluate the potential of mirnas as therapeutic targets. Although we have begun to understand mirna biogenesis and have identified key effectors of mirna function, the mechanisms of action of mirnas in animal cells are still being hotly debated. We consider it unlikely, though, that the diverse mechanisms observed result from different technical approaches. Rather, we favor a view whereby mirna repression is manifested in distinct ways depending upon the specific features and composition of the mrnp target. Currently, we cannot determine whether these different modes of repression represent multiple independent silencing mechanisms or multiple outputs from a unique primary inhibitory event. Acknowledgments We would like to thank W. Filipowicz and D. Weigel for helpful comments. Cell 132, January 11, Elsevier Inc. 13
6 References Alemán, L.M., Doench, J., and Sharp, P.A. (2007). RNA 13, Bagga, S., Bracht, J., Hunter, S., Massirer, K., Holtz, J., Eachus, R., and Pasquinelli, A.E. (2005). Cell 122, Behm-Ansmant, I., Rehwinkel, J., Doerks, T., Stark, A., Bork, P., and Izaurralde, E. (2006). Genes Dev. 20, Beitzinger, M., Peters, L., Zhu, J.Y., Kremmer, E., and Meister, G. (2007). RNA Biol. 4, Bushati, N., and Cohen, S.M. (2007). Annu. Rev. Cell Dev. Biol. 23, Chendrimada, T.P., Finn, K.J., Ji, X., Baillat, D., Gregory, R.I., Liebhaber, S.A., Pasquinelli, A.E., and Shiekhattar, R. (2007). Nature 447, Chu, C.Y., and Rana, T.M. (2006). PLoS Biol. 4, e /journal.pbio Ding, L., and Han, M. (2007). Trends Cell Biol. 17, Eulalio, A., Rehwinkel, J., Stricker, M., Huntzinger, E., Yang, S.-F., Doerks, T., Dorner, S., Peer Bork, P., Boutros, M., and Izaurralde, E. (2007a). Genes Dev. 21, Eulalio, A., Behm-Ansmant, I., and Izaurralde, E. (2007b). Nat. Rev. Mol. Cell Biol. 8, Eulalio, A., Behm-Ansmant, I., Schweizer, D., and Izaurralde, E. (2007c). Mol. Cell. Biol. 27, Giraldez, A.J., Mishima, Y., Rihel, J., Grocock, R.J., Van Dongen, S., Inoue, K., Enright, A.J., and Schier, A.F. (2006). Science 312, Grimson, A., Farh, K.K., Johnston, W.K., Garrett- Engele, P., Lim, L.P., and Bartel, D.P. (2007). Mol. Cell 27, Humphreys, D.T., Westman, B.J., Martin, D.I., and Preiss, T. (2005). Proc. Natl. Acad. Sci. USA 102, Kiriakidou, M., Tan, G.S., Lamprinaki, S., De Planell-Saguer, M., Nelson, P.T., and Mourelatos, Z. (2007). Cell 129, Leung, A.K., and Sharp, P.A. (2007). Cell 130, Lim, L.P., Lau, N.C., Garrett-Engele, P., Grimson, A., Schelter, J.M., Castle, J., Bartel, D.P., Linsley, P.S., and Johnson, J.M. (2005). Nature 433, Maroney, P.A., Yu, Y., Fisher, J., and Nilsen, T.W. (2006). Nat. Struct. Mol. Biol. 13, Mathonnet, G., Fabian, M.R., Svitkin, Y.V., Parsyan, A., Huck, L., Murata, T., Biffo, S., Merrick, W.C., Darzynkiewicz, E., Pillai, R.S., et al. (2007). Science 17, Mishima, Y., Giraldez, A.J., Takeda, Y., Fujiwara, T., Sakamoto, H., Schier, A.F., and Inoue, K. (2006). Curr. Biol. 16, Nottrott, S., Simard, M.J., and Richter, J.D. (2006). Nat. Struct. Mol. Biol. 13, Parker, R., and Sheth, U. (2007). Mol. Cell 25, Petersen, C.P., Bordeleau, M.E., Pelletier, J., and Sharp, P.A. (2006). Mol. Cell 21, Pillai, R.S., Bhattacharyya, S.N., Artus, C.G., Zoller, T., Cougot, N., Basyuk, E., Bertrand, E., and Filipowicz, W. (2005). Science 309, Rehwinkel, J., Natalin, P., Stark, A., Brennecke, J., Cohen, S.M., and Izaurralde, E. (2006). Mol. Cell. Biol. 26, Schmitter, D., Filkowski, J., Sewer, A., Pillai, R.S., Oakeley, E.J., Zavolan, M., Svoboda, P., and Filipowicz, W. (2006). Nucleic Acids Res. 34, Seggerson, K., Tang, L., and Moss, E.G. (2002). Dev. Biol. 243, Thermann, R., and Hentze, M.W. (2007). Nature 447, Tomari, Y., Du, T., and Zamore, P.D. (2007). Cell 130, Wakiyama, M., Takimoto, K., Ohara, O., and Yokoyama, S. (2007). Genes Dev. 21, Wang, B., Love, T.M., Call, M.E., Doench, J.G., and Novina, C.D. (2006). Mol. Cell 22, Wu, L., and Belasco, J.G. (2005). Mol. Cell. Biol. 25, Wu, L., Fan, J., and Belasco, J.G. (2006). Proc. Natl. Acad. Sci. USA 103, Cell 132, January 11, Elsevier Inc.
MicroRNAs, mrnas, and Translation
 MicroRNAs, mrnas, and Translation P.A. MARONEY, Y. YU, AND T.W. NILSEN Center for RNA Molecular Biology and Department of Biochemistry, Case Western Reserve University School of Medicine, Cleveland, Ohio
MicroRNAs, mrnas, and Translation P.A. MARONEY, Y. YU, AND T.W. NILSEN Center for RNA Molecular Biology and Department of Biochemistry, Case Western Reserve University School of Medicine, Cleveland, Ohio
MicroRNAs Silence Gene Expression by Repressing Protein Expression and/or by Promoting mrna Decay
 MicroRNAs Silence Gene Expression by Repressing Protein Expression and/or by Promoting mrna Decay I. BEHM-ANSMANT, J. REHWINKEL, AND E. IZAURRALDE MPI for Developmental Biology, D-72076 Tuebingen, Germany
MicroRNAs Silence Gene Expression by Repressing Protein Expression and/or by Promoting mrna Decay I. BEHM-ANSMANT, J. REHWINKEL, AND E. IZAURRALDE MPI for Developmental Biology, D-72076 Tuebingen, Germany
Let Me Count the Ways: Mechanisms of Gene Regulation by mirnas and sirnas
 Let Me Count the Ways: Mechanisms of Gene Regulation by mirnas and sirnas Ligang Wu 1,2 and Joel G. Belasco 1,2, * 1 Kimmel Center for Biology and Medicine, Skirball Institute for Biomolecular Medicine
Let Me Count the Ways: Mechanisms of Gene Regulation by mirnas and sirnas Ligang Wu 1,2 and Joel G. Belasco 1,2, * 1 Kimmel Center for Biology and Medicine, Skirball Institute for Biomolecular Medicine
Bi 8 Lecture 17. interference. Ellen Rothenberg 1 March 2016
 Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Date: Approved: Matthias Gromeier, Chair. Bryan Cullen. Christopher Nicchitta. Michael Hauser. Jack Keene
 The Role of Poly(A)-binding Protein in microrna-mediated Repression by Robert Wheeler Walters Department of Molecular Genetics and Microbiology Duke University Date: Approved: Matthias Gromeier, Chair
The Role of Poly(A)-binding Protein in microrna-mediated Repression by Robert Wheeler Walters Department of Molecular Genetics and Microbiology Duke University Date: Approved: Matthias Gromeier, Chair
Small Interfering RNA-Mediated Translation Repression Alters Ribosome Sensitivity to Inhibition by Cycloheximide in Chlamydomonas reinhardtii
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Dissertations and Theses in Biological Sciences Biological Sciences, School of Spring 2013 Small Interfering RNA-Mediated
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Dissertations and Theses in Biological Sciences Biological Sciences, School of Spring 2013 Small Interfering RNA-Mediated
Chapter 10 - Post-transcriptional Gene Control
 Chapter 10 - Post-transcriptional Gene Control Chapter 10 - Post-transcriptional Gene Control 10.1 Processing of Eukaryotic Pre-mRNA 10.2 Regulation of Pre-mRNA Processing 10.3 Transport of mrna Across
Chapter 10 - Post-transcriptional Gene Control Chapter 10 - Post-transcriptional Gene Control 10.1 Processing of Eukaryotic Pre-mRNA 10.2 Regulation of Pre-mRNA Processing 10.3 Transport of mrna Across
GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation
 Manuscript EMBO-2012-83275 GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation Latifa Zekri, Duygu Kuzuoğlu-Öztürk and Elisa Izaurralde Corresponding author:
Manuscript EMBO-2012-83275 GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation Latifa Zekri, Duygu Kuzuoğlu-Öztürk and Elisa Izaurralde Corresponding author:
Chapter 1 Understanding How mirnas Post-Transcriptionally Regulate Gene Expression
 Chapter 1 Understanding How mirnas Post-Transcriptionally Regulate Gene Expression Marc R. Fabian, Thomas R. Sundermeier, and Nahum Sonenberg Contents 1.1 Introduction... 2 1.1.1 Eukaryotic Translation...
Chapter 1 Understanding How mirnas Post-Transcriptionally Regulate Gene Expression Marc R. Fabian, Thomas R. Sundermeier, and Nahum Sonenberg Contents 1.1 Introduction... 2 1.1.1 Eukaryotic Translation...
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions. 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19
 Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003)
 MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins.
 Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins. The RNA transcribed from a complex transcription unit
Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins. The RNA transcribed from a complex transcription unit
Computational Analysis of mirna and Target mrna Interactions: Combined Effects of The Quantity and Quality of Their Binding Sites *
 www.pibb.ac.cn!"#$%!""&'( Progress in Biochemistry and Biophysics 2009, 36(5): 608~615 Computational Analysis of mirna and Target mrna Interactions: Combined Effects of The Quantity and Quality of Their
www.pibb.ac.cn!"#$%!""&'( Progress in Biochemistry and Biophysics 2009, 36(5): 608~615 Computational Analysis of mirna and Target mrna Interactions: Combined Effects of The Quantity and Quality of Their
MicroRNA and Male Infertility: A Potential for Diagnosis
 Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Concordant Regulation of Translation and mrna Abundance for Hundreds of Targets of a Human microrna
 Concordant Regulation of Translation and mrna Abundance for Hundreds of Targets of a Human microrna David G. Hendrickson 1., Daniel J. Hogan 2,3., Heather L. McCullough 2,3,4., Jason W. Myers 2,3., Daniel
Concordant Regulation of Translation and mrna Abundance for Hundreds of Targets of a Human microrna David G. Hendrickson 1., Daniel J. Hogan 2,3., Heather L. McCullough 2,3,4., Jason W. Myers 2,3., Daniel
mirna Dr. S Hosseini-Asl
 mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
V16: involvement of micrornas in GRNs
 What are micrornas? V16: involvement of micrornas in GRNs How can one identify micrornas? What is the function of micrornas? Elisa Izaurralde, MPI Tübingen Huntzinger, Izaurralde, Nat. Rev. Genet. 12,
What are micrornas? V16: involvement of micrornas in GRNs How can one identify micrornas? What is the function of micrornas? Elisa Izaurralde, MPI Tübingen Huntzinger, Izaurralde, Nat. Rev. Genet. 12,
Novel RNAs along the Pathway of Gene Expression. (or, The Expanding Universe of Small RNAs)
 Novel RNAs along the Pathway of Gene Expression (or, The Expanding Universe of Small RNAs) Central Dogma DNA RNA Protein replication transcription translation Central Dogma DNA RNA Spliced RNA Protein
Novel RNAs along the Pathway of Gene Expression (or, The Expanding Universe of Small RNAs) Central Dogma DNA RNA Protein replication transcription translation Central Dogma DNA RNA Spliced RNA Protein
Utility of Circulating micrornas in Cardiovascular Disease
 Utility of Circulating micrornas in Cardiovascular Disease Pil-Ki Min, MD, PhD Cardiology Division, Gangnam Severance Hospital, Yonsei University College of Medicine Introduction Biology of micrornas Circulating
Utility of Circulating micrornas in Cardiovascular Disease Pil-Ki Min, MD, PhD Cardiology Division, Gangnam Severance Hospital, Yonsei University College of Medicine Introduction Biology of micrornas Circulating
MICRORNAS IN THE DROSOPHILA EGG AND EARLY EMBRYO
 MICRORNAS IN THE DROSOPHILA EGG AND EARLY EMBRYO by Sarah M. Votruba A thesis submitted in conformity with the requirements for the degree of Master of Science, Department of Molecular Genetics University
MICRORNAS IN THE DROSOPHILA EGG AND EARLY EMBRYO by Sarah M. Votruba A thesis submitted in conformity with the requirements for the degree of Master of Science, Department of Molecular Genetics University
Reduced Expression of Ribosomal Proteins Relieves MicroRNA-Mediated Repression
 Article Reduced Expression of Ribosomal Proteins Relieves MicroRNA-Mediated Repression Maja M. Janas,,, Eric Wang, 5,6 Tara Love,,,,8 Abigail S. Harris, 7 Kristen Stevenson, Karlheinz Semmelmann, 7 Jonathan
Article Reduced Expression of Ribosomal Proteins Relieves MicroRNA-Mediated Repression Maja M. Janas,,, Eric Wang, 5,6 Tara Love,,,,8 Abigail S. Harris, 7 Kristen Stevenson, Karlheinz Semmelmann, 7 Jonathan
Molecular Biology (BIOL 4320) Exam #2 April 22, 2002
 Molecular Biology (BIOL 4320) Exam #2 April 22, 2002 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Molecular Biology (BIOL 4320) Exam #2 April 22, 2002 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
The influence of micrornas and poly(a) tail length on endogenous mrna protein complexes
 Rissland et al. Genome Biology (7 8: DOI.86/s359-7-33-z RESEARCH Open Access The influence of micrornas and poly(a tail length on endogenous mrna protein complexes Olivia S. Rissland,,3,4,5,6*, Alexander
Rissland et al. Genome Biology (7 8: DOI.86/s359-7-33-z RESEARCH Open Access The influence of micrornas and poly(a tail length on endogenous mrna protein complexes Olivia S. Rissland,,3,4,5,6*, Alexander
MicroRNA in Cancer Karen Dybkær 2013
 MicroRNA in Cancer Karen Dybkær RNA Ribonucleic acid Types -Coding: messenger RNA (mrna) coding for proteins -Non-coding regulating protein formation Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
MicroRNA in Cancer Karen Dybkær RNA Ribonucleic acid Types -Coding: messenger RNA (mrna) coding for proteins -Non-coding regulating protein formation Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
Phenomena first observed in petunia
 Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Research Article Base Composition Characteristics of Mammalian mirnas
 Journal of Nucleic Acids Volume 2013, Article ID 951570, 6 pages http://dx.doi.org/10.1155/2013/951570 Research Article Base Composition Characteristics of Mammalian mirnas Bin Wang Department of Chemistry,
Journal of Nucleic Acids Volume 2013, Article ID 951570, 6 pages http://dx.doi.org/10.1155/2013/951570 Research Article Base Composition Characteristics of Mammalian mirnas Bin Wang Department of Chemistry,
Lecture 4 Regulation of Initiation: mrna-binding
 Institut für Biochemie und Molekulare Medizin Lecture 4 Regulation of Initiation: mrna-binding Michael Altmann FS 2010 Regulation of mrna binding to ribosomes mrna structure Cap accessibility RNA secondary
Institut für Biochemie und Molekulare Medizin Lecture 4 Regulation of Initiation: mrna-binding Michael Altmann FS 2010 Regulation of mrna binding to ribosomes mrna structure Cap accessibility RNA secondary
MicroRNAs: Biogenesis and Molecular Functions
 Brain athology ISSN 1015-6305 SYMOSIUM: MicroRNAs: Biology and Roles in Neurodegeneration and Brain Tumors Xuhang Liu*; Kristine Fortin*; Zissimos Mourelatos Department of athology and Laboratory Medicine,
Brain athology ISSN 1015-6305 SYMOSIUM: MicroRNAs: Biology and Roles in Neurodegeneration and Brain Tumors Xuhang Liu*; Kristine Fortin*; Zissimos Mourelatos Department of athology and Laboratory Medicine,
Investigating the regulation of mirna biogenesis and Argonaute2 by RNA binding proteins
 Investigating the regulation of mirna biogenesis and Argonaute2 by RNA binding proteins Patrick Peter Connerty Supervisor: Gyorgy Hutvagner Thesis Submitted for the Degree of Doctor of Philosophy (Science)
Investigating the regulation of mirna biogenesis and Argonaute2 by RNA binding proteins Patrick Peter Connerty Supervisor: Gyorgy Hutvagner Thesis Submitted for the Degree of Doctor of Philosophy (Science)
Objectives: Prof.Dr. H.D.El-Yassin
 Protein Synthesis and drugs that inhibit protein synthesis Objectives: 1. To understand the steps involved in the translation process that leads to protein synthesis 2. To understand and know about all
Protein Synthesis and drugs that inhibit protein synthesis Objectives: 1. To understand the steps involved in the translation process that leads to protein synthesis 2. To understand and know about all
Messenger RNA (mrna) is never alone
 Messenger RNA (mrna) is never alone mrna is coated and compacted by RNA-binding proteins (RBPs), forming large messenger ribonucleoprotein particles (mrnps). RBPs assemble on nascent and mature mrnas.
Messenger RNA (mrna) is never alone mrna is coated and compacted by RNA-binding proteins (RBPs), forming large messenger ribonucleoprotein particles (mrnps). RBPs assemble on nascent and mature mrnas.
Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression. Chromatin Array of nuc
 Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression Chromatin Array of nuc 1 Transcriptional control in Eukaryotes: Chromatin undergoes structural changes
Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression Chromatin Array of nuc 1 Transcriptional control in Eukaryotes: Chromatin undergoes structural changes
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Circular RNAs (circrnas) act a stable mirna sponges
 Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Regulation of Gene Expression in Eukaryotes
 Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Marta Puerto Plasencia. microrna sponges
 Marta Puerto Plasencia microrna sponges Introduction microrna CircularRNA Publications Conclusions The most well-studied regions in the human genome belong to proteincoding genes. Coding exons are 1.5%
Marta Puerto Plasencia microrna sponges Introduction microrna CircularRNA Publications Conclusions The most well-studied regions in the human genome belong to proteincoding genes. Coding exons are 1.5%
MicroRNAs, RNA Modifications, RNA Editing. Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM
 MicroRNAs, RNA Modifications, RNA Editing Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM Expanding world of RNAs mrna, messenger RNA (~20,000) trna, transfer
MicroRNAs, RNA Modifications, RNA Editing Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM Expanding world of RNAs mrna, messenger RNA (~20,000) trna, transfer
Translation. Host Cell Shutoff 1) Initiation of eukaryotic translation involves many initiation factors
 Translation Questions? 1) How does poliovirus shutoff eukaryotic translation? 2) If eukaryotic messages are not translated how can poliovirus get its message translated? Host Cell Shutoff 1) Initiation
Translation Questions? 1) How does poliovirus shutoff eukaryotic translation? 2) If eukaryotic messages are not translated how can poliovirus get its message translated? Host Cell Shutoff 1) Initiation
High AU content: a signature of upregulated mirna in cardiac diseases
 https://helda.helsinki.fi High AU content: a signature of upregulated mirna in cardiac diseases Gupta, Richa 2010-09-20 Gupta, R, Soni, N, Patnaik, P, Sood, I, Singh, R, Rawal, K & Rani, V 2010, ' High
https://helda.helsinki.fi High AU content: a signature of upregulated mirna in cardiac diseases Gupta, Richa 2010-09-20 Gupta, R, Soni, N, Patnaik, P, Sood, I, Singh, R, Rawal, K & Rani, V 2010, ' High
Ch. 18 Regulation of Gene Expression
 Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
RNA Processing in Eukaryotes *
 OpenStax-CNX module: m44532 1 RNA Processing in Eukaryotes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you
OpenStax-CNX module: m44532 1 RNA Processing in Eukaryotes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you
The meaning of nonsense
 Opinion The meaning of nonsense Lukas Stalder and Oliver Mühlemann Institute of Cell Biology, University of Berne, Baltzerstrabe 4, 3012 Berne, Switzerland To ensure the accuracy of gene expression, eukaryotes
Opinion The meaning of nonsense Lukas Stalder and Oliver Mühlemann Institute of Cell Biology, University of Berne, Baltzerstrabe 4, 3012 Berne, Switzerland To ensure the accuracy of gene expression, eukaryotes
Cross species analysis of genomics data. Computational Prediction of mirnas and their targets
 02-716 Cross species analysis of genomics data Computational Prediction of mirnas and their targets Outline Introduction Brief history mirna Biogenesis Why Computational Methods? Computational Methods
02-716 Cross species analysis of genomics data Computational Prediction of mirnas and their targets Outline Introduction Brief history mirna Biogenesis Why Computational Methods? Computational Methods
PROTEIN SYNTHESIS. It is known today that GENES direct the production of the proteins that determine the phonotypical characteristics of organisms.
 PROTEIN SYNTHESIS It is known today that GENES direct the production of the proteins that determine the phonotypical characteristics of organisms.» GENES = a sequence of nucleotides in DNA that performs
PROTEIN SYNTHESIS It is known today that GENES direct the production of the proteins that determine the phonotypical characteristics of organisms.» GENES = a sequence of nucleotides in DNA that performs
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004
 Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION. Ana M. Martinez
 NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
MicroRNAs regulate de novo DNA methylation and histone mrna 3 end formation in mammalian cells
 MicroRNAs regulate de novo DNA methylation and histone mrna 3 end formation in mammalian cells Inauguraldissertation zur Erlangung der Würde eines Doktors der Philosophie vorgelegt der Philosophisch-Naturwissenschaftlichen
MicroRNAs regulate de novo DNA methylation and histone mrna 3 end formation in mammalian cells Inauguraldissertation zur Erlangung der Würde eines Doktors der Philosophie vorgelegt der Philosophisch-Naturwissenschaftlichen
MicroRNA target interactions: new insights from genome-wide approaches
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Nutrition and Physical Activity in Aging, Obesity, and Cancer MicroRNA target interactions: new insights from genome-wide
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Nutrition and Physical Activity in Aging, Obesity, and Cancer MicroRNA target interactions: new insights from genome-wide
THE EFFECT OF HSA-MIR-105 ON PROSTATE CANCER GROWTH
 THE EFFECT OF HSA-MIR-105 ON PROSTATE CANCER GROWTH by David Rice Honeywell Bachelor of Science, Queen s University, 2010 THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER
THE EFFECT OF HSA-MIR-105 ON PROSTATE CANCER GROWTH by David Rice Honeywell Bachelor of Science, Queen s University, 2010 THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER
Plasticity at the synapse A role for micrornas
 Plasticity at the synapse A role for micrornas Anil Ori Universiteit Utrecht Biology of Disease 04-07-2011 Table of contents 1. Preface... 3 2. Abstract... 4 3. Background... 5 4. Neurons and their synapses...
Plasticity at the synapse A role for micrornas Anil Ori Universiteit Utrecht Biology of Disease 04-07-2011 Table of contents 1. Preface... 3 2. Abstract... 4 3. Background... 5 4. Neurons and their synapses...
An oxygen-regulated switch in the protein synthesis machinery
 An oxygen-regulated switch in the protein synthesis machinery 1 Inhibition of translation during acute hypoxia is mediated by eif2α phosphorilation mouse embryo fibroblasts (MEFs) 1 Watson et al., BIOLOGIA
An oxygen-regulated switch in the protein synthesis machinery 1 Inhibition of translation during acute hypoxia is mediated by eif2α phosphorilation mouse embryo fibroblasts (MEFs) 1 Watson et al., BIOLOGIA
SUPPLEMENTARY INFORMATION
 Table S1 Regulators of mirna processing and function Name Description Effect on mirnas Ref. Regulators of mirna processing p68/p72 SMADs SNIP1 DEAD-box RNA helicases Signal transducers; transcription regulators
Table S1 Regulators of mirna processing and function Name Description Effect on mirnas Ref. Regulators of mirna processing p68/p72 SMADs SNIP1 DEAD-box RNA helicases Signal transducers; transcription regulators
Mechanisms of alternative splicing regulation
 Mechanisms of alternative splicing regulation The number of mechanisms that are known to be involved in splicing regulation approximates the number of splicing decisions that have been analyzed in detail.
Mechanisms of alternative splicing regulation The number of mechanisms that are known to be involved in splicing regulation approximates the number of splicing decisions that have been analyzed in detail.
Controlling mirna Regulation in Disease. Willemijn M. Gommans and Eugene Berezikov
 Chapter 1 Controlling mirna Regulation in Disease Willemijn M. Gommans and Eugene Berezikov Abstract Our understanding of the importance of noncoding RNA molecules is steadily growing. One such important
Chapter 1 Controlling mirna Regulation in Disease Willemijn M. Gommans and Eugene Berezikov Abstract Our understanding of the importance of noncoding RNA molecules is steadily growing. One such important
MicroRNAs: Critical Regulators of mrna Traffic and Translational Control with Promising Biotech and Therapeutic Applications
 Iranian Journal of Biotechnology. 2013 August; 11(3): 147-55. Published Online 2013 August 01. Review Article MicroRNAs: Critical Regulators of mrna Traffic and Translational Control with Promising Biotech
Iranian Journal of Biotechnology. 2013 August; 11(3): 147-55. Published Online 2013 August 01. Review Article MicroRNAs: Critical Regulators of mrna Traffic and Translational Control with Promising Biotech
Human Genome: Mapping, Sequencing Techniques, Diseases
 Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
RNA (Ribonucleic acid)
 RNA (Ribonucleic acid) Structure: Similar to that of DNA except: 1- it is single stranded polunucleotide chain. 2- Sugar is ribose 3- Uracil is instead of thymine There are 3 types of RNA: 1- Ribosomal
RNA (Ribonucleic acid) Structure: Similar to that of DNA except: 1- it is single stranded polunucleotide chain. 2- Sugar is ribose 3- Uracil is instead of thymine There are 3 types of RNA: 1- Ribosomal
scientific report Defining a new role of GW182 in maintaining mirna stability scientificreport
 scientificreport Defining a new role of in maintaining mirna stability Bing Yao 1,w,LanB.La 1,Ying-ChiChen 2, Lung-Ji Chang 2 &EdwardK.L.Chan 1,+ 1 Department of Oral Biology, and 2 Department of Molecular
scientificreport Defining a new role of in maintaining mirna stability Bing Yao 1,w,LanB.La 1,Ying-ChiChen 2, Lung-Ji Chang 2 &EdwardK.L.Chan 1,+ 1 Department of Oral Biology, and 2 Department of Molecular
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Genetics and Genomics in Medicine Chapter 6 Questions
 Genetics and Genomics in Medicine Chapter 6 Questions Multiple Choice Questions Question 6.1 With respect to the interconversion between open and condensed chromatin shown below: Which of the directions
Genetics and Genomics in Medicine Chapter 6 Questions Multiple Choice Questions Question 6.1 With respect to the interconversion between open and condensed chromatin shown below: Which of the directions
Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Chapter 18 Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development and is responsible for differences
Chapter 18 Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development and is responsible for differences
Post-transcriptional and translational regulation of mrna-like long non-coding RNAs by micrornas in early developmental stages of zebrafish embryos
 BMB Reports BMB Rep. 2017; 50(4): 226-231 www.bmbreports.org Post-transcriptional and translational regulation of mrna-like long non-coding RNAs by micrornas in early developmental stages of zebrafish
BMB Reports BMB Rep. 2017; 50(4): 226-231 www.bmbreports.org Post-transcriptional and translational regulation of mrna-like long non-coding RNAs by micrornas in early developmental stages of zebrafish
Intrinsic cellular defenses against virus infection
 Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
MicroRNAs and Cancer
 MicroRNAs and Cancer MicroRNAs are an abundant class of small (20-25 nucleotides) non-protein coding RNAs that function as negative gene regulators. The human genome contains up to 1000 micrornas which
MicroRNAs and Cancer MicroRNAs are an abundant class of small (20-25 nucleotides) non-protein coding RNAs that function as negative gene regulators. The human genome contains up to 1000 micrornas which
Selective filtering defect at the axon initial segment in Alzheimer s disease mouse models. Yu Wu
 Selective filtering defect at the axon initial segment in Alzheimer s disease mouse models Yu Wu Alzheimer s Disease (AD) Mouse models: APP/PS1, PS1δE9, APPswe, hps1 Wirths, O. et al, Acta neuropathologica
Selective filtering defect at the axon initial segment in Alzheimer s disease mouse models Yu Wu Alzheimer s Disease (AD) Mouse models: APP/PS1, PS1δE9, APPswe, hps1 Wirths, O. et al, Acta neuropathologica
The Biology and Genetics of Cells and Organisms The Biology of Cancer
 The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
Post-transcriptional regulation of an intronic microrna
 Post-transcriptional regulation of an intronic microrna Carl Novina Dana-Farber Cancer Institute Harvard Medical School Broad Institute of Harvard and MIT Qiagen Webinar 05-17-11 Outline 1. The biology
Post-transcriptional regulation of an intronic microrna Carl Novina Dana-Farber Cancer Institute Harvard Medical School Broad Institute of Harvard and MIT Qiagen Webinar 05-17-11 Outline 1. The biology
Genetics. Instructor: Dr. Jihad Abdallah Transcription of DNA
 Genetics Instructor: Dr. Jihad Abdallah Transcription of DNA 1 3.4 A 2 Expression of Genetic information DNA Double stranded In the nucleus Transcription mrna Single stranded Translation In the cytoplasm
Genetics Instructor: Dr. Jihad Abdallah Transcription of DNA 1 3.4 A 2 Expression of Genetic information DNA Double stranded In the nucleus Transcription mrna Single stranded Translation In the cytoplasm
Computational Biology I LSM5191
 Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture Notes: Transcriptome: Molecular Biology of Gene Expression II TRANSLATION RIBOSOMES: protein synthesizing machines Translation takes place on defined
Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture Notes: Transcriptome: Molecular Biology of Gene Expression II TRANSLATION RIBOSOMES: protein synthesizing machines Translation takes place on defined
Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral
 doi:10.1038/nature14171 SUPPLEMENTARY DISCUSSION Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral signaling 1. Stress-induced oligomerization of the extracellular domain of
doi:10.1038/nature14171 SUPPLEMENTARY DISCUSSION Supplementary Discussion 1: The mechanistic model of NIK1-mediated antiviral signaling 1. Stress-induced oligomerization of the extracellular domain of
MicroRNAs: molecular features and role in cancer.
 MicroRNAs: molecular features and role in cancer. Elodie Lages, Hélène Ipas, Audrey Guttin, Houssam Nesr, François Berger, Jean-Paul Issartel To cite this version: Elodie Lages, Hélène Ipas, Audrey Guttin,
MicroRNAs: molecular features and role in cancer. Elodie Lages, Hélène Ipas, Audrey Guttin, Houssam Nesr, François Berger, Jean-Paul Issartel To cite this version: Elodie Lages, Hélène Ipas, Audrey Guttin,
Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN
 Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN Institute for Brain Disorders and Neural Regeneration F.M. Kirby Program in Neural
Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN Institute for Brain Disorders and Neural Regeneration F.M. Kirby Program in Neural
MicroRNAs control mrna fate by compartmentalization based on 3 UTR length in male germ cells
 Zhang et al. Genome Biology (2017) 18:105 DOI 10.1186/s13059-017-1243-x RESEARCH MicroRNAs control mrna fate by compartmentalization based on 3 UTR length in male germ cells Ying Zhang 1, Chong Tang 1,
Zhang et al. Genome Biology (2017) 18:105 DOI 10.1186/s13059-017-1243-x RESEARCH MicroRNAs control mrna fate by compartmentalization based on 3 UTR length in male germ cells Ying Zhang 1, Chong Tang 1,
number Done by Corrected by Doctor Ashraf
 number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Assembly of mrnp Complexes During Stress and Nonsense-Mediated mrna Decay Quality Control in Saccharomyces cerevisiae
 Assembly of mrnp Complexes During Stress and Nonsense-Mediated mrna Decay Quality Control in Saccharomyces cerevisiae Item Type text; Electronic Dissertation Authors Swisher, Kylie Publisher The University
Assembly of mrnp Complexes During Stress and Nonsense-Mediated mrna Decay Quality Control in Saccharomyces cerevisiae Item Type text; Electronic Dissertation Authors Swisher, Kylie Publisher The University
RNA interference (RNAi)
 RN interference (RNi) Natasha aplen ene Silencing Section Office of Science and Technology Partnerships Office of the Director enter for ancer Research National ancer Institute ncaplen@mail.nih.gov Plants
RN interference (RNi) Natasha aplen ene Silencing Section Office of Science and Technology Partnerships Office of the Director enter for ancer Research National ancer Institute ncaplen@mail.nih.gov Plants
TRANSCRIPTION. DNA à mrna
 TRANSCRIPTION DNA à mrna Central Dogma Animation DNA: The Secret of Life (from PBS) http://www.youtube.com/watch? v=41_ne5ms2ls&list=pl2b2bd56e908da696&index=3 Transcription http://highered.mcgraw-hill.com/sites/0072507470/student_view0/
TRANSCRIPTION DNA à mrna Central Dogma Animation DNA: The Secret of Life (from PBS) http://www.youtube.com/watch? v=41_ne5ms2ls&list=pl2b2bd56e908da696&index=3 Transcription http://highered.mcgraw-hill.com/sites/0072507470/student_view0/
Chapter 11 How Genes Are Controlled
 Chapter 11 How Genes Are Controlled PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture by Mary
Chapter 11 How Genes Are Controlled PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture by Mary
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
Chapter 2 Biogenesis of Mammalian mirna
 Chapter 2 Biogenesis of Mammalian mirna Stephen L. Clarke, McKale R. Davis and Ramanjulu Sunkar Abstract MicroRNA (mirna) are small noncoding molecules approximately 22 nucleotides in length that mediate
Chapter 2 Biogenesis of Mammalian mirna Stephen L. Clarke, McKale R. Davis and Ramanjulu Sunkar Abstract MicroRNA (mirna) are small noncoding molecules approximately 22 nucleotides in length that mediate
Where Splicing Joins Chromatin And Transcription. 9/11/2012 Dario Balestra
 Where Splicing Joins Chromatin And Transcription 9/11/2012 Dario Balestra Splicing process overview Splicing process overview Sequence context RNA secondary structure Tissue-specific Proteins Development
Where Splicing Joins Chromatin And Transcription 9/11/2012 Dario Balestra Splicing process overview Splicing process overview Sequence context RNA secondary structure Tissue-specific Proteins Development
Translation Specificity and Ribosomal Heterogeneity
 Translation Specificity and Ribosomal Heterogeneity Dylan Mooijman 1 Control of gene expression is essential for cellular processes to proceed correctly as disturbances in both RNA and protein levels have
Translation Specificity and Ribosomal Heterogeneity Dylan Mooijman 1 Control of gene expression is essential for cellular processes to proceed correctly as disturbances in both RNA and protein levels have
Eukaryotic mrna is covalently processed in three ways prior to export from the nucleus:
 RNA Processing Eukaryotic mrna is covalently processed in three ways prior to export from the nucleus: Transcripts are capped at their 5 end with a methylated guanosine nucleotide. Introns are removed
RNA Processing Eukaryotic mrna is covalently processed in three ways prior to export from the nucleus: Transcripts are capped at their 5 end with a methylated guanosine nucleotide. Introns are removed
Life Sciences 1A Midterm Exam 2. November 13, 2006
 Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
LESSON 4.4 WORKBOOK. How viruses make us sick: Viral Replication
 DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
DNA codes for RNA, which guides protein synthesis.
 Section 3: DNA codes for RNA, which guides protein synthesis. K What I Know W What I Want to Find Out L What I Learned Vocabulary Review synthesis New RNA messenger RNA ribosomal RNA transfer RNA transcription
Section 3: DNA codes for RNA, which guides protein synthesis. K What I Know W What I Want to Find Out L What I Learned Vocabulary Review synthesis New RNA messenger RNA ribosomal RNA transfer RNA transcription
Removal of Shelterin Reveals the Telomere End-Protection Problem
 Removal of Shelterin Reveals the Telomere End-Protection Problem DSB Double-Strand Breaks causate da radiazioni stress ossidativo farmaci DSB e CROMATINA Higher-order chromatin packaging is a barrier to
Removal of Shelterin Reveals the Telomere End-Protection Problem DSB Double-Strand Breaks causate da radiazioni stress ossidativo farmaci DSB e CROMATINA Higher-order chromatin packaging is a barrier to
Lecture 2: Virology. I. Background
 Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Eukaryotic Gene Regulation
 Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
MicroRNA expression profiling and functional analysis in prostate cancer. Marco Folini s.c. Ricerca Traslazionale DOSL
 MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
Sections 12.3, 13.1, 13.2
 Sections 12.3, 13.1, 13.2 Now that the DNA has been copied, it needs to send its genetic message to the ribosomes so proteins can be made Transcription: synthesis (making of) an RNA molecule from a DNA
Sections 12.3, 13.1, 13.2 Now that the DNA has been copied, it needs to send its genetic message to the ribosomes so proteins can be made Transcription: synthesis (making of) an RNA molecule from a DNA
Insulin mrna to Protein Kit
 Insulin mrna to Protein Kit A 3DMD Paper BioInformatics and Mini-Toober Folding Activity Student Handout www.3dmoleculardesigns.com Insulin mrna to Protein Kit Contents Becoming Familiar with the Data...
Insulin mrna to Protein Kit A 3DMD Paper BioInformatics and Mini-Toober Folding Activity Student Handout www.3dmoleculardesigns.com Insulin mrna to Protein Kit Contents Becoming Familiar with the Data...
Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design
 Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design Crick's "Central Dogma"? DNA makes RNA makes protein Crick's "Central Dogma"? DNA makes
Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design Crick's "Central Dogma"? DNA makes RNA makes protein Crick's "Central Dogma"? DNA makes
scientific report Kinetic analysis reveals successive steps leading to mirna-mediated silencing in mammalian cells scientificreport
 scientific report scientificreport Kinetic analysis reveals successive steps leading to mirn-mediated silencing in mammalian cells Julien Béthune 1+,CarolineG.rtus-Revel 1 & Witold Filipowicz 1,2++ 1 Friedrich
scientific report scientificreport Kinetic analysis reveals successive steps leading to mirn-mediated silencing in mammalian cells Julien Béthune 1+,CarolineG.rtus-Revel 1 & Witold Filipowicz 1,2++ 1 Friedrich
Protein Synthesis
 Protein Synthesis 10.6-10.16 Objectives - To explain the central dogma - To understand the steps of transcription and translation in order to explain how our genes create proteins necessary for survival.
Protein Synthesis 10.6-10.16 Objectives - To explain the central dogma - To understand the steps of transcription and translation in order to explain how our genes create proteins necessary for survival.
L I F E S C I E N C E S
 1a L I F E S C I E N C E S 5 -UUA AUA UUC GAA AGC UGC AUC GAA AAC UGU GAA UCA-3 5 -TTA ATA TTC GAA AGC TGC ATC GAA AAC TGT GAA TCA-3 3 -AAT TAT AAG CTT TCG ACG TAG CTT TTG ACA CTT AGT-5 NOVEMBER 2, 2006
1a L I F E S C I E N C E S 5 -UUA AUA UUC GAA AGC UGC AUC GAA AAC UGU GAA UCA-3 5 -TTA ATA TTC GAA AGC TGC ATC GAA AAC TGT GAA TCA-3 3 -AAT TAT AAG CTT TCG ACG TAG CTT TTG ACA CTT AGT-5 NOVEMBER 2, 2006
The Characterization of microrna-mediated Gene Regulation as Impacted by Both Target Site Location and Seed Match Type
 The Characterization of microrna-mediated Gene Regulation as Impacted by Both Target Site Location and Seed Match Type Wenlong Xu 1, Zixing Wang 1, Yin Liu 1,2 * 1 Department of Neurobiology and Anatomy,
The Characterization of microrna-mediated Gene Regulation as Impacted by Both Target Site Location and Seed Match Type Wenlong Xu 1, Zixing Wang 1, Yin Liu 1,2 * 1 Department of Neurobiology and Anatomy,
Computational Identification and Prediction of Tissue-Specific Alternative Splicing in H. Sapiens. Eric Van Nostrand CS229 Final Project
 Computational Identification and Prediction of Tissue-Specific Alternative Splicing in H. Sapiens. Eric Van Nostrand CS229 Final Project Introduction RNA splicing is a critical step in eukaryotic gene
Computational Identification and Prediction of Tissue-Specific Alternative Splicing in H. Sapiens. Eric Van Nostrand CS229 Final Project Introduction RNA splicing is a critical step in eukaryotic gene
Explain that each trna molecule is recognised by a trna-activating enzyme that binds a specific amino acid to the trna, using ATP for energy
 7.4 - Translation 7.4.1 - Explain that each trna molecule is recognised by a trna-activating enzyme that binds a specific amino acid to the trna, using ATP for energy Each amino acid has a specific trna-activating
7.4 - Translation 7.4.1 - Explain that each trna molecule is recognised by a trna-activating enzyme that binds a specific amino acid to the trna, using ATP for energy Each amino acid has a specific trna-activating
