Eukaryotic cells use internal membrane-bound compartments
|
|
|
- Gregory Clarke
- 5 years ago
- Views:
Transcription
1 Exo1: A new chemical inhibitor of the exocytic pathway Yan Feng*, Sidney Yu, Troy K. R. Lasell, Ashutosh P. Jadhav*, Eric Macia, Pierre Chardin, Paul Melancon, Michael Roth, Timothy Mitchison*, and Tomas Kirchhausen* *Institute of Chemistry and Cell Biology, Department of Cell Biology, and Center for Blood Research, Harvard Medical School, Boston, MA 02115; Department of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX 75235; Department of Cell Biology, University of Alberta, Edmonton, AB, Canada T6G 2H7; and Institut de Pharmacologie Moleculaire et Cellulaire, Centre National de la Recherche Scientifique Unité Mixte de Recherche 6097, Valbonne Sophia-Antipolis, France Communicated by Marc W. Kirschner, Harvard Medical School, Boston, MA, March 27, 2003 (received for review November 5, 2002) A phenotypic screen was used to search for drug-like molecules that can interfere with specific steps in membrane traffic. 2-(4- Fluorobenzoylamino)-benzoic acid methyl ester (Exo1), identified in this screen, induces a rapid collapse of the Golgi to the endoplasmic reticulum, thus acutely inhibiting the traffic emanating from the endoplasmic reticulum. Like Brefeldin A (BFA), Exo1 induces the rapid release of ADP-ribosylation factor (ARF) 1 from Golgi membranes but has less effect on the organization of the trans-golgi network. Our data indicate that Exo1 acts by a different mechanism from BFA. Unlike BFA, Exo1 does not induce the ADP-ribosylation of CtBP Bars50 and does not interfere with the activity of guanine nucleotide exchange factors specific for Golgibased ARFs. Thus, Exo1 allows the fatty acid exchange activity of Bars50 to be distinguished from ARF1 activity in the control of Golgi tubulation. Golgi ADP-ribosylation factor endoplasmic reticulum (ER) imaging-based screen Bars50 Eukaryotic cells use internal membrane-bound compartments to spatially segregate metabolic reactions and to create different chemical environments. The distinct identity of these compartments must be established and maintained. This process is presumably particularly difficult for the organelles of the secretory and endocytic pathways, which must exchange membrane components as part of their function. We know that small vesicles and tubules are used to transport the membrane components between organelles so that the parent organelles remain separated (1, 2). We do not yet fully understand, however, the functions of proteins that have been identified as important for secretion and how their functions are integrated. In part this lack of knowledge is because most of the steps in secretion and endocytosis are highly dynamic and highly regulated, and therefore hard to study by using the tools of genetics and biochemistry. We are interested in the potential of small molecule inhibitors or activators for studying secretion. Because of the dynamic nature of membrane traffic, specific, reversible, fast-acting modulators of secretion would be particularly useful research tools. Unfortunately, very few small molecule modulators of the secretory pathway are known. This may partly be because most small molecule discovery is done in the pharmaceutical industry, and inhibitors of secretion are generally too toxic to be useful as clinical drugs. Brefeldin A (BFA), a particularly useful reagent for study of Golgi function (3, 4) is an example: it was originally discovered at a pharmaceutical company (5) but was essentially ignored for 25 years due to its toxicity. BFA causes dramatic changes in the morphology and function of the Golgi; the Golgi tubulates, then redistributes its entire contents and membrane to the endoplasmic reticulum (ER). A subset of the endosomal compartment also tubulates and mixes with the trans-golgi network (TGN; refs. 6 and 7). After much study, the mechanism of BFA has been at least partly elucidated. It stabilizes a transient complex formed between the small GTPase ADP-ribosylation factor (ARF) 1 in its GDP-bound form and some of its exchange factors, preventing GDP GTP exchange (8, 9). Thus, ARF1-GTP is consumed and not replaced, therefore processes that depend on ARF1-GTP (such as membrane recruitment or activation of various proteins involved in regulation of vesicular traffic) are blocked (10). BFA has been a powerful tool, and we owe a good deal of our understanding of the Golgi as a dynamic organelle to the use of BFA. However, it has pleiotropic effects in mammalian cells that are confusing and often of unknown mechanism. For example, in addition to inhibiting ARF1-GTP GDP exchange, BFA is known to cause ADP-ribosylation of CtBP Bars50 (11, 12). CtBP Bars50 is an enzyme involved in the transfer of palmitate from palmitoyl CoA to lysophosphatidic acid. This ADPribosylation event has been suggested to contribute to tube formation in the Golgi collapse phenomenon, by a mechanism involving changes in membrane curvature due to reduction in CtBP Bars50 activity (13). An alternative hypothesis is that tubule formation is a consequence of the release of ARF1 from Golgi membranes (14, 15), and these hypotheses have been hard to differentiate. To find a small molecule tool that would complement BFA in the study of membrane trafficking, we used a phenotypic screen to identify 26 small molecules that perturb exocytosis. Here we show that one of these, 2-(4-fluorobenzoylamino)-benzoic acid methyl ester (Exo1), is a modifier of Golgi ARF1 GTPase activity. Thus far, the observed effects of Exo1 on cells are restricted to the traffic between the ER and Golgi apparatus. Because Exo1 affects a subset of the membrane events that are blocked by BFA, but seems to have a different protein target (and presumably has different side effects), it provides an independent means to interfere with Golgi activity. Methods High-Throughput Phenotypic Screen. BSC1 fibroblast cells were mixed with vesicular stomatitis virus fused to GFP (VSVG ts - GFP) adenovirus, plated in 384-well clear bottom plates at 1,500 cells per 30 l per well and grown overnight at 40 C in a5%co 2 incubator. Compounds (10,240 from the DIVERSet E, Chembridge, San Diego) were screened by sampling 100 nl of 10 mg ml of stocks dissolved in DMSO that were transferred into wells in duplicate plates by using a pin-transfer robot (http: iccb.med.harvard.edu screening). Cells were incubated further at 40 C for 60 min, transferred to 40 C or 32 C for 2 h, fixed with 4% paraformaldehyde, and imaged by using an automated Abbreviations: BFA, brefeldin A; TGN, trans-golgi network; VSVG ts -GFP, vesicular stomatitis virus fused to GFP; Exo1, 2-(4-fluorobenzoylamino)-benzoic acid methyl ester; m-exo1, 3-(4-fluorobenzoylamino)-benzoic acid methyl ester; GAP, GTPase-activating protein; ARF, ADP-ribosylation factor; CFP, cyan fluorescent protein; COPI, coatomer protein I; GGA, Golgi-localizing, gamma-adaptin ear homology domain, ARF-binding; GEF, guanine nucleotide exchange factor; GalT, galactosyltransferase; ER, endoplasmic reticulum. To whom correspondence should be addressed. yfeng@hms.harvard.edu and kirchhausen@crystal.harvard.edu. CELL BIOLOGY cgi doi pnas PNAS May 27, 2003 vol. 100 no
2 Fig. 1. Exit of VSVG ts -GFP from the ER is inhibited by Exo1. (A) Chemical structures of Exo1, the analog m-exo1, and BFA. (B E) VSVG ts -GFP accumulates in the ER at 40 C (B) and reaches the Golgi apparatus after a 30-min shift to 32 C (C) but remains in the ER in the presence of 100 M Exo1 (D and E). Movies 1 and 2 show time-lapse data for these experiments. (F) The effect of different concentrations of Exo1 and m-exo1 on the relative efficiency of delivery of VSVG ts -GFP from the ER to the cell surface. BSC1 cells expressing VSVG ts -GFP were incubated at 40 C with different amounts of the compounds, then shifted to 32 C and incubated for 3 h. Under these conditions Exo1 has an IC 50 of about 20 M whereas m-exo1 has no effect at 300 M (for dose response with BFA see Appendix II). (G) Results from a pulse chase experiment after the acquisition of endoglycosidase H resistance by VSVG ts -GFP in the absence (Top) or presence (Middle)of 100 M Exo1 or 5 M BFA (Bottom). fluorescence microscope (autoscope, http: iccb.med.harvard. edu screening) at a rate of about 1 plate per h. Images were visually scored for inhibition of VSVG ts -GFP traffic, and positive compounds were retested at different concentrations. Supporting Information. For additional information, see Appendices I VIII and Movies 1 6, which are published as supporting information on the PNAS web site, Results An Image-Based Phenotypic Screen to Identify Small Molecule Inhibitors of the Exocytic Pathway. We developed an automated fluorescence microscopy imaging-based screen to search for small molecules affecting the exocytic traffic of membrane proteins from the ER to the plasma membrane. The screen was based on the behavior of the temperature-sensitive mutant ts045 of the surface glycoprotein of VSVG ts -GFP. At the nonpermissive temperature of 40 C, VSVG ts -GFP remains in the ER. Shift of the cells to 32 C results in the synchronous export of VSVG ts - GFP, first to the Golgi apparatus and then to the plasma membrane (see Appendix I A and B) (16, 17). We screened 10,240 compounds (part of the DIVERSet E, Chembridge library) for effects on the localization of VSVG ts -GFP at 40 C and 32 C. Most had no effect, or toxic effects. Twenty-six compounds were considered hits. These elicited four distinct phenotypes on the intracellular distribution of VSVG ts -GFP (K). These phenotypes can be summarized as ER exit block (C, D), Golgi exit block (E, F), Golgi fragmentation (G, H), and vacuole formation (I, J). We believe these compounds are relatively specific for processes involved in membrane traffic. None of these compounds has been selected as high-priority hits in any of the other 100 phenotypic or enzymatic screens performed at Institute of Chemistry and Cell Biology (Harvard Medical School, Boston) with the same set of compounds, and none of them has shown major effects on the appearance of the actin-based cytoskeleton, the rate of transferrin uptake from the plasma membrane to endosome, or the appearance of endosomes and lysosomes (data not shown). Exo1 Disrupts Vesicular Traffic from the ER to the Golgi Apparatus by Disrupting Golgi Structures. Here we focus on Exo1 (Fig. 1A), one of the two compounds that blocked VSVG ts -GFP exit from the ER (Fig. 1 B E and Movies 1 and 2). We confirmed the identity and purity of the compound by using routine analytical chemistry techniques and by de novo synthesis. The closely related analog 3-(4-fluorobenzoylamino)-benzoic acid methyl ester (m-exo1; Fig. 1A) has no apparent effect in this assay. BFA also blocks the same process (3) (see Appendix III) but has a very different structure (Fig. 1A). Exo1 inhibits exocytosis with an IC 50 of 20 M (Fig. 1F). A small SAR study (234 related compounds) was performed but failed to identify more potent analogs (Appendix II). The effects of Exo1 are similar on all cells we tested. It blocks ER exit of VSVG ts -GFP in nonpolarized mammalian cells such as Chinese hamster ovary, human HeLa and 293, rat NRK, hamster BHK, and polarized canine MDCK cells (data not shown). Likewise, Exo1 prevents acquisition of endoglycosidase H resistance by newly synthesized proteins such as VSVG ts -GFP (Fig. 1G), transferrin, and MHC class I (data not shown). To understand the mechanism of these effects, we first asked whether Exo1 treatment alters the distribution of a series of markers specific for different organelles along the exocytic and cgi doi pnas Feng et al.
3 endocytic pathways. Exo1 treatment induces rapid redistribution of Golgi content back to ER [galactosyltransferase-gfp (GalT- GFP); see Fig. 2 A and B and Appendix IV], whereas it has essentially no effect on endocytic organelle structures, demonstrated by staining of transferrin-positive early and recycling endosomes (Fig. 2C) and cathepsin D-positive late endosomes and lysosomes (Appendix IV). The effects of Exo1 on the Golgi apparatus are reversible. Golgi markers (residential enzyme GalT and matrix protein GM130) reappear in the perinuclear area 30 min after Exo1 is removed from the medium (Appendix IV). The Effects of Exo1 Are Similar but Not Identical to Those of BFA. Treatment with Exo1 results in massive tubulation of the Golgi apparatus, formation of contacts between the Golgi tubules, and massive and uncontrolled transfer of Golgi contents primarily to the ER as followed by the movement of GalT-GFP (Fig. 2A and Movie 3). Similar effects were described with BFA treatment (3) (Movie 4). The primary target of BFA in cells was shown to be ARF1 GTPase (18). BFA treatment causes a rapid decrease of the levels of activated ARF1 on the Golgi membrane and therefore release of many Golgi- and TGN-associated proteins such as coatomer protein I (COPI) coatomers, clathrin coat, and adaptors AP1, AP3, and Golgi-localizing, gamma-adaptin ear homology domain, ARF-binding protein (GGAs) (15). All these events precede the general collapse of the Golgi apparatus. We therefore examined the release of several markers from intracellular membranes in response to Exo1 and BFA. Kinetic analysis by time-lapse microscopy showed that the average half-life for disappearance of ARF1-GFP from Golgi membranes after treatment with either Exo1 or BFA was less than 1 min (Fig. 2B and Movies 5 and 6). In both cases, the release of COPI is also very rapid after ARF1 dissociation (Appendix V) and this precedes the general collapse of the Golgi apparatus indicated here by GalT-GFP (Fig. 2B and Appendix V). The effects of the two chemicals are not identical, however. Exo1 addition did not induce rapid release of TGN-associated coat proteins such as GGA3, AP-1, AP-3, and clathrin (Fig. 2C and Appendix V), whereas BFA caused release of these proteins to the cytosol within 2 min of treatment. Longer treatments with Exo1 (30 min) led to a dispersal of AP-1, AP-3, and GGA3 signals to small punctate structures, with partial release to the cytosol (data not shown). A second distinction is that Exo1 does not tubulate endosomal membranes labeled with internalized transferrin (Fig. 2C), whereas BFA does. Because of these differences, we conclude that either Exo1 binds to a subset of the protein targets of BFA or the target of Exo1 is different from that of BFA. Because Exo1 is relatively specific for Golgi membranes, we assume that the target of Exo1 is relatively abundant in the Golgi. CELL BIOLOGY Fig. 2. Exo1 induces tubulation and collapse of the Golgi apparatus but not endosomes and TGN. (A) The intracellular location of GalT-GFP was monitored by time-lapse fluorescence microscopy in BSC1 cells treated with 100 M Exo1 or 5 M BFA. Both compounds induced the appearance of tubules emanating from the Golgi apparatus at around 4 min. The tubes eventually reach the ER and this leads to the dramatic redistribution of Golgi contents to the ER at 10 min (Movies 3 and 4). (B) The effect of 100 M Exo1 or 5 M BFA on the distribution of ARF1-GFP or GalT-GFP in BSC1 cells was followed as a function of time by time-lapse fluorescence microscopy. The perinuclear Golgiassociated fluorescent signal was monitored for 5 min before addition of the compounds to the media. Each curve represent the average data recorded from 6 movies. Also see Movies 5 and 6. (C) Effects of BFA or Exo1 on the distribution of endosomes (Tfn) and the TGN (GGA3). The early recycling endosomes were visualized by following the distribution of Texas red-labeled transferrin internalized for 20 min by receptor-mediated endocytosis in the ARF-Guanine Nucleotide Exchange Factor (GEF) Is Not the Direct Target of Exo1. The steady-state distribution of ARF1-GTP bound to Golgi membranes depends on the balance between the exchange activity of ARF-GEFs (generating ARF-GTP) and the rate of GTP hydrolysis by ARF. We therefore examined whether Exo1 decreases GTP loading onto ARF1 or increases GTP hydrolysis by ARF1. Either activity would explain the observed release of ARF1 from Golgi membranes. Unlike BFA, Exo1 did not interfere with the guanine nucleotide exchange activity of a number of ARF-GEFs in vitro (Fig. 3A and Appendix VI). Nucleotide exchange in vivo is directly linked to stable binding presence of compounds. BFA induced enlargement and tubulation of transferrin-positive endosomes (Inset). Exo1 had no effect. The TGN-associated adaptor protein GGA3 was visualized after a 2-min treatment of BFA or Exo1. BFA induced GGA3 redistribution to cytosol, whereas Exo1 did not. Feng et al. PNAS May 27, 2003 vol. 100 no
4 Fig. 3. (Legend appears at the bottom of the opposite page.) cgi doi pnas Feng et al.
5 of ARF1 to the Golgi membrane. We also showed that, in the presence of a sufficient level of Exo1 to rapidly release all wild-type ARF1 from Golgi, ARF1 Q71L -GFP could be reloaded to Golgi membrane after it was released from Golgi with BFA pretreatment (Fig. 3B). Thus, Exo1 does not seem to directly inhibit ARF1-GDP GTP exchange. It is most complicated to determine whether Exo1 accelerates GTP hydrolysis. In vitro GTPase-activating protein (GAP)- stimulated ARF1-GTP hydrolysis was not sensitive to Exo1 (data not shown). The in vivo ARF1-GTP hydrolysis rate is several thousand-fold higher than the level achievable in vitro, however. We therefore used two indirect approaches to ask whether Exo1 might affect ARF1-GTP hydrolysis in vivo. First, we preincubated cells with AlF 4, which has been shown to lock several small GTPases in their GDP.AlF 4 -binding transition state together with a limiting cofactor GAP (19, 20). It is not yet clear whether AlF 4 has the same effect on ARF1. Although an early experiment suggests that AlF 4 stabilizes a subset of ARF1 on Golgi membranes (21), recent evidence from Presley et al. (22) indicates that the main effect of AlF 4 on membrane is to irreversibly block dissociation of COPI but not the bulk of ARF1. However, we observed that in the presence of BFA, AlF 4 slowed the dissociation rate of ARF1 from the Golgi significantly, and AlF 4 treatment itself increased the steady-state level of ARF1wt-GFP on the Golgi membrane (Fig. 3C). It is possible that this result indicates that AlF 4 has a similar effect on ARF1 as it does on other small GTPases, i.e., tethering the limiting GAP factor in a relatively more stable complex and thus reducing the overall rate of GTP hydrolysis. In this case, the presence of AlF 4 should slow (but not entirely prevent) dissociation of ARF1 from the membrane. Because reloading is blocked in the presence of BFA, the dissociation should eventually reach the same extent as in the absence of AlF 4. That BFA is able to release all ARF1 from the Golgi membrane and that the rate of release is only slightly reduced, indicate that the AlF 4 -induced tethering of ARF1, whatever its mechanism, must be short term (Fig. 3C). This finding is in agreement with the observation by Kahn (23). In contrast, AlF 4 blocked the ability of Exo1 to induce dissociation of membrane-bound ARF1 wt -GFP (Fig. 3C). This result is consistent with the idea that Exo1 might increase the rate of GTP hydrolysis, and that this increase depends on the availability of GAP. It is also possible, however, that Exo1 is having an indirect inhibitory effect on ARF-GEF via another GTPase or via some unknown mechanism. In any case, the effects of Exo1 and BFA are dramatically different. In the second approach, we used cells transiently expressing a low level of ARF1 Q71L -GFP, a mutated form of ARF1 with reduced GTPase activity (24). The dissociation of ARF1 Q71L - GFP in response to BFA is slower than the release of wild-type ARF1 (Fig. 3D). COPI, which binds to ARF1-GTP, also dissociates from the membrane (Fig. 3E). Exo1 caused only a Fig. 4. Exo1 does not induce the ADP-ribosylation of Bars50. The supernatant from a cell lysate of BSC1 cells subjected to high-speed centrifugation was incubated with 10 M BFA or 100 M Exo1 in the presence of 32 P-ADP-ribose for 30 min at 37 C. The proteins in the lysate were then subjected to denaturing SDS PAGE, as demonstrated (11). BFA (lane 2) induces the ADPribosylation of Bars50. In contrast, Exo1 (lane 3) had no effect. No ADPribosylation is detected in the presence of 1% DMSO (lane 1). transient and incomplete dissociation of the mutated ARF1 Q71L - GFP (Fig. 3D) and minimal dissociation of COPI (Fig. 3E). Thus, a mutation that slows the GTPase activity of ARF1 also reduces the sensitivity of the enzyme to Exo1. The presence of biologically active concentrations of Exo1 in these experiments can be confirmed by examining nontransfected cells in the field, which were observed to lose Golgi-associated COPI (Fig. 3E). Consistent with these data, ARF1 Q71L rescued ER export of VSVG ts -GFP in the presence of Exo1 but not in the presence of BFA (Fig. 3F). These results strongly support the hypothesis that Exo1 and BFA have different targets, and that the target of Exo1 is likely to be downstream from the ARF1-GTP-loading step. These results are consistent with the notion that Exo1 works by increasing the rate of GTP hydrolysis through the activation of an ARF-GAP-dependent step, although many other models are possible. Golgi Tubulation Does Not Require ADP-Ribosylation of CtBP Bars50. As one example of the utility of this tool, we explored the question of whether Golgi tubulation induced by Exo1, like with BFA, also involves ADP-ribosylation of CtBP Bars50. We found that CtBP Bars50 is not detectably modified in response to Exo1, whereas it is modified in the presence of BFA (Fig. 4). Therefore, tubulation of the Golgi and migration of Golgi membranes back to the ER do not require ADP-ribosylation of CtBP Bars50. Discussion As a first step toward developing new tools with which to probe events in vesicle trafficking, we describe the identification of CELL BIOLOGY Fig. 3. Exo1 acts on ARF1-GTPase through a different mechanism compared with BFA. (A) Exo1 has no effect on GEF-stimulated GDP GTP exchange on ARF1. The figure shows the effect of Exo1 and BFA on GTP loading into ARF1 by a recombinant SEC7 domain of GEF. The loading of GTP was followed as a function of time by the increase in relative fluorescence. BFA (10 M) inhibited the exchange rate by 60% (bar graph, Inset) whereas 100 M of Exo1 showed no inhibition. (B) Exo1 does not block the loading of ARF1 onto Golgi membrane. Mutant ARF1 Q71L -GFP was released from Golgi in cells treated with 5 M of BFA for 30 min. BFA was then washed out and exchanged for control medium or 100 M of Exo1. Relocalization of ARF1 Q71L -GFP from cytosol to Golgi was measured over the period of 1 h. (C) The effect of Exo1 is blocked by a chemical that masks the activity of the ARF1-GTPase. The extent of association of wild-type ARF1 (ARF1 wt -GFP) with Golgi membranes was determined in sequential images recorded by time-lapse fluorescence microscopy. The measurements were made in BSC1 cells preincubated with 50 M AlCl 3 and 30 mm NaF for 30 min, in the absence or presence of 5 M BFA or 100 M Exo1. The Golgi association of ARF1 during the preincubation period is shown (Inset). (D) A GTPase-deficient ARF1 mutant is insensitive to Exo1 treatment. The extent of association of a mutant of ARF1 defective in GTP hydrolysis (ARF1 Q71L -GFP) with Golgi membranes was determined as in C in the absence or presence of 5 M BFA or 100 M Exo1. (E) The expression of ARF1 Q71L -GFP in BSC1 cells prevents the dissociation of COPI from the Golgi. The fluorescence images show that 5 M BFA induces the dissociation of COPI in the presence of ARF1 wt -GFP or ARF1 Q71L -GFP. In contrast, Exo1 promotes dissociation of COPI only in the presence of ARF1 wt -GFP but not when ARF1 Q71L -GFP is expressed. (F) Expression of ARF1 Q71L -CFP protects cells from the block of ER to Golgi traffic and prevents collapse of the Golgi induced by Exo1. BSC1 cells coexpressing VSVG tso45 -yellow fluorescent protein (YFP) with ARF1 wt -CFP or with ARF1 Q71L -CFP were treated with 5 M BFA or 100 M Exo1 for 30 min followed by a temperature shift from 40 C to32 C. BFA promotes dissociation of ARF1 wt -CFP and ARF1 Q71L -CFP from Golgi membranes and prevents exit of VSVG tso45 -YFP from the ER. Exo1 had the same effects on cells expressing ARF1 wt -CFP, but were blocked after expression of ARF1 Q71L -CFP. Feng et al. PNAS May 27, 2003 vol. 100 no
6 Exo1 as an inhibitor of vesicular traffic from the ER to the Golgi. We have not identified the relevant protein target of Exo1 but we have shown that it is distinct from that of BFA. We present two alternative models to explain the differences between Exo1 and BFA. One possibility is that Exo1 activates a currently unidentified inhibitor of the ARF-GEF. In AlF 4 - treated cells, like coatomers, this ARF1 effector would be unable to dissociate from Golgi membranes and recycle back for a new round of GEF activity. Under these circumstances, the target of Exo1 would be sequestered and unavailable for inhibition. Similarly, expression of ARF1 Q71L with a slower rate of GTP hydrolysis could also sequester coatomer and the target of Exo1, thus decreasing the effectiveness of Exo1 to prevent GTPloading of ARF. A second possibility, which we currently prefer, is that Exo1 might act on a step other than ARF1-GDP GTP exchange. We consider it likely that Exo1 affects the balance of the futile cycle with the functional cycle of ARF1-GTP hydrolysis (see Appendix VII). ARF-GAP1 has two possible fates when it is recruited to Golgi membranes from the cytosol: either it associates with unloaded COPI coatomers and ARF1-GTP, in which case it is highly active and ARF1 has high GTPase activity (the futile cycle), or it associates with COPI coatomers that are bound to cargo receptors, in which case it gives lower activity and allows ARF1-GTP to persist for longer (functional cycle, Appendix VII) (25 27). The effects of Exo1 on GAP-dependent ARF1-GTP hydrolysis might result from interference with the futile cycle hydrolysis step, the functional cycle hydrolysis step, or both. Furthermore, Exo1 could affect the functional cycle directly by acting on the GAP, or indirectly by interfering with the formation of the complex of cargo-copi with ARF1-GTP-GAP that is required to set off the functional cycle. Such interference would cause a decrease in the amount of ARF1-GTP on the membrane because the futile cycle is faster than the functional cycle. Our current favored model is that Exo1 interferes with the formation of the ARF1-GTP-GAP-cargo-COPI complex required to initiate the functional cycle. We do not rule out the possibility that Exo1 directly or indirectly enhances GAP activity in the futile cycle but it seems unlikely because in the futile cycle, GTPase activity is already very high (at least 5 times the rate in the functional cycle) and one would expect that it would be hard to enhance this rate further. It is also possible that Exo1 selectively increases the GAP activity in the ARF1-GTP-GAPcargo-COPI complex, causing the release of ARF1 without allowing coat assembly. More work will be needed to distinguish between these models. It is clear from our results that at least one confounding effect of BFA, its ability to cause ADP-ribosylation of CtBP Bars50, can be avoided by using Exo1. Because Exo1 has no apparent effects on the TGN and endosomes, it will be a valuable reagent to study the consequences of perturbations of ER and Golgi traffic for the movement of ligands from the plasma membrane to the ER. We thank H. Vandertol-Vanier for purification of GST-Golgi-associated BFA-resistant GEF 1 (GBF1), Dr. R. Ward for editorial help, and Mr. I. Levesque for administrative assistance. We acknowledge National Institutes of Health Grants R01 GM and P01 GM (to T.K.), the Alberta Heritage Foundation for Medical Research (P.M.), the Canadian Institutes of Health Research (P.M. and T.K.R.L.), and the Howard Hughes Medical Institute Medical Student Research Fellowship (to A.P.J.). 1. Palade, G. (1975) Science 189, Rothman, J. E. (1994) Nature 372, Klausner, R. D., Donaldson, J. G. & Lippincott-Schwartz, J. (1992) J. Cell Biol. 116, Jackson, C. L. (2000) Subcell. Biochem. 34, Harri, E., Loeffler, W., Sigg, H. P. & Tamm, H. (1963) Helv. Chim. Acta 46, Lippincott-Schwartz, J., Yuan, L. C., Bonifacino, J. S. & Klausner, R. D. (1989) Cell 56, Lippincott-Schwartz, J., Yuan, L., Tipper, C., Amherdt, M., Orci, L. & Klausner, R. D. (1991) Cell 67, Peyroche, A., Antonny, B., Robineau, S., Acker, J., Cherfils, J. & Jackson, C. L. (1999) Mol. Cell 3, Mansour, S. J., Skaug, J., Zhao, X. H., Giordano, J., Scherer, S. W. & Melancon, P. (1999) Proc. Natl. Acad. Sci. USA 96, Dascher, C. & Balch, W. E. (1994) J. Biol. Chem. 269, Mironov, A., Colanzi, A., Silletta, M. G., Fiucci, G., Flati, S., Fusella, A., Polishchuk, R., Mironov, A., Jr., Di Tullio, G., Weigert, R., et al. (1997) J. Cell Biol. 139, Spano, S., Silletta, M. G., Colanzi, A., Alberti, S., Fiucci, G., Valente, C., Fusella, A., Salmona, M., Mironov, A., Luini, A., et al. (1999) J. Biol. Chem. 274, Weigert, R., Silletta, M. G., Spano, S., Turacchio, G., Cericola, C., Colanzi, A., Senatore, S., Mancini, R., Polishchuk, E. V., Salmona, M., et al. (1999) Nature 402, Scheel, J., Pepperkok, R., Lowe, M., Griffiths, G. & Kreis, T. E. (1997) J. Cell Biol. 137, Donaldson, J. G., Lippincott-Schwartz, J., Bloom, G. S., Kreis, T. E. & Klausner, R. D. (1990) J. Cell Biol. 111, Presley, J. F., Cole, N. B., Schroer, T. A., Hirschberg, K., Zaal, K. J. & Lippincott-Schwartz, J. (1997) Nature 389, Scales, S. J., Pepperkok, R. & Kreis, T. E. (1997) Cell 90, Donaldson, J. G., Finazzi, D. & Klausner, R. D. (1992) Nature 360, Rittinger, K., Walker, P. A., Eccleston, J. F., Smerdon, S. J. & Gamblin, S. J. (1997) Nature 389, Scheffzek, K., Ahmadian, M. R., Kabsch, W., Wiesmuller, L., Lautwein, A., Schmitz, F. & Wittinghofer, A. (1997) Science 277, Finazzi, D., Cassel, D., Donaldson, J. G. & Klausner, R. D. (1994) J. Biol. Chem. 269, Presley, J. F., Ward, T. H., Pfeifer, A. C., Siggia, E. D., Phair, R. D. & Lippincott-Schwartz, J. (2002) Nature 417, Kahn, R. A. (1991) J. Biol. Chem. 266, Zhang, C. J., Rosenwald, A. G., Willingham, M. C., Skuntz, S., Clark, J. & Kahn, R. A. (1994) J. Cell Biol. 124, Goldberg, J. (1999) Cell 96, Szafer, E., Rotman, M. & Cassel, D. (2001) J. Biol. Chem. 276, Lanoix, J., Ouwendijk, J., Stark, A., Szafer, E., Cassel, D., Dejgaard, K., Weiss, M. & Nilsson, T. (2001) J. Cell Biol. 155, cgi doi pnas Feng et al.
Chapter 1: Vesicular traffic. Biochimica cellulare parte B 2017/18
 Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Mechanism of Vesicular Transport
 Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Maintenance of Golgi structure and function depends on the integrity of ER export
 JCB Article Published November 12, 2001 Maintenance of Golgi structure and function depends on the integrity of ER export Theresa H. Ward, Roman S. Polishchuk, Steve Caplan, Koret Hirschberg, and Jennifer
JCB Article Published November 12, 2001 Maintenance of Golgi structure and function depends on the integrity of ER export Theresa H. Ward, Roman S. Polishchuk, Steve Caplan, Koret Hirschberg, and Jennifer
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic
 Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Legionella pneumophila: an intracellular pathogen of phagocytes Prof. Craig Roy
 an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
In the previous chapter we explored how proteins are targeted
 17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson
 Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Vesicle Transport. Vesicle pathway: many compartments, interconnected by trafficking routes 3/17/14
 Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
ArfGAP1 dynamics and its role in COPI coat assembly on Golgi membranes of living cells
 JCB: ARTICLE ArfGAP1 dynamics and its role in COPI coat assembly on Golgi membranes of living cells Wei Liu, 1 Rainer Duden, 3 Robert D. Phair, 2 and Jennifer Lippincott-Schwartz 1 1 Cell Biology and Metabolism
JCB: ARTICLE ArfGAP1 dynamics and its role in COPI coat assembly on Golgi membranes of living cells Wei Liu, 1 Rainer Duden, 3 Robert D. Phair, 2 and Jennifer Lippincott-Schwartz 1 1 Cell Biology and Metabolism
Intracellular Vesicular Traffic Chapter 13, Alberts et al.
 Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/7/334/rs4/dc1 Supplementary Materials for Rapidly rendering cells phagocytic through a cell surface display technique and concurrent Rac activation Hiroki Onuma,
www.sciencesignaling.org/cgi/content/full/7/334/rs4/dc1 Supplementary Materials for Rapidly rendering cells phagocytic through a cell surface display technique and concurrent Rac activation Hiroki Onuma,
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest.
 Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human
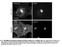 Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
endomembrane system internal membranes origins transport of proteins chapter 15 endomembrane system
 endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
7.06 Spring of PROBLEM SET #6
 7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS
 Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
04_polarity. The formation of synaptic vesicles
 Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Molecular Cell Biology 5068 In class Exam 1 October 2, Please print your name: Instructions:
 Molecular Cell Biology 5068 In class Exam 1 October 2, 2012 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
Molecular Cell Biology 5068 In class Exam 1 October 2, 2012 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
ab CytoPainter Golgi/ER Staining Kit
 ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Dynamics of proteins in Golgi membranes: comparisons between mammalian and plant cells highlighted by photobleaching techniques
 CMLS, Cell. Mol. Life Sci. 61 (2004) 172 185 1420-682X/04/020172-14 DOI 10.1007/s00018-003-3355-6 Birkhäuser Verlag, Basel, 2004 CMLS Cellular and Molecular Life Sciences Dynamics of proteins in Golgi
CMLS, Cell. Mol. Life Sci. 61 (2004) 172 185 1420-682X/04/020172-14 DOI 10.1007/s00018-003-3355-6 Birkhäuser Verlag, Basel, 2004 CMLS Cellular and Molecular Life Sciences Dynamics of proteins in Golgi
Advanced Cell Biology. Lecture 33
 Advanced Cell Biology. Lecture 33 Alexey Shipunov Minot State University April 22, 2013 Shipunov (MSU) Advanced Cell Biology. Lecture 33 April 22, 2013 1 / 38 Outline Questions and answers Intracellular
Advanced Cell Biology. Lecture 33 Alexey Shipunov Minot State University April 22, 2013 Shipunov (MSU) Advanced Cell Biology. Lecture 33 April 22, 2013 1 / 38 Outline Questions and answers Intracellular
regulation of polarized membrane transport by rab GTPases Peter van der Sluijs;
 regulation of polarized membrane transport by rab GTPases Peter van der Sluijs; p.vandersluijs@umcutrecht.nl cytotoxic T cell recognizes complex of viral peptide with MHC class I and kills infected cell
regulation of polarized membrane transport by rab GTPases Peter van der Sluijs; p.vandersluijs@umcutrecht.nl cytotoxic T cell recognizes complex of viral peptide with MHC class I and kills infected cell
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
Appendix: Subcellular localization of MIG-14::GFP in C. elegans body wall muscle cells. Pei-Tzu Yang and Hendrik C. Korswagen
 Appendix: Subcellular localization of MIG-14::GFP in C. elegans body wall muscle cells Pei-Tzu Yang and Hendrik C. Korswagen 129 SUBCELLULAR LOCALIZATION OF MIG-14 Abstract MIG-14/Wls is a Wnt binding
Appendix: Subcellular localization of MIG-14::GFP in C. elegans body wall muscle cells Pei-Tzu Yang and Hendrik C. Korswagen 129 SUBCELLULAR LOCALIZATION OF MIG-14 Abstract MIG-14/Wls is a Wnt binding
JCB. Evidence that the entire Golgi apparatus cycles in interphase HeLa cells: sensitivity of Golgi matrix proteins to an ER exit block.
 JCB Published Online: 5 November, 2001 Supp Info: http://doi.org/10.1083/jcb.200103104 Downloaded from jcb.rupress.org on October 8, 2018 Article Evidence that the entire Golgi apparatus cycles in interphase
JCB Published Online: 5 November, 2001 Supp Info: http://doi.org/10.1083/jcb.200103104 Downloaded from jcb.rupress.org on October 8, 2018 Article Evidence that the entire Golgi apparatus cycles in interphase
Project report October 2012 March 2013 CRF fellow: Principal Investigator: Project title:
 Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
2013 John Wiley & Sons, Inc. All rights reserved. PROTEIN SORTING. Lecture 10 BIOL 266/ Biology Department Concordia University. Dr. S.
 PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Intracellular vesicular traffic. B. Balen
 Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
The contribution of proteins and lipids to COPI vesicle formation and consumption. Fredrik Kartberg
 The contribution of proteins and lipids to COPI vesicle formation and consumption Fredrik Kartberg Institute of Biomedicine Department of Medical Genetics 2008 A doctoral thesis at a Swedish University
The contribution of proteins and lipids to COPI vesicle formation and consumption Fredrik Kartberg Institute of Biomedicine Department of Medical Genetics 2008 A doctoral thesis at a Swedish University
Zool 3200: Cell Biology Exam 4 Part II 2/3/15
 Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Molecular Trafficking
 SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
Downloaded from:
 Nichols, BJ; Kenworthy, AK; Polishchuk, RS; Lodge, R; Roberts, TH; Hirschberg, K; Phair, RD; Lippincott-Schwartz, J (2001) Rapid cycling of lipid raft markers between the cell surface and Golgi complex.
Nichols, BJ; Kenworthy, AK; Polishchuk, RS; Lodge, R; Roberts, TH; Hirschberg, K; Phair, RD; Lippincott-Schwartz, J (2001) Rapid cycling of lipid raft markers between the cell surface and Golgi complex.
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
Shaping tubular carriers for intracellular membrane transport
 FEBS Letters 583 (2009) 3847 3856 journal homepage: www.febsletters.org Review Shaping tubular carriers for intracellular membrane transport Roman S. Polishchuk a,b, *, Mariagrazia Capestrano a, Elena
FEBS Letters 583 (2009) 3847 3856 journal homepage: www.febsletters.org Review Shaping tubular carriers for intracellular membrane transport Roman S. Polishchuk a,b, *, Mariagrazia Capestrano a, Elena
SUPPLEMENTARY INFORMATION
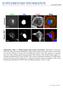 doi:10.1038/nature12004 Supplementary Figure 1 Shigella disrupts Golgi structure and function. Representative fluorescence microscopy images showing the redistribution of Golgi resident proteins upon Shigella
doi:10.1038/nature12004 Supplementary Figure 1 Shigella disrupts Golgi structure and function. Representative fluorescence microscopy images showing the redistribution of Golgi resident proteins upon Shigella
Supplemental information contains 7 movies and 4 supplemental Figures
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
Renata Schipp Medical Biology Department
 Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
Summary and Discussion antigen presentation
 Summary and Discussion antigen presentation 247 248 Summary & Discussion Summary and discussion: antigen presentation For a cell to communicate information about its internal health and status to the immune
Summary and Discussion antigen presentation 247 248 Summary & Discussion Summary and discussion: antigen presentation For a cell to communicate information about its internal health and status to the immune
Dissociation of Coatomer from Membranes Is Required for Brefeldin A induced Transfer of Golgi Enzymes to the Endoplasmic Reticulum
 Dissociation of Coatomer from Membranes Is Required for Brefeldin A induced Transfer of Golgi Enzymes to the Endoplasmic Reticulum Jochen Scheel, Rainer Pepperkok, Martin Lowe, Gareth Griffiths,* and Thomas
Dissociation of Coatomer from Membranes Is Required for Brefeldin A induced Transfer of Golgi Enzymes to the Endoplasmic Reticulum Jochen Scheel, Rainer Pepperkok, Martin Lowe, Gareth Griffiths,* and Thomas
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
Intracellular Compartments and Protein Sorting
 Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Practice Exam 2 MCBII
 1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for
 Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
Molecular Cell Biology 5068 In Class Exam 1 September 29, Please print your name:
 Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Subcellular biochemistry
 Department of Medical Biochemistry Semmelweis University Subcellular biochemistry February-March 2017 Subcellular biochemistry (biochemical aspects of cell biology) Miklós Csala Semmelweis University Dept.
Department of Medical Biochemistry Semmelweis University Subcellular biochemistry February-March 2017 Subcellular biochemistry (biochemical aspects of cell biology) Miklós Csala Semmelweis University Dept.
AP Biology
 Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Study Guide for Biology Chapter 5
 Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Cellular compartments
 Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Essential Cell Biology
 Alberts Bray Hopkin Johnson Lewis Raff Roberts Walter Essential Cell Biology FOURTH EDITION Chapter 15 Intracellular Compartments and Protein Transport Copyright Garland Science 2014 CHAPTER CONTENTS MEMBRANE-ENCLOSED
Alberts Bray Hopkin Johnson Lewis Raff Roberts Walter Essential Cell Biology FOURTH EDITION Chapter 15 Intracellular Compartments and Protein Transport Copyright Garland Science 2014 CHAPTER CONTENTS MEMBRANE-ENCLOSED
Lecture 6 - Intracellular compartments and transport I
 01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
Arf family GTP loading is activated by, and generates, positive membrane curvature
 Biochem. J. (2008) 414, 189 194 (Printed in Great Britain) doi:10.1042/bj20081237 189 ACCELERATED PUBLICATION Arf family GTP loading is activated by, and generates, positive membrane curvature Richard
Biochem. J. (2008) 414, 189 194 (Printed in Great Britain) doi:10.1042/bj20081237 189 ACCELERATED PUBLICATION Arf family GTP loading is activated by, and generates, positive membrane curvature Richard
Enzymes Part III: regulation II. Dr. Mamoun Ahram Summer, 2017
 Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER
 Article A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER Matteo Fossati 1,, Sara F Colombo 1 & Nica Borgese 1,2,* Abstract The Golgi complex and ER are dynamically
Article A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER Matteo Fossati 1,, Sara F Colombo 1 & Nica Borgese 1,2,* Abstract The Golgi complex and ER are dynamically
Overview of clathrin-mediated endocytosis
 Overview of clathrin-mediated endocytosis Accessory and adaptor proteins promote clathrin nucleation on the plasma membrane and some help deform membrane. Clathrin assembly into lattices stabilize the
Overview of clathrin-mediated endocytosis Accessory and adaptor proteins promote clathrin nucleation on the plasma membrane and some help deform membrane. Clathrin assembly into lattices stabilize the
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
Visualization of TGN to Endosome Trafficking through Fluorescently Labeled MPR and AP-1 in Living Cells V
 Molecular Biology of the Cell Vol. 14, 142 155, January 2003 Visualization of TGN to Endosome Trafficking through Fluorescently Labeled MPR and AP-1 in Living Cells V Satoshi Waguri,* Frédérique Dewitte,*
Molecular Biology of the Cell Vol. 14, 142 155, January 2003 Visualization of TGN to Endosome Trafficking through Fluorescently Labeled MPR and AP-1 in Living Cells V Satoshi Waguri,* Frédérique Dewitte,*
Lysosomes, Peroxisomes and Centrioles. Hüseyin Çağsın
 Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
Endocytosis and Intracellular Trafficking of Notch and Its Ligands
 CHA P T E R F IVE Endocytosis and Intracellular Trafficking of Notch and Its Ligands Shinya Yamamoto, *,1 Wu-Lin Charng, *,1 and Hugo J. Bellen *,,, Contents 1. Notch Signaling and its Regulation by Endocytosis
CHA P T E R F IVE Endocytosis and Intracellular Trafficking of Notch and Its Ligands Shinya Yamamoto, *,1 Wu-Lin Charng, *,1 and Hugo J. Bellen *,,, Contents 1. Notch Signaling and its Regulation by Endocytosis
Chapter 17: Vesicular traffic, secretion, and endocytosis
 Chapter 17: Vesicular traffic, secretion, and endocytosis SEM of the formation of clathrin-coated vesicles on the cytosolic face of the plasma membrane Outline: 1. Techniques for studying the secretory
Chapter 17: Vesicular traffic, secretion, and endocytosis SEM of the formation of clathrin-coated vesicles on the cytosolic face of the plasma membrane Outline: 1. Techniques for studying the secretory
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head
 CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
Module 3 Lecture 7 Endocytosis and Exocytosis
 Module 3 Lecture 7 Endocytosis and Exocytosis Endocytosis: Endocytosis is the process by which cells absorb larger molecules and particles from the surrounding by engulfing them. It is used by most of
Module 3 Lecture 7 Endocytosis and Exocytosis Endocytosis: Endocytosis is the process by which cells absorb larger molecules and particles from the surrounding by engulfing them. It is used by most of
PHSI3009 Frontiers in Cellular Physiology 2017
 Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
Lectures 18: Biological Membranes: Life in Two Dimensions (contd.)
 Lectures 18: Biological Membranes: Life in Two Dimensions (contd.) Lecturer: Brigita Urbanc Office: 1-909 (E-mail: brigita@drexel.edu) Course website: www.physics.drexel.edu/~brigita/courses/biophys_011-01/
Lectures 18: Biological Membranes: Life in Two Dimensions (contd.) Lecturer: Brigita Urbanc Office: 1-909 (E-mail: brigita@drexel.edu) Course website: www.physics.drexel.edu/~brigita/courses/biophys_011-01/
Renáta Schipp Gergely Berta Department of Medical Biology
 The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
Laboratory 10 Factors Controlling Phagocytosis in Tetrahymena,
 BIO354: Cell Biology Laboratory 1 Laboratory 1 Factors Controlling Phagocytosis in Tetrahymena, I. Introduction A characteristic feature of all eukaryotic cells is the ability to pinch off portions of
BIO354: Cell Biology Laboratory 1 Laboratory 1 Factors Controlling Phagocytosis in Tetrahymena, I. Introduction A characteristic feature of all eukaryotic cells is the ability to pinch off portions of
Introduction and protein sorting
 Introduction and protein sorting Membrane proteins Major components of cells Nucleic acids Carbohydrates Proteins Lipids (50% of mass of plasma membranes, 30% of mitochondrial membranes, 80% of myelin
Introduction and protein sorting Membrane proteins Major components of cells Nucleic acids Carbohydrates Proteins Lipids (50% of mass of plasma membranes, 30% of mitochondrial membranes, 80% of myelin
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
Chapter 13: Vesicular Traffic
 Chapter 13: Vesicular Traffic Know the terminology: ER, Golgi, vesicle, clathrin, COP-I, COP-II, BiP, glycosylation, KDEL, microtubule, SNAREs, dynamin, mannose-6-phosphate, M6P receptor, endocytosis,
Chapter 13: Vesicular Traffic Know the terminology: ER, Golgi, vesicle, clathrin, COP-I, COP-II, BiP, glycosylation, KDEL, microtubule, SNAREs, dynamin, mannose-6-phosphate, M6P receptor, endocytosis,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
Cells: The Living Units
 Chapter 3 Part B Cells: The Living Units Annie Leibovitz/Contact Press Images PowerPoint Lecture Slides prepared by Karen Dunbar Kareiva Ivy Tech Community College 3.4 Active Membrane Transport Two major
Chapter 3 Part B Cells: The Living Units Annie Leibovitz/Contact Press Images PowerPoint Lecture Slides prepared by Karen Dunbar Kareiva Ivy Tech Community College 3.4 Active Membrane Transport Two major
A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER
 Article A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER Matteo Fossati 1,, Sara F Colombo 1 & Nica Borgese 1,2,* Abstract The Golgi complex and ER are dynamically
Article A positive signal prevents secretory membrane cargo from recycling between the Golgi and the ER Matteo Fossati 1,, Sara F Colombo 1 & Nica Borgese 1,2,* Abstract The Golgi complex and ER are dynamically
Chapt. 10 Cell Biology and Biochemistry. The cell: Student Learning Outcomes: Describe basic features of typical human cell
 Chapt. 10 Cell Biology and Biochemistry Cell Chapt. 10 Cell Biology and Biochemistry The cell: Lipid bilayer membrane Student Learning Outcomes: Describe basic features of typical human cell Integral transport
Chapt. 10 Cell Biology and Biochemistry Cell Chapt. 10 Cell Biology and Biochemistry The cell: Lipid bilayer membrane Student Learning Outcomes: Describe basic features of typical human cell Integral transport
Thursday, October 16 th
 Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
Intracellular Vesicle Trafficking
 Intracellular Vesicle Trafficking Chi-Kuang Yao (IBC, Academia Sinica) 11-6-2017 ckyao@gate.sinica.edu.tw 1 Compartmentalization makes difference between bacteria and yeast 1. More compartments with specific
Intracellular Vesicle Trafficking Chi-Kuang Yao (IBC, Academia Sinica) 11-6-2017 ckyao@gate.sinica.edu.tw 1 Compartmentalization makes difference between bacteria and yeast 1. More compartments with specific
Transport through the Golgi Apparatus by Rapid Partitioning within a Two-Phase Membrane System
 Transport through the Golgi Apparatus by Rapid Partitioning within a Two-Phase Membrane System George H. Patterson, 1,6 Koret Hirschberg, 3,6 Roman S. Polishchuk, 4 Daniel Gerlich, 5 Robert D. Phair, 2,6
Transport through the Golgi Apparatus by Rapid Partitioning within a Two-Phase Membrane System George H. Patterson, 1,6 Koret Hirschberg, 3,6 Roman S. Polishchuk, 4 Daniel Gerlich, 5 Robert D. Phair, 2,6
LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role in Alzheimer s disease
 Brodeur et al. Molecular Neurodegeneration 2012, 7:31 RESEARCH ARTICLE Open Access LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role
Brodeur et al. Molecular Neurodegeneration 2012, 7:31 RESEARCH ARTICLE Open Access LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role
/07/$15.00/0 Molecular Endocrinology 21(12): Printed in U.S.A.
 0888-8809/07/$15.00/0 Molecular Endocrinology 21(12):3087 3099 Printed in U.S.A. Copyright 2007 by The Endocrine Society doi: 10.1210/me.2006-0476 The Glucose Transporter 4 FQQI Motif Is Necessary for
0888-8809/07/$15.00/0 Molecular Endocrinology 21(12):3087 3099 Printed in U.S.A. Copyright 2007 by The Endocrine Society doi: 10.1210/me.2006-0476 The Glucose Transporter 4 FQQI Motif Is Necessary for
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
CELL BIOLOGY - CLUTCH CH INTRACELLULAR PROTEIN TRANSPORT.
 !! www.clutchprep.com CONCEPT: MEMBRANE ENCLOSED ORGANELLES Table of eukaryotic organelles and their functions Organelle Function % volume of cell Cytosol Aqueous fluid where metabolic pathways and chemical
!! www.clutchprep.com CONCEPT: MEMBRANE ENCLOSED ORGANELLES Table of eukaryotic organelles and their functions Organelle Function % volume of cell Cytosol Aqueous fluid where metabolic pathways and chemical
Name Class Date. cell theory organelle eukaryotic cell. MAIN IDEA: Early studies led to the development of the cell theory.
 Section 1: Cell Theory KEY CONCEPT Cells are the basic unit of life. VOCABULARY cell theory organelle eukaryotic cell cytoplasm prokaryotic cell MAIN IDEA: Early studies led to the development of the cell
Section 1: Cell Theory KEY CONCEPT Cells are the basic unit of life. VOCABULARY cell theory organelle eukaryotic cell cytoplasm prokaryotic cell MAIN IDEA: Early studies led to the development of the cell
Dynamic Interaction of HIV-1 Nef with the Clathrin-Mediated Endocytic Pathway at the Plasma Membrane
 Traffic 2007; 8: 61 76 Blackwell Munksgaard Dynamic Interaction of HIV-1 Nef with the Clathrin-Mediated Endocytic Pathway at the Plasma Membrane # 2006 The Authors Journal compilation # 2006 Blackwell
Traffic 2007; 8: 61 76 Blackwell Munksgaard Dynamic Interaction of HIV-1 Nef with the Clathrin-Mediated Endocytic Pathway at the Plasma Membrane # 2006 The Authors Journal compilation # 2006 Blackwell
PROTEIN TRAFFICKING. Dr. SARRAY Sameh, Ph.D
 PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
Organization of ATPases
 The Primary Active Transporter II: The ATPase Objectives: Organization P type with NPA domains Proton pumps of the rotary V type ATPase 1 Organization of P type, solute transport, found in plasma membranes
The Primary Active Transporter II: The ATPase Objectives: Organization P type with NPA domains Proton pumps of the rotary V type ATPase 1 Organization of P type, solute transport, found in plasma membranes
C) You find that the Raf kinase is not constitutively active. What was necessary in the previous assay to show any Raf kinase activity?
 PROBLEM SET 3 1. You have obtained immortalized liver cells from a patient who died of Wilson s disease, an inherited disorder of copper metabolism marked by neuronal degeneration and hepatic cirrhosis.
PROBLEM SET 3 1. You have obtained immortalized liver cells from a patient who died of Wilson s disease, an inherited disorder of copper metabolism marked by neuronal degeneration and hepatic cirrhosis.
Antigen presenting cells
 Antigen recognition by T and B cells - T and B cells exhibit fundamental differences in antigen recognition - B cells recognize antigen free in solution (native antigen). - T cells recognize antigen after
Antigen recognition by T and B cells - T and B cells exhibit fundamental differences in antigen recognition - B cells recognize antigen free in solution (native antigen). - T cells recognize antigen after
Cell Membranes and Signaling
 5 Cell Membranes and Signaling Concept 5.1 Biological Membranes Have a Common Structure and Are Fluid A membrane s structure and functions are determined by its constituents: lipids, proteins, and carbohydrates.
5 Cell Membranes and Signaling Concept 5.1 Biological Membranes Have a Common Structure and Are Fluid A membrane s structure and functions are determined by its constituents: lipids, proteins, and carbohydrates.
Rewiring of Cellular Membrane Homeostasis by Picornaviruses. 1. Department of Veterinary Medicine, Virginia-Maryland Regional College of Veterinary
 JVI Accepts, published online ahead of print on 11 June 2014 J. Virol. doi:10.1128/jvi.00922-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 3 Rewiring of Cellular Membrane
JVI Accepts, published online ahead of print on 11 June 2014 J. Virol. doi:10.1128/jvi.00922-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 3 Rewiring of Cellular Membrane
