Deletion of the Receptor for Advanced Glycation Endproducts Reduces Glomerulosclerosis and Preserves Renal Function in the Diabetic OVE26 Mouse
|
|
|
- Laureen Hill
- 6 years ago
- Views:
Transcription
1 Diabetes Publish Ahead of Print, published online July 13, 2010 Deletion of the Receptor for Advanced Glycation Endproducts Reduces Glomerulosclerosis and Preserves Renal Function in the Diabetic OVE26 Mouse Nina Reiniger 1, Kai Lau 6, Daren McCalla 1, Bonnie Eby 6, Bin Cheng 2, Yan Lu 1, Wu Qu 1, Nosirudeen Quadri 1, Radha Ananthakrishnan 1, Maryana Furmansky 1, Rosa Rosario 1, Fei Song 1, Vivek Rai 1, Alan Weinberg 2, Richard Friedman 4,5, Ravichandran Ramasamy 1, Vivette D'Agati 3 and Ann Marie Schmidt 1. Departments of 1 Surgery, 2 Biostatistics, 3 Pathology and 4 Biomedical Informatics and 5 Biomedical Informatics Shared Resource, Herbert Irving Comprehensive Cancer Center, Columbia University, New York, NY, United States. 6 Section of Nephrology, Department of Medicine, University of Oklahoma Health Sciences Center, Oklahoma City, OK, United States. Running title: RAGE deletion preserves function in DN in OVE26 Corresponding author contact information: Nina Reiniger nr2207@columbia.edu Additional information for this article can be found in an online appendix at Submitted 2 December 2009 and accepted 4 May This is an uncopyedited electronic version of an article accepted for publication in Diabetes. The American Diabetes Association, publisher of Diabetes, is not responsible for any errors or omissions in this version of the manuscript or any version derived from it by third parties. The definitive publisher-authenticated version will be available in a future issue of Diabetes in print and online at Copyright American Diabetes Association, Inc., 2010
2 Objective Previous studies showed that genetic deletion or pharmacological blockade of the Receptor for Advanced Glycation Endproducts (RAGE) prevents the early structural changes in the glomerulus associated with diabetic nephropathy (DN). To overcome limitations of mouse models that lack the progressive glomerulosclerosis observed in humans, we studied the contribution of RAGE to DN in the OVE26 type 1 mouse, a model of progressive glomerulosclerosis and decline of renal function. Research Design and Methods We bred OVE26 mice with homozygous RAGE knock out (RKO) mice and examined structural changes associated with DN and used inulin clearance studies and albumin:creatinine measurements to assess renal function. Transcriptional changes in the Tgf-β1 and Plasminogen activator inhibitor 1 gene products were measured to investigate mechanisms underlying accumulation of mesangial matrix in OVE26 mice. Results - Deletion of RAGE in OVE26 mice reduced nephromegaly, mesangial sclerosis, cast formation, glomerular basement membrane thickening, podocyte effacement, and albuminuria. The significant 29% reduction in glomerular filtration rate observed in OVE26 mice was completely prevented by deletion of RAGE. Increased transcription of the genes for Plasminogen activator inhibitor 1, Tgf-β1, Tgf-β induced, α1- (IV) collagen observed in OVE26 renal cortex was significantly reduced in OVE26 RKO kidney cortex. ROCK1 activity was significantly lower in OVE26 RKO compared to OVE26 kidney cortex. Conclusions - These data provide compelling evidence for critical roles for RAGE in the pathogenesis of DN and suggest that strategies targeting RAGE in long-term diabetes may prevent loss of renal function. T he Receptor for Advanced Glycation Endproducts (RAGE), a member of the immunoglobulin superfamily, is upregulated in tissues subjected to the longterm impact of diabetes.(1; 2) The ligands of RAGE, including advanced glycation endproducts (AGEs), S100/calgranulins and High mobility group box-1 (HMGB1) display elevated expression in diabetic tissues.(1; 2) In diabetic nephropathy (DN), RAGE is upregulated in cells such as glomerular podocytes and endothelial cells in both humans and mice.(2; 3) Previous studies provided evidence for roles for RAGE and its ligands in mouse models of early DN. Overexpression of RAGE in vascular endothelial cells of hypo-insulinemic mice led to increased mesangial matrix expansion and glomerulosclerosis.(4) Pharmacological blockade of RAGE, using the soluble extracellular ligand binding domain of RAGE (srage), in type 2 insulinresistant db/db diabetic mice, protected against glomerulosclerosis and other classical lesions of early DN.(3) In addition, the kidneys of streptozotocin (STZ)-injected RAGE knockout (RKO) mice were protected from early mesangial matrix expansion and thickening of the glomerular basement membrane (GBM) seen in wild-type diabetic mice.(3) Myint and colleagues demonstrated protection from DN in RKO mice crossed with transgenic mice expressing inos under the control of the insulin promoter.(5) Others 2
3 have shown that blocking antibodies to RAGE suppressed DN in mouse models of type 1 and type 2 diabetes.(6; 7) Until recently, very few mouse models of DN progress beyond the early disease stages of microalbuminuria and mild mesangial expansion. Transgenic over-expression of calmodulin specifically in pancreatic β cells, the OVE26 model of type 1 diabetes(8), resulted in nephromegaly, albuminuria, glomerulosclerosis, tubulointerstitial fibrosis, occasional occurrence of arteriolar hyalinosis, and the suggestion of decreased glomerular filtration rate (GFR).(9-11). Hence, the OVE26 mouse is considered one of the most human-relevant models of DN studied to date. We tested the hypothesis that RAGE contributes to advanced glomerulosclerosis and loss of renal function in long-term diabetes by crossing OVE26 mice with homozygous RAGE null mice to investigate the role of RAGE in this robust model of DN. RESEARCH DESIGN AND METHODS Additional information is also available in online supplemental information section at Mice: OVE26 mice (strain FVB(Cg)-Tg(Ins2- CALM1)26Ove Tg(Cryaa-TAg)1Ove/PneJ), and FVB controls (strain FVB/NJ) were obtained from The Jackson Laboratory (Bar Harbor, ME) and bred in house. FVB RKO mice, backcrossed >10 generations into FVB, were bred in house and crossed with OVE26 resulting in OVE26 RKO mice. Changes associated with DN were studied in male mice at 7 months based on our identification of this time point as one when moderate to severe histologic renal changes were observed in most male OVE26 mice. Mice were sacrificed at 1 month to test RAGE expression. Polyclonal chicken anti-mouse RAGE IgY antibody production: Soluble (s)rage was produced as previously described.(1) Anti- RAGE Chicken IgY was generated and purified from serum and specificity for RAGE was confirmed. Fluorescence Microscopy: Frozen kidney sections were obtained from 1 month old (for RAGE staining) and 7 months old mice (for Glyoxalase1 (Glo1) staining) and stained with polyclonal chicken anti-mouse RAGE IgY or Rat monoclonal [6F10] to Glo1 (Abcam, Cambridge, MA). Fluorescence microscopy was performed using a Lasersharp 2000 BioRad scanning confocal microscope (Bio- Rad, Hercules, CA). Blood glucose and glycosylated hemoglobin measurements: Blood glucose measurements were made from non-fasted mice at 4-week intervals and prior to sacrifice at 7 months using a Freestyle Blood Glucose Meter (Alameda, CA). Levels above 499 mg/dl ( HI reading), the maximum reading for the glucometer, were denoted as 500 mg/dl for calculation purposes. Blood Hgb A1c was measured using Cholestech GDX A1c test cartridges for human Hgb because the last 7 amino acids in the N terminus are identical between human and mice (Cholestech, Harward, California). Methylglyoxal measurement: Levels of methylglyoxal were determined in frozen kidney cortex tissue obtained from six female 7 month-old mice, as previously described.(12) Glyoxalase 1 Western: 30μg of kidnedy cortex lysate was run on a 12% NuPAGE gel using MES running buffer (Invitrogen, Carlsbad, CA) under reducing conditions. After transfer to nitrocellulose membrane (Invitrogen, Carlsbad, CA), staining was performed using Rat monoclonal [6F10] to Glo1 (1:1000) (Abcam, Cambridge, MA) followed by chicken anti-rat (1:1000) (Santa Cruz Biotechnology, Santa Cruz, Ca) Blots were then stripped with Restore Western Blot Stripping Buffer (Pierce, Rockford, IL) and probed with anti-β actin antibody (1:2500) (Becton Dickinson, Franklin Lakes, NJ). 3
4 Bands were quantified using Alpha imager software (Alpha Innotech, San Leandro, CA). Morphometry: Kidneys were harvested and bisected longitudinally. One half kidney was fixed in 10% formalin overnight then dehydrated, embedded in paraffin, sectioned at 3 μm, mounted on 3-aminopropyltriethoxy silane-coated glass slides (Sigma, St Louis, MO), stained with periodic acid Schiff (PAS) (Sigma) and analyzed by light microscopy. Pathology scoring: Pathology was scored in a semi-quantitative manner by a renal pathologist (V D A), blinded to the genotypes of the animals as described in the supplemental information. Glomerular basement membrane (GBM) and podocyte effacement measurements: Glutaraldehyde fixed kidney cortex tissue was analyzed by electron microscopy. The thickness of the GBM of multiple capillaries was measured in 6 8 glomeruli per mouse (N=5 per group). A mean of 55 measurements was taken per mouse (from podocyte to endothelial cell membranes) at random sites where the GBM was displayed in best crosssection. The same glomeruli were scored for degree of podocyte effacement, defined as the percentage of total glomerular capillary surface area over which the podocyte foot processes were effaced. Urine collection and measurement of albuminuria: Urine was collected over 24 hours from each mouse in individual mouse metabolic cages (Nalgene, Rochester, NY) and frozen at -80 C for subsequent analyses. Urinary albumin and creatinine levels were measured using the murine specific Albuwell ELISA kit and the Creatinine Companion kit (Exocell, Inc, Philadelphia, PA) according to manufacturer s instructions. Measurement of glomerular filtration rate (GFR) and effective renal plasma flow (RPF): Standard renal inulin clearance studies were performed during continuous hemodynamic monitoring as published,(13; 14) in male mice at 7 months of age. Details are provided in supplemental methods. Glomerular isolation: Glomeruli were isolated by perfusing anesthetized mice with Dynabeads (Invitrogen, Carlsbad, CA), digesting the kidney tissue and performing a series of sieving steps. Glomeruli were picked under the microscope using a pipette and placed in RNAlater (Ambion, Austin, TX). RNA isolation, reverse transcription and real time: RNA was isolated from ~20-30mg of kidney cortex tissue (for all real time with the exception of Glyoxalase 1) or ~1000 isolated glomeruli (for Glyoxalase 1 real time and for microarray sample preparation) per mouse using the RNaqueous kit (Ambion) and reverse transcription was performed with the Superscript III kit (Invitrogen) according to manufacturers instructions. Real time primers and probes for murine 18s, Glyoxalase1, Tgfb1, Tgfbi, Serpine1, and α1-(iv) collagen were purchased from PE Applied Biosystems (Foster City, CA). Realtime PCR was performed with an ABI Prism 7900HT Sequence Detection System with TaqMan PCR Master Mix (PE Applied Biosystems). ROCK1 activity assays: Activation of ROCK1 was evaluated on lysates of kidneys isolated from OVE26 or OVE26 RKO mice, as previously described.(15) Details provided in supplemental information. Microarray experimental methods: RNA was isolated from ~1000 isolated glomeruli per mouse using the RNaqueous kit (Ambion, Austin, TX). Total RNA concentration was assessed using a ND-1000 NanoDrop Spectrophotometer (Thermo Scientific, Wilmington, DE) and quality was assessed on a 2100 Bioanalyzer system (Agilent Technologies, Santa Clara, CA) using the Agilent RNA 6000 Nano kit. The RNA was then amplified, biotin labeled and fragmented using the Gene Chip 3 IVT Express Kit (Affymetrix, Santa Clara, CA). 10 μg of fragmented RNA was hybridized to Mouse 4
5 Genome GeneChips (Affymetrix). All samples were prepared with the two-cycle protocol recommended by the manufacturer. One array per mouse was used. 3 arrays (mice) for each genotype were used. Microarray data analysis: Microarray data was analyzed as described in the supplemental information section. All microarray data has been deposited in Gene Expression Omnibus. The GEO accession number is GSE Statistical analysis: For the statistical analysis of all the data sets comparing 4 groups, a one-way ANOVA model followed by paired comparisons was performed. Bonferroni correction due to multiple comparisons was used to control the overall type one error rate. Log transformation was used for the urine albumin:creatinine analysis (Fig 5). For Fig 3d&e, the comparison was done only between OVE26 and OVE26 RKO groups because the values for other groups were 0. The severity of mesangial sclerosis data was compared using a Fisher s exact test for contingency table (Fig 3e). All other data comparing 2 groups were analyzed using an independent two-sample t-test. Analyses were performed in SAS version 9.1 (SAS Institute Inc., Cary, NC), with the exception of the ANOVA and t-tests, which were performed using Stat View (Adept Scientific, Acton, MA). RESULTS RAGE is expressed in glomeruli of OVE26 mice. Previous studies reported that RAGE is expressed in human and murine glomeruli(2), but expression had not been studied in the OVE26 model. RAGE was detected in the glomeruli of kidney cortex from male OVE26 mice at 1 month of age and a baseline level of RAGE in the FVB glomerulus is provided for comparison (Fig 1a). This time point was chosen in order to determine whether RAGE expression was present in the OVE26 mouse prior to observable structural changes in the kidney, thus correlating changes in RAGE with the timing of early stages of DN development. Precursor to RAGE ligands is reduced by RKO in OVE26 mice despite persistent hyperglycemia. OVE26 mice displayed significant increases in blood glucose levels compared to the non-diabetic FVB mice (498 ± 6 vs. 138 ± 36 mg/dl) (P<0.001) (Fig 1b). RKO did not alter the degree of hyperglycemia in OVE26 mice (494 ± 11 mg/dl) (P=0.7438). Blood glucose levels measured at 4-week intervals beginning at 8 weeks of age showed the same trends among groups as the measurements at 7 months (data not shown). Hemoglobin (Hgb) A1c was significantly increased in OVE26 mice versus FVB controls (8.87 ± 0.33 vs ± 0.25 %) (P<0.0001) (Fig 1c), confirming sustained hyperglycemia (Fig 1b). Hgb A1c was normal in FVB mice, with or without RAGE (5.35 ± 0.25% vs ± 0.57%) (Fig 1c). Importantly, Hgb A1c was comparably elevated among OVE26 mice with or without RAGE (8.87 ± 0.33 vs ± 0.41 %) (Fig 1c), confirming that RKO did not impact glycemic control. We studied the effect of RAGE deletion in the context of hyperglycemia on the levels of methylglyoxal (MG), a key precursor of AGEs. Levels of MG were 3.25 fold higher in OVE26 kidney cortex compared to FVB (P<0.05), but were unaffected by RKO in FVB mice. However, levels of MG were 5.93 fold lower in OVE26 RKO cortex compared to OVE26 cortex (P<0.05), despite equivalent degrees of hyperglycemia in both groups (Fig 1d). Glyoxalase 1 levels are increased by RKO in OVE26 mice. To further probe the differences in MG levels among OVE26 and OVE26 RKO cortex, we assessed the levels of mrna and protein for glyoxalase 1, an important regulator of MG levels in vivo. Our data revealed that both mrna transcript 5
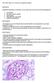
6 levels (P<0.05) (Fig 1e) and protein levels for glyoxalase 1 were higher in OVE26 RKO compared to OVE26 (P<0.05) (Fig 1f) Immunofluorescence staining localized glyoxalase 1 particularly to the glomeruli of OVE26 and OVE26 RKO mice (Fig 1g). OVE26 RKO Mice are Partially Protected from Glomerulosclerosis. To establish the optimal time point for testing the role of RAGE in DN in the OVE26 model in mice raised in our facility, we monitored the pathological changes in the kidney at 5, 6 and 7 months of age. At 5 months of age, mesangial sclerosis was mild in most OVE26 mice. At 6 months of age, more OVE26 mice revealed moderate pathology. At 7 months of age, we consistently observed a range of moderate to severe mesangial sclerosis including the formation of mesangial nodules causing capillary luminal narrowing (Fig 2a), focal tubular atrophy, interstitial fibrosis and chronic inflammation (Fig 2b), and progression to segmental (Fig 2c) and global glomerulosclerosis (Fig 2d). Glomeruli of OVE26 mice appeared enlarged compared to FVB controls (Fig 3a,b), owing to diffuse and global mesangial sclerosis (Fig 3b, right panel). The cortical tubules contained focal proteinaceous casts (Fig 3b, left panel). Thus, to investigate the role of RAGE in advanced DN, we studied OVE26 mice crossed with FVB RKO mice at age 7 months. OVE26 RKO mice were partially protected from these pathological alterations including significantly fewer casts (Fig 3c,d), and reduced mesangial matrix expansion (Fig 3c,e) (P<0.05). Tubular atrophy that was present in OVE26 mice (Fig 2b) was completely prevented in OVE26 RKO mice (data not shown). The histology of FVB RKO mice did not differ significantly from that of FVB mice (data not shown). At the ultrastructural level, the mesangial areas in OVE26 glomeruli were expanded by increased matrix and hyalinosis (Fig 2e), in some cases obliterating capillary lumens (Fig 2h). In OVE26 mice, the podocyte foot processes appeared effaced (Fig 2f-h & 4b) and there was significant GBM thickening (Fig 2f and 4e). In contrast, OVE26 RKO demonstrated less podocyte effacement compared to the OVE26 (Fig 4c,d) and no significant increase in GBM thickness compared to FVB mice (Fig 4c,e) (P<0.05). The glomeruli of FVB RKO mice were indistinguishable ultrastructurally from those of FVB mice (Fig 4d,e, image not shown). Albuminuria in OVE26 mice is markedly ameliorated by RKO. Urinary albumin and creatinine were measured at 7 months of age. OVE26 mice displayed significantly increased albumin:creatinine ratios compared to FVB mice (822 ± 159 mg/mg vs. 46 ± 14 mg/mg) (P<0.001) (Fig 5). OVE26 RKO mice demonstrated significantly lower albumin:creatinine ratios (359 ± 152 mg/mg), reduced by ~56%, compared to OVE26 mice (Fig 5) (P=0.0085). FVB RKO albumin:creatinine ratios were not significantly different from FVB ratios (P=0.9793). Renal insufficiency is present in OVE26 mice but prevented by RKO. (a) Metabolic, hematocrit (Hct), hemodynamic, and urine flow data in mice subjected to inulin clearance studies - Hct was comparable among the 4 groups, from the beginning to the end of the experiment (Table 1), indicating negligible blood loss. In response to body weight-based mild saline infusion (to prevent volume depletion, especially in diabetic mice), all groups experienced a small but similar decline in Hct (Table 1). Throughout the 3-hour experiment, Hct, averaged from 5 serial readings per mouse, was also comparable (Table 1), suggesting similar degrees of volume expansion and stability of blood volume. Mean systolic blood pressure (SBP) was comparable among the 4 groups (Table 1), specifically without hypotension in any mice. Mean urine flow rates were similar between the FVB and OVE26 mice, without 6
7 or with RKO (Table 1). Urine flow rate, however, was lower in the FVB RKO group vs. the OVE26 RKO group (P= ). Despite this decrease in flow rate, their absolute urine volumes (>75 µl per period) posed no technical problems for counting inulin or quantification by mass. Overall, these hemodynamic parameters suggested good stability of the surgical preparations during anesthesia. Body weight (BW) was comparable among the 4 groups (Table 1). Weights of both kidneys (KW) in OVE26 mice were markedly increased (0.82 ± 0.03 vs ± 0.02 g in FVB controls, P<0.0001) (Fig 6a), consistent with the well-known nephromegaly in diabetes. Furthermore, KW/BW was also markedly increased in OVE26 vs. FVB controls (2.82 ± 0.11 versus 1.54 ± 0.08 %, respectively; P<0.0001) (Fig 6b). Although nephromegaly was prominent in OVE26 diabetic mice, this was partially attenuated by RAGE deletion (0.71 ± 0.02 g, P <0.005, Δ=14% reduction) (Fig 6a). KW/BW of OVE26 mice was reduced by RKO (2.35 ± 0.05 versus 2.82 ± 0.11 %, p< 0.001, Δ=17 % reduction) (Fig 6b). (b) Absence of impaired renal functional phenotypes in FVB RKO mice - While RKO imparted major structural (Figs 3 & 4) and functional effects in OVE26 mice (Fig 5 & 6, vide infra), we could not detect any significant alterations in BW, Hct, SBP, KW, or renal function in FVB RKO controls. More specifically, GFR was essentially unchanged by RKO in FVB mice whether expressed in units per mouse (Fig 6c), per 100 g BW (Fig 6d), or per g KW (Fig 6e). (c) Renal Insufficiency in OVE26 versus non-diabetic controls - Compared to ageand sex-matched FVB controls, 7 month-old OVE26 showed a significant reduction in GFR (456 ± 18 vs. 325 ± 24 µl/min/mouse, P<0.0003, Δ=29% reduction, Fig 6c). The decline in GFR remained significant whether factored for BW (1106 vs µl/min/100 g BW, P<0.003, Δ=24% reduction, Fig 6d) or factored for KW (401 vs. 940 µl/min/g KW, p<0.001, Δ=57% reduction, Fig 6e). (d) Preservation of renal function in OVE26 mice by RKO - In OVE26 mice, RKO prevented loss of GFR, which otherwise developed in OVE26 mice (436 ± 37 vs. 325 ± 24 µl/min per mouse, P<0.005, Δ=34% increase, Fig 6c). Further, in OVE26 mice, RKO preserved GFR, as levels were comparable to those of FVB and FVB RKO mice (456 ± 18 and 404 ± 21 µl/min per mouse, respectively, Fig 6c). Likewise, reduction in renal function was prevented in OVE26 RKO mice versus OVE26 mice, whether GFR was factored by BW (1457 ± 134 vs ± 78 µl/min/100 g BW, P<0.006, Δ=32% increase, Fig 6d) or factored by KW (620 ± 60 vs. 401 ± 35 µl/min/g KW, P<0.004, Δ=55% increase, Fig 6e). Significantly, in OVE26 RKO mice, GFR was preserved when factored for BW at levels similar to those of FVB controls, with or without RAGE (1457 ± 134 vs ± 56 and 1287 ± 64 µl/min/100 g BW, Fig 6d). Although OVE26 RKO mice did not appear to have fully normalized GFR when factored by KW compared to levels of FVB controls (Fig 6e), this was primarily a mathematical aberration, and due to the marked nephromegaly (Fig 6a). Although RKO significantly reduced KW of diabetic mice, the attenuation was only 14 % (Fig 6a). Thus, RKO is reno-protective in OVE26 mice. The prevention of renal insufficiency conferred by RKO is independent of glycemic control and systemic hemodynamic alterations. Increases in PAI-1, Tgfb1, Tgfb induced, and α1- (IV) collagen transcripts are found in OVE26 kidney cortex but are prevented by RAGE deletion. To explore the pathogenic mechanisms linked to DN in OVE26 mice, we identified significant changes in Serpine1 expression (the gene for Plasminogen activator inhibitor 1 (PAI-1)) through a microarray study comparing expression of genes in isolated glomeruli 7
8 from FVB and OVE26 mouse kidneys at 2 months of age. Serpine1 levels were significantly increased by 1.46 fold in OVE26 glomeruli (False discovery rate (FDR)=0.02) (Table S1, see supplemental information). To determine whether PAI-1 could be a downstream effector of RAGE in the diabetic kidney, we performed real time PCR on kidney cortex RNA from mice at 7 months of age and found that levels of PAI-1 mrna were increased 4.3 fold in OVE26 compared to FVB kidney cortex (P<0.0001), whereas the levels in PAI-1 mrna in OVE26 RKO were significantly lower than those in OVE26 (P<0.0005), and were not significantly different from FVB cortex (P=0.3723) (Fig 7a). One of the central pathways that has been implicated in the pathogenesis of DN is the Tgf-β pathway. Previous studies illustrated that PAI-1 expression may be induced by Tgfβ, leading to inhibition of extracellular matrix degradation.(16-18) PAI-1 can also directly stimulate Tgf-β expression leading to increased synthesis of extracellular matrix.(17) We thus performed real time PCR on kidney cortex RNA from mice for Tgfb1 at 7 months of age and found that levels of Tgfb1 were increased 1.9 fold in OVE26 compared to FVB cortex (P<0.01), whereas levels in OVE26 RKO were much lower and not significantly different from FVB cortex (P=0.8904) (Fig 7B). To further investigate the impact of RAGE on the Tgf-β1 pathway in the OVE26 and OVE26 RKO cortex, we performed real time PCR on TGFβ induced transcript as a measure of activity level and found significantly lower levels in OVE26 RKO compared to OVE26 cortex (P<0.0005) (Fig 7C). In parallel, to explore the effects of RAGE on extracellular matrix deposition, we performed real time PCR for α1-(iv) collagen, one of the collagen IV species secreted by podocytes of the glomeruli,(19) and found significantly lower levels in OVE26 RKO compared to OVE26 cortex (P<0.001) (Fig 7D). The type I Tgf-β receptor may interact with a number of different molecules including RhoA which activates ROCK1.(20; 21) ROCK1 has recently been shown to be potentially important in DN(22; 23) and has been shown to be downstream of RAGE signaling in the diabetic vasculature. (15) We thus measured ROCK1 activity in OVE26 and OVE26 RKO kidney cortex and found that ROCK1 activity was significantly lower in OVE26 RKO compared to OVE26 cortex lysates, N=3 per group (P< 0.005) (Fig 7E). DISCUSSION In this study, we employed OVE26 mice to investigate the role of RAGE for the first time in a model displaying progressive and advanced features that closely resemble human DN, including loss of GFR.(10; 11; 24; 25) Several previous models used to study RAGE, such as STZ injection and db/db mice,(3) do not progress beyond microalbuminuria and mild mesangial expansion. Diabetic mice due to the inos transgene display advanced glomerulosclerosis, but no tubulointerstitial fibrosis or arteriolar hyalinosis, (4; 26) features that are present in OVE26 mice.(9) Here, we have illustrated that several of these advanced features of DN in the OVE26 model are impacted by deletion of RAGE, such as the occurrence of segmental and global glomerular sclerosis, nodule formation, tubular atrophy, podocyte effacement and thickening of the GBM. Importantly, the decline in GFR suggested previously in OVE26 mice,(9) and validated in our study, was completely prevented by RAGE deletion. In the previous study, however, although a 17% decrease in GFR in OVE26 vs. nondiabetic control mice was demonstrated, this occurred in the presence of hypotension, thus supporting that the decline in GFR in OVE26 mice was due to the structural pathology 8
9 associated with DN and not hemodynamic factors.(9), A recent study reported increased serum creatinine in OVE26 mice but paradoxically increased creatinine clearance compared to FVB mice at ages 3, 6 and 9 months.(11) This disparity underscores the limitations of using creatinine clearance to assess GFR, which could be inaccurate due to artifacts from urine collection or due to the use of non-hplc methods for determination of serum creatinine in diabetic animals.(24; 26; 27) To overcome these limitations, we used inulin clearance, the gold standard for measuring GFR (28). Beyond confirming previous findings of segmental and global sclerosis, nodule formation, and tubular atrophy, the observed decline in GFR in OVE26 mice in the absence of hypotension in our studies suggests a causal relationship with the pathology associated with DN. This inference is supported by the degree of mesangial matrix expansion and the accompanying reduction in capillary lumen diameter, which would be expected to decrease ultrafiltration surface area and hence GFR. In addition, the focal tubular atrophy and interstitial fibrosis, which are known to correlate with declining renal function,(29) may have contributed to renal insufficiency. This close functionalpathologic correlation helps establish the utility and validity of OVE26 mice to study clinical DN, providing a basis to test the hypothesis of renoprotection by RKO. Our results indicate that renal insufficiency in OVE26 mice was prevented by RKO, associated with significant ameliorations of pathology and albuminuria. Their nephromegaly was only minimally improved, possibly due to other upstream factors directly consequent to hyperglycemia. Over a shorter duration of STZ-induced diabetes, we previously found normalization of KW/BW by RKO.(3) Another study found persistently increased glomerular volume despite marked podocyte loss in advanced DN of OVE26 mice,(25) making it conceivable for the coexistence of nephromegaly and preserved glomerular function. Despite limitations of previously used DN models, it has been reported that deletion or blockade of RAGE or inhibitors of AGE consistently led to partial preservation of renal structure and/or improved serum creatinine.(3; 5-7; 30-34) The diversity of these models provides further evidence for the role of RAGE, as srage,(3) anti-rage antibodies,(6; 7) or frank RAGE deletion exert salutary effects on the increased albuminuria and histopathology in early stages of mouse DN. Previously, in 27 weekold db/db mice exhibiting low creatinine clearance, we found significant benefits of srage administration,(3) corroborating the adverse impact of RAGE on raising serum creatinine(4) and the salutary action by RKO on reducing serum creatinine in a type 1 DN model.(5) Our present studies, however, provide the first direct evidence for functional preservation in diabetic mice by RKO, as assessed by GFR. Methylglyoxal, a highly reactive α- oxoaldehyde, is formed in cells primarily from the triose phosphate intermediates of glycolysis, dihydroxyacetone phosphate and glyceraldehyde 3-phosphate. In diabetes, hyperglycemia triggers enhanced production of methylglyoxal, one consequence of which is the rapid modification of proteins and other substrates to generate AGEs, which may trigger signaling pathways leading to structural and functional changes of DN, at least in part via the actions of TGF-β and PAI-1. The finding that methylglyoxal is significantly lower in OVE26 RKO mice, which have fewer structural changes and less functional impairment than OVE26 mice, supports this hypothesis. Notably, blood glucose levels in the OVE26 RKO did not differ significantly from the OVE26 mice. This suggests that the difference in levels of methylglyoxal did not result from a reduction 9
10 in plasma glucose. Glyoxalase 1, a defense against glycation in vivo,(35) detoxifies reactive α-oxoaldehydes, thereby, removing deleterious species such as methylglyoxal. Our finding that glyoxalase 1 mrna and protein levels are higher in OVE26 RKO mouse kidney cortex suggests that reduced activation of RAGE by AGEs may occur because of enhanced removal of methylglyoxal. The importance of the link between RAGE and glyoxalase 1 is that this may set up a positive feedback loop with relevant consequences. We have shown that by deleting RAGE, glyoxalase 1 mrna and protein levels increase significantly, which decreases the tissue concentration of MG and suppressed related AGE formation. These data suggest that disruption of RAGE signaling may be a beneficial therapeutic target in prevention of progression of DN. To begin to explore potential molecular mechanisms accounting for the reduction in mesangial sclerosis noted in OVE26 RKO mice, we assessed mrna levels of PAI-1, Tgf-β1, Tgf-β induced, and α1- (IV) collagen. PAI-1 has been implicated previously in the pathogenesis of DN. In PAI-1 knock out mice treated with stz or crossed with type 2 diabetic db/db mice, albuminuria was improved and glomerular injury was reduced compared to the diabetic, PAI-1- expressing animals.(17; 36; 37) A link between PAI-1 induction and RAGE has been previously demonstrated, but only in cell culture. Berrou et al. measured PAI-1 protein expression in cultured mesangial cells in response to both glycated albumin and carboxymethyl lysine (CML) AGE, two RAGE ligands identified through in vitro experiments. An anti-rage antibody partially blocked the AGE and CML induced PAI-1 expression in these cells.(38) The significant reduction we find in Tgf-β1, Tgf-β induced and PAI-1 in OVE26 RKO mice suggests that deletion of RAGE may both reduce mesangial matrix accumulation and facilitate matrix degradation, both factors linked to reduced glomerulosclerosis. Reduced levels of α1-(iv) collagen expression in OVE26 RKO kidney cortex confirm these findings, as they are indicative of mesangial matrix accumulation. Here we have shown that ROCK1 activity is significantly decreased in OVE26 RKO compared to OVE26 kidney cortex lysates. The importance of this finding is highlighted not only by known roles of ROCK1 in DN,(22; 23), the involvement of ROCK1 in Tgf-β and PAI-1 downstream signaling,(20; 21) but also our recent finding that activation of the ROCK1 branch of the Tgf-β pathway contributes to RAGE-dependent acceleration of atherosclerosis in diabetic ApoE null mice.(15) The significant changes in ROCK1 activity in the kidney in the absence of RAGE suggest a novel mechanism by which RAGE may promote pathological change in DN. In future studies, exploration of the relevance of the OVE26 model of DN to human DN could be explored by administering renoprotective agents that are effective in humans, such as ACE inhibitors, to RAGEexpressing and RKO OVE26 mice. AGE formation and the renin-angiotensin system have been shown to interact in the progression of renal disease. Wilkinson-Berka et al. have shown that blocking AGE formation can ameliorate angiotensin II-dependent renal injury.(39) In addition, Thomas et al have shown that advanced glycation endproducts can activate ACE and other components of the intra-renal renin-angiotensin system.(40) We have shown that increased levels of methylglyoxal are found in OVE26 kidney cortex and that the decreased pathology found in OVE26 RKO mice is accompanied by lower levels of methylglyoxal and increased glyoxalase 1 activity. We expect that treatment of OVE26 mice with ACE inhibitors could serve to prevent progression of renal disease in this model by potentially blocking ACE levels that may be elevated by 10
11 the presence of excess AGEs in the OVE26 mice. In conclusion, our data underscore important roles for RAGE in the structural and functional deteriorations in advanced and progressive DN. Our findings support the notion that RAGE blockade may confer significant benefits by ameliorating severe renal histopathology and by preventing renal insufficiency in mice or patients at risk for DN. Author contributions: Nina Reiniger: prepared the initial main draft of the manuscript subsequently distributed to all coauthors for editing and comments, substantial contribution to initial design of study, urine and tissue collection, blood glucose measurements, albumin:creatinine measurements, fluorescent staining imaging, glomerular isolation, RNA preparation, labeling and fragmentation of cdna for microarray, real time PCRs, breeding and maintenance of mouse colony, some statistical analysis excluding microarray, coordination of mouse and mouse sample distribution to other coauthors. Kai Lau: GFR, Hct, BP and HgA1C measurements, contributed to manuscript, reviewed and edited manuscript. Daren McCalla: urine and tissue collection, blood glucose measurements, albumin: creatinine measurements. Bonnie Eby: GFR, Hct, BP and HgA1C measurements. Bin Cheng: statistical analyses, excluding the microarray. Yan Lu: sections of frozen kidney tissue, fluorescent staining and imaging. Wu Qu: generated the anti-rage antibody. Nosirudeen Quadri: methyl glyoxal measurements. Radha Ananthakrishnan: contributed to the development and validation of the assays in the laboratory for the measurement of methylglyoxal, performed methyl glyoxal measurements, reviewed and edited manuscript. Maryana Furmansky: tissue collection, glomerular isolation. Rosa Rosario: breeding, genotyping and maintenance of the mouse colony. Fei Song: breeding, genotyping and maintenance of the mouse colony. Vivek Rai: ROCK1 activity assay. Alan Weinberg: statistical analyses, excluding the microarray. Richard Friedman: data analysis on the microarray results, reviewed and edited manuscript. Ravichandran Ramasamy: contributed to initial design of study, arranged funding, supervised research, developed and validated in the laboratory the assays for measurement of methylglyoxal, reviewed all primary data on measurements of methylglyoxal and glyoxalase 1, contributed to manuscript, reviewed and edited manuscript. Vivette D Agati: analysis of the PAS stained tissue sections and electron micrographs including scoring of mesangial sclerosis, cast formation, GBM thickness measurements, and podocyte effacement, contributed to initial design of study, contributed to manuscript, reviewed and edited manuscript. Ann Marie Schmidt: substantial contribution to initial design of study, arranged funding, supervised research, reviewed all primary data with Dr. Reiniger and determined the direction of the research, reviewed and edited the first draft of the manuscript prepared by Dr. Reiniger prior to distribution to all co-authors, reviewed and edited manuscript ACKNOWLEDGEMENTS The authors would like to thank Alexander Lau, University of Oklahoma Health Sciences Center, for help with the inulin clearance experiments and Joshua Karas, Columbia University, for help with mouse tissue collection and processing. In addition, we are grateful to Ms. Latoya Woods, Columbia University, for her expert assistance in preparation of this manuscript. Portions of this study were presented at 2007, 2008 and 2009 annual meetings of the American Society of Nephrology. This work was made possible with funding by the National 11
12 Institutes of Health Ruth L. Kirschstein National Research Service Award 1F32DK and the Juvenile Diabetes Research Foundation. REFERENCES 1. Park L, Raman KG, Lee KJ, Lu Y, Ferran LJ, Jr., Chow WS, Stern D, Schmidt AM: Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat Med 4: , Tanji N, Markowitz GS, Fu C, Kislinger T, Taguchi A, Pischetsrieder M, Stern D, Schmidt AM, D'Agati VD: Expression of advanced glycation end products and their cellular receptor RAGE in diabetic nephropathy and nondiabetic renal disease. J Am Soc Nephrol 11: , Wendt TM, Tanji N, Guo J, Kislinger TR, Qu W, Lu Y, Bucciarelli LG, Rong LL, Moser B, Markowitz GS, Stein G, Bierhaus A, Liliensiek B, Arnold B, Nawroth PP, Stern DM, D'Agati VD, Schmidt AM: RAGE drives the development of glomerulosclerosis and implicates podocyte activation in the pathogenesis of diabetic nephropathy. Am J Pathol 162: , Yamamoto Y, Kato I, Doi T, Yonekura H, Ohashi S, Takeuchi M, Watanabe T, Yamagishi S, Sakurai S, Takasawa S, Okamoto H, Yamamoto H: Development and prevention of advanced diabetic nephropathy in RAGE-overexpressing mice. J Clin Invest 108: , Myint KM, Yamamoto Y, Doi T, Kato I, Harashima A, Yonekura H, Watanabe T, Shinohara H, Takeuchi M, Tsuneyama K, Hashimoto N, Asano M, Takasawa S, Okamoto H, Yamamoto H: RAGE control of diabetic nephropathy in a mouse model: effects of RAGE gene disruption and administration of low-molecular weight heparin. Diabetes 55: , Jensen LJ, Denner L, Schrijvers BF, Tilton RG, Rasch R, Flyvbjerg A: Renal effects of a neutralising RAGE-antibody in long-term streptozotocin-diabetic mice. J Endocrinol 188: , Flyvbjerg A, Denner L, Schrijvers BF, Tilton RG, Mogensen TH, Paludan SR, Rasch R: Long-term renal effects of a neutralizing RAGE antibody in obese type 2 diabetic mice. Diabetes 53: , Epstein PN, Overbeek PA, Means AR: Calmodulin-induced early-onset diabetes in transgenic mice. Cell 58: , Zheng S, Noonan WT, Metreveli NS, Coventry S, Kralik PM, Carlson EC, Epstein PN: Development of late-stage diabetic nephropathy in OVE26 diabetic mice. Diabetes 53: , Powell DW, Bertram CC, Cummins TD, Barati MT, Zheng S, Epstein PN, Klein JB: Renal tubulointerstitial fibrosis in OVE26 type 1 diabetic mice. Nephron Exp Nephrol 111:e11-19, Yuzawa Y, Niki I, Kosugi T, Maruyama S, Yoshida F, Takeda M, Tagawa Y, Kaneko Y, Kimura T, Kato N, Yamamoto J, Sato W, Nakagawa T, Matsuo S: Overexpression of calmodulin in pancreatic beta cells induces diabetic nephropathy. J Am Soc Nephrol 19: , Ohmori S, Mori M, Kawase M, Tsuboi S: Determination of methylglyoxal as 2- methylquinoxaline by high-performance liquid chromatography and its application to biological samples. J Chromatogr 414: , Cowgill LD, Goldfarb S, Lau K, Slatopolsky E, Agus ZS: Evidence for an intrinsic renal tubular defect in mice with genetic hypophosphatemic rickets. J Clin Invest 63: ,
13 14. Lau K, Eby BK: Tubular mechanism for the spontaneous hypercalciuria in laboratory rat. J Clin Invest 70: , Bu DX, Rai V, Shen X, Rosario R, Lu Y, D'Agati V, Yan SF, Friedman RA, Nuglozeh E, Schmidt AM: Activation of the ROCK1 Branch of the Transforming Growth Factor-{beta} Pathway Contributes to RAGE-Dependent Acceleration of Atherosclerosis in Diabetic ApoE- Null Mice. Circ Res, Kutz SM, Hordines J, McKeown-Longo PJ, Higgins PJ: TGF-beta1-induced PAI-1 gene expression requires MEK activity and cell-to-substrate adhesion. J Cell Sci 114: , Nicholas SB, Aguiniga E, Ren Y, Kim J, Wong J, Govindarajan N, Noda M, Wang W, Kawano Y, Collins A, Hsueh WA: Plasminogen activator inhibitor-1 deficiency retards diabetic nephropathy. Kidney Int 67: , Baricos WH, Cortez SL, el-dahr SS, Schnaper HW: ECM degradation by cultured human mesangial cells is mediated by a PA/plasmin/MMP-2 cascade. Kidney Int 47: , Iglesias-de la Cruz MC, Ziyadeh FN, Isono M, Kouahou M, Han DC, Kalluri R, Mundel P, Chen S: Effects of high glucose and TGF-beta1 on the expression of collagen IV and vascular endothelial growth factor in mouse podocytes. Kidney Int 62: , Mulder KM: Role of Ras and Mapks in TGFbeta signaling. Cytokine Growth Factor Rev 11:23-35, Derynck R, Zhang YE: Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 425: , Peng F, Wu D, Gao B, Ingram AJ, Zhang B, Chorneyko K, McKenzie R, Krepinsky JC: RhoA/Rho-kinase contribute to the pathogenesis of diabetic renal disease. Diabetes 57: , Gojo A, Utsunomiya K, Taniguchi K, Yokota T, Ishizawa S, Kanazawa Y, Kurata H, Tajima N: The Rho-kinase inhibitor, fasudil, attenuates diabetic nephropathy in streptozotocin-induced diabetic rats. Eur J Pharmacol 568: , Breyer MD: Stacking the deck for drug discovery in diabetic nephropathy: in search of an animal model. J Am Soc Nephrol 19: , Teiken JM, Audettey JL, Laturnus DI, Zheng S, Epstein PN, Carlson EC: Podocyte loss in aging OVE26 diabetic mice. Anat Rec (Hoboken) 291: , Breyer MD, Bottinger E, Brosius FC, 3rd, Coffman TM, Harris RC, Heilig CW, Sharma K: Mouse models of diabetic nephropathy. J Am Soc Nephrol 16:27-45, Dunn SR, Qi Z, Bottinger EP, Breyer MD, Sharma K: Utility of endogenous creatinine clearance as a measure of renal function in mice. Kidney Int 65: , Gaspari F, Perico N, Remuzzi G: Measurement of glomerular filtration rate. Kidney Int Suppl 63:S , Ziyadeh FN: The extracellular matrix in diabetic nephropathy. Am J Kidney Dis 22: , Miyoshi H, Taguchi T, Sugiura M, Takeuchi M, Yanagisawa K, Watanabe Y, Miwa I, Makita Z, Koike T: Aminoguanidine pyridoxal adduct is superior to aminoguanidine for preventing diabetic nephropathy in mice. Horm Metab Res 34: , Peppa M, Brem H, Cai W, Zhang JG, Basgen J, Li Z, Vlassara H, Uribarri J: Prevention and reversal of diabetic nephropathy in db/db mice treated with alagebrium (ALT-711). Am J Nephrol 26: , Ellis EN, Good BH: Prevention of glomerular basement membrane thickening by aminoguanidine in experimental diabetes mellitus. Metabolism 40: ,
14 33. Soulis-Liparota T, Cooper M, Papazoglou D, Clarke B, Jerums G: Retardation by aminoguanidine of development of albuminuria, mesangial expansion, and tissue fluorescence in streptozocin-induced diabetic rat. Diabetes 40: , Vlassara H, Striker LJ, Teichberg S, Fuh H, Li YM, Steffes M: Advanced glycation end products induce glomerular sclerosis and albuminuria in normal rats. Proc Natl Acad Sci U S A 91: , Thornalley PJ: The glyoxalase system in health and disease. Mol Aspects Med 14: , Collins SJ, Alexander SL, Lopez-Guisa JM, Cai X, Maruvada R, Chua SC, Zhang G, Okamura DM, Matsuo S, Eddy AA: Plasminogen activator inhibitor-1 deficiency has renal benefits but some adverse systemic consequences in diabetic mice. Nephron Exp Nephrol 104:e23-34, Lassila M, Fukami K, Jandeleit-Dahm K, Semple T, Carmeliet P, Cooper ME, Kitching AR: Plasminogen activator inhibitor-1 production is pathogenetic in experimental murine diabetic renal disease. Diabetologia 50: , Berrou J, Tostivint I, Verrecchia F, Berthier C, Boulanger E, Mauviel A, Marti HP, Wautier MP, Wautier JL, Rondeau E, Hertig A: Advanced glycation end products regulate extracellular matrix protein and protease expression by human glomerular mesangial cells. Int J Mol Med 23: , Wilkinson-Berka JL, Kelly DJ, Koerner SM, Jaworski K, Davis B, Thallas V, Cooper ME: ALT-946 and aminoguanidine, inhibitors of advanced glycation, improve severe nephropathy in the diabetic transgenic (mren-2)27 rat. Diabetes 51: , Thomas MC, Tikellis C, Burns WM, Bialkowski K, Cao Z, Coughlan MT, Jandeleit-Dahm K, Cooper ME, Forbes JM: Interactions between renin angiotensin system and advanced glycation in the kidney. J Am Soc Nephrol 16: , 2005 Table 1: Metabolic, hemodynamic and urine flow data during inulin clearance studies Groups Genotypes N Body weight Hct initial Δ Hct Hct final Ave. of 5 Hct during clearance Ave. systolic BP Ave. urine flow rate (g) (%) (%) (%) (%) (torr) (μl/min) I FVB Mean ± SE II FVB RKO Mean ± SE III OVE26 Mean ± SE IV OVE26 RKO Mean * 9.08 ± SE *P < 0.01 vs. group II. 14
15 Figure Legends Figure 1. (a) RAGE is expressed in OVE26 glomeruli: age 1 month. Original magnification: 1000x. (b) Blood glucose levels ( ### P<0.0001) (N=12-15/group, except OVE26 RKO, N=6). (c) Hg A1c levels were measured at age 7 months on randomly selected N=4, 3, 9, and 6 mice used in the GFR studies in groups FVB, FVB RKO, OVE26 and OVE26 RKO, respectively ( ## P<0.0005, ### P<0.0001). (d) Levels of methylglyoxal (MG) and in the kidney cortex of FVB, FVB RKO, OVE26 and OVE26 RKO mice at age 7 months (*P<0.05). N=6 per group. (e) Real time PCR for Glyoxalase 1 was performed, normalized to 18s transcript levels and expressed as fold change compared to the OVE26 group (*P<0.05). N=6 per group. (f) Glyoxalase 1 protein levels measured by Western blot, normalized to Actin levels and expressed as fold change compared to the OVE26 group (*P<0.05)(N=3 per group). (g) Glyoxalase 1 staining in OVE26 and OVE26 RKO glomeruli: age 7 months. Original magnification: 1000x. Figure 2. Histology and Ultrastructural Pathology in Diabetic OVE mice: age 7 months. By light microscopy, the glomeruli of 7-month-old male OVE26 mice display global mesangial sclerosis with nodularity (a), with progression in some glomeruli to segmental glomerulosclerosis (c) and global glomerulosclerosis (d). There is focal tubular atrophy, interstitial fibrosis and chronic inflammation (b). By electron microscopy, the mesangial areas are expanded by increased matrix and electron dense hyaline material, consistent with insuded plasma proteins (e). There is thickening of glomerular basement membranes with overlying effacement of foot processes (f). Some mesangial areas have marked mesangial sclerosis with a nodular aspect (g). In areas of severe mesangial sclerosis, the glomerular capillary lumina are narrowed and focally occluded by the mesangial encroachment (h, L= lumen) (Arrow indicates focal lumenal occlusion). Original magnifications are marked above each image. Figure 3. Deletion of RAGE in OVE26 mice imparts partial protection from the structural abnormalities of DN at age 7 months. (a). No histologic abnormalities were detected in FVB RKO mice (not shown). By contrast, OVE26 mice display well developed features of diabetic nephropathy including diffuse and global mesangial sclerosis and focal hyaline casts (b). OVE26 RKO mice are markedly protected from the development of mesangial sclerosis and tubular cast formation (c). There were significant differences between OVE26 mice and OVE26 RKO mice with respect to % cortical area occupied by casts (d) and the severity of mesangial sclerosis (e), where 0=no mesangial sclerosis; 1=mild; 2=moderate; 3=severe. (*P<0.05). Original magnifications are marked above each image. Semi-quantitative scoring (d,e) was performed on N=7 OVE26 RKO and N=13 OVE26 mice. Figure 4. Deletion of RAGE in OVE26 mice imparts partial protection from the ultrastructural abnormalities of DN at age 7 months. Electron microscopy was performed on 7 month old male kidney cortex samples. FVB mice display normal glomerular ultrastructural features with wellpreserved foot processes and glomerular basement membranes of normal thickness (a). By comparison, OVE26 glomeruli display increased mesangial matrix forming focal nodules, thickened glomerular basement membranes and prominent foot process effacement (b). OVE26 RKO mice were partially protected from these changes, leading to less mesangial sclerosis and partial restoration of foot processes (FP) (c). Semi-quantitative scoring of podocyte effacement 15
16 (N=5) (d). Measurements of GBM thickness (N=5) (e). (***P<0.005, ### P<0.0001). Original magnifications are marked above each image. Figure 5. Deletion of RAGE in OVE26 mice imparts partial protection from functional abnormalities of DN at age 7 months. Albumin:creatinine levels were measured in male FVB, FVB RKO, OVE26 and OVE26 RKO mouse urine retrieved from metabolic cages at 7 months of age. (***P<0.005, ### P<0.0001). N=7-13 per group. Figure 6. Deletion of RAGE in OVE26 mice preserves GFR in OVE26 mice at age 7 months. Body weight and glomerular function: age 7 months. (a) Weights of both kidneys from 7-mo old male mice. (b) Ratios of kidney weight to body weight of 7-mo old mice. (c-e) Clearance of inulin (CIN) expressed as (c) CIN per mouse, (d) CIN per 100g body weight and (e) CIN per g kidney weight. (**P<0.01, ***P<0.005, ## P<0.0005, ### P<0.0001). N=6-10 per group, as indicated in Table 1. Figure 7. PAI-1 (Serpine1), Tgfb1, Tgfb induced, α1-(iv) collagen mrna transcripts and ROCK1 activity are lower in OVE26 RKO kidney cortex compared to OVE26 kidney cortex levels at age 7 months. Real time PCR for (a) PAI-1 (b) Tgfb1, (c) Tgfb induced, and (d) α1 - (IV) collagen gene products was performed, normalized to 18s transcript levels and expressed as fold change compared to the FVB or OVE26 group (**P<0.01, ***P<0.005, ## P<0.0005, ### P<0.0001). N=6 per group. (e) ROCK1 activity was measured as the amount of phosphorylated MYPT1 compared to total ROCK1 levels by Western blot. Relative activity is expressed as fold change compared to the OVE26 group (***P<0.005)(N=3 per group). 16
17 17 RAGE deletion preserves function in DN in OVE26
18 18 RAGE deletion preserves function in DN in OVE26
19 19 RAGE deletion preserves function in DN in OVE26
20 20 RAGE deletion preserves function in DN in OVE26
21 21 RAGE deletion preserves function in DN in OVE26
22 22 RAGE deletion preserves function in DN in OVE26
The receptor for advanced glycation end products
 ORIGINAL ARTICLE Deletion of the Receptor for Advanced Glycation End Products Reduces Glomerulosclerosis and Preserves Renal Function in the Diabetic Mouse Nina Reiniger, Kai Lau, 2 Daren McCalla, Bonnie
ORIGINAL ARTICLE Deletion of the Receptor for Advanced Glycation End Products Reduces Glomerulosclerosis and Preserves Renal Function in the Diabetic Mouse Nina Reiniger, Kai Lau, 2 Daren McCalla, Bonnie
Diabetic Nephropathy in Spontaneously Diabetic Torii (SDT) Rats
 The Open Diabetes Journal, 2011, 4, 45-49 45 Diabetic Nephropathy in Spontaneously Diabetic Torii (SDT) Rats Takeshi Ohta * and Tomohiko Sasase Open Access Biological/Pharmacological Research Laboratories,
The Open Diabetes Journal, 2011, 4, 45-49 45 Diabetic Nephropathy in Spontaneously Diabetic Torii (SDT) Rats Takeshi Ohta * and Tomohiko Sasase Open Access Biological/Pharmacological Research Laboratories,
Mouse Models of Diabetic Nephropathy. Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI
 Mouse Models of Diabetic Nephropathy Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI Group Members Mount Sinai: Phenotyping, Molecular Pathology & Validation Erwin
Mouse Models of Diabetic Nephropathy Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI Group Members Mount Sinai: Phenotyping, Molecular Pathology & Validation Erwin
Surgical Pathology Report
 Louisiana State University Health Sciences Center Department of Pathology Shreveport, Louisiana Accession #: Collected: Received: Reported: 6/1/2012 09:18 6/2/2012 09:02 6/2/2012 Patient Name: Med. Rec.
Louisiana State University Health Sciences Center Department of Pathology Shreveport, Louisiana Accession #: Collected: Received: Reported: 6/1/2012 09:18 6/2/2012 09:02 6/2/2012 Patient Name: Med. Rec.
renoprotection therapy goals 208, 209
 Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Supplementary Figure 1:
 Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
Diabetic Nephropathy
 Diabetic Nephropathy Objectives: Know what Diabetic Nephropathy means. Know how common is Diabetic nephropathy in Saudi Arabia and to appreciate how bad are this complications. Know the risk factors of
Diabetic Nephropathy Objectives: Know what Diabetic Nephropathy means. Know how common is Diabetic nephropathy in Saudi Arabia and to appreciate how bad are this complications. Know the risk factors of
Case # 2 3/27/2017. Disclosure of Relevant Financial Relationships. Clinical history. Clinical history. Laboratory findings
 Case # 2 Christopher Larsen, MD Arkana Laboratories Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to influence or control the content
Case # 2 Christopher Larsen, MD Arkana Laboratories Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to influence or control the content
RENAL HISTOPATHOLOGY
 RENAL HISTOPATHOLOGY Peter McCue, M.D. Department of Pathology, Anatomy & Cell Biology Sidney Kimmel Medical College There are no conflicts of interest. 1 Goals and Objectives! Goals Provide introduction
RENAL HISTOPATHOLOGY Peter McCue, M.D. Department of Pathology, Anatomy & Cell Biology Sidney Kimmel Medical College There are no conflicts of interest. 1 Goals and Objectives! Goals Provide introduction
Ordering Physician. Collected REVISED REPORT. Performed. IgG IF, Renal MCR. Lambda IF, Renal MCR. C1q IF, Renal. MCR Albumin IF, Renal MCR
 RenalPath Level IV Wet Ts IgA I Renal IgM I Renal Kappa I Renal Renal Bx Electron Microscopy IgG I Renal Lambda I Renal C1q I Renal C3 I Renal Albumin I Renal ibrinogen I Renal Mayo Clinic Dept. of Lab
RenalPath Level IV Wet Ts IgA I Renal IgM I Renal Kappa I Renal Renal Bx Electron Microscopy IgG I Renal Lambda I Renal C1q I Renal C3 I Renal Albumin I Renal ibrinogen I Renal Mayo Clinic Dept. of Lab
Physiopathologie de la Néphropathie Diabétique
 Physiopathologie de la Néphropathie Diabétique Philippe Rieu Service de Néphrologie et de Transplantation UMR CNRS 6198 Reims Development of diabetic nephropathy 160 14 12 GFR (ml/min) 120 80 40 I II III
Physiopathologie de la Néphropathie Diabétique Philippe Rieu Service de Néphrologie et de Transplantation UMR CNRS 6198 Reims Development of diabetic nephropathy 160 14 12 GFR (ml/min) 120 80 40 I II III
Diabetic Nephropathy. Introduction/Clinical Setting. Pathologic Findings Light Microscopy. J. Charles Jennette
 12 Diabetic Nephropathy J. Charles Jennette Introduction/Clinical Setting Diabetic nephropathy is a clinical syndrome in a patient with diabetes mellitus that is characterized by persistent albuminuria,
12 Diabetic Nephropathy J. Charles Jennette Introduction/Clinical Setting Diabetic nephropathy is a clinical syndrome in a patient with diabetes mellitus that is characterized by persistent albuminuria,
Mouse Models of Diabetic Nephropathy. Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI
 Mouse Models of Diabetic Nephropathy Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI Group Members Mount Sinai: Phenotyping, Molecular Pathology & Validation Erwin
Mouse Models of Diabetic Nephropathy Mount Sinai / Jefferson / Einstein / Minnesota Erwin Böttinger, PI Kumar Sharma, Co-PI Group Members Mount Sinai: Phenotyping, Molecular Pathology & Validation Erwin
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Light and electron microscopical studies of focal glomerular sclerosis
 J. clin. Path., 1971, 24, 846-850 Light and electron microscopical studies of focal glomerular sclerosis A. H. NAGI, F. ALEXANDER, AND R. LANNIGAN From the Department of Pathology, Queen's University of
J. clin. Path., 1971, 24, 846-850 Light and electron microscopical studies of focal glomerular sclerosis A. H. NAGI, F. ALEXANDER, AND R. LANNIGAN From the Department of Pathology, Queen's University of
Introduction. Acute sodium overload produces renal tubulointerstitial inflammation in normal rats
 Acute sodium overload produces renal tubulointerstitial inflammation in normal rats MI Roson, et al. Kidney International (2006) Introduction Present by Kanya Bunnan and Wiraporn paebua Tubular sodium
Acute sodium overload produces renal tubulointerstitial inflammation in normal rats MI Roson, et al. Kidney International (2006) Introduction Present by Kanya Bunnan and Wiraporn paebua Tubular sodium
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Effects of beraprost sodium on renal function and inflammatory factors of rats with diabetic nephropathy
 Effects of beraprost sodium on renal function and inflammatory factors of rats with diabetic nephropathy J. Guan 1,2, L. Long 1, Y.-Q. Chen 1, Y. Yin 1, L. Li 1, C.-X. Zhang 1, L. Deng 1 and L.-H. Tian
Effects of beraprost sodium on renal function and inflammatory factors of rats with diabetic nephropathy J. Guan 1,2, L. Long 1, Y.-Q. Chen 1, Y. Yin 1, L. Li 1, C.-X. Zhang 1, L. Deng 1 and L.-H. Tian
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
RENOPROTECTIVE EFFECTS OF COMBINED ALISKIREN AND VALSARTAN IN PROGRESSIVE DIABETIC NEPHROPATHY IN RATS
 Research Article RENOPROTECTIVE EFFECTS OF COMBINED ALISKIREN AND VALSARTAN IN PROGRESSIVE DIABETIC NEPHROPATHY IN RATS Hemanth Kumar. Nyathani, Prashanth. Srirangam*, Vidya Sagar Jinugu Vaagdevi College
Research Article RENOPROTECTIVE EFFECTS OF COMBINED ALISKIREN AND VALSARTAN IN PROGRESSIVE DIABETIC NEPHROPATHY IN RATS Hemanth Kumar. Nyathani, Prashanth. Srirangam*, Vidya Sagar Jinugu Vaagdevi College
12. GLYCATION AND AGE IN DIABETES AND COMPLICATIONS
 12. GLYCATION AND AGE IN DIABETES AND COMPLICATIONS Dr. Zdenka Turk Vuk Vrhovac University Clinic, Zagreb, Croatia Epidemiological studies have confirmed that hyperglycaemia is the most important factor
12. GLYCATION AND AGE IN DIABETES AND COMPLICATIONS Dr. Zdenka Turk Vuk Vrhovac University Clinic, Zagreb, Croatia Epidemiological studies have confirmed that hyperglycaemia is the most important factor
Diabetic Nephropathy 2009
 Diabetic Nephropathy 2009 Michael T McDermott MD Director, Endocrinology and Diabetes Practice University of Colorado Hospital Michael.mcdermott@ucdenver.edu Diabetic Nephropathy Clinical Stages Hyperfunction
Diabetic Nephropathy 2009 Michael T McDermott MD Director, Endocrinology and Diabetes Practice University of Colorado Hospital Michael.mcdermott@ucdenver.edu Diabetic Nephropathy Clinical Stages Hyperfunction
Year 2004 Paper one: Questions supplied by Megan
 QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
Interesting case seminar: Native kidneys Case Report:
 Interesting case seminar: Native kidneys Case Report: Proximal tubulopathy and light chain deposition disease presented as severe pulmonary hypertension with right-sided cardiac dysfunction and nephrotic
Interesting case seminar: Native kidneys Case Report: Proximal tubulopathy and light chain deposition disease presented as severe pulmonary hypertension with right-sided cardiac dysfunction and nephrotic
Glomerular Pathology- 1 Nephrotic Syndrome. Dr. Nisreen Abu Shahin
 Glomerular Pathology- 1 Nephrotic Syndrome Dr. Nisreen Abu Shahin The Nephrotic Syndrome a clinical complex resulting from glomerular disease & includes the following: (1) massive proteinuria (3.5 gm /day
Glomerular Pathology- 1 Nephrotic Syndrome Dr. Nisreen Abu Shahin The Nephrotic Syndrome a clinical complex resulting from glomerular disease & includes the following: (1) massive proteinuria (3.5 gm /day
Histopathology: Hypertension and diabetes in the kidney These presentations are to help you identify basic histopathological features.
 Histopathology: Hypertension and diabetes in the kidney These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you need
Histopathology: Hypertension and diabetes in the kidney These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you need
Reversal of Microalbuminuria A Causative Factor of Diabetic Nephropathy is Achieved with ACE Inhibitors than Strict Glycemic Control
 ISSN 0976 3333 Available Online at www.ijpba.info International Journal of Pharmaceutical & Biological Archives 2013; 4(5): 923-928 ORIGINAL RESEARCH ARTICLE Reversal of Microalbuminuria A Causative Factor
ISSN 0976 3333 Available Online at www.ijpba.info International Journal of Pharmaceutical & Biological Archives 2013; 4(5): 923-928 ORIGINAL RESEARCH ARTICLE Reversal of Microalbuminuria A Causative Factor
Glomerular pathology in systemic disease
 Glomerular pathology in systemic disease Lecture outline Lupus nephritis Diabetic nephropathy Glomerulonephritis Associated with Bacterial Endocarditis and Other Systemic Infections Henoch-Schonlein Purpura
Glomerular pathology in systemic disease Lecture outline Lupus nephritis Diabetic nephropathy Glomerulonephritis Associated with Bacterial Endocarditis and Other Systemic Infections Henoch-Schonlein Purpura
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Pathogenesis of diabetic nephropathy
 MINI REVIEW Zemin Cao, Mark E Cooper* ABSTRACT As the increasing prevalence of diabetes reaches epidemic proportions worldwide, diabetic nephropathy and associated end-stage renal failure will be an unavoidable
MINI REVIEW Zemin Cao, Mark E Cooper* ABSTRACT As the increasing prevalence of diabetes reaches epidemic proportions worldwide, diabetic nephropathy and associated end-stage renal failure will be an unavoidable
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Diabetic Nephropathy
 Diabetic Nephropathy Outline Introduction of diabetic nephropathy Manifestations of diabetic nephropathy Staging of diabetic nephropathy Microalbuminuria Diagnosis of diabetic nephropathy Treatment of
Diabetic Nephropathy Outline Introduction of diabetic nephropathy Manifestations of diabetic nephropathy Staging of diabetic nephropathy Microalbuminuria Diagnosis of diabetic nephropathy Treatment of
ORIGINAL ARTICLE Urinary type IV collagen levels in diabetes mellitus
 Malaysian J Pathol 21; 32(1) : 43 47 ORIGINAL ARTICLE Urinary type IV collagen levels in diabetes mellitus Pavai STHANESHWAR MBBS, MD and *Siew-Pheng CHAN MBBS, FRCP Departments of Pathology and *Medicine,
Malaysian J Pathol 21; 32(1) : 43 47 ORIGINAL ARTICLE Urinary type IV collagen levels in diabetes mellitus Pavai STHANESHWAR MBBS, MD and *Siew-Pheng CHAN MBBS, FRCP Departments of Pathology and *Medicine,
The Ebf1 knockout mouse and glomerular maturation
 Repository of the Max Delbrück Center for Molecular Medicine (MDC) Berlin (Germany) http://edoc.mdc-berlin.de/14009/ The Ebf1 knockout mouse and glomerular maturation Schmidt-Ott, K.M. Published in final
Repository of the Max Delbrück Center for Molecular Medicine (MDC) Berlin (Germany) http://edoc.mdc-berlin.de/14009/ The Ebf1 knockout mouse and glomerular maturation Schmidt-Ott, K.M. Published in final
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Chapter 2. Standardization of rat model for diabetic nephropathy
 Chapter 2 Standardization of rat model for diabetic nephropathy 35 2.1 BACKGROUND Several animal models have been developed to elucidate the pathophysiology of DNP and to identify innovative therapies
Chapter 2 Standardization of rat model for diabetic nephropathy 35 2.1 BACKGROUND Several animal models have been developed to elucidate the pathophysiology of DNP and to identify innovative therapies
Methylglyoxal Level In Type-2 Diabetes Mellitus Patients: A Prospective, Randomized and Case Control S
 Methylglyoxal Level In Type-2 Diabetes Mellitus Patients: A Prospective, Randomized and Case Control S Methylglyoxal Level In Type-2 Diabetes Mellitus Patients: A Prospective, Randomized and Case Control
Methylglyoxal Level In Type-2 Diabetes Mellitus Patients: A Prospective, Randomized and Case Control S Methylglyoxal Level In Type-2 Diabetes Mellitus Patients: A Prospective, Randomized and Case Control
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
A clinical syndrome, composed mainly of:
 Nephritic syndrome We will discuss: 1)Nephritic syndrome: -Acute postinfectious (poststreptococcal) GN -IgA nephropathy -Hereditary nephritis 2)Rapidly progressive GN (RPGN) A clinical syndrome, composed
Nephritic syndrome We will discuss: 1)Nephritic syndrome: -Acute postinfectious (poststreptococcal) GN -IgA nephropathy -Hereditary nephritis 2)Rapidly progressive GN (RPGN) A clinical syndrome, composed
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Eremina V, Jefferson JA, Kowalewska J, et al. VEGF inhibition
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Eremina V, Jefferson JA, Kowalewska J, et al. VEGF inhibition
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY
 JCD REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY SAPTARSHI MUKHOPADHYAY* *FACULTY (DEPARTMENT OF MEDICINE), B R SINGH HOSPITAL (EASTERN RAILWAY), KOLKATA. INTRODUCTION renal disease. It
JCD REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY SAPTARSHI MUKHOPADHYAY* *FACULTY (DEPARTMENT OF MEDICINE), B R SINGH HOSPITAL (EASTERN RAILWAY), KOLKATA. INTRODUCTION renal disease. It
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
3. PODOCYTE INJURY IN GLOMERULAR DISEASES
 How to Cite this article: Podocyte Injury in Glomerular Diseases - ejifcc 20/01 2009 http://www.ifcc.org 3. PODOCYTE INJURY IN GLOMERULAR DISEASES Mirjana Sabljar Matovinović Podocytes are injured in diabetic
How to Cite this article: Podocyte Injury in Glomerular Diseases - ejifcc 20/01 2009 http://www.ifcc.org 3. PODOCYTE INJURY IN GLOMERULAR DISEASES Mirjana Sabljar Matovinović Podocytes are injured in diabetic
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
Nephropathy is a morbid complication of diabetes.
 Rapid Publications Progression of Nephropathy in Spontaneous Diabetic Rats Is Prevented by OPB-9195, a Novel Inhibitor of Advanced Glycation Sakurako Nakamura, Zenji Makita, Shintaro Ishikawa, Koichi Yasumura,
Rapid Publications Progression of Nephropathy in Spontaneous Diabetic Rats Is Prevented by OPB-9195, a Novel Inhibitor of Advanced Glycation Sakurako Nakamura, Zenji Makita, Shintaro Ishikawa, Koichi Yasumura,
Ablation of carbohydrate-responsive element-binding protein improves kidney injury in streptozotocin-induced diabetic mice
 European Review for Medical and Pharmacological Sciences 2017; 21: 42-47 Ablation of carbohydrate-responsive element-binding protein improves kidney injury in streptozotocin-induced diabetic mice W. ZHANG,
European Review for Medical and Pharmacological Sciences 2017; 21: 42-47 Ablation of carbohydrate-responsive element-binding protein improves kidney injury in streptozotocin-induced diabetic mice W. ZHANG,
Michigan/Chicago unit
 Michigan/Chicago unit Modifications in Mouse Models to Enhance Nephropathy/Neuropathy- GLUT1 overexpression Increased oxidative stress Increased glucose metabolic flux or alteration in GLUT expression
Michigan/Chicago unit Modifications in Mouse Models to Enhance Nephropathy/Neuropathy- GLUT1 overexpression Increased oxidative stress Increased glucose metabolic flux or alteration in GLUT expression
Relationship between renal injury and the antagonistic roles of angiotensin-converting enzyme (ACE) and ACE2
 Relationship between renal injury and the antagonistic roles of angiotensin-converting enzyme (ACE) and ACE2 C. Ma, H. Xin, X.-Y. Jiang, Y.-X. Wang and Y.-S. Zhang Key Laboratory of Animal Physiology and
Relationship between renal injury and the antagonistic roles of angiotensin-converting enzyme (ACE) and ACE2 C. Ma, H. Xin, X.-Y. Jiang, Y.-X. Wang and Y.-S. Zhang Key Laboratory of Animal Physiology and
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
The topic of normal vascular and glomerular anatomy is introduced
 Normal Vascular and Glomerular Anatomy Arthur H. Cohen Richard J. Glassock The topic of normal vascular and glomerular anatomy is introduced here to serve as a reference point for later illustrations of
Normal Vascular and Glomerular Anatomy Arthur H. Cohen Richard J. Glassock The topic of normal vascular and glomerular anatomy is introduced here to serve as a reference point for later illustrations of
Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients.
 Supplementary Materials Supplementary Figures Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients. Figure S2. Expression level of podocyte
Supplementary Materials Supplementary Figures Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients. Figure S2. Expression level of podocyte
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the
 Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Mohammad Husain Department of Biotechnology, Jamia Millia Islamia New Delhi
 Role of Vitamin D receptor (VDR) in HIV induced tubular injury Mohammad Husain Department of Biotechnology, Jamia Millia Islamia New Delhi 07/10/2015 INTRODUCTION Vitamin D is technically not a Vitamin;
Role of Vitamin D receptor (VDR) in HIV induced tubular injury Mohammad Husain Department of Biotechnology, Jamia Millia Islamia New Delhi 07/10/2015 INTRODUCTION Vitamin D is technically not a Vitamin;
A Central Role of MG53 in Metabolic Syndrome. and Type-2 Diabetes
 A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Thin basement membrane nephropathy cannot be diagnosed reliably in deparaffinized, formalin-fixed tissue
 Nephrol Dial Transplant (2007) 22: 1228 1232 doi:10.1093/ndt/gfl838 Advance Access publication 3 February 2007 Technical Note Thin basement membrane nephropathy cannot be diagnosed reliably in deparaffinized,
Nephrol Dial Transplant (2007) 22: 1228 1232 doi:10.1093/ndt/gfl838 Advance Access publication 3 February 2007 Technical Note Thin basement membrane nephropathy cannot be diagnosed reliably in deparaffinized,
Familial DDD associated with a gain-of-function mutation in complement C3.
 Familial DDD associated with a gain-of-function mutation in complement C3. Santiago Rodríguez de Córdoba, Centro de investigaciones Biológicas, Madrid Valdés Cañedo F. and Vázquez- Martul E., Complejo
Familial DDD associated with a gain-of-function mutation in complement C3. Santiago Rodríguez de Córdoba, Centro de investigaciones Biológicas, Madrid Valdés Cañedo F. and Vázquez- Martul E., Complejo
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS
 FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
Ανάπτυξη Βιοτράπεζας για την Ανίχνευση Πρώιμων Βιοδεικτών σε Ασθενείς με Χρόνια Νεφρική Νόσο
 Ανάπτυξη Βιοτράπεζας για την Ανίχνευση Πρώιμων Βιοδεικτών σε Ασθενείς με Χρόνια Νεφρική Νόσο ΔΗΜΗΤΡΙΟΣ Σ. ΓΟΥΜΕΝΟΣ Νεφρολογικό και Μεταμοσχευτικό Κέντρο Πανεπιστημιακό Νοσοκομείο Πατρών Causes of chronic
Ανάπτυξη Βιοτράπεζας για την Ανίχνευση Πρώιμων Βιοδεικτών σε Ασθενείς με Χρόνια Νεφρική Νόσο ΔΗΜΗΤΡΙΟΣ Σ. ΓΟΥΜΕΝΟΣ Νεφρολογικό και Μεταμοσχευτικό Κέντρο Πανεπιστημιακό Νοσοκομείο Πατρών Causes of chronic
The use of pathology surrogate markers in Fabry Disease. Beth L. Thurberg MD PhD Vice President of Pathology Genzyme
 Disclaimer: Presentation slides from the Rare Disease Workshop Series are posted by the EveryLife Foundation for Rare Diseases for educational purposes only. They are for use by drug development professionals
Disclaimer: Presentation slides from the Rare Disease Workshop Series are posted by the EveryLife Foundation for Rare Diseases for educational purposes only. They are for use by drug development professionals
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Podocyte Biology and clinical applications Dr. F. Ahmadi Professor Of Nephrology TUMS
 Podocyte Biology and clinical applications Dr. F. Ahmadi Professor Of Nephrology TUMS Proteinuria is a major healthcare problem that affects several hundred million people worldwide. Proteinuria is a cardinal
Podocyte Biology and clinical applications Dr. F. Ahmadi Professor Of Nephrology TUMS Proteinuria is a major healthcare problem that affects several hundred million people worldwide. Proteinuria is a cardinal
Histopathology: Glomerulonephritis and other renal pathology
 Histopathology: Glomerulonephritis and other renal pathology These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you
Histopathology: Glomerulonephritis and other renal pathology These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you
As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the
 3 RESULTS As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the DKFZ in Heidelberg (Dept. of Cellular and Molecular pathology) contributed to this work by performing
3 RESULTS As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the DKFZ in Heidelberg (Dept. of Cellular and Molecular pathology) contributed to this work by performing
Ο ρόλος των τριγλυκεριδίων στην παθογένεια των μικροαγγειοπαθητικών επιπλοκών του σακχαρώδη διαβήτη
 Ο ρόλος των τριγλυκεριδίων στην παθογένεια των μικροαγγειοπαθητικών επιπλοκών του σακχαρώδη διαβήτη Κωνσταντίνος Τζιόμαλος Επίκουρος Καθηγητής Παθολογίας Α Προπαιδευτική Παθολογική Κλινική, Νοσοκομείο
Ο ρόλος των τριγλυκεριδίων στην παθογένεια των μικροαγγειοπαθητικών επιπλοκών του σακχαρώδη διαβήτη Κωνσταντίνος Τζιόμαλος Επίκουρος Καθηγητής Παθολογίας Α Προπαιδευτική Παθολογική Κλινική, Νοσοκομείο
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author):
 Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
CHAPTER 2. Primary Glomerulonephritis
 2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
Dr. Hayam Gad Associate Professor of Physiology College of Medicine King Saud University
 Dr. Hayam Gad Associate Professor of Physiology College of Medicine King Saud University INTRODUCTION Diabetic nephropathy (DN) DN is defined as the appearance of persistent clinical albuminuria in an
Dr. Hayam Gad Associate Professor of Physiology College of Medicine King Saud University INTRODUCTION Diabetic nephropathy (DN) DN is defined as the appearance of persistent clinical albuminuria in an
Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients
 Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients S. Yang, B. Chen, J. Shi, F. Chen, J. Zhang and Z. Sun Department of Nephrology, Huaihe Hospital
Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients S. Yang, B. Chen, J. Shi, F. Chen, J. Zhang and Z. Sun Department of Nephrology, Huaihe Hospital
For more information about how to cite these materials visit
 Author(s): Frank Brosius, M.D, 2011 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Frank Brosius, M.D, 2011 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Supplemental Figure 1
 Supplemental Figure 1 A S100A4: SFLGKRTDEAAFQKLMSNLDSNRDNEVDFQEYCVFLSCIAMMCNEFFEGFPDK Overlap: SF G DE KLM LD N D VDFQEY VFL I M N FF G PD S100A2: SFVGEKVDEEGLKKLMGSLDENSDQQVDFQEYAVFLALITVMCNDFFQGCPDR
Supplemental Figure 1 A S100A4: SFLGKRTDEAAFQKLMSNLDSNRDNEVDFQEYCVFLSCIAMMCNEFFEGFPDK Overlap: SF G DE KLM LD N D VDFQEY VFL I M N FF G PD S100A2: SFVGEKVDEEGLKKLMGSLDENSDQQVDFQEYAVFLALITVMCNDFFQGCPDR
Fine structural appearances of glomerular capillaries in a case of malignant hypertension
 J. clin. Path. (1969), 22, 579-583 Fine structural appearances of glomerular capillaries in a case of malignant hypertension R. F. MACADAM From the University Department of Pathology, Western Infirmary,
J. clin. Path. (1969), 22, 579-583 Fine structural appearances of glomerular capillaries in a case of malignant hypertension R. F. MACADAM From the University Department of Pathology, Western Infirmary,
RENAL HISTOPATHOLOGY FOLLOWING RUSSELL'S VIPER (VIPERA RUSSELLI) BITE
 RENAL HISTOPATHOLOGY FOLLOWING RUSSELL'S VIPER (VIPERA RUSSELLI) BITE Soe ~oe', May Mya win2, Than Than Htwe', Myint win', Swe Swe ~het* and Wynn Wynn Kyawl 'Department of Medical Research No. 5. Ziwaka
RENAL HISTOPATHOLOGY FOLLOWING RUSSELL'S VIPER (VIPERA RUSSELLI) BITE Soe ~oe', May Mya win2, Than Than Htwe', Myint win', Swe Swe ~het* and Wynn Wynn Kyawl 'Department of Medical Research No. 5. Ziwaka
Is the new Mayo Clinic Quadratic (MCQ) equation useful for the estimation of glomerular filtration rate in type 2 diabetic patients?
 Diabetes Care Publish Ahead of Print, published online October 3, 2008 The MCQ equation in DM2 patients Is the new Mayo Clinic Quadratic (MCQ) equation useful for the estimation of glomerular filtration
Diabetes Care Publish Ahead of Print, published online October 3, 2008 The MCQ equation in DM2 patients Is the new Mayo Clinic Quadratic (MCQ) equation useful for the estimation of glomerular filtration
Pathology of Complement Mediated Renal Disease
 Pathology of Complement Mediated Renal Disease Mariam Priya Alexander, MD Associate Professor of Pathology GN Symposium Hong Kong Society of Nephrology July 8 th, 2017 2017 MFMER slide-1 The complement
Pathology of Complement Mediated Renal Disease Mariam Priya Alexander, MD Associate Professor of Pathology GN Symposium Hong Kong Society of Nephrology July 8 th, 2017 2017 MFMER slide-1 The complement
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events
 Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Glomerular diseases mostly presenting with Nephritic syndrome
 Glomerular diseases mostly presenting with Nephritic syndrome 1 The Nephritic Syndrome Pathogenesis: proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls escape of RBCs
Glomerular diseases mostly presenting with Nephritic syndrome 1 The Nephritic Syndrome Pathogenesis: proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls escape of RBCs
Inflammatory basis of diabetic retinopathy. Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH
 Inflammatory basis of diabetic retinopathy Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH Diabetic retinopathy currently is diagnosed based on vascular abnormalities
Inflammatory basis of diabetic retinopathy Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH Diabetic retinopathy currently is diagnosed based on vascular abnormalities
Obese Type 2 Diabetic Model. Rat Model with Early Onset of Diabetic Complications. SDT fatty rats. SDT fatty rats
 Obese Type 2 Diabetic Model rats Rat Model with Early Onset of Diabetic Complications rats Background and Origin In 24, Dr. Masuyama and Dr. Shinohara (Research Laboratories of Torii Pharmaceutical Co.,
Obese Type 2 Diabetic Model rats Rat Model with Early Onset of Diabetic Complications rats Background and Origin In 24, Dr. Masuyama and Dr. Shinohara (Research Laboratories of Torii Pharmaceutical Co.,
CHAPTER I INTRODUCTION. Nowadays chronic kidney disease (CKD) becomes one. of the most common diseases found in the population.
 CHAPTER I INTRODUCTION I.1 Background Nowadays chronic kidney disease (CKD) becomes one of the most common diseases found in the population. Based on community survey that is held by PERNEFRI (Perhimpunan
CHAPTER I INTRODUCTION I.1 Background Nowadays chronic kidney disease (CKD) becomes one of the most common diseases found in the population. Based on community survey that is held by PERNEFRI (Perhimpunan
Renal Pathology 1: Glomerulus. With many thanks to Elizabeth Angus PhD for EM photographs
 Renal Pathology 1: Glomerulus With many thanks to Elizabeth Angus PhD for EM photographs Anatomy of the Kidney http://www.yalemedicalgroup.org/stw/page.asp?pageid=stw028980 The Nephron http://www.beltina.org/health-dictionary/nephron-function-kidney-definition.html
Renal Pathology 1: Glomerulus With many thanks to Elizabeth Angus PhD for EM photographs Anatomy of the Kidney http://www.yalemedicalgroup.org/stw/page.asp?pageid=stw028980 The Nephron http://www.beltina.org/health-dictionary/nephron-function-kidney-definition.html
Dr Ian Roberts Oxford. Oxford Pathology Course 2010 for FRCPath Illustration-Cellular Pathology. Oxford Radcliffe NHS Trust
 Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Present the basic diagnostic features of the commonest conditions causing proteinuria & haematuria Highlight diagnostic pitfalls Nephrotic
Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Present the basic diagnostic features of the commonest conditions causing proteinuria & haematuria Highlight diagnostic pitfalls Nephrotic
Dr. Mehmet Kanbay Department of Medicine Division of Nephrology Istanbul Medeniyet University School of Medicine Istanbul, Turkey.
 The uric acid dilemma: causal risk factor for hypertension and CKD or mere bystander? Mehmet Kanbay, Istanbul, Turkey Chairs: Anton H. van den Meiracker, Rotterdam, The Netherlands Claudia R.C. Van Roeyen,
The uric acid dilemma: causal risk factor for hypertension and CKD or mere bystander? Mehmet Kanbay, Istanbul, Turkey Chairs: Anton H. van den Meiracker, Rotterdam, The Netherlands Claudia R.C. Van Roeyen,
Role of Tyk-2 in Th9 and Th17 cells in allergic asthma
 Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Collin County Community College RENAL PHYSIOLOGY
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
Familial Nephropathy (FN) in [English] Cocker Spaniel Dogs Introduction Clinical signs Laboratory features
![Familial Nephropathy (FN) in [English] Cocker Spaniel Dogs Introduction Clinical signs Laboratory features Familial Nephropathy (FN) in [English] Cocker Spaniel Dogs Introduction Clinical signs Laboratory features](/thumbs/77/75043585.jpg) Familial Nephropathy (FN) in [English] Cocker Spaniel Dogs George E. Lees, DVM, MS, DACVIM; Texas A&M University, USA Email: glees@cvm.tamu.edu Telephone: 979-845-2351 Introduction An inherited renal disease
Familial Nephropathy (FN) in [English] Cocker Spaniel Dogs George E. Lees, DVM, MS, DACVIM; Texas A&M University, USA Email: glees@cvm.tamu.edu Telephone: 979-845-2351 Introduction An inherited renal disease
hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and function in
 SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure 1. Fbn1 C1039G/+ hearts display normal cardiac function in the absence of hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and
SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure 1. Fbn1 C1039G/+ hearts display normal cardiac function in the absence of hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
The CARI Guidelines Caring for Australasians with Renal Impairment. ACE Inhibitor and Angiotensin II Antagonist Combination Treatment GUIDELINES
 ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
References. Plasma renin activity (PRA) PRA was measured by a radioimmunoassay kit (Wallac, Tokyo, Japan).
 Detailed Methods Experiment I enos / mice were purchased from Jackson Laboratory (Bar Harbor, USA). C57BL/6J mice on the same genetic background were purchased from KBT Oriental (Hamamatsu, Japan). Eleven-week-old
Detailed Methods Experiment I enos / mice were purchased from Jackson Laboratory (Bar Harbor, USA). C57BL/6J mice on the same genetic background were purchased from KBT Oriental (Hamamatsu, Japan). Eleven-week-old
The CARI Guidelines Caring for Australians with Renal Impairment. Specific effects of calcium channel blockers in diabetic nephropathy GUIDELINES
 Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
