SUPPLEMENTARY INFORMATION
|
|
|
- Janis Booth
- 5 years ago
- Views:
Transcription
1 doi: /nature12004 Supplementary Figure 1 Shigella disrupts Golgi structure and function. Representative fluorescence microscopy images showing the redistribution of Golgi resident proteins upon Shigella infection are presented. To assess the trafficking of enzymes from the Endoplasmic reticulum to the Golgi apparatus, we monitored HeLa cells stably expressing green fluorescent protein (GFP) attached to NH 2 -terminal retention signal of N- acetylglucosaminyltransferase I 1 (HeLa NAGFP). HeLa cells were stimulated for production of NAGFP marker and were infected by wild type Shigella flexneri. Infection with Shigella results in the inhibition of ER to Golgi trafficking and redistribution of NAGFP marker into ER/Golgi fragments. Scale bar = 20µm. 1
2 Supplementary Figure 2 Identification of IpaJ and VirA as effectors that inhibit Golgi structure and function. a. HEK293A were transfected with Shigella flexneri Type 3 effector proteins and Golgi morphology 2
3 was then assayed with fluorescence microscopy. Transfection of IpaJ or VirA resulted in complete Golgi disruption whereas 16 other effector proteins had no effect. IpgD effector did not express in HEK293A or HeLa cells. Cis-Golgi was detected with GM130 antibodies. Scale bar = 5µm. b. Although IcsB transfection induces Golgi disruption, our data indicate that this is a non-specific affect. IcsB effector is required for lysis of cell membrane protrusion and facilitates Shigella s intercellular spreading. IcsB is distantly related to and is predicted to share papain-like thiol protease fold with Rho GTPase inactivation domain (RID) of RtxA toxin from Vibrio cholera 2. Since overexpression of IcsB induces a severe cell rounding phenotype and disappearance of plasma membrane filopodia, Golgi disruption in this case likely results from non-specific toxicity caused by alterations of the cytoskeleton. Scale bar = 20µm.c. Diagram (left) and Graph (right) showing the amount of human Growth Hormone (hgh) released from Hela cells (after drug addition) transfected with indicated expression plasmids. Experiments were performed as previously described 3,4. See extended material and methods for additional experimental details accompanying this figure. Error bars represent means ± SEM. Supplementary Figure 3 Shigella ipaj/ vira divides and exhibits actin-based motility in host cells, but does not disrupt Golgi structure. Fluorescence microscopy showing that Shigella ipaj/ vira double knockout strain has no effect on Golgi morphology. Golgi ribbon remains intact despite high pathogen burden suggesting that there are no other Shigella effectors disrupting Golgi besides IpaJ and VirA. Double deletion strain exhibits normal intracellular movement evidenced by actin comet tails. These data indicate that Golgi disruption does not result from either high intracellular bacterial replication or intracellular spread, but rather from the specific activities of IpaJ and VirA type 3 effector proteins. Shigella was detected by Dapi stain (blue), cis-golgi was detected with GM130 antibodies (green) and actin was detected by rhodamine phalloidin (red). Scale bar = 10µm. 3
4 Supplementary Figure 4 The IpaJ-family. Multiple PSI-BLAST iterations identified bacterial proteins with homology to IpaJ. Six representative family members are shown. Most of the proteins identified are putative and hypothetical proteins with unknown function. IpaJ family includes proteins from Shigella spp, Salmonella spp, Burkholderia spp, Yersinia spp, Pseudomonas spp. Invariant Cys/His/Asp residues (see Fig. 2a) are colored in red. 4
5 Supplementary Figure 5 Complementation of the Shigella ipaj/vira mutant strain. a. Fluorescence microscopy of Hela cells infected with Shigella ipaj/vira strain harboring control pbad plasmid, pbad expressing IpaJ, or pbad expressing IpaJ C64A as indicated. Golgi morphology was assessed by visualizing the localization of GM130 (green) in Shigella infected cells (Dapi). F-actin is shown (red). b. Quantification of data presented in a. The graph shows the percentage of HeLa cells with Golgi disrupted after infection with Shigella ipaj/vira double mutants complemented with the arabinose inducible pbad plasmid expressing the indicated protein. Error bars represent means ± SEM. 5
6 Supplementary Figure 6 IpaJ does not regulate the guanine-nucleotide exchange cycle, substrate binding or GTPase activity of ARF1. We performed three different experiments to test the function of IpaJ on the ARF activation and signaling cycle (Supplementary Fig. 6a). First, recombinant IpaJ was incubated with GST-tagged GDP-ARF1 followed by induction GTP-nucleotide exchange (Supplementary Fig. 6b). The ability of ARF1 to exchange nucleotide in the presence of IpaJ was assessed by the ability of GST-ARF1 pulldown GGA, a GTPdependent substrate of ARF1 5 (top panel, experiments 1). IpaJ did not inhibit GTP exchange and GGA binding under these conditions. Second, we tested the ability of IpaJ to compete for substrate interaction by incubating GST-tagged GTP-ARF1 with GGA in the presence of IpaJ. GST-pulldown experiments indicated that IpaJ did not affect GGA substrate binding under these conditions (top panel, experiment 2). Third, we asked if IpaJ stimulated GTPase activity of ARF1 by first forming complex between GST-tagged GTP-ARF1 and GGA and then adding IpaJ. IpaJ did not induce the release of GGA from ARF1-GTP, indicating that it does not function as a GTPase activating protein (GAP). Equal loading of GST-tagged ARF1 (ARF1 17 was used as this truncation mutant of ARF1 that harbors an intact GTPase domain) is shown in the bottom panel. 6
7 Supplementary Figure 7 Identification of the Glycine-2 cleavage site on ARF1 by top-down mass spectrometry. Here, we present the tandem MS/MS fragmentation maps for N-myristoylated ARF1 in untreated (a), IpaJ treated (b), and IpaJ C64A treated (c) samples (left). Proteins were subjected to online LC-MS and online tandem LC-MS/MS by source-induced disassociation. Two sequence tags, EIVTTI and EGLDWLS, were identified from the ARF1-strep sample MS/MS data. Searching these two sequence tags against the default human top-down protein database (official_human_td, basic sequences, protein forms) in ProSightPC 2.0TM identified ARF1 with a score of ProSightPC 2.0TM was used to assign fragmentation 7
8 datasets to theoretical ARF1 sequences with < 10 ppm mass accuracy (lists of matching fragment ions are presented in the corresponding Tables). These data provide an unambiguous identification of N-myristoylated ARF1-strep from these samples. a. The N-myristoylated ARF1-strep (untreated sample) had 30 fragments with 8 b-ions showing the myristoyl group being localized to the N-terminal region of the protein. b. The IpaJ treated ARF1 had 39 fragments with 12 b-ions. These provide an unambiguous assignment of ARF1-strep residues Thus IpaJ induces the cleavage between Gly2 and Asn3, thereby liberating N-myritoyl glycine from the intact ARF1 protein. The Δm mode of ProSightPC 2.0TM facilitated the assignment of a 1 Da change in molecular mass on all b-ions at the protein N-terminus. Evidence for a 1 Da shift in molecular weight is observed at the intact protein level as well (Fig. 3b). We presume that the 1Da shift is caused by a deamidation event, a known effect of the cysteine protease fold 6, occurring at the amino terminus of the ARF1 protein. Because a similar shift is not observed in untreated (a) or IpaJ C64A treated (b) sample, we assume that ARF1 deamidation is caused by IpaJ activity, yet the location and functional consequences of this event is currently unknown. c. The myristoylated ARF1-strep (IpaJ C64A sample) had 56 fragments among which 18 b-ions show the myristoyl group being localized to the N-terminal region of the protein. 8
9 Supplementary Figure 8 Free myristic acid does not inhibit IpaJ cleavage of N-myristoylated ARF1. a. Diagram illustrating the procedure for the reconstitution of cleavage of myristoylated ARF1-His by IpaJ in the presence of exogenous myristic acid. ARF1-His was co-expressed with NMTp in the presence of azide myristic acid. After cell lysis, the cleavage reaction was performed by adding lysates isolated from E. coli expressing IpaJ (wild type or C64A mutant). To determine if myristic acid inhibits the proteolytic reaction, increasing concentrations of free myristic acid were added before mixing of lysates and the cleavage reaction was performed at 37 C for 30 minutes. ARF1-His was purified Ni-NTA resin. Azide conjugated myristoylated ARF1 (green) was covalently labeled with Alex Fluor 647 Alkyne (red) by Click Chemistry and visualized by in gel fluorescence. Equal ARF1 loading was then confirmed by Coommassie Brilliant Blue staining. b. In gel fluorescence assay (top panel) visualizing Alex Fluor 647 myristoylated ARF1-His isolated from bacteria expressing NMTp. Bacterial cell lysates were left either untreated (lane 1) or were treated with IpaJ (lane2). Neither vehicle control (DMSO) (lane 3) nor free myristic acid (1 nm, 100 nm, 10 um, 100 um, 1 mm*, lanes 4-8) inhibited the proteolytic cleavage. *1 mm concentration was beyond the solubility of myristic acid and some precipitate was observed. The demyristoylation of ARF1-His occurred by proteolysis since the IpaJ C64A mutant had no effect on ARF1 N-myristoylation (lane 9). After the in gel fluorescence, the gel was stained with Coommassie Brilliant Blue to confirm equal protein loading in each reaction. This assay demonstrates that free myristic acid cannot inhibit proteolytic cleavage of N-myristoylated proteins by IpaJ. Thus, IpaJ likely recognizes myristoyl group only the protein context. 9
10 Supplementary Figure 9 IpaJ does not cleave non-myristoylated ARF1. The cleavage of ARF1-His was tested as in Figure 3e and 3f (with the exception that E. coli was not expressing yeast NMTp and was not exposed to exogenous myristic acid). Lysates of E. coli cultures producing full-length ARF1 were co-incubated with E. coli lysates producing IpaJ (wild type of C64A mutant) to allow IpaJ-dependent cleavage reaction. ARF1 was then purified using Ni-NTA resin and intact molecular weight was determined by mass spectrometry. a. 10
11 Diagram showing the theoretical molecular mass of expected ARF1-His forms purified from E. coli cultures. Full-length ARF1 (Met1-end) and ARF1-His processed by methionine aminopeptidase (Glycine2-end) are expected in all samples examined. Cleavage by IpaJ would also remove glycine-2 and expose asparagine-3, therefore producing a shorter form of ARF1 (Asn3-end). b. E. coli cellular extracts expressing ARF1-His were incubated at 37 C for 30 minutes. ARF1-His was subsequently purified on Ni-NTA resin and analyzed by mass spectrometry. As expected, only the peaks representing Met1-end ( ) and Gly2-end ( Da) forms were found and the Gly2-end peak was predominant. No Asn3-end form was found, as shown in the inset (red box). All of the peaks in the red box represent background levels of protein. c-d. E. coli cellular extracts expressing ARF1-His were incubated with the lysate containing IpaJ (c) or IpaJ C64A (d) protein at 37 C for 30 minutes and subsequently purified on Ni-NTA resin and analyzed by mass spectrometry. Importantly the mass spectrometry profile was identical to that of untreated samples shown in (b). No Asn3-end form was found in either of the treated samples, as shown in the insets. Thus IpaJ requires ARF1 N-myristoylation to perform the cleavage reaction. Supplementary Figure 10 The ARF1 myristoyl switch. The structure of yeast N-myrisoylated ARF1p in the GDP-inactive conformation (PDB ID: 2K5U) and the GTP-active conformation (PDB ID: 2KSQ). The N- myristoyl group is deeply buried in GDP-inactive ARF1p (upper panel) whereas it is extended toward the membrane in the GTP-ARF1p structure (lower panel). 11
12 Supplementary Figure 11 IpaJ C64A mutant localizes to Golgi in an ARF1 dependent manner. Imaging studies lead us to the observation that IpaJ C64A localized to the Golgi apparatus whereas neither WT IpaJ nor other catalytic mutants localized to this site (see Figure 2b). These data suggest that IpaJ C64A may function as a substrate trap by binding to its host cellular targets in a catalytically inactive complex. It is therefore 12
13 intriguing to speculate that GTP-active ARF1 recruits IpaJ C64A to the Golgi. Alternatively, IpaJ C64A may bind to other host cell targets at this site. To distinguish between these possibilities, we monitored the subcellular distribution IpaJ C64A in cells treated with Brefeldin A, fungal metabolite that locks GDP-inactive ARF1 in an unproductive complex with its cellular GEF 7. The rationale behind this experiment is that as BFA titrates away GTP-active ARF1 from the Golgi (by limiting the excess pool of GDP-ARF1 available for activation), IpaJ C64A mutant would be released from membranes in an ARF1 dependent manner. We found that egfp-ipaj C64A co-localized with mcherry ARF1 at the Golgi in untreated Hela cells (Supplementary Figure 11a). Addition of BFA rapidly displaced both IpaJ C64A and ARF1 from the Golgi membranes (Supplementary Fig. 11b). Importantly, GM130, a peripheral membrane protein that associates with the cis-golgi, was not displaced by BFA. Similar results were found when assaying IpaJ C64A localization in cells expressing endogenous ARF1 (data not shown). To assure that the loss of IpaJ C64A localization was not due to non-specific changes in Golgi membrane morphology, we treated cells with Nocodazole, a microtubule depolymerizing agent that potently disrupt ER to Golgi trafficking. Both IpaJ C64A and ARF1 co-localized on membranes in Nocodazole treated cells. As expected after microtubule depolymerization, GM130 localized to Golgi fragments, yet it did not co-localize with IpaJ C64A or ARF1. These findings support our biochemical results (Fig. 4c) that IpaJ recognizes N-mryistoylated ARF1 in its membrane-bound GTP-active conformation. a. Fluorescence microscopy of HeLa cells transfected with egfp-ipaj C64A (1) and ARF1-mCherry (2). The Golgi apparatus monitored by immunocytochemistry of GM130, a membrane-associated Golgi matrix proteins (3). We also show an illustration of the natural ARF1 activation cycle at the Golgi. Scale bar = 10µm. b. Fluorescence microscopy of HeLa cells transfected with egfp-ipaj C64A (1) and ARF1-mCherry (2) and treated for 20 minutes with BFA. The Golgi membranes were monitored by immunocytochemistry of GM130, membrane associated Golgi matrix proteins (3). We also show an illustration highlighting the effects of BFA on both ARF1 and IpaJ C64A localization as well as Golgi fragmentation. Scale bar = 10µm. c. Fluorescence microscopy of HeLa cells transfected with egfp-ipaj C64A (1) and ARF1-mCherry (2) and treated for 1 hour with Nocodazole (10µg/ml). The Golgi apparatus monitored by immunocytochemistry of GM130, a membrane-associated Golgi matrix proteins (3). We also show an illustration highlighting the effects of Nocodazole on both ARF1 and IpaJ C64A localization as well as Golgi fragmentation. Scale bar = 10µm. 13
14 Supplementary Figure 12 The cellular action of IpaJ phenocopies Brefeldin A. ARF GTPases are essential to maintain the structural and functional organization of the Golgi complex. In fact, a hallmark of ARF1 disruption by Brefeldin A (a fungal metabolite that locks ARF1 GTPase in an unproductive complex with 14
15 cellular GEFs) is the rapid redistribution of Golgi enzymes to the ER. To therefore determine if IpaJ functions in a similar fashion as BFA, we compared the subcellular distribution of the medial Golgi enzyme N- acetylglucosaminyltransferase I (NAGFP) in HeLa cells treated with BFA to cells infected with Shigella flexneri. As expected, BFA induced the redistribution of NAGFP from the Golgi to the Endoplasmic reticulum (ER) (Supplementary Fig. 12a). To directly compare this phenotype to bacterial infection, we infected NAGFP expressing HeLa cells with Shigella vira (a strain that secretes IpaJ), ipaj (a strain that secretes VirA), and the double ipaj/vira deletion mutant. As shown in Supplementary Fig. 12b, NAGFP redistributed to the ER upon Type 3 secretion of IpaJ. The ER redistribution of NAGFP phenocopies BFA, providing support that ARF1 GTPase is a cellular target of the bacterial protease. In contrast, secretion of VirA did not phenocopy BFA treatment consistent with its role as a Rab1 specific GAP 8. Importantly, Shigella ipaj/vira double mutant had no effect on NAGFP localization. In total, these data are consistent with ARF1 as a physiological target of IpaJ during Shigella infection. a. Fluorescence microscopy of NAGFP (green) in untreated (upper image) and Brefeldin A (lower image) treated cells. Enlarged regions show the clear Golgi and ER morphologies, respectively. Cartoon diagram (right panel) shows the expected localization of NAGFP before (upper diagram) and after (lower diagram) BFA treatment. Under normal conditions NAGFP resides in medial Golgi compartment. Cellular treatment with BFA induces its redistribution into the Endoplasmic Reticulum (ER). b. NAGFP (green) expressing HeLa cells were infected with the indicated mutant Shigella strains for 4 hours and visualized by fluorescence microscopy. Importantly, NAGFP redistributes to ER in Shigella vira infected cells, a gene deletion mutant that secretes IpaJ (enlarged image). The ER distribution of NAGFP induced by IpaJ phenocopies BFA treatment (compare to a.). To compare the Golgi disruption phenotype caused by IpaJ to that of VirA, we examined the localization of NAGFP in cells infected with Shigella ipaj, a gene deletion mutant that secretes VirA. NAGFP formed large membrane aggregations in Shigella ipaj infected cells (enlarged image). This phenotype did not resemble BFA, which is consistent with the function of VirA as a Rab1 specific GAP 8. In control experiments, we found that Shigella ipaj/vira double mutant had no effect on NAGFP localization. The merged image shows NAGFP (green), Dapi (blue, shows both Hela nuclei and Shigella DNA), and F-actin (red, Rhodamine phalloidin). 15
16 Supplementary Figure 13 Subcellular localization of N-myristoylated GRASP65 proteins during Shigella infection. Data presented in Figures 4d and 4e indicate that Shigella displaces ARF1 GTPase from host membranes as mechanism of Golgi disruption by IpaJ. However, several N-myristoylated proteins reside at the Golgi and are required for its structure and function 9,10. N-myristoylation of GRASP65 is required for its initial recruitment to Golgi, where it is subsequently maintained at this site through both lipidation and secondary protein:protein interactions 11. For example, GRASP65 forms a complex with GM130, which together maintain the structural organization of Golgi cisternae 9. To determine if Shigella also displaces these N-myristoylated substrates from the Golgi, we examined the subcellular localization of GRASP65 during Shigella infection. As shown in Supplementary Fig. 13a, GRASP65 co-localizes with GM130 at the cis-golgi. While Shigella infection induces severe Golgi fragmentation, both GRASP65 and GM130 maintain their membrane association (Supplementary Fig. 13b). Similar results were also observed with Shigella ipaj (which secretes VirA) mutants (data not shown). These data indicate that IpaJ does not cause the release of N-myristoylated GRASP65 from Golgi as a mechanism of organelle destruction. It is important to point out that we do not rule out the possibility that cleavage of GRASP65 results in a loss of membrane orientation that is required for Golgi cisternae organization 11. Even in this unlikely event, when coupled with our findings in Supplementary Figures 11 and 12 these data suggest that elimination of ARF1 N-myristoylation by IpaJ expression is largely responsible for the 16
17 Golgi fragmentation induced by Shigella infection. a. Fluorescence microscopy of GRASP65 (green) and GM130 (blue) in untreated cells. Enlarged regions show the localization of these proteins to the Golgi apparatus. b. Fluorescence microscopy of GRASP65 (green) and GM130 (blue) in cells infected with WT Shigella M90T. Enlarged regions show the localization of these proteins to disrupted Golgi membranes. Scale bar = 10µm. References 1 Shima, D. T., Haldar, K., Pepperkok, R., Watson, R. & Warren, G. Partitioning of the Golgi apparatus during mitosis in living HeLa cells. The Journal of cell biology 137, (1997). 2 Pei, J. & Grishin, N. V. The Rho GTPase inactivation domain in Vibrio cholerae MARTX toxin has a circularly permuted papain-like thiol protease fold. Proteins 77, , doi: /prot (2009). 3 Rivera, V. M. et al. Regulation of protein secretion through controlled aggregation in the endoplasmic reticulum. Science (New York, N.Y 287, (2000). 4 Selyunin, A. S. et al. The assembly of a GTPase-kinase signalling complex by a bacterial catalytic scaffold. Nature 469, , doi: /nature09593 (2011). 5 Puertollano, R., Randazzo, P. A., Presley, J. F., Hartnell, L. M. & Bonifacino, J. S. The GGAs promote ARFdependent recruitment of clathrin to the TGN. Cell 105, (2001). 6 Schmidt, G., Selzer, J., Lerm, M. & Aktories, K. The Rho-deamidating cytotoxic necrotizing factor 1 from Escherichia coli possesses transglutaminase activity. Cysteine 866 and histidine 881 are essential for enzyme activity. The Journal of biological chemistry 273, (1998). 7 Chardin, P. & McCormick, F. Brefeldin A: the advantage of being uncompetitive. Cell 97, (1999). 8 Dong, N. et al. Structurally Distinct Bacterial TBC-like GAPs Link Arf GTPase to Rab1 Inactivation to Counteract Host Defenses. Cell 150, , doi: /j.cell (2012). 9 Barr, F. A., Nakamura, N. & Warren, G. Mapping the interaction between GRASP65 and GM130, components of a protein complex involved in the stacking of Golgi cisternae. The EMBO journal 17, , doi: /emboj/ (1998). 10 Shorter, J. et al. GRASP55, a second mammalian GRASP protein involved in the stacking of Golgi cisternae in a cell-free system. The EMBO journal 18, , doi: /emboj/ (1999). 11 Bachert, C. & Linstedt, A. D. Dual anchoring of the GRASP membrane tether promotes trans pairing. The Journal of biological chemistry 285, , doi: /jbc.m (2010). 17
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest.
 Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
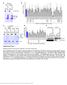 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
SUPPLEMENTARY FIGURE LEGENDS
 SUPPLEMENTARY FIGURE LEGENDS Supplemental FIG. 1. Localization of myosin Vb in cultured neurons varies with maturation stage. A and B, localization of myosin Vb in cultured hippocampal neurons. A, in DIV
SUPPLEMENTARY FIGURE LEGENDS Supplemental FIG. 1. Localization of myosin Vb in cultured neurons varies with maturation stage. A and B, localization of myosin Vb in cultured hippocampal neurons. A, in DIV
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human
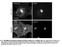 Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/7/334/rs4/dc1 Supplementary Materials for Rapidly rendering cells phagocytic through a cell surface display technique and concurrent Rac activation Hiroki Onuma,
www.sciencesignaling.org/cgi/content/full/7/334/rs4/dc1 Supplementary Materials for Rapidly rendering cells phagocytic through a cell surface display technique and concurrent Rac activation Hiroki Onuma,
Nature Methods: doi: /nmeth.4257
 Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic
 Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Legionella pneumophila: an intracellular pathogen of phagocytes Prof. Craig Roy
 an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
Supplementary Figure S1
 Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS
 Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles
 Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles Amin Feizpour Reinhard Lab Department of Chemistry and the Photonics Center, Boston University, Boston, MA May 2014
Quantifying Lipid Contents in Enveloped Virus Particles with Plasmonic Nanoparticles Amin Feizpour Reinhard Lab Department of Chemistry and the Photonics Center, Boston University, Boston, MA May 2014
IP: anti-gfp VPS29-GFP. IP: anti-vps26. IP: anti-gfp - + +
 FAM21 Strump. WASH1 IP: anti- 1 2 3 4 5 6 FAM21 Strump. FKBP IP: anti-gfp VPS29- GFP GFP-FAM21 tail H H/P P H H/P P c FAM21 FKBP Strump. VPS29-GFP IP: anti-gfp 1 2 3 FKBP VPS VPS VPS VPS29 1 = VPS29-GFP
FAM21 Strump. WASH1 IP: anti- 1 2 3 4 5 6 FAM21 Strump. FKBP IP: anti-gfp VPS29- GFP GFP-FAM21 tail H H/P P H H/P P c FAM21 FKBP Strump. VPS29-GFP IP: anti-gfp 1 2 3 FKBP VPS VPS VPS VPS29 1 = VPS29-GFP
Appendix. Table of Contents
 Appendix Table of Contents Appendix Figures Figure S1: Gp78 is not required for the degradation of mcherry-cl1 in Hela Cells. Figure S2: Indel formation in the MARCH6 sgrna targeted HeLa clones. Figure
Appendix Table of Contents Appendix Figures Figure S1: Gp78 is not required for the degradation of mcherry-cl1 in Hela Cells. Figure S2: Indel formation in the MARCH6 sgrna targeted HeLa clones. Figure
Ras and Cell Signaling Exercise
 Ras and Cell Signaling Exercise Learning Objectives In this exercise, you will use, a protein 3D- viewer, to explore: the structure of the Ras protein the active and inactive state of Ras and the amino
Ras and Cell Signaling Exercise Learning Objectives In this exercise, you will use, a protein 3D- viewer, to explore: the structure of the Ras protein the active and inactive state of Ras and the amino
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Supplementary figure legends
 Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
SUPPLEMENTARY INFORMATION
 sirna pool: Control Tetherin -HA-GFP HA-Tetherin -Tubulin Supplementary Figure S1. Knockdown of HA-tagged tetherin expression by tetherin specific sirnas. HeLa cells were cotransfected with plasmids expressing
sirna pool: Control Tetherin -HA-GFP HA-Tetherin -Tubulin Supplementary Figure S1. Knockdown of HA-tagged tetherin expression by tetherin specific sirnas. HeLa cells were cotransfected with plasmids expressing
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR
 Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Supplementary Figure 1
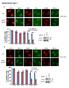 Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes
 Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes Armen Petrosyan 1*, Pi-Wan Cheng 1,3, Dahn L. Clemens 2,3 & Carol A. Casey 2,3 1 Department
Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes Armen Petrosyan 1*, Pi-Wan Cheng 1,3, Dahn L. Clemens 2,3 & Carol A. Casey 2,3 1 Department
Signal Transduction: G-Protein Coupled Receptors
 Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Nature Biotechnology: doi: /nbt.3828
 Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
crossmark Ca V subunits interact with the voltage-gated calcium channel
 crossmark THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 291, NO. 39, pp. 20402 20416, September 23, 2016 Author s Choice 2016 by The American Society for Biochemistry and Molecular Biology, Inc. Published in
crossmark THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 291, NO. 39, pp. 20402 20416, September 23, 2016 Author s Choice 2016 by The American Society for Biochemistry and Molecular Biology, Inc. Published in
Supplementary Materials
 Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) (b) (c) (d) (e) (f) (g) .
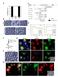 Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) The inhibitory effects of new antibodies (Mab17 and Mab18). They were investigated in in vitro fertilization
Supplementary Figure 1. Properties of various IZUMO1 monoclonal antibodies and behavior of SPACA6. (a) The inhibitory effects of new antibodies (Mab17 and Mab18). They were investigated in in vitro fertilization
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson
 Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
genome edited transient transfection, CMV promoter
 Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Vesicle Transport. Vesicle pathway: many compartments, interconnected by trafficking routes 3/17/14
 Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb /DC1
 Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
A Bacterial Virulence Protein Suppresses Host Innate Immunity to Cause Plant Disease
 A Bacterial Virulence Protein Suppresses Host Innate Immunity to Cause Plant Disease Nomura, K., Debroy, S., Lee, Y.H., Pumplin, N., Jones, J., and He, S.Y. (2006). Science 313, 220-223. Presented by:
A Bacterial Virulence Protein Suppresses Host Innate Immunity to Cause Plant Disease Nomura, K., Debroy, S., Lee, Y.H., Pumplin, N., Jones, J., and He, S.Y. (2006). Science 313, 220-223. Presented by:
a b G75 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server.
 a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
SUPPLEMENTARY INFORMATION
 Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
Double charge of 33kD peak A1 A2 B1 B2 M2+ M/z. ABRF Proteomics Research Group - Qualitative Proteomics Study Identifier Number 14146
 Abstract The 2008 ABRF Proteomics Research Group Study offers participants the chance to participate in an anonymous study to identify qualitative differences between two protein preparations. We used
Abstract The 2008 ABRF Proteomics Research Group Study offers participants the chance to participate in an anonymous study to identify qualitative differences between two protein preparations. We used
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Chapter 1: Vesicular traffic. Biochimica cellulare parte B 2017/18
 Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Effects of Second Messengers
 Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Supplementary Figure 1. Spatial distribution of LRP5 and β-catenin in intact cardiomyocytes. (a) and (b) Immunofluorescence staining of endogenous
 Supplementary Figure 1. Spatial distribution of LRP5 and β-catenin in intact cardiomyocytes. (a) and (b) Immunofluorescence staining of endogenous LRP5 in intact adult mouse ventricular myocytes (AMVMs)
Supplementary Figure 1. Spatial distribution of LRP5 and β-catenin in intact cardiomyocytes. (a) and (b) Immunofluorescence staining of endogenous LRP5 in intact adult mouse ventricular myocytes (AMVMs)
SUPPLEMENTARY INFORMATION
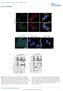 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
Supplemental Information. Helicobacter pylori Employs a Unique Basolateral. Type IV Secretion Mechanism for CagA Delivery
 Cell Host & Microbe, Volume 22 Supplemental Information Helicobacter pylori Employs a Unique Basolateral Type IV Secretion Mechanism for CagA Delivery Nicole Tegtmeyer, Silja Wessler, Vittorio Necchi,
Cell Host & Microbe, Volume 22 Supplemental Information Helicobacter pylori Employs a Unique Basolateral Type IV Secretion Mechanism for CagA Delivery Nicole Tegtmeyer, Silja Wessler, Vittorio Necchi,
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures
 Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/7/308/ra4/dc1 Supplementary Materials for Antipsychotics Activate mtorc1-dependent Translation to Enhance Neuronal Morphological Complexity Heather Bowling, Guoan
www.sciencesignaling.org/cgi/content/full/7/308/ra4/dc1 Supplementary Materials for Antipsychotics Activate mtorc1-dependent Translation to Enhance Neuronal Morphological Complexity Heather Bowling, Guoan
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Lu et al., http://www.jcb.org/cgi/content/full/jcb.201012063/dc1 Figure S1. Kinetics of nuclear envelope assembly, recruitment of Nup133
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Lu et al., http://www.jcb.org/cgi/content/full/jcb.201012063/dc1 Figure S1. Kinetics of nuclear envelope assembly, recruitment of Nup133
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering
 nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
In the previous chapter we explored how proteins are targeted
 17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
CELLS. Cells. Basic unit of life (except virus)
 Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
04_polarity. The formation of synaptic vesicles
 Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Nature Immunology: doi: /ni eee Supplementary Figure 1
 eee Supplementary Figure 1 Hyphae induce NET release, but yeast do not. (a) NET release by human peripheral neutrophils stimulated with a hgc1 yeast-locked C. albicans mutant (yeast) or pre-formed WT C.
eee Supplementary Figure 1 Hyphae induce NET release, but yeast do not. (a) NET release by human peripheral neutrophils stimulated with a hgc1 yeast-locked C. albicans mutant (yeast) or pre-formed WT C.
Dramatic increase in SHP2 binding activity of Helicobacter. pylori Western CagA by EPIYA-C duplication: its
 Supplementary Information Dramatic increase in SHP2 binding activity of Helicobacter pylori Western CagA by EPIYA-C duplication: its implications in gastric carcinogenesis Lisa Nagase, Takeru Hayashi,
Supplementary Information Dramatic increase in SHP2 binding activity of Helicobacter pylori Western CagA by EPIYA-C duplication: its implications in gastric carcinogenesis Lisa Nagase, Takeru Hayashi,
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
endomembrane system internal membranes origins transport of proteins chapter 15 endomembrane system
 endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
Project report October 2012 March 2013 CRF fellow: Principal Investigator: Project title:
 Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
JCB. Evidence that the entire Golgi apparatus cycles in interphase HeLa cells: sensitivity of Golgi matrix proteins to an ER exit block.
 JCB Published Online: 5 November, 2001 Supp Info: http://doi.org/10.1083/jcb.200103104 Downloaded from jcb.rupress.org on October 8, 2018 Article Evidence that the entire Golgi apparatus cycles in interphase
JCB Published Online: 5 November, 2001 Supp Info: http://doi.org/10.1083/jcb.200103104 Downloaded from jcb.rupress.org on October 8, 2018 Article Evidence that the entire Golgi apparatus cycles in interphase
Effects of UBL5 knockdown on cell cycle distribution and sister chromatid cohesion
 Supplementary Figure S1. Effects of UBL5 knockdown on cell cycle distribution and sister chromatid cohesion A. Representative examples of flow cytometry profiles of HeLa cells transfected with indicated
Supplementary Figure S1. Effects of UBL5 knockdown on cell cycle distribution and sister chromatid cohesion A. Representative examples of flow cytometry profiles of HeLa cells transfected with indicated
7.06 Spring of PROBLEM SET #6
 7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
Supplementary Information. Supplementary Figure 1
 Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Supplements. Figure S1. B Phalloidin Alexa488
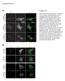 Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role in Alzheimer s disease
 Brodeur et al. Molecular Neurodegeneration 2012, 7:31 RESEARCH ARTICLE Open Access LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role
Brodeur et al. Molecular Neurodegeneration 2012, 7:31 RESEARCH ARTICLE Open Access LDLR-related protein 10 (LRP10) regulates amyloid precursor protein (APP) trafficking and processing: evidence for a role
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC
 Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
A. Generation and characterization of Ras-expressing autophagycompetent
 Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
Nature Medicine: doi: /nm.4322
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
AP Biology
 Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Supplemental information contains 7 movies and 4 supplemental Figures
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
Name: Multiple choice questions. Pick the BEST answer (2 pts ea)
 Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
SUPPLEMENTARY INFORMATION
 SUPPLEENTRY INFORTION DOI: 1.138/ncb2577 Early Telophase Late Telophase B icrotubules within the ICB (percent of total cells in telophase) D G ultinucleate cells (% total) 8 6 4 2 2 15 1 5 T without gaps
SUPPLEENTRY INFORTION DOI: 1.138/ncb2577 Early Telophase Late Telophase B icrotubules within the ICB (percent of total cells in telophase) D G ultinucleate cells (% total) 8 6 4 2 2 15 1 5 T without gaps
Chapter 6. Antigen Presentation to T lymphocytes
 Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Intracellular Compartments and Protein Sorting
 Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
10/13/11. Cell Theory. Cell Structure
 Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature13418 Supplementary Results: USP30 opposes autophagic flux In HEK-293 cells, USP30 overexpression increased basal LC3-II levels, dependent on enzymatic activity,
SUPPLEMENTARY INFORMATION doi:10.1038/nature13418 Supplementary Results: USP30 opposes autophagic flux In HEK-293 cells, USP30 overexpression increased basal LC3-II levels, dependent on enzymatic activity,
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Prolonged mitotic arrest induces a caspase-dependent DNA damage
 SUPPLEMENTARY INFORMATION Prolonged mitotic arrest induces a caspase-dependent DNA damage response at telomeres that determines cell survival Karolina O. Hain, Didier J. Colin, Shubhra Rastogi, Lindsey
SUPPLEMENTARY INFORMATION Prolonged mitotic arrest induces a caspase-dependent DNA damage response at telomeres that determines cell survival Karolina O. Hain, Didier J. Colin, Shubhra Rastogi, Lindsey
Nature Structural & Molecular Biology: doi: /nsmb.3218
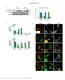 Supplementary Figure 1 Endogenous EGFR trafficking and responses depend on biased ligands. (a) Lysates from HeLa cells stimulated for 2 min. with increasing concentration of ligands were immunoblotted
Supplementary Figure 1 Endogenous EGFR trafficking and responses depend on biased ligands. (a) Lysates from HeLa cells stimulated for 2 min. with increasing concentration of ligands were immunoblotted
Supplementary Figure 1. MAT IIα is Acetylated at Lysine 81.
 IP: Flag a Mascot PTM Modified Mass Error Position Gene Names Score Score Sequence m/z [ppm] 81 MAT2A;AMS2;MATA2 35.6 137.28 _AAVDYQK(ac)VVR_ 595.83-2.28 b Pre-immu After-immu Flag- WT K81R WT K81R / Flag
IP: Flag a Mascot PTM Modified Mass Error Position Gene Names Score Score Sequence m/z [ppm] 81 MAT2A;AMS2;MATA2 35.6 137.28 _AAVDYQK(ac)VVR_ 595.83-2.28 b Pre-immu After-immu Flag- WT K81R WT K81R / Flag
Chapter 9. Cellular Signaling
 Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al
 Supplementary Material Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al Supplementary Figure 1. AICAR improves P23H rod opsin
Supplementary Material Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al Supplementary Figure 1. AICAR improves P23H rod opsin
