Department of Nephrology and Medical Intensive Care, Klinikum Charité, Campus Virchow, Humboldt University, Berlin, Germany 3
|
|
|
- Jody Harmon
- 5 years ago
- Views:
Transcription
1 Heterozygous deficiency of hypoxia-inducible factor 2α protects mice against pulmonary hypertension and right ventricular dysfunction during prolonged hypoxia Koen Brusselmans, 1 Veerle Compernolle, 1 Marc Tjwa, 1 Michael S. Wiesener, 2 Patrick H. Maxwell, 3 Désiré Collen, 1 and Peter Carmeliet 1 1 Center for Transgene Technology and Gene Therapy, Flanders Interuniversity Institute for Biotechnology, Katholieke Universiteit Leuven, Leuven, Belgium 2 Department of Nephrology and Medical Intensive Care, Klinikum Charité, Campus Virchow, Humboldt University, Berlin, Germany 3 Renal Section, Imperial College of Science, Technology and Medicine, Hammersmith Campus, London, United Kingdom Chronic hypoxia induces pulmonary vascular remodeling, leading to pulmonary hypertension, right ventricular hypertrophy, and heart failure. Heterozygous deficiency of hypoxia-inducible factor 1α (HIF-1α), which mediates the cellular response to hypoxia by increasing expression of genes involved in erythropoiesis and angiogenesis, has been previously shown to delay hypoxia-induced pulmonary hypertension. HIF-2α is a homologue of HIF-1α and is abundantly expressed in the lung, but its role in pulmonary hypertension remains unknown. Therefore, we analyzed the pulmonary response of WT and viable heterozygous HIF-2α deficient (Hif2α +/ ) mice after exposure to 10% O 2 for 4 weeks. In contrast to WT mice, Hif2α +/ mice were fully protected against pulmonary hypertension and right ventricular hypertrophy, unveiling a critical role of HIF-2α in hypoxia-induced pulmonary vascular remodeling. Pulmonary expression levels of endothelin-1 and plasma catecholamine levels were increased threefold and 12-fold respectively in WT but not in Hif2α +/ mice after hypoxia, suggesting that HIF-2α mediated upregulation of these vasoconstrictors contributes to the development of hypoxic pulmonary vascular remodeling. J. Clin. Invest. 111: (2003). doi: /jci Introduction Pulmonary hypertension is a formidable health problem, as it often leads to right ventricular (RV) hypertrophy and heart failure (1, 2). Current treatment includes the administration of oxygen, bronchodilators, vasodilators (e.g., prostacyclin, NO, and endothelin-1 antagonists), and, eventually, mechanical ventilation (2 4). However, since oxygenation and vasodilatation merely delay the progression of this disease, a better understanding of its pathogenesis is required (2). The pathophysiology of hypoxic pulmonary hypertension is complex and poorly understood. It is characterized by increased levels of the vasoconstrictors angiotensin II and endothelin-1 (ET-1), Received for publication March 21, 2002, and accepted in revised form March 11, Address correspondence to: Peter Carmeliet, Center for Transgene Technology and Gene Therapy, Flanders Interuniversity Institute of Biotechnology, Katholieke Universiteit Leuven, Gasthuisberg, Herestraat 49, B-3000 Leuven, Belgium. Phone: ; Fax: ; peter.carmeliet@med.kuleuven.ac.be. Conflict of interest: The authors have declared that no conflict of interest exists. Nonstandard abbreviations used: right ventricular (RV); endothelin-1 (ET-1); hypoxia-inducible factor 1α (HIF-1α); hypoxia-response element (HRE); internal elastic lamina (IEL); external elastic lamina (EEL); smooth muscle cell (SMC); norepinephrine (NE). impaired production of the vasodilators NO and prostacyclin, and an unbalanced production of factors affecting growth, migration, and differentiation of VSMCs, including FGF-2, PDGF-B, TGF-β, IGF-I and -II, and EGF (5, 6). As a result, peripheral, normally nonmuscular arteries become muscularized, and the media and adventitia enlarge (5 7). Loss of enos (8) or prostacyclin receptor (9) aggravates hypoxic pulmonary vascular disease, whereas ET-1 receptor blockade (4), overexpression of prostacyclin (10), and gene transfer of inos (11) reduce hypoxia-induced pulmonary hypertension. In addition, deficiency of the serotonin transporter, a pulmonary VSMC mitogen that is upregulated during hypoxia, also attenuates hypoxic pulmonary hypertension (12). Furthermore, serine elastase, plasminogen, and MMPs have been implicated in growth and migration of VSMCs via degradation of the ECM and release of mitogens or differentiation factors (13). Proteinases are involved in the pathology of pulmonary hypertension, since mice deficient in plasminogen or urokinase-type plasminogen activator are partially protected against pulmonary vascular remodeling (14). Very recently, gene transfer of VEGF was also shown to reduce pulmonary hypertension in rats (15). Hypoxia-inducible factor 1α (HIF-1α) is a key regulator in the cellular adaptation to hypoxia (16). During hypoxia, HIF-1α upregulates the expression of a number of genes involved in erythropoiesis, glycolysis, and angiogenesis by binding, as a heterodimer with HIF-1β, The Journal of Clinical Investigation May 2003 Volume 111 Number
2 to a hypoxia-response element (HRE) in the promoter of these target genes (16, 17). Loss of HIF-1α or HIF-1β impaired gene expression in response to hypoxia and/or hypoglycemia and caused embryonic lethality around embryonic day 10.5 (16, 17). Recently, a novel homologue, HIF-2α (also known as EPAS1 [ref. 18], HLF [ref. 19], or HRF [ref. 20]), was identified, which also binds as a heterodimer with HIF-1β to the HRE in the promoter of target genes. Gene-inactivation studies revealed a role of HIF-2α in cardiovascular development and angiogenesis in the embryo (21, 22), but its role in adult pathologies remains unknown. HIF-1α was recently demonstrated to be involved in the pulmonary response to chronic hypoxia, since pulmonary hypertension was delayed in heterozygous deficient Hif1α +/ mice (23). In addition, Hif1α +/ pulmonary arterial myocytes showed impaired electrophysiological responses to chronic hypoxia (24). Although HIF-2α is abundantly expressed in the lung (19, 20, 25), its role in pulmonary hypertension has thus far not been studied. We previously inactivated the Hif2α gene in embryonic stem cells (26) and used them to generate transgenic mice (25). Since homozygous deficient Hif2α / mice died during gestation or immediately after birth (21, 22, 25), viable heterozygous Hif2α +/ mice were used in the present study to analyze the role of HIF-2α during pulmonary hypertension and vascular remodeling. Methods Animal protocol. Animal experiments were approved by the institutional review board and were performed as previously described (14), according to the guidelines for animal experiments of the NIH. Eight-week-old mice (littermates; mixed-background Swiss/129Sv) were weighed and placed in a tightly sealed chamber under normobaric hypoxia (10% O 2 ), which was maintained by a continuous inflow of 2 l/min N 2 and 2 l/min normal air (21% O 2 ). Control mice were kept in normal air (21% O 2 ). After exposure to hypoxia for the indicated period, mice were weighed and immediately used for determination of RV hypertrophy, hematocrit, plasma catecholamine levels, gene expression, and histology. For the hemodynamic measurements, mice were first equilibrated to room air for 1 hour. Hemodynamic measurements after exposure to chronic hypoxia. Hemodynamic measurements were performed as previously described (14). Mice were first equilibrated by returning them to room air for 1 hour, to avoid acute vasomotor responses (11, 23), and were then anesthetized with urethane (1.4 mg/kg). While the mice were ventilated (Minivent 845; Hugo Sachs Elektronik, March-Hugstetten, Germany; 150 strokes per minute, volume 250 µl), RV pressures were measured via transthoracic puncture using a 1.4F high-fidelity pressure micromanometer (SPR-671; Millar Instruments Inc., Houston, Texas, USA) at controlled normal body temperatures. Manometer reference settings were calibrated before each measurement. Correct manometer position in the right ventricle was verified by pressure readings and confirmed by postmortem examination. After hemodynamic stabilization, pressure measurements were amplified (Siemens Pressure Amplifier 863; Siemens-Elema AB, Solna, Sweden) and unfiltered digitalized (Dataq DA Convert DI-205; Dataq Instruments Inc., Akron, Ohio, USA) (sampling rate 2,000 Hz). Digital files were recorded and analyzed (WinDaq software; Dataq Instruments Inc.) as previously described (14). For each measurement, an average of ten sequential beats was calculated. Contractile performance indices included maximal isovolumetric rate of development of ventricular pressure (dp/dt max ) and contractility (dp/dt max P). We calculated τ, the time constant of ventricular relaxation during isovolumic diastole, by fitting the decay of RV pressure (from dp/dt min ) to a monoexponential equation assuming a zero asymptote: P(t) = P(0).e t/τ (27). Functional assessment of the right ventricle in the setting of chronic pulmonary hypertension was performed as described previously (28). Hemodynamic responses of normoxic mice to acute hypoxia and to agonists. Normoxic mice were anesthetized with urethane, ventilated, and maintained at controlled normal body temperature. Calibration of 1.4F micromanometer reference settings was performed before each measurement. Introduction of the manometer was performed as previously described (29). Briefly, the pressure catheter was introduced into the RV cavity via the right jugular vein. Continuous recording of pressure parameters was performed during initial ventilation with room air (15 minutes), and during ventilation with a hypoxic gas mixture (7% O 2, 93% N 2 ; 20 minutes) or after agonist administration. Acute hypoxic vasoconstriction was defined as the change in RV systolic pressure, expressed as percentage of base-line. The vasodepressor response was evaluated as previously described (30). Briefly, vasoconstriction was induced by i.v. injection of prostaglandin F 2α (0.3 µg/kg). After 3 minutes, bradykinin (1 µg/kg) was injected i.v., and vasodepressor responses, defined as the RV systolic pressure at injection of bradykinin minus the RV systolic pressure 3 minutes later, were measured. Hematocrit and plasma catecholamine analysis. Blood samples were collected from the vena cava, and hematocrit was analyzed using an automated cell counter (Cell-Dyn 1330; Abbott Laboratories, Abbott Park, Illinois, USA) (14). Plasma norepinephrine levels were quantified by HPLC in the University Hospital Laboratory (Katholieke Universiteit Leuven, Leuven, Belgium) as previously described (22). RV hypertrophy measurement. Hearts were dissected, and the RV wall was removed from the left ventricle and septum after removal of the atria (14). Ventricles were dried at 55 C (until weight difference between two consecutive days was less than 0.1 mg) and weighed. Results were expressed as the ratio of right ventricle weight to left ventricle plus septum weight [RV/(LV+S)] or as the ratio of right ventricle weight to total body weight (RV/B) The Journal of Clinical Investigation May 2003 Volume 111 Number 10
3 Histological analysis. As described previously (14), we performed tissue preparations; Hart s elastin staining, for visualization of internal and external elastic lamina (IEL and EEL); staining of smooth muscle cells (SMCs) with mouse antibodies against murine SMC α-actin; and staining of endothelial cells with rabbit antibodies against thrombomodulin. To assess pulmonary vascular remodeling, we counted the number of nonmuscularized (only IEL), partially muscularized (IEL plus incomplete EEL), and fully muscularized (IEL and complete EEL) peripheral vessels that were located distally to the bronchi. Vessel numbers were quantified using the Quantimet Q600 imaging system (Leica Imaging Systems Ltd., Cambridge, United Kingdom) and expressed per 100 alveoli (14, 31). Wall thickness of completely muscularized pulmonary arterioles (diameter less than 80 µm) was calculated as the percentage of the diameter of the EEL minus the diameter of the IEL divided by the diameter of the EEL (14, 31). Capillary density and intercapillary distance were measured as previously described (14), using the Quantimet Q600 imaging system. Gene expression analysis. Protein extraction and Western blot analysis were performed as previously described (17, 26). Briefly, frozen tissues were homogenized in extraction buffer, and extracts were quantified with the Bio- Rad DC protein assay (Bio-Rad Laboratories GmbH, Munich, Germany). Fifty micrograms of total protein was separated by 6% SDS-PAGE, transferred onto Immobilon-P membranes (Millipore Corp., Bedford, Massachusetts, USA), and incubated with antibodies against enos (Calbiochem-Novabiochem Corp., San Diego, California, USA) or against HIF-1α or HIF-2α (previously generated [ref. 32]). Detection was performed by chemiluminescence (SuperSignal West Dura; Pierce Chemical Co., Rockford, Illinois, USA) with HRP-conjugated secondary antibodies. ET-1 protein levels were determined by ELISA (Immuno-Biological Laboratories Hamburg, Hamburg, Germany). RNA extractions from lungs and hearts and TaqMan quantitative real-time RT-PCR (Applied Biosystems, Lennik, Belgium) were performed as previously described (26). Gene expression was determined as the amount of mrna copies per 100 mrna copies of the HPRT gene (hypoxanthine-guanine phosphoribosyl transferase), which was used as an internal control. We used the following forward (F) and reverse (R) primers, and probes (P) labeled with fluorescent dyes (FAM or JOE) and quenchers (TAMRA): for HPRT, F, 5 -TTATCAGACTGAAGAGCTACTGTAATGATC- 3, R, 5 -TTACCAGTGTCAATTATATCTTCAACAATC-3, and P, 5 -JOE-TGAGAGATCATCTCCACCAATAACTTTTATGTC- CC-TAMRA-3 ; for enos, F, 5 -AGCCCGGGACTTCAT- CAATC-3, R, 5 -TGAAGCCGCTGCTCATGAG-3, and P, 5 - FAM-ACTATAACTCCATCAAAAGGAGTGGCTCCCAG-TAM RA-3 ; for ET-1, F, 5 -CTTCTGCCACCTGGACATCA-3, R, 5 -CTCCCAGTCCATACGGTACGA-3, and P, 5 -FAM- CTGGGTCAACACTCCCGAGCGC-TAMRA-3 ; for PDGF-B, F, 5 -CGGTCCAGGTGAGAAAGATTG-3, R, 5 -CGTCTTG- GCTCGCTGCTC-3, and P, 5 -FAM-CCCATCTTCAAGAAG- GCCACAGTGACCT-TAMRA-3 ; for HIF-1α, F, 5 -TG- AGCTCACATCTTGATAAAGCTTCT-3, R, 5 -GGGCTTT- CAGATAAAAACAGTCCAT-3, and P, 5 -FAM-AGACCACCG- GCATCCAGAAGTTTTCTCA-TAMRA-3, for HIF-2α, F, 5 -CCTGGCCATCAGCTT-3, R, 5 -CTGGTCGGCCTCA- GCTTC-3, and P, 5 -FAM-CACATAAGCTCCTCAGTCT- GCTCTGA-TAMRA-3. Statistical analysis. Data are expressed as mean ± SEM. Comparisons of values were made using a nonparametric Mann-Whitney U test. Results Generation of heterozygous HIF-2α deficient (Hif2α +/ ) mice. To study the role of HIF-2α in vivo, we previously inactivated the HIF-2α gene in embryonic stem cells by homologous recombination (26) with deletion of the second exon, encoding the basic helixloop-helix domain, which is essential for DNA binding and dimerization (16, 18, 22). Transgenic mice lacking a single HIF-2α allele (Hif2α +/ ) were generated using diploid embryo aggregation and subsequent breeding of high-percentage chimeras as previously described (25). Hif2α +/ mice were viable and healthy and, upon macroscopic and microscopic analysis, did not exhibit obvious vascular abnormalities in the heart (see below) and kidneys (afferent and efferent arterioles per longitudinal kidney section: 170 ± 4 in HIF-2α +/+ mice hereafter referred to as WT mice and 168 ± 10 in Hif2α +/ mice; n = 3 5; P = NS). Half of the Hif2α / embryos died during gestation, while the remaining Hif2α / mice died at birth, consistent with previous reports (21, 22, 25). Expression of HIF-1α and HIF-2α was analyzed in WT and Hif2α +/ mice. HIF-2α transcript levels, expressed as mrna copy number per 100 HPRT mrna copies, were reduced by 50% in Hif2α +/ mice in the lungs (23 ± 4 and 20 ± 2 in WT mice vs. 12 ± 2 and 11 ± 1 in Hif2α +/ mice in normoxia and hypoxia, respectively; n = 6; P < 0.05) and in the heart (3.3 ± 0.7 and 3.0 ± 0.5 in WT mice vs. 1.7 ± 0.3 and 1.6 ± 0.4 in Hif2α +/ mice in normoxia and hypoxia, respectively; n = 6; P < 0.05). Heterozygous loss of HIF-2α was not compensated by HIF-1α, since HIF-1α mrna levels (mrna copy number per 100 HPRT mrna copies) were comparable in both genotypes in the lungs (16 ± 2 and 12 ± 1 in WT mice vs. 13 ± 1 and 13 ± 3 in Hif2α +/ mice in normoxia and hypoxia, respectively; n = 6; P = NS) and in the heart (55 ± 3 and 39 ± 5 in WT mice vs. 48 ± 9 and 44 ± 3 in Hif2α +/ mice in normoxia and hypoxia, respectively; n = 6; P = NS). Western blot analysis revealed that both normoxic and hypoxic pulmonary HIF-2α protein levels were significantly decreased in Hif2α +/ mice compared with WT mice (Figure 1a). HIF-1α protein was undetectable in both normoxic and hypoxic lungs (data not shown), consistent with previous reports (23). Hif2α +/ mice survive long-term exposure to severe hypoxia. To study the role of HIF-2α in the response to chronic hypoxia, 8-week-old WT and Hif2α +/ littermates were The Journal of Clinical Investigation May 2003 Volume 111 Number
4 Figure 1 (a) Western blot analysis showed that pulmonary HIF-2α protein levels were decreased in Hif2α +/ mice compared with WT mice after normoxia (N) and after 1 week of hypoxia (H). Extracts from PC12 cells, exposed to normoxia or hypoxia (1% oxygen, 4 hours) as previously described (17, 26), were used as controls. (b) Systolic RV pressures in WT and Hif2α +/ mice under normoxia or after hypoxic exposure. Hemodynamic analysis revealed no differences in RV pressure under normoxia; however, WT mice showed increased RV pressure after 4 weeks of hypoxia, but Hif2α +/ mice did not. (c) RV hypertrophy analysis in WT and Hif2α +/ mice revealed no differences under normoxic conditions. After hypoxic exposure for 4 weeks, the ratio of the mass of the right ventricle to the mass of the left ventricle plus septum [RV/(LV+S)] was increased in WT mice but not in Hif2α +/ mice. (d) Pulmonary ET-1 transcript levels were increased in WT mice after exposure to chronic hypoxia for 6 days and were still elevated after 4 weeks of hypoxia. In contrast, chronic hypoxia failed to induce ET-1 expression in lungs of Hif2α +/ mice. Data represent means ± SEM of systolic RV pressure (mmhg; n = 4 7) (b), of the ratio of the mass of the right ventricle to the mass of the left ventricle plus septum [RV/(LV+S); n = 6 7] (c), and of the ET-1 mrna copy number per 100 copies of HPRT (n = 5 6) (d). *Statistically significant (P < 0.05) vs. control (WT, normoxia). housed under normobaric hypoxia (10% O 2 ) for 4 weeks. Control groups of both genotypes were maintained in normal air (21% O 2 ). At the start of the experiment, mice of both genotypes appeared healthy, actively moved in their cages, and had comparable body weights (about 26 g). After 4 weeks in normoxia, WT mice gained more weight than Hif2α +/ mice (22% ± 3% for WT mice vs. 10% ± 3% for Hif2α +/ mice; n = 6 8; P = 0.04). When exposed to hypoxia, most WT mice became immobile and lost 29% ± 2% of their body weight, but none of the mice died after 4 weeks of hypoxia. In contrast, Hif2α +/ mice did not exhibit any signs of discomfort, behaved normally, moved around actively, and lost only 16% ± 2% of their body weight after 4 weeks of hypoxia (P < vs. WT mice; n = 15 17). Hif2α +/ mice are protected against pulmonary hypertension. No genotypic differences in systolic RV pressure were observed in normoxia (Figure 1b). Chronic hypoxia increased the systolic RV pressure in WT mice but, remarkably, not in Hif2α +/ mice (Figure 1b). Additional hemodynamic measurements demonstrated that exposure of normoxic mice to acute hypoxia (7% O 2 ) for 20 minutes caused a similar increase in systolic RV pressure in WT mice (25% ± 9%; n = 5) and Hif2α +/ mice (29% ± 7%; n = 4;P = NS). In addition, bradykinin-induced vasodepressor responses were also comparable in WT and Hif2α +/ mice (2.0 ± 0.2 mmhg in WT mice vs. 2.2 ± 0.2 mmhg in Hif2α +/ mice; n = 4;P = NS). These data indicate that acute vasoconstriction and vasodepressor responses were comparable in WT and Hif2α +/ mice and that protection of Hif2α +/ mice against pulmonary hypertension was not attributable to differences in acute vasomotor responses. Taken together, these results suggest that any possible additional increase of RV pressure due to hypoxia-induced vasoconstriction may indeed have also contributed to the RV hypertrophy in WT mice but had, obviously, no effect on pulmonary vascular remodeling and RV hypertrophy in Hif2α +/ mice The Journal of Clinical Investigation May 2003 Volume 111 Number 10
5 RV contractility, measured by the maximal dp/dt corrected for the RV pressure (dp/dt max P), was comparable in both genotypes in normoxia (180 ± 32 s 1 in WT mice vs. 160 ± 29 s 1 in Hif2α +/ mice; n = 4 5; P = NS). After 4 weeks of hypoxia, RV contractility tended to be impaired in WT mice (110 ± 27 s 1 ; n = 4; P = NS vs. normoxic WT mice), while it was improved in Hif2α +/ mice (290 ± 41 s 1 ; n = 6; P < 0.02 vs. normoxic WT and Hif2α +/ mice). RV diastolic function, evaluated by measuring the relaxation time constant τ, was comparable in both genotypes in normoxia (τ = 9 ± 1 ms in WT mice vs. 7 ± 1 ms in Hif2α +/ mice; n = 4 5; P = NS). In contrast, after 4 weeks of hypoxia, RV relaxation was significantly impaired in WT but not in Hif2α +/ mice (τ = 17 ± 5 ms in WT mice vs. 6 ± 1 ms in Hif2α +/ mice; n = 4 6; P = 0.01). Heart rates were comparable in WT and Hif2α +/ mice in normoxia and after exposure to hypoxia (data not shown). Hif2α +/ mice are protected against RV hypertrophy. To evaluate RV hypertrophy, the weights of the RV and of the left ventricle plus septum (LV+S) were determined. No genotypic differences were observed in the RV/(LV+S) weight ratio in normoxia (Figure 1c). After 4 weeks of hypoxia, the RV/(LV+S) weight ratio increased in WT but not in Hif2α +/ mice (Figure 1c). Similar findings were obtained when the RV weight was normalized for the total body weight (B). Whereas no differences in the RV/B weight ratio were observed in normoxia (0.25 ± 0.02 mg/g in WT mice vs ± 0.01 mg/g in Hif2α +/ mice; n = 7 10; P = NS), the RV/B weight ratio was higher in WT than in Hif2α +/ mice after hypoxia (0.44 ± 0.03 mg/g in WT mice vs ± 0.02 mg/g in Hif2α +/ mice; n = 6 7; P = 0.001). As a result of the hypoxia-induced RV hypertrophy in WT mice, significant changes in the coronary vasculature developed in the RV myocardial wall: the intercapillary distance enlarged (11 ± 0.3 µm in normoxia vs. 14 ± 0.1 µm in hypoxia; n = 5; P < 0.05), while the capillary density decreased (number of capillaries/mm 2 : 7,200 ± 240 in normoxia vs. 5,600 ± 390 in hypoxia; n = 5; P < 0.05). Since there was no compensatory angiogenesis, as evidenced by the normal capillary/cardiomyocyte ratio (1.2 ± 0.08 in normoxia vs. 1.1 ± 0.04 in hypoxia; n = 5; P = NS), the hypertrophic cardiomyocytes may have suffered some degree of ischemia. In contrast, no such signs of vascular insufficiency were detectable in Hif2α +/ mice (intercapillary distance: 12 ± 0.3 µm in normoxia vs. 11 ± 0.3 µm in hypoxia; capillaries/mm 2 : 7,000 ± 310 in normoxia vs. 7,600 ± 410 in hypoxia; capillary/cardiomyocyte ratio: 1.1 ± 0.05 in normoxia vs. 1.2 ± 0.08 in hypoxia; n = 5; P = NS). Lack of pulmonary vascular remodeling in Hif2α +/ mice. Since pulmonary hypertension is often associated with vascular remodeling of pulmonary arteries (5, 6), we investigated whether HIF-2α affected pulmonary vascular remodeling. Counting of vessels at the level of alveoli and alveolar ducts after elastin staining revealed that the densities of nonmuscularized vessels (containing only an IEL) or partially muscularized vessels (containing an IEL plus an incomplete EEL) were comparable in WT and Hif2α +/ mice in normoxia (Table 1). After 4 weeks of hypoxia, WT mice had more thick-walled muscularized vessels containing an IEL and a complete EEL (Table 1). In contrast, vascular remodeling failed to occur in hypoxic Hif2α +/ mice, in which the fraction of muscularized vessels with an IEL and a complete EEL was comparable to that of normoxic WT and Hif2α +/ mice (Table 1; Figure 2, a e). The wall thickness of completely muscularized pulmonary arterioles, measured as the percentage of the EEL diameter minus the IEL diameter divided by the EEL diameter, was comparable in both genotypes in normoxia (7.6% ± 0.5% in WT mice vs. 8.0% ± 0.5% in Hif2α +/ mice; n = 5; P = NS). Hypoxia increased the wall thickness of completely muscularized arterioles in WT mice but not in Hif2α +/ mice (16% ± 0.4% in WT mice vs. 7.4% ± 0.2% in Hif2α +/ mice; n = 5; P = 0.02). The genotypic differences in vascular remodeling were confirmed by immunostaining for SMC α-actin on lung sections. Compared with Hif2α +/ mice, WT mice contained significantly more fully muscularized vessels that were completely surrounded by SMCs after hypoxic exposure. In Hif2α +/ mice, most of the pulmonary vessels were only partially surrounded by SMCs after hypoxic exposure (Table 1; Figure 2, f j). Normal hematocrit levels in Hif2α +/ mice. Hematocrit levels were comparable in WT and Hif2α +/ mice in normoxia (36% ± 1% in WT mice vs. 34% ± 1% in Hif2α +/ Table 1 Hypoxia-induced pulmonary vascular remodeling Normoxia Hypoxia WT Hif2α +/ WT Hif2α +/ Single IEL 1.9 ± ± ± 0.06 A 1.9 ± 0.07 IEL + incomplete EEL 0.98 ± ± ± ± 0.09 IEL + complete EEL 0.19 ± ± ± 0.11 A 0.17 ± 0.02 Total 3.0 ± ± ± ± 0.13 Partial SMC coverage 1.3 ± ± ± ± 0.04 Complete SMC coverage 0.65 ± ± ± 0.09 A 0.66 ± 0.02 The data represent means ± SEM (n = 6) of the number of vessels per 100 alveoli that contained a single IEL (nonmuscularized), an IEL and an incomplete EEL (partially muscularized), or an IEL and a complete EEL (fully muscularized), and means ± SEM (n = 6) of the number of vessels per 100 alveoli that were partially covered by SMCs (partially muscularized) or completely covered by SMCs (fully muscularized). A Statistically significant (P < 0.05) vs. control (WT, normoxia). The Journal of Clinical Investigation May 2003 Volume 111 Number
6 Figure 2 (a e) Hart s elastin staining revealed the presence of vessels, located distally to the bronchi, at the level of alveoli and alveolar ducts, that contained only an IEL (or an IEL plus an incomplete EEL) (arrows) in lungs of normoxic (N) WT (a) and Hif2α +/ mice (b). Lungs of hypoxic (H) WT mice showed the presence of thick-walled vessels containing both an IEL and a complete EEL (arrows) (c and d), whereas no hypoxia-induced vascular remodeling occurred in Hif2α +/ mice (arrows) (e). (f j) SMC α-actin staining shows the presence of partially muscularized peripheral vessels (arrows) in lungs of normoxic WT (f) and Hif2α +/ mice (g). Chronic hypoxia caused pulmonary vascular remodeling in WT mice, as revealed by the presence of fully muscularized vessels (arrows) (h and i), but not in Hif2α +/ mice (arrows) (j). Bar = 50 µm in all panels. mice; n = 6; P = NS). After 4 weeks of hypoxia, hematocrit levels were comparably increased in both genotypes (46% ± 1% in 12 WT mice vs. 46% ± 1% in 16 Hif2α +/ mice; P = NS). These findings indicate that the reduced pulmonary hypertension in Hif2α +/ mice was not attributable to a lower vascular resistance caused by reduced erythropoiesis in hypoxia. Role of HIF-2α in pulmonary and cardiac gene expression. ET-1 may contribute to pulmonary vasoconstriction and pulmonary vascular remodeling by stimulating growth and proliferation of VSMCs (33), while ET-1 receptor blockade ameliorates pulmonary hypertension (4). Moreover, the promoter of the ET-1 gene harbors a HIF-binding site, and ET-1 is upregulated by hypoxia in vitro and in hypoxic lungs in vivo (4, 16). Therefore, pulmonary expression of ET-1 was determined by quantitative RT-PCR. No genotypic differences were observed in pulmonary ET-1 transcript levels in normoxia. After 6 days and 4 weeks of hypoxia, pulmonary ET-1 transcript levels were increased in WT but not in Hif2α +/ mice (Figure 1d). Measurements of ET-1 protein levels by ELISA confirmed that hypoxic upregulation of pulmonary ET-1 protein was impaired in Hif2α +/ mice (pg ET-1/mg protein in normoxia and hypoxia, respectively: 10 ± 2 and 31 ± 2 in WT mice vs. 11 ± 3 and 19 ± 3 in Hif2α +/ mice; n = 4; P < 0.05 in hypoxia). Because NO has a critical role in vasodilation of pulmonary vessels and inhibits VSMC growth (34), we also determined enos expression in the lungs. Pulmonary enos transcript levels were comparable in both genotypes under normoxia and after 6 days of hypoxia (mrna copy number per 100 copies of HPRT mrna in normoxia and hypoxia, respectively: 4.3 ± 0.5 and 3.7 ± 0.1 in WT mice vs. 3.2 ± 0.4 and 3.1 ± 0.4 in Hif2α +/ mice; n = 6; P = NS). Western blot analysis (not shown) revealed that pulmonary enos protein was increased by hypoxia in WT mice (consistent with previous findings; ref. 35); pulmonary enos protein was upregulated to the same degree in Hif2α +/ mice (data not shown), indicating that pulmonary enos expression in hypoxia is not regulated by HIF-2α. ET-1 has also been implicated in cardiac hypertrophy (36). Cardiac ET-1 transcript levels were comparable under normoxia (mrna copy number per 100 copies of HPRT mrna: 90 ± 10 in WT mice vs. 100 ± 10 in Hif2α +/ mice; n = 6; P = NS) but were upregulated to a higher level in WT than in Hif2α +/ mice after 4 weeks of hypoxia (240 ± 20 in WT mice vs. 160 ± 30 in Hif2α +/ mice; n =5; P < 0.05). ELISA measurements demonstrated that hypoxic upregulation of cardiac ET-1 protein was also impaired in Hif2α +/ mice (normoxia: 3.9 ± 0.3 pg/mg protein in WT mice vs. 4.4 ± 1.4 pg/mg protein in Hif2α +/ mice; n = 4; P = NS; hypoxia: 7.1 ± 0.3 pg/mg protein in WT mice vs. 5.1 ± 1.5 pg/mg protein in Hif2α +/ mice; n = 4; P < 0.05). Cardiac PDGF-B expression was also elevated by hypoxia in WT but not in Hif2α +/ mice (mrna copy number per 100 copies of HPRT mrna in normoxia: 0.5 ± 0.1 in WT mice vs. 0.5 ± 0.1 in Hif2α +/ mice; n = 6; P = NS; mrna copy number per 100 copies of HPRT mrna in hypoxia: 1.2 ± 0.3 in WT mice vs. 0.5 ± 0.1 in Hif2α +/ mice; n = 5; P < 0.05). Role of HIF-2α in catecholamine homeostasis. Expression of tyrosine hydroxylase, an essential gene for catecholamine biosynthesis, is upregulated by hypoxia (16). In addition, plasma catecholamines are increased during hypoxia and in patients with chronic obstructive pulmonary disease and/or pulmonary hypertension (37, 38). Since loss of HIF-2α impairs fetal catecholamine production (22) and norepinephrine (NE) stimulates vasoconstriction of isolated pulmonary arteries (39) and pulmonary microvessels in rat lung explants (40), plasma NE levels were determined. Under normoxia, plasma NE levels were comparable in both genotypes (0.5 ± 0.1 ng/ml in WT mice vs. 0.6 ± 0.1 ng/ml in Hif2α +/ mice; n = 5; P = NS). Chronic hypoxia for 4 weeks elevated plasma NE 12-fold in WT mice but only 3.5-fold in Hif2α +/ mice (5.8 ± 1.5 ng/ml in WT mice vs. 2.2 ± 0.5 ng/ml in Hif2α +/ mice; n = 5; P = 0.022). After 4 days of hypoxia, 1524 The Journal of Clinical Investigation May 2003 Volume 111 Number 10
7 when no pulmonary vascular remodeling was observed in WT mice (data not shown), plasma NE levels were already increased 3.5-fold in WT mice (1.8 ± 0.3 ng/ml), suggesting that the increased plasma NE levels in WT mice occurred prior to pulmonary vascular remodeling. Discussion The principal finding of this study is that heterozygous deficiency of HIF-2α protects mice against pulmonary hypertension, RV hypertrophy, and dysfunction upon exposure to prolonged hypoxia. These findings unveil a critical role for HIF-2α in pulmonary vascular remodeling, possibly by upregulation of ET-1 and NE. Pulmonary vascular remodeling. Hypoxia caused a significant increase in neomuscularization of the pulmonary resistance vessels in WT mice, as evidenced by the increased fraction of pulmonary vessels that were completely covered by SMCs and contained both an IEL and a complete EEL. Since pulmonary veins exhibit minimal structural changes during chronic hypoxia (31), the increase in thick-walled pulmonary vessels likely corresponded to an increase in muscularized arterioles. Remarkably, vessel muscularization in Hif2α +/ mice after hypoxia was comparable to that in Hif2α +/ or WT mice after normoxia. Similar to heterozygous deficient Hif1α +/ mice (23), heterozygous deficient Hif2α +/ mice were also protected against pulmonary vascular remodeling. Although our data demonstrate that pulmonary hypertension during chronic hypoxia is mediated by HIF-2α, it remains to be determined whether the vascular remodeling is the result of a direct effect of HIF-2α in the vessel wall or is caused, indirectly, by a HIF-2α mediated pressure increase in the pulmonary arterioles. Another intriguing question is why the reduced HIF-2α levels in Hif2α +/ mice were sufficient to sustain embryonic development but not to induce pulmonary hypertension, suggesting that different thresholds of HIF-2α expression may be required for distinct biological processes. A similar phenomenon has been previously observed in angiogenesis, where placental growth factor seems to affect pathological angiogenesis but not vascular development (41). Pulmonary vascular remodeling is a complex, poorly understood process. Pulmonary vessels react to local hypoxia with constriction via upregulation of vasoactive substances as part of a natural response to normalize the oxygenation/perfusion ratio. When hypoxia is sustained and generalized, pulmonary vessels undergo significant structural changes involving neomuscularization and medial thickening of terminal bronchiolar and alveolar duct arterioles. Inhibitors of NAPDH oxidoreductases, which block HIF-1α (16), reduce pulmonary vasoconstriction (42), suggesting that hypoxia-induced pulmonary vasoconstriction may be mediated by HIFs. Furthermore, pulmonary expression levels of ET-1, a wellknown HIF-1α target gene (16), are increased in patients with pulmonary hypertension (4, 7), and inhibition of ET-1 or its receptor prevents hypoxiainduced pulmonary hypertension and vascular remodeling (4). By causing vasoconstriction, ET-1 may initially increase the shear stress and initiate pulmonary vascular remodeling (7). Since ET-1 is known to stimulate growth of VSMCs (4, 33), it may also contribute to the muscularization of pulmonary vessels during hypoxia. As pulmonary ET-1 levels were markedly upregulated during hypoxia in WT but not in Hif2α +/ mice, HIF-2α may regulate the development of pulmonary hypertension at least in part via upregulation of ET-1. While ET-1 is a well-known target gene of HIF-1α (16), our findings now demonstrate that expression of ET-1 is also inducible by HIF-2α. This is not entirely surprising, since both HIF-1α and HIF-2α bind to the same HRE in the ET-1 promoter (18, 19) and HIF-2α is abundantly expressed in the lung (19, 20). Whereas our findings suggest an involvement of HIF-2α in pulmonary hypertension by regulation of ET-1 expression, they obviously do not exclude that HIF-2α regulates the expression of additional vasoactive substances. RV heart hypertrophy and dysfunction. Pulmonary hypertension leads to RV hypertrophy and dysfunction, ultimately progressing to RV failure (cor pulmonale) (1, 2). When WT mice were exposed for prolonged periods to hypoxia, RV pressures were significantly increased and caused RV hypertrophy. RV pressures might even have been underestimated, as they were measured after equilibration of the mice to room air for 1 hour, which likely reduced or eliminated a hypoxia-induced vasoconstrictor response. WT mice also suffered signs of systolic and diastolic RV dysfunction, likely representing an insufficient myocardial adaptation to the prolonged increase in hemodynamic stress. In contrast, RV diastolic function was preserved, while RV systolic function improved in hypoxic Hif2α +/ mice as compared with normoxic mice. This is remarkable since RV loading remained normal in hypoxic mutant mice, as suggested by the lack of remodeling of pulmonary vascular resistance vessels and the normal RV pressures. Possibly, cardiac function is enhanced in these mice to compensate for the hypoxemia and impaired oxygenation of peripheral tissues when maintained for prolonged periods in a low-oxygen environment. Expression of ET-1 and PDGF-B, previously implicated in cardiac hypertrophy (36, 43), was increased in hypertrophic hearts of hypoxic WT mice, suggesting that these molecules may have contributed, at least in part, to the hypertension-induced cardiomyocyte hypertrophy. In addition, ET-1 is known to induce hypertrophy with enhanced expression of muscle-specific genes in cultured rat cardiomyocytes in vitro (36). While HIF-2α could directly stimulate ET-1 gene transcription, no HIF-binding site has thus far been identified in the PDGF-B promoter, suggesting that upregulation of PDGF-B occurs indirectly. However, the increased cardiac expression of ET-1 and/or PDGF-B may also have been a direct effect of the increased RV pressure. The Journal of Clinical Investigation May 2003 Volume 111 Number
8 Catecholamine homeostasis. HIF-2α has been implicated in the regulation of catecholamine biosynthesis during development (22), but a possible role during adult life has not been reported thus far. Hypoxia upregulated plasma NE levels 12-fold in WT mice but only 3.5- fold in Hif2α +/ mice. Plasma NE levels are upregulated in patients with chronic obstructive pulmonary disease and/or pulmonary hypertension (37, 38). Since NE stimulates vasoconstriction of isolated pulmonary arteries (39) and of pulmonary microvessels in adult rat lung explants (40), and since NE is also known to stimulate growth of pulmonary SMCs (44), the impaired upregulation of NE under hypoxic conditions may also have contributed to the impaired pulmonary vascular remodeling and hypertension in Hif2α +/ mice. Although we cannot exclude that NE levels were upregulated due to an increase in pulmonary pressure, plasma NE levels were already elevated after 4 days of hypoxia in WT mice, i.e., prior to onset of significant pulmonary vascular remodeling. The observation that both WT and Hif2α +/ mice had a similar increase in systolic RV pressure during acute hypoxia indicates that the acute hypoxic vasoconstriction response is unlikely to be mediated by HIF-2α. Instead, HIF-2α is essential for upregulation of ET-1 and NE during chronic hypoxic conditions and thereby mediates SMC proliferation and pulmonary vascular remodeling. Hematocrit. Yu et al. documented a delay in the development of polycythemia in Hif1α +/ mice (23). We did not observe any difference in hematocrit levels in Hif2α +/ mice after 4 weeks of hypoxia. Possibly, hypoxic induction of erythropoietin expression might be more dependent on HIF-1α than on HIF-2α, at least under the present conditions. This contrasts with the significant role of HIF-2α in the hypoxic upregulation of ET-1 and NE and in the induction of pulmonary hypertension and vascular remodeling. While we cannot exclude that partial loss of HIF-2α might have delayed polycythemia in the initial stage, the comparable hematocrit levels in combination with the impaired vascular remodeling in Hif2α +/ mice after 4 weeks of hypoxia suggest that the lower vascular resistance in these mice was not attributable to the reduced hematocrit levels. Weight loss. Both genotypes had comparable body weights at the start of the experiment (about 26 g). Changes in body weight were more pronounced in WT than in Hif2α +/ mice both the weight gain in normoxia and the weight loss in hypoxia. As a result, WT mice weighed 18 g after 4 weeks of hypoxia and 32 g after 4 weeks of normoxia, while corresponding body weights of Hif2α +/ mice were 22 g in hypoxia and 29 g in normoxia. Another remarkable observation is that both Hif1α +/ and Hif2α +/ mice gained less weight during normoxia, while Hif1α +/ mice, unlike Hif2α +/ mice, lost more weight than WT mice during hypoxia (23). These findings suggest that both HIFs have distinct roles. This may relate to their different cell-specific expression. Possibly, deficiency of HIF-1α impairs glycolysis during hypoxia, thereby reducing the overall metabolism in Hif1α +/ mice. In contrast, we recently showed that HIF-2α plays a less important role than HIF-1α in hypoxic upregulation of glycolytic genes (26), suggesting that HIF-1α could play a more important role than HIF-2α in hypoxic regulation of particular processes influencing weight gain. Weight loss has also been documented in patients suffering from chronic obstructive pulmonary disease (45). Medical significance. The role of HIF-2α in the development of pulmonary hypertension and RV dysfunction may have clinical relevance. Overall, Hif2α +/ mice tolerated hypoxic stress much better than did WT mice, as they lost less weight, remained more active, and showed no pulmonary vascular remodeling or hypertension. Therefore, our data would encourage evaluation of the possible therapeutic potential of HIF-2α inhibitors for this disease. Retrovirally mediated inhibition of HIFs, via blocking of the interaction of the cofactor p300 with HIF-1α and HIF-2α, has been previously demonstrated to suppress tumor growth (46). Considering the limitations of current treatments of pulmonary hypertension (2, 3), inhibition of HIFs might be considered to prevent or reduce hypoxiainduced pulmonary hypertension. Since heterozygous deficiency of HIF-2α already protected mice, partial inhibition of hypoxia-inducible factors may be therapeutically sufficient. Acknowledgments The authors thank S. Janssens (Center for Transgene Technology and Gene Therapy) for helpful discussion and A. Bouché, K. Bijnens, E. Gils, S. Jansen, L. Kieckens, A. Manderveld, K. Maris, T. Vancoetsem, A. Vandenhoeck, B. Vanwetswinkel, and S. Wyns (all from the Center for Transgene Technology and Gene Therapy) for technical assistance. This work was supported by a European BIOMED grant (no. PL963380) and a grant from the Interuniversitaire Attractiepolen. 1. Barbera, J.A., Peinado, V.I., and Santos, S Pulmonary hypertension in COPD: old and new concepts. Monaldi Arch. Chest Dis. 55: Hida, W., Tun, Y., Kikuchi, Y., Okabe, S., and Shirato, K Pulmonary hypertension in patients with chronic obstructive pulmonary disease: recent advances in pathophysiology and management. Respirology. 7: Barnes, P.J Chronic obstructive pulmonary disease. N. Engl. J. Med. 343: Chen, Y.F., and Oparil, S Endothelin and pulmonary hypertension. J. Cardiovasc. Pharmacol. 35(4 Suppl. 2):S49 S Durmowicz, A.G., and Stenmark, K.R Mechanisms of structural remodeling in chronic pulmonary hypertension. Pediatr. Rev. 20:e91 e Stenmark, K.R., and Mecham, R.P Cellular and molecular mechanisms of pulmonary vascular remodeling. Annu. Rev. Physiol. 59: Voelkel, N.F., and Tuder, R.M Hypoxia-induced pulmonary vascular remodeling: a model for what human disease? J. Clin. Invest. 106: Fagan, K.A., et al The pulmonary circulation of homozygous or heterozygous enos-null mice is hyperresponsive to mild hypoxia. J. Clin. Invest. 103: Hoshikawa, Y., et al Prostacyclin receptor-dependent modulation of pulmonary vascular remodeling. Am. J. Respir. Crit. Care Med. 164: Geraci, M.W., et al Pulmonary prostacyclin synthase overexpression in transgenic mice protects against development of hypoxic pulmonary hypertension. J. Clin. Invest. 103: The Journal of Clinical Investigation May 2003 Volume 111 Number 10
9 11. Budts, W., et al Aerosol gene transfer with inducible nitric oxide synthase reduces hypoxic pulmonary hypertension and pulmonary vascular remodeling in rats. Circulation. 102: Eddahibi, S., et al Attenuated hypoxic pulmonary hypertension in mice lacking the 5-hydroxytryptamine transporter gene. J. Clin. Invest. 105: Carmeliet, P., and Collen, D Vascular development and disorders: molecular analysis and pathogenic insights. Kidney Int. 53: Levi, M., et al Deficiency of urokinase-type plasminogen activatormediated plasmin generation impairs vascular remodeling during hypoxia-induced pulmonary hypertension in mice. Circulation. 103: Campbell, A.I., Zhao, Y., Sandhu, R., and Stewart, D.J Cell-based gene transfer of vascular endothelial growth factor attenuates monocrotaline-induced pulmonary hypertension. Circulation. 104: Semenza, G.L Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu. Rev. Cell Dev. Biol. 15: Carmeliet, P., et al Role of HIF-1α in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature. 394: Tian, H., McKnight, S.L., and Russell, D.W Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev. 11: Ema, M., et al A novel bhlh-pas factor with close sequence similarity to hypoxia-inducible factor 1α regulates the VEGF expression and is potentially involved in lung and vascular development. Proc. Natl. Acad. Sci. U. S. A. 94: Flamme, I., et al HRF, a putative basic helix-loop-helix-pas-domain transcription factor is closely related to hypoxia-inducible factor-1α and developmentally expressed in blood vessels. Mech. Dev. 63: Peng, J., Zhang, L., Drysdale, L., and Fong, G.H The transcription factor EPAS-1/hypoxia-inducible factor 2α plays an important role in vascular remodeling. Proc. Natl. Acad. Sci. U. S. A. 97: Tian, H., Hammer, R.E., Matsumoto, A.M., Russell, D.W., and McKnight, S.L The hypoxia-responsive transcription factor EPAS1 is essential for catecholamine homeostasis and protection against heart failure during embryonic development. Genes Dev. 12: Yu, A.Y., et al Impaired physiological responses to chronic hypoxia in mice partially deficient for hypoxia-inducible factor 1α. J. Clin. Invest. 103: Shimoda, L.A., Manalo, D.J., Sham, J.S., Semenza, G.L., and Sylvester, J.T Partial HIF-1α deficiency impairs pulmonary arterial myocyte electrophysiological responses to hypoxia. Am. J. Physiol. Lung Cell. Mol. Physiol. 281:L202 L Compernolle, V., et al Loss of HIF-2α and inhibition of VEGF-signaling impair fetal lung maturation, while VEGF treatment prevents fatal respiratory distress in premature mice. Nat. Med. 8: Brusselmans, K., et al Hypoxia-inducible factor-2α (HIF-2α) is involved in the apoptotic response to hypoglycemia but not to hypoxia. J. Biol. Chem. 276: Hoit, B.D Invasive hemodynamic studies in open and closed chest mice. In Cardiovascular physiology in the genetically engineered mouse. B.D. Hoit and R.A. Walsh, editors. Kluwer Academic Publishers. Norwell, Massachusetts, USA Chen, E.P., Craig, D.M., Bittner, H.B., Davis, R.D., and Van Trigt, P Pharmacological strategies for improving diastolic dysfunction in the setting of chronic pulmonary hypertension. Circulation. 97: Steudel, W., et al Sustained pulmonary hypertension and right ventricular hypertrophy after chronic hypoxia in mice with congenital deficiency of nitric oxide synthase 3. J. Clin. Invest. 101: Champion, H.C., et al Gene transfer of endothelial nitric oxide synthase to the lung of the mouse in vivo. Circ. Res. 84: Roberts, J.D., Jr., Roberts, C.T., Jones, R.C., Zapol, W.M., and Bloch, K.D Continuous nitric oxide inhalation reduces pulmonary arterial structural changes, right ventricular hypertrophy, and growth retardation in the hypoxic newborn rat. Circ. Res. 76: Wiesener, M.S., et al Widespread, hypoxia-inducible expression of HIF-2α in distinct cell populations of different organs. FASEB J. 17: Wort, S.J., Woods, M., Warner, T.D., Evans, T.W., and Mitchell, J.A Endogenously released endothelin-1 from human pulmonary artery smooth muscle promotes cellular proliferation: relevance to pathogenesis of pulmonary hypertension and vascular remodeling. Am. J. Respir. Cell Mol. Biol. 25: Nathan, C., and Xie, Q.W Nitric oxide synthases: roles, tolls, and controls. Cell. 78: Le Cras, T.D., and McMurtry, I.F Nitric oxide production in the hypoxic lung. Am. J. Physiol. Lung Cell. Mol. Physiol. 280:L575 L Ito, H., et al Endothelin-1 induces hypertrophy with enhanced expression of muscle-specific genes in cultured neonatal rat cardiomyocytes. Circ. Res. 69: Zaloga, G.P., et al Increased circulating plasma norepinephrine concentrations in noncardiac causes of pulmonary hypertension. Crit. Care Med. 12: Bratel, T., Wennlund, A., and Carlstrom, K Impact of hypoxaemia on neuroendocrine function and catecholamine secretion in chronic obstructive pulmonary disease (COPD). Effects of long-term oxygen treatment. Respir. Med. 94: Jin, N., Packer, C.S., English, D., and Rhoades, R.A Inositol trisphosphate is involved in norepinephrine- but not in hypoxia-induced pulmonary arterial contraction. Am. J. Physiol. 264:L160 L Davies, P., Maddalo, F., and Reid, L The response of microvessels in rat lung explants to incubation with norepinephrine. Exp. Lung Res. 7: Carmeliet, P., et al Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat. Med. 7: Jones, R.D., Thompson, J.S., and Morice, A.H The NADPH oxidase inhibitors iodonium diphenyl and cadmium sulphate inhibit hypoxic pulmonary vasoconstriction in isolated rat pulmonary arteries. Physiol. Res. 49: Suzuki, J., et al Immunohistochemical analysis of platelet-derived growth factor-b expression in myocardial tissues in hypertrophic cardiomyopathy. Cardiovasc. Pathol. 8: Nakaki, T., Nakayama, M., Yamamoto, S., and Kato, R Alpha 1-adrenergic stimulation and beta 2-adrenergic inhibition of DNA synthesis in vascular smooth muscle cells. Mol. Pharmacol. 37: Gross, N.J Extrapulmonary effects of chronic obstructive pulmonary disease. Curr. Opin. Pulm. Med. 7: Kung, A.L., Wang, S., Klco, J.M., Kaelin, W.G., and Livingston, D.M Suppression of tumor growth through disruption of hypoxia-inducible transcription. Nat. Med. 6: The Journal of Clinical Investigation May 2003 Volume 111 Number
Hypoxia-induced pulmonary vascular remodeling: a model for what human disease?
 PERSPECTIVE SERIES Tissue responses to ischemia Hypoxia-induced pulmonary vascular remodeling: a model for what human disease? Norbert F. Voelkel and Rubin M. Tuder Pulmonary Hypertension Center, University
PERSPECTIVE SERIES Tissue responses to ischemia Hypoxia-induced pulmonary vascular remodeling: a model for what human disease? Norbert F. Voelkel and Rubin M. Tuder Pulmonary Hypertension Center, University
Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia!
 Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia! Lorenz Poellinger! Dept. of Cell and Molecular Biology! Karolinska Institutet, Stockholm, Sweden! Normoxia - O 2 availability is in balance
Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia! Lorenz Poellinger! Dept. of Cell and Molecular Biology! Karolinska Institutet, Stockholm, Sweden! Normoxia - O 2 availability is in balance
Nox-Dependent Mechanisms of Cardiomyocyte Dysfunction in a Model of Pressure Overload
 Nox-Dependent Mechanisms of Cardiomyocyte Dysfunction in a Model of Pressure Overload Giovanna Frazziano, PhD Vascular Medicine Institute Department of Pharmacology and Chemical Biology University of Pittsburgh
Nox-Dependent Mechanisms of Cardiomyocyte Dysfunction in a Model of Pressure Overload Giovanna Frazziano, PhD Vascular Medicine Institute Department of Pharmacology and Chemical Biology University of Pittsburgh
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy
 Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
Supplemental Figure I
 Supplemental Figure I Kl ( mmol/l)-induced Force orta M (mn) 1 (mn) 1 Supplemental Figure I. Kl-induced contractions. and, Kl ( mmol/l)-induced contractions of the aorta () and those of mesenteric arteries
Supplemental Figure I Kl ( mmol/l)-induced Force orta M (mn) 1 (mn) 1 Supplemental Figure I. Kl-induced contractions. and, Kl ( mmol/l)-induced contractions of the aorta () and those of mesenteric arteries
SUPPLEMENTAL DATA. Lumen area ( m 2 )
 Elastin Lumen area ( m 2 ) Media to lumen ratio (x1) H.E. Medium thickness ( m) Medium area ( m 2 ) SUPPLEMENTAL DATA A (Bmal1 flox/flox ) (SM-Bmal1 -/- ) B 1 8 8 6 6 4 4 2 2 1µm 5 8 4 6 3 2 4 1 2 Supplemental
Elastin Lumen area ( m 2 ) Media to lumen ratio (x1) H.E. Medium thickness ( m) Medium area ( m 2 ) SUPPLEMENTAL DATA A (Bmal1 flox/flox ) (SM-Bmal1 -/- ) B 1 8 8 6 6 4 4 2 2 1µm 5 8 4 6 3 2 4 1 2 Supplemental
PULMONARY CIRCULATION AT HIGH ALTITUDE. Jean COUDERT
 PULMONARY CIRCULATION AT HIGH ALTITUDE Jean COUDERT Laboratoire de Physiologie Biologie du Sport Faculté de Médecine Université d Auvergne CLERMONT-FERRAND, FRANCE The most striking changes at High Altitude
PULMONARY CIRCULATION AT HIGH ALTITUDE Jean COUDERT Laboratoire de Physiologie Biologie du Sport Faculté de Médecine Université d Auvergne CLERMONT-FERRAND, FRANCE The most striking changes at High Altitude
Research article. Department of Pharmacology and Toxicology, Medical College of Georgia, Georgia Health Sciences University, Augusta, Georgia, USA.
 Research article Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension, and its inhibition attenuates pathologic features of disease Wanli Ma, 1 Weihong Han, 1 Peter
Research article Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension, and its inhibition attenuates pathologic features of disease Wanli Ma, 1 Weihong Han, 1 Peter
Caffeine Modulates Hyperoxia - Induced Angiogenesis in Newborn Mice
 Caffeine Modulates Hyperoxia - Induced Angiogenesis in Newborn Mice Vikramaditya Dumpa, MD Lori C Nielsen, MS Huamei Wang, MD Vasanth HS Kumar, MD Supported by AAP Marshall Klaus Perinatal Research Grant
Caffeine Modulates Hyperoxia - Induced Angiogenesis in Newborn Mice Vikramaditya Dumpa, MD Lori C Nielsen, MS Huamei Wang, MD Vasanth HS Kumar, MD Supported by AAP Marshall Klaus Perinatal Research Grant
Pulmonary circulation. Lung Blood supply : lungs have a unique blood supply system :
 Dr. Ali Naji Pulmonary circulation Lung Blood supply : lungs have a unique blood supply system : 1. Pulmonary circulation 2. Bronchial circulation 1- Pulmonary circulation : receives the whole cardiac
Dr. Ali Naji Pulmonary circulation Lung Blood supply : lungs have a unique blood supply system : 1. Pulmonary circulation 2. Bronchial circulation 1- Pulmonary circulation : receives the whole cardiac
ino in neonates with cardiac disorders
 ino in neonates with cardiac disorders Duncan Macrae Paediatric Critical Care Terminology PAP Pulmonary artery pressure PVR Pulmonary vascular resistance PHT Pulmonary hypertension - PAP > 25, PVR >3,
ino in neonates with cardiac disorders Duncan Macrae Paediatric Critical Care Terminology PAP Pulmonary artery pressure PVR Pulmonary vascular resistance PHT Pulmonary hypertension - PAP > 25, PVR >3,
renoprotection therapy goals 208, 209
 Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
RAPID COMMUNICATION. Vascular Reactivity in Isolated Lungs of Rats with Spontaneous Systemic Hypertension
 Physiol. Res. 40:367-371,1991 RAPID COMMUNICATION Vascular Reactivity in Isolated Lungs of Rats with Spontaneous Systemic Hypertension V. HAMPL, J. HERGET Department of Physiology, 2nd Medical School,
Physiol. Res. 40:367-371,1991 RAPID COMMUNICATION Vascular Reactivity in Isolated Lungs of Rats with Spontaneous Systemic Hypertension V. HAMPL, J. HERGET Department of Physiology, 2nd Medical School,
Tissue repair. (3&4 of 4)
 Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Control of blood tissue blood flow. Faisal I. Mohammed, MD,PhD
 Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
Circulation. Blood Pressure and Antihypertensive Medications. Venous Return. Arterial flow. Regulation of Cardiac Output.
 Circulation Blood Pressure and Antihypertensive Medications Two systems Pulmonary (low pressure) Systemic (high pressure) Aorta 120 mmhg Large arteries 110 mmhg Arterioles 40 mmhg Arteriolar capillaries
Circulation Blood Pressure and Antihypertensive Medications Two systems Pulmonary (low pressure) Systemic (high pressure) Aorta 120 mmhg Large arteries 110 mmhg Arterioles 40 mmhg Arteriolar capillaries
BNP mrna expression in DR and DS rat left ventricles (n = 5). (C) Plasma norepinephrine
 Kanazawa, et al. Supplementary figure legends Supplementary Figure 1 DS rats had congestive heart failure. (A) DR and DS rat hearts. (B) QRT-PCR analysis of BNP mrna expression in DR and DS rat left ventricles
Kanazawa, et al. Supplementary figure legends Supplementary Figure 1 DS rats had congestive heart failure. (A) DR and DS rat hearts. (B) QRT-PCR analysis of BNP mrna expression in DR and DS rat left ventricles
hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and function in
 SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure 1. Fbn1 C1039G/+ hearts display normal cardiac function in the absence of hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and
SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure 1. Fbn1 C1039G/+ hearts display normal cardiac function in the absence of hemodynamic stress. A. Echocardiographic quantification of cardiac dimensions and
In Vivo Models and Cell Delivery for Lung Indications NO DISCLOSURES
 In Vivo Models and Cell Delivery for Lung Indications Marlowe Eldridge MD Department of Pediatrics and Biomedical Engineering University of Wisconsin School of Medicine and Public Health NO DISCLOSURES
In Vivo Models and Cell Delivery for Lung Indications Marlowe Eldridge MD Department of Pediatrics and Biomedical Engineering University of Wisconsin School of Medicine and Public Health NO DISCLOSURES
c Ischemia (30 min) Reperfusion (8 w) Supplementary Figure bp 300 bp Ischemia (30 min) Reperfusion (4 h) Dox 20 mg/kg i.p.
 a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
Supplementary Figure 1:
 Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Role of Inflammatory and Progenitor Cells in Pulmonary Vascular Remodeling: Potential Role for Targeted Therapies. Traditional Hypothesis Stress
 3/1/212 Role of Inflammatory and Progenitor Cells in Pulmonary Vascular Remodeling: Potential Role for Targeted Therapies K.R. Stenmark University of Colorado Denver, CO 845 Prominent Fibroproliferative
3/1/212 Role of Inflammatory and Progenitor Cells in Pulmonary Vascular Remodeling: Potential Role for Targeted Therapies K.R. Stenmark University of Colorado Denver, CO 845 Prominent Fibroproliferative
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
SUPPLEMENTARY INFORMATION
 a c e doi:10.1038/nature10407 b d f Supplementary Figure 1. SERCA2a complex analysis. (a) Two-dimensional SDS-PAGE gels of SERCA2a complexes. A silver-stained SDSPAGE gel is shown, which reveals a 12 kda
a c e doi:10.1038/nature10407 b d f Supplementary Figure 1. SERCA2a complex analysis. (a) Two-dimensional SDS-PAGE gels of SERCA2a complexes. A silver-stained SDSPAGE gel is shown, which reveals a 12 kda
HYPERTENSION: Sustained elevation of arterial blood pressure above normal o Systolic 140 mm Hg and/or o Diastolic 90 mm Hg
 Lecture 39 Anti-Hypertensives B-Rod BLOOD PRESSURE: Systolic / Diastolic NORMAL: 120/80 Systolic = measure of pressure as heart is beating Diastolic = measure of pressure while heart is at rest between
Lecture 39 Anti-Hypertensives B-Rod BLOOD PRESSURE: Systolic / Diastolic NORMAL: 120/80 Systolic = measure of pressure as heart is beating Diastolic = measure of pressure while heart is at rest between
Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 TAC 8W TAC 2W. Postn MCM ; R26 mtmg Sham GFP Col 1/3 TAC 8W TAC 2W
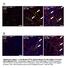 A Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 Tcf21 MCM ; R26 mtmg TAC 2W Tcf21 MCM ; R26 mtmg TAC 8W B Postn MCM ; R26 mtmg Sham GFP Col 1/3 Postn MCM ; R26 mtmg TAC 2W Postn MCM ; R26 mtmg TAC 8W Supplementary
A Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 Tcf21 MCM ; R26 mtmg TAC 2W Tcf21 MCM ; R26 mtmg TAC 8W B Postn MCM ; R26 mtmg Sham GFP Col 1/3 Postn MCM ; R26 mtmg TAC 2W Postn MCM ; R26 mtmg TAC 8W Supplementary
BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1
 BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1 Terms you should understand: hemorrhage, intrinsic and extrinsic mechanisms, anoxia, myocardial contractility, residual
BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1 Terms you should understand: hemorrhage, intrinsic and extrinsic mechanisms, anoxia, myocardial contractility, residual
Left atrial function. Aliakbar Arvandi MD
 In the clinic Left atrial function Abstract The left atrium (LA) is a left posterior cardiac chamber which is located adjacent to the esophagus. It is separated from the right atrium by the inter-atrial
In the clinic Left atrial function Abstract The left atrium (LA) is a left posterior cardiac chamber which is located adjacent to the esophagus. It is separated from the right atrium by the inter-atrial
Cardiovascular Physiology
 Cardiovascular Physiology Lecture 1 objectives Explain the basic anatomy of the heart and its arrangement into 4 chambers. Appreciate that blood flows in series through the systemic and pulmonary circulations.
Cardiovascular Physiology Lecture 1 objectives Explain the basic anatomy of the heart and its arrangement into 4 chambers. Appreciate that blood flows in series through the systemic and pulmonary circulations.
Kidney. Heart. Lung. Sirt1. Gapdh. Mouse IgG DAPI. Rabbit IgG DAPI
 a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
1. Distinguish among the types of blood vessels on the basis of their structure and function.
 Blood Vessels and Circulation Objectives This chapter describes the structure and functions of the blood vessels Additional subjects contained in Chapter 13 include cardiovascular physiology, regulation,
Blood Vessels and Circulation Objectives This chapter describes the structure and functions of the blood vessels Additional subjects contained in Chapter 13 include cardiovascular physiology, regulation,
The cardiovascular system is composed of a pump the heart and blood
 5 E X E R C I S E Cardiovascular Dynamics O B J E C T I V E S 1. To understand the relationships among blood flow, pressure gradient, and resistance 2. To define resistance and describe the main factors
5 E X E R C I S E Cardiovascular Dynamics O B J E C T I V E S 1. To understand the relationships among blood flow, pressure gradient, and resistance 2. To define resistance and describe the main factors
Left ventricular hypertrophy: why does it happen?
 Nephrol Dial Transplant (2003) 18 [Suppl 8]: viii2 viii6 DOI: 10.1093/ndt/gfg1083 Left ventricular hypertrophy: why does it happen? Gerard M. London Department of Nephrology and Dialysis, Manhes Hospital,
Nephrol Dial Transplant (2003) 18 [Suppl 8]: viii2 viii6 DOI: 10.1093/ndt/gfg1083 Left ventricular hypertrophy: why does it happen? Gerard M. London Department of Nephrology and Dialysis, Manhes Hospital,
Aberrant Promoter CpG Methylation is a Mechanism for Lack of Hypoxic Induction of
 Aberrant Promoter CpG Methylation is a Mechanism for Lack of Hypoxic Induction of PHD3 in a Diverse Set of Malignant Cells Abstract The prolyl-hydroxylase domain family of enzymes (PHD1-3) plays an important
Aberrant Promoter CpG Methylation is a Mechanism for Lack of Hypoxic Induction of PHD3 in a Diverse Set of Malignant Cells Abstract The prolyl-hydroxylase domain family of enzymes (PHD1-3) plays an important
Weeks 1-3:Cardiovascular
 Weeks 1-3:Cardiovascular Cardiac Output The total volume of blood ejected from the ventricles in one minute is known as the cardiac output. Heart Rate (HR) X Stroke Volume (SV) = Cardiac Output Normal
Weeks 1-3:Cardiovascular Cardiac Output The total volume of blood ejected from the ventricles in one minute is known as the cardiac output. Heart Rate (HR) X Stroke Volume (SV) = Cardiac Output Normal
Cardiovascular System B L O O D V E S S E L S 2
 Cardiovascular System B L O O D V E S S E L S 2 Blood Pressure Main factors influencing blood pressure: Cardiac output (CO) Peripheral resistance (PR) Blood volume Peripheral resistance is a major factor
Cardiovascular System B L O O D V E S S E L S 2 Blood Pressure Main factors influencing blood pressure: Cardiac output (CO) Peripheral resistance (PR) Blood volume Peripheral resistance is a major factor
Pulmonary Hypertension. Murali Chakinala, M.D. Washington University School of Medicine
 Pulmonary Hypertension Murali Chakinala, M.D. Washington University School of Medicine Pulmonary Circulation Alveolar Capillary relationship Pulmonary Circulation High flow, low resistance PVR ~1/15 of
Pulmonary Hypertension Murali Chakinala, M.D. Washington University School of Medicine Pulmonary Circulation Alveolar Capillary relationship Pulmonary Circulation High flow, low resistance PVR ~1/15 of
Pulmonary hypertension in chronic obstructive pulmonary disease: current theories of pathogenesis and their implications for treatment
 605 OCCASIONAL REVIEW Pulmonary hypertension in chronic obstructive pulmonary disease: current theories of pathogenesis and their implications for treatment J L Wright, R D Levy, A Churg... The development
605 OCCASIONAL REVIEW Pulmonary hypertension in chronic obstructive pulmonary disease: current theories of pathogenesis and their implications for treatment J L Wright, R D Levy, A Churg... The development
Cardiovascular System
 Cardiovascular System Purpose Transport oxygen and nutrients Take waste products away from tissues & organs Things we learned Blood pressure: the force of blood pushing against the walls of blood vessels
Cardiovascular System Purpose Transport oxygen and nutrients Take waste products away from tissues & organs Things we learned Blood pressure: the force of blood pushing against the walls of blood vessels
Role of Inflammation in Pulmonary Hypertension
 Role of Inflammation in Pulmonary Hypertension K. R. Stenmark University of Colorado Denver, USA Prominent Fibroproliferative Changes are Observed in the Lung Vasculature of Infants With Pulmonary Arterial
Role of Inflammation in Pulmonary Hypertension K. R. Stenmark University of Colorado Denver, USA Prominent Fibroproliferative Changes are Observed in the Lung Vasculature of Infants With Pulmonary Arterial
Remodeling the failing heart: : the biology and future treatment options
 Remodeling the failing heart: : the biology and future treatment options J-L Balligand (UCL-Brussels, BE) jl.balligand@uclouvain.be Myocardial remodeling: definitions phenotypic plasticity : remodeling
Remodeling the failing heart: : the biology and future treatment options J-L Balligand (UCL-Brussels, BE) jl.balligand@uclouvain.be Myocardial remodeling: definitions phenotypic plasticity : remodeling
Pharmacology - Problem Drill 11: Vasoactive Agents
 Pharmacology - Problem Drill 11: Vasoactive Agents Question No. 1 of 10 1. Vascular smooth muscle contraction is triggered by a rise in. Question #01 (A) Luminal calcium (B) Extracellular calcium (C) Intracellular
Pharmacology - Problem Drill 11: Vasoactive Agents Question No. 1 of 10 1. Vascular smooth muscle contraction is triggered by a rise in. Question #01 (A) Luminal calcium (B) Extracellular calcium (C) Intracellular
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
d) Cardiovascular System Higher Human Biology
 d) Cardiovascular System Higher Human Biology What can your remember about the heart and blood vessels? What is the Cardiovascular System? The cardiovascular system, also known as the circulatory system,
d) Cardiovascular System Higher Human Biology What can your remember about the heart and blood vessels? What is the Cardiovascular System? The cardiovascular system, also known as the circulatory system,
Fetal gene upregulation by 1-wk TAC is significantly increased in mice lacking RGS2.
 3562-RG-1 Supplementary Figure 1 Fetal gene upregulation by 1-wk is significantly increased in mice lacking RGS2. ANP(Nppa) /BNP(Nppb) A-type and B-type natriuretic peptide; β-mhc (Myh7) beta myosin heavy
3562-RG-1 Supplementary Figure 1 Fetal gene upregulation by 1-wk is significantly increased in mice lacking RGS2. ANP(Nppa) /BNP(Nppb) A-type and B-type natriuretic peptide; β-mhc (Myh7) beta myosin heavy
PHM142 Lecture 4: Platelets + Endothelial Cells
 PHM142 Lecture 4: Platelets + Endothelial Cells 1 Hematopoiesis 2 Platelets Critical in clotting - activated by subendothelial matrix proteins (e.g. collagen, fibronectin, von Willebrand factor) and thrombin
PHM142 Lecture 4: Platelets + Endothelial Cells 1 Hematopoiesis 2 Platelets Critical in clotting - activated by subendothelial matrix proteins (e.g. collagen, fibronectin, von Willebrand factor) and thrombin
Exercise in Adverse Cardiac Remodeling: of Mice and Men
 Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
End of chapter exercises
 End of chapter exercises Problem 1: The following diagrams show the heart during the cardiac cycle. The arrows represent the flow of blood. Study the diagrams and answer the questions that follow: Figure
End of chapter exercises Problem 1: The following diagrams show the heart during the cardiac cycle. The arrows represent the flow of blood. Study the diagrams and answer the questions that follow: Figure
REGULATION OF CARDIOVASCULAR SYSTEM
 REGULATION OF CARDIOVASCULAR SYSTEM Jonas Addae Medical Sciences, UWI REGULATION OF CARDIOVASCULAR SYSTEM Intrinsic Coupling of cardiac and vascular functions - Autoregulation of vessel diameter Extrinsic
REGULATION OF CARDIOVASCULAR SYSTEM Jonas Addae Medical Sciences, UWI REGULATION OF CARDIOVASCULAR SYSTEM Intrinsic Coupling of cardiac and vascular functions - Autoregulation of vessel diameter Extrinsic
EPO, VEGF, chronic hypoxia adaptations and metabolism in the heart
 EPO, VEGF, chronic hypoxia adaptations and metabolism in the heart BSc Catherine Privat M. Laboratorio de Transporte de Oxígeno Instituto de Investigaciones de Altura Dpto de Ciencias Biológicas y Fisiológicas
EPO, VEGF, chronic hypoxia adaptations and metabolism in the heart BSc Catherine Privat M. Laboratorio de Transporte de Oxígeno Instituto de Investigaciones de Altura Dpto de Ciencias Biológicas y Fisiológicas
THE CIRCULATORY SYSTEM
 Biology 30S THE CIRCULATORY SYSTEM Name: This module adapted from bblearn.merlin.mb.ca 1 Introduction to Circulation The first organ to form, and the last organ to die. The heart is the pump of life. The
Biology 30S THE CIRCULATORY SYSTEM Name: This module adapted from bblearn.merlin.mb.ca 1 Introduction to Circulation The first organ to form, and the last organ to die. The heart is the pump of life. The
SCVMC RESPIRATORY CARE PROCEDURE
 Page 1 of 7 New: 12/08 R: 4/11 R NC: 7/11, 7/12 B7180-63 Definitions: Inhaled nitric oxide (i) is a medical gas with selective pulmonary vasodilator properties. Vaso-reactivity is the evidence of acute
Page 1 of 7 New: 12/08 R: 4/11 R NC: 7/11, 7/12 B7180-63 Definitions: Inhaled nitric oxide (i) is a medical gas with selective pulmonary vasodilator properties. Vaso-reactivity is the evidence of acute
Copyright 2011, 2007 by Mosby, Inc., an affiliate of Elsevier Inc. Normal Cardiac Anatomy
 Mosby,, an affiliate of Elsevier Normal Cardiac Anatomy Impaired cardiac pumping Results in vasoconstriction & fluid retention Characterized by ventricular dysfunction, reduced exercise tolerance, diminished
Mosby,, an affiliate of Elsevier Normal Cardiac Anatomy Impaired cardiac pumping Results in vasoconstriction & fluid retention Characterized by ventricular dysfunction, reduced exercise tolerance, diminished
PUBLICATIONS. cells. J. Physiol. (London) 517P:91P (Manchester, England, UK).
 277 PUBLICATIONS Abstracts Haddad JJ, Land SC (1999). Differential activation of oxygen-responsive transcription factors over fetal-to-neonatal alveolar oxygen tensions in rat fetal distal lung epithelial
277 PUBLICATIONS Abstracts Haddad JJ, Land SC (1999). Differential activation of oxygen-responsive transcription factors over fetal-to-neonatal alveolar oxygen tensions in rat fetal distal lung epithelial
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events
 Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
Title: Smooth muscle cell-specific Tgfbr1 deficiency promotes aortic aneurysm formation by stimulating multiple signaling events Pu Yang 1, 3, radley M. Schmit 1, Chunhua Fu 1, Kenneth DeSart 1, S. Paul
Age-related changes in cardiovascular system. Dr. Rehab Gwada
 Age-related changes in cardiovascular system Dr. Rehab Gwada Objectives explain the main structural and functional changes in cardiovascular system associated with normal aging Introduction aging results
Age-related changes in cardiovascular system Dr. Rehab Gwada Objectives explain the main structural and functional changes in cardiovascular system associated with normal aging Introduction aging results
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
HIF-inducible mir-191 promotes migration in breast cancer through complex regulation of TGFβ-signaling in hypoxic microenvironment.
 HIF-inducible mir-9 promotes migration in breast cancer through complex regulation of TGFβ-signaling in hypoxic microenvironment. Neha Nagpal, Hafiz M Ahmad, Shibu Chameettachal3, Durai Sundar, Sourabh
HIF-inducible mir-9 promotes migration in breast cancer through complex regulation of TGFβ-signaling in hypoxic microenvironment. Neha Nagpal, Hafiz M Ahmad, Shibu Chameettachal3, Durai Sundar, Sourabh
Nothing to Disclose. Severe Pulmonary Hypertension
 Severe Ronald Pearl, MD, PhD Professor and Chair Department of Anesthesiology Stanford University Rpearl@stanford.edu Nothing to Disclose 65 year old female Elective knee surgery NYHA Class 3 Aortic stenosis
Severe Ronald Pearl, MD, PhD Professor and Chair Department of Anesthesiology Stanford University Rpearl@stanford.edu Nothing to Disclose 65 year old female Elective knee surgery NYHA Class 3 Aortic stenosis
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Unit 1 Exam: Respiratory and Circulatory Systems
 Name Date Unit 1 Exam: Respiratory and Circulatory Systems Multiple Choice. Please make your best selection. 1. Respiration is the process of: (a) Sucking air into the lungs (b) Converting glucose into
Name Date Unit 1 Exam: Respiratory and Circulatory Systems Multiple Choice. Please make your best selection. 1. Respiration is the process of: (a) Sucking air into the lungs (b) Converting glucose into
Cor pulmonale. Dr hamid reza javadi
 1 Cor pulmonale Dr hamid reza javadi 2 Definition Cor pulmonale ;pulmonary heart disease; is defined as dilation and hypertrophy of the right ventricle (RV) in response to diseases of the pulmonary vasculature
1 Cor pulmonale Dr hamid reza javadi 2 Definition Cor pulmonale ;pulmonary heart disease; is defined as dilation and hypertrophy of the right ventricle (RV) in response to diseases of the pulmonary vasculature
Pathology of Coronary Artery Disease
 Pathology of Coronary Artery Disease Seth J. Kligerman, MD Pathology of Coronary Artery Disease Seth Kligerman, MD Assistant Professor Medical Director of MRI University of Maryland Department of Radiology
Pathology of Coronary Artery Disease Seth J. Kligerman, MD Pathology of Coronary Artery Disease Seth Kligerman, MD Assistant Professor Medical Director of MRI University of Maryland Department of Radiology
International Graduate Research Programme in Cardiovascular Science
 1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
Pulmonary Hypertension Perioperative Management
 Pulmonary Hypertension Perioperative Management Bruce J Leone, MD Professor of Anesthesiology Chief, Neuroanesthesiology Vice Chair for Academic Affairs Mayo Clinic Jacksonville, Florida Introduction Definition
Pulmonary Hypertension Perioperative Management Bruce J Leone, MD Professor of Anesthesiology Chief, Neuroanesthesiology Vice Chair for Academic Affairs Mayo Clinic Jacksonville, Florida Introduction Definition
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Outline. Pathophysiology: Heart Failure. Heart Failure. Heart Failure: Definitions. Etiologies. Etiologies
 Outline Pathophysiology: Mat Maurer, MD Irving Assistant Professor of Medicine Definitions and Classifications Epidemiology Muscle and Chamber Function Pathophysiology : Definitions An inability of the
Outline Pathophysiology: Mat Maurer, MD Irving Assistant Professor of Medicine Definitions and Classifications Epidemiology Muscle and Chamber Function Pathophysiology : Definitions An inability of the
Cardiovascular Responses to Exercise
 CARDIOVASCULAR PHYSIOLOGY 69 Case 13 Cardiovascular Responses to Exercise Cassandra Farias is a 34-year-old dietician at an academic medical center. She believes in the importance of a healthy lifestyle
CARDIOVASCULAR PHYSIOLOGY 69 Case 13 Cardiovascular Responses to Exercise Cassandra Farias is a 34-year-old dietician at an academic medical center. She believes in the importance of a healthy lifestyle
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Exam KEY. NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 23, 2015 Total POINTS: % of grade in class
 NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 23, 2015 Total POINTS: 100 20% of grade in class 1) Arterial and venous blood samples are taken, and other physiological measures are obtained, from a
NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 23, 2015 Total POINTS: 100 20% of grade in class 1) Arterial and venous blood samples are taken, and other physiological measures are obtained, from a
Squeeze, Squeeze, Squeeze: The Importance of Right Ventricular Function and PH
 Squeeze, Squeeze, Squeeze: The Importance of Right Ventricular Function and PH Javier Jimenez MD PhD FACC Director, Advanced Heart Failure and Pulmonary Hypertension Miami Cardiac & Vascular Institute
Squeeze, Squeeze, Squeeze: The Importance of Right Ventricular Function and PH Javier Jimenez MD PhD FACC Director, Advanced Heart Failure and Pulmonary Hypertension Miami Cardiac & Vascular Institute
Cardiovascular System. Heart
 Cardiovascular System Heart Electrocardiogram A device that records the electrical activity of the heart. Measuring the relative electrical activity of one heart cycle. A complete contraction and relaxation.
Cardiovascular System Heart Electrocardiogram A device that records the electrical activity of the heart. Measuring the relative electrical activity of one heart cycle. A complete contraction and relaxation.
Unit 1 Exam: Respiratory and Circulatory Systems
 Name Date Unit 1 Exam: Respiratory and Circulatory Systems Multiple Choice. Please make your best selection. 1. Respiration is the process of: (a) Sucking air into the lungs (b) Converting glucose into
Name Date Unit 1 Exam: Respiratory and Circulatory Systems Multiple Choice. Please make your best selection. 1. Respiration is the process of: (a) Sucking air into the lungs (b) Converting glucose into
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D.
 Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
Control of blood tissue blood flow. Faisal I. Mohammed, MD,PhD
 Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
Control of blood tissue blood flow Faisal I. Mohammed, MD,PhD 1 Objectives List factors that affect tissue blood flow. Describe the vasodilator and oxygen demand theories. Point out the mechanisms of autoregulation.
30.1 Respiratory and Circulatory Functions. KEY CONCEPT The respiratory and circulatory systems bring oxygen and nutrients to the cells.
 30.1 Respiratory and Circulatory Functions KEY CONCEPT The respiratory and circulatory systems bring oxygen and nutrients to the cells. 30.1 Respiratory and Circulatory Functions The respiratory and circulatory
30.1 Respiratory and Circulatory Functions KEY CONCEPT The respiratory and circulatory systems bring oxygen and nutrients to the cells. 30.1 Respiratory and Circulatory Functions The respiratory and circulatory
The Study of Endothelial Function in CKD and ESRD
 The Study of Endothelial Function in CKD and ESRD Endothelial Diversity in the Human Body Aird WC. Circ Res 2007 Endothelial Diversity in the Human Body The endothelium should be viewed for what it is:
The Study of Endothelial Function in CKD and ESRD Endothelial Diversity in the Human Body Aird WC. Circ Res 2007 Endothelial Diversity in the Human Body The endothelium should be viewed for what it is:
For more information about how to cite these materials visit
 Author: Thomas Sisson, MD, 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author: Thomas Sisson, MD, 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
In the name of GOD. Animal models of cardiovascular diseases: myocardial infarction & hypertension
 In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
Lecture Notes. Chapter 2: Introduction to Respiratory Failure
 Lecture Notes Chapter 2: Introduction to Respiratory Failure Objectives Define respiratory failure, ventilatory failure, and oxygenation failure List the causes of respiratory failure Describe the effects
Lecture Notes Chapter 2: Introduction to Respiratory Failure Objectives Define respiratory failure, ventilatory failure, and oxygenation failure List the causes of respiratory failure Describe the effects
Special circulations, Coronary, Pulmonary. Faisal I. Mohammed, MD,PhD
 Special circulations, Coronary, Pulmonary Faisal I. Mohammed, MD,PhD 1 Objectives Describe the control of blood flow to different circulations (Skeletal muscles, pulmonary and coronary) Point out special
Special circulations, Coronary, Pulmonary Faisal I. Mohammed, MD,PhD 1 Objectives Describe the control of blood flow to different circulations (Skeletal muscles, pulmonary and coronary) Point out special
Familial PAH. Genetics and pulmonary arterial hypertension. PAH and mutations in the bone morphogenetic protein type II receptor (BMPR-II)
 athology of vascular lesions in idiopathic pulmonary arterial hypertension Genetics and pulmonary arterial hypertension Concentric intimal lesion Nick Morrell British Heart Foundation rofessor of Cardiopulmonary
athology of vascular lesions in idiopathic pulmonary arterial hypertension Genetics and pulmonary arterial hypertension Concentric intimal lesion Nick Morrell British Heart Foundation rofessor of Cardiopulmonary
Heart Failure with Preserved Ejection Fraction: Mechanisms and Management
 Heart Failure with Preserved Ejection Fraction: Mechanisms and Management Jay N. Cohn, M.D. Professor of Medicine Director, Rasmussen Center for Cardiovascular Disease Prevention University of Minnesota
Heart Failure with Preserved Ejection Fraction: Mechanisms and Management Jay N. Cohn, M.D. Professor of Medicine Director, Rasmussen Center for Cardiovascular Disease Prevention University of Minnesota
GAS EXCHANGE IB TOPIC 6.4 CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM. Terminal bronchiole Nasal cavity. Pharynx Left lung Alveoli.
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
In vivo bromodeoxyuridine (BrdU) incorporation was performed to analyze cell
 Supplementary Methods BrdU incorporation in vivo In vivo bromodeoxyuridine (BrdU) incorporation was performed to analyze cell proliferation in the heart. Mice were subjected to LI-TAC, and 5 days later
Supplementary Methods BrdU incorporation in vivo In vivo bromodeoxyuridine (BrdU) incorporation was performed to analyze cell proliferation in the heart. Mice were subjected to LI-TAC, and 5 days later
IB TOPIC 6.4 GAS EXCHANGE
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
Lung Diseases of the Newborn
 Lung Diseases of the Newborn PD Dr. med. Anne Hilgendorff Comprehensive Pneumology Center Perinatal Center LMU ispz Hauner Ludwig-Maximilians University und Helmholtz Zentrum München Prematurity 1 % of
Lung Diseases of the Newborn PD Dr. med. Anne Hilgendorff Comprehensive Pneumology Center Perinatal Center LMU ispz Hauner Ludwig-Maximilians University und Helmholtz Zentrum München Prematurity 1 % of
more than 50% of adults weigh more than 20% above optimum
 In the US: more than 50% of adults weigh more than 20% above optimum >30 kg m -2 obesity >40 kg m -2 morbid obesity BMI = weight(kg) / height(m 2 ) Pounds X 2.2 Inches divided by 39, squared From 2000
In the US: more than 50% of adults weigh more than 20% above optimum >30 kg m -2 obesity >40 kg m -2 morbid obesity BMI = weight(kg) / height(m 2 ) Pounds X 2.2 Inches divided by 39, squared From 2000
Abhd2 regulates alveolar type Ⅱ apoptosis and airway smooth muscle remodeling: a key target of COPD research
 Abhd2 regulates alveolar type Ⅱ apoptosis and airway smooth muscle remodeling: a key target of COPD research Shoude Jin Harbin Medical University, China Background COPD ------ a silent killer Insidious,
Abhd2 regulates alveolar type Ⅱ apoptosis and airway smooth muscle remodeling: a key target of COPD research Shoude Jin Harbin Medical University, China Background COPD ------ a silent killer Insidious,
HYPERTENSIVE VASCULAR DISEASE
 HYPERTENSIVE VASCULAR DISEASE Cutoffs in diagnosing hypertension in clinical practice sustained diastolic pressures >90 mm Hg, or sustained systolic pressures >140 mm Hg Malignant hypertension A small
HYPERTENSIVE VASCULAR DISEASE Cutoffs in diagnosing hypertension in clinical practice sustained diastolic pressures >90 mm Hg, or sustained systolic pressures >140 mm Hg Malignant hypertension A small
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Supplemental methods Pericardium In several studies, it has been shown that the pericardium significantly modulates ventricular interaction. 1-4 Since ventricular interaction has
SUPPLEMENTAL MATERIAL Supplemental methods Pericardium In several studies, it has been shown that the pericardium significantly modulates ventricular interaction. 1-4 Since ventricular interaction has
Blood flows away from the heart in arteries, to the capillaries and back to the heart in the veins
 Cardiovascular System Summary Notes The cardiovascular system includes: The heart, a muscular pump The blood, a fluid connective tissue The blood vessels, arteries, veins and capillaries Blood flows away
Cardiovascular System Summary Notes The cardiovascular system includes: The heart, a muscular pump The blood, a fluid connective tissue The blood vessels, arteries, veins and capillaries Blood flows away
Disclosures. Objectives 6/16/2016. A Look at the Other Side: Focus on the Right Ventricle and Pulmonary Hypertension
 A Look at the Other Side: Focus on the Right Ventricle and Pulmonary Hypertension Susan P. D Anna MSN, APN-BC, CHFN June 24, 2016 Disclosures Objectives Differentiate structure and function of RV and LV
A Look at the Other Side: Focus on the Right Ventricle and Pulmonary Hypertension Susan P. D Anna MSN, APN-BC, CHFN June 24, 2016 Disclosures Objectives Differentiate structure and function of RV and LV
Level 2 Anatomy and Physiology Bite size revision. Respiratory System. The order of the passage of oxygen into the body (inhalation/inspiration) is: -
 Respiratory System Function of the respiratory system The lungs are located in the chest cavity and their function is to intake oxygen and remove carbon dioxide from the body. Structure of the respiratory
Respiratory System Function of the respiratory system The lungs are located in the chest cavity and their function is to intake oxygen and remove carbon dioxide from the body. Structure of the respiratory
Blood Vessel Mechanics
 Blood Vessel Mechanics Ying Zheng, Ph.D. Department of Bioengineering BIOEN 326 11/01/2013 Blood Vessel Structure A Typical Artery and a Typical Vein Pressure and Blood Flow Wall stress ~ pressure Poiseuille
Blood Vessel Mechanics Ying Zheng, Ph.D. Department of Bioengineering BIOEN 326 11/01/2013 Blood Vessel Structure A Typical Artery and a Typical Vein Pressure and Blood Flow Wall stress ~ pressure Poiseuille
Expression of hypoxia inducible factor 1α, endothelin 1 and adrenomedullin in newborn rats with hypoxia induced pulmonary hypertension
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 8: 335-339, 2014 Expression of hypoxia inducible factor 1α, endothelin 1 and adrenomedullin in newborn rats with hypoxia induced pulmonary hypertension LE WANG, YING
EXPERIMENTAL AND THERAPEUTIC MEDICINE 8: 335-339, 2014 Expression of hypoxia inducible factor 1α, endothelin 1 and adrenomedullin in newborn rats with hypoxia induced pulmonary hypertension LE WANG, YING
Echocardiography as a diagnostic and management tool in medical emergencies
 Echocardiography as a diagnostic and management tool in medical emergencies Frank van der Heusen MD Department of Anesthesia and perioperative Care UCSF Medical Center Objective of this presentation Indications
Echocardiography as a diagnostic and management tool in medical emergencies Frank van der Heusen MD Department of Anesthesia and perioperative Care UCSF Medical Center Objective of this presentation Indications
