Remote Control of Insulin Secretion by Fat Cells in Drosophila
|
|
|
- Augustine Shelton
- 6 years ago
- Views:
Transcription
1 Article Remote Control of Insulin Secretion by Fat Cells in Drosophila Charles Géminard, Eric J. Rulifson, and Pierre Léopold, * Institute for Developmental Biology and Cancer, CNRS/University of Nice-Sophia Antipolis, Parc Valrose, 68 Nice, France Institute for Regeneration Medicine and Diabetes Center, University of California, San Francisco, 53 Parnassus Avenue, San Francisco, CA 9443, USA *Correspondence: leopold@unice.fr DOI.6/j.cmet.9.8. SUMMARY Insulin-like peptides (ILPs) couple growth, metabolism, longevity, and fertility with changes in nutritional availability. In Drosophila, several ILPs called Dilps are produced by the brain insulin-producing cells (IPCs), from which they are released into the hemolymph and act systemically. We show here that in response to nutrient deprivation, brain Dilps are no longer secreted and accumulate in the IPCs. We further demonstrate that the larval fat body, a functional homolog of vertebrate liver and white fat, couples the level of circulating Dilps with dietary amino acid levels by remotely controlling Dilp release through a TOR/RAPTOR-dependent mechanism. We finally use ex vivo tissue coculture to demonstrate that a humoral signal emitted by the fat body transits through the hemolymph and activates Dilp secretion in the IPCs. Thus, the availability of nutrients is remotely sensed in fat body cells and conveyed to the brain IPCs by a humoral signal controlling ILP release. INTRODUCTION Insulin and insulin-like growth factors (IGFs) separated functionally 6 million years ago and share the regulation of complex physiological processes like growth, carbohydrate metabolism, longevity, and reproduction. The mechanisms of action of both molecules are very similar at the cell level, where closely related receptors activate conserved signaling pathways (Nakae et al., ). By contrast, their mechanisms of storage and release have considerably diverged. IGF-I is produced and released freely into the blood, where it accumulates in the form of stable complexes with binding partners, forming an extracellular reservoir that can be used by peripheral tissues. The liver is the main site of IGF-I production. IGF-I transcription is directly controlled by growth hormone (GH) and its receptor on liver cells. Among other environmental factors, nutrition strongly influences the response of liver cells to GH and, therefore, IGF-I transcript levels (Thissen et al., 994). Unlike IGF-I, insulin is stored in intracellular pools in the form of dense granules whose secretion in the blood is highly regulated. Misregulation of insulin release has a strong impact on metabolic homeostasis, leading to type diabetes mellitus (Hosker et al., 989). Due to the growing incidence of this disease, the study of the molecular mechanisms controlling insulin release from pancreatic b cells is the subject of considerable interest (Eliasson et al., 8; Rutter, ). Insulin is produced as a precursor, and its mature form is stored in dense granules. Each granule contains approximately 6 molecules of insulin organized in a semicrystalline arrangement with Zn + ions. Granule exocytosis is induced by increases in plasma glucose. After entry into b cells through specific transporters, glucose is metabolized to produce ATP, which inhibits the Sur-regulatory subunit of K ATP channels and induces membrane depolarization and calcium fluxes. This results in fusion of predocked granules with the plasma membrane, insulin release into the surrounding lymph, and its transcytosis in the capillary vessels. Although glucose is the most relevant physiological regulator of insulin secretion, hormones and nutrients play important additional roles in amplifying glucose-stimulated insulin secretion (Nolan et al., 6; Rutter, ). Pancreatic b cells therefore constitute the node of a general fuel-sensor mechanism. Interestingly, a flurry of recent work has established that insulin/igf functions are evolutionarily conserved in simpler organisms amenable to systematic genetic analysis, like the invertebrate Drosophila melanogaster. In the brain of the fruit fly, two separate groups of cells, the insulin-producing cells (IPCs) and the adipokinetic hormone (AKH)-producing cells (APCs), are functionally analogous to b and a cells, respectively (Geminard et al., 6). These separate endocrine clusters are physically linked by axon-like cellular processes emanating from the IPCs and projecting on the APCs, therefore constituting a functional invertebrate islet. Interestingly, these two groups of cells originate from two pairs of embryonic neuroblasts, expressing genes orthologous to those expressed in the vertebrate anterior pituitary and neurosecretory hypothalamic precursors (Wang et al., 7). IPCs express 4 of the 7 Drosophila insulin-like peptides (DILP,, 3, and 5) and are required for both larval growth and carbohydrate metabolism, since their genetic ablation leads to adult survival with severe growth reduction and increased levels of trehalose (the main circulating carbohydrate in insects) (Brogiolo et al., ; Rulifson et al., ). Although insulin/igf signaling (IIS) is described as allowing coupling organismal growth with energy status (Britton et al., ), it is not clear whether the IPCs directly respond to circulating trehalose, since these cells do not express the Drosophila sur ortholog (Kim and Rulifson, 4). By contrast, the APCs express sur, respond to sulfonylurea, and are crucial regulators Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc. 99
2 of glucose homeostasis (Kim and Rulifson, 4). Dilp function could be controlled by other nutritional inputs, like amino acids or fat. Indeed, starvation blocks larval growth and strongly suppresses IPC-derived DILP3/5 transcription (Ikeya et al., ). However, DILP expression, which contributes approximately 8% of DILP gene transcription in the IPCs (Buch et al., 8), remains unchanged (Ikeya et al., ). This suggests that the production of Dilp peptides is controlled posttranscriptionally by nutritional inputs. In this line, recent genetic analysis of the nucleostemin family gene NS3 has revealed that NS3 function is required in brain serotonergic neurons for full activation of the insulin pathway in peripheral tissues. In particular, ns3 mutant larvae are small and accumulate in the IPCs despite normal levels of DILP transcripts, therefore suggesting that Dilp secretion is controlled by a serotonergic neuronal input (Kaplan et al., 8). Nevertheless, the existence of a possible link between this input and the nutritional information has not been explored yet. Previous data indicate that changes in the nutritional status of the larva are relayed by an important endocrine organ called the fat body, functionally related to the vertebrate liver and white adipose. When amino acid import is specifically restricted in fat body cells by genetic manipulation, general IIS is decreased in peripheral tissues, leading to a systemic reduction of larval growth (Colombani et al., 3). The mechanisms relaying the information from the fat body to the peripheral tissues are currently unknown. As a starting point to our present work, we have hypothesized that the fat body remotely controls the level of circulating Dilps, which in turn control peripheral tissue growth. We show that Dilp secretion from the brain IPCs is controlled by nutrition. Moreover, we present evidence that amino acid shortage or inhibition of the TOR-signaling pathway, both targeted in fat body cells, are sufficient to induce Dilp retention in the IPCs. Finally, we use an ex vivo tissue coculture system to demonstrate that Dilp secretion is controlled by a direct humoral link between the fat body and the brain. RESULTS The Secretion of Brain Dilps Is Controlled by Diet In order to follow the behavior of Dilp molecules in larval brain, we designed specific antibodies directed against and Dilp5, two ILPs produced in the brain IPCs. The specificity of these antibodies is presented in Figure SA. In normally fed larvae (Rich), and Dilp5 present indistinguishable patterns, accumulating in dotted structures in the IPC cell body, along the axonal projections, and in the axonal termini on the corpora cardiaca (the site of AKH production) and the aorta. Upon feeding on low-aminoacid diet (Poor,.3 yeast, see Experimental Procedures) or acute starvation (Starved, PBS/% sucrose for 4 hr), the IPCs of third-instar larvae showed strong accumulation of and Dilp5 in the cell bodies and in the axonal termini (Figures A, B, and SB). This accumulation could be efficiently reverted by transferring back starving larvae on food (Rich, 3 yeast). Indeed, brain fluorescence decreased after only 5 min refeeding, reaching fed levels after hr (Figure D). Interestingly, refeeding with tryptone extract (proteolyzed casein) reduced Dilp accumulation, whereas sucrose or soy lipids did not have any effect (Figure E). We then carried out refeeding experiments using single-amino-acid diets (amino acid in agar/% sucrose), which revealed that only L-leucine and L-isoleucine can promote Dilp release (Figure E and data not shown). This indicates that amino acids (and specifically the branched-chain amino acids leucine and isoleucine) are key in controlling this event. The rapid response obtained upon refeeding suggests that the mechanism of Dilp accumulation does not involve increased gene expression. Accordingly, the measurement of DILP transcripts by quantitative RT-PCR on dissected larval brains showed no significant change of DILP expression and a decrease in DILP5 transcription upon culture on food (Figure C). A FLAG-tagged form of ( F ) expressed in the IPCs using the DILP-Gal4 driver was subjected to the same regulation, showing strong accumulation in the IPCs under restrictive diet (Figure A). Western blot experiments using anti- FLAG antibodies confirmed that F accumulates in the brain of underfed larvae and revealed that, in these conditions, its concentration in the hemolymph is reduced by 7% (Figure B). This demonstrates that increased labeling as seen in IPCs of underfed larvae is the sign of a retention in the brain and that levels of circulating in the hemolymph are decreased upon restrictive diet. Accordingly, in these conditions, trehalosemia was increased (Figure C). Taken together, our results indicate that diet and in particular amino acids control Dilp secretion in larval IPCs and, as a consequence, larval growth. The Brain IPCs Couple Neurosecretion with Nutritional Inputs To test whether the 4 IPCs are specialized neurosecretory cells controlling Dilp secretion in response to low diet, we expressed DILP either in a control set of neurons that do not express the DILP genes (using the kurs6-gal4 driver) (Siegmund and Korge, ) or in the IPCs (using DILP-Gal4) and measured the weight gain obtained after raising the larvae on (3) or (.33) medium (Figure 3A). Larvae expressing DILP in kurs6 neurons were % bigger than control animals, indicating that when expressed in these neurons, is secreted and activates systemic growth. Interestingly, kurs6 > DILP animals presented low diet-resistant growth increase with commensurate developmental delay, reaching adult sizes comparable to the one observed under normal food condition (Figure 3A). By contrast, overexpression of DILP in the larval IPCs led to moderate weight increase in the fed condition and was barely able to induce weight gain under low diet, suggesting that DILP expression leads to a very limited increase in circulating levels of peptide (Figure 3A). This indicates that the IPCs have the specialized function of coupling Dilp secretion with nutritional input. Moreover, expression of in the kurs6 neurons upon severely restrictive diet (.3) lead to % larval lethality, whereas strong overexpression in the IPCs was fully viable (data not shown). This further indicates that the regulation of secretion by the brain IPCs is a major physiological regulation allowing modulating IIS in response to diet in the larva. To confirm this, we forced membrane depolarization and neurosecretion in the IPCs by using the targeted expression of a bacterial sodium channel (NaChBac). In these conditions, accumulation normally observed in response to restrictive diet was Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc.
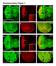
3 A B Mean fluorescence (f.c.) RNA levels (f.c.) C,4,,8,6,4, starved Dilp5 Dilp5 Dilp5 starved DILP DILP5 D E Mean fluorescence (f.c.) Mean fluorescence (f.c.) * * Time (min) starved refed x tryptone sugar high fat Ile Leu Figure. Diet Controls the Level of Dilp Accumulation in the Brain Neurosecretory Cells (A) Insulin-producing cells (IPCs, labeled with GFP) are visualized in the brain hemisphere. Brains from third-instar larvae fed, raised on medium (.3), or starved for 4 hr on PBS/% sucrose were immunostained with anti- antibodies and imaged by confocal microscopy, using laser and scan settings adjusted on the accumulation state to avoid saturation. With these settings, Dilp staining is not visible in the fed (3) state. Low-magnification Nomarski image of a brain is presented in the upper left panel, showing IPCs as green-labeled cells. (B) Quantified fluorescence intensities for and Dilp5 in the IPCs of larvae raised on different diets during all development (n = 3 larvae for each plot, p <.; error bars represent SEM). (C) Measurement of larval brain DILP and DILP5 transcript levels during starvation by quantitative RT-PCR. Larvae were reared on normal or restricted food during all development. Fold changes (f.c.) are indicated; RP-49 is used as an internal reference; error bars represent SEM. (D) Kinetics of Dilp accumulation upon refeeding. Third-instar larvae (7 hr AED) were starved on PBS/% sucrose for 4 hr then transferred to medium for indicated times. fluorescence intensity in the IPCs was quantified as previously (n = 3 larvae for each plot; error bars represent SEM). (E) Ingestion of amino acids is sufficient to induce insulin secretion from IPCs. Third-instar larvae starved on PBS/% sucrose for 4 hr were refed for 6 hr with 3 food, 7 g/l tryptone, % sucrose or glucose, 7 g/l soy lipids mix,.3% L-isoleucine, or.% L-leucine. Brains were immunostained for, and fluorescence intensity was quantified (n = larvae, *p <.5, p <. compared to nonfed control; error bars represent SEM). prevented, and significant lethality (5%) was observed (Figures 3B and 3C). In order to characterize the regulation of Dilp secretion in the IPCs, we first tested whether other secreted molecules were subjected to a control of secretion comparable to the one operating for endogenous Dilps. For this purpose, a secreted GFP (secgfp) construct was expressed in the IPCs using the Gal4 system, and the behavior of the protein was followed in and restrictive-diet conditions. As in the case of FLAG-tagged, secgfp presented a strong accumulation in low diet (Figure A). By contrast, neither secgfp nor F ectopically produced in the Kurs6 neurons was subjected to low-dietinduced accumulation (data not shown), indicating that the IPCs have the specific ability to couple the release of secreted molecules to a nutritional input. We next tested whether IPC membrane depolarization was important for the control of Dilp release by inducing expression of mammalian Kir., a constitutively active potassium channel, specifically in the IPCs. Kir.-induced hyperpolarization of IPCs resulted in increased accumulation of in the cell bodies and Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc.
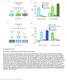
4 A DILP-Gal4 > F B C DILP-Gal4 >secgfp LSP F quant. (a.u.) hemolymph hemolymph glycemia (mg/ml) F quant. (a.u.) brains 5 5 brains Flag tubulin Figure. Dilp Accumulation in the IPCs Reflects a Control of Their Release in the Hemolymph (A) Restrictive diet induces the accumulation of secreted proteins in the IPCs. A GFP linked to a signal peptide (secgfp) or a FLAG-tagged ( F ) were overexpressed in the IPCs using the DILP-Gal4 driver. Both proteins show accumulation under restrictive diet, similar to that observed for the endogenous Dilps. (B) Hemolymph or brain extracts from larvae expressing F with the DILP- Gal4 driver were immunoblotted for F in or medium conditions (western blot image and quantification are shown). For hemolymph, relative concentrations of circulating F were calculated (arbitrary units/ml of hemolymph). The amount of F is normalized to LSP in hemolymph or tubulin in brains; error bars represent SEM. (C) Glycemia is severely increased under starvation. Glucose circulating levels of normally fed and starved third-instar larvae were measured after conversion of hemolymph trehalose into glucose (n = 8, p <.; error bars represent SEM). in the axonal termini and was accompanied by a drastic inhibition of larval growth, leading to adults with 4% weight reduction and increased glycemia (Figure 3D). These data demonstrate that Dilp secretion is coupled to changes in membrane polarity in the IPC. Altogether, these results establish that the brain IPCs have the special ability to couple neurosecretion with nutrition and that Dilp release from the IPCs is controlled by membrane voltage-dependent neurosecretory mechanisms similar to those operating for the secretion of insulin and other neuropeptides. The Fat Body Remotely Controls Insulin Release from Brain IPCs While our results suggest that the molecular mechanism of Dilp secretion in the IPCs involves changes in IPC membrane potential, the upstream events leading to its activation/inhibition remain unknown. Interestingly, it has been previously found that the fly IPCs do not express the unique Drosophila sur ortholog, therefore suggesting that these cells do not respond directly to carbohydrate levels (Kim and Rulifson, 4). Therefore, a relay mechanism must integrate the nutritional information and convey it to the IPCs for proper insulin release. We had previously demonstrated a role for the fat body in relaying the nutritional information to general IIS in peripheral tissues. Hence, we asked whether the nutritional sensing operating in the fat body could control the release of Dilps from the brain IPCs. For this purpose, we used a genetic starvation tool, making use of the Gal4/UAS system to induce the silencing of an amino acid transporter encoded by the slimfast (slif) gene in a tissuespecific manner (Colombani et al. 3). When expression of slif is knocked down in the fat body, mimicking amino acid depletion, a local downregulation of TOR signaling is induced, followed by a systemic downregulation of IIS and, ultimately, growth inhibition. We therefore monitored the level of Dilp release upon slif downregulation in the fat body (referred to as ppl > slif A )(Colombani et al. 3). Interestingly, the brain IPCs of ppl > slif A larvae showed a retention of similar to that observed upon restrictive diet or starvation (Figures 4A and 4B). Similar retention was observed upon inhibition of TORC signaling in the fat body (overexpression of the TOR inhibitors TSC and TSC or knockdown of RAPTOR), but not upon InR/ PI3K inhibition (overexpression of the inhibitor PTEN), consistent with our previous finding that TOR is the main relay for the amino acid signal in fat-body cells (Figures 4A and 4B) (Colombani et al., 3). slif inhibition targeted to other larval tissues had no systemic effect on larva growth (Colombani et al. 3) and did not promote Dilp accumulation in the IPCs (data not shown). We then asked whether restoring the level of TOR signaling in fat cells of underfed larvae would be sufficient to restore Dilp release. To test this, we activated TOR signaling in the fat body of underfed animals by knocking down the TSC gene (ppl > TSCi) and analyzed the level of in the brain IPCs. Indeed, restoring TOR activation in fat cells was sufficient to prevent accumulation in brain cells under restrictive diet (Figure 4C). Similar results were obtained using anti-dilp5 antibodies (data not shown), suggesting again that all Dilps are subjected to the same regulation in the IPCs. In addition, although ppl > TSCi larvae developed normally on medium (3), these animals died as larvae on.3 medium, suggesting that bypassing the regulation on Dilp secretion under food limitation is strongly Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc.
5 A Adult weight (%) DILP>w +6% +% DILP>DILP kurs6>w kurs6>dilp DILP>w +9% +47% DILP>DILP kurs6>w kurs6>dilp B Mean fluorescence (f.c.) DILP-Gal4 ctrl >NaChBac C DILP-Gal4 D DILP-Gal4 adult emergence (%) ctrl >NaChBac ctrl Adult weight (%) >Kir. 4 Glycemia (mg/ml) ctrl >Kir. ctrl >Kir. Figure 3. The Electric Activity of the Brain IPCs Links Dilp Secretion with the Nutritional Status (A) In restrictive diet conditions, expression of in the IPC induces only mild overgrowth, as compared to its expression in control neurons. DILP was overexpressed either in IPCs or in control neurons (Kurs6-Gal4). Larvae were reared on or medium, and adult flies were weighted (n = 3 3, p <.; error bars represent SEM). (B) Neurosecretion was forced using targeted expression of a bacterial sodium channel (NaChBac), provoking membrane depolarization in the IPCs. In this background, no accumulation was observed upon starvation; error bars represent SEM. (C) On restrictive diet, expression of a bacterial sodium channel (NaChBac) induces larval/pupal death. Larvae were reared on or medium (5 per vial), and emerging adult flies were counted (n = 3 3 5; error bars represent SEM). (D) Brains from normally fed larvae overexpressing the Kir. channel in the IPCs present a strong accumulation of (upper panel), associated with a severe decrease in adult size (8 C, n = 3 3, p <.) and an increased glycemia (n = 8, p <.; error bars represent SEM). detrimental to larval development. Taken together, these results indicate that the fat body integrates nutritional inputs and remotely controls brain Dilp release and that TOR signaling is the molecular effector for this regulation in fat cells. A Direct Humoral Link between the Fat Body and the Brain This regulation could be the consequence of a direct humoral link between the fat body and the brain or an indirect effect of fatbody metabolism on general homeostasis leading to perturbed IPC function. To distinguish between these two possibilities, we set up ex vivo cocultures of larval brains and fat-body explants (Figure 5A). In a first series of experiments, brains from starved third-instar larvae were cultured in Schneider medium in the presence of serum. In these conditions, was present at high levels in the IPCs of dissected brains and remained stable even after 5 hr of culture. Addition of fat bodies dissected from underfed animals in the culture did not modify levels, indicating that underfed fat bodies are unable to stimulate Dilp release. By contrast, addition of fat bodies from fed animals induced a strong reduction of staining (Figure 5B). This staining did not fade in fed brains incubated with fed fat bodies Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc. 3
6 A B C ppl-gal4 ctrl >Slif a >TSC/ >PTEN Mean fluorescence (f.c.) c t r l > S l i f a ppl-gal4>tsci > T S C / > P T E N > R A P T O R i (data not shown), indicating that the reduction of staining observed in underfed brain/fed fat body cocultures is not due to a specific degradation induced by the coculture, but reflects Dilp release from the IPCs. Accordingly, incubation of underfed brains in the presence of mm KCl, a condition that forced depolarization of brain cells, also induced Dilp release, in line with our previous results obtained upon ectopic expression of the Kir. potassium channel (Figure 5B). Incubation of fed and underfed fat bodies together with larval brains induced Dilp release similarly to that observed with fed fat bodies alone. Therefore, our experiments suggest that fat bodies in the fed state emit a diffusible factor stimulating brain Dilp release that is dominant over the underfed state. We next reasoned that the secretion signal produced by the fat body should be released and circulate into the larval hemolymph in order to reach the brain IPCs. For this, we tested the effect of larval hemolymph on Dilp release in our culture system. Interestingly, hemolymph from fed but not from underfed or ppl > slif A larvae was able to promote efficient release from starved brains (Figure 5B). This established that a secretion factor emanating from the fat body is released in the hemolymph in fed conditions. This secretion signal could be either a metabolite or a specific factor produced by the fat body. We tested the effects of different metabolites added directly to the brain culture. Neither carbohydrates (glucose, sucrose, or trehalose), lipids (complete soy lipid extract), nor amino acids (already present in the Schneider medium) were able to promote Dilp release when added in the culture with larval brains (Figure 5B). Similar results were obtained using anti-dilp5 antibodies (data not shown). Overall, this demonstrates that the IPCs do not respond directly to nutrient inputs, but instead the fat body relays the nutritional information to the IPCs through the production of a specific secretion signal. DISCUSSION Mean fluorescence (f.c.) Figure 4. The Larval Fat Body Uses TOR Signaling to Relay the Nutritional Signal to the Brain IPCs (A) Reducing amino acid import or TOR signaling in fat-body cells remotely induces accumulation in the IPCs. The slif A genetic background (Colombani et al., 3) was used to suppress amino acid import in fat cells. Overexpression of TSC and TSC or Raptor silencing was used to specifically reduce TORC signaling, and overexpression of PTEN was used to reduce PI3K activity. slif A and TOR knockdown directed in fat cells induced Dilp accumulation, whereas PI3K inhibition did not. Experiments were carried out with Due to the lack of immunoassay, the study of the regulation of Dilp levels in Drosophila has been limited so far to the analysis of their expression level in response to nutritional conditions. Here, we present evidence that the secretion of and Dilp5 as well as a secgfp is controlled by the nutritional status of the larva. Our data also indicate that the IPCs have the specific ability to couple secretion with nutritional input. This suggests that all Dilps produced in the IPCs could be subjected to a common control on their secretion that could therefore the fat-body ppl-gal4 driver; identical results were obtained with the cg-gal4 driver (data not shown). (B) Anti- fluorescence in the IPCs was quantified as described (n = larvae, p <. compared to control; error bars represent SEM). (C) Activation of TOR signaling in the larval fat body is sufficient to block Dilp accumulation induced by restrictive diet. Larvae expressing a TSC RNAi construct in the fat body (ppl > TSCi) were reared on (3) and (.3) medium. In these conditions, accumulation of in the IPCs on medium was no more observed. For both conditions, IPCs were imaged by confocal microscopy using laser and scan settings adjusted on the fed state, which explains the difference in intensity in the fed state when compared to other images. Anti- fluorescence in the IPCs was quantified as described previously (n = 5 larvae; error bars represent SEM). Experiments were carried out with the fat-body ppl-gal4 driver; identical results were obtained with the cg-gal4 driver (data not shown). 4 Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc.
7 Figure 5. A Humoral Link between the Fat Body and the Brain Remotely Controls Insulin Secretion (A) Coculture of dissected larval brains and fat bodies in Schneider medium. The brains sink to the bottom of the microfuge tube and are therefore never in direct contact with the fat body explants that float at the surface. (B) A positive signal for Dilp secretion is sent by the fat body to the brain. After culture with/without fat-body explants, the brains were immunostained for, and anti- fluorescence was quantified as described. Brains dissected from underfed animals and cultured in Schneider medium without fat bodies served as control fluorescence levels. Same cultures were carried out using hemolymph instead of fat bodies (Hemolymph). Finally, underfed brains were cultured in mm KCl or % sucrose or 7 g/l tryptone solutions (n = brains, p <.; error bars represent SEM). (C) A model for the remote control of brain Dilp release by the fat body. Amino acids are delivered by the gut and taken up by fat-body cells. In the presence of amino acids in the diet, TORC signaling in fat cells generates a positive messenger that is released into the hemolymph. This signal reaches the brain IPCs, where it remotely controls Dilp secretion. override differences in their transcriptional regulation. We further show that the regulation of Dilp secretion plays a key role in controlling Dilp circulating levels and biological functions, since blocking neurosecretion in the IPCs led to growth and metabolic defects, and conversely, expression of in nonregulated neurosecretory cells is lethal upon starvation. Interestingly, previous reports suggest that Dilp release could also be controlled in the adult IPCs, raising the possibility that this type of regulation contributes to controlling metabolic homeostasis, reproduction, and aging during adult life (Gershman et al., 7; Tu and Tatar, 3). Dilp release is not activated by high-carbohydrate or -fat diets, but rather depends on the level of amino acids and in particular on the presence of branched-chain amino acids like leucine and isoleucine. This finding is consistent with the described mechanism of TOR activation by leucine in mammalian cells (Avruch et al., 9; Nicklin et al., 9). In particular, it was recently shown that Rag GTPases can physically interact with mtorc and regulate its subcellular localization in response to L-leucine (Sancak et al., 8). Interestingly, our present work indicates that amino acids do not directly signal to the IPCs, but rather they act on fat-body cells to control Dilp release. TOR signaling has been previously shown to relay the nutritional input in fatbody cells (Colombani et al., 3). We show here that it is required for the remote control of Dilp secretion, since inhibition of Raptor-dependent TOR activity in fat cells provokes Dilp retention. Surprisingly, activation of TOR signaling in fat cells of underfed larvae is sufficient to induce Dilp release, indicating that TOR signaling is the major pathway relaying the nutrition signal from the fat body to the brain IPCs. In contrast, inhibition of PI3K activity in fat cells does not appear to influence Dilp secretion in the brain. This result is in line with our previous in vivo data showing that reduction of PI3K levels in fat cells does not induce systemic growth defects (Colombani et al., 3). Altogether, this suggests that the nutritional signal is read by a TOR-dependent mechanism in fat cells, leading to the production of a secretion signal that is conveyed to the brain by the hemolymph (Figure 5D). Our ex vivo brain culture experiments demonstrate that hemolymph or dissected fat bodies from fed larvae constitute an efficient source for the Dilp secretion factor. This signal is absent in underfed animals, suggesting that it could be identified by comparative analysis of fed and underfed states. The nature of the secretion signal is unknown. It is produced and released in the hemolymph by fat cells, and its production relies on TORC function. Given the role of TORC in protein translation, one could envisage that the secretion factor is a protein or a peptide for which translation is limited by TORC activity and relies on amino acid input in fat-body cells. In mammals, fatty acids and other lipid molecules have the capacity to amplify glucose-stimulated insulin secretion in pancreatic b cells (Nolan et al., 6). The fly fat body carries important functions related to lipid metabolism, and a recent link has been established between TOR signaling and lipid metabolism in flies (Porstmann et al., 8), leaving open the possibility that a TOR-dependent lipid-based signal could also operate in this regulation. Interestingly, carbohydrates do not appear to contribute to the regulation of insulin secretion by brain cells in flies. This finding is reminiscent of the absence of expression of the Sur ortholog in the IPCs and suggests that global carbohydrate levels are controlled by the glucagon-like AKH produced by the corpora cardiaca cells (Kim and Rulifson, 4). Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc. 5
8 Our experiments demonstrate that Dilp secretion is linked to the polarization state of the IPC membrane, suggestive of a calcium-dependent granule exocytosis, like the one observed for insulin and many other neuropeptides. The nature of the upstream signal controlling membrane depolarization is not known. Recent data concerning the function of the nucleostemin gene ns3 in Drosophila suggest that a subset of serotonergic neurons in the larval brain act on the IPCs to control insulin secretion (Kaplan et al., 8). Therefore, it remains to be known whether the IPCs or upstream serotonergic neurons constitute a direct target for the secretion signal. So far, no link has been established between the serotonergic stimulation of IPC function and the nutritional input. In 998, J. Britton and B. Edgar presented experiments where starved brain and fed fat bodies were cocultured, allowing arrested brain neuroblasts to resume proliferation in the presence of nutrients (Britton and Edgar, 998). From these experiments, the authors proposed that quiescent neuroblasts were induced to re-enter the cell cycle by a mitogenic factor emanating from the fed fat bodies. Our present data extend these pioneer findings and suggest the possibility that the factor sent by the fed fat bodies is the secretion factor that triggers Dilp release from the IPCs, allowing neuroblasts to continue their growth and proliferation program through paracrine Dilp-dependent activation. In conclusion, our work combines genetic and physiology approaches on a model organism to decipher key physiological regulations and opens the route for a genetic study of the molecular mechanisms controlling insulin secretion in Drosophila. EXPERIMENTAL PROCEDURES Nomenclature Gal4 driver lines were named after the gene used for Gal4 expression specificity (for example DILP) followed by either -Gal4 or > (DILP-Gal4 or DILP >). The name DILP > DILP is therefore an abbreviation for the genotype DILP-Gal4;UAS-DILP. Ctrl stands for control, i.e., Gal4 driver lines crossed to wild-type strain w 8. Fly Strains and Food The following fly lines were used: w 8 ; ppl-gal4 (Colombani et al. 3), cg-gal4 (Bloomington Stock Center; Indiana University), DILP-Gal4 (Brogiolo et al., ; Rulifson et al., ), Kurs6-Gal4 (Siegmund and Korge, ), UAS-FLAG-DILP (Honegger et al. 8), UAS-SecGFP (Entchev et al., ), UAS-DILP; UAS-Kir. (Brogiolo et al., ; Rulifson et al., ), UAS-Slif a (Colombani et al. 3), UAS-Raptor RNAi and UAS-TSC RNAi (VDRC) (Dietzl et al., 7), UAS-dPTEN (Huang et al. 999), yw 8 ; UAS-TSC and UAS- TSC (Tapon et al. ), and UAS-NaChBac (Bloomington Stock Center). Both ppl-gal4 and cg-gal4 express in non-fat-body-related tissues, but their expression domains intersect only in the fat body. No trace of expression was found in the IPCs. In all the presented experiments, both drivers produced identical results, ensuring that the effects are due to fat-body expression. The Kurs6-Gal4 driver line is a P[Gal4] enhancer trap line that expresses Gal4 in a groupof neurosecretory neurons innervating the corporacardiacaand located in the lateral protocerebrum, the pars intercerebralis, and the medial subesophageal ganglion. The expression level of Kurs6-Gal4 is comparable to that of the endogenous DILP promoter, as shown in Figure S. Animals were reared at 5 Con3 food containing, per liter: 34 g inactivated yeast powder, 83 g corn flour, g agar, 6 g white sugar, and 4.6 g Nipagin M (in ethanol) (referred to Rich medium). Restrictive diets (also labeled medium) correspond to.3 or.33 medium containing.7 g/l or 5. g/l inactivated yeast powder, respectively. For acute starvation, third-instar larvae (7 hr after egg deposition [AED]) were shifted on agar plates containing PBS/ % sucrose and collected for tissue dissection after 4 hr. Quantitative RT-PCR Larvae were collected 74 hr after egg laying and dissected, and brains were flash-frozen (5 brains per sample). Total RNA was extracted using QIAGEN RNeasy Lipid Tissue Mini Kit according to the manufacturer s protocol. RNA samples (5 mg per reaction) were reverse transcribed using SuperScript II (Invitrogen), and the generated cdna was used for real-time RT-PCR (ABI Prism 7 system, qpcr Mastermix Plus for SYBRGreen I, Eurogentec France; Angers, France), using.8 ng of cdna template and a primer concentration of 3 nm. Rp49 was used as a normalizer. Three separate samples were collected from each condition, triplicate measurements were conducted, and two independent sets of primers were used for each gene. Primers were designed using the Primer Express software (Applied Biosystems; Foster City, CA). Statistics For all experiments, error bars represent SEM, and p values are the results of a Student s test provided by Microsoft Excel (*p <.5; p <.). Antibodies and Immunostaining To generate antisera specific for the protein, a peptide corresponding to amino acids 8 8 of the sequence (TRQRQGIVERC) was used as immunogen in rats (Eurogentec). For Dilp5 antisera generation, two peptides corresponding to amino acids 4 5 and 77 9 (CPNGFNSMFA, SLSSIRRDFRGVVDSC) were used as immunogen in rabbits (Eurogentec). Brains were dissected from larvae in PBS, fixed in PBS containing 4% formaldehyde for 5 min at room temperature, and extensively washed in PBS containing.3% Triton X- (PBT). Tissues were then blocked for hr in PBT containing 5% BSA. Primary antibodies were incubated overnight at 4 C, and secondary antibodies for hr at room temperature after extensive washes. Tissues were mounted in Vectashield mounting medium (Vector Laboratories, Inc.; Peterborough, UK), and fluorescence images were acquired using a Zeiss LSM 5 META confocal laser scanning microscope. Antibodies used: rat anti-, M anti-flag (Sigma-Ald; St. Louis), anti-lsp (Burmester et al., 999), anti-rat Alexa 546, and anti-mouse Alexa 546 conjugated (/6; Molecular Probes). Weighing Flies L larvae were collected 4 hr after egg deposition (AED, 4 hr egg collections) and reared at 3 animals per tube. Groups of 3 adult males were weighed with an Adventurer Pro Precision balance (Ohaus; Pine Brook, NJ). Trehalose Measurements For each genotype, 3 L larvae were incubated at 5 C until L3. Hemolymph from eight groups of ten larvae was used for each condition. Hemolymph was diluted (:) in homogenization buffer (37 mm NaCl,.7 mm KCl, 5 mm Tris [ph 6.6]) and heated for 5 min at 7 C, and trehalose was converted into glucose after incubation with porcine trehalase (Sigma, T8778) at 37 C overnight. Total glucose was measured using the Thermo Glucose GOD-POD assay kit (Thermo Fisher Scientific; Waltham, MA). Quantifications were performed using a Sunrise spectrophotometer plate reader at 5 nm (Tecan; Mannedorf, Switzerland). Fluorescence Quantification To quantify levels, confocal Z series of the IPCs were obtained using a mm step size and identical laser power and scan settings. ImageJ software was used to generate sum-intensity 3D projections of the Z stacks ( bit scanned images) and to measure total fluorescent intensity across the IPCs. Ex Vivo Organ Coculture Ex vivo organ culture was performed using a method based on that of Britton and Edgar (998). Larvae were cultured on standard medium, surface-sterilized in a solution of 7% ethanol for min, rinsed in sterile water, and dissected in Schneider insect cell-culture medium (Sigma) using sterile tools. Brains were transferred to a microcentrifuge tube containing a ml Schneider medium with or without 5% FBS. A dissecting needle was used to transfer the tissues in a minimal volume of medium. In coculture experiments, the entire fat bodies from 5 feeding mid-third-instar larvae were added to the culture. Coculture experiments were also performed with the hemolymph collected 6 Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc.
9 from 5 mid-third-instar larvae (5 ml) and diluted in Schneider medium (:) prior to addition to the brains culture. Cultures were incubated at room temperature for the indicated times. Western Blotting Fed or underfed mid-third-instar larvae were collected and washed in cold PBS. Brains were dissected and hemolymph collected. Tissues and total larvae were homogenized in lysis buffer (5 mm Tris [ph 8], 5 mm NaCl, mm EGTA, % NP-4, Complete Roche protease inhibitor). Samples were quantified using the Bradford method and denatured in x Laemmli buffer with mm DTT, and equal amounts of protein were loaded for each lane onto NuPAGE Novex gel (4% % Tris-Glycine, Invitrogen). Enhanced chemoluminescence (Advanced ECL, Invitrogen; Carlsbad, CA) was used for antibody detection after blotting on Immobilon-P SQ membrane (Millipore; Billerica, MA). Signal quantification was performed using Fujifilm Multi Gauge software. SUPPLEMENTAL DATA Supplemental Data include one figure and can be found online at cell.com/cell-metabolism/supplemental/s55-43(9)3-9. ACKNOWLEDGMENTS We are grateful to Julien Colombani, whose early experiments first suggested a control of Dilp secretion by nutrients. We thank Gisèle Jarretou and Aurore Dussert for technical help and all members of the laboratory for fruitful discussions and comments on the manuscript. We thank PRISM Platform for the imaging facility. We thank the Vienna Drosophila RNAi Center (VDRC) and the Bloomington Stock Center for providing Drosophila lines. This work was supported by the Centre National de la Recherche Scientifique, the Institut National de la Santé et de la Recherche Médicale, the Fondation pour la Recherche Médicale, the Fondation de France, and the Agence Nationale pour la Recherche for C.G. and P.L. and NIH grant NIH RDK6949 for E.J.R. Received: April, 9 Revised: July 7, 9 Accepted: August, 9 Published: September, 9 REFERENCES Avruch, J., Long, X., Ortiz-Vega, S., Rapley, J., Papageorgiou, A., and Dai, N. (9). Amino acid regulation of TOR complex. Am. J. Physiol. Endocrinol. Metab. 96, E59 6. Britton, J.S., and Edgar, B.A. (998). Environmental control of the cell cycle in Drosophila: nutrition activates mitotic and endoreplicative cells by distinct mechanisms. Development 5, Britton, J.S., Lockwood, W.K., Li, L., Cohen, S.M., and Edgar, B.A. (). Drosophila s insulin/pi3-kinase pathway coordinates cellular metabolism with nutritional conditions. Dev. Cell, Brogiolo, W., Stocker, H., Ikeya, T., Rintelen, F., Fernandez, R., and Hafen, E. (). An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr. Biol., 3. Buch, S., Melcher, C., Bauer, M., Katzenberger, J., and Pankratz, M.J. (8). Opposing effects of dietary protein and sugar regulate a transcriptional target of Drosophila insulin-like peptide signaling. Cell Metab. 7, Burmester, T., Antoniewski, C., and Lepesant, J.A. (999). Ecdysone-regulation of synthesis and processing of fat body protein, the larval serum protein receptor of Drosophila melanogaster. Eur. J. Biochem. 6, Colombani, J., Raisin, S., Pantalacci, S., Radimerski, T., Montagne, J., and Leopold, P. (3). A nutrient sensor mechanism controls Drosophila growth. Cell 4, Dietzl, G., Chen, D., Schnorrer, F., Su, K.C., Barinova, Y., Fellner, M., Gasser, B., Kinsey, K., Oppel, S., Scheiblauer, S., et al. (7). A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, Eliasson, L., Abdulkader, F., Braun, M., Galvanovskis, J., Hoppa, M.B., and Rorsman, P. (8). Novel aspects of the molecular mechanisms controlling insulin secretion. J. Physiol. 586, Entchev, E.V., Schwabedissen, A., and Gonzalez-Gaitan, M. (). Gradient formation of the TGF-beta homolog Dpp. Cell 3, Geminard, C., Arquier, N., Layalle, S., Bourouis, M., Slaidina, M., Delanoue, R., Bjordal, M., Ohanna, M., Ma, M., Colombani, J., et al. (6). Control of metabolism and growth through insulin-like peptides in Drosophila. Diabetes 55 (Suppl ), S5 S8. Gershman, B., Puig, O., Hang, L., Peitzsch, R.M., Tatar, M., and Garofalo, R.S. (7). High-resolution dynamics of the transcriptional response to nutrition in Drosophila: a key role for dfoxo. Physiol. Genomics 9, Honegger, B., Galic, M., Köhler, K., Wittwer, F., Brogiolo, W., Hafen, E., and Stocker, H. (8). Imp-L, a putative homolog of vertebrate IGF-binding protein 7, counteracts insulin signaling in Drosophila and is essential for starvation resistance. J. Biol. 7,. Hosker, J.P., Rudenski, A.S., Burnett, M.A., Matthews, D.R., and Turner, R.C. (989). Similar reduction of first- and second-phase B-cell responses at three different glucose levels in type II diabetes and the effect of gliclazide therapy. Metabolism 38, Huang, H., Potter, C.J., Tao, W., Li, D.M., Brogiolo, W., Hafen, E., Sun, H., and Xu, T. (999). PTEN affects cell size, cell proliferation and apoptosis during Drosophila eye development. Development 6, Ikeya, T., Galic, M., Belawat, P., Nairz, K., and Hafen, E. (). Nutrient-dependent expression of insulin-like peptides from neuroendocrine cells in the CNS contributes to growth regulation in Drosophila. Curr. Biol., Kaplan, D.D., Zimmermann, G., Suyama, K., Meyer, T., and Scott, M.P. (8). A nucleostemin family GTPase, NS3, acts in serotonergic neurons to regulate insulin signaling and control body size. Genes Dev., Kim, S.K., and Rulifson, E.J. (4). Conserved mechanisms of glucose sensing and regulation by Drosophila corpora cardiaca cells. Nature 43, Nakae, J., Kido, Y., and Accili, D. (). Distinct and overlapping functions of insulin and IGF-I receptors. Endocr. Rev., Nicklin, P., Bergman, P., Zhang, B., Triantafellow, E., Wang, H., Nyfeler, B., Yang, H., Hild, M., Kung, C., Wilson, C., et al. (9). Bidirectional transport of amino acids regulates mtor and autophagy. Cell 36, Nolan, C.J., Madiraju, M.S., Delghingaro-Augusto, V., Peyot, M.L., and Prentki, M. (6). Fatty acid signaling in the beta-cell and insulin secretion. Diabetes 55 (Suppl ), S6 S3. Porstmann, T., Santos, C.R., Griffiths, B., Cully, M., Wu, M., Leevers, S., Griffiths, J.R., Chung, Y.L., and Schulze, A. (8). SREBP activity is regulated by mtorc and contributes to Akt-dependent cell growth. Cell Metab. 8, Rulifson, E.J., Kim, S.K., and Nusse, R. (). Ablation of insulin-producing neurons in flies: growth and diabetic phenotypes. Science 96, 8. Rutter, G.A. (). Nutrient-secretion coupling in the pancreatic islet betacell: recent advances. Mol. Aspects Med., Sancak, Y., Peterson, T.R., Shaul, Y.D., Lindquist, R.A., Thoreen, C.C., Bar-Peled, L., and Sabatini, D.M. (8). The Rag GTPases bind raptor and mediate amino acid signaling to mtorc. Science 3, Siegmund, T., and Korge, G. (). Innervation of the ring gland of Drosophila melanogaster. J. Comp. Neurol. 43, Tapon, N., Ito, N., Dickson, B.J., Treisman, J.E., and Hariharan, I.K. (). The Drosophila tuberous sclerosis complex gene homologs restrict cell growth and cell proliferation. Cell 5, Thissen, J.P., Ketelslegers, J.M., and Underwood, L.E. (994). Nutritional regulation of the insulin-like growth factors. Endocr. Rev. 5, 8. Tu, M.P., and Tatar, M. (3). Juvenile diet restriction and the aging and reproduction of adult Drosophila melanogaster. Aging Cell, Wang, S., Tulina, N., Carlin, D.L., and Rulifson, E.J. (7). The origin of islet-like cells in Drosophila identifies parallels to the vertebrate endocrine axis. Proc. Natl. Acad. Sci. USA 4, Cell Metabolism, 99 7, September, 9 ª9 Elsevier Inc. 7
Supplementary Information
 Supplementary Information Supplementary s Supplementary 1 All three types of foods suppress subsequent feeding in both sexes when the same food is used in the pre-feeding test feeding. (a) Adjusted pre-feeding
Supplementary Information Supplementary s Supplementary 1 All three types of foods suppress subsequent feeding in both sexes when the same food is used in the pre-feeding test feeding. (a) Adjusted pre-feeding
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
50mM D-Glucose. Percentage PI. L-Glucose
 Monica Dus, Minrong Ai, Greg S.B Suh. Taste-independent nutrient selection is mediated by a brain-specific Na+/solute cotransporter in Drosophila. Control + Phlorizin mm D-Glucose 1 2mM 1 L-Glucose Gr5a;Gr64a
Monica Dus, Minrong Ai, Greg S.B Suh. Taste-independent nutrient selection is mediated by a brain-specific Na+/solute cotransporter in Drosophila. Control + Phlorizin mm D-Glucose 1 2mM 1 L-Glucose Gr5a;Gr64a
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Drosophila Cytokine Unpaired 2 Regulates Physiological Homeostasis by Remotely Controlling Insulin Secretion
 Drosophila Cytokine Unpaired 2 Regulates Physiological Homeostasis by Remotely Controlling Insulin Secretion Akhila Rajan 1, * and Norbert Perrimon 1,2, * 1 Department of Genetics 2 Howard Hughes Medical
Drosophila Cytokine Unpaired 2 Regulates Physiological Homeostasis by Remotely Controlling Insulin Secretion Akhila Rajan 1, * and Norbert Perrimon 1,2, * 1 Department of Genetics 2 Howard Hughes Medical
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila
 SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila Ayako Kohyama-Koganeya, 1,2 Takuji Nabetani, 1 Masayuki Miura, 2,3 Yoshio Hirabayashi
SUPPLEMENTARY INFORMATION Glucosylceramide synthase (GlcT-1) in the fat body controls energy metabolism in Drosophila Ayako Kohyama-Koganeya, 1,2 Takuji Nabetani, 1 Masayuki Miura, 2,3 Yoshio Hirabayashi
Supplementary Figures
 J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Head of College Scholars List Scheme. Summer Studentship Report Form
 Head of College Scholars List Scheme Summer Studentship 2019 Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the
Head of College Scholars List Scheme Summer Studentship 2019 Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Table S1. Gal4 Driver Lines in This Study, Related to Figures 1, 2, 3, 4, 5, 6, S1, S2, S3, S4, S5, and S6
 Table S1. Gal4 Driver Lines in This Study, Related to s 1, 2, 3, 4, 5, 6, S1, S2, S3, S4, S5, and S6 Genotype Abbreviation Expression patterns Ubi-Gal4 Ubi Ubiquitous dome-gal4;elav-gal80 dome MZ Progenitor-specific
Table S1. Gal4 Driver Lines in This Study, Related to s 1, 2, 3, 4, 5, 6, S1, S2, S3, S4, S5, and S6 Genotype Abbreviation Expression patterns Ubi-Gal4 Ubi Ubiquitous dome-gal4;elav-gal80 dome MZ Progenitor-specific
supplementary information
 Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
100 mm Sucrose. +Berberine +Quinine
 8 mm Sucrose Probability (%) 7 6 5 4 3 Wild-type Gr32a / 2 +Caffeine +Berberine +Quinine +Denatonium Supplementary Figure 1: Detection of sucrose and bitter compounds is not affected in Gr32a / flies.
8 mm Sucrose Probability (%) 7 6 5 4 3 Wild-type Gr32a / 2 +Caffeine +Berberine +Quinine +Denatonium Supplementary Figure 1: Detection of sucrose and bitter compounds is not affected in Gr32a / flies.
Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld
 Journal of Biology BioMed Central Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld Address: Department of Genetics, Cell Biology, and Development,
Journal of Biology BioMed Central Minireview Shrinkage control: regulation of insulin-mediated growth by FOXO transcription factors Thomas P Neufeld Address: Department of Genetics, Cell Biology, and Development,
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation.
 ` Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation. (a) Overexpression of hlrrk2 in HeLa cells enhances total protein synthesis in [35S] methionine/cysteine incorporation assays.
` Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation. (a) Overexpression of hlrrk2 in HeLa cells enhances total protein synthesis in [35S] methionine/cysteine incorporation assays.
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
effects on organ development. a-f, Eye and wing discs with clones of ε j2b10 show no
 Supplementary Figure 1. Loss of function clones of 14-3-3 or 14-3-3 show no significant effects on organ development. a-f, Eye and wing discs with clones of 14-3-3ε j2b10 show no obvious defects in Elav
Supplementary Figure 1. Loss of function clones of 14-3-3 or 14-3-3 show no significant effects on organ development. a-f, Eye and wing discs with clones of 14-3-3ε j2b10 show no obvious defects in Elav
Genetic analysis of insulin signaling in Drosophila
 156 Review Genetic analysis of insulin signaling in Drosophila Robert S. Garofalo Studies in the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans have revealed that components
156 Review Genetic analysis of insulin signaling in Drosophila Robert S. Garofalo Studies in the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans have revealed that components
Supplementary Fig. 1 V-ATPase depletion induces unique and robust phenotype in Drosophila fat body cells.
 Supplementary Fig. 1 V-ATPase depletion induces unique and robust phenotype in Drosophila fat body cells. a. Schematic of the V-ATPase proton pump macro-complex structure. The V1 complex is composed of
Supplementary Fig. 1 V-ATPase depletion induces unique and robust phenotype in Drosophila fat body cells. a. Schematic of the V-ATPase proton pump macro-complex structure. The V1 complex is composed of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Insulin Signaling, Lifespan and Stress Resistance Are Modulated by Metabotropic GABA Receptors on Insulin Producing Cells in the Brain of Drosophila
 Insulin Signaling, Lifespan and Stress Resistance Are Modulated by Metabotropic GABA Receptors on Insulin Producing Cells in the Brain of Drosophila Lina E. Enell., Neval Kapan., Jeannette A. E. Söderberg,
Insulin Signaling, Lifespan and Stress Resistance Are Modulated by Metabotropic GABA Receptors on Insulin Producing Cells in the Brain of Drosophila Lina E. Enell., Neval Kapan., Jeannette A. E. Söderberg,
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Ch45: Endocrine System
 Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = coordinate and control the body with hormones to maintain homeostasis Works with
Ch45: Endocrine System Endocrine System Homeostasis is the tendency to maintain a stable internal environment. Function = coordinate and control the body with hormones to maintain homeostasis Works with
Basics of Signal Transduction. Ebaa M Alzayadneh, PhD
 Basics of Signal Transduction Ebaa M Alzayadneh, PhD What is signal transduction? Cell signaling The science of understanding how individual cells sense their environments and respond to stimuli... how
Basics of Signal Transduction Ebaa M Alzayadneh, PhD What is signal transduction? Cell signaling The science of understanding how individual cells sense their environments and respond to stimuli... how
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Endocrine System Hormones (Ch. 45)
 Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3
 Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Glucose Homeostasis. Liver. Glucose. Muscle, Fat. Pancreatic Islet. Glucose utilization. Glucose production, storage Insulin Glucagon
 Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
Supplementary Figure 1. Procedures to independently control fly hunger and thirst states.
 Supplementary Figure 1 Procedures to independently control fly hunger and thirst states. (a) Protocol to produce exclusively hungry or thirsty flies for 6 h water memory retrieval. (b) Consumption assays
Supplementary Figure 1 Procedures to independently control fly hunger and thirst states. (a) Protocol to produce exclusively hungry or thirsty flies for 6 h water memory retrieval. (b) Consumption assays
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Endocrine Notes Mrs. Laux AP Biology I. Endocrine System consists of endocrine glands (ductless), cells, tissues secrete hormones
 I. Endocrine System consists of endocrine glands (ductless), cells, tissues secrete hormones regulates metabolism, fluid balance, growth, reproduction A. Hormones 1. chemical signals-cell to cell communication
I. Endocrine System consists of endocrine glands (ductless), cells, tissues secrete hormones regulates metabolism, fluid balance, growth, reproduction A. Hormones 1. chemical signals-cell to cell communication
Nutritional Requirement for Protein Production from Trehalose Synthesis in the Male Accessory Gland of Tribolium castaneum
 Kaleidoscope Volume 11 Article 29 July 2014 Nutritional Requirement for Protein Production from Trehalose Synthesis in the Male Accessory Gland of Tribolium castaneum Ashlee Anciro Follow this and additional
Kaleidoscope Volume 11 Article 29 July 2014 Nutritional Requirement for Protein Production from Trehalose Synthesis in the Male Accessory Gland of Tribolium castaneum Ashlee Anciro Follow this and additional
Functional studies of Drosophila zinc transporters reveal the mechanism for dietary zinc absorption and regulation
 Qin et al. BMC Biology 2013, 11:101 RESEARCH ARTICLE Open Access Functional studies of Drosophila zinc transporters reveal the mechanism for dietary zinc absorption and regulation Qiuhong Qin, Xiaoxi Wang
Qin et al. BMC Biology 2013, 11:101 RESEARCH ARTICLE Open Access Functional studies of Drosophila zinc transporters reveal the mechanism for dietary zinc absorption and regulation Qiuhong Qin, Xiaoxi Wang
Endocrine System Hormones
 Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation metabolism
Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation metabolism
Endocrine System Hormones. AP Biology
 Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? u chemical messages from one body part to another u communication needed to coordinate whole body u daily homeostasis & regulation
Endocrine System Hormones 2007-2008 Regulation Why are hormones needed? u chemical messages from one body part to another u communication needed to coordinate whole body u daily homeostasis & regulation
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h)
 Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets
 Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Supplementary Information for
 Supplementary Information for Acid-Resistant Mesoporous Metal Organic Framework Toward Oral Insulin Delivery: Protein Encapsulation, Protection & Release Yijing Chen, Peng Li, Justin A. Modica, Riki J.
Supplementary Information for Acid-Resistant Mesoporous Metal Organic Framework Toward Oral Insulin Delivery: Protein Encapsulation, Protection & Release Yijing Chen, Peng Li, Justin A. Modica, Riki J.
Xenoestrogen-induced Regulation of EZH2 and Histone Methylation via Non-Genomic Estrogen
 Xenoestrogen-induced Regulation of EZH2 and Histone Methylation via Non-Genomic Estrogen Receptor Signaling to PI3K/AKT Tiffany G. Bredfeldt, Kristen L. Greathouse, Stephen H. Safe, Mien-Chie Hung, Mark
Xenoestrogen-induced Regulation of EZH2 and Histone Methylation via Non-Genomic Estrogen Receptor Signaling to PI3K/AKT Tiffany G. Bredfeldt, Kristen L. Greathouse, Stephen H. Safe, Mien-Chie Hung, Mark
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used for PCR and qpcr Primer Name
 Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
were isolated from the freshly drawn blood of healthy donors and ACS patients using the
 Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
SLOB, a SLOWPOKE channel binding protein, regulates insulin pathway signaling and metabolism in Drosophila.
 Thomas Jefferson University Jefferson Digital Commons Department of Neuroscience Sidney Kimmel Medical College 8-5-2011 SLOB, a SLOWPOKE channel binding protein, regulates insulin pathway signaling and
Thomas Jefferson University Jefferson Digital Commons Department of Neuroscience Sidney Kimmel Medical College 8-5-2011 SLOB, a SLOWPOKE channel binding protein, regulates insulin pathway signaling and
Molecular mechanisms of metabolic regulation by insulin in Drosophila
 Biochem. J. (2010) 425, 13 26 (Printed in Great Britain) doi:10.1042/bj20091181 13 REVIEW ARTICLE Molecular mechanisms of metabolic regulation by insulin in Drosophila Aurelio A. TELEMAN 1 German Cancer
Biochem. J. (2010) 425, 13 26 (Printed in Great Britain) doi:10.1042/bj20091181 13 REVIEW ARTICLE Molecular mechanisms of metabolic regulation by insulin in Drosophila Aurelio A. TELEMAN 1 German Cancer
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
Chapter 20 Endocrine System
 Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
AMPK Phosphorylation Assay Kit
 AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Requires Signaling though Akt2 Independent of the. Transcription Factors FoxA2, FoxO1, and SREBP1c
 Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
Cell Communication and Cell Signaling
 Cell Communication and Cell Signaling Why is cell signaling important? Why is cell signaling important? Allows cells to communicate and coordinate functions/activities of the organism Usually involves
Cell Communication and Cell Signaling Why is cell signaling important? Why is cell signaling important? Allows cells to communicate and coordinate functions/activities of the organism Usually involves
Development Supplementary information
 Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
General Principles of Endocrine Physiology
 General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or
 Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
18. PANCREATIC FUNCTION AND METABOLISM. Pancreatic secretions ISLETS OF LANGERHANS. Insulin
 18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
Midgut-Derived Activin Regulates Glucagon-like Action in the Fat Body and Glycemic Control
 Article Midgut-Derived Activin Regulates Glucagon-like Action in the Fat Body and Glycemic Control Graphical Abstract Authors Wei Song, Daojun Cheng, Shangyu Hong,..., Michael B. O Connor, Pavlos Pissios,
Article Midgut-Derived Activin Regulates Glucagon-like Action in the Fat Body and Glycemic Control Graphical Abstract Authors Wei Song, Daojun Cheng, Shangyu Hong,..., Michael B. O Connor, Pavlos Pissios,
Hormonal regulation of. Physiology Department Medical School, University of Sumatera Utara
 Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
Chapter 26. Hormones and the Endocrine System. Lecture by Edward J. Zalisko
 Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
Campbell's Biology: Concepts and Connections, 7e (Reece et al.) Chapter 26 Hormones and the Endocrine System Multiple-Choice Questions
 Campbell's Biology: Concepts and Connections, 7e (Reece et al.) Chapter 26 Hormones and the Endocrine System 26.1 Multiple-Choice Questions 1) Hormones are chemicals produced by the endocrine system that
Campbell's Biology: Concepts and Connections, 7e (Reece et al.) Chapter 26 Hormones and the Endocrine System 26.1 Multiple-Choice Questions 1) Hormones are chemicals produced by the endocrine system that
Endocrine secretion cells secrete substances into the extracellular fluid
 Animal Hormones Concept 30.1 Hormones Are Chemical Messengers Endocrine secretion cells secrete substances into the extracellular fluid Exocrine secretion cells secrete substances into a duct or a body
Animal Hormones Concept 30.1 Hormones Are Chemical Messengers Endocrine secretion cells secrete substances into the extracellular fluid Exocrine secretion cells secrete substances into a duct or a body
Homeostasis. Endocrine System Nervous System
 Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Table S1. Sequence of human and mouse primers used for RT-qPCR measurements.
 Table S1. Sequence of human and mouse primers used for RT-qPCR measurements. Ca9, carbonic anhydrase IX; Ndrg1, N-myc downstream regulated gene 1; L28, ribosomal protein L28; Hif1a, hypoxia inducible factor
Table S1. Sequence of human and mouse primers used for RT-qPCR measurements. Ca9, carbonic anhydrase IX; Ndrg1, N-myc downstream regulated gene 1; L28, ribosomal protein L28; Hif1a, hypoxia inducible factor
Diagnosis and prevention of metabolic diseases in Drosophila melanogaster
 Southern Methodist University SMU Scholar Engaged Learning Collection Engaged Learning Spring 2013 Diagnosis and prevention of metabolic diseases in Drosophila melanogaster Claire Marie Coogan Southern
Southern Methodist University SMU Scholar Engaged Learning Collection Engaged Learning Spring 2013 Diagnosis and prevention of metabolic diseases in Drosophila melanogaster Claire Marie Coogan Southern
The Endocrine System. PowerPoint Lecture Presentations prepared by Jason LaPres. Lone Star College North Harris
 18 The Endocrine System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris NOTE: Presentations extensively modified for use in MCB 244 & 246 at the University of Illinois
18 The Endocrine System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris NOTE: Presentations extensively modified for use in MCB 244 & 246 at the University of Illinois
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original
 Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Chapter 16: Endocrine System 1
 Ch 16 Endocrine System Bi 233 Endocrine system Endocrine System: Overview Body s second great controlling system Influences metabolic activities of cells by means of hormones Slow signaling Endocrine glands
Ch 16 Endocrine System Bi 233 Endocrine system Endocrine System: Overview Body s second great controlling system Influences metabolic activities of cells by means of hormones Slow signaling Endocrine glands
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/430/ra57/dc1 Supplementary Materials for The 4E-BP eif4e axis promotes rapamycinsensitive growth and proliferation in lymphocytes Lomon So, Jongdae Lee, Miguel
www.sciencesignaling.org/cgi/content/full/9/430/ra57/dc1 Supplementary Materials for The 4E-BP eif4e axis promotes rapamycinsensitive growth and proliferation in lymphocytes Lomon So, Jongdae Lee, Miguel
A post-ingestive amino acid sensor promotes food consumption in Drosophila
 www.nature.com/cr www.cell-research.com ARTICLE OPEN A post-ingestive amino acid sensor promotes food consumption in Drosophila Zhe Yang 1,2, Rui Huang 3,4, Xin Fu 5,6,7, Gaohang Wang 1,2, Wei Qi 1,2,
www.nature.com/cr www.cell-research.com ARTICLE OPEN A post-ingestive amino acid sensor promotes food consumption in Drosophila Zhe Yang 1,2, Rui Huang 3,4, Xin Fu 5,6,7, Gaohang Wang 1,2, Wei Qi 1,2,
Testosterone and other male hormones seem to be related to aggressive behavior in some species
 Testosterone and Male Aggression Testosterone and other male hormones seem to be related to aggressive behavior in some species In the fish species Oreochromis mossambicus, elevated levels have been found
Testosterone and Male Aggression Testosterone and other male hormones seem to be related to aggressive behavior in some species In the fish species Oreochromis mossambicus, elevated levels have been found
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment
 Supplementary Information Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment Robin A. Kimmel, Stefan Dobler, Nicole Schmitner, Tanja Walsen, Julia
Supplementary Information Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment Robin A. Kimmel, Stefan Dobler, Nicole Schmitner, Tanja Walsen, Julia
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION
 Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
SUPPLEMENTARY DATA. Supplementary Table 2. Antibodies used for Immunofluoresence. Supplementary Table 3. Real-time PCR primer sequences.
 Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Regulation of Metabolism
 Regulation of Metabolism Pratt and Cornely Chapter 19 Regulation by Compartmentalization Form of reciprocal regulation Degradation vs biosynthesis Requires transporters 1 Specialization of organs Fuel
Regulation of Metabolism Pratt and Cornely Chapter 19 Regulation by Compartmentalization Form of reciprocal regulation Degradation vs biosynthesis Requires transporters 1 Specialization of organs Fuel
Hormones. Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6
 Hormones Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6 Tel. 030-8385-6920 (Sekret.) 030-8385-6922 (direkt) e-mail: vhaucke@chemie.fu-berlin.de http://userpage.chemie.fu-berlin.de/biochemie/aghaucke/teaching.html
Hormones Prof. Dr. Volker Haucke Institut für Chemie-Biochemie Takustrasse 6 Tel. 030-8385-6920 (Sekret.) 030-8385-6922 (direkt) e-mail: vhaucke@chemie.fu-berlin.de http://userpage.chemie.fu-berlin.de/biochemie/aghaucke/teaching.html
Human Leptin ELISA Kit
 Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
