necessity for an investigation into possible different types of urine acidity. In
|
|
|
- Maximilian Marshall
- 5 years ago
- Views:
Transcription
1 456 J. Physiol. (I947) io6, I2.46i SOME FACTORS AFFECTING THE ACIDITY OF URINE IN MAN BY M. GRACE EGGLETON From the Department of Physiology, University College, London (Received 22 February 1947) The recent failure to fid any satisfactory explanation of the increase in urine acidity resulting from ingestion of alcohol (Eggleton, 1946) suggested the necessity for an investigation into possible different types of urine acidity. In the experiments with alcohol, the usual criterion of urine acidity, namely changes in hydrogen-ion concentration, was adopted. In such a buffered solution as urine, however, it is apparent that an increase in hydrogen-ion concentration need not necessarily be due entirely to an increase in excretion of acidic ions: a reduction in the overall buffering power of the solution would enable an unchanged excretion of acidic ions to produce a shift in hydrogen-ion concentration. Hitherto, little attention has been paid to changes in the total buffering capacity of the urine under various physiological conditions, interest being centred rather on the 'titratable acidity' value. This value is obtained by titration of the sample with standard alkali to about ph 8 (phenolphthalein first pink) and represents the buffering power of the urine over the range ph 8 to the ph at which it was secreted. It is found, naturally enough, to vary directly with the hydrogen-ion concentration, i.e. the lower the ph of the urine sample, the higher its 'titratable acidity' value. It gives no indication of the total buffering capacity of any sample over the whole physiological range ph 4*8-8*0 except for those samples secreted at ph 4X8. Approach from a completely different angle, that of determination of changes in output of phosphate, suggests that an increase in hydrogen-ion concentration of the urine may in some circumstances be associated with an increase in total buffering power. Thus Haldane (1921) noted an increase in phosphate excretion following ingestion of ammonium chloride in man, a result later confirmed by Loeb, Atchley, Richards, Benedict & Driscoll (1932), while Gamble, Ross & Tisdall (1923) observed a similar result after fasting, the increase in phosphate output being greater than that accounted for by wastage of the muscles. Under both sets of conditions, ammonium chloride acidosis and fasting, the prime need of the body is to rid itself of excess acidic ions: by
2 URINE ACIDITY IN MAN 457 simultaneously increasing the buffering power of the urine, this end can be achieved without increasing the hydrogen-ion concentration beyond the physiological limits to which the kidney is accustomed. The results to be presented are concerned with experiments designed to test the possible existence of two distinct types of urine acidity on the lines suggested above. METHODS The general procedure used in previous investigations was adhered to, with only slight modifications. The meal previous to the experiment, either breakfast or lunch, was omitted, whether the experimental substance was to be ingested or injected, and a glass of water was usually taken 2-2j hr. before the experiment began. At least one urine sample was collected during the last min. of this resting period, and five or six samples during the 2-3 hr. following the experimental injection or ingestion. A large number of experiments were performed on one subject and these were repeated, whenever feasible, on groups of other subjects. In addition to determinations of ph and ammonia concentration (formol titration method) as detailed elsewhere (Eggleton, 1946), the buffering power of the urine was determined. To the 'titratable acidity' value (urine titrated to ph 8 0) was added what might reasonably be called the 'titratable alkalinity' value (urine titrated to ph 4-8, using methyl red-methylene blue indicator), the sum of the two representing the buffering of the urine over the physiological range ph Neither titration is extremely accurate, owing partly to the yellow pigmentation of the urine. This inaccuracy was partially counteracted, and reasonably large titration values obtained even on dilute urine samples, by the use of volumes varying from c.c. according to the rate of urine flow, the smaller samples being diluted to 50 c.c. in each case. In a few experiments determinations were also made of phosphate, output. RESULTS The results obtained by Haldane and his successors in regard to the action of acidifying agents such as ammonium chloride were fully confirmed. Two experiments on one subject and one experiment on a second subject were made with ammonium chloride, and one experiment on each of three subjects with ammonium sulphate. In all six a result similar to that shown in Fig. 1 was obtained. Since these substances can be ingested only in dilute solution without inducing vomiting, and since it was desirable, for a reason to be explained later, that they should be taken with a minimum amount of water, they were ingested in the form of enteric-coated capsules. It is clear that, under such conditions of acidosis, a fall in urine ph is accompanied by a pronounced increase in output of buffering substances, of which phosphate accounts for %. An attempt was made at a later date to give an intravenous injection of hypertonic ammonium sulphate (20% solution) but proved fruitless: nervous symptoms (incipient black-out) and thrombosis occurred after the first c.c. had been injected. Under all other conditions in which a change in urine ph was observed, a relationship, the exact converse of that shown in Fig. 1, was seen to exist. In Fig. 2, for example, is shown the effect of an intravenous injection of hypertonic sucrose. As the urine acidity increases, the buffer output falls. This experiment was not attempted on any further subjects since it induced
3 458 M. GRACE EGGLETON Fig. 1. The effect of ingestion of 20 g. ammonium sulphate (40 enteric-coated capsules) on urine ph, output of phosphate and total buffers v Buffer outpul \ (m.equiv./hr.) -Urine D5 Fig c. 50% sucrose intravenously The effect of intravenous injection of concentrated sucrose solution on urine ph and buffer output.
4 thrombosis. URINE ACIDITY IN MAN 459 Hypertonic sodium sulphate, however, was found to produce no ill effects and the average results of an intravenous injection of this substance into four subjects is shown in Fig. 3. Again, the buffer output falls as the urine acidity increases. In a fifth subject the ph fell from 5-6 to 4-8 following the injection, but began to rise again after min., and the result, therefore, was not included with those of the remaining four who each individually showed the more prolonged fall seen in Fig. 3. In this figure also is shown the Buffer NH3 ~~~~~~~~~~~~~ /OAmmonia otutput \ / (m.equiv.jhr.) ph 2-0 *.../ / ~~~~~ _ O 16-6; Buffer output 1-5 d *1(mequiv.fhr.) 6-2 Fig ±5- \1Urine ph 2-0 Urine Now t c.c. I0%NO a2sot Min intravenously The effect of intravenous injection of hypertonic sodium sulphate solution on urine ph, buffer output and ammonia output. Average results from four subjects. inverse relationship which is regularly observed between the outputs of buffer substances and ammonia, a result confirming that previously established between urine ph and ammonia output in alcohol diuresis (Eggleton, 1946). In contrast with the increase in urine acidity and decrease in buffer output induced by intravenous injection of hypertonic solutions of sucrose or sodium sulphate are the effects, shown in Fig. 4, of the administration by mouth of hypertonic urea solution (the average results of five subjects), but again it is seen that urine ph and buffer output run hand in hand. In one subject an attempt was made to give a rapid intravenous injection of 20% solution of urea; this procedure, however, was not only followed by thrombosis but was so painful that the somewhat erratic course of both urine ph and buffer output which resulted was discounted as being probably connected with the initial nervous disturbance: nor was the experiment repeated on any other subjects. PH. CVI
5 460 M. GRACE EGGLETON Ingestion of hypertonic solutions does not necessarily lead to the results shown in Fig. 4. On one occasion, administration of 200 g. glucose in 200 c.c. solution resulted in a fall of urine ph from 6-6 to 4-8, which persisted for the following 2 hr., during most of which time glycosuria was present and the rate of urine flow never greater than 0*63 c.c./min. The experiment was performed with another end in view and buffer output was not determined; the results, therefore, do not merit more than passing mention. The relationship between buffer output and urine acidity demonstrated in the preceding figures has been observed in some thirty to forty subjects under a variety of conditions, including the naturally occurring variations in acidity 2.6 oo 284.'\ o 22 * 0 2.6/X Buffer output (m.equiv./hr.) 2 + 4= UrinepH flow Fig g. urea by mouth Miin. (in 200c.c.) The effect of hypertonic urea solution taken by mouth on urine ph and buffer output. Average results from five subjects. encountered during the course of the day. Two factors may, however, mask this relationship at times. Ingestion of tea or coffee results in a disproportionately great output of buffer substances in relation to the ph, but this factor has been eliminated in the series of experiments under consideration. The second factor is that of diuresis itself. A hint of this is to be seen in Figs. 3 and 4 and is shown in pronounced form in Fig. 5. These results again are the average from four subjects who on one occasion took water alone and on another day water, immediately preceded by subcutaneous injection of 1 unit 'pituitrin'. Considerable variations in urine acidity occurred in the different subjects, but in each individual the curve of buffer output followed fairly closely that of urine ph when the rate of urine flow was low. In the water diuresis, however, the large increase in rate of flow, accompanied by a slight fall in urine ph, induced a large increase in buffer output.
6 URINE ACIDITY IN MAN 461 This 'flushing-out' effect on buffer substances at high rates of urine flow is sufficiently great to mask the increased acidity following injection of hypertonic sodium sulphate solutions under certain conditions. Two experiments were performed with a different technique from that used on earlier occasions, in an effort to reduce the lag in time between collection of the resting urine sample and injection of the solution; also, the solution was more concentrated (100 c.c. of 20% sodium sulphate) and given in a shorter time in the hope that a more dramatic change in urine acidity might be observed. But these precautions defeated their own ends. In one subject the rate of urine flow rose to 2- Buffer output + 26 (m.equiv./hr) \ * 48 wturine ph itu6-2 Fig. 5. TheeffectrUa rine o flow t, ~~~~(c.c./min.) L It 90 12_ 5 S560c.c. water -or Min. wae + I unit pituitrin Fig. 5. The effect of rate of urine flow on buffer output. Average results from four subjects c.c. water;. --- * 560 c.c. water preceded by 1 unit 'pituitrin' subcutaneously. 7 c.c./min. and in the other to 4-7 c.c./min. following the injection, and in both subjects this relatively large flow was accompanied by an increase in buffer output and no detectable change in urine ph (7-25 and 7-1 respectively in the two subjects). Incidentally, both subjects noted a sensation of thirst before the end of the injection and both developed a severe headache. The latter symptom had also been observed previousry in a subject (probably a mild case of diabetes insipidus) in whom both alcohol and water produced a rapid shift in body water, as indicated by an unusually large and speedy diuresis. Ammonia excretion. Earlier results (Eggleton, 1946) indicated that the output of ammonia is fairly closely correlated with urine ph, the ammonia rising 30-2
7 462 M. GRACE EGGLETON as the ph falls: and that, although this ammonia output is affected to some extent by varying rate of urine flow, no definite relationship exists between ammonia concentration and urine ph. Those conclusions were drawn from experiments on one individual, and it has been deemed advisable to extend the observations by experiments on a group of other subjects and in reference to urine acidity induced by a number of different agencies. The results already presented in Fig. 3 show the relationship between ammonia output and urine ph in a group of four subjects after injection of sodium sulphate. Ammonia concentration can readily be calculated from the data given in the figure, and shows no consistent relationship with the ph; s.f(17) i.i.(27) >~~~~~~~~ +(33) 5.5 (24) I+0(7)_ Ammonia output (m.equiv./hr.) Fig. 6. The relationship between ammonia output and urine pe in one subject, under a variety of conditions. Average values are given for each half unit of ph. The numbers in brackets indicate the number of observations included in each average: the distance between the small vertical lines the standard error of each average. Rate of urine flow varied fifty-fold, from 025 to 11.0 c.c./min. in different experiments. during the stage of increased rate of urine flow, the ammonia concentration falls as the ph falls (from 21 to 15 m.equiv./l.) and during the later stage of diminishing rate of flow, the concentration rises considerably (from 33-5 to 46 m.equiv./l.) as the ph changes only from 5-15 to 5 0. In thirteen out of thirty subjects on whom at least two experiments have been performed, this relationship between ammonia output and urine ph has been observed. In regard to the remaining seventeen subjects, no such definite conclusion could be drawn. Considerable variation was noted among the whole group of subjects in the extent to which rate of urine flow affected the ammonia output, and only a more intensive study of each subject over a wide range of ph and rate of flow could decide which in each is the more fundamental relationship.
8 URINE ACIDITY IN MAN 463 In the subject, for whom data have already been presented from experiments with water and alcohol diuresis (Eggleton, 1946), it has been found that the relation between ammonia output and urine ph remains unaffected when changes in ph are induced by ingestion of urea, or injection of urea, sucrose or sulphate. This relationship, obtained from a large number of experiments of different types, is shown in Fig. 6 in the form of average values for each half unit of ph. The values for ammonia were all obtained by use of the formol titration method, so that the curve for true ammonia would lie about 0-2 m.equiv./hr. to the left of that given. The results of the experiments in which acidifying agents were ingested have not been included in the averages shown in Fig. 6, for under those conditions the ammom"a output is greatly in excess of the values found under other conditions of urine acidity. In the experiment illustrated in Fig. 1, for example, the ammonia output reached a value of over 3 m.equiv./hr. DISCUSSION The main point of interest in the results presented is the practical one-that increased urine acidity (hydrogen-ion concentration) may in some circumstances be associated with an increase, and in other circumstances with a decrease in output of buffer substances. The former of these two conditions has been recognized by many workers, but attention has not previously been called to the latter, which appears to be of more general occurrence. This differentiation between two distinct, types of urine acidity argues against Briggs's theory (1934) that the acidifying action of sodium sulphate is due to preferential excretion of the foreign sulphate ion. If that were the case, the increased acidity should be accompanied by an increase in buffer output as with ammonium sulphate, but the reverse is observed. The fact that sodium sulphate and sucrose, both slowly diffusing substances, lead to an increased acidity of the urine whereas rapidly diffusing urea has no such action suggests that the acidity may be connected with dehydration of the tissues in general. It is tempting to speculate that the post-pituitary antidiuretic hormone may itself be concerned, though the experiments reported here provide no direct evidence on the point. The relatively small diuresis resulting from injection of sodium sulphate or sucrose (although both should be strong osmotic diuretics) as compared with the larger diuresis after ingestion of urea suggests that secretion of the anti-diuretic hormone has been stimulated by the former. This interpretation is fully in accord with Verney's results (1946) on the unanaesthetized dog; intra-carotid injection of hypertonic urea was found to be without effect on a water diuresis whereas hypertonic sucrose, glucose or NaCl produced immediate inhibition. The result of injections of 'pituitrin' might also be cited as additional evidence in favour of this interpretation. The average results of four subjects, given in Fig. 5, show no large
9 464 M. GRACE EGGLETON variations, either in rate of urine flow or in urine acidity, owing to individual variations in the degree of response to water plus 'pituitrin', but in each subject the urine ph followed closely the changes in rate of flow. If this tentative hypothesis be accepted at the moment for lack of a better one, it should be stressed that the acidifying action of 'pituitrin' cannot override an alkalosis of the body. In two subjects to whom 10 g. potassium citrate had been administered, the resulting diuresis was partially counteracted by subcutaneous injection of 1 unit of 'pituitrin'. Under these conditions the diminished rate of urine flow was accompanied by a decrease in acidity: the ph, which had fallen to 7-5 at the height of diuresis, returned to ph 8-0 after the 'pituitrin' injection. Any attempt at a theoretical consideration of the results from the renal point of view at first seemed impossible, since the excretion of individual buffer substances, and carbon dioxide partial pressures, had not been determined. Some further analysis can, however, be made and I am indebted to Dr L. E. Bayliss for surveying the results and presenting the following interpretation. The rate of acid or alkali excretion may be readily calculated from the data in this paper if some assumptions are made as to the initial hydrogen-ion concentration of the glomerular fluid, and as to the changes in hydrogen-ion concentration that would result from the re-absorption of water alone. It is reasonable to suppose that the glomerular fluid has a ph of about 7-4, and that the increase in ionic strength, and reduced activity of the water, 'during elaboration of the urine will not change this by more than 0.1 ph, but this requires further investigation. The amount of buffer substances excreted, as given in this paper, is measured as the amount of acid required to change the ph of the urine from 4-8 to 8-0, i.e. by 3-2 ph units. As a first approximation, therefore, the amount of acid that must have been added by the tubules, or alkali removed, to bring the urine to any observed value of ph is given by (744-pH) x (buffer output) 3-2 (it is assumed that the titration curve of urine is a straight line, which is justifiable over the range of ph considered). It may be remarked in parenthesis that, if this viewpoint be adopted, the rate of acid excretion can be directly measured by titrating the urine to ph 7.4, the titre then giving the acid output, and this, divided by (7-4 minus ph of urine as excreted) giving the buffer value. Calculation of the rate of acid excretion by this method leads to the following conclusions: (1) Administration of ammonium sulphate led to a considerable increase in the acid output (approximately three-fold), as is obvious from the simultaneous rise in buffer output and fall in ph; this increase was maintained after the diuresis had subsided.
10 URINE ACIDITY IN MAN 465 (2) Administration of sucrose or sodium sulphate also led to an increase in acid output (by %); this acid output fell as the diuresis subsided, but less rapidly. The two types of response to administration of sodium sulphate in respect to changes in ph, the one shown in Fig. 3 (four subjects) and the other in a fifth subject mentioned in the text, had the same type of response in respect to acid output; it is the excretion of buffer substances that behaved differently. (3) Administration of urea led to no appreciable change in the output of acid, the fluctuations in ph being due entirely to fluctuations in buffer output. (4) During simultaneous administration of water and pituitary extract, there were small and irregular changes in acid output, which were associated with inverse changes in the rate of urine flow. This association broke down on the onset of water diuresis, and in one subject in which the urine went alkaline to ph 7-2. SUMMARY 1. The increased acidity of the urine after ingestion of ammonium sulphate or chloride is accompanied by an increase in buffer output (titration range ), of which phosphate accounts for about two-thirds (Fig. 1). 2. Intravenous injection of hypertonic sucrose or sodium sulphate solutions also results in increased acidity of the urine, but this is accompanied by a decrease in buffer output (Figs. 2 and 3). 3. The more pronounced diuresis resulting from ingestion of hypertonic urea solution is accompanied by a decrease in urine acidity, with increase in buffer output (Fig. 4). 4. This inverse relationship between urine acidity (hydrogen-ion concentration) and buffer output may be masked at high rates of urine flow by a 'flushing-out' effect on buffer substances (Fig. 5). 5. Evidence is adduced in favour of the tentative hypothesis that increase in urine acidity, accompanied by decrease in buffer output, may be connected with secretion of anti-diuretic hormone by the post-pituitary. 6. A direct relationship is found between urine acidity and ammonia output under all conditions (Fig. 6). I wish to thank Dr R. A. Gregory and Dr D. R. Wilkie for their generous help, during the earlier and later stages of this research respectively, in giving all the intravenous injections. I am greatly indebted to Dr J. W. Trevan for a supply of enteric-coated capsules prepared at the Wellcome Laboratories. REFERENCES Briggs, A. P. (1934). J. biol. Chem. 104, 231. Eggleton, M. Grace (1946). J. Physiol. 104, 312. Gamble, J. L., Ross, S. G. & Tisdall, F. F. (1923). J. biol. Chem. 57, 633. Haldane, J. B. S. (1921). J. Phy8iol. 55, 265. Loeb, R. F., Atchley, D. W., Richards, D. W. Jr., Benedict, E. M.- & Driscoll, M. E. (1932). J. clin. Inve8t. 11, 621. Verney, E. B. (1946). Lancet, no. 251, p. 781.
(ethanol) suggests that it is similar to the diuresis following ingestion of water.
 435 J. Physiol. (I946) I04, 435-442 6I2.464.I THE EFFECT OF ETHYL ALCOHOL AND SOME OTHER DIURETICS ON CHLORIDE EXCRETION IN MAN BY M. GRACE EGGLETON AND ISABEL G. SMITH, From the Physiology Department,
435 J. Physiol. (I946) I04, 435-442 6I2.464.I THE EFFECT OF ETHYL ALCOHOL AND SOME OTHER DIURETICS ON CHLORIDE EXCRETION IN MAN BY M. GRACE EGGLETON AND ISABEL G. SMITH, From the Physiology Department,
estimates were made of the normal rate of increase in plasma urea over periods in skin and in plasma, hypertonic sodium chloride solution was
 482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
simultaneously excreted. They also brought forward some evidence to
 THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
GLUCOSE is the most important diffusible substance in the blood which
 ON THE ACTION OF PHLORHIZIN ON THE KIDNEY. By E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) GLUCOSE is the most important diffusible substance in the blood which is completely held back
ON THE ACTION OF PHLORHIZIN ON THE KIDNEY. By E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) GLUCOSE is the most important diffusible substance in the blood which is completely held back
excreted, in spite of its constant presence in the blood. Similarly, a salt-free diet will rapidly cause the practical disappearance of chlorides
 THE REGULATION OF EXCRETION OF WATER BY THE KIDNEYS. I. By J. S. HALDANE, M.D., F.R.S. AND J. G. PRIESTLEY, B.M., Captain R.A.M.C., Beit Memorial Research Fellow. NUMEROUS observations tend to show that
THE REGULATION OF EXCRETION OF WATER BY THE KIDNEYS. I. By J. S. HALDANE, M.D., F.R.S. AND J. G. PRIESTLEY, B.M., Captain R.A.M.C., Beit Memorial Research Fellow. NUMEROUS observations tend to show that
capillaries, and a consequent increased transudation, without necessarily altering to any marked extent the total circulation of blood
 612.463.4 THE CONTROL OF THE GLOMERULAR PRESSURE BY VASCULAR CHANGES WITHIN THE ISOLATED MAMMALIAN KIDNEY, DEMONSTRATED BY THE ACTIONS OF ADRENALINE. BY F. R. WINT0N (Beit Memorial Research Fellow). (Depaortment
612.463.4 THE CONTROL OF THE GLOMERULAR PRESSURE BY VASCULAR CHANGES WITHIN THE ISOLATED MAMMALIAN KIDNEY, DEMONSTRATED BY THE ACTIONS OF ADRENALINE. BY F. R. WINT0N (Beit Memorial Research Fellow). (Depaortment
for Medical Research. (Received May 10th, 1922.)
 XLV. NOTE ON URINARY TIDES AND EXCRETORY RHYTHM. BY JAMES ARGYLL CAMPBELL AND THOMAS ARTHUR WEBSTER. From the Department of Applied Physiology, National Institute for Medical Research. (Received May 10th,
XLV. NOTE ON URINARY TIDES AND EXCRETORY RHYTHM. BY JAMES ARGYLL CAMPBELL AND THOMAS ARTHUR WEBSTER. From the Department of Applied Physiology, National Institute for Medical Research. (Received May 10th,
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
principles. laboratory [Stehle & Fraser, 1935] and contains 200 pressor units and (Received 20 November 1940)
![principles. laboratory [Stehle & Fraser, 1935] and contains 200 pressor units and (Received 20 November 1940) principles. laboratory [Stehle & Fraser, 1935] and contains 200 pressor units and (Received 20 November 1940)](/thumbs/92/110006249.jpg) .#Lil-RAFY 4 233 J. Physiol. (I94I) IOO, 233-238 4 V>6x2.492.8:577.I52 I THE RATIO BETWEEN ANTIDIURETIC AND PRESSOR ACTIVITIES OF POSTERIOR PITUITARY EXTRACT SUBJECTED TO MILD HYDROLYSIS BY A. M. FRASER
.#Lil-RAFY 4 233 J. Physiol. (I94I) IOO, 233-238 4 V>6x2.492.8:577.I52 I THE RATIO BETWEEN ANTIDIURETIC AND PRESSOR ACTIVITIES OF POSTERIOR PITUITARY EXTRACT SUBJECTED TO MILD HYDROLYSIS BY A. M. FRASER
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
PHOSPHORUS CONTENT OF THE BLOOD IN DIABETES
 10 BLOOD PHOSPHORUS IN HEALTH AND DISEASE: IV-THE PHOSPHORUS CONTENT OF THE BLOOD IN DIABETES MELLITUS F B BYROM From the Dunn Laboratories, London Hospital Received for publication November 24th, 1928
10 BLOOD PHOSPHORUS IN HEALTH AND DISEASE: IV-THE PHOSPHORUS CONTENT OF THE BLOOD IN DIABETES MELLITUS F B BYROM From the Dunn Laboratories, London Hospital Received for publication November 24th, 1928
following experiments were designed to show the effects of changes in CO2 combining power in the blood of dogs after the administration of acid
 OBSERVATIONS ON THE FORMATION OF WHEALS V. THE EFFECTS OF VARIATION OF THE CO2 COMBINING POWER OF THE BLOOD ON HISTAMINE WHEALS By F. S. McCONNELL, W. K. WEAVER AND H. L. ALEXANDER (From the Department
OBSERVATIONS ON THE FORMATION OF WHEALS V. THE EFFECTS OF VARIATION OF THE CO2 COMBINING POWER OF THE BLOOD ON HISTAMINE WHEALS By F. S. McCONNELL, W. K. WEAVER AND H. L. ALEXANDER (From the Department
Medicine, Cambridge, England, and Wuppertal, B.A.O.R.
 182 J. Physiol. (I948) I07, i82-i86 6I2.46I.62 PHOSPHATE CLEARANCES IN INFANTS AND ADULTS BY R. F. A. DEAN AND R. A. McCANCE From the Medical Research Council, Department. of Experimental Medicine, Cambridge,
182 J. Physiol. (I948) I07, i82-i86 6I2.46I.62 PHOSPHATE CLEARANCES IN INFANTS AND ADULTS BY R. F. A. DEAN AND R. A. McCANCE From the Medical Research Council, Department. of Experimental Medicine, Cambridge,
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
(From the Departments of Physiology, Pharmacology and Anatomy, State University of Iowa, Iowa City) (Received for publication, October 18, 1937)
 SERUM SODIUM, POTASSIUM AND CHLORIDE AFTER SUPRARENALECTOMY IN CATS WITH DIABETES INSIPIDUS BY CHARLES A. WINTER, PH.D., E. G. GROSS, M.D., AND W. R. INGRAM, P~t.D. (From the Departments of Physiology,
SERUM SODIUM, POTASSIUM AND CHLORIDE AFTER SUPRARENALECTOMY IN CATS WITH DIABETES INSIPIDUS BY CHARLES A. WINTER, PH.D., E. G. GROSS, M.D., AND W. R. INGRAM, P~t.D. (From the Departments of Physiology,
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES
 HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES TERUO NAKAYAMA* Institute of Physiology, School of Medicine, University of Nagoya It is known that electrical
HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES TERUO NAKAYAMA* Institute of Physiology, School of Medicine, University of Nagoya It is known that electrical
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
HUMAN SUBJECT 1. Syracuse, N. Y.) the urine of increasing quantities of these buffers, it has been found in man as in the dog that (1)
 THE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. II. FACTORS AFFECTING THE EXCRETION OF TITRATABLE ACID BY THE NORMAL HUMAN SUBJECT 1 By W. A. SCHIESS, J. L. AYER, W. D. LOTSPEICH AND R. F. PITTS WITH
THE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. II. FACTORS AFFECTING THE EXCRETION OF TITRATABLE ACID BY THE NORMAL HUMAN SUBJECT 1 By W. A. SCHIESS, J. L. AYER, W. D. LOTSPEICH AND R. F. PITTS WITH
hold for the human kidney.2 Shannon and Smith (4) have rightfully stressed
 THE RENAL EXCRETION OF INULIN AT LOW PLASMA CONCEN- TRATIONS OF THIS COMPOUND, AND ITS RELATIONSHIP TO THE GLOMERULAR FILTRATION RATE IN NORMAL, NEPHRITIC AND HYPERTENSIVE INDIVIDUALS' By BENJAMIN F. MILLER,
THE RENAL EXCRETION OF INULIN AT LOW PLASMA CONCEN- TRATIONS OF THIS COMPOUND, AND ITS RELATIONSHIP TO THE GLOMERULAR FILTRATION RATE IN NORMAL, NEPHRITIC AND HYPERTENSIVE INDIVIDUALS' By BENJAMIN F. MILLER,
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
entirely by glomerular filtration and was neither reabsorbed nor secreted
 6I2.46I.63 INORGANIC SULPHATE EXCRETION BY THE HUMAN KIDNEY. BY CUTHBERT L. COPE. (From the Biochemistry Department, Oxford, and the Radcliffe Infirmary, Oxford.) IN putting forward his modern theory of
6I2.46I.63 INORGANIC SULPHATE EXCRETION BY THE HUMAN KIDNEY. BY CUTHBERT L. COPE. (From the Biochemistry Department, Oxford, and the Radcliffe Infirmary, Oxford.) IN putting forward his modern theory of
Use the following diagram to answer the next question. 1. In the diagram above, pressure filtration occurs in a. W b. X c. Y d. Z
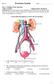 Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
Part A: Multiple Choice Questions Value: 32 Marks Suggested time: 40 minutes Instructions: For each question select the best answer and record your choice on the Scantron card provided. Using an HB pencil,
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
McCANCE, From The Department of Medicine, Cambridge
 196 J. Physiol. (I945) I04, I96-209 6I2.4632.2 THE EXCRETION OF UREA, SALTS AND WATER DURING PERIODS OF HYDROPAENIA IN MAN BY R. A. McCANCE, From The Department of Medicine, Cambridge (Received 20 February
196 J. Physiol. (I945) I04, I96-209 6I2.4632.2 THE EXCRETION OF UREA, SALTS AND WATER DURING PERIODS OF HYDROPAENIA IN MAN BY R. A. McCANCE, From The Department of Medicine, Cambridge (Received 20 February
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
technique by Hemingway [1931] makes it possible to
![technique by Hemingway [1931] makes it possible to technique by Hemingway [1931] makes it possible to](/thumbs/90/101795941.jpg) 6I2.464 THE ACTION OF CYANIDE ON THE ISOLATED MAMMALIAN KIDNEY. BY L. E. BAYLISS' AND E. LUNDSGAARD2 (Copenhagen). (From the Department of Physiology and Biochemistry, University College, London.) THE
6I2.464 THE ACTION OF CYANIDE ON THE ISOLATED MAMMALIAN KIDNEY. BY L. E. BAYLISS' AND E. LUNDSGAARD2 (Copenhagen). (From the Department of Physiology and Biochemistry, University College, London.) THE
Cutler, Power & Wilder, 1938; Hall & Langley, 1940), in the dog (Winkler &
 8 J. Physiol. (I948) I07, 8-I3 6I2.46I.6 RENAL EXCRETION OF SODIUM AND POTASSIUM IN RATS BY S. E. DICKER (Beit Memorial Fellow) From the Department of Pharmacology, University of Bristol (Received 30 December
8 J. Physiol. (I948) I07, 8-I3 6I2.46I.6 RENAL EXCRETION OF SODIUM AND POTASSIUM IN RATS BY S. E. DICKER (Beit Memorial Fellow) From the Department of Pharmacology, University of Bristol (Received 30 December
(Received 20 March 1940)
 336 J. Physiol. (I940) 98, 336-360 612.464. I THE MECHANISMS OF DILUTION DIURESIS IN THE ISOLATED KIDNEY AND THE ANAESTHETIZED DOG BY M. GRACE EGGLETON,1 J. R. PAPPENHEIMER AND F. R. WINTON Departments
336 J. Physiol. (I940) 98, 336-360 612.464. I THE MECHANISMS OF DILUTION DIURESIS IN THE ISOLATED KIDNEY AND THE ANAESTHETIZED DOG BY M. GRACE EGGLETON,1 J. R. PAPPENHEIMER AND F. R. WINTON Departments
(Received 27 September 1937)
 222 J. Physiol. (I937) 9I, 222-23I 6I2.46I:6I2.392.6 THE SECRETION OF URINE IN MAN DURING EXPERIMENTAL SALT DEFICIENCY BY R. A. McCANCE AND E. M. WIDDOWSON From the Biochemical Laboratory, King's College
222 J. Physiol. (I937) 9I, 222-23I 6I2.46I:6I2.392.6 THE SECRETION OF URINE IN MAN DURING EXPERIMENTAL SALT DEFICIENCY BY R. A. McCANCE AND E. M. WIDDOWSON From the Biochemical Laboratory, King's College
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
factors. directly. There are many, however, who regard the hydrochloric acid degree of stimulation of the stomach cells [Roseman, 1927; Katsch &
 308 J. Physiol. (I940) 97, 308-3I5 6I2.323.3 ON THE PRIMARY ACIDITY OF THE GASTRIC JUICE BY TORSTEN TEORELL From the Department of Medical Chemistry, University of Uppsala, Sweden (Received 10 July 1939)
308 J. Physiol. (I940) 97, 308-3I5 6I2.323.3 ON THE PRIMARY ACIDITY OF THE GASTRIC JUICE BY TORSTEN TEORELL From the Department of Medical Chemistry, University of Uppsala, Sweden (Received 10 July 1939)
M6ller, McIntosh and Van Slyke (5) has been employed. The cases. changes in functional activity. Indications suggesting that such changes
 STUDIES OF UREA EXCRETION. VIII. THE EFFECTS ON THE UREA CLEARANCE OF CHANGES IN PROTEIN AND SALT CONTENTS OF THE DIET BY CUTHBERT L. COPE I (From the Hospital of the Rockefeller Institute for Medical
STUDIES OF UREA EXCRETION. VIII. THE EFFECTS ON THE UREA CLEARANCE OF CHANGES IN PROTEIN AND SALT CONTENTS OF THE DIET BY CUTHBERT L. COPE I (From the Hospital of the Rockefeller Institute for Medical
only. Analysis of human muscle obtained by biopsy suggests that changes
 A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
RELEASES TOXIC FUMES!
 Name(s): Block: Date: Urinary System Lab Activity: Urinalysis Background: Urinalysis, the physical, chemical, and microscopic analysis or examination of urine can provide information about a person s carbohydrate
Name(s): Block: Date: Urinary System Lab Activity: Urinalysis Background: Urinalysis, the physical, chemical, and microscopic analysis or examination of urine can provide information about a person s carbohydrate
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
uric acid itself. VARIATIONS IN THE EXCRETION OF URIC ACID PRODUCED BY ADMINISTRATION OF ACIDS AND
 VARIATIONS IN THE EXCRETION OF URIC ACID PRODUCED BY ADMINISTRATION OF ACIDS AND ALKALIES. BY A. HAIG, M.B., Oxon., M.R.C.P. P1. VI. Figs. 1-3. WIIILE investigating the relationship of a certain form of
VARIATIONS IN THE EXCRETION OF URIC ACID PRODUCED BY ADMINISTRATION OF ACIDS AND ALKALIES. BY A. HAIG, M.B., Oxon., M.R.C.P. P1. VI. Figs. 1-3. WIIILE investigating the relationship of a certain form of
ACID BASE BALANCE & BODY FLUID. Ani Retno Prijanti Renal and Body Fluids Module Juni 2008
 ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
(Foulerton Student of the Royal Society).
 6I2.62I:6I2.OI8 STUDIES ON OVULATION. VI. Relative importance of concentration and absolute amount of the ovulation-producing hormone. BY F. W. ROGERS BRAMBELL AD A. S. PARKES (Foulerton Student of the
6I2.62I:6I2.OI8 STUDIES ON OVULATION. VI. Relative importance of concentration and absolute amount of the ovulation-producing hormone. BY F. W. ROGERS BRAMBELL AD A. S. PARKES (Foulerton Student of the
Physical Characteristics of
 Physical Characteristics of Urine Bởi: OpenStaxCollege The urinary system s ability to filter the blood resides in about 2 to 3 million tufts of specialized capillaries the glomeruli distributed more or
Physical Characteristics of Urine Bởi: OpenStaxCollege The urinary system s ability to filter the blood resides in about 2 to 3 million tufts of specialized capillaries the glomeruli distributed more or
blue, buffer excretion by titrating back to ph 3*7 with.1 N hydrochloric
 CALCIUM CHLORIDE ACIDOSIS. BY J. B. S. HALDANE, R. HILL, AND J. M. LUCK. (From the Biochemical Laboratory, Cambridge.) GYORGY(1) has shown that calcium chloride, when administered to babies, causes an
CALCIUM CHLORIDE ACIDOSIS. BY J. B. S. HALDANE, R. HILL, AND J. M. LUCK. (From the Biochemical Laboratory, Cambridge.) GYORGY(1) has shown that calcium chloride, when administered to babies, causes an
fluid in the muscles of the rat and the frog following violent
 612.766.1: 612.014.461.3 THE CHANGES IN PLASMA AND TISSUE FLUID VOLUME FOLLOWING EXERCISE. By H. CULLUMBINE and A. C. E. KoCH. From the Department of Physiology and Pharmacology, University of Ceylon,
612.766.1: 612.014.461.3 THE CHANGES IN PLASMA AND TISSUE FLUID VOLUME FOLLOWING EXERCISE. By H. CULLUMBINE and A. C. E. KoCH. From the Department of Physiology and Pharmacology, University of Ceylon,
Technical University of Mombasa Faculty of Applied and Health Sciences
 Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
already been published [O'Connor, 1958 b]. emphasized that the most prominent action of adrenaline on the kidney is to
![already been published [O'Connor, 1958 b]. emphasized that the most prominent action of adrenaline on the kidney is to already been published [O'Connor, 1958 b]. emphasized that the most prominent action of adrenaline on the kidney is to](/thumbs/88/116101114.jpg) THE EFFECT ON THE VOLUME AND COMPOSITION OF THE URINE OF THE INFUSION OF ADRENALINE AND NORADRENALINE. By W. J. O'CoNNoR. From the Department of Physiology, School of Medicine, University of Leeds. (Received
THE EFFECT ON THE VOLUME AND COMPOSITION OF THE URINE OF THE INFUSION OF ADRENALINE AND NORADRENALINE. By W. J. O'CoNNoR. From the Department of Physiology, School of Medicine, University of Leeds. (Received
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
THE TOXICITY OF THE DOUBLE CHLORIDES OF MERCURY AND SODIUM
 325 THE TOXICITY OF THE DOUBLE CHLORIDES OF MERCURY AND SODIUM I. EXPERIMENTS WITH THE MINNOW PHOXINUS PHOXINUS (L.) BY J. R. ERICHSEN JONES Department of Zoology, University College of Wales, Aberystwyth
325 THE TOXICITY OF THE DOUBLE CHLORIDES OF MERCURY AND SODIUM I. EXPERIMENTS WITH THE MINNOW PHOXINUS PHOXINUS (L.) BY J. R. ERICHSEN JONES Department of Zoology, University College of Wales, Aberystwyth
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE*
 ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
Objectives Body Fluids Electrolytes The Kidney and formation of urine
 Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Cushny(4) has shown, however, that the amount of urea in the kidney. by some vital process, retain those diflusible substances which are of
 THE FUNCTION OF THE TUBULES IN KIDNEY EXCRETION. BY E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) IT is becoming generally recognised that filtration through the glomeruli and some degree
THE FUNCTION OF THE TUBULES IN KIDNEY EXCRETION. BY E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) IT is becoming generally recognised that filtration through the glomeruli and some degree
: : (From the Department of Physiology, University of Toronto.)
 94 612.352.2:547.435:612.34.089.87 CHOLINE AND LIVER FAT IN DIABETIC DOGS. BY C. H. BEST, G. C. FERGUSON AND J. M. HERSHEY. (From the Department of Physiology, University of Toronto.) IN the first completely
94 612.352.2:547.435:612.34.089.87 CHOLINE AND LIVER FAT IN DIABETIC DOGS. BY C. H. BEST, G. C. FERGUSON AND J. M. HERSHEY. (From the Department of Physiology, University of Toronto.) IN the first completely
STUDIES ON THE PHYSIOLOGY OF THE PARATHYROID GLANDS
 STUDIES ON THE PHYSIOLOGY OF THE PARATHYROID GLANDS V. ACTION OF PARATHYROID EXTRACT ON THE RENAL THRESHOLD FOR PHOSPHORUS By READ ELLSWORTH (From the Medical Clinic, the School of Medicine, Johns Hopkins
STUDIES ON THE PHYSIOLOGY OF THE PARATHYROID GLANDS V. ACTION OF PARATHYROID EXTRACT ON THE RENAL THRESHOLD FOR PHOSPHORUS By READ ELLSWORTH (From the Medical Clinic, the School of Medicine, Johns Hopkins
J. Physiol. (I944) I02; :6I2.0I4.46I.2
 415 J. Physiol. (I944) I02; 45-428 612.463:6I2.0I4.46I.2 THE SECRETION OF URINE DURING DEHYDRATION AND REHYDRATION BY R. A. McCANCE AND W. F. YOUNG WITH THE ASSISTANCE OF D. A. K. BLACK From the Department
415 J. Physiol. (I944) I02; 45-428 612.463:6I2.0I4.46I.2 THE SECRETION OF URINE DURING DEHYDRATION AND REHYDRATION BY R. A. McCANCE AND W. F. YOUNG WITH THE ASSISTANCE OF D. A. K. BLACK From the Department
College, London. NITROGEN IN THE URINE. BY OTTO VERAGUTH, steadily hour by hour to the fifth hour, when it reached its maximum
 THE EFFECT OF A MEAL ON THE EXCRETION OF NITROGEN IN THE URINE. BY OTTO VERAGUTH, M.D., Zurich. (Twelve Figures in Text.) From the Department of Pathological Chemistry, University College, London. DR TSCHLENOFF1
THE EFFECT OF A MEAL ON THE EXCRETION OF NITROGEN IN THE URINE. BY OTTO VERAGUTH, M.D., Zurich. (Twelve Figures in Text.) From the Department of Pathological Chemistry, University College, London. DR TSCHLENOFF1
The Excretory System. Biology 20
 The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE
 PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE - OBJECTIVES: 1- The simple examination of urine. 2- To detect some of the normal organic constituents of urine. 3- To detect some of the
PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE - OBJECTIVES: 1- The simple examination of urine. 2- To detect some of the normal organic constituents of urine. 3- To detect some of the
PARTS OF THE URINARY SYSTEM
 EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
PHYSIOLOGICAL SOCIETY,
 PROCEEDINGS OF THE PHYSIOLOGICAL SOCIETY, March 16, 1929. A central chemical control of the heart rate. By R. J. S. MCDOWALL. (King's College, London.) Provided adequate measures are taken to avoid the
PROCEEDINGS OF THE PHYSIOLOGICAL SOCIETY, March 16, 1929. A central chemical control of the heart rate. By R. J. S. MCDOWALL. (King's College, London.) Provided adequate measures are taken to avoid the
Chapter 12. Excretion and the Interaction of Systems
 Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-
 Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
Excretory System. Biology 2201
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System. Excretory System
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
RENAL TUBULAR ACIDOSIS An Overview
 RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
PHYSIOLOGICAL DISTURBANCES DURING EXPERIMENTAL
 PHYSIOLOGICAL DISTURBANCES DURING EXPERIMENTAL DIPHTHERITIC INTOXICATION. III. RESPIRATORY QUOTIENTS AND METABOLIC RATE 1 By HERMAN YANNET AND WALTER GOLDFARB (From the Department of Pediatrics and Physiology,
PHYSIOLOGICAL DISTURBANCES DURING EXPERIMENTAL DIPHTHERITIC INTOXICATION. III. RESPIRATORY QUOTIENTS AND METABOLIC RATE 1 By HERMAN YANNET AND WALTER GOLDFARB (From the Department of Pediatrics and Physiology,
Physiology week 16 Renal 2 (volume/buffers)
 Physiology week 16 Renal 2 (volume/buffers) Defense of Tonicity and Volume Defense of tonicity Tonicity = osmolality of a solution relative to plasma Osmolality measures [ ] all particles in solution,
Physiology week 16 Renal 2 (volume/buffers) Defense of Tonicity and Volume Defense of tonicity Tonicity = osmolality of a solution relative to plasma Osmolality measures [ ] all particles in solution,
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Introduction. Acids, Bases and ph; a review
 0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
The technique of multiple clearance was chosen. because it supplied us with the filtration rate, the
 RENAL REABSORPTION OF CHLORIDE AND PHOSPHATE IN NORMAL SUBJECTS AND IN PATIENTS WITH ESSENTIAL ARTERIAL HYPERTENSION' By EDITH B. FARNSWORTH (From the Department of Medicine, Northwestern University Medical
RENAL REABSORPTION OF CHLORIDE AND PHOSPHATE IN NORMAL SUBJECTS AND IN PATIENTS WITH ESSENTIAL ARTERIAL HYPERTENSION' By EDITH B. FARNSWORTH (From the Department of Medicine, Northwestern University Medical
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
College of Medicine, Newcastle-upon-Tyne.)
 GLUCOSE ABSORPTION IN THE RENAL TUBULES OF THE FROG. BY G. A. CLARK. (From the Physiological Laboratory of the University of Durham College of Medicine, Newcastle-upon-Tyne.) OPINION is divided on the
GLUCOSE ABSORPTION IN THE RENAL TUBULES OF THE FROG. BY G. A. CLARK. (From the Physiological Laboratory of the University of Durham College of Medicine, Newcastle-upon-Tyne.) OPINION is divided on the
AJl the experiments of the following series were done on rabbits. THE secretion of glucose is considered to depend on a " threshold " value
 THE INFLUENCE OF DIURETICS ON THE EXCRETION OF SUGAR. BYE. J. CONWAY. (From the Physiological Department, University College, Dublin.) THE secretion of glucose is considered to depend on a " threshold
THE INFLUENCE OF DIURETICS ON THE EXCRETION OF SUGAR. BYE. J. CONWAY. (From the Physiological Department, University College, Dublin.) THE secretion of glucose is considered to depend on a " threshold
Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
Excretion Chapter 29. The Mammalian Excretory System consists of. The Kidney. The Nephron: the basic unit of the kidney.
 Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
University College, London.)
 6I2.I2I:547.472*3 LACTIC ACID FORMATION AND REMOVAL WITH CHANGE OF BLOOD REACTION. BY M. GRACE EGGLETON1 AND C. LOVATT EVANS. (From the Department of Physiology and Biochemistry, University College, London.)
6I2.I2I:547.472*3 LACTIC ACID FORMATION AND REMOVAL WITH CHANGE OF BLOOD REACTION. BY M. GRACE EGGLETON1 AND C. LOVATT EVANS. (From the Department of Physiology and Biochemistry, University College, London.)
(essential, spontaneous) pentosuria is rare. Less than fifty cases are on
 THE UTILIZATION OF CARBOHYDRATES IN A CASE OF CHRONIC PENTOSURIA By I. M. RABINOWITCH (From the Department of Metabolism, Montreal General Hospital. Montreal, Canada) (Received for publication April 23,
THE UTILIZATION OF CARBOHYDRATES IN A CASE OF CHRONIC PENTOSURIA By I. M. RABINOWITCH (From the Department of Metabolism, Montreal General Hospital. Montreal, Canada) (Received for publication April 23,
THE ABSORPTION OF VOLATILE FATTY ACIDS FROM THE RUMEN
 VOL. 24, Nos. 1 & 2 SEPTEMBER 1947 THE ABSORPTION OF VOLATILE FATTY ACIDS FROM THE RUMEN BY F. V. GRAY From the Division of Biochemistry and General Nutrition of the Council for Scientific and Industrial
VOL. 24, Nos. 1 & 2 SEPTEMBER 1947 THE ABSORPTION OF VOLATILE FATTY ACIDS FROM THE RUMEN BY F. V. GRAY From the Division of Biochemistry and General Nutrition of the Council for Scientific and Industrial
RENAL FUNCTION An Overview
 RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
AP Biology. Homeostasis. Chapter 44. Regulating the Internal Environment. Homeostasis
 Chapter 44. Regulating the Internal Environment omeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
Chapter 44. Regulating the Internal Environment omeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
Excretory Lecture Test Questions Set 1
 Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
hydrocortisone (cortisol) appeared to increase potassium excretion without
 J. Phy8iol. (1961), 156, pp. 415-423 415 With 1 text-figure Printed in Great Britain THE EFFECTS OF INTRAVENOUS ALDOSTERONE AND HYDRORTISONE ON THE URINARY ELECTROLYTES OF THE RECUMBENT HUMAN SUBJECT BY
J. Phy8iol. (1961), 156, pp. 415-423 415 With 1 text-figure Printed in Great Britain THE EFFECTS OF INTRAVENOUS ALDOSTERONE AND HYDRORTISONE ON THE URINARY ELECTROLYTES OF THE RECUMBENT HUMAN SUBJECT BY
Chapter 23. Composition and Properties of Urine
 Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
to assess the renal reabsorptive capacity after restoration of normal carbon dioxide tension in
 Journal of Clinical nvestigation Vol. 41, No. 12, 1962 EFFECTS OF CHRONC HYPERCAPNA ON ELECTROLYTE AND ACD-BASE EQULBRUM.. CHARACTERSTCS OF THE ADAPTVE AND RECOVERY PROCESS AS EVALUATED BY PROVSON OF ALKAL
Journal of Clinical nvestigation Vol. 41, No. 12, 1962 EFFECTS OF CHRONC HYPERCAPNA ON ELECTROLYTE AND ACD-BASE EQULBRUM.. CHARACTERSTCS OF THE ADAPTVE AND RECOVERY PROCESS AS EVALUATED BY PROVSON OF ALKAL
Disclaimer. Chapter 3 Disorder of Water, Electrolyte and Acid-base Professor A. S. Alhomida. Disorder of Water and Electrolyte
 Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
EXCRETORY SYSTEM E. F. G. H.
 XRTORY SYSTM 1. Label the following parts of the nephron in the diagram below:..... F. G. H. I. J. K. L. 2. Identify the following as either True or False: There is a greater osmotic concentration in the
XRTORY SYSTM 1. Label the following parts of the nephron in the diagram below:..... F. G. H. I. J. K. L. 2. Identify the following as either True or False: There is a greater osmotic concentration in the
Renal Reabsorption & Secretion
 Renal Reabsorption & Secretion Topics for today: Nephron processing of filtrate Control of glomerular filtration Reabsorption and secretion Examples of solute clearance rates Hormones affecting kidney
Renal Reabsorption & Secretion Topics for today: Nephron processing of filtrate Control of glomerular filtration Reabsorption and secretion Examples of solute clearance rates Hormones affecting kidney
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
1.2 Synonyms There are several synonyms e.g. diaminomethanal, but in a medical context, this substance is always referred to as urea.
 Urea (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Urea 1.2 Synonyms There are several synonyms e.g. diaminomethanal, but in a medical context, this substance is always referred
Urea (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Urea 1.2 Synonyms There are several synonyms e.g. diaminomethanal, but in a medical context, this substance is always referred
Chapter 44. Regulating the Internal Environment. AP Biology
 Chapter 44. Regulating the Internal Environment Homeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
Chapter 44. Regulating the Internal Environment Homeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
April 08, biology 2201 ch 11.3 excretion.notebook. Biology The Excretory System. Apr 13 9:14 PM EXCRETORY SYSTEM.
 Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
