Gonadal Developmental Systems Biology
|
|
|
- Randolf Nelson
- 5 years ago
- Views:
Transcription
1 Spring 2018 Systems Biology of Reproduction Discussion Outline Gonadal Developmental Systems Biology Michael K. Skinner Biol 475/575 CUE 418, 10:35-11:50 am, Tuesday & Thursday February 15, 2018 Week 6 Gonadal Developmental Systems Biology Primary Papers: 1. Jameson, et al. (2012) PLoS Genetics 8(3):e Rakoczy, et al. (2013) Biol Reprod 88(6):143, Nilsson, et al. (2013) BMC Genomics 14:496 Discussion Student 15: Reference 1 above What is the technical approach? What sex specific transcriptome observations were made? Why is the female fate a default? Student 16: Reference 2 above What are the technology used and objectives? What did the expression patterns show? What basic information on Leydig cells was obtained? Student 18: Reference 3 above What is the experimental and systems approach? What is a cluster analysis? What gene networks were identified for primordial follicle assembly?
2 Temporal Transcriptional Profiling of Somatic and Germ Cells Reveals Biased Lineage Priming of Sexual Fate in the Fetal Mouse Gonad Samantha A. Jameson, Anirudh Natarajan, Jonah Cool, Tony DeFalco, Danielle M. Maatouk, Lindsey Mork, Steven C. Munger, Blanche Capel* Department of Cell Biology, Duke University Medical Center, Durham, North Carolina, United States of America Abstract The divergence of distinct cell populations from multipotent progenitors is poorly understood, particularly in vivo. The gonad is an ideal place to study this process, because it originates as a bipotential primordium where multiple distinct lineages acquire sex-specific fates as the organ differentiates as a testis or an ovary. To gain a more detailed understanding of the process of gonadal differentiation at the level of the individual cell populations, we conducted microarrays on sorted cells from XX and XY mouse gonads at three time points spanning the period when the gonadal cells transition from sexually undifferentiated progenitors to their respective sex-specific fates. We analyzed supporting cells, interstitial/stromal cells, germ cells, and endothelial cells. This work identified genes specifically depleted and enriched in each lineage as it underwent sex-specific differentiation. We determined that the sexually undifferentiated germ cell and supporting cell progenitors showed lineage priming. We found that germ cell progenitors were primed with a bias toward the male fate. In contrast, supporting cells were primed with a female bias, indicative of the robust repression program involved in the commitment to XY supporting cell fate. This study provides a molecular explanation reconciling the female default and balanced models of sex determination and represents a rich resource for the field. More importantly, it yields new insights into the mechanisms by which different cell types in a single organ adopt their respective fates. Citation: Jameson SA, Natarajan A, Cool J, DeFalco T, Maatouk DM, et al. (2012) Temporal Transcriptional Profiling of Somatic and Germ Cells Reveals Biased Lineage Priming of Sexual Fate in the Fetal Mouse Gonad. PLoS Genet 8(3): e doi: /journal.pgen Editor: Gregory S. Barsh, Stanford University School of Medicine, United States of America Received November 30, 2011; Accepted January 17, 2012; Published March 15, 2012 Copyright: ß 2012 Jameson et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: NIH/GUDMAP ( provided funding to the laboratory (as a subcontract to GUDMAP U01 DK070219) and influenced the design of the data collection as part of the GUDMAP initiative to establish a public resource for gene expression in the urogenital tract. GUDMAP did not influence the analysis of the data, the decision to publish, or the preparation of this manuscript. BC and SAJ were funded by this GUDMAP grant. SCM, AN, and DMM were funded by NIH-P50GM ( BC, SAJ, DMM, and LM were funded by NIH-HD TD was funded by an NIH-NRSA post-doctoral fellowship (F32 HD ). JC was supported by a March of Dimes ( research grant to BC (MOD 1-FY10-355) and an American Heart Association ( predoctoral fellowship. These funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing Interests: The authors have declared that no competing interests exist. * blanche.capel@duke.edu Introduction Little is known about how the transcriptome of an undifferentiated progenitor is poised between two fates and resolves to one of two stable, differentiated outcomes during the process of cell fate determination. The bipotential gonad is a unique system for studying cell fate decisions during mammalian organ development. In mice, the gonads arise around embryonic day 10 (E10.0). For the first 36 hours the gonad primordia harbor the potential to become testes or ovaries irrespective of genetic sex. The process by which the bipotential gonad adopts the ovarian or testicular fate is known as primary sex determination, and involves a binary fate decision within cells of each gonadal lineage. The early gonad is composed of four lineages: supporting cells, interstitial/stromal cells, germ cells, and endothelial cells. The determinant of gonadal fate in mammals is the Y-chromosome gene Sry. In XY individuals, expression of Sry in supporting cell progenitors triggers commitment to a testicular ( male ) fate, whereas the absence of Sry expression in XX supporting cells results in ovarian ( female ) development [1,2]. Supporting cells and interstitial/stromal cells arise within the urogenital ridge from a common mesodermal progenitor, while the primordial germ cells and endothelial cells migrate into the developing gonad [3 6]. Despite their distinct origins, cells of each lineage in the early gonad are bipotential progenitors capable of adopting either a male or female fate, which they do in a coordinated manner to form a functional testis or ovary [7 9]. Temporal examination of the transcriptomes of these diverse progenitors as they make their parallel, binary fate decisions provides an opportunity to understand how cell fate decisions are made in the context of organ development. Some gonadal lineages have been studied at the transcriptome level in independent experiments [10 15], resulting in the identification of genes that are up-regulated in a sex- or (in some cases) lineage-specific manner. However, the molecular relationship between the somatic lineages (i.e., supporting cells versus interstitial/stromal cells) has never been examined, as these lineages were not separated in previous studies. Previous studies did not fully characterize the undifferentiated progenitors or the temporal sequence for the divergence of the multiple progenitors PLoS Genetics 1 March 2012 Volume 8 Issue 3 e
3 Lineage Priming of Sexual Fate in Gonadal Cells Author Summary How cells diverge from a common progenitor and adopt specific fates is still poorly understood. We analyzed gene expression profiles in the distinct cell lineages of the gonad over the period when sex determination occurs. The undifferentiated progenitor cells expressed genes characteristic of both sexual fates, explaining the plasticity of the gonadal cells to differentiate as male or female cell types. The establishment of sex-specific fate in both the germ cells and somatic cells involved activation of some genes; but, importantly, we show that an active repression of genes associated with the alternative pathway is also a characteristic of cell fate commitment. Although germ cell progenitors expressed genes associated with both possible fates, genes characteristic of the male fate were overrepresented in the progenitors, giving them a male bias. However, in somatic cell progenitors, which control sex determination, genes associated with the female fate were over-represented. These results suggest an explanation for why the female fate is the developmental default for the gonad, and they advance our understanding of how complex transcriptional networks regulate fate determination during organ development. to their sexual fates. Furthermore, other potentially important transcriptional patterns associated with differentiation and fate commitment, such as the specific transcript depletion previously noted in the Arabidopsis root and in early primordial germ cells [16,17], have not been characterized. As part of the GenitoUrinary Molecular Anatomy Project (GUDMAP, we undertook a comprehensive transcriptome analysis of the four principle gonadal lineages in XX and XY gonads at three time points, spanning the period from the undifferentiated bipotential stage until the cells adopt sex-specific fates. While this type of comprehensive transcriptome analysis has been performed in other developing systems [16,18], the relative simplicity of the gonad and the theoretical framework for sex determination allowed us to extend our analysis to test distinct models for the process of cell fate determination, and to evaluate the fit of these models to the theories of sex determination that have been proposed in the past 50 years. To explore how the cells of the gonad adopt their sex-specific fates, we considered the various theories that have been proposed for gonadal sex determination. It has been suggested that the female fate is the default state because expression of Sry is required to divert the cells to the male fate [19 21]. The concept of a female default state originated in the secondary sex determination literature [19], but that language crept into the field of primary sex determination and became a way to conceptualize female gonad development [20,21]. Others proposed that a female-promoting Z gene normally blocks an underlying male developmental program and that Z is itself blocked by Sry [22]. Still others have proposed that both the female and male programs require an active switch to initiate differentiation from their initially bipotential state (i.e., that there is an ovarydetermining gene as well as a testis-determining gene ) [23]. More recently, it has been suggested that the gonad is balanced between the male and female fates by antagonistic signaling pathways [24 27]. We wanted to determine whether one of these models could describe the differentiation of the gonadal cells at the level of the transcriptome. Therefore, we re-framed and logically extended these models so that we could test them against our transcriptional data, and we used the concept of lineage priming to do so. Studies suggest that multipotent cells are not a blank slate, but rather are lineage primed by expressing markers of all potential fates they can adopt [28 35]. During their differentiation, multipotent cells repress markers of specific fates that were not adopted while maintaining gene expression associated with the fate that was adopted [28 35]. A similar phenomenon has also been observed in the early embryo, where individual blastomeres express transcripts that later become restricted to the specific lineages of the blastocyst [36 38]. It is possible for progenitor cells to be equally balanced between their multiple fates, expressing similar numbers of genes associated with each alternative differentiated fate. However, the progenitors need not have all differentiation programs equally represented [28]. Instead a progenitor may show biased priming if markers characteristic of one of its possible fates predominate, indicating the closer relationship of the progenitor to that fate. We investigated the transcriptional profiles of the gonadal cell lineages as they differentiate. Interestingly, we identified different variations of biased lineage priming: while germ cells showed male-biased priming, supporting cells showed female-biased priming. This study provides a molecular explanation reconciling the female default and balanced models of sex determination and represents a rich resource for the field. In addition, it affords insight into the mechanisms by which different cell types in a single organ adopt their respective fates. Results Sorted cell microarrays accurately reflect known gene expression patterns We quantified global gene expression in four lineages of the XX and XY developing mouse gonad at E11.5, E12.5, and E13.5. To isolate individual lineages, we utilized mouse lines expressing fluorescent cell-specific markers (Figure 1A, Figure S1). The cells from separately pooled XX and XY gonads were isolated by fluorescence-activated cell sorting (FACS). Sry-EGFP [7] and Sox9- ECFP [39] were used as markers for supporting cells (see Materials and Methods for a full explanation). XY interstitial cells and XX stromal cells were isolated using Mafb-EGFP [40,41]. The XY interstitial cells are excluded from testis cords and give rise to steroidogenic Leydig cells [41]. The XX stroma is not defined morphologically, but for the purposes of this analysis, is defined as the population labeled with Mafb-EGFP. Germ cells were isolated using Oct4-EGFP [42], and endothelial cells were isolated using Flk1-mCherry [43,44]. In general, cells were pooled from gonads of multiple embryos on non-inbred genetic backgrounds (see Materials and Methods). RNA purified from each XX and XY cell population was used to measure transcript abundance with Affymetrix Mouse Genechip Gene 1.0 ST Arrays. We produced 3 biological replicates for each population. The data are available in GEO (accession number GSE27715) and at org/. RMA normalized values used in our analysis are provided with the capability to generate an expression graph for any gene, as a user-friendly resource for the community (Dataset S1). We also isolated cells from the asma-eyfp transgenic mouse [45] with the expectation that this reporter would label a larger population of cells in the interstitium and stroma (Figure S2A). While this population transcriptionally resembled the interstitial/stromal population isolated with the Mafb-EGFP line (data not shown), unlike the Mafb-EGFP positive cells, the asma-eyfp cells also expressed high levels of Sry at E11.5, a gene predicted to be specific to XY supporting cells [46] (Figure S2B). Indeed, E11.5 asma-eyfp cells stained positive for both SRY and PLoS Genetics 2 March 2012 Volume 8 Issue 3 e
4 Lineage Priming of Sexual Fate in Gonadal Cells Figure 1. Sorted cell lineages and microarray validation. (A) Illustration of the developing XX and XY gonad with supporting cells (blue), interstitial/stromal cells (purple), germ cells (green), and endothelial cells (red). (B, C, and F) Graphs of the log-transformed, normalized intensity values from the microarrays for control genes known to be specific to each lineage. The color for each lineage is conserved in all figures and matches the illustration (A), with XX (R) values shown as dashed lines, and XY (=) values shown as solid lines. The error bars are standard error of the mean ( standard error ) of the log transformed values. The Y-axis scale differs for each graph because each transcript cluster has its own intensity range. (B) The control genes were found in the expected lineage, except for (C) genes characteristic of Leydig cells. Leydig cell genes were highly expressed in both the interstitium (as expected) and the endothelial cell fraction. (D) Immunofluorescence of E13.5 XY gonads with Flk1-mCherry (red), PECAM1 (germ and endothelial cells, blue), and 3b-HSD (Leydig cells, green). Arrowheads indicate Flk1-mCherry and PECAM1 double positive endothelial cells. Arrows indicate Flk1-mCherry positive, PECAM1 negative cells that were positive for 3b-HSD, confirming aberrant reporter expression in some Leydig cells. Asterisks indicate germ cells positive for PECAM1 alone. Scale bar = 25 mm. (E) The XY interstitial cells have very low expression of the endogenous Flk1 (Kdr) transcript at E13.5, supporting our conclusion that the Flk1-mCherry transgene is aberrantly expressed in Leydig cells. doi: /journal.pgen g001 SOX9 protein (Figure S2C). Consequently, at least early in development, it appears that asma-eyfp labels a heterogeneous population of cells containing supporting cells as well as interstitial/stromal cells. While this finding may have biological significance, it could also result from leaky expression of the asma-eyfp transgene. Thus, we excluded the asma-eyfp data from this analysis but have made the data available with the rest of the microarrays. PLoS Genetics 3 March 2012 Volume 8 Issue 3 e
5 Lineage Priming of Sexual Fate in Gonadal Cells To validate the cell sorting and microarray data, we examined the expression of genes known to be specific to each of the cell populations (Figure 1B). The expression of each control gene was consistent with previous reports: Dhh (desert hedgehog) was enriched in XY supporting cells [10], Wnt4 (wingless-related MMTV integration site 4) was enriched in XX supporting cells [10], Inhba (inhibin beta-a) was enriched in the XY interstitium [47,48], Nanos2 (nanos homolog 2) was enriched in XY germ cells [49], Stra8 (stimulated by retinoic acid gene 8) was enriched in XX germ cells [50], and Flt1 (FMS-like tyrosine kinase 1, VEGFR1) was enriched in endothelial cells [51]. Therefore, the microarrays on the sorted cell populations accurately reflected gene expression patterns that were previously characterized for each of the cell lineages. An unexpected expression pattern was observed in XY Flk1- mcherry-positive cells (Figure 1C). We found that genes expressed in Leydig cells, such as Cyp11a1 (Scc) [52], were enriched in both XY interstitial (containing steroidogenic Leydig cells) and Flk1- mcherry-positive endothelial populations. This was surprising given that steroidogenic enzymes have not previously been reported in endothelial cells [41]. When we immunostained the Flk1-mCherry-positive cells purified by FACS for a Leydig cell marker, we found that Leydig cells were present in this sorted population (data not shown). To investigate the basis for this finding, we stained E13.5 Flk1-mCherry gonads with the vascular marker PECAM1 and an antibody against the Leydig cell marker 3b-HSD. We found Flk1-mCherry-positive cells that were negative for the vascular marker, but positive for the Leydig marker (Figure 1D). This suggests that the Flk1-mCherry transgene was ectopically expressed in some Leydig cells. Because the corresponding Flk1 transcript (Kdr) was expressed at low levels in the XY interstitial cells at E13.5 (Figure 1E), we do not believe that the Flk1-mCherry-positive, 3b-HSD-positive cells express endogenous Kdr/Flk1. We concluded that the source of the Leydig cell contamination was leaky expression of the Flk1-mCherry transgene in Leydig cells. While this result complicated our analysis of the endothelial population, it provided strong evidence that the array data accurately reflected gene expression in the sorted populations. Lineage, sex, and stage all influence gene expression To investigate the effects of cell lineage, sex, and stage on global gene expression and to validate the consistency of expression measurements in biological replicates, we clustered all 72 individual microarrays based on the expression of transcript clusters that met our inclusion criteria outlined in the Materials and Methods (Figure 2A). With the exception of one E11.5 XY endothelial array (see Materials and Methods), biological replicates, and even samples of different sexes (early XX and XY germ cells) or stages (late XX supporting cells) that were expected to be similar, showed consistent expression patterns as indicated by the tight clustering of those samples (Figure 2A). This again validates the quality of the microarray data. The dendrogram indicates that cell lineage is the most significant factor affecting gene expression. The same general patterns were observed using other clustering methods (Figure S3A, S3B). This result was confirmed by an analysis of the sources of variation (ANOVA), in which lineage was identified as the most significant factor influencing gene expression variation (Figure 2B, Figure S3C). Interstitial/stromal cells were distinct from supporting cells at all stages, indicating that, despite their shared origin [3], these are separate lineages by E11.5. Sex and stage were also sources of expression variation in the gonadal cell populations, albeit to a lesser extent than lineage (Figure 2A, 2B). XY and XX supporting cells clustered in distinct groups at E11.5, confirming that these cells embark on their sexspecific differentiation by E11.5 [10,11]. There was no distinction between the sexes in the other cell types until E12.5 or E13.5. While the late stage XY endothelial cells clustered away from the early XX and XY endothelial cells, this could be due to the sexspecific contamination by Leydig cells. In summary, this analysis confirms the high quality of the data and shows that each lineage is distinct from E11.5 onwards. Each lineage has uniquely expressed enriched and depleted genes that provide insight into the biology of the cells To explore the differences between the cell types apparent in the dendrogram, we identified lineage specific genes that were specifically enriched and depleted in each lineage relative to the other lineages at each stage. We then determined whether these genes were expressed in a sex-specific (expression was different between XY and XX samples) or a sex-independent (expression was similar in XY and XX samples) manner. We identified these genes by performing multiple pairwise comparisons on the normalized array values (similar to previously described methods [53]) with a p-value cutoff of 0.05 and a fold change cutoff of 1.5. Enriched genes were more highly expressed than in other lineages, and depleted genes were less highly expressed than in other lineages. Examples of genes showing these different patterns include the sex-specific enrichment of Dhh in XY supporting cells (Figure 1B, Figure 3A), the sex-independent enrichment of Flt1 in XX and XY endothelial cells (Figure 1B, Figure 3A), the sexspecific depletion of Ccna2 (cyclin A2) in XX supporting cells (Figure 3A, 3B), and the sex-independent depletion of Gata6 (GATA binding protein 6) in germ cells (Figure 3A, 3B). A full description of our statistical methods is provided in the Materials and Methods and a list of all genes identified appears in Dataset S2A. Due to the sex-specific Leydig cell contamination of the endothelial population after E11.5 (Figure 1C 1E), we present the gene lists (Dataset S2A) but not the graphs (Figure 3A) because we could not determine whether sex-specific gene expression was due to an artifact of the contamination or genuine differences in endothelial cells. We deduced a list of Leydig cell genes (Figure 3A, Dataset S2A) by identifying genes specifically over-expressed or under-expressed in both XY endothelial cells and interstitial cells at E12.5 and E13.5 compared to other lineages. These genes were removed from XY endothelial gene lists at E12.5 and E13.5 (Dataset S2A). To evaluate these lists, we first performed permutation testing (Dataset S2B), and considered those with a false positive rate,20% as acceptable (lists that did not pass this test are marked ns ) (Figure 3A). Second, we determined whether positive control genes with known expression patterns, such as the expression of Sox9 in XY supporting cells, were found in the expected lists (Dataset S2A). Third, we interrogated all significant lists in Figure 3A (not ns or 0 ) for enrichment of BioCarta and KEGG pathways (Dataset S2C) as well as GO terms (Dataset S2D). As evidence of the high quality of the sorted cell microarray data, functional annotation of the enriched/depleted lists identified expected terms for each cell type: germ cell development in the germ cell lists, steroid production in interstitial and Leydig lists, vascular development in the endothelial list, and sex determination in supporting cell lists (Dataset S2C, S2D). All enriched terms for significant lists are provided to facilitate the discovery of new pathways involved in the functions of these cells (Dataset S2C, S2D). PLoS Genetics 4 March 2012 Volume 8 Issue 3 e
6 Lineage Priming of Sexual Fate in Gonadal Cells Figure 2. Gene expression was affected by lineage, sex, and stage. (A) Clustering dendrogram of individual microarray samples. The E11.5, E12.5, and E13.5 samples are represented by short, intermediate, and long bars, respectively. The dashed bars indicate XX samples, and the solid bars indicate XY samples. Ward s method with squared Euclidean distance as the distance metric was used. The arrays cluster primarily by lineage, and secondarily by sex and stage. (B) Analysis of the sources of variation confirmed that the primary source of variation is lineage, and secondarily sex and stage. doi: /journal.pgen g002 This analysis identified several novel expression features of these cells. For example, all of the lineages had sex-specific and sexindependent cohorts of depleted genes, with the exception of interstitial/stromal cells (Figure 3A). The genes identified as depleted appear biologically relevant based on individual genes and enriched categories of genes. Both XY and XX germ cells repressed Gata6 (Figure 3B, Dataset S2A), which can drive embryonic stem cells to adopt the extraembryonic endoderm fate [54]. Thus, the repression of Gata6 may be important for maintaining a totipotent transcriptional state in germ cells. Similarly, Lef1 became sex-specifically depleted in XY supporting cells (Dataset S2A). Lef1 can interact with b-catenin in a transcriptional complex downstream of Wnt signaling [55], and this pathway antagonizes aspects of testis development [24,56]. Thus, the depletion of Lef1 may be important for maintaining the male supporting cell fate. The sex-specifically depleted genes in XX supporting cells were enriched for cell cycle-related pathways and GO terms (Dataset S2C, S2D). Interestingly, both XX and XY supporting cells are arrested at E11.5 and only XY supporting cells re-enter the cell cycle; XX supporting cells remain nonproliferative [57,58]. It was previously shown that XX cells express higher levels of cell cycle inhibitors [11] and that cell cycle genes are over-represented in XY supporting cells [15]. However, our data suggest a mechanism of cell cycle arrest involving the active repression of multiple genes important for cell cycle progression in XX supporting cells. Additionally, we identified transcripts associated with a sexually undifferentiated progenitor cell for each lineage. All cell types had a large number of genes that were lineage-specific and sexindependent at E11.5 (Figure 3A, =+R category). The identification of shared expression in XX and XY cells demonstrates that there is a sexually undifferentiated progenitor for each lineage with a distinct transcriptional state. This was consistent with previous data suggesting that XX and XY supporting cells have a common origin [7] as well as the clustering results showing that supporting cells and interstitial/stromal cells are distinct lineages by E11.5 (Figure 2A). Supporting cells exhibited the largest number of sexually dimorphic genes at E11.5 (Figure 3A, = and R categories), in accord with previous evidence that the supporting cell lineage adopts a sex-specific fate early in gonad development and instructs the other lineages as to which fate they should adopt [8,10,46,59]. Although it was clear that supporting cells began sex-specific differentiation by E11.5, XX and XY supporting cells still expressed sex-independent genes. Since the supporting cells are in the midst of their sex-specific differentiation at E11.5, the sexindependent genes likely represent remnants of the sexuallyundifferentiated progenitor state. The XX and XY supporting cells adopt their distinct sex-specific states by E12.5. The other three cell types exhibited few sex-specific genes at E11.5 (Figure 3A, categories = and R ), showing that the differentiation of these lineages is delayed relative to that of the PLoS Genetics 5 March 2012 Volume 8 Issue 3 e
7 Lineage Priming of Sexual Fate in Gonadal Cells Figure 3. Lineage-specific enriched and depleted genes revealed distinct differentiation programs. (A) Graphs of the number of genes specific to each lineage. The gene lists and permutation tests are provided in Dataset S2. The = and R symbols indicate lineage-specific and sexspecific genes, while the =+R symbol indicates genes that are lineage-specific and sex-independent. Pale bars below the axis indicate genes that are depleted relative to other lineages. The E11.5 graphs are on the top row, the E12.5 graphs are in the middle, and the E13.5 graphs are on the bottom row. The germ cell Y-axis is scaled to accommodate the larger number of genes specific to this lineage. Leydig cell genes (burgundy) were separately identified by cross-referencing the endothelial and interstitial data and added to the bars for the XY interstitium. Lists with.20% false positives are indicated by ns. Lists with no genes are marked with 0. Some bars also have a colored gene name exemplifying the pattern within that category (the graphs for Dhh and Flt1 appear in Figure 1B). (B) Graphs of the log-transformed, normalized intensity values for genes that are sexspecifically (Ccna2) and sex-independently (Gata6) depleted. The error bars are standard error. Three lineages showed specific gene depletion in addition to enrichment. Each lineage had transcriptionally distinct progenitors as indicated by =+R genes at E11.5. Supporting cells were already in the midst of their sex-specific differentiation by E11.5 as indicated by genes in the = or R columns at E11.5, but the other cell types were sexually undifferentiated at E11.5. doi: /journal.pgen g003 supporting cells. Germ cells and interstitial/stromal cells began to adopt lineage-specific, sex-specific transcriptional states by E12.5, a process that was further advanced by E13.5. This was again consistent with the dendrogram (Figure 2A). We identified few lineage-specific genes in the XX stroma at these stages. While some genes were over-expressed in XX stromal cells when compared only to XY interstitial cells, most were also dimorphic in another lineage (Figure S4). This may indicate that the XX stromal cells are delayed in their differentiation and/or closely related to the supporting cell lineage based on shared expression of female-associated genes (like Irx3 and Wnt4; Figure S4). Predictions of the gonad differentiation models These analyses defined the transcriptome shared between XX and XY progenitors for each lineage, and traced the timing of differentiation and acquisition of sex specific fate for each cell type. Informed by this analysis, we investigated whether the transcriptome shared by XX and XY progenitors in the germ cell and supporting cell lineages showed evidence of lineage priming toward the female or male fate. Progenitors that show lineage priming express markers of the various fates into which they can differentiate and subsequently silence genes associated with the fate not adopted as they differentiate [28 35]. Four predominant models have been proposed to account for gonad differentiation that have important historical antecedents in the sex determination literature, and make different predictions as to the ground state of gonadal progenitors (Figure 4A). It has been proposed that (1) the female state is a default pathway [19 21], (2) a female Z gene actively represses the male program [22], (3) both the female and male programs are actively initialized [23], and (4) the gonad is balanced between the male and female fates [24 27]. We logically extended and reframed these models in PLoS Genetics 6 March 2012 Volume 8 Issue 3 e
8 Lineage Priming of Sexual Fate in Gonadal Cells Figure 4. Germ cells showed lineage priming with a male bias. (A) Models tested and their predictions for the relationship between the differentiated cells and their undifferentiated progenitors. (B, I) Graphs of the log-transformed, normalized intensity values. The error bars are standard error. Only the values for germ cells are shown, except in the depleted and primed example where all lineages are shown for comparison (B). (B) Esrp1 and Pcgf5 are examples of male- and female-primed genes, and Mosc2 is an example of a male-primed depleted gene. We used three different methods to identify primed genes: (C, D, and J) all primed genes were considered, (E, F, and K) only primed and lineage-specifically enriched genes were considered, and (G, H, and L) lineage-specifically depleted primed genes were analyzed. (C, E, and G) The percentages of primed genes that were male-primed and female-primed: all methods showed male-biased priming. The boxes contain the p-values from the binomial test with the expected percentages of the extreme models: 90% male genes ( Male ), 50% male and female genes ( Balanced ), and 90% female genes ( Female ) (see A). All of the extreme models were excluded because p-values were,0.05. (D, F, and H) The percentages of male or female genes that were primed. Significance (*) was determined with the hypergeometric test (p-value,0.05). (I) Graphs illustrating two primed genes whose expression in PLoS Genetics 7 March 2012 Volume 8 Issue 3 e
9 Lineage Priming of Sexual Fate in Gonadal Cells progenitors is similar to the differentiated cell in one sex or intermediate between the two sexes. (J L) In all cases, for both sexes, the majority of primed genes were similarly expressed in germ cell progenitors and differentiated cells of one sex. Gene lists and permutation tests are provided in Dataset S4. doi: /journal.pgen g004 the context of lineage priming so that we could test their applicability to the differentiation of the individual cell types in the gonad at the level of the whole transcriptome. The female model predicts that the transcriptome shared by XY and XX progenitors should be predominately associated with the differentiated female fate (i.e., female primed ). Conversely, the male model predicts that the transcriptome shared by XY and XX progenitors should be predominately associated with the differentiated male fate (i.e., male primed ). If both the male and female programs are activated de novo as progenitors differentiate, those progenitors should be a blank slate in that they would not express transcripts associated with either the differentiated male or female cells. Alternatively, the progenitors could fit the balanced priming model and express a similar number of both male- and female-associated transcripts at the time when they are poised to adopt either fate. Finally, the progenitors could be primed to adopt either fate, but there could be more genes associated with the female ( female-biased priming ) or the male ( male-biased priming ) fate (Figure 4A). Germ cell progenitors are primed with a bias toward the male fate We analyzed the relationship between XX and XY germ cell progenitors and their final sexually differentiated fates in multiple ways to ensure that our methods did not skew the results. We defined a gene as primed if it showed identical levels of expression in XY and XX germ cells at E11.5 that were retained or elevated in one sex and not in the other at E13.5 (Figure 4B, Esrp1 and Pcgf5). This was an unbiased method of identifying genes characteristic of the female and male fates [60]. Similarly, we determined expression in the E11.5 sexually undifferentiated progenitors in an unbiased way. We inferred expression from the fact that these genes showed down-regulation in one of the sexes. In this approach, all primed genes were analyzed, regardless of whether they were specific to germ cells (Figure 4C, 4D). In a second approach, we adopted more stringent, but still unbiased, definitions for the set of genes analyzed (Figure 4E, 4F). In this approach, we restricted our analysis to primed genes that were lineage-specifically enriched in E11.5 germ cells and in differentiated E13.5 germ cells of one sex (Figure 3A). Using an analogous method, we also explored depleted and primed genes that were specifically depleted in XY and XX E11.5 progenitors, were activated by one sex, and remained sex-specifically and lineage-specifically depleted in the other (Figure 4B, Mosc2, Figure 4G 4H). Gene lists and associated permutation tests are provided (Dataset S4A, S4C). With all methods tested, germ cells showed a similar pattern indicating male-biased priming. When comparing the percentage of primed genes that were male or female primed, we observed a clear bias toward male genes (Figure 4C, 4E, 4G). We performed a binomial test using the expected values provided with the models (Figure 4A) to determine if we could exclude the extreme models. In all cases, the pattern of genes observed was significantly different from the extreme female and male models as well as the completely balanced priming model (Figure 4C, 4E, 4G). Thus, a male-biased priming model best described the transcriptome in undifferentiated XX and XY germ cell progenitors. This finding was consistent with the clustering dendrogram showing a closer relationship of the undifferentiated early germ cells to the late XY germ cells than to the late XX germ cells (Figure 2A). We also wanted to ensure that this result was not a statistical artifact of the size of the underlying lists of male and female markers. For example, if the male program contained a larger number of genes than the female program, seeing a male bias in the number of primed genes could reflect the higher relative percentage of male pathway genes, rather than a real priming bias in the progenitor. Thus, we examined the percentage of male and female genes that showed priming (Fig 4D, 4F, and 4H). Again, in all cases, we saw that the same male bias was preserved. Given the large number and percentage (nearly 40% of male germ cell markers) of genes that showed priming (Figure 4C, 4D), the blank slate model could be discarded. Thus, germ cell progenitors showed male-biased priming, including the priming of depleted genes. Lastly, we were interested in determining whether genes that showed priming were expressed at high or low levels. The expression levels of differentiation markers in the progenitor cells are low in the hematopoietic system [29], but high levels of expression of differentiation markers were observed in progenitors in the early embryo [36]. For our analysis, expression level was defined relative to the differentiated cell. A gene with similar expression was expressed in progenitors at a level similar to the level in sexually differentiated cells maintaining expression (Figure 4I, Ddx18), analogous to the high expression observed in the early embryo [36]. A gene with intermediate expression was expressed in progenitors at a level between the levels observed in the two sexes (Figure 4I, Cdh1), analogous to the low expression observed in hematopoietic cells [29]. We analyzed the expression level for genes identified as primed by each method (Figure 4J 4L). A majority of both male and female primed genes were similarly expressed in the undifferentiated germ cell progenitors and the sexually differentiated cells, regardless of how the set of primed genes was defined or whether enriched or depleted genes were considered (Figure 4J 4L). Thus, not only did germ cells show male-biased priming, but the progenitors frequently expressed these primed genes at the same level as the sexually differentiated cells. We analyzed the lists of genes that exhibited a primed expression pattern (including all primed genes), and were regulated in the same way (similar expression in the progenitor and sexually differentiated cells, Figure 4J) for enrichment of GO terms (Dataset S4D). The genes primed toward the male germ cell fate showed a strong enrichment for categories related to RNA biology, such as RNA binding (Dataset S4D). This is consistent with the previously reported importance of gene control at the RNA level during germ cell development, especially in the male [49,61,62]. All the data indicate that the germ cells are primed with a malebias, and that these primed genes are generally expressed at a similar level in the progenitor and differentiated cell. Supporting cell progenitors are primed with a bias toward the female fate We also examined the relationship between supporting cell progenitors and their sexually differentiated states. We used the same method and tested the same models as with germ cells, but the end point of the analysis for supporting cells was E12.5 because PLoS Genetics 8 March 2012 Volume 8 Issue 3 e
10 Lineage Priming of Sexual Fate in Gonadal Cells Figure 5. Supporting cells showed lineage priming with a female bias. (A and H) Graphs of the log-transformed, normalized intensity values of genes. The error bars are standard error. Only the values for supporting cells are shown, except in the depleted and primed example where all cell types are shown. (A) Mdk and Rasgrp1 are examples of male- and female-primed genes and Cenpa is an example of a female-primed depleted gene. As in the germ cell analysis, we examined all primed genes (B, C, and I), primed and lineage-specifically enriched genes (D, E, and J), and primed and lineage-specifically depleted genes (F, G, and K). (B, D, and F) The percentages of primed genes that were male-primed and female-primed. The boxes contain the p-values from the binomial test with the expected percentages of the extreme models. (B) Using the first method, all of the extreme models could be excluded because they had a p-value,0.05. (D and F) However, using the second and third methods, the balanced and female models could not be excluded, respectively. (C, E, and G) Nevertheless, examining the percentage of male or female genes that were primed, all methods showed a significant (*) bias toward the female pathway, as determined by the hypergeometric test (p-value,0.05). Taken together, the data supported female-biased priming. (H) Graphs illustrating two primed genes, whose expression in the progenitor is similar to the differentiated cell of one sex, or intermediate between the two sexes. (I K) The female-primed genes were predominantly similarly expressed, but the maleprimed genes showed more variability. Gene lists and permutation tests are provided in Dataset S4. doi: /journal.pgen g005 their transcriptome changed little between E12.5 and E13.5 (Figure 3A, Figure S4, Dataset S2A, Dataset S3A). As in the germ cell analysis, we defined a gene as primed if it showed identical levels of expression in XY and XX supporting cells at E11.5, which were retained or elevated in one sex and not in the other by E12.5 (Figure 5A). The same variations of the analytical methods used for germ cells were also used to identify supporting cellspecifically enriched and depleted primed genes (Figure 5A). The PLoS Genetics 9 March 2012 Volume 8 Issue 3 e
11 Lineage Priming of Sexual Fate in Gonadal Cells gene lists, permutation test results, and GO terms are provided (Dataset S4B-S4D). The pattern in supporting cells was less consistent than the pattern observed for germ cells, but was indicative of femalebiased priming. Examining all of the primed genes, there was a clear female bias both for the primed genes as a percentage of the priming program (Figure 5B) and the primed genes as a percentage of the XY and XX sexually dimorphic genes (Figure 5C). When restricting our analysis to the genes specifically enriched in supporting cells (Figure 5D, 5E), we observed a similar female bias in the percentage of XX and XY genes that were primed (Figure 5E), but the bias in the percentage of primed genes associated with the male and female pathways was not sufficient to exclude the balanced priming model (Figure 5D). This is likely a statistical artifact due to the small number of primed genes and the difference in size between the E12.5 XX and XY enriched supporting cell gene lists (Figure 3A). The genes specifically depleted in supporting cells showed a strong female bias (Figure 5F, 5G), such that the female model could not be excluded (Figure 5F). All of these analyses suggested female-biased priming, consistent with the clustering showing the closer relationship between the less differentiated early supporting cells and the late XX supporting cells compared to the late XY supporting cells (Figure 2A). We also determined if the primed genes showed similar or intermediate expression in undifferentiated supporting cell progenitors (Figure 5H). For the female-primed genes, we consistently observed that most genes were similarly expressed (Figure 5I 5K). Expression levels of male-primed genes were more variable (Figure 5I 5K), which again could be due to the small number of genes analyzed. Nevertheless, supporting cell progenitors expressed at least some of the primed genes at levels similar to the sexually differentiated supporting cells. However, the XX and XY supporting cells were already expressing sexually dimorphic genes by E11.5 (Figure 3A) and therefore were not fully undifferentiated at the start of our analysis. With the reporters available to us, collection of a pure population of progenitors from an earlier stage was not feasible. To determine whether our results were affected by the differentiation process already in progress, we reanalyzed a publically available microarray time course on sorted Sf1-EGFP cells from the urogenital ridge that included earlier time points than were collected here [11] (Figure 6, Figure S5, Dataset S5). While the Sf1-EGFP reporter allows for the collection of earlier time points, it also labels a mixed population that includes at least supporting cells and interstitial/stromal cells [11,52]. This is concerning because different cell types can have different priming patterns (Figure 4, Figure 5). Therefore, we identified genes from the Sf1-EGFP data set that showed priming, and then restricted our analysis to only those genes whose expression patterns were associated with the supporting cell lineage as defined by our data set. Using the same approach as in the analysis of the Sry-EGFP/ Sox9-ECFP sorted supporting cell progenitors, we identified primed genes within the Sf1-EGFP undifferentiated progenitor population. To limit the contribution of genes from interstitial/ stromal cells, we set the end point of the analysis at E12.5. E11.0 Sf1-EGFP cells were selected as the starting point for the analysis presented in Figure 6 because it was closest to the sexual divergence point of the primed genes, although the analysis from E10.5 is also provided (Figure S5, Dataset S5B). While lists of all primed genes identified in the Sf1-EGFP cells are provided (Dataset S5A), we limited our analysis to only those genes associated with supporting cells. To define this gene set, we used two different methods utilizing our purified supporting cell (Sry-EGFP/Sox9-ECFP) and interstitial/stromal cell data as a reference. First, we used a rigorous threshold for inclusion and retained only those genes that were also found to be lineagespecifically and sex-specifically enriched in our E12.5 XX or XY Sry-EGFP/Sox9-ECFP supporting cells (Dataset S2A; Figure 6A, 6C, 6D, 6G). In a second approach, we used less rigorous criteria and removed genes sexually dimorphic in the interstitial/stromal cells (Figure 6B, 6E, 6F, 6H; Dataset S5A, S5C). Genes that were sexually dimorphic in both the Sry-EGFP/Sox9-ECFP supporting cells and the interstitial/stromal population were only removed if they were expressed at higher levels in interstitial/stromal cells than in supporting cells. Regardless of the method used, these data also supported female-biased priming of the supporting cells. Most of the primed genes were female, although the progenitors expressed some male genes as well (Figure 6C 6F, Dataset S5A). Many of these genes were also similarly expressed (Figure 6G, 6H; Dataset S5A). This analysis is therefore consistent with the findings from the Sry- EGFP/Sox9-ECFP data. To investigate the overlap in these data sets, we determined whether similar transcripts were identified as primed in both the Sf1-EGFP and Sry-EGFP/Sox9-ECFP data sets (Figure S6). While a small percentage of the genes primed in the Sf1-EGFP cells were also identified as primed in the Sry-EGFP/ Sox9-ECFP data, a larger proportion were already sexually dimorphic by E11.5, although indications of priming could be observed in some of the expression patterns (Figure S6). This analysis was also consistent with previous findings showing that individual genes we identified as primed were expressed in the supporting cell progenitors and then became sex-specific (Dax1/ Nr0b1, Wnt4, Sox9, and Cbln4 [26,63 66], Dataset S4B, Dataset S5A). Thus, the analysis of these two independent data sets produced consistent results for individual genes and reached the same overall conclusion that the supporting cells are primed with a female-bias. Because nearly 30% of female genes showed priming in the Sf1- EGFP data (Figure 6D, 6F), the blank slate model can be rejected. The E11.5 Sry-EGFP/Sox9-ECFP progenitors showed a lower percentage of primed transcripts (Figure 5C, 5E) than the E11.0 Sf1-EGFP progenitors (Figure 6D, 6F), which may be explained by the fact that the Sry-EGFP/Sox9-ECFP supporting cell progenitors were already partially sexually differentiated by E11.5 (Figure 3A). Discussion A comprehensive understanding of organogenesis requires systems-level knowledge of transcriptional network dynamics underlying cell differentiation. By performing a microarray analysis on sorted cell populations in the fetal mouse gonad over the course of sex-specific differentiation, we quantified the transcription dynamics of diverse cell types as they build one of two different organs from similar pools of progenitors. This study provided an expression resource for the field of gonad development, but more importantly, it characterized the features of the biological system that could only be appreciated at the whole transcriptome level. By examining the system as a whole, we obtained new insights into patterns of cell fate determination and lineage commitment. Insights from whole transcriptome characterization of multiple gonadal lineages We characterized the transcriptomes of undifferentiated progenitors and analyzed their transition to sexually differentiated cells. All four lineages analyzed (including interstitial/stromal cells) have a sexually undifferentiated progenitor cell with a distinct transcriptome. Although we detected some overlapping sexually PLoS Genetics 10 March 2012 Volume 8 Issue 3 e
12 Lineage Priming of Sexual Fate in Gonadal Cells Figure 6. Data from sorted Sf1-EGFP cells also supported female-biased priming for supporting cells. (A B) Graphical illustrations of the genes included in our analysis of priming in the Sf1-EGFP data. Because the Sf1-positive population is a mixture of lineages, we used two methods to identify the primed genes associated with supporting cells. XY cells are illustrated in this example, but the same operations were also performed for XX cells. (A) Sf1 primed and supporting cell enriched genes were both male-primed in the Sf1-EGFP data (comparing E11.0 and E12.5) and lineagespecifically enriched in our XY Sry-EGFP/Sox9-ECFP purified supporting cells at E12.5. Red indicates genes being removed from the analysis, and green indicates genes being retained. (B) For the Sf1 primed, removing interstitial/stromal genes, we removed genes associated with the interstitial/ stromal cells at E12.5 (i.e., sexually dimorphic in the interstitium/stroma) from the Sf1-EGFP primed genes. Genes that were expressed sexually dimorphically in both the interstitial/stromal cells and the supporting cells were removed only if expression was higher in the interstitial/stromal cells than in the Sry-EGFP/Sox9-ECFP supporting cells. The Sf1-EGFP primed genes that were enriched in the Sry-EGFP/Sox9-ECFP supporting cells (C, D, and G) and those that were identified by removing interstitial/stromal genes (E, F, and H) were analyzed separately. (C and E) The percentages of primed genes that were male-primed and female-primed. Both methods showed a female bias. The boxes contain the p-values from the binomial test with the expected percentages of the extreme models, and all extreme models could be rejected as having a p-value,0.05. (D and F) The percentage of male or female genes that were primed showed a significant (*) bias toward the female pathway, as determined by the hypergeometric test (pvalue,0.05). (G and H) However, primed genes in both sexes were predominantly expressed at similar levels in progenitors and E12.5 supporting cells of one sex. While supporting cell progenitors have a female bias, they also express some markers of the male pathway at levels similar to male supporting cells at E12.5. Gene lists and permutation tests are provided in Dataset S5. doi: /journal.pgen g006 dimorphic expression patterns that may have biological significance between the supporting cells and interstitial/stromal cells (Figure S4), these lineages have transcriptionally distinct progenitors at E11.5 (Figure 3A). However, the differentiation of the interstitial and stromal cells is sexually asymmetric over this time period (Figure 7). Whereas the XY interstitium expressed lineagespecific transcripts, there were few lineage-specific transcripts in XX stromal cells even at E13.5 (Figure 3A). Thus, the XX stroma may not fully differentiate until after E13.5. The XX stroma also showed overlapping sexually dimorphic expression with XX supporting cells, in part due to pathways downstream of widespread Wnt signaling in the ovary (Figure S4). We also provided global transcriptional evidence that the supporting cells are the first cell type in the gonad to adopt a sexspecific fate (Figure 3A), as predicted by previous experiments [10,11,46,59]. While there are some gene expression differences between E12.5 and E13.5, the supporting cells appear to have adopted their sex-specific fates by E12.5 (Figure 3A, Figure S4, Dataset S2, Dataset S3). The sex-specific differentiation of interstitial/stromal cells and germ cells began at E12.5, when PLoS Genetics 11 March 2012 Volume 8 Issue 3 e
13 Lineage Priming of Sexual Fate in Gonadal Cells Figure 7. Models of differentiation for the different gonadal lineages. The interstitial/stromal cells differentiate asymmetrically over the time period examined, as we detected few genes specific to the XX stroma by E13.5, whereas, the XY interstitial population acquired a larger set of lineage-specific genes. Supporting cells are primed with a female bias. The natural progression of the primed state may be to adopt the female differentiated state, but in the presence of Sry the cells repress the female program and adopt the male fate. Conversely, germ cells are primed with a male bias. An extrinsic signal may be required from the mesonephros to induce the adoption of the female fate; otherwise, germ cells adopt the male fate. doi: /journal.pgen g007 the supporting cells had essentially completed their differentiation process (Figure 3A). Lineage- and sex-specific transcriptional depletion in the differentiating gonad For supporting cells, germ cells, and endothelial cells, our methods were sensitive enough to detect lineage-specific transcript depletion that could be sex-specific or sex-independent (Figure 3A). Given that other studies have also reported specific gene depletion [16,17], this likely represents a common regulatory logic in the transcription network of differentiating cells. For example, unique cell fate specification in the sea urchin involves the repression of widelyexpressed genes to lock-down the selected fate [67]. We detected the lineage-specific repression of transcription factors likely involved in specifying alternative fates in both germ and supporting cells (i.e., Gata6 and Lef1, Figure 3, Dataset S2A). We also found evidence for lineage-specific repression of genes that regulate cell behavior. The transcriptome of XX supporting cells is characterized by the sex- and lineage-specific repression of cell cycle genes (Figure 3, Dataset S2), which is correlated with the failure of XX supporting cells to reenter the cell cycle [57,58]. A similar phenomenon was reported in senescence and DNA damage arrest [68,69], indicating this may be a widely-used mechanism of cell cycle arrest. Depleted genes also showed evidence of lineage priming in progenitors with similar patterns as genes expressed and primed (Figure 4G, 4H; Figure 5F, 5G), particularly in germ cells. The association between components of the network that are expressed and repressed suggests that both are important for specifying cell fate and may be co-regulated as parts of the same transcriptional program [67]. Contrary to our findings in the other lineages, we did not detect lineage-specifically depleted genes in the XY interstitial or XX stromal cells. However, there may be heterogeneity within this population that masks repression characteristic of any one subfraction. Since the purity of the germ and supporting cell populations were likely important for detecting depletion, the ability to isolate distinct populations within the interstitium/ stroma may be necessary to do the same for this population. A role for priming in the bipotential supporting cell lineage We determined that the supporting cell progenitors are primed with a female bias, indicating that both male and female genes are expressed in the progenitor, but that the female program is overrepresented (Figure 7). This female-biased priming model bridges the more recent evidence for a balance in the gonad between the male and female fates [24 27] and the classic theory of the female default state [19 21]. Although we found genes characteristic of both the male and female programs in supporting cell progenitors, the over-representation of the female program in progenitors explains why the female fate is the default state in the absence of Sry. The female fate may be the default state because the overrepresented female program of supporting cell progenitors is selfsustaining without additional inputs and leads to silencing of the alternative testis pathway. The high level expression of primed genes may make the primed state in the progenitors unstable. Whereas, low-level expression of fate determinates has been associated with a stable primed state, the expression of determinates at high levels has been associated with instability of the primed state [70]. As priming with high level expression has also been noted in the early embryo [36], this may be a common developmental mechanism to ensure that development progresses and does not become stalled. Thus, this unstable primed state would naturally lead to female differentiation in the absence of an intervention from Sry. Under these circumstances, an ovarydetermining gene, as proposed by Eicher and Washburn [23], would not be necessary. Differences in the strength of the female bias may explain differences between humans and mice with respect to the effects of mutations in individual genes in the female pathway. For example, mutation of RSPO1 in humans resulted in female-to-male sex reversal [71]. However, deletion of Wnt4, Rspo1, or b-catenin in the mouse did not result in full female-to-male sex reversal [25,66,72,73]. The female bias in mice may be sufficiently robust to rebound from the loss of any one of these genes. Conversely, a lower level of female-biased priming in humans could allow deletion of just one key factor in the female program to disrupt female development. Bipotential supporting cells show priming toward the male and female fates (Figure 5, Figure 6). A previous study identified a female subnetwork in E11.5 XY gonads [27]. We identified many of these genes as female-primed (Wnt4, Fst, Rspo1, Dapk1, Pld1, Actr6, and Dock4, Dataset S5A). The expression of female genes in XX and XY supporting cell progenitors could be consistent with a female transcriptional state in the progenitors that is repressed by the activation of Sry in XY cells rather than by the concept of male and female priming. However, the coexpression of male genes in both XX and XY supporting cell progenitors strongly supports the idea that these cells are primed to adopt either of their potential fates. These genes likely represent a male subnetwork operating in the early XX and XY cells independent of Sry, assry is not present in the XX cells. This work provides a molecular explanation for the concept of bipotentiality characterized by the coexpression of both male and female transcripts in the XX and XY supporting cell progenitors. The mechanism of priming remains to be determined. It has been speculated that priming is a byproduct of open chromatin [29,33,70]. Bivalent chromatin has been reported in embryonic stem cells at loci expressed at low levels [74]. However, this is inconsistent with our findings in the gonad where primed genes in bipotential supporting cell precursors tended to be expressed at high levels. Other studies in the laboratory are aimed at investigating the state of chromatin at primed loci. While priming may be important to establish a bipotential state, mounting evidence suggests that repression of the primed genes PLoS Genetics 12 March 2012 Volume 8 Issue 3 e
14 Lineage Priming of Sexual Fate in Gonadal Cells associated with the alternative sex is important for supporting or maintaining cell fate commitment. Genes important for testis (Sox9 and Dmrt1) [75 77] and ovary (Wnt4, Fst, and Rspo1) [25,66,78] development were identified as expressed in XX and XY supporting cell precursors and repressed in the opposite sex (Dataset S4B, Dataset S5A). Over-expression of Sox9 is known to result in female-to-male sex reversal [79]. Ectopic activation of the downstream target of WNT4, b-catenin, can reverse differentiation of XY supporting cells and trigger their differentiation as female cells [56]. Thus, the repression of genes associated with the opposite sex (which we identified in these experiments) may be as essential to the cell fate decision as the genes that are expressed. Priming during germ cell development The testicular-biased primed state of germ cell progenitors was surprising because germ cell fate is determined by the somatic environment [8]. Germ cells that enter an ovarian environment initiate meiosis (the female fate), while germ cells that enter a testis environment undergo mitotic arrest (the male fate) [80]. Historically, entry into meiosis was thought to be the default state for germ cell differentiation [8,81 84]. However, the weight of current evidence indicates that meiosis (the female fate) is the result of an external inducing signal produced in the mesonephros and specifically degraded in the testis by Cyp26b1 [85 89]. While there is some evidence for a signal promoting the male fate [90 94], this signal may act by antagonizing the female-promoting signal [85,91] and/or providing a permissive environment for male germ cell development [90]. The male-biased transcriptome of germ cell progenitors is consistent with a male developmental default state in the absence of the female-promoting signal (Figure 7). Interestingly, in both supporting and germ cells, the dominant fate-determining signal is associated with the fate under-represented in the progenitor s transcriptome (Figure 7). Sry expression in XY supporting cells is required to stabilize the male program and repress the female program. Similarly, the external female signal initiates the meiotic program in germ cells and represses the alternative male program. XY germ cells adopt the female fate if Cyp26b1 activity is eliminated (resulting in the presence of the meiosis-inducing signal), even in an otherwise male environment [85,86]. Our priming model suggests that the over-represented male program requires only subtle reinforcement from the somatic environment. On the other hand, the under-represented female program cannot be stabilized without its instructive cue, but once that input is received, it is able to suppress the male program. One reason why E13.5 XY germ cells share more transcriptional features with the progenitor than XX germ cells may be due to their maintenance in a more stem cell-like state (Dataset S4A) [95]. However, XY germ cells at E13.5 are not identical to the sexually undifferentiated germ cell progenitors at E11.5. Specifically, by E13.5, XY germ cells have repressed genes associated with the female germ cell program (Figure 4), which may explain why, even when put into a female environment after E11.5, XY germ cells can no longer adopt the female fate [8,81]. Priming during differentiation This study revealed previously unknown systems-level aspects of the differentiation of two critical cell types during gonad development, with implications for other developing cells. Supporting and germ cells arise from different embryonic origins and respond to different cues during their terminal differentiation, and yet both show priming. Priming may be a common feature of differentiation from multipotent progenitors at all levels, as it has now been identified in the early embryo [36], multipotent hematopoietic cells [29],bone marrow mesenchymal stem cells [28], germ cells, and somatic gonadal cells. However, each priming program appears surprisingly lineage specific. Even the cells within the gonad do not share a common bias in their priming programs (Figure 7). Priming may limit the developmental potential of cells by preparing them to respond in a unique manner to the same signals used throughout development. Only certain avenues of differentiation are available while others are closed [28,30,60]. For example, male supporting cells and male germ cells exposed to similar Fgf9 signals adopt different fates [24,91,92,96] because they have different underlying transcriptional networks that prepare the cell to respond differently. In vivo, a supporting cell progenitor cannot become a germ cell because the required transcriptional avenues are not available. The ability to induce pluripotent cells from differentiated cells in vitro may be related to the ability to return the cell to a primitive primed state, where many avenues of differentiation are open. We identified priming patterns using simple, yet flexible, statistical methods that can be applied to any microarray time course on a single purified cell population isolated immediately preceding and following differentiation. While we were able to analyze the priming of lineage-specific enriched and depleted transcripts to validate our results (Figure 4, Figure 5), the results were similar regardless of the method used. Because having other cell types for comparison is not required, this method can be broadly applied to other systems exploring differentiation. Systems biology entails the use of both whole genome analysis and molecular genetic approaches to inform each other [97]. While studies disrupting the function of individual genes have clearly identified critical components of the system, they are unlikely to be sufficient on their own to fully elucidate the combinatorial interactions within the complex transcriptional network governing organ development. Recent studies show that developmental transcriptional networks are highly buffered and contain redundant factors, suggesting that many important network players may not have a developmental phenotype when disrupted [27,98]. While some transcriptional profiles uncovered in this study may have no functional relevance, others likely contribute to robustness of the system and allow it to rebound from perturbation. In conjunction with the traditional functional studies examining individual genes, our understanding of gonad organogenesis (and development in general) is facilitated by a whole system view of the process as this approach reveals novel phenomena that cannot be identified by studying single genes. This analysis leads to many new and exciting hypotheses related to the role of priming in the differentiation of gonadal cells and provides new insight into the processes of cellular differentiation and lineage commitment. Materials and Methods Mice All animals were maintained and experiments were conducted according to DUMC-IACUC and NIH guidelines, based on existing protocols. We used six different transgenic mouse lines with fluorescent reporters: Sry-EGFP [Tg(Sry-EGFP)92Ei] (sex determining region of Chr Y) [7], Sox9-ECFP (SRY-box containing gene 9) [39], Mafb-EGFP [Mafb tm1jeng ] (a gift from S. Takahashi; v-maf musculoaponeurotic fibrosarcoma oncogene family, protein B) [40], asma-eyfp (a gift from J. Lessard; the official gene name of asma is Acta2; actin, alpha 2, smooth muscle) [45], Flk1-mCherry [Tg(Kdr-mCherry)1Medi] (a gift from M. Dickinson; the official gene name of Flk1 is Kdr; kinase insert domain protein receptor) [43,44], and Oct4-EGFP [Tg(Pou5f1-EGFP)2Mnn] (the official PLoS Genetics 13 March 2012 Volume 8 Issue 3 e
15 Lineage Priming of Sexual Fate in Gonadal Cells gene name of Oct4 is Pou5f1; POU domain, class 5, transcription factor 1) [42]. In most cases, males from these lines were crossed to CD-1 females (an outbreed line from Charles River), and gonads from multiple embryos were generally pooled to reduce the impact of strain variation. All of the males were homozygous for the marker, with the exception of Mafb-EGFP males. This line is a targeted insertion of GFP into the Mafb locus, which results in a Mafb mutant when homozygous. Mafb-EGFP embryos collected in this study were therefore heterozygous for Mafb; however, we know of no defects in gonad development in Mafb heterozygotes (data not shown). To increase the fluorescence intensity for both the asma-eyfp (maintained on an FVB/CD-1 mixed line) and Flk1-mCherry (maintained on the outbreed CD-1 line) reporters, homozygous males were crossed to homozygous females. Sry-EGFP and Sox9-ECFP reporters were used to collect supporting cells. In the Sry-EGFP line, the Sry promoter drives expression of GFP in cells competent to activate the Sry promoter in both XX and XY gonads. This labels the supporting cell lineage in both sexes, but because the transgene lacks the SRY open reading frame, transgenic XX gonads express no SRY protein and develop as normal ovaries [7]. While Sry-EGFP expression persists in XX supporting cell precursors through E13.5, expression of the transgene is reduced in XY supporting cell precursors after E11.5 in our hands (data not shown), similar to endogenous Sry expression [99]. Therefore, Sry-EGFP was used to isolate supporting cells from XX and XY samples at E11.5, and XX E12.5 and E13.5 samples. Sox9-ECFP was used to sort XY supporting cells at E12.5 and E13.5. All cells that later express Sox9 can be lineage traced from Sry-positive cells [46]. Sox9 is also the direct downstream target of SRY [100]. This Sox9-ECFP reporter specifically labels XY supporting cells(figures1).thus,wedid notexpectthattheuseofsox9-ecfp would affect our analysis of the supporting cells. We validated that this was the case by ensuring the genes we identified in the Sox9-ECFP population substantially overlapped those identified in a previous study using the Sry-EGFP reporter [10], and that some genes continued to be similarly expressed in the XX Sry-EGFP and XY Sox9-ECFP supporting cells (data not shown). Collection of gonadal lineages Timed matings were performed, with the day the vaginal plug was detected considered E0.5. Embryos were collected at E11.5, E12.5, and E13.5. For the Mafb-EGFP line, only GFP-positive embryos were used for sorting. The sex of the gonad is obvious by eye at E12.5 and E13.5. The E11.5 embryos were genotyped to determine the sex as previously described using primers to detect Kdm5c (X chromosome) and Kdm5d (Y chromosome) [27,101]. To collect gonadal cells, the urogenital ridge and dorsal aorta were removed, and the gonad/mesonephric complex was isolated. In most cases, the gonad was separated from the mesonephros. However, for the Oct4-EGFP sorts, the gonad was left attached because Oct4 expression is highly specific to germ cells (data not shown). For the E11.5 Flk1-mCherry sorts, only the anterior and posterior portions of the mesonephros were removed by cutting at a45u angle from the end of the gonad. The gonad vasculature arises from a plexus in the mesonephros [5]; thus, the gonadal and a portion of the mesonephric endothelial cells represent one population. This procedure retained the mesonephric plexus while removing most of the vasculature associated with the mesonephric ducts. At E12.5 and E13.5, the mesonephros was removed completely for the Flk1-mCherry sorts. XY and XX gonads were separately pooled from one or more litters and incubated in 250 ml 0.25% Trypsin EDTA (Gibco #25200) at 37uC for 5 10 minutes. The trypsin was removed and replaced with 400 ml PBS with or without 4 ml RNase-free DNase (Promega #M6101). The tissue was dissociated, and the cells were passed through a strainer (BD Falcon #352235). FACS was performed by the Duke Comprehensive Cancer Center Flow Cytometry Shared Resource. The positive fraction was pelleted, the liquid supernatant was removed, and the cells were immediately frozen at 280uC. Preparation of samples and arrays Generally, cells from multiple embryos were pooled. RNA was extracted from over 100,000 cells to as few as 10,000 cells using the RNeasy Micro kit (Qiagen #74004) following the manufacturer s instructions for Cells. However, the protocol was started at step 2 (disruption with RLT), and b-me was not added. The cells from multiple sorts were pooled during disruption with RLT (if necessary), step 3 (homogenization) was skipped, and in step 10 three RPE washes were performed. Samples were prepared for the Affymetrix Genechip Mouse Gene 1.0 ST Arrays (#901168) using the Nugen WT-Ovation Pico RNA Amplification System (#3300), WT-Ovation Exon Module (#2000), and the Encore Biotin Module (#4200), following the manufacturer s instructions. For purification following the Pico and Exon kits, the Qiagen QIAquick PCR Purification Kit (#28104) was used following the instructions provided by Nugen. Fragmented and labeled product was submitted to the Duke Institute for Genome Sciences and Policy Microarray Facility for hybridization and reading. We ran a total of 91 arrays. This included the arrays on our five sorted cell types at the three time points with separate XX and XY samples in biological triplicate as well as one array on a whole P1 mouse RNA sample(a gift from S. Potter) for normalization across GUDMAP. Immunofluorescence Samples were fixed, stained, and imaged as whole gonads with the mesonephros attached as previously described [102]. Some samples were first processed through a methanol series and stored at 280uC prior to rehydration and staining [103]. Primary antibodies used were: anti-3b-hsd (Santa Cruz sc-30820, 1:100 in samples processed through methanol; TransGenic Inc. KO607, 1:500; K. Morohashi generously provided a non-commercial antibody before the TransGenic Inc. antibody was available), anti- PECAM1 (BD Pharmingen , 1:250), anti-sry (a gift from P. Koopman and D. Wilhelm), and anti-sox9 (a gift from P. Koopman and D. Wilhelm). Secondary antibodies used included Alexa 647- and 488- conjugated secondary antibodies (Molecular Probes, 1:500) and Cy3- and Cy5-conjugated secondary antibodies (Jackson ImmunoResearch, 1:500). DAPI (Sigma-Aldrich) was used to label nuclei. Initial array processing and analysis The.cel files were processed with Partek Genomics Suite version 6.5 ( ) by RMA with quantile normalization and median polish summarization at the transcript cluster (gene) level. Probes were adjusted for GC content and probe sequence. The data were transformed into log base 2. We removed all transcript clusters that did not have a cross hybridization category of 1 (perfect match) in the Affymetrix annotation, that did not have a gene symbol, or that did not have a log base 2 normalized expression value.6 in at least two out of three replicates of at PLoS Genetics 14 March 2012 Volume 8 Issue 3 e
16 Lineage Priming of Sexual Fate in Gonadal Cells least one sample. The genes that passed this filtering step, and only these genes, were used in subsequent analyses. In the case of the analysis of the asma-eyfp cells (Figure S2), this initial processing of the arrays included all 91. However, for all other analyses, we used data generated by processing only 72 of our arrays because the asma-eyfp and P1 whole mouse data were not included. These processed data on the 72 arrays were used in all portions of the analysis and are provided as a resource for the community (Dataset S1). Partek Genomics Suite was used to generate the hierarchical clustering dendrograms and perform the ANOVA sources of variation analysis (Figure 2, Figure S3). The clustering methods used are described in the figure legends. One of the E11.5 XY endothelial samples was somewhat of an outlier in the clustering. This may be due in part to its processing, which resulted in unusually low (but still adequate) yield after the amplification with the Pico kit. However, we do not believe this compromised the sample as it still clustered with endothelial cells. Pairwise comparisons used to identify genes of interest This analysis was performed at the level of the transcript cluster (gene), but some genes have multiple transcript clusters. Thus, the lists of transcript clusters may include multiple entries for the same gene. For graphical display of the numbers of genes identified (Figure 3, Figure 4, Figure 5, Figure 6, Figure S4, Figure S5, Figure S6), each gene was counted only once (i.e., duplicates were removed). However, in tables, all transcript clusters are shown (i.e., duplicates are not removed) (Dataset S2, Dataset S3, Dataset S4, Dataset S5). To identify genes of interest, we adopted a simple and flexible method using multiple or single pairwise comparisons between samples (analogous to methods used previously [53]). The same p- value and fold change cutoffs were used throughout. In all cases where we identified a difference between samples, we used a p- value cutoff of 0.05, and a fold change cutoff of 1.5 for each comparison. A gene was deemed to be identically expressed in two samples if the p-value was.0.05 and the fold change was between 21.5 and 1.5. When multiple pairwise comparisons were done, the intersection of the multiple lists generated was taken as the genes of interest. To identify sex-specifically and lineage-specifically enriched genes (using E12.5 XY supporting cells as an example), we used the following pairwise comparisons with the above cutoffs: 1. The gene was more highly expressed in XY supporting cells than XX supporting cells at E12.5 (i.e., sex-specific expression). 2. The gene was more highly expressed in XY supporting cells than the XY interstitium, germ cells, and endothelial cells at E12.5 (XY supporting cells versus XY interstitium, XY supporting cells versus XY germ cells, etc.) (i.e., lineage-specific expression). The intersection of these multiple lists were the genes considered enriched in XY supporting cells at E12.5 (Figure 3A). Because the XY supporting cells were not compared to other XX lineages (e.g., XX germ cells), genes could be identified as enriched in both the XY supporting cells and a different XX lineage. Similar comparisons were also used to identify sex- and lineagespecifically depleted genes, but a gene was deemed depleted when its expression was higher in all other cell lineages than the E12.5 XY supporting cells in the example. To identify sex-independent and lineage-specifically enriched genes indicative of sexually undifferentiated progenitor expression (Figure 3A), we used the following pairwise comparisons (using the E11.5 supporting cells as an example): 1. The gene was more highly expressed in XY supporting cells than the XY interstitium, germ cells, and endothelial cells at E11.5 (i.e., lineage-specific expression among XY cells). 2. The gene was more highly expressed in XX supporting cells than the XX stroma, germ cells, and endothelial cells at E11.5 (i.e., lineage-specific expression among XX cells). 3. The gene was identically expressed in XX and XY supporting cells at E11.5 (i.e., identically expressed in progenitors). Again, similar comparisons were used to identify the lineagespecifically depleted genes, but a gene was deemed depleted when its expression was higher in all other cell lineages than the E11.5 XY and XX supporting cells in the example. Occasionally, multiple transcript clusters for a gene may behave differently. In this case that gene may be identified in multiple lists. For example, different Myo9a transcript clusters were identified as sex-specifically enriched in E11.5 XX supporting cells and sex-independently enriched in E11.5 XX and XY supporting cells (Dataset S2A). The different behavior of the different transcript clusters in the gene could be caused by off-target probe binding or alterative splicing. Leydig cell genes were a special case because both our sorted interstitial and endothelial cells contained Leydig cells (Figure 1C 1E). To identify Leydig cell genes (Figure 3A), using E13.5 as an example, we used the following pairwise comparisons: 1. The gene was more highly expressed in the XY interstitium than XY supporting cells and germ cells at E13.5 (i.e., lineagespecific expression). 2. The gene was more highly expressed in XY endothelial cells than XY supporting cells and germ cells at E The gene was more highly expressed in the XY interstitium than the XX stroma at E13.5 (i.e., sex-specific expression). 4. The gene was more highly expressed in XY endothelial cells than XX endothelial cells at E13.5. Depleted genes were identified similarly, but a gene was deemed depleted when its expression was higher in all other cell lineages than the E13.5 XY interstitium and endothelial cells in the example. The genes identified as E13.5 Leydig cell genes were removed from the E12.5 and E13.5 XY endothelial cell gene lists (Dataset S2A). In all cases, we used the E13.5 Leydig cell lists to remove the maximum number of genes associated with Leydig cells. Some of these genes were also identified in the XY interstitial lists, and they appear in both lists of identified genes (Dataset S2A), but the overlapping genes were removed from the Leydig cell bar for the graphical depiction (Figure 3A). Permutation testing was done to estimate the false discovery rate in the gene lists. The array data from the samples being used in the generation of a gene list (in the first example above: XY supporting cells, XX supporting cells, XY interstitium, XY germ cells and XY endothelial cells; all at E12.5) were permuted. The series of operations was run on the permuted columns, and the number of genes generated from each permutation was stored. The permutations were performed 200 times, and the mean number of genes was used to compute the false discovery rate. In all cases, we considered a false discovery rate of 20% or less as acceptable (Dataset S2B). Most lists actually had a much lower false discovery rate. In any case where some of the genes were removed from the list, such as the removal of Leydig genes from endothelial cell lists, this operation was ignored in the permutation tests. PLoS Genetics 15 March 2012 Volume 8 Issue 3 e
17 Lineage Priming of Sexual Fate in Gonadal Cells The lists of transcript clusters for the lists considered significant were inputted into DAVID ( [104,105] to identify pathway and GO term enrichment. The full list of transcript clusters used in the analysis (Dataset S1A) was inputted as the background. We included all KEGG and BioCarta pathways as well as GO_FAT molecular function (MF) and Biological Process (BP) terms with a p-value.0.05 (Dataset S2C, S2D). Primed genes were identified by multiple methods (Figure 4, Figure 5). In general, we defined a gene as primed when it was expressed in the progenitor, then repressed by one sex and maintained or activated by the other sex. To identify all maleprimed genes, using germ cells as an example, we used the following pairwise comparisons: 1. The gene was identically expressed in XX and XY germ cells at E11.5 (i.e., identically expressed in progenitors). 2. The gene was more highly expressed in XY than XX germ cells at E13.5 (i.e., specific to XY cells). 3. The gene was more highly expressed in XX germ cells at E11.5 than at E13.5 (i.e., this gene is repressed in XX cells). 4. If the gene was also more highly expressed in XY germ cells at E11.5 than at E13.5, it was removed (i.e., genes showing differential repression were eliminated). In the second analysis, we used more stringent criteria to define genes characteristic of the progenitor cells at E11.5 and differentiated cells at E13.5 by incorporating information on lineage-specific expression. To identify the enriched and primed genes, we used the above comparisons in addition to requiring that: 5. The gene was more highly expressed in XY germ cells at E11.5 than XY supporting cells, interstitium, and endothelial cells at E The gene was more highly expressed in XX germ cells at E11.5 than XX supporting cells, stroma, and endothelial cells at E The gene was more highly expressed in XY germ cells at E13.5 than XY supporting cells, interstitium, and endothelial cells at E13.5. Finally, we also wanted to explore the possibility of depleted gene priming. Continuing with the example of XY germ cells, we used the following pairwise comparisons to identify depleted and primed genes: 1. The gene was identically expressed in XX and XY germ cells at E11.5 (i.e., identically expressed in progenitors). 2. The gene was more highly expressed in XY supporting cells, interstitium, and endothelial cells at E11.5 than XY germ cells at E11.5 (i.e., lineage specific repression). 3. The gene was more highly expressed in XX supporting cells, stroma, and endothelial cells at E11.5 than XX germ cells at E The gene was more highly expressed in XX than XY germ cells at E13.5 (i.e., remains repressed in XY cells). 5. The gene was more highly expressed in XY supporting cells, interstitium, and endothelial cells at E13.5 than XY germ cells at E13.5 (i.e., lineage-specific repression). 6. The gene was more highly expressed in XX germ cells at E13.5 than at E11.5 (i.e., this gene is activated in XX cells). 7. If the gene was also more highly expressed in XY germ cells at E13.5 than at E11.5, it was removed (i.e., genes showing differential activation were eliminated). The same methods were used to analyze priming in the supporting cells, but E12.5 was used as the end point of the analysis. Permutation tests were run on all of these lists of primed genes. We then examined these primed genes in two ways. First, we compiled all genes identified as male or female primed and determined the percentage associated with each sex (Figure 4C, 4E, 4G; Figure 5B, 5D, and 5F). We used a binomial test for the different extreme models to determine whether priming showed a sex-specific bias. A one-tailed test was used for the female and male models, which were defined as predicting that 90% of the genes were female-primed or male-primed, respectively. A twotailed binomial test was used for the balanced model, which predicts 50% male and 50% female genes. Any model resulting in a p-value,0.05 was excluded. If all models were excluded, an intermediate model was selected. To ensure that this result was not a statistical artifact of the size of the underlying lists of male and female markers, we also displayed primed genes as a percentage of the total male or female genes (Figure 4D, 4F, 4H; Figure 5C, 5E, 5G). These male and female genes were determined in different ways to account for differences in the method of defining primed genes. When identifying all primed genes, the list of all testis genes included everything identified in step 2 alone of the process for generating the primed genes. For the enriched and primed genes, testis genes were required to meet the requirements of step 2 and 7. For the depleted and primed genes, testis genes were required to meet steps 4 and 5. A 262 contingency table (with the actual numbers of genes, not percentages) and a two-tailed hypergeometric test were used to determine if there were a significant difference in the percentage of primed genes in the male and female programs. A p-value,0.05 was considered significant. A significant p-value meant that sex and priming were not independent variables and that there was a bias in the representation of the two programs in the progenitor cells. We further characterized the primed genes identified by each method based on their expression level. We divided these into two categories: similar and intermediate expression relative to the expression in the differentiated cells (Figures 4J 4L, Figure 5I 5K). For similarly expressed genes, using XY germ cells as an example, the gene was required to be primed and identically expressed in XY germ cells at E11.5 and E13.5. For intermediate expression genes, the gene was required to be primed and more highly expressed in XY germ cells at E13.5 than E11.5 (or more highly expressed at E11.5 for depleted and primed genes). Any gene that did not fall into one of these categories, or had transcript clusters that fell into both similar and intermediate categories, was counted as other. If a gene had transcript clusters that fell into the defined similar or intermediate categories and others that did not fall into either, the gene was still counted as similar or intermediate. GO term enrichment for all of the primed genes with similar expression for both supporting and germ cells was determined using DAVID as described above. Sf1-EGFP array analysis To examine earlier expression in supporting cells, we reanalyzed previously generated microarray data from Sf1-EGFP sorted cells [11] (raw data available at = Nef&expandefo = on). Because these data used a different array format (Affymetrix Mouse PLoS Genetics 16 March 2012 Volume 8 Issue 3 e
18 Lineage Priming of Sexual Fate in Gonadal Cells Genome arrays), they were analyzed separately. The.cel files were processed with Partek Genomics Suite in the same manner as our own, and the same criteria were used to remove probe sets. The only difference was that rather than using the cross-hybridization category, the annotation grade was obtained from NetAffx ( [106], and only probes with unambiguous A and B grade annotation were retained. Control probes were also removed. We used the same method to identify and analyze primed genes as described above. This analysis used the Sf1-EGFP data at E11.0 (or E10.5) and E12.5 (Figure 6, Figure S5, Dataset S5). The priming analysis was limited to probes associated with the supporting cells by two methods, although the list of all primed genes is provided (Dataset S5A, S5B). First, we examined only genes that were lineage-specifically enriched in our XX or XY E12.5 Sry-EGFP/Sox9-ECFP data (Figure 3A, Dataset S2A) and Sf1-EGFP primed (Figure 6A, 6C, 6D, 6G; Figure S5A, S5B, S5E). Because different arrays were used, we compared between the two datasets using the gene symbol. If the gene symbol for an Sf1- EGFP primed gene was also found in the lineage-specific Sry- EGFP/Sox9-ECFP lists, the gene was retained in the analysis. Second, we removed the genes associated with the interstitial/ stromal cells at E12.5 from the Sf1-EGFP primed genes (Figure 6B, 6E, 6F, 6H; Figure S5C, S5D, S5F). Using XY cells as an example, we identified the genes to remove using the following pairwise comparisons and removal steps: 1. We identified genes more highly expressed in XY interstitial cells than XY supporting cells at E We identified genes more highly expressed in XY supporting cells than XX supporting cells and also XY interstitial cells than XX stromal cells, then removed the genes identified in step We identified all genes more highly expressed in XY interstitial cells than the XX stromal cells, and removed all of the genes remaining in step 2. This list contains genes sexually dimorphic in the interstitial cells and not the supporting cells, and genes sexually dimorphic in both the interstitial/stromal cells and the supporting cells if expression was higher in the interstitial/ stromal cells. The lists of the genes identified for removal is provided in Dataset S5C. Again, the comparison between the two data sets was based on the gene symbol: gene symbols found in the interstitial/stromal-associated lists were removed from the Sf1- EGFP primed list. To calculate the percentage of male and female genes showing priming (Figure 6D, 6F; Figure S5B, S5D), we used the same method used in analyzing all primed genes in our own data. However, in addition to being more highly expressed in one sex at E12.5, the gene was also required to pass the appropriate filter used for identifying supporting cell-associated genes. Supporting Information Dataset S1 RMA normalized values and intensity value graphing. We provided an excel file with (A) the log-transformed RMA normalized intensity values used in our analysis and (B) the graphic format used to display gene expression in each cell type over time. The user can copy any row from (A) with values into row 2 of (B) (yellow) to generate the graph for that gene. Only transcript clusters included in the analysis are provided, and the grounds for removing transcript clusters are provided in the Materials and Methods. (XLSX) Dataset S2 Lineage-specific gene lists, permutation tests, pathway enrichment, and GO term enrichment. (A) Gene lists generated by multiple pairwise comparisons, graphically illustrated in Figure 3A. The lists are arranged in order of lineage, stage, enrichment or depletion, and sex. Genes are in alphabetical order. (B) Results of permutation tests for all the gene lists displayed in Figure 3A. Separate entries are listed for the median and mean number of transcript clusters and individual genes found in the permutation tests. The number of genes in the actual list and the percentage of false positives based on the mean number of genes are also provided. (C) The lists of KEGG and BioCarta pathways enriched with a p-value,0.05 in each list with a false discovery rate,20%. (D) The list of biological process ( BP ) and molecular function ( MF ) GO terms enriched with a p-value,0.05 in each significant list. In both cases, the lists are arranged in order of lineage, stage, sex, and enrichment or depletion. The terms in the list are ranked from lowest to highest p-value. (XLSX) Dataset S3 Lists of sexually dimorphic genes, overlapping expression, and permutation tests. (A) Gene lists generated by a single pairwise comparison between XY and XX cells of a lineage graphically illustrated in Figure S4A. Lists of all sexually dimorphic genes in each lineage at each stage are provided first, followed by lists of genes sexually dimorphic in multiple lineages. Genes are in alphabetical order. The gene lists are arranged by stage, sex, and lineage. When transcript clusters for a single gene behaved differently, in all cases the gene was counted in the highest overlapping category. For example, if a gene had one transcript cluster sexually dimorphic in only one lineage and another that was dimorphic in two lineages, it was counted as being dimorphic in both lineages. 1 next to a gene name indicates the gene was counted as R E13.5 over-expressed in all cell types (the triple overlap in the Venn diagram in Figure S4A) even though it appeared in other categories (Syngap1 and Myo9a). (B) Results of permutation tests for the single pairwise comparisons. (C) Lists of genes that were sexually dimorphic in only two lineages, expressed identically in both lineages, and enriched in these two lineages compared to the remaining two lineages (Figure S4E). Genes are in alphabetical order. The gene lists are arranged by lineage, sex, and stage. The gene considered in the triple overlap (Syngap1) is again marked by the 1 and removed from the analysis. (XLSX) Dataset S4 Germ cell and supporting cell primed gene lists, permutation tests, and GO term enrichment. (A and B) Gene lists generated by multiple pairwise comparisons to identify germ cell (A) and supporting cell (B) primed genes, graphically illustrated in Figure 4 and Figure 5. Genes are in alphabetical order. The gene lists are arranged by method of identifying primed genes, and sex. The genes with similar and intermediate expression are provided after each list of primed genes. 1 next to a gene name indicates its transcript clusters appeared in both the similar and intermediate expression lists, and so was counted in Figure 4 and Figure 5 as Other. (C) Results of permutation tests for the lists of primed genes. (D) The lists of biological process ( BP ) and molecular function ( MF ) GO terms enriched with a p-value,0.05 in all of the primed genes with similar expression (Figure 4J, Figure 5I). The lists are arranged in order of lineage and sex. The terms in the lists are ranked from lowest to highest p-value. (XLSX) Dataset S5 Sf1-EGFP primed gene lists and permutation tests. (A and B) Gene lists generated by multiple pairwise comparisons, graphically illustrated in Figure 6 and Figure S5. Genes are in alphabetical order. The gene lists are arranged by the method of PLoS Genetics 17 March 2012 Volume 8 Issue 3 e
19 Lineage Priming of Sexual Fate in Gonadal Cells selecting supporting cell primed genes, and sex. The list of all primed genes identified in the Sf1-EGFP data without limiting the list to genes associated with supporting cells, all, are also provided. Lists of similar and intermediate expression primed genes follow each list of primed genes (except for the first). Lists are provided starting the priming analysis at both E11.0 (A) and E10.5 (B). (C) Gene lists of interstitial/stromal associated genes that were used to remove interstitial/stromal genes from the priming analysis as illustrated in Figure 6B. (D) Lists of genes illustrated in Figure S5G that were primed at E10.5 or E11.0 and became sexually dimorphic 12 hours later. Genes are in alphabetical order. (E) Lists of genes illustrated in Figure S5H. Of the genes that were primed at E10.5 or E11.0 and remained similarly expressed in XX and XY cells 12 hours later, we identified genes that showed a significant and similar change in expression in both XX and XY cells. Genes are in alphabetical order. (F) Lists of genes illustrated in Figure S6A that were primed in the Sf1-EGFP and Sry-EGFP/ Sox9-ECFP data, were primed in the Sf1-EGFP data and dimorphic in the Sry-EGFP/Sox9-ECFP data, and a subset of these E11.5 dimorphic genes that showed indications of previous priming in the Sry-EGFP/Sox9-ECFP cells. Genes are in alphabetical order. (XLSX) Figure S1 Lineage-specific fluorescent tags used for FACS. Images of E13.5 XY and XX gonads with DAPI (blue) and each fluorescent marker used: Sox9-ECFP and Sry-EGFP (cyan) labeling supporting cells, Mafb-EGFP (purple) labeling the interstitial/ stromal cells, Oct4-EGFP (green) labeling germ cells, and Flk1- mcherry (red) labeling endothelial cells. Scale bar = 100 mm. (TIF) Figure S2 asma-eyfp labeled a heterogeneous population containing supporting cell precursors. (A) Images of E13.5 XY and XX gonads with DAPI (blue) and asma-eyfp (pink) labeling the interstitial/stromal cells. (B) Graphs of the log-transformed, normalized intensity values. The error bars are standard error. The Sry transcript is expressed at similarly high levels in both XY supporting cells and asma-eyfp cells at E11.5, and declines rapidly in both cell types. Expression of Sry is lower in the Mafb- EGFP cells. However, the pattern seen with Sry did not hold true for most supporting cell markers: Sox9 is expressed at a lower level in both asma-eyfp and Mafb-EGFP cells than in supporting cells. (C) SRY and SOX9 proteins are also present in asma-eyfp cells. Antibodies against SRY (red) and SOX9 (blue) co-label asma- EYFP (green) cells. Cells with asma-eyfp and SRY alone are indicated with arrows, whereas cells with asma-eyfp, SRY, and SOX9 are indicated with arrowheads. Scale bar = 25 mm. This suggests that asma-eyfp is expressed in a heterogeneous population of early gonadal cells containing supporting cell progenitors. (TIF) Figure S3 Alternative methods showed generally similar patterns indicating the importance of lineage, sex, and stage. Clustering dendrograms of the individual arrays generated using (A) Average linkage with Euclidean distance as a distance metric and (B) Complete linkage with Pearson s dissimilarity as a distance metric. Consistent with Figure 2A, the arrays cluster primarily by lineage, and secondarily by sex and stage. The largest differences were in the relationship of the somatic populations to each other, although the same clusters could always be identified. (C) Examining the sources of variation with the median F ratio shows a similar pattern to the mean F ratio (Figure 2B) with the primary source of variation being lineage. (TIF) Figure S4 Overlap of genes that are sexually dimorphic in at least one lineage. (A) The number of genes over-expressed ( sexually dimorphic ) in XY or XX cells of each lineage at each stage. Genes were identified by a single pairwise comparison between XX and XY cells for each lineage at each stage (Dataset S3A). Many more genes are identified here than in Figure 3A because additional pairwise comparisons were performed to restrict the analysis to genes that showed lineage-specificity in Figure 3A. The area proportional Venn diagrams were generated using Venn Diagram Plotter v from PNNL and OMICS.PNL.GOV ( The sizing of the Venn diagrams relative to each other is approximate. Endothelial cells were not analyzed. The numbers shown indicate the number of genes exclusively in each portion of the diagram (except for E11.5 for which the total number of genes dimorphic in the supporting cells is shown). Lists marked ns had a false positive rate.20% (Dataset S3B). The overlapping areas on the Venn diagrams indicate genes sexually dimorphic in multiple lineages. Most genes dimorphic in the XX stroma were also dimorphic in another lineage. (B D) Graphs of the log-transformed, normalized intensity values. The error bars are standard error. Endothelial cell values are not shown. (B) Many genes sexually dimorphic in multiple lineages were overexpressed in one of the lineages, as was the case for Sox9. However, this could be explained by the low and variable contamination expected after FACS. To address this issue, we used antibody stains of sorted cells to estimate that the XY E13.5 germ cells had,1% contamination with supporting cells, but the XY E13.5 interstitium was more variable and had between 1% and 15% supporting cell contamination (data not shown). Therefore, patterns similar to Sox9 were not further analyzed. (C D) However, not all genes sexually dimorphic in multiple lineages had a pattern consistent with low level contamination. (C) Axin2, a Wnt/b-catenin transcriptional target gene [107], was highly expressed in the three XX cell types examined, indicative of widespread Wnt signaling in the ovary. (D) Similarly, Irx3 (Iroquois related homeobox 3), a gene known to be ovary-specific [108], showed convergent expression in the XX stroma and supporting cells. We identified more genes with a pattern similar to Irx3 as sexually dimorphic in only two lineages, expressed identically in both lineages, and enriched in these two lineages compared to the remaining two (Dataset S3C). Genes sexually dimorphic in all three lineages were not analyzed. There were few genes meeting these criteria in the overlap between the germ cells and other lineages (Dataset S3C), but (E) more genes met these criteria in the overlap between interstitial/stromal cells and supporting cells (Dataset S3C). The XX stroma in particular expressed several transcripts at similar levels to the XX supporting cells, although the populations are not identical as the XX supporting cells have many more lineage-specific transcripts (Figure 3A). Some of these shared transcripts are downstream of Wnt4 signaling in the ovary (Calb1, Fgfr2, Irx3, Sema3a, and Tkt) [109], and Wnt4 itself showed this pattern. Thus, at least some of these similarities may be attributable to widespread Wnt signaling in the ovary, as reported by Axin2 expression in all 3 lineages (C). (TIF) Figure S5 E10.5 Sf1-EGFP primed genes generally supported female-biased priming, but the E11.0 analysis was more informative. The analysis of the Sf1-EGFP primed genes (beginning the analysis at E10.5 and comparing to Sf1-EGFP cells at E12.5) was also limited to genes enriched in the Sry-EGFP/Sox9- ECFP supporting cells at E12.5 (A, B, and E) or to those genes identified by removing interstitial/stromal genes (C, D, and F). (A and C) The percentages of primed genes that were male-primed PLoS Genetics 18 March 2012 Volume 8 Issue 3 e
20 Lineage Priming of Sexual Fate in Gonadal Cells and female-primed. The first, but not the second, method showed a female bias. The boxes contain the p-values from the binomial test with the expected percentages of the extreme models. The balanced model can be rejected with the first (A), but not the second (C), method. (B and D) Examining the percentage of male or female genes that were primed similarly showed a significant (*) bias toward the female pathway, as determined by the hypergeometric test (p-value,0.05), for the first (B), but not the second (D), method (ns). (E and F) The primed genes for both sexes are predominantly similarly expressed in progenitors and E12.5 differentiated cells. However, E10.5 may not be the appropriate starting point for the priming analysis. (G) No primed genes (identified by removing interstitial/stromal genes) became dimorphic between E10.5 and E11.0 ( 0 ). Thus, starting the analysis at E11.0 does not result in the loss of any information. Starting at E11.0 is also preferable because it is closer to the divergence point. (H) Between E10.5 and E11.0, 98% of the primed genes were identically expressed in XX and XY samples at both E10.5 and E11.0, and 76% of the primed genes were identically expressed in XX and XY samples at both E11.0 and E11.5 (data not shown). Of these genes, 16% were changing expression level in the same way (see I) in both XX and XY cells between E10.5 and E11.0, whereas only 4% fell in this category between E11.0 and E11.5. This difference was significant (*), as determined by the hypergeometric test with a p-value,0.05, and affects how primed genes are called. This problem is illustrated in (I) by the graphs of the log-transformed, normalized intensity values from Sf1-EGFP cells (black) of two genes identified as primed at E10.5, but not at E11.0. Tpx2 showed significant identical changes in expression in XX and XY cells between E10.5 and E11.0. A similar pattern was observed in many other genes that did not reach significance, such as Ppil5, and so this pattern may be more pervasive than indicated by the number meeting the significance thresholds shown in H. This type of expression pattern confounds the analysis when we compare expression at E10.5 to E12.5 to identify primed genes. This may be caused by changes in the number of supporting cell progenitors, or alternatively, the continued transcriptional development of the progenitors between E10.5 and E11.0 may establish the priming program immediately preceding sexual divergence. For these reasons, we chose to start the analysis at E11.0. The gene lists for these analyses are provided in Dataset S5. (TIF) Figure S6 Overlap of primed genes between the Sf1-EGFP and Sry-EGFP/Sox9-ECFP data sets. (A) To cross-validate the analysis, we determined whether genes primed in the Sf1-EGFP data (removing interstitial/stromal genes, Figure 6E) were also identified References 1. Koopman P, Gubbay J, Vivian N, Goodfellow P, Lovell-Badge R (1991) Male development of chromosomally female mice transgenic for Sry. Nature 351: Gubbay J, Collignon J, Koopman P, Capel B, Economou A, et al. (1990) A gene mapping to the sex-determining region of the mouse Y chromosome is a member of a novel family of embryonically expressed genes. Nature 346: Karl J, Capel B (1998) Sertoli cells of the mouse testis originate from the coelomic epithelium. Dev Biol 203: Wylie C (1999) Germ cells. Cell 96: Coveney D, Cool J, Oliver T, Capel B (2008) Four-dimensional analysis of vascularization during primary development of an organ, the gonad. Proc Natl Acad Sci U S A 105: Martineau J, Nordqvist K, Tilmann C, Lovell-Badge R, Capel B (1997) Malespecific cell migration into the developing gonad. Curr Biol 7: Albrecht KH, Eicher EM (2001) Evidence that Sry is expressed in pre-sertoli cells and Sertoli and granulosa cells have a common precursor. Dev Biol 240: Adams IR, McLaren A (2002) Sexually dimorphic development of mouse primordial germ cells: switching from oogenesis to spermatogenesis. Development 129: as primed in the Sry-EGFP/Sox9-ECFP data (all genes with a priming pattern, Figure 5B). Some of the same primed genes were identified in both data sets (blue). Many more of the Sf1 primed genes were already sexually dimorphic in the Sry-EGFP/Sox9-ECFP cells by E11.5 (light and dark green). This is not surprising since primed genes were already becoming dimorphic at E11.5 based on the Sf1-EGFP data (Figure S5G). Some of the genes that were already sexually dimorphic in the E11.5 Sry-EGFP/Sox9-ECFP cells showed some indication of previous priming in the Sry-EGFP/Sox9- ECFP data (light green). These genes met the same requirements for defining a primed pattern outlined in the Materials and Methods, but rather than being identical at E11.5, the sex for which the gene was primed had higher expression at E11.5. This pattern is illustrated in (B) by the graphs of the log-transformed, normalized intensity values for the Sf1-EGFP (black) and Sry-EGFP/Sox9-ECFP (blue) cells for the gene Dock4. The error bars are standard error. Together, these primed or E11.5 dimorphic categories account for over half of the Sf1-EGFP primed genes, indicating both arrays showed consistent results for many genes. A number of Sf1-EGFP primed genes (48%) were not identified as primed or sexually dimorphic in Sry-EGFP/Sox9-ECFP cells at E11.5 (yellow). This was expected as probe sets for the two arrays, as well as the cell types collected, were different. The identification of similar patterns for the same genes in these two different data sets despite their differences gives us confidence in the results. The gene lists for these analyses are provided in Dataset S5F. (TIF) Acknowledgments We gratefully acknowledge Bruce Aronow s helpful advice in selecting appropriate methods for processing our arrays. We thank Serge Nef for the outstanding publically available Sf1-EGFP data set that he produced. We would also like to thank Nugen for their excellent customer service and technical support at all stages of the sample processing, the Duke Institute for Genome Sciences and Policy Microarray Facility for their assistance collecting these data, and Duke Comprehensive Cancer Center Flow Cytometry Shared Resource for their exceptional efforts to assist us in collecting the cells we required to make this study possible. Finally, we appreciate the supportive environment of the Duke Center for Systems Biology. Author Contributions Conceived and designed the analysis: SAJ AN BC. Provided input in writing the manuscript: AN DMM LM SCM. Conceived and designed the experiments: BC. Performed the experiments: SAJ JC TD DMM. Analyzed the data: SAJ AN. Wrote the paper: SAJ BC. 9. Swain A, Lovell-Badge R (1999) Mammalian sex determination: a molecular drama. Genes Dev 13: Bouma GJ, Hudson QJ, Washburn LL, Eicher EM (2010) New candidate genes identified for controlling mouse gonadal sex determination and the early stages of granulosa and Sertoli cell differentiation. Biol Reprod 82: Nef S, Schaad O, Stallings NR, Cederroth CR, Pitetti JL, et al. (2005) Gene expression during sex determination reveals a robust female genetic program at the onset of ovarian development. Dev Biol 287: Mise N, Fuchikami T, Sugimoto M, Kobayakawa S, Ike F, et al. (2008) Differences and similarities in the developmental status of embryo-derived stem cells and primordial germ cells revealed by global expression profiling. Genes Cells 13: Beverdam A, Koopman P (2006) Expression profiling of purified mouse gonadal somatic cells during the critical time window of sex determination reveals novel candidate genes for human sexual dysgenesis syndromes. Hum Mol Genet 15: Rolland AD, Lehmann KP, Johnson KJ, Gaido KW, Koopman P (2011) Uncovering gene regulatory networks during mouse fetal germ cell development. Biol Reprod 84: PLoS Genetics 19 March 2012 Volume 8 Issue 3 e
21 Lineage Priming of Sexual Fate in Gonadal Cells 15. Bouma GJ, Affourtit JP, Bult CJ, Eicher EM (2007) Transcriptional profile of mouse pre-granulosa and Sertoli cells isolated from early-differentiated fetal gonads. Gene Expr Patterns 7: Brady SM, Orlando DA, Lee JY, Wang JY, Koch J, et al. (2007) A highresolution root spatiotemporal map reveals dominant expression patterns. Science 318: Saitou M, Barton SC, Surani MA (2002) A molecular programme for the specification of germ cell fate in mice. Nature 418: Brunskill EW, Aronow BJ, Georgas K, Rumballe B, Valerius MT, et al. (2008) Atlas of gene expression in the developing kidney at microanatomic resolution. Dev Cell 15: Jost A (1947) Recherches sur la diff erenciation sexuelle de l embryon de lapin. Arch Anat Microsc Morph Exp. pp McLaren A (1991) Development of the mammalian gonad: the fate of the supporting cell lineage. Bioessays 13: Capel B (1998) Sex in the 90s: SRY and the switch to the male pathway. Annu Rev Physiol 60: McElreavey K, Vilain E, Abbas N, Herskowitz I, Fellous M (1993) A regulatory cascade hypothesis for mammalian sex determination: SRY represses a negative regulator of male development. Proc Natl Acad Sci U S A 90: Eicher EM, Washburn LL (1986) Genetic control of primary sex determination in mice. Annu Rev Genet 20: Kim Y, Kobayashi A, Sekido R, DiNapoli L, Brennan J, et al. (2006) Fgf9 and Wnt4 act as antagonistic signals to regulate mammalian sex determination. PLoS Biol 4: e187. doi: /journal.pbio Chassot AA, Ranc F, Gregoire EP, Roepers-Gajadien HL, Taketo MM, et al. (2008) Activation of beta-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Hum Mol Genet 17: Swain A, Narvaez V, Burgoyne P, Camerino G, Lovell-Badge R (1998) Dax1 antagonizes Sry action in mammalian sex determination. Nature 391: Munger SC, Aylor DL, Syed HA, Magwene PM, Threadgill DW, et al. (2009) Elucidation of the transcription network governing mammalian sex determination by exploiting strain-specific susceptibility to sex reversal. Genes Dev 23: Delorme B, Ringe J, Pontikoglou C, Gaillard J, Langonne A, et al. (2009) Specific lineage-priming of bone marrow mesenchymal stem cells provides the molecular framework for their plasticity. Stem Cells 27: Hu M, Krause D, Greaves M, Sharkis S, Dexter M, et al. (1997) Multilineage gene expression precedes commitment in the hemopoietic system. Genes Dev 11: Ng SY, Yoshida T, Zhang J, Georgopoulos K (2009) Genome-wide lineagespecific transcriptional networks underscore Ikaros-dependent lymphoid priming in hematopoietic stem cells. Immunity 30: Zipori D (2004) The nature of stem cells: state rather than entity. Nat Rev Genet 5: Miyamoto T, Iwasaki H, Reizis B, Ye M, Graf T, et al. (2002) Myeloid or lymphoid promiscuity as a critical step in hematopoietic lineage commitment. Dev Cell 3: Enver T, Greaves M (1998) Loops, lineage, and leukemia. Cell 94: Hipp JA, Hipp JD, Atala A, Soker S (2010) Functional genomics: new insights into the function of low levels of gene expression in stem cells. Curr Genomics 11: Golan-Mashiach M, Dazard JE, Gerecht-Nir S, Amariglio N, Fisher T, et al. (2005) Design principle of gene expression used by human stem cells: implication for pluripotency. FASEB J 19: Guo G, Huss M, Tong GQ, Wang C, Li Sun L, et al. (2010) Resolution of cell fate decisions revealed by single-cell gene expression analysis from zygote to blastocyst. Dev Cell 18: Dietrich JE, Hiiragi T (2007) Stochastic patterning in the mouse preimplantation embryo. Development 134: Plusa B, Piliszek A, Frankenberg S, Artus J, Hadjantonakis AK (2008) Distinct sequential cell behaviours direct primitive endoderm formation in the mouse blastocyst. Development 135: Kim Y, Bingham N, Sekido R, Parker KL, Lovell-Badge R, et al. (2007) Fibroblast growth factor receptor 2 regulates proliferation and Sertoli differentiation during male sex determination. Proc Natl Acad Sci U S A 104: Moriguchi T, Hamada M, Morito N, Terunuma T, Hasegawa K, et al. (2006) MafB is essential for renal development and F4/80 expression in macrophages. Mol Cell Biol 26: DeFalco T, Takahashi S, Capel B (2011) Two distinct origins for Leydig cell progenitors in the fetal testis. Dev Biol 352: Szabo PE, Hubner K, Scholer H, Mann JR (2002) Allele-specific expression of imprinted genes in mouse migratory primordial germ cells. Mech Dev 115: Larina IV, Shen W, Kelly OG, Hadjantonakis AK, Baron MH, et al. (2009) A membrane associated mcherry fluorescent reporter line for studying vascular remodeling and cardiac function during murine embryonic development. Anat Rec (Hoboken) 292: Poche RA, Larina IV, Scott ML, Saik JE, West JL, et al. (2009) The Flk1- myr::mcherry mouse as a useful reporter to characterize multiple aspects of ocular blood vessel development and disease. Dev Dyn 238: Cool J, Carmona FD, Szucsik JC, Capel B (2008) Peritubular myoid cells are not the migrating population required for testis cord formation in the XY gonad. Sex Dev 2: Sekido R, Bar I, Narvaez V, Penny G, Lovell-Badge R (2004) SOX9 is upregulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev Biol 274: Yao HH, Aardema J, Holthusen K (2006) Sexually dimorphic regulation of inhibin beta B in establishing gonadal vasculature in mice. Biol Reprod 74: Jeanes A, Wilhelm D, Wilson MJ, Bowles J, McClive PJ, et al. (2005) Evaluation of candidate markers for the peritubular myoid cell lineage in the developing mouse testis. Reproduction 130: Tsuda M, Sasaoka Y, Kiso M, Abe K, Haraguchi S, et al. (2003) Conserved role of nanos proteins in germ cell development. Science 301: Menke DB, Koubova J, Page DC (2003) Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. Dev Biol 262: Fong GH, Rossant J, Gertsenstein M, Breitman ML (1995) Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 376: Ikeda Y, Shen WH, Ingraham HA, Parker KL (1994) Developmental expression of mouse steroidogenic factor-1, an essential regulator of the steroid hydroxylases. Mol Endocrinol 8: Beckervordersandforth R, Tripathi P, Ninkovic J, Bayam E, Lepier A, et al. (2010) In vivo fate mapping and expression analysis reveals molecular hallmarks of prospectively isolated adult neural stem cells. Cell Stem Cell 7: Fujikura J, Yamato E, Yonemura S, Hosoda K, Masui S, et al. (2002) Differentiation of embryonic stem cells is induced by GATA factors. Genes Dev 16: Huber O, Korn R, McLaughlin J, Ohsugi M, Herrmann BG, et al. (1996) Nuclear localization of beta-catenin by interaction with transcription factor LEF-1. Mech Dev 59: Maatouk DM, DiNapoli L, Alvers A, Parker KL, Taketo MM, et al. (2008) Stabilization of beta-catenin in XY gonads causes male-to-female sex-reversal. Hum Mol Genet 17: Schmahl J, Eicher EM, Washburn LL, Capel B (2000) Sry induces cell proliferation in the mouse gonad. Development 127: Mork L, Maatouk DM, McMahon JA, Guo JJ, Zhang P, et al. (2011) Temporal differences in granulosa cell specification in the ovary reflect distinct follicle fates in mice. Biol Reprod. 59. Burgoyne PS, Buehr M, Koopman P, Rossant J, McLaren A (1988) Cellautonomous action of the testis-determining gene: Sertoli cells are exclusively XY in XX----XY chimaeric mouse testes. Development 102: Mansson R, Hultquist A, Luc S, Yang L, Anderson K, et al. (2007) Molecular evidence for hierarchical transcriptional lineage priming in fetal and adult stem cells and multipotent progenitors. Immunity 26: Raz E (2000) The function and regulation of vasa-like genes in germ-cell development. Genome Biol 1: REVIEWS Cook MS, Munger SC, Nadeau JH, Capel B (2011) Regulation of male germ cell cycle arrest and differentiation by DND1 is modulated by genetic background. Development 138: Kent J, Wheatley SC, Andrews JE, Sinclair AH, Koopman P (1996) A malespecific role for SOX9 in vertebrate sex determination. Development 122: Morais da Silva S, Hacker A, Harley V, Goodfellow P, Swain A, et al. (1996) Sox9 expression during gonadal development implies a conserved role for the gene in testis differentiation in mammals and birds. Nat Genet 14: Bradford ST, Hiramatsu R, Maddugoda MP, Bernard P, Chaboissier MC, et al. (2009) The cerebellin 4 precursor gene is a direct target of SRY and SOX9 in mice. Biol Reprod 80: Vainio S, Heikkila M, Kispert A, Chin N, McMahon AP (1999) Female development in mammals is regulated by Wnt-4 signalling. Nature 397: Davidson EH, Rast JP, Oliveri P, Ransick A, Calestani C, et al. (2002) A genomic regulatory network for development. Science 295: Larsson O, Scheele C, Liang Z, Moll J, Karlsson C, et al. (2004) Kinetics of senescence-associated changes of gene expression in an epithelial, temperaturesensitive SV40 large T antigen model. Cancer Res 64: Badie C, Itzhaki JE, Sullivan MJ, Carpenter AJ, Porter AC (2000) Repression of CDK1 and other genes with CDE and CHR promoter elements during DNA damage-induced G(2)/M arrest in human cells. Mol Cell Biol 20: Laslo P, Spooner CJ, Warmflash A, Lancki DW, Lee HJ, et al. (2006) Multilineage transcriptional priming and determination of alternate hematopoietic cell fates. Cell 126: Parma P, Radi O, Vidal V, Chaboissier MC, Dellambra E, et al. (2006) R- spondin1 is essential in sex determination, skin differentiation and malignancy. Nat Genet 38: Liu CF, Bingham N, Parker K, Yao HH (2009) Sex-specific roles of betacatenin in mouse gonadal development. Hum Mol Genet 18: Jeays-Ward K, Hoyle C, Brennan J, Dandonneau M, Alldus G, et al. (2003) Endothelial and steroidogenic cell migration are regulated by WNT4 in the developing mammalian gonad. Development 130: PLoS Genetics 20 March 2012 Volume 8 Issue 3 e
22 Lineage Priming of Sexual Fate in Gonadal Cells 74. Bernstein BE, Mikkelsen TS, Xie X, Kamal M, Huebert DJ, et al. (2006) A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125: Matson CK, Murphy MW, Sarver AL, Griswold MD, Bardwell VJ, et al. (2011) DMRT1 prevents female reprogramming in the postnatal mammalian testis. Nature 476: Raymond CS, Murphy MW, O Sullivan MG, Bardwell VJ, Zarkower D (2000) Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation. Genes Dev 14: Chaboissier MC, Kobayashi A, Vidal VI, Lutzkendorf S, van de Kant HJ, et al. (2004) Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development 131: Yao HH, Matzuk MM, Jorgez CJ, Menke DB, Page DC, et al. (2004) Follistatin operates downstream of Wnt4 in mammalian ovary organogenesis. Dev Dyn 230: Vidal VP, Chaboissier MC, de Rooij DG, Schedl A (2001) Sox9 induces testis development in XX transgenic mice. Nat Genet 28: Ewen KA, Koopman P (2010) Mouse germ cell development: from specification to sex determination. Mol Cell Endocrinol 323: McLaren A, Southee D (1997) Entry of mouse embryonic germ cells into meiosis. Dev Biol 187: Upadhyay S, Zamboni L (1982) Ectopic germ cells: natural model for the study of germ cell sexual differentiation. Proc Natl Acad Sci U S A 79: Chuma S, Nakatsuji N (2001) Autonomous transition into meiosis of mouse fetal germ cells in vitro and its inhibition by gp130-mediated signaling. Dev Biol 229: Farini D, Scaldaferri ML, Iona S, La Sala G, De Felici M (2005) Growth factors sustain primordial germ cell survival, proliferation and entering into meiosis in the absence of somatic cells. Dev Biol 285: Bowles J, Knight D, Smith C, Wilhelm D, Richman J, et al. (2006) Retinoid signaling determines germ cell fate in mice. Science 312: Kumar S, Chatzi C, Brade T, Cunningham TJ, Zhao X, et al. (2011) Sexspecific timing of meiotic initiation is regulated by Cyp26b1 independent of retinoic acid signalling. Nat Commun 2: Koubova J, Menke DB, Zhou Q, Capel B, Griswold MD, et al. (2006) Retinoic acid regulates sex-specific timing of meiotic initiation in mice. Proc Natl Acad Sci U S A 103: Byskov AG (1974) Does the rete ovarii act as a trigger for the onset of meiosis? Nature 252: O W, Baker TG (1976) Initiation and control of meiosis in hamster gonads in vitro. J Reprod Fertil 48: DiNapoli L, Batchvarov J, Capel B (2006) FGF9 promotes survival of germ cells in the fetal testis. Development 133: Bowles J, Feng CW, Spiller C, Davidson TL, Jackson A, et al. (2010) FGF9 suppresses meiosis and promotes male germ cell fate in mice. Dev Cell 19: Barrios F, Filipponi D, Pellegrini M, Paronetto MP, Di Siena S, et al. (2010) Opposing effects of retinoic acid and FGF9 on Nanos2 expression and meiotic entry of mouse germ cells. J Cell Sci 123: Francavilla S, Zamboni L (1985) Differentiation of mouse ectopic germinal cells in intra- and perigonadal locations. J Exp Zool 233: Best D, Sahlender DA, Walther N, Peden AA, Adams IR (2008) Sdmg1 is a conserved transmembrane protein associated with germ cell sex determination and germline-soma interactions in mice. Development 135: Phillips BT, Gassei K, Orwig KE (2010) Spermatogonial stem cell regulation and spermatogenesis. Philos Trans R Soc Lond B Biol Sci 365: Colvin JS, Green RP, Schmahl J, Capel B, Ornitz DM (2001) Male-to-female sex reversal in mice lacking fibroblast growth factor 9. Cell 104: Ideker T, Galitski T, Hood L (2001) A new approach to decoding life: systems biology. Annu Rev Genomics Hum Genet 2: Brady SM, Zhang L, Megraw M, Martinez NJ, Jiang E, et al. (2011) A steleenriched gene regulatory network in the Arabidopsis root. Mol Syst Biol 7: Bullejos M, Koopman P (2001) Spatially dynamic expression of Sry in mouse genital ridges. Dev Dyn 221: Sekido R, Lovell-Badge R (2008) Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature 453: Mroz K, Carrel L, Hunt PA (1999) Germ cell development in the XXY mouse: evidence that X chromosome reactivation is independent of sexual differentiation. Dev Biol 207: Cook MS, Coveney D, Batchvarov I, Nadeau JH, Capel B (2009) BAXmediated cell death affects early germ cell loss and incidence of testicular teratomas in Dnd1(Ter/Ter) mice. Dev Biol 328: Barske LA, Capel B (2010) Estrogen represses SOX9 during sex determination in the red-eared slider turtle Trachemys scripta. Dev Biol 341: Huang da W, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4: Huang da W, Sherman BT, Lempicki RA (2009) Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 37: Liu G, Loraine AE, Shigeta R, Cline M, Cheng J, et al. (2003) NetAffx: Affymetrix probesets and annotations. Nucleic Acids Res 31: Jho EH, Zhang T, Domon C, Joo CK, Freund JN, et al. (2002) Wnt/betacatenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol 22: Jorgensen JS, Gao L (2005) Irx3 is differentially up-regulated in female gonads during sex determination. Gene Expr Patterns 5: Coveney D, Ross AJ, Slone JD, Capel B (2008) A microarray analysis of the XX Wnt4 mutant gonad targeted at the identification of genes involved in testis vascular differentiation. Gene Expr Patterns 8: PLoS Genetics 21 March 2012 Volume 8 Issue 3 e
23 BIOLOGY OF REPRODUCTION (2013) 88(6):143, 1 11 Published online before print 24 April DOI /biolreprod MicroRNAs-140-5p/140-3p Modulate Leydig Cell Numbers in the Developing Mouse Testis 1 Joanna Rakoczy, 3,5 Selene L. Fernandez-Valverde, 3,6 Evgeny A. Glazov, 6 Elanor N. Wainwright, 5 Tempei Sato, 7 Shuji Takada, 7 Alexander N. Combes, 5 Darren J. Korbie, 6 David Miller, 6 Sean M. Grimmond, 6 Melissa H. Little, 5 Hiroshi Asahara, 7 John S. Mattick, 4,5 Ryan J. Taft, 3,6 and Dagmar Wilhelm 2,3,5 5 Division of Molecular Genetics and Development, Institute for Molecular Biosciences, The University of Queensland, Brisbane, Queensland, Australia 6 Genomics & Computational Biology, Institute for Molecular Biosciences, The University of Queensland, Brisbane, Queensland, Australia 7 Department of Systems BioMedicine, National Institute for Child Health and Development, Okura, Setagaya, Tokyo, Japan ABSTRACT MicroRNAs (mirnas) have been shown to play key regulatory roles in a range of biological processes, including cell differentiation and development. To identify mirnas that participate in gonad differentiation, a fundamental and tightly regulated developmental process, we examined mirna expression profiles at the time of sex determination and during the early fetal differentiation of mouse testes and ovaries using highthroughput sequencing. We identified several mirnas that were expressed in a sexually dimorphic pattern, including several members of the let-7 family, mir-378, and mir-140-3p. We focused our analysis on the most highly expressed, sexually dimorphic mirna, mir-140-3p, and found that both mir-140-3p and its more lowly expressed counterpart, the previously annotated guide strand, mir-140-5p, are testis enriched and expressed in testis cords. Analysis of the mir-140-5p/mir-140-3p-null mouse revealed a significant increase in the number of Leydig cells in the developing XY gonad, strongly suggesting an important role for mir-140-5p/mir-140-3p in testis differentiation in mouse. gonad development, high-throughput sequencing, microrna, sex determination, strand selection, testis differentiation INTRODUCTION Sex determination, which is regulated by a number of molecular pathways, is a fundamental process for the maintenance and continuation of life. In mammals, sex is determined genetically through the inheritance of an X or a Y 1 Supported by research grants from the Australian Research Council (ARC; grant DP ) and National Health and Medical Research Council of Australia (NHMRC; grant ). R.J.T is supported by an ARC Discovery Early Career Research Award (DE ). D.W. is a Future Fellow of the ARC (FT ). 2 Correspondence and current address: Dagmar Wilhelm, Department of Anatomy and Developmental Biology, Monash University, Bldg. 76 Wellington Rd, Clayton, VIC 3800, Australia. dagmar.wilhelm@monash.edu 3 These authors contributed equally to this work. 4 Current address: Garvan Institute of Medical Research, Darlinghurst, NSW 2010, Australia. Received: 1 January First decision: 25 January Accepted: 19 April Ó 2013 by the Society for the Study of Reproduction, Inc. eissn: ISSN: chromosome from the father. The main embryonic process that is influenced by the sex chromosomes is the differentiation of the bipotential genital ridge into a testis or an ovary. The Y chromosome harbors the testis-determining gene Sry, which, in mouse, is transiently expressed in the supporting cell precursors of the indifferent genital ridge from 10.5 to 12.5 days postcoitum (dpc) [1 3]. SRY directly up-regulates the related factor SOX9 [4], which drives the differentiation of these precursor cells into Sertoli cells [5, 6]. Sertoli cells then assemble around clusters of primordial germ cells (PGCs) to form the testis cords [7]. Sertoli cells also coordinate the differentiation of other cell types in the developing testis, including the migration of endothelial cells from the underlying mesonephros to form the testis-specific vasculature [8, 9] and the differentiation of interstitial cells into long, flattened peritubular myoid cells that will form a contractile layer around the testis cords [10]. Another cell type that differentiates from interstitial precursor cells is the steroidogenic Leydig cell. Leydig cells are initially specified by the morphogen desert hedgehog (DHH) secreted by Sertoli cells [11]. Additional Leydig cells are then recruited from undifferentiated progenitors, with the Notch signaling pathway maintaining the progenitor population and restricting their differentiation into fetal Leydig cells [12]. In the absence of the Y chromosome, or if Sry is nonfunctional, ovarian determining pathways are activated, and the bipotential genital ridge will develop into an ovary. In contrast to the testis, the ovary changes very little morphologically during the first few days during embryonic development. At around 13.5 dpc, PGCs in an ovary will start to form germ cell nests or cysts and, as part of their differentiation process, start to enter meiosis in an anterior-to-posterior wave [13, 14]. Only after birth will the germ cell cysts break apart, with single germ cells becoming surrounded by granulosa cells, the supporting cell lineage of the ovary, to form the primordial follicle [15]. While less than 1.5% of the mammalian transcriptome encodes proteins, at least 80% is transcribed (ENCODE 2012), suggesting that the mammalian genome encodes a vast suite of RNAs, most of which may be regulatory [16] and are likely to play key roles in developmental processes, including gonad formation. MicroRNAs (mirnas) are the most well-studied class of small nonprotein-coding RNA and are now known to affect nearly all fundamental cellular processes, including cell differentiation, proliferation, cell-cycle regulation, and development [17 19]. These 20- to 22-nucleotide (nt)-long RNAs 1 Article 143 Downloaded from
24 RAKOCZY ET AL. are transcribed in the nucleus as long primary transcripts and cleaved to form a 70- to 80-nt stem-loop precursor. This premirna is further processed in the cytoplasm into a ragged-end RNA duplex, normally thought to consist of the so-called mature mirna, and its presumed nonfunctional counter part, dubbed the mirna* [20]. The most widely accepted models posit that the mature mirna is selected as the guide strand, which mediates gene regulation by directing the RNAinduced silencing complex RISC to a target mrna, while the mirna* strand is quickly degraded [21]. Previous expression profiling experiments investigating fetal and postnatal mouse ovaries and testes have demonstrated that mirnas are expressed in a sexually dimorphic pattern [22 25], including high expression of mir-106b and mir-107 in testes and high levels of let-7a, let-7b, and let-7c in ovaries [22]. To identify the role mirnas play in gonad development, a conditional Dicer1 knockout mouse was used to completely ablate mirnas in the gonad. These mice displayed a relatively late phenotype after birth [26 29]. This late phenotype is due to the fact that the Cre deleter in mice used in these experiments is activated at around 13.5 dpc in the differentiating gonad, 2 days after sex is determined by the expression of Sry. In addition, Papaioannou et al. [29] found that mirna levels decreased only very slowly following Dicer1 deletion, demonstrating that mirnas are stable for days. It is, therefore, clear that studies of mirna function at early gonad development time points must be restricted to individual mirnas that can be selectively ablated. In the present study, we investigated the mirna expression profiles between the developing XY and XX gonad at 11.5, 12.5, and 13.5 dpc using high-throughput sequencing. This approach allowed us to successfully identify sexually dimorphic expression of 46 mirnas. Candidates were validated, and further characterization of the mir-140 knockout mouse established a role for mir-140-5p/mir-140-3p in testis differentiation. MATERIALS AND METHODS Animal Collection, Gonad Tissue Collection, and Embryo Sexing Embryos were collected from timed matings of outbred CD1-strain mice, with noon of the day on which the mating plug was observed designated as 0.5 dpc. Embryo sex was determined using PCR, with tail DNA for the Y chromosome gene Zfy (5P: 5 0 -GAC TAG ACA TGT CTT AAC ATC TGT CC- 3 0 ; 3P: 5 0 -CCT ATT GVA TGG ACT GVA GCT TAT G-3 0 ) and the X chromosome gene Ube1x (5P: 5 0 -TGG TCT GGA CCC AAA CGC TGT CCA CA-3 0 ; 3P:5 0 -GGC AGC AGC CAT CAC ATA ATC CAG ATG-3 0 ). Protocols and use of animals were approved by the Animal Welfare Unit of the University of Queensland (approval IMB/131/09/ARC), which is registered as an institution that uses animals for scientific purposes under the Queensland Animal Care and Protection Act. The generation of the pre-mir-140-null mice has been described previously [30], and experiments were performed according to protocols approved by the Institutional Animal Care and Use Committee at the National Institute for Child Health and Development. Small RNA Library Construction and Sequencing To analyze differences between XY and XX developing gonads, gonads (mesonephroi removed) were dissected from 11.5, 12.5, and 13.5 dpc mouse embryos. Small RNAs from all samples were prepared in parallel using the mirneasy Mini Kit (Qiagen) from pooled testes and ovaries from 11.5 dpc to 13.5 dpc, with 15 genital ridges per pool at 11.5 dpc and gonad pairs per pool at 12.5 and 13.5 dpc. The quality of the RNA was analyzed using a BioAnalyzer and was of high quality (RIN score.8). RNA (1.5 lg) was used for each sex and stage to prepare six libraries, which were sequenced on the Illumina Genome Analyzer II platform (Illumina) using a standard single-read 36-cycle sequencing protocol and Illumina s sequencing reagents according to the manufacturer s recommendations. Each library was sequenced individually on a single-flow cell lane (Supplemental Table S1; all Supplemental Data are available online at To further quantify mirna expression, we investigated small RNAs (15 36 nt) from testes and, for comparison, limb buds from 13.5 dpc mouse embryos using the MiSeq platform (Illumina; Supplemental Table S1). Independent, directional small RNA libraries suitable for sequencing on the MiSeq platform were prepared using TruSeq Small RNA Sample Preparation Kit (part# RS ; Illumina) following the manufacturer s recommendations. Libraries were size selected as per the standard protocol and were quantified on the Agilent BioAnalyzer 2100 (G2939AA; Agilent) using the High Sensitivity DNA kit ( ; Agilent). Quantified libraries were subsequently sequenced on the Illumina MiSeq sequencing platform running software version and a titration point of 8pM for cluster generation. Each library was sequenced as per the manufacturer s recommendations using MiSeq Reagent Kit 50 Cycles (MS ; Illumina), with matched lot numbers generating reads of 50 bp. A single index read of 6 bp was generated in addition to the 50-bp read to positively identify samples and minimize cross talk. Small RNA Mapping and Normalization In all cases, high-throughput sequencing tags were adaptor trimmed using the fastx toolkit ( and mapped to the latest version of the mouse genome (NCBI37/mm9) using Bowtie [31]. To avoid erroneous identification of mirnas, no mismatches were allowed. To enable comparison between libraries, tags were normalized by library depth, or counts per million (cpm). Briefly, the abundance of each sequence detected was divided by the total abundance of small RNAs in each library (see Supplemental Table S1), and the result was divided by mirna Expression Comparison The mapped positions of the small RNA sequencing tags were intersected with the annotated genomic positions of mature and star (*) mouse mirnas in mirbase (Release 15.0; [32]. Only tags that mapped within an mirna locus (mature or star) flanked by six nt on both sides of the mirbase annotation were classified as mirnas. Only mirnas mapping to a single location in the genome or mapping within a known mirna precursor were used for differential expression analysis. Normalized expression level (cpm) of each mirna was compared between XY and XX gonads for each sequenced developmental stage. All analyses were performed using a series of in-house-developed Perl scripts and the R statistical analysis software ( Target Prediction Messenger RNA targets for mir-140-3p and mir-140-5p within the Notch signaling pathway were predicted in silico using MirWalk [33]. To identify other potential gene targets for mir-140-3p, the target predictions for the two most prevalent isomirs were passed into TargetScan 5.2 Custom [34], MirWalk, and RNA22 [35], and the prediction sets were intersected. Whenever a target was predicted by at least two programs, it was considered as a gene likely to be targeted by mir-140-3p. Quantitative Reverse Transcription PCR Gonads from 11.5 dpc to 13.5 dpc were dissected, and RNA was extracted using an mirneasy RNA Mini Kit following the manufacturer s instructions and eluted in 30 ll RNase-free water. RNA was reverse transcribed using the Multiscribe Reverse Transcription enzyme (Applied Biosystems), with the following parameters: 30 min at 168C, 30 min at 428C, 5 min at 858C. Real-time PCR was performed using the Taqman Small RNA Assay (Applied Biosystems) following the manufacturer s instructions and the ABI- PRISM 7000 sequence detection system (Applied Biosystems) with the following parameters: 958C for 10 min, 958C for 15 sec, and 608C for 60 sec, repeated over 40 cycles. Quantitative reverse transcription PCR (qrt-pcr) was performed in triplicate with at least three independent biological samples. Results were normalized to the small nucleolar RNA snorna202. Significance was tested using Student t-test for samples of unequal variance. Locked Nucleic Acid In Situ Hybridization The double digoxigenin-labeled, predesigned mircury locked nucleic acid (LNA) probes for section in situ hybridization (ISH) detection of mir-140-5p and mir-140-3p, as well as an mir-scramble probe, were obtained from Exiqon. Embryos were fixed in 4% paraformaldehyde (PFA) in PBS (PFA/ PBS), embedded in OCT compound (Sakura Finetek), and cryo-sectioned at 10 2 Article 143 Downloaded from
25 ROLE OF mir-140-3p IN TESTIS DEVELOPMENT lm. LNA-ISH was conducted as previously described [36], with the following modifications. Hybridization of the probe occurred overnight with 1 pmol/ll probe diluted in prehybridization solution at 588C. Postantibody washes, three times for 5 min each, were performed in NT buffer (150 mm NaCl and 50 mm Tris-HCl [ph 7.5]), followed by incubation for 10 min in NTM buffer (100 mm NaCl, 100 mm Tris-HCl [ph 9.5], and 50 mm MgCl 2 ). The color reaction was prepared with 3.5 ll nitroblue tetrazolium and 3.5 ll 5-bromo-4-chloro-3- indolyl-phosphate (Roche) per milliliter NTM and incubated in the dark until color developed. The reaction was terminated in PBS, and samples were postfixed in 4% PFA/PBS for 10 min at room temperature. Slides were mounted with Mount-Quick aqueous mounting medium (SPI Supplies) and photographed using an Olympus BX-51 microscope (Olympus). Whole-Mount Immunofluorescence and Quantification Mouse embryonic gonads were collected at 13.5 and 15.5 dpc, fixed for several hours with 4% PFA/PBS, washed three times with PBS þ 0.1% Triton 3-100, and stored in 100% MeOH. To reduce the background, samples were incubated in a 3:2:1 solution of (respectively) methanol, dimethyl sulfoxide, and H 2 O 2. Immunofluorescence was performed as described before [8]. Anti- SCC antibody [37] was used at a 1:300 dilution and detected with anti-rabbit alexa 647 (Invitrogen). Samples were imaged on a Zeiss LSM 510 Meta confocal microscope (Zeiss) using a 403 oil objective. Data were acquired as nonoverlapping, consecutive, 2-lm optical sections until the depth at which the working distance for the objective was reached. Overlapping fields of view were stitched using the Fiji image analysis program ( All SCCpositive cells were counted on every third optical section for the first 72 lm of each sample and then averaged. This process was repeated for three wild-type and three knockout samples at 13.5 dpc and four wild-type and four knockout samples at 15.5 dpc. Data are represented as an average of all samples within a genotype and age, and error bars represent the SEM. Significance was tested using Student t-test for samples of unequal variance. Northern Blot Gonads of 13.5 dpc mouse embryos were dissected, and RNA was extracted using the mirneasy RNA Mini Kit following the manufacturer s instructions. To concentrate the RNA, samples were pooled, and 400 ll of TRL buffer (Qiagen) and 1 ml of 100% EtOH were added before being loaded onto a microspin column (Qiagen), washed with 500 ll of RPE buffer (Qiagen), washed with 80% EtOH, and eluted in 20 ll of RNase-free water. Northern blot analysis was conducted following the protocol previously described [38]. DNA oligonucleotides (10 pmol) were radiolabeled with 2 ll 32 P c-atp (3000 Ci/ mmol; Perkin Elmer) and T4 Polynucleotide Kinase (NEB) at 378C f or 1 h. Free nt were removed through a G-50 spin column (GE Healthcare Life Sciences). The labeled probe was heated to 858C for 5 min and hybridized to the membrane. Hybridization was carried out following the protocol reviewed by the Bartel laboratory [39]. RESULTS Small RNAs Show Sexually Dimorphic Expression During Gonad Development To identify small RNAs that expressed a sexually dimorphic manner, we sequenced small RNAs extracted from XY and XX mouse fetal gonads at 11.5, 12.5 and 13.5 dpc. From a total of 717 mirnas annotated in mirbase 15 [32], 331 mirnas were expressed during gonad development. Of these, 46 mirnas were differentially expressed more than 1.5-fold at 11.5 dpc and more than 2-fold at 12.5 and 13.5 dpc, between testes and ovaries (Supplemental Table S2). These mirnas included members of the well-characterized let-7 family; let-7c and let-7e were enriched in XY genital ridges at 11.5 dpc (Supplemental Table S2), and let-7d was expressed at higher levels in ovaries compared to testes at 13.5 dpc (Supplemental Table S2). However, to eliminate the potentially confounding effects of multimapping small RNA deep-sequencing reads, we chose to restrict our analysis to mirnas that could be analyzed unambiguously, i.e., mirna loci that were entirely unique within the mouse genome. Twenty-three of the 46 differentially expressed mirnas fulfilled this requirement (Fig. 1, Table 1, and Supplemental Fig. S1), enabling us to unequivocally TABLE 1. Uniquely mapping, sexually dimorphic, expressed mirnas. mirna Stages Chromosome Testis-enriched mir-130a 11.5 dpc 2 mir-434-5p 11.5 dpc 12 mir dpc 4 mir-202-3p 12.5 and 13.5 dpc 7 mir-202-5p 12.5 and 13.5 dpc 7 mir-140-5p 13.5 dpc 8 mir-140-3p 13.5 dpc 8 mir dpc X mir dpc X mir-743a 13.5 dpc X mir-540-3p 13.5 dpc 12 mir-465a-5p 13.5 dpc X mir dpc 9 mir dpc X mir-743b-3p 13.5 dpc X mir-878-5p 13.5 dpc X mir dpc 12 Ovary-enriched mir dpc 12 mir-423-5p 11.5 dpc 11 mir-878-5p 12.5 dpc X mir dpc 16 mir dpc 18 mir dpc 1 quantify their expression. Among these mirnas (Fig. 1), expression of 17 was testis enriched, and six ovary enriched (Table 1), consistent with the fact that during this time period the testis undergoes major morphological changes, whereas the ovary remains relatively quiescent. Of the 17 testis-enriched, expressed mirnas, seven are encoded on the X chromosome (Table 1). The testis-enriched mirna mir-140-3p (formerly mir-140*) and the ovary-enriched mirna mir-378 showed the highest overall expression levels. Interestingly, mir-485 and mir-878-5p expression switched from being ovary enriched at 11.5 dpc and 12.5 dpc, respectively, to testisenriched expression at 13.5 dpc, thereby representing the first examples of genetic elements whose expression switches between these two organs. mir-140-3p Is Expressed in the Developing Testis We chose the candidate with the highest overall expression level, mir-140-3p, for detailed analysis. To confirm the dynamics and sex-specific up-regulation of this mirna, we performed qrt-pcr of embryonic gonads from 11.5 dpc to 13.5 dpc (Fig. 2A, right panel) and compared the results with those obtained from high-throughput sequencing (Fig. 2A, left panel). Both approaches showed that mir-140-3p was expressed at low levels at 11.5 dpc in XX and XY genital ridges, but that by 12.5 dpc there was a significant increase in mir-140-3p expression in the developing testis, but not the ovary, and that this expression was maintained until at least 13.5 dpc. To identify the spatial and cellular expression patterns of mir-140-3p, we performed LNA-ISH on mouse embryo sections from 11.5 dpc (data not shown) to 13.5 dpc (Fig. 2B). mir-140-3p was not detectable in the developing ovary at any stage investigated (Fig. 2B, top panel) or at 11.5 dpc in the XY genital ridge (data not shown). At 12.5 dpc, mir-140-3p was detected at very low levels in the developing testis, which increased to robust levels at 13.5 dpc (Fig. 2B, bottom panel). The expression of this mirna was clearly restricted to testis 3 Article 143 Downloaded from
26 RAKOCZY ET AL. Downloaded from FIG. 1. Differentially expressed mirnas during mouse gonad development. Normalized abundance (cpm) of mirnas that are sexually dimorphic expressed between testes (blue bars) and ovaries (pink bars) at 11.5 dpc (A), 12.5 dpc (B), and 13.5 dpc (C). with more than 1.5-fold expression difference (A) and 2-fold difference (B, C), respectively. Fold difference is shown above each pair of bars; negative numbers indicate higher expression in testes, while a positive number indicates higher expression in ovaries. cords (Fig. 2B, arrowheads), which comprise germ cells surrounded by supporting Sertoli cells. To further validate the testis-enriched expression, we also performed Northern blot hybridization of small RNAs isolated from 13.5 dpc testes and ovaries using radiolabeled DNA oligonucleotides antisense to the corresponding mirna. These experiments not only confirmed that mir-140-3p is more highly expressed in testes compared to ovaries, but also 4 Article 143
27 ROLE OF mir-140-3p IN TESTIS DEVELOPMENT FIG. 2. Testis-specific expression of mir-140-3p. A) High-throughput sequencing (left panel) and qrt-pcr (right panel) of mouse embryonic testes (XY) and ovaries (XX) at 11.5, 12.5, and 13.5 dpc demonstrate testis-enriched expression of mir-140-3p. High-throughput sequencing expression is shown in cpm. At least three independent experiments were performed for qrt-pcr analysis (mean 6 SEM; *P 0.5, **P 0.01, ***P 0.001; ns, not statistically significant), with individual experiments performed in triplicate on RNA obtained from pooled gonads from three to four littermates and expression levels shown relative to sno202 RNA. B) LNA-ISH on sagittal sections of mouse embryos at 12.5 and 13.5 dpc for mir-140-3p detected expression in testis cords (arrowheads) at 13.5 dpc. O, ovary; T, testis. Bar ¼ 100 lm. C) Northern blot hybridization for mir-140-3p (top panel) in 13.5 dpc mouse testes (XY) and ovaries (XX) demonstrated testis-enriched expression as well as the presence of at least two isomirs (arrowheads). Lysine-tRNA served as loading control (bottom panel). Downloaded from TABLE 2. Sequences and expression levels (cpm) of highly expressed mir-140-3p isomirs detected by high-throughput sequencing. Counts per million (CPM) RNA sequence a T11.5 O11.5 T12.5 O12.5 T13.5 O13.5 mir-140-3p UACCACAGGGUAGAACCACGG ACCACAGGGUAGAACCACGGA ACCACAGGGUAGAACCACGGAC UACCACAGGGUAGAACCACGGA UACCACAGGGUAGAACCACGGAC a Bolded text indicates the seed sequence of the micornas. 5 Article 143
28 RAKOCZY ET AL. FIG. 3. Testis-specific expression of mir-140-5p. High-throughput sequencing (A) and qrt-pcr (B) of mouse embryonic testes (XY) and ovaries (XX) at 11.5, and 13.5 dpc demonstrate testis-enriched expression of mir-140-5p. High-throughput sequencing expression is shown in cpm. At least three independent experiments were performed for qrt-pcr analysis (mean 6 SEM; *P 0.5, **P 0.01, ***P 0.001; ns, not statistically significant) with individual experiments performed in triplicate on RNA obtained from pooled gonads from three to four littermates and expression levels shown relative to sno202 RNA. (C) LNA-ISH on sagittal section of mouse embryos at 12.5 and 13.5 dpc for mir-140-5p detected expression in testis cords (arrowheads) at 13.5 dpc. O, ovary; T, testis. Bar ¼ 100 lm. detected at least two variants, or isomirs, of slightly different lengths (Fig. 2C). The presence of these isomirs was confirmed in the high-throughput sequencing data, which identified a total of four mir-140-3p isomirs (Table 2). When compared to the canonical mir-140-3p sequence annotated in mirbase, we found that the most abundant mir-140-3p isomir was one nt shorter at its 5 0 end and two nt longer at its 3 0 end (Table 2). This and a second mir-140-3p isomir varied in their 5 0 end from the annotated sequence, resulting in a different seed sequence, i.e., the region spanning nt 2 to 8 that is essential for target recognition [40]. It is likely that each mir-140-3p isomir targets different gene subsets [41], which could further fine-tune the testis-specific developmental program. mir-140-5p Is Expressed in the Developing Testis Until recently, mir-140-3p was the annotated passenger strand, or star strand, of the mir-140 duplex. We therefore also analyzed the expression of the opposing strand, mir-140-5p, during gonad development. High-throughput sequencing analysis showed that mir-140-5p was expressed at low levels in ovaries and testes at 11.5 dpc, before its expression increased in gonads of both sexes by 12.5 dpc (Fig. 3A). At 13.5 dpc, mir-140-5p expression further increased in the developing testes but not ovaries (Fig. 3A). We validated the testisenriched expression of mir-140-5p by qrt-pcr, which showed statistically significant higher expression in testes compared to ovaries at all three stages (Fig. 3B). Finally, we performed LNA-ISH of mouse embryonic gonads from 11.5 dpc (data not shown) to 13.5 dpc (Fig. 3C). We detected clear expression of mir-140-5p in testis cords at 13.5 dpc (Fig. 3C, bottom right panel). The Predicted Passenger Strand, mir-140-3p, Is the Predominant Strand in Testes and Chondrocytes Interestingly, the presumed guide strand, mir-140-5p, has been previously reported as the dominant mir-140 strand in mouse and zebrafish cartilage [42, 43], and has been reported to have a role in cartilage development and homeostasis in mouse [30] and palate development in fish [44]. Section LNA- ISH of whole mouse embryos at 13.5 dpc showed that, however, similar to the embryonic testis, both mir-140-5p (Fig. 4A, left panel) and mir-140-3p (Fig. 4A, right panel) were expressed at sites of cartilage formation, such as the ribs (Fig. 4A, arrowheads). To independently confirm that mir-140-3p is the strand predominantly expressed in the testis and to determine which of the two mirna strands, mir-140-5p or mir-140-3p, is predominantly expressed in cartilage, we performed highthroughput sequencing of 13.5 dpc mouse testes and limb buds using the Illumina MiSeq platform. Surprisingly, we found that mir-140-3p is expressed at higher levels than mir-140-5p in both tissues (Fig. 4B), contradicting previous studies reporting mir-140-5p as the prevalent strand expressed at sites of chondrogenesis and further strengthening our results that show 6 Article 143 Downloaded from
29 ROLE OF mir-140-3p IN TESTIS DEVELOPMENT marker OCT4 and absence of the meiosis-associated marker SCP3 (Fig. 5A, bottom panel). While initial analysis suggested an increase in Sertoli cell number as observed by the expression of SOX9 (Fig. 5A, upper panel), this was not confirmed using additional samples. However, a detailed examination of SCC expression revealed a reproducible increase in Leydig cell numbers in the mir-140-null compared to wild-type testes (Fig. 5B). Quantification of this phenotype using SCC whole mount immunofluorescence confirmed a significant 2.6- and 1.6-fold increase in Leydig cell numbers at 13.5 and 15.5 dpc, respectively (Fig. 5C). FIG. 4. mir-140-3p is the predominantly expressed strand in embryonic testes and limb buds. A) LNA-ISH on sagittal sections of 13.5 dpc XX mouse embryos for mir-140-5p (left panel) and mir-140-3p (right panel) showed that both mirna strands are expressed at sites of chondrogenesis, such as the developing ribs (arrowheads). Bar ¼ 1 mm. B) High-throughput sequencing of mouse embryonic limb buds (LB) and testes (XY) at 13.5 dpc demonstrate higher accumulation of mir-140-3p over mir-140-5p in both tissues. High-throughput sequencing expression is shown in cpm. mir-140-3p is the most prevalent strand in the developing testes (Fig. 1). mir-140-5p/mir-140-3p Modulate Leydig Cell Numbers Having detected expression of predominantly mir-140-3p, but also lower levels of mir-140-5p, in the developing testis, we next examined the role of these mirnas in gonad differentiation by analyzing a mouse line with targeted deletion of pre-mir-140, i.e., a deletion of the hairpin that encodes both mir-140-5p and mir-140-3p [30]. We confirmed that mir-140-3p and mir-140-5p are absent in the developing gonads using qrt-pcr on 13.5 dpc testes from heterozygous and knockout embryos (Supplemental Fig. S2). Using analysis of marker gene expression, we found that XX fetuses developed ovaries that appeared normal at 13.5 dpc (data not shown). Similarly, XY fetuses developed testes, as shown by the formation of testis cords, expression of the Sertoli cell marker SOX9 (Fig. 5A, upper panel) and the Leydig cell marker SCC (Fig. 5A, middle panel), and absence of the ovary-specific protein FOXL2 (data not shown). Also, PGCs appeared to differentiate normally, with the continued expression of the pluripotency mir-140 Targets The testicular phenotype observed in the mir-140-null embryos closely resembles the phenotype observed in disrupted Notch signaling [12]. To investigate this further, we queried for targets of mir-140 using mirwalk [33] and identified nine genes associated with Notch signaling that were predicted to be targeted by canonical mir-140-5p or mir-140-3p (Supplemental Table 3). Here we have shown, however, that the most highly expressed mir-140-3p species in the testis is not the annotated mature mirna, but is in fact two isomirs with different seed sequences (Table 2). To assess the target genes of these species, we queried for potential mir-140-3p isomir targets using mirwalk, TargetScan, and RNA22 (see Materials and Methods). Intriguingly, even if using the stringent criteria that a gene must be identified as a potential target by at least two algorithms, this analysis resulted in the identification of a total of 26 putative genes targeted by either or both of the two most abundant mir-140-3p isomir, and the annotated mature mirna sequence (Table 3). Intriguingly, this set of putative targets includes the X-linked MECP2, a methyl- CpG binding protein that functions as both an activator and repressor of transcription [45]. Recent work has shown that duplication of MECP2 is associated with undescended testes and micropenis [46]. DISCUSSION MicroRNAs have been shown to play a role in almost all developmental processes (for review see [47, 48]). However, their function during mouse sex determination and early gonad differentiation has not been studied in detail. Here, we identified mirnas that were differentially expressed in developing mouse gonads at 11.5 dpc, the time when sex is determined, to 13.5 dpc, when testes and ovaries begin to differentiate. We focused our analysis on mir-140 and have shown that, in contrast to the published literature, the assumed passenger strand, mir-140* or mir-140-3p, is the predominantly expressed strand not only in the developing testis but also at sites of chondrogenesis. In addition, we demonstrated that mir-140-5p/mir-140-3p play a role during testis differentiation by regulating the Leydig cell number. Sexually Dimorphic mirnas Using Illumina high-throughput sequencing, we identified 23 mirnas that were differentially expressed between XX and XY embryonic gonads. Of these, 17 were testis enriched and six were ovary enriched. Comparison with other mirna expression studies of testes and ovaries from various stages of development and species revealed that approximately half of the identified mirnas were also detected to be sexually dimorphic in other systems, corroborating our results. For example, seven of the testis-enriched mirnas, mir-130a, mir- 434, mir-202, mir-140-5p, mir-470, mir-743a, and mir Article 143 Downloaded from
30 RAKOCZY ET AL. FIG. 5. mir-140-null mouse embryos at 13.5 dpc have an increased number of Leydig cells in the developing testis. A) Double immunofluorescence for SOX9 (green; Sertoli cells) and E-cadherin (ECAD; red; germ cells, mesonephric tubules, top panel); SCC (green; Leydig cells) and WT1 (red; Sertoli cells; middle panel); and OCT4 (red, pluripotency marker of germ cells) and SCP3 (green; meiotic germ cells; bottom panel) on sagittal sections of gonads from wild-type and mir-140-null fetuses at 13.5 dpc. Wild-type and mir-140-null testes showed conspicuous testis cords with strong expression of SOX9 protein, no meiotic germ cells (demonstrated by the lack of SCP3 expression) and Leydig cells in the interstitium marked by SCC expression. B) Closer examination of at least eight different wild-type and mir-140-null testes at 13.5 dpc using SCC immunofluorescence revealed a consistent increase in Leydig cell numbers in testes lacking pre-mir-140. Bars ¼ 100 lm (A) and 50 lm (B). C) Quantification of SCC-positive cells by whole-mount anti-scc immunofluorescence of three wild-type and three knockout samples at 13.5 dpc and four wild-type and four knockout samples at 15.5 dpc. Data are represented as an average of all samples within a genotype and age, and error bars represent the SEM. Significance was tested using Student t-test for samples of unequal variance (*P 0.5, **P 0.01). Downloaded from were cloned from postnatal testes at Day 8 and/or Day 15 [49], mir-872 was shown to be expressed by Sertoli cells [29], and for mir-184, a functional role through the regulation of Ncor2 in spermatogenesis has been demonstrated [50]. Similarly, mir-202 and mir-140-5p are also testis enriched, and mir-423, mir-28, mir-378, and mir-206 are ovary enriched, in cattle and/or sheep [51, 52]. In addition, prior work investigating embryonic mouse gonads confirmed the transient expression of mir-378 at 12.5 dpc in the developing ovary [22], but this work failed to directly compare mirna expression levels between ovaries and testes, making it difficult to assess sexually dimorphic expression [22]. Since all of these studies used different techniques (e.g., cloning, microarray, qrt- PCR), different stages (ranging from embryonic to adult gonads), and different species (e.g., mouse, cattle, sheep), any mirna that has been identified by two or more of these studies is a good candidate for having a role in gonad biology. Almost half of the testis-enriched mirnas are located on the X chromosome. This phenomenon mirrors a previous study showing that genes expressed in testes before spermatogenesis occurs are enriched on the X chromosome [53]. It supports the hypothesis in Rice [54] that any recessive allele, which arises on the X chromosome and gives males an advantage, is immediately open to positive selection because there is only one X. Interestingly, many mirnas encoded on the X chromosome escape meiotic sex-chromosome inactivation (MSCI) [55], which occurs only in male meiotic germ cells during spermatogenesis and is characterized by transcriptional silencing of genes on both the X and Y chromosome [56]. The seven X-chromosomal mirnas, mir-470, mir-871, mir-741, mir-743a, mir-465a-5p, mir-743b, and mir-878-5p, identified to have testis-enriched expression during embryonic gonad development in this study, all escape MSCI [55], suggesting that they play a role during gonad differentiation as well as spermatogenesis. Regulation of mir-140-5p/mir-140-3p Expression The mirna that showed the highest expression in testes in the work presented here was mir-140-3p. We also found that its opposing strand, mir-140-5p, was testis enriched, albeit at a much lower level of expression. Intriguingly, mir-140-5p was previously identified as highly expressed in chondrocytes [42], which suggests that a transcription factor common to both 8 Article 143
31 ROLE OF mir-140-3p IN TESTIS DEVELOPMENT TABLE 3. Gene symbol Target predictions for mir-140-3p. Target prediction program TargetScan MirWalk RNA22 Hic1 U U U Kif21b U U U Mecp2 U U U Slc17a9 U U AI U U Acad11 U U Acta1 U U U Ankrd49 U U U Ccnj U U Clec16a U U Dot1l U U Galnt2 U U Hdlbp U U Hmgcll1 U U Limk2 U U Lrig1 U U Lrrc58 U U Nr2f2 U U Nr6a1 U U Pcdh19 U U Pcdh8 U U Serf2 U U Smarcc1 U U Tgfbr3 U U Urm1 U U Wnt9a U U tissues might transcriptionally regulate the expression of the primary mir-140 transcript. For sites of cartilage formation, one regulator, SOX9, has been shown in zebrafish and mouse to directly regulate pri-mir-140 expression [57, 58]. SOX9 is not only a master regulator of chondrogenesis but is also necessary and sufficient for testis differentiation [59 62]. Sox9 is expressed in Sertoli cells [5, 6], the supporting cell lineage in the testis. We detected mir-140-5p/mir-140-3p expression in testis cords, which are comprised of a cluster of germ cells surrounded by Sertoli cells, suggesting that in the XY gonad SOX9 also regulates the expression of these mirnas in Sertoli cells. Target Genes of mir-140-3p and mir-140-5p in Chondrocytes and Testis A number of publications have presented evidence of high expression of mir-140-5p in chondrocytes in human, mouse, and zebrafish and have characterized the role of this mirna in chondrocyte development and function [30, 42, 57, 58, 63, 64]. Only one of these studies also reported the expression of mir p, the opposing and presumed passenger strand [65]. Surprisingly, we found mir-140-3p to be the strand that is predominantly expressed not only in the developing testis but also in chondrocytes. It appears that most, if not all, of the studies characterizing mir-140-5p in cartilage development failed to investigate mir-140-3p [30, 42, 57, 58, 63, 64] and, therefore, did not compare levels between the two strands in these cells. The two opposing strands of an mirna have different seed sequences and are, therefore, predicted to regulate a different set of target genes. Several target genes for mir-140-5p in chondrocytes have been identified, such as Hdac4 [42], Pdgfra [63], Igfbp5 [66], Adamts5 [30], Smad3 [67], and Cxcl12 [68]. The fact that mir-140-5p is expressed in these cells means that such regulation may indeed take place. However, mir-140-3p is also expressed, and at higher levels than the mir-140-5p strand. Therefore, it is feasible that a number of target genes that play important roles during chondrogenesis are yet to be discovered and may in fact be revealed by further investigation of mir-140-3p behavior and function. The genes identified and validated to be targeted by mir p include genes that are known to play a role in gonad development and disease, such as Bmp2, a gene that is expressed in the ovary during gonad development [69]; Pdgfra, which has been implicated in the formation of testis-specific vasculature [70]; and Smad3, which plays a role in testicular and ovarian cancer [71]. It is possible that regulation of these genes by mir-140-5p also plays a role during sex differentiation. However, mir-140-5p is weakly expressed in the developing testes, suggesting that mir-140-3p may be equally, if not more, important in gonad development. Using a combination of three different algorithms, we have predicted 26 genes that are likely to be regulated by mir-140-3p. These include the genes encoding MECP2 (as discussed above), LIMkinase 2, the nuclear receptor NR2F2, betaglycan (TGFBR3), and germ cell nuclear factor (GCNF or NR6A1), all of which have been shown to play a role in testis development, Leydig cell differentiation, and function and spermatogenesis [72 75]. However, adult mir-140-5p/mir140-3p-null mice are fertile [30], suggesting that mir-140 must only weakly target these transcripts or that other regulatory RNA species compensate for its loss the latter of which is likely, given that any given transcript is generally targeted by a suite of mirnas. This would also be consistent with the fact that very mild effects been observed for a number of mirna knockouts [76]. Role of mir-140-5p/mir-140-3p During Testis Development The deletion of mir-140-5p/mir-140-3p resulted in an increase in Leydig cell numbers at 13.5 dpc. Two pathways are involved in the differentiation of Leydig cells, the hedgehog [11] and the Notch signaling pathway [12]. While hedgehog signaling is important for the initial specification of Leydig cells, Notch signaling restricts further differentiation of Leydig progenitor cells. The observed phenotype of the mir-140-5p/ mir-140-3p knockout mouse is very similar to that from blocking Notch signaling, and we have identified a number of putative target genes that are associated with this signaling pathway that are predicted to be targeted by either mir-140-5p or mir-140-3p. Expression analysis of these genes in the mir- 140-null mice could provide evidence to support the hypothesis that they are indeed target genes. Certainly, the fact that mir p and mir-140-5p are expressed in Sertoli cells, while a phenotype is observed in Leydig cell differentiation, indicates the involvement of a signaling pathway, i.e., these mirnas are likely to regulate the expression of factors produced by Sertoli cells that influence the Leydig cells differentiation. ACKNOWLEDGMENT We thank Dr. Cassy Spiller (Institute for Molecular Bioscience, The University of Queensland) for technical help with gonad dissection and RNA preparation. We thank Dr. Paul Waters (Australian National University) for comments on the manuscript. Confocal microscopy was performed at the Australian Cancer Research Foundation Dynamic Imaging Centre for Cancer Biology. REFERENCES 1. Bullejos M, Koopman P. Spatially dynamic expression of Sry in mouse genital ridges. Dev Dyn 2001; 221: Article 143 Downloaded from
32 RAKOCZY ET AL. 2. Hacker A, Capel B, Goodfellow P, Lovell-Badge R. Expression of Sry, the mouse sex determining gene. Development 1995; 121: Jeske YW, Bowles J, Greenfield A, Koopman P. Expression of a linear Sry transcript in the mouse genital ridge. Nat Genet 1995; 10: Sekido R, Lovell-Badge R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature 2008; 453: Sekido R, Bar I, Narvaez V, Penny G, Lovell-Badge R. SOX9 is upregulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev Biol 2004; 274: Wilhelm D, Martinson F, Bradford S, Wilson MJ, Combes AN, Beverdam A, Bowles J, Mizusaki H, Koopman P. Sertoli cell differentiation is induced both cell-autonomously and through prostaglandin signaling during mammalian sex determination. Dev Biol 2005; 287: Combes AN, Lesieur E, Harley VR, Sinclair AH, Little MH, Wilhelm D, Koopman P. Three-dimensional visualization of testis cord morphogenesis, a novel tubulogenic mechanism in development. Dev Dyn 2009; 238: Combes AN, Wilhelm D, Davidson T, Dejana E, Harley V, Sinclair A, Koopman P. Endothelial cell migration directs testis cord formation. Dev Biol 2009; 326: Coveney D, Cool J, Oliver T, Capel B. Four-dimensional analysis of vascularization during primary development of an organ, the gonad. Proc Natl Acad Sci U S A 2008; 105: Skinner MK, Tung PS, Fritz IB. Cooperativity between Sertoli cells and testicular peritubular cells in the production and deposition of extracellular matrix components. J Cell Biol 1985; 100: Barsoum IB, Bingham NC, Parker KL, Jorgensen JS, Yao HH. Activation of the Hedgehog pathway in the mouse fetal ovary leads to ectopic appearance of fetal Leydig cells and female pseudohermaphroditism. Dev Biol 2009; 329: Tang H, Brennan J, Karl J, Hamada Y, Raetzman L, Capel B. Notch signaling maintains Leydig progenitor cells in the mouse testis. Development 2008; 135: Bullejos M, Koopman P. Germ cells enter meiosis in a rostro-caudal wave during development of the mouse ovary. Mol Reprod Dev 2004; 68: Menke DB, Koubova J, Page DC. Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. Dev Biol 2003; 262: Liu CF, Liu C, Yao HH. Building pathways for ovary organogenesis in the mouse embryo. Curr Top Dev Biol 2010; 90: Mattick JS. The functional genomics of noncoding RNA. Science 2005; 309: Chen CZ, Li L, Lodish HF, Bartel DP. MicroRNAs modulate hematopoietic lineage differentiation. Science 2004; 303: Kloosterman WP, Plasterk RH. The diverse functions of micrornas in animal development and disease. Dev Cell 2006; 11: Stefani G, Slack FJ. Small non-coding RNAs in animal development. Nat Rev Mol Cell Biol 2008; 9: Lee Y, Jeon K, Lee JT, Kim S, Kim VN. MicroRNA maturation: stepwise processing and subcellular localization. EMBO J 2002; 21: Khvorova A, Reynolds A, Jayasena SD. Functional sirnas and mirnas exhibit strand bias. Cell 2003; 115: Aguilar AL, Piskol R, Beitzinger M, Zhu JY, Kruspe D, Aszodi A, Moser M, Englert C, Meister G. The small RNA expression profile of the developing murine urinary and reproductive systems. FEBS Lett 2010; 584: Ahn HW, Morin RD, Zhao H, Harris RA, Coarfa C, Chen ZJ, Milosavljevic A, Marra MA, Rajkovic A. MicroRNA transcriptome in the newborn mouse ovaries determined by massive parallel sequencing. Mol Hum Reprod 2010; 16: Takada S, Berezikov E, Choi YL, Yamashita Y, Mano H. Potential role of mir-29b in modulation of Dnmt3a and Dnmt3b expression in primordial germ cells of female mouse embryos. RNA 2009; 15: Yan N, Lu Y, Sun H, Tao D, Zhang S, Liu W, Ma Y. A microarray for microrna profiling in mouse testis tissues. Reprod 2007; 134: Gonzalez G, Behringer RR. Dicer is required for female reproductive tract development and fertility in the mouse. Mol Reprod Dev 2009; 76: Kim GJ, Georg I, Scherthan H, Merkenschlager M, Guillou F, Scherer G, Barrionuevo F. Dicer is required for Sertoli cell function and survival. Int J Dev Biol 2010; 54: Nagaraja AK, Andreu-Vieyra C, Franco HL, Ma L, Chen R, Han DY, Zhu H, Agno JE, Gunaratne PH, DeMayo FJ, Matzuk MM. Deletion of Dicer in somatic cells of the female reproductive tract causes sterility. Mol Endocrinol 2008; 22: Papaioannou MD, Pitetti JL, Ro S, Park C, Aubry F, Schaad O, Vejnar CE, Kuhne F, Descombes P, Zdobnov EM, McManus MT, Guillou F, et al. Sertoli cell Dicer is essential for spermatogenesis in mice. Dev Biol 2009; 326: Miyaki S, Sato T, Inoue A, Otsuki S, Ito Y, Yokoyama S, Kato Y, Takemoto F, Nakasa T, Yamashita S, Takada S, Lotz MK, et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev 2010; 24: Langmead B, Trapnell C, Pop M, Salzberg SL. Ultrafast and memoryefficient alignment of short DNA sequences to the human genome. Genome Biol 2009; 10:R Griffiths-Jones S, Saini HK, van Dongen S, Enright AJ. mirbase: tools for microrna genomics.nucleic Acids Res 2008; 36:D154 D Dweep H, Sticht C, Pandey P, Gretz N. mirwalk database: prediction of possible mirna binding sites by walking the genes of three genomes.j Biomed Inform 2011; 44: Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microrna targets. Cell 2005; 120: Miranda KC, Huynh T, Tay Y, Ang YS, Tam WL, Thomson AM, Lim B, Rigoutsos I. A pattern-based method for the identification of MicroRNA binding sites and their corresponding heteroduplexes. Cell 2006; 126: Pena JT, Sohn-Lee C, Rouhanifard SH, Ludwig J, Hafner M, Mihailovic A, Lim C, Holoch D, Berninger P, Zavolan M, Tuschl T. mirna in situ hybridization in formaldehyde and EDC-fixed tissues.nat Methods 2009; 6: Svingen T, Francois M, Wilhelm D, Koopman P. Three-dimensional imaging of Prox1-EGFP transgenic mouse gonads reveals divergent modes of lymphangiogenesis in the testis and ovary. PLoS One 2012; 7: e Pall GS, Hamilton AJ. Improved northern blot method for enhanced detection of small RNA. Nat Protoc 2008; 3: Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE, Horvitz HR, Ruvkun G. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature 2000; 403: Brennecke J, Stark A, Russell RB, Cohen SM. Principles of micrornatarget recognition. PLoS Biol 2005; 3:e Cloonan N, Wani S, Xu Q, Gu J, Lea K, Heater S, Barbacioru C, Steptoe AL, Martin HC, Nourbakhsh E, Krishnan K, Gardiner B, et al. MicroRNAs and their isomirs function cooperatively to target common biological pathways. Genome Biol 2011; 12:R Tuddenham L, Wheeler G, Ntounia-Fousara S, Waters J, Hajihosseini MK, Clark I, Dalmay T. The cartilage specific microrna-140 targets histone deacetylase 4 in mouse cells. FEBS Lett 2006; 580: Wienholds E, Kloosterman WP, Miska E, Alvarez-Saavedra E, Berezikov E, de Bruijn E, Horvitz HR, Kauppinen S, Plasterk RH. MicroRNA expression in zebrafish embryonic development. Science 2005; 309: He X, Eberhart JK, Postlethwait JH. MicroRNAs and micromanaging the skeleton in disease, development and evolution. J Cell Mol Med 2009; 13: Chahrour M, Jung SY, Shaw C, Zhou X, Wong ST, Qin J, Zoghbi HY. MeCP2, a key contributor to neurological disease, activates and represses transcription. Science 2008; 320: Sanmann JN, Bishay DL, Starr LJ, Bell CA, Pickering DL, Stevens JM, Kahler SG, Olney AH, Schaefer GB, Sanger WG. Characterization of six novel patients with MECP2 duplications due to unbalanced rearrangements of the X chromosome. Am J Med Genet A 2012; 158A: Pauli A, Rinn JL, Schier AF. Non-coding RNAs as regulators of embryogenesis. Nat Rev Genet 2011; 12: Suh N, Blelloch R. Small RNAs in early mammalian development: from gametes to gastrulation. Development 2011; 138: Watanabe T, Takeda A, Tsukiyama T, Mise K, Okuno T, Sasaki H, Minami N, Imai H. Identification and characterization of two novel classes of small RNAs in the mouse germline: retrotransposon-derived sirnas in oocytes and germline small RNAs in testes. Genes Dev 2006; 20: Wu J, Bao J, Wang L, Hu Y, Xu C. MicroRNA-184 downregulates nuclear receptor corepressor 2 in mouse spermatogenesis. BMC Dev Biol 2011; 11: Huang J, Ju Z, Li Q, Hou Q, Wang C, Li J, Li R, Wang L, Sun T, Hang S, Gao Y, Hou M, et al. Solexa sequencing of novel and differentially expressed micrornas in testicular and ovarian tissues in Holstein cattle. Int J Biol Sci 2011; 7: Torley KJ, da Silveira JC, Smith P, Anthony RV, Veeramachaneni DN, 10 Article 143 Downloaded from
33 ROLE OF mir-140-3p IN TESTIS DEVELOPMENT Winger QA, Bouma GJ. Expression of mirnas in ovine fetal gonads: potential role in gonadal differentiation. Reprod Biol Endocrinol 2011; 9: Khil PP, Smirnova NA, Romanienko PJ, Camerini-Otero RD. The mouse X chromosome is enriched for sex-biased genes not subject to selection by meiotic sex chromosome inactivation. Nat Genet 2004; 36: Rice WR. Sex chromosomes and the evolution of sexual dimorphism. Evolution 1984; 38: Song R, Ro S, Michaels JD, Park C, McCarrey JR, Yan W. Many X- linked micrornas escape meiotic sex chromosome inactivation. Nat Genet 2009; 41: McKee BD, Handel MA. Sex chromosomes, recombination, and chromatin conformation. Chromosoma 1993; 102: Nakamura Y, He X, Kato H, Wakitani S, Kobayashi T, Watanabe S, Iida A, Tahara H, Warman ML, Watanapokasin R, Postlethwait JH. Sox9 is upstream of microrna-140 in cartilage. Appl Biochem Biotechnol 2012; 166: Yang J, Qin S, Yi C, Ma G, Zhu H, Zhou W, Xiong Y, Zhu X, Wang Y, He L, Guo X. MiR-140 is co-expressed with Wwp2-C transcript and activated by Sox9 to target Sp1 in maintaining the chondrocyte proliferation. FEBS Lett 2011; 585: Barrionuevo F, Bagheri-Fam S, Klattig J, Kist R, Taketo MM, Englert C, Scherer G. Homozygous inactivation of Sox9 causes complete XY sex reversal in mice. Biol Reprod 2006; 74: Bishop CE, Whitworth DJ, Qin Y, Agoulnik AI, Agoulnik IU, Harrison WR, Behringer RR, Overbeek PA. A transgenic insertion upstream of Sox9 is associated with dominant XX sex reversal in the mouse. Nat Genet 2000; 26: Chaboissier MC, Kobayashi A, Vidal VI, Lutzkendorf S, van de Kant HJ, Wegner M, de Rooij DG, Behringer RR, Schedl A. Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development 2004; 131: Foster JW, Dominguez-Steglich MA, Guioli S, Kwok C, Weller PA, Weissenbach J, Mansour S, Young ID, Goodfellow PN, Brook JD, Schafer AJ. Campomelic dysplasia and autosomal sex reversal caused by mutations in an SRY-related gene. Nature 1994; 372: Eberhart JK, He X, Swartz ME, Yan YL, Song H, Boling TC, Kunerth AK, Walker MB, Kimmel CB, Postlethwait JH. MicroRNA Mirn140 modulates Pdgf signaling during palatogenesis. Nat Genet 2008; 40: Miyaki S, Nakasa T, Otsuki S, Grogan SP, Higashiyama R, Inoue A, Kato Y, Sato T, Lotz MK, Asahara H. MicroRNA-140 is expressed in differentiated human articular chondrocytes and modulates interleukin-1 responses. Arthritis Rheum 2009; 60: Swingler TE, Wheeler G, Carmont V, Elliott HR, Barter MJ, Abu-Elmagd M, Donell ST, Boot-Handford RP, Hajihosseini MK, Munsterberg A, Dalmay T, Young DA, et al. The expression and function of micrornas in chondrogenesis and osteoarthritis. Arthritis Rheum 2012; 64: Tardif G, Hum D, Pelletier JP, Duval N, Martel-Pelletier J. Regulation of the IGFBP-5 and MMP-13 genes by the micrornas mir-140 and mir- 27a in human osteoarthritic chondrocytes. BMC Musculoskelet Disord 2009; 10: Pais H, Nicolas FE, Soond SM, Swingler TE, Clark IM, Chantry A, Moulton V, Dalmay T. Analyzing mrna expression identifies Smad3 as a microrna-140 target regulated only at protein level. RNA 2010; 16: Nicolas FE, Pais H, Schwach F, Lindow M, Kauppinen S, Moulton V, Dalmay T. mrna expression profiling reveals conserved and nonconserved mir-140 targets. RNA Biol 2011; 8: Kashimada K, Pelosi E, Chen H, Schlessinger D, Wilhelm D, Koopman P. FOXL2 and BMP2 act cooperatively to regulate follistatin gene expression during ovarian development. Endocrinol 2011; 152: Brennan J, Tilmann C, Capel B. Pdgfr-alpha mediates testis cord organization and fetal Leydig cell development in the XY gonad. Genes Dev 2003; 17: Li Q, Graff JM, O Connor AE, Loveland KL, Matzuk MM. SMAD3 regulates gonadal tumorigenesis. Mol Endocrinol 2007; 21: Takahashi H, Koshimizu U, Miyazaki J, Nakamura T. Impaired spermatogenic ability of testicular germ cells in mice deficient in the LIM-kinase 2 gene. Dev Biol 2002; 241: Martin LJ, Tremblay JJ. Nuclear receptors in Leydig cell gene expression and function. Biol Reprod 2010; 83: Qin J, Tsai MJ, Tsai SY. Essential roles of COUP-TFII in Leydig cell differentiation and male fertility. PLoS One 2008; 3:e Sarraj MA, Escalona RM, Umbers A, Chua HK, Small C, Griswold M, Loveland K, Findlay JK, Stenvers KL. Fetal testis dysgenesis and compromised Leydig cell function in Tgfbr3 (beta glycan) knockout mice. Biol Reprod 2010; 82: Park CY, Choi YS, McManus MT. Analysis of microrna knockouts in mice. Hum Mol Genet 2010; 19:R169 R175. Downloaded from 11 Article 143
34
35
36
37
38
39
40
41
42
43
44
45
46
47
Bi-potent Gonads. Sex Determination
 יצירת הגונדות Primordial Germ Cells (PGCs) Somatic cells Genital ridge Bi-potent Gonads Sex Determination Testis and Sperm Ovary and Oocyte Migration of Primordial Germ Cells in the Chick Embryo The
יצירת הגונדות Primordial Germ Cells (PGCs) Somatic cells Genital ridge Bi-potent Gonads Sex Determination Testis and Sperm Ovary and Oocyte Migration of Primordial Germ Cells in the Chick Embryo The
Identification of candidate gonadal sex differentiation genes in the chicken embryo using RNA-seq
 Ayers et al. BMC Genomics (2015)16:704 DOI 10.1186/s12864-015-1886-5 RESEARCH ARTICLE Identification of candidate gonadal sex differentiation genes in the chicken embryo using RNA-seq Open Access Katie
Ayers et al. BMC Genomics (2015)16:704 DOI 10.1186/s12864-015-1886-5 RESEARCH ARTICLE Identification of candidate gonadal sex differentiation genes in the chicken embryo using RNA-seq Open Access Katie
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2638 Figure S1 Morphological characteristics of fetal testes and ovaries from 6.5-20 developmental weeks. Representative images of Hematoxylin and Eosin staining of testes and ovaries over
DOI: 10.1038/ncb2638 Figure S1 Morphological characteristics of fetal testes and ovaries from 6.5-20 developmental weeks. Representative images of Hematoxylin and Eosin staining of testes and ovaries over
Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a,
 Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a, Transverse sections of E17.5 ovary and mesonephros from Gli1-LacZ reporter embryos (n=3) after LacZ staining (blue). The
Supplementary Figure 1: Expression of Gli1-lacZ in E17.5 ovary and mesonephros. a, Transverse sections of E17.5 ovary and mesonephros from Gli1-LacZ reporter embryos (n=3) after LacZ staining (blue). The
Nature Immunology: doi: /ni.3412
 Supplementary Figure 1 Gata1 expression in heamatopoietic stem and progenitor populations. (a) Unsupervised clustering according to 100 top variable genes across single pre-gm cells. The two main cell
Supplementary Figure 1 Gata1 expression in heamatopoietic stem and progenitor populations. (a) Unsupervised clustering according to 100 top variable genes across single pre-gm cells. The two main cell
Title: Human breast cancer associated fibroblasts exhibit subtype specific gene expression profiles
 Author's response to reviews Title: Human breast cancer associated fibroblasts exhibit subtype specific gene expression profiles Authors: Julia Tchou (julia.tchou@uphs.upenn.edu) Andrew V Kossenkov (akossenkov@wistar.org)
Author's response to reviews Title: Human breast cancer associated fibroblasts exhibit subtype specific gene expression profiles Authors: Julia Tchou (julia.tchou@uphs.upenn.edu) Andrew V Kossenkov (akossenkov@wistar.org)
Topics for this lecture: Sex determination Sexual differentiation Sex differences in behavior and CNS development. 1) organizational effects of
 Topics for this lecture: Sex determination Sexual differentiation Sex differences in behavior and CNS development. 1) organizational effects of gonadal steroids on CNS development 2) our model system:
Topics for this lecture: Sex determination Sexual differentiation Sex differences in behavior and CNS development. 1) organizational effects of gonadal steroids on CNS development 2) our model system:
Wnt4 is required for proper male as well as female sexual development
 Developmental Biology 276 (2004) 431 440 www.elsevier.com/locate/ydbio Wnt4 is required for proper male as well as female sexual development Katherine Jeays-Ward 1, Mathieu Dandonneau 1, Amanda Swain*
Developmental Biology 276 (2004) 431 440 www.elsevier.com/locate/ydbio Wnt4 is required for proper male as well as female sexual development Katherine Jeays-Ward 1, Mathieu Dandonneau 1, Amanda Swain*
11. SEXUAL DIFFERENTIATION. Germinal cells, gonocytes. Indifferent stage INDIFFERENT STAGE
 11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants
 SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
DAX1, testes development role 7, 8 DFFRY, spermatogenesis role 49 DMRT genes, male sex differentiation role 15
 Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Nature Genetics: doi: /ng Supplementary Figure 1. Assessment of sample purity and quality.
 Supplementary Figure 1 Assessment of sample purity and quality. (a) Hematoxylin and eosin staining of formaldehyde-fixed, paraffin-embedded sections from a human testis biopsy collected concurrently with
Supplementary Figure 1 Assessment of sample purity and quality. (a) Hematoxylin and eosin staining of formaldehyde-fixed, paraffin-embedded sections from a human testis biopsy collected concurrently with
Strategic delivery: Setting standards Increasing and. Details: Output: Demonstrating efficiency. informing choice.
 Strategic delivery: Setting standards Increasing and informing choice Demonstrating efficiency economy and value Details: Meeting Scientific and Clinical Advances Advisory Committee Agenda item 6 Paper
Strategic delivery: Setting standards Increasing and informing choice Demonstrating efficiency economy and value Details: Meeting Scientific and Clinical Advances Advisory Committee Agenda item 6 Paper
Nature Neuroscience: doi: /nn Supplementary Figure 1. Behavioral training.
 Supplementary Figure 1 Behavioral training. a, Mazes used for behavioral training. Asterisks indicate reward location. Only some example mazes are shown (for example, right choice and not left choice maze
Supplementary Figure 1 Behavioral training. a, Mazes used for behavioral training. Asterisks indicate reward location. Only some example mazes are shown (for example, right choice and not left choice maze
W.S. O University of Hong Kong
 W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
SUPPLEMENTARY INFORMATION
 Supplementary text Collectively, we were able to detect ~14,000 expressed genes with RPKM (reads per kilobase per million) > 1 or ~16,000 with RPKM > 0.1 in at least one cell type from oocyte to the morula
Supplementary text Collectively, we were able to detect ~14,000 expressed genes with RPKM (reads per kilobase per million) > 1 or ~16,000 with RPKM > 0.1 in at least one cell type from oocyte to the morula
Sex Determination and Gonadal Sex Differentiation in Fish
 Sex Determination and Gonadal Sex Differentiation in Fish Yoshitaka Nagahama Okazaki National Research Institutes, Japan This first slide shows the processes of gonadal sex differentiation and gametogenesis
Sex Determination and Gonadal Sex Differentiation in Fish Yoshitaka Nagahama Okazaki National Research Institutes, Japan This first slide shows the processes of gonadal sex differentiation and gametogenesis
Sex Differentiation. Course Outline. Topic #! Topic lecture! Silverthorn! Membranes (pre-requisite material)!!
 Sex Differentiation The goal of these lectures is to discuss how a control system is formed. For this, we will use basic physiology associated with the control of reproduction (from sexual differentiation
Sex Differentiation The goal of these lectures is to discuss how a control system is formed. For this, we will use basic physiology associated with the control of reproduction (from sexual differentiation
Conserved properties of dentate gyrus neurogenesis across postnatal development revealed by single-cell RNA sequencing
 SUPPLEMENTARY INFORMATION Resource https://doi.org/10.1038/s41593-017-0056-2 In the format provided by the authors and unedited. Conserved properties of dentate gyrus neurogenesis across postnatal development
SUPPLEMENTARY INFORMATION Resource https://doi.org/10.1038/s41593-017-0056-2 In the format provided by the authors and unedited. Conserved properties of dentate gyrus neurogenesis across postnatal development
SUPPLEMENTAL INFORMATION
 SUPPLEMENTAL INFORMATION GO term analysis of differentially methylated SUMIs. GO term analysis of the 458 SUMIs with the largest differential methylation between human and chimp shows that they are more
SUPPLEMENTAL INFORMATION GO term analysis of differentially methylated SUMIs. GO term analysis of the 458 SUMIs with the largest differential methylation between human and chimp shows that they are more
SUPPLEMENTARY APPENDIX
 SUPPLEMENTARY APPENDIX 1) Supplemental Figure 1. Histopathologic Characteristics of the Tumors in the Discovery Cohort 2) Supplemental Figure 2. Incorporation of Normal Epidermal Melanocytic Signature
SUPPLEMENTARY APPENDIX 1) Supplemental Figure 1. Histopathologic Characteristics of the Tumors in the Discovery Cohort 2) Supplemental Figure 2. Incorporation of Normal Epidermal Melanocytic Signature
DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2018
 DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2018 Director:, DVM, Ph.D. 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve
DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2018 Director:, DVM, Ph.D. 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Sexual Reproduction. For most diploid eukaryotes, sexual reproduction is the only mechanism resulting in new members of a species.
 Sex Determination Sexual Reproduction For most diploid eukaryotes, sexual reproduction is the only mechanism resulting in new members of a species. Meiosis in the sexual organs of parents produces haploid
Sex Determination Sexual Reproduction For most diploid eukaryotes, sexual reproduction is the only mechanism resulting in new members of a species. Meiosis in the sexual organs of parents produces haploid
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Inferring Biological Meaning from Cap Analysis Gene Expression Data
 Inferring Biological Meaning from Cap Analysis Gene Expression Data HRYSOULA PAPADAKIS 1. Introduction This project is inspired by the recent development of the Cap analysis gene expression (CAGE) method,
Inferring Biological Meaning from Cap Analysis Gene Expression Data HRYSOULA PAPADAKIS 1. Introduction This project is inspired by the recent development of the Cap analysis gene expression (CAGE) method,
Animal Reproduction. Hypothalamic-pituitary-gonadal axis. # lectures for cumulative test # 01 book 01
 Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 05 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 05 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/7/283/283ra54/dc1 Supplementary Materials for Clonal status of actionable driver events and the timing of mutational processes in cancer evolution
www.sciencetranslationalmedicine.org/cgi/content/full/7/283/283ra54/dc1 Supplementary Materials for Clonal status of actionable driver events and the timing of mutational processes in cancer evolution
Stem cells: units of development and regeneration. Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research.
 Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Detection of aneuploidy in a single cell using the Ion ReproSeq PGS View Kit
 APPLICATION NOTE Ion PGM System Detection of aneuploidy in a single cell using the Ion ReproSeq PGS View Kit Key findings The Ion PGM System, in concert with the Ion ReproSeq PGS View Kit and Ion Reporter
APPLICATION NOTE Ion PGM System Detection of aneuploidy in a single cell using the Ion ReproSeq PGS View Kit Key findings The Ion PGM System, in concert with the Ion ReproSeq PGS View Kit and Ion Reporter
MRC-Holland MLPA. Description version 08; 07 May 2015
 mix P185-C1 Intersex Lot C1-0611: As compared to the previous version B2 (lot B2-0311), s for CYP21A2 have been removed and s for the CXorf21 gene as well as additional s for NR0B1, NR5A1 and the Y chromosome
mix P185-C1 Intersex Lot C1-0611: As compared to the previous version B2 (lot B2-0311), s for CYP21A2 have been removed and s for the CXorf21 gene as well as additional s for NR0B1, NR5A1 and the Y chromosome
Haematopoietic stem cells
 Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Normal and Abnormal Development of the Genital Tract. Dr.Raghad Abdul-Halim
 Normal and Abnormal Development of the Genital Tract Dr.Raghad Abdul-Halim objectives: Revision of embryology. Clinical presentation, investigations and clinical significance of most common developmental
Normal and Abnormal Development of the Genital Tract Dr.Raghad Abdul-Halim objectives: Revision of embryology. Clinical presentation, investigations and clinical significance of most common developmental
Results and Problems in Cell Differentiation
 Results and Problems in Cell Differentiation Volume 58 Series editors Jacek Z. Kubiak, Rennes CX, France Malgorzata Kloc, Houston, TX, USA More information about this series at http://www.springer.com/series/400
Results and Problems in Cell Differentiation Volume 58 Series editors Jacek Z. Kubiak, Rennes CX, France Malgorzata Kloc, Houston, TX, USA More information about this series at http://www.springer.com/series/400
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/487/eaag2476/dc1 Supplementary Materials for Gene expression profiles of brain endothelial cells during embryonic development at bulk and single-cell levels
www.sciencesignaling.org/cgi/content/full/10/487/eaag2476/dc1 Supplementary Materials for Gene expression profiles of brain endothelial cells during embryonic development at bulk and single-cell levels
CHAPTER 6 SUMMARIZING DISCUSSION
 CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
Cluster Dendrogram. dist(cor(na.omit(tss.exprs.chip[, c(1:10, 24, 27, 30, 48:50, dist(cor(na.omit(tss.exprs.chip[, c(1:99, 103, 104, 109, 110,
 A Transcriptome (RNA-seq) Transcriptome (RNA-seq) 3. 2.5 2..5..5...5..5 2. 2.5 3. 2.5 2..5..5...5..5 2. 2.5 Cluster Dendrogram RS_ES3.2 RS_ES3. RS_SHS5.2 RS_SHS5. PS_SHS5.2 PS_SHS5. RS_LJ3 PS_LJ3..4 _SHS5.2
A Transcriptome (RNA-seq) Transcriptome (RNA-seq) 3. 2.5 2..5..5...5..5 2. 2.5 3. 2.5 2..5..5...5..5 2. 2.5 Cluster Dendrogram RS_ES3.2 RS_ES3. RS_SHS5.2 RS_SHS5. PS_SHS5.2 PS_SHS5. RS_LJ3 PS_LJ3..4 _SHS5.2
SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer.
 Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
SALSA MLPA probemix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four probes have been adjusted.
 mix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four s have been adjusted. The sex-determining region on chromosome Y (SRY) is the most important
mix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four s have been adjusted. The sex-determining region on chromosome Y (SRY) is the most important
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Illustration of the working of network-based SVM to confidently predict a new (and now confirmed) ASD gene. Gene CTNND2 s brain network neighborhood that enabled its prediction by
Supplementary Figure 1 Illustration of the working of network-based SVM to confidently predict a new (and now confirmed) ASD gene. Gene CTNND2 s brain network neighborhood that enabled its prediction by
DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2011
 DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2011 Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke
DRB666 Applied Developmental and Reproductive Biology Spring Semester, 2011 Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke
DRB666 Applied Developmental and Reproductive Biology (Spring 2013)
 DRB666 Applied Developmental and Reproductive Biology (Spring 2013) Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke Marikawa
DRB666 Applied Developmental and Reproductive Biology (Spring 2013) Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke Marikawa
Nature Genetics: doi: /ng Supplementary Figure 1. SEER data for male and female cancer incidence from
 Supplementary Figure 1 SEER data for male and female cancer incidence from 1975 2013. (a,b) Incidence rates of oral cavity and pharynx cancer (a) and leukemia (b) are plotted, grouped by males (blue),
Supplementary Figure 1 SEER data for male and female cancer incidence from 1975 2013. (a,b) Incidence rates of oral cavity and pharynx cancer (a) and leukemia (b) are plotted, grouped by males (blue),
Supplementary Materials for
 www.sciencemag.org/content/355/6332/eaai8478/suppl/dc1 Supplementary Materials for Decoupling genetics, lineages, and microenvironment in IDH-mutant gliomas by single-cell RNA-seq Andrew S. Venteicher,
www.sciencemag.org/content/355/6332/eaai8478/suppl/dc1 Supplementary Materials for Decoupling genetics, lineages, and microenvironment in IDH-mutant gliomas by single-cell RNA-seq Andrew S. Venteicher,
Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell and patient-derived tumor organoids
 Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell and patient-derived tumor organoids Ling Huang 1, Audrey Holtzinger 1,2,11, Ishaan Jagan 1,11, Michael BeGora 1, Ines
Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell and patient-derived tumor organoids Ling Huang 1, Audrey Holtzinger 1,2,11, Ishaan Jagan 1,11, Michael BeGora 1, Ines
Loss of Mitogen-Activated Protein Kinase Kinase Kinase 4 (MAP3K4) Reveals a Requirement for MAPK Signalling in Mouse Sex Determination
 Loss of Mitogen-Activated Protein Kinase Kinase Kinase 4 (MAP3K4) Reveals a Requirement for MAPK Signalling in Mouse Sex Determination Debora Bogani 1, Pam Siggers 1, Rachel Brixey 1, Nick Warr 1, Sarah
Loss of Mitogen-Activated Protein Kinase Kinase Kinase 4 (MAP3K4) Reveals a Requirement for MAPK Signalling in Mouse Sex Determination Debora Bogani 1, Pam Siggers 1, Rachel Brixey 1, Nick Warr 1, Sarah
Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells
 Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells Christopher Sturgeon February 14, 2017 Pluripotent Stem Cells self-renewal hpsc Mesoderm blood cardiovascular muscle Endoderm lung liver
Modeling Developmental Hematopoiesis Using Pluripotent Stem Cells Christopher Sturgeon February 14, 2017 Pluripotent Stem Cells self-renewal hpsc Mesoderm blood cardiovascular muscle Endoderm lung liver
Convergence of cmyc and β-catenin on Tcf7l1 enables endoderm specification
 Manuscript EMBO-2015-92116 Convergence of cmyc and β-catenin on Tcf7l1 enables endoderm specification Gillian Morrison, Roberta Scognamiglio, Andreas Trumpp, and Austin Smith Corresponding author: Gillian
Manuscript EMBO-2015-92116 Convergence of cmyc and β-catenin on Tcf7l1 enables endoderm specification Gillian Morrison, Roberta Scognamiglio, Andreas Trumpp, and Austin Smith Corresponding author: Gillian
Biology 4361 Developmental Biology. October 11, Multiple choice (one point each)
 Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Supplemental Figures Supplemental Figure 1:
 Supplemental Figures Supplemental Figure 1: Representative FACS data showing Concurrent Brain cell type Acquisition using either Percoll PLUS (top row) or myelin removal beads (bottom two rows). Debris
Supplemental Figures Supplemental Figure 1: Representative FACS data showing Concurrent Brain cell type Acquisition using either Percoll PLUS (top row) or myelin removal beads (bottom two rows). Debris
RASA: Robust Alternative Splicing Analysis for Human Transcriptome Arrays
 Supplementary Materials RASA: Robust Alternative Splicing Analysis for Human Transcriptome Arrays Junhee Seok 1*, Weihong Xu 2, Ronald W. Davis 2, Wenzhong Xiao 2,3* 1 School of Electrical Engineering,
Supplementary Materials RASA: Robust Alternative Splicing Analysis for Human Transcriptome Arrays Junhee Seok 1*, Weihong Xu 2, Ronald W. Davis 2, Wenzhong Xiao 2,3* 1 School of Electrical Engineering,
Growth pattern of the sex ducts in foetal mouse hermaphrodites
 /. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
/. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
Discovery of Novel Human Gene Regulatory Modules from Gene Co-expression and
 Discovery of Novel Human Gene Regulatory Modules from Gene Co-expression and Promoter Motif Analysis Shisong Ma 1,2*, Michael Snyder 3, and Savithramma P Dinesh-Kumar 2* 1 School of Life Sciences, University
Discovery of Novel Human Gene Regulatory Modules from Gene Co-expression and Promoter Motif Analysis Shisong Ma 1,2*, Michael Snyder 3, and Savithramma P Dinesh-Kumar 2* 1 School of Life Sciences, University
IN SUMMARY HST 071 NORMAL & ABNORMAL SEXUAL DIFFERENTIATION Fetal Sex Differentiation Postnatal Diagnosis and Management of Intersex Abnormalities
 Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
Chapter 16: Steroid Hormones (Lecture 17)
 Chapter 16: Steroid Hormones (Lecture 17) A) 21 or fewer carbon atoms B) Precursor: 27 carbon cholesterol C) major classes of steroid hormones 1) progestagens a) progesterone- prepares lining of uterus
Chapter 16: Steroid Hormones (Lecture 17) A) 21 or fewer carbon atoms B) Precursor: 27 carbon cholesterol C) major classes of steroid hormones 1) progestagens a) progesterone- prepares lining of uterus
Research Strategy: 1. Background and Significance
 Research Strategy: 1. Background and Significance 1.1. Heterogeneity is a common feature of cancer. A better understanding of this heterogeneity may present therapeutic opportunities: Intratumor heterogeneity
Research Strategy: 1. Background and Significance 1.1. Heterogeneity is a common feature of cancer. A better understanding of this heterogeneity may present therapeutic opportunities: Intratumor heterogeneity
Nature Immunology: doi: /ni Supplementary Figure 1
 Supplementary Figure 1 A β-strand positions consistently places the residues at CDR3β P6 and P7 within human and mouse TCR-peptide-MHC interfaces. (a) E8 TCR containing V β 13*06 carrying with an 11mer
Supplementary Figure 1 A β-strand positions consistently places the residues at CDR3β P6 and P7 within human and mouse TCR-peptide-MHC interfaces. (a) E8 TCR containing V β 13*06 carrying with an 11mer
PCxN: The pathway co-activity map: a resource for the unification of functional biology
 PCxN: The pathway co-activity map: a resource for the unification of functional biology Sheffield Institute for Translational Neurosciences Center for Integrative Genome Translation GENOME INFORMATICS
PCxN: The pathway co-activity map: a resource for the unification of functional biology Sheffield Institute for Translational Neurosciences Center for Integrative Genome Translation GENOME INFORMATICS
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. Heatmap of GO terms for differentially expressed genes. The terms were hierarchically clustered using the GO term enrichment beta. Darker red, higher positive
Supplementary Figures Supplementary Figure 1. Heatmap of GO terms for differentially expressed genes. The terms were hierarchically clustered using the GO term enrichment beta. Darker red, higher positive
Defects in ErbB-Dependent Establishment of Adult Melanocyte Stem Cells Reveal Independent Origins for Embryonic and Regeneration Melanocytes
 Defects in ErbB-Dependent Establishment of Adult Melanocyte Stem Cells Reveal Independent Origins for Embryonic and Regeneration Melanocytes Keith A. Hultman 1, Erine H. Budi 2, Daniel C. Teasley 1, Andrew
Defects in ErbB-Dependent Establishment of Adult Melanocyte Stem Cells Reveal Independent Origins for Embryonic and Regeneration Melanocytes Keith A. Hultman 1, Erine H. Budi 2, Daniel C. Teasley 1, Andrew
Supplemental Information. Aryl Hydrocarbon Receptor Controls. Monocyte Differentiation. into Dendritic Cells versus Macrophages
 Immunity, Volume 47 Supplemental Information Aryl Hydrocarbon Receptor Controls Monocyte Differentiation into Dendritic Cells versus Macrophages Christel Goudot, Alice Coillard, Alexandra-Chloé Villani,
Immunity, Volume 47 Supplemental Information Aryl Hydrocarbon Receptor Controls Monocyte Differentiation into Dendritic Cells versus Macrophages Christel Goudot, Alice Coillard, Alexandra-Chloé Villani,
Abstract. Optimization strategy of Copy Number Variant calling using Multiplicom solutions APPLICATION NOTE. Introduction
 Optimization strategy of Copy Number Variant calling using Multiplicom solutions Michael Vyverman, PhD; Laura Standaert, PhD and Wouter Bossuyt, PhD Abstract Copy number variations (CNVs) represent a significant
Optimization strategy of Copy Number Variant calling using Multiplicom solutions Michael Vyverman, PhD; Laura Standaert, PhD and Wouter Bossuyt, PhD Abstract Copy number variations (CNVs) represent a significant
Supplementary Figure 1. Chimeric analysis of inner ears. (A-H) Chimeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears.
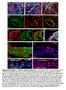 Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
Supplementary Figure 1. himeric analysis of inner ears. (A-H) himeric inner ears with fluorescent ES cells and (I,J) Rainbow inner ears. (A,B) omposite images showing three colors in different vestibular
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Figure S1. Clinical significance of ZNF322A overexpression in Caucasian lung cancer patients. (A) Representative immunohistochemistry images of ZNF322A protein expression in tissue
SUPPLEMENTARY FIGURES Figure S1. Clinical significance of ZNF322A overexpression in Caucasian lung cancer patients. (A) Representative immunohistochemistry images of ZNF322A protein expression in tissue
under its influence, male development occurs; in its absence, female development is established.
 Sex differentiation is a complex process that involves many genes, including some that are autosomal. The key to sexual dimorphism is the Y chromosome, which contains the testis determining gene called
Sex differentiation is a complex process that involves many genes, including some that are autosomal. The key to sexual dimorphism is the Y chromosome, which contains the testis determining gene called
Sexual Development. 6 Stages of Development
 6 Sexual Development 6 Stages of Development Development passes through distinct stages, the first of which is fertilization, when one sperm enters one ovum. To enter an ovum, a sperm must undergo the
6 Sexual Development 6 Stages of Development Development passes through distinct stages, the first of which is fertilization, when one sperm enters one ovum. To enter an ovum, a sperm must undergo the
Gene co-expression networks in the mouse, monkey, and human brain July 16, Jeremy Miller Scientist I
 Gene co-expression networks in the mouse, monkey, and human brain July 16, 2013 Jeremy Miller Scientist I jeremym@alleninstitute.org Outline 1. Brief introduction to previous WGCNA studies in brain 2.
Gene co-expression networks in the mouse, monkey, and human brain July 16, 2013 Jeremy Miller Scientist I jeremym@alleninstitute.org Outline 1. Brief introduction to previous WGCNA studies in brain 2.
Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of four noncompliance methods
 Merrill and McClure Trials (2015) 16:523 DOI 1186/s13063-015-1044-z TRIALS RESEARCH Open Access Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of
Merrill and McClure Trials (2015) 16:523 DOI 1186/s13063-015-1044-z TRIALS RESEARCH Open Access Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Urinary system development. Male ( ) and Female ( ) Reproductive Systems Development
 Urinary system development Male ( ) and Female ( ) Reproductive Systems Development Urogenital system develops from mesodermal uro-genital ridge (intermediate mesoderm) development of male and female genital
Urinary system development Male ( ) and Female ( ) Reproductive Systems Development Urogenital system develops from mesodermal uro-genital ridge (intermediate mesoderm) development of male and female genital
Hierarchical Bayesian Modeling of Individual Differences in Texture Discrimination
 Hierarchical Bayesian Modeling of Individual Differences in Texture Discrimination Timothy N. Rubin (trubin@uci.edu) Michael D. Lee (mdlee@uci.edu) Charles F. Chubb (cchubb@uci.edu) Department of Cognitive
Hierarchical Bayesian Modeling of Individual Differences in Texture Discrimination Timothy N. Rubin (trubin@uci.edu) Michael D. Lee (mdlee@uci.edu) Charles F. Chubb (cchubb@uci.edu) Department of Cognitive
Animal Science 434 Reproductive Physiology"
 Animal Science 434 Reproductive Physiology" Embryogenesis of the Pituitary and Sexual Development: Part A Development of the Pituitary Gland" Infundibulum" Brain" Rathke s Pouch" Stomodeum" Germ Cell Migration"
Animal Science 434 Reproductive Physiology" Embryogenesis of the Pituitary and Sexual Development: Part A Development of the Pituitary Gland" Infundibulum" Brain" Rathke s Pouch" Stomodeum" Germ Cell Migration"
Reliability of Ordination Analyses
 Reliability of Ordination Analyses Objectives: Discuss Reliability Define Consistency and Accuracy Discuss Validation Methods Opening Thoughts Inference Space: What is it? Inference space can be defined
Reliability of Ordination Analyses Objectives: Discuss Reliability Define Consistency and Accuracy Discuss Validation Methods Opening Thoughts Inference Space: What is it? Inference space can be defined
Module 3: Pathway and Drug Development
 Module 3: Pathway and Drug Development Table of Contents 1.1 Getting Started... 6 1.2 Identifying a Dasatinib sensitive cancer signature... 7 1.2.1 Identifying and validating a Dasatinib Signature... 7
Module 3: Pathway and Drug Development Table of Contents 1.1 Getting Started... 6 1.2 Identifying a Dasatinib sensitive cancer signature... 7 1.2.1 Identifying and validating a Dasatinib Signature... 7
Breast cancer. Risk factors you cannot change include: Treatment Plan Selection. Inferring Transcriptional Module from Breast Cancer Profile Data
 Breast cancer Inferring Transcriptional Module from Breast Cancer Profile Data Breast Cancer and Targeted Therapy Microarray Profile Data Inferring Transcriptional Module Methods CSC 177 Data Warehousing
Breast cancer Inferring Transcriptional Module from Breast Cancer Profile Data Breast Cancer and Targeted Therapy Microarray Profile Data Inferring Transcriptional Module Methods CSC 177 Data Warehousing
Sex determination and SRY: down to a wink and a nudge?
 Review Feature Review Sex determination and SRY: down to a wink and a nudge? Ryohei Sekido and Robin Lovell-Badge Division of Developmental Genetics, Medical Research Council National Institute for Medical
Review Feature Review Sex determination and SRY: down to a wink and a nudge? Ryohei Sekido and Robin Lovell-Badge Division of Developmental Genetics, Medical Research Council National Institute for Medical
Fibroblasts cell lines misclassified as cancer cell lines
 Fibroblasts cell lines misclassified as cancer cell lines Antoine de Weck 1, Hans Bitter 2, Audrey Kauffmann 1 1. Novartis Institutes for Biomedical Research, Basel CH-4002, Switzerland. 2. Novartis Institutes
Fibroblasts cell lines misclassified as cancer cell lines Antoine de Weck 1, Hans Bitter 2, Audrey Kauffmann 1 1. Novartis Institutes for Biomedical Research, Basel CH-4002, Switzerland. 2. Novartis Institutes
Protein SD Units (P-value) Cluster order
 SUPPLEMENTAL TABLE AND FIGURES Table S1. Signature Phosphoproteome of CD22 E12 Transgenic Mouse BPL Cells. T-test vs. Other Protein SD Units (P-value) Cluster order ATPase (Ab-16) 1.41 0.000880 1 mtor
SUPPLEMENTAL TABLE AND FIGURES Table S1. Signature Phosphoproteome of CD22 E12 Transgenic Mouse BPL Cells. T-test vs. Other Protein SD Units (P-value) Cluster order ATPase (Ab-16) 1.41 0.000880 1 mtor
Genome-Wide DNA Methylation Analysis Predicts an Epigenetic Switch for GATA Factor Expression in Endometriosis
 Genome-Wide DNA Methylation Analysis Predicts an Epigenetic Switch for GATA Factor Expression in Endometriosis Matthew T. Dyson 1, Damian Roqueiro 2, Diana Monsivais 1, C. Mutlu Ercan 1, Mary Ellen Pavone
Genome-Wide DNA Methylation Analysis Predicts an Epigenetic Switch for GATA Factor Expression in Endometriosis Matthew T. Dyson 1, Damian Roqueiro 2, Diana Monsivais 1, C. Mutlu Ercan 1, Mary Ellen Pavone
Using Bayesian Networks to Analyze Expression Data. Xu Siwei, s Muhammad Ali Faisal, s Tejal Joshi, s
 Using Bayesian Networks to Analyze Expression Data Xu Siwei, s0789023 Muhammad Ali Faisal, s0677834 Tejal Joshi, s0677858 Outline Introduction Bayesian Networks Equivalence Classes Applying to Expression
Using Bayesian Networks to Analyze Expression Data Xu Siwei, s0789023 Muhammad Ali Faisal, s0677834 Tejal Joshi, s0677858 Outline Introduction Bayesian Networks Equivalence Classes Applying to Expression
HOMOSEXYALITY AS A CONSEQUENCE OF EPIGENETICALLY CANALIZED SEXUAL DEVELOPMENT. W.R.Rice, U.Freiberg, S.Gavrilets. December 2012
 HOMOSEXYALITY AS A CONSEQUENCE OF EPIGENETICALLY CANALIZED SEXUAL DEVELOPMENT W.R.Rice, U.Freiberg, S.Gavrilets December 2012 Used terms: Epi-marks means changes in chromatin structure that influence the
HOMOSEXYALITY AS A CONSEQUENCE OF EPIGENETICALLY CANALIZED SEXUAL DEVELOPMENT W.R.Rice, U.Freiberg, S.Gavrilets December 2012 Used terms: Epi-marks means changes in chromatin structure that influence the
Histone Modifications Are Associated with Transcript Isoform Diversity in Normal and Cancer Cells
 Histone Modifications Are Associated with Transcript Isoform Diversity in Normal and Cancer Cells Ondrej Podlaha 1, Subhajyoti De 2,3,4, Mithat Gonen 5, Franziska Michor 1 * 1 Department of Biostatistics
Histone Modifications Are Associated with Transcript Isoform Diversity in Normal and Cancer Cells Ondrej Podlaha 1, Subhajyoti De 2,3,4, Mithat Gonen 5, Franziska Michor 1 * 1 Department of Biostatistics
GENDER James Bier
 GENDER 2005-2008 James Bier Objectives 1. State the method of determining gender in several genetic systems. 2. List the three regions of the Y chromosome. 3. Describe the events that promote sexual development
GENDER 2005-2008 James Bier Objectives 1. State the method of determining gender in several genetic systems. 2. List the three regions of the Y chromosome. 3. Describe the events that promote sexual development
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3443 In the format provided by the authors and unedited. Supplementary Figure 1 TC and SC behaviour during ISV sprouting. (a) Predicted outcome of TC division and competitive Dll4-Notch-mediated
DOI: 10.1038/ncb3443 In the format provided by the authors and unedited. Supplementary Figure 1 TC and SC behaviour during ISV sprouting. (a) Predicted outcome of TC division and competitive Dll4-Notch-mediated
he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003)
 MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
Expanded View Figures
 Solip Park & Ben Lehner Epistasis is cancer type specific Molecular Systems Biology Expanded View Figures A B G C D E F H Figure EV1. Epistatic interactions detected in a pan-cancer analysis and saturation
Solip Park & Ben Lehner Epistasis is cancer type specific Molecular Systems Biology Expanded View Figures A B G C D E F H Figure EV1. Epistatic interactions detected in a pan-cancer analysis and saturation
Animal Science 434 Reproductive Physiology
 Animal Science 434 Reproductive Physiology Development of the Pituitary Gland Lec 5: Embryogenesis of the Pituitary and Sexual Development Stomodeum Brain Infundibulum Rathke s Pouch Germ Cell Migration
Animal Science 434 Reproductive Physiology Development of the Pituitary Gland Lec 5: Embryogenesis of the Pituitary and Sexual Development Stomodeum Brain Infundibulum Rathke s Pouch Germ Cell Migration
Nature Genetics: doi: /ng Supplementary Figure 1. Country distribution of GME samples and designation of geographical subregions.
 Supplementary Figure 1 Country distribution of GME samples and designation of geographical subregions. GME samples collected across 20 countries and territories from the GME. Pie size corresponds to the
Supplementary Figure 1 Country distribution of GME samples and designation of geographical subregions. GME samples collected across 20 countries and territories from the GME. Pie size corresponds to the
SUPPLEMENTARY INFORMATION In format provided by Javier DeFelipe et al. (MARCH 2013)
 Supplementary Online Information S2 Analysis of raw data Forty-two out of the 48 experts finished the experiment, and only data from these 42 experts are considered in the remainder of the analysis. We
Supplementary Online Information S2 Analysis of raw data Forty-two out of the 48 experts finished the experiment, and only data from these 42 experts are considered in the remainder of the analysis. We
Scientific Principles for Studying Sex Differences in Health and Disease
 Scientific Principles for Studying Sex Differences in Health and Disease Art Arnold Department of Integrative Biology and Physiology Lab of Neuroendocrinology Brain Research Institute UCLA FCG Objectives
Scientific Principles for Studying Sex Differences in Health and Disease Art Arnold Department of Integrative Biology and Physiology Lab of Neuroendocrinology Brain Research Institute UCLA FCG Objectives
Supplementary Figure S1. Gene expression analysis of epidermal marker genes and TP63.
 Supplementary Figure Legends Supplementary Figure S1. Gene expression analysis of epidermal marker genes and TP63. A. Screenshot of the UCSC genome browser from normalized RNAPII and RNA-seq ChIP-seq data
Supplementary Figure Legends Supplementary Figure S1. Gene expression analysis of epidermal marker genes and TP63. A. Screenshot of the UCSC genome browser from normalized RNAPII and RNA-seq ChIP-seq data
Comparison of open chromatin regions between dentate granule cells and other tissues and neural cell types.
 Supplementary Figure 1 Comparison of open chromatin regions between dentate granule cells and other tissues and neural cell types. (a) Pearson correlation heatmap among open chromatin profiles of different
Supplementary Figure 1 Comparison of open chromatin regions between dentate granule cells and other tissues and neural cell types. (a) Pearson correlation heatmap among open chromatin profiles of different
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Supplemental Information. Derivation of Human Trophoblast Stem Cells
 Cell Stem Cell, Volume 22 Supplemental Information Derivation of Human Trophoblast Stem Cells Hiroaki Okae, Hidehiro Toh, Tetsuya Sato, Hitoshi Hiura, Sota Takahashi, Kenjiro Shirane, Yuka Kabayama, Mikita
Cell Stem Cell, Volume 22 Supplemental Information Derivation of Human Trophoblast Stem Cells Hiroaki Okae, Hidehiro Toh, Tetsuya Sato, Hitoshi Hiura, Sota Takahashi, Kenjiro Shirane, Yuka Kabayama, Mikita
Development of the Genital System
 Development of the Genital System Professor Alfred Cuschieri Department of Anatomy University of Malta The mesonephros develops primitive nephrotomes draining into a mesonephric duct nephrotome mesonephric
Development of the Genital System Professor Alfred Cuschieri Department of Anatomy University of Malta The mesonephros develops primitive nephrotomes draining into a mesonephric duct nephrotome mesonephric
