Biological Half-life of Bromide in the Rat Depends Primarily on the Magnitude of Sodium Intake
|
|
|
- Maude Elliott
- 6 years ago
- Views:
Transcription
1 Physiol. Res. 54: , 2005 Biological Halflife of Bromide in the Rat Depends Primarily on the Magnitude of Sodium Intake S. PAVELKA 1,2, A. BABICKÝ 3, M. VOBECKÝ 3 1 Institute of Physiology, Academy of Sciences of the Czech Republic, Prague, 2 Department of Biochemistry, Faculty of Science, Masaryk University, Brno and 3 Institute of Analytical Chemistry, Academy of Sciences of the Czech Republic, Prague, Czech Republic Received August 23, 2004 Accepted January 21, 2005 Online available February 16, 2005 Summary The parallel course of the excretion rates of bromide and sodium ions was demonstrated in adult male and female rats administered simultaneously with potassium 82 Brbromide and 24 Nasodium chloride. The animals were exposed to various intakes of sodium ions accompanied with five different anions: Br, Cl, HCO 3, ClO4, and SCN. Regardless of the anion accompanying the sodium ion, the excretion rates of 82 Br and 24 Na + ions were proportional to the magnitude of sodium intake in the animals. Hence, we have proved our hypothesis that the biological halflife of bromide depends on the magnitude of sodium intake rather than on the intake of chloride. Key words Biological halflife Bromide Chloride Sodium Rat Introduction The mammalian body regulates both volume and ionic composition of the extracellular fluid (ECF), i.e. blood plasma and interstitial fluid. The major osmotically active solutes in the ECF are sodium and its attendant anions. Provided that the osmolality of intra and extracellular fluids remains constant, changes in ECF volume will primarily reflect changes in the total body sodium content. The constancy of electrolyte concentration is achieved by active reabsorption of sodium in kidney tubules, whereas chloride (and bromide) mostly follows passively the movement of sodium. Thus, the control of urinary salt and water excretion is important for maintaining homeostasis of the body. Elements from the same group of the periodic system have repeatedly been shown to exhibit certain similarities of metabolic behavior. This is particularly pronounced for the elements in group 7 (fluorine, chlorine, bromine, and iodine). The physiological significance of chloride ion has long been recognized. Rats fed a diet low in chloride had a retarded growth rate and symptomatology different from that observed with a sodiumdeficient diet (Greenberg and Cuthbertson 1942). The therapeutic as well as the toxic effects of the element bromine are exerted by bromide, the ionic form of bromine. Although bromidecontaining sedative drugs are now infrequently employed, intoxication with this halide and its attendant CNS symptoms remain a serious problem. The distribution (Pavelka et al. 2000) and action of bromide appears to be directly related to its PHYSIOLOGICAL RESEARCH ISSN Institute of Physiology, Academy of Sciences of the Czech Republic, Prague, Czech Republic Fax physres@biomed.cas.cz
2 640 Pavelka et al. Vol. 54 resemblance to other halides, chloride and, to a lesser extent, iodide (Pavelka 2004a,b). In the intestine, both chloride and bromide ions are completely passively absorbed by the paracellular pathway. The distribution of chloride and that of bromide are almost exclusively extracellular (Ullberg et al. 1964). Exceptions concern erythrocytes and acinar cells of the gastric wall. In the extracellular fluid, bromide ions replace an equivalent amount of chloride ions, the molar sum of total halides remaining constant at approximately 110 mmol/l. Both ions are predominantly excreted by the kidneys. The greater part of both halogen ions is reabsorbed in renal tubules after glomerular filtration and, due to the similarity in their physicochemical properties, bromide and chloride compete for tubular reabsorption (Rauws 1983). The biological halflife of bromide can be shortened markedly by administering surplus of sodium chloride (Langley Czerwinski 1958). In fact, this is one of the major actions in the treatment of bromide intoxication (van Leeuwen and Sangster 1987). On the other hand, the already long halflife of bromide, which is about 12 days in humans (Söremark 1960) and approximately 3 to 8 days in the rat (Rauws and van Logten 1975, Pavelka et al. 2000), may be increased considerably by a saltdeficient diet. In the rat, bromide halflife was prolonged to 25 days on a saltfree diet (Rauws and van Logten 1975). This finding was interpreted by the authors as a marked dependence of the biological halflife of bromide on chloride concentration in the diet. However, keeping in mind the differences between the metabolism of sodium and chloride ions and the fact that sodium excretion rate depends on the magnitude of sodium intake, we have hypothesized (Babický et al. 2005) that the biological halflife of bromide depends on the magnitude of sodium intake rather than on the intake of chloride. In order to test our hypothesis, in the present studies we simultaneously determined the biological halflife of bromide (using radionuclide 82 Br) and sodium (radionuclide 24 Na) which was accompanied with five different anions. Methods Animals and diets Experiments were performed on adult Wistar rats: 15 females and 10 males of an average body weight of 268±17 g and 441±27 g, respectively. All the animals were fed a special, lowsodium pelleted diet (Bergman, Kocanda, Czech Republic) with a mean concentration of sodium of 284±20 mg/kg (12.3 meq/kg) and normal levels of potassium and magnesium of about 6730 and 1750 mg/kg, respectively. Dietary concentration of bromine was 3.5±0.2 mg/kg (0.04 meq/kg) and that of chlorine was 588±33 mg/kg (16.6 meq/kg). Concentrations of Na, K, Mg, Br, and Cl in the diet were determined by instrumental neutron activation analysis in a mode of shortterm activation (Vobecký et al. 2000), and in the case of Na and K also by a longterm activation. The experimental design is shown in Table 1. The rats were arranged into five groups, each consisting of three females and two males. Animals in the first group drank distilled water with the addition of NaBr, in the second group with NaCl, in the third group with NaHCO 3, in the fourth group with NaClO 4, and animals in the fifth group drank water with the addition of NaSCN. Concentrations of the solutions of these salts were 50 and 100 mmol/l in the case of females and males, respectively. During the whole experimental period, diet and drinking fluids were provided ad libitum. The rats were housed individually in plastic cages under standard conditions. The body weight of each rat as well as their consumption of diet and drinking fluid were recorded at regular intervals. Table 1. Experimental arrangement of different sodium salts administered in drinking fluid. Group Sex Number of animals Salt Concentration (mmol/l) 1 F 3 NaBr 50 2 F 3 NaCl 50 3 F 3 NaHCO F 3 NaClO F 3 NaSCN 50 Simultaneous determination of biological halflives of bromide and sodium After establishing constant drinking regimens,
3 2005 Biological HalfLive of Bromide in the Rat 641 on the 14th experimental day all the animals were given simultaneously approximately 1.7 MBq 82 Br (about 0.25 mg Br) in the form of potassium bromide and 1.8 MBq 24 Na (about 0.35 mg Na) in the form of sodium chloride in 0.3 ml saline by subcutaneous injection. Obviously, these amounts of Br and Na + ions injected with the radioactive preparations were negligible. The retained 82 Br and 24 Na radioactivity was measured in vivo (at the time intervals of 15, 39, 63, 87, 111, 135, 159, and 183 h after the application of radionuclides) by means of a computerized gammaspectrometric system equipped with an HPGe detector (Canberra). Details of the wholebody measurement of radioactivity are given in our previous paper (Vobecký et al. 2005). The time course of the changes in the wholebody 82 Br ( 24 Na) radioactivity of the experimental animal can be expressed by the equation: R t = A [exp ( t/t el )] (1) where R t is the 82 Br ( 24 Na) radioactivity retained in the rat at time t expressed as the % of the applied dose A, T el is the halflife of the 82 Br ( 24 Na) elimination from the animal s body (given in hours), and t is the time which elapsed from the moment of the 82 Br ( 24 Na) application to the animal (given in hours). The radionuclides 82 Br (halflife of radioactive decay equal to 35.3 h) and 24 Na (physical halflife equal to 15 h) were prepared by irradiating the KBr and NaCl targets with neutrons in the core of the LWR15 research nuclear reactor (Nuclear Research Institute, Řež near Prague) for 1 h at 9 MW power outputs. Statistics The correlation between the biological halflife of bromide and the magnitude of sodium intake was evaluated by Pearson s correlation coefficient, applied to values determined in individual animals exposed to various intakes of sodium ions accompanied with four different anions. Likewise, the correlation between bromide and sodium halflives was evaluated by Pearson s correlation coefficient, but applied to group mean values. Table 2. Mean daily intakes of diet and drinking solutions throughout the experimental period, mean body weight of rats on the 14th and the 22nd day of the experiment, and average values of the 8day body weight gain in individual groups of animals Group Sex Mean daily intake Body weight (g) Diet (g) Solution (ml) 14th day 22nd day 8day gain 1 F 21.3 ± ± ± ± 18 4 M 14.8 ± ± ± ± F 17.4 ± ± ± ± 14 4 M 30.7 ± ± ± ± F 17.4 ± ± ± ± 18 5 M 28.1 ± ± ± ± F 15.3 ± ± ± ± 20 1 M 25.6 ± ± ± ± F 18.0 ± ± ± ± 11 5 M 21.2 ± ± ± ± 6 4 Values are means ± S.D. Results Daily intake of the diet and salt solutions throughout the experimental period as well as mean body weight of rats on the 14th and the 22nd day of the experiment (at the end of the experimental period) in particular groups of animals are summarized in Table 2. It is evident that an increased intake of various sodium salts did not noticeably influence the growth of the animals. Table 3 gives the estimated values of mean daily intake of sodium and the corresponding average values of simultaneously determined biological halflives of bromide and sodium ions and the Br/Na halflife ratios in individual groups.
4 642 Pavelka et al. Vol. 54 Br Cl ClO 4 SCN Linear (All Nasalts) Halflife of Br (h) Halflife of Br (h) y = 1.03x Na intake (mg/day) Fig. 1. Dependence of the biological halflife of bromide on mean daily intake of sodium. The straight line is fitted by linear regression analysis of the values determined in individual animals receiving sodium accompanied with the specified anions Halflife of Na (h) Fig. 2. Correlation between the rate of excretion of bromide and sodium ions. Each symbol represents the mean of bromide and sodium halflife values, measured in 23 animals receiving sodium in the form of solutions of sodium bromide, chloride, perchlorate and thiocyanate in the amounts specified in Table 3. Table 3. Mean daily intake of sodium throughout the experimental period, mean values of the halflife of 82 Brbromide and 24 Nasodium excretion, and average values of the Br/Na halflife ratios in individual groups of animals receiving sodium accompanied with different anions (Br, Cl, HCO 3, ClO4, and SCN ) Group Sex Na intake Halflife (mg/day) Br (h) Na (h) Br/Na 1 F 49.4 ± ± ± M 52.6 ± ± ± F 41.3 ± ± ± M ± ± ± F 39.8 ± ± ± M ± 12.4 (330 ± 12) (99 ± 4) 4 F 37.8 ± ± ± M ± ± ± F 35.8 ± ± ± M 61.4 ± ± ± Values are means ± SD. Not included in the subsequent data evaluation. Figure 1 shows that the measured values of the biological halflife of bromide are inversely proportional to the magnitude of sodium intake in the animals, regardless of the type of anion accompanying sodium ions. The correlation between the halflife of bromide and the intake of sodium was found to be significant (r = 0.76, p<0.05). At the same time, the average values of the biological halflife of bromide closely follow those of sodium halflife, again irrespectively of the type of sodium salt solutions administered to the rats in the individual experimental groups (Fig. 2), with the exception of sodium bicarbonate (see later). The correlation between the determined values of bromide and sodium halflives was highly significant (r = 0.96, p<0.001). Discussion The data obtained in the present experiments (Table 3, Figs 1 and 2) clearly show that the rates of excretion of bromide and sodium ions run in parallel in the rats maintained under the same conditions as regards
5 2005 Biological HalfLive of Bromide in the Rat 643 the magnitude of sodium intake and metabolic activity. The only exception was the biological halflife of bromide in male rats in the third experimental group. These animals were characterized by enormously long biological halflife of bromide, markedly different from the short halflife of sodium in this group. Most probably, an immediate cause of this discrepancy was the large amount of administered bicarbonate ions that altered the blood acidbase equilibrium and produced alkalosis in the animals. The kidneys of these animals obviously excreted the surplus bicarbonate ions at the expense of bromide and chloride ions. Therefore, the discrepant values obtained in these animals maintained under nonphysiological conditions were omitted in Figures 1 and 2. In our previous paper (Babický et al. 2005), we have already demonstrated the parallel course of the excretion rates of sodium and bromide ions in rats which had simultaneously received the radionuclides 24 Na and 82 Br. However, in that paper the experimental animals were maintained on various sodium intakes but sodium ions were applied solely in the form of differently concentrated sodium chloride solutions. Obviously, the present arrangement of feeding sodium ions accompanied with other than chloride anions (Table 1) reasonably extended the validity of our previous conclusions and refuted any objections against possible interference of chloride anions with the effect of sodium ions. Because bromide is excreted slowly and practically completely in the urine, total body chloride may be followed for a considerable time after a single dose of bromide (Hellerstein et al. 1960). As the rat tissues show consistent agreement between the ratios of chloride to bromide in the serum, liver, skin and muscle (Hellerstein et al. 1960), we assume that it is justifiable to use radioactive 82 Brbromide for following the excretion rate of chloride as well. In addition, due to the close relation of sodium ions and the attendant anions, the excretion rate of sodium could also be followed by using 82 Brbromide. The achieved results of the simultaneous determination of biological halflives of sodium and bromide in the rats maintained on various sodium intakes in the present experiments (Figs 1 and 2) and also in our previous experiments (Babický et al. 2005) corroborate this assumption. Acknowledgements This work was supported by the Academy of Sciences of the Czech Republic (research project No. AV0Z ). The authors are indebted to Dr. J. Vorlíček (Institute of Physiology, Academy of Sciences of the Czech Republic, Prague) for his help with statistical evaluation of the data. Preliminary results of this study were presented at the 80 th Meeting of the Czech and Slovak Physiological Societies in Prague, February 36, 2004 (Pavelka et al. 2004). References BABICKÝ A, PAVELKA S, VOBECKÝ M: Biological halflives of bromide and sodium in the rat are connected and dependent on the physiological state. Biol Trace Elem Res 103: 4958, GREENBERG DM, CUTHBERTSON EM: Dietary chloride deficiency and alkalosis in the rat. J Biol Chem 145: , HELLERSTEIN S, KAISER C, DARROW DD, DARROW DC: The distribution of bromide and chloride in the body. J Clin Invest 39: , LANGLEY CZERWINSKI A: Bromide excretion as affected by chloride administration. J Am Pharm Assoc 47: , PAVELKA S: Bromine. In: Elements and Their Compounds in the Environment. Occurrence, Analysis and Biological Relevance, E MERIAN, M ANKE, M IHNAT, M STOEPPLER (eds), Vol. 3, Part IV. WileyVCH Verlag, Weinheim, 2004a, pp PAVELKA S: Metabolism of bromide and its interference with the metabolism of iodine. Physiol Res 53 (Suppl 1): S81S90, 2004b. PAVELKA S, BABICKÝ A, VOBECKÝ M, LENER J, ŠVANDOVÁ E: Bromide kinetics and distribution in the rat. I. Biokinetics of 82 Brbromide. Biol Trace Elem Res 76: 5766, PAVELKA S, BABICKÝ A, VOBECKÝ M: Biological halflives of bromide and sodium in the rat are related and depend on the physiological state. Physiol Res 53: 28P, RAUWS AG: Pharmacokinetics of bromide ion an overview. Food Chem Toxicol 21: , 1983.
6 644 Pavelka et al. Vol. 54 RAUWS AG, VAN LOGTEN MJ: The influence of dietary chloride on the bromide excretion in the rat. Toxicology 3: 2932, SÖREMARK R: The biological halflife of bromide ions in human blood. Acta Physiol Scand 50: , ULLBERG S, APPELGREN LE, CLEMEDSON CJ, ERICSSON Y, EWALDSSON B, SORBÖ B, SÖREMARK R: A comparison of the distribution of some halide ions in the body. Biochem Pharmacol 13: , VAN LEEUWEN FXR, SANGSTER B: The toxicology of bromide ion. CRC Crit Rev Toxicol 18: , VOBECKÝ M, BABICKÝ A, PAVELKA S, LENER J: Determination of bromine and iodine in the rat thyroid by shortterm INAA. J Trace Microprobe Techn 18, , VOBECKÝ M, PAVELKA S, BABICKÝ A: Bromide transfer through mother s milk and its impact on the suckling rat. Biol Trace Elem Res 103: 3748, Reprint requests S. Pavelka, Institute of Physiology, Academy of Sciences of the Czech Republic, Vídeňská 1083, Prague 4, Czech Republic. pavelka@biomed.cas.cz
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
simultaneously excreted. They also brought forward some evidence to
 THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are
 Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Mannitol-induced Metabolic Alkalosis
 Electrolyte & Blood Pressure :, 00 ) Mannitolinduced Metabolic Alkalosis Kyung Pyo Kang, M.D., Sik Lee, M.D., Kyung Hoon Lee, M.D., and Sung Kyew Kang, M.D. Department of Internal Medicine, Research Institute
Electrolyte & Blood Pressure :, 00 ) Mannitolinduced Metabolic Alkalosis Kyung Pyo Kang, M.D., Sik Lee, M.D., Kyung Hoon Lee, M.D., and Sung Kyew Kang, M.D. Department of Internal Medicine, Research Institute
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Fluid, Electrolyte, and Acid Base Balance
 25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
Cutler, Power & Wilder, 1938; Hall & Langley, 1940), in the dog (Winkler &
 8 J. Physiol. (I948) I07, 8-I3 6I2.46I.6 RENAL EXCRETION OF SODIUM AND POTASSIUM IN RATS BY S. E. DICKER (Beit Memorial Fellow) From the Department of Pharmacology, University of Bristol (Received 30 December
8 J. Physiol. (I948) I07, 8-I3 6I2.46I.6 RENAL EXCRETION OF SODIUM AND POTASSIUM IN RATS BY S. E. DICKER (Beit Memorial Fellow) From the Department of Pharmacology, University of Bristol (Received 30 December
Ch 17 Physiology of the Kidneys
 Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Don t Forget the Strong Ions
 Don t Forget the s Rich Erdman University of Maryland Department of Animal & Avian Sciences erdman@umd.edu Don t Forget the s Rich Erdman Department of Animal & Avian Sciences erdman@umd.edu K + 39.1 Na
Don t Forget the s Rich Erdman University of Maryland Department of Animal & Avian Sciences erdman@umd.edu Don t Forget the s Rich Erdman Department of Animal & Avian Sciences erdman@umd.edu K + 39.1 Na
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
9.3 Bromine. Stanislav Pavelka
 1445 9.3 Bromine Stanislav Pavelka 9.3.1 Introduction Bromine belongs to the group of halogens, and is one of the most abundant and ubiquitous of the recognized trace elements in the biosphere. However,
1445 9.3 Bromine Stanislav Pavelka 9.3.1 Introduction Bromine belongs to the group of halogens, and is one of the most abundant and ubiquitous of the recognized trace elements in the biosphere. However,
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
NATURAL HISTORY AND SURVIVAL OF PATIENTS WITH ASCITES. PATIENTS WHO DO NOT DEVELOP COMPLICATIONS HAVE MARKEDLY BETTER SURVIVAL THAN THOSE WHO DEVELOP
 PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
1. 09/07/16 Ch 1: Intro to Human A & P 1
 Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
RENAL TUBULAR ACIDOSIS An Overview
 RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
Electrolytes Solution
 Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
The kidneys are excretory and regulatory organs. By
 exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
exercise 9 Renal System Physiology Objectives 1. To define nephron, renal corpuscle, renal tubule, afferent arteriole, glomerular filtration, efferent arteriole, aldosterone, ADH, and reabsorption 2. To
DIURETICS-3 Dr. Shariq Syed
 DIURETICS-3 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Diuretics primarily prevent the reabsorption of K Na Cl I Don t know, Too busy with periodic exams! AIKTC - Knowledge
DIURETICS-3 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Diuretics primarily prevent the reabsorption of K Na Cl I Don t know, Too busy with periodic exams! AIKTC - Knowledge
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Objectives Body Fluids Electrolytes The Kidney and formation of urine
 Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Effect of Iodine Status and Gender on Perchlorate Inhibition of Iodide Uptake by the Thyroid in Adults: New Data from the Greer Study
 Effect of Iodine Status and Gender on Perchlorate Inhibition of Iodide Uptake by the Thyroid in Adults: New Data from the Greer Study Gay Goodman, Ph.D., D.A.B.T. Human Health Risk Resources, Inc., Seattle,
Effect of Iodine Status and Gender on Perchlorate Inhibition of Iodide Uptake by the Thyroid in Adults: New Data from the Greer Study Gay Goodman, Ph.D., D.A.B.T. Human Health Risk Resources, Inc., Seattle,
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
Body Fluid Compartments
 Yıldırım Beyazıt University Faculty of Medicine Department of Physiology Body Fluid Compartments Dr. Sinan Canan Body fluid balance 1 Body fluid compartments 2 Water distribution Tissue % Water Blood 83,0
Yıldırım Beyazıt University Faculty of Medicine Department of Physiology Body Fluid Compartments Dr. Sinan Canan Body fluid balance 1 Body fluid compartments 2 Water distribution Tissue % Water Blood 83,0
The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency *
 lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
lournal of Clinical Investigation Vol. 45, No. 4, 1966 The Induction of Metabolic Alkalosis by Correction of Potassium Deficiency * HOWARD L. BLEICH,t RICHARD L. TANNEN,t AND WILLIAM B. SCHWARTZ t (From
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Disclaimer. Chapter 3 Disorder of Water, Electrolyte and Acid-base Professor A. S. Alhomida. Disorder of Water and Electrolyte
 Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
H 2 O, Electrolytes and Acid-Base Balance
 H 2 O, Electrolytes and Acid-Base Balance Body Fluids Intracellular Fluid Compartment All fluid inside the cells 40% of body weight Extracellular Fluid Compartment All fluid outside of cells 20% of body
H 2 O, Electrolytes and Acid-Base Balance Body Fluids Intracellular Fluid Compartment All fluid inside the cells 40% of body weight Extracellular Fluid Compartment All fluid outside of cells 20% of body
Renal physiology V. Regulation of acid-base balance. Dr Alida Koorts BMS
 Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
Chapter 21. Diuretic Agents. Mosby items and derived items 2008, 2002 by Mosby, Inc., an affiliate of Elsevier Inc.
 Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
1. remove: waste products: urea, creatinine, and uric acid foreign chemicals: drugs, water soluble vitamins, and food additives, etc.
 Making Water! OR is it really Just Water Just Ask the Nephron!! Author: Patricia L. Ostlund ostlundp@faytechcc.edu (910) 678-9892 Fayetteville Technical Community College Fayetteville, NC 28303 Its just
Making Water! OR is it really Just Water Just Ask the Nephron!! Author: Patricia L. Ostlund ostlundp@faytechcc.edu (910) 678-9892 Fayetteville Technical Community College Fayetteville, NC 28303 Its just
Therapy of Bicarbonate-losing Renal Tubular Acidosis
 Archives of Disease in Childhood, 1970, 45, 774. Therapy of Bicarbonate-losing Renal Tubular Acidosis R. A. DONCKERWOLCKE, G. J. VAN STEKELENBURG, and H. A. TIDDENS From the Wilhelmina Kinderziekenhuis,
Archives of Disease in Childhood, 1970, 45, 774. Therapy of Bicarbonate-losing Renal Tubular Acidosis R. A. DONCKERWOLCKE, G. J. VAN STEKELENBURG, and H. A. TIDDENS From the Wilhelmina Kinderziekenhuis,
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline
 BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE*
 ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
ELECTROLYTE DISTURBANCES IN CONGESTIVE HEART FAILURE* DAVID P. B.AUMANN, M.D. Department of Medicine, University of Arkansas School of Medicine, Little Rock, Arkansas The retention of salt and water secondary
Fluid and electrolyte balance, imbalance
 Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Collin College. BIOL Anatomy & Physiology. Urinary System. Summary of Glomerular Filtrate
 Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
Collin College BIOL. 2402 Anatomy & Physiology Urinary System 1 Summary of Glomerular Filtrate Glomerular filtration produces fluid similar to plasma without proteins GFR ~ 125 ml per min If nothing else
IMPACT OF DIETARY SALT CONCENTRATION ON WATER INTAKE AND PHYSIOLOGICAL MEASUREMENTS OF FEEDLOT CATTLE. Authors:
 IMPACT OF DIETARY SALT CONCENTRATION ON WATER INTAKE AND PHYSIOLOGICAL MEASUREMENTS OF FEEDLOT CATTLE 1999 Animal Science Research Report Authors: Story in Brief Pages 159-164 A.F. La Manna, F.N. Owens,
IMPACT OF DIETARY SALT CONCENTRATION ON WATER INTAKE AND PHYSIOLOGICAL MEASUREMENTS OF FEEDLOT CATTLE 1999 Animal Science Research Report Authors: Story in Brief Pages 159-164 A.F. La Manna, F.N. Owens,
Fluid, electrolyte, and acid-base balance
 Fluid, electrolyte, and acid-base balance Chapter 50 Ra'eda Almashaqba 1 Fluid, electrolyte, and acid-base balance About 46% to 60%of the average adult's weight is water, which is vital to health and normal
Fluid, electrolyte, and acid-base balance Chapter 50 Ra'eda Almashaqba 1 Fluid, electrolyte, and acid-base balance About 46% to 60%of the average adult's weight is water, which is vital to health and normal
Osmoregulation and Osmotic Balance
 OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
Renal System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly
PARAMEDIC RESOURCE MANUAL
 ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
ACID BASE BALANCE & BODY FLUID. Ani Retno Prijanti Renal and Body Fluids Module Juni 2008
 ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
Water compartments inside and outside cells maintain a balanced distribution of total body water.
 Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
Selected Water Quality Topics Related to Larval Shrimp Culture
 Selected Water Quality Topics Related to Larval Shrimp Culture Claude E. Boyd Professor Emeritus School of Fisheries, Aquaculture and Aquatic Sciences Auburn University, Alabama 36849 USA Chlorination
Selected Water Quality Topics Related to Larval Shrimp Culture Claude E. Boyd Professor Emeritus School of Fisheries, Aquaculture and Aquatic Sciences Auburn University, Alabama 36849 USA Chlorination
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Unit 2b: EXCRETION OF DRUGS. Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK)
 Unit 2b: EXCRETION OF DRUGS By Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK) Excretion, along with metabolism and tissue redistribution,
Unit 2b: EXCRETION OF DRUGS By Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK) Excretion, along with metabolism and tissue redistribution,
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA
 J. Exp. Biol. (1971). 54. 269-374 269 With 1 text-figure Printed in Great Britain ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA BY W. R. HARVEY AND K. ZERAHN* Department of Biology,
J. Exp. Biol. (1971). 54. 269-374 269 With 1 text-figure Printed in Great Britain ACTIVE TRANSPORT OF SODIUM BY THE ISOLATED MIDGUT OF HYALOPHORA CECROPIA BY W. R. HARVEY AND K. ZERAHN* Department of Biology,
Metabolic Alkalosis: Vomiting
 RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
DIURETICS-4 Dr. Shariq Syed
 DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
Pressure Diuresis 9 Sample Student Essays
 Pressure Diuresis 9 Sample Student Essays Below please find assembled consecutively in one document the brief analyses submitted by nine students in Mammalian Physiology 08 to the Teach Yourself Pressure
Pressure Diuresis 9 Sample Student Essays Below please find assembled consecutively in one document the brief analyses submitted by nine students in Mammalian Physiology 08 to the Teach Yourself Pressure
Cadmium Binding Components in the Supernatant Fraction of Liver, Kidney and Intestinal Mucosa Homogenates of Cadmium-Administered Rats
 Cadmium Binding Components in the Supernatant Fraction of Liver, Kidney and Intestinal Mucosa Homogenates of Cadmium-Administered Rats Keiichi Tanaka and Kaori Sueda Department of Public Health, Faculty
Cadmium Binding Components in the Supernatant Fraction of Liver, Kidney and Intestinal Mucosa Homogenates of Cadmium-Administered Rats Keiichi Tanaka and Kaori Sueda Department of Public Health, Faculty
only. Analysis of human muscle obtained by biopsy suggests that changes
 A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
A PROPOSED MECHANISM OF EXTRACELLLAR REGLATION OF MSCLE COMPOSITION* ROBERT E. COOKE** AND WILLIAM E. SEGAR The changes in muscle composition which occur in disturbances of extracellular electrolyte composition
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
Chapter 24 Water, Electrolyte and Acid-Base Balance
 Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
estimates were made of the normal rate of increase in plasma urea over periods in skin and in plasma, hypertonic sodium chloride solution was
 482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
Fluids and electrolytes
 Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Terminology. Terminology. Terminology. Molarity number of moles of solute / Liter of solution. a) Terminology b) Body Fluid Compartments
 Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
preliminaryfinding. Current theories of iron metabolism would explain dispute over urinary iron, which is agreed by all to be very small and
 148 J. Physiol. (I938) 94, I48-I54 6I5.739.I3:6I2.386 THE ABSORPTION AND EXCRETION OF IRON FOLLOWING ORAL AND INTRAVENOUS ADMINISTRATION BY R. A. McCANCE AND E. M. WIDDOWSON From the Biochemical Laboratory,
148 J. Physiol. (I938) 94, I48-I54 6I5.739.I3:6I2.386 THE ABSORPTION AND EXCRETION OF IRON FOLLOWING ORAL AND INTRAVENOUS ADMINISTRATION BY R. A. McCANCE AND E. M. WIDDOWSON From the Biochemical Laboratory,
Fal Fal P h y s i o l o g y 6 1 1, S a n F r a n c i s c o S t a t e U n i v e r s i t y
 Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
After studying this lecture, you should be able to...
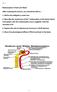 Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
Reabsorption of Salt and Water After studying this lecture, you should be able to... 1. Define the obligatory water loss. 2. Describe the mechanism of Na ++ reabsorption in the distal tubule and explain
BASIC PHARMACOKINETICS
 BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
RENAL FUNCTION An Overview
 RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
11/05/1431. Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate
 Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Renal physiology D.HAMMOUDI.MD
 Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
The kidney. (Pseudo) Practical questions. The kidneys are all about keeping the body s homeostasis. for questions Ella
 The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
Urinary System. Dr. ZHANG Xiong. Dept. of Physiology. ZJU School of Medicine. QUESTION 6
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Dept. of Physiology. ZJU School of Medicine.
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151/Able.Acc2.Web/Template/View.aspx?action =view&coursetype=0&courseid=26519 QUESTION 6 How is the filtrate reabsorbed
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151/Able.Acc2.Web/Template/View.aspx?action =view&coursetype=0&courseid=26519 QUESTION 6 How is the filtrate reabsorbed
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure
 EXCRETORY SYSTEM WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure These wastes include: Carbon dioxide Mostly through breathing
EXCRETORY SYSTEM WHY DO WE NEED AN EXCRETORY SYSTEM? Function: To eliminate waste To maintain water and salt balance To maintain blood pressure These wastes include: Carbon dioxide Mostly through breathing
Kidneys in regulation of homeostasis
 Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
Testing Protocol. Iodine Estimation of Salt
 Testing Protocol Iodine Estimation of Salt 1 Laboratory Procedure for Iodine Estimation of Salt Background material: Iodine is one of the first minerals recognized as essential for human health. Iodine
Testing Protocol Iodine Estimation of Salt 1 Laboratory Procedure for Iodine Estimation of Salt Background material: Iodine is one of the first minerals recognized as essential for human health. Iodine
Acid-Base Physiology. Dr. Tamás Bense Dr. Alexandra Turi
 Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
DIETARY CHLORIDE DEFICIENCY AND ALKALOSIS IN THE RAT. (Received for publication, June 29, 1942)
 DIETARY CHLORIDE DEFICIENCY AND ALKALOSIS IN THE RAT BY DAVID M. GREENBERG AND ELIZABETH M. CUTHBERTSON (From the Division of Biochemistry, University of California Medical School, Berkeley) (Received
DIETARY CHLORIDE DEFICIENCY AND ALKALOSIS IN THE RAT BY DAVID M. GREENBERG AND ELIZABETH M. CUTHBERTSON (From the Division of Biochemistry, University of California Medical School, Berkeley) (Received
Lab 19 The Urinary System
 Lab 19 The Urinary System Laboratory Objectives Identify and describe the micro- and macroscopic anatomy of the kidney. Track the blood flow in and out of the kidney. Compare blood, glomerular filtrate,
Lab 19 The Urinary System Laboratory Objectives Identify and describe the micro- and macroscopic anatomy of the kidney. Track the blood flow in and out of the kidney. Compare blood, glomerular filtrate,
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Libromide 325 mg tablets for dogs 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 tablet contains: Active substance: Potassium
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Libromide 325 mg tablets for dogs 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 tablet contains: Active substance: Potassium
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
