GTP/GDP exchange by Sec12p enables COPII vesicle bud formation on synthetic liposomes
|
|
|
- Cecil Norris
- 5 years ago
- Views:
Transcription
1 he EMBO Journal (24) 23, & 24 European Molecular Biology Organization All Rights Reserved /4 GP/GP exchange by Sec12p enables COPII vesicle bud formation on synthetic liposomes HE EMBO JOURNAL Eugene Futai 1, Susan Hamamoto 1,2, Lelio Orci 3 and Randy Schekman 1,2, * 1 epartment of Molecular and Cell Biology, University of California, Berkeley, CA, USA, 2 Howard Hughes Medical Institute, University of California, Berkeley, CA, USA and 3 epartment of Cellular Physiology and Metabolism, University of Geneva Medical School, Geneva, Switzerland he generation of COPII vesicles from synthetic liposome membranes requires the minimum coat components Sar1p,, Sec13/31p, and a nonhydrolyzable GP analog such as GMP-PNP. However, in the presence of GP and the full complement of coat subunits, nucleotide hydrolysis by Sar1p renders the coat insufficiently stable to sustain vesicle budding. In order to recapitulate a more authentic, GP-dependent budding event, we introduced the Sar1p nucleotide exchange catalyst, Sec12p, and evaluated the dynamics of coat assembly and disassembly by light scattering and tryptophan fluorescence measurements. he catalytic, cytoplasmic domain of Sec12p (Sec12Cp) activated Sar1p with a turnover 1-fold higher than the GAP activity of Sec23p stimulated by the full coat. COPII assembly was stabilized on liposomes incubated with Sec12Cp and GP. Numerous COPII budding profiles were visualized on membranes, whereas a parallel reaction conducted in the absence of Sec12Cp produced no such profiles. We suggest that Sec12p participates actively in the growth of COPII vesicles by charging new Sar1p- GP molecules that insert at the boundary between a bud and the surrounding endoplasmic reticulum membrane. he EMBO Journal (24) 23, doi:1.138/ sj.emboj.76428; Published online 3 September 24 Subject Categories: membranes & transport Keywords: COPII; GAP; GEF; vesicle transport Introduction In eukaryotic cells, transport between the compartments of the secretory pathway is mediated by vesicles, which are produced by budding from donor membranes and fuse with acceptor membranes. he mechanisms of vesicle formation and fusion have been investigated in great detail in recent years, largely through the in vitro reconstitution of transport steps with pure components (Bonifacino and Glick, 24). Cytoplasmic coat complexes drive vesicle biogenesis and act during different transport steps: clathrin and its assembly proteins function in the late secretory pathway, the COPI coat *Corresponding author. epartment of Molecular and Cell Biology, University of California, Berkeley, CA , USA. el.: þ ; Fax: þ ; schekman@berkeley.edu Received: 14 July 24; accepted: 6 September 24; published online: 3 September 24 acts in intra-golgi and retrograde Golgi endoplasmic reticulum (ER) transport, and the COPII coat acts in ER Golgi transport. hese coat proteins interact directly with cargo proteins (Cosson and Letourneur, 1994; Hoflack, 1998; Springer and Schekman, 1998) and also deform the membrane to form a bud (Matsuoka et al, 1998b; Spang et al, 1998; Peter et al, 24), thereby performing the essential aspects of transport: cargo concentration and membrane deformation. he Ras family G-proteins, ARF and Sar1p, appear to be the main regulators of vesicle formation. ARF and Sar1p are activated by binding GP, which stimulates coat assembly. Small GPases have very low intrinsic nucleotide exchange or GPase activity; thus, these events are regulated by guanine nucleotide exchange factors (GEFs) and GPase-activating proteins (GAPs), which are required for activation and inactivation of the GPase, respectively (Vetter and Wittinghofer, 21). Genetic studies in yeast revealed that Sec12p, Sec13p, Sec16p, Sec23p, Sec24p, Sec31p, and Sar1p are essential for vesicle formation at the ER. hese proteins contribute to the formation of the COPII coat, which is composed of heteromeric protein complexes, comprising Sec23p/Sec24p (heterodimer) and Sec13p/Sec31p (heterotetramer), and the Sar1p GPase. Sec23p acts as a GAP specific for Sar1p (Yoshihisa et al, 1993). Sec23p/Sec24p is responsible for the selection of cargo proteins for packaging, providing binding sites for multiple cargo proteins (Miller et al, 23; Mossessova et al, 23). Sec13/31p bridges molecules bound to cargo proteins to create a coat that envelops the membrane into a bud (Matsuoka et al, 21). he entire process is localized and initiated by Sec12p, an ER-resident membrane GEF that recruits and activates Sar1p (Barlowe and Schekman, 1993). An additional organizing factor, Sec16p, associates with activated Sar1p to provide binding sites for Sec23p, Sec24p, and Sec31p (Espenshade et al, 1995; Gimeno et al, 1996; Shaywitz et al, 1997; Supek et al, 22). he mechanism of COPII coat and vesicle formation has been probed with liposomes formulated with defined, synthetic phospholipids. However, an authentic budding reaction that reproduces the normal physiologic event has yet to be recapitulated with the full set of Sec proteins known to operate in vivo. he minimum requirements for synthetic COPII vesicle formation are Sar1p, the core coat subunits, and a nonhydrolyzable analog of GP, GMP-PNP (Matsuoka et al, 1998b). In conditions of low Mg 2 þ, Sar1p is activated by spontaneous nucleotide exchange and recruited to the membrane. he next two coat complexes are recruited sequentially. interacts with both Sar1p-GMP-PNP and the charged head groups of acidic phospholipids. Sec13/31p is then recruited, presumably through interactions with both Sar1p and. uring the process of coat recruitment, the membrane is deformed to generate vesicles (Matsuoka et al, 1998b). Even in the absence of liposomes, high concentrations of and Sec13/31p polymerize into a 4146 he EMBO Journal VOL 23 NO & 24 European Molecular Biology Organization
2 cage structure, suggesting that these complexes have an intrinsic ability to assemble (Antonny et al, 23). COPII coats formed on liposomes with GP are very unstable and dissociate rapidly from the membrane due to the high GAP activity of the assembled coat (Antonny et al, 21). he GAP activity of is stimulated B1-fold by Sec13/31p, resulting in a built-in coat disassembly mechanism (Antonny et al, 21). hus, although coated vesicles form in a synthetic budding reaction conducted in the presence of GMP-PNP, no evidence of budding vesicles is seen in a reaction containing GP. Our goal has been to reconstitute the salient features of this process and thereby resolve the essential mechanistic questions. How can the intrinsic instability of the COPII coat be reconciled with the efficient budding of COPII vesicles from microsomes in the presence of GP (Barlowe et al, 1994), and indeed with the use of GP by living cells? One hypothesis is that proteins within the ER membrane inhibit the GAP activity or stabilize the coat after GP hydrolysis. Cargo proteins may fulfill this role; peptides of p24a protein inhibit Arf1p-GAP activity during COPI coat formation (Goldberg, 2). Other candidates are the essential genes for COPII formation, Sec16p and Sec12p. Sec16p has binding sites for individual coat subunits and is thought to nucleate the COPII budding sites. However, Sec16p showed no effect on Sec23/ 24p GAP activity and only partial coat stabilization after GP hydrolysis (Supek et al, 22). In this study, we show that the GEF activity of Sec12p surpasses the GAP activity of Sec23/ 24p and promotes coat stability and vesicle bud formation on liposomes in reactions containing GP. Results Sec12p keeps COPII coats with Sar1p activated by GP he real-time assembly of the COPII coat on liposomes can be monitored and quantified by light scattering (Antonny et al, 21). he intensity of scattered light depends on a mass increase associated with coat polymerization on the liposome surface (Antonny et al, 21). Using the light scattering assay, we directly assessed the effect of the cytoplasmic domain of Sec12p (Sec12Cp) on coat formation on liposomes (major minor mix of lipids) previously established as optimal for COPII recruitment (Matsuoka et al, 1998b). Coat assembly was monitored by the sequential recruitment of each coat component. In the absence of Sec12Cp, Sar1p exchanged GP for GP slowly and bound to liposomes to produce a gradual increase in the light scattering signal. Addition of yielded a sharp increase in the light scattering followed by a slow decay with a half-life of B2 min (Figure 1A). Addition of Sec13/31p induced a rapid assembly/disassembly response as previously reported (Figure 1A; Antonny et al, 21). he spontaneous exchange of nucleotide by Sar1p is facilitated in incubations containing a low concentration of Mg 2 þ. Addition of EA to a standard incubation accelerated the rise in the Sar1p-GP-liposome light scattering signal and stabilized the, Sar1p-GP-liposome complex. However, subsequent addition of Sec13/31p produced the usual unstable coat complex. Addition of Sec12Cp produced a more rapid rise in the light scattering signal, consistent with faster nucleotide exchange on Sar1p and enhanced recruitment of Sar1p-GP by the membrane. he coat intermediate formed on addition of, and the completion of coat assembly by Sec13/ 31p were significantly stabilized by Sec12Cp. (Figure 1A) his stabilization was more obvious in incubations containing Sec13/31p from the outset (Figure 1B). In such a reaction, the coat formed and disassembled rapidly on addition of in the absence of Sec12Cp (t 1/2 B2 s), but depolymerized slowly (t 1/2 B15 min) in the presence of Sec12Cp (Figure 1B). As before, EA did not promote stabilization of the fully assembled coat. hese results suggest that the continuous generation of Sar1p-GP promotes COPII coat stability. In the absence of Sec12Cp, the order of addition of coat proteins with respect to the nucleotide was critical, because coat formation was limited by the slow Sar1p activation by spontaneous nucleotide exchange. When we added GP to a mixture containing all three coat components, no binding signal was observed (Figure 1C). However, in the presence of Sec12Cp, the addition of GP induced an instant increase in the signal followed by a slow decay (Figure 1C). hus, the potent exchange of nucleotide catalyzed by Sec12Cp more than compensated for the high GAP activity of the full coat. Under these conditions, coat assembly and stability were similar in incubations containing Sec12Cp with GP or GMP-PNP (Figure 1C). However, at lower concentrations of GP (1 mm), we observed a decrease in coat stability (t 1/2 B45 s) (Figure 1), suggesting that GP turnover enhanced by continuous nucleotide exchange and hydrolysis limited coat longevity. We examined the activity of Sec12Cp on liposomes composed of neutral phospholipids (PC/PE), a formulation that does not permit the stable recruitment of coat subunits. Whereas Sec12Cp bound well to major minor mix liposomes, it was not recruited significantly to PC/PE liposomes in a liposome flotation assay (Figure 1E) (Matsuoka et al, 1998b). Although coat subunits induced a transient light scattering signal on PC/PE liposomes, Sec12Cp was unable to stabilize the coat on these membranes (Figure 1F). hese results suggest that Sec12Cp acts from a membrane surface, in this case most likely bound electrostatically, rather than from solution to promote coat assembly and stability. Relative rates of Sec12pGEF and Sec23pGAP activity o better quantify the effect of Sec12p on coat assembly, we employed a tryptophan fluorescence assay that detects a fluorescence change in Sar1p upon nucleotide exchange or hydrolysis (Antonny et al, 21). he experiment in Figure 2A shows the relative rates of nucleotide exchange in incubations of Sar1p with GP, GP þ EA, and GP þ Sec12Cp. In addition to a substantial rate enhancement, Sec12Cp increased the amplitude of Sar1p-GP exchange; Sec12Cp promoted reasonably stable retention of Sar1p-GP in these incubations, which contained the full set of COPII proteins. he retention of Sar1-GP declined in incubations with coat proteins and lower concentrations of GP (data not shown). Sec12Cp did not support exchange activity in incubations containing neutral (PC/PE) liposomes, consistent with the diminished binding of the protein to such membranes (data not shown). We determined the GEF and GAP activities from the rates of Sar1p activation and inactivation (Figure 2B). he exchange rate catalyzed by Sec12Cp was 77.6 & 24 European Molecular Biology Organization he EMBO Journal VOL 23 NO
3 A Light scattering (normalized) Light scattering (normalized) B EA ( or EA at t =) major minor liposome Sec 23/24p Sec 13/31p Sar1pGP GP (+Sec13/31p and Sec12ΔCp or EA at t =) Sar1pGP GP EA major minor liposome Light scattering (normalized) E (+Sec13/31p and Sec12ΔCp at t =) Sar1pGP GP Major minor 2 PC-PE ime (s) 6 Input 1. M.75 M Float + μm GP +1 μm GP Without GP 7 8 C Light scattering (normalized) ( at t =) Sec13/31p Sar1p GP or GMP-PNP 2 ime (s) 3 major minor liposome 4 5 F Light scattering (normalized) (+Sec13/31p and Sec12ΔCp at t =) Sar1pGP GP 2 ime (s) 3 PC-PE liposome Figure 1 Sec12Cp stabilizes GP-driven COPII coat assembly on liposomes. (A ) he light scattering of a suspension of major minor mix liposomes ( mgml 1 ) was continuously monitored upon the addition of 95 nm Sar1p GP, 16 nm, 26 nm Sec13/31p, and mm GP or GMP-PNP, at specific time points as indicated by arrows. (A, B) he initial mixture contained 5 nm Sec12Cp (red trace), or 2 mm EA (blue traces) in HKM buffer (2 mm Hepes-K, ph 6.8, 16 mm KOAc, 1 mm MgCl 2 ), or buffer alone (black trace). EA decreases the concentration of free Mg 2 þ from 1 mm to 1 mm, and accelerates GP GP exchange by Sar1p. Sec13/31p was added to the initial mixture in (B). (C) Sar1p-GP was activated by addition of GP (bold trace) or GMP-PNP (thin trace). he initial mixture contained 5 nm Sec12Cp in HKM buffer (red trace), or buffer alone (black trace). () ifferent levels of GP were added to the reaction mixture: mm GP (black trace), 1 mm GP (red trace), or without GP (gray trace). he initial mixture contained Sec13/31p and 5 nm Sec12Cp. (E) Sec12Cp binding to liposomes. Major minor or PC/PE liposomes (4 mgml 1 ) were incubated with Sec12Cp (5 nm) and separated by flotation centrifugation through sucrose step gradients (1.,.75, and M sucrose in HKM buffer). Proteins recovered in fractions (input (1. M sucrose), 1. or.75 M sucrose, and float fractions (without sucrose)) were analyzed by SS PAGE. (F) Light scattering of a suspension of PC/PE liposomes was monitored upon addition of Sar1p-GP, GP, and. he initial mixture contained Sec13/31p and 5 nm Sec12Cp (red trace) or HKM buffer alone (black trace). 4 (1 6 s 1 M 1 ), whereas the spontaneous exchange rate was (1 3 s 1 ). he activation rate saturated at 3 nm Sec12Cp (data not shown). Sec12Cp is remarkably active as an exchange factor, comparable to the activities of the ARNO-Sec7 domain on [17]ARF GPase (k ex ¼ (1 5 s 1 M 1 )) (Beraud-ufour et al, 1998) and RCC1 on Ran GPase (k ex ¼ 2.2 (1 6 s 1 M 1 )) (Klebe et al, 1995). he catalytic rate of Sec23GAP, measured in the presence of the full coat ensemble, was (1 5 s 1 M 1 ) (Figure 2B). hus the catalytic rate of Sec12GEF is 1-fold higher than that of Sec23GAP. A 1:1 relative rate was in good agreement with light scattering experiments measuring coat stability in a range of Sec12Cp concentrations. At reduced levels of Sec12Cp, coat dissociation was biphasic (Figure 2C). his result suggested that at limiting Sec12Cp, coat disassembly progressed rapidly to a new equilibrium characterized by slow dissociation linked to depletion of the GP pool. Quantification of COPII retained on membranes at the end of the first phase (t ¼ 4 s) indicated that B5% of maximum binding was obtained at B11.17 nm Sec12Cp in the presence of 16 nm and saturating Sec13/31p (Figure 2). his stoichiometry is roughly consistent with the measured catalytic rates of Sec12GEF and Sec23GAP. Morphology of COPII vesicle formed in the presence of Sec12Cp and GP o visualize coat assembly and vesicle formation in the presence of Sec12Cp, we examined the morphology of liposomes by thin-section electron microscopy (EM). When the liposomes were incubated with GP and COPII proteins, we observed virtually no coats on membranes (Figure 3A). In 4148 he EMBO Journal VOL 23 NO & 24 European Molecular Biology Organization
4 the liposome mixture with Sec12Cp, GP, and COPII proteins, numerous coated surfaces and coated buds but few coated vesicles were observed (Figure 3B). Incubations containing GMP-PNP with and without Sec12Cp produced broadly coated surfaces, coated buds, and a size range of coated vesicles. he ratio of buds/completed vesicles in three separate experiments was quantified by thin-section EM (able I), confirming the preponderance of COPII buds in incubation with Sec12Cp and GP. We conclude that Sec12Cp promotes coat stability and coated bud formation in incubations containing GP, but it appears that such reactions do not progress efficiently through coated vesicle fission. N4 as a potential catalytic site We performed a structure function analysis to test the notion that the nucleotide exchange activity of Sec12Cp is responsible for COPII assembly and stability. he cytosolic region of A B C Light scattering (normalized) kactivation (1 3 S 1 ) ime (s) Sec12ΔCp (nm) (nm) (+Sec13/31p and Sec12ΔCp at t =) Sec12ΔCp 2 nm nm (16 nm) 5 nm Fluorescence (AU) Sar1pG (+Sec13/31p and Sec12ΔCp or EA at t =) +EA major minor liposome Sar1pGP GP GP 2 COPII binding (%) Nucleotide exchange 5 kactivation (1 3 S 1 ) ime (s) GP hydrolysis 8 Without Sec12ΔCp 6 2 Sec12ΔCp (nm) nm 15 nm 1 nm 5 nm 1 nm Sec12p is predicted to form a b-propeller structure with seven W repeats (Chardin and Callebaut, 22). he crystal structure of another GEF with W repeats, RCC1, was solved in complex with the Ran GPase (Renault et al, 21). In the case of RCC1, loop regions between b-strands interact with Ran. In particular, the asparagine-rich b-wedge region is in contact with the P-loop of Ran and is thought to facilitate nucleotide removal. In the predicted seven-bladed b-propeller structure of Sec12p, four residues (P13, N4, H38, and 313) on loops of one face are completely conserved among species (Chardin and Callebaut, 22). he original temperaturesensitive mutant sec12-1/4 contains a P73L mutation (d Enfert et al, 1991). P73 is also in a loop region on the same face, but it is not well conserved. Mutational analysis of these residues was performed and we found that the N4A mutant had the most severe defect in exchange activity, B2% of wild type (Figure 4A). Other mutants also showed substantial defects, with the exception of H38A, which reproducibly stimulated nucleotide exchange approximately 1% above wild type but had a marginally lower COPII coat stabilizing activity. P73L showed less activity (13%) at 371C than at 31C (39.3%), consistent with the temperature-sensitive character of sec12-1 mutant cells. hese results support a conclusion that the nucleotide exchange activity of Sec12 is directly responsible for stabilizing COPII coat assembly. We analyzed the interaction of Sar1p with wild-type and mutant Sec12Cp in the presence and absence of nucleotide. For this purpose, GS-Sec12Cp hybrid proteins were employed and the recruitment of Sar1p was evaluated using GSH beads. Wild-type Sec12Cp bound best to nucleotide-free Sar1p, with background binding (compared to GS alone) in the presence of nucleotide (GMP-PNP or GP) (Figure 4B). Sec12Cp mutants with exchange defects showed reduced binding to Sar1p, and the binding correlated well with the defective exchange activity. Control experiments showed that Figure 2 Exchange activity of Sec12Cp on Sar1p is B1-fold higher than GAP activity of complex with saturating Sec13/31p. (A) he GEF activity of Sec12Cp and the GAP activity of were measured by tryptophan fluorescence of Sar1p. A large tryptophan fluorescence change accompanies a conformational change by Sar1p from the GP-bound to the GP-bound state. Sar1p-GP (2 mm) was added to major minor liposomes (3 mgml 1 ) and activated by the addition of GP. After 6 min, (55 nm) was added to activate GP hydrolysis. he initial mixture contained 5 nm Sec12Cp (red trace), 2 mm EA (blue traces) in HKM buffer, or buffer alone (black trace) and 9 nm Sec13/31p, which stimulates GAP activity at saturation. EA decreases the concentration of free Mg 2 þ from 1 mm to 1 mm, and accelerates GP GP exchange by Sar1p. (B) he rate constants for Sar1p activation were plotted against Sec12Cp concentration (left) to determine the nucleotide exchange activity (k activation ¼ k spontaspontaneousþ kexchange [Sec12Cp]). he specific nucleotide exchange activity (k exchange /[Sec12Cp]) corresponds to the rate constant of the activation of Sar1p normalized to the concentration of Sec12Cp (see Materials and methods). he rate constant for Sar1p inactivation by with saturating Sec13/31p was plotted against concentration (right) to determine catalytic activity (k inactivation ¼ k catalyze []). (C) Light scattering assays with the indicated concentration of Sec12Cp ( 2 nm). he experimental conditions were as in Figure 1. () In the experiments (C), light scattering signal decays instantly after addition. he intensity of signals after instant decay was measured at 4 s. Percentage of the maximal amplitude was calculated, and plotted against Sec12Cp concentration. & 24 European Molecular Biology Organization he EMBO Journal VOL 23 NO
5 A B C Figure 3 Morphology of major minor liposomes incubated with COPII proteins and Sec12Cp. (A ) he reaction contains major minor liposomes ( mgml 1 ), 95 nm Sar1p, 16 nm, and 26 nm Sec13/31p. he reaction was initiated by the addition of 3 mm GP and incubated at 271C. After 15 min, the samples were processed for EM. On thin-section electron micrographs, characteristic profiles observed without (A) or with (B) 5 nm Sec12Cp are shown. he inset in (B) depicts liposome with multiple coated buds. Comparison of COPII-like vesicles formed with GMP-PNP in the absence (C) or presence () of Sec12Cp is shown. Scale bars, 2 nm. he arrows point to coated buds (B, C). the mutant proteins were not defective in binding to the major minor mix liposomes used in the nucleotide exchange assays (not shown). Exchange factors operate by binding to the nucleotide-free form of GP-binding proteins. hus the mutants are substrate binding and catalytic mutants. N4, P73, 313, and P13 are important for the interaction of Sec12p with Sar1p. Light scattering assays were performed to evaluate the influence of Sec12Cp mutations on coat assembly and stabilization. he N4A mutant was inert and other mutants reduced coat stability in relation to a defect in Sar1p interaction (Figure 4C). hese results suggest that one face of Sec12Cp contains a binding site for Sar1p, and the asparagine-rich loop of the first blade, containing N4, is critically important, similar to the b-wedge region in RCC1. COPII vesicle formation from Sec12p proteoliposomes In order to examine the incorporation of Sec12p into COPII vesicles, we employed a COPII vesicle budding assay (Matsuoka et al, 1998b) using proteoliposomes. Although Sec12p is not enriched in COPII vesicles formed from ER membranes (Barlowe et al, 1994), it was important to document this property with synthetic membranes. We reconstituted proteoliposomes with Sec12Mp (1 373), which 415 he EMBO Journal VOL 23 NO & 24 European Molecular Biology Organization
6 contains the transmembrane domain, with the SNARE proteins, Bet1p, Bos1p, and Ufe1p, and with exas red-phosphatidylethanolamine (exas red-pe) to permit quantification of phospholipid distribution. he proteoliposomes were reconstituted by a detergent removal method (Parlati et al, 2), which produced vesicles with an average diameter of nm as determined by dynamic light scattering measurements. Carbonate extraction of the proteoliposomes able I Morphogenesis of COPII coat maintained with Sec12C Experiment a /incubation Number of buds+vesicles Ratio of buds/vesicles 1 GMP-PNP+sec GP+sec GMP-PNP+sec GP+sec GMP-PNP sec GMP-PNP+sec GP+sec GMP-PNP sec a In all, mm 2 of pellet sections was evaluated in each sample. Pictures (15 for each sample) were printed at a final magnification of 76. showed that reconstituted proteins remained membrane bound (data not shown), suggesting that the proteins were incorporated into the lipid bilayler. We probed the orientation of two proteins integrated into the proteoliposomes: Bet1p and Sec12p. An N-terminal GS- Bet1p fusion protein, membrane integrated through the C- terminal membrane anchor domain of Bet1p, was accessible to thrombin, which cleaved 7% of the GS at a thrombin linker sequence to Bet1p. Reconstituted Sec12M liposomes were assayed in Sar1p nucleotide exchange reactions to measure enzyme latency. Comparing the specific activity of Sec12M liposomes to soluble Sec12Cp showed that 62% of the Sec12 active site was accessible to Sar1p. Furthermore, incubations containing liposomes with 5 nm Sec12Mp and GP produced COPII light scattering signal stability comparable to that promoted by a similar level of Sec12Cp (Supplementary Figure S1). Reaction mixtures with COPII protein, proteoliposomes, and nucleotide were subjected to centrifugation through sucrose gradients (Figure 5A), which separate partially coated liposomes and coated COPII vesicles (Matsuoka et al, 1998b). For nucleotide, we compared GMP-PNP and GP because reactions driven by GP failed to retain membranebound coat proteins during sedimentation to equilibrium on a sucrose density gradient. he separated fractions were A Sec12ΔC 27 C Wild type Exchange activity, k exchange /[Sec12ΔCp] (1 6 s 1 M 1 ) 1.46±.15 Relative activity (%) B P73L.576± P13A 1.8± N4A.27± H38A 1.79± A.745± C Wild type 3.59±.12 P73L.466± C Light scattering (normalized) (+Sec13/31p and Sec12ΔCp at t =) Sar1pGP GP Sec12ΔCp Wild type H38A P13A 313A P73L Without Sec12ΔCp N4A ime (s) Figure 4 Exchange activity of Sec12Cp mutants on Sar1p. (A) For each mutant, experiments similar to that shown in Figure 2A were performed to determine value 7s.e. of the specific exchange activity (k exchange /[Sec12Cp]). Except for [N4A] Sec12Cp, the mutants were assayed at a concentration up to 2 nm. A very weak stimulation of Sar1p activation as detected by varying concentrations of [N4A] Sec12Cp up to 6 nm. (B) Sar1p binding to GSSec12Cp wild type or mutants. GSSec12Cp (3 mm) was incubated with Sar1p-GP (3 mm) and GMP- PNP (3 mm) (left), GP (3 mm) (middle), or EA (2 mm) with alkaline phosphatase (calf intestine, Promega) (right) in HKM buffer containing.2% octylglucoside. he incubation with EA and alkaline phosphatase renders Sar1p nucleotide free. GSSec12Cp was recovered with glutathione agarose and washed extensively with HKM buffer (.2% octylglucoside) containing GMP-PNP (left), GP (middle), or EA (right). Sar1p bound to the beads was analyzed by SS PAGE and quantified by Sypro-Ruby staining. (C) Light scattering assays with Sec12Cp wild type or mutants. he experimental conditions were as in Figure 1. Sec12Cp (5 nm) wild-type or mutant proteins were added to the initial mixture. & 24 European Molecular Biology Organization he EMBO Journal VOL 23 NO
7 A Coated liposomes Vesicles the proteoliposomes. However, Sec12Mp and Ufe1p, ERresident proteins, and phospholipids were neither enriched nor excluded in vesicle fractions (Figure 5B and C). hese results suggest that the proteoliposome reaction reconstitutes a faithful protein sorting reaction and that the integral membrane-bound form of Sec12p is not enriched in COPIIcoated vesicles during the synthetic budding reaction. iscussion B α-bet1p α-bos1p α-sec12p C α-gs (Ufe1p) GMP-PNP GP GMP-PNP GP GMP-PNP GP GMP-PNP GP Fr 1 9 Fr GMP-PNP Fractions Lipid +GP Proteoliposome +COPII Major minor +COPII Figure 5 Sorting of Sec12Mp and cargo proteins into COPII vesicles. (A) Proteoliposomes ( mgml 1 ) with Sec12Mp, Bet1p, Bos1p, and GS-Ufe1p were incubated for 3 min at 271C with COPII proteins (13 mgml 1, 15 mgml 1 sec13/31p, and 8 mg ml 1 Sar1p) and nucleotide (GMP-PNP or GP). COPII vesicles were separated from donor proteoliposomes by sucrose density gradient sedimentation (2.2.2 M sucrose). After separation, lipid recovery was monitored by fluorescence of exas red-pe. (B) Bet1p, Bos1p, Sec12Mp, and GS-Ufe1p in each fraction were detected by immunoblot. (C) From the blot in (B), recovery of Bet1p and Sec12Mp was quantified using 35 S-labeled anti-igg and a phosphorimager. he recovery in the fractions for liposomes (Fr 1 9) or COPII vesicles (Fr 1 15) was calculated. Bos1 and Bet1 were enriched in the COPII vesicles, but Sec12M and Ufe1 were neither enriched nor excluded in the vesicle fractions. evaluated by EM to confirm the recovery of partially coated liposomes or COPII vesicles (data not shown). Qualitative and quantitative protein detection by dot blot analysis showed that Bet1p and Bos1p were enriched in the COPII vesicle fraction (B7% recovery) (Figure 5B and C), indicating that the selection of cargo proteins was reproduced with Lipid SEC12 was identified as an essential gene required for protein transport from the ER to the Golgi apparatus in yeast (Nakano et al, 1988). Sec12p is an ER-resident transmembrane protein (Sato et al, 1996) and its cytosolic domain catalyzes guanine nucleotide exchange (GEF) specifically on Sar1p (Barlowe and Schekman, 1993). Recently, a mammalian Sec12 homolog (PREB) was also identified and shown to have GEF activity on Sar1 and was required for COPII vesicle formation in vitro (Weissman et al, 21). Sec12p is thought to initiate coat polymerization and to ensure that coat proteins are recruited specifically to the ER. his hypothesis extends to the localization of coating reactions initiated by ARF, which employs Sec7 domain GEF proteins that are peripherally associated with specific membranes through interaction with lipids or protein (Jackson et al, 2; Chantalat et al, 24). Here, using liposome reconstitution, we directly show that the high nucleotide exchange activity of Sec12p is required to keep Sar1p activated at membranes engaged in COPII vesicle budding. In an effort to probe the details of cargo sorting and vesicle budding, we developed a reconstituted liposome reaction that reproduces some but not all aspects of COPII vesicle formation (Matsuoka et al, 1998b). One obvious limitation of our reaction is the rapid rate of coat disassembly observed when the coat polymerizes in the presence of GP (Antonny et al, 21). Although coats and coated vesicles form in the presence of GMP-PNP, no evidence of vesicles is seen in incubations with GP (Matsuoka et al, 1998b). In an effort to mitigate the effect of rapid GP hydrolysis mediated by coat assembly, we explored the effect of membrane cargo proteins and Sec16p, a peripheral membrane protein that organizes COPII vesicle budding. SNAREs and other cargo molecules do not dampen the rate of GP hydrolysis (unpublished observations). Although Sec16p and Sar1p-GP stabilizes the recruitment of to liposomes, no diminished rate of GP hydrolysis or stable recruitment of the full coat is detected (Supek et al, 22). In contrast, addition of a soluble cytoplasmic domain fragment of Sec12 (Sec12Cp) or proteoliposome reconstitution of an integral membrane form of Sec12 (Sec12Mp) promoted the stable formation of coated membranes and coated buds. We used light scattering and tryptophan fluorescence assays to document the catalysis of nucleotide exchange and its effect on the recruitment of Sar1p to liposome membranes. Light scattering and thin-section EM revealed a stable coat and coated buds, most likely due to a continuous recharging of Sar1p in the area of a nascent vesicle. he exchange activity of Sec12p is much higher than we previously estimated and instead is comparable to the activity reported for other GEFs (77.6 (1 6 s 1 M 1 )). Our previous assays were conducted in the presence of.1% riton X-, whereas here we used liposomes. Under these 4152 he EMBO Journal VOL 23 NO & 24 European Molecular Biology Organization
8 COPII vesicle budding with Sec12p GEF conditions, the specific GEF activity was approximately 1- fold greater than the Sar1p GPase activity stimulated by Sec23 GAP and the rest of the COPII coat. A ratio of 1:15 of Sec12 to COPII components produced half-maximal coat stabilization in an incubation conducted in the presence of GP. As a consequence of the greatly enhanced cycle of GP hydrolysis stimulated by these conditions, coat stability is limited at lower concentrations of nucleotide (e.g. 1 mm). In previous work, we showed that coated buds and coated vesicles are observed in thin sections of liposome budding reactions conducted in the presence of GMP-PNP. We now find that similar profiles of coated membranes and coated buds are visible in preparations incubated with Sec12Cp and GP. Interestingly, few clear profiles of separate coated vesicles are observed (Figure 3B). It may be that these conditions are insufficient to reproduce the fission reaction that completes vesicle budding. Unfortunately, we were unable to preserve and fractionate coated membranes in these preparations and thus we have no means of quantifying the fraction of membrane included in separate coated vesicles. Resolution of this issue awaits the development of a real-time vesicle budding reaction suitable for assays of liposome and proteoliposome populations. Sec12p is largely excluded from COPII vesicles formed from microsomes incubated with GP or GMP-PNP (Barlowe et al, 1994). However, some low level, perhaps representing the concentration prevailing in the ER membrane, is packaged because Sec12p is known to cycle between the ER and Golgi and requires a membrane protein, Rer1p, to facilitate recycling (Sato et al, 1996). Similarly, Sec12Mp and Ufe1p, an ER membrane t-snare, were neither enriched nor depleted in a proteoliposome budding reaction. In contrast, membrane cargo proteins, such as the SNAREs Bet1p and Bos1p, are enriched in COPII vesicles by virtue of direct interactions with binding sites on the membrane proximal surface of Sec24p (Miller et al, 23). We reproduced this sorting event with GS hybrid proteins linked to the surface of a liposome through glutathione-derivatized PE (Matsuoka et al, 1998a), and now using integral membrane forms of these proteins (Figure 5). he enrichment of cargo proteins and of clustered COPII subunits on the surface of a bud results in a diminished ratio of Sec12GEF/Sec23GAP. As a result, GP hydrolysis would tend to destabilize the coat except at the growing boundary between a bud and the surrounding ER membrane where the ratio of GEF/GAP is higher than elsewhere (Figure 6A and B). Coat subunits remain transiently bound to completed COPII vesicles even after GP hydrolysis has rendered the vesicles devoid of Sar1p (Barlowe et al, 1994). his simple consideration may explain how a coat remains relatively intact until the moment of fission separates a budded vesicle (Figure 6B and C). Other factors may facilitate the exclusion of Sec12p from budded vesicles. In Pichia pastoris and in mammalian cells, Sec12p is organized in a transitional ER membrane segregated from the bulk ER (Rossanese et al, 1999; Ellgaard and Helenius, 23). Although this segregation is not apparent in Saccharomyces cerevisiae, small domains of Sec12p may be 1 COPII coat assembly promoted by Sec12pGEF GP Coat disassembly Sar1p GP Sec12p Sec13/31p Coat assembly GP GP GP P i P i P i GP GP GP GP Cargo proteins 2 Vesicle budding (1Sec12p>15) 3 isassembly of the coats after budding (1Sec12p<15) Figure 6 Coordination of COPII coating and uncoating by localized action of Sec12 GEF. & 24 European Molecular Biology Organization he EMBO Journal VOL 23 NO
9 organized by the recruitment of Sar1p and Sec16p to the ER at the onset of a budding event (Supek et al, 22; Bonifacino and Glick, 24). We probed the GEF activity of Sec12p using a structural model based on the interaction of the RCC1 GEF and its cognate GPase, Ran (Renault et al, 21). Sec12Cp binds well to nucleotide-free Sar1p and less well to Sar1p-GP or - GP just as has been shown for binding of RCC1 to Ran (Klebe et al, 1995) and ARNO to 17ARF (Beraud-ufour et al, 1998) (Figure 4). We mutated residues conserved in a variety of Sec12s (P13, N4, and 313, H38A), which are located to putative loop regions between blades of the proposed Sec12 b-propeller structure (Chardin and Callebaut, 22). Several of these, as well as a residue identified in the initial selection for temperature-sensitive sec mutants (P73L), reduced or eliminated Sar1p binding, GEF activity, and stable COPII polymerization on liposomes (Figure 5A). he most dramatic effect was produced by the N4A mutation, which is predicted to reside in a b-wedge region similar to RCC1, a position known to be associated with catalysis. A structure of Sec12p-Sar1p would allow this prediction to be tested. We conclude that the nucleotide exchange activity of Sec12p, rather than some other structural feature of the protein, is actively involved in building the COPII coat, most likely by stabilizing the growing boundary of the coat polymer. Materials and methods Proteins and liposomes Sar1p, Sec23/24 (His 6 )p, and Sec13/31 (His 6 )p were prepared as described (Barlowe et al, 1994; Salama et al, 1997). Sar1p was purified in the presence of 5 mm GP. Sec12C(1 354)p, Sec12M(1 373)p, Bet1p, Bos1p, and Ufe1p were produced as N- terminal GS fusion proteins in Escherichia coli from the vectors pgex-2 (for GS-Sec12C, GS-Sec12M, GS-Bos1, GS-Ufe1) (Pharmacia) and pegex (for GS-Bet1) (Sharrocks, 1994) and purified as described (Parlati et al, 2). GS tags were removed by thrombin cleavage for Sec12Cp, Sec12Mp, Bet1p, and Bos1p. Ufe1p was purified as a GS fusion (GS-Ufe1p). he composition of phospholipids in the major minor or PC/PE mix liposomes is essentially the same as previously described (Matsuoka et al, 1998b) with some changes: we incorporated 1% (mol%) exas red-pe instead of NB-phospholipids and 1% (w/ w) cholesterol instead of ergosterol. exas red-pe was used to quantify lipid recovery. Liposomes were prepared as described (Antonny et al, 21). Light scattering measurement and tryptophan fluorescence he light scattering and tryptophan fluorescence assays were performed as described previously (Antonny et al, 21). For better temporal resolution, all experiments were performed in a cylindrical cuvette (sample volume 55 ml) in which the reaction mixtures were mixed by a magnetic stir bar, and injection of reactants (COPII proteins and nucleotides) was performed through a Hamilton syringe. With this device, the recording was not interrupted by the injections and the mixing time was o2 s, allowing kinetic measurement in the range of few seconds. Kinetic measurements Activation of Sar1p upon GP/GP exchange was measured by tryptophan fluorescence. he apparent rate constant of Sar1p activation (k act ) was determined by fitting the fluorescence change with a single exponential and plotted as a function of [Sec12Cp]. k act increases linearly with the concentration of Sec12Cp according to the equation k act ¼ a þ b [Sec12Cp], where a is the rate constant of spontaneous nucleotide exchange, which was measured in the absence of Sec12Cp, and b is the specific exchange activity of Sec12Cp, that is, the rate constant of catalyzed GP/GP exchange normalized to the concentration of Sec12Cp (k exchange / [Sec12Cp]). Inactivation of Sar1p upon GAP-catalyzed GP hydrolysis was also measured by tryptophan fluorescence. he apparent rate constant for Sar1p inactivation was difficult to determine by fitting to single exponential and was determined by the value t inact (k inact ¼ 1/t inact ), which is obtained graphically, and plotted as a function of []. Assuming k inact is in the absence of, k inact increases linearly with the concentration of according to the equation k inact ¼ c [], where c is the specific catalytic activity of, that is, the rate constant of catalyzed GP hydrolysis normalized to the concentration of saturated with Sec13/31p (k catalyze / []). hese parameters (k exchange, k catalyze ) correspond to the apparent second-order rate constant k cat /K m of a Michaelis Menten mechanism. Note that we did not determine the individual k cat and K m values. Liposome binding and budding assays Recruitment of Sec12Cp proteins to synthetic liposomes was performed as described previously (Matsuoka et al, 1998b). Sec12Cp (5 nm) was incubated for 3 min at 271C with liposomes (4 mgml 1 ) in HKM buffer (17 ml) (2 mm Hepes-K, ph 6.8, 16 mm KOAc, 1 mm MgCl 2 ) and mixed with M sucrose (11 ml) in HKM to make a 1. M sucrose solution. he mixture was aliquoted into mm polycarbonate centrifuge tubes (Beckman) and overlaid with.75 M sucrose in HKM (2 ml) and HKM (5 ml). he samples were centrifuged in a LS55 rotor in an Optima LX table top ultracentrifuge (Beckman) at 55 r.p.m. for 2 h at 41C. After centrifugation, fractions floated to the.75 and 1. M sucrose interfaces were collected and proteins were analyzed by SS PAGE. Budding of COPII vesicles from synthetic liposomes was performed as described previously (Matsuoka et al, 1998b). Protein reconstitution into liposomes Proteoliposomes were prepared by detergent depletion as described previously (Parlati et al, 2) with modifications. A lipid film was dissolved with buffer A (25 mm ris HCl, ph 8., 4 mm KCl, 1% glycerol, 1% octyl-b--glucopyranoside (octylglucoside), 2 mm 2- mercaptoethanol) and mixed with proteins to make a solution of 1 mm phospholipids and.33 mm proteins. he detergent lipid protein mixture was incubated for 15 min at room temperature. Octylglucoside was removed by extensive dialysis to buffer B (25 mm Hepes-K, ph 7.4, 4 mm KCl, 1% glycerol, 1 mm ) at 41C for 3 h with four buffer changes, using 1 MWCO dialysis cassettes (Pierce). Each B5 ml dialysate was mixed with 33 ml of M sucrose in HK (2 mm Hepes-K, ph 6.8, 25 mm KOAc) and aliquoted into mm polycarbonate centrifuge tubes (Beckman). Each was overlaid with 66 ml.75 M sucrose in HK and 2 ml HK. he samples were centrifuged in an SW55 rotor in the Optima table top ultracentrifuge (Beckman) at 55 r.p.m. for 4 h at 41C. Proteoliposomes were harvested from the top fraction and extruded through a 4 nm polycarbonate filter to remove lipid aggregates. he lipid concentration was determined by the fluorescence of exas red. Electron microscopy Liposomes were made from a mixture of phospholipids and cholesterol by extrusion through 4 nm polycarbonate filters. he experimental conditions were the same as those used for the light scattering experiments. After incubations, samples were processed for conventional EM, as described (Matsuoka et al, 1998b). Supplementary data Supplementary data are available at he EMBO Journal Online. Acknowledgements We thank Crystal Chan and Robert Lesch for COPII proteins and Matthew Welsh and avid G rubin for sharing their equipment. We thank Elizabeth Miller and Marcus Lee for improving the manuscript and Bruno Antonny for kinetic calculations of realtime assays. We thank members of the Schekman lab for discussions and encouragement. his work was supported by the HHMI (RS), the Swiss National Science Foundation (LO), and postdoctoral research fellowships from JSPS and ACS (EF) he EMBO Journal VOL 23 NO & 24 European Molecular Biology Organization
10 References Antonny B, Gounon P, Schekman R, Orci L (23) Self-assembly of minimal COPII cages. EMBO Reports 4: Antonny B, Madden, Hamamoto S, Orci L, Schekman R (21) ynamics of the COPII coat with GP and stable analogues. Nat Cell Biol 3: Barlowe C, Orci L, Yeung, Hosobuchi M, Hamamoto S, Salama N, Rexach MF, Ravazzola M, Amherdt M, Schekman R (1994) COPII: a membrane coat formed by Sec proteins that drive vesicle budding from the endoplasmic reticulum. Cell 77: Barlowe C, Schekman R (1993) SEC12 encodes a guanine nucleotide exchange factor essential for transport vesicle budding from ER. Nature 365: Beraud-ufour S, Robineau S, Chardin P, Paris S, Chabre M, Cherfils J, Antonny B (1998) A glutamic finger in the guanine nucleotide exchange factor ARNO displaces Mg 2+ and the beta-phosphate to destabilize GP on ARF1. EMBO J 17: Bonifacino JS, Glick BS (24) he mechanism of vesicle budding and fusion. Cell 116: Chantalat S, Park SK, Hua Z, Liu K, Gobin R, Peyroche A, Rambourg A, Graham R, Jackson CL (24) hearf activator Gea2p and the P-type APase rs2p interact at the Golgi in Saccharomyces cerevisiae. J Cell Sci 117: Chardin P, Callebaut I (22) he yeast Sar1 exchange factor Sec12, and its higher organism orthologs, fold as b-propellers. FEBS Lett 525: Cosson P, Letourneur F (1994) Coatomer interaction with di-lysine endoplasmic reticulum retention motifs. Science 263: d Enfert C, Wuestehube LJ, Lila, Schekman R (1991) Sec12pdependent membrane binding of the small GP-binding protein Sar1p promotes formation of transport vesicles from the ER. J Cell Biol 114: Ellgaard L, Helenius A (23) Quality control in the endoplasmic reticulum. Nat Rev Mol Cell Biol 4: Espenshade P, Gimeno RE, Holzmacher E, eung P, Kaiser CA (1995) Yeast Sec16 gene encodes a multidomain vesicle coat protein that interact with Sec23p. J Cell Biol 131: Gimeno RE, Espenshade P, Kaiser CA (1996) COPII coat subunit interactions: Sec24p and Sec23p bind to adjacent regions of Sec16p. Mol Biol Cell 7: Goldberg J (2) ecoding of sorting signals by coatomer through a GPase switch in the COPI coat complex. Cell : Hoflack B (1998) Mechanism of protein sorting and coat assembly; clathrin coated vesicle pathways. Curr Opin Cell Biol 1: Jackson R, Kearns BG, heibert AB (2) Cytohesins and centaurins: mediators of PI 3-kinase regulated Arf signaling. rends Biochem Sci 25: Klebe C, Bischoff FR, Ponstingl H, Wittinghofer A (1995) Interaction of the nuclear GP-binding protein Ran with its regulatory proteins RCC1 and RanGAP1. Biochemistry 34: Matsuoka K, Morimitsu Y, Uchida K, Schekman R (1998a) Coat assembly directs v-snare concentration into synthetic COPII vesicles. Mol Cell 2: Matsuoka K, Orci L, Amherdt M, Bednarek SY, Hamamoto S, Schekman R, Yeung (1998b) COPII-coated vesicle formation reconstituted with purified coat proteins and chemically defined liposomes. Cell 93: Matsuoka K, Schekman R, Orci L, Heuser JE (21) Surface structure of the COPII-coated vesicle. Proc Natl Acad Sci USA 98: Miller EA, Beilharz H, Malkus PN, Lee MC, Hamamoto S, Orci L, Schekman R (23) Multiple cargo binding sites on the COPII subunit Sec24p ensure capture of diverse membrane proteins into transport vesicles. Cell 114: Mossessova E, Bickford LC, Goldberg J (23) SNARE selectivity of COPII coat. Cell 114: Nakano A, Brada, Schekman R (1988) A membrane glycoprotein, Sec12p, required for protein transport from the endoplasmic reticulum to the Golgi apparatus in yeast. J Cell Biol 17: Parlati F, McNew JA, Fukuda R, Miller R, Sollner H, Rothman JE (2) opological restriction of SNARE dependent membrane fusion. Nature 47: Peter BJ, Kent HM, Mills IG, Vallis Y, Butler PJ, Evans PR, McMahon H (24) BAR domains as sensors of membrane curvature: the amphiphysin BAR structure. Science 33: Renault L, Kuhlmann J, Henkel A, Wittinghofer A (21) Structural basis for guanine nucleotide exchange on Ran by the regulator of chromosome condensation (RCC1). Cell 15: Rossanese OW, Soderholm J, Bevis BJ, Sears IB, O Conner J, Williamson EK, Glick BS (1999) Golgi structure correlates with traditional endoplasmic reticulum organization in Pichia pastoris and Saccharomyces cerevisiae. J Cell Biol 145: Salama NR, Chuang JS, Schekman RW (1997) Sec31 encodes an essential component of the COPII coat required for transport vesicle budding from the endoplasmic reticulum. Mol Biol Cell 8: Sato M, Sato K, Nakano A (1996) Endoplasmic reticulum localization of Sec12p is activated by two mechanisms: Rer1p-depenedent retrieval that requires the transmembrane domain and Rer1p-independent retrieval that involves the cytoplasmic domain. J Cell Biol 134: Sharrocks A (1994) A 7 expression vector for providing N- and C- terminal fusion proteins with glutathione S-transferase. Gene 138: Shaywitz A, Espenshade PJ, Gimeno RE, Kaiser CA (1997) COPII subunit interactions in the assembly of the vesicle coat. J Biol Chem 272: Spang A, Matsuoka K, Hamamoto S, Schekman R, Orci L (1998) Coatomer, Arf1p, and nucleotide are required to bud coat protein complex I-coated vesicles from large synthetic liposomes. Proc Natl Acad Sci USA 95: Springer S, Schekman R (1998) Nucleation of COPII vesicular coat complex by endoplasmic reticulum to Golgi vesicle SNAREs. Science 281: Supek F, Madden, Hamamoto S, Orci L, Schekman R (22) Sec16p potentiate the action of COPII proteins to bud transport vesicles. J Cell Biol 158: Vetter IR, Wittinghofer A (21) he guanine nucleotide-binding switch in three dimensions. Science 294: Weissman J, Plutner H, Balch WE (21) he mammalian guanine nucleotide exchange factor msec12 is essential for activation of the Sar1 GPase directing endoplasmic reticulum export. raffic 2: Yoshihisa, Barlowe C, Schekman R (1993) Requirement for a GPase-activating protein in vesicle budding from the endoplasmic reticulum. Science 259: & 24 European Molecular Biology Organization he EMBO Journal VOL 23 NO
Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Coat Assembly Directs v-snare Concentration into Synthetic COPII Vesicles
 Molecular Cell, Vol. 2, 703 708, November, 1998, Copyright 1998 by Cell Press Coat Assembly Directs v-snare Concentration into Synthetic COPII Vesicles Ken Matsuoka,* Yasujiro Morimitsu, Koji Uchida, and
Molecular Cell, Vol. 2, 703 708, November, 1998, Copyright 1998 by Cell Press Coat Assembly Directs v-snare Concentration into Synthetic COPII Vesicles Ken Matsuoka,* Yasujiro Morimitsu, Koji Uchida, and
Mechanism of Vesicular Transport
 Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
Mechanism of Vesicular Transport Transport vesicles play a central role in the traffic of molecules between different membrane-enclosed enclosed compartments. The selectivity of such transport is therefore
Section 6. Junaid Malek, M.D.
 Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Supplementary Information
 Gureaso et al., Supplementary Information Supplementary Information Membrane-dependent Signal Integration by the Ras ctivator Son of Sevenless Jodi Gureaso, William J. Galush, Sean Boyevisch, Holger Sondermann,
Gureaso et al., Supplementary Information Supplementary Information Membrane-dependent Signal Integration by the Ras ctivator Son of Sevenless Jodi Gureaso, William J. Galush, Sean Boyevisch, Holger Sondermann,
Supplementary Materials. High affinity binding of phosphatidylinositol-4-phosphate. by Legionella pneumophila DrrA
 Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Vesicle Transport. Vesicle pathway: many compartments, interconnected by trafficking routes 3/17/14
 Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
Vesicle Transport Vesicle Formation Curvature (Self Assembly of Coat complex) Sorting (Sorting Complex formation) Regulation (Sar1/Arf1 GTPases) Fission () Membrane Fusion SNARE combinations Tethers Regulation
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Practice Exam 2 MCBII
 1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
In the previous chapter we explored how proteins are targeted
 17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
17 VESICULAR TRAFFIC, SECRETION, AND ENDOCYTOSIS Electron micrograph of clathrin cages, like those that surround clathrin-coated transport vesicles, formed by the in vitro polymerization of clathrin heavy
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
Chapter 1: Vesicular traffic. Biochimica cellulare parte B 2017/18
 Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
Chapter 1: Vesicular traffic Biochimica cellulare parte B 2017/18 Major Protein-sorting pathways in eukaryotic cells Secretory and endocytic pathways Unifying principle governs all protein trafficking
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
Supplementary Figure 1. Overview of steps in the construction of photosynthetic protocellular systems
 Supplementary Figure 1 Overview of steps in the construction of photosynthetic protocellular systems (a) The small unilamellar vesicles were made with phospholipids. (b) Three types of small proteoliposomes
Supplementary Figure 1 Overview of steps in the construction of photosynthetic protocellular systems (a) The small unilamellar vesicles were made with phospholipids. (b) Three types of small proteoliposomes
Zool 3200: Cell Biology Exam 4 Part I 2/3/15
 Name: Key Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name: Key Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic
 Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Molecular Cell Biology - Problem Drill 17: Intracellular Vesicular Traffic Question No. 1 of 10 1. Which of the following statements about clathrin-coated vesicles is correct? Question #1 (A) There are
Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013
 Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
Transport from the endoplasmic reticulum to the Golgi Chris Kaiser* and Susan Ferro-Novick
 477 Transport from the endoplasmic reticulum to the Golgi Chris Kaiser* and Susan Ferro-Novick Two crucial aspects of transport vesicle function have recently been reconstituted using purified proteins
477 Transport from the endoplasmic reticulum to the Golgi Chris Kaiser* and Susan Ferro-Novick Two crucial aspects of transport vesicle function have recently been reconstituted using purified proteins
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 2/21/17 glycosylation is a non-template derived phenomenon - the presence of
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation
 Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
Localization and Retention of Glycosyltransferases And the Role of Vesicle Trafficking in Glycosylation Richard Steet, Ph.D. 3/8/2011 glycosylation is a non-template derived phenomenon - the presence of
04_polarity. The formation of synaptic vesicles
 Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Thursday, October 16 th
 Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
endomembrane system internal membranes origins transport of proteins chapter 15 endomembrane system
 endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
endo system chapter 15 internal s endo system functions as a coordinated unit divide cytoplasm into distinct compartments controls exocytosis and endocytosis movement of molecules which cannot pass through
Intracellular vesicular traffic. B. Balen
 Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
Intracellular vesicular traffic B. Balen Three types of transport in eukaryotic cells Figure 12-6 Molecular Biology of the Cell ( Garland Science 2008) Endoplasmic reticulum in all eucaryotic cells Endoplasmic
Intracellular Vesicular Traffic Chapter 13, Alberts et al.
 Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Intracellular Vesicular Traffic Chapter 13, Alberts et al. The endocytic and biosynthetic-secretory pathways The intracellular compartments of the eucaryotic ell involved in the biosynthetic-secretory
Overview of clathrin-mediated endocytosis
 Overview of clathrin-mediated endocytosis Accessory and adaptor proteins promote clathrin nucleation on the plasma membrane and some help deform membrane. Clathrin assembly into lattices stabilize the
Overview of clathrin-mediated endocytosis Accessory and adaptor proteins promote clathrin nucleation on the plasma membrane and some help deform membrane. Clathrin assembly into lattices stabilize the
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest.
 Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Chapter 7: Membranes
 Chapter 7: Membranes Roles of Biological Membranes The Lipid Bilayer and the Fluid Mosaic Model Transport and Transfer Across Cell Membranes Specialized contacts (junctions) between cells What are the
Chapter 7: Membranes Roles of Biological Membranes The Lipid Bilayer and the Fluid Mosaic Model Transport and Transfer Across Cell Membranes Specialized contacts (junctions) between cells What are the
Nature Biotechnology: doi: /nbt.3828
 Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
AP Biology
 Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Molecular Cell Biology 5068 In Class Exam 1 September 29, Please print your name:
 Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Fusion (%) = 100 (B-A)/(C-A)
 6 Fusion (%) = 1 (B-A)/(C-A) fluorescence, a.u. x 1 C 1 B A 6 1 A Supplementary Figure 1. Fusion of lipid vesicles studied with cobalt-calcein liquid content transfer assay. An example of fusion % calibration
6 Fusion (%) = 1 (B-A)/(C-A) fluorescence, a.u. x 1 C 1 B A 6 1 A Supplementary Figure 1. Fusion of lipid vesicles studied with cobalt-calcein liquid content transfer assay. An example of fusion % calibration
The Cell Organelles. Eukaryotic cell. The plasma membrane separates the cell from the environment. Plasma membrane: a cell s boundary
 Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Lecture 6 - Intracellular compartments and transport I
 01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
01.25.10 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
Molecular Trafficking
 SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
SCBM 251 Molecular Trafficking Assoc. Prof. Rutaiwan Tohtong Department of Biochemistry Faculty of Science rutaiwan.toh@mahidol.ac.th Lecture outline 1. What is molecular trafficking? Why is it important?
7.06 Spring of PROBLEM SET #6
 7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
Chapter 3. Expression of α5-megfp in Mouse Cortical Neurons. on the β subunit. Signal sequences in the M3-M4 loop of β nachrs bind protein factors to
 22 Chapter 3 Expression of α5-megfp in Mouse Cortical Neurons Subcellular localization of the neuronal nachr subtypes α4β2 and α4β4 depends on the β subunit. Signal sequences in the M3-M4 loop of β nachrs
22 Chapter 3 Expression of α5-megfp in Mouse Cortical Neurons Subcellular localization of the neuronal nachr subtypes α4β2 and α4β4 depends on the β subunit. Signal sequences in the M3-M4 loop of β nachrs
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Tala Saleh. Ahmad Attari. Mamoun Ahram
 23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson
 Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
CMB621: Cytoskeleton. Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved
 CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine
 Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Supplementary Information
 Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Instructions. Fuse-It-Color. Overview. Specifications
 Membrane fusion is a novel and highly superior method for incorporating various molecules and particles into mammalian cells. Cargo-specific liposomal carriers are able to attach and rapidly fuse with
Membrane fusion is a novel and highly superior method for incorporating various molecules and particles into mammalian cells. Cargo-specific liposomal carriers are able to attach and rapidly fuse with
Zool 3200: Cell Biology Exam 4 Part II 2/3/15
 Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f
![1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f 1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f](/thumbs/77/75734355.jpg) 1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. S k f k r P a) Calculate K eq (the equilibrium constant) and k r. b) A catalyst increases k f by a factor of 10
1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. S k f k r P a) Calculate K eq (the equilibrium constant) and k r. b) A catalyst increases k f by a factor of 10
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
Supplementary Material
 Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
Signal Transduction: G-Protein Coupled Receptors
 Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Organization of ATPases
 The Primary Active Transporter II: The ATPase Objectives: Organization P type with NPA domains Proton pumps of the rotary V type ATPase 1 Organization of P type, solute transport, found in plasma membranes
The Primary Active Transporter II: The ATPase Objectives: Organization P type with NPA domains Proton pumps of the rotary V type ATPase 1 Organization of P type, solute transport, found in plasma membranes
Subcellular biochemistry
 Department of Medical Biochemistry Semmelweis University Subcellular biochemistry February-March 2017 Subcellular biochemistry (biochemical aspects of cell biology) Miklós Csala Semmelweis University Dept.
Department of Medical Biochemistry Semmelweis University Subcellular biochemistry February-March 2017 Subcellular biochemistry (biochemical aspects of cell biology) Miklós Csala Semmelweis University Dept.
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human
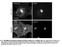 Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
Fig. S1. Subcellular localization of overexpressed LPP3wt-GFP in COS-7 and HeLa cells. Cos7 (top) and HeLa (bottom) cells expressing for 24 h human LPP3wt-GFP, fixed and stained for GM130 (A) or Golgi97
Chapter 1 Membrane Structure and Function
 Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
A Primer on G Protein Signaling. Elliott Ross UT-Southwestern Medical Center
 A Primer on G Protein Signaling Elliott Ross UT-Southwestern Medical Center Receptor G Effector The MODULE Rhodopsins Adrenergics Muscarinics Serotonin, Dopamine Histamine, GABA b, Glutamate Eiscosanoids
A Primer on G Protein Signaling Elliott Ross UT-Southwestern Medical Center Receptor G Effector The MODULE Rhodopsins Adrenergics Muscarinics Serotonin, Dopamine Histamine, GABA b, Glutamate Eiscosanoids
Legionella pneumophila: an intracellular pathogen of phagocytes Prof. Craig Roy
 an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
an intracellular pathogen of phagocytes Section of Microbial Pathogenesis, Yale University School of Medicine 1 Legionella pneumophila Gram-negative bacterium Facultative intracellular pathogen Protozoa
Enzymes: The Catalysts of Life
 Chapter 6 Enzymes: The Catalysts of Life Lectures by Kathleen Fitzpatrick Simon Fraser University Activation Energy and the Metastable State Many thermodynamically feasible reactions in a cell that could
Chapter 6 Enzymes: The Catalysts of Life Lectures by Kathleen Fitzpatrick Simon Fraser University Activation Energy and the Metastable State Many thermodynamically feasible reactions in a cell that could
RECAP (1)! In eukaryotes, large primary transcripts are processed to smaller, mature mrnas.! What was first evidence for this precursorproduct
 RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
BIOLOGY 103 Spring 2001 MIDTERM LAB SECTION
 BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
Ras and Cell Signaling Exercise
 Ras and Cell Signaling Exercise Learning Objectives In this exercise, you will use, a protein 3D- viewer, to explore: the structure of the Ras protein the active and inactive state of Ras and the amino
Ras and Cell Signaling Exercise Learning Objectives In this exercise, you will use, a protein 3D- viewer, to explore: the structure of the Ras protein the active and inactive state of Ras and the amino
Instructions. Fuse-It-mRNA easy. Shipping and Storage. Overview. Kit Contents. Specifications. Note: Important Guidelines
 Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
The Cell and Cellular transport
 Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Information. Supplementary Figures
 Supplementary Information Supplementary Figures Supplementary Figure 1: Mutational analysis of the ADP-based coupled ATPase-AK activity. (a) Proposed model for the coupled ATPase/AK reaction upon addition
Supplementary Information Supplementary Figures Supplementary Figure 1: Mutational analysis of the ADP-based coupled ATPase-AK activity. (a) Proposed model for the coupled ATPase/AK reaction upon addition
Name: Multiple choice questions. Pick the BEST answer (2 pts ea)
 Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
Problem Set #5 4/3/ Spring 02
 Question 1 Chloroplasts contain six compartments outer membrane, intermembrane space, inner membrane, stroma, thylakoid membrane, and thylakoid lumen each of which is populated by specific sets of proteins.
Question 1 Chloroplasts contain six compartments outer membrane, intermembrane space, inner membrane, stroma, thylakoid membrane, and thylakoid lumen each of which is populated by specific sets of proteins.
Lecture 15. Membrane Proteins I
 Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
/searchlist/6850.html Tour of the Cell 1
 http://www.studiodaily.com/main /searchlist/6850.html Tour of the Cell 1 2011-2012 Cytology: science/study of cells To view cells: Light microscopy resolving power: measure of clarity Electron microscopy
http://www.studiodaily.com/main /searchlist/6850.html Tour of the Cell 1 2011-2012 Cytology: science/study of cells To view cells: Light microscopy resolving power: measure of clarity Electron microscopy
Enzymes Part III: regulation II. Dr. Mamoun Ahram Summer, 2017
 Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
Biology 2180 Laboratory #3. Enzyme Kinetics and Quantitative Analysis
 Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
Biol110L-Cell Biology Lab Spring Quarter 2012 Module 1-4 Friday April 13, 2012 (Start promptly; work fast; the protocols take ~4 h)
 Biol110L-Cell Biology Lab Spring Quarter 2012 Module 1-4 Friday April 13, 2012 (Start promptly; work fast; the protocols take ~4 h) A. Microscopic Examination of the Plasma Membrane and Its Properties
Biol110L-Cell Biology Lab Spring Quarter 2012 Module 1-4 Friday April 13, 2012 (Start promptly; work fast; the protocols take ~4 h) A. Microscopic Examination of the Plasma Membrane and Its Properties
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
MAE 545: Lecture 14 (11/10) Mechanics of cell membranes
 MAE 545: ecture 14 (11/10) Mechanics of cell membranes Cell membranes Eukaryotic cells E. Coli FIBROBAST 10 mm E. COI nuclear pore complex 1 mm inner membrane plasma membrane secretory complex ribosome
MAE 545: ecture 14 (11/10) Mechanics of cell membranes Cell membranes Eukaryotic cells E. Coli FIBROBAST 10 mm E. COI nuclear pore complex 1 mm inner membrane plasma membrane secretory complex ribosome
The Molecular Mechanism of Intracellular Membrane Fusion. Richard H. Scheller
 The Molecular Mechanism of Intracellular Membrane Fusion Richard H. Scheller The human brain contains approximately 10 15 connections between nerve cells (Figure 2). The specific formation and modulation
The Molecular Mechanism of Intracellular Membrane Fusion Richard H. Scheller The human brain contains approximately 10 15 connections between nerve cells (Figure 2). The specific formation and modulation
2013 John Wiley & Sons, Inc. All rights reserved. PROTEIN SORTING. Lecture 10 BIOL 266/ Biology Department Concordia University. Dr. S.
 PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
EDUCATIONAL OBJECTIVES
 EDUCATIONAL OBJECTIVES The lectures and reading assignments of BIS 2A are designed to convey a large number of facts and concepts that have evolved from modern studies of living organisms. In order to
EDUCATIONAL OBJECTIVES The lectures and reading assignments of BIS 2A are designed to convey a large number of facts and concepts that have evolved from modern studies of living organisms. In order to
The contribution of proteins and lipids to COPI vesicle formation and consumption. Fredrik Kartberg
 The contribution of proteins and lipids to COPI vesicle formation and consumption Fredrik Kartberg Institute of Biomedicine Department of Medical Genetics 2008 A doctoral thesis at a Swedish University
The contribution of proteins and lipids to COPI vesicle formation and consumption Fredrik Kartberg Institute of Biomedicine Department of Medical Genetics 2008 A doctoral thesis at a Swedish University
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Chapter 3. Protein Structure and Function
 Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Structural vs. nonstructural proteins
 Why would you want to study proteins associated with viruses or virus infection? Receptors Mechanism of uncoating How is gene expression carried out, exclusively by viral enzymes? Gene expression phases?
Why would you want to study proteins associated with viruses or virus infection? Receptors Mechanism of uncoating How is gene expression carried out, exclusively by viral enzymes? Gene expression phases?
Types of cells. Cell size comparison. The Jobs of Cells 10/5/2015. Cells & Cell Organelles. Doing Life s Work
 Types of cells Prokaryote Cells & Cell Organelles bacteria cells Doing Life s Work Eukaryotes 2009-2010 animal cells plant cells Cell size comparison Animal cell Bacterial cell most bacteria (prokaryotic)
Types of cells Prokaryote Cells & Cell Organelles bacteria cells Doing Life s Work Eukaryotes 2009-2010 animal cells plant cells Cell size comparison Animal cell Bacterial cell most bacteria (prokaryotic)
Dynamic Partitioning of a GPI-Anchored Protein in Glycosphingolipid-Rich Microdomains Imaged by Single-Quantum Dot Tracking
 Additional data for Dynamic Partitioning of a GPI-Anchored Protein in Glycosphingolipid-Rich Microdomains Imaged by Single-Quantum Dot Tracking Fabien Pinaud 1,3, Xavier Michalet 1,3, Gopal Iyer 1, Emmanuel
Additional data for Dynamic Partitioning of a GPI-Anchored Protein in Glycosphingolipid-Rich Microdomains Imaged by Single-Quantum Dot Tracking Fabien Pinaud 1,3, Xavier Michalet 1,3, Gopal Iyer 1, Emmanuel
Zool 3200: Cell Biology Exam 4 Part I 2/3/15
 Name: Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name: Trask Zool 3200: Cell Biology Exam 4 Part I 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
20S Proteasome Activity Assay Kit
 20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
Six Types of Enzyme Catalysts
 Six Types of Enzyme Catalysts Although a huge number of reactions occur in living systems, these reactions fall into only half a dozen types. The reactions are: 1. Oxidation and reduction. Enzymes that
Six Types of Enzyme Catalysts Although a huge number of reactions occur in living systems, these reactions fall into only half a dozen types. The reactions are: 1. Oxidation and reduction. Enzymes that
CELL PARTS TYPICAL ANIMAL CELL
 AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
Using a proteomic approach to identify proteasome interacting proteins in mammalian
 Supplementary Discussion Using a proteomic approach to identify proteasome interacting proteins in mammalian cells, we describe in the present study a novel chaperone complex that plays a key role in the
Supplementary Discussion Using a proteomic approach to identify proteasome interacting proteins in mammalian cells, we describe in the present study a novel chaperone complex that plays a key role in the
