CARBOHYDRATE BOOKLET ANSWERS
|
|
|
- Domenic Hardy
- 5 years ago
- Views:
Transcription
1 CARBOHYDRATE BOOKLET ANSWERS FOR: QER QUESTION APPLICATION AND EXTENSION QUESTIONS. FEEDBACK CAN BE VIEWED AT:
2 LONG ANSWER QUESTIONS WITH QER The long answer question requires you to write at length about a topic. The quality of your extended responses (QER) will also be assessed. The mark scheme will have the following areas: 1. Indicative Content this is the biological concepts and terminology you are expected to use in your answer marks to gain these top marks your answer will show: The candidate constructs an articulate, integrated account correctly linking relevant points, such as those in the indicative content, which shows sequential reasoning. The answer fully address the question with no irrelevant inclusions or significant omissions. The candidate uses scientific conventions and vocabulary appropriately and accurately marks to gain these marks your answer will show: The candidate constructs an account correctly linking some relevant points, such as those in the indicative content showing some reasoning. The answer addresses the question with some omissions. The candidate uses scientific conventions and vocabulary appropriately and accurately marks to gain these marks your answer will show: The candidate makes some relevant points, such as those in the indicative content, showing limited reasoning. The answer addresses the question with significant omissions. The candidate has limited use of scientific conventions and vocabulary.
3 Complete the QER questions number 4 below. 4. Carbohydrates are a large group of biological molecules with diverse structures and functions. Plants and animals can use different carbohydrates to perform a common function. In addition to this, plants and animals can use carbohydrates that are unique to them and for unique functions. The microscope images below show carbohydrates that fit the description stated in this question. Image A Image B State the names of the carbohydrates shown in image A and Image B. Describe the structure of these carbohydrates and explain how this structure permits their function. [9] Image A is a plant cell and contains starch grains and cellulose cell wall. Image B is of an animal cell which contains glycogen granules. Starch consists of two polysaccharides called amylose and amylopectin which are both polymers of α-glucose. Amylose is a coiled structure with only α-1,4 glycosidic bonds but amylopectin is branched and has both α-1,4 and α-1,6 glycosidic bonds. Starch is a storage of energy in plant cells. It can perform this function as it is insoluble and so cannot alter the water potential of the cell, is compact so large amount of starch can be stored as starch grains in the cell. The starch grains also prevent the starch form leaving the cell. In animal cells glycogen acts as the storage of energy. It s structure is identical to amylopectin in being a polymer of α-glucose and having both α-1,4 and α-1,6 glycosidic bonds.
4 However, glycogen is highly branched unlike amylopectin. Glycogen is a good storage polysaccharide for the same reasons as those for starch stated above, but glycogen forms glycogen granules. Cellulose is a structural polysaccharides and forms part of the cell wall of plants. It is a polymer of β-glucose which form long straight chains joined by β-1,4 glycosidic bonds. The β-glucose is rotated by 180 o so causing the glycosidic bonds to alternate up and down. Hydrogen bonds can form between the chains of β-glucose to form microfibrils. Microfibrils join together to form macrofibrils. The large number of hydrogen bonds in cellulose gives it a high tensile strength and makes it ridged and inelastic. These properties provide strength and support to plant cells. The inelastic nature of cellulose also causes plant cells to become turgid as the cytoplasm swells with water and pushes against the cell wall.
5 APPLICATION AND EXTENSION 1. Rare sugar syrup containing D -Allulose but not high-fructose corn syrup maintains glucose tolerance and insulin sensitivity partly via hepatic glucokinase translocation in Wistar rats. The above title is from a scientific paper published in the journal of agricultural and food chemistry (2017, 64, , Shintani et al). Below are extracts taken from this paper (some slightly modified). Using information in these extracts and your own knowledge answer the questions that follow. High-fructose corn syrup (HFCS) is commercially produced by isomerizing glucose to fructose. The taste of HFCS is similar to that of sucrose, and its price is affordable. Thus, HFCS has been used in various foods, such as beverages, desserts and confectionary and bakery products. However, the consumption of HFCS has been reported to be one of the risk factors for developing diabetes and obesity. From a preventative point of view it is advisable to reduce sugar intake or replace sugar with other low-calorie sweeteners. Thus, the development of new types of sweeteners is awaiting. D-Allulose is a C 3 epimer of D -fructose and is one of the rare sugars that are present in a limited quantity in nature. Studies have shown that D -allulose has various functions, such as reducing postprandial glucose elevation this has been confirmed in a number of studies and there have been two mechanisms proposed for the glucose lowering effect of D -allulose. These are: 1. The inhibition of α-glucosidase resulting in the decreased absorption of glucose. 2. The effect on the glycaemic response in the liver. D -Allulose promotes the conversion of blood glucose to glycogen in the liver via the use of
6 glucokinase. When glucose enters the hepatocytes, it is phosphorylated to glucose-phosphate by glucokinase which is a key step to forming glycogen. Thus, glucokinase increases the amount of glycogen in the liver, suppressing postprandial blood glucose elevation. D -Allulose has the effect of activating the glucokinase, causing its transport out of the nucleus and into the cytoplasm where it will then increase the amount of glycogen and thus lower the blood glucose level. Rare sugar syrup (RSS) is a mixture of 45% glucose, 29% fructose, 5% D - allulose and a number of other carbohydrates. Therefore, RSS containing D-allulose may activate glucokinase and lead to improved glycaemic control. The anti-obesity effect of RSS has been confirmed in both humans and animals in a clinical trial the results of which showed that the intake of 40g of RSS for 3 months lead to a 1.8Kg reduction in body weight compared to that observed after HFCS intake. It is of interest to assess whether RSS, a new sweetener derived from HFCS, maintains glucose tolerance and insulin sensitivity and, if so, whether anti-hyperglycaemic action of RSS is mediated by exporting glucokinase from the nucleus to the cytoplasm. To this end, glucose tolerance and the degree of glucokinase translocation in rats administered water, HFCS or RSS in drinking water were investigated. (a). State three controlled variables for this investigation. Same gender of Wistar rat, same age of Wistar rat, same concentration of RSS.
7 (b). Which of the following molecules are mentioned in the passage? A B C D E F A, B,C,D, E (c). What are the ethical issues of this investigation. The investigation uses rats. Rats may have been harmed in the investigation, e.g. been in pain. Results would not be usefull as the effects may be different in humans. (d). Explain why the statement glucokinase increases the amount of glycogen in the liver is scientifically inaccurate. Amount is the term meaning moles. The term concentration should have been used.
8 (e). Below is some selective data from this investigation (the data has been significantly modified for the purposes of this question). Figure 1. Effects of water, HFCS and RSS on glucose levels expressed as a percentage ± range. Figure 2. Effects of water, HFCS and RSS on Plasma glucose levels ± range.
9 Figure 3. Effects of water, HFCS and RSS on Plasma insulin levels ± range. Figure 4. Effects of water, HFCS and RSS on cytoplasmic glucokinase levels ± range. (a) Using figure 1 describe the trend for the effect of HFCS on glucose levels. At time 0 minutes the glucose concentration was 100%. The percentage of glucose gradually reduced to approximately 90% at 60 minutes. After 60 minutes the glucose percentage raised to approximately 80% at 180 minutes.
10 (ii) Explain which set of data: water, RSS or HFCS from figure 1 has the greatest consistency. RSS shows the greatest consistency because the range bars are very short and do not overlap. This means that the replicates results were close together. Due to this we have confidence in the trend and confidence that there is a true difference in the mean values. (iii) Insulin sensitivity is a measure of how sensitive the body is to the effects of insulin. Glucose tolerance is a measure of glucose levels in the blood. Using both figure 2 and figure 3 explain whether water, HFCS or RSS shows the greatest glucose tolerance and greatest insulin sensitivity. The blood glucose levels for RSS treatment are the lowest compared to water and HFCS in figure 2. In figure 3 RSS treatment caused the lowest levels of blood insulin. This lowest level of insulin caused the greatest reduction in blood glucose with RSS treatment indicating high insulin sensitivity and the greatest glucose tolerance was caused by RSS. (iv) It has been suggested that the polymerisation of glucose to glycogen is achieved by the action of glucokinase in the cytoplasm of hepatic cells and that the glucokinase levels in the cytoplasm are greatest with d -allulose present. Does the data presented in this question fully confirm this suggestion? Explain your answer. The data in figure 3 shows that the greatest percentage of cytoplasmic glucokinase is achieved by RSS which contains d -allulose. However, there is no data to support that its glucokinase that converts glucose to glycogen.
11 (v) Compare the structure of glycogen and starch. Starch is a polymer of α-glucose and so is glycogen. Glycogen is branched and so is amylopectin. Starch is made up of two different polysaccharides, but glycogen is made up of just one polysaccharide. (vi) Does the data disprove the prediction stated at the start of this question? Explain your answer. No, it does not disprove it. The data proves the prediction in that RSS with D-Allulose maintains glucose tolerance and insulin sensitivity due to the data in figure 2 and 3 as well as the data in figure 4 that shows the highest cytoplasmic kinase concentration.
12 Q2. Below is the structure of a carbohydrate called raffinose. Raffinose can be found in beans, cabbage, Brussel sprouts and other vegetables. Study this structure carefully and answer the following questions. (a) (i) What type of carbohydrate is raffinose? Trisaccharide (ii) State the names of the two bonds in raffinose including carbon numbers. α-1,6 and α 1,6 glycosidic bonds. (b) The enzyme α-galactosidase can breakdown raffinose but is not found in the human digestive tract. (i) What is the name of the reaction catalysed by α-galactosidase? Hydrolysis (ii) Place an arrow on the diagram of raffinose to show the site of action of α-galactosidase and state the names of the products of this reaction. Sucrose and galactose.
13 (c) The molar mass for raffinose is g/mol. You need to make up 5 standard solutions of concentration: 0.60 moldm -3, 0.45 moldm -3, 0.25 moldm -3, 0.15 moldm -3, 0.08 moldm -3. (i) Describe and calculate how you would make up the above 5 standard solutions. Each standard solution must be in a total volume of 100cm g/mol x 0.6 = g dissolved in 1 dm 3 = 0.6moldm -3. This is the stock solution that will be used to make the other concentrations. Its volume is 1dm 3 As the question asked for a total volume of 100cm 3 the V1xC1 = V2xC2 equation can be used to calculate the volumes needed to do the dilution. V1 and C1 are the concentration and volume of your stock solution and V2 and C2 are the concentration and volume of the solution that you require. Worked example for 0.45 moldm -3 So V1 = this is what we need to calculate, C1 = 0.6moldm -3 V2 = 100cm 3, C2 = 0.45moldm -3 We need to make V1 the subject of the equation: V1 = V2 C2 C1 V1 = 100cm3 0.45moldm 3 0.6moldm 3 = 75cm 3 So, we need 7.5cm 3 of the 0.6moldm -3 stock solution and add this to a volume of water to make a total volume of 100cm 3. The volume of water is calculated by 100cm 3 75cm 3 = 25cm 3.
14 The above stages are done for the other 3 standard solutions. The answers are: 0.25 moldm -3 V1 = 41.6cm 3, volume of water = 58.3cm moldm -3, V1 = 25cm 3, volume of water = 75cm moldm -3 V1 = 13.3cm 3, volume of water = 86.7cm 3 (ii) Explain why you would need to make up standard solutions. To calibrate an instrument and plot a calibration curve.
BIOLOGICAL MOLECULES 1
 Name: Option Group: BIOLOGICAL MOLECULES 1 GENERAL CHEMISTRY, CARBOHYDRATES, MATHS AND PRAC WORKBOOK You realise when you know how to think, because it empowers you far beyond those who know only what
Name: Option Group: BIOLOGICAL MOLECULES 1 GENERAL CHEMISTRY, CARBOHYDRATES, MATHS AND PRAC WORKBOOK You realise when you know how to think, because it empowers you far beyond those who know only what
SPECIFICATION CONTINUED Glucose has two isomers, α-glucose and β-glucose, with structures:
 alevelbiology.co.uk SPECIFICATION Monosaccharides are the monomers from which larger carbohydrates are made. Glucose, galactose and fructose are common monosaccharides. A condensation reaction between
alevelbiology.co.uk SPECIFICATION Monosaccharides are the monomers from which larger carbohydrates are made. Glucose, galactose and fructose are common monosaccharides. A condensation reaction between
Carbohydrates. Organic compounds which comprise of only C, H and O. C x (H 2 O) y
 Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates. 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds.
 Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
Biological Molecules
 SIM Tuition Biological Molecules I won t lie. This is probably the most boring topic you have ever done in any science. It s pretty much as simple as this: learn the material deal with it. Enjoy don t
SIM Tuition Biological Molecules I won t lie. This is probably the most boring topic you have ever done in any science. It s pretty much as simple as this: learn the material deal with it. Enjoy don t
Structural Polysaccharides
 Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
Carbohydrates. Monosaccharides
 Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
IB Biology BIOCHEMISTRY. Biological Macromolecules SBI3U7. Topic 3. Thursday, October 4, 2012
 + IB Biology SBI3U7 BIOCHEMISTRY Topic 3 Biological Macromolecules Essential Questions: 1.What are the 4 main types of biological macromolecules and what is their function within cells? 2.How does the
+ IB Biology SBI3U7 BIOCHEMISTRY Topic 3 Biological Macromolecules Essential Questions: 1.What are the 4 main types of biological macromolecules and what is their function within cells? 2.How does the
Waseem Abu Obeida. Salsabeel Fleifal. Mamoon Ahram
 8 Waseem Abu Obeida Salsabeel Fleifal Mamoon Ahram Anomers Anomers cyclic monosaccharides or glycosides that are epimers, they differ from each other in the configuration of C-1 if they are aldoses or
8 Waseem Abu Obeida Salsabeel Fleifal Mamoon Ahram Anomers Anomers cyclic monosaccharides or glycosides that are epimers, they differ from each other in the configuration of C-1 if they are aldoses or
Carbohydrates. Lecture2
 Carbohydrates Lecture2 Disaccharides Consist of two monosaccharides covalently bound to each other. All of which are isomers with the molecular formula C 12 22 O 11. The differences in these disaccharides
Carbohydrates Lecture2 Disaccharides Consist of two monosaccharides covalently bound to each other. All of which are isomers with the molecular formula C 12 22 O 11. The differences in these disaccharides
Definition of a Carbohydrate
 * Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
* Atoms held together by covalent bonds Definition of a Carbohydrate * Organic macromolecules * Consist of C, H, & O atoms * Usually in a 1:2:1 ratio of C:H : O Functions Performed by Carbohydrates Used
Carbs: The Staff of Life, or The Stuff of Death? Ed Cox, M.D.
 Carbs: The Staff of Life, or The Stuff of Death? Ed Cox, M.D. Pyramid, or Paleo? Carbs defined Carbohydrates (abbrev. CHO) = saccharides Saccharide from Greek for sugar Compounds of carbon, oxygen and
Carbs: The Staff of Life, or The Stuff of Death? Ed Cox, M.D. Pyramid, or Paleo? Carbs defined Carbohydrates (abbrev. CHO) = saccharides Saccharide from Greek for sugar Compounds of carbon, oxygen and
CLASS 11th. Biomolecules
 CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
Carbohydrates Dr. Ameerah M. Zarzoor
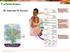 Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
Can you explain that monomers are smaller units from which larger molecules are made?
 Biological molecules Can you explain that all living things have a similar biochemical basis? Can you explain that monomers are smaller units from which larger molecules are made? Can you describe polymers
Biological molecules Can you explain that all living things have a similar biochemical basis? Can you explain that monomers are smaller units from which larger molecules are made? Can you describe polymers
Chapter 1. Chemistry of Life - Advanced TABLE 1.2: title
 Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
BIOCHEMISTRY UNIT 2 Part 4 ACTIVITY #4 (Chapter 5) CARBOHYDRATES
 AP BIOLOGY BIOCHEMISTRY UNIT 2 Part 4 ACTIVITY #4 (Chapter 5) NAME DATE PERIOD CARBOHYDRATES GENERAL CHARACTERISTICS: Polymers of simple sugars Classified according to number of simple sugars Sugars 3
AP BIOLOGY BIOCHEMISTRY UNIT 2 Part 4 ACTIVITY #4 (Chapter 5) NAME DATE PERIOD CARBOHYDRATES GENERAL CHARACTERISTICS: Polymers of simple sugars Classified according to number of simple sugars Sugars 3
Biochemistry: Macromolecules
 1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
Chemistry B11 Chapters 13 Esters, amides and carbohydrates
 Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic
 Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Topic 3: Molecular Biology
 Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
What are the most common elements in living organisms? What is the difference between monomers, dimers and polymers?
 What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
Bridging task for 2016 entry. AS/A Level Biology. Why do I need to complete a bridging task?
 Bridging task for 2016 entry AS/A Level Biology Why do I need to complete a bridging task? The task serves two purposes. Firstly, it allows you to carry out a little bit of preparation before starting
Bridging task for 2016 entry AS/A Level Biology Why do I need to complete a bridging task? The task serves two purposes. Firstly, it allows you to carry out a little bit of preparation before starting
Lecture 2 Carbohydrates
 Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
Disaccharides. Three Important Disaccharides Maltose, Lactose, and Sucrose. The formation of these three common disaccharides are:
 DISACCHARIDES Disaccharides Three Important Disaccharides Maltose, Lactose, and Sucrose The formation of these three common disaccharides are: 2 Disaccharides Maltose (Malt Sugar) Maltose is known as malt
DISACCHARIDES Disaccharides Three Important Disaccharides Maltose, Lactose, and Sucrose The formation of these three common disaccharides are: 2 Disaccharides Maltose (Malt Sugar) Maltose is known as malt
Carbohydrates suga. AP Biology
 Carbohydrates suga Carbohydrates energyo molecules C 2 O O O O O *4 Cal/gram Carbohydrates Carbohydrates are composed of C,, O carbo - hydr - ate C 2 O (C 12 2 O) x C 6 12 O 6 Function: energy energy storage
Carbohydrates suga Carbohydrates energyo molecules C 2 O O O O O *4 Cal/gram Carbohydrates Carbohydrates are composed of C,, O carbo - hydr - ate C 2 O (C 12 2 O) x C 6 12 O 6 Function: energy energy storage
Carbohydrates. Sugars, Starches, and Fibers. Chapter 4
 Carbohydrates Sugars, Starches, and Fibers Chapter 4 Introduction Brain Glucose Muscles Glucose Glycogen Fat Sources of carbohydrates Whole grains, vegetables, legumes, fruits, milk Fattening mistaken
Carbohydrates Sugars, Starches, and Fibers Chapter 4 Introduction Brain Glucose Muscles Glucose Glycogen Fat Sources of carbohydrates Whole grains, vegetables, legumes, fruits, milk Fattening mistaken
Molecular Structure and Function Polysaccharides as Energy Storage. Biochemistry
 1 1.Objectives Dr. Vijaya Khader Dr. MC Varadaraj To understand how polysaccharides act as energy source To understand the structure and energy generation process from glycogen To understand the structure
1 1.Objectives Dr. Vijaya Khader Dr. MC Varadaraj To understand how polysaccharides act as energy source To understand the structure and energy generation process from glycogen To understand the structure
Quiz 4 Review Guide Fall 2018
 Quiz 4 Review Guide Fall 2018 Major Topics: Enzyme Kinetics: o reaction rates and catalysis; transition state binding theory o Michaelis-Menten equation and interpretation o Inhibitors types and explanations
Quiz 4 Review Guide Fall 2018 Major Topics: Enzyme Kinetics: o reaction rates and catalysis; transition state binding theory o Michaelis-Menten equation and interpretation o Inhibitors types and explanations
CARBOHYDRATES 8/25/2014. Lesson Objectives. NUTR 2050 Nutrition for Nursing Professionals. Mrs. Deborah A. Hutcheon, MS, RD, LD.
 CARBOHYDRATES NUTR 2050 Nutrition for Nursing Professionals Mrs. Deborah A. Hutcheon, MS, RD, LD Lesson Objectives At the end of the lesson, the student will be able to: 1. Discuss the broad functions
CARBOHYDRATES NUTR 2050 Nutrition for Nursing Professionals Mrs. Deborah A. Hutcheon, MS, RD, LD Lesson Objectives At the end of the lesson, the student will be able to: 1. Discuss the broad functions
The building blocks for this molecule are A) amino acids B) simple sugars C) fats D) molecular bases
 1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS
 BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
The Structure and Func.on of Macromolecules: GRU1L4 Carbohydrates
 The Structure and Func.on of Macromolecules: GRU1L4 Carbohydrates Do Now: WHAT IS TABLE SUGAR MADE UP OF? Sucrose (table sugar) Composed of a glucose molecule and a fructose molecule Please draw the structure
The Structure and Func.on of Macromolecules: GRU1L4 Carbohydrates Do Now: WHAT IS TABLE SUGAR MADE UP OF? Sucrose (table sugar) Composed of a glucose molecule and a fructose molecule Please draw the structure
ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES Carbohydrate Chemistry
 I. General structures A. D-Aldoses ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
I. General structures A. D-Aldoses ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES
 I. General structures ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES A. D-Aldoses 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
I. General structures ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES A. D-Aldoses 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
Name a property of. water why is it necessary for life?
 02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
Dehydration Synthesis and Hydrolysis Reactions. ne_content/animations/reaction_types.ht ml
 Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
OCR (A) Biology A-level
 OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins
 I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
Ch13. Sugars. What biology does with monosaccharides disaccharides and polysaccharides. version 1.0
 Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
What Are Carbohydrates? Chapter 4 Carbohydrates: Simple Sugars and Complex Chains. Monosaccharides. Simple Sugars 6/17/16. BIOL 103, Spring 2016
 What Are Carbohydrates? Chapter 4 Carbohydrates: Simple Sugars and Complex Chains BIOL 103, Spring 2016 Sugars, starches, and fibers Major food sources: Plants Produced during photosynthesis Two main carbohydrate
What Are Carbohydrates? Chapter 4 Carbohydrates: Simple Sugars and Complex Chains BIOL 103, Spring 2016 Sugars, starches, and fibers Major food sources: Plants Produced during photosynthesis Two main carbohydrate
Ch. 5 Macromolecules. Overview: The Molecules of Life. Macromolecules BIOL 222. Macromolecules
 Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Macromolecules. Ch. 5 Macromolecules BIOL 222. Overview: The Molecules of Life. Macromolecules
 Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
The Chemistry of Carbohydrates
 Name Period Date The Chemistry of Carbohydrates Biologists today depend upon chemists for much of their understanding of life and life processes. Therefore, an understanding of some chemical concepts important
Name Period Date The Chemistry of Carbohydrates Biologists today depend upon chemists for much of their understanding of life and life processes. Therefore, an understanding of some chemical concepts important
1. Denaturation changes which of the following protein structure(s)?
 Chem 11 Fall 2008 Examination #5 ASWER KEY MULTIPLE CICE (20 pts. total; 2 pts. each) 1. Denaturation changes which of the following protein structure(s)? a. primary b. secondary c. tertiary d. both b
Chem 11 Fall 2008 Examination #5 ASWER KEY MULTIPLE CICE (20 pts. total; 2 pts. each) 1. Denaturation changes which of the following protein structure(s)? a. primary b. secondary c. tertiary d. both b
Topic 4 - #2 Carbohydrates Topic 2
 Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Haworth structure for fructose
 BIOMOLECULES carbohydrates may be defined as optically active polyhydroxy aldehydes or ketones or the compounds which produce such units on hydrolysis. Some of the carbohydrates, which are sweet in taste,
BIOMOLECULES carbohydrates may be defined as optically active polyhydroxy aldehydes or ketones or the compounds which produce such units on hydrolysis. Some of the carbohydrates, which are sweet in taste,
For more info visit
 Carbohydrates Classification of carbohydrates: Monosaccharides: Polyhydroxy aldehydes or polyhydroxy ketones which cannot be decomposed by hydrolysis to give simpler carbohydrates.examples: Glucose, Fructose,
Carbohydrates Classification of carbohydrates: Monosaccharides: Polyhydroxy aldehydes or polyhydroxy ketones which cannot be decomposed by hydrolysis to give simpler carbohydrates.examples: Glucose, Fructose,
January 31, Chemistry of Life. Carbohydrates. Lipids. Proteins. Biologically Important Macromolecules. Nucleic Acids
 Chemistry of Life Carbohydrates Lipids Proteins Biologically Important Macromolecules Nucleic Acids Polymers Polymers are large molecules of repeating sub units (building blocks) Individual Building Blocks......can
Chemistry of Life Carbohydrates Lipids Proteins Biologically Important Macromolecules Nucleic Acids Polymers Polymers are large molecules of repeating sub units (building blocks) Individual Building Blocks......can
Carbohydrates. Mark Scheme. Save My Exams! The Home of Revision. Exam Board 3.1 Biological Molecules Carbohydrates. Page 1.
 Carbohydrates Mark Scheme Level Subject Exam Board Module Topic Booklet A Level Biology AQA 3.1 Biological Molecules 3.1. Carbohydrates Mark Scheme Time Allowed: 59 minutes Score: /4 Percentage: /100 Grade
Carbohydrates Mark Scheme Level Subject Exam Board Module Topic Booklet A Level Biology AQA 3.1 Biological Molecules 3.1. Carbohydrates Mark Scheme Time Allowed: 59 minutes Score: /4 Percentage: /100 Grade
Lesson 2. Biological Molecules. Introduction to Life Processes - SCI 102 1
 Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Chemistry of Carbon. All living things rely on one particular type of molecule: carbon
 Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
Topic 2 Test B [40 marks]
![Topic 2 Test B [40 marks] Topic 2 Test B [40 marks]](/thumbs/80/82239443.jpg) Topic 2 Test B [40 marks] 1. The graph shows the effect of increasing the substrate concentration on the rate of an enzyme-catalysed reaction. What is occurring during the phase indicated by section Y
Topic 2 Test B [40 marks] 1. The graph shows the effect of increasing the substrate concentration on the rate of an enzyme-catalysed reaction. What is occurring during the phase indicated by section Y
2.1.1 Biological Molecules
 2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
Carbohydrate Chemistry 2016 Family & Consumer Sciences Conference Karin Allen, PhD
 Carbohydrate Chemistry 2016 Family & Consumer Sciences Conference Karin Allen, PhD Overview Carbohydrate chemistry General characteristics Sugar chemistry Starch chemistry 10 minute break Iodine test for
Carbohydrate Chemistry 2016 Family & Consumer Sciences Conference Karin Allen, PhD Overview Carbohydrate chemistry General characteristics Sugar chemistry Starch chemistry 10 minute break Iodine test for
Thursday, September 5. Why are carbohydrates important for us to consume?
 Bell Work: Thursday, September 5 Carbon Video Why are carbohydrates important for us to consume? 1 O 2 CARBON What is it...and WY is it important? Carbon the building block of life, basis for most molecules
Bell Work: Thursday, September 5 Carbon Video Why are carbohydrates important for us to consume? 1 O 2 CARBON What is it...and WY is it important? Carbon the building block of life, basis for most molecules
Chapter 18. Carbohydrates with an Introduction to Biochemistry. Carbohydrates with an Introduction to Biochemistry page 1
 Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Not long ago the world was caught up in an anti-carbohydrate craze.
 Carbohydrates Not long ago the world was caught up in an anti-carbohydrate craze. What was or is the problem? Why are carbohydrates so unpopular? Carbohydrates are sugar compounds that plants make when
Carbohydrates Not long ago the world was caught up in an anti-carbohydrate craze. What was or is the problem? Why are carbohydrates so unpopular? Carbohydrates are sugar compounds that plants make when
Carbohydrates. Mark Scheme 1. Save My Exams! The Home of Revision For more awesome GCSE and A level resources, visit us at
 Carbohydrates Scheme 1 Level A Level Subject Biology Exam Board Edexcel Topic Biological Molecules Sub Topic Carbohydrates Booklet Scheme 1 Time Allowed: 52 minutes Score: /43 Percentage: /100 Grade Boundaries:
Carbohydrates Scheme 1 Level A Level Subject Biology Exam Board Edexcel Topic Biological Molecules Sub Topic Carbohydrates Booklet Scheme 1 Time Allowed: 52 minutes Score: /43 Percentage: /100 Grade Boundaries:
Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol
 Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol (polyhydroxyaldehyde or polyhyroxyketone); their polymers,which
Dr. Entedhar Carbohydrates Carbohydrates are carbon compounds that have aldehyde (C-H=0) or ketone (C=O) moiety and comprises polyhyroxyl alcohol (polyhydroxyaldehyde or polyhyroxyketone); their polymers,which
Statement Starch Cellulose Glycogen glycosidic bonds present polymer of α-glucose unbranched chains only only found in plants
 1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
Wallington County Grammar School
 Wallington County Grammar School Y11 to Lower Sixth Bridging Work Subject: Subject Leader to direct questions to (email enquiries@wcgs.org.uk): Estimated hours of work needed to complete this work successfully:
Wallington County Grammar School Y11 to Lower Sixth Bridging Work Subject: Subject Leader to direct questions to (email enquiries@wcgs.org.uk): Estimated hours of work needed to complete this work successfully:
A BEGINNER S GUIDE TO BIOCHEMISTRY
 A BEGINNER S GUIDE TO BIOCHEMISTRY Life is basically a chemical process Organic substances: contain carbon atoms bonded to other carbon atom 4 classes: carbohydrates, lipids, proteins, nucleic acids Chemical
A BEGINNER S GUIDE TO BIOCHEMISTRY Life is basically a chemical process Organic substances: contain carbon atoms bonded to other carbon atom 4 classes: carbohydrates, lipids, proteins, nucleic acids Chemical
Tuesday 21 May 2013 Afternoon
 Tuesday 21 May 2013 Afternoon AS GCE UMAN BIOLOGY F221/01 Molecules, Blood and Gas Exchange *F210790613* Candidates answer on the Question Paper. OCR supplied materials: None Other materials required:
Tuesday 21 May 2013 Afternoon AS GCE UMAN BIOLOGY F221/01 Molecules, Blood and Gas Exchange *F210790613* Candidates answer on the Question Paper. OCR supplied materials: None Other materials required:
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud
 BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
ARE ALL SUGARS CREATED EQUAL?
 Fundamentals Module 21 ARE ALL SUGARS CREATED EQUAL? 2 SUGAR Sugars are carbohydrates, composed of carbon, hydrogen, and oxygen. Sugars come in a variety of forms, but can be distinguished easily (at least
Fundamentals Module 21 ARE ALL SUGARS CREATED EQUAL? 2 SUGAR Sugars are carbohydrates, composed of carbon, hydrogen, and oxygen. Sugars come in a variety of forms, but can be distinguished easily (at least
Organic Compounds. Biology-CP Mrs. Bradbury
 Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
not to be republished NCERT BIOMOLECULES CHAPTER 9 BIOMOLECULES 43 MULTIPLE CHOICE QUESTIONS
 BIOMOLECULES 43 43 CHAPTER 9 BIOMOLECULES MULTIPLE CHOICE QUESTIONS 1. It is said that elemental composition of living organisms and that of inanimate objects (like earth s crust) are similar in the sense
BIOMOLECULES 43 43 CHAPTER 9 BIOMOLECULES MULTIPLE CHOICE QUESTIONS 1. It is said that elemental composition of living organisms and that of inanimate objects (like earth s crust) are similar in the sense
Name: Class: Honors Biology Period: Question: What is the molecular formula of this molecule?
 Chapter 3: The Chemistry of Organic Molecules Exercise 1 Diversity of Carbon-Based Molecules (3.1) The great variety of organic compounds results from the ability of carbon atoms to bond with four other
Chapter 3: The Chemistry of Organic Molecules Exercise 1 Diversity of Carbon-Based Molecules (3.1) The great variety of organic compounds results from the ability of carbon atoms to bond with four other
GENERAL TESTS FOR CARBOHYDRATE. By Sandip Kanazariya
 GENERAL TESTS FOR CARBOHYDRATE By Sandip Kanazariya Introduction Carbohydrates are of great importance to human beings. They are major part of our diet, providing 60-70% of total energy required by the
GENERAL TESTS FOR CARBOHYDRATE By Sandip Kanazariya Introduction Carbohydrates are of great importance to human beings. They are major part of our diet, providing 60-70% of total energy required by the
3.9 Carbohydrates. Provide building materials and energy storage. Are molecules that contain carbon, hydrogen and oxygen in a 1:2:1 ratio
 3.9 Carbohydrates Provide building materials and energy storage Are molecules that contain carbon, hydrogen and oxygen in a 1:2:1 ratio Are of two main types Simple carbohydrates Complex carbohydrates
3.9 Carbohydrates Provide building materials and energy storage Are molecules that contain carbon, hydrogen and oxygen in a 1:2:1 ratio Are of two main types Simple carbohydrates Complex carbohydrates
!"#$%&'()*+(!,-./012-,345(
 (!"#$%&'()*+(!,-./012-,345( (!"#"$%&'()$*%#+,'(-(.+/&/*+,%&(01"2+34$5( 6%#+,"(!/$75#38+(92+41( CAPTER 20: Learning Objectives:! >
(!"#$%&'()*+(!,-./012-,345( (!"#"$%&'()$*%#+,'(-(.+/&/*+,%&(01"2+34$5( 6%#+,"(!/$75#38+(92+41( CAPTER 20: Learning Objectives:! >
Lecture Outline Chapter 4- Part 2: The Carbohydrates
 Lecture Outline Chapter 4- Part 2: The Carbohydrates I Types of Carbohydrates If someone told you "My carbohydrate intake is too high", what would you assume about what they're eating? A. SIMPLE CARBOHYDRATES:
Lecture Outline Chapter 4- Part 2: The Carbohydrates I Types of Carbohydrates If someone told you "My carbohydrate intake is too high", what would you assume about what they're eating? A. SIMPLE CARBOHYDRATES:
Carbohydrates. Chapter 12
 Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Chapter 4: Sugars, Starches and Fibers. Copyright 2012 John Wiley & Sons, Inc. All rights reserved.
 Chapter 4: Sugars, Starches and Fibers Copyright Student learning outcomes: At the end of this chapter, you should be able to: Compare and contrast whole grains and refined sugars Compare and contrast
Chapter 4: Sugars, Starches and Fibers Copyright Student learning outcomes: At the end of this chapter, you should be able to: Compare and contrast whole grains and refined sugars Compare and contrast
BIOCHEMISTRY NOTES Pre AP
 BIOCHEMISTRY NOTES Pre AP I. Chemistry study of what are made of and how they (text pages 35 43) A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron B.
BIOCHEMISTRY NOTES Pre AP I. Chemistry study of what are made of and how they (text pages 35 43) A. Atom fundamental unit of matter 1. Subatomic particles: n o = neutron p + = proton e - = electron B.
Carbohydrates- Disaccharides. By Dr. Bhushan R. Kavimandan
 Carbohydrates- Disaccharides By Dr. Bhushan R. Kavimandan Disaccharides ofbiological importance: Disaccharides consist of two monosaccharides joined by glycosidic linkages. They are crystalline, water-soluble
Carbohydrates- Disaccharides By Dr. Bhushan R. Kavimandan Disaccharides ofbiological importance: Disaccharides consist of two monosaccharides joined by glycosidic linkages. They are crystalline, water-soluble
2/25/2015. Chapter 6. Carbohydrates. Outline. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates
 Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Introduction to Macromolecules. If you were to look at the nutrition label of whole milk, what main items stick out?
 Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
Carbohydrates. Sugars. Monosaccharides. Fig. 3.23b(TE Art) 8/27/2014. Carbohydrates
 8/7/0 arbohydrates AIE BILGY JNES & FSBERY APTER arbohydrates arbohydrates are composed of,, carbo - hydr - ate ( ) x 6 6 Function: fast energy raw materials u energy storage u structural materials Monomer:
8/7/0 arbohydrates AIE BILGY JNES & FSBERY APTER arbohydrates arbohydrates are composed of,, carbo - hydr - ate ( ) x 6 6 Function: fast energy raw materials u energy storage u structural materials Monomer:
Chapter 20 Carbohydrates Chapter 20
 Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Carbon Compounds (2.3) (Part 1 - Carbohydrates)
 Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
I. ROLE OF CARBON IN ORGANISMS:
 Name: Period: Date: I. ROLE OF CARBON IN ORGANISMS: = compounds that contain carbon Ex: Carbohydrates, lipids, proteins = compounds that DO NOT contain carbon Ex: Vitamins, minerals, water Carbon forms
Name: Period: Date: I. ROLE OF CARBON IN ORGANISMS: = compounds that contain carbon Ex: Carbohydrates, lipids, proteins = compounds that DO NOT contain carbon Ex: Vitamins, minerals, water Carbon forms
BIOLOGICAL MOLECULES. Although many inorganic compounds are essential to life, the vast majority of substances in living things are organic compounds.
 BIOLOGY 12 BIOLOGICAL MOLECULES NAME: Although many inorganic compounds are essential to life, the vast majority of substances in living things are organic compounds. ORGANIC MOLECULES: Organic molecules
BIOLOGY 12 BIOLOGICAL MOLECULES NAME: Although many inorganic compounds are essential to life, the vast majority of substances in living things are organic compounds. ORGANIC MOLECULES: Organic molecules
Carbohydrates: The Energy Nutrient Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license.
 Carbohydrates: The Energy Nutrient 2001 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license. CARBOHYDRATES Functions of Carbohydrates 1. Energy 2.
Carbohydrates: The Energy Nutrient 2001 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning is a trademark used herein under license. CARBOHYDRATES Functions of Carbohydrates 1. Energy 2.
Tests for Carbohydrates
 Goals bserve physical and chemical properties of some common carbohydrates. Use physical and chemical tests to distinguish between monosaccharides, disaccharides, and polysaccharides. Identify an unknown
Goals bserve physical and chemical properties of some common carbohydrates. Use physical and chemical tests to distinguish between monosaccharides, disaccharides, and polysaccharides. Identify an unknown
AS BIOLOGY 7401/1R. Surname. Other Names. Centre Number. Candidate Number. Candidate Signature. Paper 1. Time allowed: 1 hour 30 minutes
 B Surname Other Names Centre Number Candidate Number Candidate Signature AS BIOLOGY Paper 1 7401/1R Thursday 26 May 2016 Afternoon Time allowed: 1 hour 30 minutes For this paper you must have: a ruler
B Surname Other Names Centre Number Candidate Number Candidate Signature AS BIOLOGY Paper 1 7401/1R Thursday 26 May 2016 Afternoon Time allowed: 1 hour 30 minutes For this paper you must have: a ruler
Chapter 22 Carbohydrates
 Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Topic 3.1 Nutrients. - Lipids are an essential part of the and are a part of cell in the body.
 Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Chapter 16: Carbohydrates
 Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
2-3 Carbon Compounds 10/22/2013. The Chemistry of Carbon. More Carbon. Chemistry (cont) More Macromolecules. Macromolecules
 The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds:
 Biochemistry Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic
Biochemistry Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic
CHEMISTRY OF LIFE 05 FEBRUARY 2014
 CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids
 9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids o o o Food is a good source of one or more of the following: protein,
9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids o o o Food is a good source of one or more of the following: protein,
Carbohydrates Chemical Composition and Identification
 Carbohydrates Chemical Composition and Identification Introduction: Today, scientists use a combination of biology and chemistry for their understanding of life and life processes. Thus, an understanding
Carbohydrates Chemical Composition and Identification Introduction: Today, scientists use a combination of biology and chemistry for their understanding of life and life processes. Thus, an understanding
BIOMOLECULES. Ms. Bosse Fall 2015
 BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
1. Substances in Cells
 1. Substances in Cells Students: Investigate cell requirements, including but not limited to: - Suitable forms of energy, including light energy and chemical energy in complex molecules. - Matter, including
1. Substances in Cells Students: Investigate cell requirements, including but not limited to: - Suitable forms of energy, including light energy and chemical energy in complex molecules. - Matter, including
