Bone Morphogenetic Protein Signaling Is Essential for Terminal Differentiation of the Intestinal Secretory Cell Lineage
|
|
|
- Tyrone Lloyd
- 6 years ago
- Views:
Transcription
1 GASTROENTEROLOGY 2007;133: Bone Morphogenetic Protein Signaling Is Essential for Terminal Differentiation of the Intestinal Secretory Cell Lineage BENOIT A. AUCLAIR,* YANNICK D. BENOIT,* NATHALIE RIVARD,* YUJI MISHINA, and NATHALIE PERREAULT* *Canadian Institutes of Health Research Team on Digestive Epithelium, Département d Anatomie et Biologie Cellulaire, Faculté de Médecine et des Sciences de la Santé, Université de Sherbrooke, Sherbrooke, Quebec, Canada; and the Molecular Developmental Biology Section, Laboratory of Reproductive and Developmental Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina See editorial on page Background & Aims: Bone morphogenetic proteins (Bmps) are morphogens known to play key roles in gastrointestinal development and pathology. Most Bmps are produced primarily by the mesenchymal compartment and activate their signaling pathways following a paracrine or autocrine route. The aim of this study was to investigate the role of epithelial Bmp signaling in intestinal morphogenesis and maintenance of adult epithelial cell functions. Methods: With the use of tissue-specific gene ablation, we generated mice lacking the Bmp receptor type IA (Bmpr1a) exclusively in the intestinal epithelium. Bmpr1a mutant and control mice were sacrificed for histology, immunofluorescence, Western blot analysis, electron microscopy, and quantitative polymerase chain reaction. Results: As well as showing increased proliferation and altered intestinal epithelial morphology, Bmpr1a mutant mice revealed that epithelial Bmp signaling is associated with impaired terminal differentiation of cells from the secretory lineage but not with the determination of cell fate. Loss of Bmp signaling exclusively in the epithelial compartment is not sufficient for the initiation of the de novo crypt phenomenon associated with juvenile polyposis syndrome. Conclusions: Epithelial Bmp signaling plays an important role in the terminal differentiation of the intestinal secretory cell lineage but not in de novo crypt formation. These findings emphasize the importance of delineating the contribution of the stroma vs the epithelium in gastrointestinal physiology and pathology. The adult intestinal mucosa is composed of undifferentiated pluripotent stem cells as well as undifferentiated daughter cells located in the lower portion of the intestinal crypt and differentiated functional epithelial cells found along the villus. The commitment of the daughter cell to differentiate is acquired in the upper third of the crypt where the cell loses its ability to divide. 1,2 There are 4 types of terminally differentiated intestinal cells derived from stem cells that can be divided into 2 categories. First are the absorptive cells, which play a role in the absorption of nutrients, and, second, cells of the secretory lineage including the mucin-secreting goblet cells, the hormone-secreting enteroendocrine cells, and the antimicrobial peptide secreting Paneth cells. 1,2 Such cellular dynamics and specification of cell fate requires a complex and precise regulation of gene expression along the anterior/posterior and crypt villus axes. 1,2 The defined expression of certain genes at a specific time or at a precise location requires a specialized microenvironment that depends on a variety of growth factors, cell cell interactions, and epithelial mesenchymal interactions. 3 5 Recent studies have identified the Hedgehog, platelet-derived growth factor, and bone morphogenetic protein (Bmp) signaling pathways as key mediators in crypt and villus morphogenesis as well as maintenance of intestinal stem cells. 4,6 8 The Bmps are morphogens belonging to the transforming growth factor superfamily. Bmps play active roles in many cellular functions in adult animals, 9 signaling through the serine/threonine kinase receptor subtypes I and II. The main Bmps found in the small intestine, Bmp2 and Bmp4, 5 perform their signaling through the type IA receptor (Bmpr1a). 9 Recently, 3 research groups generated mice with impaired Bmp signaling affecting the epithelial and stromal compartments of the gut simultaneously. 4,7,8 All showed the phenomenon of de novo crypt in the intestinal mucosa because the intestinal epithelium of these mice resembles the mucosa of patients with juvenile polyposis syndrome. This shows a significant contribution of both epithelial and mesenchymal Bmp signaling in intestinal pathology. Interestingly, a recent study by Kim et al 10 revealed that the selective loss of Smad4 in the T cell, a prominent stromal component, led to epithelial tumors throughout the gastro- Abbreviations used in this paper: Bmps, bone morphogenetic proteins; Bmpr1a, bone morphogenetic protein receptor type IA; BrdU, bromodeoxyuridine; Ngn3, Neurogenin by the AGA Institute /07/$32.00 doi: /j.gastro
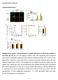
2 888 AUCLAIR ET AL GASTROENTEROLOGY Vol. 133, No. 3 intestinal tract, whereas its epithelial-specific deletion did not. Deletion of Smad4 in T cells suggests that loss of Smad4-dependent signaling in cells within the epithelial microenvironment has an important role in the evolution of juvenile polyposis syndrome. However, a specific role and the molecular relevance of excluding the Bmp pathway from one particular cell compartment over the other in gastrointestinal maintenance of adult epithelial cell functions remain unknown. This study shows that the loss of Bmp signaling exclusively in the intestinal epithelium has a dramatic consequence on epithelial proliferation and terminal differentiation and maturation of cells from the secretory lineage, but not on the initiation of, de novo crypts. Materials and Methods Animals SVEV-Bmpr1a loxp/loxp mice were provided by Dr. Mishina and the C57BL/6 12.4KbVilCre transgenic line was provided by Dr. Gumucio. Both mutations were genotyped following protocols already published. 11,12 Tissue Collection, RNA Extraction, and Gene Expression Analysis Total RNA was isolated and processed using the Totally RNA extraction kit (Ambion, Austin, TX). Reverse-transcription polymerase chain reaction analysis was performed using AMV-RT (Roche Diagnostic, Laval, Quebec, Canada) following the manufacturer s protocol. Quantitative real-time polymerase chain reaction was performed using the Quantitech SYBR Green Kit from Qiagen (Mississauga, Ontario, Canada) with the Roche Diagnostic Light Cycler. Primer sequences are available on request. Tissue Preparation and Histologic Staining Jejunum were fixed in 4% paraformaldehyde overnight at 4 C, then dehydrated and embedded in paraffin. Alcian blue and H&E stainings were performed as previously described. 13 Bromodeoxyuridine Incorporation, Immunofluorescence, and In Situ Hybridization Mice were injected with 10 L of bromodeoxyuridine (BrdU) (Invitrogen, Burlington, Ontario, Canada) per gram of body weight 90 minutes before death. Immunofluorescence staining was performed as previously described. 13 The following antibodies were used: anti-brdu (1:50; Roche Diagnostic), anti-lysozyme (1:2; DakoCytomation, Mississauga, Ontario, Canada), anti phosphorylated-smad (1:200; Cell Signaling, Boston, MA), anti chromogranin A (1:2; Zymed), anti intestinal fatty acid binding protein (1:5000; kindly provided by Dr. Gordon), anti-bmpr1a (1:50; Abgent, San Diego, CA), anti activated-caspase3 (1:1500; Cell Signaling), and fluorescein isothiocyanate conjugated anti-rabbit or mouse immunoglobulin G (1:200; Vector, Burlington, Ontario, Canada). In situ hybridization was performed according to the Wilkinson 14 procedure, modified by Lantz et al, 15 and sucrase-isomaltase riboprobes were synthesized as previously described. 16 Protein Extraction and Western Blot Analysis Total protein was isolated from the jejunum as described previously. 3 Nuclear proteins were isolated using the ProteoExtract Subcellular Proteome Extraction kit (EMD Biosciences, San Diego, CA) following the manufacturer s protocol. Fifty (total) or 15 (nuclear) micrograms of protein were separated as described previously. 3 The following antibodies were used and incubated overnight at 4 C: anti c-myc (1:200), anti cyclin D1/D2 (1: 1000), anti-actin (1:5000), anti histone H1 (1:1000), antirabbit or goat horseradish peroxidase (1:3000) (all from Santa Cruz Biotech, Santa Cruz, CA), and anti -catenin (1:2500) (Cell Signaling). Quantification and Statistical Tests Images of alcian blue labeled goblet cells and chromogranin A stained enteroendocrine cells were acquired and positive cells were counted per crypt/villus axis. Nuclei were counted using 4=,6-diamidino-2-phenylindole. Variation of crypt number and crypt fission was assessed by counting the total number of crypts, villi, and crypt fissions in a blind manner. Paneth cells were quantified with lysozyme immunofluorescence. Proliferation was measured by counting BrdU-labeled cells per crypt. Statistics were calculated using the Mann Whitney or the Student 2-tailed t test. All statistical analyses were performed using Systat10 (Systat software Inc., San Jose, CA). Results Loss of Epithelial Intestinal Bmp Signaling Increases Epithelial Proliferation But Is Insufficient to Initiate De Novo Crypts To determine the specific contribution of epithelial Bmp signaling on intestinal physiology and cell function, we used the tissue-specific gene deletion of Bmpr1a using the Cre-loxP system. To delete the Bmpr1a gene exclusively in the epithelium, we crossed Bmpr1a loxp/loxp mice with a Villin-Cre transgenic line, which directs Cre expression in the epithelium of the small intestine and colon, but not in the mesenchymal compartment. 11,12 Villin-Cre;Bmpr1a loxp/loxp mice survived to at least 1 year of age and grew normally. Bmpr1a immunofluorescence of control mice (Figure 1A) revealed a strong expression in the upper portion of the crypt unit and villus, with weaker staining in cells at the bottom of the crypt (Figure 1E). Bmpr1a expression was lost exclusively in the intestinal epithelium of the
3 September 2007 BMP SIGNALING IN INTESTINAL CELL LINEAGE 889 crypt-villus axis in Villin-Cre;Bmpr1aloxP/loxP mice but not in the mesenchymal cells of the lamina propria (Figure 1B and F). Loss of Bmp signaling exclusively in the intestinal epithelium was assessed by immunofluorescence of phosphorylated-smad Phosphorylation and nuclear translocation of these Bmp effectors are features of an active Bmp signaling pathway.9 Phosphorylated-Smad immunostaining in control mice is observed mainly in villus epithelial cells, as well as at the Figure 1. Loss of Bmp signaling cascade and histologic analysis of the intestinal architecture in VillinCre;Bmpr1aloxP/loxP mice. Bmpr1a immunofluorescence in control mice revealed a (A) strong expression in the upper portion of the crypt and villus and a (E) weaker staining in the cells at the bottom of the crypt. (B and F) Expression was lost exclusively in the intestinal epithelial cells of the crypt villus axis in Villin-Cre;Bmpr1aloxP/loxP mice. (A and B) Note the mesenchymal cells of the lamina propria express Bmpr1a (white arrowheads). Phosphorylated-Smad immunostaining in control mice is observed mainly in (C) villus epithelial cells and at the crypt villus junction, and to a lesser extent at the (G) bottom of the crypt near the stem and Paneth cell regions. The epithelial Bmp signaling cascade was lost in the (D and H) intestinal epithelium of Villin-Cre; Bmpr1aloxP/loxP mice, (D) but not in the mesenchymal cells of the lamina propria (white arrowheads). (J) H&E staining performed on the jejunum of mutant animals shows an important increase in the length of the villi and multiplication of the crypt units, but an absence of the de novo crypt phenomenon as well as polyp growth as compared with (I) controls. (J) Note the presence of crypt fission in the mutant animal (white arrow). (K) Crypt units and (L) crypt fission were counted from the jejunum of control (n 8) and Villin-Cre;Bmpr1aloxP/loxP (n 8) animals. The number of crypt units and fissions were increased significantly in the mutant (Mann Whitney; P.05). Magnification, 200 (A, B, C, D, I, and J) and 400 (E, F, G, and H). crypt villus junction, and to a lesser extent at the bottom of the crypt near the stem and Paneth cell region (Figure 1C and G). Immunoreactivity to phosphorylated-smad is lost in Villin-Cre;Bmpr1aloxP/loxP mice (Figure 1D and H). The expression pattern of phosphorylated-smad is predominantly in regions of the crypt villus axis composed of differentiating and terminally differentiated cells, suggesting a possible role for Bmp in intestinal epithelial cytodifferentiation.
4 890 AUCLAIR ET AL GASTROENTEROLOGY Vol. 133, No. 3 Histologic analysis showed that the intestine of Villin- Cre;Bmpr1a loxp/loxp mice displayed abnormal epithelial morphology with elongated villi and multiplication of crypt units (Figure 1J). Statistical analysis of the number of crypt units over the number of villi shows a significant 1.37-fold increase of crypt units (Figure 1K) invillin-cre; Bmpr1a loxp/loxp mice, suggesting a role for Bmp signaling in crypt fission. A statistical analysis of the percentage of crypt fission found in mutant compared with control animals revealed a 4.62-fold increase in crypt fission in Villin-Cre;Bmpr1a loxp/loxp mice (Figure 1L). We noticed the absence of de novo crypts along the villus as well as polyp growth in Villin-Cre;Bmpr1a loxp/loxp mice, even in 1-yearold animals. The re-initiating of crypt morphogenesis in the villi associated with juvenile polyposis syndrome has been reported in other mouse models with impaired Bmp signaling. 4,7,8 These results suggest different roles for mesenchymal as opposed to epithelial Bmp signaling in crypt morphogenesis and epithelial cell function. We next analyzed whether proliferation and/or programmed cell death were altered in these mice. Proliferation assays in Villin-Cre;Bmpr1a loxp/loxp mice showed a significant increase in the number of proliferating cells, which was not confined to the bottom but was scattered throughout the crypt (Figure 2B). Statistical analysis revealed a significant increase in proliferative cells of fold in Bmpr1a mutant mice (Figure 2E). No difference in activated caspase-3 immunostaining was observed between control and mutant mice (data not shown). These results confirmed that the thickening of the intestinal mucosa seen in the Villin-Cre;Bmpr1a loxp/loxp mice was a direct outcome of deregulation of the proliferation program of the epithelial cells. Loss of Epithelial Bmp Signaling Does Not Affect the Wnt/ -Catenin Signaling Pathway To verify the involvement of the Wnt/ -catenin signaling pathway in the deregulation of epithelial cell proliferation in Villin-Cre;Bmpr1a loxp/loxp mice, we performed Western blot analysis with nuclear epithelial intestinal extracts. As shown in Figure 2F, loss of epithelial Bmp signaling does not affect -catenin nuclear expression, suggesting no increase in Wnt/ -catenin signaling activity. We then analyzed c-myc and cyclin D1 (Figure 2G) and showed that c-myc is not modulated, whereas cyclin D1 is up-regulated in Villin-Cre;Bmpr1a loxp/loxp mice. Cyclin D1 expression is up-regulated not only by the Wnt/ -catenin pathway, but also by many other signaling pathways such as the Ras/Erk/mitogene activated protein kinase pathway. 17 Figure 2. Epithelial Bmp signaling regulates epithelial intestinal proliferation without interfering with the Wnt/ -catenin pathway. (A and C) Proliferating cells stained by BrdU incorporation are found at the bottom of the crypts in control animals. (B and D) Mutant mice display abnormal cell proliferation. BrdU-positive cells were counted from the jejunum of control (n 3) and Villin-Cre;Bmpr1a loxp/loxp (n 3) animals. (E) Statistical analysis of the number of positive BrdU cells revealed a significant increase in the mutant (Mann Whitney; P.05). No modulation of nuclear -catenin was observed between the jejunum of the Villin-Cre;Bmpr1a loxp/loxp and control animals. (F) Histone H1 served as loading control for the nuclear extracts. An increase of cyclin D1 but no modulation of c-myc protein levels was observed in mutant compared with control animals. (G) Actin served as the loading control for the total protein extracts. Magnification, 200 (A and B) and 400 (C and D).
5 September 2007 BMP SIGNALING IN INTESTINAL CELL LINEAGE 891 Bmpr1a Mutant Mice Have Impaired Terminal Differentiation of the Secretory Cell Lineage To characterize changes in the differentiation and maturation of the 4 intestinal cell types in Villin-Cre; Bmpr1a loxp/loxp mice, we stained the mucosa with specific markers for each cell type. Goblet cells were stained with alcian blue (Figure 3A and B) and counted. No significant change was found between Bmpr1a mutant mice and control littermates (Figure 3K), however, the goblet cells were consistently smaller in the mutant mice, suggesting a lack of maturity of these cells. Indeed, smaller mucigen granules and reduction in size were observed in the mutant animals (Figure 3H). No significant change was observed in the expression of the goblet cell specific marker Muc2 between the mutant and control animals (Table 1). Comparative analysis by quantitative polymerase chain reaction of both Klf4 and Tff3, factors that regulate goblet cell terminal differentiation, 18,19 revealed a decrease in expression of 3.47-fold and 2.06-fold, respectively, in the mutant animals (Table 1). Hence, these data show that mice with impaired Bmp signaling show alterations in goblet cell terminal differentiation and maturity. Bmp signaling activity at the bottom of the crypt near the stem and Paneth cell regions (Figure 1G) has been associated exclusively with stem cell renewal. 8 Herein, we observed a more compact staining of lysozyme, a Paneth cell marker, in Bmpr1a mutant mice (Figure 3D) as compared with the control (Figure 3C), however, the location Figure 3. Epithelial Bmp signaling is not required for the determination of the secretory precursor cell but is necessary for proper differentiation of cells from the secretory lineage. Alcian blue staining revealed no variation in the number of goblet cells between the (A) control and (B) mutant animals. However, goblet cells were consistently smaller in the (H) mutant when compared with the (G) control as confirmed by electron microscopy (n 4). Immunostaining with an anti-lysozyme antibody suggested a reduction in Paneth cell secretory granule content in (D) mutant compared with (C) control animals. Electron microscopy analysis showed a reduction in the number of secretory granules in (J) mutant compared with (I) control animals. Immunostaining with chromogranin A revealed that the number of chromogranin A expressing cells was decreased in the (F) mutant intestinal epithelium when compared with the (E) controls. Goblet, Paneth, and enteroendocrine cells were counted from the jejunum of control and Villin-Cre;Bmpr1a loxp/loxp animals (n 4). (K) Statistical analysis of the number of chromogranin A positive cells revealed a significant decrease in mutant mice but no significant variation for goblet and Paneth cells (2-tailed Student t test; P.05). Magnification, 200 (A, B, C, D, E, and F). Bar 10 m (G, H, I, and J).
6 892 AUCLAIR ET AL GASTROENTEROLOGY Vol. 133, No. 3 Table 1. Gene Expression Changes in the Jejunum of Villin- Cre;Bmpr1a loxp/loxp Mice Gene description Gene symbol Fold P Cell determination genes Math 1 Math Hairy and enhancer of split 1 Hes 1.32 NS Enterocyte markers E74-like factor 3 Elf NS Sucrase-isomaltase SI 1.75 NS Enteroendocrine cell markers Neurogenin 3 Ngn Neurogenic differentiation 1 Neurod Chromogranin A Chga Paneth cell markers Cryptdin Defcr Lysozyme Lyz Matrix metallopeptidase 7 MMP Goblet cell markers Mucin 2 Muc NS Kruppel-like factor 4 Klf Trefoil factor 3 Tff Hedgehog signaling pathway Indian Hedgehog IHH 1.49 NS GLI-Kruppel family member 1 Gli NS Patched 1 Ptc 1.89 NS NOTE. Fold changes represent the ratio of mean expression values (control/mutant). Negative values indicate reduction in Villin-Cre; Bmpr1a loxp/loxp intestines. NS, nonsignificant fold change (Mann Whitney test). of these cells was not affected by the loss of epithelial Bmp signaling. Statistical analysis revealed no significant decrease in the number of Paneth cells in the mutant animals (Figure 3K), suggesting that the determination of Paneth precursor cells is not affected by the loss of epithelial Bmp signaling. Thus, we hypothesized that Paneth cells were not differentiated fully in the mutant mice. Indeed, a reduction in the amount of Paneth cell secretory granule content was noted in mutant mice (Figure 3J). Finally, a decrease of 2.28-fold in matrilysin, 4.8-fold in lysozyme, and 5.24-fold in cryptdin messenger RNA (mrna) expressions, all important markers for Paneth cell functionality and maturation (Table 1), 20,21 was observed in the mutant mice. Expression of Indian Hedgehog, which negatively regulates the differentiation of Paneth precursor cells into fully mature cells, 22 as well as its downstream targets Gli-1 and Patched-1, were not affected by the loss of epithelial Bmp signaling (Table 1). These results support that Paneth cell terminal differentiation as well as maintenance can be linked to the instructive interaction of epithelial Bmp signaling. We next analyzed whether enteroendocrine cells were affected by the loss of epithelial Bmp signaling. Immunostaining with chromogranin A revealed fewer positive cells in Villin-Cre;Bmpr1a loxp/loxp mice (Figure 3F). We found a significant decrease of 75% of enteroendocrine cells per crypt villus axis in the mutant mice (Figure 3K), strongly suggesting that the enteroendocrine precursor is affected by the loss of epithelial Bmp signaling. Accordingly, we noted a 2.04-fold decrease in chromogranin A mrna levels in the mutant animals (Table 1). Comparative analysis by quantitative polymerase chain reaction of proenteroendocrine factors, such as Neurogenin 3 (Ngn3) and BETA2/NeuroD1, 13,23 revealed a significant decrease of 2.25-fold and 3.33-fold, respectively, in the mutant animals (Table 1). Thus, loss of epithelial Bmp signaling caused a reduction in Ngn3 and BETA2/NeuroD expression, leading to improper specification and differentiation of enteroendocrine cells. Absorptive Cell Differentiation Is Not Affected by Loss of Bmp Signaling We then investigated whether absorptive cell differentiation also was affected in Bmpr1a mutant mice. Differentiated cells of the absorptive lineage express specific markers such as intestinal fatty acid binding protein and sucrase-isomaltase. 1 In situ hybridization for sucrase-isomaltase (Figure 4A and B) and immunostaining for intestinal fatty acid binding protein (Figure 4C and D) revealed no modulation in these markers. No ultrastructural modification was noted between absorptive cells from mutant or control mice (Figure 4E and F) and no significant modulation of sucrase-isomaltase mrna levels was observed (Table 1). Thus, lack of terminal differentiation in mice with intestinal epithelial impaired Bmp signaling is restricted to cells of the secretory lineage. Bmp Signaling Does Not Affect the Secretory Precursor Cells Given the central role of hairy enhancer of split-1 and Math1 in intestinal epithelial specification, 24 and that Bmpr1a mutant mice have impaired terminal differentiation of the secretory cell lineage, we analyzed their levels of expression. A slight 1.95-fold decrease of Math1 but no significant modulation of hairy enhancer of split mrna levels were noted in the mutant animals (Table 1). Hence, loss of epithelial Bmp signaling is not sufficient to directly affect the secretory precursor cells or to change the fate of cells from the secretory to the absorptive cell lineage. Discussion Intercellular communications are decisive for intestinal morphogenesis as well as for the establishment and functional maintenance of the crypt villus axis. 3 5 Studies have identified the Bmp signaling pathway as a possible key mediator in crypt and villus morphogenesis as well as maintenance of intestinal stem cells. 4,7,8 However, the specific roles and contributions of the Bmp signaling pathway on one particular cell compartment compared with another in the maintenance of adult epithelial intestinal cell function has not yet been delineated. The current study generated a murine model with
7 September 2007 BMP SIGNALING IN INTESTINAL CELL LINEAGE 893 impaired Bmp signaling exclusively in the epithelial compartment of the intestine. In this study, we provide evidence that epithelial intestinal Bmp signaling plays a critical role in the maintenance of epithelial architecture within the crypt villus axis. We showed that loss of epithelial Bmp signaling leads to elongated villi and multiplication of the crypt units. The latter observation suggests a possible role for Bmp signaling in crypt fission. The role of Bmp signaling in stem cell renewal and consequently on crypt fission already has been addressed in previous work. 8 We have found the same occurrence in our Villin-Cre;Bmpr1a loxp/loxp mice, suggesting that the regulation of the crypt fission found at the bottom of the crypt unit is associated with epithelial Bmp signaling. Interestingly, and in major contrast with previous studies, 4,7,8 there is an absence of the de novo crypt phenomenon as well as polyp growth in Villin-Cre; Bmpr1a loxp/loxp mice even in 1-year-old animals. The key difference between the present study and those referred to previously is that herein, the Bmp signaling pathway is affected exclusively in the intestinal epithelium, whereas in previous studies both the epithelial and mesenchymal compartments were affected. These results suggest that the Bmp signaling pathway in the mesenchyme plays a significant role in crypt morphogenesis and juvenile polyposis syndrome whereas epithelial signalization plays a role in cell proliferation, stem cell renewal, 8 and maintenance of epithelial cell differentiation. We have investigated how epithelial Bmp signaling can suppress or restrain epithelial cell proliferation. Unexpectedly, we showed that the loss of epithelial Bmp signaling does not lead to an increase in -catenin nuclear expression, suggesting no increase in Wnt/ -catenin signaling activity. These results are contradictory to those recently reported by He et al, 8 who reported an increase in nuclear -catenin levels associated with an activation of target genes involved in cell-cycle progression. This increase in nuclear -catenin appears to be the consequence of the inhibition of phosphatase and tensin homologue deleted on chromosome 10 (PTEN) activity in the intestinal crypt epithelium of Mx1-Cre;Bmpr1a loxp/loxp mice, resulting in the activation of the phosphatidylinositol 3-kinase (PI3K)/Akt pathway and the inhibition of Gsk3 activity. He et al suggested that Bmp signaling inhibits intestinal stem cell self-renewal through 4 Figure 4. Epithelial Bmp signaling does not affect differentiation of absorptive cells. In situ hybridization revealed no variation in sucraseisomaltase mrna expression between the (A) control and (B) mutant animals. No variations in the level of intestinal fatty acid binding protein staining were seen between the (C) control and (D) mutant mice. (E and F) Electron microscopy confirmed there was no difference in the size, polarization, or morphology of absorbent cells. Magnification, 200 (A, B, C, and D). Bar 10 m (E and F).
8 894 AUCLAIR ET AL GASTROENTEROLOGY Vol. 133, No. 3 Figure 5. (A) Comparative representation of phenotypes observed in mice with either total or epithelium-specific loss of intestinal Bmp signaling. The total loss of Bmp in the intestine leads to multiplication and fission of crypt units, increased epithelial proliferation, deregulation of the Wnt/ -catenin pathway, presence of de novo crypts in the villus and polyps (combined phenotypes from Madison et al, 4 Haramis et al, 7 and He et al 8 ). The loss of Bmp signaling exclusively in the epithelium leads to increased length of villi, multiplication, and fission of crypt units; increased epithelial proliferation; and impaired terminal differentiation of cells from the secretory lineage. We found no evidence of de novo crypts and polyps as well as no modulation of the Wnt/ -catenin pathway. (B) Epithelial Bmp signaling action on the specification of intestinal secretory cell lineages. Epithelial Bmp signaling is not essential for maintenance or determination of the secretory precursor. It is needed for the proliferation of the enteroendocrine precursor cell through regulation of Ngn3 gene expression as well as their terminal differentiation given its effect on BETA2/Neurod1 gene expression. Epithelial Bmp signaling does not interfere with the maintenance or determination of the Paneth/goblet precursor cells but affects the gene expression necessary for their respective terminal differentiation, namely MMP7, Defcr, and Lyz for Paneth cells and Klf4 and Tff3 for goblet cells. EE, enteroendocrine; G, goblet; P, Paneth. activation of PTEN, leading to suppression of Wnt/ catenin signaling. However, their conclusions have raised some controversy in this field of research. Bjerknes and Cheng 25 have shown that the phosphorylated form of PTEN (p-pten)-positive cells identified by He et al 8 as intestinal stem cells are more likely to be the postmitotic secreting cells frequently found at the crypt base. After our result, we analyzed 2 known target genes of the Wnt/ -catenin pathway in our Villin-Cre;Bmpr1a loxp/loxp mice, namely c-myc and cyclin D1. Interestingly, we showed that c-myc is not modulated, whereas cyclin D1 is up-regulated in Villin-Cre;Bmpr1a loxp/loxp mice. Of note, cyclin D1 expression is enhanced not only by the Wnt/ -catenin pathway but also by many other signaling pathways such as the Ras/Erk/mitogene activated protein kinase pathway, 17 which also controls intestinal epithelial crypt cell proliferation. 26,27 Hence, one could speculate that epithelial Bmp signaling may inhibit proliferation of intestinal epithelial cells by interfering with the activation of this pathway instead of the Wnt/ -catenin pathway. In addition, epithelial Bmp signaling may directly control intestinal cell-cycle progression by modulating gene expression of cell inhibitors such as p21 Cip/Waf, p27 Kip, and p57 as reported previously in colon cancer cells 28 and in human keratinocytes. 29 Because the Bmpr1a gene ablation in the study with the Mx1-Cre;Bmpr1a loxp/loxp mice was in both the epithelial and stromal compartments of the gut, we think it is possible that the activation of the Wnt/ -catenin pathway is associated with the loss of mesenchymal Bmp signaling. Such action of the mesenchyme on the epithelial Wnt/ -catenin pathway has been shown previously and contributes to gastrointestinal epithelial tumor initiation. 3 Our study revealed an important and novel function for epithelial intestinal Bmp signaling in the proper terminal differentiation of cells from the intestinal secretory lineage. Our data suggest that key regulators of goblet cell terminal differentiation such as Tff3 and Klf4 are down-regulated in the mutant mice. Interestingly, Klf4 has been shown to be a target of Bmp signaling and to play a role in the maintenance of the vascular smooth muscle cell phenotype. 30 Here, we show that epithelial loss of Bmp signaling results in a significant decrease of Klf4 mrna and protein levels (data not shown), which leads to an important reduction in terminal differentiation of intestinal goblet cells. Our analysis exposed a dramatic decrease of 75% of enteroendocrine cells per crypt villus axis in the jejunum of Bmpr1a mutant mice as well as a significant decrease in proenteroendocrine factors such as Ngn3 and BETA2/NeuroD. These results strongly suggest that the enteroendocrine precursor cell, which is derived from the secretory precursor, is affected by the loss of epithelial Bmp signaling. This could be
9 September 2007 BMP SIGNALING IN INTESTINAL CELL LINEAGE 895 explained partially by the reduction in Ngn3 because this proenteroendocrine factor is known to play a role in the proliferation of the endocrine precursor. 13 Our data show a previously unknown role for epithelial Bmp signaling in enteroendocrine cell determination, as well as final specification through its involvement with the proenteroendocrine factors Ngn3 and BETA2/NeuroD. The Bmp signaling activity found at the bottom of the crypts thus far has been associated with stem cell renewal. 8 The differentiated Paneth cells found at the bottom of the crypts do not arise from direct differentiation of nearby stem or progenitor cells, but arrive there by the downward migration of already committed cells of the Paneth phenotype. 2 Our study shows that Paneth cell terminal differentiation can be linked to the instructive interaction of Bmp signaling. Because we observed no modulation in the number of Paneth cells in the mutant animals, this strongly suggests that the determination of Paneth precursor cells is not affected by the loss of epithelial Bmp signaling. Interestingly, Indian Hedgehog, the main representative of the hedgehog pathway in the gut, negatively regulates differentiation of Paneth precursor cells into fully mature cells. 22 The investigation of Indian Hedgehog expression revealed no significant modulation for this gene in mice with impaired epithelial Bmp signaling. Hence, Bmp signaling affects terminal differentiation and maturation of Paneth cells by a different mechanism than either the hedgehog or Wnt pathways. However, the decrease in matrilysin expression, which is also an important regulator of Paneth cell terminal differentiation, 20 could provide some explanation for a possible mechanism for the impaired Paneth cell terminal differentiation. Thus, our results reveal a distinct role for Bmp signaling at the bottom of the crypt in the terminal differentiation of Paneth cells. The phenotypes observed in the Bmpr1a mutant may suggest a greater role for Bmp signaling in the specification of intestinal precursor cells. Still, the change seen in Math1 expression in animals with impaired epithelial Bmp signaling might not be sufficient to directly affect precursor cells or to change the fate of cells from the secretory to the absorptive cell lineage. These observations clearly show that the loss of epithelial Bmp signaling affects the differentiation potential of the precursor cells, preventing them from fully acquiring their mature and functional phenotype. In summary, we show that epithelial Bmp signaling actively plays a role in terminal differentiation and maturation of cells from the secretory lineage. Most importantly, we have shown that the loss of Bmp signaling exclusively in the intestinal epithelial compartment is not responsible for the de novo crypt phenomenon, suggesting a greater role for mesenchymal Bmp signaling in juvenile polyposis syndrome. References 1. Ménard D, Beaulieu J-F, Boudreau F, et al. Gastrointestinal tract. In: Unsicker K, Krieglstein K, eds. Cell signaling and growth factors in development: from molecules to organogenesis. Volume 2. Weinheim: Wiley-Vch, 2006: Bjerknes M, Cheng H. Gastrointestinal stem cells. II. Intestinal stem cells. Am J Physiol 2005;289:G381 G Perreault N, Sackett SD, Katz JP, et al. Foxl1 is a mesenchymal modifier of min in carcinogenesis of stomach and colon. Genes Dev 2005;19: Madison BB, Braunstein K, Kuizon E, et al. Epithelial hedgehog signals pattern the intestinal crypt-villus axis. Development 2005;132: Roberts DJ. Molecular mechanisms of development of the gastrointestinal tract. Dev Dyn 2000;219: Karlsson L, Lindahl P, Heath JK, et al. Abnormal gastrointestinal development in PDGF-A and PDGFR-(alpha) deficient mice implicates a novel mesenchymal structure with putative instructive properties in villus morphogenesis. Development 2000;127: Haramis AP, Begthel H, van den Born M, et al. De novo crypt formation and juvenile polyposis on BMP inhibition in mouse intestine. Science 2004;303: He XC, Zhang J, Tong WG, et al. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat Genet 2004;36: Mishina Y. Function of bone morphogenetic protein signaling during mouse development. Front Biosci 2003;8:d855 d Kim BG, Li C, Qiao W, et al. Smad4 signalling in T cells is required for suppression of gastrointestinal cancer. Nature 2006;441: Madison BB, Dunbar L, Qiao XT, et al. Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J Biol Chem 2002;277: Mishina Y, Hanks MC, Miura S, et al. Generation of Bmpr/Alk3 conditional knockout mice. Genesis 2002;32: Lee CS, Perreault N, Brestelli JE, et al. Neurogenin 3 is essential for the proper specification of gastric enteroendocrine cells and the maintenance of gastric epithelial cell identity. Genes Dev 2002;16: Wilkinson DG. In situ hybridization: a practical approach. IRL Press, NY: Oxford University Press, Lantz KA, Vatamaniuk MZ, Brestelli JE, et al. Foxa2 regulates multiple pathways of insulin secretion. J Clin Invest 2004;114: Traber PG. Regulation of sucrase-isomaltase gene expression along the crypt-villus axis of rat small intestine. Biochem Biophys Res Commun 1990;173: Lavoie JN, L Allemain G, Brunet A, et al. Cyclin D1 expression is regulated positively by the p42/p44mapk and negatively by the p38/hogmapk pathway. J Biol Chem 1996;271: Katz JP, Perreault N, Goldstein BG, et al. The zinc-finger transcription factor Klf4 is required for terminal differentiation of goblet cells in the colon. Development 2002;129: Podolsky DK, Lynch-Devaney K, Stow JL, et al. Identification of human intestinal trefoil factor. Goblet cell-specific expression of a peptide targeted for apical secretion. J Biol Chem 1993;268: Weeks CS, Tanabe H, Cummings JE, et al. Matrix metalloproteinase-7 activation of mouse paneth cell pro-alpha-defensins: SER43 down arrow ILE44 proteolysis enables membrane-disruptive activity. J Biol Chem 2006;281: van Es JH, Jay P, Gregorieff A, et al. Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nat Cell Biol 2005;7:
10 896 AUCLAIR ET AL GASTROENTEROLOGY Vol. 133, No Varnat F, Heggeler BB, Grisel P, et al. PPARbeta/delta regulates paneth cell differentiation via controlling the hedgehog signaling pathway. Gastroenterology 2006;131: Mutoh H, Fung BP, Naya FJ, et al. The basic helix-loop-helix transcription factor BETA2/NeuroD is expressed in mammalian enteroendocrine cells and activates secretin gene expression. Proc Natl Acad Sci U S A 1997;94: Yang Q, Bermingham NA, Finegold MJ, et al. Requirement of Math1 for secretory cell lineage commitment in the mouse intestine. Science 2001;294: Bjerknes M, Cheng H. Re-examination of P-PTEN staining patterns in the intestinal crypt. Nat Genet 2005;37: Rivard N, Boucher MJ, Asselin C, et al. MAP kinase cascade is required for p27 downregulation and S phase entry in fibroblasts and epithelial cells. Am J Physiol 1999;277:C652 C Aliaga JC, Deschenes C, Beaulieu JF, et al. Requirement of the MAP kinase cascade for cell cycle progression and differentiation of human intestinal cells. Am J Physiol 1999;277:G631 G Beck SE, Jung BH, Del Rosario E, et al. BMP-induced growth suppression in colon cancer cells is mediated by p21(waf1) stabilization and modulated by RAS/ERK. Cell Signal 2007;19: Gosselet FP, Magnaldo T, Culerrier RM, et al. BMP2 and BMP6 control p57(kip2) expression and cell growth arrest/terminal differentiation in normal primary human epidermal keratinocytes. Cell Signal 2007;19: King KE, Iyemere VP, Weissberg PL, et al. Kruppel-like factor 4 (KLF4/GKLF) is a target of bone morphogenetic proteins and transforming growth factor beta 1 in the regulation of vascular smooth muscle cell phenotype. J Biol Chem 2003;278: Received September 21, Accepted May 17, Address requests for reprints to: Nathalie Perreault, Département d Anatomie et Biologie Cellulaire, Faculté de Médecine et des Sciences de la Santé, Université de Sherbrooke, e Ave Nord, Sherbrooke, Quebec, Canada, J1H 5N4. Nathalie.Perreault@ USherbrooke.ca; fax: (819) Supported by the Canadian Institutes of Health Research and by the Natural Sciences and Engineering Research Council of Canada ( ) (N.P.) and the Intramural Research Program of the National Institutes of Health, National Institute of Environmental Health Sciences (Y.M.). N.P. is a scholar from the Fonds de la recherche en santé du Québec. N.R. is a recipient of a Canadian Research Chair in Signaling and Digestive Physiopathology. The authors thank Pierre Chailler for his contribution to Figure 5, Elizabeth Herring for critical reading of the manuscript, Dr. Gumucio for providing the 12.4KbVilCre transgenic line, Dr. Gordon for providing the intestinal fatty acid binding protein antibody, and Denis Martel for assistance with electron microscopy. B.A.A. and Y.D.B. contributed equally to this work.
CHAPTER 6 SUMMARIZING DISCUSSION
 CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
Summary and Concluding Remarks
 Summary and Concluding Remarks Chapter 6 The intestinal epithelium provides an excellent model system for investigating molecular mechanisms regulating cell lineage establishment, stem cell proliferation,
Summary and Concluding Remarks Chapter 6 The intestinal epithelium provides an excellent model system for investigating molecular mechanisms regulating cell lineage establishment, stem cell proliferation,
Beuling _SupplFig. 1
 euling _SupplFig. 1 Gata6: F: 5 -GGGTGTGG-3 R: 5 -GTGGTTGT-3 hga: F: 5 -GT-3 R: 5 -TTTTTTTT-3 Gcg: F: 5 -TTGGGTGGTTTG-3 R: 5 -TGGGGTGTTGTGGTG-3 Pyy: F: 5 -GGGGGG-3 R: 5 -GGGGGGTG-3 Muc2: F: 5 -TTGTGTGTG-3
euling _SupplFig. 1 Gata6: F: 5 -GGGTGTGG-3 R: 5 -GTGGTTGT-3 hga: F: 5 -GT-3 R: 5 -TTTTTTTT-3 Gcg: F: 5 -TTGGGTGGTTTG-3 R: 5 -TGGGGTGTTGTGGTG-3 Pyy: F: 5 -GGGGGG-3 R: 5 -GGGGGGTG-3 Muc2: F: 5 -TTGTGTGTG-3
CHAPTER 3. Expression pattern of Wnt signaling components in the adult intestine. Gastroenterology 23: (2005)
 CHAPTER 3 Expression pattern of Wnt signaling components in the adult intestine Gastroenterology 23:1825-1833 (2005) Chapter 3 Expression pattern of Wnt signaling components in the adult intestine Alex
CHAPTER 3 Expression pattern of Wnt signaling components in the adult intestine Gastroenterology 23:1825-1833 (2005) Chapter 3 Expression pattern of Wnt signaling components in the adult intestine Alex
From crypt stem cell to colorectal cancer
 19 3 2007 6 Chinese Bulletin of Life Sciences Vol. 19, No. 3 Jun., 2007 1004-0374(2007)03-0321-05 ( 510405) Wnt Notch BMP R735.35; R730.21 A From crypt stem cell to colorectal cancer WEN Bin*, CHEN Weiwen
19 3 2007 6 Chinese Bulletin of Life Sciences Vol. 19, No. 3 Jun., 2007 1004-0374(2007)03-0321-05 ( 510405) Wnt Notch BMP R735.35; R730.21 A From crypt stem cell to colorectal cancer WEN Bin*, CHEN Weiwen
Genotype analysis by Southern blots of nine independent recombinated ES cell clones by
 Supplemental Figure 1 Selected ES cell clones show a correctly recombined conditional Ngn3 allele Genotype analysis by Southern blots of nine independent recombinated ES cell clones by hybridization with
Supplemental Figure 1 Selected ES cell clones show a correctly recombined conditional Ngn3 allele Genotype analysis by Southern blots of nine independent recombinated ES cell clones by hybridization with
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is
 Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Supplemental Figure 1: Lrig1-Apple expression in small intestine. Lrig1-Apple is observed at the crypt base and in insterstial cells of Cajal, but is not co-expressed in DCLK1-positive tuft cells. Scale
Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H.
 UvA-DARE (Digital Academic Repository) Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H. Link to publication Citation for published version (APA):
UvA-DARE (Digital Academic Repository) Lactase, sucrase-isomaltase, and carbamoyl phosphate synthase I expression in human intestine van Beers, E.H. Link to publication Citation for published version (APA):
icamp: Cancer biology tutorial II: recent developments in tumor biology, experimental methodology, and reference identification
 icamp: Cancer biology tutorial II: recent developments in tumor biology, experimental methodology, and reference identification Stem cells and the environment in the adenoma-carcinoma sequence (Medema,
icamp: Cancer biology tutorial II: recent developments in tumor biology, experimental methodology, and reference identification Stem cells and the environment in the adenoma-carcinoma sequence (Medema,
Anatomy & Histology of The Small intestine
 Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
Anatomy & Histology of The Small intestine Prof. Abdulameer Al-Nuaimi E-mail: a.al-nuaimi@sheffield.ac.uk E. mail: abdulameerh@yahoo.com Jejunum Ileum Histology: Duodenum, jejunum, and ileum
General Structure of Digestive Tract
 Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
SUPPLEMENTARY INFORMATION
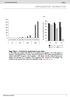 b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Principles of cell signaling Lecture 4
 Principles of cell signaling Lecture 4 Johan Lennartsson Molecular Cell Biology (1BG320), 2014 Johan.Lennartsson@licr.uu.se 1 Receptor tyrosine kinase-induced signal transduction Erk MAP kinase pathway
Principles of cell signaling Lecture 4 Johan Lennartsson Molecular Cell Biology (1BG320), 2014 Johan.Lennartsson@licr.uu.se 1 Receptor tyrosine kinase-induced signal transduction Erk MAP kinase pathway
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
ulcer healing role 118 Bicarbonate, prostaglandins in duodenal cytoprotection 235, 236
 Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Generation of a conditional allele of the Kindlin-2 gene. (A) A restriction map of the relevant genomic region of Kindlin-2 (top), the targeting construct
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Generation of a conditional allele of the Kindlin-2 gene. (A) A restriction map of the relevant genomic region of Kindlin-2 (top), the targeting construct
without LOI phenotype by breeding female wild-type C57BL/6J and male H19 +/.
 Sakatani et al. 1 Supporting Online Material Materials and methods Mice and genotyping: H19 mutant mice with C57BL/6J background carrying a deletion in the structural H19 gene (3 kb) and 10 kb of 5 flanking
Sakatani et al. 1 Supporting Online Material Materials and methods Mice and genotyping: H19 mutant mice with C57BL/6J background carrying a deletion in the structural H19 gene (3 kb) and 10 kb of 5 flanking
RAS Genes. The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes.
 ۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION. Androgen deprivation therapy is the most used treatment of de novo or recurrent
 CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION Stathmin in Prostate Cancer Development and Progression Androgen deprivation therapy is the most used treatment of de novo or recurrent metastatic PCa.
CHAPTER VII CONCLUDING REMARKS AND FUTURE DIRECTION Stathmin in Prostate Cancer Development and Progression Androgen deprivation therapy is the most used treatment of de novo or recurrent metastatic PCa.
Colonic polyps and colon cancer. Andrew Macpherson Director of Gastroentology University of Bern
 Colonic polyps and colon cancer Andrew Macpherson Director of Gastroentology University of Bern Improtance of the problem of colon cancers - Epidemiology Lifetime risk 5% Incidence/10 5 /annum (US Detroit
Colonic polyps and colon cancer Andrew Macpherson Director of Gastroentology University of Bern Improtance of the problem of colon cancers - Epidemiology Lifetime risk 5% Incidence/10 5 /annum (US Detroit
Small intestine. Small intestine
 General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
The Wnt target MASH-2 regulates expression of the putative intestinal specific stemcell markers Sox4 and Sfrp5.
 The Wnt target MASH-2 regulates expression of the putative intestinal specific stemcell markers Sox4 and Sfrp5. Submitted for publication: Irma M. Oving *, Marielle E. van Gijn *, Johan H. van Es, Marc
The Wnt target MASH-2 regulates expression of the putative intestinal specific stemcell markers Sox4 and Sfrp5. Submitted for publication: Irma M. Oving *, Marielle E. van Gijn *, Johan H. van Es, Marc
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
DIGESTIVE TRACT ESOPHAGUS
 DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
Supporting Information
 Supporting Information Fujishita et al. 10.1073/pnas.0800041105 SI Text Polyp Scoring. Intestinal polyps were counted as described (1). Briefly, the small and large intestines were excised, washed with
Supporting Information Fujishita et al. 10.1073/pnas.0800041105 SI Text Polyp Scoring. Intestinal polyps were counted as described (1). Briefly, the small and large intestines were excised, washed with
The Beauty of the Skin
 The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
The Beauty of the Skin Rose-Anne Romano, Ph.D Assistant Professor Department of Oral Biology School of Dental Medicine State University of New York at Buffalo The Big Question How do approximately 50 trillion
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
SUPPLEMENTARY FIGURES 1 Supplementary Figure 1, Adult hippocampal QNPs and TAPs uniformly express REST a-b) Confocal images of adult hippocampal mouse sections showing GFAP (green), Sox2 (red), and REST
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2607 Figure S1 Elf5 loss promotes EMT in mammary epithelium while Elf5 overexpression inhibits TGFβ induced EMT. (a, c) Different confocal slices through the Z stack image. (b, d) 3D rendering
DOI: 10.1038/ncb2607 Figure S1 Elf5 loss promotes EMT in mammary epithelium while Elf5 overexpression inhibits TGFβ induced EMT. (a, c) Different confocal slices through the Z stack image. (b, d) 3D rendering
EMBO REPORT SUPPLEMENTARY SECTION. Quantitation of mitotic cells after perturbation of Notch signalling.
 EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer.
 Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
AP VP DLP H&E. p-akt DLP
 A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
Small Intestine, Large Intestine and anal cannel
 Small Intestine, Large Intestine and anal cannel 32409 Small intestine Large intestine Small intestine General Structure of the Digestive Tract rat 32409 Epithelium with goblet cells and absorptive cells
Small Intestine, Large Intestine and anal cannel 32409 Small intestine Large intestine Small intestine General Structure of the Digestive Tract rat 32409 Epithelium with goblet cells and absorptive cells
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
SUPPLEMENTARY FIG. S2. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of dnscs.
 Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Targeting the cgmp Pathway to Treat Colorectal Cancer
 Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 29 Targeting the cgmp
Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 29 Targeting the cgmp
Dr Nadine Gravett School of Anatomical Sciences Room 2B10B
 Dr Nadine Gravett School of Anatomical Sciences Room 2B10B Nadine.Gravett@wits.ac.za Oral cavity Mechanical breakdown Formation of bolus Oesophagus Conduit from mouth to stomach Stomach Digestion Temporary
Dr Nadine Gravett School of Anatomical Sciences Room 2B10B Nadine.Gravett@wits.ac.za Oral cavity Mechanical breakdown Formation of bolus Oesophagus Conduit from mouth to stomach Stomach Digestion Temporary
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS
 ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
ODONTOGENESIS- A HIGHLY COMPLEX CELL-CELL INTERACTION PROCESS AMBRISH KAUSHAL, MALA KAMBOJ Department of Oral and Maxillofacial Pathology Career Post Graduate Institute of Dental Sciences and Hospital
Signaling. Dr. Sujata Persad Katz Group Centre for Pharmacy & Health research
 Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
SUPPLEMENTARY DATA. Supplementary Table 2. Antibodies used for Immunofluoresence. Supplementary Table 3. Real-time PCR primer sequences.
 Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
Enterprise Interest Nothing to declare
 Enterprise Interest Nothing to declare Update of mixed tumours of the GI tract, the pancreas and the liver Introduction to the concept of mixed tumours and clinical implication Jean-Yves SCOAZEC Surgical
Enterprise Interest Nothing to declare Update of mixed tumours of the GI tract, the pancreas and the liver Introduction to the concept of mixed tumours and clinical implication Jean-Yves SCOAZEC Surgical
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
11 -Hydroxysteroid dehydrogenase, expression, corticotroph adenomas 68 70
 Subject Index Activin activity decrease, gonadotroph 78 pituitary tumorigenesis, role 99 Adenoma, see also specific cell types angiogenesis, see Angiogenesis clonality, see Clonality, pituitary tumors
Subject Index Activin activity decrease, gonadotroph 78 pituitary tumorigenesis, role 99 Adenoma, see also specific cell types angiogenesis, see Angiogenesis clonality, see Clonality, pituitary tumors
Alimentary Canal (I)
 Alimentary Canal (I) Esophagus and Stomach (Objectives) By the end of this lecture, the student should be able to discuss the microscopic structure in correlation with the function of the following organs:
Alimentary Canal (I) Esophagus and Stomach (Objectives) By the end of this lecture, the student should be able to discuss the microscopic structure in correlation with the function of the following organs:
INSIGHTS INTO THE DEVELOPMENT OF GASTROINTESTINAL BRUNNER S GLANDS: CRITICAL STEM CELLS AND DIFFERENTIATION FACTORS
 INSIGHTS INTO THE DEVELOPMENT OF GASTROINTESTINAL BRUNNER S GLANDS: CRITICAL STEM CELLS AND DIFFERENTIATION FACTORS by MICHAEL S. PARAPPILLY A THESIS Presented to the Department of Human Physiology and
INSIGHTS INTO THE DEVELOPMENT OF GASTROINTESTINAL BRUNNER S GLANDS: CRITICAL STEM CELLS AND DIFFERENTIATION FACTORS by MICHAEL S. PARAPPILLY A THESIS Presented to the Department of Human Physiology and
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE
 LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
F-box and WD Repeat Domain-Containing 7 Regulates Intestinal Cell Lineage Commitment and Is a Haploinsufficient Tumor Suppressor
 GASTROENTEROLOGY 2010;139:929 941 F-box and WD Repeat Domain-Containing 7 Regulates Intestinal Cell Lineage Commitment and Is a Haploinsufficient Tumor Suppressor ROCIO SANCHO,* ANETT JANDKE,* HAYLEY DAVIS,
GASTROENTEROLOGY 2010;139:929 941 F-box and WD Repeat Domain-Containing 7 Regulates Intestinal Cell Lineage Commitment and Is a Haploinsufficient Tumor Suppressor ROCIO SANCHO,* ANETT JANDKE,* HAYLEY DAVIS,
Dana Alrafaiah. Dareen Abu Shalbak. Mohammad Almuhtaseb. 1 P a g e
 2 Dana Alrafaiah Dareen Abu Shalbak Mohammad Almuhtaseb 1 P a g e Esophagus: A muscular tube that is 25 cm long, but if measured from the incisors it would be 45cm long. Extends from C6 of cervical vertebra,
2 Dana Alrafaiah Dareen Abu Shalbak Mohammad Almuhtaseb 1 P a g e Esophagus: A muscular tube that is 25 cm long, but if measured from the incisors it would be 45cm long. Extends from C6 of cervical vertebra,
Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 )
 770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
Cellular Physiology and Biochemistry
 Original Paper 2015 The Author(s). 2015 Published The Author(s) by S. Karger AG, Basel Published online: November 27, 2015 www.karger.com/cpb Published by S. Karger AG, Basel 2194 1421-9778/15/0376-2194$39.50/0
Original Paper 2015 The Author(s). 2015 Published The Author(s) by S. Karger AG, Basel Published online: November 27, 2015 www.karger.com/cpb Published by S. Karger AG, Basel 2194 1421-9778/15/0376-2194$39.50/0
27
 26 27 28 29 30 31 32 33 34 35 Diagnosis:? Diagnosis: Juvenile Polyposis with BMPR1A Mutation 36 Juvenile Polyposis Syndrome Rare Autosomal Dominant Disorder with Multiple Juvenile Polyps in GI Tract Juvenile
26 27 28 29 30 31 32 33 34 35 Diagnosis:? Diagnosis: Juvenile Polyposis with BMPR1A Mutation 36 Juvenile Polyposis Syndrome Rare Autosomal Dominant Disorder with Multiple Juvenile Polyps in GI Tract Juvenile
Development of the Rodent Gastrointestinal Tract:
 White Paper Development of the Rodent Gastrointestinal Tract: The regulation of the development of the GI tract is unique and complex. With respect to the GI tract there are several considerations that
White Paper Development of the Rodent Gastrointestinal Tract: The regulation of the development of the GI tract is unique and complex. With respect to the GI tract there are several considerations that
BIO360 Fall 2013 Quiz 1
 BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence
 Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Cell Death & Renewal (part 2)
 17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
Anatomy of the Intes.ne: Epithelium, Lympha.cs, Vessels and Nerves
 Master Course Gastroenterology 2015 Anatomy of the Intes.ne: Epithelium, Lympha.cs, Vessels and Nerves Dr. Stephanie Ganal Department Klinische Forschung University of Bern stephanie.ganal@dkf.unibe.ch
Master Course Gastroenterology 2015 Anatomy of the Intes.ne: Epithelium, Lympha.cs, Vessels and Nerves Dr. Stephanie Ganal Department Klinische Forschung University of Bern stephanie.ganal@dkf.unibe.ch
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature11463 %Sox17(+) 9 8 7 6 5 4 3 2 1 %Sox17(+) #Sox17(+) d2 d4 d6 d8 d1 d12 d14 d18 25 2 15 1 5 Number of Sox17(+) cells X 1 Supplementary Figure 1: Expression of
SUPPLEMENTARY INFORMATION doi:1.138/nature11463 %Sox17(+) 9 8 7 6 5 4 3 2 1 %Sox17(+) #Sox17(+) d2 d4 d6 d8 d1 d12 d14 d18 25 2 15 1 5 Number of Sox17(+) cells X 1 Supplementary Figure 1: Expression of
Hyperplastische Polyps Innocent bystanders?
 Hyperplastische Polyps Innocent bystanders?? K. Geboes P th l i h O tl dk d Pathologische Ontleedkunde, KULeuven Content Historical Classification Relation Hyperplastic polyps carcinoma The concept cept
Hyperplastische Polyps Innocent bystanders?? K. Geboes P th l i h O tl dk d Pathologische Ontleedkunde, KULeuven Content Historical Classification Relation Hyperplastic polyps carcinoma The concept cept
COPD lungs show an attached stratified mucus layer that separate. bacteria from the epithelial cells resembling the protective colonic
 COPD lungs show an attached stratified mucus layer that separate bacteria from the epithelial cells resembling the protective colonic mucus SUPPLEMENTARY TABLES AND FIGURES Tables S1 S8, page 1 and separate
COPD lungs show an attached stratified mucus layer that separate bacteria from the epithelial cells resembling the protective colonic mucus SUPPLEMENTARY TABLES AND FIGURES Tables S1 S8, page 1 and separate
The Digestive System Laboratory
 The Digestive System Laboratory 1 The Digestive Tract The alimentary canal is a continuous tube stretching from the mouth to the anus. Liver Gallbladder Small intestine Anus Parotid, sublingual, and submaxillary
The Digestive System Laboratory 1 The Digestive Tract The alimentary canal is a continuous tube stretching from the mouth to the anus. Liver Gallbladder Small intestine Anus Parotid, sublingual, and submaxillary
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
DIGESTIVE. CHAPTER 17 Lecture: Part 1 Part 2 BIO 212: ANATOMY & PHYSIOLOGY II
 BIO 212: ANATOMY & PHYSIOLOGY II CHAPTER 17 Lecture: DIGESTIVE Part 1 Part 2 Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. SMALL INTESTINE DUODENUM > JEJUNUM
BIO 212: ANATOMY & PHYSIOLOGY II CHAPTER 17 Lecture: DIGESTIVE Part 1 Part 2 Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. SMALL INTESTINE DUODENUM > JEJUNUM
Supplemental Data Tamoxifen administration to Vil-Scap- mice.
 Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
Nutrition & Gut Immunity
 Nutrition & Gut Immunity Kenneth A. Kudsk, MD FACS Professor of Surgery University of Wisconsin - Madison Madison, Wisconsin No conflicts of interest to disclose % Survival Survival vs. Malnutrition (after
Nutrition & Gut Immunity Kenneth A. Kudsk, MD FACS Professor of Surgery University of Wisconsin - Madison Madison, Wisconsin No conflicts of interest to disclose % Survival Survival vs. Malnutrition (after
Megan Lawless. Journal Club. January 20 th, 2011
 Megan Lawless Journal Club January 20 th, 2011 Gut-expressed gustducin and taste receptors regulate secretion of glucagon-like peptide-1 Proceedings of the National Academy of Sciences September 2007 Abstract
Megan Lawless Journal Club January 20 th, 2011 Gut-expressed gustducin and taste receptors regulate secretion of glucagon-like peptide-1 Proceedings of the National Academy of Sciences September 2007 Abstract
Gastric adenocarcinoma of the
 Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse
 Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22
 Supplementary Information High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22 Max Gulhane 1, Lydia Murray 1, Rohan Lourie 1, Hui Tong 1, Yong H. Sheng 1, Ran
Supplementary Information High Fat Diets Induce Colonic Epithelial Cell Stress and Inflammation that is Reversed by IL-22 Max Gulhane 1, Lydia Murray 1, Rohan Lourie 1, Hui Tong 1, Yong H. Sheng 1, Ran
Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Basement membrane: What is the basement membrane? - It is a layer of ECM separating the epithelial cells from the underlying connective tissue Basement
Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Basement membrane: What is the basement membrane? - It is a layer of ECM separating the epithelial cells from the underlying connective tissue Basement
Dual epithelial and immune cell function of Dvl1 regulates gut microbiota composition and intestinal homeostasis
 Dual epithelial and immune cell function of Dvl1 regulates gut microbiota composition and intestinal homeostasis Haim Belinson,, Richard M. Locksley, Ophir D. Klein JCI Insight. 2016;1(10):e85395. https://doi.org/10.1172/jci.insight.85395.
Dual epithelial and immune cell function of Dvl1 regulates gut microbiota composition and intestinal homeostasis Haim Belinson,, Richard M. Locksley, Ophir D. Klein JCI Insight. 2016;1(10):e85395. https://doi.org/10.1172/jci.insight.85395.
C. elegans Embryonic Development
 Autonomous Specification in Tunicate development Autonomous & Conditional Specification in C. elegans Embryonic Development Figure 8.36 Bilateral Symmetry in the Egg of the Tunicate Styela partita Fig.
Autonomous Specification in Tunicate development Autonomous & Conditional Specification in C. elegans Embryonic Development Figure 8.36 Bilateral Symmetry in the Egg of the Tunicate Styela partita Fig.
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and
 Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Am J Physiol Gastrointest Liver Physiol 298: G504 G517, First published February 4, 2010; doi: /ajpgi
 Am J Physiol Gastrointest Liver Physiol 298: G504 G517, 2010. First published February 4, 2010; doi:10.1152/ajpgi.00265.2009. Cooperation between HNF-1, Cdx2, and GATA-4 in initiating an enterocytic differentiation
Am J Physiol Gastrointest Liver Physiol 298: G504 G517, 2010. First published February 4, 2010; doi:10.1152/ajpgi.00265.2009. Cooperation between HNF-1, Cdx2, and GATA-4 in initiating an enterocytic differentiation
The role of Hepatitis C Virus in hepatocarcinogenesis
 The role of Hepatitis C Virus in hepatocarcinogenesis Laura Beretta Fred Hutchinson Cancer Research Center l8 Incidence and mortality of the five most common cancers worldwide, 2000 Incidence Lung Breast
The role of Hepatitis C Virus in hepatocarcinogenesis Laura Beretta Fred Hutchinson Cancer Research Center l8 Incidence and mortality of the five most common cancers worldwide, 2000 Incidence Lung Breast
Molecular Regulation of Intestinal Stem Cells
 Molecular Regulation of Intestinal Stem Cells Review Tulay Bayram Biomedical Sciences University of Utrecht Table of contents Abstract... 3 1. INTRODUCTION... 4 1.1 Structure of the small intestine and
Molecular Regulation of Intestinal Stem Cells Review Tulay Bayram Biomedical Sciences University of Utrecht Table of contents Abstract... 3 1. INTRODUCTION... 4 1.1 Structure of the small intestine and
INTRODUCTION: The intestinal epithelium is one of the most rapidly dividing tissues in the
 INTRODUCTION: The intestinal epithelium is one of the most rapidly dividing tissues in the mammalian organism, with near complete replacement of the epithelial monolayer every 7-10 days (Wright et al.,
INTRODUCTION: The intestinal epithelium is one of the most rapidly dividing tissues in the mammalian organism, with near complete replacement of the epithelial monolayer every 7-10 days (Wright et al.,
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
Lab activity manual - Histology of the digestive system. Lab activity 1: esophagus stomach - small intestines
 Lab activity manual - Histology of the digestive system Jeanne Adiwinata Pawitan Prerequisite: Histology of the 4 basic tissues In this module we learn about the histology of the digestive system, from
Lab activity manual - Histology of the digestive system Jeanne Adiwinata Pawitan Prerequisite: Histology of the 4 basic tissues In this module we learn about the histology of the digestive system, from
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU. PhD THESIS Summary
 ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU PhD THESIS Summary INVOLVEMENT OF ALARMIN HMGB1 IN INFLAMMATORY PROCESSES ASSOCIATED WITH VASCULAR DYSFUNCTION IN HYPERLIPIDEMIA
ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU PhD THESIS Summary INVOLVEMENT OF ALARMIN HMGB1 IN INFLAMMATORY PROCESSES ASSOCIATED WITH VASCULAR DYSFUNCTION IN HYPERLIPIDEMIA
Div. of Endocrinology and Metabolism Konyang University, School of Medicine. Byung-Joon Kim M.D., Ph.D.
 Diabetes and Thyroid Center Div. of Endocrinology and Metabolism Konyang University, School of Medicine Byung-Joon Kim M.D., Ph.D. Background Incretin effect and GLP 1 Receptor in L cells TGR5 receptor
Diabetes and Thyroid Center Div. of Endocrinology and Metabolism Konyang University, School of Medicine Byung-Joon Kim M.D., Ph.D. Background Incretin effect and GLP 1 Receptor in L cells TGR5 receptor
Hunk is required for HER2/neu-induced mammary tumorigenesis
 Research article Hunk is required for HER2/neu-induced mammary tumorigenesis Elizabeth S. Yeh, 1 Thomas W. Yang, 1 Jason J. Jung, 1 Heather P. Gardner, 1 Robert D. Cardiff, 2 and Lewis A. Chodosh 1 1 Department
Research article Hunk is required for HER2/neu-induced mammary tumorigenesis Elizabeth S. Yeh, 1 Thomas W. Yang, 1 Jason J. Jung, 1 Heather P. Gardner, 1 Robert D. Cardiff, 2 and Lewis A. Chodosh 1 1 Department
HISTOLOGY. GIT Block 432 Histology Team. Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih
 HISTOLOGY Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih Color Guide: Black: Slides. Red: Important. Green: Doctor s notes. Blue: Explanation.
HISTOLOGY Lecture 1: Alimentary Canal (1) (Esophagus & Stomach) Done by: Ethar Alqarni Reviewed by: Ibrahim Alfuraih Color Guide: Black: Slides. Red: Important. Green: Doctor s notes. Blue: Explanation.
RECENT ADVANCES IN BASIC SCIENCE STEM CELL IN GASTROINTESTINAL STRUCTURE AND NEOPLASTIC DEVELOPMENT
 See end of article for authors affiliations Correspondence to: M Brittan, Histopathology Unit, Cancer Research UK, 44 Lincoln s Inn Fields, London WC2A 3PX, UK; mairi.brittan@cancer.org.uk RECENT ADVANCES
See end of article for authors affiliations Correspondence to: M Brittan, Histopathology Unit, Cancer Research UK, 44 Lincoln s Inn Fields, London WC2A 3PX, UK; mairi.brittan@cancer.org.uk RECENT ADVANCES
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
