Transport of the outer dynein arm complex to cilia requires a cytoplasmic protein Lrrc6
|
|
|
- Edwin Singleton
- 6 years ago
- Views:
Transcription
1 Genes to Cells Transport of the outer dynein arm complex to cilia requires a cytoplasmic protein Lrrc6 Yasuko Inaba 1, Kyosuke Shinohara 1a, Yanick Botilde 1, Ryo Nabeshima 1, Katsuyoshi Takaoka 1, Rieko Ajima 1b, Lynda Lamri 1, Hiroyuki Takeda 2, Yumiko Saga 3, Tetsuya Nakamura 1c and Hiroshi Hamada 1 * 1 Developmental Genetics Group, Graduate School of Frontier Biosciences, Osaka University, and CREST, Japan Science and Technology Corporation (JST), 1-3 Yamada-oka, Suita, Osaka , Japan 2 Department of Biological Sciences, Graduate School of Science, University of Tokyo, Hongo, Bunkyo-ku, Tokyo , Japan 3 Division of Mammalian Development, Genetic Strains Research Center, National Institute of Genetics, Yata 1111, Mishima, Shizuoka , Japan Lrrc6 encodes a cytoplasmic protein that is expressed specifically in cells with motile cilia including the node, trachea and testes of the mice. A mutation of Lrrc6 has been identified in human patients with primary ciliary dyskinesia (PCD). Mutant mice lacking Lrrc6 show typical PCD defects such as hydrocephalus and laterality defects. We found that in the absence of Lrrc6, the morphology of motile cilia remained normal, but their motility was completely lost. The arrangement of microtubules remained normal in Lrrc6 -/- mice, but the outer dynein arms (ODAs), the structures essential for the ciliary beating, were absent from the cilia. In the absence of Lrrc6, ODA proteins such as DNAH5, DNAH9 and IC2, which are assembled in the cytoplasm and transported to the ciliary axoneme, remained in the cytoplasm and were not transported to the ciliary axoneme. The IC2 IC1 interaction, which is the first step of ODA assembly, was normal in Lrrc6 / mice testes. Our results suggest that ODA proteins may be transported from the cytoplasm to the cilia by an Lrrc6- dependent mechanism. Introduction Cilia are hairlike organelles present in nearly all cell types. There are two types of mammalian cilia: motile and nonmotile cilia (also known as primary cilia). Motile cilia are found in the trachea, oviduct, sperm and brain. They have a characteristic structure with nine peripheral microtubule doublets arranged around a central pair of microtubules. In motile cilia, dynein complexes outer dynein arms (ODAs) and inner dynein arms (IDAs) are attached to the Communicated by: Hisato Kondoh *Correspondence: hamada@fbs.osaka-u.ac.jp a Present address: Institute of Engineering, Tokyo University of Agriculture and Technology, Koganei, Tokyo , Japan b Present address: National Institute of Genetics, Yata 1111, Mishima, Shizuoka , Japan c Present address: Department of Organismal Biology and Anatomy, University of Chicago, Chicago, IL 60637, USA peripheral microtubule doublets. However, primary cilia structurally lack the central pair of microtubules (9 + 0). However, some primary cilia with a arrangement such as the cilia present in the mouse embryonic node are exceptionally motile (Nonaka et al. 2002; Ishikawa and Marshall, 2011). The nodal cilia play a critical role in left right patterning (Hirokawa et al. 2009). Cilia mediate diverse functions in signaling and fluid dynamics. Nonmotile cilia are implicated in sensing environmental cues. They act as sensory antennae that receive signals from the outside of cells. Motile cilia are involved in the movement of extracellular fluids. In the respiratory tract, for example, motile cilia are responsible for mucociliary clearance of external pathogens. Defects in the structure and function of motile cilia result in primary ciliary dyskinesia (PCD, OMIM242650), a disorder characterized by recurrent respiratory infections and male infertility. 728 Genes to Cells (2016) 21, DOI: /gtc Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
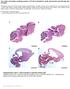
2 Transport of dynein to cilia Outer dynein arms (ODAs) and inner dynein arms (IDAs) are essential structures for cilia motility. ODAs hydrolyze ATP to power ciliary movement, whereas IDAs regulate the ciliary beating pattern. ODAs and IDAs are composed of many proteins including heavy (HC)-, light (LC)- and intermediate-chain (IC) proteins. ODA complexes are preassembled in the cytoplasm before being transported into cilia (Fowkes & Mitchell 1998). Assembly of ODA complexes has been extensively studied in Chlamydomonas. Cytoplasmic preassembly of ODA complexes has been found to require at least two steps. The first step involves the folding of globular dynein head domains that are required for HCs stability. The second step involves the formation of assembled complex between HCs and intermediate chains (ICs) (Mitchison et al. 2012). ODA assembly factors are known as dynein axonemal assembly factors (DNAAFs) and currently include four proteins: ODA7 (also known as DNAAF1 or LRRC50) (Loges et al. 2009), PF13 (also known as DNAAF2 or Ktu) (Omran et al. 2008), PF22 (also known as DNAAF3) (Mitchison et al. 2012) and DNAAF4 (also known as DYX1C1) (Tarkar et al. 2013). DNAAF1 4 work together with heat-shock protein (HSP)-based molecular chaperone to direct the proper folding of ODA subunits. Additional proteins including ZMYND10, HEATR2, C21orf59, SPAG1 and Pih1d3 may also be involved in ODA assembly. Like DNAAF1 4, these proteins are localized in the cytoplasm and mutations in each of these genes leads to ODA deficiency (Austin-Tse et al. 2013; Knowles et al. 2013; Zariwala et al. 2013; Diggle et al. 2014; Dong et al. 2014). However, the precise function of these proteins is not well understood. A mutagenesis screen in zebrafish identified Lrrc6/Seahorse as a gene responsible for cystic kidney (Sun et al. 2004). Lrrc6 encodes a coiled-coil domain containing protein. Lrrc6 mutant zebrafish showed aberrant left right (LR) patterning (Kishimoto et al. 2008), suggesting that Lrrc6 is involved in the regulation of cilia-mediated processes. Recent report (Zariwala et al. 2013) showed that Lrrc6 interacts with reptin in zebrafish and Zmynd10 in mice. Reptin is involved in epigenetic and transcriptional regulation (Zhao et al. 2013), whereas Zmynd10 is one of the dynein arm assembly factor (Zariwala et al. 2013). However, the precise function of Lrrc6 remains to be elucidated. To understand the function of Lrrc6, we generated and analyzed mutant mouse lacking Lrrc6. Results Lrrc6 is specifically expressed in cells with motile cilia We first examined Lrrc6 expression using a BAC transgene (Lrrc6-lacZ), in which IRES-lacZ was inserted after the stop codon of an Lrrc6 BAC clone. At the embryonic day 8.0 (E8.0), Lrrc6 was expressed at the node (Fig. 1A,B), especially in pit cells, which are located at the central region of the node and possess motile cilia (Fig. 1C). At later stages, Lrrc6 expression was detected in the notochord at E9.0 (Fig. 1D), in the hindbrain, the branchial arches and neural tube at E11.0 (Fig. 1E), in the hindbrain at E12.0 (Fig. 1F) and in the forebrain at E13.0 (Fig. 1G). In adult, Lrrc6 was found to be expressed in epithelial cells of the trachea (Fig. 1H), testis (Fig. 1I) and ependymal cells of the cerebral ventricles (Fig. 1J), all of which are known to possess motile cilia. Lrrc6 expression was not detected in other cell types, including those with immotile cilia. In all, Lrrc6 expression was mainly expressed in cells with motile cilia. Lrrc6 is required for the motility of various motile cilia To investigate Lrrc6 function, we generated an Lrrc6 mutant mouse (Fig. S1 in Supporting Information) Figure 1 Expression of Lrrc6 in cells that have motile cilia. (A-C) E8.0 transgenic embryos harboring Lrrc6-lacZ were stained with X-gal. (A) Lateral view and (B) posterior view. The arrowhead indicates the node in (A). (C) Transverse section of the node. The expression of Lrrc6-lacZ is highly specific to node pit cells. (D J) Lrrc6-lacZ expression at later stages. Lrrc6-lacZ expression was detected in the notochord at E9.0 (D) (indicated by the arrowhead), in the hindbrain (indicated by the black arrowhead), branchial arches (indicated by the blue arrowhead) and neural tube (indicated by the arrow) at E11.0 (E), in the hindbrain (indicated by the arrowhead) at E12.0 (F), and in the fore brain (indicated by the arrowhead) at E13.0 (G). X-gal staining of trachea (H), testis (I) and brain (J) of adult Lrrc6-lacZ mouse. (H, I) Pictures of whole-mount staining are shown in the left panels and of section staining are shown in the right panel. Lrrc6-lacZ expression is observed in multiciliated epithelial cells of the trachea (H) and in seminiferous tube of testis (I). (J) Coronal sections of adult brains showing expression of Lrrc6-lacZ in the lateral ventricle (indicated by the arrowhead) Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
3 Y Inaba et al. (A) (B) (C) (F) (G) (D) (E) (H) (I) (J) 730 Genes to Cells (2016) 21, Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
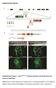
4 Transport of dynein to cilia and examined its phenotype. Lrrc6 protein was absent in the trachea of Lrrc6 / mice (Fig. S1E in Supporting Information), suggesting that Lrrc6 is a null allele. Upon intercrosses of Lrrc6 +/ mice, Lrrc6 / embryos were detected at the expected frequency at E8.0. However, Lrrc6 / mice were found at a lower frequency at birth, suggesting that a significant portion (approximately 60%) of Lrrc6 / mice die in utero. Mutant newborn mice showed laterality defects (6/6 newborn mice) with variable severities. Thus, some of them (2/6) showed reversal of the heart apex, stomach, aortic arch and azygos vein, whereas others showed less severe defects with abnormal liver lobation (Table S1 in Supporting Information). Surviving mutant mice (6/6 mice) developed hydrocephalus 3 weeks after the birth, and all Lrrc6 / mice eventually died within 5 weeks of birth. Laterality defects in the Lrrc6 / mice prompted us to examine Nodal expression at E8.0. Nodal expression was found exclusively on the left side of the lateral plate in the wild-type (WT) embryos. In the Lrrc6 / embryos, however, Nodal expression was randomized so that it was found either on the left (3/9 embryos), right (1/9 embryos) or both (2/9 embryos) side(s) or was absent (3/9 embryos) (Fig. 2J). These defects, typical of patients with PCD and PCD animals, suggest that ciliary motility may be impaired in the Lrrc6 / mice. This was indeed the case. Node cilia, which rotate into the clockwise direction in the WT embryos (Movie S1 in Supporting Information), were completely immotile in the Lrrc6 / mice (Fig. 3A; Movie S2 in Supporting Information). The cilia of trachea epithelial cells and brain ependymal cells, which show planar beating in WT mice (Movie S3 and S5 in Supporting Information), also lost motility in the Lrrc6 / mice (Fig. 3A; Movie S4 and S6 in Supporting Information). Finally, Lrrc6 was specifically deleted from male germ cells using Nanos3-Cre (Suzuki et al. 2008). Sperm obtained from the testis-specific Lrrc6 knockout (Lrrc6 f/f ;Nanos3 Cre/+ ) mouse were completely immotile (Fig. 3A; Movie S8 in Supporting Information). Thus, all types of the motile cilia that were examined lost motility in the absence of Lrrc6. Lrrc6 is essential for transporting the ODA complex from the cytoplasm to the cilia To understand why ciliary motility was lost in the absence of Lrrc6, we examined the ultrastructure of trachea cilia by transmission electron microscopy (TEM). In the WT trachea cilia, their inner structure showed microtubules containing nine peripheral doublet microtubules and two central microtubules (Fig. 3B). Peripheral microtubules contained outer and inner dynein arms (Fig. 3B), which are essential for ciliary motility. In the trachea cilia of the Lrrc6 / mice, the configuration of the microtubules remained normal, but ODAs were absent (Fig. 3B). We next examined the subcellular distribution of ODA proteins in airway epithelial cells by immunostaining. In the WT mouse, DNAH5 and DNAH9 proteins were specifically expressed in the multiciliated cells and, these proteins were localized exclusively to the ciliary axoneme (Fig. 4A,B). In the Lrrc6 / mice, however, both DNAH5 and DNAH9 were absent from the cilia but were detected in the cytoplasm as punctuated spots (Fig. 4A,B). IC2 protein was also expressed specifically in the multiciliated cells, and was localized predominantly in the ciliary axoneme in the WT mice (Fig. 4C). In the Lrrc6 / mice, however, IC2 was absent from the cilia and instead was diffusely distributed throughout the cytoplasm (Fig. 4C). These results suggest that ODA proteins are not transported to the ciliary axoneme in the absence of Lrrc6. Lrrc6 is a cytoplasmic protein, but assembly of the IC1 IC2 complex does not require Lrrc6 To investigate subcellular localization of Lrrc6 protein in cells with motile cilia, we generated a BAC Figure 2 Primary ciliary dyskinesia in Lrrc6 / mice. (A-D) Laterality of visceral organs in Lrrc6 +/+ (A, B) and Lrrc6 / (C, D) mice at P0. The position of the heart apex in Lrrc6 +/+ mouse is located on the left (A), but it is reversed in Lrrc6 / mice (C). The stomach is located in the left side in Lrrc6 +/+ mouse (B), but it is on the right side in the Lrrc6 / mouse (D, arrow). (E, F) Lrrc6 +/+ (E) and Lrrc6 / (F) mice at P21. Compared to Lrrc6 +/+ mouse, Lrrc6 / mouse shows domed crown head (indicated by the arrow in F). (G, H) Dorsal (G) and coronal (H) views of the brain of Lrrc6 +/+ and Lrrc6 / mice. Lrrc6 / brains show significant deformation with collapsed cortices. (I) Whole-mount in situ hybridization of Nodal in E8.0 Lrrc6 +/+ and Lrrc6 / embryos. The black arrows indicate left lateral plate mesoderm (LPM). The blue arrows indicate aberrant expression in right LPM. (J) The numbers of embryos showing each pattern of gene expression are summarized. L, left; R, right Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
5 Y Inaba et al. (A) (B) (E) (C) (D) (F) (G) (H) (I) (J) 732 Genes to Cells (2016) 21, Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
6 Transport of dynein to cilia (A) Node Brain Sperm Lrrc6 / Lrrc6 +/+ Trachea Lrrc6 / Lrrc6 +/+ (B) Lrrc6 +/+ Lrrc6 / Figure 3 Motile cilia lose motility in the absence of Lrrc6. (A) Movement of cilia in the node, brain ependymal cells and trachea of Lrrc6 +/+ and Lrrc6 / mice was traced with TEMA software. Sperm motility was examined with Lrrc6 +/+ and Lrrc6 f/f ; Nanos3 Cre/+ mice. Colored lines (red, light blue and blue) indicate the movement of individual cilia. (B) Transmission electron microscope (TEM) images of tracheal cilia from Lrrc6 +/+ and Lrrc6 / mice. ODA in tracheal cilia from the Lrrc6 +/+ mouse is indicated by the red arrowhead, whereas those from the Lrrc6 / mouse lacked ODAs (indicated by the white arrowhead). Scale bars: 100 nm Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
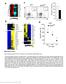
7 Y Inaba et al. transgenic mouse that expressed an Lrrc6::Venus fusion protein (Venus is a version of yellow fluorescent protein) (Fig. S2 in Supporting Information). This fusion protein was fully functional because it was able to rescue lethality of Lrrc6 / mouse (Fig. S2 in Supporting Information). In the node cells of E8.0 embryos harboring this transgene, the Lrrc6::Venus protein was localized only in the cytoplasm, whereas it was not detected in the axoneme (Ift46 + region) of the node cilia (Fig. 5A,B). Similarly, the Lrrc6::Venus protein was localized to the cytoplasm of airway epithelial cells, in particular to the apical side of the cytoplasm (Fig. 5C,D). However, the Lrrc6::Venus protein was not detected in the ciliary axoneme, which was positive for acetylated tubulin (Fig. 5C,D). Finally, we also examined the Lrrc6::Venus protein in the testes. The Lrrc6::Venus protein was expressed in germ cells ranging from primary spermatocytes and spermatids, and its subcellular localization was again restricted to the cytoplasm (Fig. 5E,F). Cytoplasmic localization of the Lrrc6 protein and the absence of ODA in the axoneme of Lrrc6 / motile cilia suggest that this protein acts in the cytoplasm, and may be involved in a process such as cytoplasmic preassembly of the ODAs or transport of ODA complex from the cytoplasm to the ciliary axoneme. ODAs are assembled in the cytoplasm through several steps by DNAAFs. The IC1 IC2 complex is formed first followed by HC IC complex formation. In the testes lacking Lrrc6 (testes from the Lrrc6 f/ ; Nanos3 Cre/+ mice), the level of IC2 was reduced (Fig. 6). However, interaction between IC1 and IC2 remained normal (Fig. 6), suggesting that the IC1 IC2 complex formation, the first step of ODA assembly, does not require Lrrc6. Discussion ODA proteins are preassembled in the cytoplasm and are subsequently transported to the ciliary axoneme. Several proteins, collectively called DNAAFs, are required for cytoplasmic preassembly of the ODA complex. The cytoplasmic assembly of ODA is achieved through multiple steps. The first step is the folding of the head domain of the dynein heavy chain by DNAAF1/Lrrc50, DNAAF2/PF13 and Hsp70. This is followed by formation of a stable IC1 IC2 complex, which involves Pih1d3 in sperm (Dong et al. 2014). Finally, the IC HC (heavy chain) complex is formed via DNAAF3. Whereas the mechanism of ODA preassembly is becoming clear, it is totally unknown how assembled ODA complex is transported from the cytoplasm to the cilia. Although our current data do not clarify whether Lrrc6 is involved in the preassembly or transport, several lines of evidence suggest that Lrrc6 is more likely involved in the transport of the preassembled ODA complex to the axoneme. First, the interaction between IC1 and IC2 remained normal in the Lrrc6 / testes, suggesting that the early step of preassembly does not require Lrrc6. Furthermore, Lrrc6::Venus expression in the testes was found mainly between the spermatocyte and spermatid stages, which is later than the stage for expression of Pih1d3, one of the cytoplasmic DNAAF proteins required for the stability of the IC1 IC2 complex in the testes (Dong et al. 2014). Second, ODA proteins are not transported if cytoplasmic preassembly is impaired. Thus, in airway epithelial cells in human patients with PCD lacking DNAAF, ODA proteins are absent in the ciliary axoneme (Loges et al. 2009; Mitchison et al. 2012; Panizzi et al. 2012; Tarkar et al. 2013). However, these proteins are often absent in the cytoplasm, probably because unassembled ODA proteins undergo rapid degradation. For example, DNAH5 and DNAH9 are absent when DNAAF1 is mutated (Loges et al. 2009); and DNAH9 is absent in respiratory cells with DNAAF4 mutations (Tarkar et al. 2013). In Lrrc6 / trachea, however, DNAH5, DNAH9 and IC2 proteins remained in the cytoplasm. In particular, there was no sign of IC2 degradation in the Lrrc6 mutant cells, as the level of cytoplasmic IC2 protein was maintained or even increased in the Lrrc6 / airway cells (Fig. 4C). However, a low level of DNAH5 protein was detected in the cytoplasm of airway cells from some patients with PCD, such as a patient with a mutation in DNAAF4 (Tarkar et al. 2013), suggesting that the fate of ODA proteins in patients with PCD may depend on individual mutations. In all, our data suggest that Lrrc6 may be required for the transport of the ODA complex to the axoneme, but Lrrc6 may also be involved in cytoplasmic preassembly of ODA proteins. A significant portion of Lrrc6 / mice die before birth (between E13.5 and E18.5), but the exact cause of embryonic lethality is unknown. Randomization of LR asymmetry at E8.0 would account for the death at the newborn stage, but not for the embryonic lethality. Interestingly, whereas Lrrc6 was expressed in cells with motile cilia, it was also expressed in the branchial arch of E11.0 embryos (Fig. 1E), which do not have motile cilia. In this regard, Lrrc6 may have an additional function 734 Genes to Cells (2016) 21, Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
8 Transport of dynein to cilia (A) (B) (C) 2016 Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
9 Y Inaba et al. Figure 4 Cilia of Lrrc6 / trachea epithelial cells lack ODA proteins. (A-C) Immunofluorescence analysis of trachea epithelial cells at P14 in Lrrc6 +/+ and Lrrc6 / mice. Acetylated a-tubulin (red); DAPI (blue). ODA heavy-chain proteins DNAH5 (A) and DNAH9 (B) are localized in cilia of the Lrrc6 +/+ airway cells, but both proteins were absent from cilia of Lrrc6 / airway cells. Note that both proteins remained localized in the cytoplasm as punctuated spots (indicated by arrows) in the Lrrc6 / airway cells. ODA intermediate-chain protein DNAIC2 (C) was mainly localized in the cilia of Lrrc6 +/+ airway cells, whereas DNAIC2 was absent from cilia but remained in the cytoplasm of Lrrc6 / airway cells. Note that the level of cytoplasmic DNAIC2 was increased in some of the Lrrc6 / airway cells (white arrowheads). Scale bars: 5 lm. unrelated to ciliary motility. For example, Lrrc6 may be involved in intracellular transport of some other proteins in nonciliated cell. In conclusion, whereas our data suggest that ODA may be transported to the cilia by an Lrrc6-dependent mechanism, the precise mechanism of ODA transport from the cytoplasm to cilia and the exact role of Lrrc6 require further investigation. Experimental procedures Mice The targeting vector for Lrrc6 was constructed with bacterial artificial chromosome (BAC) clone RP24-333G17. A single loxp site was inserted 200 bp upstream of exon 1 of Lrrc6 gene. The Frt-Pgk-neo-Frt-loxP cassette was inserted 100 bp downstream of the exon 1. The construct was introduced into G4 mouse embryonic stem (ES) cells (C57BL/6 and 129/Sv hybrid). After selection of transformed clones with G418, homologous recombinants were detected by Southern blot hybridization with a specific two probes, external and internal probes. Chimeric mice were generated by aggregation of ES clones with ICR morulas. Null allele was generated by cross with CAG-Cre or CAG-Flp transgenic mice, to remove Lrrc6 exon 1 and the neo cassette. PCR primers for the Lrrc6 + allele were Lrrc6-WT-F (AGCTCAGACATAAGCTCCCAAG) and Lrrc6-WT-R (GACAGCACTGGCAATATCTCTG), whereas PCR primers for Lrrc6 allele were Lrrc6-Mut-F (TGCCAGTGGGTGTAAGTAAGG) and Lrrc6-Mut-R (CACATCGCTTTGAGGACAGTT). To generate Lrrc6:: Venus BAC transgene, the Venus-coding sequence was inserted before the stop codon of Lrrc6 in BAC clone RP24-333G17. To generate Lrrc6-lacZ transgene, the IRES-lacZ cassette was inserted after the stop codon of Lrrc6 in the same BAC clone. X- gal (5-bromo-4-chloro-3-indolyl-b-D-galactopyranoside) staining was carried out as described previously (Saijoh et al. 1999). Serial sections (10 lm) of trachea, testis and brain were prepared after X-gal staining. All animal experiments were approved by the animal experiment committee of Osaka University. Whole-mount in situ hybridization Whole-mount in situ hybridization was carried out according to standard procedures specific with digoxigenin-labeled riboprobes for Nodal. Antibodies Polyclonal antibodies that recognize a COOH-terminal sequence (QPRDEPSEEDPDFEDNPEV) of mouse Lrrc6 were recovered from a corresponding antigen injected rabbit and purified by affinity chromatography (Immuno-Biological Laboratories). Additional antibodies used in this study are rabbit anti-ift46 (Abcam, ab122422), rabbit anti-gfp (MBL, 598), rat anti-gfp (Nacalai, ), mouse anti-acetylated tubulin (SIGMA, T6793) and mouse anti-ic2 (Abnova, H MO1). DNAH5 and DNAH9 antibodies have been described previously (Matsuo et al. 2013). Immunofluorescence staining E8.0 embryos were collected and fixed in 4% PFA for 1 h at 4 C. The embryos were then dehydrated in methanol, blocked with blocking solution for 1 h at 4 C, incubated overnight at 4 C with primary antibodies diluted in the blocking solution, washed for 6 h with phosphate-buffered saline (PBS) containing 0.1% Triton X-100, incubated overnight at 4 C with secondary antibodies diluted in the blocking solution and washed. Node of stained embryos was excised and placed to slide glasses with silicone rubber spacers, covered with cover glasses and observed with an Olympus FV1000 confocal microscope. Trachea were collected and cut longitudinally in ice-cold PBS and fixed in 4% PFA for 1 h at 4 C. They were then permeabilized with 1% Triton X-100 in PBS for 10 min at 4 C, blocked with blocking solution 1 h at 4 C, and then incubated overnight at 4 C with primary antibodies diluted in the blocking solution, washed with PBS containing 0.1% Triton X-100, incubated overnight at 4 C with secondary antibodies diluted in the blocking solution and washed. The samples were placed on a slide glasses with silicone rubber spacers, covered with cover glasses and observed with an Olympus FV1000 confocal microscope. Testis were collected and fixed in 4% PFA for 24 h at 4 C and then mounted in OCT compound and sectioned into 15-lm slices. They were treated in the same way as tracheal samples. Western blotting Testis and trachea samples from adult mice were lysed in the lysis buffer (50 mm Tris-HCl, 150 mm NaCl, 5 mm MgCl2, 0.2% NP-40 and 19 protease inhibitor), homogenized using Dounce Tissue Grinder (WHEATON ) and incubated 736 Genes to Cells (2016) 21, Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
10 Transport of dynein to cilia (A) (B) (C) (D) (E) (G) (F) (H) 2016 Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
11 Y Inaba et al. Figure 5 Lrrc6 is a cytoplasmic protein. (A, B) The node from Lrrc6::Venus Tg + or Tg mouse was immunostained for Ift46 (red) and GFP (green). The node was also stained with DAPI (blue). (C-H) The trachea (C, D) and testis (E-H) from Lrrc6::Venus Tg + and Tg mice were stained for acetylated tubulin (red), GFP (green) and DAPI (blue). The boxed regions (E, F) are shown at higher magnification (G, H). Note that GFP signals were detected in the cytoplasm in all cases examined: node, epithelial cells of trachea and seminiferous tube of testis from Lrrc6::Venus Tg + mouse. Scale bars: 10 lm (A D) and 50 lm (E H). were added to 20 ll of Dynabeads protein G diluted in 1 ml IP buffer and incubated with rotation for 3 h at 4 C to make antibody-coupled beads. In the meantime, protein samples from testis extracts were also incubated with Dynabeads to reduce nonspecific binding. After 3 h, protein samples were incubated with anti-ic2/mouse IgG beads for 3 h at 4 C and then washed briefly two times with IP buffer and three times for 10 min each with rotation at 4 C. Immunoprecipitates were eluted using 50 ll of19 SDS buffer after 30 min of incubation at 37 C water bath. Transmission electron microscopy (TEM) Figure 6 IC1 and IC2 are normally assembled in Lrrc6 f/ ; Nanos3 Cre/+ testis. Testis extracts were prepared from adult Lrrc6 +/+, Lrrc6 f/ and Lrrc6 f/ ;Nanos3 Cre/+ mice. The extracts were subjected to immunoprecipitation with either mouse anti-ic2 antibody or control mouse immunoglobulin G (migg). The immunoprecipitated samples were subjected to Western blot analysis using an anti-ic1 antibody or an anti-ic2 antibody. Testis extracts (input) were subjected to Western blot analysis using anti-ic2, IC1, Lrrc6 and Hsp70 (loading control) antibodies. on ice for 30 min. Lysates were centrifuged at g for 10 min at 4 C, and the supernatant was used for Western blotting and immunoprecipitation assays. Total protein concentration was measured using BCA Protein Assay Kit (Thermo #23224). Motility of cilia Motility of cilia was examined (100 frames/second for node cilia and sperm, 500 frames/second for airway cilia and brain ependymal cilia) with a high-speed CMOS camera (HAS- 500M, Detect). Motion pattern of cilia was traced with TEMA software. Immunoprecipitation and interaction protein Testes were used for immunoprecipitation of IC2 antibody. Dynabeads protein G was washed three times with IP buffer (50 mm Tris-HCl, 150 mm NaCl, 5 mm MgCl 2, 0.2% NP- 40). Anti-IC2 (5 lg) and mouse control IgG (5 lg) antibodies E8.0 embryos and trachea from 3-week-old mice were fixed overnight with 2% PFA, 2.5% glutaraldehyde and 0.1% tannic acid in 0.1M cacodylate buffer. They were then washed with 8% sucrose in 0.1M cacodylate buffer, fixed with 2% osmium tetroxide for 1 h at room temperature and blocked with 0.5% uranyl acetate. They were dehydrated with ethanol, incubated in QY-1 (Okenshoji, #50751) and embedded in epoxy resin. The area of interest was selected by observation of semithin (1 lm) sections stained with 1% methylene blue. Ultrathin (80 nm) sections were then prepared with a diamond knife and dried on 100-lm grid meshes. These sections were stained with 2% uranyl acetate and 3% lead citrate solution and then observed with a transmission electron microscope (JEM-1200 EX; JEOL). Acknowledgements We thank Y. Ikawa and H. Nishimura for technical assistance. This work was supported by a grant from CREST (Core Research for Evolutional Science and Technology) of the Japan Science and Technology Corporation as well as by a Grant-in- Aid from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (Project No. 13J04993 and ). Y.I. was supported by a fellowship from the Japan Society for the Promotion of Science for Japanese Junior Scientists. References Austin-Tse, C., Halbritter, J., Zariwala, M.A. et al. (2013) Zebrafish ciliopathy screen plus human mutational analysis identifies C21orf59 and CCDC65 defects as causing primary ciliary dyskinesia. Am. J. Hum. Genet. 93, Diggle, C.P., Moore, D.J. & Mill, P. (2014) HEATR2 Plays a Conserved Role in Assembly of the Ciliary Motile Apparatus. PLoS Genet. 10, Genes to Cells (2016) 21, Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd
12 Transport of dynein to cilia Dong, F., Shinohara, K., Botilde, Y., Nabeshima, R., Asai, Y., Fukumoto, A., Hasegawa, T., Matsuo, M., Takeda, H., Shiratori, H., Nakamura, T. & Hamada, H. (2014) Pih1d3 is required for cytoplasmic preassembly of axonemal dynein in mouse sperm. J. Cell Biol. 204, Fowkes, M.E. & Mitchell, D.R. (1998) The role of preassembled cytoplasmic complexes in assembly of flagellar dynein subunits. Mol. Biol. Cell 9, Hirokawa, N., Tanaka, Y. & Okada, Y. (2009) Left-right determination: involvement of molecular motor KIF3, cilia, and nodal flow. Cold Spring Harb. Perspect. Biol. 1, a Ishikawa & Marshall (2011) Ciliogenesis: building the cell s antenna. Nat. Rev. Mol. Cell Biol. 12, Kishimoto, N., Cao, Y., Park, A. & Sun, Z. (2008) Cystic kidney gene seahorse regulates cilia-mediated processes and Wnt pathways. Dev. Cell 14, Knowles, M.R., Ostrowski, L.E., Loges, N.T. et al. (2013) Mutations in SPAG1 cause primary ciliary dyskinesia associated with defective outer and inner dynein arms. Am. J. Physiol. Lung Cell. Mol. Physiol. 304, L736 L745. Loges, N.T., Olbrich, H., Becker-Heck, A. et al. (2009) Deletions and point mutations of LRRC50 cause primary ciliary dyskinesia due to dynein arm defects. Am. J. Hum. Genet. 85, Matsuo, M., Shimada, A., Koshida, S., Saga, Y. & Takeda, H. (2013) The establishment of rotational polarity in the airway and ependymal cilia. Am. J. Physiol. 304, L736 L745. Mitchison, H.M., Schmidts, M., Loges, N.T. et al. (2012) Mutations in axonemal dynein assembly factor DNAAF3 cause primary ciliary dyskinesia. Nat. Genet. 44, Nonaka, S., Shiratori, H., Saijoh, Y. & Hamada, H. (2002) Determination of left-right patterning of the mouse embryo by artificial nodal flow. Nature 418, Omran, H., Kobayashi, D., Olbrich, H. et al. (2008) Ktu/ PF13 is required for cytoplasmic pre-assembly of axonemal dyneins. Nature 456, Panizzi, J.R., Becker-Heck, A., Castleman, V.H., et al. (2012) CCDC103 mutations cause primary ciliary dyskinesia by disrupting assembly of ciliary dynein arms. Nat. Genet. 44, Saijoh, Y., Adachi, H., Mochida, K., Ohishi, S., Hirao, A. & Hamada, H. (1999) Distinct transcriptional regulatory mechanisms underlie left-right asymmetric expression of lefty-1 and lefty-2. Genes Dev. 13, Sun, Z., Amsterdam, A., Pazour, G.J., Cole, D.G., Miller, M.S. & Hopkins, N. (2004) A genetic screen in zebrafish identifies cilia genes as a principal cause of cystic kidney. Development 131, Suzuki, H., Tsuda, M., Kiso, M. & Saga, Y. (2008) Nanos3 maintains the germ cell lineage in the mouse by suppressing both Bax-dependent and independent apoptotic pathways. Dev. Biol. 318, Tarkar, A., Loges, N.T., Slagle, C.E. et al. (2013) DYX1C1 is required for axonemal dynein assembly and ciliary motility. Nat. Genet. 45, Zariwala, M.A., Gee, H.Y., Kurkowiak, M. et al. (2013) ZMYND10 is mutated in primary ciliary dyskinesia and interacts with LRRC6. Am. J. Hum. Genet. 93, Zhao, L., Yuan, S., Cao, Y., Kallakuri, S., Li, Y., Kishimoto, N., Dibella, L. & Sun, Z. (2013) Reptin/Ruvbl2 is a Lrrc6/Seahorse interactor essential for cilia motility. Proc. Natl Acad. Sci. USA 110, Received: 23 March 2016 Accepted: 19 April 2016 Supporting Information Additional Supporting Information may be found online in the supporting information tab for this article: Figure S1 Generation of Lrrc6 / mice. Figure S2 Lrrc6(IRES-LacZ) and Lrrc6::Venus BAC transgenes. Table S1 Laterality defects in visceral organs of the Lrrc6 / mouse Movie S1 Motility of Node Cilia in the Lrrc6 +/+ mouse. Movie S2 Motility of Node Cilia in the Lrrc6 / mouse. Movie S3 Motility of tracheal Cilia in the Lrrc6 +/+ mouse. Movie S4 Motility of tracheal Cilia in the Lrrc6 / mouse. Movie S5 Cilia of brain ependymal cells from the Lrrc6 +/+ mouse are motile. Movie S6 Cilia of brain ependymal cells from the Lrrc6 / mouse are immotile. Movie S7 Motility of sperm from Lrrc6 +/+ mouse. Movie S8 Motility of sperm from Lrrc6 f/f ;Nanos3 Cre/+ mouse Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd Genes to Cells (2016) 21,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
University of Chicago 2012-Present Postdoctoral Research Associate Dept. of Organismal Biology and Anatomy,
 Tetsuya Nakamura, Ph.D. Organismal Biology and Anatomy, University of Chicago Culver Hall 108, 1027 E, 57 th Street Chicago, IL 60637 (773) 834-4774 tetsuya@uchicago.edu http://tetsuyanakamura.work Research
Tetsuya Nakamura, Ph.D. Organismal Biology and Anatomy, University of Chicago Culver Hall 108, 1027 E, 57 th Street Chicago, IL 60637 (773) 834-4774 tetsuya@uchicago.edu http://tetsuyanakamura.work Research
The coiled-coil domain containing protein CCDC40 is essential for motile cilia function and left-right axis formation
 The coiled-coil domain containing protein CCDC40 is essential for motile cilia function and left-right axis formation Anita Becker-Heck#, Irene Zohn#, Noriko Okabe#, Andrew Pollock#, Kari Baker Lenhart,
The coiled-coil domain containing protein CCDC40 is essential for motile cilia function and left-right axis formation Anita Becker-Heck#, Irene Zohn#, Noriko Okabe#, Andrew Pollock#, Kari Baker Lenhart,
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
Primary Ciliary Dyskinesia Clinical Presentation and Diagnosis. Douglas Conrad California Thoracic Society January, 2018
 Primary Ciliary Dyskinesia Clinical Presentation and Diagnosis Douglas Conrad California Thoracic Society January, 2018 Disclosures Current Funding: NIH RO1 National CF Foundation Gilead Sciences Potential
Primary Ciliary Dyskinesia Clinical Presentation and Diagnosis Douglas Conrad California Thoracic Society January, 2018 Disclosures Current Funding: NIH RO1 National CF Foundation Gilead Sciences Potential
Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal
 Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus Fadel TISSIR, Yibo QU, Mireille MONTCOUQUIOL, Libing ZHOU, Kouji KOMATSU, Dongbo SHI, Toshihiko FUJIMORI,
Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus Fadel TISSIR, Yibo QU, Mireille MONTCOUQUIOL, Libing ZHOU, Kouji KOMATSU, Dongbo SHI, Toshihiko FUJIMORI,
SUPPLEMENTARY INFORMATION
 Supplementary Information included with Nature MS 2008-02-01484B by Colantonio et al., entitled The dynein regulatory complex is required for ciliary motility and otolith biogenesis in the inner ear. This
Supplementary Information included with Nature MS 2008-02-01484B by Colantonio et al., entitled The dynein regulatory complex is required for ciliary motility and otolith biogenesis in the inner ear. This
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
DIAGNOSIS OF PRIMARY CILIARY DYSKINESIA
 DIAGNOSIS OF PRIMARY CILIARY DYSKINESIA Kris De Boeck, MD PHD Pediatric Pulmonology, University of Leuven, Belgium Introduction Cause Congenital dysfunction of motile cilia Absence/dysfunction of one of
DIAGNOSIS OF PRIMARY CILIARY DYSKINESIA Kris De Boeck, MD PHD Pediatric Pulmonology, University of Leuven, Belgium Introduction Cause Congenital dysfunction of motile cilia Absence/dysfunction of one of
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
A study of cilia density in sea urchin embryos during gastrulation
 A study of cilia density in sea urchin embryos during gastrulation Steve Das Independent Research Project Report Bio 254 Developmental Biology May 3, 2012 Introduction Embryo growth and development is
A study of cilia density in sea urchin embryos during gastrulation Steve Das Independent Research Project Report Bio 254 Developmental Biology May 3, 2012 Introduction Embryo growth and development is
Supplemental Information. Supernumerary Centrosomes. Nucleate Extra Cilia and Compromise. Primary Cilium Signaling. Current Biology, Volume 22
 Current Biology, Volume 22 Supplemental Information Supernumerary Centrosomes Nucleate Extra Cilia and Compromise Primary Cilium Signaling Moe R. Mahjoub and Tim Stearns Supplemental Inventory 1. Supplemental
Current Biology, Volume 22 Supplemental Information Supernumerary Centrosomes Nucleate Extra Cilia and Compromise Primary Cilium Signaling Moe R. Mahjoub and Tim Stearns Supplemental Inventory 1. Supplemental
Supplementary Figure 1.
 Supplementary Figure 1. Visualization of endoplasmic reticulum-mitochondria interaction by in situ proximity ligation assay. A) Illustration of targeted proteins in mitochondria (M), endoplasmic reticulum
Supplementary Figure 1. Visualization of endoplasmic reticulum-mitochondria interaction by in situ proximity ligation assay. A) Illustration of targeted proteins in mitochondria (M), endoplasmic reticulum
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Primary Ciliary Dyskinesia Pathogenesis, diagnosis and treatment DR CLAIRE HOGG DEPT. RESPIRATORY PAEDIATRICS ROYAL BROMPTON HOSPITAL LONDON.
 Primary Ciliary Dyskinesia Pathogenesis, diagnosis and treatment DR CLAIRE HOGG DEPT. RESPIRATORY PAEDIATRICS ROYAL BROMPTON HOSPITAL LONDON. Pathogenesis of PCD Autosomal recessive Heterogeneous Defect
Primary Ciliary Dyskinesia Pathogenesis, diagnosis and treatment DR CLAIRE HOGG DEPT. RESPIRATORY PAEDIATRICS ROYAL BROMPTON HOSPITAL LONDON. Pathogenesis of PCD Autosomal recessive Heterogeneous Defect
Génétique des DCP. Deciphering the molecular bases of ciliopathies. Estelle Escudier, INSERM U 681 Serge Amselem, INSERM U 654
 Génétique des DCP Estelle Escudier, INSERM U 681 Serge Amselem, INSERM U 654 Deciphering the molecular bases of ciliopathies Linkage analyses Candidate gene approaches Chromosome abnormalities Comparative
Génétique des DCP Estelle Escudier, INSERM U 681 Serge Amselem, INSERM U 654 Deciphering the molecular bases of ciliopathies Linkage analyses Candidate gene approaches Chromosome abnormalities Comparative
RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM
 J. exp. Biol. 192, 73 81 (1994) Printed in Great Britain The Company of Biologists Limited 1994 73 RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM II. ASYMMETRY OF THE CILIARY
J. exp. Biol. 192, 73 81 (1994) Printed in Great Britain The Company of Biologists Limited 1994 73 RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM II. ASYMMETRY OF THE CILIARY
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Axis Formation and Mesoderm Induction
 Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
Zhu et al, page 1. Supplementary Figures
 Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Supporting Information
 Supporting Information Kim et al. 10.1073/pnas.0912180106 SI Materials and Methods DNA Constructs. The N-terminally-tagged mouse Gli2 expression construct, pcefl/3 HA-Gli2, was constructed by ligating
Supporting Information Kim et al. 10.1073/pnas.0912180106 SI Materials and Methods DNA Constructs. The N-terminally-tagged mouse Gli2 expression construct, pcefl/3 HA-Gli2, was constructed by ligating
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
High prevalence of Primary Ciliary Dyskinesia in a British Asian population
 High prevalence of Primary Ciliary Dyskinesia in a British Asian population Chris O Callaghan 1, Phil Chetcuti 2, Eduardo Moya 3 1. University of Leicester, Leicester, United Kingdom 2. Leeds General Infirmary,
High prevalence of Primary Ciliary Dyskinesia in a British Asian population Chris O Callaghan 1, Phil Chetcuti 2, Eduardo Moya 3 1. University of Leicester, Leicester, United Kingdom 2. Leeds General Infirmary,
Asymmetry of the central apparatus defines the location of active microtubule sliding in Chlamydomonas flagella
 Asymmetry of the central apparatus defines the location of active microtubule sliding in Chlamydomonas flagella Matthew J. Wargo and Elizabeth F. Smith* Department of Biological Sciences, Dartmouth College,
Asymmetry of the central apparatus defines the location of active microtubule sliding in Chlamydomonas flagella Matthew J. Wargo and Elizabeth F. Smith* Department of Biological Sciences, Dartmouth College,
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein
 Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
SUPPLEMENTARY INFORMATION
 DOI: 1.138/ncb222 / b. WB anti- WB anti- ulin Mitotic index (%) 14 1 6 2 T (h) 32 48-1 1 2 3 4 6-1 4 16 22 28 3 33 e. 6 4 2 Time (min) 1-6- 11-1 > 1 % cells Figure S1 depletion leads to mitotic defects
DOI: 1.138/ncb222 / b. WB anti- WB anti- ulin Mitotic index (%) 14 1 6 2 T (h) 32 48-1 1 2 3 4 6-1 4 16 22 28 3 33 e. 6 4 2 Time (min) 1-6- 11-1 > 1 % cells Figure S1 depletion leads to mitotic defects
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Mitochondrial DNA Isolation Kit
 Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Supplementary Figure 1. Baf60c and baf180 are induced during cardiac regeneration in zebrafish. RNA in situ hybridization was performed on paraffin
 Supplementary Figure 1. Baf60c and baf180 are induced during cardiac regeneration in zebrafish. RNA in situ hybridization was performed on paraffin sections from sham-operated adult hearts (a and i) and
Supplementary Figure 1. Baf60c and baf180 are induced during cardiac regeneration in zebrafish. RNA in situ hybridization was performed on paraffin sections from sham-operated adult hearts (a and i) and
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Generation and validation of mtef4-knockout mice.
 Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Supplementary Table 1. List of primers used in this study
 Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
SWISS SOCIETY OF NEONATOLOGY. Kartagener s syndrome: Neonatal respiratory distress as initial symptom of primary ciliary dyskinesia
 SWISS SOCIETY OF NEONATOLOGY Kartagener s syndrome: Neonatal respiratory distress as initial symptom of primary ciliary dyskinesia MAY 2005 2 Christen M, and Berger TM, Neonatal and Pediatric Intensive
SWISS SOCIETY OF NEONATOLOGY Kartagener s syndrome: Neonatal respiratory distress as initial symptom of primary ciliary dyskinesia MAY 2005 2 Christen M, and Berger TM, Neonatal and Pediatric Intensive
IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia
 Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Sperm dysfunction and ciliopathy
 Reprod Med Biol (2016) 15:77 94 DOI 10.1007/s12522-015-0225-5 REVIEW ARTICLE Sperm dysfunction and ciliopathy Kazuo Inaba 1 Katsutoshi Mizuno 1,2 Received: 30 July 2015 / Accepted: 26 September 2015 /
Reprod Med Biol (2016) 15:77 94 DOI 10.1007/s12522-015-0225-5 REVIEW ARTICLE Sperm dysfunction and ciliopathy Kazuo Inaba 1 Katsutoshi Mizuno 1,2 Received: 30 July 2015 / Accepted: 26 September 2015 /
Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants
 SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
Supplementary Figure 1. Gene schematics of hyls-1, gasr-8 and k10g6.4, and TEM analysis of TFs in WT and hyls-1 cilia. (a) Gene structure of hyls-1,
 Supplementary Figure 1. Gene schematics of hyls-1, gasr-8 and k10g6.4, and TEM analysis of TFs in WT and hyls-1 cilia. (a) Gene structure of hyls-1, gasr-8 and k10g6.4 based on WormBase (http://wormbase.org),
Supplementary Figure 1. Gene schematics of hyls-1, gasr-8 and k10g6.4, and TEM analysis of TFs in WT and hyls-1 cilia. (a) Gene structure of hyls-1, gasr-8 and k10g6.4 based on WormBase (http://wormbase.org),
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
SUPPLEMENTAL INFORMATION FOR. PAX7 expression defines germline stem cells in the adult testis
 SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION 60611, USA
 Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
Developmental Biology
 Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
ASSET Student Laboratory
 ASSET Student Laboratory The Effect of Cigarette Smoke and Alcohol on Tetrahymena Background Information While the general effects of smoking and alcohol use on humans are well documented, it is useful
ASSET Student Laboratory The Effect of Cigarette Smoke and Alcohol on Tetrahymena Background Information While the general effects of smoking and alcohol use on humans are well documented, it is useful
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
Chromatin IP (Isw2) Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles.
 Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments:
 Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
Edinburgh Research Explorer
 Edinburgh Research Explorer HEATR2 Plays a Conserved Role in Assembly of the Ciliary Motile Apparatus Citation for published version: Diggle, CP, Moore, DJ, Mali, G, Zur Lage, P, Ait-Lounis, A, Schmidts,
Edinburgh Research Explorer HEATR2 Plays a Conserved Role in Assembly of the Ciliary Motile Apparatus Citation for published version: Diggle, CP, Moore, DJ, Mali, G, Zur Lage, P, Ait-Lounis, A, Schmidts,
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplementary Figure 1
 Supplementary Figure 1 Kif1a RNAi effect on basal progenitor differentiation Related to Figure 2. Representative confocal images of the VZ and SVZ of rat cortices transfected at E16 with scrambled or Kif1a
Supplementary Figure 1 Kif1a RNAi effect on basal progenitor differentiation Related to Figure 2. Representative confocal images of the VZ and SVZ of rat cortices transfected at E16 with scrambled or Kif1a
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
Diagnosis of primary ciliary dyskinesia: searching for a gold standard
 EDITORIAL PRIMARY CILIARY DYSKINESIA Diagnosis of primary ciliary dyskinesia: searching for a gold standard Jane S. Lucas 1,2 and Margaret W. Leigh 3,4 Affiliations: 1 Primary Ciliary Dyskinesia Centre,
EDITORIAL PRIMARY CILIARY DYSKINESIA Diagnosis of primary ciliary dyskinesia: searching for a gold standard Jane S. Lucas 1,2 and Margaret W. Leigh 3,4 Affiliations: 1 Primary Ciliary Dyskinesia Centre,
Supplemental Data. Septin-Mediated Uniform Bracing. of Phospholipid Membranes. Supplemental Experimental Procedures. Preparation of giant liposomes
 Supplemental Data Septin-Mediated Uniform Bracing of Phospholipid Membranes Yohko Tanaka-Takiguchi, Makato Kinoshita, and Kingo Takiguchi Supplemental Experimental Procedures Preparation of giant liposomes
Supplemental Data Septin-Mediated Uniform Bracing of Phospholipid Membranes Yohko Tanaka-Takiguchi, Makato Kinoshita, and Kingo Takiguchi Supplemental Experimental Procedures Preparation of giant liposomes
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Microtubule Teardrop Patterns
 Supporting Information Microtubule Teardrop Patterns Kosuke Okeyoshi 1, Ryuzo Kawamura 1, Ryo Yoshida 2, and Yoshihito Osada 1 * 1 RIKEN Advanced Science Institute, 2-1 Hirosawa, Wako-shi, Saitama 351-0198,
Supporting Information Microtubule Teardrop Patterns Kosuke Okeyoshi 1, Ryuzo Kawamura 1, Ryo Yoshida 2, and Yoshihito Osada 1 * 1 RIKEN Advanced Science Institute, 2-1 Hirosawa, Wako-shi, Saitama 351-0198,
ab CytoPainter Golgi/ER Staining Kit
 ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Identification of predicted human outer dynein arm genes- candidates for primary ciliary dyskinesia genes
 JMG Online First, published on June 3, 2005 as 10.1136/jmg.2005.033001 Identification of predicted human outer dynein arm genes- candidates for primary ciliary dyskinesia genes Gregory J. Pazour 1, Nathan
JMG Online First, published on June 3, 2005 as 10.1136/jmg.2005.033001 Identification of predicted human outer dynein arm genes- candidates for primary ciliary dyskinesia genes Gregory J. Pazour 1, Nathan
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Muscular Dystrophy. Biol 405 Molecular Medicine
 Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Luminescent platforms for monitoring changes in the solubility of amylin and huntingtin in living cells
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Development Supplementary information
 Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
Supplemental Materials and Methods Mosaic clonal analysis GSC and SP clones were induced with the FLP/FRT-mediated mitotic recombination technique (Xu and Rubin, 1993) in files with following genotypes:
A protocol for enhancement of the AAV-mediated expression of transgenes
 A protocol for enhancement of the AAV-mediated expression of transgenes Hiroaki Mizukami, Takeharu Kanazawa, Takashi Okada, and Keiya Ozawa Division of Genetic Therapeutics, Center for Molecular Medicine,
A protocol for enhancement of the AAV-mediated expression of transgenes Hiroaki Mizukami, Takeharu Kanazawa, Takashi Okada, and Keiya Ozawa Division of Genetic Therapeutics, Center for Molecular Medicine,
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information
 Supporting Information Chen et al. 10.1073/pnas.0807991106 SI Methods Sucrose Gradient Fractionation. Fractionation by sucrose gradient was performed as described by Gasparini et al. [(2001) J Neurosci
Supporting Information Chen et al. 10.1073/pnas.0807991106 SI Methods Sucrose Gradient Fractionation. Fractionation by sucrose gradient was performed as described by Gasparini et al. [(2001) J Neurosci
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
tom tom 24hpf tom tom 48hpf tom 60hpf tom tom 72hpf tom
 a 24hpf c 48hpf d e 60hpf f g 72hpf h i j k ISV ISV Figure 1. Vascular integrity defects and endothelial regression in mutant emryos. (a,c,e,g,i) Bright-field and (,d,f,h,j) corresponding fluorescent micrographs
a 24hpf c 48hpf d e 60hpf f g 72hpf h i j k ISV ISV Figure 1. Vascular integrity defects and endothelial regression in mutant emryos. (a,c,e,g,i) Bright-field and (,d,f,h,j) corresponding fluorescent micrographs
Supplementary Information. Glycogen shortage during fasting triggers liver-brain-adipose. neurocircuitry to facilitate fat utilization
 Supplementary Information Glycogen shortage during fasting triggers liver-brain-adipose neurocircuitry to facilitate fat utilization Supplementary Figure S1. Liver-Brain-Adipose neurocircuitry Starvation
Supplementary Information Glycogen shortage during fasting triggers liver-brain-adipose neurocircuitry to facilitate fat utilization Supplementary Figure S1. Liver-Brain-Adipose neurocircuitry Starvation
Maoqing Ye, Yan Yin, Kazumi Fukatsu, and Paul Grossfeld
 Evidence That Deletion of ETS-1, a Gene in the Jacobsen Syndrome (11q-) Cardiac Critical Region, Causes Congenital Heart Defects through Impaired Cardiac Neural Crest Cell Function 52 Maoqing Ye, Yan Yin,
Evidence That Deletion of ETS-1, a Gene in the Jacobsen Syndrome (11q-) Cardiac Critical Region, Causes Congenital Heart Defects through Impaired Cardiac Neural Crest Cell Function 52 Maoqing Ye, Yan Yin,
Epithelium-1. Hanan Jafar BDS.MSc.PhD
 Epithelium-1 Hanan Jafar BDS.MSc.PhD General features Epithelium is an avascular tissue composed of cells that cover the exterior body surfaces and line internal closed cavities and tubes. It also forms
Epithelium-1 Hanan Jafar BDS.MSc.PhD General features Epithelium is an avascular tissue composed of cells that cover the exterior body surfaces and line internal closed cavities and tubes. It also forms
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Primer DNA sequence (5 to 3 )
 Supplemental Table 1 Oligonucleotide primers used in the study Primer DNA sequence (5 to 3 ) Plasmodium falciparum 18s forward Plasmodium falciparum 18s reverse Plasmodium falciparum MSP1 forward Plasmodium
Supplemental Table 1 Oligonucleotide primers used in the study Primer DNA sequence (5 to 3 ) Plasmodium falciparum 18s forward Plasmodium falciparum 18s reverse Plasmodium falciparum MSP1 forward Plasmodium
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
CASE REPORT. 1. Assistant Professor. Department of Paediatrics, Vinayaka Missions Medical College, Karaikal
 A CASE OF KARTAGENER SYNDROME Pagadpally Srinivas 1. Assistant Professor. Department of Paediatrics, Vinayaka Missions Medical College, Karaikal CORRESPONDING AUTHOR: Pagadpally Srinivas, 72, Vellai Pillaiyar
A CASE OF KARTAGENER SYNDROME Pagadpally Srinivas 1. Assistant Professor. Department of Paediatrics, Vinayaka Missions Medical College, Karaikal CORRESPONDING AUTHOR: Pagadpally Srinivas, 72, Vellai Pillaiyar
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Stelter et al., http://www.jcb.org/cgi/content/full/jcb.201105042/dc1 S1 Figure S1. Dyn2 recruitment to edid-labeled FG domain Nups.
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Stelter et al., http://www.jcb.org/cgi/content/full/jcb.201105042/dc1 S1 Figure S1. Dyn2 recruitment to edid-labeled FG domain Nups.
High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart.
 High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart. Daniel Aston, Rebecca A. Capel, Kerrie L. Ford, Helen C. Christian,
High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart. Daniel Aston, Rebecca A. Capel, Kerrie L. Ford, Helen C. Christian,
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR
 Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
SUPPLEMENTARY INFORMATION
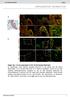 Suppl. Fig. 1 in vivo expression of ISL1 in the human fetal heart. a, Hematoxylin eosin staining showing structures of left atrium and left atrium appendage (*) of a human fetal heart at 11 weeks of gestation.
Suppl. Fig. 1 in vivo expression of ISL1 in the human fetal heart. a, Hematoxylin eosin staining showing structures of left atrium and left atrium appendage (*) of a human fetal heart at 11 weeks of gestation.
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
LC2, the chlamydomonas homologue of the t complex-encoded protein Tctex2, is essential for outer dynein arm assembly
 University of Massachusetts Medical School escholarship@umms Open Access Articles Open Access Publications by UMMS Authors 10-8-1999 LC2, the chlamydomonas homologue of the t complex-encoded protein Tctex2,
University of Massachusetts Medical School escholarship@umms Open Access Articles Open Access Publications by UMMS Authors 10-8-1999 LC2, the chlamydomonas homologue of the t complex-encoded protein Tctex2,
supplementary information
 Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
SUPPLEMENTARY LEGENDS...
 TABLE OF CONTENTS SUPPLEMENTARY LEGENDS... 2 11 MOVIE S1... 2 FIGURE S1 LEGEND... 3 FIGURE S2 LEGEND... 4 FIGURE S3 LEGEND... 5 FIGURE S4 LEGEND... 6 FIGURE S5 LEGEND... 7 FIGURE S6 LEGEND... 8 FIGURE
TABLE OF CONTENTS SUPPLEMENTARY LEGENDS... 2 11 MOVIE S1... 2 FIGURE S1 LEGEND... 3 FIGURE S2 LEGEND... 4 FIGURE S3 LEGEND... 5 FIGURE S4 LEGEND... 6 FIGURE S5 LEGEND... 7 FIGURE S6 LEGEND... 8 FIGURE
